Abstract
West Nile virus (WNV) is a mosquito-borne flavivirus with one of the widest global distributions. Since its discovery in Uganda in 1937, it has become a major zoonotic pathogen, and after its introduction into the United States in 1999, it spread rapidly across the Americas, becoming the leading cause of neuroinvasive arboviral disease. Its expansion illustrates a remarkable ecological adaptability, further intensified by climate change. In Latin America and the Caribbean, WNV circulation has been consistently documented in birds, horses, and mosquitoes; however, confirmed human cases remain disproportionately scarce compared with North America and Europe. Reports include sporadic human cases in Brazil (>100 since 2014), Mexico (~13), Argentina (2006–2007), Puerto Rico (2007), Nicaragua, and Haiti, while animal and vector evidence extends to Guatemala, El Salvador, Belize, Costa Rica, Bolivia, Paraguay, Colombia, Venezuela, Cuba, and Ecuador. This paradox likely reflects structural limitations within regional health systems, including underdiagnosis, restricted diagnostic capacity, and significant surveillance gaps, particularly in contexts where mild febrile syndromes may be misclassified as dengue, Zika, or Chikungunya. The regional risk of emergence is further amplified by climatic variability, ecological change, and intensifying human–wildlife interactions. Experiences from Europe highlight the importance of early detection, transfusion safety, and integrated surveillance within a One Health framework. Strengthening preparedness in Latin America will require investments in diagnostic infrastructure, implementation of standardized seroepidemiological surveys, development of predictive models tailored to local ecological contexts, and robust intersectoral collaboration.
1. Introduction
West Nile virus (WNV) is a mosquito-borne, zoonotic flavivirus and one of the most widely distributed arboviruses globally. It was first isolated in 1937 from the blood of a febrile patient in the West Nile District of Uganda and belongs to the Japanese encephalitis virus serocomplex, which also includes St. Louis encephalitis virus (SLEV), Murray Valley encephalitis virus (MVEV), and Alfuy virus (ALFV) [1,2].
Because West Nile virus and St. Louis encephalitis virus belong to the same Japanese encephalitis serocomplex, serological cross-reactivity driven by conserved epitopes is common and represents a major diagnostic challenge in flavivirus-endemic regions [3,4]. Clinical presentation alone is insufficient to reliably distinguish between these infections, in this context, initial screening is often based on IgM or IgG enzyme-linked immunosorbent assays; however, definitive differentiation between WNV and SLEV requires confirmatory plaque reduction neutralization tests (PRNTs), which remain the reference standard [5]. Molecular methods, including RT-PCR and genomic sequencing, allow specific identification during the acute viremic phase but are limited by the short duration of detectable viremia [6].
Initially regarded as a pathogen of limited human relevance due to its predominantly mild or subclinical presentations, WNV is now recognized as a major public health threat capable of causing significant morbidity and mortality in humans and a broad range of vertebrates, including birds, horses, reptiles, and small mammals [7].
The global epidemiology of WNV has shifted markedly in the past decades. In the Americas, following its introduction into the United States in 1999, the virus spread rapidly across the continent, becoming the leading cause of neuroinvasive arboviral disease [8]. Between 1999 and 2017, nearly 22,999 neuroinvasive cases were reported in the U.S., arising from millions of infections [9]. In Europe, the Middle East, and Russia, multiple large outbreaks have occurred, including the unprecedented 2018 European epidemic, which resulted in more than 1900 human cases [10]. More recently, autochthonous infections have been documented in regions previously unaffected, such as Germany and the Netherlands, underscoring the expanding ecological niche of WNV [11].
Recent epidemiological transitions highlight the remarkable adaptability of WNV to changing environments [12]. In Greece, evidence of overwintering in active mosquito populations during 2022 raised the possibility of year-round circulation in temperate regions [13]. Similarly, unusually high numbers of neuroinvasive cases reported in Israel as early as June 2024 illustrate how warmer winters and springs may accelerate viral amplification [14]. Modeling studies in southern Europe have projected that rising temperatures extend the seasonal window of transmission, while in the U.S., precipitation patterns and large-scale spatial synchrony of mosquito populations have been linked to outbreak intensity [15]. Collectively, these findings position climate change as a critical driver of WNV dynamics, reshaping vector distribution and geographic spread.
In contrast, in Latin America and the Caribbean, WNV circulation has been repeatedly documented in vectors and animal hosts, yet the number of confirmed human cases remains disproportionately low compared with North America and Europe. This paradox refers to the marked contrast between the extensive and sustained enzootic circulation of West Nile virus documented in birds, horses, and mosquito vectors across Latin America and the Caribbean, and the disproportionately low number of confirmed human cases reported in the region, particularly when compared with North America and Europe. This discrepancy raises critical questions regarding underdiagnosis, ecological and immunological factors, and structural gaps in regional surveillance capacity. Against this backdrop, this article aims to provide a comprehensive overview of current evidence on WNV infection in Latin America, and to examine its broader implications for public health and One Health strategies in the region.
2. Virology and Transmission Dynamics
The WNV is maintained in nature through a bird–mosquito transmission cycle. Birds act as the principal amplifying hosts, sustaining high-titer viremia that enables efficient transmission to mosquitoes, particularly those of the genus Culex [16]. In contrast, humans and horses are considered dead-end hosts, as their viremia levels are insufficient to perpetuate the cycle [17]. While mosquito bites remain the predominant route of transmission, alternative pathways have been documented in experimental and clinical contexts, including blood transfusion, organ transplantation, intrauterine transmission, and breastfeeding [1]. Viral shedding in urine further suggests that environmentally mediated transmission may be possible.
In the Americas, Cx. pipiens, Cx. quinquefasciatus, and Cx. tarsalis are regarded as the most competent vectors in North America, where their abundance in urban and peri-urban environments coincides with epidemic peaks [18]. In Latin America, Cx. quinquefasciatus and related species are widely distributed and ecologically suited to sustain WNV transmission [16]. Studies in Mexico and Central America suggest that viremic migratory birds, together with the widespread presence of Culex mosquitoes, facilitate regional introduction and persistence of the virus [7]. The detection of Culex mosquitoes in international airports further highlights the potential for human-mediated dispersal [19].
From a genetic perspective, WNV comprises multiple lineages, but only lineages 1 and 2 are considered of major global relevance [20]. Lineage 1 is the most widespread, associated with outbreaks across Africa, Europe, the Middle East, and the Americas. Lineage 2, once restricted to sub-Saharan Africa, has emerged as an important cause of human disease in Europe [1,21]. Experimental and field evidence suggests that lineage-specific differences in virulence, vector competence, and ecological adaptation may partially explain the observed geographic heterogeneity in human disease incidence.
3. Current Evidence in Latin America and the Caribbean
3.1. Circulation in Animals and Vectors
Since the early 2000s, circulation of WNV in Latin America and the Caribbean has been primarily documented through findings in animals and vectors. The first confirmed isolation in South America occurred in Argentina in 2006 from horses with encephalitis, where viral isolation and molecular detection provided definitive evidence of active infection, marking the official introduction of the virus into the region [22]. Since then, multiple serological studies, most commonly based on the detection of neutralizing antibodies, have identified evidence of WNV exposure in resident birds, horses, non-human primates, and reptiles, often with confirmation by plaque reduction neutralization tests (PRNTs) to address flavivirus cross-reactivity, as well as viral isolations from mosquitoes, confirming active transmission in diverse ecosystems [23].
In Mexico, the evidence is broad and consistent, serological surveys have detected antibodies against WNV have been identified in wild birds, reptiles, and mammals, and the virus has been isolated from Culex quinquefasciatus and other species, suggesting established enzootic circulation [24,25]. In the Caribbean, monitoring studies conducted after the first introductions from North America confirmed local transmission: resident birds from Puerto Rico and Cuba showed neutralizing antibodies to WNV confirmed by PRNT, ruling out infections solely in migratory species [26].
In Central America, evidence is also strong. In Guatemala (2005–2008), WNV was isolated from Culex quinquefasciatus and annual circulation was detected through entomological studies, although without clinical evidence in humans [27]. In El Salvador, between 2001 and 2003, an epizootic caused the death of 203 horses; subsequent analyses demonstrated that 25% of surviving stablemates had antibodies against WNV, with 10 confirmed by PRNT, thereby confirming the expansion of the virus into Central America [28]. In Belize, an early 2003 report documented a clinically infected horse with laboratory confirmation, constituting the first evidence of circulation in the country [29]. In Costa Rica, serological studies using WNV-specific assays, with confirmatory neutralization testing, have shown co-circulation of flaviviruses in birds, horses, and wild primates, with specific detection of neutralizing antibodies against WNV in some animals, though no human cases have been reported to date [30,31].
In South America, beyond Argentina, circulation has been documented in several countries. In Bolivia, a 2011 study showed that 59.4% of 160 horses had antibodies using competitive ELISA, one-third of which were confirmed by PRNT, with no evidence of recent infections [32]. In Paraguay (2016–2018), serological surveys in resident birds identified neutralizing antibodies against WNV in a barred antshrike (Thamnophilus doliatus), representing the first serological evidence of the virus in the country [33]. In Colombia and Venezuela, multiple studies have demonstrated circulation in birds, horses, and other animals, including antibodies in captive flamingos and equines, largely through serological detection confirmed by neutralization assays, although no confirmed human cases have been reported [34,35,36]. In Ecuador, Culex quinquefasciatus populations from the Galápagos Islands have been shown experimentally competent to transmit WNV, and serological studies in horses have so far yielded negative results, reflecting potential but not yet established circulation [16] (Figure 1).
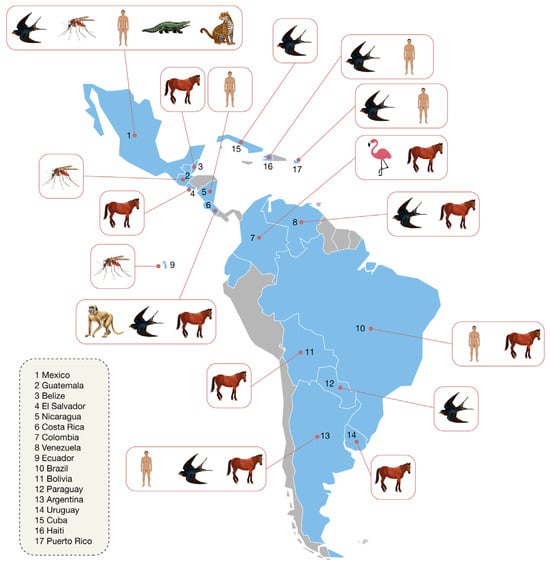
Figure 1.
Summary of WNV evidence distribution in Latin America and the Caribbean (2001–2024). This figure summarizes evidence of WNV across Latin America and the Caribbean from 2001 to 2024. Icons denote the type of finding by country (human, horse, bird, mosquito, reptile, mammal). For vectors, the “mosquito” icon refers primarily to Culex quinquefasciatus, with virus isolations in Guatemala (2005–2008) and demonstrated vector competence for Galápagos (Ecuador) strains. In Mexico, beyond isolation in Cx. quinquefasciatus, neutralizing antibodies have been documented in resident birds, reptiles (Crocodylus acutus and C. acutus × C. moreletii hybrids), and mammals (e.g., Panthera onca or jaguar), Canis latrans or coyote), indicating heterogeneous enzootic circulation. Among equines, evidence includes the El Salvador epizootic (2001–2003), a single horse case in Belize (2003), seropositivity in Bolivia, and in Argentina both seropositivity and virus isolation in 2006; Colombia and Venezuela also report equine seropositivity. In birds, WNV has been confirmed in Paraguay (Thamnophilus doliatus), with neutralizing antibodies in Cuba (resident birds) and seropositivity reported in Colombia and Venezuela. Regarding humans, confirmed cases have been reported in Brazil (>100; 2014–2024), Mexico (~13), Argentina (2006–2007), Puerto Rico (2007), Nicaragua (anti-prM serologic evidence in children with infections predating 2007–2009), and Haiti (two historical cases). The figure synthesizes evidence published in peer-reviewed articles and technical reports for the indicated period.
3.2. Human Cases: Confirmed but Limited
Unlike North America and Europe, Latin America has not recorded large-scale human epidemics of WNV. Confirmed cases remain sporadic, totaling only a few dozen across the region [23,24].
In Brazil, a recent multicenter study reported 110 confirmed cases between 2014 and 2024 in 13 states, with clinical presentations including febrile and neuroinvasive forms, and four fatalities [37]. Cases were identified using serological assays (IgM/IgG) complemented by molecular detection (RT-PCR) in acute presentations, and the first internationally confirmed case occurred in 2015 in Piauí, associated with acute flaccid paralysis [38]. In Mexico, despite extensive circulation in animals, only 13 human cases were confirmed between 2002 and 2023, confirmation relying on RT-PCR or viral isolation in early cases and serology with neutralization testing in later investigations, with the first viral isolation in 2005 and a fatal case in 2009 [24,25,39,40].
In Argentina, sporadic human cases were reported in 2006–2007 across at least five northeastern and central provinces, diagnosed primarily through serological assays and, in selected cases, molecular confirmation, coinciding with the first equine isolation of the virus in South America [41]. In Nicaragua, a pediatric cohort study identified anti-prM antibodies to WNV in three children, with infections predating 2007–2009, providing the first human evidence of WNV in Central America [42].
In the Caribbean, Puerto Rico documented human transmission in 2007 through laboratory detection in a blood donor and a symptomatic patient, using serological screening with confirmatory testing, within a context of active surveillance that also included mosquitoes and raptors [43]. In Haiti, a population-based study detected IgG antibodies against WNV by serological testing in 7 of 673 participants (≈1%), although cross-reactivity with dengue was suggested; historically, only two human WNV cases have been confirmed in the country [44] (Figure 1).
Available data suggest that the clinical spectrum of human WNV infection in Latin America mirrors that observed in other regions, ranging from asymptomatic infection and mild febrile illness to neuroinvasive disease, including meningitis, encephalitis, and acute flaccid paralysis [45]. However, systematic characterization of disease severity in the region remains limited. In Brazil, where the largest number of confirmed cases has been reported, both febrile and neuroinvasive presentations have been documented, with a small number of fatalities. In other countries, human cases have been identified primarily through targeted investigations, blood donor screening, or retrospective serological studies, rather than through routine clinical surveillance [46,47,48]. The apparent scarcity of reported neuroinvasive disease likely could reflect a scenario of limited case detection and reporting, rather than true absence of severe infection in Latin America, underscoring the need for improved clinical awareness and surveillance integration in the region.
4. Public Health Implications
The panorama of WNV in Latin America is paradoxical: while serological and entomological evidence confirms widespread circulation in wildlife and vectors, confirmed human cases remain rare [23]. This discrepancy likely reflects substantial underreporting and diagnostic limitations, as mild febrile cases may be misclassified as dengue, Zika, or Chikungunya [49,50]. Although notification of WNV is officially mandatory across nearly all countries in the region (Table 1), human surveillance is predominantly passive and restricted to neurological or encephalitic syndromes, which hampers timely detection of sporadic or mild cases.

Table 1.
Surveillance systems of WNV in Latin America countries.
In addition to underdiagnosis and surveillance limitations, several complementary hypotheses may also contribute to the relatively low number of reported human West Nile virus cases in Latin America. These include partial cross-protective immunity resulting from high background exposure to other endemic flaviviruses, such as dengue, Zika, and St. Louis encephalitis virus, which may modulate susceptibility to symptomatic or neuroinvasive WNV infection [69]. Differences in vector competence, feeding behavior, and host preference among Culex species circulating in the region may further influence the likelihood of human exposure and transmission [70]. Moreover, lineage-specific viral characteristics and host–virus interactions can shape pathogenicity and neuroinvasive potential, contributing to heterogeneity in clinical presentation and detection [71,72]. Importantly, these mechanisms are not mutually exclusive and likely interact with structural diagnostic and surveillance gaps, collectively shaping the observed epidemiological pattern.
From a public health perspective, the consequences are significant. Climate change acts as a critical driver by expanding the habitats of Culex mosquitoes, shortening the extrinsic incubation period of the virus, and altering bird migration patterns [73]. Experiences in Europe illustrate these risks: in the Netherlands and Germany, climatic and land-use changes have been linked to outbreaks of WNV and Usutu virus (USUV) [74,75]. Such findings suggest that viral expansion into new areas of Latin America is both possible and likely underestimated.
Another concern is transfusion-related transmission, already documented in North America and Europe, which led to the implementation of nucleic acid testing (NAT) in blood donations in affected regions [76,77]. Similar measures may become essential in Latin America, particularly if viral activity in animals or vectors is detected near blood collection centers.
Finally, European experience highlights that integrated surveillance under a One Health framework—combining entomological monitoring, veterinary surveillance, and human case detections—is the most effective approach to anticipate outbreaks. Italy’s regional programs, initiated in 2008 and later scaled nationally, successfully correlated mosquito, bird, and human data to guide real-time control measures. Comparable models in the Netherlands, involving citizen science, zoos, and migratory bird sampling, proved crucial for early detection and prediction [78].
In Latin America, however, integrated approaches remain incipient. The documented circulation in fauna, favorable environmental conditions, and fragmented surveillance systems suggest that human outbreak risk is underestimated. Strengthening intersectoral collaboration across veterinary, public health, and conservation sectors is fundamental to ensure early detection of new transmission areas, safeguard transfusion safety, and reduce the probability of large-scale epidemics in the region [49].
5. Research and Surveillance Priorities in a One Health Framework
Building on the implications above, we propose a shift from fragmented activities to an integrated One Health preparedness agenda that couples technical rigor with community engagement and intersectoral governance.
5.1. Population-Based Seroepidemiological Studies
Implement standardized, population-based serosurveys—stratified by eco-regions and urban/rural gradients—to estimate true infection prevalence, including asymptomatic and mild infections frequently misclassified as other arboviruses [79]. Incorporate community-based sampling and participatory designs to expand coverage in hard-to-reach settings and reduce selection bias.
5.2. Strengthening Diagnostic Capacity and Clinical Awareness
Among the proposed research and surveillance priorities, strengthening diagnostic capacity across public health and animal health sectors represents the highest-yield and most immediately actionable intervention in resource-limited settings. Improved diagnostic availability underpins early case detection, accurate burden estimation, timely outbreak response, and the effectiveness of all subsequent surveillance, modeling, and preparedness efforts [80,81]. Therefore, investments in diagnostic capacity should precede or accompany all other One Health interventions aimed at mitigating the risk of WNV emergence. Overcoming current molecular and serological capacity gaps, as well as the cross-reactivity that complicates case attribution in flavivirus-endemic areas, is essential. Key priorities include expanding access to RT-PCR and genomic sequencing, validating highly specific serological platforms (e.g., PRNT), and investing in decentralized hub laboratories linked to national reference centers to accelerate confirmation and reporting [12].
Concurrently, clinical awareness should be strengthened to raise suspicion for WNV in both febrile and neurological syndromes and to improve differentiation from dengue, Zika, and chikungunya. This requires targeted training of interdisciplinary teams—including clinicians, neurologists, microbiologists, veterinarians, entomologists, and public health officers—alongside community leaders, focusing on case recognition, biosafety, sampling, and timely notification [1].
5.3. Predictive Climate and Ecological Modeling
Develop region-specific predictive models that integrate temperature, precipitation, land-use change, Culex dynamics, and bird migration/phenology to anticipate hotspots and guide seasonal vector control, transfusion safety policies, and resource allocation. Lessons from the Mediterranean Basin show that climate anomalies (e.g., warmer springs, earlier seasonal peaks) can precede major outbreaks. Incorporate local and ancestral knowledge (e.g., phenological cues, shifts in breeding sites) as qualitative covariates to improve model performance and operational relevance [82].
5.4. Toward Integrated One Health and Community Surveillance
As demonstrated in Europe and the Mediterranean, surveillance is most effective when human, animal, and vector data are integrated. In Latin America, establish multisectoral and transnational frameworks to enable early viral detection, enhance transfusion safety, and generate actionable indicators for decision-makers. Define a One Health governance architecture (roles, decision flows, accountability) with sustainable financing (contingency funds and climate-linked lines) and deploy regional dashboards with near-real-time data and cross-border sharing agreements [7,75].
In parallel, co-design surveillance with community organizations and Indigenous peoples (“salud propia”), leveraging citizen science to report sick/dead birds, map Culex breeding sites, and document environmental signals, and provide multilingual materials—including Spanish and Portuguese, as well as relevant Indigenous languages in settings where Indigenous and rural communities play a central role in environmental monitoring and early detection—to increase timely reporting, acceptability, and local ownership of surveillance activities [41].
6. Conclusions
WNV has undergone a marked transition, evolving from a pathogen once regarded as of limited relevance to humans into a global zoonotic threat, characterized by a wide geographic distribution and a remarkable capacity to adapt to changing ecological and climatic conditions. While its epidemiological impact has been clearly documented in regions such as North America and Europe, the picture in Latin America remains less well defined. Here, consistent evidence of viral circulation in vectors and animal reservoirs has been reported across several countries, yet confirmed human cases remain unusually scarce. This discrepancy likely reflects regional health system limitations, including underdiagnosis, constrained diagnostic capacity, and significant gaps in surveillance.
The convergence of climate change, ecological modifications, and increasing human–wildlife interactions amplifies the risk of WNV emergence and expansion into new territories of Latin America and the Caribbean. Lessons from Europe underscore the importance of early detection, transfusion safety, and the integration of entomological, veterinary, and human data into surveillance programs. Strengthening regional preparedness through a One Health framework is essential to confront WNV, requiring investments in diagnostic infrastructure, the implementation of standardized seroepidemiological surveys, and the development of predictive models tailored to local ecological contexts.
Author Contributions
Conceptualization, J.S.I.-C.; methodology, J.S.I.-C., E.O.-P.; software, J.S.I.-C., J.C.G., J.S.S.-T., H.A.N.-C., J.J.M.P., C.V.N., A.L.-C., M.A.-I.; validation, J.S.I.-C., E.O.-P.; formal analysis, J.S.I.-C.; investigation, J.S.I.-C., J.C.G., J.S.S.-T., H.A.N.-C., J.J.M.P., C.V.N., A.L.-C., M.A.-I.; resources, J.S.I.-C., J.C.G., J.S.S.-T., H.A.N.-C., J.J.M.P., C.V.N., A.L.-C., M.A.-I.; data curation, J.S.I.-C., J.S.S.-T., H.A.N.-C.; writing—original draft preparation, J.S.I.-C., J.S.S.-T., H.A.N.-C., J.J.M.P., C.V.N., A.L.-C., M.A.-I.; writing—review and editing, J.S.I.-C., J.C.G., E.O.-P.; visualization, J.S.I.-C., A.L.-C.; supervision, J.S.I.-C., E.O.-P.; project administration, J.S.I.-C.; funding acquisition, J.S.I.-C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
This study did not generate new datasets. All information analyzed is available in the published literature referenced in this article.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| Abbreviation | Definition |
| ALFV | Alfuy virus |
| CC BY | Creative Commons Attribution |
| CDC | Centers for Disease Control and Prevention |
| CRG | Cancer Research Group |
| Cx. | Culex |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| GIS | Grupo de Investigación en Salud |
| GIMI | Interinstitutional Internal Medicine Group |
| ICA | Instituto Colombiano Agropecuario |
| IgG | Immunoglobulin G |
| INS | Instituto Nacional de Salud |
| MoH | Ministry of Health |
| MVEV | Murray Valley encephalitis virus |
| NAT | Nucleic Acid Testing |
| OIE | World Organisation for Animal Health |
| PAHO | Pan American Health Organization |
| PRNT | Plaque Reduction Neutralization Test |
| RT-PCR | Reverse Transcription Polymerase Chain Reaction |
| SENASA | Servicio Nacional de Sanidad y Calidad Agroalimentaria |
| SINAN | Sistema de Informação de Agravos de Notificação |
| SINAVE | Sistema Nacional de Vigilancia Epidemiológica |
| SIVIGILA | Sistema Nacional de Vigilancia en Salud Pública |
| SLEV | St. Louis encephalitis virus |
| USUV | Usutu virus |
| WHO | World Health Organization |
| WNV | West Nile virus |
References
- Habarugira, G.; Suen, W.W.; Hobson-Peters, J.; Hall, R.A.; Bielefeldt-Ohmann, H. West Nile Virus: An Update on Pathobiology, Epidemiology, Diagnostics, Control and “One Health” Implications. Pathogens 2020, 9, 589. [Google Scholar] [CrossRef] [PubMed]
- Popović, N.; Milošević, B.; Urošević, A.; Poluga, J.; Lavadinović, L.; Nedelijković, J.; Jevtović, D.; Dulović, O. Outbreak of West Nile Virus Infection among Humans in Serbia, August to October 2012. Eurosurveillance 2013, 18, 20613. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.R.; Ismail, A.A.; Thergarajan, G.; Raju, C.S.; Yam, H.C.; Rishya, M.; Sekaran, S.D. Serological Cross-Reactivity among Common Flaviviruses. Front. Cell. Infect. Microbiol. 2022, 12, 975398. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Condoy, J.S.; Vásconez-Gonzáles, J.; Morales-Lapo, E.; Tello-De-la-Torre, A.; Naranjo-Lara, P.; Fernández, R.; Hidalgo, M.R.; Escobar, A.; Yépez, V.H.; Díaz, A.M.; et al. Beyond the Acute Phase: A Comprehensive Literature Review of Long-Term Sequelae Resulting from Infectious Diseases. Front. Cell. Infect. Microbiol. 2024, 14, 1293782. [Google Scholar] [CrossRef]
- Curren, E.J.; Venkat, H.; Sunenshine, R.; Fitzpatrick, K.; Kosoy, O.; Krow-Lucal, E.; Zabel, K.; Adams, L.; Kretschmer, M.; Fischer, M.; et al. Assessment of Immunoglobulin M Enzyme-Linked Immunosorbent Assay Ratios to Identify West Nile Virus and St. Louis Encephalitis Virus Infections During Concurrent Outbreaks of West Nile Virus and St. Louis Encephalitis Virus Diseases, Arizona 2015. Vector Borne Zoonotic Dis. 2020, 20, 619–623. [Google Scholar] [CrossRef]
- Zuddas, C.; Piras, S.; Cappai, S.; Loi, F.; Murgia, G.; Puggioni, G.; Savini, G.; Monaco, F.; Polci, A.; Valleriani, F.; et al. First Detection of West Nile Virus by Nasopharyngeal Swab, Followed by Phylogenetic Analysis. Pathogens 2024, 13, 1023. [Google Scholar] [CrossRef]
- Bruno, L.; Nappo, M.A.; Frontoso, R.; Perrotta, M.G.; Di Lecce, R.; Guarnieri, C.; Ferrari, L.; Corradi, A. West Nile Virus (WNV): One-Health and Eco-Health Global Risks. Vet. Sci. 2025, 12, 288. [Google Scholar] [CrossRef]
- Murray, K.O.; Mertens, E.; Desprès, P. West Nile Virus and Its Emergence in the United States of America. Vet. Res. 2010, 41, 67. [Google Scholar] [CrossRef]
- Petersen, L.R. Epidemiology of West Nile Virus in the United States: Implications for Arbovirology and Public Health. J. Med. Entomol. 2019, 56, 1456–1462. [Google Scholar] [CrossRef]
- Camp, J.V.; Nowotny, N. The Knowns and Unknowns of West Nile Virus in Europe: What Did We Learn from the 2018 Outbreak? Expert Rev. Anti-Infect. Ther. 2020, 18, 145–154. [Google Scholar] [CrossRef]
- Erazo, D.; Grant, L.; Ghisbain, G.; Marini, G.; Colón-González, F.J.; Wint, W.; Rizzoli, A.; Van Bortel, W.; Vogels, C.B.F.; Grubaugh, N.D.; et al. Contribution of Climate Change to the Spatial Expansion of West Nile Virus in Europe. Nat. Commun. 2024, 15, 1196. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Khatib, M.N.; Ballal, S.; Kaur, M.; Nathiya, D.; Sharma, S.; Prasad, G.V.S.; Sinha, A.; Gaidhane, A.M.; Mohapatra, P.; et al. West Nile Virus in a Changing Climate: Epidemiology, Pathology, Advances in Diagnosis and Treatment, Vaccine Designing and Control Strategies, Emerging Public Health Challenges—A Comprehensive Review. Emerg. Microbes Infect. 2025, 14, 2437244. [Google Scholar] [CrossRef] [PubMed]
- Balatsos, G.; Beleri, S.; Tegos, N.; Bisia, M.; Karras, V.; Zavitsanou, E.; Papachristos, D.P.; Papadopoulos, N.T.; Michaelakis, A.; Patsoula, E. Overwintering West Nile Virus in Active Culex Pipiens Mosquito Populations in Greece. Parasit. Vectors 2024, 17, 286. [Google Scholar] [CrossRef] [PubMed]
- Mor, Z.; Omari, H.; Indenbaum, V.; Kirstein, O.D.; Shatach Catabi, O.; Reicher, S.; Lustig, Y.; Davidovich-Cohen, M.; Kaliner, E.; Sheffer, R.; et al. Early Rise of West Nile Fever in Israel, June 2024. Eurosurveillance 2024, 29, 2400457. [Google Scholar] [CrossRef]
- Costa, E.d.F.; Streng, K.; de Souza Santos, M.A.; Counotte, M.J. The Effect of Temperature on the Boundary Conditions of West Nile Virus Circulation in Europe. PLoS Neglected Trop. Dis. 2024, 18, e0012162. [Google Scholar] [CrossRef]
- Eastwood, G.; Kramer, L.D.; Goodman, S.J.; Cunningham, A.A. West Nile Virus Vector Competency of Culex Quinquefasciatus Mosquitoes in the Galápagos Islands. Am. J. Trop. Med. Hyg. 2011, 85, 426–433. [Google Scholar] [CrossRef]
- García-Bocanegra, I.; Jaén-Téllez, J.A.; Napp, S.; Arenas-Montes, A.; Fernández-Morente, M.; Fernández-Molera, V.; Arenas, A. West Nile Fever Outbreak in Horses and Humans, Spain, 2010. Emerg. Infect. Dis. 2011, 17, 2397–2399. [Google Scholar] [CrossRef]
- Diaz-Badillo, A.; Bolling, B.G.; Perez-Ramirez, G.; Moore, C.G.; Martinez-Munoz, J.P.; Padilla-Viveros, A.A.; Camacho-Nuez, M.; Diaz-Perez, A.; Beaty, B.J.; de Lourdes Munoz, M. The Distribution of Potential West Nile Virus Vectors, Culex Pipiens Pipiens and Culex Pipiens Quinquefasciatus (Diptera: Culicidae), in Mexico City. Parasit. Vectors 2011, 4, 70. [Google Scholar] [CrossRef]
- Ibáñez-Justicia, A.; Smitz, N.; den Hartog, W.; van de Vossenberg, B.; De Wolf, K.; Deblauwe, I.; Van Bortel, W.; Jacobs, F.; Vaux, A.G.C.; Medlock, J.M.; et al. Detection of Exotic Mosquito Species (Diptera: Culicidae) at International Airports in Europe. Int. J. Environ. Res. Public Health 2020, 17, 3450. [Google Scholar] [CrossRef]
- Lara, M.; Casimiro-Soriguer, C.S.; Pedrosa-Corral, I.; Gómez-Camarasa, C.; Lorusso, N.; Navarro-Marí, J.M.; Dopazo, J.; Perez-Florido, J.; Sanbonmatsu-Gámez, S. First Autochthonous Transmission of West Nile Virus (WNV) Lineage 2 to Humans in Spain. One Health 2025, 20, 101036. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Triana, L.M.; Jeffries, C.L.; Mansfield, K.L.; Carnell, G.; Fooks, A.R.; Johnson, N. Emergence of West Nile Virus Lineage 2 in Europe: A Review on the Introduction and Spread of a Mosquito-Borne Disease. Front. Public Health 2014, 2, 271. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.A.; Barrandeguy, M.; Fabbri, C.; Garcia, J.B.; Vissani, A.; Trono, K.; Gutierrez, G.; Pigretti, S.; Menchaca, H.; Garrido, N.; et al. West Nile Virus Isolation from Equines in Argentina, 2006. Emerg. Infect. Dis. 2006, 12, 1559–1561. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, C.; Chiaravalloti-Neto, F. Why Are There No Human West Nile Virus Outbreaks in South America? Lancet Reg. Health Am. 2022, 12, 100276. [Google Scholar] [CrossRef]
- Elizondo-Quiroga, D.; Elizondo-Quiroga, A. West Nile Virus and Its Theories, a Big Puzzle in Mexico and Latin America. J. Glob. Infect. Dis. 2013, 5, 168–175. [Google Scholar] [CrossRef]
- Correa-Morales, F.; González-Acosta, C.; Ibarra-Ojeda, D.; Moreno-García, M. West Nile Virus in Mexico: Why Vectors Matter for Explaining the Current Absence of Epidemics. Acta Trop. 2024, 249, 107065. [Google Scholar] [CrossRef]
- Dupuis, A.P.; Marra, P.P.; Reitsma, R.; Jones, M.J.; Louie, K.L.; Kramer, L.D. Serologic Evidence for West Nile Virus Transmission in Puerto Rico and Cuba. Am. J. Trop. Med. Hyg. 2005, 73, 474–476. [Google Scholar] [CrossRef]
- Morales-Betoulle, M.E.; Komar, N.; Panella, N.A.; Alvarez, D.; López, M.R.; Betoulle, J.-L.; Sosa, S.M.; Müller, M.L.; Kilpatrick, A.M.; Lanciotti, R.S.; et al. West Nile Virus Ecology in a Tropical Ecosystem in Guatemala. Am. J. Trop. Med. Hyg. 2013, 88, 116–126. [Google Scholar] [CrossRef][Green Version]
- Cruz, L.; Cardenas, V.M.; Abarca, M.; Rodriguez, T.; Reyna, R.F.; Serpas, M.V.; Fontaine, R.E.; Beasley, D.W.C.; Da Rosa, A.P.A.T.; Weaver, S.C.; et al. Short Report: Serological Evidence of West Nile Virus Activity in El Salvador. Am. J. Trop. Med. Hyg. 2005, 72, 612–615. [Google Scholar] [CrossRef]
- Komar, N.; Clark, G.G. West Nile Virus Activity in Latin America and the Caribbean. Rev. Panam. Salud Publica 2006, 19, 112–117. [Google Scholar] [CrossRef]
- Hobson-Peters, J.; Arévalo, C.; Cheah, W.Y.; Blitvich, B.J.; Tan, C.S.E.; Sandis, A.; Araya, L.N.; Hernández, J.L.; Toye, P.; Hall, R.A. Detection of Antibodies to West Nile Virus in Horses, Costa Rica. 2004. Available online: https://www.liebertpub.com/doi/10.1089/vbz.2010.0198 (accessed on 30 September 2025).
- Piche-Ovares, M.; Romero-Vega, M.; Vargas-González, D.; Murillo, D.F.B.; Soto-Garita, C.; Francisco-Llamas, J.; Alfaro-Alarcón, A.; Jiménez, C.; Corrales-Aguilar, E. Serosurvey in Two Dengue Hyperendemic Areas of Costa Rica Evidence Active Circulation of WNV and SLEV in Peri-Domestic and Domestic Animals and in Humans. Pathogens 2022, 12, 7. [Google Scholar] [CrossRef]
- Mazzei, M.; Savini, G.; Di Gennaro, A.; Macchioni, F.; Prati, M.C.; Guzmàn, L.R.; Tolari, F. West Nile Seroprevalence Study in Bolivian Horses, 2011. Vector Borne Zoonotic Dis. 2013, 13, 894–896. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, F.; Bernal, C.; Rojas, A.; Diaz, A.; Spinsanti, L.; Páez, M.; Guillén, Y.; Lesterhuis, A.; Yanosky, A.; Contigiani, M.; et al. Detection of Neutralizing Antibodies against Flaviviruses in Free-Ranging Birds, Paraguay (2016–2018). Trans. R. Soc. Trop. Med. Hyg. 2023, 117, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Mattar, S.; Edwards, E.; Laguado, J.; González, M.; Alvarez, J.; Komar, N. West Nile Virus Antibodies in Colombian Horses. Emerg. Infect. Dis. 2005, 11, 1497–1498. [Google Scholar] [CrossRef]
- Bosch, I.; Herrera, F.; Navarro, J.-C.; Lentino, M.; Dupuis, A.; Maffei, J.; Jones, M.; Fernández, E.; Perez, N.; Pérez-Emán, J.; et al. West Nile Virus, Venezuela. Emerg. Infect. Dis. 2007, 13, 651–653. [Google Scholar] [CrossRef]
- López, R.H.; Soto, S.U.; Gallego-Gómez, J.C. Evolutionary Relationships of West Nile Virus Detected in Mosquitoes from a Migratory Bird Zone of Colombian Caribbean. Virol. J. 2015, 12, 80. [Google Scholar] [CrossRef]
- de Lima, S.T.S.; Claro, I.M.; Hua, X.; de Jesus, R.; Serres, K.; Simões Mello, L.M.; Forato, J.; Moreira, F.R.R.; Kato, R.B.; Guimarães, G.N.; et al. Active West Nile Virus Transmission in Brazil: An Epidemiological Study. Lancet Reg. Health Am. 2025, 51, 101229. [Google Scholar] [CrossRef]
- Vieira, M.A.C.S.; Romano, A.P.M.; Borba, A.S.; Silva, E.V.P.; Chiang, J.O.; Eulálio, K.D.; Azevedo, R.S.S.; Rodrigues, S.G.; Almeida-Neto, W.S.; Vasconcelos, P.F.C. West Nile Virus Encephalitis: The First Human Case Recorded in Brazil. Am. J. Trop. Med. Hyg. 2015, 93, 377–379. [Google Scholar] [CrossRef]
- Rios-Ibarra, C.; Blitvich, B.J.; Farfan-Ale, J.; Ramos-Jimenez, J.; Muro-Escobedo, S.; Martínez-Rodriguez, H.R.; Torres-López, E.; Rivas-Estilla, A.M. Fatal Human Case of West Nile Disease, Mexico, 2009. Emerg. Infect. Dis. 2010, 16, 741–743. [Google Scholar] [CrossRef]
- Elizondo-Quiroga, D.; Davis, C.T.; Fernandez-Salas, I.; Escobar-Lopez, R.; Olmos, D.V.; Gastalum, L.C.S.; Acosta, M.A.; Elizondo-Quiroga, A.; Gonzalez-Rojas, J.I.; Cordero, J.F.C.; et al. West Nile Virus Isolation in Human and Mosquitoes, Mexico. Emerg. Infect. Dis. 2005, 11, 1449–1452. [Google Scholar] [CrossRef] [PubMed]
- Goenaga, S.; Goenaga, J.; Boaglio, E.R.; Enria, D.A.; Levis, S.d.C. Superinfection Exclusion Studies Using West Nile Virus and Culex Flavivirus Strains from Argentina. Mem. Inst. Oswaldo Cruz 2020, 115, e200012. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.-Y.; Tseng, A.C.; Chen, G.-H.; Hsieh, S.-C.; Balmaseda, A.; Nerurkar, V.R.; Harris, E.; Wang, W.-K. Identification of West Nile Virus Infection by Anti-Premembrane Antibodies in Nicaraguan Children Prior to 2007–2009. Microbiol. Spectr. 2025, 13, e0004725. [Google Scholar] [CrossRef] [PubMed]
- Center for Disease Control and Prevention. Detection of West Nile Virus in Blood Donations—Puerto Rico, 2007. Morb. Mortal. Wkly. Rep. 2008, 57, 577–580. [Google Scholar]
- Weppelmann, T.A.; Burne, A.; von Fricken, M.E.; Elbadry, M.A.; De Rochars, M.B.; Boncy, J.; Okech, B.A. A Tale of Two Flaviviruses: A Seroepidemiological Study of Dengue Virus and West Nile Virus Transmission in the Ouest and Sud-Est Departments of Haiti. Am. J. Trop. Med. Hyg. 2017, 96, 135–140. [Google Scholar] [CrossRef]
- Sejvar, J.J. West Nile Virus Infection. Microbiol. Spectr. 2016, 4, 175–199. [Google Scholar] [CrossRef]
- Najafi, S.; Jojani, M.; Najafi, K.; Costanzo, V.; Vicidomini, C.; Roviello, G.N. West Nile Virus: Epidemiology, Surveillance, and Prophylaxis with a Comparative Insight from Italy and Iran. Vaccines 2026, 14, 57. [Google Scholar] [CrossRef]
- Gobbo, F.; Chiarello, G.; Sgubin, S.; Toniolo, F.; Gradoni, F.; Danca, L.I.; Carlin, S.; Capello, K.; De Conti, G.; Bortolami, A.; et al. Integrated One Health Surveillance of West Nile Virus and Usutu Virus in the Veneto Region, Northeastern Italy, from 2022 to 2023. Pathogens 2025, 14, 227. [Google Scholar] [CrossRef]
- Grard, G.; Franke, F.; Laperche, S.; Cochet, A.; Paty, M.-C.; Gonzalez, G.; Chaubenit, F.; Migné, C.V.; Fontaine, A.; Le Cam, S.; et al. Blood Donation Screening and West Nile Virus Surveillance Strategy in France. JAMA Netw. Open 2025, 8, e2524494. [Google Scholar] [CrossRef]
- Lessa, C.L.S.; Fiuza, B.S.D.; Hodel, K.V.S.; Minafra, C.; de Souza Gonçalves, M.; Machado, B.A.S. Understanding Dengue Underreporting: An Analysis of the Impacts for the World, Latin America and Brazil. Sci. World J. 2025, 2025, 9993911. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Izquierdo-Condoy, J.S.; Vásconez-González, J. Urgent Response Needed: Addressing the Dengue Crisis in the Andean and Southern Cone Latin American Regions. Am. J. Trop. Med. Hyg. 2024, 111, 714–718. [Google Scholar] [CrossRef]
- Sistema Nacional de Vigilancia de la Salud; Ministerio de Salud Argentina. Listado de Eventos de Notificación Obligatoria Actualización 2022. Buenos Aires, Argentina; 2022. Available online: https://servicios.infoleg.gob.ar/infolegInternet/anexos/195000-199999/195093/res2827-1.pdf (accessed on 4 February 2026).
- Sistema Nacional de Información en Salud—Vigilancia Epidemiológica. In Boletín de Vigilancia Epidemiológica; La Paz, Bolivia; 2024. Available online: https://snis.minsalud.gob.bo/publicaciones (accessed on 4 February 2026).
- Secretaria da Saúde; Governo do Estado do Ceará. Nota Técnica Febre Do Nilo Ocidental. Estado do Ceará, Brasil; 2019. Available online: https://www.saude.ce.gov.br/wp-content/uploads/sites/9/2018/06/nota_febre_nilo_ocidental_26_07_2019.pdf (accessed on 4 February 2026).
- División Planificación Sanitaria; Subsecretaria de Salud Pública; Ministerio de Salud. Sistema De Vigilancia De Encefalitis Del Nilo Occidental (West Nile). Chile; 2006. Available online: https://www.ispch.gob.cl/wp-content/uploads/2022/05/Circular-B51-17-2006.pdf (accessed on 4 February 2026).
- Instituto Nacional de Salud (Colombia); Dirección de Vigilancia y Análisis del Riesgo en Salud Pública. Encefalitis Equina En Colombia. Colombia; 2025. Available online: https://www.ins.gov.co/buscador-eventos/BoletinEpidemiologico/2025_Boletin_epidemiologico_semana_8.pdf (accessed on 4 February 2026).
- Piche Ovares, M.M. Detección del virus del oeste del Nilo en dos localidades con antecedentes de enfermedad neurológica en equinos: Determinación molecular en aves y mosquitos y serología en humanos, aves y equinos. Kerwa Repos. 2019, 8, 45–57. [Google Scholar]
- Pupo-Antunez, M.; Vázquez, Y.; Andonova, M.; Vázquez, S.; Morier, L. Field Study: Searching for West Nile Virus in Cuba. J. Emerg. Dis. Virol. 2018, 4, 1–8. [Google Scholar] [CrossRef]
- Amador, M.; Díaz, A.; Smith, J. El periodismo científico en América Latina: ¿se informa correctamente a la población? Rev. Panam. Salud Publica 2008, 23, 426–427. [Google Scholar] [CrossRef]
- Coello Peralta, R.D.; González González, M.; Martínez Cepeda, G.E. Virus del Nilo Occidental en Ecuador. Rev. MVZ Córdoba 2018, 24, 7151–7156. [Google Scholar] [CrossRef]
- Escalante-Hernández, M.N.; Quintanilla-Moreno, G.A.; López-Salazar, C.D.; Romero-Pérez, L.E.; Avendaño-Romero, N.O. Evidencia serológica del Virus del Nilo Occidental en equinos (Equus caballus), en el área natural protegida laguna El Jocotal, departamento de San Miguel, El Salvador. Rev. Agrociencia 2018, 1, 44–51. [Google Scholar]
- Jaramillo, M.; Peña, J.; Berrocal, L.; Komar, N.; González Tous, M.; Ponce, C.; Ariza, K.; Mattar, S. Vigilancia centinela para el virus del oeste del Nilo en culícidos y aves domésticas en el departamento de Córdoba. Rev. MVZ Córdoba 2005, 10, 633–638. [Google Scholar] [CrossRef]
- Salud Digital. Fundación Carlos Slim Honduras fortalece el uso de plataforma del sistema de vigilancia de la salud. Salud Digit. 2024, 15, 89–94. [Google Scholar]
- Secretaría de Salud; Subsecretaría de Prevención y Promoción de la Salud; Dirección General de Epidemiología. Sistema De Vigilancia Epidemiológica Convencional, Manual de Procedimientos Estandarizados Para La Vigilancia Epidemiológica Convencional. 2021. Available online: https://www.minsa.gob.ni/index.php/repositorio/guia-vigilancia-2020 (accessed on 4 February 2026).
- Ministerio de Salud Nicaragua. Guía Para La Vigilancia Epidemiológica Integrada. 2020. Available online: https://www.gob.mx/salud/documentos/manual-de-procedimientos-epidemiologia (accessed on 4 February 2026).
- Departamento de Vigilancia de Factores Protectores y de Riesgo a la Salud y Enfermedades; Ministerio De Salud Panama; Dirección General De Salud Pública. Sistema Nacional De Vigilancia Epidemiológica De Las Encefalitis Virales Incluye La Vigilancia De La Enfermedad Por El Virus Del Nilo Occidental. 2013. Available online: https://www.minsa.gob.pa/sites/default/files/programas/guia_encefalitis_2013.pdf (accessed on 4 February 2026).
- Sistema Nacional de Vigilancia Epidemiológica; Ministerio de Salud Publica y Bienestar Social de Paraguay. Guía Nacional de Vigilancia y Control de Enfermedades. 2022. Available online: https://www.mspbs.gov.py/dependencias/portal/adjunto/guia-vigilancia-2022.pdf (accessed on 4 February 2026).
- Servicio Nacional de Sanidad Agraria (SENASA). Senasa: Vigilancia Epidemiológica Pasiva Para Determinar Enfermedades en Ganado Bovino; SENASA: Buenos Aires, Argentina, 2016.
- Ministerio de Salud Pública Urugay; División Epidemiología; Departamento de Vigilancia en Salud. Guía Nacional de Vigilancia y Control de Enfermedades y Eventos Sanitarios de Notificación Obligatoria. 2015. Available online: https://www.gub.uy/ministerio-salud-publica/sites/ministerio-salud-publica/files/documentos/publicaciones/guia_vigilancia_2015.pdf (accessed on 4 February 2026).
- Rathore, A.P.S.; St. John, A.L. Cross-Reactive Immunity Among Flaviviruses. Front. Immunol. 2020, 11, 334. [Google Scholar] [CrossRef]
- Rodríguez-Valencia, V.; Olive, M.-M.; Le Goff, G.; Faisse, M.; Bourel, M.; L’Ambert, G.; Vollot, B.; Tolsá-García, M.J.; Paupy, C.; Roiz, D. Host-Feeding Preferences of Culex Pipiens and Its Potential Significance for Flavivirus Transmission in the Camargue, France. Med. Vet. Entomol. 2025, 39, 614–625. [Google Scholar] [CrossRef]
- Fiacre, L.; Pagès, N.; Albina, E.; Richardson, J.; Lecollinet, S.; Gonzalez, G. Molecular Determinants of West Nile Virus Virulence and Pathogenesis in Vertebrate and Invertebrate Hosts. Int. J. Mol. Sci. 2020, 21, 9117. [Google Scholar] [CrossRef]
- Byas, A.D.; Ebel, G.D. Comparative Pathology of West Nile Virus in Humans and Non-Human Animals. Pathogens 2020, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Caminade, C.; McIntyre, K.M.; Jones, A.E. Impact of Recent and Future Climate Change on Vector-borne Diseases. Ann. N. Y Acad. Sci. 2019, 1436, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Simonin, Y. Circulation of West Nile Virus and Usutu Virus in Europe: Overview and Challenges. Viruses 2024, 16, 599. [Google Scholar] [CrossRef] [PubMed]
- Münger, E.; Atama, N.C.; van Irsel, J.; Blom, R.; Krol, L.; van Mastrigt, T.; van den Berg, T.J.; Braks, M.; de Vries, A.; van der Linden, A.; et al. One Health Approach Uncovers Emergence and Dynamics of Usutu and West Nile Viruses in the Netherlands. Nat. Commun. 2025, 16, 7883. [Google Scholar] [CrossRef]
- Pisani, G.; Cristiano, K.; Pupella, S.; Liumbruno, G.M. West Nile Virus in Europe and Safety of Blood Transfusion. Transfus. Med. Hemother 2016, 43, 158–167. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention. Update: West Nile Virus Screening of Blood Donations and Transfusion-Associated Transmission—United States, 2003. Morb. Mortal. Wkly. Rep. 2004, 53, 281–284. [Google Scholar]
- Tamba, M.; Bonilauri, P.; Galletti, G.; Casadei, G.; Santi, A.; Rossi, A.; Calzolari, M. West Nile Virus Surveillance Using Sentinel Birds: Results of Eleven Years of Testing in Corvids in a Region of Northern Italy. Front. Vet. Sci. 2024, 11, 1407271. [Google Scholar] [CrossRef]
- Vargas Campos, C.A.; García-Pérez, S.; Figuerola, J.; Martínez-de la Puente, J.; Polo, I.; Rodríguez-de-Fonseca, B.; Fernández-Álvarez, S.; Galván Fraile, V.; Martín-Rey, M.; Lacasaña, M.; et al. Comprehensive Analysis of West Nile Virus Transmission: Environmental, Ecological, and Individual Factors. An Umbrella Review. One Health 2025, 20, 100984. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Izquierdo-Condoy, J.S.; Salazar-Santoliva, C.; Vasconez-Gonzalez, J. Leptospirosis Deaths in Children in the Amazon: Syndemic Inequities. Lancet 2025, 405, 2275. [Google Scholar] [CrossRef]
- Izquierdo Condoy, J.S.; Tello-De-la-Torre, A.; Espinosa Del Pozo, P.; Ortiz-Prado, E. Advancing Global Health Equity: The Transformative Potential of Community-Based Surveillance in Developing Countries. Front. Public Health 2023, 11, 1294686. [Google Scholar] [CrossRef]
- Abbas, I.; Ahmed, F.; Muqaddas, H.; Alberti, A.; Varcasia, A.; Sedda, L. Epidemiology and Surveillance of West Nile Virus in the Mediterranean Basin during 2010–2023: A Systematic Review. Curr. Res. Parasitol. Vector Borne Dis. 2025, 7, 100277. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.