Abstract
(1) Since its introduction to China, pine wilt disease (PWD) has caused severe damage to coniferous forests in affected regions. The disease continues to expand northwestward, posing a significant threat to the ecological security of Xinjiang. (2) This study employed the maximum entropy (MaxEnt, version 3.4.4) model to predict potential areas for PWD transmission and suitable habitats for its vector insect, Monochamus saltuarius (Gebler, 1830). By integrating these results, the potential occurrence areas of PWD in Xinjiang were identified. (3) Human activities were the primary drivers of PWD spread, with factors related to scenic areas and overall human influence playing key roles. Altitude and isothermality were the main limiting factors for the vector insect. Potential PWD occurrence areas were identified, covering approximately 88% of the total coniferous forest area in Xinjiang. (4) High-risk regions include Urumqi City, Ili Kazakh Autonomous Prefecture, and Altay Prefecture. This study clarifies potential transmission routes and analyzes high-risk areas, providing a scientific basis for forestry authorities to implement targeted prevention and control measures.
1. Introduction
Pine wilt disease (PWD) is a devastating systemic disease caused by the pine wood nematode, Bursaphelenchus xylophilus (Steiner et Bührer, 1934, Aphelenchida, Aphelenchidae), which infects Pinus species and other conifers [1]. While native to North America, where it causes limited damage [2], PWD has become a globally significant pathogen since its discovery in Japan in the 20th century, causing extensive mortality of coniferous forests across Asia and Europe [3,4,5]. Since its first detection in Nanjing, China, in 1982 [6], PWD has established 577 epidemic areas across 18 provinces/autonomous regions, resulting in substantial economic and ecological losses (http://www.forestry.gov.cn).
PWD is transmitted by vector beetles carrying the nematode to healthy trees during feeding [2,7,8,9,10,11]. This often leads to blockage of the tree’s vascular system, with symptoms including needle discoloration, reduced resin exudation, and ultimately wilting and death [12]. Notably, tree mortality is influenced by the specific symbiotic bacteria carried by B. xylophilus; strains carrying Pseudomonas spp. are associated with higher host mortality [13,14]. In China, Monochamus alternatus (Hope, 1843, Coleoptera, Cerambycidae) and M. saltuarius (Gebler, 1830, Coleoptera, Cerambycidae) are confirmed vectors [15]. M. saltuarius is distributed in Xinjiang, where it primarily damages conifers such as Picea, Pinus, and Larix [16]. This univoltine species overwinters in the larval stage. Adults emerge around June, and females create oviposition grooves in the bark, a behavior that facilitates nematode transmission [17,18]. The strong flight capacity of M. saltuarius enables rapid local dissemination of PWD, potentially leading to widespread infection in host stands.
Current PWD research focuses on control methods and pathogenic mechanisms. Control strategies include eradication of infected wood, vector control, trunk-injection prophylaxis, quarantine, and epidemic prediction [1]. While direct measures such as tree removal and insect control can eliminate sources, they are resource-intensive. Surveillance and prediction offer a more efficient means to mitigate threats but require accurate data to ensure reliability. Predictive modeling is thus a crucial component of PWD management. The maximum entropy model (MaxEnt, version 3.4.4; American Museum of Natural History, New York, NY, USA, 2024) estimates species distribution probabilities by combining occurrence data with environmental variables [19]. It is widely used due to its operational simplicity and high predictive accuracy [20]. For instance, Xu et al. (2023) used MaxEnt to predict PWD risk in a non-infected county, supporting subsequent control efforts [6]. Aierken et al. (2024) employed ensemble models, including MaxEnt, to forecast the global potential distribution of PWD for 2050–2080 [21]. Similarly, studies by Han et al. (2017), Liu et al. (2017), and Hao et al. (2022) applied MaxEnt to predict PWD distribution across China and specific regions such as Chongqing and Yichang [22,23,24], demonstrating its flexibility across spatiotemporal scales. However, limitations such as overfitting and multicollinearity can affect model transferability [25].
Xinjiang, located in northwestern China, features a unique “three mountains flanking two basins” topography. The Tianshan, Altai, and Kunlun Mountains harbor rich coniferous forest resources vital for regional water conservation and soil protection [26]. Previous studies have classified coniferous forests in the Tianshan and Altai Mountains as moderately and highly susceptible to PWD, respectively [27]. Furthermore, M. saltuarius is distributed in both mountain ranges [28]. As the primary vector for PWD in northern China, M. saltuarius could facilitate the rapid spread of the disease if PWD were introduced [29,30]. In 2021, PWD was first reported in Gansu Province, neighboring Xinjiang. While no PWD occurrence has been confirmed in Xinjiang to date (http://www.forestry.gov.cn), the disease remains a serious potential threat to the region’s economy and ecology, warranting concern from forestry authorities.
As one of the few PWD-free regions in China with susceptible hosts, Xinjiang faces a persistent invasion risk. Beyond natural factors, increased human-mediated pathways—such as infrastructure development associated with the “Silk Road,” growing domestic and international trade, and rapid tourism expansion—have elevated the movement of people and goods, raising the probability of PWD introduction. Given the critical ecological role of Xinjiang’s coniferous forests and the devastating impact of PWD [7], predicting its potential distribution in the region is imperative. However, relevant predictive studies for Xinjiang remain scarce.
To address this gap, explore transmission mechanisms, and identify high-risk areas, this study considers Xinjiang’s unique context by incorporating climatic, anthropogenic, vector-related, and proximity-based factors (e.g., proximity to Grade A scenic spots). Predictions were made at two spatial scales: China (for PWD transmission zones) and Xinjiang (for M. saltuarius habitat suitability). These were integrated to map current potential PWD occurrence areas in Xinjiang, aiming to clarify risk zones, analyze transmission routes, and pinpoint high-risk regions [16].
Based on existing knowledge of PWD transmission and Xinjiang’s environmental conditions, we hypothesize the following: (1) Human-mediated factors are the primary drivers of PWD introduction into Xinjiang. (2) The distribution of M. saltuarius in Xinjiang is primarily constrained by specific bioclimatic factors, particularly temperature and precipitation patterns. (3) A coupled model integrating potential transmission zones (anthropogenically driven) and vector suitability areas (climatically driven) can effectively identify high-risk regions for PWD establishment in Xinjiang.
This research enhances the understanding of PWD transmission dynamics and provides a scientific basis for local forestry authorities to develop preventive strategies.
2. Materials and Methods
2.1. Study Area
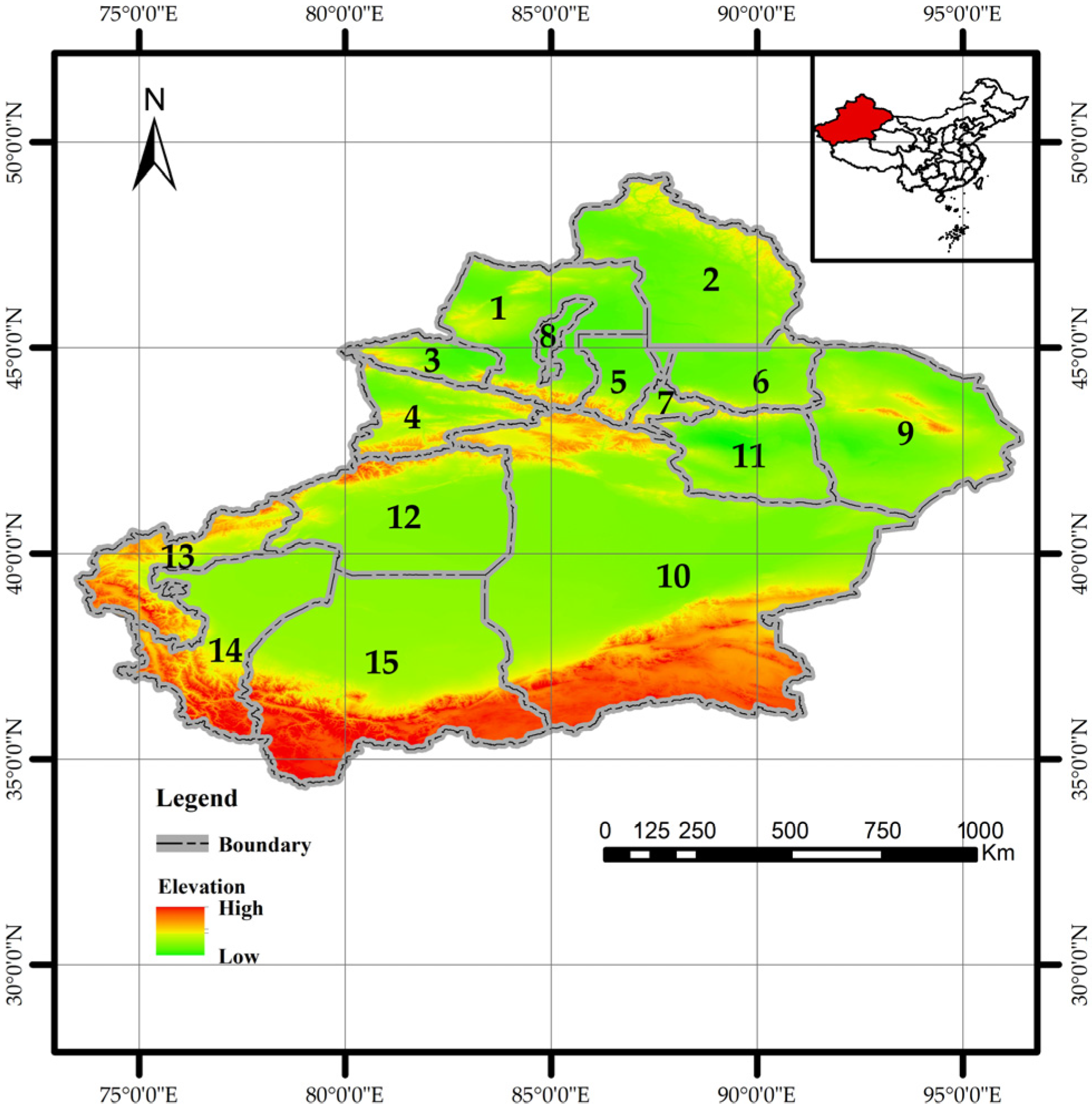
Xinjiang is located in northwestern China (34.25°–49.17° N, 73.33°–96.42° E) (Figure 1). With a land border of approximately 5600 km adjoining eight countries, it covers about 1.66 million km2, making it China’s largest provincial-level administrative region. It is a key hub for the modern “Silk Road.” The region is characterized by a “three mountains flanking two basins” terrain, encompassing a forest area of approximately 83,300 km2 (including the Xinjiang Production and Construction Corps), with a forest coverage rate of 5.05% and a timber stock volume of 445 million m3, indicating a relatively fragile ecosystem. The Altai, Tianshan, and Kunlun Mountains contain rich coniferous forest resources, accounting for over 97% of the region’s total timber stock. Larix sibirica Ledeb. and Pinus schrenkiana Fisch. & C.A. Mey. dominate these forests (http://lcj.xinjiang.gov.cn).
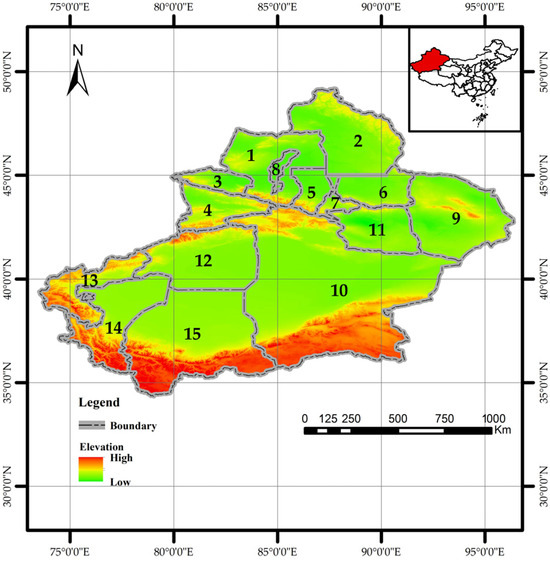
Figure 1.
Geographical map of Xinjiang showing administrative divisions. The numbered labels (1–15) correspond to the following prefectures, autonomous prefectures, and cities: 1: Tarbagatay Prefecture. 2: Altay Prefecture. 3: Bortala Mongolian Autonomous Prefecture. 4: Ili Kazakh Autonomous Prefecture. 5,6: Changji Hui Autonomous Prefecture. 7: Urumqi City. 8: Karamay. 9: Hami City. 10: Bayingolin Mongolian Autonomous Prefecture. 11: Turpan City. 12: Aksu City. 13: Kizilsu Kirgiz Autonomous Prefecture. 14: Kashgar Prefecture. 15: Hotan Prefecture. Note: Map approval number: GS (2022) No. 4308 (the same below).
Altai Mountain coniferous forests are primarily composed of L. sibirica, Picea obovata Ledeb., Abies sibirica Korsh., and P. sylvestris L. The Tianshan Mountains are mainly covered by P. schrenkiana var. tianschanica (Rupr.) W.C. Cheng & S.H. Fu, with L. sibirica present in the eastern ranges. The Kunlun Mountains host species such as Juniperus centrasiatica Kom., J. jarkendensis Kom., and J. pseudosabina var. Turkestanica [26]. Although species composition varies across ranges, forest stands are often naturally pure and structurally simple. These coniferous forests hold significant ecological and economic value for Xinjiang and China.
2.2. Occurrence Data
2.2.1. PWD Occurrence Data
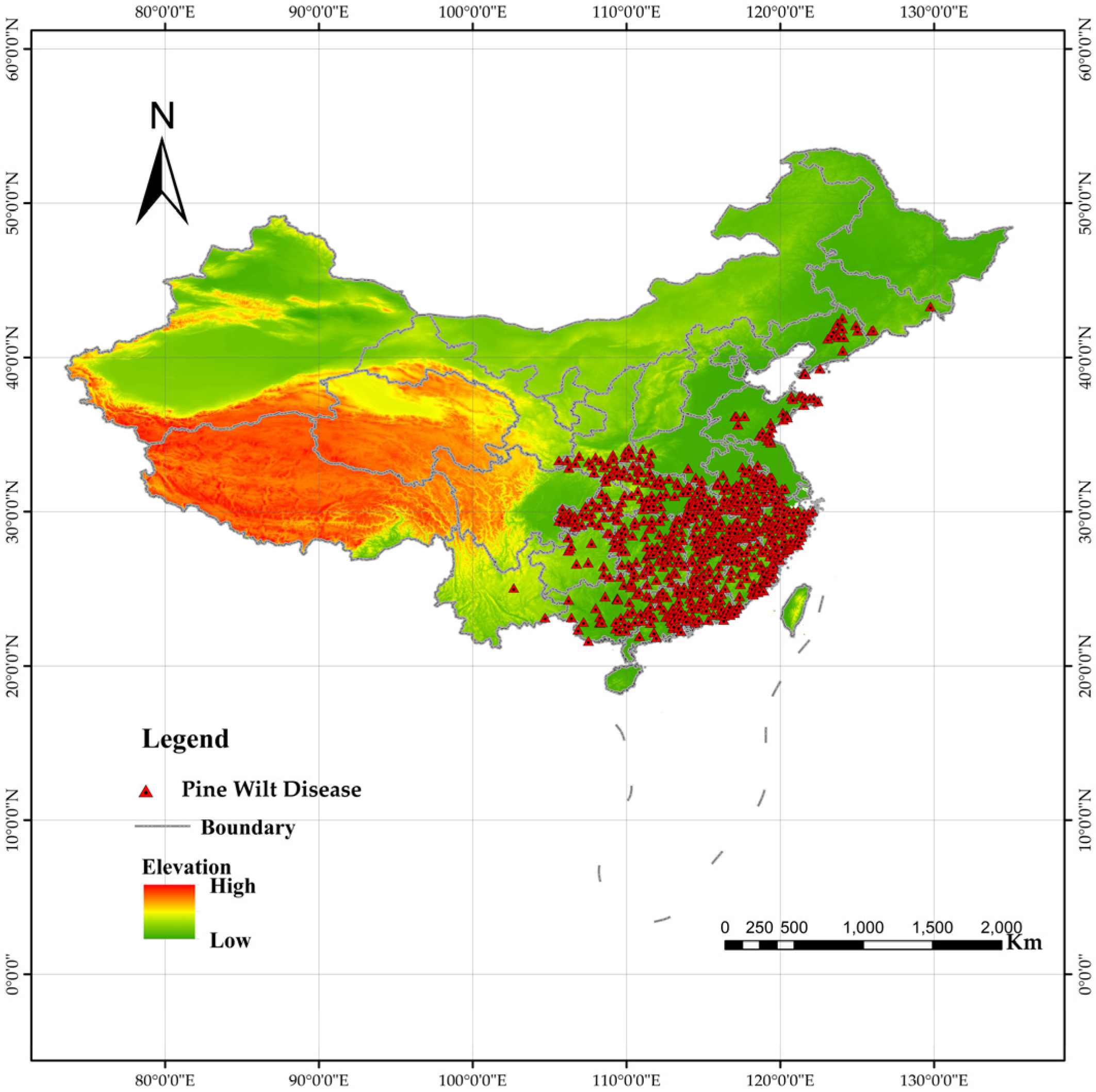
Since 1990, China has established a national forest pest monitoring network. In 2003, a comprehensive pest census was conducted, creating an extensive database. PWD distribution data for this study were obtained from the annual lists of affected counties published by the China National Forestry and Grassland Administration (http://www.forestry.gov.cn). Following model requirements, each affected county was represented by coordinate points adjusted to corresponding small-scale coniferous forest plots within the county. These plots were identified using coniferous forest data extracted from a national vegetation map. This yielded 685 occurrence points [21] (Figure 2). Compared to using county centroids, this method reduces model uncertainty [31,32].
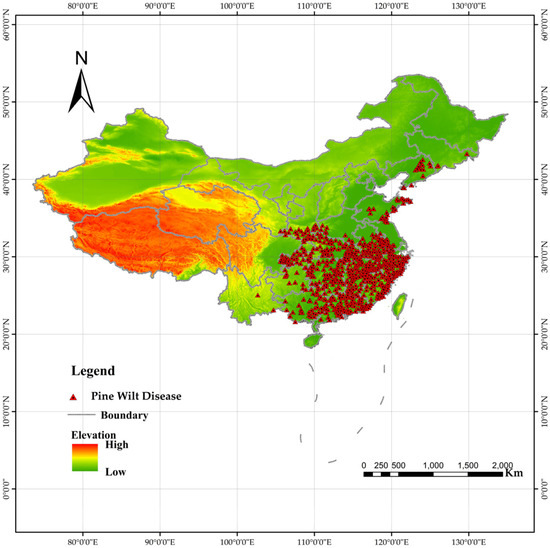
Figure 2.
Distribution of pine wilt disease (PWD) outbreak sites in China. Red dots indicate confirmed occurrence locations used for model training (n = 685).
2.2.2. Vector Insect Occurrence Data
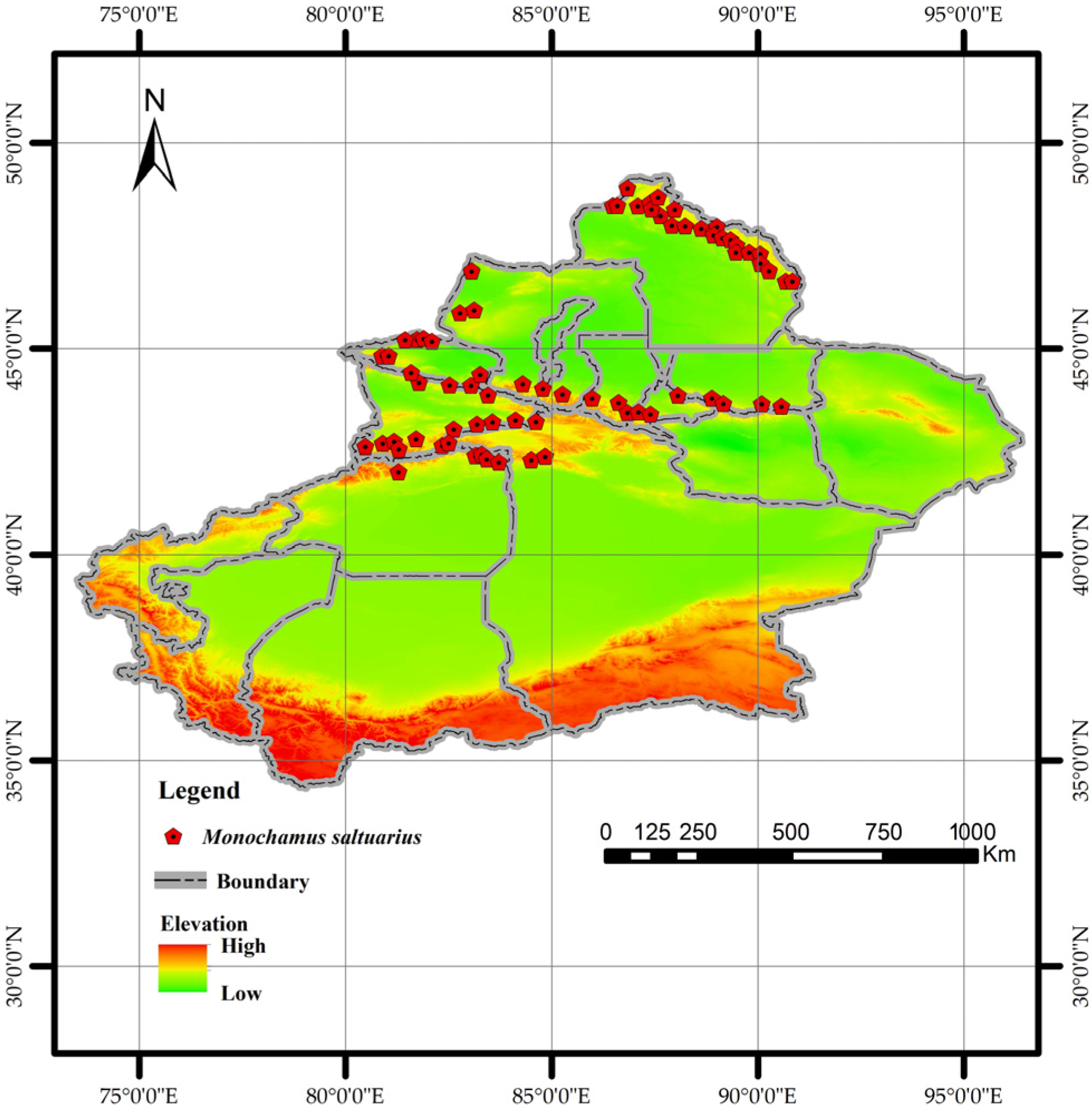
National occurrence data for M. saltuarius were sourced from the China National Forestry and Grassland Administration, the Global Biodiversity Information Facility (GBIF) [33], the National Animal Collection Resource Center (http://museum.ioz.ac.cn/), the Field Identification Manual for Forest Pests in Xinjiang, and the published literature. Given the challenges of direct field identification in Xinjiang’s complex terrain, the known distribution range of the insect’s host plants (as recorded in the manual) was used as a proxy for its habitat. A total of 72 random points were generated within this range using the same methodology as for PWD [34,35,36,37,38,39,40] (Figure 3).
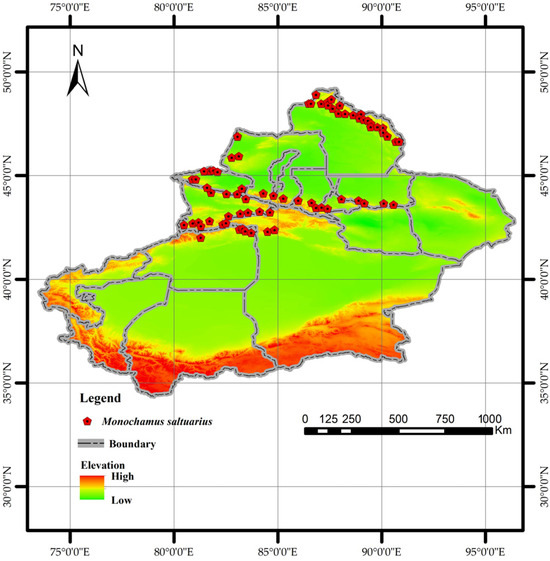
Figure 3.
Distribution of Monochamus saltuarius occurrence points used in this study.
2.3. Environmental Variables
2.3.1. Variables for PWD Transmission
PWD transmission is categorized as natural or human-mediated [41]. Natural transmission includes vector insect feeding, nematode movement, and root contact [42,43]. Considering data availability and relevance, coniferous forest areas within the potential influence range of vector insects were selected as the natural transmission factor.
Human activities are the primary drivers of long-distance PWD spread. Therefore, this study included a composite human influence factor (HIF) raster (integrating road networks and settlement density). Furthermore, given historical links between improper wood packaging and outbreaks and the increased human mobility associated with Xinjiang’s growing tourism sector, two additional anthropogenic factors were incorporated [44,45]: distance to power lines and proximity to Grade A scenic spots (including those below Grade 3A rating).
The HIF raster (1 km resolution, representing 1995–2004) was downloaded from NASA Earthdata (https://search.earthdata.nasa.gov/, accessed 2 April 2025). Power line vector data were extracted from a GitHub repository (https://github.com/OpenQGIS/OMap-GeoDB, accessed 25 April 2025). Locations of all Grade-A tourist attractions in China were obtained as shapefiles from the Beijing Municipal Government open data portal (https://data.beijing.gov.cn/, accessed 25 April 2025). All vector data were converted to 1 km resolution rasters to match the HIF layer.
2.3.2. Variables for M. saltuarius Habitat Suitability
Bioclimatic variables and elevation data were used to predict vector habitat suitability. We downloaded 19 bioclimatic variables (Bio1–Bio19) and elevation data at 30-s (~1 km) resolution, representing the 1970–2000 climate normals, from WorldClim (https://www.worldclim.org/, accessed 7 May 2025). Although wind speed influences insect dispersal [46,47,48], its effect was indirectly considered via the vector insect factor and thus not included as a separate variable.
2.3.3. Other Variables
A land cover map was required to define the natural transmission factor. The 1 km resolution National Vegetation Distribution Map of China was obtained from the Resource and Environment Science and Data Center (https://www.resdc.cn/, accessed 6 April 2025) to extract coniferous forest distribution.
2.4. Methodology
Successful disease establishment typically requires a combination of vector, pathogen, host, and suitable climate. However, existing studies have demonstrated that PWD can occur when both the pathogen and host trees coexist [5]. For PWD, vector insects play an irreplaceable role in transmission [49]. Therefore, predicting potential PWD occurrence areas in Xinjiang was decomposed into two components: predicting potential transmission areas and predicting potential suitable habitats for M. saltuarius. The outputs were given equal weight and coupled to determine the final potential occurrence areas.
2.4.1. MaxEnt Model
MaxEnt is a machine learning model based on the maximum entropy principle, requiring only species occurrence points and environmental variables to predict potential distribution. Its strength lies in maintaining good performance with limited occurrence data. However, excessive or correlated variables can lead to multicollinearity and overfitting [50]. We used MaxEnt version 3.4.4. Model parameters were set as follows: 75% of occurrence points for training, 25% for testing; other parameters were default; output format was logistic (.asc). The model was run 10 times, with the results averaged for stability, and visualized in Quantum GIS (QGIS, version 3.34.1; QGIS Development Team, 2023) using the natural breaks classification method.
2.4.2. Predicting Potential PWD Transmission Areas
As no direct data product exists for PWD’s natural transmission factor, we created it using QGIS, vector insect occurrence points, and the national vegetation map. The process involved (1) extracting coniferous forest data; (2) identifying areas overlapping with vector insect occurrence points; and (3) applying a 2.4 km buffer (based on a study by Liu et al., 2023, demonstrating most affected coniferous forests were within this distance from an outbreak epicenter) to define coniferous forests potentially influenced by vectors [51]. A new field was added, assigning a value of 1 to affected forests and 0 to others. The selection of vector insect points balanced model accuracy, integrating literature, book records, PWD occurrence points, and additional data from epidemic areas.
For anthropogenic factors, the processed rasters for HIF, distance to power lines, and distance to scenic spots were unified in terms of the coordinate system, resolution, and extent with the natural transmission factor. All data were converted to ASCII format for MaxEnt [52].
Given the absence of PWD in Xinjiang, historical PWD occurrence points across China and the processed transmission factors were imported into MaxEnt to model potential transmission areas across China. The Xinjiang portion of the result was then extracted. Model settings mirrored those in Section 2.4.1.
2.4.3. Predicting Potential Habitat for M. saltuarius
To reduce multicollinearity [53], 20 initial bioclimatic and elevation variables were standardized in QGIS, clipped to Xinjiang’s boundary, and correlated with M. saltuarius occurrence points. The MaxEnt model (parameters identical to Section 2.4.1) was run 10 times, and factors contributing less than 1% were eliminated. This yielded 11 core factors, including Mean Temperature of the Warmest Quarter (Bio10), Annual Precipitation (Bio12), Precipitation of the Driest Quarter (Bio17), and Altitude (elev). The final model used these 11 factors and the insect occurrence points.
2.4.4. Model Accuracy Evaluation
The jackknife test was used to analyze variable contributions. Model accuracy was evaluated using the Receiver Operating Characteristic (ROC) curve and the Area Under the Curve (AUC). AUC values range from 0 to 1, with higher values indicating better performance: ≤0.6 (fail), 0.6–0.7 (poor), 0.7–0.8 (fair), 0.8–0.9 (good), and 0.9–1.0 (excellent) [49].
2.4.5. Coupling Method
The results for potential PWD transmission areas and potential M. saltuarius suitable habitats were combined using a 1:1 weighting scheme. The final composite layer was then masked by the distribution map of coniferous forests in Xinjiang to delineate the ultimate potential PWD occurrence areas.
3. Results
3.1. Model Accuracy
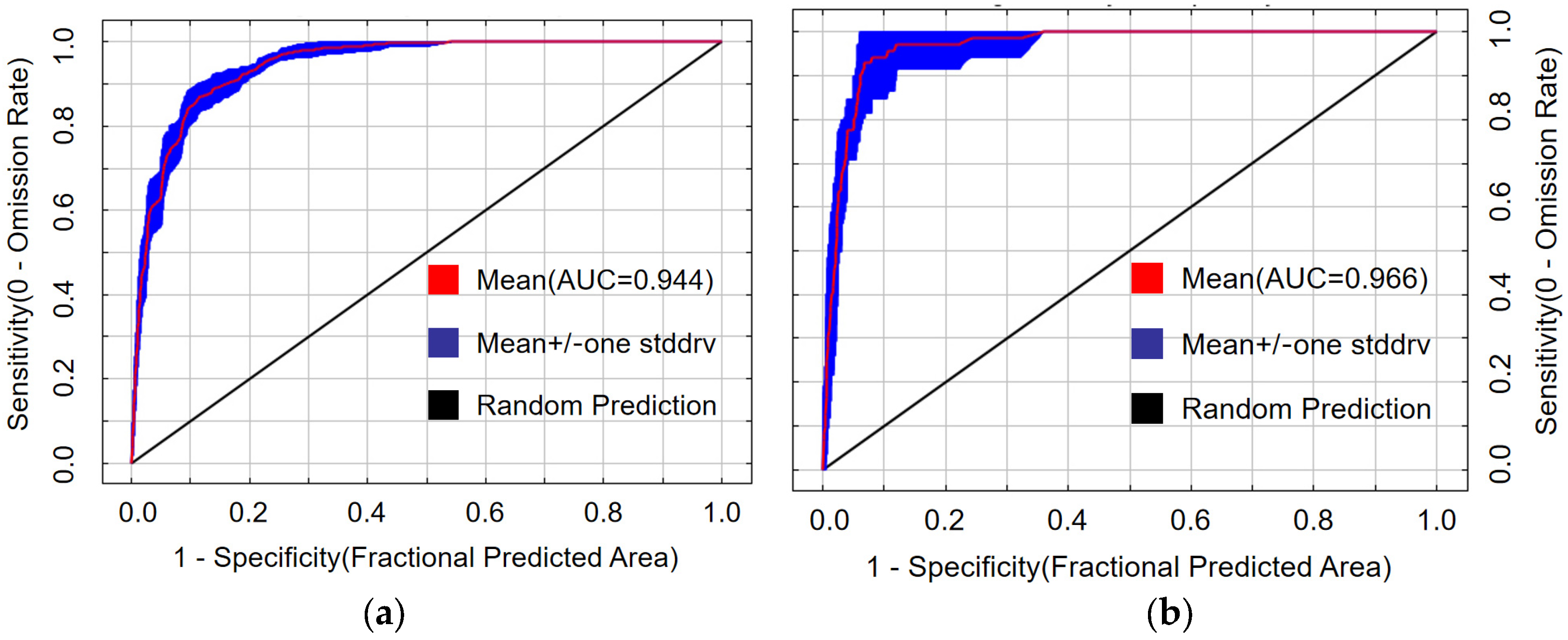
Both predictive models performed excellently. The mean AUC value for the PWD transmission model was 0.944 (Figure 4a), and for the M. saltuarius habitat model, 0.966 (Figure 4b). Both values exceed the 0.9 threshold for ‘excellent’ predictive accuracy. The slightly higher AUC for the vector model indicates a well-defined relationship between its distribution and the selected bioclimatic variables.
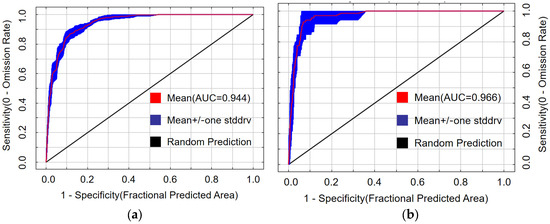
Figure 4.
Receiver operating characteristic curves for the maximum entropy models: (a) potential transmission areas of pine wilt disease; (b) potential suitable habitat of Monochamus saltuarius.
3.2. Model Drivers
3.2.1. Contribution of Factors to PWD Transmission Potential
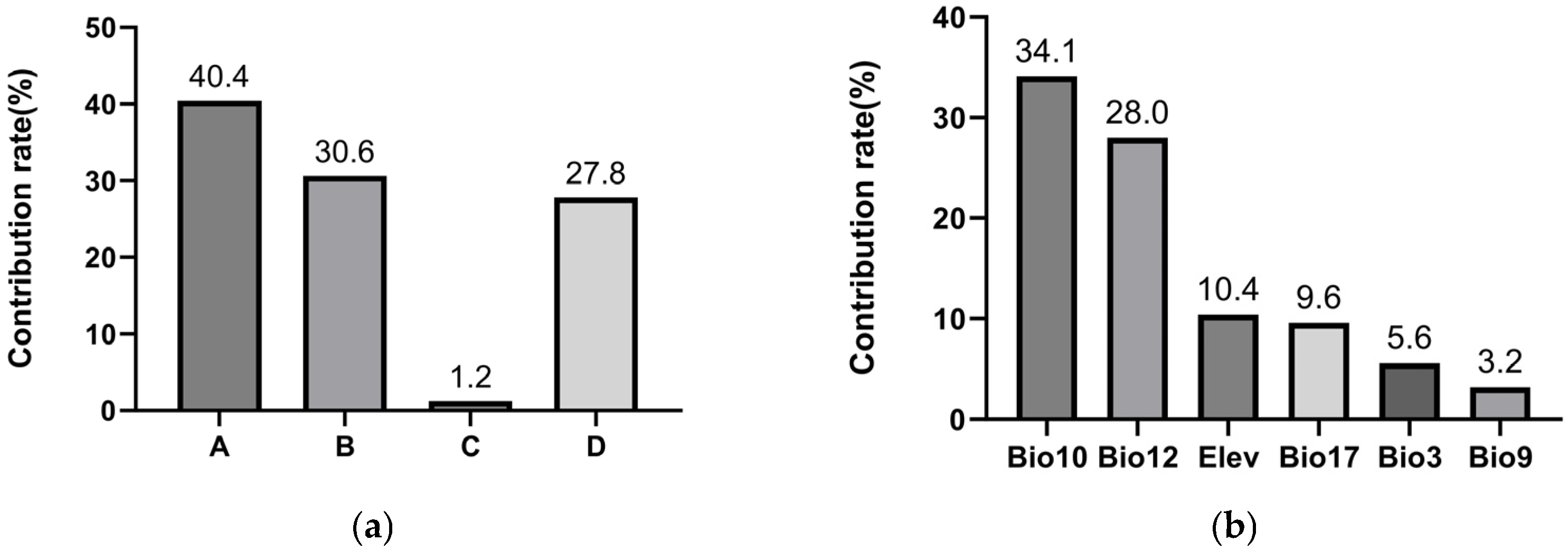
Jackknife analysis revealed that anthropogenic factors dominated the potential spread of PWD to Xinjiang (Figure 5a). The composite human influence factor (40.4%) and proximity to Grade A scenic spots (30.6%) together accounted for 71.0% of the model’s explanatory power. In contrast, the area of coniferous forest within the natural dispersal range of vector insects (proxy for natural transmission) contributed 27.8%. This strongly supports hypothesis H1, indicating human-mediated pathways pose the greatest introduction risk. The contribution of distance to power lines was negligible (1.2%).
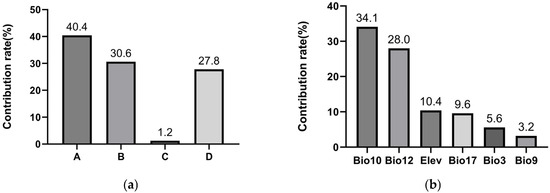
Figure 5.
Contribution rates of environmental variables in the maximum entropy models: (a) Variables associated with PWD transmission (A = human influence factor, B = proximity to Grade A scenic spots, C = distance to power lines, D = short-distance dispersal mediated by vector insects. Human-related factors (A + B) accounted for 71% of the model’s explanatory power, indicating that human activities are the primary pathway for PWD introduction into Xinjiang. (b) The top six contributing variables for Monochamus saltuarius habitat suitability.
3.2.2. Contribution of Factors to M. saltuarius Habitat Suitability
After removing variables with contribution rates below 1%, nine of the 20 initial bioclimatic factors were excluded. These included Min Temperature of Coldest Month (Bio6), Temperature Annual Range (Bio7), Mean Temperature of Wettest Quarter (Bio8), Mean Temperature of Coldest Quarter (Bio11), Precipitation of Driest Month (Bio14), Precipitation Seasonality (Bio15), Precipitation of Wettest Quarter (Bio16), Precipitation of Warmest Quarter (Bio18), and Precipitation of Coldest Quarter (Bio19).
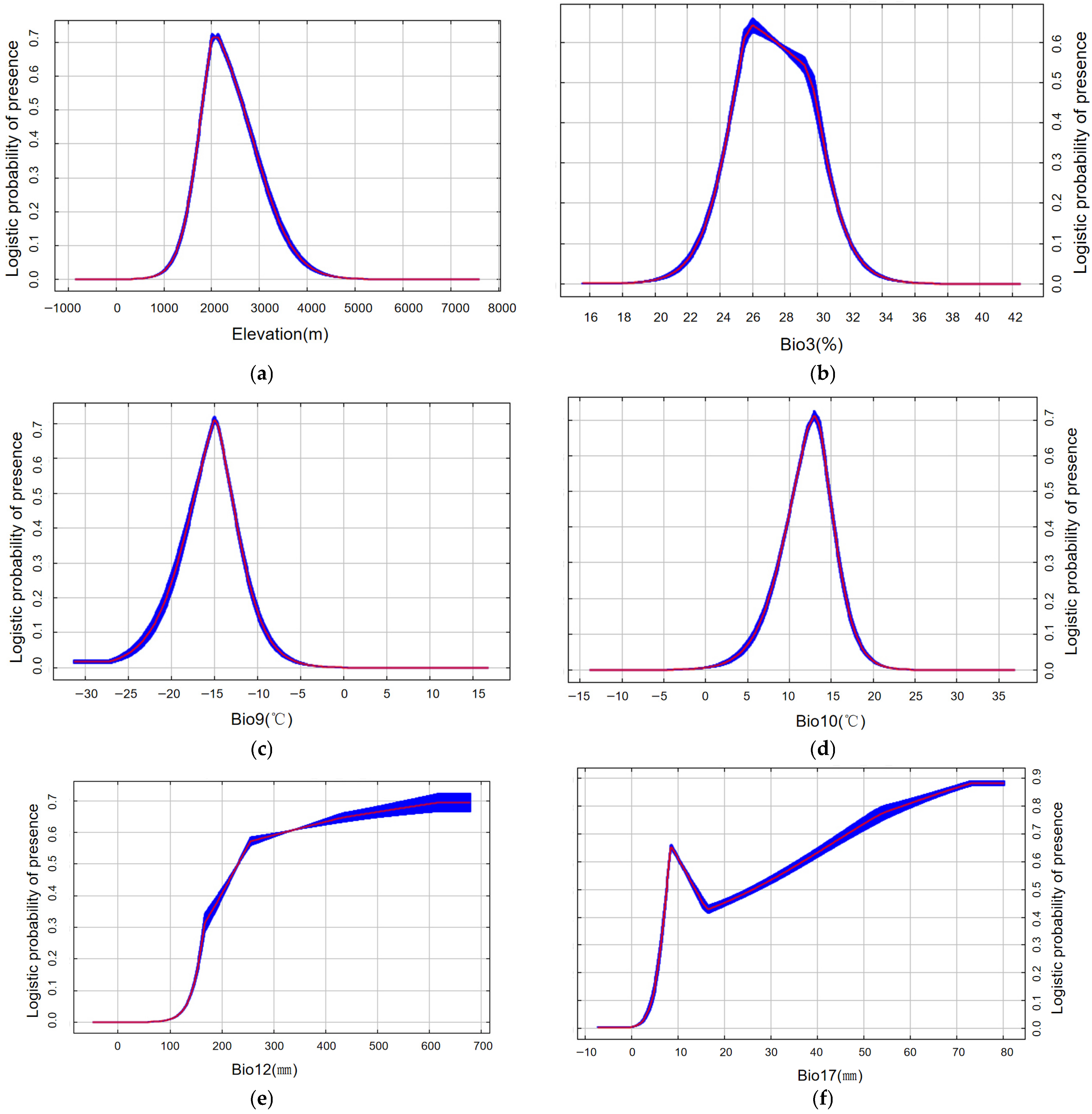
Six variables were identified as core predictors of suitable habitat for M. saltuarius, with a cumulative contribution rate of 90.9% (Figure 5b): Mean Temperature of the Warmest Quarter (Bio10), Annual Precipitation (Bio12), Precipitation of the Driest Quarter (Bio17), Altitude (Elev), Isothermality (Bio3), and Mean Temperature of the Driest Quarter (Bio9). Using a habitat suitability threshold of 0.5, the optimal environmental ranges were defined as follows: Altitude, 1797–2676 m (Figure 6a); Isothermality, 0.25–0.29 (Figure 6b); Mean Temperature of the Driest Quarter, −17 °C to −13.1 °C (Figure 6c); Mean Temperature of the Warmest Quarter, 10.6–14.7 °C (Figure 6d); Annual Precipitation, 232–679 mm (Figure 6e); and Precipitation of the Driest Quarter, 7.6–13.7 mm or 26–80 mm (Figure 6f).
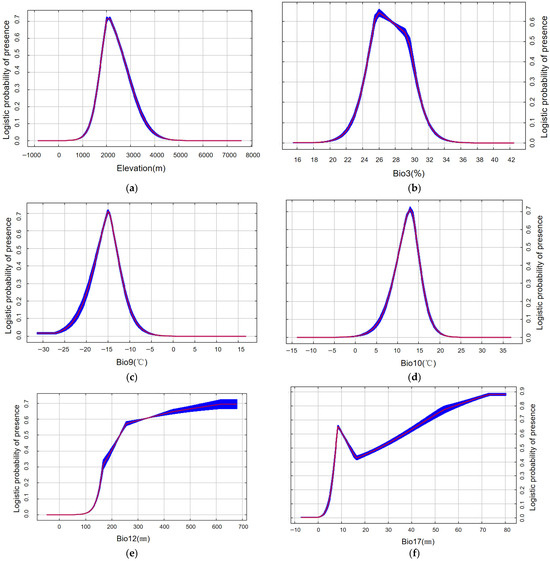
Figure 6.
Response curves of Monochamus saltuarius under current climate scenario (red indicates the average value, while blue represents the range of fluctuation.): (a) elevation response curve (m); (b) Isothermality Response curve (%); (c) Mean Temperature of Driest Quarter response curve (°C); (d) Mean Temperature of Warmest Quarter response curve (°C); (e) Annual Precipitation response curve (mm); (f) Precipitation of Driest Quarter response curve (mm). Note: The response curve for Precipitation of Driest Quarter (Bio17) exhibits multiple peaks, an atypical pattern for a biologically critical variable. The bimodal distribution reflects differences in water niches among host species and represents the integrated suitable range of the host–vector complex.
For M. saltuarius, climatic factors were paramount. Mean Temperature of the Warmest Quarter (Bio10) and Annual Precipitation (Bio12) were the most influential, contributing 34.1% and 28.0%, respectively. This aligns with hypothesis H2. Response curves defined optimal ranges: Bio10 between 10.6 and 14.7 °C and Bio12 between 232 and 679 mm. The results imply that the distribution of M. saltuarius is primarily constrained by cool summer conditions and year-round moisture availability.
The Precipitation of the Driest Quarter (Bio17) showed a bimodal response curve, likely reflecting the methodology of using host plant distributions as insect occurrence proxies, which integrates the slightly different hydrological niches of multiple host species. The two optimal ranges (7.6–13.7 mm and 26–80 mm) thus represent aggregated suitable conditions for the host–plant complex.
3.3. Spatial Representation of Model Results
3.3.1. Potential Transmission Areas of PWD
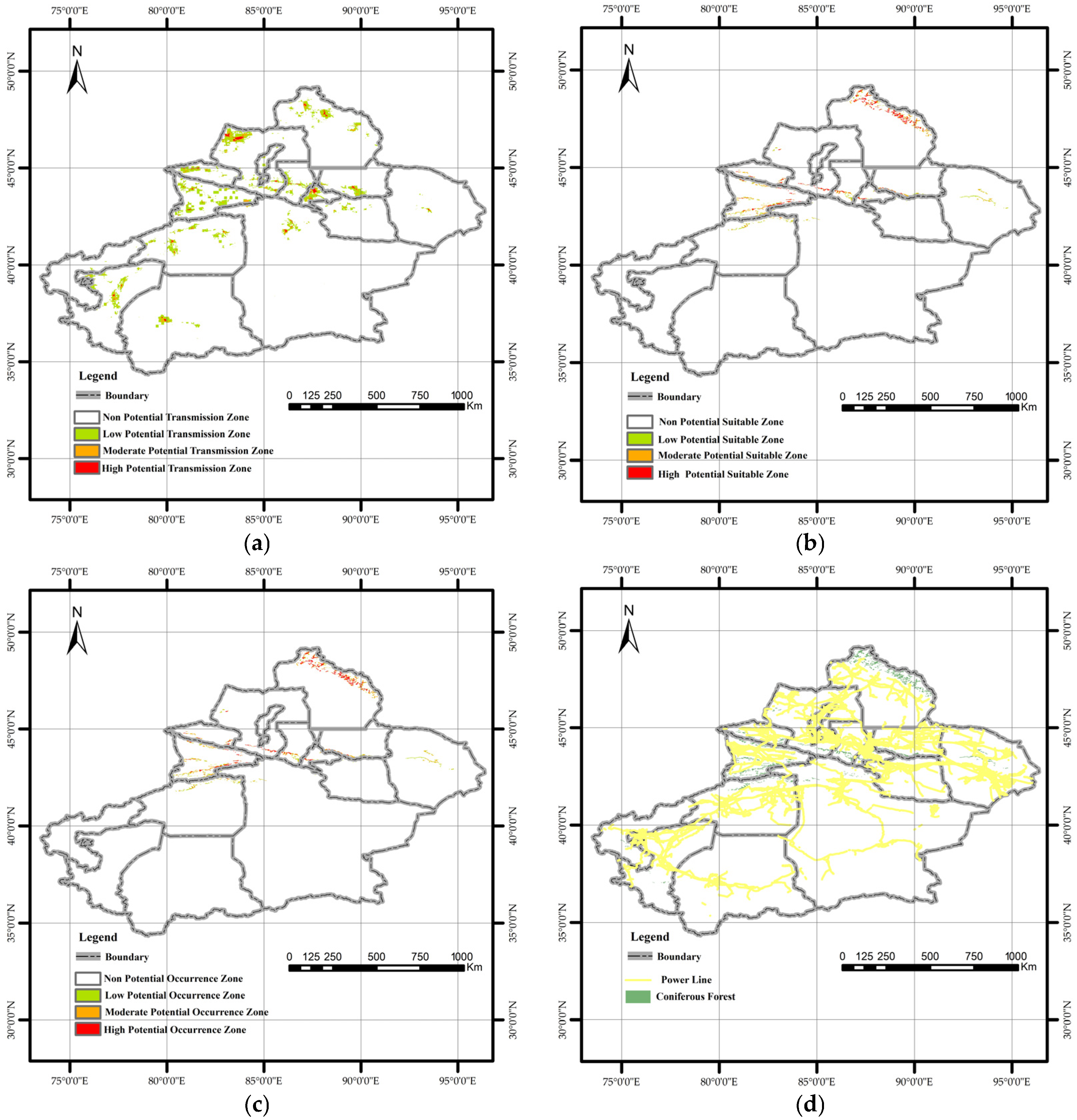
The MaxEnt model, parameterized with four transmission-related factors, indicated that PWD has the potential to spread into Xinjiang (Figure 7a). The high-potential transmission zone (probability 0.55–1) covered approximately 3000 km2, the moderate-potential transmission zone (probability 0.27–0.55) encompassed about 7000 km2, and the low-potential transmission zone (probability 0.1–0.27) extended over roughly 54,000 km2. All prefectures and cities in Xinjiang were classified as being at risk of PWD transmission. The high-potential transmission zone covered about 3000 km2 (<2% of Xinjiang) and was strategically concentrated in key transportation and economic hubs: Urumqi (the regional capital), Tacheng, Altay Prefectures (bordering Kazakhstan), and the Ili Valley. This spatial overlap with major “Silk Road” nodes visually corroborates the dominant role of human activity.
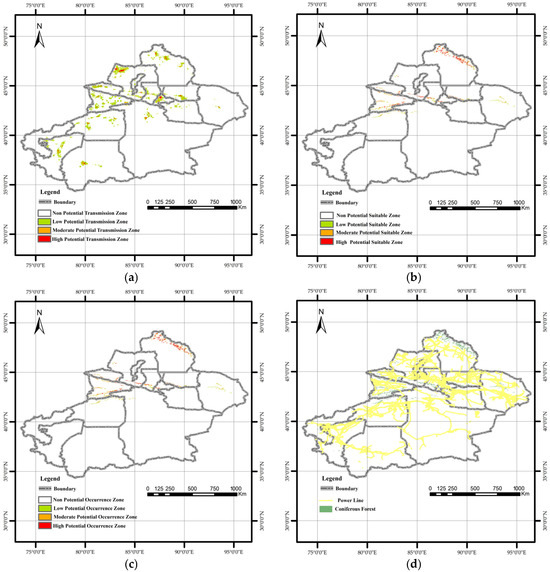
Figure 7.
Spatial visualization of maximum entropy model outputs in Quantum GIS: (a) Potential transmission areas of pine wilt disease. The high-potential transmission area covers approximately 3000 km2, representing about 1.9% of Xinjiang’s total land area. Urumqi City, Tacheng Prefecture, and Altay Prefecture are identified as the primary high-risk transmission zones and should be prioritized for future quarantine efforts. (b) Potential suitable habitat of Monochamus saltuarius. The total potential habitat area for the vector insect is 71,200 km2, of which 24,000 km2 is classified as high-potential habitat. This high-suitability area is primarily concentrated in the Altai Mountains. In the Ili River Valley of the Tianshan Mountains, high-potential habitat exhibits a decreasing trend from west to east. (c) Integrated potential occurrence areas of PWD in Xinjiang. The potential occurrence area of pine wilt disease comprises approximately 88% of the total coniferous forest area in Xinjiang. (d) Spatial distribution of power lines and coniferous forests across the study area.
3.3.2. Potential Suitable Habitats of M. saltuarius
Suitable habitat for M. saltuarius was more spatially confined and ecologically defined, covering 71,200 km2 (Figure 7b). The high-potential suitable zone (24,000 km2) was concentrated in mid-elevation forests of the Altai Mountains. A second important zone existed in the Tianshan Mountains, particularly the Ili Valley, with suitability decreasing eastward. This trend can be attributed to climatic differences between the eastern and western Tianshan Mountains. In general, the western Tianshan experiences higher temperatures and drier conditions during summer, whereas the eastern Tianshan has relatively cool and humid summers. This mountainous distribution directly reflects the insect’s stringent climatic requirements.
3.4. Comprehensive Risk Assessment
The final risk map, generated by coupling the two models, indicated that PWD could potentially become established in 88% of Xinjiang’s total coniferous forest area (Figure 7c), highlighting the profound threat. A critical comparison of the individual and composite maps shows that the ultimate establishment risk is strongly constrained by the vector’s climatic niche. Areas with high transmission risk but low vector suitability (e.g., parts of the Tarim Basin) did not emerge as high-occurrence zones. Synthesizing these patterns, three high-risk hotspots were identified where conditions for both introduction and establishment converge. Urumqi City possesses the highest anthropogenic introduction risk and is proximate to suitable vector habitat in the Tianshan Mountains. The Ili Kazakh Autonomous Prefecture contains extensive, highly suitable vector habitats overlapping with moderate introduction risk pathways related to trade and tourism. The Altay Prefecture hosts the most concentrated area of highly suitable vector habitat and is intersected by high-risk introduction points along the northern border. These regions represent the most likely epicenters for initial invasion and subsequent spread if PWD is introduced.
4. Discussion
Identifying high-risk areas and transmission routes is crucial for early PWD prevention in Xinjiang. This study considered both the introduction and establishment processes, decomposing the prediction into transmission zones and vector habitats. This approach accounts for the differences in vector species and significant climatic variations between Xinjiang and the southern epidemic regions in China. Results suggest PWD could occur in Xinjiang, with most coniferous forests potentially at risk. Urumqi City, Ili Kazakh Autonomous Prefecture, and Altay Prefecture are the most likely future transmission sources.
4.1. Possibility of PWD Occurrence in Xinjiang
According to official announcements, Xinjiang remains one of the few regions in China with susceptible Pinaceae hosts but no reported PWD invasion. However, outbreaks have occurred at similar latitudes in Liaoning and neighboring Gansu Province. Despite no current detection, national occurrence data show a northwestward expansion trend. PWD has demonstrated strong environmental adaptability, causing damage in high-altitude (Qinling Mountains) and cold (Liaoning) regions [24], continually exceeding previous understanding of its suitable range. Establishment requires both host trees and the nematode [5], with local vector insect adaptability being a critical determinant [51].
4.2. Significance Compared to Previous Research
A prior study suggested no potential PWD area in northwestern China [2]. However, this is often the result of considering only abiotic factors, omitting critical biotic factors such as vector insects and human activities, which can significantly impact prediction accuracy. The global-scale prediction by Aierken et al. [21] yielded results somewhat similar to this study but lacks the granularity for targeted guidance by Xinjiang’s forestry authorities. Large-scale studies may also face data availability challenges for specific transmission factors, potentially introducing bias. Furthermore, climate data versions and suitability may differ. Our methodology, tailored to Xinjiang’s PWD-free status and distinct vector insect characteristics, offers a more targeted approach for regional prediction.
4.3. Potential Transmission of PWD
This study confirms that human activities account for nearly 75% of the potential spread of PWD in Xinjiang, consistent with previous research [45]. The human influence factor was the most significant factor, likely linked to improper timber handling and transport [54]. Proximity to Grade A tourist attractions was the second most influential factor, as these sites in Xinjiang often feature coniferous forests, and high human traffic can facilitate pathogen spread [45]. The minimal contribution (<2%) of distance to power lines is likely because power lines in Xinjiang are predominantly in Gobi Desert areas with little overlap with coniferous forests (Figure 7d). Enhanced quarantine measures for wood packaging materials may further reduce this pathway’s relevance.
4.4. Potential Habitat of M. saltuarius
The prediction for M. saltuarius identified precipitation (Annual Precipitation: 232–679 mm) and temperature (Mean Temperature of Warmest Quarter: 10.6–14.7 °C) as key survival factors, indicating a preference for cool, humid summer conditions. This is a key reason why southern Xinjiang (characterized by an arid climate) lacks potential PWD occurrence zones. The climatic thresholds identified here are similar to those in previous PWD prediction studies [6,22,23], likely because PWD distribution in northern China is closely linked to the niche of its primary vector, M. saltuarius, narrowing the overall suitable range. The optimal habitat for the vector insect occurs at elevations between 1797 and 2676 m, corresponding to the mid-elevation zones of the Tianshan and Altai Mountains, where host tree species are abundant. The mean temperature of the driest quarter (Bio9: −17 °C to −13.1 °C) represents an extreme climatic condition that remains suitable for overwintering. Collectively, these findings indicate that temperature and precipitation regimes during summer and winter impose stronger constraints on vector distribution than those of other seasons.
The bimodal response curve for Precipitation of the Driest Quarter (Bio17) likely stems from using host plant distributions as a proxy for insect occurrences. This dataset inherently encompasses the environmental niches of multiple host species with slightly different drought tolerances. The model identified two precipitation ranges (7.6–13.7 mm and 26–80 mm) representing the aggregated suitable conditions for the host community. Therefore, these peaks should be interpreted cautiously as representing the collective niche breadth of the M. saltuarius-host system.
4.5. Recommendations from Local Forestry Authorities and Relevant Departments
In China, the primary vectors of PWD differ between northern and southern regions: M. saltuarius is the dominant vector in the north, while M. alternatus prevails in the south. Studies have shown that a lack of climatic adaptation in introduced vector populations can disrupt the PWD transmission cycle [55]. Consequently, Xinjiang—particularly Urumqi City, Ili Kazakh Autonomous Prefecture, and Altay Prefecture—should strengthen quarantine inspections of timber originating from northern epidemic areas and improve the efficiency of rapid PWD detection in logistics and trade. Moreover, since most scenic areas in Xinjiang are situated within natural forests, forestry authorities and relevant departments need to enhance PWD surveillance in these regions to reduce the risk of introduction [45]. Given the critical role of vector insects in disease spread, monitoring and managing the population density of M. saltuarius also constitutes an effective strategy for preventing the establishment and dissemination of PWD [49].
4.6. Limitations and Future Directions
It is important to note that PWD severity is also influenced by the characteristics of the invading nematode population (e.g., symbiotic bacteria). This factor was not included due to data constraints and may cause slight prediction deviations. Furthermore, incomplete vector insect distribution data could introduce errors. Future studies incorporating these factors and refining vector data will yield more accurate and scientifically robust predictions.
5. Conclusions
This study employed the MaxEnt model to predict potential PWD occurrence areas in Xinjiang. Given the disease’s absence in the region, predictions of potential transmission zones and vector insect habitats were combined to delineate final risk zones. Results demonstrated that human-mediated transmission was the predominant spread mode, accounting for nearly 75% of the risk. Mean Temperature of the Warmest Quarter (Bio10) and Annual Precipitation (Bio12) were key limiting factors for the vector insect. The findings indicate that 88% of Xinjiang’s coniferous forests are potential risk zones, with Urumqi City, Ili Kazakh Autonomous Prefecture, and Altay Prefecture classified as high-risk areas. These results provide a scientific foundation for local forestry authorities to develop targeted prevention and control strategies.
Author Contributions
Conceptualization, H.G.; methodology, T.H. and Z.X.; software, T.H., Z.X. and F.H.; formal analysis, Z.X.; investigation, Z.X.; data curation, H.G.; writing—original draft preparation, Z.X.; writing—review and editing, L.D., Z.X. and H.G.; visualization, Z.X.; supervision, H.G.; project administration, H.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Xinjiang Forestry Development Subsidy Funds (Forestry and Grass Science and Technology (XJLYKJ-2023-08)).
Data Availability Statement
Data are available upon request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ye, J.; Wu, X. Research progress of Pine Wilt Disease. For. Pest Dis. 2022, 41, 1–10. [Google Scholar]
- Futai, K. Pine wood nematode, Bursaphelenchus xylophilus. Annu. Rev. Phytopathol. 2013, 51, 61–83. [Google Scholar] [CrossRef] [PubMed]
- Dwinell, L.D. First report of pinewood nematode (Bursaphelenchus xylophilus) in Mexico. Plant Dis. 1993, 77, 846. [Google Scholar] [CrossRef]
- Robinet, C.; David, G.; Jactel, H. Modeling the distances traveled by flying insects based on the combination of flight mill and mark-release-recapture experiments. Ecol. Modell. 2019, 402, 85–92. [Google Scholar] [CrossRef]
- Pimentel, C. Bursaphelenchus xylophilus (Pinewood Nematode); CABI Compendium: Oxfordshire, UK, 2022. [Google Scholar] [CrossRef]
- Xu, G.; Yu, R.; Yang, C.; Liu, H.; Zhou, Z.; Shen, Y. Prediction of invasion risk of Pine Wilt Disease based on GIS spatial technology and MaxEnt model in western Sichuan Province of southwestern China. J. Beijing For. Univ. 2023, 45, 104–115. [Google Scholar]
- Ye, J. Epidemic Status of Pine Wilt Disease in China and Its Prevention and Control Techniques and Counter Measures. Sci. Silvae Sin. 2019, 55, 1–10. [Google Scholar]
- Mamiya, Y.; Enda, N. Transmission of Bursaphelenchus Lignicolus (Nematoda: Aphelenchoididae) by Monochamus Alternatus (Coleoptera: Cerambycidae). Nematologica 1972, 18, 159–162. [Google Scholar] [CrossRef]
- Fu, D.; Hu, S.; Ye, H.; Haack, R.A.; Zhou, P. Pine Wilt Disease in Yunnan, China: Evidence of non-local pine sawyer Monochamus alternatus (Coleoptera: Cerambycidae) populations revealed by mitochondrial DNA. Insect Sci. 2010, 17, 439–447. [Google Scholar] [CrossRef]
- Kobayashi, F.; Yamane, A.; Ikeda, T. The Japanese Pine Sawyer Beetle as the Vector of Pine Wilt Disease. Annu. Rev. Entomol. 1984, 29, 115–135. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, S.; Wei, W.; Hao, H.; Zhang, B.; Butcher, R.A.; Sun, J. Chemical Signals Synchronize the Life Cycles of a Plant-Parasitic Nematode and Its Vector Beetle. Curr. Biol. 2013, 23, 2038–2043. [Google Scholar] [CrossRef]
- Back, M.A.; Bonifácio, L.; Inácio, M.L.; Mota, M.; Boa, E. Pine Wilt Disease: A global threat to forestry. Plant Pathol. 2014, 73, 1026–1041. [Google Scholar] [CrossRef]
- Kiyohara, T.; Bolla, R.I. Pathogenic variability among populations of the pinewood nematode, Bursaphelenchus xylophilus. For. Sci. 1990, 36, 1061–1076. [Google Scholar] [CrossRef]
- Han, Z.M.; Hong, Y.D.; Zhao, B.G. A study on pathogenicity of bacteria carried by pine wood nematodes. J. Phytopathol. 2003, 151, 683–689. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, Y.; Fan, L.; Liu, P.; Zheng, Y. Investigation on long-horned beetle species in Pine Wilt Disease epidemic area in Dalian. For. Pest Dis. 2021, 40, 36–39. [Google Scholar]
- Yusufukadier, A.; Ren, L.; Hasimajie, P.; Deli, E.; Lieke, T. Determination of flight ability of Monochamus sartor urussovi. For. Pest Dis. 2025, 44, 27–31. [Google Scholar]
- Ye, W.; Li, T.; Weng, M.; Guo, W.; Xin, F.; Yu, W.; Wu, S.; Guo, Y. RNAi-Mediated Silencing of Chitin Synthase 1 (CHS1) Disrupts Molting and Growth in Monochamus alternatus. Forests 2025, 16, 922. [Google Scholar] [CrossRef]
- Uchiyama, Y.; Futai, K. Assessing the Role of Asymptomatic Infected Trees in Pine Wilt Disease Spread in Japan—Insights from Tree Health Monitoring. Forests 2025, 16, 583. [Google Scholar] [CrossRef]
- Ning, H. Impacts of Climate Change on The Suitable Distribution of Dendroctonus armandi, Trypophloeus klimeschi and Their Host in China. Ph.D. Thesis, Northwest A&F University, Shanxi, China, 2022. [Google Scholar] [CrossRef]
- Wei, S.; Li, T.; Lin, Y. Prediction of the Potential Distribution of Bursaphelenchus xylophilus in Sichuan Province Using MaxEnt Model. Sichuan J. Zool. 2019, 38, 37–46. [Google Scholar]
- Aierken, N.; Wang, G.; Chen, M.; Chai, G.; Han, X.; Qian, Z.; Zhang, X. Assessing global Pine Wilt Disease risk based on ensemble species distribution models. Ecol. Indic. 2024, 167, 112691. [Google Scholar] [CrossRef]
- Han, Y.; Wang, Y.; Xiang, Y.; Ye, J. Prediction of potential distribution of Bursaphelenchus xylophius in China based on Maxent ecological niche model. J. Nanjing For. Univ. (Nat. Sci. Ed.) 2015, 39, 6–10. [Google Scholar] [CrossRef]
- Liu, X.; Pu, Y.; Li, H.; Liu, X.; Yang, Q.; Ding, S. A study on Potential Biotope of Pine Wilt Disease in Chongqing by using MaxEnt Model. Ecol. Environ. Monit. Three Gorges 2017, 3, 75–80. [Google Scholar]
- Hao, Z.; Fang, G.; Huang, W.; Ye, H.; Zhang, B.; Li, X. Risk Prediction and Variable Analysis of Pine Wilt Disease by a Maximum Entropy Model. Forests 2022, 13, 342. [Google Scholar] [CrossRef]
- Li, X. Prediction of Potential Distribution of Quercus fabri in China Based on Optimized MaxEnt Model. Sci. Silvae Sin. 2018, 54, 153–164. [Google Scholar] [CrossRef]
- Li, L.-P.; Liu, Y.-N.; Tang, Z.-Y.; Guo, Z.-D.; Fang, J.-Y. Community structure and influencing factors of mountain coniferous forest in Xinjiang. Arid Zone Res. 2011, 28, 31–39. [Google Scholar] [CrossRef]
- Hu, B.; Huang, W.; Hao, Z.; Guo, J.; Huang, Y.; Cheng, X.; Zhao, J.; Jiao, Q.; Zhang, B. Invasion of Pine Wilt Disease: A threat to forest carbon storage in China. Ecol. Indic. 2024, 169, 112819. [Google Scholar] [CrossRef]
- Forestry Pest Control and Quarantine Bureau of Xinjiang Uygur Autonomous Region. Wildlife Identification Manual of Forest Pests in Xinjiang; China Forestry Publishing House: Beijing, China, 2014; p. 347. [Google Scholar]
- Zou, Y.; Zhou, Y.; Guo, S.; Wang, T.; Zong, S. Risk of Monochamus saltuarius spreading in China. Chin. J. Appl. Entomol. 2023, 60, 287–297. [Google Scholar]
- Zheng, Y.; Wang, J.; Wang, W.; Li, Y.; Fan, L.; Shi, Y.; Zhang, Y. Parasitic Effect of Dastarcus helophoroides of Monochamus alternatus Biotype on Monochamus saltuarius. Chin. J. Biol. Control 2022, 38, 587–594. [Google Scholar]
- Wang, W.; Zhu, Q.; He, G.; Liu, X.; Peng, W.; Cai, Y. Impacts of climate change on Pine Wilt Disease outbreaks and associated carbon stock losses. Agric. For. Meteorol. 2023, 334, 109426. [Google Scholar] [CrossRef]
- Zhao, H.; Xian, X.; Yang, N.; Guo, J.; Zhao, L.; Shi, J.; Liu, W.-X. Risk assessment framework for Pine Wilt Disease: Estimating the introduction pathways and multispecies interactions among the pine wood nematode, its insect vectors, and hosts in China. Sci. Total Environ. 2023, 905, 167075. [Google Scholar] [CrossRef]
- GBIF Backbone Taxonomy. Available online: https://doi.org/10.15468/39omei (accessed on 1 April 2025).
- Fu, Y. Control technology of Monochamus saltuarius in Guan censhan forest area. For. Shanxi 2025, 1, 50–51. [Google Scholar] [CrossRef]
- Li, Y.; Deng, X.; Zhang, Y.; Song, X.; Ma, X.; Wang, Z.; Du, W. Survey of Longicorn Species in Pinus koraiensis Plantation in Dongning City, Heilongjiang Province. For. Sci. Technol. 2025, 50, 55–58. [Google Scholar]
- Wang, S. Ecological characteristics and control methods of Monochamus saltuarius—Taking Zuojialin Farm, Xiaolongshan Forestry Protection Center, Gansu Province as an example. Guangdong Canye 2024, 58, 34–36. [Google Scholar] [CrossRef]
- Zhu, Z.; Sun, F.; Cao, C.; Liu, Y.; Huo, Q.; Xu, Y. The catching effect of two means on Monochamus saltuarius in field experiments. J. Nanjing For. Univ. (Nat. Sci. Ed.) 2025, 49, 89–94. [Google Scholar]
- Wang, J.; Zhang, Y.; Shu, H.; Zheng, Y. Study on the number of Bursaphelenchus xylophilus carried by Monochamus saltuarius adult. J. Beijing For. Univ. 2023, 45, 142–147. [Google Scholar]
- Dong, Z.; Nie, H.; Zhong, C.; Chen, Z.; Chen, Q.; Jiao, S. Feeding preferences of adult Monochamus saltuarius on four host pine trees. Contemp. Hortic. 2022, 45, 18–19. [Google Scholar] [CrossRef]
- Fan, L.; Shi, Y.; Jiang, S.; Zheng, Y. Study on the species of Longhorned beetles carrying Bursaphelenchus xylophilus in Liaoning. For. Res. 2021, 34, 174–181. [Google Scholar] [CrossRef]
- Haran, J.; Rousselet, J.; Tellez, D.; Roques, A.; Roux, G. Phylogeography of Monochamus galloprovincialis, the European vector of the pinewood nematode. Pest Manag. Sci. 2018, 91, 247–257. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, X.; Mo, X.; Wu, J.; Zhou, Y.; Fang, G. Host vegetation connectivity is decisive for the natural spread of Pine Wilt Disease. Pest Manag. Sci. 2024, 80, 5141–5156. [Google Scholar] [CrossRef]
- Lu, X.; Huang, J.; Li, X.; Fang, G.; Liu, D. The interaction of environmental factors increases the risk of spatiotemporal transmission of Pine Wilt Disease. Ecol. Indic. 2021, 133, 108394. [Google Scholar] [CrossRef]
- Chongqing Forestry Bureau. Strengthen quarantine law enforcement of infected trees and make every effort to control the spread of pine wood nematode. Land Greening 2019, 9, 50–51. [Google Scholar]
- Hao, Z.; Huang, J.; Zhou, Y.; Fang, G. Spatiotemporal Pattern of Pine Wilt Disease in the Yangtze River Basin. Forests 2021, 12, 731. [Google Scholar] [CrossRef]
- Gao, R.; Wang, Z.; Wang, H.; Hao, Y.; Shi, J. Relationship Between Pine Wilt Disease Outbreaks and Climatic Variables in the Three Gorges Reservoir Region. Forests 2019, 10, 816. [Google Scholar] [CrossRef]
- Xi, Y.; Niu, S. The Effects of Climatic Factors on Pine Wilt Disease. For. Resour. Manag. 2008, 4, 70–76. [Google Scholar] [CrossRef]
- Osada, Y.; Yamakita, T.; Shoda-Kagaya, E.; Liebhold, A.M.; Yamanaka, T. Disentangling the drivers of invasion spread in a vector-borne tree disease. J. Anim. Ecol. 2018, 87, 1512–1524. [Google Scholar] [CrossRef]
- Xiao, Y.; Guo, Q.; Xie, N.; Yuan, G.; Liao, M.; Gui, Q.; Ding, G. Predicting the global potential distribution of Bursaphelenchus xylophilus using an ecological niche model: Expansion trend and the main driving factors. BMC Ecol. Evol. 2024, 24, 48. [Google Scholar] [CrossRef]
- Huang, T.; Yang, T.; Wang, K.; Huang, W. Assessing the Current and Future Potential Distribution of Solanum rostratum Dunal in China Using Multisource Remote Sensing Data and Principal Component Analysis. Remote Sens. 2024, 16, 271. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, J.; Yang, T. Natural Factors Play a Dominant Role in the Short-Distance Transmission of Pine Wilt Disease. Forests 2023, 14, 1059. [Google Scholar] [CrossRef]
- Eli, S.; Abdukerim, R.; Mamtimin, Y.; Mamat, T.; Ismail, Z.; Halik, M. Assessment of Habitat Suitability for Gazella subgutturosa in the Xinjiang Based on the MaxEnt Modeling. Chin. J. Wildl. 2019, 40, 27–32. [Google Scholar] [CrossRef]
- Feng, X.; Park, D.S.; Liang, Y.; Pandey, R.; Papeş, M. Collinearity in ecological niche modeling: Confusions and challenges. Ecol. Evol. 2019, 9, 10365–10376. [Google Scholar] [CrossRef]
- Hu, G. Study on the Harm and Comprehensive Control Measures of Pine Wood Nematode Disease. J. Agric. Catastrophol. 2021, 11, 31–32. [Google Scholar]
- Zhou, H.; Xie, M.; Koski, T.-M.; Li, Y.; Zhou, H.; Song, J.; Gong, C.; Fang, G.; Sun, J. Epidemiological model including spatial connection features improves prediction of the spread of Pine Wilt Disease. Ecol. Indic. 2024, 163, 112103. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.