Compounding of Liquid and Solid Dose Adjustable Formulations with Pantoprazole: Comparison of Stability, Applicability and Suitability
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Compounding of Syrup Formulations
2.3. Characterization of Compounded Syrup Formulations
2.3.1. Determination of Pantoprazole Concentration, Physical Appearance and Measurements of pH Value
2.3.2. Microbiological Studies
2.4. Compounding of Capsule Formulations
2.5. Characterization of Compounded Capsule Formulations
2.5.1. Uniformity of Mass, Uniformity of Content and Assay
2.5.2. Dissolution Testing
2.6. Chemical Stability of Pantoprazole
2.7. Analytical Methods
2.7.1. UV/Vis Spectrophotometry
2.7.2. High-Performance Liquid Chromatography (HPLC)
2.8. Statistical Analyses
3. Results
3.1. Characterization of Compounded Syrup Formulations
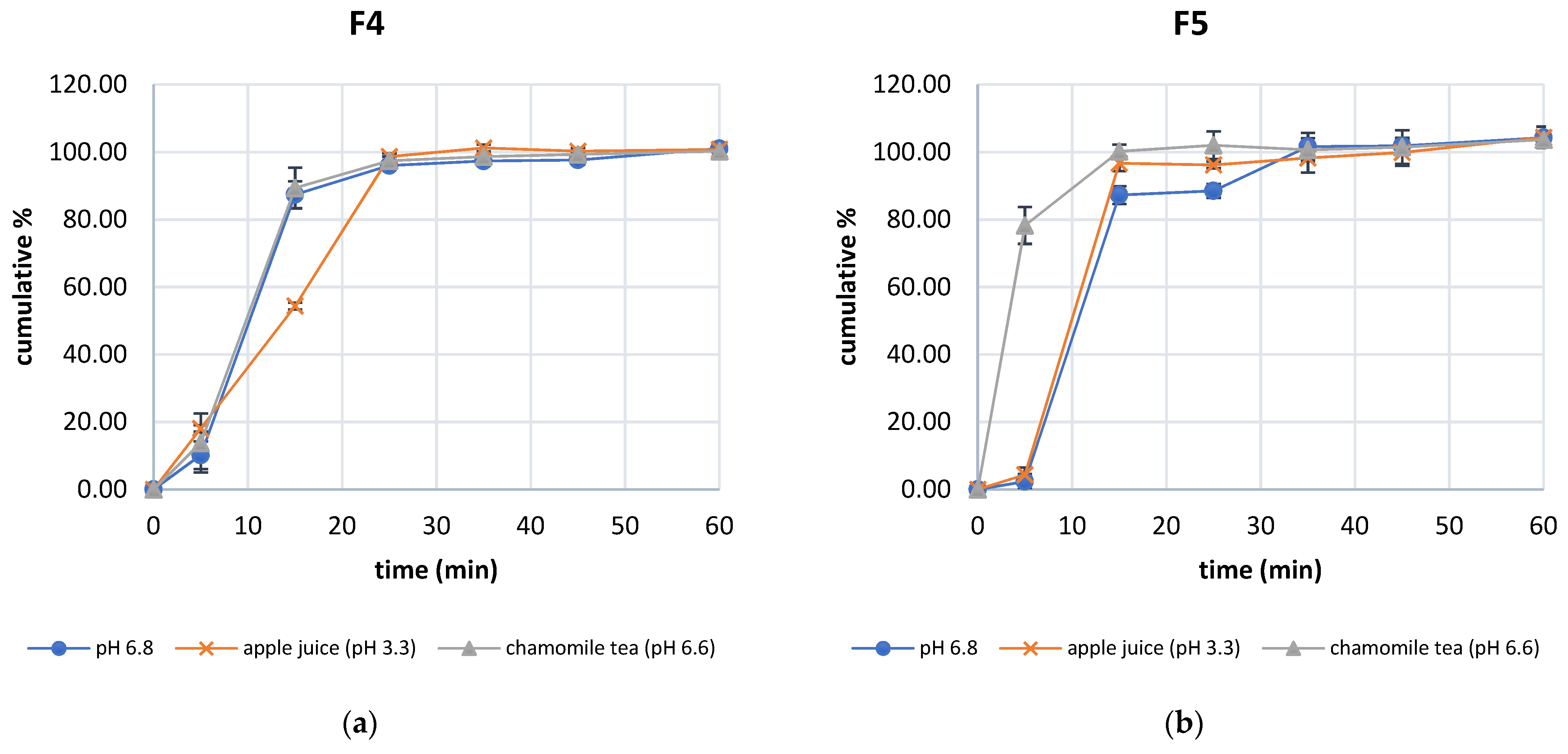
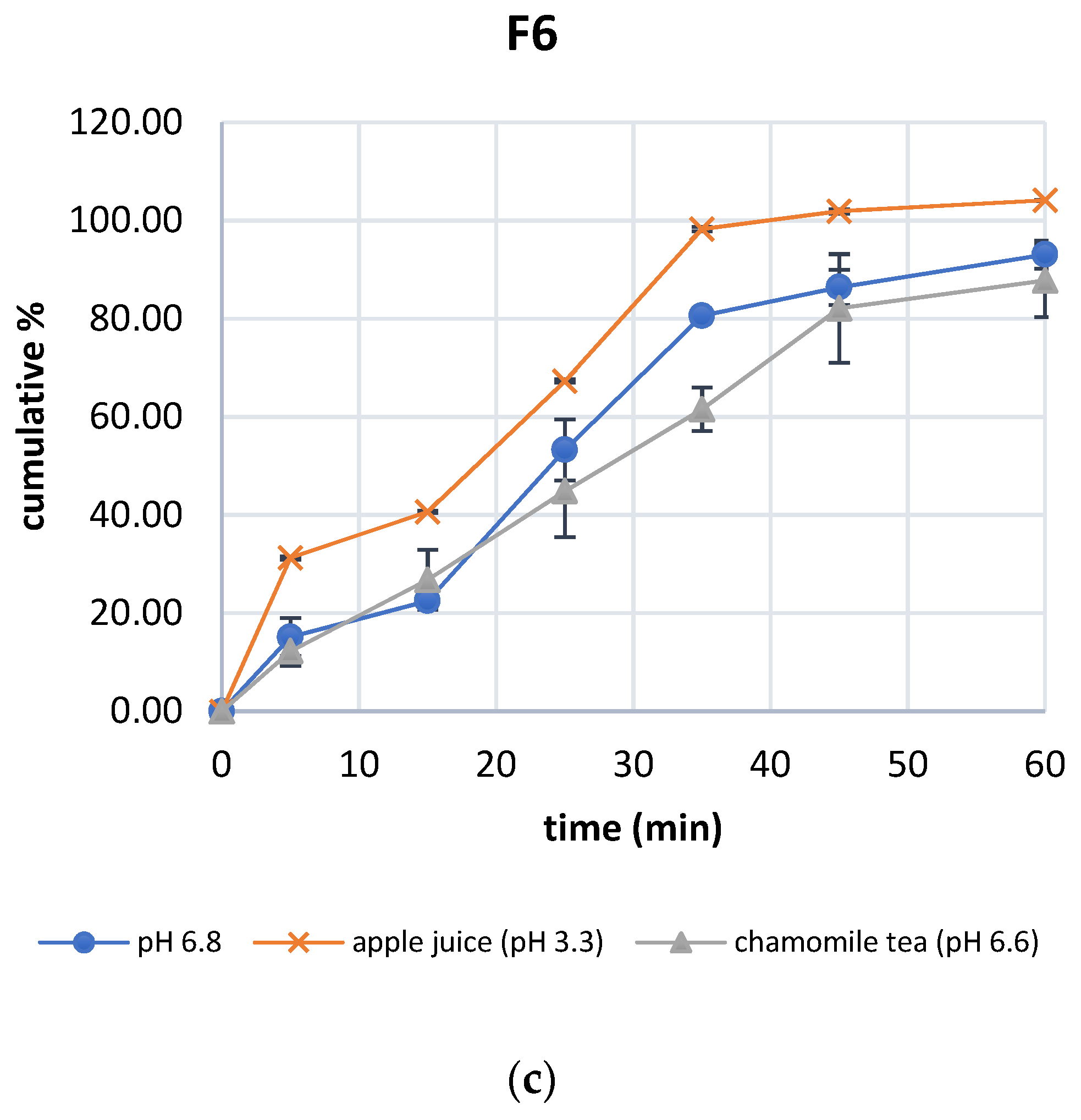
3.2. Characterization of Compounded Capsule Formulations
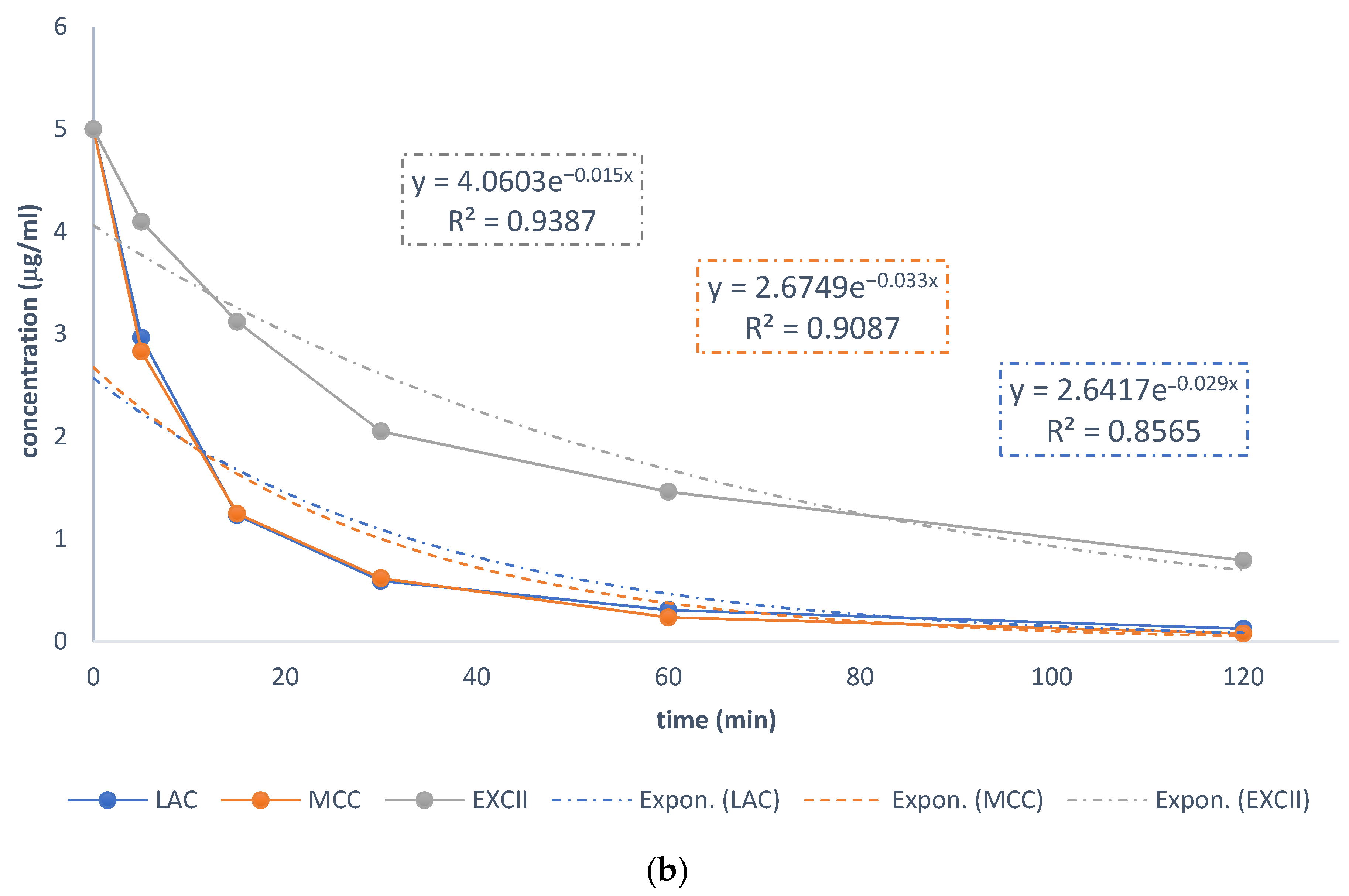
3.3. Stability of Pantoprazole in Various Tested Media
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, S.-H.; Kim, J.-E. Quality by Design Applied Development of Immediate-Release Rabeprazole Sodium Dry-Coated Tablet. Pharmaceutics 2021, 13, 259. [Google Scholar] [CrossRef]
- Geng, L.; Han, L.; Huang, L.; Wu, Z.; Wu, Z.; Qi, X. High Anti-Acid Omeprazole Lightweight Capsule for Gastro-Enteric System Acid-Related Disorders Treatment. J. Clin. Gastroenterol. Treat. 2019, 5, 1–11. [Google Scholar] [CrossRef]
- Kan, S.-L.; Lu, J.; Liu, J.-P.; Zhao, Y. Preparation and in vitro/in vivo evaluation of esomeprazole magnesium-modified release pellets. Drug Deliv. 2014, 23, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Srebro, J.; Brniak, W.; Mendyk, A. Formulation of Dosage Forms with Proton Pump Inhibitors: State of the Art, Challenges and Future Perspectives. Pharmaceutics 2022, 14, 2043. [Google Scholar] [CrossRef] [PubMed]
- Peery, A.F.; Dellon, E.S.; Lund, J.; Crockett, S.D.; McGowan, C.E.; Bulsiewicz, W.J.; Gangarosa, L.M.; Thiny, M.T.; Stizenberg, K.; Morgan, D.R.; et al. Burden of Gastrointestinal Disease in the United States: 2012 Update. Gastroenterology 2012, 143, 1179–1187.e3. [Google Scholar] [CrossRef]
- de Campo, D.R.; Klein, S.; Zolle, T.; Vieri, N.R.; Barro, F.A.; Meure, E.C.; Coelho, E.C.; Marchiorett, M.A.; Pedrazzoli, J. Evaluation of pantoprazole formulations in different dissolution apparatus using biorelevant medium. Arzneim. Forsch. Drug Res. 2010, 60, 42. [Google Scholar] [CrossRef]
- Shin, J.M.; Sachs, G. Pharmacology of proton pump inhibitors. Curr. Gastroenterol. Rep. 2008, 10, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Ekpe, A.; Jacobsen, T. Effect of Various Salts on the Stability of Lansoprazole, Omeprazole, and Pantoprazole as Determined by High-Performance Liquid Chromatography. Drug Dev. Ind. Pharm. 1999, 25, 1057–1065. [Google Scholar] [CrossRef] [PubMed]
- Roche, V.F. The Chemically Elegant Proton Pump Inhibitors. Am. J. Pharm. Educ. 2006, 70, 101. [Google Scholar] [CrossRef]
- European Pharmacopoeia, 10th ed.; European Directorate for the Quality of Medicines & Healthcare; Council of Europe: Strasbourg, France, 2019.
- Sachs, G.; Shin, J.M.; Briving, C.; Wallmark, B.; Hersey, S.J. The Pharmacology of the Gastric Acid Pump: The H+,K+ ATPase. Annu. Rev. Pharmacol. Toxicol. 1995, 35, 277–305. [Google Scholar] [CrossRef]
- Horn, J. The proton-pump inhibitors: Similarities and differences. Clin. Ther. 2000, 22, 266–280. [Google Scholar] [CrossRef]
- Escourrou, J.; Deprez, P.H.; Saggioro, A.; Geldof, H.; Fischer, R.; Maier, C.; The european pantoprazole study group. Maintenance therapy with pantoprazole 20 mg prevents relapse of reflux oesophagitis. Aliment. Pharmacol. Ther. 1999, 13, 1481–1491. [Google Scholar] [CrossRef] [PubMed]
- Lanza, F.L.; The Practice Parameters Committee of the American College of Gastroenterology; Chan, F.K.L.; Quigley, E.M.M. Guidelines for Prevention of NSAID-Related Ulcer Complications. Am. J. Gastroenterol. 2009, 104, 728–738. [Google Scholar] [CrossRef]
- Mansour-Ghanaei, F.; Pedarpour, Z.; Shafaghi, A.; Joukar, F. Clarithromycin versus Gemifloxacin in Quadruple Therapeutic Regimens for Helicobacter Pylori Infection Eradication. Middle East J. Dig. Dis. 2017, 9, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.M.; Kearns, G.L. Proton Pump Inhibitors in Pediatrics. Pediatr. Drugs 2013, 15, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Agency (FDA). Table of Pharmacogenetic Associations. Available online: https://www.fda.gov/medical-devices/precision-medicine/table-pharmacogenetic-associations (accessed on 22 December 2022).
- Gawrońska-Szklarz, B.; Adamiak-Giera, U.; Wyska, E.; Kurzawski, M.; Gornik, W.; Kaldonska, M.; Drozdzik, M. CYP2C19 polymorphism affects single-dose pharmacokinetics of oral pantoprazole in healthy volunteers. Eur. J. Clin. Pharmacol. 2012, 68, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Medicines and Medical Devices Agency of Serbia. Available online: https://www.alims.gov.rs/ (accessed on 22 December 2022).
- Standing, J.F.; Tuleu, C. Paediatric formulations—Getting to the heart of the problem. Int. J. Pharm. 2005, 300, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Malkawi, W.A.; AlRafayah, E.; AlHazabreh, M.; AbuLaila, S.; Al-Ghananeem, A.M. Formulation Challenges and Strategies to Develop Pediatric Dosage Forms. Children 2022, 9, 488. [Google Scholar] [CrossRef]
- Sivén, M.; Kovanen, S.; Siirola, O.; Hepojoki, T.; Isokirmo, S.; Laihanen, N.; Eränen, T.; Pellinen, J.; Juppo, A.M. Challenge of paediatric compounding to solid dosage forms sachets and hard capsules—Finnish perspective. J. Pharm. Pharmacol. 2016, 69, 593–602. [Google Scholar] [CrossRef]
- Reis, H.H.; Lima, L.M.; Reis, V.E.; Mota-Júnior, R.J.; Soares-Júnior, D.T.; Sillero-Quintana, M.; Rasmussen, H.E.; Carneiro-Júnior, M.A.; Marins, J.B. Effects of conventional and sugar-free energy drinks intake in runners: A double-blind, randomized, placebo-controlled crossover clinical trial. J. Sport. Med. Phys. Fit. 2021, 61, 928–934. [Google Scholar] [CrossRef]
- Barnes, A.R.; Nash, S. Stability of bendrofluazide in a low-dose extemporaneously prepared capsule. J. Clin. Pharm. Ther. 1994, 19, 89–93. [Google Scholar] [CrossRef]
- Fortner, J.; Salton, J.; Carlson, C.; Wheeler, R.; Cote, B.; Rao, D. Solid State Stability of Extemporaneously Prepared Levothyroxine Aliquots and Capsules. Int. J. Pharm. Compd. 2015, 19, 414–419. [Google Scholar]
- Brion, F.; Nunn, A.J.; Rieutord, A. Extemporaneous (magistral) preparation of oral medicines for children in European hospitals. Acta Paediatr. 2007, 92, 486–490. [Google Scholar] [CrossRef] [PubMed]
- The United States Pharmacopoea 43, The National Formulary 38, Rockville, Md: United States Pharmacopoeial Convention, Inc; NF Monographs: Vehicles for Oral Solution. 2020. Available online: http://www.pharmacopeia.cn/v29240/usp29nf24s0_m382.html (accessed on 6 August 2022).
- European Medicines Agency. European Union Herbal Monograph on Matricaria recutita L., flos. Available online: https://www.ema.europa.eu/en/documents/herbal-monograph/final-european-union-herbal-monograph-matricaria-recutita-l-flos_en.pdf (accessed on 22 December 2022).
- Bundesvereinigung Deutscher Apothek-erverbände ABDA (Ed.) Deutscher Arzneimittel-Codex/Neues Rezeptur-Formularium (DAC/NRF); Avoxa–Mediengruppe Deutscher Apotheker GmbH: Eschborn, Germany, 2018. [Google Scholar]
- Official Journal of the European Union. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2003:159:0046:0094:en:PDF (accessed on 22 December 2022).
- Pfizer Medical Information. Available online: https://www.pfizermedicalinformation.com/en-us/protonix/instructions (accessed on 22 December 2022).
- Ferron, G.M.; Ku, S.; Abell, M.; Unruh, M.; Getsy, J.; Mayer, P.R.; Paul, J. Oral bioavailability of pantoprazole suspended in sodium bicarbonate solution. Am. J. Health Pharm. 2003, 60, 1324–1329. [Google Scholar] [CrossRef]
- Milic, J.; Radojkovic, B.; Jancic-Stojanovic, B.; Draskovic, J.; Mirasevic, S.; Čalija, B. Investigation of omeprazole stability in oral suspensions for pediatric use prepared extemporaneously from omeprazole capsules. Arh. Za Farm. 2017, 67, 14–25. [Google Scholar] [CrossRef]
- Makeen, H.A.; Pancholi, S.S.; Ali, M.S.; Alam, M.I.; Alam, M.S.; Redaie, A.M.; Albarraq, A.A.; Al-Bratty, M.; Kadomi, Y.J.; Attiya, M.A.Q.; et al. Shelf life study of extemporaneously prepared omeprazole oral suspension. Saudi J. Health Sci. 2018, 7, 28. [Google Scholar] [CrossRef]
- Rafiq, A.; Gul, S.; Ajaz, A.; Fatima, S.; Mirza, A.Z. Quantitative Analysis of Pantoprazole Sodium Sesquihydrate in Bulk and Solid Dosage Form via UV-Spectrophotometric Method. Indian J. Pharm. Educ. Res. 2020, 54, 448–455. [Google Scholar] [CrossRef]
- Rahic, O.; Vranic, E.; Mujein, I. Development and Validation of HPLC Method for Determination of Pantoprazole in Pantoprazole Pellets. Int. J. Pharm. Teach. Pract. 2013, 4, 793–796. [Google Scholar]
- Kollipara, S.; Boddu, R.; Ahmed, T.; Chachad, S. Simplified Model-Dependent and Model-Independent Approaches for Dissolution Profile Comparison for Oral Products: Regulatory Perspective for Generic Product Development. AAPS PharmSciTech 2022, 23, 53. [Google Scholar] [CrossRef] [PubMed]
- Parrish, R.H.; Ashworth, L.D.; Löbenberg, R.; Benavides, S.; Cies, J.J.; MacArthur, R.B. Compounded Nonsterile Prepa-rations and FDA-Approved Commercially Available Liquid Products for Children: A North American Update. Pharmaceutics 2022, 14, 1032. [Google Scholar] [CrossRef] [PubMed]
- Dentinger, P.J.; Swenson, C.F.; Anaizi, N.H. Stability of pantoprazole in an extemporaneously compounded oral liquid. Am. J. Health Pharm. 2002, 59, 953–956. [Google Scholar] [CrossRef] [PubMed]
- Ley, L.; Stahlheber-Dilg, B.; Sander, P.; Huber, R.; Mascher, H.; Lücker, P. Bioavailability of a crushed pantoprazole tablet after buffering with sodium hydrogencarbonate or magaldrate relative to the intact enteric coated pantoprazole tablet. Methods Find. Exp. Clin. Pharmacol. 2001, 23, 41. [Google Scholar] [CrossRef]
- Yao, C.-L.; Ding, A.-M. Saccharides with Different Molecular Weight Affects Crystallization of Calcium Carbonate. Asian J. Chem. 2013, 25, 2939–2940. [Google Scholar] [CrossRef]
- Quercia, R.A.; Fan, C.; Liu, X.; Chow, M.S.S. Stability of omeprazole in an extemporaneously prepared oral liquid. Am. J. Health Pharm. 1997, 54, 1833–1836. [Google Scholar] [CrossRef]
- Polonini, H.C.; Silva, S.L.; Loures, S.; Almy, R.; Balland, A.; Brandão, M.A.F.; Ferreira, A.O. Compatibility of proton pump inhibitors in a preservative-free suspending vehicle. Eur. J. Hosp. Pharm. 2016, 25, 150–156. [Google Scholar] [CrossRef] [PubMed]
- FDA. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020987s045lbl.pdf (accessed on 28 December 2022).
- Campos, C.A.; Gerschenson, L.N. Effect of certain additives on sorbate stability. Food Res. Int. 1996, 29, 147–154. [Google Scholar] [CrossRef]
- Arya, S.; Thakur, B. Degradation products of sorbic acid in aqueous solutions. Food Chem. 1988, 29, 41–49. [Google Scholar] [CrossRef]
- Karbane, M.E.; ELalami, A.; Bouchafra, H.; Bourichi, H.; Cherrah, Y.; Azougagh, M.; Bouklouze, A. Stability study of parabens in pharmaceutical formulations containing Paracetamol or Carbocisteine by high-performance liquid chromtatography. Mor. J. Chem. 2015, 3, 3. [Google Scholar] [CrossRef]
- Van Riet-Nales, D.A.; De Neef, B.J.; Schobben, A.F.A.M.; Ferreira, J.A.; Egberts, T.C.G.; Rademaker, C.M.A. Acceptability of different oral formulations in infants and preschool children. Arch. Dis. Child. 2013, 98, 725–731. [Google Scholar] [CrossRef]
- World Health Organization. The Selection and Use of Essential Medicines. Available online: https://apps.who.int/iris/bitstream/handle/10665/112729/WHO_TRS_985_eng.pdf?sequence=1&isAllowed=y (accessed on 28 December 2022).
- European Medicines Agency. Reflection Paper: Formulations of Choice for the Paediatric Population. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-formulations-choice-paediatric-population_en.pdf (accessed on 28 December 2022).
- Patel, S.A.; Patel, N.G.; Joshi, A.B. Multiple unit pellet system (mups) based fast disintegrating delayed-release tablets for pantoprazole delivery. Int. J. Pharm. Pharm. Sci. 2018, 10, 77. [Google Scholar] [CrossRef]
- Bigoniya, P.; Shukla, A.; Singh, C.S.; Gotiya, P. Comparative anti-ulcerogenic study of pantoprazole formulation with and without sodium bicarbonate buffer on pyloric ligated rat. J. Pharmacol. Pharmacother. 2011, 2, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Rathnanand, M. Stability enhancement of proton pump inhibitor in stomach: Formulation and in-vitro evaluation of stabilized proton pump inhibitor. Asian J. Pharm. Clin. Res. 2017, 10, 88. [Google Scholar] [CrossRef]
- Hedayati, S.; Shahidi, F.; Koocheki, A.; Farahnaky, A.; Majzoobi, M. Physical properties of pregelatinized and granular cold water swelling maize starches at different pH values. Int. J. Biol. Macromol. 2016, 91, 730–735. [Google Scholar] [CrossRef] [PubMed]

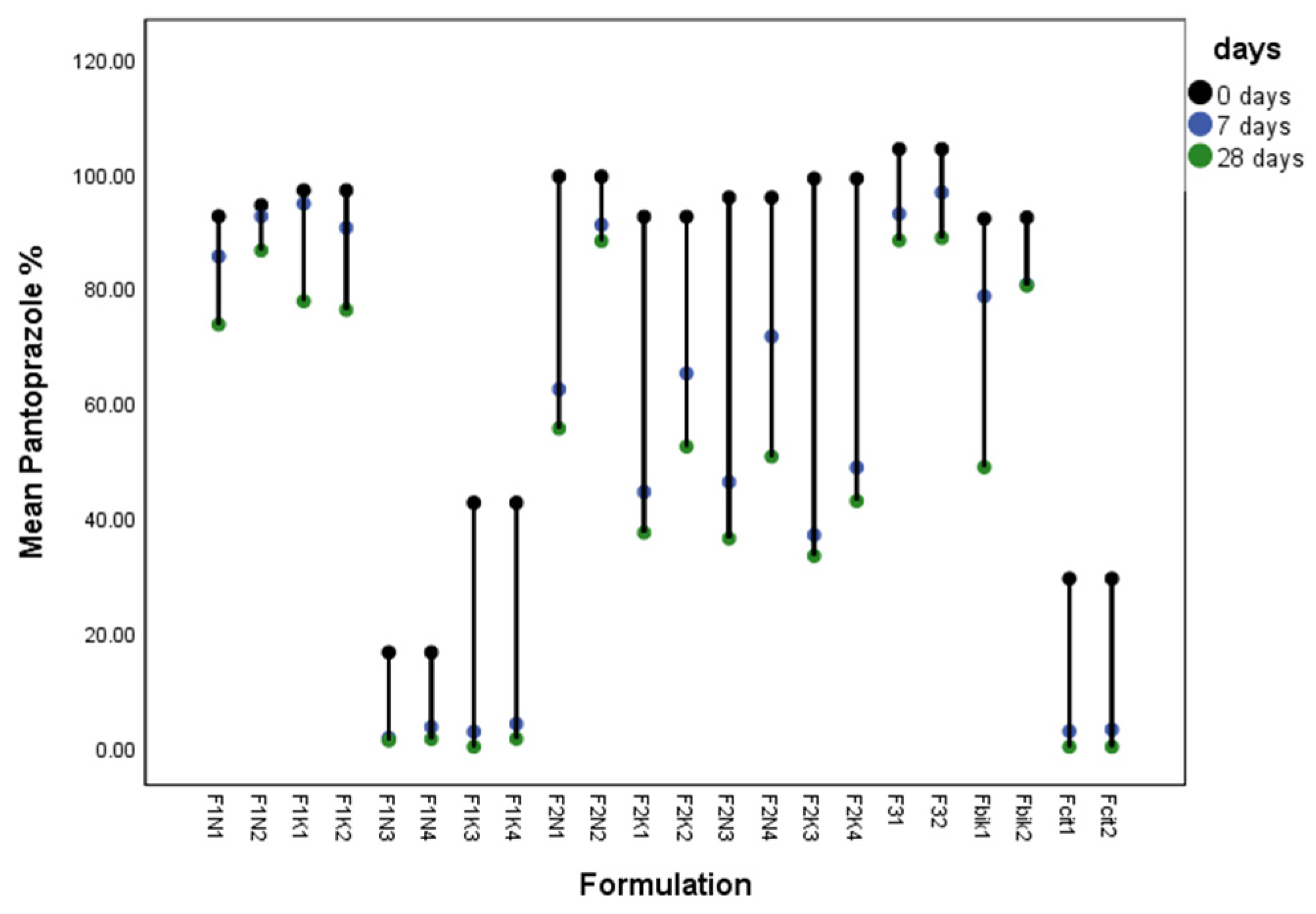

| Ingredients | F1 (USP43-NF38) | F2 (DAC/NRF2018) | F3 (SyrSpend) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH 8.4 | pH 4.5 | pH 8.4 | pH 6.5 | |||||||||||||||
| Presence of Preservative | ||||||||||||||||||
| no | yes | no | yes | no | yes | no | yes | |||||||||||
| Storage Temperature (°C) | ||||||||||||||||||
| 25 | 2–8 | 25 | 2–8 | 25 | 2–8 | 25 | 2–8 | 2–8 | 25 | 25 | 2–8 | 25 | 2–8 | 25 | 2–8 | 25 | 2–8 | |
| Labels | ||||||||||||||||||
| F1N1 | F1N2 | F1K1 | F1K2 | F1N3 | F1N4 | F1K3 | F1K4 | F2N1 | F2N2 | F2K1 | F2K2 | F2N3 | F2N4 | F2K3 | F2K4 | F3.1 | F3.2 | |
| PSS | 200 mg | 200 mg | 200 mg | 200 mg | 200 mg | 200 mg | 200 mg | 200 mg | 200 mg | |||||||||
| xanthan gum | 0.05 | 0.05 | 0.05 | 0.05 | ||||||||||||||
| glycerol | 10 mL | 10 mL | 10 mL | 10 mL | ||||||||||||||
| sorbitol | 25 mL | 25 mL | 25 mL | 25 mL | ||||||||||||||
| saccharin | 2 tbl | 2 tbl | 2 tbl | 2 tbl | ||||||||||||||
| HEC | 0.5 | 0.5 | 0.5 | 0.5 | ||||||||||||||
| glucose | 11.0 | 11.0 | 11.0 | 11.0 | ||||||||||||||
| potassium sorbate | 0.1 | 0.14 | ||||||||||||||||
| methyl paraben | 0.1 | 0.1 | 0.1 | |||||||||||||||
| citric acid | 1.5 | 1.5 | 0.07 | 0.07 | ||||||||||||||
| sodium citrate | 2.0 | 2.0 | ||||||||||||||||
| sodium bicarbonate | 8.4 | 8.4 | 8.4 | 8.4 | ||||||||||||||
| SyrSpend base | 100 mL | |||||||||||||||||
| distilled water | up to 100 mL | up to 100 mL | up to 100 mL | up to 100 mL | up to 100 g | up to 100 g | up to 100 g | up to 100 g | ||||||||||
| Ingredient | F4 | F5 | F6 |
|---|---|---|---|
| hard gelatin capsule (number) | 8 | 8 | 8 |
| tablet containing 40 mg pantoprazole * (number) | 1 | 1 | 1 |
| LAC (g) | 4.88 | ||
| MCC (g) | 2.68 | ||
| EXCPII ** (g) | 3.48 |
| F1N2 | F1K2 | F2N2 | F2K2 | F3.2 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| days | 0 | 7 | 28 | 0 | 7 | 28 | 0 | 7 | 28 | 0 | 7 | 28 | 0 | 7 | 28 |
| Total aerobic microbial count (TAMC) * | 10 | 10 | 40 | <10 | <10 | <10 | 10 | 10 | 20 | <10 | 20 | 40 | <10 | <10 | <10 |
| Total combined yeast and mold count (TYMC) ** | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 |
| Escherichia coli *** | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs | Abs |
| Sample | F4 | F5 | F6 | |||
|---|---|---|---|---|---|---|
| m (g) | % * | m (g) | % * | m (g) | % * | |
| Mass | Mass | Mass | ||||
| Content | Content | Content | ||||
| 1 | 0.505 | 5.930 | 0.363 | 0.854 | 0.478 | 0.018 |
| 2 | 0.549 | −2.325 | 0.367 | −0.113 | 0.479 | −0.087 |
| 3 | 0.558 | −3.851 | 0.367 | −0.252 | 0.477 | 0.319 |
| 4 | 0.545 | −1.476 | 0.370 | −1.181 | 0.478 | −0.038 |
| 5 | 0.521 | 3.026 | 0.367 | −0.107 | 0.475 | 0.645 |
| 6 | 0.556 | −3.530 | 0.367 | −0.129 | 0.479 | −0.120 |
| 7 | 0.546 | −1.716 | 0.367 | −0.260 | 0.477 | 0.221 |
| 8 | 0.543 | −1.202 | 0.369 | −0.700 | 0.483 | −0.917 |
| 9 | 0.554 | −3.232 | 0.368 | −0.443 | 0.478 | 0.005 |
| 10 | 0.535 | 0.420 | 0.364 | 0.485 | 0.484 | −1.302 |
| 11 | 0.533 | 0.718 | 0.367 | −0.293 | 0.486 | −1.580 |
| 12 | 0.509 | 5.137 | 0.365 | 0.234 | 0.477 | 0.300 |
| 13 | 0.549 | −2.215 | 0.361 | 1.313 | 0.476 | 0.428 |
| 14 | 0.520 | 3.060 | 0.357 | 2.468 | 0.473 | 1.091 |
| 15 | 0.536 | 0.236 | 0.374 | −2.131 | 0.476 | 0.447 |
| 16 | 0.513 | 4.494 | 0.358 | 2.173 | 0.481 | −0.576 |
| 17 | 0.538 | −0.124 | 0.364 | 0.647 | 0.472 | 1.367 |
| 18 | 0.537 | −0.084 | 0.372 | −1.691 | 0.477 | 0.248 |
| 19 | 0.544 | −1.362 | 0.372 | −1.689 | 0.479 | −0.078 |
| 20 | 0.547 | −1.902 | 0.363 | 0.814 | 0.480 | −0.392 |
| aver | 0.537 | 0.366 | 0.478 | |||
| min | −3.851 | −2.131 | −1.580 | |||
| max | 5.930 | 2.468 | 1.367 | |||
| Uniformity of Content | ||||||
|---|---|---|---|---|---|---|
| Sample | F4 | F5 | F6 | |||
| m (mg) | % * | m (mg) | % * | m (mg) | % * | |
| 1 | 4.972 | 94.82 | 5.174 | 99.71 | 5.142 | 98.53 |
| 2 | 5.595 | 106.70 | 5.168 | 99.61 | 5.585 | 107.02 |
| 3 | 5.772 | 110.07 | 5.345 | 103.01 | 4.914 | 94.16 |
| 4 | 4.865 | 92.78 | 5.221 | 100.62 | 4.899 | 93.87 |
| 5 | 4.993 | 95.22 | 5.096 | 98.21 | 5.602 | 107.34 |
| 6 | 5.012 | 95.58 | 5.321 | 102.55 | 5.009 | 95.98 |
| 7 | 5.398 | 102.94 | 4.995 | 96.27 | 5.432 | 104.08 |
| 8 | 4.998 | 95.31 | 5.563 | 107.21 | 5.113 | 97.97 |
| 9 | 5.478 | 104.47 | 4.902 | 94.47 | 5.289 | 101.34 |
| 10 | 5.354 | 102.10 | 5.102 | 98.33 | 5.205 | 99.73 |
| aver | 5.244 | 5.189 | 5.219 | |||
| min | 92.78 | 94.47 | 93.87 | |||
| max | 110.07 | 107.21 | 107.34 | |||
| Uniformity of Content after 28 days | ||||||
|---|---|---|---|---|---|---|
| Sample | F4 | F5 | F6 | |||
| m (mg) | % | m (mg) | % | m (mg) | % | |
| 1 | 5.186 | 98.92 | 5.311 | 102.23 | 5.184 | 99.41 |
| 2 | 5.699 | 108.71 | 5.167 | 99.46 | 5.396 | 103.47 |
| 3 | 5.211 | 99.40 | 5.394 | 103.82 | 5.747 | 110.20 |
| 4 | 5.138 | 98.01 | 5.018 | 96.59 | 5.059 | 97.00 |
| 5 | 5.415 | 103.29 | 5.242 | 100.90 | 5.112 | 98.02 |
| 6 | 4.998 | 95.34 | 5.285 | 101.73 | 5.086 | 97.52 |
| 7 | 5.258 | 100.30 | 5.022 | 96.67 | 5.101 | 97.81 |
| 8 | 5.263 | 100.39 | 5.386 | 103.67 | 5.247 | 100.61 |
| 9 | 5.248 | 100.11 | 5.125 | 98.65 | 5.118 | 98.14 |
| 10 | 5.009 | 95.55 | 5.001 | 96.26 | 5.101 | 97.81 |
| aver | 5.242 | 5.195 | 5.215 | |||
| min | 95.34 | 96.26 | 97.00 | |||
| max | 108.71 | 103.82 | 110.20 | |||
| Formulations | Content (% of Theoretical Value) | |
|---|---|---|
| After Compounding | After 28 Days | |
| F4 | 104.88 | 104.84 |
| F5 | 103.78 | 103.90 |
| F6 | 104.38 | 104.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Todorović, N.; Čanji Panić, J.; Zavišić, M.; Krtolica, J.; Ratajac, R.; Petrović, J.; Bosiljčić, D.; Kladar, N.; Milošević, N.; Lalić-Popović, M. Compounding of Liquid and Solid Dose Adjustable Formulations with Pantoprazole: Comparison of Stability, Applicability and Suitability. Pharmaceutics 2023, 15, 717. https://doi.org/10.3390/pharmaceutics15030717
Todorović N, Čanji Panić J, Zavišić M, Krtolica J, Ratajac R, Petrović J, Bosiljčić D, Kladar N, Milošević N, Lalić-Popović M. Compounding of Liquid and Solid Dose Adjustable Formulations with Pantoprazole: Comparison of Stability, Applicability and Suitability. Pharmaceutics. 2023; 15(3):717. https://doi.org/10.3390/pharmaceutics15030717
Chicago/Turabian StyleTodorović, Nemanja, Jelena Čanji Panić, Mina Zavišić, Jelena Krtolica, Radomir Ratajac, Jelena Petrović, Dušica Bosiljčić, Nebojša Kladar, Nataša Milošević, and Mladena Lalić-Popović. 2023. "Compounding of Liquid and Solid Dose Adjustable Formulations with Pantoprazole: Comparison of Stability, Applicability and Suitability" Pharmaceutics 15, no. 3: 717. https://doi.org/10.3390/pharmaceutics15030717
APA StyleTodorović, N., Čanji Panić, J., Zavišić, M., Krtolica, J., Ratajac, R., Petrović, J., Bosiljčić, D., Kladar, N., Milošević, N., & Lalić-Popović, M. (2023). Compounding of Liquid and Solid Dose Adjustable Formulations with Pantoprazole: Comparison of Stability, Applicability and Suitability. Pharmaceutics, 15(3), 717. https://doi.org/10.3390/pharmaceutics15030717






