Subterranean Clover Stunt Virus Revisited: Detection of Two Missing Genome Components
Abstract
:1. Introduction
2. Materials and Methods
2.1. Virus Isolates and Field Materials
2.2. DNA Extraction, RCA, PCR, Cloning, and Sequencing
2.3. Sequence Assembly and Analysis
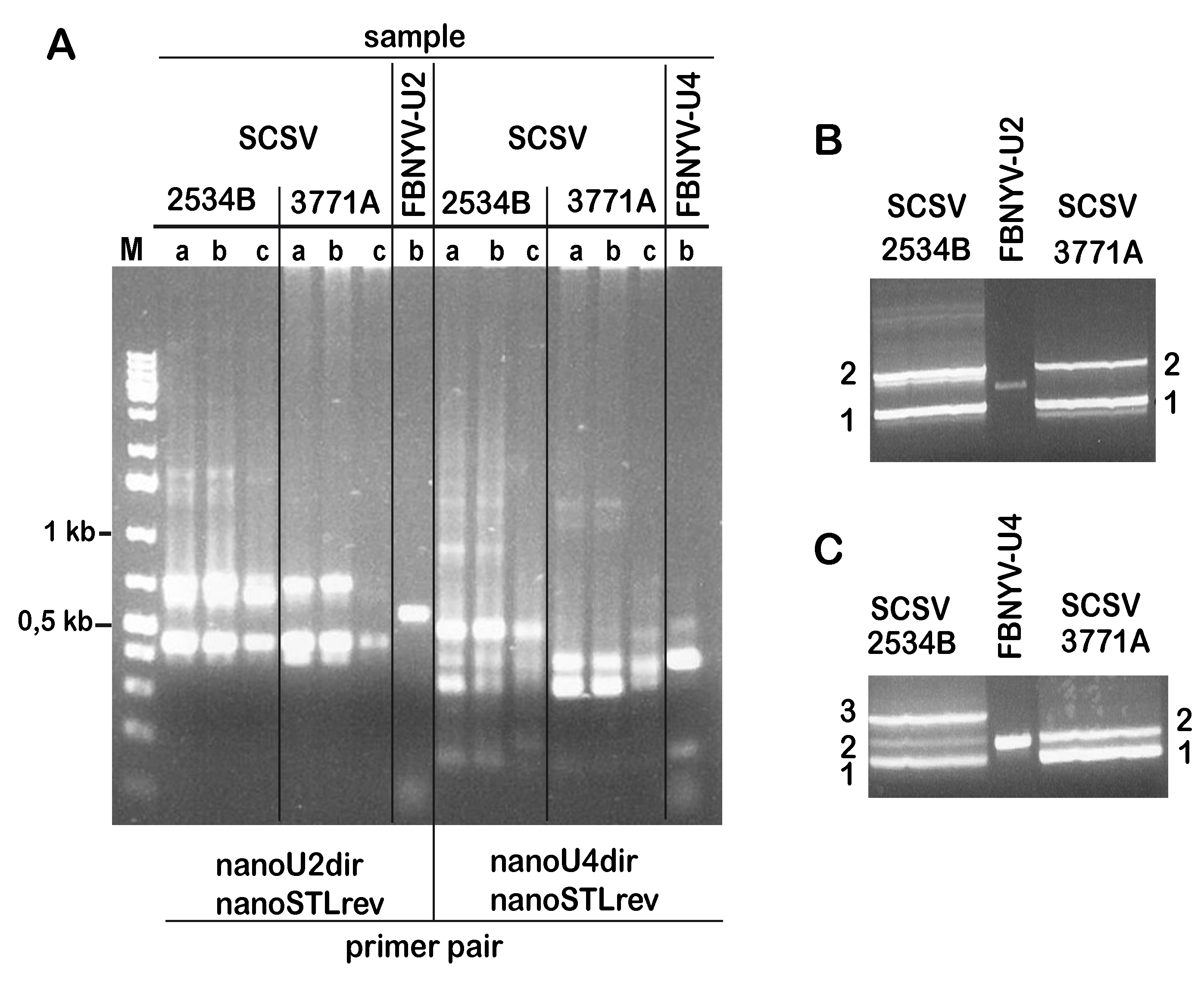
3. Results
3.1. Identification of SCSV DNA-U2 and DNA-U4
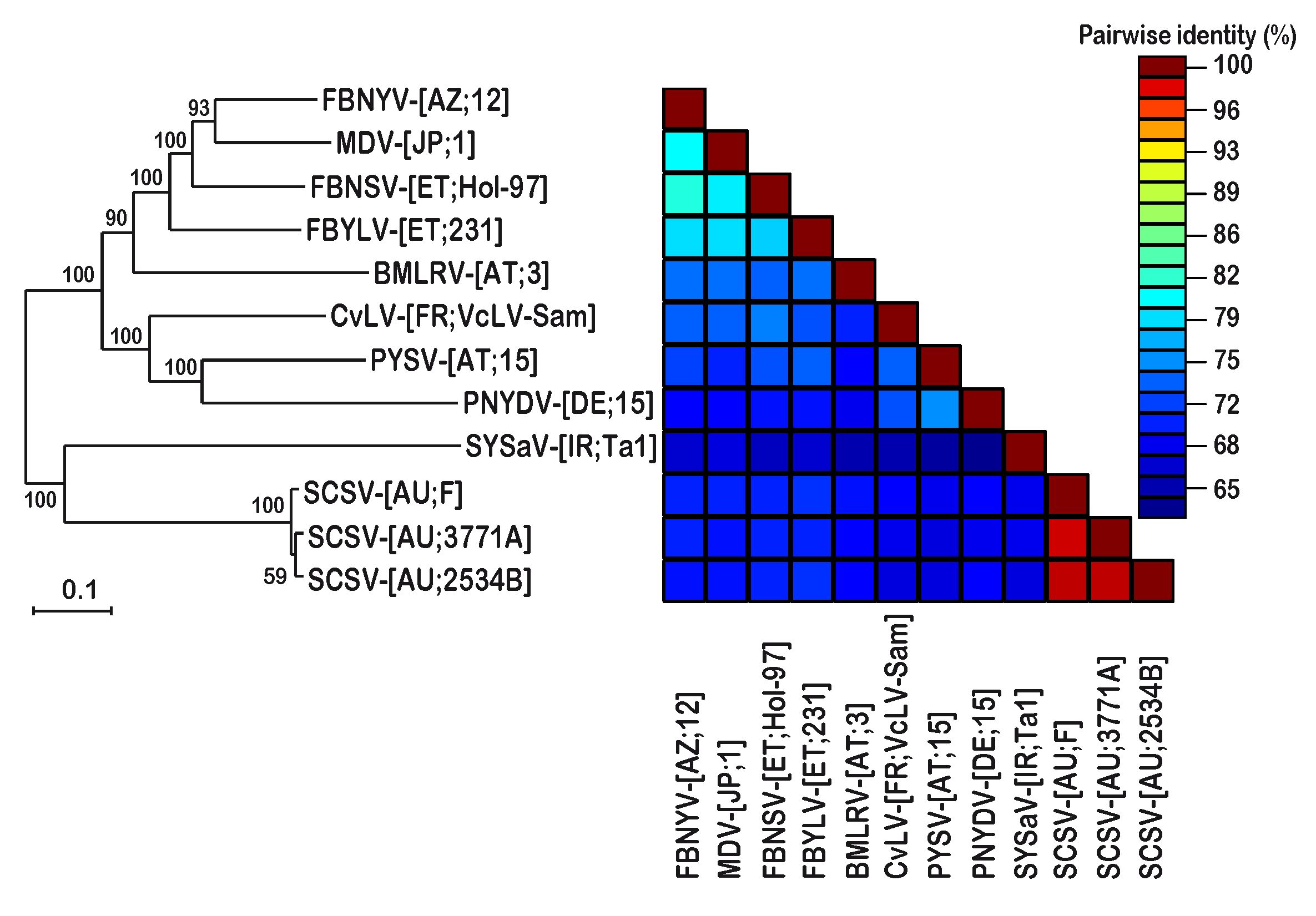
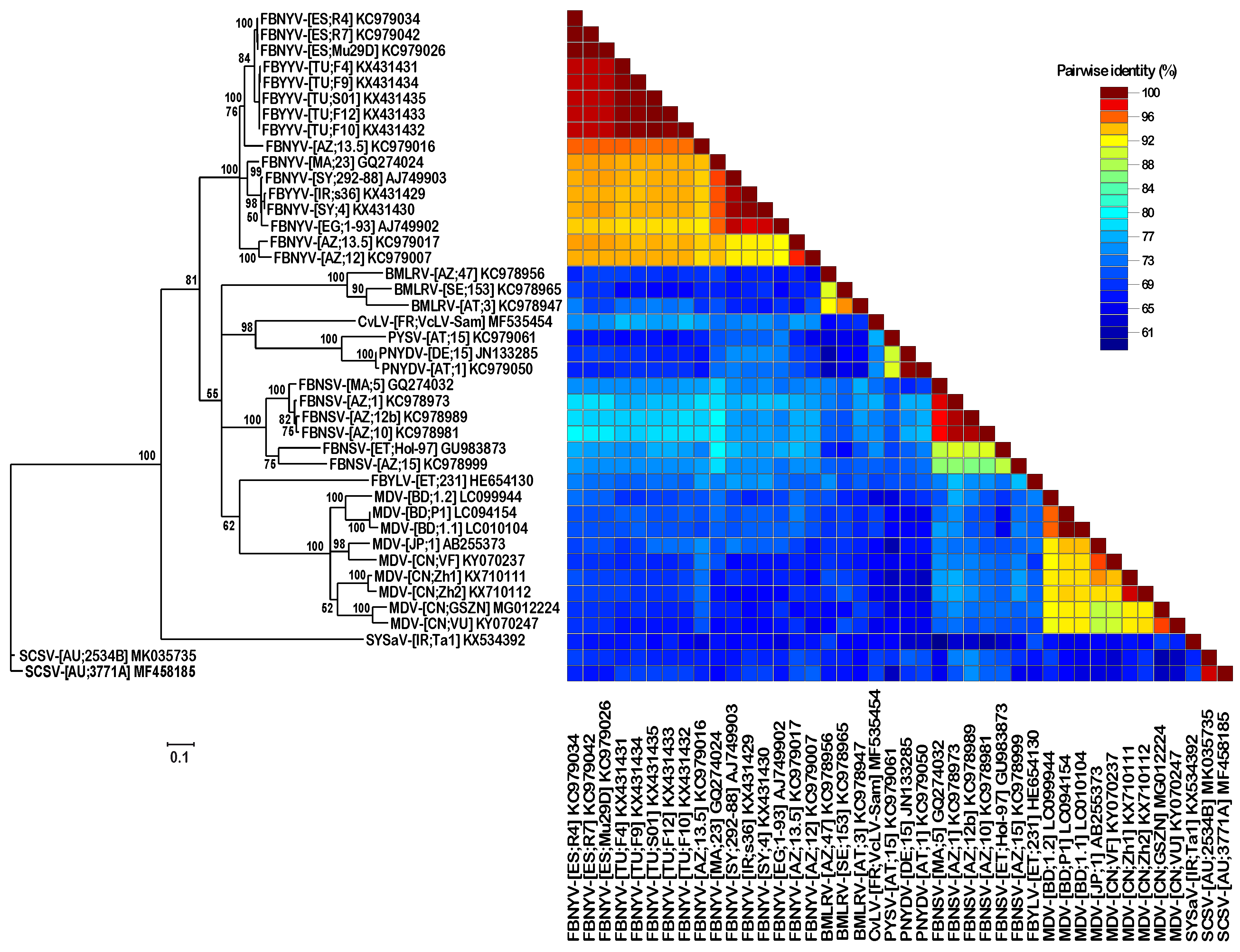
3.2. DNA Sequence Analysis and Comparison of Three Different SCSV Isolates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vetten, H.J.; Dale, J.L.; Grigoras, I.; Gronenborn, B.; Harding, R.; Randles, J.W.; Sano, Y.; Thomas, J.E.; Timchenko, T.; Yeh, H.-H. Family Nanoviridae. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.C., Lefkowitz, E.J., Eds.; Elsevier/Academic Press: London, UK, 2012; pp. 395–404. [Google Scholar]
- Vetten, H.J. Nanoviruses. In Encyclopedia of Virology, 3rd ed.; Mahy, B.W.J., Van Regenmortel, M., Eds.; Elsevier: Oxford, UK, 2008; Volume 3, pp. 385–391. [Google Scholar]
- Grigoras, I.; Ginzo, A.I.; Martin, D.P.; Varsani, A.; Romero, J.; Mammadov, A.; Huseynova, I.M.; Aliyev, J.A.; Kheyr-Pour, A.; Huss, H.; et al. Genome diversity and evidence of recombination and reassortment in nanoviruses from Europe. J. Gen. Virol. 2014, 95, 1178–1191. [Google Scholar] [CrossRef] [PubMed]
- Grigoras, I.; Timchenko, T.; Katul, L.; Grande-Pérez, A.; Vetten, H.J.; Gronenborn, B. Reconstitution of authentic nanovirus from multiple cloned DNAs. J. Virol. 2009, 83, 10778–10787. [Google Scholar] [CrossRef]
- Katul, L.; Maiss, E.; Morozov, S.Y.; Vetten, H.J. Analysis of six DNA components of the faba bean necrotic yellows virus genome and their structural affinity to related plant virus genomes. Virology 1997, 233, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Katul, L.; Timchenko, T.; Gronenborn, B.; Vetten, H.J. Ten distinct circular ssDNA components, four of which encode putative replication-associated proteins, are associated with the faba bean necrotic yellows virus genome. J. Gen. Virol. 1998, 79, 3101–3109. [Google Scholar] [CrossRef] [PubMed]
- Abraham, A.D.; Varrelmann, M.; Vetten, H.J. Three distinct nanoviruses, one of which represents a new species, infect faba bean in Ethiopia. Plant Dis. 2012, 96, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Sano, Y.; Wada, M.; Hashimoto, Y.; Matsumoto, T.; Kojima, M. Sequences of ten circular ssDNA components associated with the milk vetch dwarf virus genome. J. Gen. Virol. 1998, 79, 3111–3118. [Google Scholar] [CrossRef]
- Grigoras, I.; Gronenborn, B.; Vetten, H.J. First report of a nanovirus disease of pea in Germany. Plant Dis. 2010, 94, 642. [Google Scholar] [CrossRef]
- Boevink, P.; Chu, P.W.; Keese, P. Sequence of subterranean clover stunt virus DNA: Affinities with the geminiviruses. Virology 1995, 207, 354–361. [Google Scholar] [CrossRef]
- Chu, P.W.G.; Vetten, H.J. Subterranean clover stunt virus. In AAB Descriptions of Plant Viruses; DPV No. 396; 2003; Available online: http://www.dpvweb.net (established on 1 January 2006).
- Chu, P.W.; Helms, K. Novel virus-like particles containing circular single-stranded DNAs associated with subterranean clover stunt disease. Virology 1988, 167, 38–49. [Google Scholar] [CrossRef]
- Heydarnejad, J.; Kamali, M.; Massumi, H.; Kvarnheden, A.; Male, M.F.; Kraberger, S.; Stainton, D.; Martin, D.P.; Varsani, A. Identification of a nanovirus-alphasatellite complex in Sophora alopecuroides. Virus Res. 2017, 235, 24–32. [Google Scholar] [CrossRef]
- Gallet, R.; Kraberger, S.; Filloux, D.; Galzi, S.; Fontes, H.; Martin, D.P.; Varsani, A.; Roumagnac, P. Nanovirus-alphasatellite complex identified in Vicia cracca in the Rhone delta region of France. Arch. Virol. 2018, 163, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Timchenko, T.; de Kouchkovsky, F.; Katul, L.; David, C.; Vetten, H.J.; Gronenborn, B. A single rep protein initiates replication of multiple genome components of faba bean necrotic yellows virus, a single-stranded DNA virus of plants. J. Virol. 1999, 73, 10173–10182. [Google Scholar] [PubMed]
- Aronson, M.N.; Meyer, A.D.; Györgyey, J.; Katul, L.; Vetten, H.J.; Gronenborn, B.; Timchenko, T. Clink, a nanovirus-encoded protein, binds both pRB and SKP1. J. Virol. 2000, 74, 2967–2972. [Google Scholar] [CrossRef] [PubMed]
- Krapp, S.; Greiner, E.; Amin, B.; Sonnewald, U.; Krenz, B. The stress granule component G3BP is a novel interaction partner for the nuclear shuttle proteins of the nanovirus pea necrotic yellow dwarf virus and geminivirus abutilon mosaic virus. Virus Res. 2017, 227, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Lazarowitz, S.G.; Beachy, R.N. Viral movement proteins as probes for intracellular and intercellular trafficking in plants. Plant Cell Online 1999, 11, 535–548. [Google Scholar] [CrossRef]
- Wanitchakorn, R.; Hafner, G.J.; Harding, R.M.; Dale, J.L. Functional analysis of proteins encoded by banana bunchy top virus DNA-4 to -6. J. Gen. Virol. 2000, 81, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Krenz, B.; Schiessl, I.; Greiner, E.; Krapp, S. Analyses of pea necrotic yellow dwarf virus-encoded proteins. Virus Genes 2017, 53, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Grigoras, I.; Vetten, H.J.; Commandeur, U.; Ziebell, H.; Gronenborn, B.; Timchenko, T. Nanovirus DNA-N encodes a protein mandatory for aphid transmission. Virology 2018, 522, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Chu, P.W.; Boevink, P.; Surin, B.; Larkin, P.; Keese, P.; Waterhouse, P. Non-Geminated Single-Stranded DNA Plant Viruses; Volume Viruses and Viroids; Pergamon Press: Tarrytown, NY, USA, 1995; pp. 311–341. [Google Scholar]
- Grylls, N.E.; Butler, F.C. Subterranean clover stunt, a virus disease of pasture legumes. J. Aust. Agric. Res. 1959, 10, 145–159. [Google Scholar] [CrossRef]
- Grylls, N.E.; Peak, J.W. A virus complex of subterranean clover. Aust. J. Agric. Res. 1969, 20, 37–45. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Morgan, D.J.; Havenstein, D.E. The ecology of Aphis craccivora Koch and subterranean clover stunt virus I. The phenology of aphis populations and the epidemiology of virus in pastures in south-east Australia. J. Appl. Ecol. 1971, 8, 699–721. [Google Scholar] [CrossRef]
- Johnson, B. Studies on the dispersal by upper winds of Aphis craccivora Koch in New South Wales. Proc. Linn. Soc. N. S. W. 1957, 82, 191–198. [Google Scholar]
- Johnstone, G.R.; McLean, G.D. Virus diseases of subterranean clover. Ann. Appl. Biol. 1987, 110, 421–440. [Google Scholar] [CrossRef]
- Smith, P.R. A disease of french beans (Phaseolus vulgaris L.) caused by subterranean clover stunt virus. Aust. J. Agric. Res. 1966, 17, 875–883. [Google Scholar] [CrossRef]
- Chu, P.W.; Keese, P.; Qiu, B.S.; Waterhouse, P.M.; Gerlach, W.L. Putative full-length clones of the genomic DNA segments of subterranean clover stunt virus and identification of the segment coding for the viral coat protein. Virus Res. 1993, 27, 161–171. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Muhire, B.M.; Varsani, A.; Martin, D.P. SDT: A virus classification tool based on pairwise sequence alignment and identity calculation. PLoS ONE 2014, 9, e108277. [Google Scholar] [CrossRef]
- Guindon, S.; Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef]
- Timchenko, T.; Katul, L.; Sano, Y.; de Kouchkovsky, F.; Vetten, H.J.; Gronenborn, B. The master rep concept in nanovirus replication: Identification of missing genome components and potential for natural genetic reassortment. Virology 2000, 274, 189–195. [Google Scholar] [CrossRef]
- Grigoras, I.; Timchenko, T.; Grande-Perez, A.; Katul, L.; Vetten, H.J.; Gronenborn, B. High variability and rapid evolution of a nanovirus. J. Virol. 2010, 84, 9105–9117. [Google Scholar] [CrossRef] [PubMed]
- Briddon, R.W.; Martin, D.P.; Roumagnac, P.; Navas-Castillo, J.; Fiallo-Olive, E.; Moriones, E.; Lett, J.M.; Zerbini, F.M.; Varsani, A. Alphasatellitidae: A new family with two subfamilies for the classification of geminivirus- and nanovirus-associated alphasatellites. Arch. Virol. 2018, 163, 2587–2600. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.P.; Biagini, P.; Lefeuvre, P.; Golden, M.; Roumagnac, P.; Varsani, A. Recombination in eukaryotic single stranded DNA viruses. Viruses 2011, 3, 1699–1738. [Google Scholar] [CrossRef]
- Hyder, M.Z.; Shah, S.H.; Hameed, S.; Naqvi, S.M. Evidence of recombination in the banana bunchy top virus genome. Infect. Genet. Evol. 2011, 11, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Stainton, D.; Kraberger, S.; Walters, M.; Wiltshire, E.J.; Rosario, K.; Halafihi, M.; Lolohea, S.; Katoa, I.; Faitua, T.H.; Aholelei, W.; et al. Evidence of inter-component recombination, intra-component recombination and reassortment in banana bunchy top virus. J. Gen. Virol. 2012, 93, 1103–1119. [Google Scholar] [CrossRef] [PubMed]
- Kraberger, S.; Kumari, S.G.; Najar, A.; Stainton, D.; Martin, D.P.; Varsani, A. Molecular characterization of faba bean necrotic yellows viruses in Tunisia. Arch. Virol. 2018, 163, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Gronenborn, B.; Randles, J.W.; Knierim, D.; Barriere, Q.; Vetten, H.J.; Warthmann, N.; Cornu, D.; Sileye, T.; Winter, S.; Timchenko, T. Analysis of DNAs associated with coconut foliar decay disease implicates a unique single-stranded DNA virus representing a new taxon. Sci. Rep. 2018, 8, 5698. [Google Scholar] [CrossRef]
- Sicard, A.; Yvon, M.; Timchenko, T.; Gronenborn, B.; Michalakis, Y.; Gutierrez, S.; Blanc, S. Gene copy number is differentially regulated in a multipartite virus. Nat. Commun. 2013, 4, 2248. [Google Scholar] [CrossRef]
- Grylls, N.E.; Butler, F.C. An aphid transmitted virus affecting subterranean clover. J. Aust. Inst. Agric. Sci. 1956, 22, 73–74. [Google Scholar]

| DNA | Virus Isolate | ||||
|---|---|---|---|---|---|
| SCSV-[AU;3771A] | SCSV-[AU;2534B] | SCSV-[AU;F] | SCSV-[AU;F*] 1 | ||
| Genome components | DNA-R | MF458178 | MK035728 | AJ290434 | |
| DNA-S | MF458179 | MK035729 | U16734 | ||
| DNA-C | MF458180 | MK035730 | U16732 | ||
| DNA-M DNA-M.2 | MF458181 2 MK291271 | MK035731 | U16730 | ||
| DNA-N | MF458182 | MK035732 | U16733 | MK291272 | |
| DNA-U1 | MF458183 | MK035733 | U16736 | ||
| DNA-U2 | MF458184 | MK035734 | - | MF458186 | |
| DNA-U4 DNA-U4.2 | MF458185 | MK035735 3 MK035736 | - | - | |
| Alphasatellites | SCSA 1 | MK291268 | MK035737 | U16731 | |
| SCSA 2 | MK291269 | MK035738 | U16735 | ||
| SYSA 3 | MK291270 | - | - | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knierim, D.; Barrière, Q.; Grigoras, I.; Winter, S.; Vetten, H.-J.; Schwinghamer, M.; Thomas, J.; Chu, P.; Gronenborn, B.; Timchenko, T. Subterranean Clover Stunt Virus Revisited: Detection of Two Missing Genome Components. Viruses 2019, 11, 138. https://doi.org/10.3390/v11020138
Knierim D, Barrière Q, Grigoras I, Winter S, Vetten H-J, Schwinghamer M, Thomas J, Chu P, Gronenborn B, Timchenko T. Subterranean Clover Stunt Virus Revisited: Detection of Two Missing Genome Components. Viruses. 2019; 11(2):138. https://doi.org/10.3390/v11020138
Chicago/Turabian StyleKnierim, Dennis, Quentin Barrière, Ioana Grigoras, Stephan Winter, Heinrich-Josef Vetten, Mark Schwinghamer, John Thomas, Paul Chu, Bruno Gronenborn, and Tatiana Timchenko. 2019. "Subterranean Clover Stunt Virus Revisited: Detection of Two Missing Genome Components" Viruses 11, no. 2: 138. https://doi.org/10.3390/v11020138
APA StyleKnierim, D., Barrière, Q., Grigoras, I., Winter, S., Vetten, H.-J., Schwinghamer, M., Thomas, J., Chu, P., Gronenborn, B., & Timchenko, T. (2019). Subterranean Clover Stunt Virus Revisited: Detection of Two Missing Genome Components. Viruses, 11(2), 138. https://doi.org/10.3390/v11020138





