Abstract
The influence of forest type on soil aggregates distribution, stability, and the contribution of aggregate-associated carbon (C) to bulk soil organic carbon (SOC) remains poorly understood. This may be crucial for the accumulation and persistence of SOC in subtropical ecosystems. In this study, we examined soil aggregate distribution and stability at two depths (0–15 and 15–30 cm) in 10-, 20-, and 30-year-old Cryptomeria japonica (Japanese cedar) and Chimonobambusa quadrangularis (square bamboo) plantations. We further assessed the contribution of carbon (C) associated with distinct aggregate fractions to bulk SOC. Across all stand ages and soil depths, macroaggregates accounted for 19%–56% of total soil aggregates in Japanese cedar plantations, whereas their proportion was 30%–337% higher in square bamboo plantations. In contrast, fine aggregates constituted 3%–67% of total aggregates in Japanese cedar plantations, but were 29%–94% lower in square bamboo plantations than in Japanese cedar plantations. Compared with Japanese cedar plantations, aggregate mean weight diameter (MWD) and geometric mean diameter (GMD) increased by 17%–88% and 35%–152%, respectively, in square bamboo plantations. In Japanese cedar soils, C and nitrogen (N) were primarily concentrated in coarse macroaggregates and fine macroaggregates, whereas in square bamboo plantations, C and N were mainly associated with coarse macroaggregates only. Both aggregate-associated soil C and N varied significantly with aggregate size and forest type, and Japanese cedar soils exhibited higher aggregate C/N ratios, particularly in older stands. Bulk SOC was positively correlated with macroaggregate-associated C in both forest types and with the silt and clay fractions in Japanese cedar plantations. MWD increased with higher macroaggregate C content and declined as the proportion of C in smaller aggregate fractions increased. These findings indicate that forest type plays a critical role in regulating soil aggregation and SOC stabilization pathways, with square bamboo plantations enhancing C sequestration by promoting macroaggregate formation and stability.
1. Introduction
Soils, particularly those in forest ecosystems, represent the largest reservoirs of organic carbon (C) in terrestrial ecosystems [1]. Globally, soil C stocks are estimated to be two to three times greater than those stored in aboveground vegetation and in the atmosphere [2]. Soil CO2 efflux to the atmosphere exceeds anthropogenic emissions by more than sevenfold [3]. Even minor variations in soil C can substantially alter soil function and ecosystem services [4]. A comprehensive understanding of soil organic carbon (SOC) preservation is essential for implementing management practices aimed at maximizing SOC accumulation [5,6]. Forest type strongly regulates the accumulation and persistence of SOC, mainly through the effects of tree species composition on litter input and the soil microenvironment [7,8]. However, the impacts of different forest types remain insufficiently understood, as large uncertainties persist regarding the magnitude and direction of SOC responses to multiple interacting factors such as soil properties, stand age, and environmental conditions [9].
Soil aggregates, as primary structural units, play a crucial role in maintaining soil structure and regulating the nutrient cycling, organic matter turnover, and energy flow [10,11,12]. The formation of aggregate provides physical protection for SOC by encapsulating organic matter within mineral matrices and limiting microbial decomposition [13,14,15]. Based on the particle size, soil aggregates are commonly categorized into macroaggregates (>0.25 mm) and microaggregates (<0.25 mm). Macroaggregates can be further divided into coarse macroaggregates (>2 mm) and fine macroaggregates (0.25–2 mm), while microaggregates consist of particles ranging from 0.053 to 0.25 mm, and silt and clay fractions are <0.053 mm [16,17]. Across most terrestrial ecosystems, macroaggregates dominate the aggregate mass, accounting for 19%–56% of total soil aggregates in coniferous plantations, whereas microaggregates are less abundant but play a disproportionally important role in SOC stabilization by forming stable organo-mineral complexes [18,19,20]. Soil depth and vegetation type strongly influence both aggregate distribution and SOC stabilization. In surface soils (0–15 cm), higher biological activity, root density, and organic inputs promote macroaggregate formation and stability, whereas subsurface layers (15–30 cm) tend to have smaller aggregates and lower SOC protection. Vegetation type affects aggregate-associated C through differences in plant functional characteristics, including growth form, root patterns, litter quality, and litter quantity [21]. For example, broadleaved and bamboo forests exhibit higher macroaggregate-associated SOC than coniferous plantations [22]. Soil disturbance and land-use change can markedly alter aggregate size distribution, often reducing macroaggregate abundance and total SOC content [23]. Understanding how soil depth and forest type regulate aggregate distribution, stability, and the allocation of C across aggregate fractions is therefore essential for improving SOC sequestration, sustaining soil health, and maintaining ecosystem stability in subtropical forests.
Soil aggregate stability is an essential indicator of soil structure and plays a crucial role in regulating C sequestration and nutrient dynamics [18,24]. Enhanced aggregate stability improves the soil structure, fertility, and resistance to erosion [25]. Several indicators are commonly used to evaluate soil aggregate stability, with mean weight diameter (MWD) and geometric mean diameter (GMD) being the most widely applied [23,26]. Growing evidence suggests that tree species composition profoundly alters soil structure and properties, thereby affecting soil aggregate stability [27,28,29]. Understanding how different tree species modulate aggregate stability is fundamental for elucidating the mechanisms governing soil C and N cycling. Chen et al. [30] demonstrated that tree species with deeper root systems tended to enhance soil aggregate stability more effectively than those with shallow roots because of their greater capacity to improve soil structure and promote organic matter incorporation. Moreover, the quantity and chemical quality of litter input (e.g., C/N ratio and the relative abundance of labile versus recalcitrant organic compounds) varies among tree species, influencing the formation and stabilization of aggregates by stimulating microbial activity and the production of organic binding agents that cement the soil particles together [31]. Thus, investigating how different forest types affect soil aggregate stability can provide valuable insights into the processes driving SOC accumulation and stabilization across forest ecosystems.
In Muchuan County, Southwest China, Cryptomeria japonica (Japanese cedar) has historically been the dominant tree species. However, owing to the high economic value and rapid expansion of Chimonobambusa quadrangularis (square bamboo), large areas of Japanese cedar plantations have been converted into square bamboo stands over recent decades. This large-scale forest conversion provides a valuable opportunity to examine how shifts in forest type influence soil structural properties and C stabilization mechanisms. We hypothesized that: (1) square bamboo plantations would exhibit a higher proportion of macroaggregates and greater overall aggregate stability than Japanese cedar plantations; (2) bulk SOC is primarily contained within larger aggregate fractions, which contribute more to total SOC than smaller fractions in both square bamboo and Japanese cedar plantations; and (3) bulk SOC content and aggregate stability would be significantly related to soil aggregate-associated C content. To test these hypotheses, we aimed to (1) quantify soil aggregate distribution and stability at depths of 0–15 and 15–30 cm in 10-, 20-, and 30-year-old Japanese cedar and square bamboo plantations; (2) evaluate the contributions of C associated with different aggregate size fractions to bulk soil C; and (3) explore the relationships between aggregate-associated C content, bulk soil C, and aggregate stability.
2. Materials and Methods
2.1. Site Description
The study was conducted in Muchuan County (103°47′–103°49′ E, 28°29′–28°54′ N), Leshan city, southern Sichuan Province, China. This region is characterized by a typical humid subtropical monsoon climate, with a mean annual temperature of 12.8 °C and mean annual precipitation of 1780 mm. The geomorphology is predominantly mountainous, with elevations ranging from 1100 m to 1550 m and slope gradients of approximately 25–35°. Subtropical evergreen broadleaved forests and coniferous forests dominate the natural vegetation, while Acrisols constitute the primary soil type according to WRB soil classification. The soil pH ranges from 4.69 to 8.64, which was determined at a soil-to-water ratio of 1:5 (w:v) with a pH electrode (Starter 300; Ohaus, Parsippany, NJ, USA), reflecting moderate acidity to near-neutral conditions. Japanese cedar is one of the principal afforested species in this region. Square bamboo is an excellent bamboo species in China that is valued for its shoots and culms and possesses high economic potential. It is well-adapted to local environmental conditions; therefore, large areas of Japanese cedar plantations have been cut and replaced with square bamboo. At present, bamboo forests account for more than 50% of the total plantation area in Muchuan County.
2.2. Experimental Design and Soil Sampling
In July 2025 (summer), following a comprehensive survey of forest stands in the study region, 10-, 20-, and 30-year-old Japanese cedar and square bamboo plantations with comparable geographical locations and environmental conditions were selected for investigation. All selected plantations had been continuously managed without thinning operations or harvesting activities, and no rotation-based clear-cutting was implemented during this period. Therefore, the stands represent relatively stable plantation systems with minimal management disturbance. The basic characteristics of the selected plantations and the principal physicochemical properties of the surface soil (0–30 cm) are summarized in Table 1.

Table 1.
Basic characteristics of Japanese cedar and square bamboo plantations of different stand ages (10, 20, and 30 years) and principal physicochemical properties of surface soils (0–30 cm) in southern Sichuan Province, China.
Annual litterfall was collected using 1 m × 1 m litter traps placed randomly within each plot. Litter was collected monthly, oven-dried at 65 °C to constant weight, and weighed. Annual litter production (t·ha−1·yr−1) was calculated by summing the monthly dry weights and converting to per-hectare basis.
For each forest type and stand age, three independent plantations were selected as true field replicates. Within each plantation, three 5 × 5 m plots with similar slopes and aspects were established, with a minimum distance of 20 m between adjacent plots to ensure spatial independence. To assess how forest types influenced soil aggregates, soil samples were collected from the 0–15 and 15–30 cm layers at three randomly selected locations within each plot. At each location, soils were extracted using a stainless-steel cylinder (1178 cm3). To minimize aggregate disturbance, three subsamples from the same depth were mixed to form a composite sample. These samples were air-dried for aggregate separation and subsequent SOC and nitrogen (N) concentration analyses. For aggregate separation, the air-dried soils were carefully broken along natural structural lines into clods approximately 1 cm in size. The resulting material was fractionated into four aggregate classes following the modified method of Six et al. [32].
2.3. Soil Aggregates, Soil Organic Carbon, and Nitrogen Analysis
Using the wet sieving method [33], aggregates were separated into four size classes: coarse macroaggregates (>2 mm), fine macroaggregates (0.25–2 mm), microaggregates (0.053–0.25 mm), and silt and clay fractions (<0.053 mm). Approximately 100 g of air-dried soil was loaded onto the top screen of a sieve stack containing 2, 0.25, and 0.053 mm meshes. Prior to sieving, samples were pre-wetted by capillary action for 30 min to minimize slaking. The sieves were immersed in deionized water at room temperature (22 ± 2 °C) and vertically oscillated with a 3 cm amplitude at 50 cycles per minute for 4 min. After sieving, the materials retained on each sieve and the silt and clay fractions (<0.053 mm) that passed through the finest mesh were carefully collected. A sand correction was applied by subtracting coarse sand (>2 mm) from each fraction to ensure accurate representation of soil aggregates. The separated wet aggregates were immediately freeze-dried and weighed to determine the mass distribution of macroaggregates, microaggregates, and silt and clay fractions. To minimize disturbances, all samples were handled gently during separation.
The freeze-dried aggregates were subsequently used to measure the aggregate organic carbon and N content. Before SOC analysis, samples were placed in open containers and exposed to concentrated HCl (12 mol L−1) vapor in a sealed desiccator for 48 h to eliminate inorganic carbon. After fumigation, samples were oven-dried to constant weight and then analyzed SOC and N contents in each aggregate’s fractions were determined with an elemental analyzer (Elementar Vario EL III, Elementar, Langenselbold, Germany).
The SOC content associated with each aggregate fraction was calculated based on the mass of C per unit mass of bulk soil (g·kg−1 soil). In this way, the values simultaneously represent the C concentration within each fraction and the contribution of that fraction to the total SOC in the soil. That is, higher C content in a given aggregate fraction indicates both a higher concentration of C within that fraction and a greater contribution of that fraction to bulk SOC. This approach allows for a direct comparison of C distribution across aggregate sizes while maintaining consistency with the bulk soil C measurements.
The proportion of each soil aggregate fractions (, %) was calculated as:
where is the mass of aggregate class , is the total mass of the soil sample.
Aggregate stability was calculated using the MWD (mm) [34] and GMD (mm) [35] as follows:
In these equations, refers to the specific aggregate size fraction, is the number of fractions, and indicates the average diameter and mass percentage of fraction .
2.4. Data Analyses
Differences in aggregate proportion, stability, and aggregate-associated C, N, and C/N ratios of the same aggregate class among forest types of identical stand age were analyzed using one-way analysis of variance (ANOVA), followed by Tukey’s honestly significant difference (HSD) test for multiple comparisons. Comparisons between soil layers within the same forest type and stand age were conducted using independent samples t-tests. A univariate general linear model (GLM) was applied to determine the main effects of aggregate class, forest type, stand age, and their interactions on soil aggregate distribution, SOC, N content, and the C/N ratio. Relationships between aggregate-associated C, bulk SOC, and MWD were evaluated using simple linear regression. Data normality was verified using the Shapiro–Wilk test prior to analysis. All statistical analyses were performed in R (version 4.5.1; R Core Team, 2022), with significance defined at p < 0.05.
3. Results
3.1. Soil Aggregates Distribution
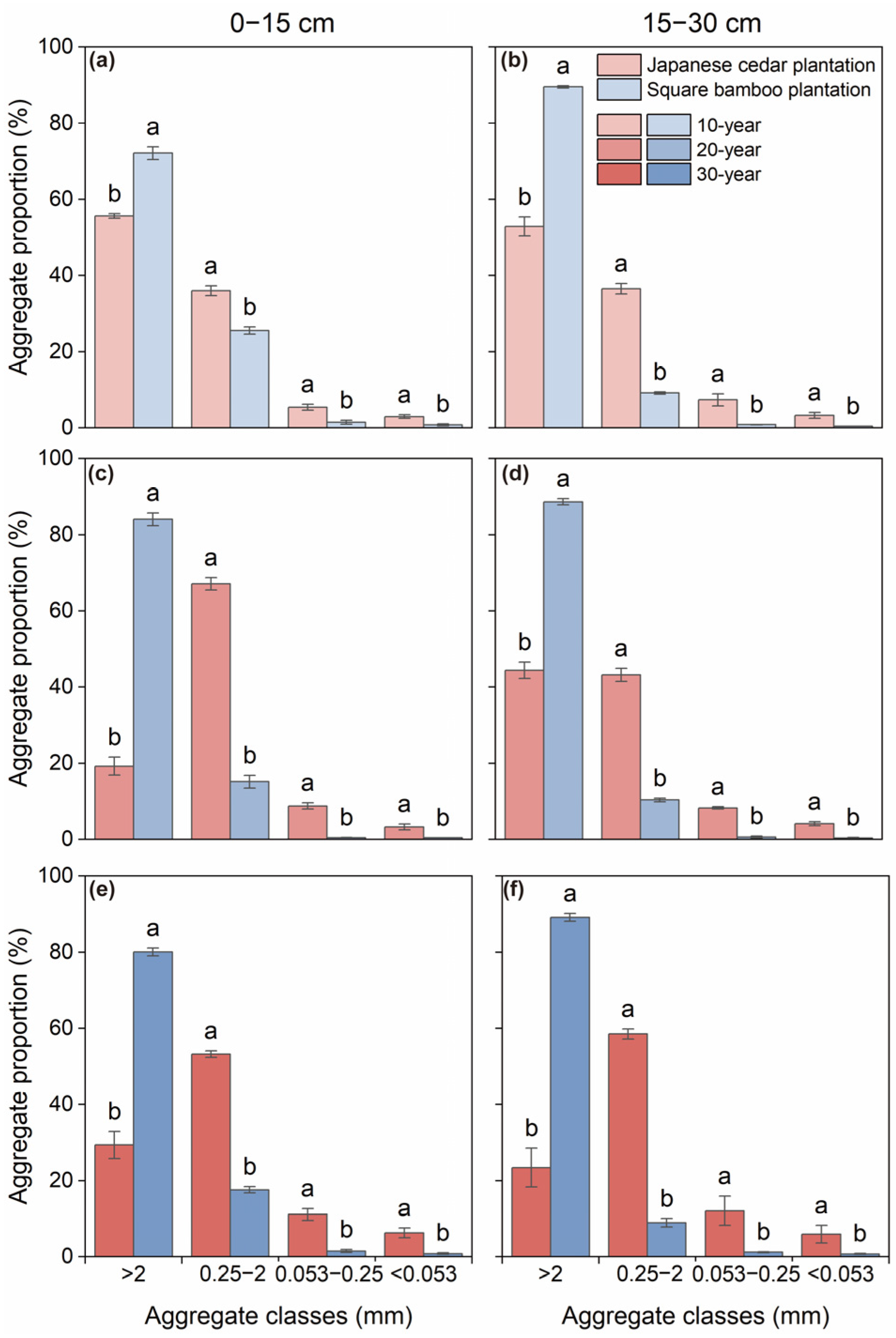
The distribution of soil aggregates varied significantly among aggregate size classes (Table 2). In both soil layers of the 10-year Japanese cedar plantations, coarse macroaggregates were the dominant fraction (52.85%–55.64%), followed by fine macroaggregates (36.01%–36.51%), microaggregates (5.40%–7.35%), and silt and clay fractions (2.95%–3.28%), respectively (Figure 1a,b). With increasing stand age, the proportion of macroaggregates declined by 47.15%–65.43%, whereas the proportion of microaggregates increased by 47.79%–111.74% (Figure 1). Across all stand ages and soil depths, square bamboo plantations significantly increased the proportion of coarse macroaggregates by 29.64%–336.92% while decreasing the proportions of fine macroaggregates, microaggregates, and silt and clay fractions by 28.95%–84.73%, 72.15%–94.42%, and 73.61%–93.633%, respectively, across different stand ages and soil depths (Figure 1). This pattern was consistent across stand ages and soil layers, suggesting that vegetation type exerted stronger control over aggregate distribution than stand age or depth (Table 2). Furthermore, significant interactive effects among aggregate class, forest type, and stand age (p < 0.01) indicated that the soil aggregate distribution was jointly influenced by vegetation type and stand age (Table 2).

Table 2.
Results of the general linear model (GLM) analysis showing the effects of soil aggregate class, forest type, stand age, and their interactions on aggregate distribution, and aggregate-associated soil organic carbon (SOC), nitrogen (N) contents, and C/N ratio at two soil depths.
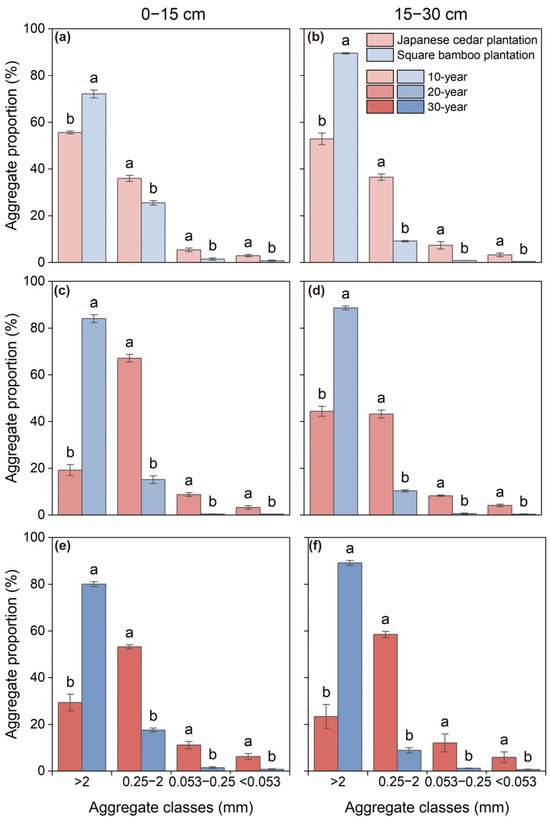
Figure 1.
Soil aggregate distribution in Japanese cedar and square bamboo plantations of different stand ages and soil layers. Panels (a,c,e) represent the 0–15 cm soil layer for 10-, 20-, and 30-year stands, respectively, and panels (b,d,f) represent the 15–30 cm soil layer. Values are presented as means ± standard deviation (n = 3). Different lowercase letters indicate significant differences (p < 0.05) between forest types within the same aggregate class.
3.2. Soil Aggregates Stability Characteristics
Aggregate stability, as indicated by MWD and GMD, varied significantly among forest types, stand ages, and soil layers (Table 3). Overall, both MWD and GMD were consistently higher in the square bamboo plantations than in Japanese cedar plantations across all stand ages and soil depths (p < 0.05). In the 0–15 cm layer, the MWD and GMD of square bamboo plantations ranged from 1.50 to 1.81 mm and from 1.50 to 1.72 mm, respectively, which were markedly higher than those of Japanese cedar plantations (0.71–1.43 mm and 0.71–1.11 mm, respectively). A similar pattern was observed in the 15–30 cm layer, where the values for square bamboo plantations were approximately 1.74–1.87 mm, compared with 0.69–1.38 mm for Japanese cedar plantations. Stand age also had a pronounced influence on aggregate stability. In Japanese cedar plantations, both MWD and GMD tended to decrease with increasing stand age, whereas in square bamboo plantations, these parameters remained relatively stable or increased slightly. Additionally, aggregate stability in the topsoil (0–15 cm) was either lower than or not significantly different from that in the subsurface layer (15–30 cm), depending on forest type and stand age.

Table 3.
Aggregates stability of Japanese cedar and square bamboo plantations of different stand ages (10, 20, and 30 years) across soil layers (0–15 and 15–30 cm) in southern Sichuan Province, China.
3.3. Contribution of Aggregate-Associated C and N to Bulk Soil
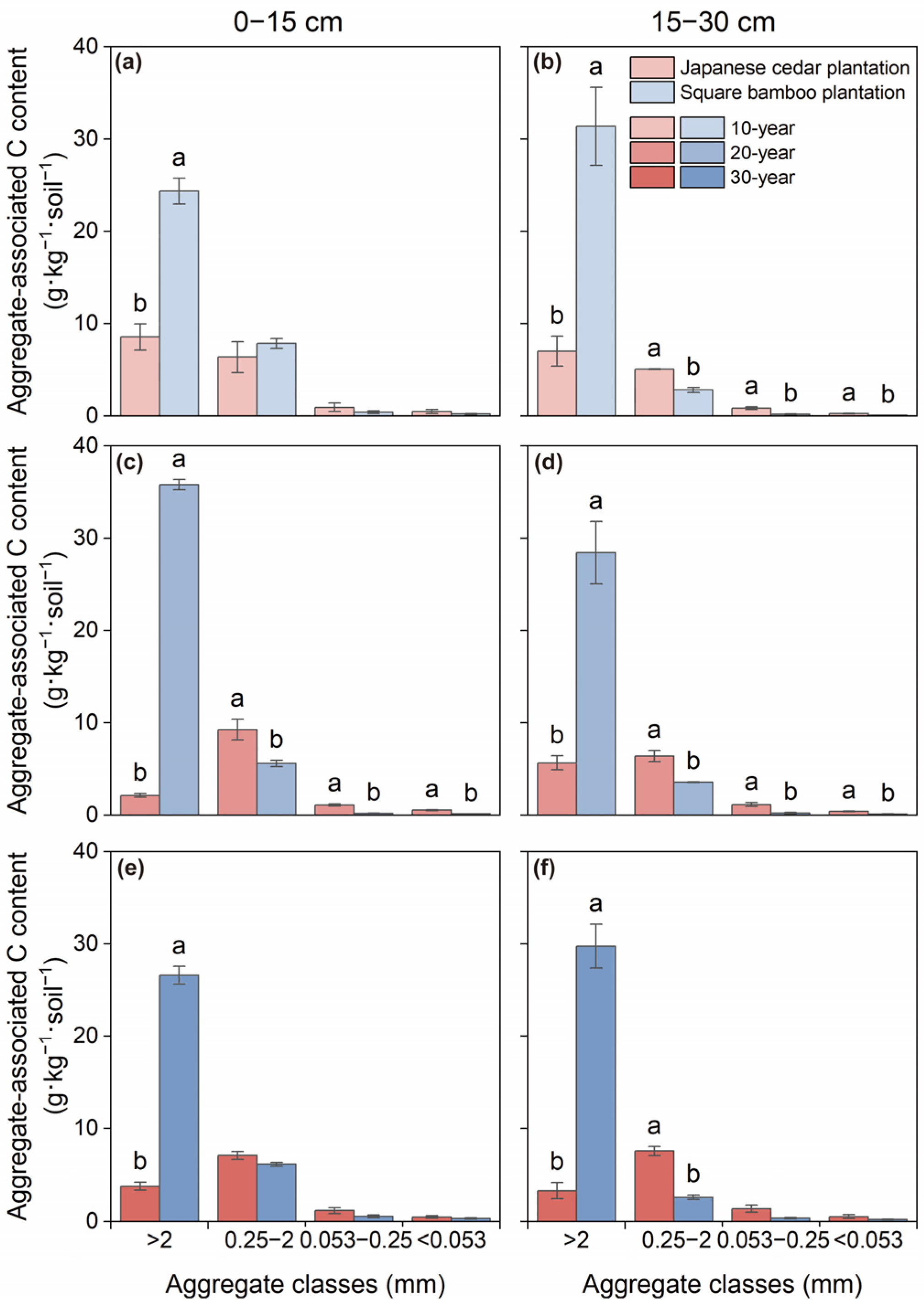
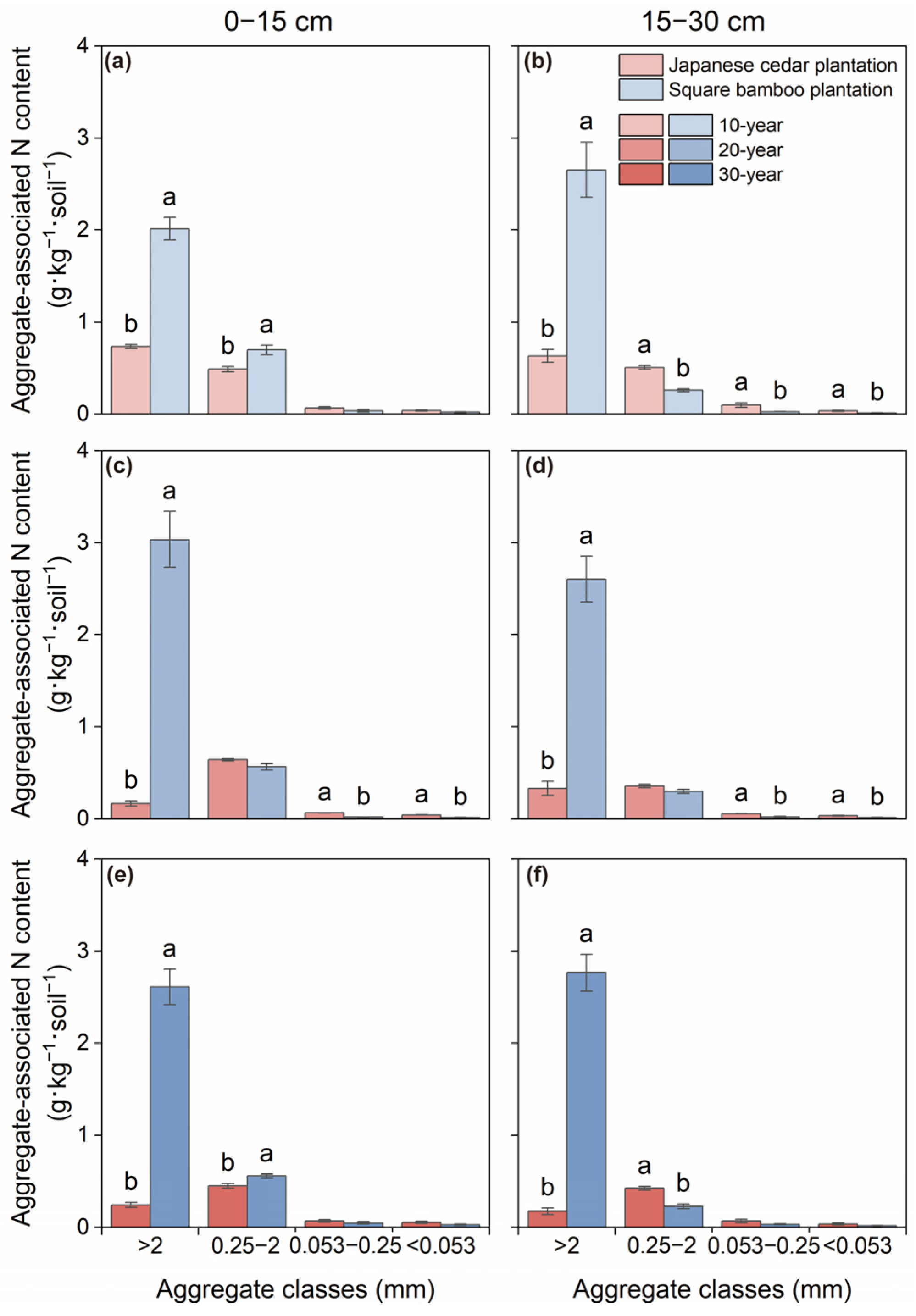
Both C and N contents varied significantly among aggregate size classes and forest type across both soil layers, while stand age affected C content only in the 0–15 cm layer (Table 2). In Japanese cedar plantations, aggregate-associated C and N were primarily concentrated in macroaggregates and microaggregates, with contents gradually declining as aggregate size decreased across both soil depths (Figure 2 and Figure 3). In square bamboo plantations, C and N were also predominantly associated with macroaggregates; however, a greater proportion of both elements were retained within macroaggregates across both soil layers, accompanied by comparatively lower contributions from microaggregates, particularly in the 15–30 cm layer.
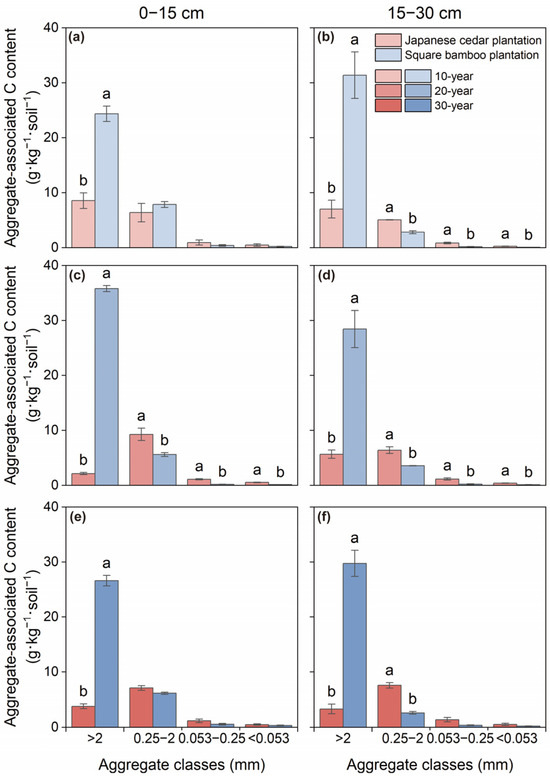
Figure 2.
Soil organic carbon content associated with different aggregate classes in Japanese cedar and square bamboo plantations of different stand ages and soil layers. Panels (a,c,e) represent the 0–15 cm soil layer for 10-, 20-, and 30-year stands, respectively, and panels (b,d,f) represent the 15–30 cm soil layer. Values are means ± standard deviation (n = 3). Different lowercase letters denote significant differences (p < 0.05) between forest types within the same aggregate class.
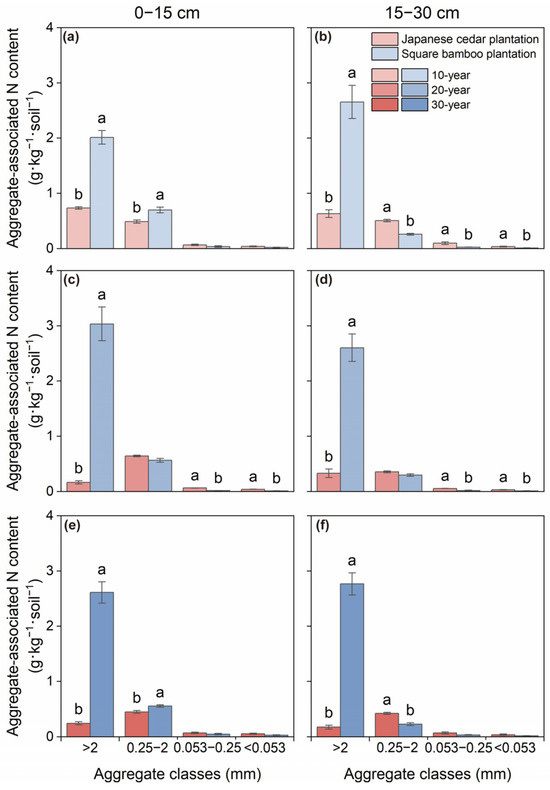
Figure 3.
Soil nitrogen content associated with different aggregate classes in Japanese cedar and square bamboo plantations of different stand ages and soil layers. Panels (a,c,e) represent the 0–15 cm soil layer for 10-, 20-, and 30-year stands, respectively, and panels (b,d,f) represent the 15–30 cm soil layer. Values are means ± standard deviation (n = 3). Different lowercase letters denote significant differences (p < 0.05) between forest types within the same aggregate class.
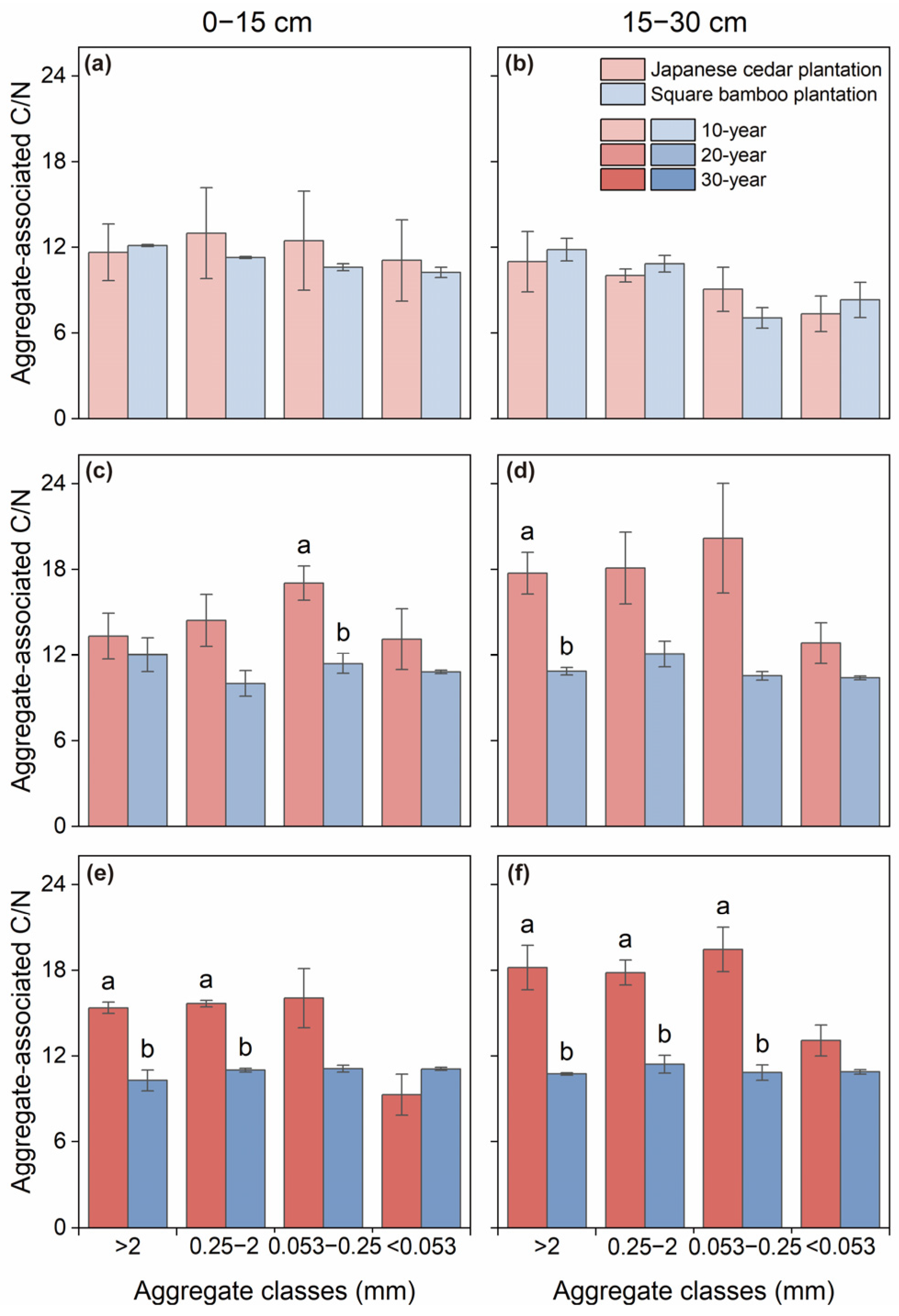
The C/N ratios of soil aggregates exhibited distinct responses to forest type and stand age (Table 2). Forest type had no significant effect on the aggregate-associated C/N ratios in either soil layer of the 10-year plantations (Figure 4a,b). However, in the 20-year stands, the C/N ratio of microaggregates in the 0–15 cm layer was significantly higher in Japanese cedar plantations than in square bamboo plantations. The same results were observed in macroaggregates in the 15–30 cm layer (Figure 4c,d). As stand age increased, the difference in soil aggregate C/N ratios between Japanese cedar plantations and square bamboo plantations became more pronounced, with Japanese cedar plantations exhibiting significantly higher soil aggregate C/N ratios (Figure 4e,f).
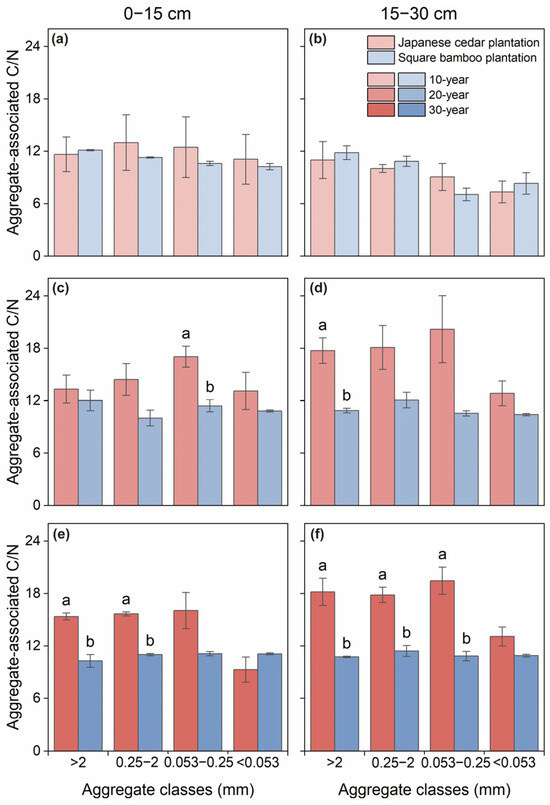
Figure 4.
Soil C/N ratios associated with different aggregate classes in Japanese cedar and square bamboo plantations of different stand ages and soil layers. Panels (a,c,e) represent the 0–15 cm soil layer for 10-, 20-, and 30-year stands, respectively, and panels (b,d,f) represent the 15–30 cm soil layer. Values are means ± standard deviation (n = 3). Different lowercase letters denote significant differences (p < 0.05) between forest types within the same aggregate class.
3.4. Bulk Soil C Content and MWD in Relation to Aggregate-Associated C Content
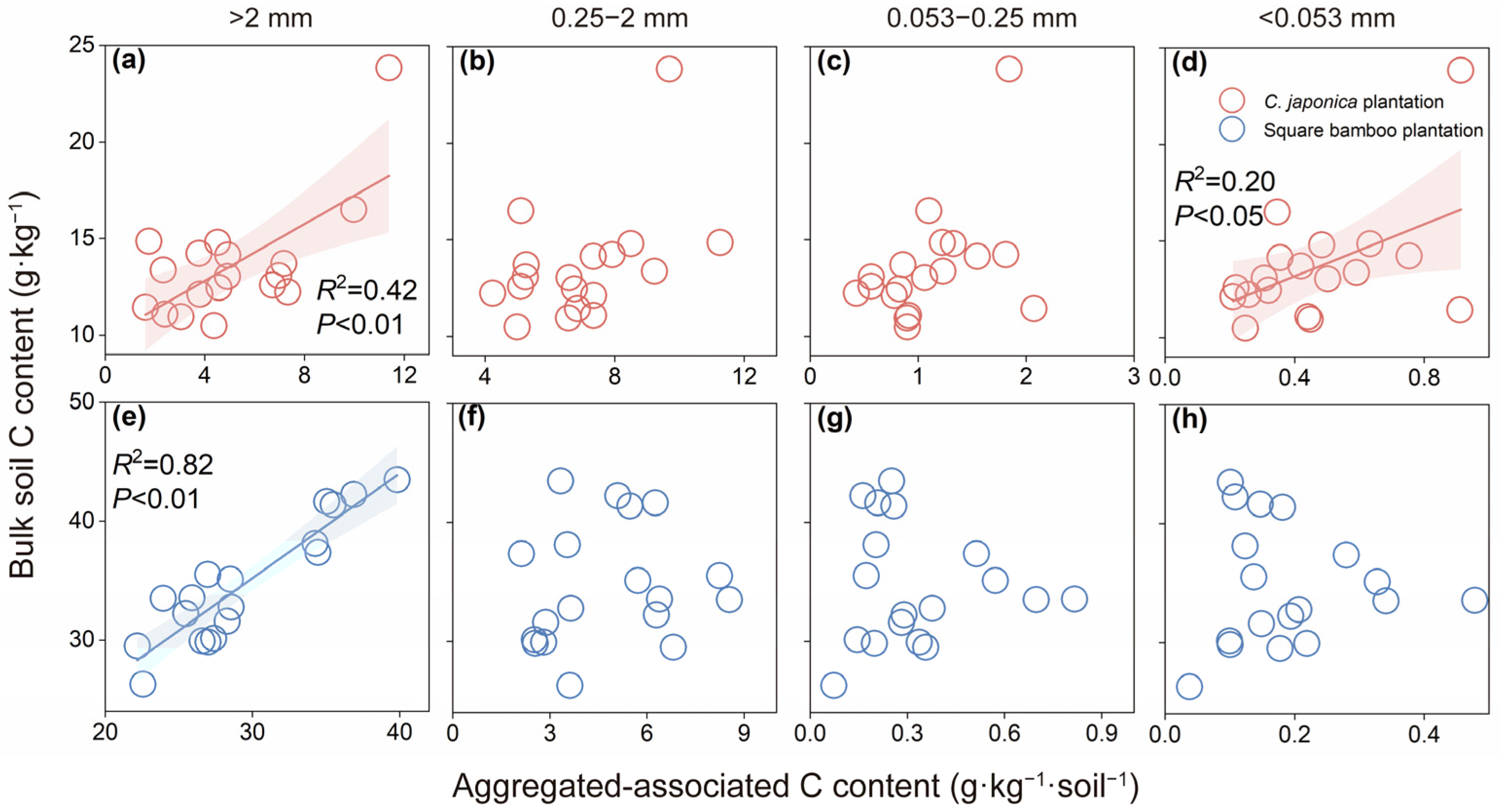
In Japanese cedar plantations, bulk soil C content was positively correlated with C content in macroaggregates (R2 = 0.42, p < 0.01) and in silt and clay fractions (R2 = 0.20, p < 0.05), whereas no significant relationships were observed between fine macroaggregate and microaggregate C content (Figure 5a–d). In square bamboo plantations, bulk soil C content was positively correlated with coarse macroaggregate C content (Figure 5a, R2 = 0.82, p < 0.01), but no significant relationships were found for other aggregate size fractions (Figure 5b–d).
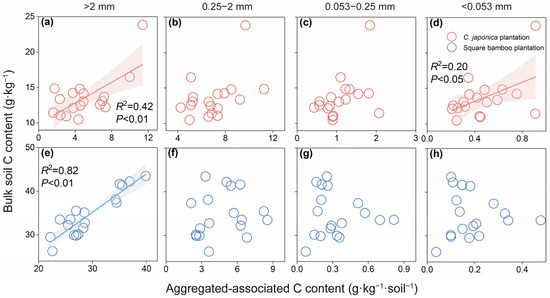
Figure 5.
Relationships between bulk soil carbon (C) content and aggregated-associated C content in Japanese cedar (a–d) and square bamboo plantations (e–h). Each point represents one observation (n = 18), including data from both soil layers (0–15 and 15–30 cm) and all stand ages (10, 20, and 30 years) within each forest type. The panels represent different aggregate size classes: (a,e) >2 mm, (b,f) 0.25–2 mm, (c,g) 0.053–0.25 mm, and (d,h) <0.053 mm. Shaded areas indicate 95% confidence intervals. Significant correlations (p < 0.05) are shown with fitted regression lines, with corresponding R2 and p values presented in each panel.
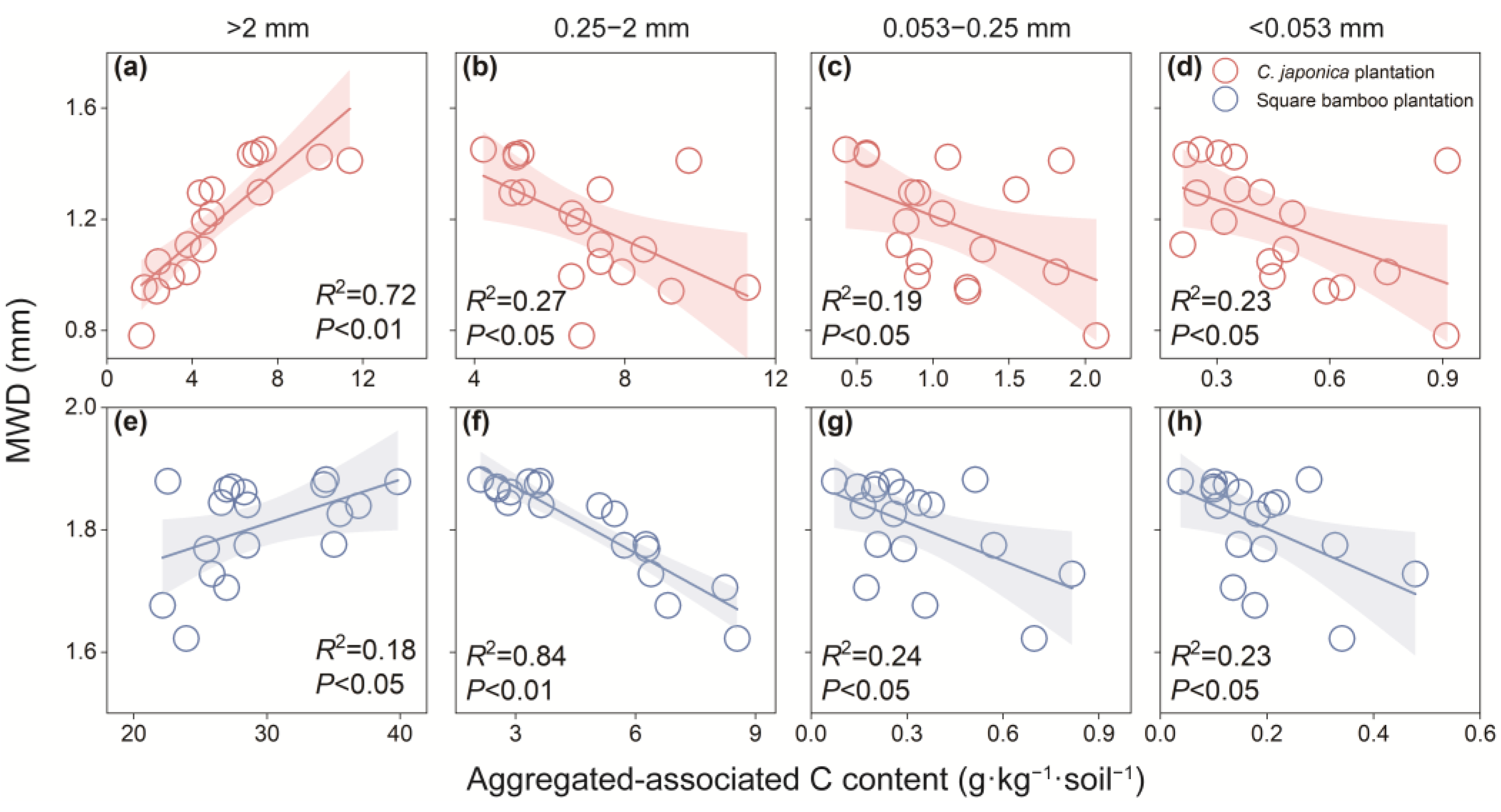
MWD as an indicator of aggregate stability exhibited contrasting patterns with aggregate-associated C among the different aggregate fractions (Figure 6). In Japanese cedar and square bamboo plantations, MWD was positively correlated with coarse macroaggregate C content, while negatively correlated with C content in fine macroaggregates, microaggregates, and silt and clay fractions.
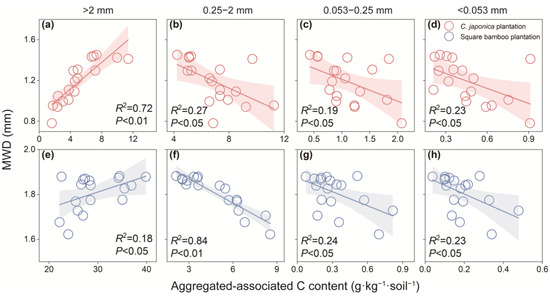
Figure 6.
Relationship between mean weight diameters (MWD) and aggregated-associated C content in Japanese cedar (a–d) and square bamboo plantations (e–h). Each point represents one observation (n = 18), including data from both soil layers (0–15 and 15–30 cm) and all stand ages (10, 20, and 30 years) within each forest type. The panels represent different aggregate size classes: (a,e) >2 mm, (b,f) 0.25–2 mm, (c,g) 0.053–0.25 mm, and (d,h) <0.053 mm. Shaded areas indicate 95% confidence intervals. Significant correlations (p < 0.05) are shown with fitted regression lines, with corresponding R2 and p values presented in each panel.
4. Discussion
4.1. Soil Aggregate Size Distribution and Stability in Different Forest Types
Our findings demonstrated that soil aggregate distribution varied significantly among aggregate size classes and was strongly influenced by forest type, stand age, and their interactions. Consistent with our first hypothesis, square bamboo plantations exhibited a significantly higher proportion of macroaggregates and a lower proportion of fine aggregates than Japanese cedar plantations across all stand ages and soil depths (Figure 1). This pattern indicates that bamboo vegetation plays a critical role in promoting soil macroaggregate formation [36,37]. The higher abundance of macroaggregates in square bamboo plantations may be partly related to differences in root biomass, as macroaggregates are primarily enmeshed by both living and decomposing roots [38,39,40], although root biomass was not directly measured in this study. Furthermore, the high litter biomass and quality of bamboo systems (Table 1) can increase soil organic inputs, stimulate microbial activity, and increase the production of extracellular polysaccharides, which serve as natural binding agents [19,41]. The proportion of macroaggregates in Japanese cedar plantations declined with increasing stand age, accompanied by a relative increase in smaller aggregate fractions (<2 mm). However, this trend was not observed in the square bamboo plantations. These findings suggest that the long-term stand development of Japanese cedar plantations may promote the breakdown of large aggregates into smaller ones.
Similar results have been reported in other coniferous forests, where slower litter decomposition, lower-quality organic inputs, and reduced soil biological activity contributed to diminished macroaggregate formation and stability [42,43]. In contrast, sustained litter and root detritus inputs in square bamboo plantations likely maintain macroaggregate turnover and stability through a continuous supply of organic binding materials, such as decomposing root residues and microbially derived organic compounds [44]. Overall, these results highlight the crucial role of vegetation type in altering soil aggregate distribution. The conversion of coniferous plantations to bamboo forests appears to enhance the soil physical quality and carbon sequestration potential by increasing the formation and persistence of macroaggregates.
Consistent with our expectations, both MWD and GMD were markedly higher in square bamboo plantations than in Japanese cedar plantations across all stand ages and soil depths, indicating overall greater soil aggregate stability in square bamboo plantations. The higher MWD values reflect the increased dominance of macroaggregates, whereas the consistently higher GMD values suggest a shift in the aggregate-size distribution toward larger and more stable size classes. Accordingly, enhanced aggregate stability in square bamboo plantations was closely associated with their higher proportion of macroaggregates (Figure 1). In Japanese cedar plantations, the decline in both MWD and GMD with increasing stand age suggests a gradual weakening of soil structural integrity, supporting previous findings that prolonged forest development may reduce aggregate stability when organic inputs and microbial activity are insufficient to sustain aggregate renewal [45]. In contrast, the relatively stable or slightly increasing MWD and GMD observed in bamboo plantations with increasing stand age indicate that sustained root growth and continuous organic matter inputs may help maintain or enhance aggregate stability over time [11,46]. Moreover, the slightly higher MWD and GMD values in the topsoil compared with the subsurface layer are consistent with earlier studies, reflecting greater biological activity, higher SOC concentrations, and more intense root-soil interactions in surface horizons that favor macroaggregates formation and stabilization [47]. These findings demonstrate that square bamboo plantations are associated with improved soil physical structure and greater resistance to structural degradation compared with Japanese cedar plantations.
4.2. Soil Aggregate–Associated C and N Contribution in Different Forest Types
In both Japanese cedar and square bamboo plantations, the contribution of C and N to bulk soil was the highest in macroaggregates and fine macroaggregates and decreased progressively with smaller aggregate sizes, which supported our second hypothesis. This finding aligns with those of previous studies, suggesting that larger aggregates provide more favorable microsites for organic matter accumulation and microbial activity, thereby enhancing C and N retention [48,49]. Compared to Japanese cedar plantations, square bamboo plantations had significantly increased C and N content within macroaggregates across both soil depths, while reducing those in microaggregates. This shift indicates that the pathways of soil C and N accumulation are regulated by stand type. The higher exogenous carbon input and root turnover rates of bamboo systems are conducive to maintaining the structure of macroaggregates and improving their stability, further promoting the macroaggregate-associated C contribution [36,38]. In contrast, Japanese cedar plantations, characterized by slower litter decomposition and lower-quality organic inputs as indicated by relatively higher C/N ratios (Table 1), exhibit a gradual decline in macroaggregate-associated C and N with increasing stand age [50]. The observed increase in microaggregate-associated C and N with stand development in Japanese cedar plantations also suggests enhanced aggregate breakdown and organic matter transfer to more stable but less dynamic pools [51].
The contrasting C/N ratios between the two forest types highlighted the differences in organic matter quality and decomposition dynamics. Higher C/N ratios in Japanese cedar aggregates, especially in older stands, indicate slower organic matter turnover and greater accumulation of recalcitrant carbon compounds [52,53]. In contrast, the lower C/N ratios observed in square bamboo plantations suggest a faster nutrient cycling rate and greater microbial processing of organic inputs. This pattern implies that bamboo conversion not only enhances aggregate stability and SOC storage, but also accelerates N cycling [54,55], with the magnitude of these effects increasing with stand age. Overall, these findings demonstrated that forest type strongly governs the spatial distribution and stabilization of aggregate-associated C and N. The conversion of Japanese cedar plantations to square bamboo forests promotes the formation of macroaggregates with higher SOC and N content, thereby enhancing soil structure, fertility, and C sequestration potential. However, lower C/N ratios under bamboo vegetation also suggest more active C and N cycling, reflecting a dynamic equilibrium between SOC stabilization and nutrient mineralization [56,57].
4.3. Bulk Soil C Content and MWD in Relation to Aggregate-Associated C Content
The relationships between bulk SOC, MWD, and aggregate-associated C provide important insights into the mechanisms governing SOC stabilization in different forest types. The significant positive correlations between bulk SOC and macroaggregate-associated C content in both Japanese cedar and square bamboo plantations support our third hypothesis, indicating the importance of macroaggregates in the regulation of SOC storage [58]. In addition, we observed a positive correlation between bulk SOC and C content in the silt and clay fractions in Japanese cedar plantations but not in square bamboo plantations. Thus, the results further emphasize that forest type exerts a strong influence on the pathways of SOC stabilization, with Japanese cedar soils showing relatively higher C in finer fractions, which may indicate greater mineral-associated C storage, and square bamboo plantations promoting macroaggregate-mediated C protection. The positive correlation between MWD and macroaggregate-associated C content across the different forests suggests that increased C accumulation within large aggregates enhances their structural integrity and resistance to disintegration [59]. Conversely, the negative correlation between MWD and C content in smaller aggregate fractions suggests that the disintegration of macroaggregates into microaggregates leads to C redistribution and a decline in overall soil structural stability [60]. Overall, these findings demonstrate that the mechanisms of SOC stabilization differ markedly between forest types. In Japanese cedar plantations, SOC is maintained through a combination of physical protection within aggregates and chemical associations with mineral surfaces, whereas in square bamboo plantations, SOC preservation is primarily governed by biologically driven macroaggregate stabilization and dynamic C turnover. These contrasting mechanisms highlight the functional importance of forest type in determining soil stability and C sequestration.
5. Conclusions
Forest type strongly influences soil aggregate distribution, stability, and the allocation of aggregate-associated C and N in subtropical plantations. Compared to Japanese cedar plantations, square bamboo plantations exhibited 30%–337% higher proportion of macroaggregates and greater aggregate stability across all stand ages and soil depths, reflecting improved soil structural integrity. In both forest types, macroaggregates were the dominant reservoirs of C and N, but their relative contribution was higher in bamboo plantations, whereas smaller aggregate fractions contributed less. This shift indicates that bamboo systems promote C and N accumulation through enhanced macroaggregate formation and stabilization. The positive relationships between macroaggregate-associated C, bulk SOC, and MWD highlight the important role of macroaggregates in SOC storage and structural stability. In addition, the positive correlation between bulk SOC and C in silt and clay fractions in Japanese cedar plantations, but not in square bamboo plantations, suggests distinct patterns of SOC distribution between Japanese cedar and square bamboo plantations. These results provide important implications for forest management and land-use planning. Specifically, vegetation conversion to bamboo systems may improve soil structural properties and the distribution of soil organic carbon, which could enhance soil health and ecosystem resilience. Such information can inform sustainable plantation management strategies, including species selection and soil conservation practices, to maintain or enhance soil quality in subtropical ecosystems.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/f17020267/s1.
Author Contributions
Conceptualization, X.W. and X.N.; methodology, X.W. and Y.G.; formal analysis, X.W., J.X., and J.C.; data curation, Y.H., W.L., and L.X.; writing—original draft preparation, writing—review and editing, visualization, X.W.; supervision, X.N.; funding acquisition, X.W. and J.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Leshan Normal University Scientific Research Start-up Project for Introduction High-level Talents, grant number RC2024012 and No. RC2025020, Leshan Science and Technology Bureau Program, grant number 24YYJC0025 and No. 24YYJC0029, and Leshan Normal University Research Incubation Program, grant number No. KYPY2025-0024.
Data Availability Statement
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| C | Carbon |
| SOC | Soil Organic Carbon |
| N | Nitrogen |
| GMD | Geometric Mean Diameter |
| MWD | Mean Weight Diameter |
| DBH | Diameter At Breast Height |
References
- Lehmann, J.; Kleber, M. The Contentious Nature of Soil Organic Matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef]
- Lorenz, K.; Lal, R. Soil Carbon Stock. In Carbon Sequestration in Agricultural Ecosystems; Springer International Publishing: Cham, Switzerland, 2018; pp. 39–136. ISBN 978-3-319-92317-8. [Google Scholar]
- Friedlingstein, P.; Jones, M.W.; O’Sullivan, M.; Andrew, R.M.; Hauck, J.; Peters, G.P.; Peters, W.; Pongratz, J.; Sitch, S.; Le Quéré, C.; et al. Global Carbon Budget 2019. Earth Syst. Sci. Data 2019, 11, 1783–1838. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Soong, J.L.; Horton, A.J.; Campbell, E.E.; Haddix, M.L.; Wall, D.H.; Parton, W.J. Formation of Soil Organic Matter via Biochemical and Physical Pathways of Litter Mass Loss. Nat. Geosci. 2015, 8, 776–779. [Google Scholar] [CrossRef]
- Dungait, J.A.J.; Hopkins, D.W.; Gregory, A.S.; Whitmore, A.P. Soil Organic Matter Turnover Is Governed by Accessibility Not Recalcitrance. Glob. Change Biol. 2012, 18, 1781–1796. [Google Scholar] [CrossRef]
- Amundson, R.; Berhe, A.A.; Hopmans, J.W.; Olson, C.; Sztein, A.E.; Sparks, D.L. Soil and Human Security in the 21st Century. Science 2015, 348, 1261071. [Google Scholar] [CrossRef]
- Zheng, Z.; Xu, Q.; Hu, Y. Effects of Forest Types on Soil Particulate Organic Carbon Contents and Distribution Along a Subtropical Climate Transect in China. Land Degrad. Dev. 2025, 36, 1860–1870. [Google Scholar] [CrossRef]
- Adair, E.C.; Parton, W.J.; Del Grosso, S.J.; Silver, W.L.; Harmon, M.E.; Hall, S.A.; Burke, I.C.; Hart, S.C. Simple Three-pool Model Accurately Describes Patterns of Long-term Litter Decomposition in Diverse Climates. Glob. Change Biol. 2008, 14, 2636–2660. [Google Scholar] [CrossRef]
- Liu, X.; Lin, T.-C.; Yang, Z.; Vadeboncoeur, M.A.; Lin, C.; Xiong, D.; Lin, W.; Chen, G.; Xie, J.; Li, Y.; et al. Increased Litter in Subtropical Forests Boosts Soil Respiration in Natural Forests but Not Plantations of Castanopsis Carlesii. Plant Soil 2017, 418, 141–151. [Google Scholar] [CrossRef]
- Six, J.; Paustian, K. Aggregate-Associated Soil Organic Matter as an Ecosystem Property and a Measurement Tool. Soil Biol. Biochem. 2014, 68, A4–A9. [Google Scholar] [CrossRef]
- Abiven, S.; Menasseri, S.; Chenu, C. The Effects of Organic Inputs over Time on Soil Aggregate Stability—A Literature Analysis. Soil Biol. Biochem. 2009, 41, 1–12. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Lal, R. Mechanisms of Carbon Sequestration in Soil Aggregates. Crit. Rev. Plant Sci. 2004, 23, 481–504. [Google Scholar] [CrossRef]
- Six, J.; Bossuyt, H.; Degryze, S.; Denef, K. A History of Research on the Link between (Micro)Aggregates, Soil Biota, and Soil Organic Matter Dynamics. Soil Tillage Res. 2004, 79, 7–31. [Google Scholar] [CrossRef]
- Guo, L.; Shen, J.; Li, B.; Li, Q.; Wang, C.; Guan, Y.; D’Acqui, L.P.; Luo, Y.; Tao, Q.; Xu, Q.; et al. Impacts of Agricultural Land Use Change on Soil Aggregate Stability and Physical Protection of Organic C. Sci. Total Environ. 2020, 707, 136049. [Google Scholar] [CrossRef]
- Vicente, L.C.; Gama-Rodrigues, E.F.; Gama-Rodrigues, A.C.; Marciano, C.R. Organic Carbon within Soil Aggregates under Forestry Systems and Pasture in a Southeast Region of Brazil. CATENA 2019, 182, 104139. [Google Scholar] [CrossRef]
- Edwards, A.P.; Bremner, J.M. Use of sonic vibration for separation of soil particles. Can. J. Soil Sci. 1964, 44, 366. [Google Scholar] [CrossRef]
- Tisdall, J.M.; Oades, J.M. Organic Matter and Water-stable Aggregates in Soils. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Six, J.; Paustian, K.; Elliott, E.T.; Combrink, C. Soil Structure and Organic Matter I. Distribution of Aggregate-Size Classes and Aggregate-Associated Carbon. Soil Sci. Soc. Am. J. 2000, 64, 681–689. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Z.; Fan, B.; Mao, X.; Luo, H.; Jiang, F.; Liang, C.; Chen, J.; Qin, H.; Xu, Q.; et al. Litter Inputs Control the Pattern of Soil Aggregate-Associated Organic Carbon and Enzyme Activities in Three Typical Subtropical Forests. Forests 2022, 13, 1210. [Google Scholar] [CrossRef]
- Totsche, K.U.; Amelung, W.; Gerzabek, M.H.; Guggenberger, G.; Klumpp, E.; Knief, C.; Lehndorff, E.; Mikutta, R.; Peth, S.; Prechtel, A.; et al. Microaggregates in Soils. J. Plant Nutr. Soil Sci. 2018, 181, 104–136. [Google Scholar] [CrossRef]
- Liu, Y.; Sui, X.; Hua, H.; Liu, X.; Chang, Q.; Xu, R.; Li, M.; Mu, L. Soil Aggregate Stability and Organic Carbon Content among Different Forest Types in Temperate Ecosystems in Northeastern China. Forests 2024, 15, 279. [Google Scholar] [CrossRef]
- He, Y.; Zhang, Q.; Jiang, C.; Lan, Y.; Zhang, H.; Ye, S. Mixed Planting Improves Soil Aggregate Stability and Aggregate-Associated C-N-P Accumulation in Subtropical China. Front. For. Glob. Chang. 2023, 6, 1141953. [Google Scholar] [CrossRef]
- Xu, C.; Lin, T.-C.; Yang, Z.; Liu, X.; Xiong, D.; Chen, S.; Wu, F.; Yang, Y. Forest Conversion Effects on Soil Organic Carbon Are Regulated by Soil Aggregate Stability and Not by Recalcitrance: Evidence from a Reforestation Experiment. CATENA 2022, 219, 106613. [Google Scholar] [CrossRef]
- Wang, X.; Yost, R.S.; Linquist, B.A. Soil Aggregate Size Affects Phosphorus Desorption from Highly Weathered Soils and Plant Growth. Soil Sci. Soc. Am. J. 2001, 65, 139–146. [Google Scholar] [CrossRef]
- Bronick, C.J.; Lal, R. Soil Structure and Management: A Review. Geoderma 2005, 124, 3–22. [Google Scholar] [CrossRef]
- Zhu, G.; Shangguan, Z.; Deng, L. Variations in Soil Aggregate Stability Due to Land Use Changes from Agricultural Land on the Loess Plateau, China. CATENA 2021, 200, 105181. [Google Scholar] [CrossRef]
- Ayoubi, S.; Mokhtari Karchegani, P.; Mosaddeghi, M.R.; Honarjoo, N. Soil Aggregation and Organic Carbon as Affected by Topography and Land Use Change in Western Iran. Soil Tillage Res. 2012, 121, 18–26. [Google Scholar] [CrossRef]
- Cheng, M.; Xiang, Y.; Xue, Z.; An, S.; Darboux, F. Soil Aggregation and Intra-Aggregate Carbon Fractions in Relation to Vegetation Succession on the Loess Plateau, China. CATENA 2015, 124, 77–84. [Google Scholar] [CrossRef]
- Zhao, D.; Xu, M.; Liu, G.; Ma, L.; Zhang, S.; Xiao, T.; Peng, G. Effect of Vegetation Type on Microstructure of Soil Aggregates on the Loess Plateau, China. Agric. Ecosyst. Environ. 2017, 242, 1–8. [Google Scholar] [CrossRef]
- Chen, X.; Gsell, T.; Yunger, J.; Randa, L.; Peng, Y.; Carrington, M. Soil Aggregation, Aggregate Stability, and Associated Soil Organic Carbon in Huron Mountains Forests, Michigan, USA. Forests 2025, 16, 219. [Google Scholar] [CrossRef]
- Ortiz, C.; Fernández-Alonso, M.J.; Kitzler, B.; Díaz-Pinés, E.; Saiz, G.; Rubio, A.; Benito, M. Variations in Soil Aggregation, Microbial Community Structure and Soil Organic Matter Cycling Associated to Long-Term Afforestation and Woody Encroachment in a Mediterranean Alpine Ecotone. Geoderma 2022, 405, 115450. [Google Scholar] [CrossRef]
- Six, J.; Callewaert, P.; Lenders, S.; De Gryze, S.; Morris, S.J.; Gregorich, E.G.; Paul, E.A.; Paustian, K. Measuring and Understanding Carbon Storage in Afforested Soils by Physical Fractionation. Soil Sci. Soc. Am. J. 2002, 66, 1981–1987. [Google Scholar] [CrossRef]
- Elliott, E.T. Aggregate Structure and Carbon, Nitrogen, and Phosphorus in Native and Cultivated Soils. Soil Sci. Soc. Am. J. 1986, 50, 627–633. [Google Scholar] [CrossRef]
- Van Bavel, C.H.M. Mean Weight-Diameter of Soil Aggregates as a Statistical Index of Aggregation. Soil Sci. Soc. Am. J. 1950, 14, 20–23. [Google Scholar] [CrossRef]
- Duan, L.; Sheng, H.; Yuan, H.; Zhou, Q.; Li, Z. Land Use Conversion and Lithology Impacts Soil Aggregate Stability in Subtropical China. Geoderma 2021, 389, 114953. [Google Scholar] [CrossRef]
- Ni, H.; Su, W. Spatial Distribution of Fine Root Traits in Relation to Soil Properties and Aggregate Stability of Intensively Managed Moso Bamboo (Phyllostachys Edulis) Plantations in Subtropical China. Plant Soil 2024, 498, 487–503. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, X.; Ni, H.; Gai, X.; Huang, Z.; Du, X.; Zhong, Z. Soil Carbon and Associated Bacterial Community Shifts Driven by Fine Root Traits along a Chronosequence of Moso Bamboo (Phyllostachys Edulis) Plantations in Subtropical China. Sci. Total Environ. 2021, 752, 142333. [Google Scholar] [CrossRef] [PubMed]
- Oades, J.M. Soil Organic Matter and Structural Stability: Mechanisms and Implications for Management. Plant Soil 1984, 76, 319–337. [Google Scholar] [CrossRef]
- Qin, H.; Chen, J.; Wu, Q.; Niu, L.; Li, Y.; Liang, C.; Shen, Y.; Xu, Q. Intensive Management Decreases Soil Aggregation and Changes the Abundance and Community Compositions of Arbuscular Mycorrhizal Fungi in Moso Bamboo (Phyllostachys Pubescens) Forests. For. Ecol. Manag. 2017, 400, 246–255. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, W.; Qi, J.; Zhang, H.; Tao, F.; Zhang, R. Priming Effects of Soil Organic Matter Decomposition with Addition of Different Carbon Substrates. J. Soils Sediments 2019, 19, 1171–1178. [Google Scholar] [CrossRef]
- Kumar, P.S.; Shukla, G.; Nath, A.J.; Chakravarty, S. Soil Properties, Litter Dynamics and Biomass Carbon Storage in Three-Bamboo Species of Sub-Himalayan Region of Eastern India. Water Air Soil Pollut. 2022, 233, 12. [Google Scholar] [CrossRef]
- Wei, C.; Wang, Q.; Ren, M.; Pei, Z.; Lu, J.; Wang, H.; Wang, W. Soil Aggregation Accounts for the Mineral Soil Organic Carbon and Nitrogen Accrual in Broadleaved Forests as Compared to That of Coniferous Forests in Northeast China: Cross-sites and Multiple Species Comparisons. Land Degrad. Dev. 2021, 32, 296–309. [Google Scholar] [CrossRef]
- Su, F.; Xu, S.; Sayer, E.J.; Chen, W.; Du, Y.; Lu, X. Distinct Storage Mechanisms of Soil Organic Carbon in Coniferous Forest and Evergreen Broadleaf Forest in Tropical China. J. Environ. Manag. 2021, 295, 113142. [Google Scholar] [CrossRef]
- Kaushal, R.; Singh, I.; Thapliyal, S.D.; Gupta, A.K.; Mandal, D.; Tomar, J.M.S.; Kumar, A.; Alam, N.M.; Kadam, D.; Singh, D.V.; et al. Rooting Behaviour and Soil Properties in Different Bamboo Species of Western Himalayan Foothills, India. Sci. Rep. 2020, 10, 4966. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Zhou, Y.; He, H. Effects of Rehabilitation through Afforestation on Soil Aggregate Stability and Aggregate-Associated Carbon after Forest Fires in Subtropical China. Geoderma 2020, 376, 114548. [Google Scholar] [CrossRef]
- Chivenge, P.; Vanlauwe, B.; Gentile, R.; Six, J. Organic Resource Quality Influences Short-Term Aggregate Dynamics and Soil Organic Carbon and Nitrogen Accumulation. Soil Biol. Biochem. 2011, 43, 657–666. [Google Scholar] [CrossRef]
- Le Bissonnais, Y.; Prieto, I.; Roumet, C.; Nespoulous, J.; Metayer, J.; Huon, S.; Villatoro, M.; Stokes, A. Soil Aggregate Stability in Mediterranean and Tropical Agro-Ecosystems: Effect of Plant Roots and Soil Characteristics. Plant Soil 2018, 424, 303–317. [Google Scholar] [CrossRef]
- Tou, C.; Liu, X.; Gan, J.; Wanek, W.; Jones, D.L.; Wu, L.; Ma, Q. Plant Residue Quality Regulates Its Incorporation into Soil Aggregates and Organic Carbon Stabilisation. Geoderma 2025, 460, 117407. [Google Scholar] [CrossRef]
- Das, B.; Chakraborty, D.; Singh, V.K.; Aggarwal, P.; Singh, R.; Dwivedi, B.S.; Mishra, R.P. Effect of Integrated Nutrient Management Practice on Soil Aggregate Properties, Its Stability and Aggregate-Associated Carbon Content in an Intensive Rice–Wheat System. Soil Tillage Res. 2014, 136, 9–18. [Google Scholar] [CrossRef]
- Laub, M.; Schlichenmeier, S.; Vityakon, P.; Cadisch, G. Litter Quality and Microbes Explain Aggregation Differences in a Tropical Sandy Soil. J. Soil Sci. Plant Nutr. 2022, 22, 848–860. [Google Scholar] [CrossRef]
- Von Fromm, S.F.; Hoyt, A.M.; Lange, M.; Acquah, G.E.; Aynekulu, E.; Berhe, A.A.; Haefele, S.M.; McGrath, S.P.; Shepherd, K.D.; Sila, A.M.; et al. Continental-Scale Controls on Soil Organic Carbon across Sub-Saharan Africa. SOIL 2021, 7, 305–332. [Google Scholar] [CrossRef]
- Long, C.; Zhang, Q.; Chen, Q.; Cheng, X. Divergent Controls on Leaf and Root Litter Decay Linking to Soil C, N, and P Pools Under a Subtropical Land-Use Change. Ecosystems 2023, 26, 1209–1223. [Google Scholar] [CrossRef]
- Castellano, M.J.; Mueller, K.E.; Olk, D.C.; Sawyer, J.E.; Six, J. Integrating Plant Litter Quality, Soil Organic Matter Stabilization, and the Carbon Saturation Concept. Glob. Change Biol. 2015, 21, 3200–3209. [Google Scholar] [CrossRef]
- Bania, J.K.; Nath, A.J.; Sileshi, G.W.; Paramesh, V.; Nandy, S.; Das, A.K. Substantial Amounts of Soil Organic Carbon and Macronutrients Are Stored in Deep Soil Layers under Bamboo Plantations. Geoderma Reg. 2025, 42, e01004. [Google Scholar] [CrossRef]
- Teng, Q.; Fang, T.; Zhang, Q.; Gunina, A.; Zheng, A.; Song, Z.; Zhou, J.; Chang, S.X.; Li, Y. Successional Transition from Broadleaf to Bamboo Forests Promotes Fungal Communities and Soil Carbon Mineralization Following the Altered Litterfall Quality. Appl. Soil Ecol. 2025, 209, 106006. [Google Scholar] [CrossRef]
- Zhao, Y.; Liang, C.; Shao, S.; Chen, J.; Qin, H.; Xu, Q. Linkages of Litter and Soil C:N:P Stoichiometry with Soil Microbial Resource Limitation and Community Structure in a Subtropical Broadleaf Forest Invaded by Moso Bamboo. Plant Soil 2021, 465, 473–490. [Google Scholar] [CrossRef]
- Ouyang, M.; Eziz, A.; Fang, W.; Cai, Q.; Ma, S.; Xiao, S.; Zhu, J.; Yang, Q.; Hu, J.; Tang, Z.; et al. Constant Isometric Scaling of Soil Carbon to Nitrogen in Moso Bamboo-Invaded Evergreen Broadleaf Forests in Subtropical China. Plant Soil 2025, 511, 299–308. [Google Scholar] [CrossRef]
- Liao, H.; Long, J.; Li, J. Conversion of Cropland to Chinese Prickly Ash Orchard Affects Soil Organic Carbon Dynamics in a Karst Region of Southwest China. Nutr. Cycl. Agroecosyst 2016, 104, 15–23. [Google Scholar] [CrossRef]
- Six, J.; Elliott, E.T.; Paustian, K. Soil Macroaggregate Turnover and Microaggregate Formation: A Mechanism for C Sequestration under No-Tillage Agriculture. Soil Biol. Biochem. 2000, 32, 2099–2103. [Google Scholar] [CrossRef]
- Degens, B.P. Macro-Aggregation of Soils by Biological Bonding and Binding Mechanisms and the Factors Affecting These: A Review. Soil Res. 1997, 35, 431–460. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.