Therapeutic Effect of Membrane Vesicle Drug Delivery Systems in Inflammatory Bowel Disease
Abstract
1. Introduction
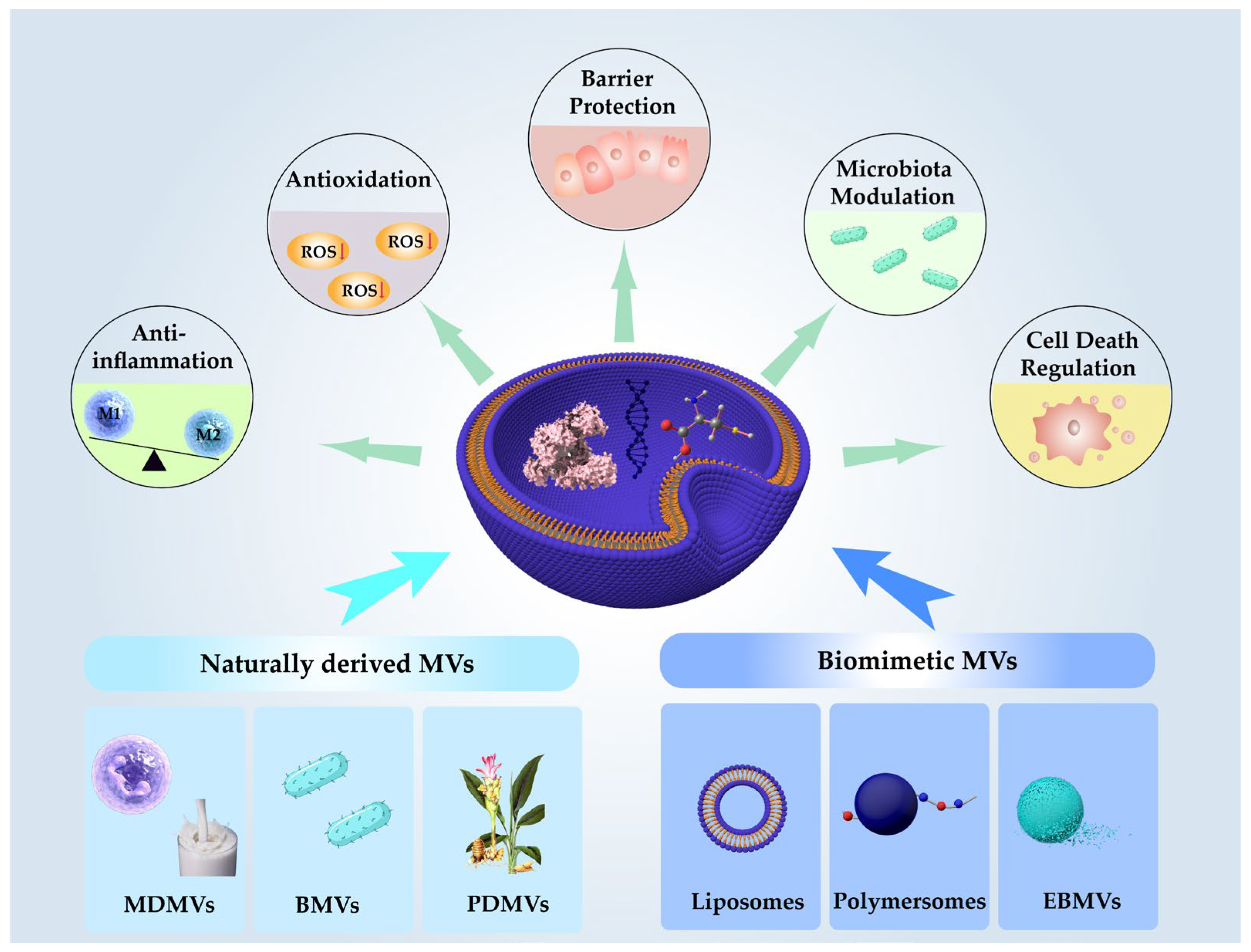
2. MVs
2.1. Overview of MVs
2.2. Classification of MVs
2.2.1. MDMVs
| Origin | Disease | Cargo | Target | Function | Ref. |
|---|---|---|---|---|---|
| hucMSCs | DSS-induced IBD | miR-378a-5p | NLRP3 | Inhibited NLRP3 inflammasome activation, reduced ASC and caspase-1, suppressed IL-1β and IL-18 | [53] |
| hucMSCs | DSS-induced IBD | N.D. | inflammatory cytokines, macrophages | Reduced TNF-α/IL-1β/IL-6/IL-7/iNOS, increased IL-10, decreased M1 macrophages, improved epithelial proliferation | [54] |
| MSCs | TNBS-induced IBD | miR-378a-3p | GATA2, AQP4, PPAR-α | Suppressed GATA2/AQP4/PPAR-α signaling, reduced apoptosis, improved tissue integrity | [55] |
| hucMSCs | DSS/TNBS-induced IBD | TSG-6 | tight junction, T cells, pro-inflammatory cytokines | Enhanced tight junctions, inhibited Th17 and pro-inflammatory cytokines, promoted Th2/anti-inflammatory cytokines, improved barrier | [56] |
| hADSC | DSS-induced IBD | N.D. | ISCs, inflammatory cytokines | Promoted Lgr5+ ISC regeneration, reduced pro-inflammatory cytokines, increased IL-10/IL-13, protected barrier | [48] |
| hucMSCs | DSS-induced IBD | miR-326 | NEDD8, NF-κB | Inhibited NEDD8-Cullin1 binding, reduced neddylation-related enzymes (NAe1/Uba3/UBC12F/DCNL1), suppressed NF-κB, downregulated IL-1β/IL-6 | [57] |
| BMSCs | DSS-induced IBD | N.D. | macrophage, inflammatory cytokines | Promoted M2 polarization, reduced M1 cytokines, inhibited JAK1/STAT1, activated STAT6 | [58] |
| HucMSCs | DSS-induced UC | miR-23b-3p | Nrf2 pathway | Activated Nrf2, increased GPX4, reduced ROS/iron/lipid peroxidation, suppressed IL--1β/IL-6 | [59] |
| hPMSCs | TNBS-induced IBD | N.D. | inflammatory cytokines, ROS, MPO, apoptosis proteins, tight junction, MMPs | Reduced ROS/MPO/cytokines, inhibited apoptosis, upregulated ZO-1/Claudin-1/Occludin), increased IL-10/TGF-β, improved epithelium | [60] |
| hucMSCs | DSS-induced IBD | N.D. | METTL3-Slc37a2-YTHDF1 axis, macrophage, inflammatory cytokines | Upregulated METTL3-m6A, stabilized Slc37a2, promoted M2 macrophages, suppressed cytokines, enhanced barrier | [61] |
| Bovine milk | DSS-induced IBD | N.D. | gut microbiota, T-AOC | Enriched Roseburia, enhanced T-AOC, restored gut diversity | [62] |
| Bovine milk | DSS-induced IBD | N.D. | TLR4-NF-κB, NLRP3, Treg/Th17, microbiota, inflammatory cytokines | Suppressed TLR4-NF-κB/NLRP3, rebalanced Treg/Th17, increased Akkermansia/Enter-orhabdus, decreased Desulfovibrionaceae/Enterococcaceae, reduced IL-1β/TNF-α/IL-6/IL-17A, increased IL-10 | [63] |
| milk | DSS-induced IBD | N.D. | TNFAIP3, COX-2, microbiota, tight junction, inflammatory cytokines | Increased Lachnospiraceae/Rumi-nococcaceae, suppressed NF-κB via TNFAIP3, restored ZO-1, reduced IL-6/IL-1β/TNF-α | [64] |
| milk | DSS-induced IBD | TGF-β1 | inflammatory cytokines, DNMT1/3 | Increased miR-320/miR-375/let-7a, downregulated DNMT1/3, increased TGF-β1, reduced IL-6/TNF-α | [65] |
| M2b macrophages | DSS-induced IBD | CCL1 | CCR8 | Promoted Th2/Treg, reduced IL-1β/IL-6/IL-17A | [66] |
2.2.2. BMVs
| Origin | Disease | Cargo | Target | Function | Ref. |
|---|---|---|---|---|---|
| Roseburia intestinalis | DSS-induced colitis | IPI | DPP4 | Suppressed DPP4 activity, increased GLP-1, downregulated NFκB/STAT3 via PI3K, promoted Bifidobacterium | [80] |
| EcN 1917 | DSS-induced colitis | N.D. | TLRs | Suppressed IL-1β/TNF-α/IL-6/IL-17, reduced MMP-9/COX-2/iNOS, increased TFF-3 | [81] |
| Clostridium butyricum | DSS-induced UC | miR-199a-3p | Map3k4 | Suppressed MAPK/NF-κB, reduced IL-6/TNF-α/IL-1β, decreased | [82] |
| Lactobacillus kefiranofaciens | TNBS-induced IBD | N.D. | NF-κB p65 | Inhibited NF-κB p65, reduced IL-8, decreased MPO | [83] |
| Odoribacter splanchnicus | DSS-induced IBD | N.D. | NLRP3 | Suppressed NLRP3, increased IL-10, upregulated ZO-1/Occludin, reduced apoptosis | [84] |
| Lactobacillus plantarum | DSS-induced UC | N.D. | Gut microbiota | Reduced Proteobacteria, increased Bifidobacteria/Muribaculaceae/Akkermansia, downregulated IL-6/IL-1β/TNF-α/IL-2, inhibited TLR4/MyD88/NF-κB | [85] |
| Faecalibacterium prausnitzii | DSS-induced colitis | N.D. | NF-κB and MAPK signaling pathways | Suppressed NF-κB/JNK/P38 (MAPK), regulated Nrf2/HO-1, upregulated ZO-1/Occludin, reduced IL-1β/IL-2/IL-6/TNF-α/IFN-γ/GM-CSF, increased IL-4/IL-10/TGF-β | [86] |
| Bacteroides fragilis | TNBS-induced colitis | PSA | TLR2 | Induced TLR2, upregulated Gadd45α, promoted CD4+CD25+Foxp3+ Tregs, suppressed TNF-α/IL-17/IFN-γ/IL-6 | [77] |
| Akkermansia muciniphila | DSS-induced colitis | N.D. | Tight junction proteins | Suppressed NO/TNF-α/L-1β/IL-6, upregulated ZO-1/MUC2, increased IgA and sIgA, reduced DAO/D-LA, increased Firmicutes, and decreased Proteobacteria | [87] |
| Bacteroides fragilis | DSS-induced colitis | miR-5119 | PD-L1 | Delivered miR-5119 to inhibit PD-L1, suppressed GSDMD-mediated NET, promoted Lgr5+ intestinal stem cells | [88] |
| Clostridium butyricum | DSS-induced UC | N.D. | Mcrophages | Promoted M2 macrophage polarization, reduced M1 macrophage infiltration, decreased Helicobacter/Escherichia-Shigella abundance; increased Lactobacillus/Akkermansia/Bacteroides, upregulated MUC2/ZO-1 | [76] |
| Lactiplantibacillus plantarum | DSS-induced colitis | small RNAs (<200 nt) | Intestinal epithelial HT29 cells, pro-inflammatory cytokine IL-8 | Delivered small RNAs into HT29 cells, suppressed IL-8, reduced neutrophil accumulation in colonic lamina propria | [89] |
| L. rhamnosus GG | DSS-induced UC | N.D. | TLR4-NF-κB-NLRP3 signaling pathway | Suppressed TLR4-NF-κB-NLRP3 axis, reduced TLR4/MyD88/p65/p-p65/NLRP3/ASC, decreased TNF-α/IL-1β/IL-6/IL-2), reduced Helicobacter/Escherichia-Shigella, increased Lachnospiraceae/Akkermansia | [90] |
| Lactobacillus kefirgranum PRCC-1301 | DSS-induced colitis | N.D. | NF-κB signaling pathway | Suppressed NF-κB by reducing phosphorylated NF-κB p65 and phosphorylated IκBα, decreased IL-2/IL-8/TNF-α, upregulated ZO-1/claudin-1/occludin | [91] |
2.2.3. PDMVs
| Origin | Disease | Cargo | Target | Function | Ref. |
|---|---|---|---|---|---|
| Garlic | DSS-induced colitis | N.D. | TLR4/MyD88/NF-κB signaling pathway, tight junction proteins, gut microbiota, pro-inflammatory cytokines | Suppressed TLR4/MyD88/NF-κB by han-miR3630-5p targeting TLR4 3′ UTR, reduced TLR4/MyD88/NF-κB p65, upregulated ZO-1/occluding/claudin-1, increased Lachnospiraceae, decreased Helicobacter/Escherichia–Shigella/Akkermansia, reduced IL-6/IL-1β/TNF-α/IFN-γ/IL-17A/NO | [102] |
| Lemon | DNBS-induced colitis | N.D. | NF-κB signaling pathway, Nrf2 antioxidant pathway, inflammatory cytokines, gut microbiota | Suppressed NF-κB, reduced IL-6/TNF-α, activated Nrf2 antioxidant pathway, reduced Pygmaiobacter/Lachnospiraceae UCG-010/Tuzzerella/Anaerofilum/Enteractinococcus/Acetatifactor, increased Enterococcus/Bacteroides_pectinophilus group/Lachnospiraceae | [103] |
| Lycium barbarum | DSS-induced UC | Phosphatidylcholine, vitexin-2-O-rhamnoside | Inflammatory cytokines, tight junction proteins, MPO | Suppressed TNF-α/IL-12, upregulated IL-10, increased occluding/ZO-1, reduced MPO | [104] |
| Turmeric | DSS-induced UC | N.D. | Inflammatory cytokines, macrophages, tight junction proteins, gut microbiota | Suppressed TNF-α/IL-6/MCP-1, reduced CD16/32, increased CD206, upregulated ZO-1/occluding/E-cadherin, increased Akkermansia/Lactobacillus, decreased Escherichia-Shigella/Helicobacter | [99] |
| Turmeric | DSS-induced UC | N.D. | Inflammatory cytokines, antioxidant gene, NF-κB signaling pathway, tight junction protein | Suppressed NF-κB, reduced TNF-α/IL-6/IL-1β, upregulated HO-1, increased E-cadherin | [100] |
| Tea leaf | DSS-induced IBD | N.D. | Inflammatory cytokines, ROS, antioxidant enzyme, tight junction proteins, gut microbiota | Suppressed TNF-α/IL-6/IL-12, increased IL-10, reduced ROS, upregulated HO-1/GSH, decreased MDA/MPO, upregulated ZO-1/MUC2, reduced Oscillibacter/elicobacter, increased Lachnospiraceae/kkermansia | [10] |
| Mulberry bark | DSS-induced colitis | HSPA8 | Aryl hydrocarbon receptor (AhR), COP9 Constitutive Photomorphogenic Homolog Subunit 8 (COPS8), CUL1, anti-microbial peptides, tight junction protein, gut microbiota | Activated AhR signaling pathway via HSPA8 binding, upregulated COPS8, promoted deneddylation of CUL1, induced anti-microbial peptides secretion, upregulated ZO-1, reduced Proteobacteria/Segmented Filamentous Bacteria, increased Firmicutes | [105] |
| Aloe | DSS-induced UC | N.D. | Oidative stress markers, tight junction proteins | Reduced p-NF-κB and p-IκB, decreased TNF-α/NO/COX2/3-NT, upregulated ZO-1/claudin4/occluding/E-cadherin/γ-catenin/α-tubulin | [106] |
| Grape | DSS-induced colitis | N.D. | Intestinal stem cells (Lgr5+), Wnt/β-catenin signaling pathway | Activated Wnt/β-catenin signaling (phosphorylated GSK-3β, nuclear translocation of β-catenin), upregulated intestinal stem cell markers (Lgr5, BMI1), promoted Lgr5+ stem cell proliferation | [107] |
| Ginseng | DSS-induced UC | Ginsenosides | Inflammatory cytokines, NF-κB signaling pathway, gut microbiota | Suppressed NF-κB signaling pathway activation, reduced TNF-α/IL-6/IL-17A, increased IL-10, decreased Firmicutes/Bacteroidota ratio, increased Lactobacillus, decreased Helicobacter/Ruminococcus | [108] |
| Broccoli | DSS-induced colitis | N.D. | Dendritic cells, AMP-activated protein kinase (AMPK), mTOR signaling pathway, inflammatory cytokines | Activated AMPK in DCs, reduced phosphorylation of p70S6K and S6, reduced TNF-α/IL-12/IFN-γ/IL-17A, increased IL-10/TGF-β | [109] |
| Ginseng | DSS-induced IBD | Small RNAs, ginsenosides | IKK/IκB/NF-κB signaling pathway | Activated autophagy (upregulated LC3/Atg7/Beclin1, downregulated p-mTOR/p-AKT), increased CD206/Arg1, decreased CD86/iNOS, reduced P-IKKα/β/P-IκBα/NF-κB, reduced TNF-α/IL-1β/IL-6, increased IL-10, increased ZO-1/occludin | [110] |
| Ginseng | DSS-induced IBD | N.D. | TLR4/MAPK signaling pathway, p62/Nrf2/Keap1 pathway | Suppressed TLR4/MAPK activation by reducing phosphorylation of ERK/JNK/p38, activated p62/Nrf2/Keap1 pathway to increase HO-1/GCLC/GCLM, decreased TNF-α/IL-6/IL-1β, enhanced ZO-1/occludin/claudin-1, promoted intestinal stem cell proliferation via Wnt/β-catenin, and reduced Firmicutes/Bacteroidetes ratio | [101] |
2.2.4. Liposomes
2.2.5. Polymersomes
2.2.6. EBMVs
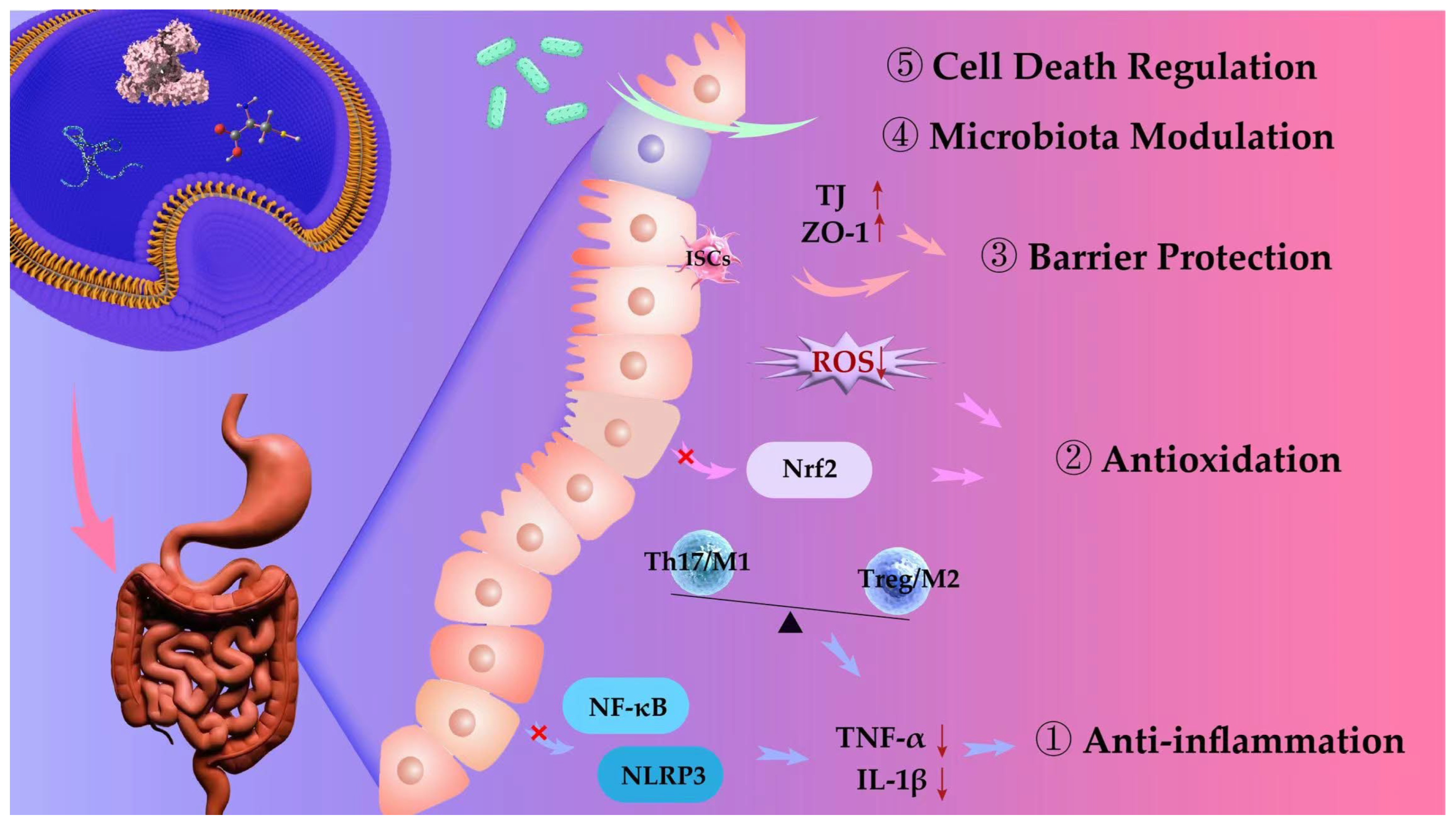
3. Therapeutic Mechanism of MV-DDSs in IBD
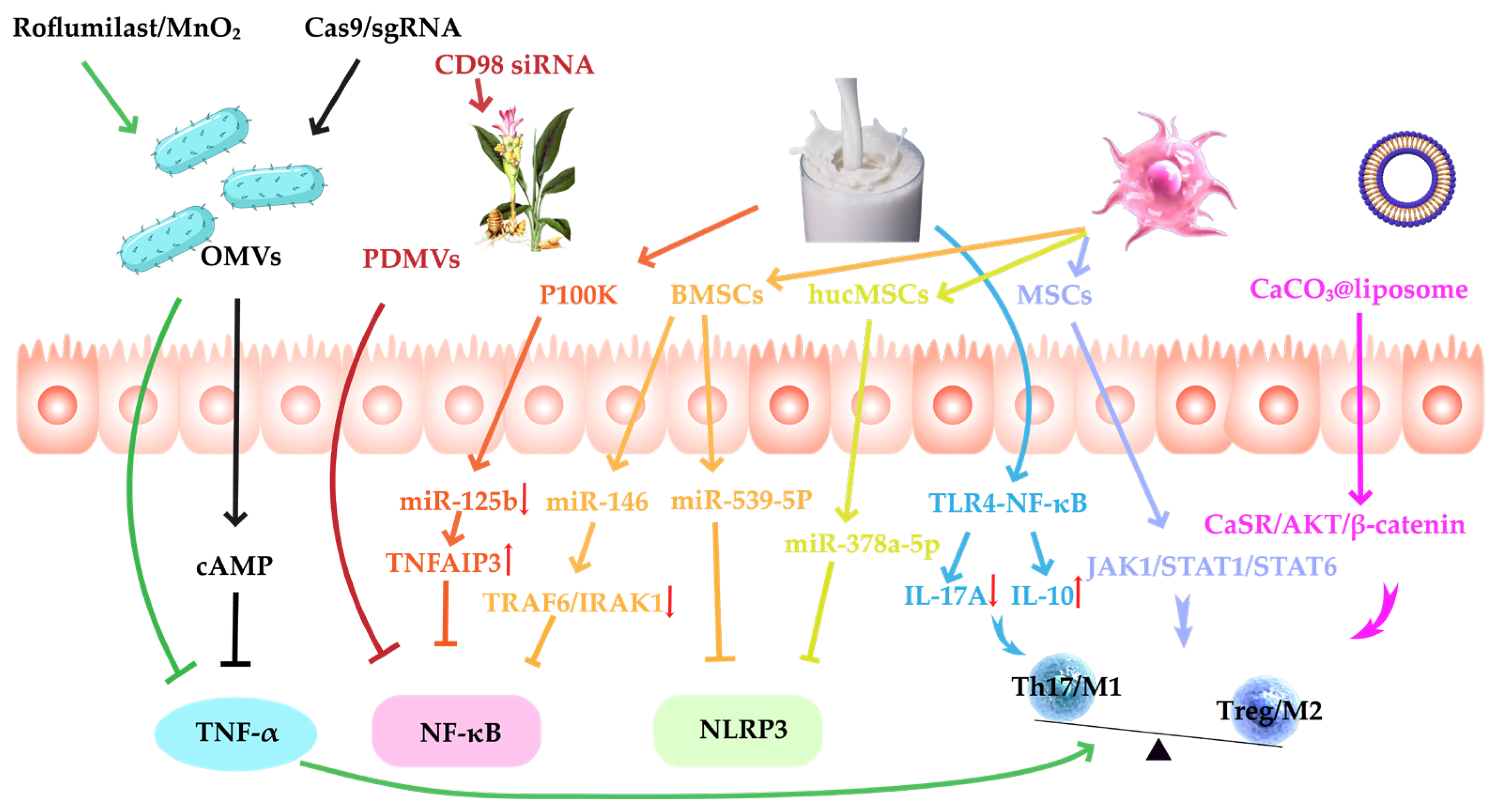
3.1. Anti-Inflammation
3.1.1. Inhibition of Pro-Inflammatory Cytokines
3.1.2. Suppression of Inflammatory Signaling Pathways
3.1.3. Modulation of Immune Homeostasis and Mucosal Tolerance
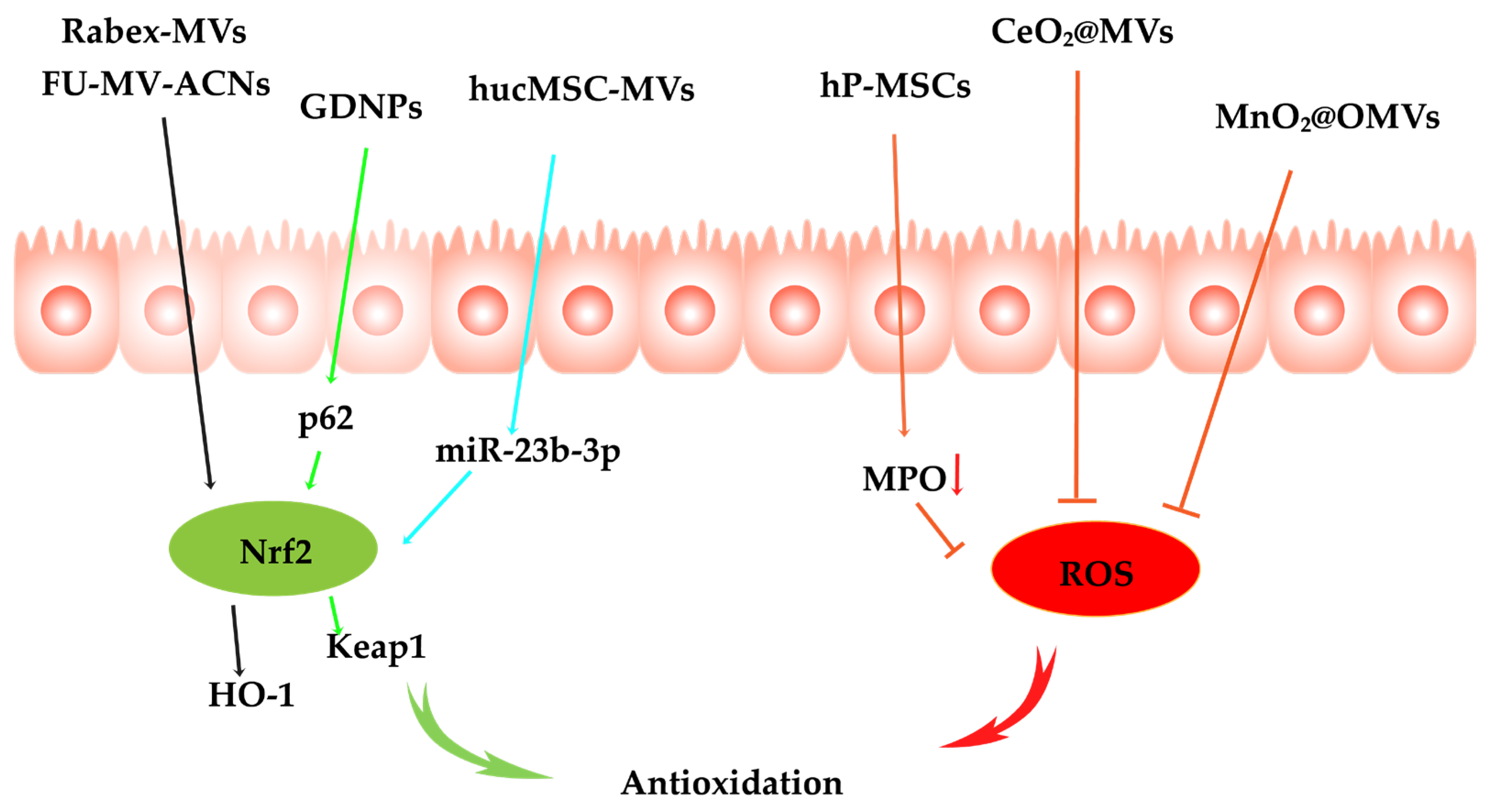
3.2. Antioxidation
3.2.1. Oxidative Stress Signaling Pathway Inhibition
3.2.2. ROS Scavenging
3.3. Barrier Protection
3.3.1. Tight Junction Protein Expression Enhancement
3.3.2. Epithelial Regeneration Promotion
3.4. Microbiota Modulation
3.5. Cell Death Regulation
4. Delivery Strategies of MV-DDSs in IBD
| Delivery Route | Vesicle Types | Advantages | Limitations | Ref. |
|---|---|---|---|---|
| Oral | Liposomes |
|
| [28] |
| Intravenous | Liposomes |
|
| [29] |
| Oral | Polymersomes |
|
| [26] |
| Intravenous | EBMVs |
|
| [33] |
| Oral | PDMVs |
|
| [32] |
| Oral | BMVs |
|
| [156] |
5. Conclusions and Outlook
- Standardization and scalability of production. The heterogeneity of vesicle sources—including plant-derived vesicles, bacterial outer membrane vesicles, milk-derived vesicles, and engineered biomimetic vesicles—poses difficulties in achieving reproducible large-scale production with consistent physicochemical properties and bioactivity. Future efforts should focus on establishing robust manufacturing processes and standardized quality control protocols.
- Biosafety and immunogenicity. Despite their generally favorable biocompatibility, the residual immunogenic components in certain vesicles, such as endotoxins in bacterial vesicles or allergenic proteins in milk-derived vesicles, remain a safety concern. Rigorous purification, detoxification, and characterization strategies are needed to ensure low immunogenicity without compromising therapeutic efficacy.
- Precise targeting and controlled release. Although vesicles exhibit inherent tropism to inflamed intestinal tissues, strategies to further improve site-specific delivery and stimuli-responsive release—such as engineering vesicles to respond to local oxidative stress or pH gradients—require continued optimization.
- Cargo loading efficiency and stability. Efficient encapsulation and stable retention of diverse therapeutic cargos, especially sensitive biological molecules like siRNA, peptides, or probiotics, remain technical bottlenecks. The development of advanced loading techniques and stabilizing formulations will be pivotal to maximize their therapeutic potential.
- In vivo tracking and pharmacokinetics. The complex intestinal microenvironment and dynamic mucus barrier present obstacles for vesicle trafficking and persistence in vivo. New imaging modalities and tracing methods are necessary to elucidate vesicles’ biodistribution, clearance, and mechanisms of action in real time.
- Regulatory pathways and translational readiness. Given the novelty and complexity of MV-DDSs, regulatory frameworks governing their safety, efficacy, and manufacturing standards are still evolving. Close collaboration among academic researchers, industry partners, and regulatory agencies will be essential to accelerate their clinical development.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| IBD | Inflammatory bowel disease |
| CD | Crohn’s disease |
| UC | Ulcerative colitis |
| MV-DDSs | Membrane vesicle-based drug delivery systems |
| MDMVs | Mammalian-derived membrane vesicles |
| PDMVs | Plant-derived membrane vesicles |
| BMVs | Bacterial membrane vesicles |
| EBMVs | Engineered biomimetic membrane vesicles |
| DDSs | Drug delivery systems |
| MVs | Membrane vesicles |
| OMVs | Outer membrane vesicles |
| ROS | Reactive oxygen species |
| 5-ASA | 5-aminosalicylic acid |
| MSCs | Mesenchymal stem cells |
| EcN | Escherichia coli Nissle |
| EPR | Enhanced permeability and retention |
| PEG | Polyethylene glycol |
| PAMPs | Pathogen-associated molecular patterns |
| PDE4 | Phosphodiesterase 4 |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| ACNs | Anthocyanins |
| FU | Fucoidan |
| MPO | Myeloperoxidase |
| CeO2 | Cerium oxide |
| hAFSCs | human amniotic fluid stem cells |
| PCD | Programmed cell death |
| Se | Selenium |
| LbL | Layer-by-layer |
| HTCC | N-(2-hydroxy)propyl-3-trimethylammonium chitosan chloride |
| OKGM | Oxidized konjac glucomannan |
| DMA | Dopamine methacrylamide |
| i.p. | Intraperitoneal |
| CCR2 | cc chemokine receptor 2 |
| MCP-1 | Monocyte chemoattractant protein-1 |
References
- Graham, D.B.; Xavier, R.J. Pathway paradigms revealed from the genetics of inflammatory bowel disease. Nature 2020, 578, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Mentella, M.C.; Scaldaferri, F.; Pizzoferrato, M.; Gasbarrini, A.; Miggiano, G.A.D. Nutrition, IBD and Gut Microbiota: A Review. Nutrients 2020, 12, 944. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Bernstein, C.N.; Iliopoulos, D.; Macpherson, A.; Neurath, M.F.; Ali, R.A.R.; Vavricka, S.R.; Fiocchi, C. Environmental triggers in IBD: A review of progress and evidence. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 39–49. [Google Scholar] [CrossRef]
- Annese, V.; Annese, M. Precision Medicine in Inflammatory Bowel Disease. Diagnostics 2023, 13, 2797. [Google Scholar] [CrossRef] [PubMed]
- Ko, I.K.; Kim, B.-G.; Awadallah, A.; Mikulan, J.; Lin, P.; Letterio, J.J.; Dennis, J.E. Targeting Improves MSC Treatment of Inflammatory Bowel Disease. Mol. Ther. 2010, 18, 1365–1372. [Google Scholar] [CrossRef]
- Lin, S.; Han, S.; Wang, X.; Wang, X.; Shi, X.; He, Z.; Sun, M.; Sun, J. Oral Microto-Nano Genome-Editing System Enabling Targeted Delivery and Conditional Activation of CRISPR-Cas9 for Gene Therapy of Inflammatory Bowel Disease. ACS Nano 2024, 18, 25657–25670. [Google Scholar] [CrossRef]
- Wang, X.; Ni, J.; You, Y.; Feng, G.; Zhang, S.; Bao, W.; Hou, H.; Li, H.; Liu, L.; Zheng, M.; et al. SNX10-mediated LPS sensing causes intestinal barrier dysfunction via a caspase-5-dependent signaling cascade. EMBO J. 2021, 40, e108080. [Google Scholar] [CrossRef]
- Gecse, K.B.; Vermeire, S. Differential diagnosis of inflammatory bowel disease: Imitations and complications. Lancet Gastroenterol. Hepatol. 2018, 3, 644–653. [Google Scholar] [CrossRef]
- Saadah, O.I.; Alameel, T.; Al Sarkhy, A.; Hasosah, M.; Al-Hussaini, A.; Almadi, M.A.; Al-Bawardy, B.; Altuwaijri, T.A.; Aledreesi, M.; Bakkari, S.A.; et al. Saudi consensus guidance for the diagnosis and management of inflammatory bowel disease in children and adolescents. Saudi J. Gastroenterol. 2025, 31, 107–136. [Google Scholar] [CrossRef]
- Zu, M.; Xie, D.; Canup, B.S.B.; Chen, N.; Wang, Y.; Sun, R.; Zhang, Z.; Fu, Y.; Dai, F.; Xiao, B. ‘Green’ nanotherapeutics from tea leaves for orally targeted prevention and alleviation of colon diseases. Biomaterials 2021, 279, 121178. [Google Scholar] [CrossRef]
- Rogler, G.; Singh, A.; Kavanaugh, A.; Rubin, D.T. Extraintestinal Manifestations of Inflammatory Bowel Disease: Current Concepts, Treatment, and Implications for Disease Management. Gastroenterology 2021, 161, 1118–1132. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Ha, C. Epidemiology and Pathogenesis of Ulcerative Colitis. Gastroenterol. Clin. N. Am. 2020, 49, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, T.; Sun, M.; Song, Y.; Huang, X.; Zhang, S.; Yang, J.; Zhang, J. Advanced Nanomedicine: Redefining Therapeutic Paradigms for Inflammatory Bowel Disease. Adv. Healthc. Mater. 2023, 12, e2300069. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Hu, Y.; Conceicao, M.; Wood, M.J.A.; Zhong, H.; Wang, Y.; Shao, P.; Chen, J.; Qiu, L. Oral delivery of layer-by-layer coated exosomes for colitis therapy. J. Control. Release 2023, 354, 635–650. [Google Scholar] [CrossRef]
- Song, G.; Zeng, C.; Li, J.; Liu, J.; Zhao, J.; Liu, B.; Fan, J.; Xie, H. Exosome-based nanomedicines for digestive system tumors therapy. Nanomedicine 2025, 20, 1167–1180. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, W.; Wu, Z.; Chen, Y. Diversity of extracellular vesicle sources in atherosclerosis: Role and therapeutic application. Angiogenesis 2025, 28, 34. [Google Scholar] [CrossRef]
- Yu, Y.; Lin, S.; Chen, Z.; Qin, B.; He, Z.; Cheng, M.; Sun, M.; Sun, J. Bacteria-driven bio-therapy: From fundamental studies to clinical trials. Nano Today 2023, 48, 101731. [Google Scholar] [CrossRef]
- Geng, T.; Ding, L.; Liu, M.; Zou, X.; Gu, Z.; Lin, H.; Sun, L. Preservation of extracellular vesicles for drug delivery: A comparative evaluation of storage buffers. J. Drug Deliv. Sci. Technol. 2025, 107, 106850. [Google Scholar] [CrossRef]
- Glassman, P.M.; Hood, E.D.; Ferguson, L.T.; Zhao, Z.; Siegel, D.L.; Mitragotri, S.; Brenner, J.S.; Muzykantov, V.R. Red blood cells: The metamorphosis of a neglected carrier into the natural mothership for artificial nanocarriers. Adv. Drug Deliv. Rev. 2021, 178, 113992. [Google Scholar] [CrossRef]
- Lei, P.; Yu, H.; Ma, J.; Du, J.; Fang, Y.; Yang, Q.; Zhang, K.; Luo, L.; Jin, L.; Wu, W.; et al. Cell membrane nanomaterials composed of phospholipids and glycoproteins for drug delivery in inflammatory bowel disease: A review. Int. J. Biol. Macromol. 2023, 249, 126000. [Google Scholar] [CrossRef]
- Liu, R. A promising area of research in medicine: Recent advances in properties and applications of Lactobacillus-derived exosomes. Front. Microbiol. 2024, 15, 1266510. [Google Scholar] [CrossRef] [PubMed]
- Barathan, M.; Ng, S.L.; Lokanathan, Y.; Ng, M.H.; Law, J.X. Milk-Derived Extracellular Vesicles: A Novel Perspective on Comparative Therapeutics and Targeted Nanocarrier Application. Vaccines 2024, 12, 1282. [Google Scholar] [CrossRef] [PubMed]
- Kameli, N.; Borman, R.; Lopez-lglesias, C.; Savelkoul, P.; Stassen, F.R.M. Characterization of Feces-Derived Bacterial Membrane Vesicles and the Impact of Their Origin on the Inflammatory Response. Front. Cell. Infect. Microbiol. 2021, 11, 1282. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Gao, B.; Xu, Y.; Zhao, Q.; Jiang, J.; Sun, D.; Zhang, Y.; Zhou, S.; Fan, J.-B.; Zhang, M.; et al. Atractylodes macrocephala-derived extracellular vesicles-like particles enhance the recovery of ulcerative colitis by remodeling intestinal microecological balance. J. Nanobiotechnol. 2025, 23, 433. [Google Scholar] [CrossRef]
- Di Gioia, S.; Hossain, M.N.; Conese, M. Biological properties and therapeutic effects of plant-derived nanovesicles. Open Med. 2020, 15, 1096–1122. [Google Scholar] [CrossRef]
- Rahman, M.; Kumar, V.; Beg, S.; Sharma, G.; Katare, O.P.; Anwar, F. Emergence of liposome as targeted magic bullet for inflammatory disorders: Current state of the art. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1597–1608. [Google Scholar] [CrossRef]
- Yang, D.; Zeng, H.; Zhuang, Y.; Jiang, S.; Pan, W. Nanodrug delivery materials for digestive system diseases. APL Mater. 2024, 12, 060603. [Google Scholar] [CrossRef]
- Xian, S.; Zhu, J.; Wang, Y.; Song, H.; Wang, H. Oral liposomal delivery of an activatable budesonide prodrug reduces colitis in experimental mice. Drug Deliv. 2023, 30, 2183821. [Google Scholar] [CrossRef]
- Tollemeto, M.; Ursulska, S.; Welzen, P.L.W.; Thamdrup, L.H.E.; Permlid, A.M.; Li, Y.; Soufi, G.; Padial, T.P.; Christensen, J.B.; Nielsen, L.H.; et al. Tailored Polymersomes for Enhanced Oral Drug Delivery: pH-Sensitive Systems for Intestinal Delivery of Immunosuppressants. Small 2024, 20, e2403640. [Google Scholar] [CrossRef]
- Zhong, H.; Luo, X.; Abdullah, X.; Liu, X.; Hussain, M.; Guan, R. Nano-targeted delivery system: A promising strategy of anthocyanin encapsulation for treating intestinal inflammation. Crit. Rev. Food Sci. Nutr. 2025, 65, 1–22. [Google Scholar] [CrossRef]
- Xu, X.; Yang, W.; Liang, Q.; Shi, Y.; Zhang, W.; Wang, X.; Meng, F.; Zhong, Z.; Yin, L. Efficient and targeted drug/siRNA co-delivery mediated by reversibly crosslinked polymersomes toward anti-inflammatory treatment of ulcerative colitis (UC). Nano Res. 2019, 12, 659–667. [Google Scholar] [CrossRef]
- Yang, W.; Lin, P.; Gao, R.; Fang, Z.; Wang, Z.; Ma, Z.; Shi, J.; Yu, W. Cell-derived biomimetic drug delivery system for inflammatory bowel disease therapy. Mater. Today Bio 2024, 29, 101332. [Google Scholar] [CrossRef]
- Corbo, C.; Cromer, W.E.; Molinaro, R.; Toledano Furman, N.E.; Hartman, K.A.; De Rosa, E.; Boada, C.; Wang, X.; Zawieja, D.C.; Agostini, M.; et al. Engineered biomimetic nanovesicles show intrinsic anti-inflammatory properties for the treatment of inflammatory bowel diseases. Nanoscale 2017, 9, 14581–14591. [Google Scholar] [CrossRef]
- Gao, F.; Chiu, S.M.; Motan, D.A.L.; Zhang, Z.; Chen, L.; Ji, H.L.; Tse, H.F.; Fu, Q.L.; Lian, Q. Mesenchymal stem cells and immunomodulation: Current status and future prospects. Cell Death Dis. 2016, 7, e2062. [Google Scholar] [CrossRef] [PubMed]
- Dabrowska, S.; Andrzejewska, A.; Janowski, M.; Lukomska, B. Immunomodulatory and Regenerative Effects of Mesenchymal Stem Cells and Extracellular Vesicles: Therapeutic Outlook for Inflammatory and Degenerative Diseases. Front. Immunol. 2021, 11, 591065. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Fan, H.; Shou, Z.; Xu, M.; Chen, Q.; Ai, C.; Dong, Y.; Liu, Y.; Nan, Z.; Wang, Y.; et al. Extracellular vesicles containing miR-146a attenuate experimental colitis by targeting TRAF6 and IRAK1. Int. Immunopharmacol. 2019, 68, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Ren, F.; Fang, X.; Yuan, L.; Liu, G.; Wang, S. Exosomal MicroRNA-181a Derived From Mesenchymal Stem Cells Improves Gut Microbiota Composition, Barrier Function, and Inflammatory Status in an Experimental Colitis Model. Front. Med. 2021, 8, 660614. [Google Scholar] [CrossRef]
- Ihara, S.; Hirata, Y.; Koike, K. TGF-β in inflammatory bowel disease: A key regulator of immune cells, epithelium, and the intestinal microbiota. J. Gastroenterol. 2017, 52, 777–787. [Google Scholar] [CrossRef]
- Yang, R.; Huang, H.; Cui, S.; Zhou, Y.; Zhang, T.; Zhou, Y. IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b. Cell Death Dis. 2020, 11, 603. [Google Scholar] [CrossRef]
- El-Badawy, A.; Amer, M.; Abdelbaset, R.; Sherif, S.N.; Abo-Elela, M.; Ghallab, Y.H.; Abdelhamid, H.; Ismail, Y.; El-Badri, N. Adipose Stem Cells Display Higher Regenerative Capacities and More Adaptable Electro-Kinetic Properties Compared to Bone Marrow-Derived Mesenchymal Stromal Cells. Sci. Rep. 2016, 6, 37801. [Google Scholar] [CrossRef]
- Guneta, V.; Tan, N.S.; Chan, S.K.J.; Tanavde, V.; Lim, T.C.; Wong, T.C.M.; Choong, C. Comparative study of adipose-derived stem cells and bone marrow-derived stem cells in similar microenvironmental conditions. Exp. Cell Res. 2016, 348, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Hong, P.; Yang, H.; Wu, Y.; Li, K.; Tang, Z. The functions and clinical application potential of exosomes derived from adipose mesenchymal stem cells: A comprehensive review. Stem Cell Res. Ther. 2019, 10, 242. [Google Scholar] [CrossRef] [PubMed]
- Heidari, N.; Abbasi-Kenarsari, H.; Namaki, S.; Baghaei, K.; Zali, M.R.; Ghaffari Khaligh, S.; Hashemi, S.M. Adipose-derived mesenchymal stem cell-secreted exosome alleviates dextran sulfate sodium-induced acute colitis by Treg cell induction and inflammatory cytokine reduction. J. Cell. Physiol. 2021, 236, 5906–5920. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Heo, J.; Ahn, E.-K.; Kim, J.H.; Kim, Y.-H.; Chang, H.-K.; Lee, S.-J.; Kim, J.; Park, S.-J. Conditioned secretome of adipose-derived stem cells improves dextran sulfate sodium-induced colitis in mice. World J. Gastroenterol. 2021, 27, 3342–3356. [Google Scholar] [CrossRef]
- Tian, J.; Zhu, Q.; Zhang, Y.; Bian, Q.; Hong, Y.; Shen, Z.; Xu, H.; Rui, K.; Yin, K.; Wang, S. Olfactory Ecto-Mesenchymal Stem Cell-Derived Exosomes Ameliorate Experimental Colitis via Modulating Th1/Th17 and Treg Cell Responses. Front. Immunol. 2020, 11, 598322. [Google Scholar] [CrossRef]
- Li, Y.; Altemus, J.; Lightner, A.L. Mesenchymal stem cells and acellular products attenuate murine induced colitis. Stem Cell Res. Ther. 2020, 11, 515. [Google Scholar] [CrossRef]
- Li, X.; Bai, J.; Ji, X.; Li, R.; Xuan, Y.; Wang, Y. Comprehensive characterization of four different populations of human mesenchymal stem cells as regards their immune properties, proliferation and differentiation. Int. J. Mol. Med. 2014, 34, 695–704. [Google Scholar] [CrossRef]
- Yu, H.; Yang, X.; Xiao, X.; Xu, M.; Yang, Y.; Xue, C.; Li, X.; Wang, S.; Zhao, R.C. Human Adipose Mesenchymal Stem Cell-derived Exosomes Protect Mice from DSS-Induced Inflammatory Bowel Disease by Promoting Intestinal-stem-cell and Epithelial Regeneration. Aging Dis. 2021, 12, 1423–1437. [Google Scholar] [CrossRef]
- Wang, S.; Song, Y.; Wang, Z.; Chang, X.; Wu, H.; Yan, Z.; Wu, J.; He, Z.; Kang, L.; Hu, W.; et al. Neutrophil-derived PAD4 induces citrullination of CKMT1 exacerbates mucosal inflammation in inflammatory bowel disease. Cell. Mol. Immunol. 2024, 21, 620–633. [Google Scholar] [CrossRef]
- Cheng, J.; Zhang, Y.; Ma, L.; Du, W.; Zhang, Q.; Gao, R.; Zhao, X.; Chen, Y.; Jiang, L.; Li, X.; et al. Macrophage-Derived Extracellular Vesicles-Coated Palladium Nanoformulations Modulate Inflammatory and Immune Homeostasis for Targeting Therapy of Ulcerative Colitis. Adv. Sci. 2023, 10, e2304002. [Google Scholar] [CrossRef]
- Han, G.; Kim, H.; Jang, H.; Kim, E.S.; Kim, S.H.; Yang, Y. Oral TNF-α siRNA delivery via milk-derived exosomes for effective treatment of inflammatory bowel disease. Bioact. Mater. 2024, 34, 138–149. [Google Scholar] [CrossRef] [PubMed]
- van Herwijnen, M.J.C.; Driedonks, T.A.P.; Snoek, B.L.; Kroon, A.M.T.; Kleinjan, M.; Jorritsma, R.; Pieterse, C.M.J.; Nolte-‘t Hoen, E.N.M.; Wauben, M.H.M. Abundantly Present miRNAs in Milk-Derived Extracellular Vesicles Are Conserved Between Mammals. Front. Nutr. 2018, 5, 81. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Zhang, Z.-Y.; Yuan, J.-T.; Ocansey, D.K.W.; Tu, Q.; Zhang, X.; Qian, H.; Xu, W.-R.; Qiu, W.; Mao, F. hucMSC-derived exosomes attenuate colitis by regulating macrophage pyroptosis via the miR-378a-5p/NLRP3 axis. Stem Cell Res. Ther. 2021, 12, 416. [Google Scholar] [CrossRef]
- Mao, F.; Wu, Y.; Tang, X.; Kang, J.; Zhang, B.; Yan, Y.; Qian, H.; Zhang, X.; Xu, W. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Relieve Inflammatory Bowel Disease in Mice. BioMed Res. Int. 2017, 2017, 5356760. [Google Scholar] [CrossRef]
- Li, P.; Zhang, H.-Y.; Gao, J.-Z.; Du, W.-Q.; Tang, D.; Wang, W.; Wang, L.-H. Mesenchymal stem cells-derived extracellular vesicles containing miR-378a-3p inhibit the occurrence of inflammatory bowel disease by targeting GATA2. J. Cell. Mol. Med. 2022, 26, 3133–3146. [Google Scholar] [CrossRef]
- Yang, S.; Liang, X.; Song, J.; Li, C.; Liu, A.; Luo, Y.; Ma, H.; Tan, Y.; Zhang, X. A novel therapeutic approach for inflammatory bowel disease by exosomes derived from human umbilical cord mesenchymal stem cells to repair intestinal barrier via TSG-6. Stem Cell Res. Ther. 2021, 12, 315. [Google Scholar] [CrossRef]
- Wang, G.; Yuan, J.; Cai, X.; Xu, Z.; Wang, J.; Ocansey, D.K.W.; Yan, Y.; Qian, H.; Zhang, X.; Xu, W.; et al. HucMSC-exosomes carrying miR-326 inhibit neddylation to relieve inflammatory bowel disease in mice. Clin. Transl. Med. 2020, 10, e113. [Google Scholar] [CrossRef]
- Cao, L.; Xu, H.; Wang, G.; Liu, M.; Tian, D.; Yuan, Z. Extracellular vesicles derived from bone marrow mesenchymal stem cells attenuate dextran sodium sulfate-induced ulcerative colitis by promoting M2 macrophage polarization. Int. Immunopharmacol. 2019, 72, 264–274. [Google Scholar] [CrossRef]
- Liu, L.; Ye, Y.; Feng, Z.; Cao, X.; Zhao, J. Human Umbilical Cord Mesenchymal Stem Cells Derived Exosomes Alleviate Ulcerative Colitis by Inhibiting Macrophage Ferroptosis Via miR-23b-3p/Nrf2 Pathway. Gastroenterology 2023, 164, S695–S696. [Google Scholar] [CrossRef]
- Duan, L.; Huang, H.; Zhao, X.; Zhou, M.; Chen, S.; Wang, C.; Han, Z.; Han, Z.-C.; Guo, Z.; Li, Z.; et al. Extracellular vesicles derived from human placental mesenchymal stem cells alleviate experimental colitis in mice by inhibiting inflammation and oxidative stress. Int. J. Mol. Med. 2020, 46, 1551–1561. [Google Scholar] [CrossRef]
- Xu, X.; Peng, J.; Wang, N.; Ocansey, D.K.W.; Zhang, X.; Mao, F. hucMSC-Ex alleviates inflammatory bowel disease in mice by enhancing M2-type macrophage polarization via the METTL3-Slc37a2-YTHDF1 axis. Cell Biol. Toxicol. 2024, 40, 74. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Chen, X.; Qi, Q.; Feng, X. Bovine Milk Derived Exosomes Affect Gut Microbiota of DSS-Induced Colitis Mice. Indian J. Microbiol. 2024, 64, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Hao, H.; Zhang, Z.; Lv, Y.; Liang, X.; Liu, Q.; Liu, T.; Gong, P.; Zhang, L.; Cao, F.; et al. Milk-derived extracellular vesicles alleviate ulcerative colitis by regulating the gut immunity and reshaping the microbiota. Theranostics 2021, 11, 8570–8586. [Google Scholar] [CrossRef]
- Benmoussa, A.; Diallo, I.; Salem, M.; Michel, S.; Gilbert, C.; Sevigny, J.; Provost, P. Concentrates of two subsets of extracellular vesicles from cow’s milk modulate symptoms and inflammation in experimental colitis. Sci. Rep. 2019, 9, 14661. [Google Scholar] [CrossRef]
- Reif, S.; Elbaum-Shiff, Y.; Koroukhov, N.; Shilo, I.; Musseri, M.; Golan-Gerstl, R. Cow and Human Milk-Derived Exosomes Ameliorate Colitis in DSS Murine Model. Nutrients 2020, 12, 2589. [Google Scholar] [CrossRef]
- Yang, R.; Liao, Y.; Wang, L.; He, P.; Hu, Y.; Yuan, D.; Wu, Z.; Sun, X. Exosomes Derived From M2b Macrophages Attenuate DSS-Induced Colitis. Front. Immunol. 2019, 10, 2346. [Google Scholar] [CrossRef] [PubMed]
- Gierynska, M.; Szulc-Dabrowska, L.; Struzik, J.; Mielcarska, M.B.; Gregorczyk-Zboroch, K.P. Integrity of the Intestinal Barrier: The Involvement of Epithelial Cells and Microbiota-A Mutual Relationship. Animals 2022, 12, 145. [Google Scholar] [CrossRef]
- Di Tommaso, N.; Gasbarrini, A.; Ponziani, F.R. Intestinal Barrier in Human Health and Disease. Int. J. Environ. Res. Public Health 2021, 18, 12836. [Google Scholar] [CrossRef]
- Kang, C.-S.; Ban, M.; Choi, E.-J.; Moon, H.-G.; Jeon, J.-S.; Kim, D.-K.; Park, S.-K.; Jeon, S.G.; Roh, T.-Y.; Myung, S.-J.; et al. Extracellular Vesicles Derived from Gut Microbiota, Especially Akkermansia muciniphila, Protect the Progression of Dextran Sulfate Sodium-Induced Colitis. PLoS ONE 2013, 8, e76520. [Google Scholar] [CrossRef]
- Qiao, K.; Chen, C.; Liu, H.; Qin, Y.; Liu, H. Pinin Induces Epithelial-to-Mesenchymal Transition in Hepatocellular Carcinoma by Regulating m6A Modification. J. Oncol. 2021, 2021, 7529164. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, X.; Wang, D.; Wang, R.; Zhu, R.; You, X.; Liu, X.; Li, Y. Probiotic nanovesicles encapsulating baicalin: A strategy to overcome colorectal cancer. Lett. Appl. Microbiol. 2025, 78, ovae117. [Google Scholar] [CrossRef]
- Behrouzi, A.; Mazaheri, H.; Falsafi, S.; Tavassol, Z.H.; Moshiri, A.; Siadat, S.D. Intestinal effect of the probiotic Escherichia coli strain Nissle 1917 and its OMV. J. Diabetes Metab. Disord. 2020, 19, 597–604. [Google Scholar] [CrossRef]
- Chen, H.; Lei, P.; Ji, H.; Yang, Q.; Peng, B.; Ma, J.; Fang, Y.; Qu, L.; Li, H.; Wu, W.; et al. Advances in Escherichia coli Nissle 1917 as a customizable drug delivery system for disease treatment and diagnosis strategies. Mater. Today Bio 2023, 18, 100543. [Google Scholar] [CrossRef] [PubMed]
- Wehkamp, J.; Harder, J.; Wehkamp, K.; Wehkamp-von Meissner, B.; Schlee, M.; Enders, C.; Sonnenborn, U.; Nuding, S.; Bengmark, S.; Fellermann, K.; et al. NF-κB- and AP-1-mediated induction of human beta defensin-2 in intestinal epithelial cells by Escherichia coli Nissle 1917: A novel effect of a probiotic bacterium. Infect. Immun. 2004, 72, 5750–5758. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Moon, C.M.; Shin, T.-S.; Kim, E.K.; McDowell, A.; Jo, M.-K.; Joo, Y.H.; Kim, S.-E.; Jung, H.-K.; Shim, K.-N.; et al. Lactobacillus paracasei-derived extracellular vesicles attenuate the intestinal inflammatory response by augmenting the endoplasmic reticulum stress pathway. Exp. Mol. Med. 2020, 52, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Yang, C.; Liu, L.; Mai, G.; Li, H.; Wu, L.; Jin, M.; Chen, Y. Commensal bacteria-derived extracellular vesicles suppress ulcerative colitis through regulating the macrophages polarization and remodeling the gut microbiota. Microb. Cell Factories 2022, 21, 88. [Google Scholar] [CrossRef]
- Shen, Y.; Torchia, M.L.G.; Lawson, G.W.; Karp, C.L.; Ashwell, J.D.; Mazmanian, S.K. Outer Membrane Vesicles of a Human Commensal Mediate Immune Regulation and Disease Protection. Cell Host Microb. 2012, 12, 509–520. [Google Scholar] [CrossRef]
- Chu, H.; Khosravi, A.; Kusumawardhani, I.P.; Kwon, A.H.K.; Vasconcelos, A.C.; Cunha, L.D.; Mayer, A.E.; Shen, Y.; Wu, W.-L.; Kambal, A.; et al. Gene-microbiota interactions contribute to the pathogenesis of inflammatory bowel disease. Science 2016, 352, 1116–1120. [Google Scholar] [CrossRef]
- Pitt, N.; Morrissette, M.; Gates, M.F.; Bargabos, R.; Krumpoch, M.; Hawkins, B.; Lewis, K. Bacterial membrane vesicles restore gut anaerobiosis. NPJ Biofilms Microbiomes 2025, 11, 48. [Google Scholar] [CrossRef]
- Han, H.S.; Hwang, S.; Choi, S.Y.; Hitayezu, E.; Humphrey, M.A.; Enkhbayar, A.; Song, D.-G.; Kim, M.; Park, J.-S.; Park, Y.-T.; et al. Roseburia intestinalis-derived extracellular vesicles ameliorate colitis by modulating intestinal barrier, microbiome, and inflammatory responses. J. Extracell. Vesicles 2024, 13, e12487. [Google Scholar] [CrossRef]
- Fabrega, M.-J.; Rodriguez-Nogales, A.; Garrido-Mesa, J.; Algieri, F.; Badia, J.; Gimenez, R.; Galvez, J.; Baldoma, L. Intestinal Anti-inflammatory Effects of Outer Membrane Vesicles from Escherichia coli Nissle 1917 in DSS-Experimental Colitis in Mice. Front. Microbiol. 2017, 8, 1274. [Google Scholar] [CrossRef]
- Ma, L.; Lyu, W.; Song, Y.; Chen, K.; Lv, L.; Yang, H.; Wang, W.; Xiao, Y. Anti-Inflammatory Effect of Clostridium butyricum-Derived Extracellular Vesicles in Ulcerative Colitis: Impact on Host microRNAs Expressions and Gut Microbiome Profiles. Mol. Nutr. Food Res. 2023, 67, e2200884. [Google Scholar] [CrossRef]
- Seo, M.K.; Park, E.J.; Ko, S.Y.; Choi, E.W.; Kim, S. Therapeutic effects of kefir grain Lactobacillus-derived extracellular vesicles in mice with 2,4,6-trinitrobenzene sulfonic acid-induced inflammatory bowel disease. J. Dairy Sci. 2018, 101, 8662–8671. [Google Scholar] [CrossRef]
- Zhuang, J.; Zhuang, Z.; Chen, B.; Yang, Y.; Chen, H.; Guan, G. Odoribacter splanchnicus-derived extracellular vesicles alleviate inflammatory bowel disease by modulating gastrointestinal inflammation and intestinal barrier function via the NLRP3 inflammasome suppression. Mol. Med. 2025, 31, 56. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Zhang, X.; Tong, L.; Liu, Q.; Liang, X.; Bu, Y.; Gong, P.; Liu, T.; Zhang, L.; Xia, Y.; et al. Effect of Extracellular Vesicles Derived from Lactobacillus plantarum Q7 on Gut Microbiota and Ulcerative Colitis in Mice. Front. Immunol. 2021, 12, 777147. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Wang, Y.; Xiao, F.; Wang, X.; Li, X.; Cao, R.; Zhang, J.; Zhang, T.F. Prausnitzii-derived extracellular vesicles attenuate experimental colitis by regulating intestinal homeostasis in mice. Microb. Cell Factories 2023, 22, 235. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Hao, H.; Liu, Q.; Li, J.; Yao, Y.; Liu, Y.; Zhang, T.; Zhang, Z.; Yi, H. Effect of Extracelluar Vesicles Derived from Akkermansia muciniphila on Intestinal Barrier in Colitis Mice. Nutrients 2023, 15, 4722. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, L.; Yang, Y.; Deng, H.; Su, S.; Xia, Y.; Su, J.; Liu, Y.; Wu, J.; Zhang, J.; et al. Bacteroides fragilis-Derived Outer Membrane Vesicles Deliver MiR-5119 and Alleviate Colitis by Targeting PD-L1 to Inhibit GSDMD-Mediated Neutrophil Extracellular Trap Formation. Adv. Sci. 2025, 12, e00781. [Google Scholar] [CrossRef]
- Yamasaki-Yashiki, S.; Kawashima, F.; Saika, A.; Hosomi, R.; Kunisawa, J.; Katakura, Y. RNA-Based Anti-Inflammatory Effects of Membrane Vesicles Derived from Lactiplantibacillus plantarum. Foods 2024, 13, 967. [Google Scholar] [CrossRef]
- Tong, L.; Zhang, X.; Hao, H.; Liu, Q.; Zhou, Z.; Liang, X.; Liu, T.; Gong, P.; Zhang, L.; Zhai, Z.; et al. Lactobacillus rhamnosus GG Derived Extracellular Vesicles Modulate Gut Microbiota and Attenuate Inflammatory in DSS-Induced Colitis Mice. Nutrients 2021, 13, 3319. [Google Scholar] [CrossRef]
- Kang, E.A.; Choi, H.-I.; Hong, S.W.; Kang, S.; Jegal, H.-Y.; Choi, E.W.; Park, B.-S.; Kim, J.S. Extracellular Vesicles Derived from Kefir Grain Lactobacillus Ameliorate Intestinal Inflammation via Regulation of Proinflammatory Pathway and Tight Junction Integrity. Biomedicines 2020, 8, 522. [Google Scholar] [CrossRef]
- Gupta, M.; Mishra, V.; Gulati, M.; Kapoor, B.; Kaur, A.; Gupta, R.; Tambuwala, M.M. Natural compounds as safe therapeutic options for ulcerative colitis. Inflammopharmacology 2022, 30, 397–434. [Google Scholar] [CrossRef]
- Liu, T.; Ning, Z.; Liu, P.; Gao, H. Cassane diterpenoid ameliorates dextran sulfate sodium-induced experimental colitis by regulating gut microbiota and suppressing tryptophan metabolism. Front. Immunol. 2023, 13, 1045901. [Google Scholar] [CrossRef]
- Yuan, S.; Li, Y.; Li, J.; Xue, J.-C.; Wang, Q.; Hou, X.-T.; Meng, H.; Nan, J.-X.; Zhang, Q.-G. Traditional Chinese Medicine and Natural Products: Potential Approaches for Inflammatory Bowel Disease. Front. Pharmacol. 2022, 13, 892790. [Google Scholar] [CrossRef] [PubMed]
- Dad, H.A.; Gu, T.-W.; Zhu, A.-Q.; Huang, L.-Q.; Peng, L.-H. Plant Exosome-like Nanovesicles: Emerging Therapeutics and Drug Delivery Nanoplatforms. Mol. Ther. 2021, 29, 13–31. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Park, J.; Sohn, Y.; Oh, C.-E.; Park, J.-H.; Yuk, J.-M.; Yeon, J.-H. Stability of Plant Leaf-Derived Extracellular Vesicles According to Preservative and Storage Temperature. Pharmaceutics 2022, 14, 457. [Google Scholar] [CrossRef]
- Zhuang, X.; Deng, Z.-B.; Mu, J.; Zhang, L.; Yan, J.; Miller, D.; Feng, W.; McClain, C.J.; Zhang, H.-G. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J. Extracell. Vesicles 2015, 4, 28713. [Google Scholar] [CrossRef]
- Gao, C.; Zhou, Y.; Chen, Z.; Li, H.; Xiao, Y.; Hao, W.; Zhu, Y.; Vong, C.T.; Farag, M.A.; Wang, Y.; et al. Turmeric-derived nanovesicles as novel nanobiologics for targeted therapy of ulcerative colitis. Theranostics 2022, 12, 5596–5614. [Google Scholar] [CrossRef]
- Liu, C.; Yan, X.; Zhang, Y.; Yang, M.; Ma, Y.; Zhang, Y.; Xu, Q.; Tu, K.; Zhang, M. Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for murine colitis therapy. J. Nanobiotechnol. 2022, 20, 206. [Google Scholar] [CrossRef]
- Yang, S.; Li, W.; Bai, X.; Di Nunzio, G.; Fan, L.; Zhao, Y.; Ren, L.; Zhao, R.; Bian, S.; Liu, M.; et al. Ginseng-derived nanoparticles alleviate inflammatory bowel disease via the TLR4/MAPK and p62/Nrf2/Keap1 pathways. J. Nanobiotechnol. 2024, 22, 48. [Google Scholar] [CrossRef]
- Zhu, Z.; Liao, L.; Gao, M.; Liu, Q. Garlic-derived exosome-like nanovesicles alleviate dextran sulphate sodium-induced mouse colitis via the TLR4/MyD88/NF-κB pathway and gut microbiota modulation. Food Funct. 2023, 14, 7520–7534. [Google Scholar] [CrossRef] [PubMed]
- Tinnirello, V.; Zizzo, M.G.; Conigliaro, A.; Tabone, M.; Ganji, N.R.; Cicio, A.; Bressa, C.; Larrosa, M.; Rappa, F.; Vergilio, G.; et al. Industrial-produced lemon nanovesicles ameliorate experimental colitis-associated damages in rats via the activation of anti-inflammatory and antioxidant responses and microbiota modification. Biomed. Pharmacother. 2024, 174, 116514. [Google Scholar] [CrossRef] [PubMed]
- Zu, M.; Song, H.; Zhang, J.; Chen, Q.; Deng, S.; Canup, B.S.B.; Yuan, Y.; Xiao, B. Lycium barbarum lipid-based edible nanoparticles protect against experimental colitis. Colloids Surf. B Biointerfaces 2020, 187, 110747. [Google Scholar] [CrossRef]
- Sriwastva, M.K.; Deng, Z.-B.; Wang, B.; Teng, Y.; Kumar, A.; Sundaram, K.; Mu, J.; Lei, C.; Dryden, G.W.; Xu, F.; et al. Exosome-like nanoparticles from Mulberry bark prevent DSS-induced colitis via the AhR/COPS8 pathway. Embo Rep. 2022, 23, e53365. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-H.; Eom, J.-Y.; Kim, H.-J.; Seo, W.; Kwun, H.-J.; Kim, D.-K.; Kim, J.; Cho, Y.-E. Aloe-derived nanovesicles attenuate inflammation and enhance tight junction proteins for acute colitis treatment. Biomater. Sci. 2023, 11, 5490–5501. [Google Scholar] [CrossRef]
- Ju, S.; Mu, J.; Dokland, T.; Zhuang, X.; Wang, Q.; Jiang, H.; Xiang, X.; Deng, Z.-B.; Wang, B.; Zhang, L.; et al. Grape Exosome-like Nanoparticles Induce Intestinal Stem Cells and Protect Mice from DSS-Induced Colitis. Mol. Ther. 2013, 21, 1345–1357. [Google Scholar] [CrossRef]
- Kim, J.; Zhang, S.; Zhu, Y.; Wang, R.; Wang, J. Amelioration of colitis progression by ginseng-derived exosome-like nanoparticles through suppression of inflammatory cytokines. J. Ginseng Res. 2023, 47, 627–637. [Google Scholar] [CrossRef]
- Deng, Z.; Rong, Y.; Teng, Y.; Mu, J.; Zhuang, X.; Sannykutty, A.; Zhang, L.; Yan, J.; Miller, D.; Suttles, J.; et al. Broccoli-derived nanoparticle inhibits mouse colitis by activating dendritic cell AMP-activated protein kinase. J. Immunol. 2017, 25, 1641–1654. [Google Scholar]
- Yang, S.; Fan, L.; Yin, L.; Zhao, Y.; Li, W.; Zhao, R.; Jia, X.; Dong, F.; Zheng, Z.; Zhao, D.; et al. Ginseng exosomes modulate M1/M2 polarisation by activating autophagy and target IKK/IкB/NF-кB to alleviate inflammatory bowel disease. J. Nanobiotechnol. 2025, 23, 198. [Google Scholar] [CrossRef]
- Wong, C.K.; Laos, A.J.; Soeriyadi, A.H.; Wiedenmann, J.; Curmi, P.M.G.; Gooding, J.J.; Marquis, C.P.; Stenzel, M.H.; Thordarson, P. Polymersomes Prepared from Thermoresponsive Fluorescent Protein-Polymer Bioconjugates: Capture of and Report on Drug and Protein Payloads. Angew. Chem.-Int. Ed. 2015, 54, 5317–5322. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Zheng, M.; Yang, W.; Meng, F.; Miyata, K.; Kim, H.J.; Kataoka, K.; Zhong, Z. Virus-Mimicking Chimaeric Polymersomes Boost Targeted Cancer siRNA Therapy In Vivo. Adv. Mater. 2017, 29, 1703285. [Google Scholar] [CrossRef] [PubMed]
- Negut, I.; Bita, B. Polymersomes as Innovative, Stimuli-Responsive Platforms for Cancer Therapy. Pharmaceutics 2024, 16, 463. [Google Scholar] [CrossRef]
- Deng, C.; Jiang, Y.; Cheng, R.; Meng, F.; Zhong, Z. Biodegradable polymeric micelles for targeted and controlled anticancer drug delivery: Promises, progress and prospects. Nano Today 2012, 7, 467–480. [Google Scholar] [CrossRef]
- Molinaro, R.; Corbo, C.; Martinez, J.O.; Taraballi, F.; Evangelopoulos, M.; Minardi, S.; Yazdi, I.K.; Zhao, P.; De Rosa, E.; Sherman, M.B.; et al. Biomimetic proteolipid vesicles for targeting inflamed tissues. Nat. Mater. 2016, 15, 1037–1046. [Google Scholar] [CrossRef]
- Steidler, L.; Neirynck, S.; Huyghebaert, N.; Snoeck, V.; Vermeire, A.; Goddeeris, B.; Cox, E.; Remon, J.P.; Remaut, E. Biological containment of genetically modified Lactococcus lactis for intestinal delivery of human interleukin 10. Nat. Biotechnol. 2003, 21, 785–789. [Google Scholar] [CrossRef]
- Braat, H.; Rottiers, P.; Hommes, D.W.; Huyghebaert, N.; Remaut, E.; Remon, J.-P.; Van Deventer, S.J.H.; Neirynck, S.; Peppelenbosch, M.P.; Steidler, L. A phase I trial with Transgenic bacteria expressing interleukin-10 in Crohn’s disease. Clin. Gastroenterol. Hepatol. 2006, 4, 754–759. [Google Scholar] [CrossRef]
- Hanson, M.L.; Hixon, J.A.; Li, W.; Felber, B.K.; Anver, M.R.; Stewart, C.A.; Janelsins, B.M.; Datta, S.K.; Shen, W.; McLean, M.H.; et al. Oral Delivery of IL-27 Recombinant Bacteria Attenuates Immune Colitis in Mice. Gastroenterology 2014, 146, 210–221.e13. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, Q.; Shen, R.; Peng, L.; Zhou, W.; Meng, T.; Hu, F.; Wang, J.; Yuan, H. ROS-responsive nanoparticles targeting inflamed colon for synergistic therapy of inflammatory bowel disease via barrier repair and anti-inflammation. Nano Res. 2024, 17, 5409–5423. [Google Scholar] [CrossRef]
- Hummel, T.; Tak, E.; Maurice-Stam, H.; Benninga, M.A.; Kindermann, A.; Grootenhuis, M.A. The Course of Life and Health-Related Quality of Life of Adolescents with Inflammatory Bowel Disease. Gastroenterology 2011, 140, S511. [Google Scholar] [CrossRef]
- Han, L.; Liu, X.-W.; Li, W.-X.; Zang, T.; Li, Z.-P.; Deng, Y.; Tan, L.-L.; Liang, D.-S.; Zhang, X.-J.; Shen, X.-F.; et al. Innovative probiotic Escherichia coli nissle 1917-derived outer membrane vesicles coupled microspheres demonstrate anti-inflammatory and barrier-enhancing properties in colitis. Chem. Eng. J. 2024, 487, 150711. [Google Scholar] [CrossRef]
- Song, C.; Wu, J.; Wu, J.; Wang, F. MnO2 and roflumilast-loaded probiotic membrane vesicles mitigate experimental colitis by synergistically augmenting cAMP in macrophage. J. Nanobiotechnol. 2024, 22, 294. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chatterjee, I.; Gujral, T.; Alakkam, A.; Coffing, H.; Anbazhagan, A.N.; Borthakur, A.; Saksena, S.; Gill, R.K.; Alrefai, W.A.; et al. Activation of Nuclear Factor—κB by Tumor Necrosis Factor in Intestinal Epithelial Cells and Mouse Intestinal Epithelia Reduces Expression of the Chloride Transporter SLC26A3. Gastroenterology 2017, 153, 1338–1350.e3. [Google Scholar] [CrossRef]
- Song, Y.; Zhao, Y.; Ma, Y.; Wang, Z.; Rong, L.; Wang, B.; Zhang, N. Biological functions of NLRP3 inflammasome: A therapeutic target in inflammatory bowel disease. Cytokine Growth Factor Rev. 2021, 60, 61–75. [Google Scholar] [CrossRef]
- Stylianou, P.; Rushwan, S.; Wang, W.; Miah, M.A.; Darweesh, O.; Mackinnon, A.; Roach, K.M.; Hitchman, C.J.; Gonchar, O.; Thorpe, S.; et al. CD98 is critical for a conserved inflammatory response to diverse injury stimuli relevant to IPF exacerbations and COVID pneumonitis. bioRxiv 2022. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, X.; Han, M.K.; Collins, J.F.; Merlin, D. Oral administration of ginger-derived nanolipids loaded with siRNA as a novel approach for efficient siRNA drug delivery to treat ulcerative colitis. Nanomedicine 2017, 12, 1927–1943. [Google Scholar] [CrossRef]
- Wang, D.; Xue, H.; Tan, J.; Liu, P.; Qiao, C.; Pang, C.; Zhang, L. Bone marrow mesenchymal stem cells-derived exosomes containing miR-539-5p inhibit pyroptosis through NLRP3/caspase-1 signalling to alleviate inflammatory bowel disease. Inflamm. Res. 2022, 71, 833–846. [Google Scholar] [CrossRef]
- Arabpour, M.; Saghazadeh, A.; Rezaei, N. Anti-inflammatory and M2 macrophage polarization-promoting effect of mesenchymal stem cell-derived exosomes. Int. Immunopharmacol. 2021, 97, 107823. [Google Scholar] [CrossRef]
- Zhang, M.; Xu, X.; Su, L.; Zeng, Y.; Lin, J.; Li, W.; Zou, Y.; Li, S.; Lin, B.; Li, Z.; et al. Oral administration of Sophora Flavescens-derived exosomes-like nanovesicles carrying CX5461 ameliorates DSS-induced colitis in mice. J Nanobiotechnol. 2024, 22, 607. [Google Scholar] [CrossRef]
- Xu, H.; Fu, J.; Chen, L.; Zhou, S.; Fang, Y.; Zhang, Q.; Chen, X.; Yuan, L.; Li, Y.; Xu, Z.; et al. TNF-α Enhances the Therapeutic Effects of MenSC-Derived Small Extracellular Vesicles on Inflammatory Bowel Disease through Macrophage Polarization by miR-24-3p. Stem Cells Int. 2023, 2023, 2988907. [Google Scholar] [CrossRef]
- Zhao, Y.; Yin, W.; Yang, Z.; Sun, J.; Chang, J.; Huang, L.; Xue, L.; Zhang, X.; Zhi, H.; Chen, S.; et al. Nanotechnology-enabled M2 macrophage polarization and ferroptosis inhibition for targeted inflammatory bowel disease treatment. J. Control. Release 2024, 367, 339–353. [Google Scholar] [CrossRef]
- Kang, S.J.; Lee, J.H.; Rhee, W.J. Engineered plant-derived extracellular vesicles for targeted regulation and treatment of colitis-associated inflammation. Theranostics 2024, 14, 5643–5661. [Google Scholar] [CrossRef]
- Jiang, Q.; Wang, L.; Si, X.; Cui, H.; Li, J.; Tian, J.; Bao, Y.; Xie, X.; Xu, A.; Sun, Y.; et al. An anthocyanins-loaded extracellular vesicles coated with fucoidan nano-delivery system for enhancing dietary defense against inflammation-related colon injury. Int. J. Biol. Macromol. 2025, 318, 144948. [Google Scholar] [CrossRef]
- Ho, G.-T.; Theiss, A.L. Mitochondria and Inflammatory Bowel Diseases: Toward a Stratified Therapeutic Intervention. Annu. Rev. Physiol. 2022, 84, 435–459. [Google Scholar] [CrossRef]
- Lu, G.; Lu, S.; Dai, H.; Zhang, F.; Wang, X.; Li, W.; Mei, L.; Tan, H. Engineered Turmeric-Derived Nanovesicles for Ulcerative Colitis Therapy by Attenuating Oxidative Stress and Alleviating Inflammation. Mol. Pharm. 2025, 22, 2159–2167. [Google Scholar] [CrossRef]
- Yue, S.; Gong, L.; Tan, Y.; Zhang, X.; Liao, F. Cerium Oxide-Loaded Exosomes Derived from Regulatory T Cells Ameliorate Inflammatory Bowel Disease by Scavenging Reactive Oxygen Species and Modulating the Inflammatory Response. J. Inflamm. Res. 2025, 18, 4395–4408. [Google Scholar] [CrossRef]
- Li, J.; Sun, M.; Liu, L.; Yang, W.; Sun, A.; Yu, J.; Liu, D.; Zhao, W.; Cheng, M.; He, Z.; et al. Nanoprobiotics for Remolding the Pro-inflammatory Microenvironment and Microbiome in the Treatment of Colitis. Nano Lett. 2023, 23, 8593–8601. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Yu, T.; Wang, W.; Wu, H.; Zhu, F.; Wei, C.; Gao, F.; Liu, C.; Fan, H. Exosomes derived from EphB2-overexpressing bone marrow mesenchymal stem cells regulate immune balance and repair barrier function. Biotechnol. Lett. 2023, 45, 601–617. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, J.S.; Lee, C.; Farhat, N.; Antounians, L.; Zani, A.; Li, B.; Pierro, A. Administration of extracellular vesicles derived from human amniotic fluid stem cells: A new treatment for necrotizing enterocolitis. Pediatr. Surg. Int. 2021, 37, 301–309. [Google Scholar] [CrossRef]
- Wei, Z.; Tang, X.; Yi, C.; Ocansey, D.K.W.; Mao, F.; Mao, Z. HucMSC-Ex alleviates DSS-induced colitis in mice by decreasing mast cell activation via the IL-33/ST2 axis. Am. J. Transl. Res. 2024, 16, 2727–2744. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.A.; Hennet, T. Mechanisms and consequences of intestinal dysbiosis. Cell. Mol. Life Sci. 2017, 74, 2959–2977. [Google Scholar] [CrossRef]
- Teng, Y.; Ren, Y.; Sayed, M.; Hu, X.; Lei, C.; Kumar, A.; Hutchins, E.; Mu, J.; Deng, Z.; Luo, C.; et al. Plant-Derived Exosomal MicroRNAs Shape the Gut Microbiota. Cell Host Microbe 2018, 24, 637–652.e8. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, L.; Sun, Y.; Liu, X.; Zhang, P.; Wang, Y.; Ding, G. Mesenchymal stem/stromal cells-derived exosomes: Possible therapeutic mechanism in inflammatory bowel disease. Hum. Cell 2025, 38, 111. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.D. Apoptosis: Implications for inflammatory bowel disease. Inflamm. Bowel Dis. 2000, 6, 191–205. [Google Scholar] [CrossRef]
- Xiao, Z.; Xie, J.; Zhao, X.; Chen, X.; Lu, Y.; Xu, Y.; Wu, M.; An, L.; Li, Q. Role of Pyroptosis in inflammatory bowel disease. Int. Immunopharmacol. 2025, 155, 114619. [Google Scholar] [CrossRef]
- Li, L.; Lam, I.; Wang, J.; Yu, H.; Chan, C.; Cai, S. Epigenetic mechanism of iPSC-MSC-EVs in colonic epithelial cell pyroptosis in ulcerative colitis cell models via modulation of ELF3/miR-342-3p/ KDM6B axis and histone methylation. Int. Immunopharmacol. 2025, 157, 114704. [Google Scholar] [CrossRef]
- Zhao, C.; Lin, S. PANoptosis in intestinal epithelium: Its significance in inflammatory bowel disease and a potential novel therapeutic target for natural products. Front. Immunol. 2025, 15, 1507065. [Google Scholar] [CrossRef]
- Tan, C.; Xiang, Z.; Wang, S.; He, H.; Li, X.; Xu, M.; Guo, X.; Pu, Y.; Zhen, J.; Dong, W. Diosmin alleviates colitis by inhibiting PANoptosis of intestinal epithelial cells and regulating gut microbiota and metabolites. Phytomedicine 2025, 141, 156671. [Google Scholar] [CrossRef]
- Gong, W.; Liu, Z.; Wang, Y.; Huang, W.; Yang, K.; Gao, Z.; Guo, K.; Xiao, Z.; Zhao, W. Reprogramming of Treg cell-derived small extracellular vesicles effectively prevents intestinal inflammation from PANoptosis by blocking mitochondrial oxidative stress. Trends Biotechnol. 2025, 43, 893–917. [Google Scholar] [CrossRef]
- Zhu, Y.; Qin, H.; Sun, C.; Shao, B.; Li, G.; Qin, Y.; Kong, D.; Ren, S.; Wang, H.; Wang, Z.; et al. Endometrial Regenerative Cell-Derived Exosomes Attenuate Experimental Colitis through Downregulation of Intestine Ferroptosis. Stem Cells Int. 2022, 2022, 3014123. [Google Scholar] [CrossRef]
- Tong, Q.; Li, K.; Huang, F.; Dai, Y.; Zhang, T.; Muaibati, M.; Abuduyilimu, A.; Huang, X. Extracellular vesicles hybrid plasmid-loaded lipid nanovesicles for synergistic cancer immunotherapy. Mater. Today Bio 2023, 23, 100845. [Google Scholar] [CrossRef]
- Nie, M.; Huang, D.; Chen, G.; Zhao, Y.; Sun, L. Bioadhesive Microcarriers Encapsulated with IL-27 High Expressive MSC Extracellular Vesicles for Inflammatory Bowel Disease Treatment. Adv. Sci. 2023, 10, e2303349. [Google Scholar] [CrossRef] [PubMed]
- Du, R.; Chen, S.; Han, C.; He, Z.; Pei, H.; Yang, Y. M1 intestinal macrophages-derived exosomes promote colitis progression and mucosal barrier injury. Aging 2024, 16, 5703–5710. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Lin, X.; Wang, Q.; An, X.; Zhao, X.; Lin, Y.; Sun, T.; Yan, C.; Cai, A.; Cao, W.; et al. Dual-targeting celecoxib nanoparticles protect intestinal epithelium and regulate macrophage polarization for ulcerative colitis treatment. Chem. Eng. J. 2023, 452, 139445. [Google Scholar] [CrossRef]
- Sun, S.; Lv, W.; Li, S.; Zhang, Q.; He, W.; Min, Z.; Teng, C.; Chen, Y.; Liu, L.; Yin, J.; et al. Smart Liposomal Nanocarrier Enhanced the Treatment of Ischemic Stroke through Neutrophil Extracellular Traps and Cyclic Guanosine Monophosphate-Adenosine Monophosphate Synthase-Stimulator of Interferon Genes (cGAS-STING) Pathway Inhibition of Ischemic Penumbra. ACS Nano 2023, 17, 17845–17857. [Google Scholar] [CrossRef]
- Wu, J.; Ye, W.; Yu, J.; Zhou, T.; Zhou, N.; Ng, D.K.P.; Li, Z. Engineered bacteria and bacterial derivatives as advanced therapeutics for inflammatory bowel disease. Essays Biochem. 2025. [Google Scholar] [CrossRef] [PubMed]
| Category | Source | Size (nm) | Markers | Biogenesis | Features | Refs. |
|---|---|---|---|---|---|---|
| MDMVs | Mammalian cells | 30–150 | CD9, CD63, CD81 | Endosomal budding to form multivesicular bodies, followed by membrane fusion |
| [21,22] |
| BMVs | Bacteria (Gram-negative, Gram-positive) | 10–400 | Gram-negative: LPS, OmpA/OmpC/OmpF; Gram-positive: LTA, Peptidoglycan | Outward budding from bacterial membrane |
| [23,24] |
| PDNVs | Plants (fruits, vegetables, herbs, etc.) | 50–500 | PM H+-ATPase, PATL1 | Plasma membrane invagination and exocytosis |
| [25] |
| Liposomes | Phospholipids, cholesterol | 50–200 | DSPC, PEG-DSPE | Self-assembly in aqueous solution |
| [26,27,28] |
| Polymersomes | synthetic amphiphilic block copolymers | 70–300 | PEG-PLA | Self-assembly in aqueous solution | [29,30,31] | |
| EBMVs | Artificial assembly (cell membrane + synthetic materials) | 20–300 | Related to molecular signatures of parent cell membranes | Coating nanoparticle cores with natural or hybrid membrane | [32,33] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Li, Z.; Gu, Y.; Gu, R. Therapeutic Effect of Membrane Vesicle Drug Delivery Systems in Inflammatory Bowel Disease. Pharmaceutics 2025, 17, 1127. https://doi.org/10.3390/pharmaceutics17091127
Zhao Z, Li Z, Gu Y, Gu R. Therapeutic Effect of Membrane Vesicle Drug Delivery Systems in Inflammatory Bowel Disease. Pharmaceutics. 2025; 17(9):1127. https://doi.org/10.3390/pharmaceutics17091127
Chicago/Turabian StyleZhao, Zhe, Ziyun Li, Yihuang Gu, and Renjun Gu. 2025. "Therapeutic Effect of Membrane Vesicle Drug Delivery Systems in Inflammatory Bowel Disease" Pharmaceutics 17, no. 9: 1127. https://doi.org/10.3390/pharmaceutics17091127
APA StyleZhao, Z., Li, Z., Gu, Y., & Gu, R. (2025). Therapeutic Effect of Membrane Vesicle Drug Delivery Systems in Inflammatory Bowel Disease. Pharmaceutics, 17(9), 1127. https://doi.org/10.3390/pharmaceutics17091127






