Randomized Clinical Trial for the Optimization of Dyslipidemia Management in Patients with End-Stage Renal Disease Undergoing In-Hospital Maintenance Hemodialysis Therapy
Abstract
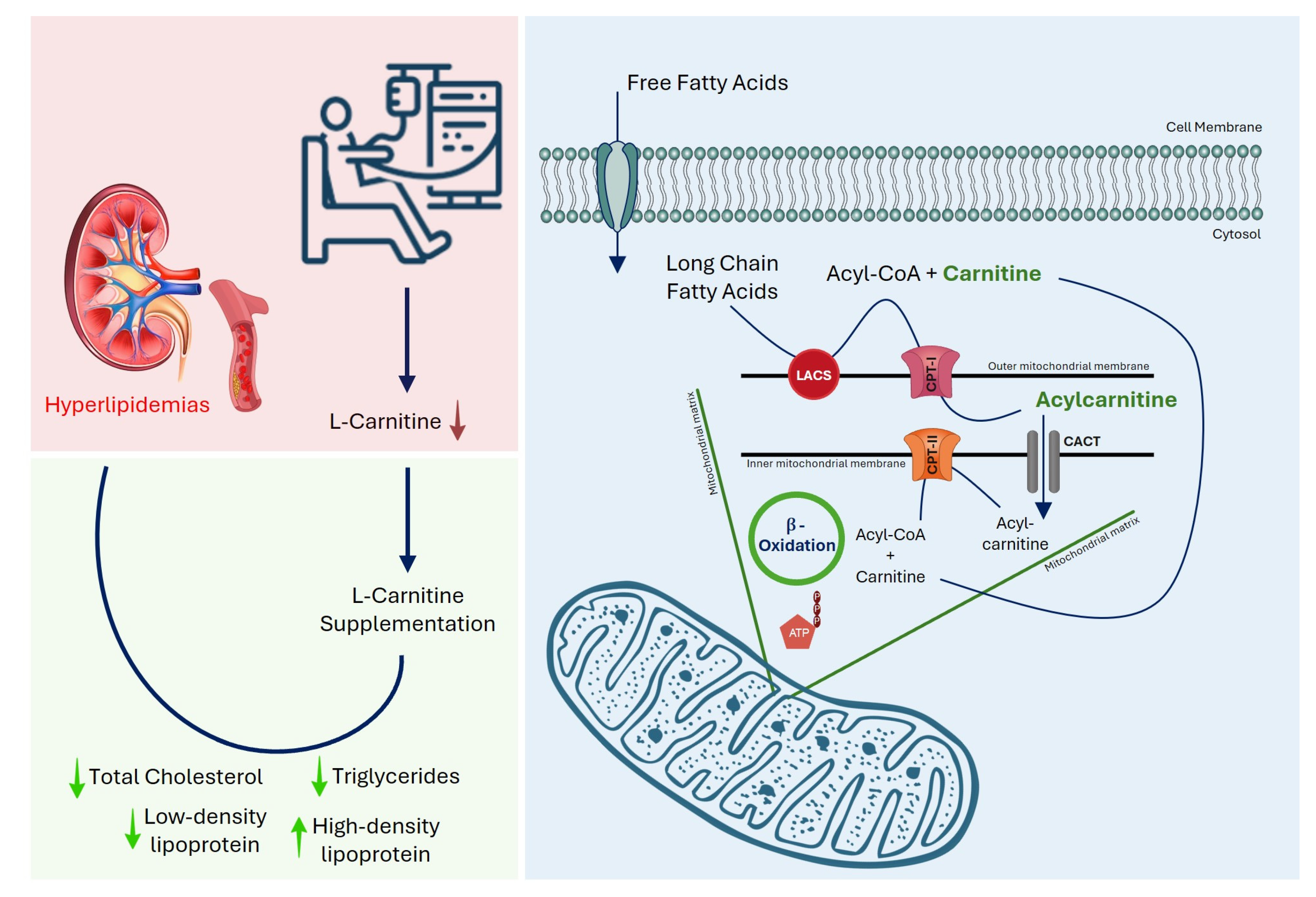
1. Introduction
2. Materials and Methods
2.1. Setting and Participants
2.2. Inclusion and Exclusion Criteria
2.3. Randomization
2.4. Interventions
2.5. Monitoring and Follow-Up
2.6. Statistical Analysis
3. Results
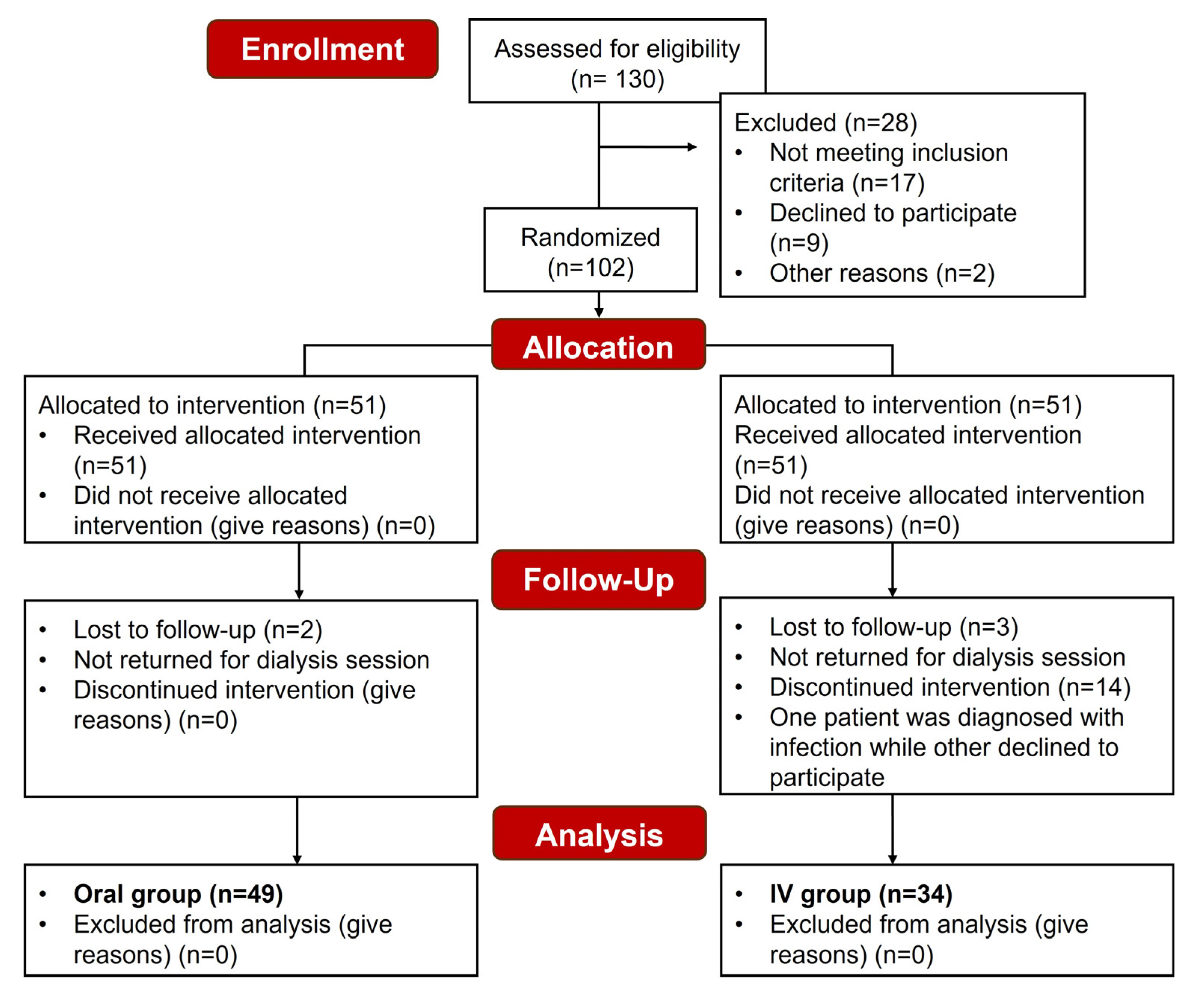
3.1. Study Population and Demographics
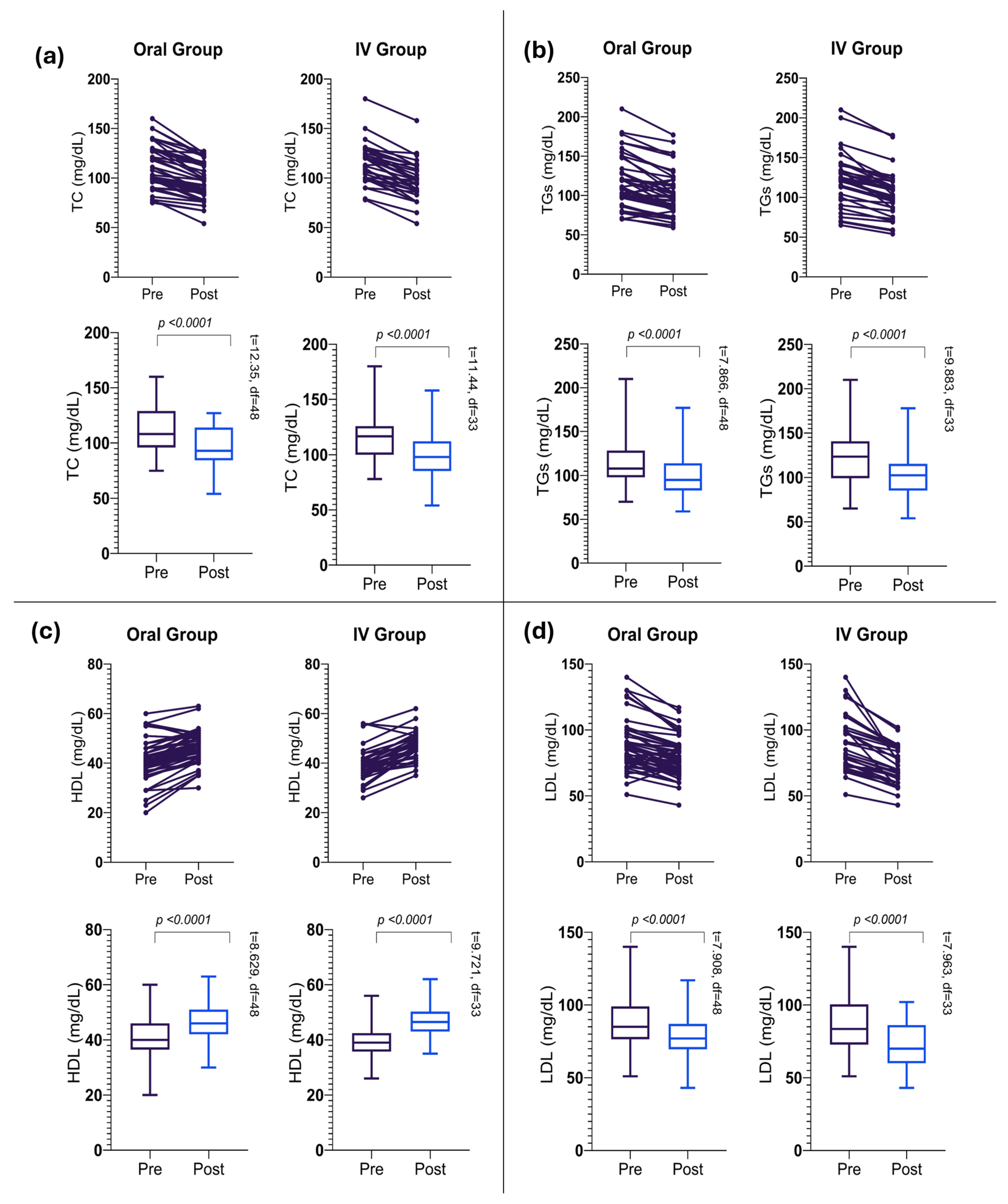
3.2. Changes in Lipid Profile Parameters in the Oral Group
3.3. Changes in Lipid Profile Parameters in the IV Group
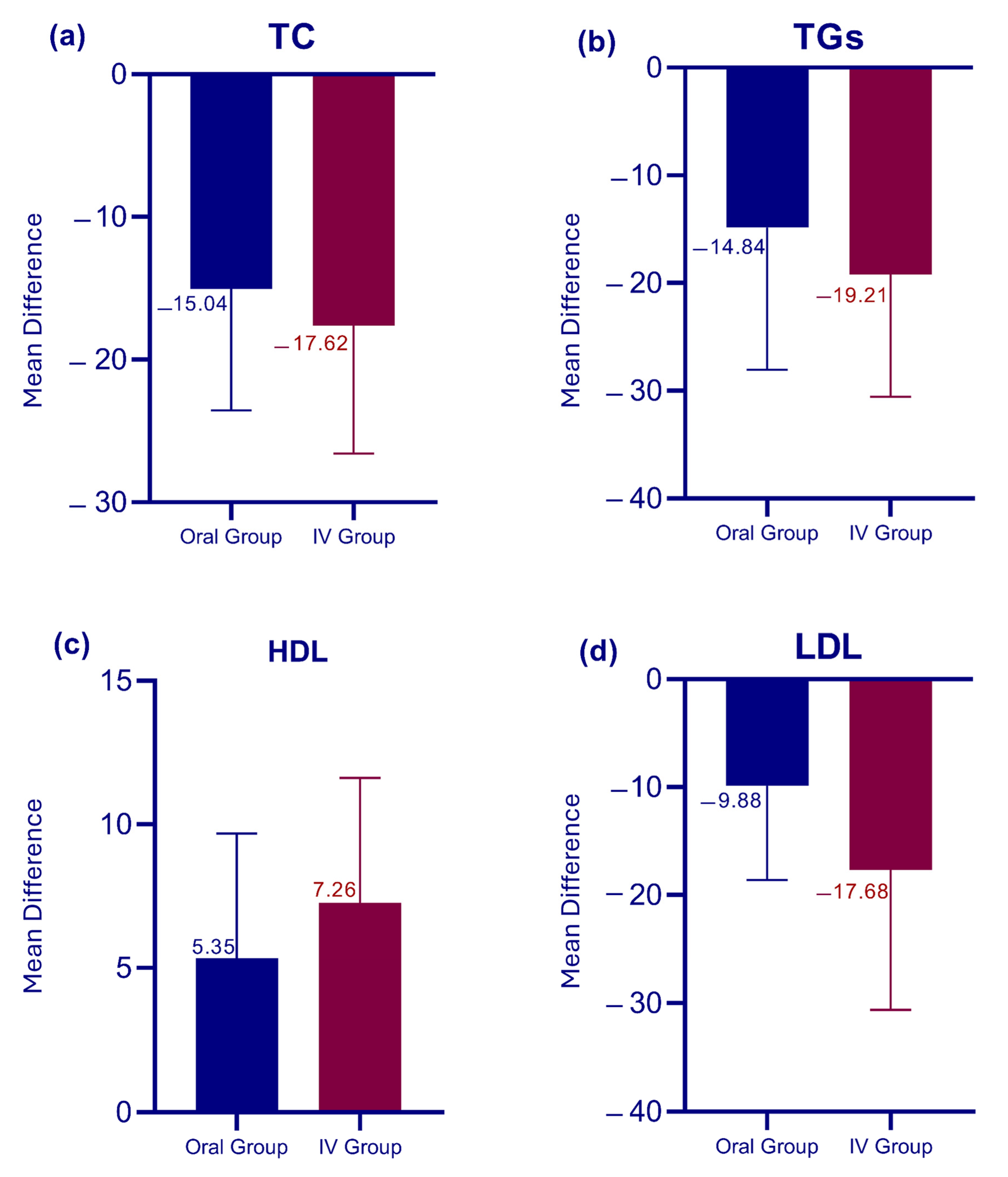
3.4. Intervention Outcomes and Group Comparison
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jain, N.; Bansal, R.; Saxena, S.; Sharma, S.; Raju, S.B. Predictors of functional impairment and mortality in patients on maintenance hemodialysis. Semin. Dial. 2024, 37, 138–144. [Google Scholar] [CrossRef]
- Montoya-Ortiz, B.; Bonilla, G. Perioperative management of renal disease, dialysis, and transplantation in arthroplasty patients. In Fast Track Surgery in Hip and Knee Arthroplasty: New Standards; Springer: Cham, Switzerland, 2024; pp. 173–179. [Google Scholar]
- Theofilis, P.; Vordoni, A.; Koukoulaki, M.; Vlachopanos, G.; Kalaitzidis, R.G. Dyslipidemia in chronic kidney disease: Contemporary concepts and future therapeutic perspectives. Am. J. Nephrol. 2021, 52, 693–701. [Google Scholar] [CrossRef]
- Shaukat, D.; Haroon, Z.H.; Aamir, M.; Binkhalid, U. Role of total carnitine to free carnitine ratio (TC/FC) in monitoring outcome of haemodialysis. Int. J. Pathol. 2021, 17, 111–116. [Google Scholar]
- Lo, H.Y.; Lin, Y.S.; Lin, D.S.; Lee, J.K.; Chen, W.J. Association of statin therapy with major adverse cardiovascular and limb outcomes in patients with end-stage kidney disease and peripheral artery disease receiving maintenance dialysis. JAMA Netw. Open 2022, 5, e2230618. [Google Scholar] [CrossRef] [PubMed]
- Pan, X. Cholesterol metabolism in chronic kidney disease: Physiology, pathologic mechanisms, and treatment. In Sphingolipid Metabolism and Metabolic Disease; Springer: Singapore, 2022; pp. 119–143. [Google Scholar]
- Leitão, A.M.F.; Silva, B.R.; Barbalho, E.C.; Paulino, L.R.; das Chagas Costa, F.; Martins, F.S.; Silva, J.R.V. The role of L-carnitine in the control of oxidative stress and lipid β-oxidation during in vitro follicle growth, oocyte maturation, embryonic development, and cryopreservation: A review. Zygote 2024, 32, 335–340. [Google Scholar] [CrossRef]
- Romero, J.M.; Acevedo, R.M.; Merino, J.I.; Vargas, F.P.; Peña, N.I.; Arreola, F.O.; Arias, A.L.A.; Quiroz, J.G.B.; Guillén, P.H.; Monroy, L.F.T. Hospitalization causes and epidemiological characteristics among patients with end-stage renal disease: A six-year retrospective study. Cureus 2024, 16, e67284. [Google Scholar]
- Ulinski, T.; Cirulli, M.; Virmani, M.A. The role of L-carnitine in kidney disease and related metabolic dysfunctions. Kidney Dial. 2023, 3, 178–191. [Google Scholar] [CrossRef]
- Yeh, Y.T.; Sung, F.C.; Tsai, C.F.; Hsu, C.C.; Tsai, W.C.; Hsu, Y.H. Statin therapy associated mortality in hyperlipidemic dialysis patients with percutaneous coronary intervention for acute myocardial infarction: A retrospective cohort study. Heliyon 2024, 10, e35287. [Google Scholar] [CrossRef]
- Lin, C.P.; Hsiao, F.C.; Tung, Y.C.; Hsu, T.J.; Chou, S.H.; Lin, Y.S.; Chen, S.W.; Chu, P.H. Intravascular imaging-guided percutaneous coronary intervention in patients with end-stage renal disease on maintenance dialysis. JACC Asia 2025, 5, 28–41. [Google Scholar] [CrossRef]
- Ma, X.; Yan, D.; Zhou, C.; Shi, Y.; Wang, Y.; Li, J.; Zhong, Q.; Li, X.; Hu, Y.; Liang, W.; et al. The correlation between protein energy wasting and the incidence of main adverse cardiovascular events in adult maintenance hemodialysis patients: A single-center retrospective cohort study. Ren. Fail. 2025, 47, 2441399. [Google Scholar] [CrossRef] [PubMed]
- Hamedi-Kalajahi, F.; Zarezadeh, M.; Mojtahedi, S.Y.; Shabbidar, S.; Fahimi, D.; Imani, H. Effect of L-carnitine supplementation on lipid profile and apolipoproteins in children on hemodialysis: A randomized placebo-controlled clinical trial. Pediatr. Nephrol. 2021, 36, 3741–3747. [Google Scholar] [CrossRef]
- Musazadeh, V.; Alinejad, H.; Esfahani, N.K.; Kavyani, Z.; Keramati, M.; Roshanravan, N.; Mosharkesh, E.; Dehghan, P. The effect of L-carnitine supplementation on lipid profile in adults: An umbrella meta-analysis on interventional meta-analyses. Front. Nutr. 2023, 10, 1214734. [Google Scholar] [CrossRef]
- Karimi, M.; Pirzad, S.; Pourfaraji, S.M.A.; Parhizkar Roudsari, P.; Shirsalimi, N.; Ahmadizad, S. Effects of L-carnitine supplementation on lipid profile in adult patients under hemodialysis: A systematic review and meta-analysis of RCTs. Front. Med. 2024, 11, 1454921. [Google Scholar] [CrossRef]
- Cohen-Hagai, K.; Benchetrit, S.; Wand, O.; Grupper, A.; Shashar, M.; Solo, O.; Pereg, D.; Zitman-Gal, T.; Haskiah, F.; Erez, D. The clinical significance of LDL-cholesterol on the outcomes of hemodialysis patients with acute coronary syndrome. Medicina 2023, 59, 1312. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xue, C.; Ou, J.; Xie, Z.; Deng, J. Effect of L-carnitine supplementation on renal anemia in patients on hemodialysis: A meta-analysis. Int. Urol. Nephrol. 2021, 53, 2149–2158. [Google Scholar] [CrossRef]
- Rehman, S.; Farhan, M.; Kumar, S.; Sarfraz, M.R.; Naveed, A.; Khan, M.T.; Huq, H. Efficacy of intravenous versus oral administration of levocarnitine in maintenance hemodialysis patients: A randomized controlled trial investigating therapeutic approaches to renal anemia. Health Sci. Rep. 2025, 8, e70297. [Google Scholar] [CrossRef]
- Rehman, S.; Farhan, M.; Naveed, A.; Mahmood, S.; Khattak, M.I. Impact of haemodialysis on plasma carnitine concentrations in haemodialysis patients. J. Pak. Med. Assoc. 2024, 74, 294. [Google Scholar] [CrossRef]
- Huang, C.; Chen, Y.; Wang, X.; Li, J.; Zhang, Y. Effects of L-carnitine supplementation on lipid profiles in hemodialysis patients: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2023, 33, 45–52. [Google Scholar]
- Li, Y.; Xie, Y.; Qiu, C.; Yu, B.; Yang, F.; Cheng, Y.; Zhong, W.; Yuan, J. Effects of L-carnitine supplementation on lipid metabolism: A systematic review and meta-analysis of randomized controlled trials. Food Funct. 2023, 14, 2502–2517. [Google Scholar] [CrossRef] [PubMed]
- Dehkordi, S.R.; Malekahmadi, M.; Nikbaf-Shandiz, M.; Rasaei, N.; Hosseini, A.M.; Bahari, H.; Rastgoo, S.; Asbaghi, O.; Shiraseb, F.; Behmadi, R. The effects of L-carnitine supplementation on lipid profiles in adults: A systematic review and dose–response meta-analysis. PharmaNutrition 2024, 27, 100374. [Google Scholar] [CrossRef]
- Lee, B.J.; Lin, J.S.; Lin, Y.C.; Lin, P.T. Effects of L-carnitine supplementation on lipid profiles in patients with coronary artery disease. Lipids Health Dis. 2016, 15, 107. [Google Scholar] [CrossRef]
- Sugiyama, H.; Maeshima, Y.; Yamasaki, Y.; Makino, H. L-Carnitine treatment of renal diseases: Mechanisms and clinical applications. Kidney Int. Suppl. 2021, 119, S16–S23. [Google Scholar]
- Li, N.; Zhao, H. Role of carnitine in non-alcoholic fatty liver disease and other related diseases: An update. Front. Med. 2021, 8, 689042. [Google Scholar] [CrossRef]
- Mercadal, L.; Coudert, M.; Vassault, A.; Pieroni, L.; Debure, A.; Ouziala, M.; Depreneuf, H.; Fumeron, C.; Servais, A.; Bassilios, N.; et al. L-carnitine treatment in incident hemodialysis patients: The multicenter, randomized, double-blinded, placebo-controlled CARNIDIAL trial. Clin. J. Am. Soc. Nephrol. 2012, 7, 1836–1842. [Google Scholar] [CrossRef]
- Fu, R.G.; Wang, L.; Zhou, J.P.; Ma, F.; Liu, X.D.; Ge, H.; Zhang, J. The effect of levocarnitine on nutritional status and lipid metabolism during long-term maintenance hemodialysis. Acad. J. Xi’an Jiaotong Univ. 2010, 22, 203–207. [Google Scholar]
- Singh, H.; Jain, D.; Bhaduri, G.; Gupta, N.; Sangwan, R. Study on effects of L-carnitine supplementation on anaemia with erythropoietin hyporesponsiveness and lipid profile in chronic kidney disease patients on maintenance haemodialysis. Indian J. Basic Appl. Med. Res. 2020, 9, 224–232. [Google Scholar]
- Yang, S.K.; Xiao, L.; Song, P.A.; Xu, X.; Liu, F.Y.; Sun, L. Effect of L-carnitine therapy on patients in maintenance hemodialysis: A systematic review and meta-analysis. J. Nephrol. 2014, 27, 317–329. [Google Scholar] [CrossRef]
- Shayanpour, S.; Ghobadi, S.; Ghaffari, S.; Ahmadi, S.; Amirkafi, A. Effect of oral L-carnitine on oxidative stress and lipid profile in hemodialysis patients: A randomized controlled trial. Gazi Med. J. 2024, 35, 20–25. [Google Scholar] [CrossRef]
- Emami Naini, A.; Moradi, M.; Mortazavi, M.; Amini Harandi, A.; Hadizadeh, M.; Shirani, F.; Ghafoori, H.B.; Naini, P.E. Effects of oral L-carnitine supplementation on lipid profile, anemia, and quality of life in chronic renal disease patients under hemodialysis: A randomized, double-blinded, placebo-controlled trial. J. Nutr. Metab. 2012, 2012, 510483. [Google Scholar] [CrossRef] [PubMed]
- Weschler, A.; Aviram, M.; Levin, M.; Better, O.S.; Brook, G. High dose of L-carnitine increases platelet aggregation and plasma triglyceride levels in uremic patients on hemodialysis. Nephron 1984, 38, 120–124. [Google Scholar] [CrossRef]
- Katalinic, L.; Krtalic, B.; Jelakovic, B.; Basic-Jukic, N. The unexpected effects of LC supplementation on lipid metabolism in hemodialysis patients. Kidney Blood Press. Res. 2018, 43, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI clinical practice guideline for nutrition in CKD: 2020 update. Am. J. Kidney Dis. 2020, 76, S1–S107. [Google Scholar] [CrossRef] [PubMed]
- Fukami, K.; Yamagishi, S.I.; Sakai, K.; Nasu, M.; Okuda, S. Effects of switching from oral administration to intravenous injection of L-carnitine on lipid metabolism in hemodialysis patients. Clin. Kidney J. 2014, 7, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Verrina, E.; Caruso, U.; Calevo, M.G.; Emma, F.; Sorino, P.; De Palo, T.; Lavoratti, G.; Dertenois, L.T.; Cassanello, M.; Cerone, R.; et al. Effect of carnitine supplementation on lipid profile and anemia in children on chronic dialysis. Pediatr. Nephrol. 2007, 22, 727–733. [Google Scholar] [CrossRef] [PubMed]
| Patient Characteristics | Oral Group (n = 49) | IV Group (n = 34) | p-Value |
|---|---|---|---|
| Male, n (%) | 25 (60.98%) | 16 (57.41%) | 0.89 1 |
| Female, n (%) | 24 (39.02%) | 18 (42.86) | |
| Age (years), Mean ± SD | 44.1 ± 8.92 | 45.62 ± 8.61 | 0.44 2 |
| BMI, Mean ± SD | 25.51 ± 4.69 | 24.55 ± 5.09 | 0.34 2 |
| Co-morbidities, n (%) | |||
| Diabetes | 8 (16.3%) | 6 (17.6%) | 0.25 1 |
| Hypertension | 23 (46.9%) | 14 (41.2%) | |
| Cardiovascular disorders | 2 (4.1%) | 6 (17.6%) | |
| None | 16 (32.7%) | 8 (23.5%) | |
| Variables | Oral Group (n = 49) | |||||
|---|---|---|---|---|---|---|
| Pre-Intervention | Post-Intervention | Differences in the Value | 95% CI | t-Value | p-Value | |
| Mean ± SD | ||||||
| TC, mg/dL | 111.33 ± 21.92 | 96.29 ± 18.11 | −15.04 ± 8.52 | −17.49 to −12.59 | 12.35 | <0.0001 * |
| TGs, mg/dL | 115.39 ± 31.27 | 100.55 ± 27.01 | −14.84 ± 13.20 | −18.63 to −11.04 | 7.87 | <0.0001 * |
| LDL, mg/dL | 88.92 ± 19.00 | 79.04 ± 14.92 | −9.87 ± 8.74 | −12.39 to −7.36 | 7.91 | <0.0001 * |
| HDL, mg/dL | 40.90 ± 8.36 | 46.24 ± 6.45 | 5.34 ± 4.33 | 4.11 to 6.59 | 8.63 | <0.0001 * |
| Variables | Oral Group (n = 34) | |||||
|---|---|---|---|---|---|---|
| Pre-Intervention | Post-Intervention | Differences in the Value | 95% CI | t-Value | p-Value | |
| Mean ± SD | ||||||
| TC, mg/dL | 114.47 ± 20.12 | 96.85 ± 19.82 | −17.62 ± 8.98 | −20.75 to −14.48 | 11.44 | <0.0001 * |
| TGs, mg/dL | 122.53 ± 33.60 | 103.32 ± 28.61 | −19.21 ± 11.33 | −23.16 to −15.25 | 9.88 | <0.0001 * |
| LDL, mg/dL | 89.59 ± 21.10 | 71.91 ± 14.37 | −17.68 ± 12.94 | −22.19 to −13.16 | 7.96 | <0.0001 * |
| HDL, mg/dL | 39.56 ± 6.95 | 46.82 ± 5.72 | 7.26 ± 4.35 | 5.74 to 8.79 | 9.72 | <0.0001 * |
| Test Results | Pre-Intervention | Post-Intervention | ||||||
|---|---|---|---|---|---|---|---|---|
| Oral Group | IV Group | t-Value | p-Value | Oral Group | IV Group | t-Value | p-Value | |
| TC, mg/dL | 111.33 ± 21.92 | 114.47 ± 20.12 | −0.66 | 0.51 | 96.29 ± 18.11 | 96.85 ± 19.82 | −0.13 | 0.89 |
| TGs, mg/dL | 115.39 ± 31.27 | 122.53 ± 33.60 | −0.99 | 0.32 | 100.55 ± 27.01 | 103.32 ± 28.61 | −0.45 | 0.65 |
| LDL, mg/dL | 88.92 ± 19.00 | 89.59 ± 21.10 | −0.16 | 0.88 | 79.04 ± 14.92 | 71.91 ± 14.37 | 2.19 | 0.03 * |
| HDL, mg/dL | 40.90 ± 8.36 | 39.56 ± 6.95 | 0.80 | 0.44 | 46.24 ± 6.45 | 46.82 ± 5.72 | −0.34 | 0.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rehman, S.; Farhan, M.; Sarfraz, M.R.; Naveed, A.; Usman, F.; Bibi, A.; Ahmed, R.; Huq, H.; Hasan, A.; Anzoom, J.; et al. Randomized Clinical Trial for the Optimization of Dyslipidemia Management in Patients with End-Stage Renal Disease Undergoing In-Hospital Maintenance Hemodialysis Therapy. Pharmaceutics 2025, 17, 1128. https://doi.org/10.3390/pharmaceutics17091128
Rehman S, Farhan M, Sarfraz MR, Naveed A, Usman F, Bibi A, Ahmed R, Huq H, Hasan A, Anzoom J, et al. Randomized Clinical Trial for the Optimization of Dyslipidemia Management in Patients with End-Stage Renal Disease Undergoing In-Hospital Maintenance Hemodialysis Therapy. Pharmaceutics. 2025; 17(9):1128. https://doi.org/10.3390/pharmaceutics17091128
Chicago/Turabian StyleRehman, Sadia, Muhammad Farhan, Muhammad Raza Sarfraz, Asma Naveed, Fahad Usman, Anila Bibi, Raheel Ahmed, Hiya Huq, Ali Hasan, Jarin Anzoom, and et al. 2025. "Randomized Clinical Trial for the Optimization of Dyslipidemia Management in Patients with End-Stage Renal Disease Undergoing In-Hospital Maintenance Hemodialysis Therapy" Pharmaceutics 17, no. 9: 1128. https://doi.org/10.3390/pharmaceutics17091128
APA StyleRehman, S., Farhan, M., Sarfraz, M. R., Naveed, A., Usman, F., Bibi, A., Ahmed, R., Huq, H., Hasan, A., Anzoom, J., & Kumar, P. (2025). Randomized Clinical Trial for the Optimization of Dyslipidemia Management in Patients with End-Stage Renal Disease Undergoing In-Hospital Maintenance Hemodialysis Therapy. Pharmaceutics, 17(9), 1128. https://doi.org/10.3390/pharmaceutics17091128








