Host Cell Calpains Can Cleave Structural Proteins from the Enterovirus Polyprotein
Abstract
1. Introduction
2. Methods
2.1. Reagents
2.2. Cells
2.3. Production of P1 and P1-2A* Constructs
2.4. Calpain In Vitro Cleavage Assays
2.5. In Vitro Cleavage Assay with Viral Proteases
2.6. Infection Assay in Cells
2.7. Immunofluorescence Labeling and Microscopy
2.8. Western Blot
2.9. Cleavage Assay with the Peptide
2.10. Mass Spectrometry
2.11. Statistical Testing
3. Results
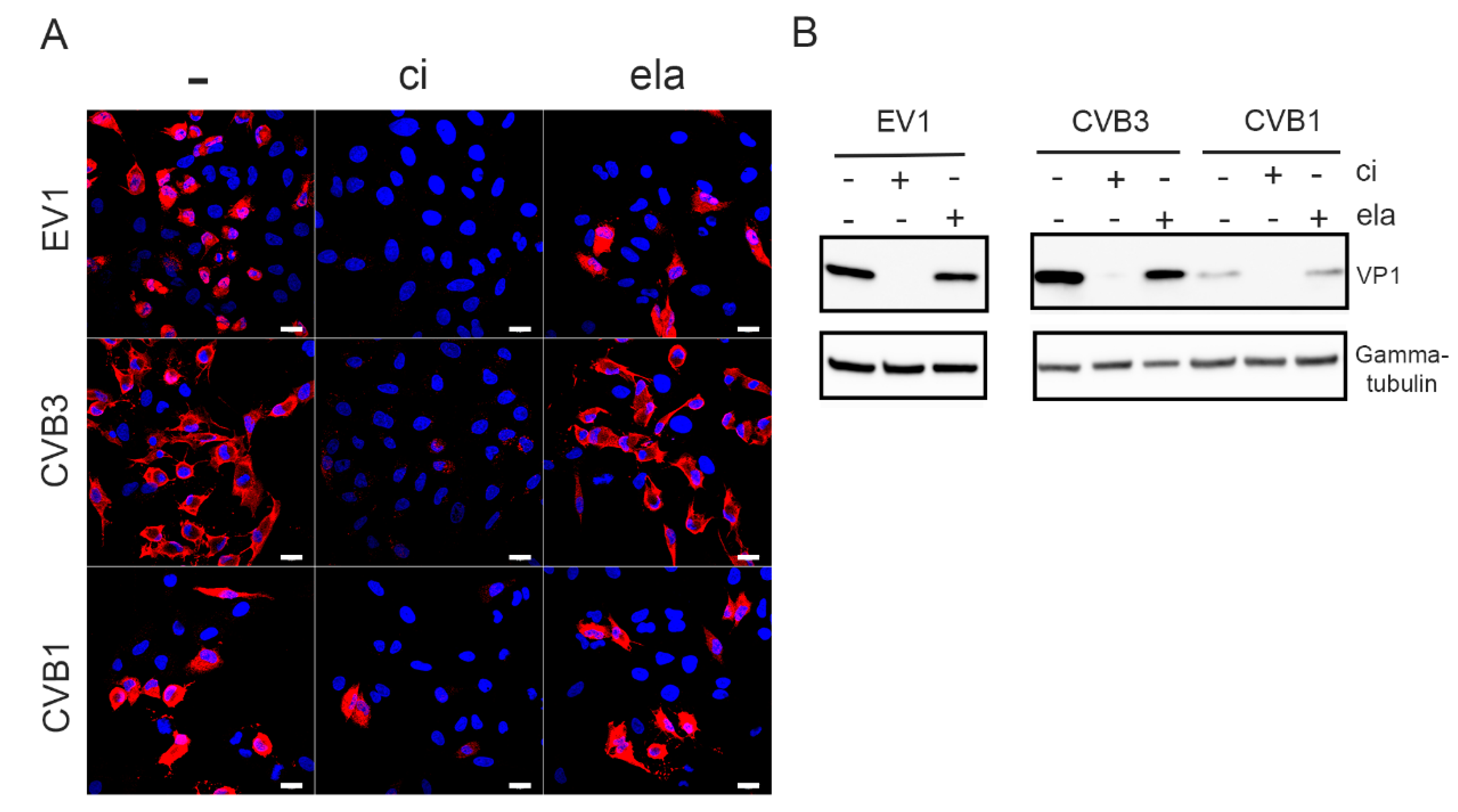
3.1. Calpain Inhibitor Prevents the Enterovirus Infection Whereas 2A Inhibitor Does Not
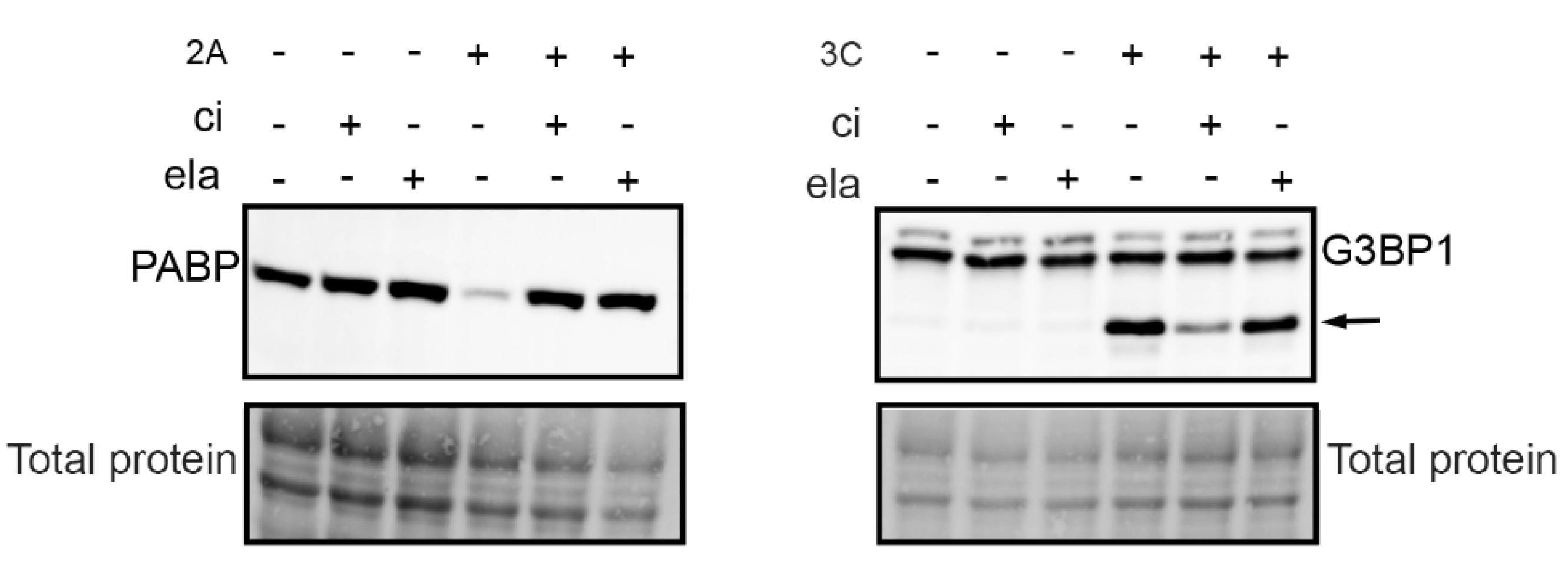
3.2. Calpain Inhibitor Cross-Reacts with Viral Proteases 2A and 3C
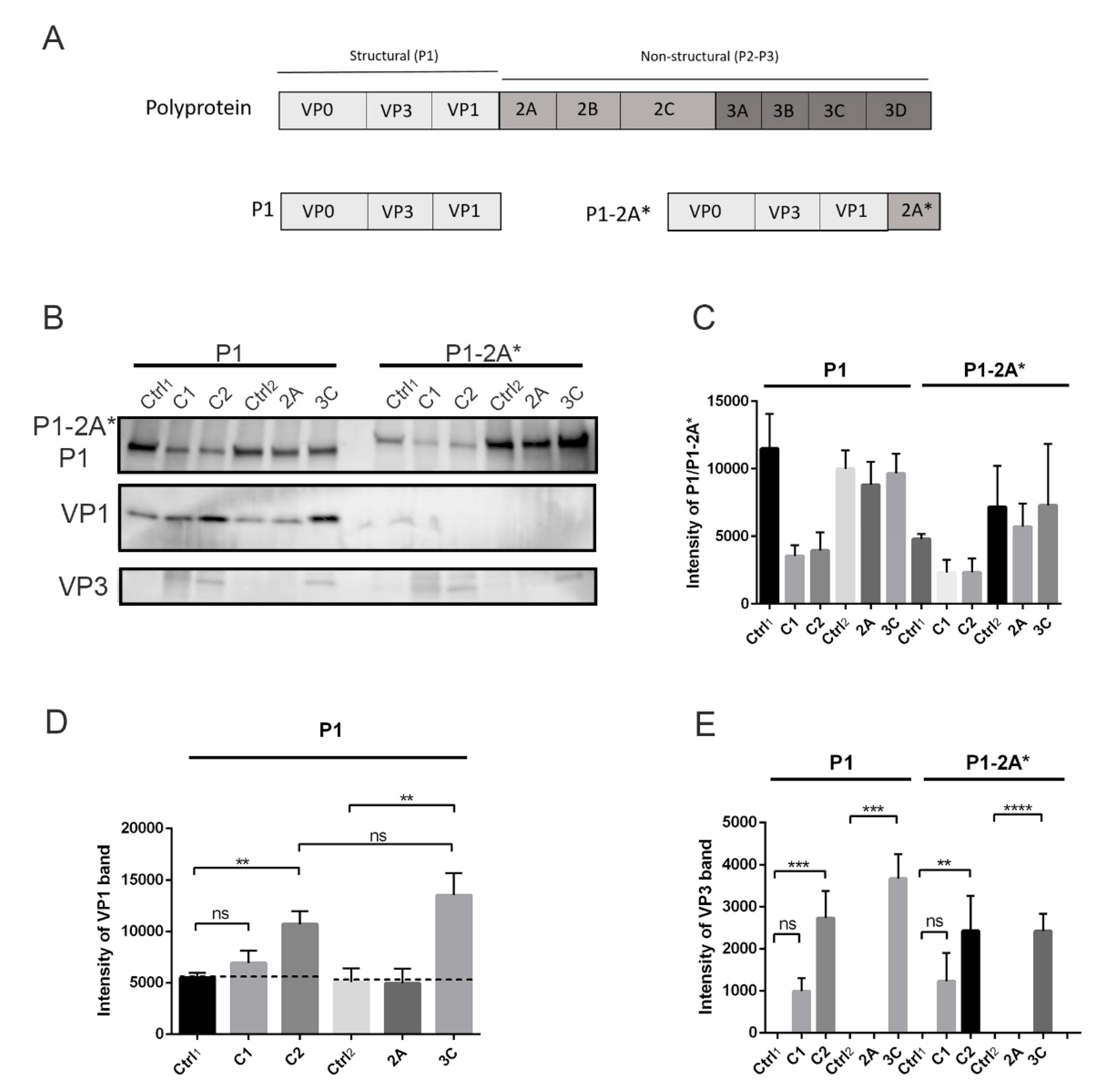
3.3. Calpains Can Cleave P1 into VP1 and VP3 In Vitro but Cannot Release P1 from the Polyprotein
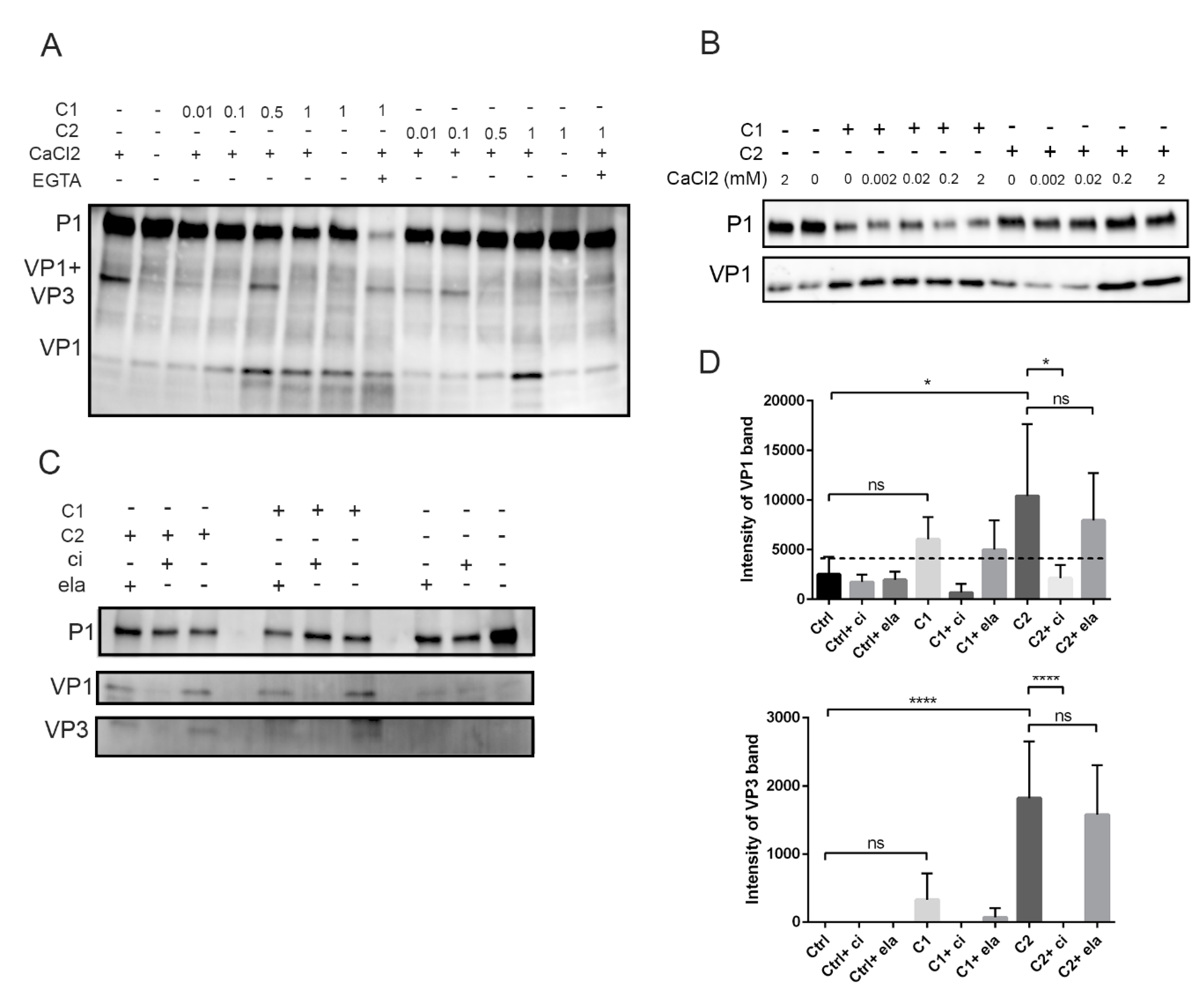
3.4. Calpain Activity is Concentration and Calcium Dependent and Can Be Prevented with Calpain Inhibitor In Vitro
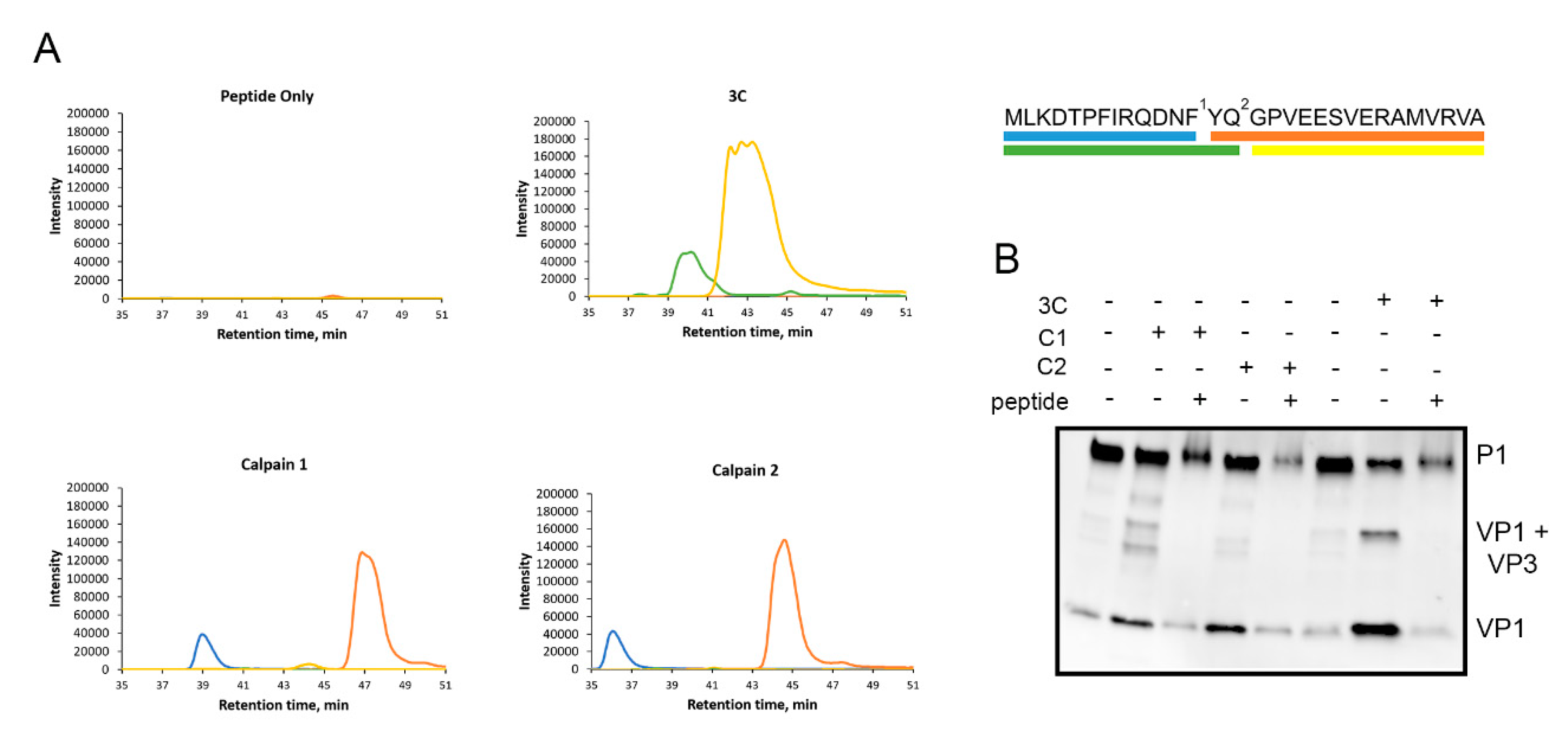
3.5. Calpains Cleave Specifically at the VP1-VP3 Cleavage Site
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tapparel, C.; Siegrist, F.; Petty, T.J.; Kaiser, L. Picornavirus and Enterovirus Diversity with Associated Human Diseases. Infect. Genet. Evol. 2013, 14, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Gaaloul, I.; Riabi, S.; Harrath, R.; Hunter, T.; Hamda, K.B.; Ghzala, A.B.; Huber, S.; Aouni, M. Coxsackievirus B Detection in Cases of Myocarditis, Myopericarditis, Pericarditis and Dilated Cardiomyopathy in Hospitalized Patients. Mol. Med. Rep. 2014, 10, 2811–2818. [Google Scholar] [CrossRef] [PubMed]
- Bouin, A.; Gretteau, P.A.; Wehbe, M.; Renois, F.; N’Guyen, Y.; Leveque, N.; Vu, M.N.; Tracy, S.; Chapman, N.M.; Bruneval, P.; et al. Enterovirus Persistence in Cardiac Cells of Patients with Idiopathic Dilated Cardiomyopathy is Linked to 5’ Terminal Genomic RNA-Deleted Viral Populations with Viral-Encoded Proteinase Activities. Circulation 2019, 139, 2326–2338. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.A.; Zheng, X.T.; Harris, M.U.; Cadichon, S.B.; Melin-Aldana, H.; Khetsuriani, N.; Oberste, M.S.; Shulman, S.T. Outbreak of Life-Threatening Coxsackievirus B1 Myocarditis in Neonates. Clin. Infect. Dis. 2009, 49, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Oikarinen, S.; Tauriainen, S.; Hober, D.; Lucas, B.; Vazeou, A.; Sioofy-Khojine, A.; Bozas, E.; Muir, P.; Honkanen, H.; Ilonen, J.; et al. Virus Antibody Survey in Different European Populations Indicates Risk Association between Coxsackievirus B1 and Type 1 Diabetes. Am. Diabetes Assoc. 2014, 63, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Sioofy-Khojine, A.B.; Lehtonen, J.; Nurminen, N.; Laitinen, O.H.; Oikarinen, S.; Huhtala, H.; Pakkanen, O.; Ruokoranta, T.; Hankaniemi, M.M.; Toppari, J.; et al. Coxsackievirus B1 Infections are Associated with the Initiation of Insulin-Driven Autoimmunity that Progresses to Type 1 Diabetes. Diabetologia 2018, 61, 1193–1202. [Google Scholar] [CrossRef]
- Laitinen, O.H.; Honkanen, H.; Pakkanen, O.; Oikarinen, S.; Hankaniemi, M.M.; Huhtala, H.; Ruokoranta, T.; Lecouturier, V.; Andre, P.; Harju, R.; et al. Coxsackievirus B1 is Associated with Induction of Beta-Cell Autoimmunity that Portends Type 1 Diabetes. Am. Diabetes Assoc. 2014, 63, 446–455. [Google Scholar] [CrossRef]
- Upla, P.; Marjomaki, V.; Nissinen, L.; Nylund, C.; Waris, M.; Hyypia, T.; Heino, J. Calpain 1 and 2 are Required for RNA Replication of Echovirus 1. J. Virol. 2008, 82, 1581–1590. [Google Scholar] [CrossRef]
- Rintanen, N.; Karjalainen, M.; Alanko, J.; Paavolainen, L.; Maki, A.; Nissinen, L.; Lehkonen, M.; Kallio, K.; Cheng, R.H.; Upla, P.; et al. Calpains Promote α2β1 Integrin Turnover in Nonrecycling Integrin Pathway. Mol. Biol. Cell 2012, 23, 448–463. [Google Scholar] [CrossRef]
- Laitinen, O.H.; Svedin, E.; Kapell, S.; Nurminen, A.; Hytonen, V.P.; Flodstrom-Tullberg, M. Enteroviral Proteases: Structure, Host Interactions and Pathogenicity. Rev. Med. Virol. 2016, 26, 251–267. [Google Scholar] [CrossRef]
- Palmenberg, A.C. Proteolytic Processing of Picornaviral Polyprotein. Annu. Rev. Microbiol. 1990, 44, 603–623. [Google Scholar] [CrossRef] [PubMed]
- Sommergruber, W.; Zorn, M.; Blaas, D.; Fessl, F.; Volkmann, P.; Maurer-Fogy, I.; Pallai, P.; Merluzzi, V.; Matteo, M.; Skern, T. Polypeptide 2A of Human Rhinovirus Type 2: Identification as a Protease and Characterization by Mutational Analysis. Virology 1989, 169, 68–77. [Google Scholar] [CrossRef]
- Toyoda, H.; Nicklin, M.J.; Murray, M.G.; Anderson, C.W.; Dunn, J.J.; Studier, F.W.; Wimmer, E. A Second Virus-Encoded Proteinase Involved in Proteolytic Processing of Poliovirus Polyprotein. Cell 1986, 45, 761–770. [Google Scholar] [CrossRef]
- Nicklin, M.J.; Krausslich, H.G.; Toyoda, H.; Dunn, J.J.; Wimmer, E. Poliovirus Polypeptide Precursors: Expression in Vitro and Processing by Exogenous 3C and 2A Proteinases. Proc. Natl. Acad. Sci. USA 1987, 84, 4002–4006. [Google Scholar] [CrossRef] [PubMed]
- Ypma-Wong, M.F.; Dewalt, P.G.; Johnson, V.H.; Lamb, J.G.; Semler, B.L. Protein 3CD is the Major Poliovirus Proteinase Responsible for Cleavage of the P1 Capsid Precursor. Virology 1988, 166, 265–270. [Google Scholar] [CrossRef]
- Sun, D.; Chen, S.; Cheng, A.; Wang, M. Roles of the Picornaviral 3C Proteinase in the Viral Life Cycle and Host Cells. Viruses 2016, 8, 82. [Google Scholar] [CrossRef]
- Seipelt, J.; Guarne, A.; Bergmann, E.; James, M.; Sommergruber, W.; Fita, I.; Skern, T. The Structures of Picornaviral Proteinases. Virus Res. 1999, 62, 159–168. [Google Scholar] [CrossRef]
- Ono, Y.; Sorimachi, H. Calpains: An Elaborate Proteolytic System. Biochim. Biophys. Acta 2012, 1824, 224–236. [Google Scholar] [CrossRef]
- Moldoveanu, T.; Campbell, R.L.; Cuerrier, D.; Davies, P.L. Crystal Structures of Calpain-E64 and -Leupeptin Inhibitor Complexes Reveal Mobile Loops Gating the Active Site. J. Mol. Biol. 2004, 343, 1313–1326. [Google Scholar] [CrossRef]
- Goll, D.E.; Thompson, V.F.; Li, H.; Wei, W.; Cong, J. The Calpain System. Physiol. Rev. 2003, 83, 731–801. [Google Scholar] [CrossRef]
- Sakai, K.; Akanuma, H.; Imahori, K.; Kawashima, S. A Unique Specificity of a Calcium Activated Neutral Protease Indicated in Histone Hydrolysis. J. Biochem. 1987, 101, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Stabach, P.R.; Cianci, C.D.; Glantz, S.B.; Zhang, Z.; Morrow, J.S. Site-Directed Mutagenesis of Alpha II Spectrin at Codon 1175 Modulates its Mu-Calpain Susceptibility. Biochemistry 1997, 36, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Bozym, R.A.; Morosky, S.A.; Kim, K.S.; Cherry, S.; Coyne, C.B. Release of Intracellular Calcium Stores Facilitates Coxsackievirus Entry into Polarized Endothelial Cells. PLoS Pathog. 2010, 6. [Google Scholar] [CrossRef] [PubMed]
- Bozym, R.A.; Patel, K.; White, C.; Cheung, K.H.; Bergelson, J.M.; Morosky, S.A.; Coyne, C.B. Calcium Signals and Calpain-Dependent Necrosis are Essential for Release of Coxsackievirus B from Polarized Intestinal Epithelial Cells. Mol. Biol. Cell 2011, 22, 3010–3021. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.Y.; Ha, Y.E.; Choi, J.E.; Ahn, J.; Lee, H.; Kweon, H.S.; Lee, J.Y.; Kim, D.H. Coxsackievirus B4 Uses Autophagy for Replication After Calpain Activation in Rat Primary Neurons. J. Virol. 2008, 82, 11976–11978. [Google Scholar] [CrossRef]
- Laitinen, O.H.; Svedin, E.; Kapell, S.; Hankaniemi, M.M.; Larsson, P.G.; Domsgen, E.; Stone, V.M.; Maatta, J.A.E.; Hyoty, H.; Hytonen, V.P.; et al. New Coxsackievirus 2A (Pro) and 3C (Pro) Protease Antibodies for Virus Detection and Discovery of Pathogenic Mechanisms. J. Virol. Methods 2018, 255, 29–37. [Google Scholar] [CrossRef]
- Feng, Q.; Langereis, M.A.; Lork, M.; Nguyen, M.; Hato, S.V.; Lanke, K.; Emdad, L.; Bhoopathi, P.; Fisher, P.B.; Lloyd, R.E.; et al. Enterovirus 2Apro Targets MDA5 and MAVS in Infected Cells. J. Virol. 2014, 88, 3369–3378. [Google Scholar] [CrossRef]
- Marjomaki, V.; Pietiainen, V.; Matilainen, H.; Upla, P.; Ivaska, J.; Nissinen, L.; Reunanen, H.; Huttunen, P.; Hyypia, T.; Heino, J. Internalization of Echovirus 1 in Caveolae. J. Virol. 2002, 76, 1856–1865. [Google Scholar] [CrossRef]
- Vahatupa, M.; Nattinen, J.; Jylha, A.; Aapola, U.; Kataja, M.; Koobi, P.; Jarvinen, T.A.H.; Uusitalo, H.; Uusitalo-Jarvinen, H. SWATH-MS Proteomic Analysis of Oxygen-Induced Retinopathy Reveals Novel Potential Therapeutic Targets. Invest. Ophthalmol. Vis. Sci. 2018, 59, 3294–3306. [Google Scholar]
- Molla, A.; Hellen, C.U.; Wimmer, E. Inhibition of Proteolytic Activity of Poliovirus and Rhinovirus 2A Proteinases by Elastase-Specific Inhibitors. J. Virol. 1993, 67, 4688–4695. [Google Scholar]
- Krausslich, H.G.; Nicklin, M.J.; Lee, C.K.; Wimmer, E. Polyprotein Processing in Picornavirus Replication. Biochimie 1988, 70, 119–130. [Google Scholar] [CrossRef]
- Lawson, M.A.; Semler, B.L. Alternate Poliovirus Nonstructural Protein Processing Cascades Generated by Primary Sites of 3C Proteinase Cleavage. Virology 1992, 191, 309–320. [Google Scholar] [CrossRef]
- Gullberg, M.; Polacek, C.; Botner, A.; Belsham, G.J. Processing of the VP1/2A Junction is Not Necessary for Production of Foot-and-Mouth Disease Virus Empty Capsids and Infectious Viruses: Characterization of “Self-Tagged” Particles. J. Virol. 2013, 87, 11591–11603. [Google Scholar] [CrossRef] [PubMed][Green Version]
- van Kuppeveld, F.J.; Hoenderop, J.G.; Smeets, R.L.; Willems, P.H.; Dijkman, H.B.; Galama, J.M.; Melchers, W.J. Coxsackievirus Protein 2B Modifies Endoplasmic Reticulum Membrane and Plasma Membrane Permeability and Facilitates Virus Release. EMBO J. 1997, 16, 3519–3532. [Google Scholar] [CrossRef] [PubMed]
- Aldabe, R.; Irurzun, A.; Carrasco, L. Poliovirus Protein 2BC Increases Cytosolic Free Calcium Concentrations. J. Virol. 1997, 71, 6214–6217. [Google Scholar] [PubMed]
- Imajoh, S.; Kawasaki, H.; Suzuki, K. Limited Autolysis of Calcium-Activated Neutral Protease (CANP): Reduction of the Ca2+-Requirement is due to the NH2-Terminal Processing of the Large Subunit. J. Biochem. 1986, 100, 633–642. [Google Scholar] [CrossRef]
- Inomata, M.; Kasai, Y.; Nakamura, M.; Kawashima, S. Activation Mechanism of Calcium-Activated Neutral Protease. Evidence for the Existence of Intramolecular and Intermolecular Autolyses. J. Biol. Chem. 1988, 263, 19783–19787. [Google Scholar]
- Molinari, M.; Anagli, J.; Carafoli, E. Ca2+-Activated Neutral Protease is Active in the Erythrocyte Membrane in its Nonautolyzed 80-kDa Form. J. Biol. Chem. 1994, 269, 27992–27995. [Google Scholar]
- Suzuki, K.; Imajoh, S.; Emori, Y.; Kawasaki, H.; Minami, Y.; Ohno, S. Calcium-Activated Neutral Protease and its Endogenous Inhibitor. Activation at the Cell Membrane and Biological Function. FEBS Lett. 1987, 220, 271–277. [Google Scholar] [CrossRef]
- Melloni, E.; Michetti, M.; Salamino, F.; Pontremoli, S. Molecular and Functional Properties of a Calpain Activator Protein Specific for Mu-Isoforms. J. Biol. Chem. 1998, 273, 12827–12831. [Google Scholar] [CrossRef]
- Pontremoli, S.; Melloni, E.; Michetti, M.; Salamino, F.; Sparatore, B.; Horecker, B.L. An Endogenous Activator of the Ca2+-Dependent Proteinase of Human Neutrophils that Increases its Affinity for Ca2+. Proc. Natl. Acad. Sci. USA 1988, 85, 1740–1743. [Google Scholar] [CrossRef] [PubMed]
- Saido, T.C.; Mizuno, K.; Suzuki, K. Proteolysis of Protein Kinase C by Calpain: Effect of Acidic Phospholipids. Biomed. Biochim. Acta 1991, 50, 485–489. [Google Scholar] [PubMed]
- Tompa, P.; Baki, A.; Schad, E.; Friedrich, P. The Calpain Cascade. Mu-Calpain Activates M-Calpain. J. Biol. Chem. 1996, 271, 33161–33164. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, X.; Yu, Y.; Yu, Y.; Xie, Y.; Zou, Y.; Ge, J.; Peng, T.; Chen, R. Coxsackievirus B3-Induced Calpain Activation Facilitates the Progeny Virus Replication Via a Likely Mechanism Related with both Autophagy Enhancement and Apoptosis Inhibition in the Early Phase of Infection: An in Vitro Study in H9c2 Cells. Virus Res. 2014, 179, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Karpe, Y.A.; Pingale, K.D.; Kanade, G.D. Activities of Proteasome and M-Calpain are Essential for Chikungunya Virus Replication. Virus Genes 2016, 52, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Howe, C.L.; LaFrance-Corey, R.G.; Mirchia, K.; Sauer, B.M.; McGovern, R.M.; Reid, J.M.; Buenz, E.J. Neuroprotection Mediated by Inhibition of Calpain during Acute Viral Encephalitis. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Xiang, Y.; Wang, Q.; Jin, F.; Chen, M.; Ma, K.; Ren, Z.; Wang, Y. Calcium-Signal Facilitates Herpes Simplex Virus Type 1 Nuclear Transport through Slingshot 1 and Calpain-1 Activation. Virus Res. 2014, 188, 32–37. [Google Scholar] [CrossRef]
- Blanc, F.; Furio, L.; Moisy, D.; Yen, H.L.; Chignard, M.; Letavernier, E.; Naffakh, N.; Mok, C.K.; Si-Tahar, M. Targeting Host Calpain Proteases Decreases Influenza A Virus Infection. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2016, 310, 689. [Google Scholar] [CrossRef]
- DeBiasi, R.L.; Edelstein, C.L.; Sherry, B.; Tyler, K.L. Calpain Inhibition Protects Against Virus-Induced Apoptotic Myocardial Injury. J. Virol. 2001, 75, 351–361. [Google Scholar] [CrossRef]
- Debiasi, R.L.; Squier, M.K.; Pike, B.; Wynes, M.; Dermody, T.S.; Cohen, J.J.; Tyler, K.L. Reovirus-Induced Apoptosis is Preceded by Increased Cellular Calpain Activity and is Blocked by Calpain Inhibitors. J. Virol. 1999, 73, 695–701. [Google Scholar]
- Kalamvoki, M.; Mavromara, P. Calcium-Dependent Calpain Proteases are Implicated in Processing of the Hepatitis C Virus NS5A Protein. J. Virol. 2004, 78, 11865–11878. [Google Scholar] [CrossRef] [PubMed]
- Bryant, P.A.; Tingay, D.; Dargaville, P.A.; Starr, M.; Curtis, N. Neonatal Coxsackie B Virus Infection-a Treatable Disease? Eur. J. Pediatr. 2004, 163, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Chiou, C.C.; Liu, W.T.; Chen, S.J.; Soong, W.J.; Wu, K.G.; Tang, R.B.; Hwang, B. Coxsackievirus B1 Infection in Infants Less than 2 Months of Age. Am. J. Perinatol. 1998, 15, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Muehlenbachs, A.; Bhatnagar, J.; Zaki, S.R. Tissue Tropism, Pathology and Pathogenesis of Enterovirus Infection. J. Pathol. 2015, 235, 217–228. [Google Scholar] [CrossRef]
- Joo, C.H.; Ahn, J.; Seo, I.; Kim, Y.K.; Kim, D.; Hong, H.; Lee, H. Characterization of Nonpolio Enteroviruses Recovered from Patients with Aseptic Meningitis in Korea. Intervirology 2005, 48, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Alidjinou, E.K.; Sane, F.; Engelmann, I.; Geenen, V.; Hober, D. Enterovirus Persistence as a Mechanism in the Pathogenesis of Type 1 Diabetes. Discov. Med. 2014, 18, 273–282. [Google Scholar]
- Flynn, C.T.; Kimura, T.; Frimpong-Boateng, K.; Harkins, S.; Whitton, J.L. Immunological and Pathological Consequences of Coxsackievirus RNA Persistence in the Heart. Virology 2017, 512, 104–112. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laajala, M.; Hankaniemi, M.M.; Määttä, J.A.E.; Hytönen, V.P.; Laitinen, O.H.; Marjomäki, V. Host Cell Calpains Can Cleave Structural Proteins from the Enterovirus Polyprotein. Viruses 2019, 11, 1106. https://doi.org/10.3390/v11121106
Laajala M, Hankaniemi MM, Määttä JAE, Hytönen VP, Laitinen OH, Marjomäki V. Host Cell Calpains Can Cleave Structural Proteins from the Enterovirus Polyprotein. Viruses. 2019; 11(12):1106. https://doi.org/10.3390/v11121106
Chicago/Turabian StyleLaajala, Mira, Minna M. Hankaniemi, Juha A. E. Määttä, Vesa P. Hytönen, Olli H. Laitinen, and Varpu Marjomäki. 2019. "Host Cell Calpains Can Cleave Structural Proteins from the Enterovirus Polyprotein" Viruses 11, no. 12: 1106. https://doi.org/10.3390/v11121106
APA StyleLaajala, M., Hankaniemi, M. M., Määttä, J. A. E., Hytönen, V. P., Laitinen, O. H., & Marjomäki, V. (2019). Host Cell Calpains Can Cleave Structural Proteins from the Enterovirus Polyprotein. Viruses, 11(12), 1106. https://doi.org/10.3390/v11121106






