Increase in SARS-CoV-2 Seroprevalence in UK Domestic Felids Despite Weak Immunogenicity of Post-Omicron Variants
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Serological Testing
2.3. Pseudotype-Based Virus Neutralisation Assay
2.4. Feline-Specific Secondary ELISA
- No serum control—casein buffer.
- Negative control—pre-pandemic VDS cat samples that had previously tested negative for SARS-CoV-2 neutralising antibodies.
- Positive control—a pool of 20 pseudotype neutralisation assay and DABA positive serum samples identified previously [14].
2.5. Data Analysis
- Positive neutralising: positive (OD ≥ 0.5) or borderline (0.5 > OD ≥ 0.1) ELISA result plus a neutralisation titre ≥ 50 (dilution which results in a 90% reduction in infectivity) against at least one variant.
- Positive non-neutralising: positive ELISA result but no detectable neutralisation titre (titre of <50).
- Negative: negative ELISA result and no detectable neutralisation titre (titre < 50).
3. Results
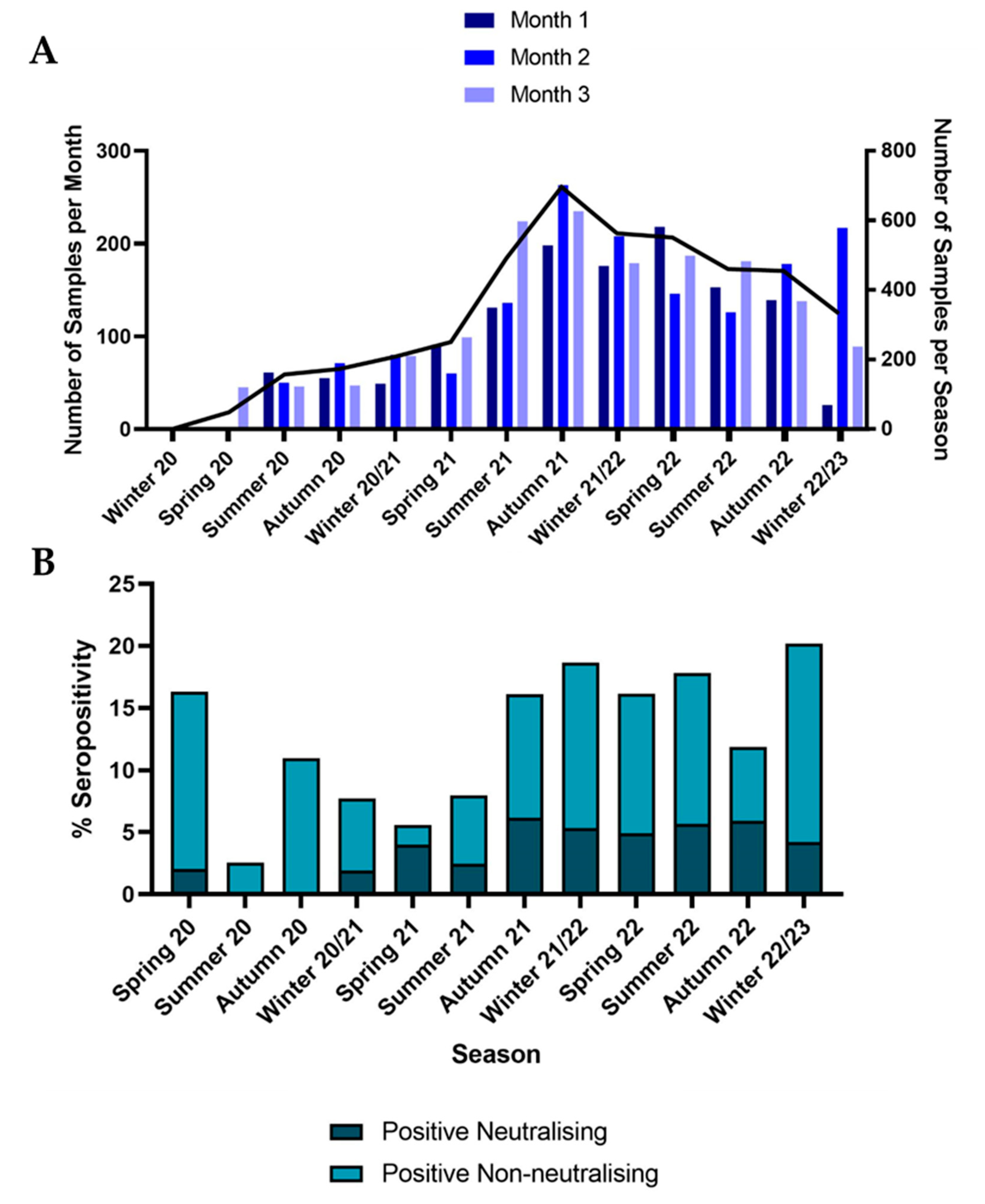
3.1. Samples
3.2. Seroprevalence
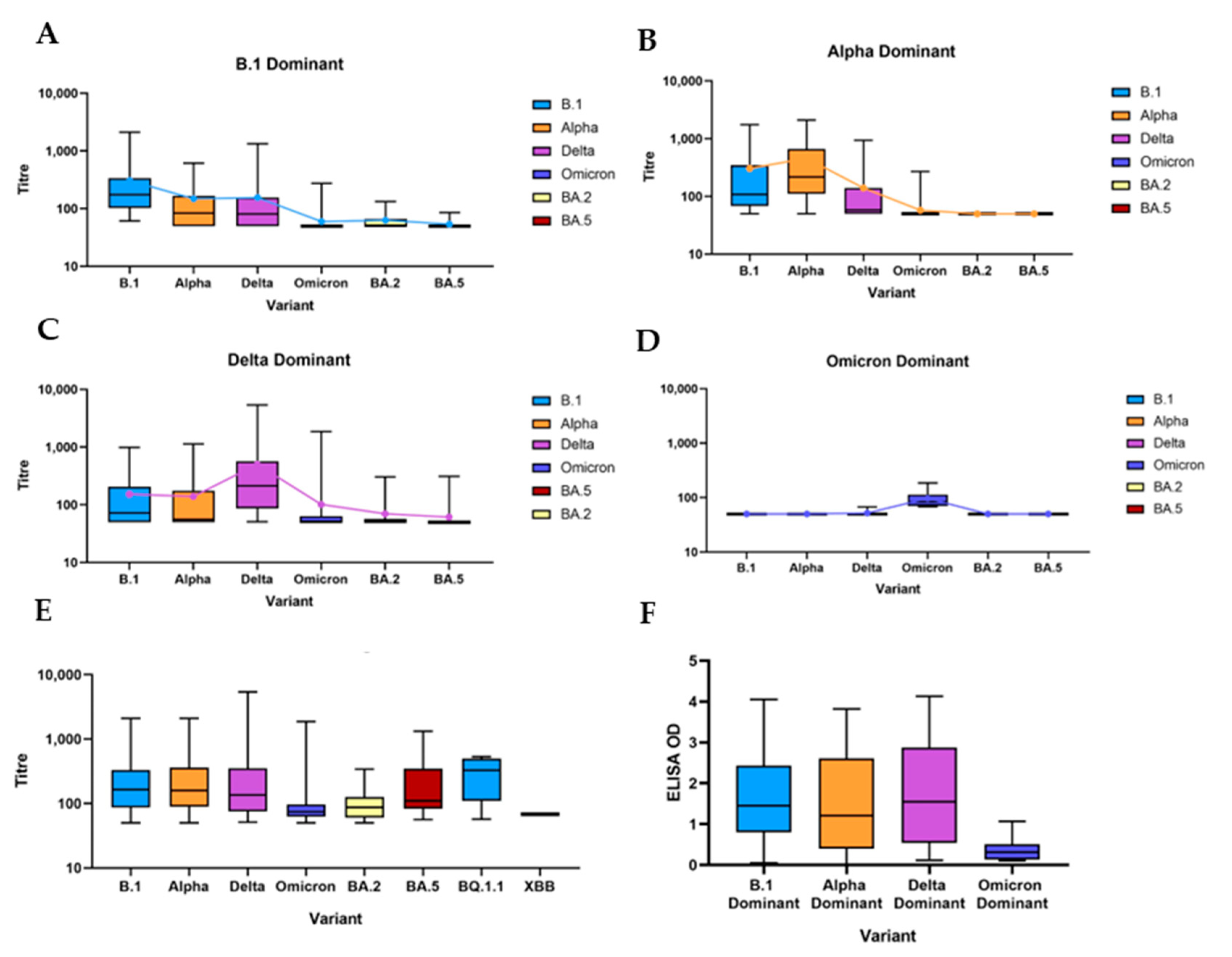
3.2.1. Comparison to Variants in Humans
3.2.2. B.1 Dominant
3.2.3. Alpha Dominant
3.2.4. Delta Dominant
3.2.5. Omicron Dominant and Post-Omicron Dominant
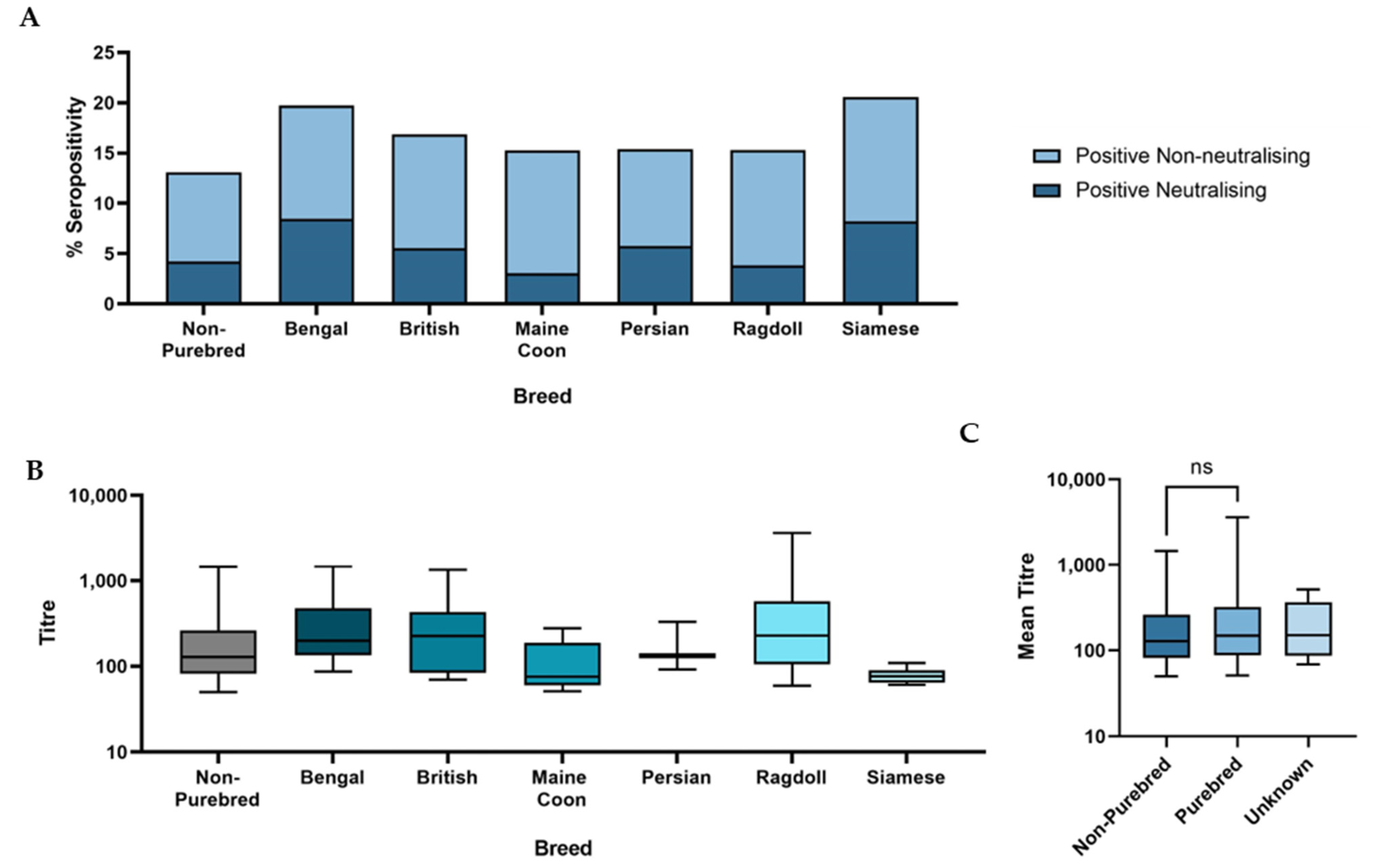
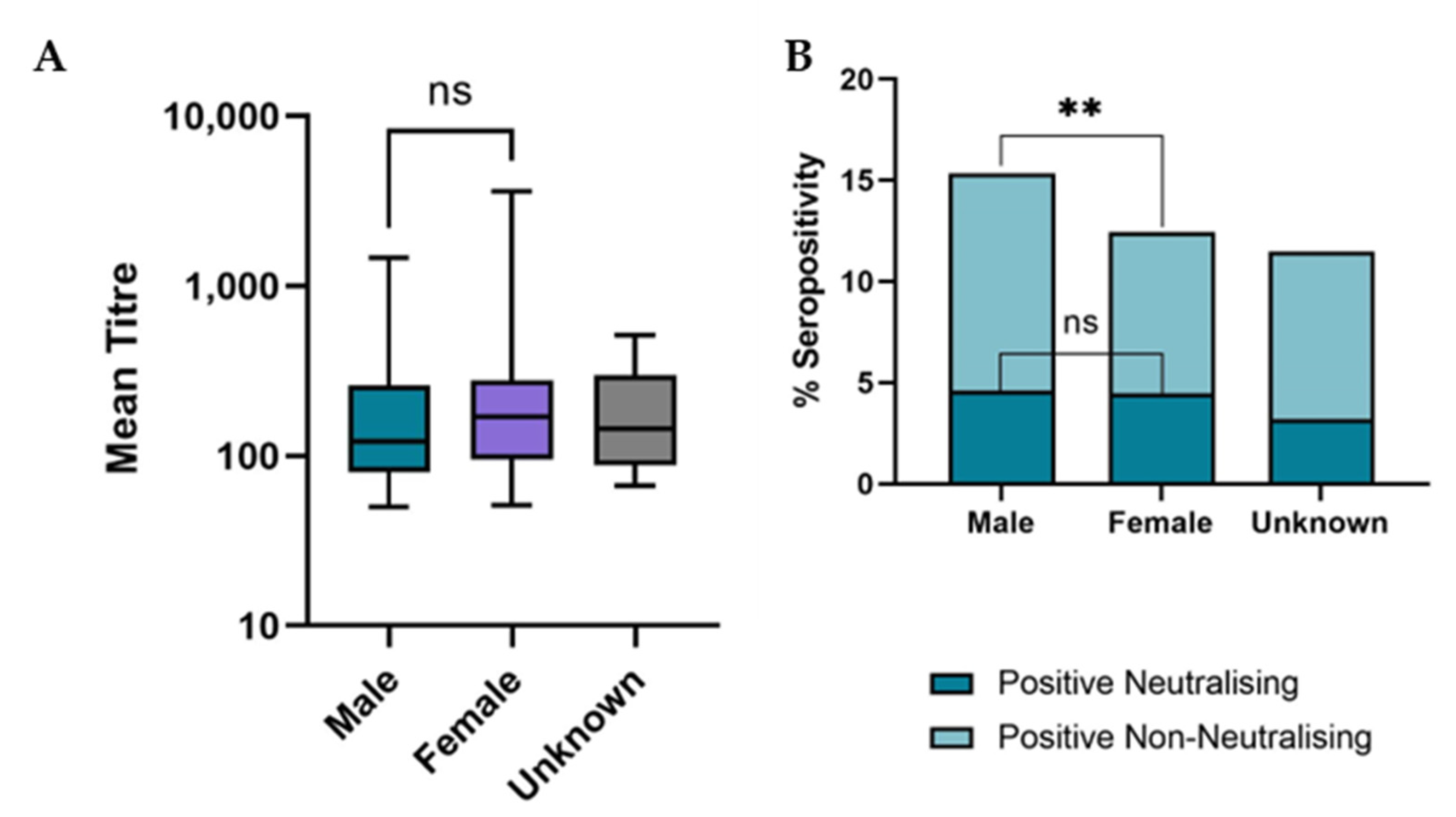
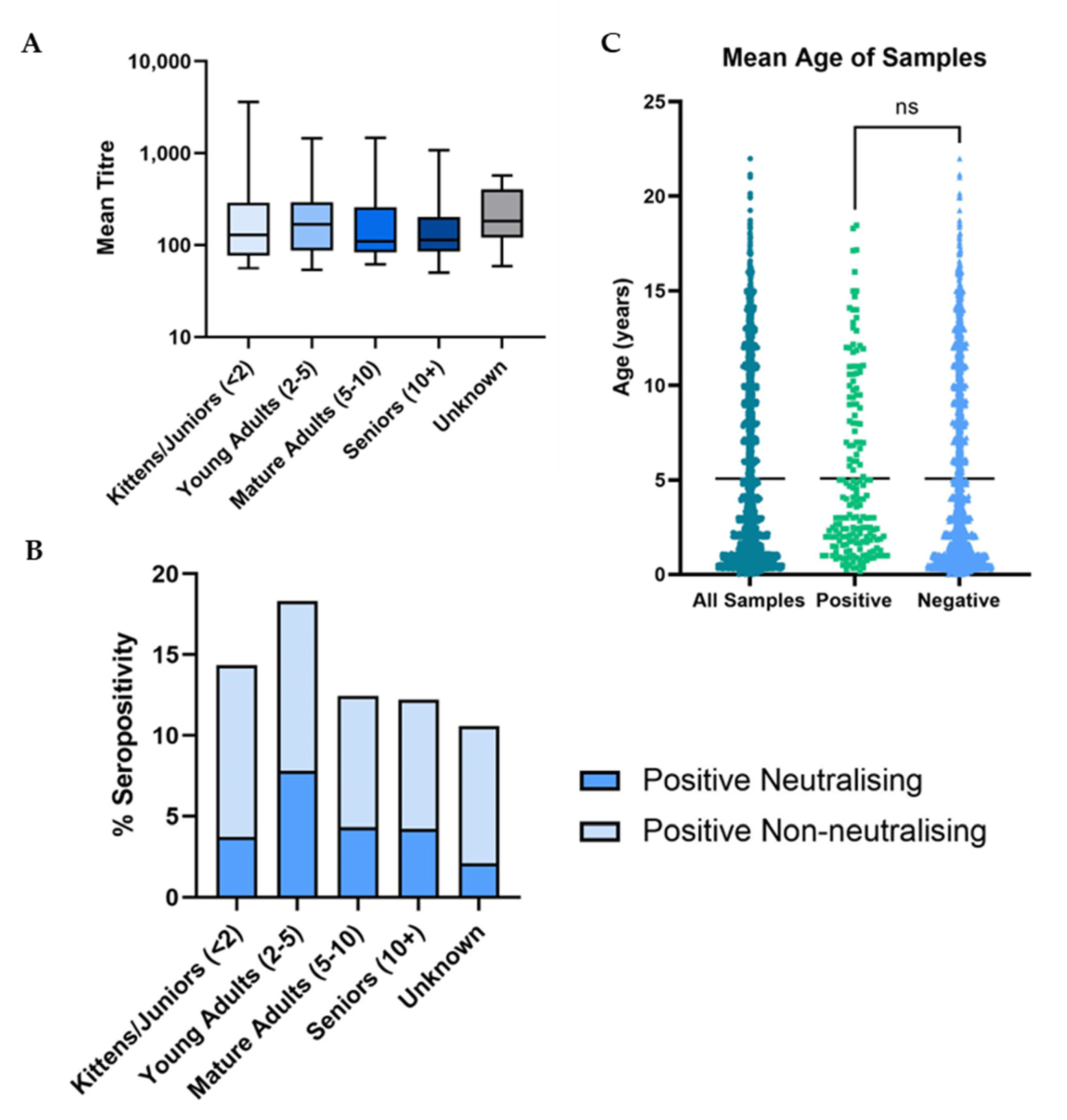
3.3. Demographic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Garigliany, M.; Van Laere, A.S.; Clercx, C.; Giet, D.; Escriou, N.; Huon, C.; van der Werf, S.; Eloit, M.; Desmecht, D. SARS-CoV-2 Natural Transmission from Human to Cat, Belgium, March 2020. Emerg. Infect. Dis. 2020, 26, 3069–3071. [Google Scholar] [CrossRef]
- Barrs, V.R.; Peiris, M.; Tam, K.W.S.; Law, P.Y.T.; Brackman, C.J.; To, E.M.W.; Yu, V.Y.T.; Chu, D.K.W.; Perera, R.; Sit, T.H.C. SARS-CoV-2 in Quarantined Domestic Cats from COVID-19 Households or Close Contacts, Hong Kong, China. Emerg. Infect. Dis. 2020, 26, 3071–3074. [Google Scholar]
- Stout, A.E.; André, N.M.; Jaimes, J.A.; Millet, J.K.; Whittaker, G.R. Coronaviruses in cats and other companion animals: Where does SARS-CoV-2/COVID-19 fit? Vet. Microbiol. 2020, 247, 108777. [Google Scholar] [PubMed]
- Krafft, E.; Denolly, S.; Boson, B.; Angelloz-Pessey, S.; Levaltier, S.; Nesi, N.; Corbet, S.; Leterrier, B.; Fritz, M.; Leroy, E.M.; et al. Report of One-Year Prospective Surveillance of SARS-CoV-2 in Dogs and Cats in France with Various Exposure Risks: Confirmation of a Low Prevalence of Shedding, Detection and Complete Sequencing of an Alpha Variant in a Cat. Viruses 2021, 13, 1759. [Google Scholar] [CrossRef]
- Kannekens-Jager, M.M.; de Rooij, M.M.T.; de Groot, Y.; Biesbroeck, E.; de Jong, M.K.; Pijnacker, T.; Smit, L.A.M.; Schuurman, N.; Broekhuizen-Stins, M.J.; Zhao, S.; et al. SARS-CoV-2 infection in dogs and cats is associated with contact to COVID-19 positive household members. Transbound. Emerg. Dis. 2022, 69, 4034–4040. [Google Scholar] [PubMed]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [PubMed]
- Bosco-Lauth, A.M.; Hartwig, A.E.; Porter, S.M.; Gordy, P.W.; Nehring, M.; Byas, A.D.; Vandewoude, S.; Ragan, I.K.; Maison, R.M.; Bowen, R.A. Pathogenesis, transmission and response to re-exposure of SARS-CoV-2 in domestic cats. Proc. Natl. Acad. Sci. USA 2020, 117, 26382–26388. [Google Scholar]
- Gaudreault, N.N.; Trujillo, J.D.; Carossino, M.; Meekins, D.A.; Morozov, I.; Madden, D.W.; Indran, S.V.; Bold, D.; Balaraman, V.; Kwon, T.; et al. SARS-CoV-2 infection, disease and transmission in domestic cats. Emerg. Microbes Infect. 2020, 9, 2322–2332. [Google Scholar] [PubMed]
- Dias, H.G.; Resck, M.E.B.; Caldas, G.C.; Resck, A.F.; da Silva, N.V.; Dos Santos, A.M.V.; Sousa, T.D.C.; Ogrzewalska, M.H.; Siqueira, M.M.; Pauvolid-Corrêa, A.; et al. Neutralizing antibodies for SARS-CoV-2 in stray animals from Rio de Janeiro, Brazil. PLoS ONE 2021, 16, e0248578. [Google Scholar]
- Zhang, Q.; Zhang, H.; Gao, J.; Huang, K.; Yang, Y.; Hui, X.; He, X.; Li, C.; Gong, W.; Zhang, Y.; et al. A serological survey of SARS-CoV-2 in cat in Wuhan. Emerg. Microbes Infect. 2020, 9, 2013–2019. [Google Scholar] [PubMed]
- van Aart, A.E.; Velkers, F.C.; Fischer, E.A.J.; Broens, E.M.; Egberink, H.; Zhao, S.; Engelsma, M.; Hakze-van der Honing, R.W.; Harders, F.; de Rooij, M.M.T.; et al. SARS-CoV-2 infection in cats and dogs in infected mink farms. Transbound. Emerg. Dis. 2022, 69, 3001–3007. [Google Scholar]
- Hosie, M.J.; Epifano, I.; Herder, V.; Orton, R.J.; Stevenson, A.; Johnson, N.; MacDonald, E.; Dunbar, D.; McDonald, M.; Howie, F.; et al. Detection of SARS-CoV-2 in respiratory samples from cats in the UK associated with human-to-cat transmission. Vet. Rec. 2021, 188, e247. [Google Scholar] [PubMed]
- Smith, S.L.; Anderson, E.R.; Cansado-Utrilla, C.; Prince, T.; Farrell, S.; Brant, B.; Smyth, S.; Noble, P.M.; Pinchbeck, G.L.; Marshall, N.; et al. SARS-CoV-2 neutralising antibodies in dogs and cats in the United Kingdom. Curr. Res. Virol. Sci. 2021, 2, 100011. [Google Scholar]
- Tyson, G.B.; Jones, S.; Logan, N.; McDonald, M.; Marshall, L.; Murcia, P.R.; Willett, B.J.; Weir, W.; Hosie, M.J. SARS-CoV-2 Seroprevalence and Cross-Variant Antibody Neutralization in Cats, United Kingdom. Emerg. Infect. Dis. 2023, 29, 1223–1227. [Google Scholar] [PubMed]
- Fritz, M.; Rosolen, B.; Krafft, E.; Becquart, P.; Elguero, E.; Vratskikh, O.; Denolly, S.; Boson, B.; Vanhomwegen, J.; Gouilh, M.A.; et al. High prevalence of SARS-CoV-2 antibodies in pets from COVID-19+ households. One Health 2021, 11, 100192. [Google Scholar]
- Tiwari, R.; Dhama, K.; Sharun, K.; Iqbal Yatoo, M.; Malik, Y.S.; Singh, R.; Michalak, I.; Sah, R.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J. COVID-19: Animals, veterinary and zoonotic links. Vet. Q. 2020, 40, 169–182. [Google Scholar] [PubMed]
- Castillo, C.; Hernández, J. The COVID pandemic should introduce new habits for pet owners. Res. Vet. Sci. 2021, 139, 1–3. [Google Scholar]
- Sila, T.; Sunghan, J.; Laochareonsuk, W.; Surasombatpattana, S.; Kongkamol, C.; Ingviya, T.; Siripaitoon, P.; Kositpantawong, N.; Kanchanasuwan, S.; Hortiwakul, T.; et al. Suspected Cat-to-Human Transmission of SARS-CoV-2, Thailand, July-September 2021. Emerg. Infect. Dis. 2022, 28, 1485. [Google Scholar]
- Siegrist, A.A.; Richardson, K.L.; Ghai, R.R.; Pope, B.; Yeadon, J.; Culp, B.; Behravesh, C.B.; Liu, L.; Brown, J.A.; Boyer, L.V. Probable Transmission of SARS-CoV-2 from African Lion to Zoo Employees, Indiana, USA, 2021. Emerg. Infect. Dis. 2023, 29, 1102–1108. [Google Scholar]
- Patterson, E.I.; Elia, G.; Grassi, A.; Giordano, A.; Desario, C.; Medardo, M.; Smith, S.L.; Anderson, E.R.; Prince, T.; Patterson, G.T.; et al. Evidence of exposure to SARS-CoV-2 in cats and dogs from households in Italy. Nat. Commun. 2020, 11, 6231. [Google Scholar]
- Barua, S.; Hoque, M.; Adekanmbi, F.; Kelly, P.; Jenkins-Moore, M.; Torchetti, M.K.; Chenoweth, K.; Wood, T.; Wang, C. Antibodies to SARS-CoV-2 in dogs and cats, USA. Emerg. Microbes Infect. 2021, 10, 1669–1674. [Google Scholar]
- Colitti, B.; Bertolotti, L.; Mannelli, A.; Ferrara, G.; Vercelli, A.; Grassi, A.; Trentin, C.; Paltrinieri, S.; Nogarol, C.; Decaro, N.; et al. Cross-Sectional Serosurvey of Companion Animals Housed with SARS-CoV-2-Infected Owners, Italy. Emerg. Infect. Dis. 2021, 27, 1919–1922. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Arrondo, I.; Portillo, A.; Palomar, A.M.; Santibáñez, S.; Santibáñez, P.; Cervera, C.; Oteo, J.A. Detection of SARS-CoV-2 in pets living with COVID-19 owners diagnosed during the COVID-19 lockdown in Spain: A case of an asymptomatic cat with SARS-CoV-2 in Europe. Transbound. Emerg. Dis. 2021, 68, 973–976. [Google Scholar] [CrossRef]
- Decaro, N.; Balboni, A.; Bertolotti, L.; Martino, P.A.; Mazzei, M.; Mira, F.; Pagnini, U. SARS-CoV-2 Infection in Dogs and Cats: Facts and Speculations. Front. Vet. Sci. 2021, 8, 619207. [Google Scholar] [CrossRef] [PubMed]
- Ferasin, L.; Fritz, M.; Ferasin, H.; Becquart, P.; Corbet, S.; Ar Gouilh, M.; Legros, V.; Leroy, E.M. Infection with SARS-CoV-2 variant B.1.1.7 detected in a group of dogs and cats with suspected myocarditis. Vet. Rec. 2021, 189, e944. [Google Scholar] [CrossRef]
- Chetboul, V.; Foulex, P.; Kartout, K.; Klein, A.M.; Sailleau, C.; Dumarest, M.; Delaplace, M.; Gouilh, M.A.; Mortier, J.; Le Poder, S. Myocarditis and Subclinical-Like Infection Associated with SARS-CoV-2 in Two Cats Living in the Same Household in France: A Case Report with Literature Review. Front. Vet. Sci. 2021, 8, 748869. [Google Scholar] [CrossRef] [PubMed]
- Gaudreault, N.N.; Carossino, M.; Morozov, I.; Trujillo, J.D.; Meekins, D.A.; Madden, D.W.; Cool, K.; Artiaga, B.L.; McDowell, C.; Bold, D.; et al. Experimental re-infected cats do not transmit SARS-CoV-2. Emerg. Microbes Infect. 2021, 10, 638–650. [Google Scholar] [PubMed]
- Zhao, S.; Schuurman, N.; Li, W.; Wang, C.; Smit, L.A.M.; Broens, E.M.; Wagenaar, J.A.; Van Kuppeveld, F.J.M.; Bosch, B.-J.; Egberink, H. Serologic Screening of Severe Acute Respiratory Syndrome Coronavirus 2 Infection in Cats and Dogs during First Coronavirus Disease Wave, the Netherlands. Emerg. Infect. Dis. 2021, 27, 1362–1370. [Google Scholar]
- Wise, J. Covid-19: Omicron sub variants driving new wave of infections in UK. BMJ 2022, 377, o1506. [Google Scholar] [CrossRef]
- Dhawan, M.; Priyanka; Choudhary, O.P. Emergence of Omicron sub-variant BA.2: Is it a matter of concern amid the COVID-19 pandemic? Int. J. Surg. 2022, 99, 106581. [Google Scholar] [CrossRef]
- Tegally, H.; Moir, M.; Everatt, J.; Giovanetti, M.; Scheepers, C.; Wilkinson, E.; Subramoney, K.; Makatini, Z.; Moyo, S.; Amoako, D.G.; et al. Emergence of SARS-CoV-2 Omicron lineages BA.4 and BA.5 in South Africa. Nat. Med. 2022, 28, 1785–1790. [Google Scholar] [CrossRef] [PubMed]
- Qu, P.; Evans, J.P.; Faraone, J.; Zheng, Y.M.; Carlin, C.; Anghelina, M.; Stevens, P.; Fernandez, S.; Jones, D.; Lozanski, G.; et al. Distinct Neutralizing Antibody Escape of SARS-CoV-2 Omicron Subvariants BQ.1, BQ.1.1, BA.4.6, BF.7 and BA.2.75.2. bioRxiv 2022. [Google Scholar] [CrossRef]
- Akerman, A.; Milogiannakis, V.; Jean, T.; Esneau, C.; Silva, M.R.; Ison, T.; Fichter, C.; Lopez, J.A.; Chandra, D.; Naing, Z.; et al. Emergence and antibody evasion of BQ, BA.2.75 and SARS-CoV-2 recombinant sub-lineages in the face of maturing antibody breadth at the population level. EBioMedicine 2023, 90. [Google Scholar] [CrossRef] [PubMed]
- Uraki, R.; Ito, M.; Furusawa, Y.; Yamayoshi, S.; Iwatsuki-Horimoto, K.; Adachi, E.; Saito, M.; Koga, M.; Tsutsumi, T.; Yamamoto, S.; et al. Humoral immune evasion of the omicron subvariants BQ.1.1 and XBB. Lancet Infect. Dis. 2023, 23, 30–32. [Google Scholar] [CrossRef]
- Planas, D.; Bruel, T.; Staropoli, I.; Guivel-Benhassine, F.; Porrot, F.; Maes, P.; Grzelak, L.; Prot, M.; Mougari, S.; Planchais, C.; et al. Resistance of Omicron subvariants BA.2.75.2, BA.4.6 and BQ.1.1 to neutralizing antibodies. bioRxiv 2022. [Google Scholar] [CrossRef]
- Willett, B.J.; Grove, J.; Maclean, O.A.; Wilkie, C.; Logan, N.; Lorenzo, G.D.; Furnon, W.; Scott, S.; Manali, M.; Szemiel, A.; et al. The hyper-transmissible SARS-CoV-2 Omicron variant exhibits significant antigenic change, vaccine escape and a switch in cell entry mechanism. Nat. Microbiol. 2022, 7, 1161–1179. [Google Scholar] [CrossRef]
- Shrestha, L.B.; Foster, C.; Rawlinson, W.; Tedla, N.; Bull, R.A. Evolution of the SARS-CoV-2 omicron variants BA.1 to BA.5: Implications for immune escape and transmission. Rev. Med. Virol. 2022, 32, e2381. [Google Scholar] [CrossRef]
- Davis, C.; Logan, N.; Tyson, G.; Orton, R.; Harvey, W.T.; Perkins, J.S.; Mollett, G.; Blacow, R.M.; Peacock, T.P.; Barclay, W.S.; et al. Reduced neutralisation of the Delta (B.1.617.2) SARS-CoV-2 variant of concern following vaccination. PLoS Pathog. 2021, 17, e1010022. [Google Scholar] [CrossRef]
- Hughes, E.C.; Amat, J.A.R.; Haney, J.; Parr, Y.A.; Logan, N.; Palmateer, N.; Nickbakhsh, S.; Ho, A.; Cherepanov, P.; Rosa, A.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 Serosurveillance in a Patient Population Reveals Differences in Virus Exposure and Antibody-Mediated Immunity According to Host Demography and Healthcare Setting. J. Infect. Dis. 2021, 223, 971–980. [Google Scholar] [CrossRef]
- Zufferey, R.; Nagy, D.; Mandel, R.J.; Naldini, L.; Trono, D. Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat. Biotechnol. 1997, 15, 871–875. [Google Scholar] [CrossRef]
- Zufferey, R.; Dull, T.; Mandel, R.J.; Bukovsky, A.; Quiroz, D.; Naldini, L.; Trono, D. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J. Virol. 1998, 72, 9873–9880. [Google Scholar] [CrossRef]
- Hyseni, I.; Molesti, E.; Benincasa, L.; Piu, P.; Casa, E.; Temperton, N.J.; Manenti, A.; Montomoli, E. Characterisation of SARS-CoV-2 Lentiviral Pseudotypes and Correlation between Pseudotype-Based Neutralisation Assays and Live Virus-Based Micro Neutralisation Assays. Viruses 2020, 12, 1011. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.; Weisblum, Y.; Muecksch, F.; Hoffmann, H.H.; Michailidis, E.; Lorenzi, J.C.C.; Mendoza, P.; Rutkowska, M.; Bednarski, E.; Gaebler, C.; et al. Measuring SARS-CoV-2 neutralizing antibody activity using pseudotyped and chimeric viruses. J. Exp. Med. 2020, 217, e20201181. [Google Scholar] [CrossRef]
- Parr, Y.A.; Beall, M.J.; Levy, J.K.; McDonald, M.; Hamman, N.T.; Willett, B.J.; Hosie, M.J. Measuring the Humoral Immune Response in Cats Exposed to Feline Leukaemia Virus. Viruses 2021, 13, 428. [Google Scholar] [CrossRef] [PubMed]
- Bahnan, W.; Wrighton, S.; Sundwall, M.; Bläckberg, A.; Larsson, O.; Höglund, U.; Khakzad, H.; Godzwon, M.; Walle, M.; Elder, E.; et al. Spike-Dependent Opsonization Indicates Both Dose-Dependent Inhibition of Phagocytosis and That Non-Neutralizing Antibodies Can Confer Protection to SARS-CoV-2. Front. Immunol. 2021, 12, 808932. [Google Scholar] [CrossRef]
- Krutikov, M.; Palmer, T.; Tut, G.; Fuller, C.; Azmi, B.; Giddings, R.; Shrotri, M.; Kaur, N.; Sylla, P.; Lancaster, T.; et al. Prevalence and duration of detectable SARS-CoV-2 nucleocapsid antibodies in staff and residents of long-term care facilities over the first year of the pandemic (VIVALDI study): Prospective cohort study in England. Lancet Healthy Longev. 2022, 3, e13–e21. [Google Scholar] [CrossRef]
- Cubuk, J.; Alston, J.J.; Incicco, J.J.; Singh, S.; Stuchell-Brereton, M.D.; Ward, M.D.; Zimmerman, M.I.; Vithani, N.; Griffith, D.; Wagoner, J.A.; et al. The SARS-CoV-2 nucleocapsid protein is dynamic, disordered, and phase separates with RNA. Nat. Commun. 2021, 12, 1936. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Wang, J.; Jian, F.; Xiao, T.; Song, W.; Yisimayi, A.; Huang, W.; Li, Q.; Wang, P.; An, R.; et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 2022, 602, 657–663. [Google Scholar] [CrossRef]
- Wang, Q.; Guo, Y.; Iketani, S.; Nair, M.S.; Li, Z.; Mohri, H.; Wang, M.; Yu, J.; Bowen, A.D.; Chang, J.Y.; et al. Antibody evasion by SARS-CoV-2 Omicron subvariants BA.2.12.1, BA.4 and BA.5. Nature 2022, 608, 603–608. [Google Scholar] [CrossRef]
- Bálint, G.; Vörös-Horváth, B.; Széchenyi, A. Omicron: Increased transmissibility and decreased pathogenicity. Signal Transduct. Target. Ther. 2022, 7, 151. [Google Scholar] [CrossRef]
- Arora, P.; Zhang, L.; Rocha, C.; Sidarovich, A.; Kempf, A.; Schulz, S.; Cossmann, A.; Manger, B.; Baier, E.; Tampe, B.; et al. Comparable neutralisation evasion of SARS-CoV-2 omicron subvariants BA.1, BA.2, and BA.3. Lancet Infect. Dis. 2022, 22, 766–767. [Google Scholar] [CrossRef] [PubMed]
- Torjesen, I. COVID-19: Omicron may be more transmissible than other variants and partly resistant to existing vaccines, scientists fear. BMJ 2021, 375, 2943. [Google Scholar]
- Ai, J.; Zhang, H.; Zhang, Y.; Lin, K.; Zhang, Y.; Wu, J.; Wan, Y.; Huang, Y.; Song, J.; Fu, Z.; et al. Omicron variant showed lower neutralizing sensitivity than other SARS-CoV-2 variants to immune sera elicited by vaccines after boost. Emerg. Microbes Infect. 2022, 11, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Peacock, T.P.; Brown, J.C.; Zhou, J.; Thakur, N.; Sukhova, K.; Newman, J.; Kugathasan, R.; Yan, A.W.C.; Furnon, W.; De Lorenzo, G.; et al. The altered entry pathway and antigenic distance of the SARS-CoV-2 Omicron variant map to separate domains of spike protein. BioRxiv 2022. [Google Scholar] [CrossRef]
- Sánchez-Morales, L.; Sánchez-Vizcaíno, J.M.; Pérez-Sancho, M.; Domínguez, L.; Barroso-Arévalo, S. The Omicron (B.1.1.529) SARS-CoV-2 variant of concern also affects companion animals. Front. Vet. Sci. 2022, 9, 940710. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Michelitsch, A.; Allendorf, V.; Conraths, F.J.; Beer, M.; Denzin, N.; Wernike, K. Dogs and cats are less susceptible to the omicron variant of concern of SARS-CoV-2—A field study. BioRxiv 2023. [Google Scholar] [CrossRef]
- Foreman-Worsley, R.; Finka, L.R.; Ward, S.J.; Farnworth, M.J. Indoors or Outdoors? An International Exploration of Owner Demographics and Decision Making Associated with Lifestyle of Pet Cats. Animals 2021, 11, 253. [Google Scholar] [CrossRef]
- Salonen, M.; Vapalahti, K.; Tiira, K.; Mäki-Tanila, A.; Lohi, H. Breed differences of heritable behaviour traits in cats. Sci. Rep. 2019, 9, 7949. [Google Scholar] [CrossRef]
- Gunn-Moore, D.; Bessant, C.; Malik, R. Breed-related disorders of cats. J. Small Anim. Pract. 2008, 49, 167–168. [Google Scholar] [CrossRef]
- Kramer, J.W.; Davis, W.C.; Prieur, D.J. The Chediak-Higashi syndrome of cats. Lab. Investig. 1977, 36, 554–562. [Google Scholar]
- Koyasu, H.; Takahashi, H.; Sasao, I.; Takagi, S.; Nagasawa, M.; Kikusui, T. Sociality of Cats toward Humans Can Be Influenced by Hormonal and Socio-Environmental Factors: Pilot Study. Animals 2022, 13, 146. [Google Scholar] [CrossRef]
- Pesteanu-Somogyi, L.D.; Radzai, C.; Pressler, B.M. Prevalence of feline infectious peritonitis in specific cat breeds. J. Feline Med. Surg. 2006, 8, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ravi, M.; Wobeser, G.A.; Taylor, S.M.; Jackson, M.L. Naturally acquired feline immunodeficiency virus (FIV) infection in cats from western Canada: Prevalence, disease associations, and survival analysis. Can. Vet. J. 2010, 51, 271–276. [Google Scholar] [PubMed]
- Levy, J.K.; Scott, H.M.; Lachtara, J.L.; Crawford, P.C. Seroprevalence of feline leukemia virus and feline immunodeficiency virus infection among cats in North America and risk factors for seropositivity. J. Am. Vet. Med. Assoc. 2006, 228, 371–376. [Google Scholar]
- Plitman, L.; Černá, P.; Farnworth, M.J.; Packer, R.M.A.; Gunn-Moore, D.A. Motivation of Owners to Purchase Pedigree Cats, with Specific Focus on the Acquisition of Brachycephalic Cats. Animals 2019, 9, 394. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, D.G.; Church, D.B.; McGreevy, P.D.; Thomson, P.C.; Brodbelt, D.C. Prevalence of disorders recorded in cats attending primary-care veterinary practices in England. Vet. J. 2014, 202, 286–291. [Google Scholar]
- Sit, T.H.C.; Brackman, C.J.; Ip, S.M.; Tam, K.W.S.; Law, P.Y.T.; To, E.M.W.; Yu, V.Y.T.; Sims, L.D.; Tsang, D.N.C.; Chu, D.K.W.; et al. Infection of dogs with SARS-CoV-2. Nature 2020, 586, 776–778. [Google Scholar] [CrossRef]
- Hamer, S.A.; Pauvolid-Corrêa, A.; Zecca, I.B.; Davila, E.; Auckland, L.D.; Roundy, C.M.; Tang, W.; Torchetti, M.; Killian, M.L.; Jenkins-Moore, M.; et al. Natural SARS-CoV-2 infections, including virus isolation, among serially tested cats and dogs in households with confirmed human COVID-19 cases in Texas, USA. bioRxiv 2020. [Google Scholar] [CrossRef]
- Cats Protection. CATS Report 2022. Available online: https://www.cats.org.uk/media/11908/cats-report-2022-uk.pdf (accessed on 26 June 2023).
- APHA (Animal and Plant Health Agency). SARS-CoV-2 in Animals—Case Definition, Testing and International Reporting Obligations, 2nd ed.; DEFRA, Ed.; HM Government: London, UK, 2021.

| Sample | Sampling Date | ELISA OD | B.1 Titre | Alpha Titre | Delta Titre | BA.1 Titre | BA.2 Titre | BA.5 Titre | BQ.1.1 Titre | XBB Titre |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 20/05/2022 | 0.94035 | 0 | 0 | 0 | 79 | 309 | 0 | ||
| B | 31/08/2022 | 0.5318 | 0 | 0 | 0 | 0 | 58 | 121 | ||
| J | 09/09/2022 | 0.5393 | 0 | 0 | 0 | 0 | 0 | 80 | ||
| M | 14/11/2022 | 3.0546 | 0 | 0 | 0 | 69 | 328 | 1320 | ||
| X | 09/01/2023 | 0.9723 | 0 | 74 | 0 | 59 | 124 | 448 | 267 | 0 |
| T | 16/01/2023 | 0.5637 | 0 | 0 | 0 | 55 | 342 | 393 | 533 | 68 |
| D | 22/02/2023 | 0.2621 | 0 | 0 | 0 | 0 | 61 | 332 | 391 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tyson, G.B.; Jones, S.; Montreuil-Spencer, C.; Logan, N.; Scott, S.; Sasvari, H.; McDonald, M.; Marshall, L.; Murcia, P.R.; Willett, B.J.; et al. Increase in SARS-CoV-2 Seroprevalence in UK Domestic Felids Despite Weak Immunogenicity of Post-Omicron Variants. Viruses 2023, 15, 1661. https://doi.org/10.3390/v15081661
Tyson GB, Jones S, Montreuil-Spencer C, Logan N, Scott S, Sasvari H, McDonald M, Marshall L, Murcia PR, Willett BJ, et al. Increase in SARS-CoV-2 Seroprevalence in UK Domestic Felids Despite Weak Immunogenicity of Post-Omicron Variants. Viruses. 2023; 15(8):1661. https://doi.org/10.3390/v15081661
Chicago/Turabian StyleTyson, Grace B., Sarah Jones, Chloe Montreuil-Spencer, Nicola Logan, Sam Scott, Hagar Sasvari, Michael McDonald, Leigh Marshall, Pablo R. Murcia, Brian J. Willett, and et al. 2023. "Increase in SARS-CoV-2 Seroprevalence in UK Domestic Felids Despite Weak Immunogenicity of Post-Omicron Variants" Viruses 15, no. 8: 1661. https://doi.org/10.3390/v15081661
APA StyleTyson, G. B., Jones, S., Montreuil-Spencer, C., Logan, N., Scott, S., Sasvari, H., McDonald, M., Marshall, L., Murcia, P. R., Willett, B. J., Weir, W., & Hosie, M. J. (2023). Increase in SARS-CoV-2 Seroprevalence in UK Domestic Felids Despite Weak Immunogenicity of Post-Omicron Variants. Viruses, 15(8), 1661. https://doi.org/10.3390/v15081661







