Abstract
Introduction: Serum metabolome changes after acute ischemic stroke (AIS), but the significance of this is poorly understood. We evaluated whether this change is associated with AIS outcomes in patients with large vessel occlusion (LVO). To improve validity, we combined cross-sectional and longitudinal designs and analyzed serum using Nuclear Magnetic Resonance (NMR) and Liquid Chromatography–Mass Spectrometry (LC-MS). Methodology: In the cross-sectional part, we compared serum metabolome from 48 LVO strokes, collected at 48–72 h, and analyzed with NMR, while in the longitudinal part, we compared metabolome from 15 LVO strokes, collected at <24 h, 48–72 h, 5–7 days, and 80–120 days, and analyzed with LC-MS between patients with modified Rankin Scores (mRS) of 0–3 and 4–6 at 90 days. We hypothesized that compounds elevated in patients with mRS 0–3 in the cross-sectional part would also be elevated in the longitudinal part, and vice versa. We used regression for the analysis and TSBH for multiple testing. Results: In the cross-sectional part, cholesterol, choline, phosphoglycerides, sphingomyelins, and phosphatidylethanolamines had lower levels in patients with an mRS of 0–3 compared to an mRS of 4–6. In the longitudinal part, lower levels of sphingomyelin (d18:1/19:0, d19:1/18:0)* significantly correlated with an mRS of 0–3 in patients with small infarction volume, while lower levels of sphingolipid N-palmitoyl-sphingosine (d18:1/16:0), 1-palmitoyl-2-docosahexaenoyl-GPC (16:0/22:6), 1-palmitoyl-2-docosahexaenoyl-GPE, palmitoyl-docosahexaenoyl-glycerol (16:0/22:6), campesterol, and 3beta-hydroxy-5-cholestenoate correlated with an mRS of 0–3 in patients with large infarction volume. Conclusions: This pilot study showed that lower levels of lipidomic components nerve cell membrane correlate with good AIS outcomes. If proven on large-scale studies, these compounds may become important AIS outcome markers.
1. Introduction
Acute ischemic stroke (AIS) is a leading cause of serious disability in the United States [1]. The annual cost of stroke care totals USD 34 billion and continues to grow [2]. Novel diagnostic and treatment techniques significantly improved AIS outcomes, but their prediction remains uncertain and purely based on clinical and radiologic data. Serum biomarkers are well developed and actively used in many areas of medicine, but nonexistent in AIS. The isolation of the central nervous system (CNS) from other body compartments by the blood–brain barrier (BBB) prevents serum from changing in many CNS disorders. The interruption of the BBB after AIS leads to free communication between the CNS and the serum compartments [3]. This may be used for the identification of AIS serum markers. Metabolomics is a novel method that investigates disease pathophysiology and carries the ability to identify many low-molecular-weight metabolites in bodily fluids. Such metabolites may serve as disease biomarkers. Many investigators found an important association of serum metabolites with AIS [4,5]. They associated these changes with post-stroke depression [6], cognitive impairment [7], and overall disease severity [8,9]. These findings, however, were often contradictory, and explanations were speculative. Our group identified common trends in the change in serum metabolome after AIS and focused on finding clinical applications for these changes. In this pilot study, we evaluated whether trends of serum metabolome are associated with outcomes of Large Vessel Occlusion (LVO) stroke. We used different technologies for metabolome analysis: Nuclear Magnetic Resonance (NMR) and Liquid Chromatography–Mass Spectroscopy (LC-MS) and a combination of cross-sectional and longitudinal study designs to improve confidence in our findings.
2. Methods
2.1. Study Goal and Design
The goal of this study was to evaluate whether changes in serum metabolome are associated with AIS outcomes. We evaluated serum metabolome on 2 sets of patients using cross-sectional and longitudinal analyses. In the cross-sectional part of this study, we evaluated the difference in serum metabolome at 48–72 h between patients with an mRS of 0–3 (good outcomes) and 4–6 (poor outcomes) at 3 months; in the longitudinal part, we evaluated the difference in the trajectory of serum metabolome change at 4 time-points: (1) <24 h; (2) 48–72 h; (3) 5–7 days; and (4) during the follow-up clinic visit (80–120 days) between patients with mRS 0–3 and 4–6 at 3 months. All collected samples were kept at −80 degrees Celsius, then processed and analyzed at the same time. Our hypothesis was that serum metabolome compounds associated with AIS outcomes would have similar changes in the cross-sectional and longitudinal parts of this study: (1) metabolites that elevated in patients with good/poor outcomes in the cross-sectional part would also elevate in the longitudinal part; (2) metabolites that decreased in patients with good/poor outcomes in the cross-sectional part would also decrease in the longitudinal part. Given that our longitudinal cohort was heterogeneous and included patients with and without successful reperfusion, we also performed analysis for patients with small (<40 mL) and large (>40 mL) infarction volume.
The study participants were a part of our ongoing, institutional board-approved, Metabolome in Ischemic Stroke Study (MISS), which investigates biomarkers using genomics, metabolomics, and other -omics technologies [4,10,11]. All participants signed their informed consent. We included patients with internal carotid, anterior, middle, or posterior cerebral artery occlusion; magnetic resonance imaging (MRI) was performed for AIS definition. Two independent investigators calculated AIS volumes using MR segmentation software [12]. This software utilizes an interactive computer-aided detection scheme, where the user identifies and draws the stroke boundary for each of the identified stroke slices, and the computer-aided detection scheme computes the infarction volume. At presentation, we evaluated patients with the National Institute of Health Stroke Scale (NIHSS) and pre-admission modified Rankin Scale (mRS). During the follow-up visit (80–120 days), we performed neurological exams, reviewed medical records for recurrent strokes, and again carried out mRS evaluations. We excluded patients with (1) hemorrhagic conversion of AIS and development of parenchymal hematoma [13]; (2) systemic infection; (3) renal disease with glomerular filtration rate <45; and (4) recurrent stroke during follow-up visits because these conditions could show distinct metabolomic profiles and confound our results.
2.2. Metabolome Analysis
We have chosen to use different analytic platforms in the cross-sectional and the longitudinal parts of this study to improve the validity of our findings. In the cross-sectional part, serum metabolome was analyzed using NMR, which provides information on 250 fundamental serum metabolites, well established and commonly used for AIS metabolomic research, including large data repositories such as UK Biobank (UKBB). In the longitudinal part, serum metabolome was analyzed using LC-MS, which provides more detailed information on 1554 metabolites (for example, while NMR shows the level of sphingomyelin, in LC-MS sphingomyelin, it is represented by 29 sphingomyelin derivatives) but is more expensive and less commonly used and has fewer AIS publications. Observing similar findings across NMR and LC-MS platforms would improve the validity of our results.
NMR metabolomics platform (Nightingale Health Ltd., Helsinki, Finland) was used to quantify the circulating metabolites of non-lipidomic and lipidomic origins. This high-throughput metabolomics platform provides the simultaneous quantification of amino acids, ketone bodies, glycol high-density metabolites, routine lipids, abundant fatty acids, and different lipoprotein subclasses, including high-density lipoproteins (HDLs); intermediate-density lipoproteins (IDLs); low-density lipoproteins (LDLs); and very-low-density lipoproteins (VLDLs)/chylomicrons in absolute concentration units. The NMR platform has been applied extensively in epidemiological studies [14,15], and details of these experiments are described elsewhere [16,17].
For the LC-MS metabolomics platform (Metabolon, Inc. Morrisville, NC, USA), the serum was separated from the red cap vacutainer by centrifugation at 2200 rpms for 15 min, after allowing the blood to clot for at least 30 min in a vertical position before spinning. EDTA plasma samples were extracted, proteins were precipitated with methanol, and aliquots were analyzed by ultra-high-performance LC-MS in the positive (one optimized for hydrophilic, the other hydrophobic compounds), negative, and polar ion modes. All methods utilized Waters ACQUITY ultra-performance liquid chromatography and Thermo Scientific Q-Exactive high resolution/accurate mass spectrometer interfaced with a heated electrospray ionization source and Orbitrap mass analyzer operated at 35,000 mass resolution. The assignment of a unique mass, signal-to-noise calculation, and compound identification were based on the mass spectral pattern compared to NIST and Wiley mass spectral libraries, followed by visual inspection for quality control [18]. For quality control and assurance, an internal matrix was used as well as several internal standards to determine instrument variability, with representative relative standard deviations of 3% (plasma) for internal standards and 8% (plasma) for endogenous biochemicals.
2.3. Statistical Analysis
The clinical and demographic variables were summarized using mean with standard deviation or error for continuous variables and numbers and percentages for categorical variables. Raw metabolome profile data were normalized through log-transformation. The analysis was performed in R4.4.2. We used multivariate linear regression for the cross-sectional part and linear mixed regression model for the longitudinal part of this study. We used age, gender, and race as covariates for cross-sectional and longitudinal analysis. We used the Two-Stage Benjamini–Hochberg (TSBH) procedure for multiple testing in order to minimize false positive correlations and, again, increase confidence in our results. We set alpha significance at <0.05.
3. Results
The cross-sectional part included 48 LVO stroke patients, 34 had an mRS of 0–3 and 14 had an mRS of 4–6. The longitudinal part included 15 LVO stroke patients. Demographic and clinical characteristics for both parts are presented in Table 1.

Table 1.
Baseline patients’ characteristics. 1 Mean (SD); n (%).
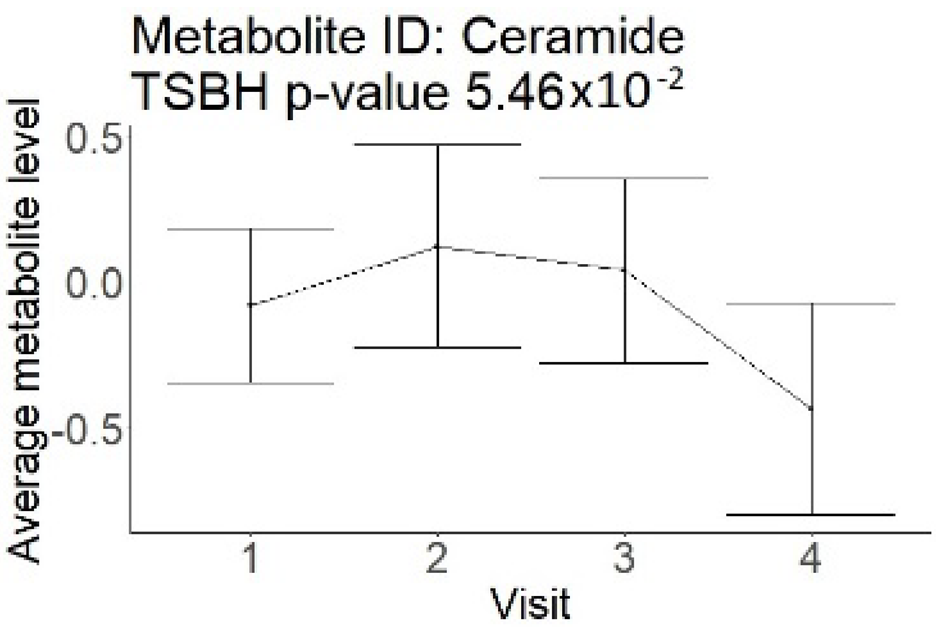
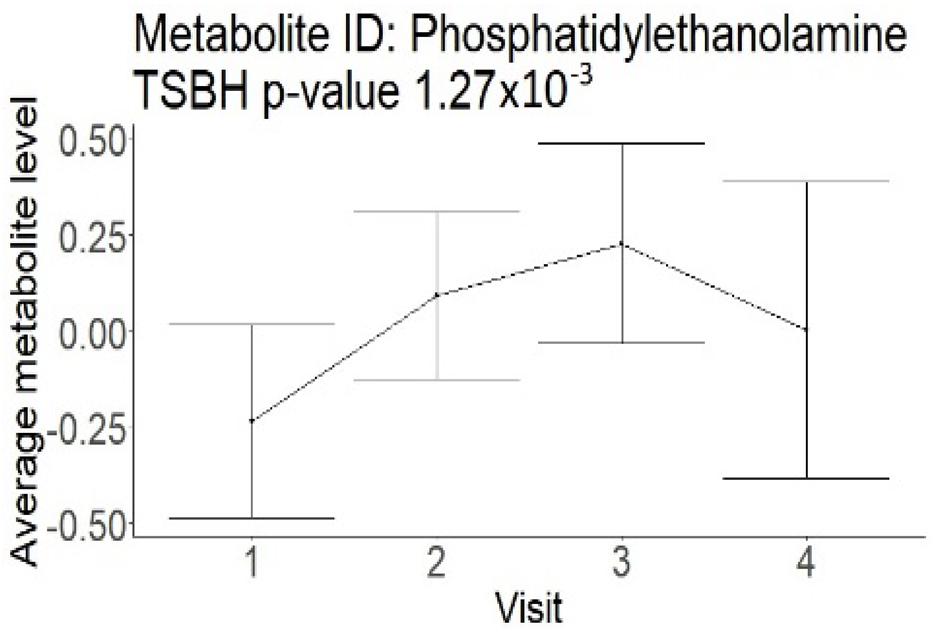
In the cross-sectional part, out of 250 compounds, 114 were significantly different in patients with an mRS of 0–3 compared to an mRS of 4–6 (Supplement Table S1). All 114 metabolites had lower levels associated with good outcomes and represented a metabolism of different lipoprotein sub-classes (100 compounds), saturated and unsaturated fatty acids (6 compounds), energy metabolites (glucose and lactate), cholesterol, choline, phosphoglycerides, sphingomyelins, and phosphatidylethanolamines.
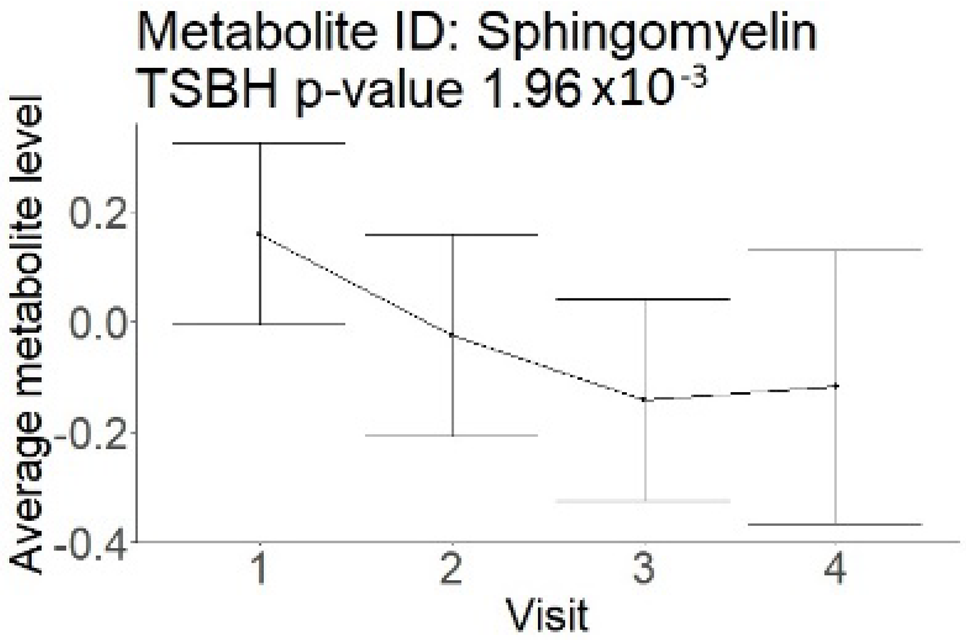
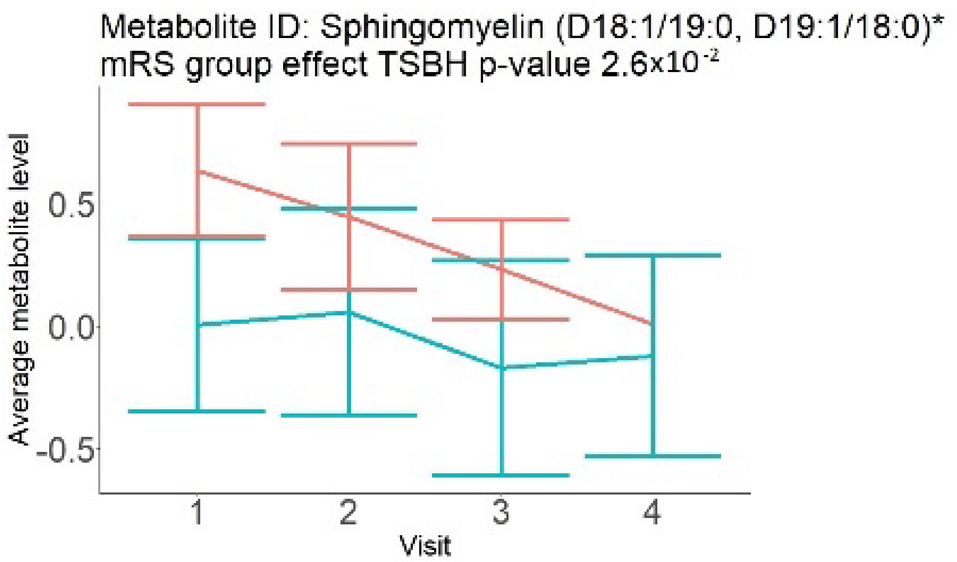
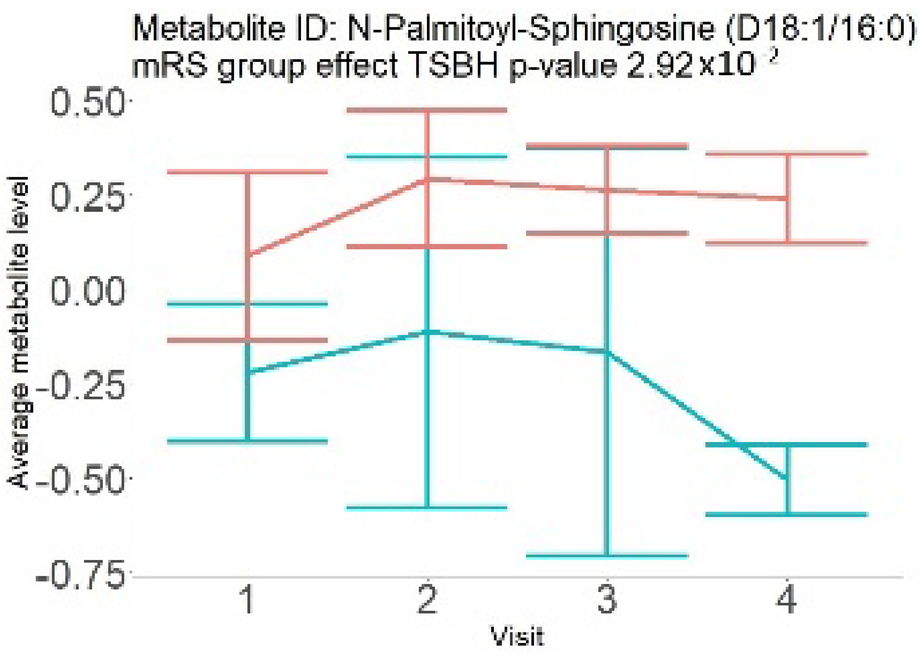
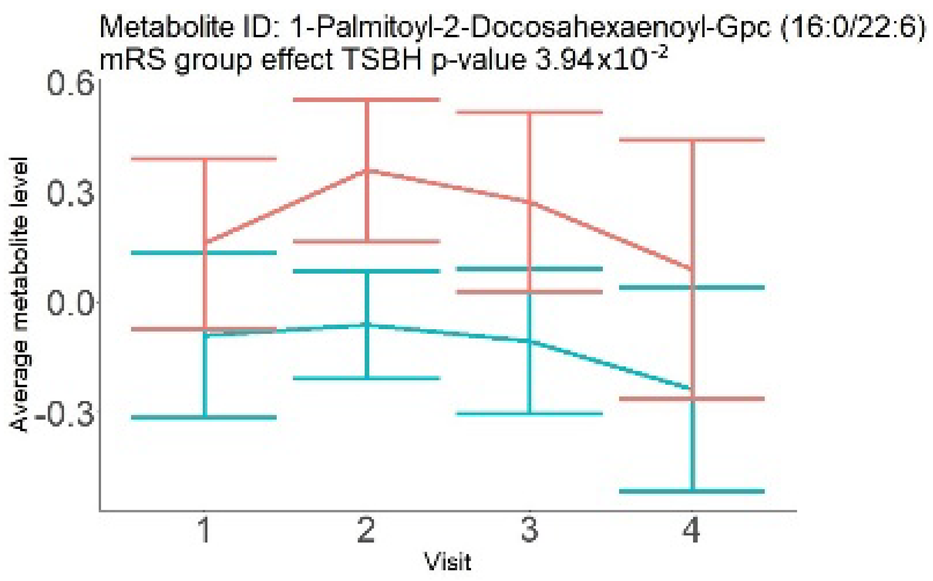
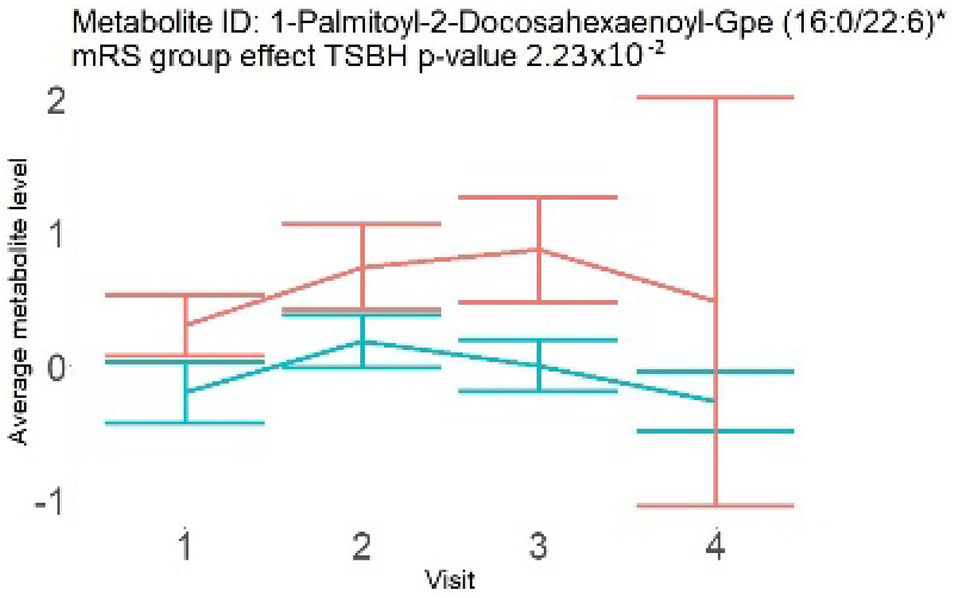
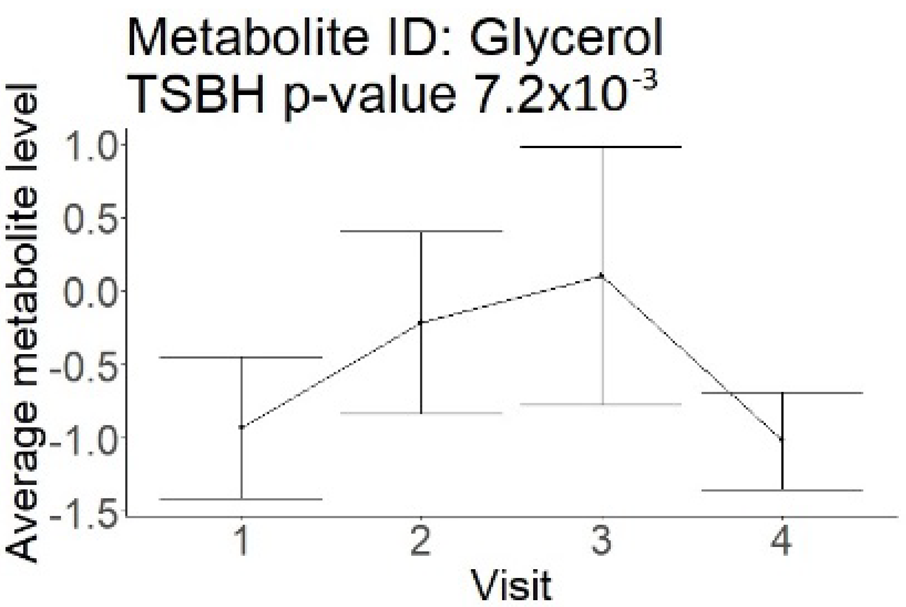
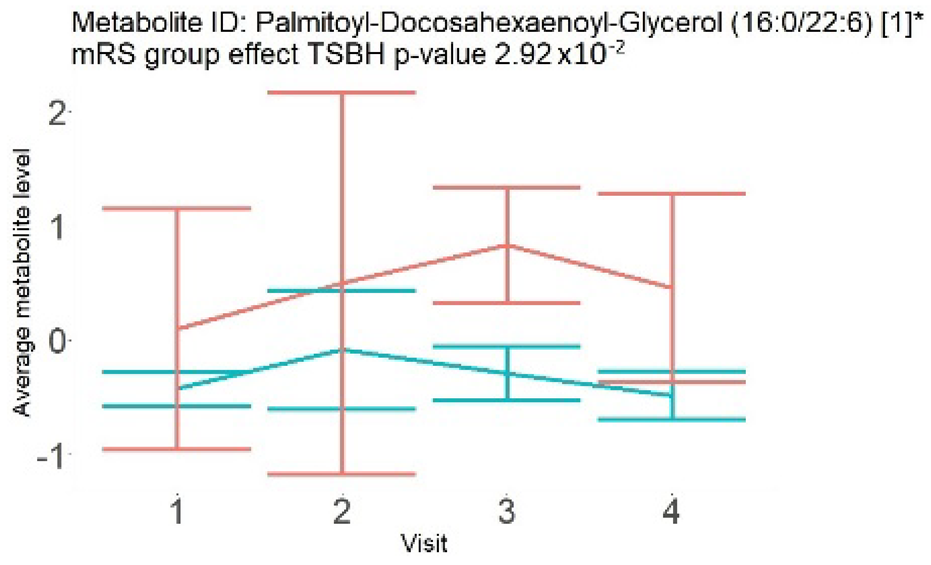
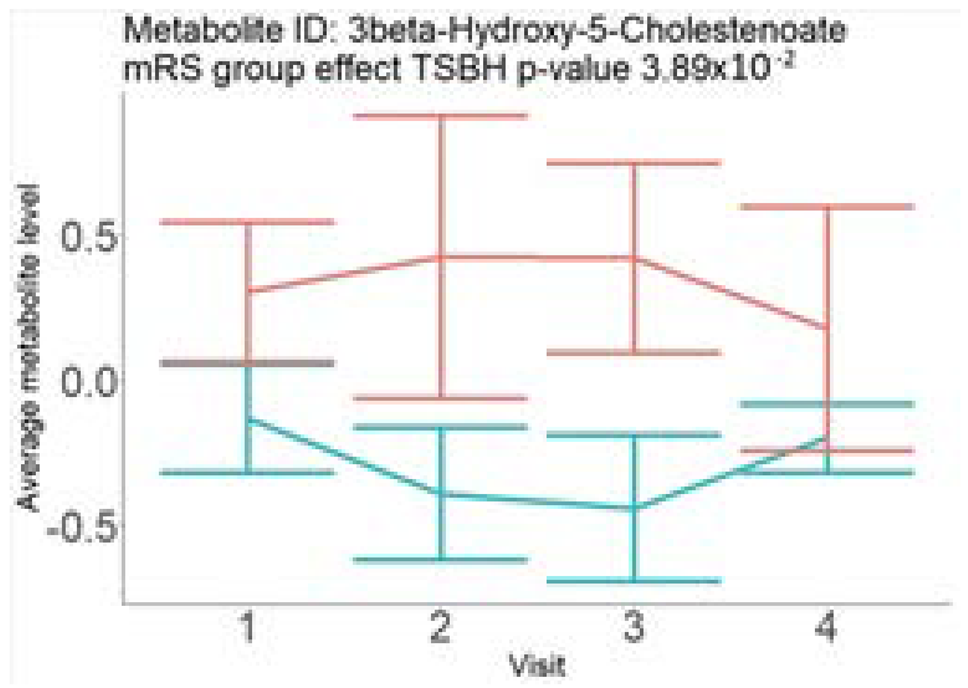
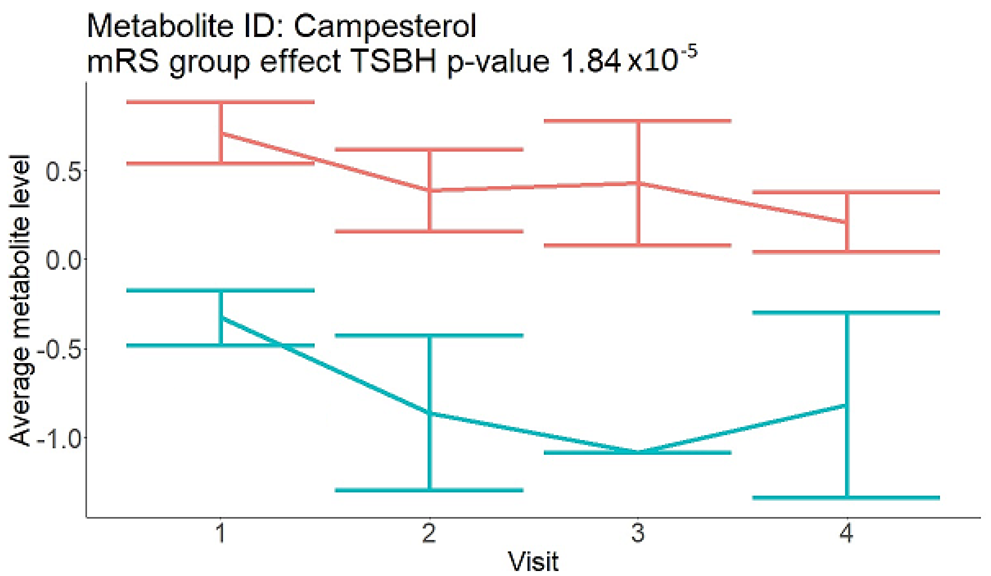
In the longitudinal part, the analysis of all 15 patients did not show metabolites associated with clinical outcomes. We identified several significant metabolites when analyzed patients with small and large infarction volume. In seven patients with small infarction volume (average infarction volume 7.99 ± 6.79 mL, follow-up mRS was 1.57 ± 2.23), we observed that lower levels of sphingomyelin (d18:1/19:0, d19:1/18:0) * correlated with good outcomes, while higher levels correlated with poor outcomes, particular at <24 h time point (p = 0.02) (Table 2). In eight patients with large infarction volume (average infarction volume 97.31 ± 49.06 mL, follow-up mRS was 3.5 ± 2.33), we observed that lower levels of six metabolites significantly correlated with good outcomes, while higher levels correlated with poor outcomes: one derivative of sphingolipids N-palmitoyl-sphingosine (d18:1/16:0) (p = 0.03); two derivatives of phosphatidylethanolamine: 1-palmitoyl-2-docosahexaenoyl-GPC (16:0/22:6) (p = 0.03) and 1-palmitoyl-2-docosahexaenoyl-GPE (p = 0.02); one derivative of glycerol: palmitoyl-docosahexaenoyl-glycerol (16:0/22:6) (p = 0.03); and two derivatives of steroids: campesterol (p < 0.01) and 3beta-hydroxy-5-cholestenoate (p = 0.03) (Table 2).

Table 2.
Change in metabolites after AIS for good and poor outcomes.
4. Discussion
In this study, we identified seven candidate markers of AIS outcomes, five of these markers showed corroborated changes in cross-sectional and longitudinal analysis. All these compounds belong to phospholipids and represent important components of the neural cell membrane. It is important that good AIS outcomes were associated with lower levels of all compounds in cross-sectional and longitudinal analysis, which may indicate the degree of neuronal membrane damage. If such hypothesis is true, there is a possibility that infarcted areas seen on diffusion-weighted MRI still contain functioning nerve cells, which may facilitate recovery.
Although the exact mechanism of change in serum metabolome after AIS is unknown, conventional explanations include diffusion through the interrupted BBB [3] and the activation of the ischemia-mediated pathways [19,20]. These explanations correspond to the findings in our study, particularly for the patients with large infarction volumes. These patients have extensive areas of BBB interruption, which led to a well-organized difference in the levels of six metabolites, observed through multiple time points. In patients with small infarction volume, the mechanism of change in metabolome may be different. These patients have small areas with BBB interruption, reflecting a change in only one metabolite at <24 h (Table 2). In the combined analysis of small- and large-volume patients, these findings could have canceled each other out. A separate analysis for large and small volumes was not possible in the cross-sectional group because all patients had similar infarction volumes.
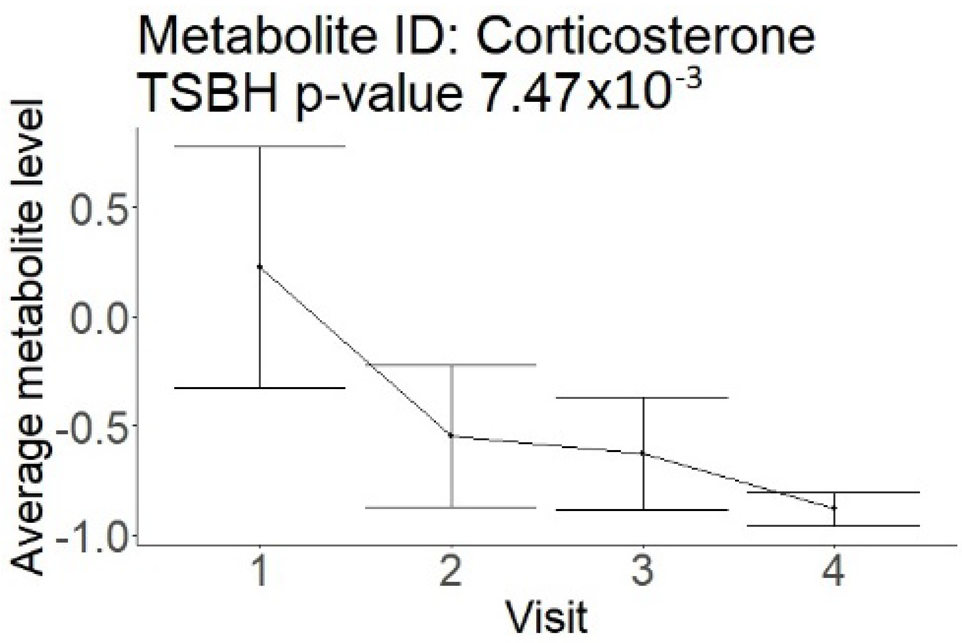
The change in serum metabolome after stroke may be also related to the ischemia-mediated inflammation [4,21,22]. This reaction may or may not be associated with the elevation in traditional inflammatory markers such as C-reactive protein and erythrocyte sedimentation rate. We did not observe a correlation of inflammatory markers with the outcomes in our study, which speaks against such a mechanism. On the other hand, we observed a correlation of good outcomes with lower levels of steroid derivatives in the longitudinal part of this study. This suggests that the diminished inflammatory response may be associated with better recovery. However, the lack of such data in the cross-sectional analysis limits our conclusions. The decrease in lipidomic compounds after AIS may also be related to the poor nutritional status of patients. This happens because AIS patients commonly develop dysphagia, and feeding through nasogastric tubes is not initiated timely. Although this could have affected the study results, poor nutrition status cannot explain the better recovery observed after AIS.
Searching through public metabolome databases, including Cayman, Avanti, PubChem, and others, could not identify the pathophysiological background for the observed changes. N-palmitoyl-sphingosine (d18:1/16:0), also known as Ceramide C16, played role in forming channels in mitochondrial membrane, increased levels in apoptosis, and acute coronary syndrome [23]; 1-palmitoyl-2-docosahexaenoyl-GPC (16:0/22:6) was associated with atherosclerotic plaques [24]. Steroid derivative 3beta-hydroxy-5-cholestenoate was related to the pathophysiology of Alzheimer disease [25]; while Campesterol serves as a steroid precursor [26].
If proven on a larger cohort, the results of our study may be used for the prognostication of ischemic stroke outcomes. Despite the general understanding that the diffusion-weighted sequence of MRI represents permanent brain damage, some case reports indicate the reversibility of such an injury and the presence of viable brain cells within the infarcted area [27,28]. In this case, the association of lower metabolite levels with better functional recovery could indicate the number of viable cells within infarcted area and explain reasonable recovery in some patients with comparatively large strokes. A gradual change in metabolites after ischemic stroke suggests the continuous transformation of ischemic penumbra into the core or metabolic changes within the core even days after the index event. A better understanding of this pathophysiology will not only allow better prediction of outcomes but may also lead to the development of neuroprotective agents in the future [29].
The strengths of our study include the homogeneous patient population of LVO strokes, and the unique study design where serum metabolome was evaluated using NMR and LC-MS technology in separate groups of patients using cross-sectional and longitudinal study designs. This marks out our study among many others that simply compared metabolome between stroke patients and controls. The most important finding of our study is that lower levels of lipidomic compounds were uniformly associated with better outcomes across different analytic platforms and study designs. Limitations include a comparatively small sample size and wide variation in the infarction volumes in the longitudinal cohort. Also, the difference between NMR and LC-MS panels did not allow for evaluating all compounds. It is important to stress that the results of this study should be viewed as corroborated changes in the cross-sectional and longitudinal parts along with significant findings after TSBH application. Separate findings from either part of this study are not convincing. Our findings need to be validated on large-scale studies before any final conclusions are made.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/neurolint17020030/s1, Table S1: Metabolites analyzed in cross-sectional part using NMR.
Author Contributions
Conceptualization, E.V.S. and D.K.S.; methodology, E.V.S.; software, C.X.; validation, K.S.; formal analysis, C.X.; resources, E.V.S. and D.K.S.; writing—original draft preparation, E.V.S.; visualization, K.S.; supervision, D.K.S.; funding acquisition E.V.S. and D.K.S. All authors have read and agreed to the published version of the manuscript.
Funding
This study was partly supported by NIH (R01DK082766), Presbyterian Health Foundation Team Science Award, College of Medicine Alumni Association Award (COMAA), and the Leinbach Seed Grant.
Institutional Review Board Statement
This study was approved by The University of Oklahoma Health Sciences Center Institutional Review Board IRB# 5349 on 28 July 2024.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
All data are available upon request.
Acknowledgments
The authors thank all the participants of MISS who made this study possible. The authors would also like to acknowledge April Vaughan for recruiting patients and the technical assistance provided by Cynthia Bejar.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the american heart association/american stroke association. Stroke 2019, 50, e344–e418. [Google Scholar] [PubMed]
- Sue Nelson, L.W. Projections of Cardiovascular Disease Prevalence and Costs: 2015–2035; RTI International: Research Triangle Park, NC, USA, 2016. [Google Scholar]
- Candelario-Jalil, E.; Dijkhuizen, R.M.; Magnus, T. Neuroinflammation, stroke, blood-brain barrier dysfunction, and imaging modalities. Stroke 2022, 53, 1473–1486. [Google Scholar] [CrossRef] [PubMed]
- Sidorov, E.; Sanghera, D.K.; Vanamala, J.K.P. Biomarker for ischemic stroke using metabolome: A clinician perspective. J. Stroke 2019, 21, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Baranovicova, E.; Kalenska, D.; Kaplan, P.; Kovalska, M.; Tatarkova, Z.; Lehotsky, J. Blood and brain metabolites after cerebral ischemia. Int. J. Mol. Sci. 2023, 24, 17302. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Gui, X.; Wu, L.; Tian, S.; Wang, H.; Xie, L.; Wu, W. Amino acid metabolism, lipid metabolism, and oxidative stress are associated with post-stroke depression: A metabonomics study. BMC Neurol. 2020, 20, 250. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhou, K.; Li, H.; Dong, X.; Tan, G.; Chai, Y.; Wang, W.; Bi, X. Potential of serum metabolites for diagnosing post-stroke cognitive impairment. Mol. Biosyst. 2015, 11, 3287–3296. [Google Scholar] [CrossRef]
- Chi, N.F.; Chang, T.H.; Lee, C.Y.; Wu, Y.W.; Shen, T.A.; Chan, L.; Chen, Y.R.; Chiou, H.Y.; Hsu, C.Y.; Hu, C.J. Untargeted metabolomics predicts the functional outcome of ischemic stroke. J. Formos. Med. Assoc. 2021, 120, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Harshfield, E.L.; Sands, C.J.; Tuladhar, A.M.; de Leeuw, F.E.; Lewis, M.R.; Markus, H.S. Metabolomic profiling in small vessel disease identifies multiple associations with disease severity. Brain 2022, 145, 2461–2471. [Google Scholar] [CrossRef] [PubMed]
- Sidorov, E.V.; Rout, M.; Xu, C.; Jordan, L.; Fields, E.; Apple, B.; Smith, K.; Gordon, D.; Chainakul, J.; Sanghera, D.K. Difference in acute and chronic stage ischemic stroke metabolic markers with controls. J. Stroke Cerebrovasc. Dis. 2023, 32, 107211. [Google Scholar] [CrossRef]
- Sidorov, E.V.; Bejar, C.; Xu, C.; Ray, B.; Gordon, D.; Chainakul, J.; Sanghera, D.K. Novel metabolites as potential indicators of ischemic infarction volume: A pilot study. Transl. Stroke Res. 2020, 12, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Santucci, J.A.; Ross, S.R.; Greenert, J.C.; Aghaei, F.; Ford, L.; Hollabaugh, K.M.; Cornwell, B.O.; Wu, D.H.; Zheng, B.; Bohnstedt, B.N.; et al. Radiological estimation of intracranial blood volume and occurrence of hydrocephalus determines stress-induced hyperglycemia after aneurysmal subarachnoid hemorrhage. Transl. Stroke Res. 2018, 10, 327–337. [Google Scholar] [CrossRef] [PubMed]
- von Kummer, R.; Broderick, J.P.; Campbell, B.C.; Demchuk, A.; Goyal, M.; Hill, M.D.; Treurniet, K.M.; Majoie, C.B.; Marquering, H.A.; Mazya, M.V.; et al. The heidelberg bleeding classification: Classification of bleeding events after ischemic stroke and reperfusion therapy. Stroke 2015, 46, 2981–2986. [Google Scholar] [CrossRef]
- Würtz, P.; Havulinna, A.S.; Soininen, P.; Tynkkynen, T.; Prieto-Merino, D.; Tillin, T.; Ghorbani, A.; Artati, A.; Wang, Q.; Tiainen, M.; et al. Metabolite profiling and cardiovascular event risk: A prospective study of 3 population-based cohorts. Circulation 2015, 131, 774–785. [Google Scholar] [CrossRef]
- Fischer, K.; Kettunen, J.; Würtz, P.; Haller, T.; Havulinna, A.S.; Kangas, A.J.; Soininen, P.; Esko, T.; Tammesoo, M.L.; Mägi, R.; et al. Biomarker profiling by nuclear magnetic resonance spectroscopy for the prediction of all-cause mortality: An observational study of 17,345 persons. PLoS Med. 2014, 11, e1001606. [Google Scholar] [CrossRef]
- Soininen, P.; Kangas, A.J.; Wurtz, P.; Suna, T.; Ala-Korpela, M. Quantitative serum nuclear magnetic resonance metabolomics in cardiovascular epidemiology and genetics. Circ. Cardiovasc. Genet. 2015, 8, 192–206. [Google Scholar] [CrossRef]
- Soininen, P.; Kangas, A.J.; Würtz, P.; Tukiainen, T.; Tynkkynen, T.; Laatikainen, R.; Järvelin, M.R.; Kähönen, M.; Lehtimäki, T.; Viikari, J.; et al. High-throughput serum nmr metabonomics for cost-effective holistic studies on systemic metabolism. Analyst 2009, 134, 1781–1785. [Google Scholar] [CrossRef] [PubMed]
- Dehaven, C.D.; Evans, A.M.; Dai, H.; Lawton, K.A. Organization of gc/ms and lc/ms metabolomics data into chemical libraries. J. Cheminform. 2010, 2, 9. [Google Scholar] [CrossRef]
- Jung, J.; Park, M.; Park, H.J.; Shim, S.B.; Cho, Y.H.; Kim, J.; Lee, H.S.; Choi, D.; Hwang, G.S. 1h nmr-based metabolic profiling of naproxen-induced toxicity in rats. Toxicol. Lett. 2011, 200, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Zhang, L.; Pu, H.; Mao, L.; Hu, X.; Jiang, X.; Xu, N.; Stetler, R.A.; Zhang, F.; Liu, X.; et al. Rapid endothelial cytoskeletal reorganization enables early blood-brain barrier disruption and long-term ischaemic reperfusion brain injury. Nat. Commun. 2016, 7, 10523. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Vaughan, A.; Sidorov, E.V.; Sanghera, D.K. Improving stroke outcome prediction using molecular and machine learning approaches in large vessel occlusion. J. Clin. Med. 2024, 13, 5917. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Vaughan, A.; Blair, A.; Stavrakis, S.; Sidorov, E.V.; Sanghera, D.K. Discovery and validation of circulating stroke metabolites by nmr-based analyses using patients from the miss and uk biobank. Neurochem. Int. 2023, 169, 105588. [Google Scholar] [CrossRef]
- Anroedh, S.; Hilvo, M.; Akkerhuis, K.M.; Kauhanen, D.; Koistinen, K.; Oemrawsingh, R.; Serruys, P.; van Geuns, R.J.; Boersma, E.; Laaksonen, R.; et al. Plasma concentrations of molecular lipid species predict long-term clinical outcome in coronary artery disease patients. J. Lipid Res. 2018, 59, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.; Koster, G.; Douet, L.J.; Scigelova, M.; Woffendin, G.; Ward, J.M.; Smith, A.; Humphries, J.; Burnand, K.G.; Macphee, C.H.; et al. Electrospray ionization mass spectrometry identifies substrates and products of lipoprotein-associated phospholipase a2 in oxidized human low density lipoprotein. J. Biol. Chem. 2008, 283, 6428–6437. [Google Scholar] [CrossRef]
- Jung, J.I.; Price, A.R.; Ladd, T.B.; Ran, Y.; Park, H.J.; Ceballos-Diaz, C.; Smithson, L.A.; Hochhaus, G.; Tang, Y.; Akula, R.; et al. Cholestenoic acid, an endogenous cholesterol metabolite, is a potent gamma-secretase modulator. Mol. Neurodegener. 2015, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Dammann, C.; Stapelfeld, C.; Maser, E. Expression and activity of the cortisol-activating enzyme 11beta-hydroxysteroid dehydrogenase type 1 is tissue and species-specific. Chem. Biol. Interact. 2019, 303, 57–61. [Google Scholar] [CrossRef]
- Tamura, G.; Ihara, S.; Morota, N. Reversible diffusion weighted imaging hyperintensities during the acute phase of ischemic stroke in pediatric moyamoya disease: A case report. Childs Nerv. Syst. 2016, 32, 1531–1535. [Google Scholar] [CrossRef] [PubMed]
- Yue, P.; Dongmei, W.; Zhenzhou, L.; Yongming, W.U.; Zhong, J.I. Diffusion-weighted imaging hyperintensity is reversible in large middle cerebral artery infarction following thrombectomy: A case report. J. South. Med. Univ. 2020, 40, 459–462. [Google Scholar]
- Li, Y.; Schappell, L.E.; Polizu, C.; DiPersio, J.; Tsirka, S.E.; Halterman, M.W.; Nadkarni, N.A. Evolving clinical-translational investigations of cerebroprotection in ischemic stroke. J. Clin. Med. 2023, 12, 6715. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).