Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Virus Stocks
2.2. Generation of Mutated Viruses and Cloning
2.3. Virus Titrations and Viral Replication Kinetic
2.4. Fluorescence Microscopy and Colocalization Analysis
2.5. Protein Expression Levels by Flow-Cytometry
2.6. Analysis of Virus Sedimentation by Ultracentrifugation
2.7. Western Blot
2.8. Transmission Electron Microscopy
2.9. Specific Infectivity
2.10. Multiple Sequences Alignment of NS3 from Orbivirus Genus
2.11. Statistical Analyzes and Software
3. Results
3.1. NS3 Proteins of Members of the Orbivirus Genus Share Two Conserved Polybasic Motifs
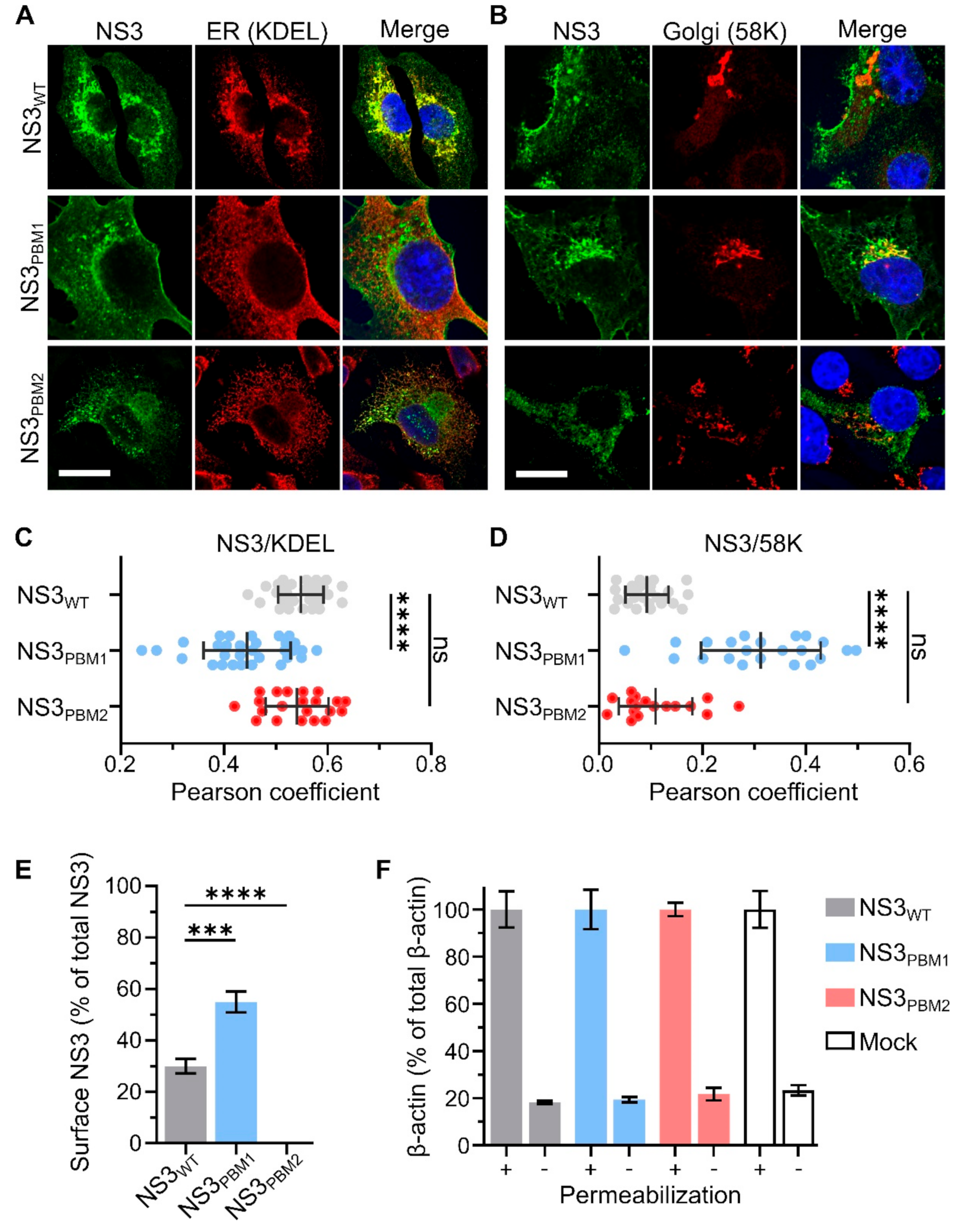
3.2. PBM1 and PBM2 Are Required for the Cellular Trafficking of NS3 and for Viral Particle Budding
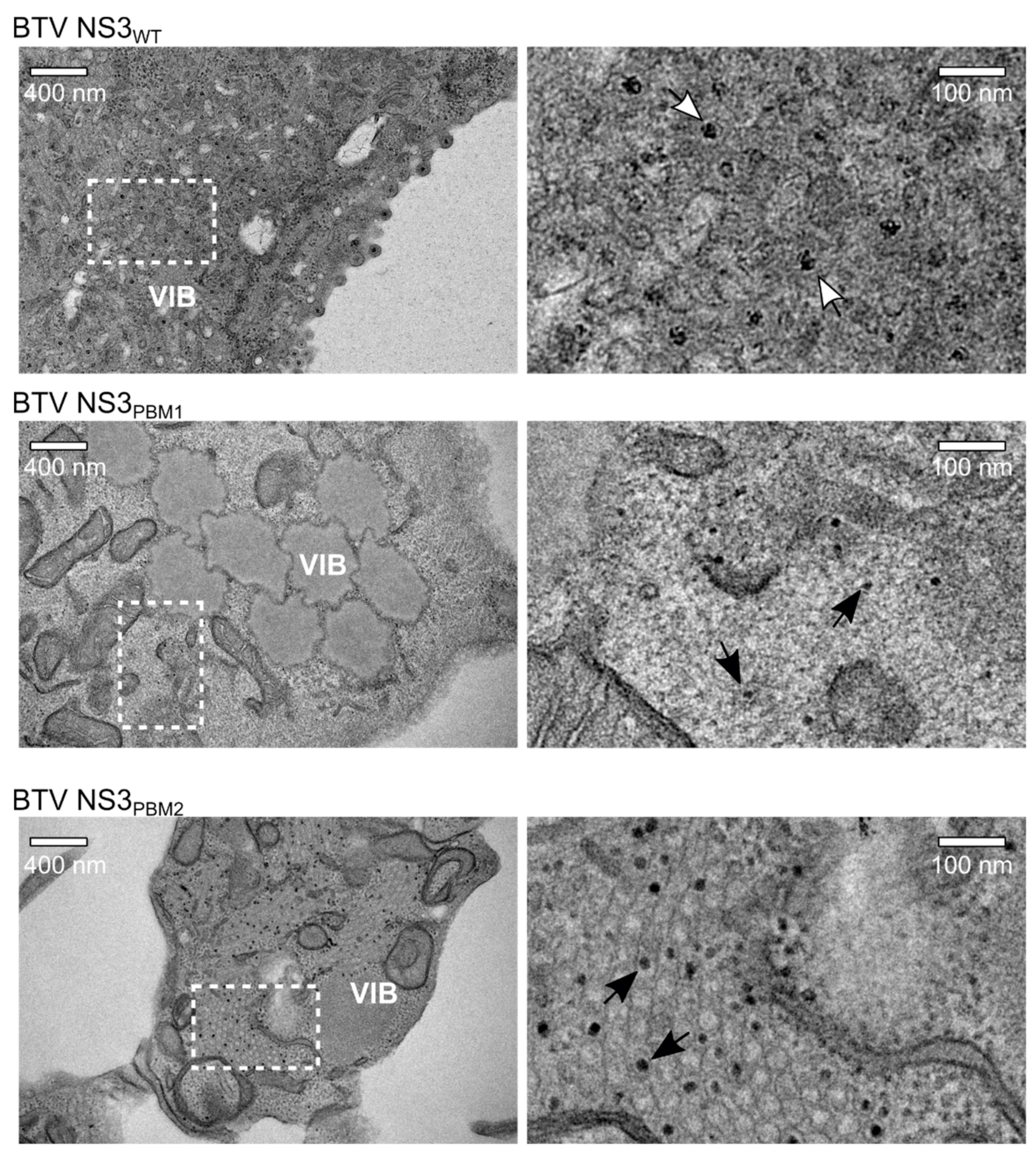
3.3. NS3 Trafficking Defect is Associated with Release of Non-Mature Core Particles
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saegerman, C.; Berkvens, D.; Mellor, P.S. Bluetongue Epidemiology in the European Union. Emerg. Infect. Dis. 2008, 14, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Drolet, B.S.; Reister, L.M.; Rigg, T.D.; Nol, P.; Podell, B.K.; Mecham, J.O.; VerCauteren, K.C.; van Rijn, P.A.; Wilson, W.C.; Bowen, R.A. Experimental infection of white-tailed deer (Odocoileus virginianus) with Northern European bluetongue virus serotype 8. Vet. Microbiol. 2013, 166, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Rojas, J.M.; Rodríguez-Martín, D.; Martín, V.; Sevilla, N. Diagnosing bluetongue virus in domestic ruminants: Current perspectives. Vet. Med. (Auckl.) 2019, 10, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Abera, T.; Bitew, M.; Gebre, D.; Mamo, Y.; Deneke, Y.; Nandi, S. Bluetongue disease in small ruminants in south western Ethiopia: Cross-sectional sero-epidemiological study. BMC Res. Notes 2018, 11, 112. [Google Scholar] [CrossRef]
- Zientara, S.; Sailleau, C.; Viarouge, C.; Höper, D.; Beer, M.; Jenckel, M.; Hoffmann, B.; Romey, A.; Bakkali-Kassimi, L.; Fablet, A.; et al. Novel Bluetongue Virus in Goats, Corsica, France, 2014. Emerg. Infect. Dis. 2014, 20, 2123–2125. [Google Scholar] [CrossRef]
- Roy, P. Bluetongue virus structure and assembly. Curr. Opin. Virol. 2017, 24, 115–123. [Google Scholar] [CrossRef]
- Mohl, B.-P.; Roy, P. Bluetongue Virus Capsid Assembly and Maturation. Viruses 2014, 6, 3250–3270. [Google Scholar] [CrossRef]
- Stewart, M.; Hardy, A.; Barry, G.; Pinto, R.M.; Caporale, M.; Melzi, E.; Hughes, J.; Taggart, A.; Janowicz, A.; Varela, M.; et al. Characterization of a second open reading frame in genome segment 10 of bluetongue virus. J. Gen. Virol. 2015, 96, 3280–3293. [Google Scholar] [CrossRef]
- Hyatt, A.D.; Zhao, Y.; Roy, P. Release of Bluetongue Virus-like Particles from Insect Cells is Mediated by BTV Nonstructural Protein NS3/NS3A. Virology 1993, 193, 592–603. [Google Scholar] [CrossRef]
- Chacko, N.; Mohanty, N.N.; Biswas, S.K.; Chand, K.; Yogisharadhya, R.; Pandey, A.B.; Mondal, B.; Shivachandra, S.B. A coiled-coil motif in non-structural protein 3 (NS3) of bluetongue virus forms an oligomer. Virus Genes 2015, 51, 244–251. [Google Scholar] [CrossRef]
- Han, Z.; Harty, R.N. The NS3 Protein of Bluetongue Virus Exhibits Viroporin-like Properties. J. Biol. Chem. 2004, 279, 43092–43097. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, B.; Celma, C.C.; Roy, P. Influence of Cellular Trafficking Pathway on Bluetongue Virus Infection in Ovine Cells. Viruses 2015, 7, 2378–2403. [Google Scholar] [CrossRef] [PubMed]
- Celma, C.C.P.; Roy, P. A Viral Nonstructural Protein Regulates Bluetongue Virus Trafficking and Release. J. Virol. 2009, 83, 6806–6816. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, B.; Roy, P. Cellular phosphoinositides and the maturation of bluetongue virus, a non-enveloped capsid virus. Virol. J. 2013, 10, 73. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wirblich, C.; Bhattacharya, B.; Roy, P. Nonstructural Protein 3 of Bluetongue Virus Assists Virus Release by Recruiting ESCRT-I Protein Tsg101. J. Virol. 2006, 80, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Beaton, A.R.; Rodriguez, J.; Reddy, Y.K.; Roy, P. The membrane trafficking protein calpactin forms a complex with bluetongue virus protein NS3 and mediates virus release. Proc. Natl. Acad. Sci. USA 2002, 99, 13154–13159. [Google Scholar] [CrossRef]
- Bhattacharya, B.; Roy, P. Bluetongue Virus Outer Capsid Protein VP5 Interacts with Membrane Lipid Rafts via a SNARE Domain. J. Virol. 2008, 82, 10600–10612. [Google Scholar] [CrossRef]
- Hyatt, A.D.; Eaton, B.T.; Brookes, S.M. The release of bluetongue virus from infected cells and their superinfection by progeny virus. Virology 1989, 173, 21–34. [Google Scholar] [CrossRef]
- Hyatt, A.D.; Gould, A.R.; Coupar, B.; Eaton, B.T. Localization of the non-structural protein NS3 in bluetongue virus-infected cells. J. Gen. Virol. 1991, 72, 2263–2267. [Google Scholar] [CrossRef]
- Kundlacz, C.; Pourcelot, M.; Fablet, A.; Moraes, R.A.D.S.; Léger, T.; Morlet, B.; Viarouge, C.; Sailleau, C.; Turpaud, M.; Gorlier, A.; et al. Novel Function of Bluetongue Virus NS3 Protein in Regulation of the MAPK/ERK Signaling Pathway. J. Virol. 2019. [Google Scholar] [CrossRef]
- Sato, M.; Tanaka, H.; Yamada, T.; Yamamoto, N. Persistent infection of BHK21/WI-2 cells with rubella virus and characterization of rubella variants. Arch. Virol. 1977, 54, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Boyce, M.; Celma, C.C.P.; Roy, P. Development of Reverse Genetics Systems for Bluetongue Virus: Recovery of Infectious Virus from Synthetic RNA Transcripts. J. Virol. 2008, 82, 8339–8348. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Okonechnikov, K.; Golosova, O.; Fursov, M. Unipro UGENE: A unified bioinformatics toolkit. Bioinformatics 2012, 28, 1166–1167. [Google Scholar] [CrossRef] [PubMed]
- De Chaumont, F.; Dallongeville, S.; Chenouard, N.; Hervé, N.; Pop, S.; Provoost, T.; Meas-Yedid, V.; Pankajakshan, P.; Lecomte, T.; Le Montagner, Y.; et al. Icy: An open bioimage informatics platform for extended reproducible research. Nat. Methods 2012, 9, 690–696. [Google Scholar] [CrossRef]
- Bansal, O.B.; Stokes, A.; Bansal, A.; Bishop, D.; Roy, P. Membrane Organization of Bluetongue Virus Nonstructural Glycoprotein NS3. J. Virol. 1998, 72, 3362–3369. [Google Scholar]
- Celma, C.C.P.; Roy, P. Interaction of Calpactin Light Chain (S100A10/p11) and a Viral NS Protein Is Essential for Intracellular Trafficking of Nonenveloped Bluetongue Virus. J. Virol. 2011, 85, 4783–4791. [Google Scholar] [CrossRef]
- Hamard-Peron, E.; Juillard, F.; Saad, J.S.; Roy, C.; Roingeard, P.; Summers, M.F.; Darlix, J.-L.; Picart, C.; Muriaux, D. Targeting of murine leukemia virus gag to the plasma membrane is mediated by PI(4,5)P2/PS and a polybasic region in the matrix. J. Virol. 2010, 84, 503–515. [Google Scholar] [CrossRef]
- Parmar, H.B.; Barry, C.; Kai, F.; Duncan, R. Golgi complex–plasma membrane trafficking directed by an autonomous, tribasic Golgi export signal. Mol. Biol. Cell 2014, 25, 866–878. [Google Scholar] [CrossRef]
- Gao, C.; Cai, Y.; Wang, Y.; Kang, B.-H.; Aniento, F.; Robinson, D.G.; Jiang, L. Retention mechanisms for ER and Golgi membrane proteins. Trends Plant Sci. 2014, 19, 508–515. [Google Scholar] [CrossRef]
- Ma, T.; Li, B.; Wang, R.; Lau, P.K.; Huang, Y.; Jiang, L.; Schekman, R.; Guo, Y. A mechanism for differential sorting of the planar cell polarity proteins Frizzled6 and Vangl2 at the trans-Golgi network. J. Biol. Chem. 2018, 293, 8410–8427. [Google Scholar] [CrossRef] [PubMed]
- Omrane, M.; Camara, A.S.; Taveneau, C.; Benzoubir, N.; Tubiana, T.; Yu, J.; Guérois, R.; Samuel, D.; Goud, B.; Poüs, C.; et al. Septin 9 has Two Polybasic Domains Critical to Septin Filament Assembly and Golgi Integrity. iScience 2019, 13, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, I.C.R.; Shikata, H.; Zourelidou, M.; Heilmann, M.; Heilmann, I.; Schwechheimer, C. Phospholipid composition and a polybasic motif determine D6 PROTEIN KINASE polar association with the plasma membrane and tropic responses. Development 2016, 143, 4687–4700. [Google Scholar] [CrossRef] [PubMed]
- Parmar, H.B.; Barry, C.; Duncan, R. Polybasic trafficking signal mediates golgi export, ER retention or ER export and retrieval based on membrane-proximity. PLoS ONE 2014, 9, e94194. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yan, C.; Xu, C.; Chua, B.T.; Li, P.; Chen, F.-J. Polybasic RKKR motif in the linker region of lipid droplet (LD)–associated protein CIDEC inhibits LD fusion activity by interacting with acidic phospholipids. J. Biol. Chem. 2018, 293, 19330–19343. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, H.C.; Matreyek, K.A.; Choi, D.Y.; Filone, C.M.; Young, S.; Lee, B. Polybasic KKR Motif in the Cytoplasmic Tail of Nipah Virus Fusion Protein Modulates Membrane Fusion by Inside-Out Signaling. J. Virol. 2007, 81, 4520–4532. [Google Scholar] [CrossRef]
- Mertens, P.P.C.; Burroughs, J.N.; Anderson, J. Purification and properties of virus particles, infectious subviral particles, and cores of bluetongue virus serotypes 1 and 4. Virology 1987, 157, 375–386. [Google Scholar] [CrossRef]
- Martin, S.A.; Zweerink, H.J. Isolation and characterization of two types of bluetongue virus particles. Virology 1972, 50, 495–506. [Google Scholar] [CrossRef]



| Orbiviruses | Genbank Accession Number |
|---|---|
| Bluetongue virus 1 | ALI51183.1 |
| Equine encephalosis virus | AEP95960.1 |
| Yunnan virus | YP_443934.1 |
| Peruvian horse sickness virus | YP_460047.1 |
| Umatilla virus | YP_009047250.1 |
| Great Island virus | YP_003896068.1 |
| St Croix River virus | YP_052951.1 |
| Eubenangee virus | AFH41518.1 |
| Palyam virus | ALW83187.1 |
| Changuinola virus | AIV43215.1 |
| Warrego virus | AIT55722.1 |
| Wallal virus | AIT55712.1 |
| Orungo virus | AFX73397.1 |
| Lebombo virus | AFX73386.1 |
| Chobar Gorge virus | YP_009158911.1 |
| Chenuda virus | YP_009158899.1 |
| Corriparta virus | AGT51064.1 |
| Wad Medani virus | YP_009158892.1 |
| African horse sickness virus 6 | AKP19850.1 |
| Epizootic hemorrhagic disease virus | AAQ62564.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Labadie, T.; Jegouic, S.; Roy, P. Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs. Viruses 2019, 11, 1107. https://doi.org/10.3390/v11121107
Labadie T, Jegouic S, Roy P. Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs. Viruses. 2019; 11(12):1107. https://doi.org/10.3390/v11121107
Chicago/Turabian StyleLabadie, Thomas, Sophie Jegouic, and Polly Roy. 2019. "Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs" Viruses 11, no. 12: 1107. https://doi.org/10.3390/v11121107
APA StyleLabadie, T., Jegouic, S., & Roy, P. (2019). Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs. Viruses, 11(12), 1107. https://doi.org/10.3390/v11121107






