Abstract
Garnet-type materials consisting of Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2), combined with Eu3+ or Ce3+ activator ions, were prepared by a solid-state method to determine the structural and optical correlations. The structure of Y3Al5-2x(Mg,Ge)xO12 (x = 1, 2) was determined to be a cubic unit cell (Ia-3d), which contains an 8-coordinated Y3+ site with octahedral (Mg,Al)O6 and tetrahedral (Al,Ge)O4 polyhedra, using synchrotron powder X-ray diffraction. When Eu3+ or Ce3+ ions were substituted for the Y3+ site in the Y3Al5-2x(Mg,Ge)xO12 host lattices, the emission spectra showed a decrease in the magnetic dipole f-f Eu3+ transition and a redshift of the d-f Ce3+ transition, related to centrosymmetry and crystal field splitting, respectively. These changes were monitored according to the increase in Mg2+ and Ge4+ contents. The dodecahedral and octahedral edge sharing was identified as a key distortion factor for the structure-correlated luminescence in the Eu3+/Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 garnet phosphors.
1. Introduction
Ce3+-doped Y3Al5O12 (YAG) phosphor has been widely utilized as a smart light source in conjunction with blue LED chips [1,2,3]. Initially developed in 1967 by G. Blasse and A. Bril, the yellow Ce3+-activated YAG phosphor was prepared for use in flying-spot cathode-ray tubes for color television, emitting intense yellow light via the 5d-4f transition of Ce3+ ions within the cubic garnet YAG structure [4]. The garnet mineral belongs to the nesosilicate subclass, characterized by isolated tetragonal polyhedra [5]. The YAG garnet structure, a cubic crystal system (Ia-3d), consists of dodecahedral YO8, octahedral AlO6, and tetrahedral AlO4 units. The local dodecahedral YO8 polyhedra within the garnet structure exhibit edge-sharing with YO8 and AlO6 polyhedra, as well as vertex-sharing with isolated AlO4 tetrahedra [6,7,8,9]. In garnet host lattices, Ce3+ activator ions can occupy the dodecahedral site, influencing crystal field splitting and resulting in a shift of the d–f transition [10,11,12]. Researchers J. Ueda and S. Tanabe have explored the effects of crystal and electronic structures on Ce3+-doped YAG, particularly regarding the crystal field splitting of the lowest 5d levels and a new distortion parameter estimated by the ratio of dodecahedral edges [6]. Similarly, Eu3+ activator ions provide insight into site-resolved luminescence, distinguishing between centrosymmetric and non-centrosymmetric sites in garnet-type phosphors using the magnetic dipole and electric dipole transitions observed in emission spectra [13,14,15]. In this study, the structure of single-phase Y3Al5-2x(Mg,Ge)xO12 (x = 1, 2) garnet materials was determined using synchrotron X-ray analysis, revealing a cubic unit cell with Iad symmetry. The cell parameters, volume, and distances within the host lattices were discussed. By substituting Ce3+ or Eu3+ activator ions into Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2), correlations between emission spectra, dipole transitions, and crystal field splitting were investigated in relation to structural distortion parameters.
2. Materials and Methods
Garnet materials doped with Eu3+- or Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) compounds were synthesized by mixing appropriate stoichiometric amounts of powdered Y2O3 (Alfa, 99.9%), MgO (Alfa, 99.95%), Al2O3 (Alfa, 99.95%), GeO2 (Alfa, 99.999%), Eu2O3 (Alfa, 99.9%), and CeO2 (Aldrich, 99.9%), along with 5 wt% LiF (Alfa, 99.98%) or Li2CO3 (Alfa, 99%) flux. The powdered precursors were mixed in an agate mortar and pestle and then heated at 950 °C and 1400 °C for 6 h in air in a box furnace. The obtained powder samples containing the CeO2 precursor underwent additional reheating at 1000 °C for 12 h in a 5% H2/95% N2 atmosphere. Phase identification was conducted using a powder diffractometer (Cu Kα radiation; Shimadzu XRD-6000, Kyoto, Japan), and structural analysis of the obtained garnet materials was performed using synchrotron powder X-ray diffraction (λ = 0.65303 Å). The new structural data were collected at the PLS-II 6D UNIST-PAL beamline of the Pohang Accelerator Laboratory (PAL) [16]. The garnet Y3Al5-2x(Mg,Ge)xO12 (x = 1 and 2) structures were refined using the Rietveld refinement program FullProf Suite [17,18]. Excitation and emission photoluminescence spectra of the powdered phosphors were obtained via UV spectroscopy using spectrofluorometers (FluoroMate FS-2, Scinco, Seoul, Korea).
3. Results and Discussion
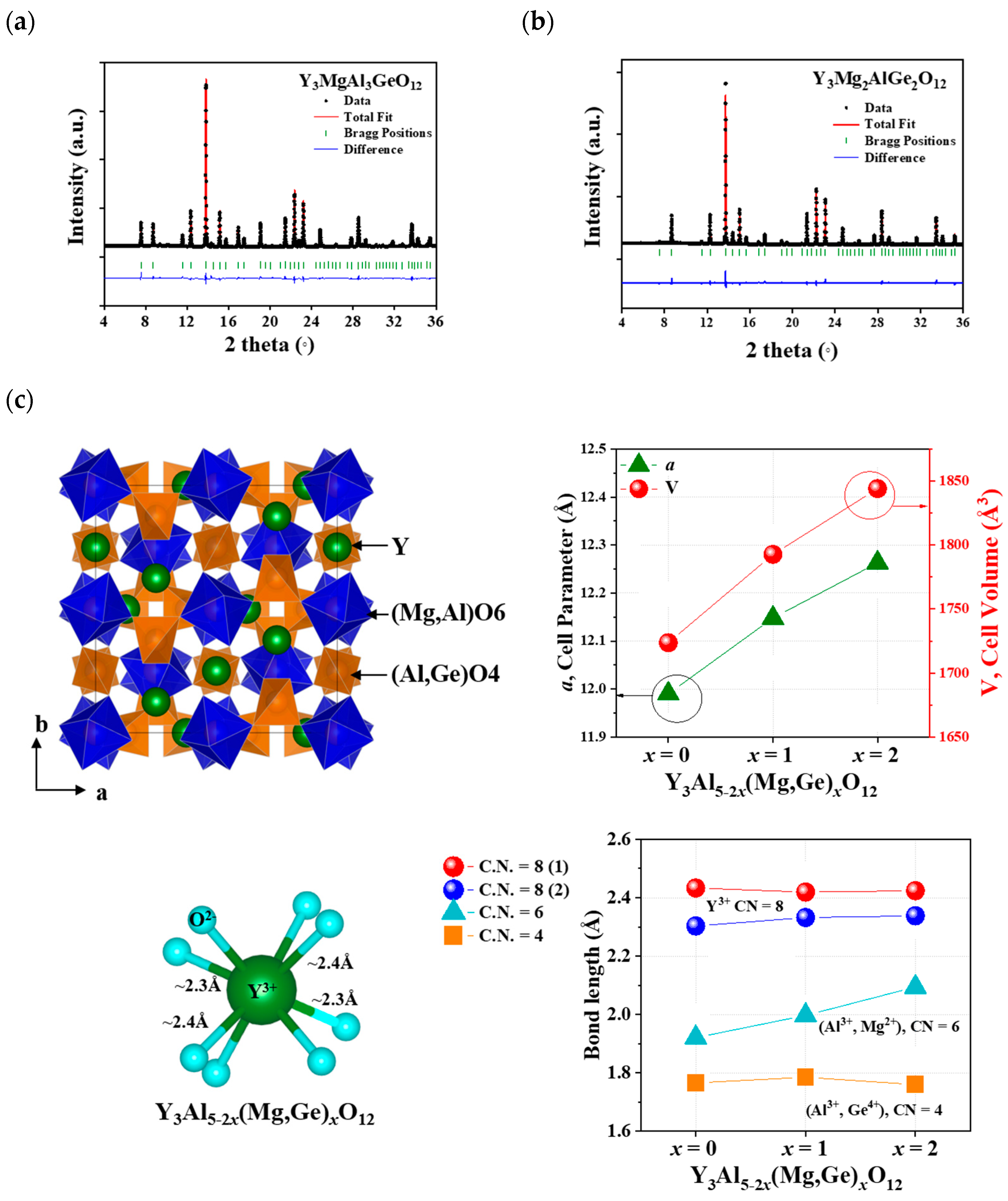
Figure 1a,b illustrate the Rietveld refinement fitting of the powdered X-ray diffraction (XRD) data for Y3Al5-2x(Mg,Ge)xO12 (x = 1 and 2). The summarized structural data are provided in Table 1, Table 2 and Table 3. A single phase of the garnet structure, determined to be a cubic unit cell (Iad), was obtained through a solid-state reaction method. This cubic phase of garnet, including Y3Al5O12 (YAG, ICSD 170157) and Y3Al5-2x(Mg,Ge)xO12 (x = 1 and 2) structures, comprises 8-, 6-, and 4-coordinated Y3+, Al3+(1)-Mg2+, and Al3+(2)-Ge4+ ions, respectively, occupying 24c, 16a, and 24d Wyckoff sites. The 6-coordinated Mg2+ (with a radius of 0.72 Å for 6 coordination number (CN)), Al3+(1) (with a radius of 0.535 Å for 6 CN), and 4-coordinated Al3+ (with a radius of 0.39 Å for 4 CN) and Ge4+ (with a radius of 0.39 Å for 4 CN) sites are suitable for substitutions in the garnet structure [19]. Therefore, the formula for the garnet-structured Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) can be expressed as Y3Al(1)2-xMgxAl(2)3-xGexO12 based on the ionic radii of the cations in the unit cell, as depicted in Figure 1c. Similar to the isolated AlO4 tetrahedra in the YAG structure, the tetrahedral (Al,Ge)O4 polyhedron in the Y3Al5-2x(Mg,Ge)xO12 structures is also isolated, with no sharing of O atoms. There are two different bond distances between Y3+ and O2− ions in the YO8 polyhedron, whereas a single bond distance is observed in the (Mg,Al)O6 and (Al,Ge)O4 polyhedra within the host lattices. The YO8 polyhedron shares edges with nearby YO8 polyhedra and (Mg,Al)O6 octahedra. YAG exhibits lattice parameters and volume, such as a = 11.9900(14) Å and V = 1723.68 Å3 (as shown in Table 1). The cell parameters and volumes of Y3Al5-2x(Mg,Ge)xO12 (x = 1 and 2) are larger than those of YAG compounds, such as Y3MgAl3GeO12 (a = 12.1479(2) Å and V = 1796.82(17) Å3) and Y3Mg2AlGe2O12 (a = 12.2628(1) Å and V = 1844.027(14) Å3). The Y–O bond distances of an 8-coordinated Y (with a radius of 1.019 Å) comprise four long distances (2.433 Å) and four short distances (2.303 Å) in the YAG structure. The Y–O bond distances in Y3MgAl3GeO12 and Y3Mg2AlGe2O12 host lattices remain consistent with those of the YAG structure, as shown in Figure 1c. However, the bond distances of a 6-coordinated (Mg,Al)-O6 and Mg-O6 exhibit distinct increases from 1.921 Å (for Al-O in YAG) to 1.997 Å and 2.094 Å, respectively, representing 4% and 9% differences. Conversely, the bond distances of Al–O4 and (Al,Ge)-O4 tetrahedra in the garnet structures remain similar, ranging from 1.766 to 1.785 Å and 1.760 Å. Interestingly, with the increase in the cell parameter and volume, the bond distance increase is observed primarily in the octahedral polyhedron in the Y3Al5-2x(Mg,Ge)xO12 structure when x = 0, 1, and 2.
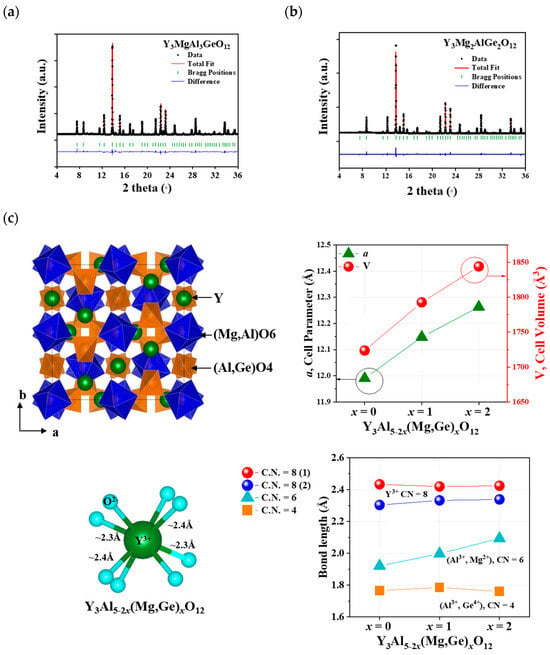
Figure 1.
Synchrotron XRD patterns of Y3Al5-2x(Mg,Ge)xO12 (a) x = 1 and (b) x = 2 and (c) the garnet Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) structure with cell parameters and cell volumes.

Table 1.
Rietveld refinement and crystal data for Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2).

Table 2.
Refined atomic coordinates of Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2).

Table 3.
Selected interatomic distances for Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2).
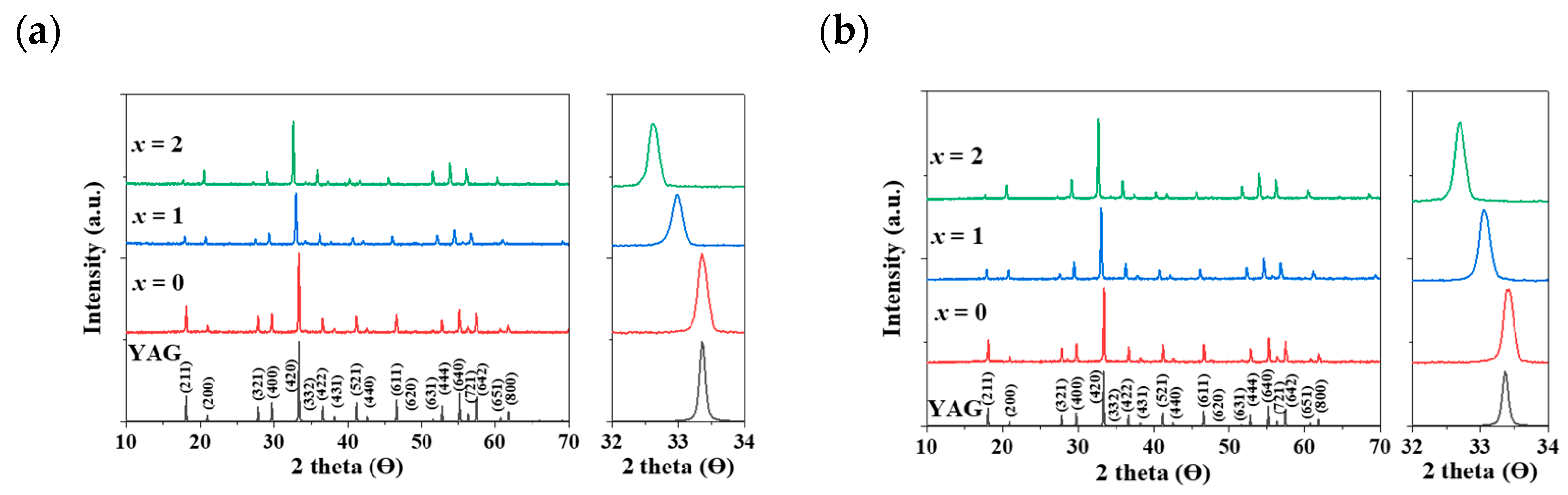
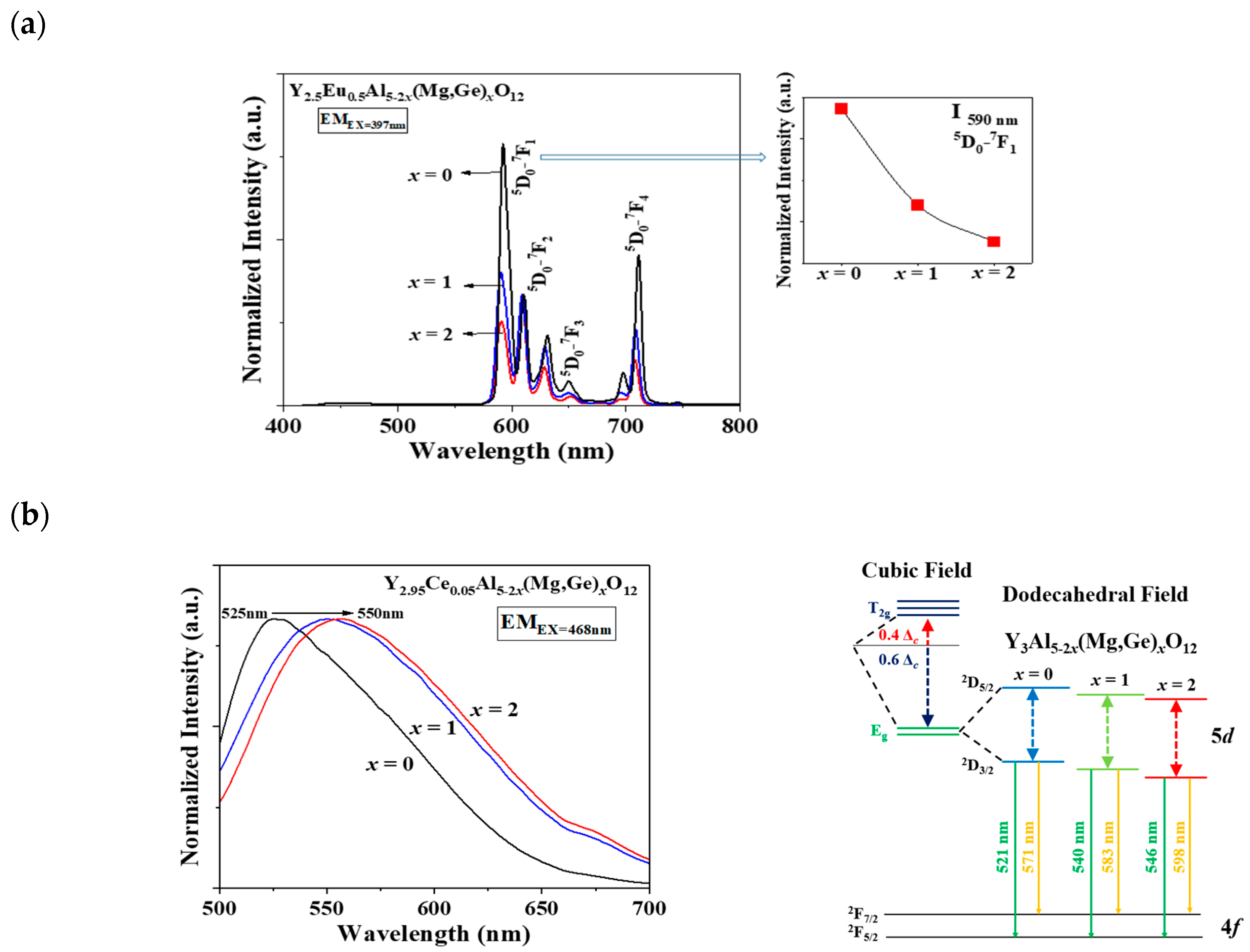
In Figure 2a,b, a distinct shift in the apparent peaks to lower angles, particularly those at 2θ = 32–34°, was observed as Al3+ ions were gradually replaced by Mg2+ and Ge4+ ions in the Eu3+- and Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors, respectively. A single phase of the garnet structure with a cubic crystal system (Iad) was obtained, free from any apparent impurities. The cell volumes of both Y2.5Eu0.5Al5-2x(Mg,Ge)xO12 and Y2.95Ce0.05Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors increased as the content of Mg2+ and Ge4+ increased. Figure 3a displays the emission photoluminescence (PL) spectra of Y2.5Eu0.5Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors. The electronic f-f transitions of Eu3+ ions in the host lattices are assigned as 5D0–7F1, 5D0–7F2, 5D0–7F3, and 5D0–7F4 within the range of 550 and 750 nm [13,14,15]. It is known that when Eu3+ ions are located at the centrosymmetric site in a crystal structure, the magnetic dipole transition (5D0–7F1) dominates, whereas, in the absence of inversion Eu3+ ions in the host lattice, the electric dipole transition (5D0–7F2) dominates [13,14,15]. The centrosymmetric symmetry of the local-environment-center Eu3+ ions in the Y3Al5-2x(Mg,Ge)xO12 structure was inferred from the normalized intensity ratio of the magnetic dipole transition, as depicted in Figure 3a, Figures S1 and S2. All intensity values in the emission spectra were normalized by dividing them by the maximum intensity value of the electric dipole transition peak. As Eu3+ ions occupy the 8-coordinated Y3+ site in the Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) structures, the dominant magnetic dipole transition around 590 nm was noticeably decreased up to x = 0, 1, and 2. This indicates that the ideal cubic field, characterized by a centrosymmetric Y3+ center (D4h point group), was gradually distorted to a dodecahedral field with the substitution of Mg2+ and Ge4+ ions. Figure 3b presents the emission spectra of Y2.95Ce0.05Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors (S1 and 2) and relative energy diagrams of cubic and distorted cubic polyhedrons, showcasing the 8-coordinated site geometry for the 5d Ce3+ orbital energy levels in the host materials. Upon substituting Mg2+ and Ge4+ ions for Al3+ ions in the structure, the distorted 8-coordinated Ce3+ ions in the YAG exhibit a significant redshift of the eg orbital splitting from a normal cubic polyhedron, resulting in emissions from the red-shifted 5d energy level caused by the high-crystal-field effect [10,11,12].
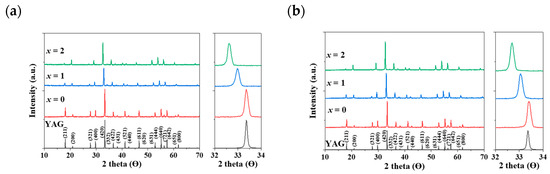
Figure 2.
The calculated XRD pattern of YAG with Miller indices and obtained XRD patterns: (a) Eu3+-doped and (b) Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors.
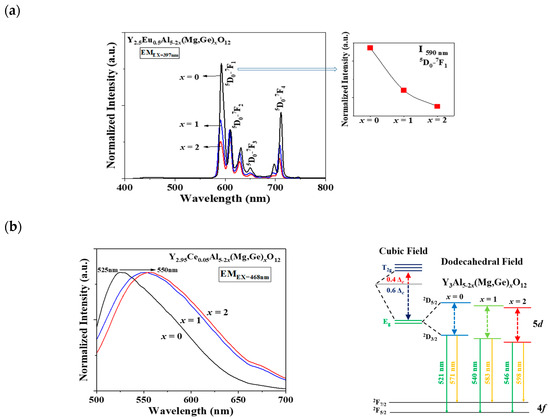
Figure 3.
The emission spectra of (a) Eu3+-doped and (b) Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors with relative centrosymmetric intensity and energy-level diagram.
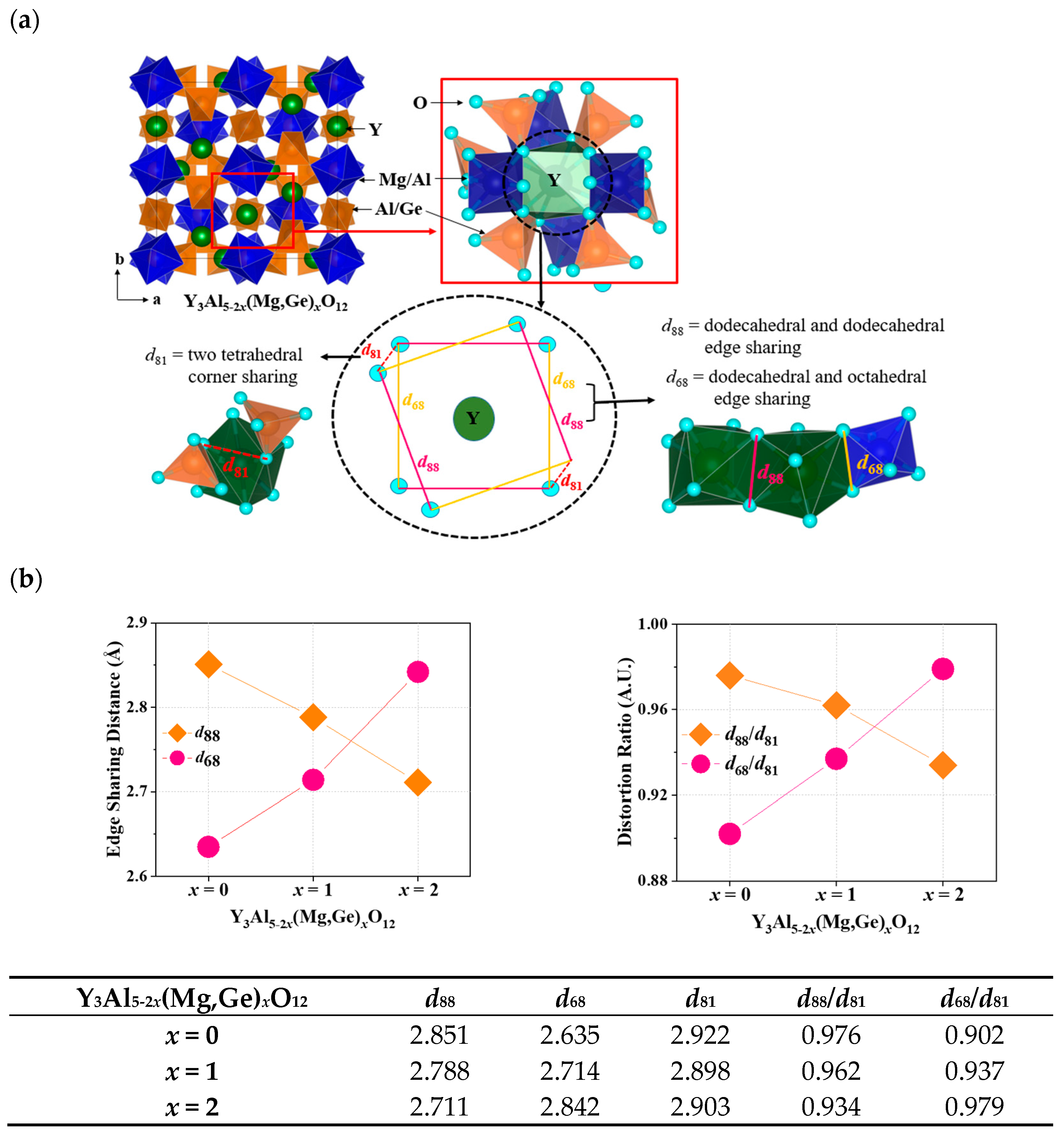
In Figure 4a, the unit cell and local structure of the dodecahedral site in Y3Al5-2x(Mg,Ge)xO12 are depicted. Ce3+ ions engaged in the Y3+ site of garnet structures can emit light due to the distortion factor expressed by the ratio of dodecahedral edges. The four short and long Y(Ce)-O bond distances in the garnet structures can be influenced by the deviation of O-O bonds shared with adjacent dodecahedra (d88) and two non-shared tetrahedra (d81) [6]. This deviation in the local structural arrangement around the Ce3+ ions within the garnet lattice can have an impact on the energy levels of the Ce3+ ions [6]. As distortion increases due to the compression on the cube, the maximum emission shifts to longer wavelengths according to the lower excited state of energy level in the cubic crystal field splitting. Figure 4b summarizes distortion parameters including d88, d68, d81, d88/d81, and d68/d81 for Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors. The edge-sharing distance of d88 and distortion parameter of d88/d81 gradually decreased by substituting Mg-Ge ions in the structures, indicating that the compression of the cube was released by the decrease in distortion. However, the maximum emission wavelength of the Ce3+ d-f transition shifts to longer wavelengths, as shown in Figure 3. Furthermore, the centrosymmetric Eu3+ emission decreased when x = 0 to 1 and 2 in Eu3+-doped Y3Al5-2x(Mg,Ge)xO12 garnet phosphors. When Mg2+-Ge4+ ions were doped into the Y3Al5O12 structure, the bond length of Mg-O among Y-O, Al-O, and Ge-O bonds from the structure analysis solely increased with increasing cell volumes, as shown in Figure 1c. For the distorted Eu3+ and Ce3+ cubic sites in the Y3Al5-2x(Mg,Ge)xO12 garnet structures, the parameter of dodecahedral and octahedral edge-sharing should be estimated as a distortion factor. Figure 4b illustrates the steady increase in edge-sharing distance d68 and distortion ratio of d68/d81 in terms of key distortion parameters, resulting in the non-centrosymmetric manners and the redshift of the maximum emission wavelength.
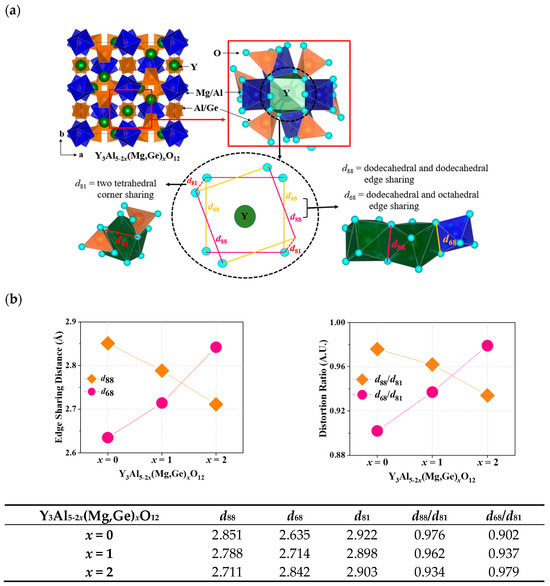
Figure 4.
(a) Unit cell and local structure of the dodecahedral site in Y3Al5-2x(Mg,Ge)xO12 (b) distortion parameters of d88, d68, d81, d88/d81, and d68/d81 for Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors.
4. Conclusions
Eu3+/Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) phosphors were synthesized using a solid-state reaction method assisted by excess LiF or Li2CO3 flux at high temperature. Synchrotron powder X-ray diffraction analysis confirmed that the single-phase of Y3Al5-2x(Mg,Ge)xO12 (x = 1 and 2) garnet materials possessed a cubic unit cell (Iad) with lattice parameters of a = 12.1479(2) Å and a = 12.2628(1) Å, respectively. These garnet structures consisted of an 8-coordination environment for Y3+ with (Mg2+,Al3+(1))O6 octahedra and (Al3+(2),Ge4+)O4 tetrahedra. Interestingly, the distance value of the Mg,Al(1)O6 octahedra only increased with the doping of Mg2+ and Ge4+ ions into the YAG host lattice. Upon doping Eu3+ or Ce3+ ions into the Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) garnet structure, the resulting phosphors exhibited increases in non-centrosymmetric and strong crystal field splitting manners with increasing contents of Mg2+ and Ge4+ ions. This indicated that the cubic polyhedra were compressed by the increase in distortion. The distortion factor of dodecahedral–dodecahedral edge sharing/non-edge sharing (d88/d81) decreased with the substitution of Mg2+-Ge4+ ions, while the distortion factor of dodecahedral–octahedral edge sharing/non-edge sharing (d68/d81) gradually increased. In Y3Al5-2x(Mg,Ge)xO12 (x = 0, 1, 2) garnet structures, when substituting ions in octahedral and tetrahedral sites, the dodecahedral–octahedral edge sharing/non-edge sharing (d68/d81) should be considered as the most crucial distortion parameter.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ma17102445/s1, Figure S1: The X-ray diffraction patterns and excitation and emission spectra of Eu3+- and Ce3+-doped Y3Al5-2x(Mg,Ge)xO12 ((A) x = 0, (B) x =1, (C) x =2) phosphors.; Figure S2: The excitation and emission spectra of (A) Y2.5Eu0.5MgxAl5-2xGexO12 and (B) Y2.95Ce0.05MgxAl5-2xGexO12 (x = 0–2).
Author Contributions
S.P.: Supervision, Conceptualization, Methodology, Writing—review and editing. H.H.: Synthesis, Data curation, Software, Writing—original draft. S.Y.: Data curation, Software, Writing—original draft. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the “Regional Innovation Strategy (RIS)” through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (MOE) (2023RIS-007).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The authors state that data supporting the study in the manuscript are available on reasonable request. Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Naira, G.B.; Swarta, H.C.; Dhoble, S.J. A review on the advancements in phosphor-converted light emitting diodes (pc-LEDs): Phosphor synthesis, device fabrication and characterization. Prog. Mater. Sci. 2020, 109, 100622. [Google Scholar] [CrossRef]
- Ye, S.; Xiao, F.; Parn, Y.X.; Ma, Y.Y.; Zhang, Q.Y. Phosphors in phosphor-converted white light-emitting diodes: Recent advances in materials, techniques and properties. Mater. Sci. Eng. R 2010, 71, 1–34. [Google Scholar] [CrossRef]
- Schubert, E.F.; Kim, J.K. Solid-state light sources getting smart. Science 2005, 308, 1274–1278. [Google Scholar] [CrossRef] [PubMed]
- Blasse, G.; Bril, A. A new phosphor for flying-spot cathode-ray tube for color television: Yellow-emitting Y3Al5O12-Ce3+. Appl. Phys. Lett. 1967, 11, 53–55. [Google Scholar] [CrossRef]
- Kim, Y.; Park, S. Eu2+, Mn2+ co-doped Ba9Y2Si6O24 phosphors based on near-UV-excitable LED lights. Mater. Res. Bull. 2014, 49, 469–474. [Google Scholar] [CrossRef]
- Ueda, J.; Tanabe, S. Review of luminescent properties of Ce3+-doped garnet phosphors: New insight into the effect of crystal and electronic structure. Opt. Mater. X 2019, 1, 100018. [Google Scholar] [CrossRef]
- Xia, Z.; Meijerink, A. Ce3+-Doped garnet phosphors: Composition modification, luminescence properties and applications. Chem. Soc. Rev. 2017, 46, 275–299. [Google Scholar] [CrossRef] [PubMed]
- Berends, A.C.; van de Haar, M.A.; Krames, M.R. YAG:Ce3+ Phosphor: From Micron-Sized Workhorse for General Lighting to a Bright Future on the Nanoscale. Chem. Rev. 2020, 120, 13461–13479. [Google Scholar] [CrossRef] [PubMed]
- Dobrzycki, Ł.; Bulska, E.; Pawlak, D.A.; Frukacz, Z.; Woźniak, K. Structure of YAG Crystals Doped/Substituted with Erbium and Ytterbium. Inorg. Chem. 2004, 43, 7656–7664. [Google Scholar] [CrossRef] [PubMed]
- Shang, M.; Fan, J.; Lian, H.; Zhang, Y.; Geng, D.; Lin, J. A Double Substitution of Mg2+−Si4+/Ge4+ for Al(1)3+−Al(2)3+ in Ce3+-Doped Garnet Phosphor for White LEDs. Inorg. Chem. 2014, 53, 7748–7755. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Li, W.; Xu, Y.; Hu, Q.; Zheng, Y. Structure and redshift of Ce3+ emission in anisotropically expanded garnet phosphor MgY2Al4SiO12:Ce3+. RSC Adv. 2016, 6, 20458–20466. [Google Scholar] [CrossRef]
- He, C.; Ji, H.; Huang, Z.; Wang, T.; Zhang, X.; Liu, Y.; Fang, M.; Wu, X.; Zhang, J.; Min, X. Red-shifted emission in Y3MgSiAl3O12:Ce3+ garnet phosphor for blue light-pumped white light-emitting diodes. J. Phys. Chem. C 2018, 122, 15659–15665. [Google Scholar] [CrossRef]
- Yen, W.M.; Shionoya, S. Phosphor Handbook; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Park, C.; Park, S. Centrosymmetric and non-centrosymmetric structural and optical study of Eu3+ ions in (Ba,Ca,Na)9(Al,Y)2Si6O24 orthosilicate phosphors. Opt. Mater. 2022, 123, 111863. [Google Scholar] [CrossRef]
- Pater, D.K.; Vishwanadh, B.; Sudarsan, V.; Kulshreshtha, S.K. Difference in the Nature of Eu3+ Environment in Eu3+-Doped BaTiO3 and BaSnO3. J. Am. Ceram. Soc. 2013, 96, 3857–3861. [Google Scholar]
- Shin, S.; Yang, S.; Lee, S.-H.; Shin, T.J.; Park, S. Distinctive occurrences of green-yellow luminescence from orthogermanate-type Ba9Y2(GeO4)6:Ce3+, Na+ phosphors under blue excitation and white-light performance with light-emitting diodes. J. Alloys Compd. 2022, 897, 163213. [Google Scholar] [CrossRef]
- Rodríguez-Carvajal, J.; Roisnel, T. FullProf.98 and WinPLOTR New Windows Applications for Diffraction. Commission on Powder Diffraction, IUCr, Newsletter, 20 May–August 1998.
- Rodríguez-Carvajal, J. Recent developments of the program FullProf. Commission on Powder Diffraction, IUCr, Newsletter, 26 December 2001. [Google Scholar]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides. Acta Cryst. 1976, A32, 751–767. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).