Figure 1.
Biochar preparation process. (a) Preparation process of primary biochar; (b) Preparation process of KOH-modified biochar. All operations were performed at room temperature.
Figure 1.
Biochar preparation process. (a) Preparation process of primary biochar; (b) Preparation process of KOH-modified biochar. All operations were performed at room temperature.
Figure 2.
Calibration curve of tetracycline hydrochloride (measured at room temperature): y = 0.0381x + 0.0093 (R2 = 0.9999).
Figure 2.
Calibration curve of tetracycline hydrochloride (measured at room temperature): y = 0.0381x + 0.0093 (R2 = 0.9999).
Figure 3.
(a) FT-IR and (b) XRD patterns of yak manure-derived biochar (YBC) prepared at different pyrolysis temperatures (500, 600, and 700 °C). All measurements were conducted at room temperature. FT-IR spectra were recorded in the range of 500–4000 cm−1 with a resolution of 4 cm−1 and 32 scans. XRD analysis was performed over a 2θ range of 5–85° with a sampling time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The results demonstrate that the raw biochar contains abundant oxygen-containing functional groups, which undergo gradual depletion as the pyrolysis temperature increases.
Figure 3.
(a) FT-IR and (b) XRD patterns of yak manure-derived biochar (YBC) prepared at different pyrolysis temperatures (500, 600, and 700 °C). All measurements were conducted at room temperature. FT-IR spectra were recorded in the range of 500–4000 cm−1 with a resolution of 4 cm−1 and 32 scans. XRD analysis was performed over a 2θ range of 5–85° with a sampling time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The results demonstrate that the raw biochar contains abundant oxygen-containing functional groups, which undergo gradual depletion as the pyrolysis temperature increases.
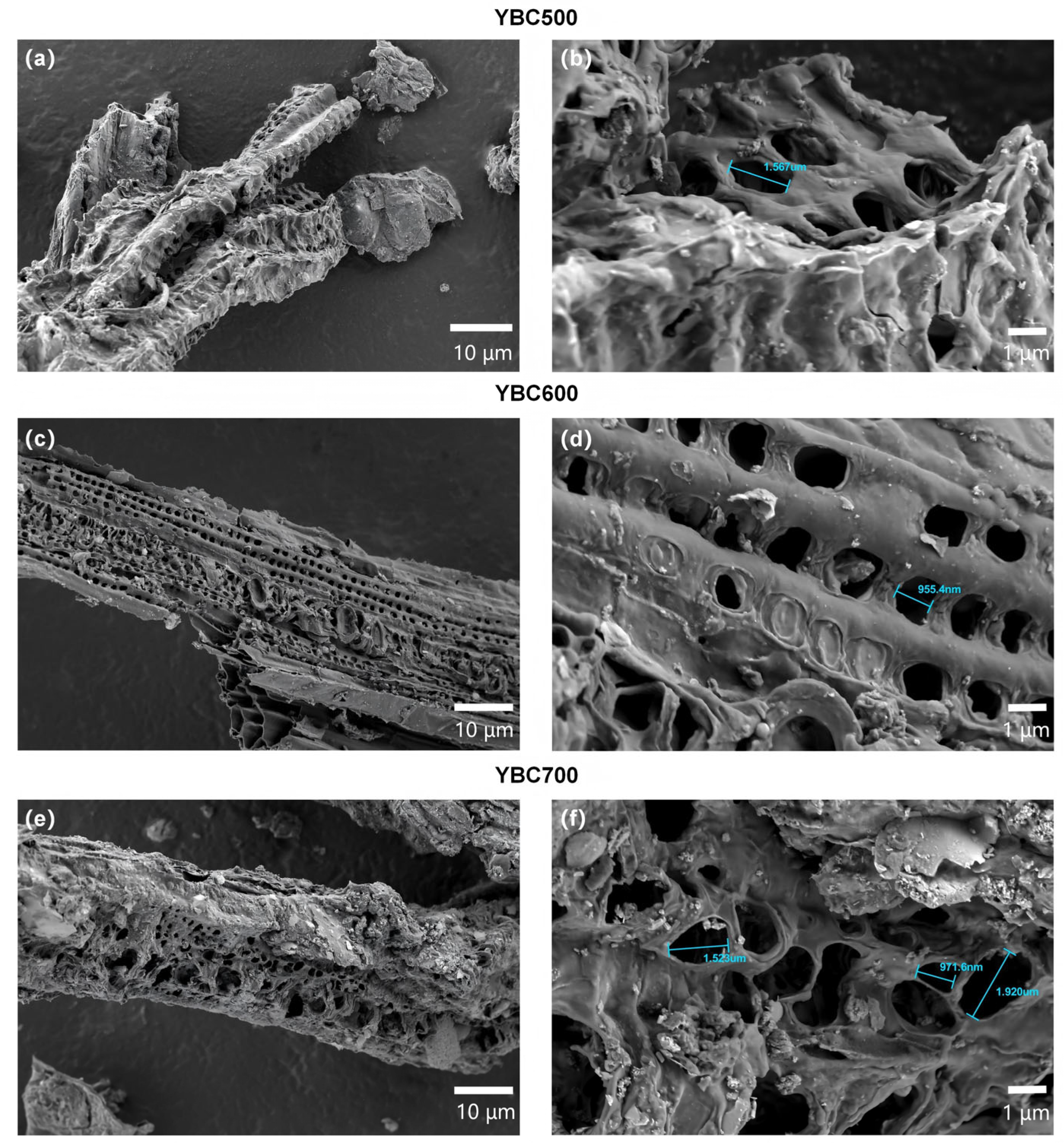
Figure 4.
The evolution of the surface pore structure in yak manure-derived biochar (YBC) with increasing pyrolysis temperature is depicted in the SEM images. All images were acquired using a scanning electron microscope operated at an accelerating voltage of 2.0 kV. As shown in panels (a,b) for YBC500, (c,d) for YBC600, and (e,f) for YBC700 at magnifications of 10 µm and 1 µm, respectively, the micrographs reveal that YBC600 exhibits a more uniform and ordered arrangement of surface pores along with a well-preserved structural integrity.
Figure 4.
The evolution of the surface pore structure in yak manure-derived biochar (YBC) with increasing pyrolysis temperature is depicted in the SEM images. All images were acquired using a scanning electron microscope operated at an accelerating voltage of 2.0 kV. As shown in panels (a,b) for YBC500, (c,d) for YBC600, and (e,f) for YBC700 at magnifications of 10 µm and 1 µm, respectively, the micrographs reveal that YBC600 exhibits a more uniform and ordered arrangement of surface pores along with a well-preserved structural integrity.
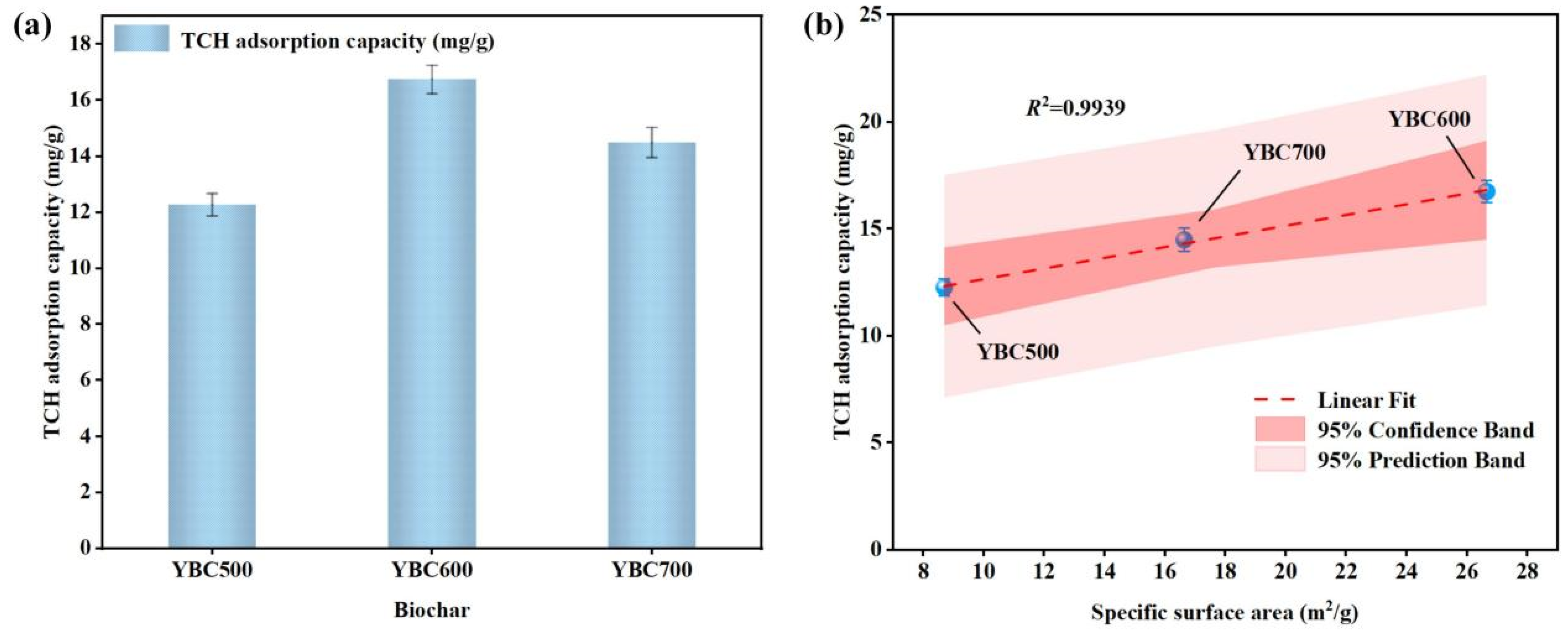
Figure 5.
(a) Adsorption capacity of pristine biochar for TCH at different pyrolysis temperatures and (b) Correlation between the specific surface area of pristine biochar and TCH adsorption capacity. The adsorption experiments were conducted under the following conditions: room temperature, an initial TCH concentration of 100 mg/L, an adsorption time of 6 h, and a biochar dosage of 1.5 g/L. The results demonstrated a positive linear correlation between the specific surface area of the pristine biochar and its tetracycline hydrochloride (TCH) adsorption capacity.
Figure 5.
(a) Adsorption capacity of pristine biochar for TCH at different pyrolysis temperatures and (b) Correlation between the specific surface area of pristine biochar and TCH adsorption capacity. The adsorption experiments were conducted under the following conditions: room temperature, an initial TCH concentration of 100 mg/L, an adsorption time of 6 h, and a biochar dosage of 1.5 g/L. The results demonstrated a positive linear correlation between the specific surface area of the pristine biochar and its tetracycline hydrochloride (TCH) adsorption capacity.
Figure 6.
(a) FTIR spectra of biochar before and after KOH modification and (b) XRD spectra of biochar before and after KOH modification. All characterizations were performed at ambient temperature. Fourier transform infrared (FT-IR) spectroscopy was conducted with a spectral range of 500–4000 cm−1, a resolution of 4 cm−1, and by averaging 32 scans. X-ray diffraction (XRD) patterns were collected over a 2θ range of 5–85° with a step time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The results demonstrate that KOH modification increased the degree of unsaturation in the biochar, thereby enhancing its aromaticity.
Figure 6.
(a) FTIR spectra of biochar before and after KOH modification and (b) XRD spectra of biochar before and after KOH modification. All characterizations were performed at ambient temperature. Fourier transform infrared (FT-IR) spectroscopy was conducted with a spectral range of 500–4000 cm−1, a resolution of 4 cm−1, and by averaging 32 scans. X-ray diffraction (XRD) patterns were collected over a 2θ range of 5–85° with a step time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The results demonstrate that KOH modification increased the degree of unsaturation in the biochar, thereby enhancing its aromaticity.
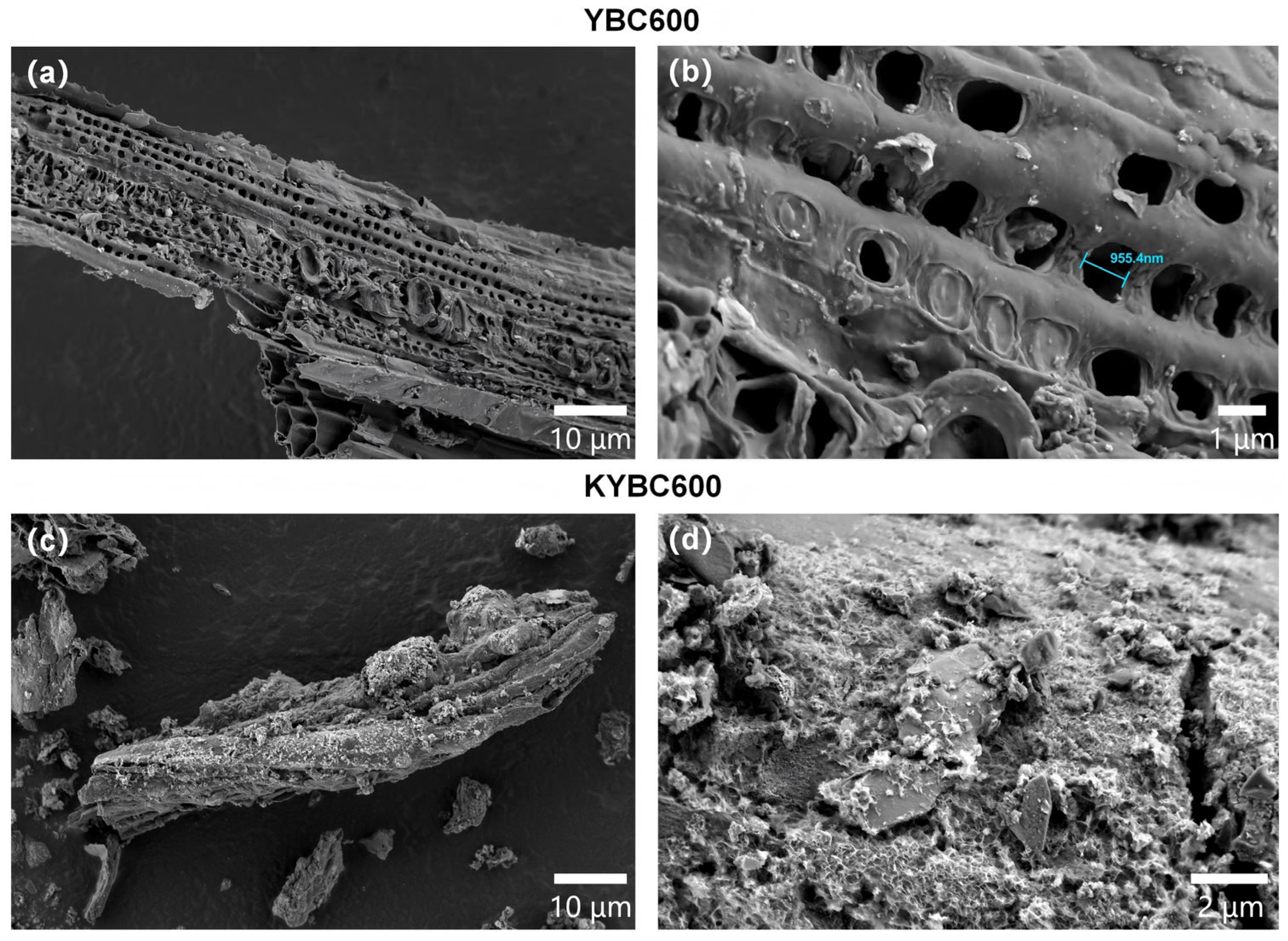
Figure 7.
Representative scanning electron microscopy (SEM) images of (a,b) the pristine biochar (YBC600) and (c,d) the KOH-modified biochar (KYBC600) are presented. All images were acquired at an accelerating voltage of 2.0 kV. The micrographs reveal that KOH modification induces significant alterations in the surface morphology of the biochar.
Figure 7.
Representative scanning electron microscopy (SEM) images of (a,b) the pristine biochar (YBC600) and (c,d) the KOH-modified biochar (KYBC600) are presented. All images were acquired at an accelerating voltage of 2.0 kV. The micrographs reveal that KOH modification induces significant alterations in the surface morphology of the biochar.
Figure 8.
(a) TCH adsorption by YBC600 and KYBC600 at different adsorption times; (b,c) TCH adsorption by biochar at different biochar dosages; (d,e) TCH adsorption by biochar at different initial TCH concentrations; (f) TCH adsorption by biochar and biochar zeta potential at different solution pH values. Reaction conditions: [TCH] = 20 mg·L−1, initial pH 5.0, temperature 25 °C. The results indicated that the optimal adsorption conditions for TCH removal by both KYBC600 and YBC600 were as follows: an adsorption time of 6 h, a biochar dosage of 0.5 g/L, an initial TCH concentration of 20 mg/L, and an initial solution pH of 5.
Figure 8.
(a) TCH adsorption by YBC600 and KYBC600 at different adsorption times; (b,c) TCH adsorption by biochar at different biochar dosages; (d,e) TCH adsorption by biochar at different initial TCH concentrations; (f) TCH adsorption by biochar and biochar zeta potential at different solution pH values. Reaction conditions: [TCH] = 20 mg·L−1, initial pH 5.0, temperature 25 °C. The results indicated that the optimal adsorption conditions for TCH removal by both KYBC600 and YBC600 were as follows: an adsorption time of 6 h, a biochar dosage of 0.5 g/L, an initial TCH concentration of 20 mg/L, and an initial solution pH of 5.
Figure 9.
(
a) Molecular structure of tetracycline hydrochloride [
72]; (
b) Species distribution of tetracycline hydrochloride at different pH values [
72]; (
c) TCH adsorption by biochar at different temperatures; (
d) Effect of NaCl concentration on TCH adsorption by KYBC600; The blue horizontal dashed line in Figure (
d) represents the TCH adsorption capacity of biochar KYBC600 without the addition of NaCl. This dashed line is included primarily for comparison purposes, serving as the control group. [TCH] = 20 mg·L
−1, initial pH 5, temperature 25 °C, dosage 0.5 g·L
−1. The results demonstrated that the optimal temperature for TCH adsorption by both KYBC600 and YBC600 was 25 °C. Furthermore, the adsorption capacity of KYBC600 for TCH was enhanced with increasing NaCl concentration.
Figure 9.
(
a) Molecular structure of tetracycline hydrochloride [
72]; (
b) Species distribution of tetracycline hydrochloride at different pH values [
72]; (
c) TCH adsorption by biochar at different temperatures; (
d) Effect of NaCl concentration on TCH adsorption by KYBC600; The blue horizontal dashed line in Figure (
d) represents the TCH adsorption capacity of biochar KYBC600 without the addition of NaCl. This dashed line is included primarily for comparison purposes, serving as the control group. [TCH] = 20 mg·L
−1, initial pH 5, temperature 25 °C, dosage 0.5 g·L
−1. The results demonstrated that the optimal temperature for TCH adsorption by both KYBC600 and YBC600 was 25 °C. Furthermore, the adsorption capacity of KYBC600 for TCH was enhanced with increasing NaCl concentration.
Figure 10.
(a) Kinetic fitting of TCH adsorption onto biochars KYBC600 and YBC600; (b) Fitting curves of the intra-particle diffusion model; (c) Linear relationship between lnK and 1/T; (d) Isotherm fitting for TCH adsorption onto KYBC600 and YBC600; (e) Values of the separation factor (RL). All experiments were conducted under the following conditions: [TCH] = 20 mg·L−1, initial pH = 5, and adsorption time t = 6 h. The results demonstrate that the adsorption process is better described by the pseudo-second-order kinetic model, indicating that chemical adsorption is the predominant mechanism. Furthermore, intra-particle diffusion is not the sole rate-limiting step, but rather, the overall process is governed by a combination of multiple factors.
Figure 10.
(a) Kinetic fitting of TCH adsorption onto biochars KYBC600 and YBC600; (b) Fitting curves of the intra-particle diffusion model; (c) Linear relationship between lnK and 1/T; (d) Isotherm fitting for TCH adsorption onto KYBC600 and YBC600; (e) Values of the separation factor (RL). All experiments were conducted under the following conditions: [TCH] = 20 mg·L−1, initial pH = 5, and adsorption time t = 6 h. The results demonstrate that the adsorption process is better described by the pseudo-second-order kinetic model, indicating that chemical adsorption is the predominant mechanism. Furthermore, intra-particle diffusion is not the sole rate-limiting step, but rather, the overall process is governed by a combination of multiple factors.
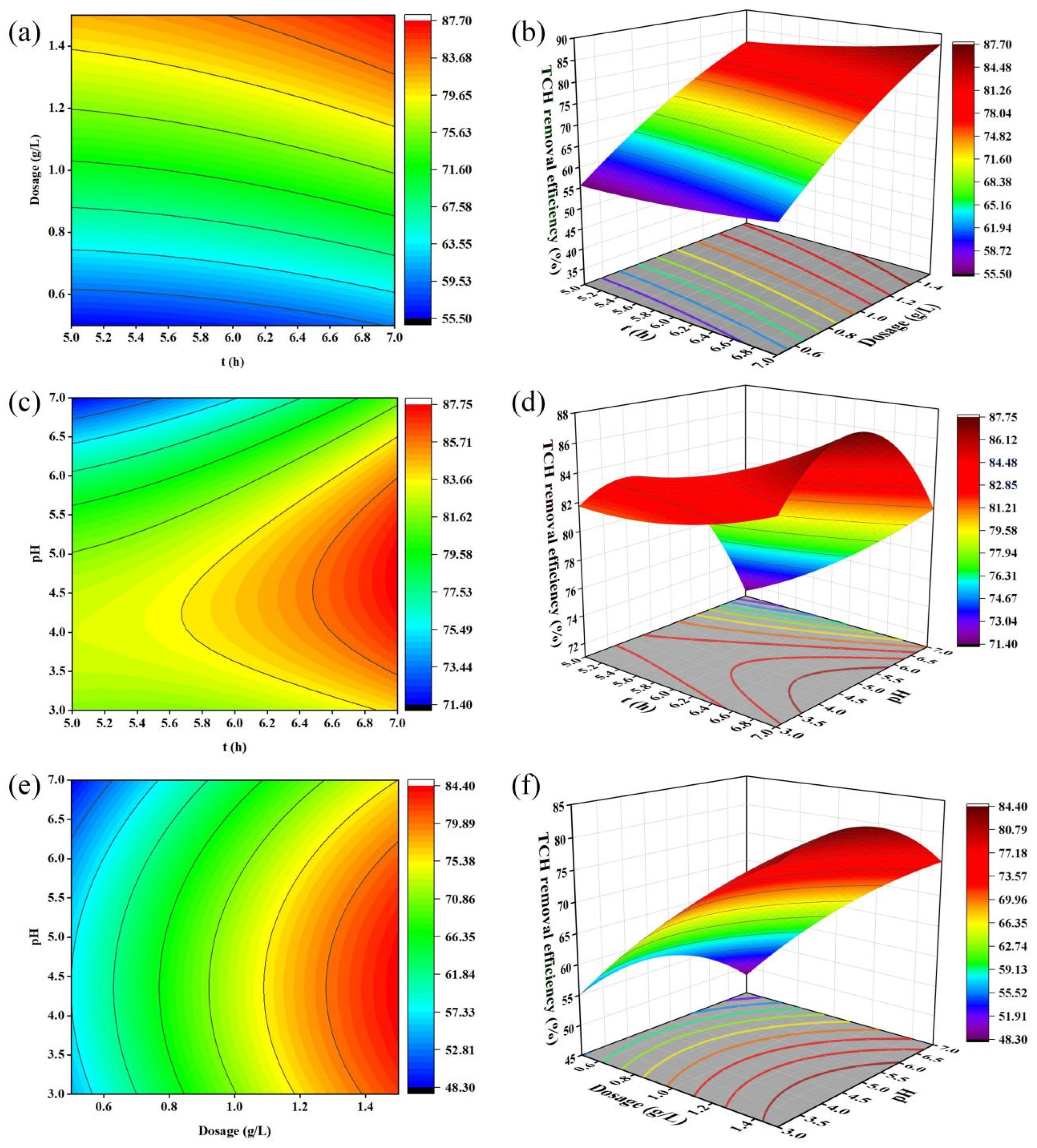
Figure 11.
Contour plots (a,c,e) and response surface plots (b,d,f) showing the interactive effects of different variable pairs on tetracycline (TCH) removal efficiency: (a,b) adsorption time (A) and biochar dosage (B); (c,d) adsorption time (A) and initial solution pH (C); (e,f) biochar dosage (B) and initial solution pH (C). All experiments were conducted with an initial TCH concentration of 20 mg/L and a temperature of 25 °C. The elliptical nature of the contour plots indicates significant interactive effects between the corresponding variables.
Figure 11.
Contour plots (a,c,e) and response surface plots (b,d,f) showing the interactive effects of different variable pairs on tetracycline (TCH) removal efficiency: (a,b) adsorption time (A) and biochar dosage (B); (c,d) adsorption time (A) and initial solution pH (C); (e,f) biochar dosage (B) and initial solution pH (C). All experiments were conducted with an initial TCH concentration of 20 mg/L and a temperature of 25 °C. The elliptical nature of the contour plots indicates significant interactive effects between the corresponding variables.
Figure 12.
(a) Adsorption–desorption cycling performance of KYBC600 for TCH; (b) TCH removal efficiency in different water matrices after KYBC600 addition. Under the experimental conditions (6 h adsorption, 1.5 g/L dosage, 20 mg/L initial TCH, pH 5, 25 °C), the results confirmed that KYBC600 exhibited excellent reusability. Furthermore, it maintained a high TCH adsorption capacity across different aqueous matrices.
Figure 12.
(a) Adsorption–desorption cycling performance of KYBC600 for TCH; (b) TCH removal efficiency in different water matrices after KYBC600 addition. Under the experimental conditions (6 h adsorption, 1.5 g/L dosage, 20 mg/L initial TCH, pH 5, 25 °C), the results confirmed that KYBC600 exhibited excellent reusability. Furthermore, it maintained a high TCH adsorption capacity across different aqueous matrices.
Figure 13.
(a) FTIR spectra of KYBC600 before and after TCH adsorption; (b) XRD patterns of KYBC600 before and after TCH adsorption. All characterizations were performed at ambient temperature. FT-IR spectra were acquired over a range of 500–4000 cm−1 with a resolution of 4 cm−1 and 32 scans. XRD patterns were collected in the 2θ range of 5–85° using a step time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The spectral data suggested the formation of hydrogen bonding and π-π interactions during the adsorption of TCH onto KYBC600.
Figure 13.
(a) FTIR spectra of KYBC600 before and after TCH adsorption; (b) XRD patterns of KYBC600 before and after TCH adsorption. All characterizations were performed at ambient temperature. FT-IR spectra were acquired over a range of 500–4000 cm−1 with a resolution of 4 cm−1 and 32 scans. XRD patterns were collected in the 2θ range of 5–85° using a step time of 0.5 s, an operating voltage of 30 kV, and a current of 20 mA. The spectral data suggested the formation of hydrogen bonding and π-π interactions during the adsorption of TCH onto KYBC600.
Figure 14.
Scanning electron microscopy (SEM) images comparing the morphology of sample KYBC600 before and after tetracycline (TCH) adsorption. (a) Surface morphology of pristine KYBC600; (b) High-resolution view of the pristine KYBC600 surface; (c) Surface morphology of KYBC600 after TCH adsorption (KYBC600-TCH); (d) High-resolution view of the post-adsorption KYBC600-TCH surface. All images were acquired at an accelerating voltage of 2.0 kV. The micrographs revealed that the surface of KYBC600 became relatively smoother after TCH adsorption, with partial coverage of the original pores.
Figure 14.
Scanning electron microscopy (SEM) images comparing the morphology of sample KYBC600 before and after tetracycline (TCH) adsorption. (a) Surface morphology of pristine KYBC600; (b) High-resolution view of the pristine KYBC600 surface; (c) Surface morphology of KYBC600 after TCH adsorption (KYBC600-TCH); (d) High-resolution view of the post-adsorption KYBC600-TCH surface. All images were acquired at an accelerating voltage of 2.0 kV. The micrographs revealed that the surface of KYBC600 became relatively smoother after TCH adsorption, with partial coverage of the original pores.
Figure 15.
(a,b) N 1s and (c) XPS survey spectra of KYBC600 before and after TCH adsorption. Measurement conditions: Monochromatic Al Kα (1486.6 eV), 150 W, 650 μm spot, 14.8 kV, 1.6 A. Results indicate successful TCH adsorption and suggest Ca-TCH complexation.
Figure 15.
(a,b) N 1s and (c) XPS survey spectra of KYBC600 before and after TCH adsorption. Measurement conditions: Monochromatic Al Kα (1486.6 eV), 150 W, 650 μm spot, 14.8 kV, 1.6 A. Results indicate successful TCH adsorption and suggest Ca-TCH complexation.
Figure 16.
High-resolution (a,c) C 1s and (b,d) O 1s XPS spectra of KYBC600 before and after TCH adsorption. Measurement conditions: Monochromatic Al Kα (1486.6 eV), 150 W, 650 μm spot. Results indicate hydrogen bonding and π-π interactions between biochar and TCH.
Figure 16.
High-resolution (a,c) C 1s and (b,d) O 1s XPS spectra of KYBC600 before and after TCH adsorption. Measurement conditions: Monochromatic Al Kα (1486.6 eV), 150 W, 650 μm spot. Results indicate hydrogen bonding and π-π interactions between biochar and TCH.
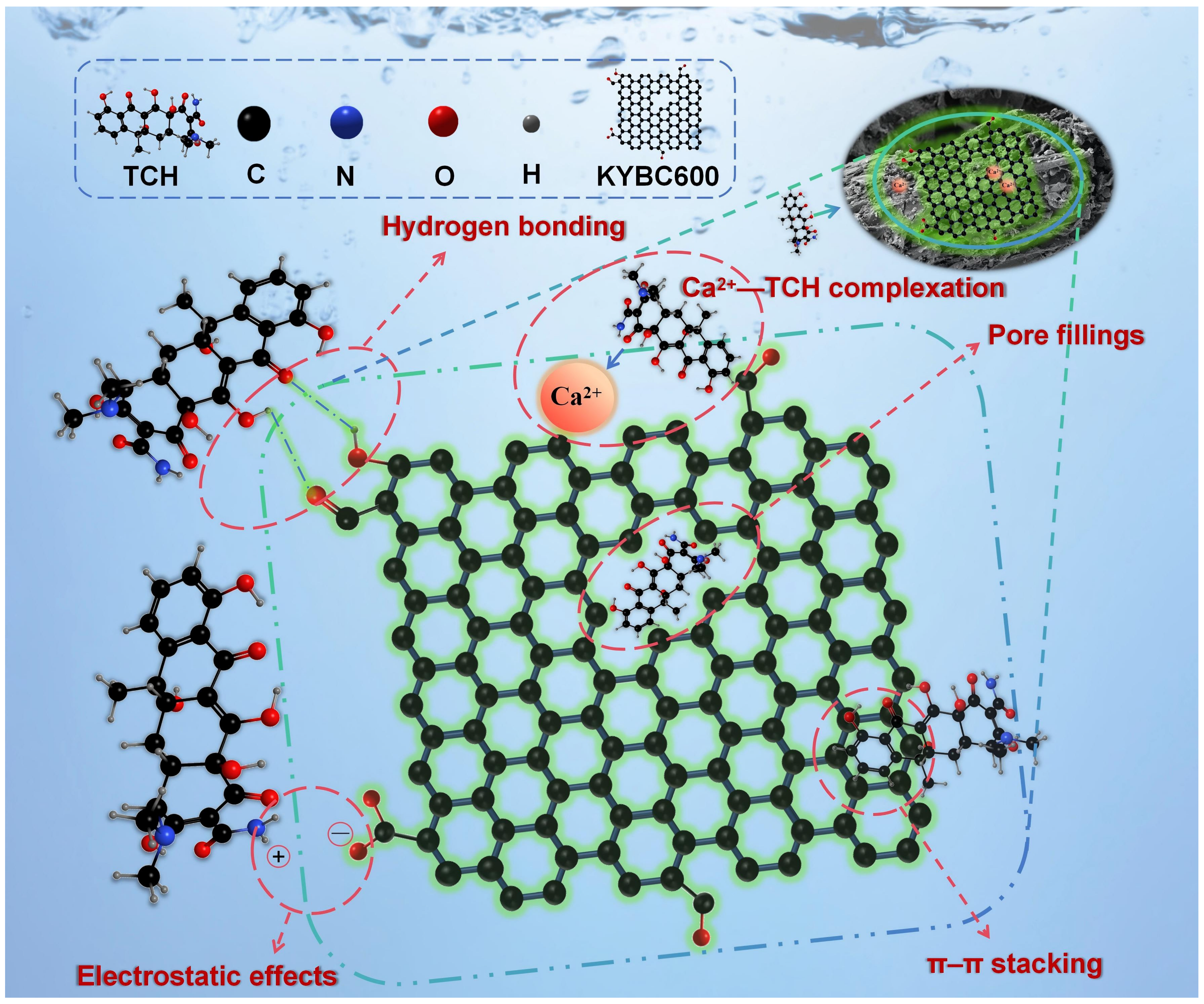
Figure 17.
Possible adsorption mechanism of KYBC600 for TCH in water. The proposed adsorption mechanism of TCH onto KYBC600 involves pore filling, hydrogen bonding, electrostatic interactions, π-π interactions, and complexation.
Figure 17.
Possible adsorption mechanism of KYBC600 for TCH in water. The proposed adsorption mechanism of TCH onto KYBC600 involves pore filling, hydrogen bonding, electrostatic interactions, π-π interactions, and complexation.
Table 1.
The BET specific surface area, pore structure parameters, and yield of yak manure-derived biochar prepared at different pyrolysis temperatures are presented. The BET measurements were conducted using nitrogen and helium gases under liquid nitrogen conditions, following a 30 min pre-treatment of the samples in an oven at 120 °C. The results indicate that YBC600 exhibits a relatively large specific surface area.
Table 1.
The BET specific surface area, pore structure parameters, and yield of yak manure-derived biochar prepared at different pyrolysis temperatures are presented. The BET measurements were conducted using nitrogen and helium gases under liquid nitrogen conditions, following a 30 min pre-treatment of the samples in an oven at 120 °C. The results indicate that YBC600 exhibits a relatively large specific surface area.
| Biochar | Specific Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Diameter (nm) | Yield (%) |
|---|
| YBC500 | 8.69 | 0.022 | 10.17 | 52.05% |
| YBC600 | 26.65 | 0.042 | 6.38 | 51.99% |
| YBC700 | 16.64 | 0.081 | 19.59 | 51.90% |
Table 2.
BET results of biochar before and after KOH modification. The BET measurements were performed using nitrogen and helium gases under liquid nitrogen conditions (77 K), with samples pre-degassed at 120 °C for 30 min prior to analysis. The results indicate that KOH modification effectively enhanced the specific surface area and pore volume of the biochar, leading to an improved pore structure.
Table 2.
BET results of biochar before and after KOH modification. The BET measurements were performed using nitrogen and helium gases under liquid nitrogen conditions (77 K), with samples pre-degassed at 120 °C for 30 min prior to analysis. The results indicate that KOH modification effectively enhanced the specific surface area and pore volume of the biochar, leading to an improved pore structure.
| Biochar | Specific Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Diameter (nm) |
|---|
| YBC600 | 26.65 | 0.042 | 6.38 |
| KYBC600 | 96.06 | 0.101 | 4.21 |
Table 3.
Adsorption kinetic parameters of biochar for TCH. The adsorption kinetics experiments were conducted under the following conditions: a temperature of 25 °C, an initial TCH concentration of 20 mg/L, an initial solution pH of 5, a biochar dosage of 0.5 g/L, and a contact time ranging from 0 to 360 min. The results revealed that the adsorption process of TCH onto both KYBC600 and YBC600 was better described by the pseudo-second-order kinetic model.
Table 3.
Adsorption kinetic parameters of biochar for TCH. The adsorption kinetics experiments were conducted under the following conditions: a temperature of 25 °C, an initial TCH concentration of 20 mg/L, an initial solution pH of 5, a biochar dosage of 0.5 g/L, and a contact time ranging from 0 to 360 min. The results revealed that the adsorption process of TCH onto both KYBC600 and YBC600 was better described by the pseudo-second-order kinetic model.
| Model | Parameter | Biochar |
|---|
| YBC600 | KYBC600 |
|---|
| Pseudo-first-order kinetics | Qe/(mg·g−1) | 11.0468 | 19.3669 |
| k1/(min−1) | 0.0361 | 0.8812 |
| R2 | 0.8920 | 0.9519 |
| Pseudo-second-order kinetics | Qe/(mg·g−1) | 12.0069 | 19.7931 |
| k2/(g·mg−1·min−1) | 0.0048 | 0.0586 |
| R2 | 0.9369 | 0.9765 |
Table 4.
Intraparticle diffusion model parameters for TCH adsorption by biochar. The adsorption experiments were conducted under the following conditions: a temperature of 25 °C, an initial TCH concentration of 20 mg/L, an initial pH of 5, a biochar dosage of 0.5 g/L, and a contact time ranging from 0 to 360 min. The results demonstrated that the TCH adsorption process involved three consecutive stages: film diffusion, intra-particle diffusion, and finally, adsorption–desorption equilibrium.
Table 4.
Intraparticle diffusion model parameters for TCH adsorption by biochar. The adsorption experiments were conducted under the following conditions: a temperature of 25 °C, an initial TCH concentration of 20 mg/L, an initial pH of 5, a biochar dosage of 0.5 g/L, and a contact time ranging from 0 to 360 min. The results demonstrated that the TCH adsorption process involved three consecutive stages: film diffusion, intra-particle diffusion, and finally, adsorption–desorption equilibrium.
| Parameter | Biochar |
|---|
| YBC600 | KYBC600 |
|---|
| ki1/(mg·(g·min1/2)−1) | 1.2293 | 1.9435 |
| C1 | 1.2002 | 9.5926 |
| R12 | 0.7442 | 0.9699 |
| ki2/(mg·(g·min1/2)−1) | 0.7737 | 0.2296 |
| C2 | 2.4690 | 17.3574 |
| R22 | 0.7886 | 0.5666 |
| ki3/(mg·(g·min1/2)−1) | 0.1743 | 0.1654 |
| C3 | 8.6101 | 17.594 |
| R32 | 0.9689 | 0.6798 |
Table 5.
Adsorption thermodynamic parameters of TCH on biochar. The adsorption experiments were conducted at temperatures of 15, 25, and 35 °C with an adsorption time of 6 h, a biochar dosage of 0.5 g/L, an initial TCH concentration of 20 mg/L, and an initial solution pH of 5. The thermodynamic analysis revealed that the adsorption of TCH onto KYBC600 was a spontaneous, endothermic, and entropy-increasing process. Furthermore, KOH modification was found to be more favorable for the adsorption of TCH onto the biochar.
Table 5.
Adsorption thermodynamic parameters of TCH on biochar. The adsorption experiments were conducted at temperatures of 15, 25, and 35 °C with an adsorption time of 6 h, a biochar dosage of 0.5 g/L, an initial TCH concentration of 20 mg/L, and an initial solution pH of 5. The thermodynamic analysis revealed that the adsorption of TCH onto KYBC600 was a spontaneous, endothermic, and entropy-increasing process. Furthermore, KOH modification was found to be more favorable for the adsorption of TCH onto the biochar.
| Biochar | Temperature T/(K) | θ/(kJ·mol−1) | θ/(kJ·mol−1) | θ/(J/mol−1·K−1) |
|---|
| YBC600 | 288.15 | −2.15 | 18.24 | 70.85 |
| 298.15 | −2.87 |
| 308.15 | −3.59 |
| KYBC600 | 288.15 | −4.02 | 25.67 | 103.12 |
| 298.15 | −4.92 |
| 308.15 | −5.81 |
Table 6.
Adsorption isotherm parameters of TCH on biochar. The experiments were conducted under controlled conditions: temperature of 25 °C, adsorption time of 6 h, biochar dosage of 0.5 g/L, initial TCH concentration of 20 mg/L, and initial solution pH of 5. The results demonstrated that the adsorption of TCH onto the biochar primarily followed monolayer chemical adsorption with significant electrostatic interactions. The modified KYBC600 surface was particularly favorable for TCH attachment due to its enhanced surface properties.
Table 6.
Adsorption isotherm parameters of TCH on biochar. The experiments were conducted under controlled conditions: temperature of 25 °C, adsorption time of 6 h, biochar dosage of 0.5 g/L, initial TCH concentration of 20 mg/L, and initial solution pH of 5. The results demonstrated that the adsorption of TCH onto the biochar primarily followed monolayer chemical adsorption with significant electrostatic interactions. The modified KYBC600 surface was particularly favorable for TCH attachment due to its enhanced surface properties.
| Model | Parameter | Biochar |
|---|
| YBC600 | KYBC600 |
|---|
| Langmuir Model | Qm/(mg·g−1) | 16.0424 | 54.1034 |
| KL/(L·mg−1) | 0.0824 | 0.0420 |
| R2 | 0.9381 | 0.9881 |
| Freundlich Model | KF/(mg·g−1)(mg·L−1)−1/n | 3.5525 | 6.2062 |
| 1/n | 3.2072 | 2.3116 |
| R2 | 0.8702 | 0.9438 |
Temkin
Model | KT/(L·g−1) | 0.8522 | 0.4032 |
| b | 741.9726 | 207.7169 |
| R2 | 0.9124 | 0.9804 |
Table 7.
Factors, levels, and codes for the Design Expert experimental design. The experiments were conducted with an initial TCH concentration of 20 mg/L and at a temperature of 25 °C.
Table 7.
Factors, levels, and codes for the Design Expert experimental design. The experiments were conducted with an initial TCH concentration of 20 mg/L and at a temperature of 25 °C.
| Factor | Code | Coded Level |
|---|
| −1 | 0 | 1 |
|---|
| Adsorption time/(h) | A | 5 | 6 | 7 |
| Biochar dosage/(g·L−1) | B | 0.5 | 1 | 1.5 |
| Initial solution pH | C | 3 | 5 | 7 |
Table 8.
Experimental design and results of the BBD model. The experiments were conducted with an initial TCH concentration of 20 mg/L and at a temperature of 25 °C.
Table 8.
Experimental design and results of the BBD model. The experiments were conducted with an initial TCH concentration of 20 mg/L and at a temperature of 25 °C.
| Code | A—Adsorption Time | B—Biochar Dosage | C—Initial Solution pH | TCH Adsorption Rate(%) |
|---|
| 1 | 0 | 1 | −1 | 87.66 |
| 2 | −1 | 1 | 0 | 82.99 |
| 3 | 0 | 0 | 0 | 73.53 |
| 4 | 0 | 0 | 0 | 72.24 |
| 5 | 1 | 0 | −1 | 73.01 |
| 6 | 0 | 1 | 1 | 74.84 |
| 7 | 1 | 0 | 1 | 70.69 |
| 8 | 0 | 0 | 0 | 71.37 |
| 9 | 0 | −1 | 1 | 48.37 |
| 10 | −1 | 0 | 1 | 59.84 |
| 11 | 0 | −1 | −1 | 55.57 |
| 12 | 1 | −1 | 0 | 58.44 |
| 13 | 1 | 1 | 0 | 86.86 |
| 14 | 0 | 0 | 0 | 72.94 |
| 15 | 0 | 0 | 0 | 72.50 |
| 16 | −1 | −1 | 0 | 56.33 |
| 17 | −1 | 0 | −1 | 69.55 |
Table 9.
ANOVA results for response surface methodology data. Under the experimental conditions of an initial TCH concentration of 20 mg/L and a temperature of 25 °C, the model demonstrated a good fit. The results revealed that the order of influence of the three examined factors on the TCH removal efficiency was as follows: biochar dosage (B) > initial solution pH (C) > adsorption time (A).
Table 9.
ANOVA results for response surface methodology data. Under the experimental conditions of an initial TCH concentration of 20 mg/L and a temperature of 25 °C, the model demonstrated a good fit. The results revealed that the order of influence of the three examined factors on the TCH removal efficiency was as follows: biochar dosage (B) > initial solution pH (C) > adsorption time (A).
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Significance |
|---|
| Model | 1747.95 | 9 | 194.22 | 108.56 | <0.0001 | Significant |
| A | 51.53 | 1 | 51.53 | 28.80 | 0.0010 | - |
| B | 1458.34 | 1 | 1458.34 | 898.24 | <0.0001 | - |
| C | 87.30 | 1 | 87.30 | 48.80 | 0.0002 | - |
| AB | 0.7765 | 1 | 0.7765 | 0.4340 | 0.5311 | - |
| AC | 13.66 | 1 | 13.66 | 7.63 | 0.0280 | - |
| BC | 0.0000 | 1 | 0.0000 | 0.0000 | 0.9962 | - |
| A2 | 3.07 | 1 | 3.07 | 1.72 | 0.2315 | - |
| B2 | 20.70 | 1 | 20.70 | 11.57 | 0.0114 | - |
| C2 | 109.41 | 1 | 109.41 | 61.15 | 0.0001 | - |
| Residual | 12.52 | 7 | 1.79 | - | - | - |
| Lack of fit | 9.94 | 3 | 3.31 | 5.14 | 0.0738 | Not significant |
| Pure error | 2.58 | 4 | 0.6448 | - | - | - |
| Total | 1760.47 | 16 | - | - | - | - |
| R2 = 0.9929, = 0.9837, = 0.9073, C.V. = 1.93%, Precision = 38.2507 |
Table 10.
Validation results under optimal adsorption conditions.
Table 10.
Validation results under optimal adsorption conditions.
| Optimal Adsorption Conditions | Actual TCH Removal Rate(%) |
|---|
| Adsorption Time(h) | Biochar Dosage(g·L−1) | Initial Solution pH | 1 | 2 | 3 | Mean Value |
|---|
| 7 | 1.5 | 4.709 | 86.493 | 85.136 | 84.891 | 85.507 |
Table 11.
Comparison of TCH Adsorption by Biochar in Aqueous Solution. Comparative analysis reveals that the TCH adsorption performance of KYBC600 is superior to that of most reported adsorbents.
Table 11.
Comparison of TCH Adsorption by Biochar in Aqueous Solution. Comparative analysis reveals that the TCH adsorption performance of KYBC600 is superior to that of most reported adsorbents.
| Feedstock | Maximum Adsorption Capacity (mg·g−1) | Actual Removal Capacity/Rate
(Reported Conditions) (mg·g−1) | TCH Residual Concentration/(mg/L) | Primary Adsorption Mechanism | References |
|---|
| Wood Ear Residue | 11.90 | 4.13 | 1.26 | π-π electron donor-acceptor interaction | [41] |
| Shiitake Spent | 37.95 | ~22.14 | 77.86 | Pore filling, π-π interaction, complexation, and hydrogen bonding | [81] |
| Grapefruit Peel | 34.58 | 37.92 | 2.6 | Pore filling, charge interaction, and chemical bonding | [106] |
| Wheat Straw | 55.23 | 31.48 | 11.85 | Pore filling, hydrogen bonding, π-π interaction, and electrostatic interaction | [107] |
| Rice Straw | 50.72 | ~96% Removal | - | π-π interaction and electrostatic interaction | [108] |
| Rice Husk | 46.95 | 39.36 | 60.64 | Hydrogen bonding, hydrophobic interaction, and π-π interaction | [109] |
| Corn Stalk | 33.53 | 17.66 | 2.34 | Hydrogen bonding and π-π interaction | [110] |
| AA Residue | 42.31 | ~40.1 | - | Hydrogen bonding interaction | [111] |
| Microalgae | 42.7 | ~90% Removal | - | Physical adsorption (attributed to enhanced specific surface area and improved pore structure), coupled with the presence of nitrogen elements | [112] |
| Waste Fiberboard | 6.37 | 5.49 | 6.28 | π-π interaction | [113] |
| Cattle Manure | 5.38 | 2.25 | 0.99 | Hydrophobic interaction and π-π electron donor-acceptor interaction | [114] |
| Yak Manure | 54.10 | ~85.51% Removal | 2.94 | Pore filling, electrostatic interaction, π–π interaction, hydrogen bonding, and Ca2+–TCH complexation | This
study |