Abstract
Heartwood proportion (HWP) and specific gravity (SG) are two important properties of Dalbergia retusa and Platymiscium curuense wood, which is considered to be of high value. The objective of this study was to establish which morphological and soil fertility parameters present the greatest influence on HWP and SG. For this, increment cores were extracted, and soil samples were collected. The results showed that D. retusa presented a lower HWP (22.65%) than P. curuense (28.75%), and D. retusa averaged a higher value (0.87) than P. curuense (0.63). The forward stepwise regression analysis for D. retusa showed that the magnesium content was the most important factor for SG, while for the HWP, the potassium content was the most important, followed by diameter at breast height (DBH). SG was most strongly influenced by total height in P. curuense, and HWP was most strongly influenced by DBH. Additional notable results showed that the SG of D. retusa was primarily determined by soil fertility conditions, whereas the SG of P. curuense was more strongly influenced by tree morphology. Meanwhile, the HWP in both species was mainly affected by DBH and total height, and to a lesser extent by soil fertility conditions. These results show that plantation management should be focused on trees with large diameters and HWP, since soil conditions demonstrated little effect on this property.
1. Introduction
Heartwood is located in the inner part of the tree and is particularly relevant in high-value commercial species [1]. It is created in the sapwood, where a series of complex biochemical processes occur that are associated with the death of parenchyma cells (radial and axial) and the deposition of extractives in the heartwood [2]. Following programmed cell death, physiologically active parenchyma cells in the sapwood contribute to the formation and transformation of heartwood [2], stages which are of greater importance when these species are established in fast-growing plantations [3]. Unlike sapwood, which is found in the outer part of the trunk and is responsible for the physiological functions of the tree, heartwood has very limited physiological activity, if any [2,3]. Heartwood is unique because it reflects the presence of low-molecular-weight, non-structural secondary metabolites, collectively known as extractives, which confer distinctive properties such as odor characteristics, specific wood color, dimensional stability, and durability [4,5]. Another highly important characteristic of wood is its specific gravity (SG), a physical property that influences many other characteristics, such as shrinkage, as well as anatomical or mechanical properties [6,7,8,9].
The variation in these two properties occurs not only within the tree but also between trees of the same species. In the case of heartwood, its proportion tends to decrease with height [2], and SG varies across the trunk and from the pith to the bark [9], showing no consistent trend in fast-growing trees: it may increase, decrease, or remain stable and is considered a mainly intrinsic genetic characteristic of each species [10,11].
Fast-growth conditions in plantations are more prevalent than in natural forests due to the higher competition among individuals [12,13]. The age at which heartwood appears is particularly relevant in high-value commercial species and varies widely among species [14]. Additionally, other factors affect heartwood proportion and SG values, including tree morphological characteristics, such as diameter, total height, crown base height, and crown size and density; plantation management practices; and site differences. The latter includes climatic, topographic, altitudinal, and soil fertility conditions [15,16,17,18].
Regarding tree morphology characteristics, trees in fast-growing plantations allow for a significant reduction in rotation cycles, reaching commercial diameters and wood volumes at lower production costs and consequently generating higher economic returns [19,20]. In relation to the influence of dasometric conditions on heartwood and SG, it was found that the effect of tree morphology on SG is variable. A positive correlation with diameter has been observed in tropical species; however, this relationship tends to weaken during juvenile stages, when cambial aging due to tree age exerts greater influence [21]. With respect to height, SG may decrease, vary beyond a certain height, or remain constant along the stem [22]. These differences arise because SG is considered an intrinsic genetic characteristic of each species [9].
The effect of soil fertility on heartwood and SG is complex, as soil condition can be characterized by multiple variables, where individually or collectively, they can influence tree growth and, consequently, wood properties [23]. Some studies report that larger trees, those with slower growth rates, or those established in acidic soils tend to develop larger heartwood areas. Conversely, trees growing in more alkaline soils (pH between 4.8 and 6.0) usually exhibit smaller heartwood areas [2]. However, each species shows correlations with soil fertility [22].
On the other hand, the establishment of commercial forest plantations has included exotic and native species [24], which are grouped into two main categories: (i) medium-density commercial species and (ii) high-value timber species. Within this context, two genera and their species are particularly relevant due to their high commercial value. The first group corresponds to the genus Dalbergia L. f., which has been of great importance in local and international markets for centuries because of the esthetic appeal of its wood and excellent wood properties [23]. The second group corresponds to species of the genus Platymiscium, whose wood is considered among the finest in Central America and is widely used in the manufacture of furniture, flooring, handicrafts, and musical instruments [25].
The variation in heartwood across these species within forest plantations has been addressed in several studies [15,16,26,27]. For instance, in Dalbergia cochinchinensis, diameter at breast height (DBH) was reportedly the most relevant variable and can be used as a predictor of heartwood content. In addition, soil fertility, including bulk density, organic matter content, and silt fraction in the surface soil layer, significantly influences heartwood content [15]. In Dalbergia sissoo, Pande [16] found that SG was influenced by both site and tree height, while Sunny et al. [26] confirmed this site effect on SG, noting that soil variables exert an even greater impact on this physical wood property. Likewise, Pande and Rawat [27] demonstrated that SG is negatively related to tree diameter and height and that heartwood proportion (HWP) gradually increases until it stabilizes once the diameter reaches 20 cm.
As mentioned earlier, few studies have analyzed the variation in HWP and SG in Dalbergia retusa and Platymiscium curuense in relation to soil fertility and tree morphology. Therefore, in the present study, we aimed to establish which morphological (DBH, total height, and commercial height) and soil fertility parameters (pH, electrical conductivity, exchangeable acidity, and macro- and micronutrients) have the greatest influence on the HWP and SG of these two high-value species in fast-growing plantations in the Central Pacific of Costa Rica, to help fill the existing information gap regarding these wood parameters. With the information gathered from this study, we expect to contribute to the understanding of factors that affect the heartwood and specific gravity of two high-value-wood trees planted in fast-growth conditions under differing soil fertility properties.
2. Materials and Methods
2.1. Study Area and Management Conditions
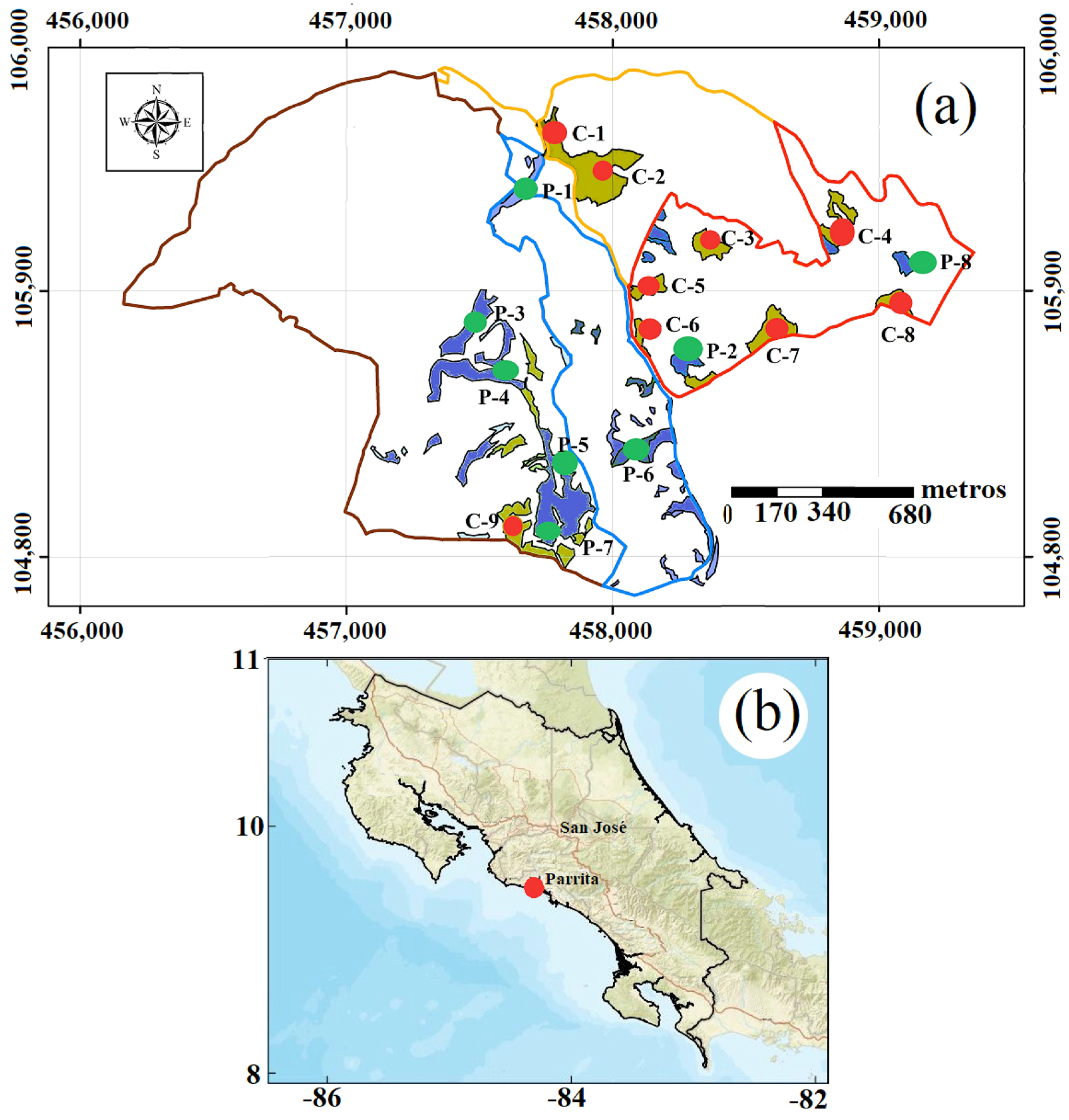
For this study, we used forest plantations of D. retusa (Figure 1a) and P. curuense (Figure 1b) located in Parrita, Puntarenas Province, Costa Rica (Figure 1b), and owned by BARCA S.A. (https://barca-agroforestal.com/bird-ecoplantations, URL Accessed on 26 November 2025). Nine plots were used for D. retusa, and eight plots were used for P. curuense, with age varying from 25 to 29 years and area from 0.87 ha to 21.12 ha (Table 1). The average morphology data of the sampled trees for each plot and age are presented in Table 1. Plantations of both species had an initial density of 1111 trees/ha, established with 3 × 3 m spacing and seed-derived seedlings produced in a nursery. No fertilization was applied, and pruning was conducted at 5 years of age, up to a height of 5 m, while thinning was carried out at 5, 10, and 20 years. In general, the plantations were established on sites with slopes between 15% and 30%.
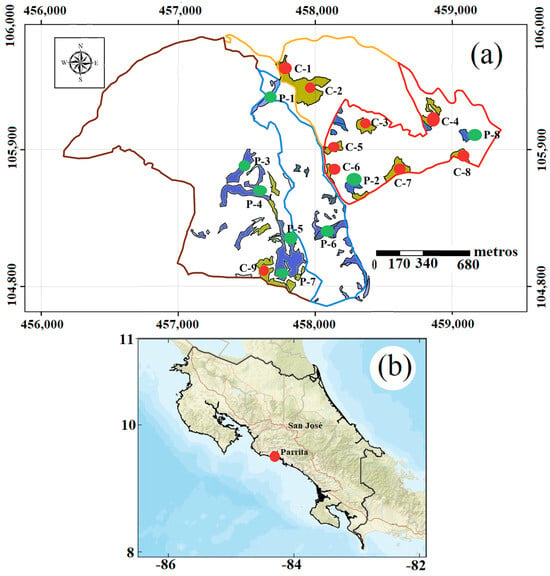
Figure 1.
(a) Localization of different plots of D. retusa and P. curuense in plantation area and (b) geographic location of the forest plantations. Legend: area of blue color and green color point represents plantation areas and plot of D. retura species and area of brown color and red color point represents plantation areas and plot of P. curuense species.

Table 1.
Morphological parameter data by plot and age of the sampled D. retusa and P. curuense trees.
2.2. Soil Fertility Conditions
From each sampled plot per species, soil samples were collected at three points, forming a triangular pattern within the plot. Samples were taken at two depth horizons, 0–30 cm and 30–60 cm, corresponding to the first and second soil horizons, respectively. Subsequently, the three samples from each horizon were composited into a single sample per horizon for each plot, resulting in a total of 18 soil samples (2 horizon × 9 plots) for D. retusa and 16 samples (2 horizon × 8 plots) for P. curuense.
The analyses were conducted at the Soils and Water Laboratory of the University of Costa Rica. The following determinations were performed for each sample: pH in water (1:2); soil electrical conductivity (CE); exchangeable acidity (EA) or aluminum by titration; exchangeable calcium (Ca) and magnesium (Mg) extracted with 1 M KCl (1:10); phosphorus (P), potassium (K), zinc (Zn), iron (Fe), manganese (Mn), and copper (Cu) extracted using the modified Olsen method (pH 8.5, 1:10); and nitrogen (N) and carbon I determined by means of atomic absorption spectrophotometry. The effective cation exchange capacity (ECEC) was expressed in centimoles of charge per kilogram (cmol(+) kg−1). The proportions of clay (<2 μm), silt (2–50 μm), and sand (>50 μm) were also determined. Acidity saturation (SA) was calculated as the sum of exchangeable base cations (Ca + Mg + K) / ECEC × 100) and exchangeable acidity saturation (EA) as (Al / ECEC × 100).
2.3. Tree Sampling
A total of 3 trees per plot were randomly selected for sampling, resulting in 27 D. retusa trees and 24 P. curuense trees (Table 1). The selected trees had straight trunks, normal branching, and no visible signs of disease or pest infestation. For each selected tree, diameter at breast height (DBH), total height, and commercial height were measured using a laser hypsometer. Subsequently, at DBH, two perpendicular increment cores per tree were extracted using an increment borer that produced samples with a 4 mm diameter (Figure 2). The boring depth was determined based on the sapwood thickness reported at DBH by Tenorio et al. [28] for trees of the same species and study site. Using this value, an additional 3 cm depth was added to ensure that each core sample included the entire amount of sapwood and a portion of the heartwood. Each core sample was placed in a plastic tube to prevent drying and was later transported to the laboratory for measurement and weighing.

Figure 2.
Tree core extraction using an increment borer (a) and different tissues measured in the tree core (b).
2.4. Heartwood and Specific Gravity Determination
Heartwood proportion (HWP) was calculated in relation to the cross-sectional area. First, bark thickness and sapwood thickness were measured in each tree core using a digital caliper (±0.001 mm) (Figure 2b). Then, the heartwood radius was obtained using Equation (1), and HWP was calculated as the ratio between the heartwood area and the total cross-sectional area, expressed as a percentage (Equation (2)):
where DBH is diameter at breast height and HWP is heartwood proportion.
Specific gravity (SG) was determined using the green volume and oven-dried weight of the wood according to ASTM D-143-22 [29]. The core volume was calculated using the green diameter of the core, and the oven-dry weight was measured after drying the core for 24 h at 103 ± 2 °C. The green volume was determined by measuring the diameter and length of the core (geometric estimation), and SG was calculated using Equation (3):
2.5. Statistical Analysis
First, the normality of the data was assessed and any outliers were identified. If such values were detected, they were removed and replaced with the average of the measurements. Thereafter, a general statistical description (mean and standard deviation) was performed for all soil fertility parameters, HWP, and SG. A two-way analysis of variance (ANOVA) was then used to test differences between species and soil horizons for the soil fertility parameters. The soil fertility parameters were treated as dependent variables, while species, soil horizon, and their interaction (species × soil horizon) were considered independent variables. Mean differences between species and soil horizons were evaluated using Tukey’s test (p < 0.05), while a classification scheme based on multivariate pattern analysis techniques was applied for each species to identify plots with similar conditions. Subsequently, ANOVA was conducted for HWP and SG, with each species analyzed separately, taking plot as the independent variable and HWP or SG as the dependent variable. Mean differences between plots were again evaluated using Tukey’s test (p < 0.05), and the effect of all soil fertility parameters on HWP and SG was examined using Pearson’s correlation coefficients, where a forward stepwise regression analysis was applied to the variables that showed statistically significant correlations (p < 0.05), and the predictors were removed according to their significance. Moreover, a correlation coefficient was mathematically and graphically established for HWP and SG with the main variables that were significant in Pearson’s correlation analysis.
3. Results
3.1. Soil Fertility Characteristics
The average soil fertility parameters for the different plots and for each species are presented in Table 2, along with the F-values obtained from the ANOVA for each independent variable. Species were significant for clay and sand content, pH, Ca, Zn, Fe, Cu, C, and C/N ratio, and soil horizon was significant for K, Zn, CE, C, N, and C/N ratio, while species × soil horizon interaction was significant only for Fe (Table 2).

Table 2.
F-value of ANOVA and difference between soil fertility conditions of different plots of Dalbergia retusa and Platymiscium curuense forest plantations in Costa Rica.
Three soil texture types were identified: clay, silt, and sandy. D. retusa plantations were established mainly in sandy clay loam and clay loam soils in seven out of the eight selected sites. In contrast, P. curuense plantations were found on clay loam soils in five sites, clay soils in two sites, and sandy clay loam soil in one site. When comparing D. retusa and P. curuense, statistically significant differences were observed in clay and sand contents (Table 2). Finally, regarding the clay, silt, and sand content for each species and site, no significant differences were detected between soil horizons (Table 2).
Plantations of both species were established on slightly acidic soils. The average soil pH of the D. retusa sites was significantly lower than that of the P. curuense sites. The levels of Ca, Zn, and Cu and the C/N ratio in soils used for P. curuense showed significantly higher values compared to those for D. retusa, but the Fe content was significantly higher in soils where D. retusa was planted (Table 2). With respect to the two soil horizons, the differences can be observed in Table 2.
3.2. Heartwood and Specific Gravity
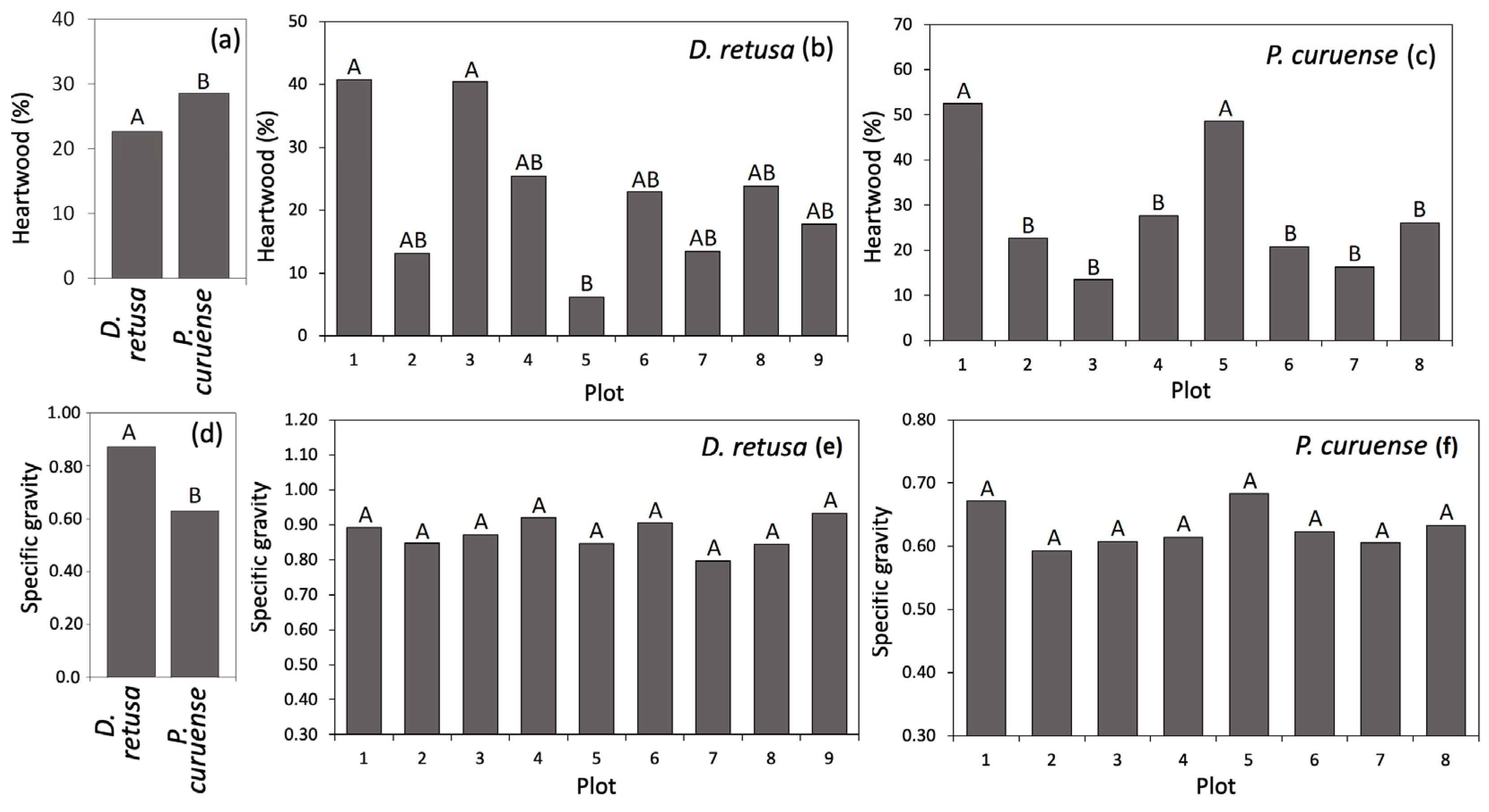
D. retusa showed a significantly lower average HWP (22.65%) than P. curuense (28.75%) (Figure 3a), and the differences between plots can be observed in Figure 3b,c. Regarding SG, D. retusa had a significantly higher average value (0.87) than P. curuense (0.63) (Figure 3d), and non-statistically significant differences were observed among plots for both species (Figure 3e,f).
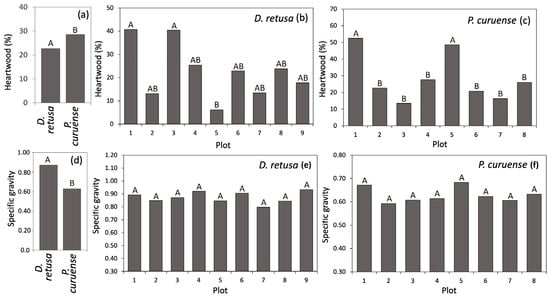
Figure 3.
Heartwood percentage and specific gravity for Dalbergia retusa and Platymiscium curuense trees from forest plantations in Costa Rica. (a) average of heartwood per species, (b) heartwood percentage for D. retusa, (c) heartwood percentage for P. curuense, (d) average of specific gravity per species, (e) specific gravity for D. retusa and (f) specific gravity for P. curuense. Legend: Different letters (A and B) for each parameter represent statistical differences between different treatments (significance of 0.05).
3.3. Effect of Soil Fertility and Morphological Properties on Heartwood and SG
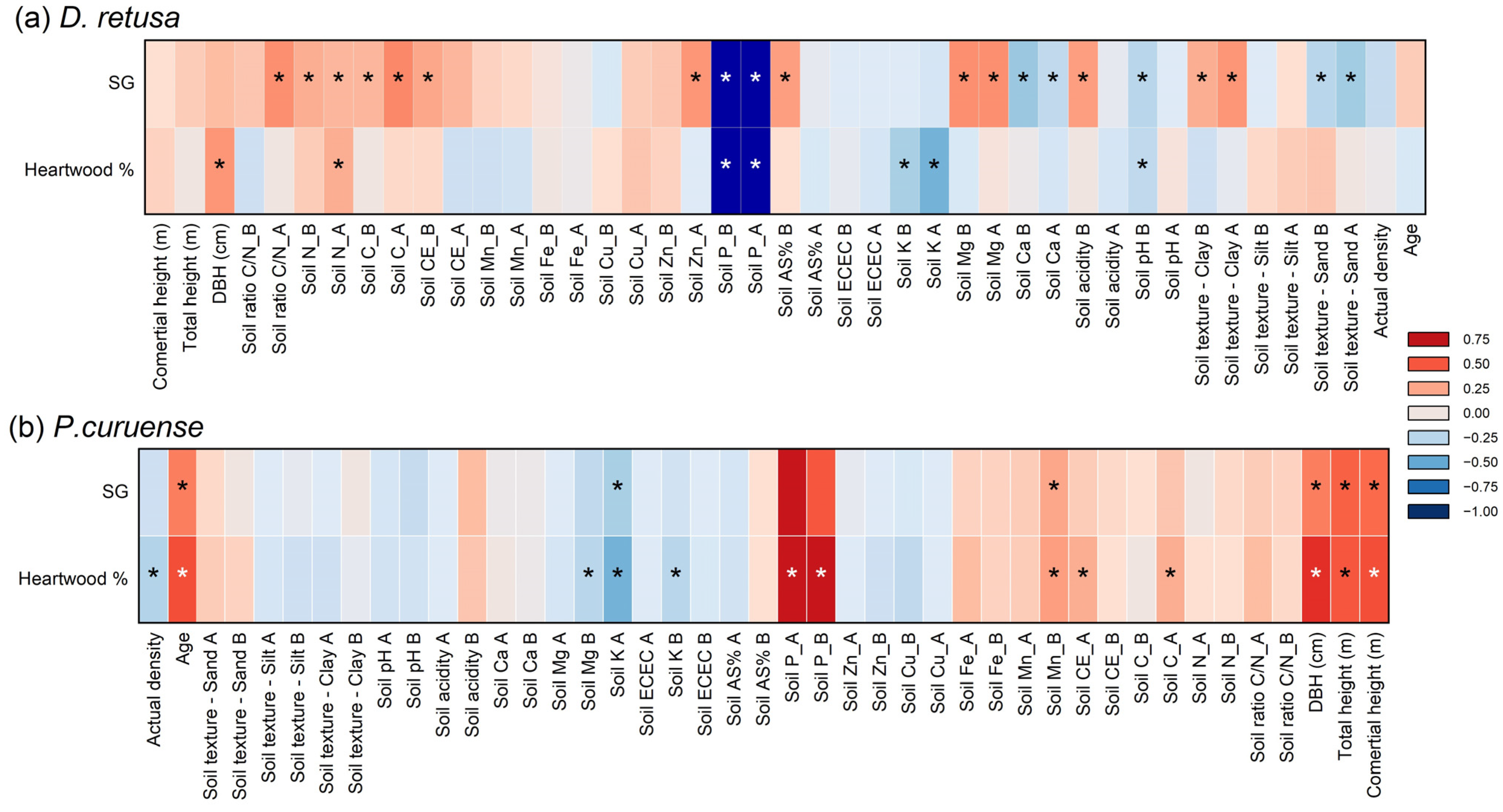
Figure 4 presents the correlation analysis between HWP and SG with soil fertility and morphological parameters for both species. Both the HWP and SG of D. retusa showed a stronger correlation with soil fertility, whereas in P. curuense, the HWP and SG were more strongly correlated with tree morphological characteristics (Figure 4).
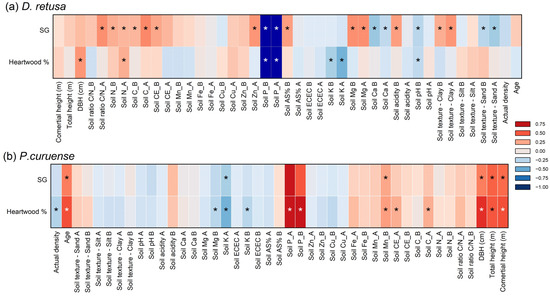
Figure 4.
Correlation coefficient of wood properties with morphological and soil fertility for Dalbergia retusa (a) and Platymiscium curuense (b) trees from forest plantations in Costa Rica. Legend: * statistically significant at 95% confidence level; first horizon; second soil horizon; soil or soil fertility parameters are non-significant for two soil horizons.
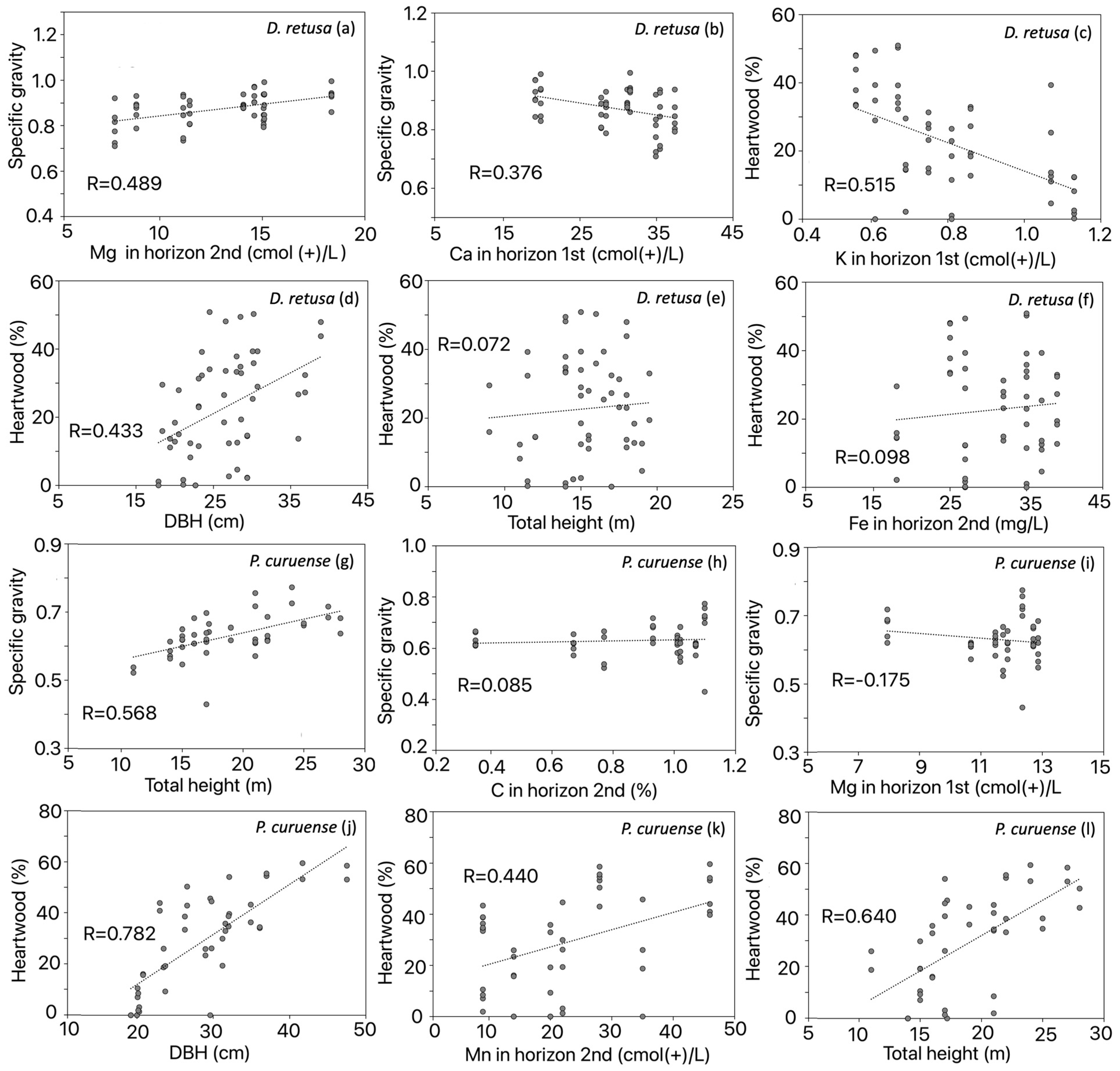
The results of the forward stepwise regression analysis with significant variables showed that Mg contributed the highest proportion to the R2 of SG, followed by Ca in the first horizon in D. retusa (Table 3). Mg was positively correlated with SG (Figure 5a), whereas Ca was negatively correlated (Figure 5b). For HWP in the same species, K explained the largest proportion of R2, followed by DBH, total height, and Fe content (Table 3). K content was negatively correlated (Figure 5c), while the DBH, total height, and Fe content in the second horizon were positively correlated with HWP (Figure 5d–f). In P. curuense, the R2 for SG was mainly explained by total height, followed by C and Mg (Table 3). Total height and C content were positively correlated with SG (Figure 5g–j), whereas Mg content was negatively correlated (Figure 5i). Finally, DBH showed the highest contribution to R2 for HWP, followed by Mn and total height (Table 4). All three factors were positively correlated with HWP (Figure 5j–l).

Table 3.
Forward stepwise regression analysis for Dalbergia retusa and Platymiscium curuense trees from forest plantations in Costa Rica.
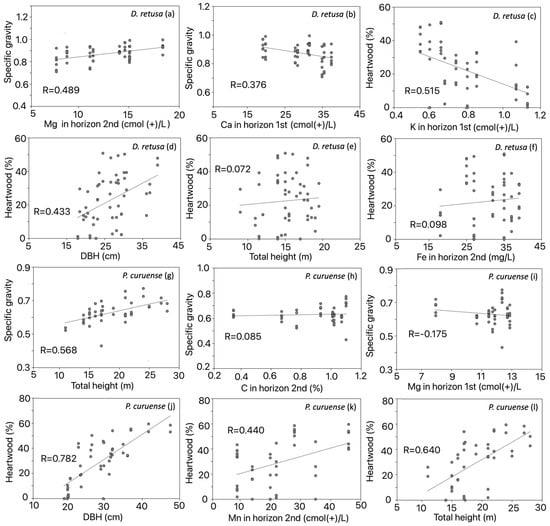
Figure 5.
Specific gravity correlation with Mg content (a) and with Ca content for D. retusa (b), heartwood correlation with K content (c), with DBH (d), with total height (e) and with Fe content in D. retusa (f), specific gravity correlation with total height (g), with C content (h) and with Mg content (i), heartwood correlation with DBH (j), with Mn content (k) and with total height (l) in P. curuense statistically significant parameters for Dalbergia retusa and Platymiscium curuense trees from forest plantations in Costa Rica.
4. Discussion
4.1. Soil Fertility
According to Camacho-Umaña et al. [30] and Mata et al. [31], the region in which the plantations were established (Parrita, Puntarenas Province, Costa Rica; Figure 1b) has four soil types: Ultisols (42%), Inceptisols (25%), Alfisols (19%), and Entisols (14%). However, based on the geographical location of the plantations (Figure 1a), Ultisols predominate, as the area comprises a foothill region. According to West et al. [32], this soil type is characterized as being clay-rich, reddish in color, and highly weathered, with a low base content and favorable physical properties. The texture, pH, EA, AS, and ECEC results obtained from laboratory analyses are consistent with the descriptions reported by West et al. [32] for these sites. Four soil textures were identified—clay, loam, sandy clay loam, and clay loam—all generally showing the presence of clay, a pH below 6.64, and an AS (acid saturation) below 2.86 (Table 2).
When evaluating the sites where the different plots of the two analyzed species were established in relation to the soil classification categories defined for Costa Rica [33], soil fertility can be classified as moderate to critical due to pH and soil acidity, which correspond to this category (Table 4). However, when considering AS, the soils are classified as critical, since the values are below 10%.

Table 4.
Parameters used for soil fertility classification in Costa Rica and comparison with the sites of D. retusa and P. curuense according to government standards.
Table 4.
Parameters used for soil fertility classification in Costa Rica and comparison with the sites of D. retusa and P. curuense according to government standards.
| Parameters | Government * | Plantation Condition | ||||
|---|---|---|---|---|---|---|
| High | Medium | Critic | D. retusa | P. curuense | ||
| Acidity parameter | pH | >6.5 | 5.5–6.5 slightly acidic | <5.5 strongly acidic | 5.4 | 6.15 |
| EA (cmol(+)/L) | >1.5 | 0.5–1.5 | <0.5 | 0.70 | 0.17 | |
| AS (%) | >50 | 10–50 | <10 | 1.78 | 0.36 | |
| Sum of bases (cmol(+)/L) | >25 | 5–25 | <5 | |||
| ECEC and Bases | ECEC (cmol(+)/L) | >25 | 5–25 | <5 | 43.00 | 46.64 |
| Ca (cmol(+)/L) | >20 | 4–20 | <4 | 29.35 | 33.78 | |
| Mg (cmol(+)/L) | >5 | 1–5 | <1 | 12.27 | 11.94 | |
| K (cmol(+)/L) | >0.6 | 0.2–0.6 | <0.2 | 0.69 | 0.75 | |
| Micronutrients | Zn (mg/L) | >10 | 2–10 | <2 | 2.86 | 2.18 |
| Mn (mg/L) | >50 | 5–50 | <5 | 25.17 | 24.44 | |
| Fe (mg/L) | >100 | 10–100 | <10 | 33.22 | 23.69 | |
| Cu (mg/L) | >20 | 2–20 | <2 | 1.83 | 4.00 | |
Note: * Information obtained from Gobierno de Costa Rica [33] and Méndez and Bertsch [34].
The evaluation of other soil parameters [33], according to Costa Rican classification criteria, indicates that the soils exhibit high fertility considering ECEC, Ca, Mg, and K values. In contrast, based on micronutrient contents, the soils are classified as having moderate fertility [33], since the measured values fall within the corresponding range (Table 4). In summary, based on the presented information, the study sites can generally be classified as having moderate-to-high fertility by Costa Rican standards [33]; however, they are characterized as being slightly to strongly acidic.
When comparing the soil fertility of the two species using the parameters established for fertility classification, it is observed that the sites of P. curuense generally present better fertility conditions than those planted with D. retusa. These sites show higher values in base sum, lower EA and AS, and a pH closer to neutrality (Table 2). Likewise, the concentrations of Ca, Zn, and Cu are statistically higher in sites where P. curuense was established, while the Fe content is lower. This result suggests that the type of species used influences the modification of soil fertility. This can be explained by the fact that each species has its own nutritional requirements and exerts distinct effects on soil properties [35]. The physical and chemical characteristics of soil vary over time due to factors such as environmental conditions and the type of vegetation present [36]. Despite these changes, they are not analyzed in detail herein, as they fall outside the scope of this study.
4.2. Wood Properties
SG is influenced by numerous factors [2], but tree age has greater influence under plantation conditions in tropical climates [21]. However, age showed little influence on SG in both species, probably due to the small age variation between them (Table 1).
Interestingly, the SG of D. retusa was strongly influenced by several soil fertility parameters (Figure 4, Table 3). In P. curuense, SG was mainly affected by morphological variables (total height, commercial height, and DBH) and by a few soil condition variables (Figure 5a, Table 3). The effect of tree morphological variables between P. curuense and D. retusa can be explained by differences in the morphology of both species [37], as well as by the distinct mechanisms trees develop to adapt and survive under varying soil conditions [38]. First, P. curuense trees exhibit greater height and diameter than those of D. retusa (Table 1). According to Woodcock and Shier [37], an increase in SG with height, as observed in P. curuense (Figure 5g), is interpreted as a higher proportion of fibers that provide mechanical support in response to stresses associated with wind load; therefore, taller individuals tend to develop higher SG. This interpretation supports the significance of height in determining SG in P. curuense (Figure 5g).
Regarding the different physiological mechanisms of trees in response to soil fertility, it has been noted that in sites with medium or low fertility (with nutrient deficiencies derived from parent rocks such as Ca, K, Mg, and P), species may differ in their nutrient acquisition and allocation strategies for wood production [38]. Mg content acts as a structural component of the cell wall and is not easily absorbed by trees compared to other nutrients [39], influencing SG via ecological strategies [40]. Sardans and Peñuelas [41] indicate that species with high SG tend to have greater longevity and must better cope with nutrient limitations than those with low SG. This could explain why Mg showed a negative correlation with SG (Figure 5i) in P. curuense, which has a low SG (Figure 3b), whereas a positive correlation between Mg and SG was observed (Figure 3a) in D. retusa, which has a high SG (Figure 3d).
The effect of soil Ca is reflected only in D. retusa, where a negative correlation is shown by SG decreasing with increasing Ca (Figure 3b). Ca in the soil is one of the most studied elements in relation to tree growth and development [42,43], with this essential macronutrient participating in plant metabolism, its absorption occurring mainly passively through the apoplast of fine roots, and its acropetal transport taking place almost exclusively via the transpiration stream [43]. Subsequently, Ca is located at multiple binding sites in the cell wall, particularly in the carboxyl groups of the middle lamella pectin [42], as well as in the flexible primary cell wall [44]. Therefore, adequate availability of this element promotes wood formation and enhances tree growth [45].
A significant relationship was observed between one of the soil fertility parameters and the SG of P. curuense, where an increase in C led to a decrease in SG (Figure 5h). Nam et al. [46] indicated that low levels of C reduce certain nutritional relationships, such as the C/N ratio, which determines the availability of N and other essential elements for plant nutrition. Specifically, in gymnosperms, a negative correlation between the C/N ratio and SG has been reported, attributed to the increase in C in the soil [46]. Moreover, Mo et al. [47] suggested that this relationship reflects a greater investment in xylem safety (increased SG) in nutrient-rich environments, where tissue construction (carbon acquisition) is a less limiting factor. This explains the negative correlation between soil C content and SG in P. curuense (Figure 5h).
Finally, it was found that SG was affected only in D. retusa trees, but not in P. curuense. Some justifications can explain this difference. Moreover, some hypotheses can be tested under controlled conditions, such as soil fertility in greenhouses or different fertilization rates. Then, a better correlation can be found, or the hypothesis that soil fertility affects SG can be tested.
It was observed that the HWP in D. retusa and P. curuense was affected by soil fertility and tree morphological variables (Figure 3). Interestingly, most variables—such as DBH and total height among morphological parameters, and Fe and Mn contents among soil fertility—positively influenced the HWP (Figure 5d–f or Figure 5 j–l), whereas an increase in K content reduced this proportion (Figure 5c).
In this regard, it is noteworthy that age does not directly determine the presence of heartwood; this is likely because in many species, the formation of this tissue occurs at early developmental stages [4,48]. In the analyzed species, it is probable that heartwood formation begins before the age of 25 years, since the sampled plantations were between 25 and 30 years old. According to Climent et al. [49], heartwood formation is not a singular event occurring at a specific age, but rather a process that begins once the tree reaches a certain diameter. This may explain the observed relationship between HWP and DBH: in D. retusa, heartwood formation starts when the tree reaches a DBH between 12 and 15 cm (Figure 5d), whereas in P. curuense, this process occurs at larger diameters, between 18 and 20 cm (Figure 5j).
As in many species, the increase in HWP shows a positive and proportional relationship with tree diameter growth [50], a pattern also observed in the two species studied (Figure 5d,j). This increase in heartwood associated with diameter growth is attributed to a regulatory mechanism controlling the thickness of the heartwood and sapwood, maintaining a specific ratio according to the conductive requirements of the crown [51]. This process enhances the physiological activity of the tree and the demand for starch and other substances, which are subsequently involved in the programmed cell death of secondary xylem cells [50].
Tree height showed a positive and proportional effect on HWP in both species (Figure 5e,l). Taller trees require a greater amount of biomass to support their structure [37]. However, during ontogeny, trees must cope with the negative effects of increasing height, such as reduced water transport efficiency, decreased metabolic activity, and the need to allocate more carbon to the maintenance of a growing xylem biomass [52]. Nevertheless, the amount of sapwood, the number of sapwood rings, and their thickness remain relatively constant in many species, keeping the sapwood–heartwood transition stable [53]. Therefore, as tree diameter increases due to greater height while sapwood thickness remains constant, the amount of HWP increases, as found in the present study (Figure 5e,l), showing a significant influence on its formation (Table 3).
The different types of micronutrients present in the soil are numerous, and each one, individually or in combination, influences tree growth [23]. In the specific case of the two analyzed species (D. retusa and P. curuense), the increase in diameter may be masking the effects of microelements on HWP. This morphological variable contributes more strongly to the variation in this tissue than the microelements do (Table 3). It was observed that only K (negative effect) and Fe influenced HWP in D. retusa (Figure 5c,f), while Mn affected the HWP of P. curuense (Figure 5k). It is expected that sites with better soil fertility conditions will exhibit greater diameter growth in trees [13] and, consequently, an increase in both diameter and HWP [23]. According to Table 3, increased availability of Fe and Mn improves soil fertility; therefore, greater diameter development and higher HWP can be expected, as evidenced by the increase in this parameter with higher Fe content in D. retusa (Figure 5f) and Mn content in P. curuense (Figure 5k).
Fe is one of the most important soil components, as it can significantly influence and actively participate in xylem formation processes. However, both excess and deficiency of Fe can cause plant diseases or alter tree growth [53,54], with its effects varying considerably due to its mobility changes with redox conditions and soil acidity levels [55,56]. The increase in tree diameter and development, and consequently in heartwood formation, can be attributed to adequate Fe availability, together with low pH and appropriate organic matter content [57].
Manganese (Mn) participates in numerous metabolic processes, such as photosynthesis, respiration, and the synthesis of fatty acids and proteins, as well as in enzymatic activation [58]. Moreover, Mn plays a key role in water-splitting reactions by providing the electrons necessary for photosynthesis, and it acts as a cofactor for various enzymes. It is also essential in multiple stages of secondary-metabolite biosynthesis, such as in lignans, flavonoids, cinnamic acids, and acyl lipids [59]. Mn is required in very low concentrations, typically between 20 and 40 mg per kilogram of dry weight; however, it is also considered a heavy metal that can be toxic to plants at excessive levels [60]. In the studied sites, Mn concentrations were below 50 mg/L—a range in which its variation promoted increased growth and, consequently, a higher HWP in P. curuense (Figure 5k).
Interestingly, it was observed that an increase in K content decreases HWP (Figure 4a), being the variable that contributed the most to the variation observed for D. retusa (Table 4). This is noteworthy, since according to the concentrations obtained from the soils where this species was planted (Table 2), the sites are classified as having good fertility based on the categorization established for Costa Rican soils [33]. In this context, it would be expected that an increase in K would promote an increase in both tree diameter and HWP. K is essential for multiple physiological processes, including enzyme activation, photosynthesis, carbohydrate metabolism, and protein synthesis [61], and its availability directly affects sapwood function, which subsequently influences heartwood formation [50]. However, one of the main issues is that only about 2–3% of the soil potassium is available to plants in a free soluble form, as the remainder is bound to soil minerals, representing approximately 95% of the total potassium [62]. Furthermore, its availability depends on multiple cationic interactions, both with other elements and with itself [63]. Therefore, this low effective availability could explain the observed decrease in heartwood proportion with increasing total K concentration in the soil.
This result showed that soil fertility, although low in some areas for growing P. curuense, was appropriate. Therefore, when establishing commercial plantations of these species, it is important to conduct soil studies to ensure conditions similar to or better than those reported in the present study. SG and HWP are two important wood properties, and it was appropriate to sample older plantations (close to the rotation schedule). However, these properties are unknown in thinned trees, so are necessary to determine. Another important property for the commercialization of this wood is heartwood color [3,5], making it crucial to evaluate this property in trees near the rotation schedule or trees on different thinning schedules.
5. Conclusions
D. retusa and P. curuense plantations were established on soils with four texture types (clay, loam, sandy clay loam, and sand), generally characterized by a high clay content, a pH below 6.64, and soluble acidity (SA) values lower than 2.86. These conditions classify the sites as having medium-to-high fertility in Costa Rica; however, the soils are classified as slightly to strongly acidic. When comparing the soils from the areas of both species, the sites where P. curuense was established generally exhibited better fertility than those of D. retusa. The P. curuense sites showed higher base saturation, lower acidity and acid saturation, and a more neutral pH. Likewise, the Ca, Zn, and Cu content was statistically higher in soils planted with P. curuense.
The wood SG of the two species studied was distinctly affected by soil fertility conditions and tree morphological variables. In D. retusa, SG was primarily determined by soil fertility conditions, whereas in P. curuense, SG depended on both morphological and soil fertility variables (total height, C, and Mg). The HWP of D. retusa and P. curuense was influenced by both soil fertility and morphological variables. Most variables—such as DBH and total height (morphological parameters), as well as Fe and Mn content (fertility conditions)—had a positive effect on HWP, while increased K content reduced it. Overall, these results indicate that heartwood development occurs mainly because of greater tree growth.
According to previous studies, two wood properties were examined in older plantations and trees close to the rotation schedule. However, the wood properties of thinned trees are unknown, so it is necessary that these are determined for this type of tree. In addition, the two species studied are commercially important due to their wood color, making it crucial to evaluate this property in trees of these ages.
Author Contributions
Conceptualization, R.M. and C.T.; methodology, R.M., R.L. and J.C.; validation, R.L.; formal analysis, R.M. and C.T.; investigation, R.M. and C.T.; resources, R.L.; data curation, R.L. and J.C.; writing—original draft preparation, R.M. and C.T.; writing—review and editing, R.M. and C.T.; supervision, J.C.; funding acquisition, R.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data presented in this article were deposited in https://doi.org/10.18845/RDA/OW1K8Q. (Accessed on 25 January 2026).
Acknowledgments
The authors wish to thank the Vicerrectoría de Investigación y Extensión of the Instituto Tecnológico de Costa Rica (ITCR) for the financial support given for this research.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| EA | exchangeable acidity |
| Ca | calcium |
| Mg | magnesium |
| P | phosphorus |
| K | potassium |
| Zn | zinc |
| Fe | iron |
| Mn | manganese |
| Cu | copper |
| N | nitrogen |
| C | carbon |
| ECEC | effective cation exchange capacity |
| AS | exchangeable acidity saturation |
| CE | soil electrical conductivity |
References
- Arisandi, R. Effect of heartwood proportion in wood properties. Wood Res. J. 2021, 12, 41–45. [Google Scholar] [CrossRef]
- Yang, S.; Qin, F.; Wang, S.; Li, X.; Zhou, Y.; Meng, S. Advances in the study of heartwood formation in trees. Life 2025, 15, 93. [Google Scholar] [CrossRef]
- Moya, R.; Bond, B.; Quesada, H. A review of heartwood properties of Tectona grandis trees from fast-growth plantations. Wood Sci. Technol. 2014, 48, 411–433. [Google Scholar] [CrossRef]
- Nakaba, S.; Funada, R. Cell death of long-lived ray parenchyma cells during heartwood formation in trees. J. Wood Sci. 2024, 70, 46. [Google Scholar] [CrossRef]
- Kirker, G.T.; Hassan, B.; Mankowski, M.E.; Eller, F.J. Critical review on the use of extractives of naturally durable woods as natural wood protectants. Insects 2024, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y. Wood specific gravity–mechanical property relationship at species level. Wood Sci. Technol. 1997, 31, 181–191. [Google Scholar] [CrossRef]
- Williamson, G.B.; Wiemann, M.C. Measuring wood specific gravity… correctly. Am. J. Bot. 2010, 97, 519–524. [Google Scholar] [CrossRef]
- Fichtler, E.; Worbes, M. Wood anatomical variables in tropical trees and their relation to site conditions and individual tree morphology. Iawa J. 2012, 33, 119–140. [Google Scholar] [CrossRef]
- Woodcock, D.; Shier, A. Wood specific gravity and its radial variations: The many ways to make a tree. Trees 2002, 16, 437–443. [Google Scholar] [CrossRef]
- Zobel, B.J.; van Buijtenen, J.P. Wood Variation and Wood Properties; Springer: Dordrecht, The Netherlands; New York, NY, USA, 1989; pp. 1–32. [Google Scholar]
- Thibaut, B.; Gril, J. Tree growth forces and wood properties. Peer Community J. 2021, 1, e46. [Google Scholar] [CrossRef]
- Bredemeier, M.; Busch, G.; Hartmann, L.; Jansen, M.; Richter, F.; Lamersdorf, N.P. Fast growing plantations for wood production—Integration of ecological effects and economic perspectives. Front. Bioeng. Biotechnol. 2021, 3, 72. [Google Scholar] [CrossRef]
- Wegner, T.; Skog, K.E.; Ince, P.J.; Michler, C.J. Uses and desirable properties of wood in the 21st century. J. For. 2010, 108, 165–173. [Google Scholar] [CrossRef]
- Tarelkina, T.V.; Galibina, N.A.; Ershova, M.A.; Moshnikov, S.A.; Nikerova, K.M.; Afoshin, N.V.; Semenova, L.I. Age of heartwood initiation in trunks of young Pinus sylvestris L. trees in the taiga zone. IAWA J. 2023, 45, 375–390. [Google Scholar] [CrossRef]
- Meunpong, P.; Penboon, C.; Kuasakun, N.; Wachrinrat, C. Tree dimension and environmental correlates of heartwood content in Siamese rosewood (Dalbergia cochinchinensis). Biodiversitas 2021, 22, 3297–3303. [Google Scholar] [CrossRef]
- Pande, P.K.; Singh, M. Inter-clonal, intra-clonal, and single tree variations of wood anatomical properties and specific gravity of clonal ramets of Dalbergia sissoo Roxb. Wood Sci. Technol. 2005, 39, 351–366. [Google Scholar] [CrossRef]
- Ghorbanian, F.M.; Ashrafi, M.; Shaabani, A.H.; Moghadam, A.Y.; Bari, E.; Niemz, P.; Hosseinpourpia, R.; Ribera, J. Physical and mechanical properties of different beech wood species grown at various climate conditions: A review. Holzforschung 2024, 78, 377–386. [Google Scholar] [CrossRef]
- Rocha, M.F.V.; Veiga, T.R.L.A.; Soares, B.C.D.; Araújo, A.C.C.D.; Carvalho, A.M.M.; Hein, P.R.G. Do the growing conditions of trees influence the wood properties? Floresta E Ambiente 2019, 26, e20180353. [Google Scholar] [CrossRef]
- Onyekwelu, J.C.; Stimm, B.; Evans, J. Review plantation forestry. In Silviculture in the Tropics. Tropical Forestry; Günter, S., Weber, M., Stimm, B., Mosandl, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 8. [Google Scholar] [CrossRef]
- Badalamenti, E.; Sferlazza, S.; Veca, D.S.L.M.; Maetzke, F.; Sala, G.; La Mantia, T. The evolution in time of the concept of fast growing tree species: Is it possible to use a definition applicable to all environmental conditions? Ann. Silvicult. Res. 2020, 45, 53–61. [Google Scholar] [CrossRef]
- Pati, P.K.; Kaushik, P.; Khan, M.L.; Khare, P.K. Inventory of wood specific gravity for Indian forests. Tree For. People 2025, 19, 100775. [Google Scholar] [CrossRef]
- Sseremba, O.E.; Mugabi, P.; Banana, A.Y.; Wessels, B.C.; Plessis, M. Variation of basic density, calorific value, and volumetric shrinkage within tree height and tree age of Ugandan grown Eucalyptus grandis wood. J. For. Res. 2021, 32, 503–512. [Google Scholar] [CrossRef]
- Klitgaard, B.B. Pterocarpum (Leguminosae: Dalbergieae): Biogeography, systematics, morphology, taxonomy and uses. Kew Bull. 2005, 60, 321–400. Available online: https://www.jstor.org/stable/4111062 (accessed on 26 January 2026).
- Rubilar, R.A.; Lee Allen, H.; Fox, T.R.; Cook, R.L.; Albaugh, T.J.; Campoe, O.C. Advances in silviculture of intensively managed plantations. Curr. For. Rep. 2018, 4, 23–34. [Google Scholar] [CrossRef]
- Jiménez, Q. Árboles Maderables en Peligro de Extinción en Costa Rica, 1st ed.; Incafo: San José, Costa Rica, 1993. [Google Scholar]
- Sunny, P.P.; Dutt, B.; Sharma, K.R.; Dhiman, B.; Sumthane, Y.Y. Wood variation in physico-mechanical properties of Dalbergia sissoo Roxb. ex DC. from local markets of Himachal Pradesh. J. Trop. Agric. 2019, 57, 17–26. [Google Scholar]
- Pande, P.; Rawat, L. Effect of growth on wood traits in seed-raised plantations of Dalbergia sissoo Roxb. J. Forest Res. 2015, 26, 241–245. [Google Scholar] [CrossRef]
- Tenorio, C.; Lujan, R.; Corrales, J.; Moya, R. Wood properties of Dalbergia retusa and Platymiscium curuense from forest plantations in Costa Rica. Eur. J. Wood Wood Prod. 2026; submitted. [Google Scholar]
- ASTM D-143-22; ASTM Standard Methods of Testing Small Clear Specimens of Timber, 2025. ASTM: West Conshohocken, PA, USA, 2016; Volume 4.10. [CrossRef]
- Camacho-Umaña, M.E.; Quesada-Román, A.; Villatoro-Sánchez, M.; Alemán-Montes, B.; Mata, R.; Henríquez-Henríquez, C.; Céspedes-Rivera, J.; Céspedes-Rivera, M.; Alvarado, A. Agricultural landscapes of Costa Rica. In Landscapes and Landforms of Costa Rica; Springer International Publishing: Cham, Switzerland, 2024; pp. 221–258. [Google Scholar]
- Mata, R.; Rosales, A.; Sandoval, D.; Vindas, E.; Alemán, B. Mapa de Órdenes de Suelos de Costa Rica, 2022; Escala 1:200,000; Universidad de Costa Rica, Centro de Investigaciones Agronómicas: San José, Costa Rica, 2022. [Google Scholar]
- West, L.T.; Beinroth, F.H.; Sumner, M.E.; Kang, B.T. Ultisols: Characteristics and impacts on society. Adv. Agron. 1997, 63, 179–236. [Google Scholar] [CrossRef]
- Gobierno de Costa Rica. Decreto Ejecutivo N° 41960-MAG-MINAE. Establecimiento de la Metodología Para la Determinación de la Capacidad de Uso de las Tierras Agroecológicas de Costa Rica; La Gaceta: Diario Oficial del Gobierno de Costa Rica 2019; Imprenta Nacional: San José, Costa Rica, 2019. [Google Scholar]
- Méndez, J.C.; Bertsch, F. Guía Para la Interpretación de la Fertilidad de los Suelos de Costa Rica; ACCS: San José, Costa Rica, 2012. [Google Scholar]
- Marais, A.; Hardy, M.; Booyse, M.; Botha, A. Effects of monoculture, crop rotation, and soil moisture content on selected soil physicochemical and microbial parameters in wheat fields. Appl. Environ. Soil Sci. 2012, 2012, 593623. [Google Scholar] [CrossRef]
- Eswaran, H.; Reich, P.F. World soil map. In Encyclopedia of Soils in the Environment; Hillel, D., Ed.; Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2005; pp. 352–365. [Google Scholar] [CrossRef]
- Woodcock, D.; Shier, A. Does canopy position affect wood specific gravity in temperate forest trees? Ann. Bot. 2003, 91, 529–537. [Google Scholar] [CrossRef]
- Heineman, K.D.; Turner, B.L.; Dalling, J.W. Variation in wood nutrients along a tropical soil fertility gradient. New Phytol. 2016, 211, 440–454. [Google Scholar] [CrossRef]
- González-Melo, A.; Posada, J.M.; Beauchêne, J.; Lehnebach, R.; Clair, B. Tropical tree species with high wood specific gravity have higher concentrations of wood phosphorus and are more efficient at resorbing it. AoB Plants 2025, 17, plaf001. [Google Scholar] [CrossRef]
- Urbina, I.; Grau, O.; Sardans, J.; Margalef, O.; Peguero, G.; Asensio, D.; Llusià, J.; Ogaya, R.; Gargallo-Garriga, A.; Van Langenhove, L.; et al. High foliar K and P resorption efficiencies in old-growth tropical forests growing on nutrient-poor soils. Ecol. Evol. 2021, 11, 8969–8982. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. Trees increase their P:N ratio with size. Glob. Ecol. Biogeogr. 2014, 24, 147–156. [Google Scholar] [CrossRef]
- Fromm, J. Wood formation of trees in relation to potassium and calcium nutrition. Tree Physiol. 2010, 30, 1140–1147. [Google Scholar] [CrossRef]
- Lautner, S.; Ehlting, B.; Windeisen, E.; Rennenberg, H.; Matyssek, R.; Fromm, J. Calcium nutrition has a significant influence on wood formation in poplar. New Phytol. 2007, 173, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, A.; Ozyhar, T. Calcium effects on structure and density of eucalyptus wood from Colombian plantation. For. Sci. 2025, 71, 123–137. [Google Scholar] [CrossRef]
- Pretzsch, H.; Biber, P.; Schütze, G.; Kemmerer, J.; Uhl, E. Wood density reduced while wood volume growth accelerated in Central European forests since 1870. For. Ecol. Manag. 2018, 429, 589–616. [Google Scholar] [CrossRef]
- Nam, V.T.; Anten, N.P.R.; van Kuijk, M. Biomass dynamics in a logged forest: The role of wood density. J. Plant Res. 2018, 131, 611–621. [Google Scholar] [CrossRef]
- Mo, L.; Crowther, T.W.; Maynard, D.S.; Van den Hoogen, J.; Ma, H.; Bialic-Murphy, L.; Liang, J.; de-Miguel, S.; Nabuurs, G.-J.; Reich, P.B.; et al. The global distribution and drivers of wood density and their impact on forest carbon stocks. Nat. Ecol. Evol. 2024, 8, 2195–2212. [Google Scholar] [CrossRef]
- Taylor, A.M.; Gartner, B.L.; Morrell, J.J. Heartwood formation and natural durability: A review. Wood Fiber Sci. 2002, 34, 587–611. [Google Scholar]
- Climent, J.; Chambel, M.R.; Pérez, E.; Gil, L.; Pardos, J. Relationship between heartwood radius and early radial growth, tree age, and climate in Pinus canariensis. Can. J. For. Res. 2002, 32, 103–111. [Google Scholar] [CrossRef]
- Kampe, A.; Magel, E. New insights into heartwood and heartwood formation. In Cellular Aspects of Wood Formation; Fromm, J., Ed.; Plant Cell Monographs; Springer: Berlin/Heidelberg, Germany, 2013; Volume 20, pp. 71–95. [Google Scholar] [CrossRef]
- Knapic, S.; Tavares, F.; Pereira, H. Heartwood and sapwood variation in Acacia melanoxylon R. Br. trees in Portugal. Forestry 2006, 79, 371–380. [Google Scholar] [CrossRef]
- Petit, G.; Mencuccini, M.; Carrer, M.; Prendin, A.L.; Hölttä, T. Axial conduit widening, tree height, and height growth rate set the hydraulic transition of sapwood into heartwood. J. Exp. Bot. 2023, 74, 5072–5087. [Google Scholar] [CrossRef]
- Beauchamp, K.; Mencuccini, M.; Perks, M.; Gardiner, B. The regulation of sapwood area, water transport and heartwood formation in Sitka spruce. Plant Ecol. Diver. 2013, 6, 45–56. [Google Scholar] [CrossRef]
- Olifir, Y.; Partyka, T.; Havryshko, O.; Konyk, H.; Panakhyd, H.; Kozak, N.; Ivaniuk, V. Iron content and qualitative composition in a waterlogged agricultural soil under long-term agrogenic influence, Western Ukraine. Agron. Res. 2025, 23, 1251–1265. [Google Scholar] [CrossRef]
- Fuss, C.B.; Driscoll, C.T.; Johnson, C.E.; Petras, R.J.; Fahey, T.J. Dynamics of oxidized and reduced iron in a northern hardwood forest. Biogeochemistry 2011, 104, 103–119. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Singh, S.; Gaur, S.; Singh, S.; Yadav, V.; Liu, S.; Singh, V.P.; Sharma, S.; Srivastava, P.; Prasad, S.M.; et al. Acquisition and homeostasis of iron in higher plants and their probable role in abiotic stress tolerance. Front. Environ. Sci. 2018, 5, 86. [Google Scholar] [CrossRef]
- Wei, X.; Shao, M.; Zhuang, J.; Horton, R. Soil iron fractionation and availability at selected landscape positions in a loessial gully region of northwestern China. J. Soil Sci. Plant Nutr. 2010, 56, 617–626. [Google Scholar] [CrossRef]
- Li, J.; Jia, Y.; Dong, R.; Huang, R.; Liu, P.; Li, X.; Wang, Z.; Liu, G.; Chen, Z. Advances in the mechanisms of plant tolerance to manganese toxicity. Int. J. Mol. Sci. 2019, 20, 5096. [Google Scholar] [CrossRef]
- Lidon, F.C.; Barreiro, M.G.; Ramalho, J.C. Manganese accumulation in rice: Implications for photosynthetic functioning. J. Plant Physiol. 2004, 161, 1235–1244. [Google Scholar] [CrossRef] [PubMed]
- Millaleo, R.; Reyes-Díaz, M.; Ivanov, A.G.; Mora, M.L.; Alberdi, M. Manganese as essential and toxic element for plants: Transport, accumulation and resistance mechanism. J. Soil Sci. Plant Nutr. 2010, 10, 470–481. [Google Scholar] [CrossRef]
- Soumare, A.; Djibril, S.A.R.R.; Diedhiou, A.G. Potassium sources, microorganisms and plant nutrition: Challenges and future research directions. Pedosphere 2023, 33, 105–115. [Google Scholar] [CrossRef]
- Olaniyan, F.T.; Alori, E.T.; Adekiya, A.O.; Ayorinde, B.B.; Daramola, F.Y.; Osemwegie, O.O.; Babalola, O.O. The use of soil microbial potassium solubilizers in potassium nutrient availability in soil and its dynamics. Annals Microbiol. 2022, 72, 45. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. Potassium control of plant functions: Ecological and agricultural implications. Plants 2021, 10, 419. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.