Abstract
To minimize chemical waste and protect the environment, our team has used green analysis with natural reagents. In this work, we designed a natural-reagent assay kit for iron determination and implemented it in chemistry education in Thailand. The iron assay method was adapted from Thai local wisdom of testing water quality using guava leaves. The guava leaf powder served as a natural reagent in the assay. The kit included equipment, standard and buffer solutions and a manual. A smart device with a built-in camera was used as a detector. Educators in six universities in Thailand implemented the kit in laboratories with modifications depending on their learning outcomes. The kit implementation was evaluated using a survey with questions in four aspects: usability, learning achievement, green chemistry and portability. The high average scores for all questions (> 4.00 of 5.00 points), with the average overall score of 4.53 ± 0.60, indicated satisfaction regarding in all aspects. Using a locally available bio-resource as a natural reagent for green analysis in chemistry education supported sustainable education in Thailand, in terms of quality education (SDG 4) and reduced inequalities (SDG 10) and environmental sustainability (SDG 6—Clean water and sanitation, 12—Responsible consumption and production and 14—Life be-low water).
1. Introduction
The green chemistry concept has provided approaches to reduce the risks of chemistry to the environment [1]. Within the twelve principles of green chemistry, proposed since 1998 by P. T. Anastas and J. C. Warner [2], analytical chemistry was a branch that was difficult to be environmentally friendly. In fact, some of the green chemistry principles, such as waste prevention, safer solvents and auxiliaries, design for energy efficiency, safer chemistry to minimize the potential of chemical accidents and development of instrumental methods, are directly related to analytical chemistry [3]. However, the need for hazardous chemical reagents for some steps in the analytical process, e.g., sample preservation and preparation and chemical analysis, have been one of the causes. Analytical chemists have endeavored to develop greener chemical analyses, leading to more sustainable chemistry [4]. The Organisation for Economic Co-operation and Development (OECD) defined the term “Sustainable chemistry” as a scientific concept that seeks to improve the efficiency with which natural resources are used to meet human needs for chemical products and services. Sustainable chemistry encompasses the design, manufacture and use of efficient, effective, safe and more environmentally benign chemical products and processes [5].
Our research group interests, since 1991, are in the development of flow-based analysis (FBA) methods for various aspects of chemical analyses. The FBA is a green analytical method that could reduce reagent consumption and, therefore, waste production, from macro- to nano-volume. Moreover, using FBA helps save energy due to its simplicity and rapidity and prevents accidents due to its closed and, sometimes, remote system. However, the use of hazardous chemicals is unavoidable in some cases. The development of green chemical analysis methods utilizing natural reagents has, hence, become our attempt to meet the sustainable chemistry goals and three of the sustainable development goals (SDGs) identified in 2015 by the United Nations (UN), i.e., goal 6‒Clean water and sanitation; goal 12‒Responsible consumption and production; and goal 14‒Life below water.
In 2005, our group reported the use of a natural reagent (guava leaf extract) as an alternative reagent for the flow injection determination of iron, based on Thai local wisdom [6,7]. This local wisdom has been passed down through generations. The ground guava leaves were put in water samples to test whether the water could be used for washing clothes. If the water turned dark purple, it indicated too much iron and the water should not be used. Recently, it was reported that these guava leaves were a source of phenolic compounds, responsible for binding to or chelating with metal ions such as Fe(III) ions [8]. Our group’s article in 2005 found that the extract from guava leaves was a promising natural reagent for the iron determination. From the discovery in this publication, we have further adapted Thai local wisdom in detecting chemicals or elements in real samples to the modern analysis, for examples; using guava leaf extract for quality control of iron content in pharmaceutical formulations [8]; exploiting green tea extracts for the iron determination by flow injection analysis (FIA) [9]; determining iron(III) using Phyllanthus emblica Linn. as a natural reagent [10]; using Indian almond (Terminalia Catappa L.) leaf extract to determine aluminum in wastewater [11]; using Morinda citrifolia root to determine aluminum in tea [12] and the attempts succeeded [13,14,15].
In order to develop sustainability in chemistry education, in this work, the guava leaf extract, among those local wisdom-based innovations, has been investigated further and applied as a green reagent in chemistry experiments for undergraduate education in six Thai universities. The extract was transformed into a ready-to-use reagent powder and packed up with a set of simple experimental equipment and a user manual, as a natural reagent iron assay kit, for the experiment. Each university adapted the kit to achieve its aim of the experiment. Implementing the natural reagent iron assay kit in chemistry courses was studied. Satisfaction with the utilization of the developed assay kit was evaluated using a questionnaire consisting of nine questions, rating satisfaction level in four aspects: the usability of the developed assay kit, the learning achievement, the green chemistry and the portability of the developed assay kit.
2. Materials and Methods
In order to standardize the natural reagent from guava leaf, it was produced in a laboratory at Chiang Mai University, as the supplier and was sent to cooperating universities. Preparation of the natural reagent from guava leaf, assembling the natural-reagent iron assay kit and the implementation of the kit are described in this part.
2.1. Natural Reagent from Guava Leaf (in Brief)
2.1.1. Production of the Natural Reagent Powder
Firstly, fresh guava leaves were washed and dried in a hot air oven at 60 °C for 24 h. The dried leaves were then ground to powder and sieved to obtain 10–20 mesh powder. Finally, a ready-to-use natural reagent powder was obtained and packed in a tea bag and sealed to an air-tight opaque package to avoid exposure to air and light as shown in Figure 1a. The natural reagent powder from guava leaves for the iron assay was so-called Nat Pow Iron (G).
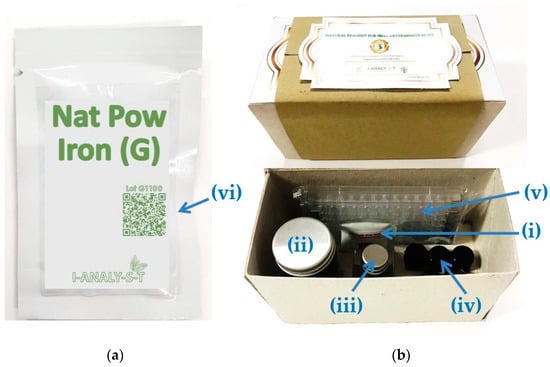
Figure 1.
(a) Nat Pow Iron (G) sachet; (b) natural-reagent iron assay kit.
2.1.2. Preparation of the Natural-Reagent Iron Assay Kit
The natural-reagent iron assay kit (Figure 1b) consists of (i) a sachet of Nat Pow Iron (G), (ii) a vial for extracting the natural reagent, (iii) a vial containing the iron(III) standard, (iv) a vial containing buffer solution, (v) a 96 wells microplate as a reaction platform and (vi) instructions for the reagent preparation as shown in Supplementary Material: Figure S1, which can be accessed by scanning the QR code on a sachet of Nat Pow Iron (G). The chemicals and conditions used were optimized as reported previously [6].
2.1.3. Quality Control of the Nat Pow Iron (G)
The quality control of the Nat Pow Iron (G) was performed using high performance liquid chromatography (HPLC). Every lot of the Nat Pow Iron (G) was sampled for HPLC analysis of active ingredients before packing as the reagent for the iron assay kit. The analysis method is described elsewhere [16]. The major components in the natural reagent extract were phenolic compounds, namely gallic acid, catechin and ellagic acid, as illustrated in Figure 2. Users could check the quality of the Nat Pow Iron (G) by scanning the QR code on the Nat Pow Iron (G) sachet for the HPLC fingerprint of the reagent. The reagent data sheet is also shown in the Supplementary Material: Figure S1.
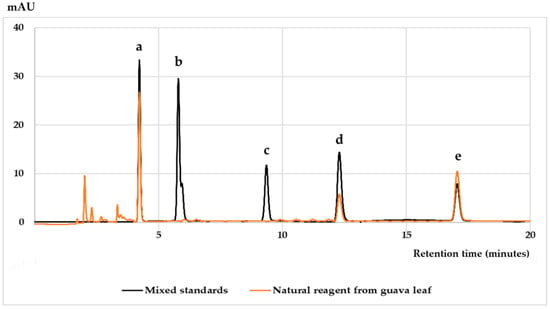
Figure 2.
HPLC chromatograms of mixed phenolic compound standards and natural reagent from guava leaf. The peaks are (a): gallic acid; (b): gallocatechin; (c): epigallocatechin; (d): catechin; and (e): ellagic acid.
2.1.4. Using a Smart Device as a Detector
A dark purple complex is the product of the reaction between iron (III) and phenolic compounds in the natural reagent. Its color intensity is directly proportional to the amount of the iron (III). Thus, the amount of iron (III could be measured by detecting the difference in the color intensity of the reaction product. To promote equitable quality education (SDG 4-Quality education), simplifying the detection using a smart device with a built-in camera, i.e., a smartphone or a tablet, was done. A photograph of the product solutions in the well-microplate was taken by a student’s smart device. The color value of each solution was analyzed using an image processing program on a computer or an application on the smart device such as ImageJ application, Chemical Sensor System by TRF and I-ANALY-S-T (CSS by TRF & I-ANALY-S-T).
2.2. Implementation of the Natural-Reagent Iron Assay Kit
The natural-reagent iron assay kit was demonstrated at six national universities in Thailand (Figure 3): (i) Faculty of Pharmacy, Chiang Mai University, (ii) Faculty of Science, King Mongkut’s University of Technology Thonburi, (iii) Faculty of Science and Engineering, Kasetsart University- Chalermphrakiat Sakon Nakhon Province Campus, (iv) Faculty of Science, Khon Kaen University, (v) School of Science, Mae Fah Luang University and (vi) School of Science, University of Phayao. The use of the developed assay kit was recommended in the procedure manual as shown in Supplementary Material: Figure S2. However, it could be customized by users and course instructors to achieve their goals or the objectives of the experiment and to suit different groups of learners at each university with different working environments. Some components of the kits could be replaced by either suitable or available equipment such as liquid measuring and handling tools.
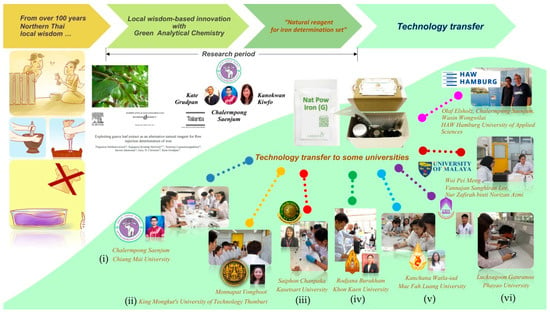
Figure 3.
Implementation of the assay kits as a learning material for a green chemical analysis topic in 6 universities: (i) Chiang Mai University (20 students); (ii) King Mongkut’s University of Technology Thonburi (25 students); (iii) Kasetsart University—Chalermphrakiat Sakon Nakhon Province Campus (11 students); (iv) Khon Kaen University (10 students); (v) Mae Fah Luang University (12 students); and (vi) University of Phayao (25 students).
Satisfaction level was evaluated using a self-administered questionnaire. The questionnaire was composed of nine issues with a 5-point Likert scale (1 = least satisfied, 2 = less satisfied, 3 = moderately satisfied, 4 = very satisfied, 5 = most satisfied). The issues covered (i) clearness and comprehensibility of the procedure manual, (ii) simplicity of the operation, (iii) suitability of the equipment, (iv) student’s comprehensibility in the principle of the iron assay, (v) student’s ability in transferring knowledge learned from this experiment to other students, (vi) operator safety, (vii) environmentally friendliness, (viii) feasibility of application for sampling-site chemical analysis and (ix) overall satisfaction with this experiment. The satisfaction was evaluated by 40 students enrolled in two chemistry subjects from two faculties, i.e., Instrumental Methods of Chemical Analysis Laboratory I (CHM363) course (20 students) at the Faculty of Science, King Mongkut’s University of Technology Thonburi, Bangkok and Quality Control for Food and Cosmetics (461571) course (20 students) at the Faculty of Pharmacy, Chiang Mai University, Chiang Mai. The difference of the satisfaction scores evaluated by students between the two groups was analyzed using independent t-test, with a p < 0.05 as significant difference. It should be noted that there was no control group of students who used a standard traditional method for iron analysis.
Moreover, we randomly selected students and divided them into three groups of four for the focus group discussion, asking about the advantages, difficulties, limitations, satisfaction and suggestions for improvement. The focus group discussion for each group took approximately 15–20 minutes and the content analysis was conducted.
3. Results
3.1. Implementation of the Developed Assay Kit in On-Site Classes
The iron assay kit using the Nat Pow Iron (G) as a natural reagent was distributed to the six universities for implementation in a hands-on experiment of chemistry courses (Figure 3). As an example, Supplementary Material: Video S1 demonstrated the use of the developed assay kit in a chemistry laboratory at the Department of Chemistry, Faculty of Science, King Mongkut’s University of Technology Thonburi. Because the procedure was designed at the beginning to be flexible, the instructors in each university were able to adjust the procedures, including adding steps and changing the provided equipment to available lab equipment, to achieve the specific learning outcomes. For example, to measure the volumes of solutions, the instructors may use different types of measuring equipment, e.g., autopipette, disposable medical syringe, or dropper. The autopipette was used at all six universities because it provided an accurate and precise volume. Four universities additionally used the disposable syringe, while one university used the dropper, both calibrated with the autopipette, to measure the volumes of the solutions. The Supplementary Material: Figure S3 illustrates the example of the laboratory instruction modified from the provided procedure manual.
Despite the fact that students used various smart devices with different operating systems (iOS and Android) and different camera quality, the assay accuracy, as measured by analysis of known-concentration samples, was not significantly different (statistical analysis by independent t-test, p = 1.00). Moreover, although the procedure manual provided in the assay kit recommended using a smart device as the detection device, the image processing application/program could actually be varied. This flexibility promoted students to learn the importance of chemical data analysis, especially the accuracy and precision of the analysis, which expressed the reliability and reproducibility of the analytical data. In all, three universities used the in-house image processing mobile application; Chemical Sensor System by TRF and I-ANALY-S-T (CSS by TRF & I-ANALY-S-T for Android-APK download) [17], whereas the other three universities used different mobile applications, including Pixel Picker [18], ImageJ [19], Color Detector [20] and Color Grab [21].
3.2. Evaluation of the Utilisation of the Developed Assay Kit as a Learning Material in Chemistry Courses
The responses from all 40 students regarding their satisfaction with the experiment using the natural-reagent iron assay kit as a learning material are shown in Figure 4. Overall, the satisfaction scores evaluated by 40 students from the chosen universities (two universities, 20 students each) showed that the average score for every question was higher than 4.00 (full score = 5.00) points, meaning very satisfied, with the average satisfaction on the natural-reagent iron assay kit of 4.55 ± 0.64. The three issues that had the highest scores were the operator safety (4.78 ± 0.42), the environmental friendliness (4.70 ± 0.46) and the suitability of the equipment (4.63 ± 0.54), respectively. The issue with the lowest score (4.20 ± 0.61) was the student’s ability in transferring knowledge learned from the experiment to other students. The score of the student’s ability in transferring knowledge given by applied science students in the university U1 was surprisingly below 4.00 points, meaning moderately satisfied. Nevertheless, the satisfaction scores evaluated by pharmacy students (U1) were lower than those by chemistry students (U2) with the significant differences (statistically tested by independent t-test, p < 0.05) in all issues, except issue 6—Operator safety (p = 0.06).
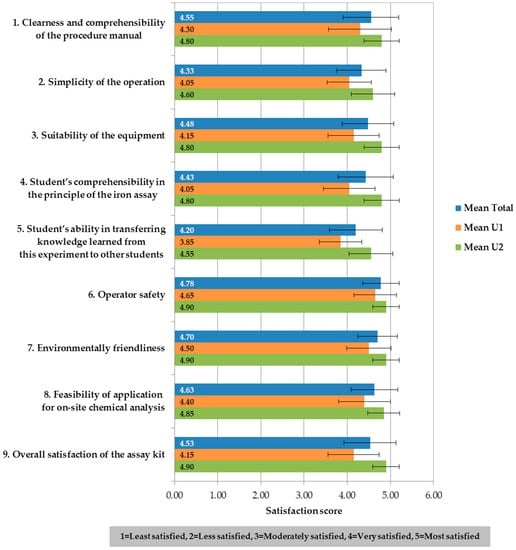
Figure 4.
Mean satisfaction on the experiment using the natural-reagent iron assay kit as a learning material evaluated by students (n = 40).
However, the results from the focus group discussion showed that all students (12 students) were very satisfied with the green chemical analysis using the developed assay kit. Most students were impressed with the innovation in green chemical analysis. Some students said that “It was a novel laboratory investigation that used less volume of chemicals, and the chemical reagent was produced from a natural bio-resource. I think it is very good and also safe for students”.
4. Discussion
4.1. Implementation of the Natural-Reagent Iron Assay Kit in Chemistry Education
An academic application of the developed natural-reagent iron assay kit was demonstrated in chemistry education at six universities in Thailand and was used as learning material for an experiment of natural and applied science courses. Although the provided procedure manual in the developed assay kit clearly directed the use, it remained flexible enough to be modified according to the desired learning outcomes of the courses. However, all six universities followed the core procedure to achieve the completion of the expected chemical reaction. The modification was made only in the steps of solution preparation. Using droppers and disposable syringes, calibrated with autopipettes, allowed students to learn with creativity and open-mindedness. They would be able to adapt any kind of volume-measuring equipment available in their laboratory or place to measure accurate and precise volume by appropriately calibrating with standard volume-measuring equipment.
In the current digital and mobile world, it could not be denied that students must possess one of the smart devices as a tool for learning and communication. The employment of smart devices as detection devices led to simplicity and even equity in education, supporting SDG 10 (Reduced inequalities). Instead of a spectrophotometer, which might be unavailable in some laboratories due to its cost, a smart phone could obviously detect the color intensity using its high-quality camera. The results showed that various smart devices with different operating systems and different camera quality did not exhibit a significant difference in the accuracy of the assay. Thus, every student would be able to analyze his or her experimental data and learn how to analyze analytical data, which is the seventh step in the chemical analytical process. Students were excited and mentioned that “The experiment was easy and fun”; It’s an exotic lab. It’s very interesting; I didn’t think I could use my mobile phone for the experiment, and it was very interesting”.
The experiment report could also be created in Microsoft Excel spreadsheet and submitted to the instructor via email or other online methods. This increased the convenience for both the students and the instructors and importantly created a paperless culture, the green mind. Moreover, as the instructors at a university created a cloud space, i.e., Google spreadsheet, for sharing the analytical results during the class, the results from students could be discussed at real-time in the class. Students were encouraged to critique as a result of this.
4.2. Evaluation of the Utilisation of the Developed Assay Kit as a Learning Material in Chemistry Courses
Because all of the students had prior experience using the traditional spectrophotometric method for other analyses, no control group was used in this study. The students were able to compare their prior experience with the developed assay kit. Overall, the evaluation results illustrated a high level of satisfaction (average overall score 4.53 ± 0.60) in the utilization of the developed assay kit as a learning material in chemistry courses. Hence, employing the assay kit in chemistry education is promising. However, the statistical t-test, comparing the satisfaction scores evaluated by pharmacy (U1) and chemistry students (U2), indicated the pharmacy students’ satisfaction scores were significantly lower than those of the chemistry students in all aspects, except the operator safety. The reason was that the pharmacy students were concerned that the assay kit would not be able to be used in pharmaceutical analysis, which needs high levels of precision and accuracy following FDA regulations and pharmaceutical pharmacopoeia. The satisfaction scores for the operator safety evaluated by students in U1 and U2 did not significantly differ. It demonstrated that the assay kit was safe for pharmaceutical and chemistry research. Therefore, the green chemical analysis may be used as an alternative analysis in the pharmaceutical industry, but may be unable to be utilized as the primary analysis. However, some pharmacy students were impressed by the use of natural extract as an environmentally friendly reagent for iron analysis, the simplicity of the analytical procedure and the use of smartphones as the detection device instead of a spectrophotometer.
The questions in the satisfaction survey were categorized, for further discussion, in four aspects: usability of the developed assay kit (questions 1, 2 and 3), learning achievement (questions 4 and 5), green chemistry (questions 6 and 7) and portability (question 8) and were discussed as follows.
4.2.1. Usability of the Developed Assay Kit
The results showed the students were very satisfied with using the assay kit in terms of the clearness and comprehensibility of the procedure manual (score 4.55 ± 0.64), the simplicity of the operation (score 4.33 ± 0.57) and the suitability of the equipment (score 4.48 ± 0.60), indicating the simplicity and user friendliness of the assay kit. Some students expressed their impression as follows:
“It was my first time discovering that natural reagents could detect iron, and was a lot easier than I imagined, with no need of expensive and complicated laboratory equipment or a teacher’s assistance. I am capable of doing all tasks on my own.”
Nonetheless, while the majority of students encountered no problems, some did. For example, because of the transparency of the solutions and the small size of the micro-volume well plate, they found it difficult to locate the wells and drop the solutions into the wells. They stated that “We got dizzy when we had to drop the solution into the well because it was so small”.
Therefore, the developed assay kit could definitely be employed as a learning material for natural and applied chemistry courses. The developed assay kit showed its flexibility so that educators could modify the procedures in order to achieve their learning outcomes of the course.
4.2.2. Learning Achievement
Most students said in the focus group session that “The experiment can be linked to the lesson and make the lesson more interesting and memorable”, which agreed with the evaluation of the developed assay kit. The average score of the issue “student’s comprehensibility in the principle of the iron assay” was 4.43 ± 0.64, meaning very satisfied. However, the average score for the issue "Student’s ability in transferring knowledge learned from this experiment to other students" was the lowest score (4.20 ± 0.61), but its interpretation was very satisfactory. According to the Learning Theory Pyramid, students will gain approximately 90% of their knowledge by teaching others, resulting in a deeper understanding and long-term memory, as opposed to learning through traditional passive methods such as lecture and reading, which only gain 5–30% of the knowledge. Furthermore, some responses from the participating students via an online survey showed that they were able to learn new things related to green chemical analysis, such as how to colorimetrically determine iron using a natural reagent, accuracy and precision, perform green analytical chemistry and obtain information technology (IT) skills. Therefore, hands-on experiments with simple but intense learning material, such as the developed assay kit, helped promote deeper understanding and long-term memory quite well. Notably, students may not have the opportunity to practice standard laboratory skills such as measuring the volumes of solutions with pipettes and preparing solutions in volumetric flasks. However, these constraints came with more benefits in terms of sustainability.
4.2.3. Green Chemistry
As mentioned earlier, the average satisfaction scores for the green chemistry aspect, i.e., the issues “Operator safety” and “Environmental friendliness”, were the highest scores (4.78 ± 0.42 and 4.70 ± 0.46 points, respectively). It indicated that students were deeply concerned with the hazards of chemicals that could harm the operator’s and the environmental health. It could be implied that utilization of natural reagents, based on Thai local wisdom, in chemistry education had raised students’ awareness of “Responsible consumption and production’’—SDG 12, which is concerned with responsible use of natural resources to avoid harmful effects to the environment.
This proposed green chemistry experiment achieved our research group’s aim to meet SDG 6—Clean water and sanitation and SDG 14—Life below water successfully. It helped ensure the availability and sustainable management of water sanitation (SDG 6) by reducing the hazardous wastewater released to water resources. Consequently, it conserved the oceans, sea and marine resources (SDG 14). Furthermore, not only does using natural reagents extracted from natural resources in Thailand reduce environmental hazards, but it also reduces inequality within and among countries (SDG 10—Reduced inequalities) by producing cost-effective natural reagents in Thailand, as well as minimizing the relatively high-cost chemical reagents imported from abroad, which some organizations rely on. Referring to the interview of Emeritus Professor in analytical chemistry, Prof. Dr. K. Grudpan, published in a national newspaper [22], the chemical reagents imported from abroad could cost four times more than those produced by using the bio-resources in Thailand. Using natural extracts as natural reagents for chemical analyses could save up to 10 million THB and save time according to the duration of long-distance transportation (up to six months) [23].
Nevertheless, because natural reagents are non-toxic and produced from a locally available bio-resource, their use for chemistry promotes the green chemistry principles 5—safer solvents and auxiliaries and 7—use of renewable feedstocks, respectively. This approach also raised awareness about the importance of utilizing green chemistry and natural reagents in experiments to save the environment and ensure sustainable education and development. This agrees with target 4.7 of SDG 4, stating “By 2030, ensure that all learners acquire the knowledge and skills needed to promote sustainable development”.
4.2.4. Portability and Future Application
The satisfaction score for the issue “Feasibility of application for sampling-site chemical analysis” was high (4.63 ± 0.54). It meant that students found the developed assay kit convenient and highly feasible for sampling-site iron analysis. Students also expressed that the developed assay kit did not require expensive and complicated scientific instruments and, thus, could be easily carried to the site. In addition, unlike the traditional analysis method for iron assay, all steps of the analysis using the developed assay kit did not have to be performed in a laboratory because there were fewer hazardous chemicals and harmful procedures.
In our further studies, the developed assay kit could be implemented in an online chemistry laboratory class. As a result of the COVID-19 pandemic, all on-site academic activities were suspended and online learning became a feasible alternative. Science educators and also students may have difficulty with laboratory lessons because students may not have the necessary equipment at home and could not afford to obtain it. Therefore, educators still need to design efficient and effective classes to continue achieving the learning outcomes of the courses. Higher education in chemistry requires hands-on laboratories to allow students to develop laboratory and 21st-century learning skills, i.e., learning and innovation, information, media and technology and life and career. During the first wave of the COVID-19 outbreak, an educator in our research team constructed an online chemistry laboratory using the developed assay kit as a learning tool [22,23]. The developed assay kit was sent to students’ homes so that all students enrolled in the course could conduct the analysis in an online laboratory class at their homes. This study will be presented as “Lab-at-Home” in the next series of the development of the natural-reagent iron assay kit based on Thai local wisdom.
5. Conclusions
Much Thai local wisdom, for over hundreds of years, has used Thai natural resources to test or analyze for poisons or toxins. Our research group has been working on utilizing this valuable wisdom to develop green chemical analysis as a green innovation. Moreover, the developed green chemical analysis method was implemented as a learning tool for chemistry education. This work presented an example of developing green innovation adapting the wisdom of detecting iron element in water using ground guava leaf extract to analyze iron(III) ion in a laboratory. The guava leaf extract was served as a natural reagent for this purpose. A set of a sachet of the ground and dried guava leaf powder (Nat Pow Iron (G)), an iron(III) standard solution, a buffer solution, a 96-wells microplate and a procedure manual were packed as a natural-reagent iron assay kit and used for a chemistry experiment of chemistry classes in six Thai universities. The detection of colored complex, the product of the chemical reaction, was simplified using smart devices with built-in cameras, instead of a relatively high-cost spectrophotometer. The instructors of the courses could modify the procedure to achieve the learning outcomes of the courses. The results showed that students were remarkably satisfied with the natural-reagent iron assay kit in terms of the usability of the developed assay kit, learning achievement, green chemistry and portability. Utilizing the developed assay kit supported SDG 4 (quality education), SDG 6 (clean water and sanitation), SDG 10 (reduced inequalities), SDG 12 (responsible consumption and production) and SDG 14 (life below water) and promoted sustainable chemistry education.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/su132011147/s1, Figure S1: instruction for the reagent preparation, Figure S2: provided procedure manual, Figure S3: example of the laboratory instruction modified from the provided procedure manual, Video S1: use of natural-reagent iron assay kit in the Instrumental Methods of Chemical Analysis Laboratory I (CHM363) course at Department of Chemistry, Faculty of Science, King Mongkut’s University of Technology Thonburi in academic year 2019.
Author Contributions
Concept and design: K.G., M.V., C.S. and K.K. (Kanokwan Kiwfo); Experiments and onsite interview: M.V., C.S., K.K. (Kanokwan Kiwfo), P.P. (Pathinan Paengnakorn), K.K. (Kullapon Kesonkan), C.Y., P.I.N.A. and D.C.; Data curation and analysis: M.V., C.S., K.K. (Kanokwan Kiwfo), P.P. (Pathinan Paengnakorn), K.K. (Kullapon Kesonkan), C.Y., P.I.N.A., N.K., D.C. and S.C.; Statistical analysis and interpretation: S.S., D.C., S.C. and P.P. (Piyatida Panitsupakamol); Writing—original draft preparation: K.K. (Kanokwan Kiwfo), P.P. (Piyatida Panitsupakamol), S.S., P.P. (Pathinan Paengnakorn), D.C., S.C., C.S. and M.V.; Writing—review and editing: S.S., D.C., S.C., P.P. (Piyatida Panitsupakamol) and K.G.; Funding Acquisition: K.G. and C.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Center of Excellence for Innovation in Analytical Science and Technology (I-ANALY-S-T) and Cluster of Excellence on Biodiversity-Based Economics and Society (B.BES-CMU), Chiang Mai University, Chiang Mai, 50200, Thailand. This research was also supported by Distinguished Research Professor Award in “Green Innovation in Chemical Analysis with Local Wisdom” (Grant No. DPG6080002; K. Grudpan), Thailand Research Found (TRF), Thailand.
Institutional Review Board Statement
Ethical review and approval were waived for this study because the study was conducted in the established educational settings, involving normal instructional practices that were unlikely to have a negative impact on students’ learning.
Informed Consent Statement
Informed consent was not applicable because this study was waived for ethical approval.
Data Availability Statement
The data presented in this study are available on reasonable request from the corresponding author.
Acknowledgments
Six universities, namely Chiang Mai University (Chalermpong Saenjum), King Mongkut’s University of Technology Thonburi (Monnapat Vongboot), Kasetsart University- Chalermphrakiat Sakon Nakhon Province Campus (Saiphon Chanpaka), Khon Kaen University (Rodjana Burakham), Mae Fah Luang University (Kanchana Watla-iad), University of Phayao (Lucksagoon Ganranoo), are acknowledged for their contributions in the implementation of the developed assay kit in chemistry courses. The authors are grateful to the Department of Pharmaceutical Sciences, Faculty of Pharmacy, Chiang Mai University and the Department of Chemistry, Faculty of Science, King Mongkut’s University of Technology Thonburi, for their supports and assistance. We also acknowledge Thomas McManamon for language editing and proofreading the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Keith, L.H.; Gron, L.U.; Young, J.L. Green Analytical Methodologies. Chem. Rev. 2007, 107, 2695–2708. [Google Scholar] [CrossRef] [PubMed]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: Oxford, UK; New York, NY, USA, 1998; ISBN 0-19-850234-6. [Google Scholar]
- Koel, M.; Kaljurand, M. Application of the Principles of Green Chemistry in Analytical Chemistry. Pure Appl. Chem. 2006, 78, 1993–2002. [Google Scholar] [CrossRef]
- Tobiszewski, M.; Mechlińska, A.; Namieśnik, J. Green Analytical Chemistry: Theory and Practice. Chem. Soc. Rev. 2010, 39, 2869–2878. [Google Scholar] [CrossRef] [PubMed]
- Sustainable Chemistry—OECD. Available online: https://www.oecd.org/chemicalsafety/risk-management/sustainablechemistry.htm (accessed on 8 September 2021).
- Settheeworrarit, T.; Hartwell, S.K.; Lapanatnoppakhun, S.; Jakmunee, J.; Christian, G.D.; Grudpan, K. Exploiting Guava Leaf Extract as an Alternative Natural Reagent for Flow Injection Determination of Iron. Talanta 2005, 68, 262–267. [Google Scholar] [CrossRef] [PubMed]
- I-ANALY-S-T CMU. Green Innovation in Chemical Analysis with Local Wisdom: EP1 from Local to Global. 2020. Available online: https://www.youtube.com/watch?v=Kka-OAzOigY (accessed on 1 September 2021).
- Siriangkhawut, W.; Ponhong, K.; Grudpan, K. A Green Colorimetric Method Using Guava Leaves Extract for Quality Control of Iron Content in Pharmaceutical Formulations. Malays. J. Anal. Sci. 2019, 23, 595–603. [Google Scholar]
- Pinyou, P.; Kradtap Hartwell, S.; Jakmunee, J.; Lapanantnoppakhun, S.; Grudpan, K. Flow Injection Determination of Iron Ions with Green Tea Extracts as a Natural Chromogenic Reagent. Anal. Sci. Int. J. Jpn. Soc. Anal. Chem. 2010, 26, 619–623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaikrajang, N.; Kruanetr, S.; Harding, D.J.; Rattanakit, P. A Simple Flow Injection Spectrophotometric Procedure for Iron(III) Determination Using Phyllanthus Emblica Linn. as a Natural Reagent. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 204, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Insain, P.; Khonyoung, S.; Sooksamiti, P.; Lapanantnoppakhun, S.; Jakmunee, J.; Grudpan, K.; Zajicek, K.; Kradtap Hartwell, S. Green Analytical Methodology Using Indian Almond (Terminalia Catappa L.) Leaf Extract for Determination of Aluminum Ion in Waste Water from Ceramic Factories. Anal. Sci. Int. J. Jpn. Soc. Anal. Chem. 2013, 29, 655–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tontrong, S.; Khonyoung, S.; Jakmunee, J. Flow Injection Spectrophotometry Using Natural Reagent from Morinda Citrifolia Root for Determination of Aluminium in Tea. Food Chem. 2012, 132, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Grudpan, K.; Hartwell, S.K.; Lapanantnoppakhun, S.; McKelvie, I. The Case for the Use of Unrefined Natural Reagents in Analytical Chemistry—A Green Chemical Perspective. Anal. Methods 2010, 2, 1651–1661. [Google Scholar] [CrossRef]
- Grudpan, K.; Hartwell, S.K.; Wongwilai, W.; Grudpan, S.; Lapanantnoppakhun, S. Exploiting Green Analytical Procedures for Acidity and Iron Assays Employing Flow Analysis with Simple Natural Reagent Extracts. Talanta 2011, 84, 1396–1400. [Google Scholar] [CrossRef] [PubMed]
- Lapanantnoppakhun, S.; Tengjaroensakul, U.; Mungkornasawakul, P.; Puangpila, C.; Kittiwachana, S.; Saengtempiam, J.; Hartwell, S.K. Green Analytical Chemistry Experiment: Quantitative Analysis of Iron in Supplement Tablets with Vis Spectrophotometry Using Tea Extract as a Chromogenic Agent. J. Chem. Educ. 2020, 97, 207–214. [Google Scholar] [CrossRef]
- Saenjum, C.; Pattananandecha, T.; Nakagawa, K. Detection of Antioxidant Phytochemicals Isolated from Camellia Japonica Seeds Using HPLC and EPR Imaging. Antioxidants 2020, 9, 493. [Google Scholar] [CrossRef] [PubMed]
- CSS by TRF & I-ANALY-S-T for Android—APK Download. Available online: https://apkpure.com/css-by-trf-i-analy-s-t/com.imagecolorpicker (accessed on 10 September 2021).
- Pixel Picker—Image Color Picker. Available online: https://apps.apple.com/us/app/pixel-picker-image-color-picker/id930804327 (accessed on 10 September 2021).
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Color Detector—Color Picker. Available online: https://apps.apple.com/us/app/color-detector-color-picker/id1478504229 (accessed on 10 September 2021).
- Sl, U.T. Color Grab (Android). Available online: https://color-grab.en.uptodown.com/android (accessed on 10 September 2021).
- CMU Researcher Creates Green Innovation in Chemical Analysis from Century-Old Wisdom of the Fang People (In Thai: นักวิจัย มช.สร้างนวัตกรรมสีเขียวในการวิเคราะห์ทางเคมี จากภูมิปัญญาบรรพชนชาวฝาง). Available online: https://www.matichon.co.th/region/news_2748857 (accessed on 10 September 2021).
- Outstanding Research Professor Develops Green Innovation for Chemical Analysis Based on the Fang People’s Century-Old Wisdom. (In Thai: ศาสตราจารย์วิจัยดีเด่นสร้างนวัตกรรมสีเขียว วิเคราะห์ทางเคมีจากภูมิปัญญาบรรพชนฝาง). Available online: https://tsri.or.th/th/news/content/601/นวัตกรรมสีเขียวในการวิเคราะห์ทางเคมีกับภูมิปัญญาท้องถิ่น (accessed on 10 September 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).