Hierarchical Hybrid Coatings with Drug-Eluting Capacity for Mg Alloy Biomaterials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. PEO Treatment
2.3. BF PCL Coating
2.4. Cell Culture
2.5. Drug Cytotoxicity
2.6. Cytocompatibility
2.7. Coating Characterization
2.8. Drug Release
2.9. Statistical Analysis
3. Results and Discussion
3.1. Coating Characterization
3.2. Paracetamol and Ciprofloxacin Release from BF PCL Layer
3.3. In Vitro Cytotoxicity Evaluation of Paracetamol and Ciprofloxacin
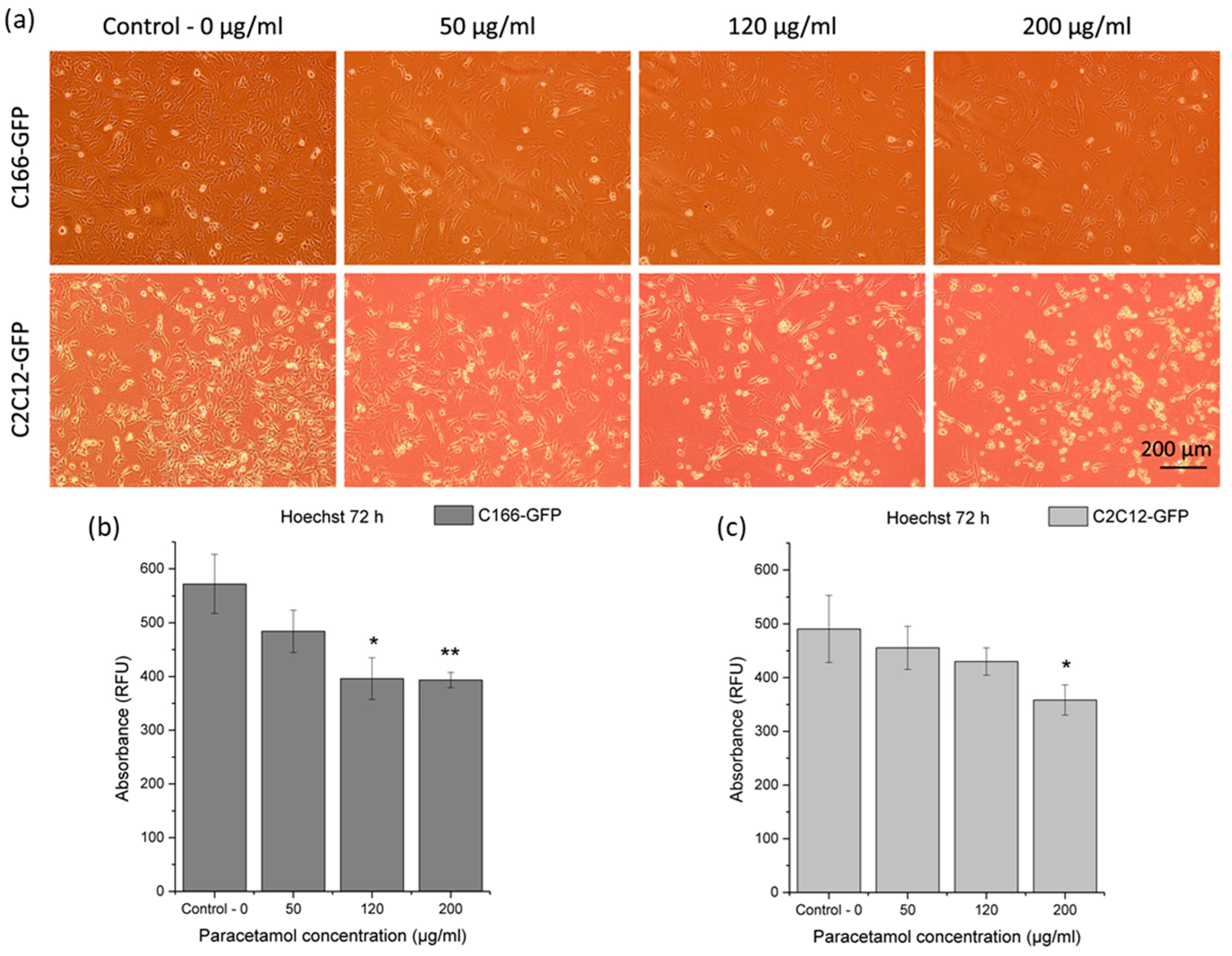
3.3.1. Cytotoxicity of Effect of Released Paracetamol on Premyoblast and Endothelial Cells
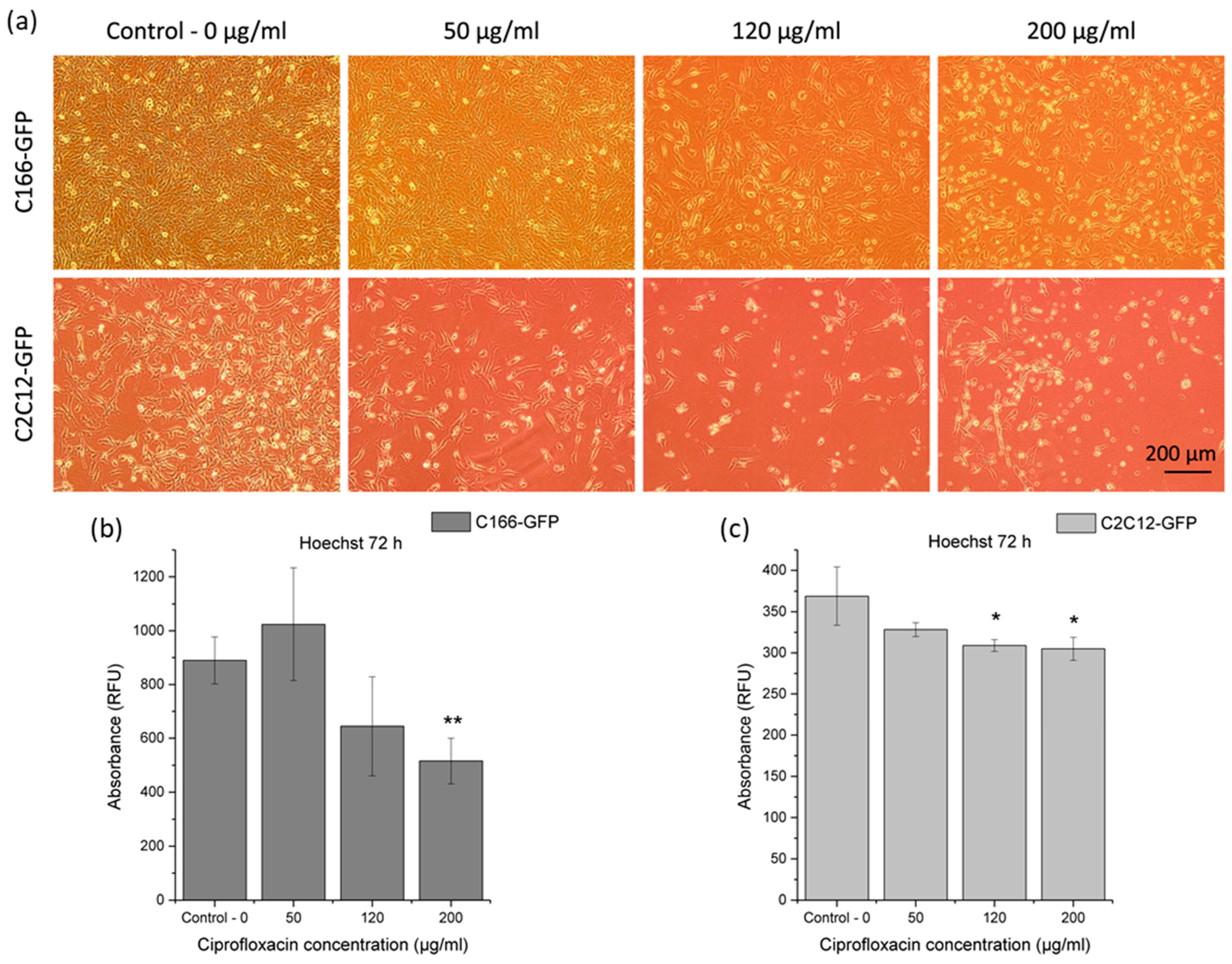
3.3.2. Cytotoxicity Effect of Released Ciprofloxacin on Premyoblast and Endothelial Cells
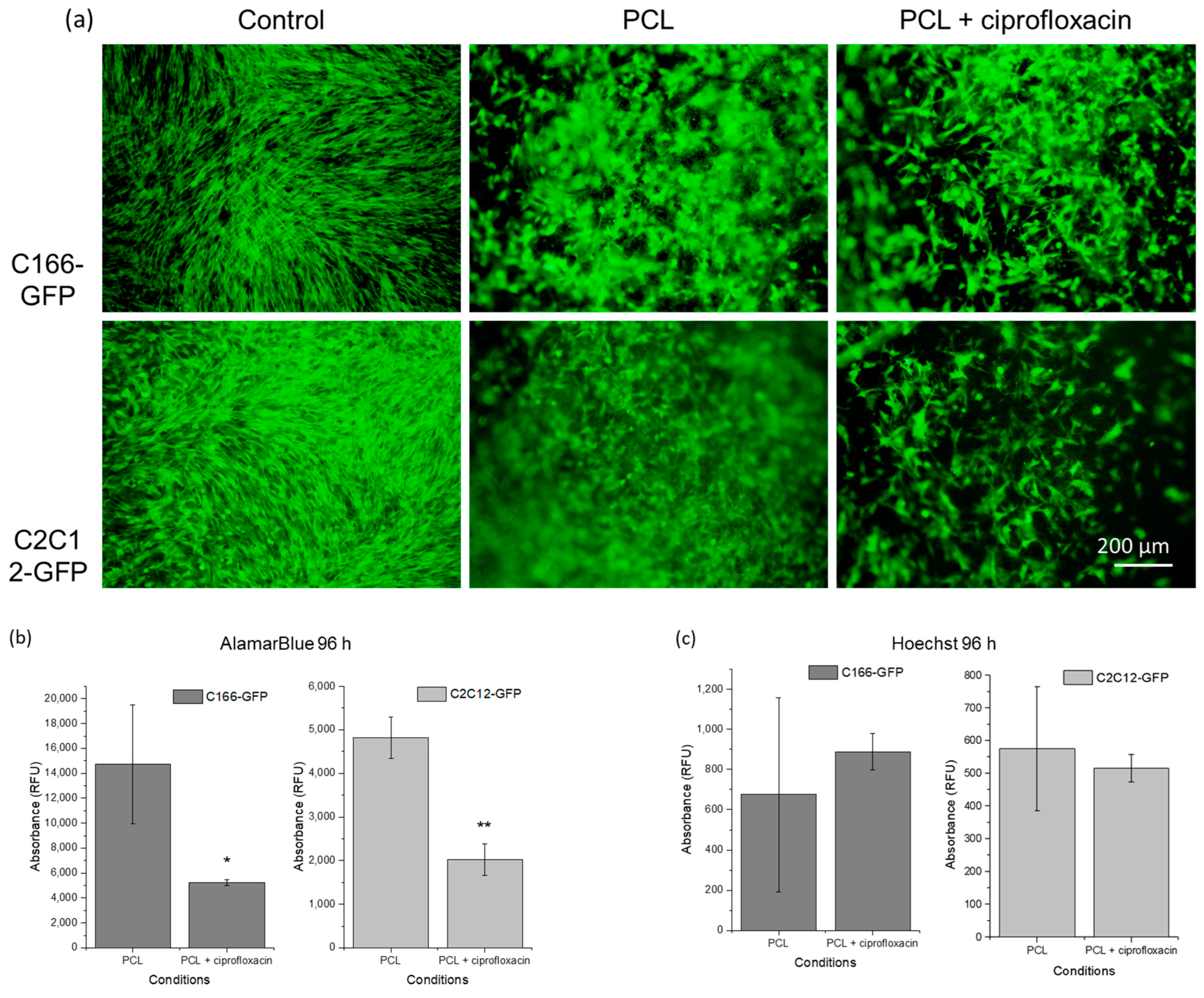
3.4. Cytocompatibility of the Drug-Eluting External Layer with the Designed Topography
3.5. Cytocompatibility of Mg-HHC System Loaded with Paracetamol or Ciprofloxacin
3.6. Coating Performance of the Mg-HHC System in Cell Culture
3.6.1. Cell Response to the Drug-Eluting Hierarchical Hybrid Coating
3.6.2. Degradation of the Mg-HHC during Sample Preparation for Cell Culture
3.6.3. Characterization of the Mg-HHC System after 96 h of Cell Culture
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tsai, D.-S.; Tsai, Y.-C.; Chou, C.-C. Corrosion Passivation of Magnesium Alloy with the Duplex Coatings of Plasma Electrolytic Oxidation and Tetrafluoroethylene-Based Polymers. Surf. Coat. Technol. 2019, 366, 15–23. [Google Scholar] [CrossRef]
- Xu, L.; Liu, X.; Sun, K.; Fu, R.; Wang, G. Corrosion Behavior in Magnesium-Based Alloys for Biomedical Applications. Materials 2022, 15, 2613. [Google Scholar] [CrossRef]
- Globig, P.; Willumeit-Römer, R.; Martini, F.; Mazzoni, E.; Luthringer-Feyerabend, B.J.C. Slow Degrading Mg-Based Materials Induce Tumor Cell Dormancy on an Osteosarcoma-Fibroblast Coculture Model. Bioact. Mater. 2022, 16, 320–333. [Google Scholar] [CrossRef]
- Kozelskaya, A.I.; Rutkowski, S.; Frueh, J.; Gogolev, A.S.; Chistyakov, S.G.; Gnedenkov, S.V.; Sinebryukhov, S.L.; Frueh, A.; Egorkin, V.S.; Choynzonov, E.L.; et al. Surface Modification of Additively Fabricated Titanium-Based Implants by Means of Bioactive Micro-Arc Oxidation Coatings for Bone Replacement. J. Funct. Biomater. 2022, 13, 285. [Google Scholar] [CrossRef] [PubMed]
- Sedelnikova, M.B.; Kashin, A.D.; Luginin, N.A.; Prosolov, K.A.; Tolkacheva, T.V.; Tolmachev, A.I.; Khimich, M.A.; Sharkeev, Y.P. Micro-Arc Sr-Calcium Phosphate Coatings on Magnesium Implants: Morphology, Structure and Corrosive Behavior. Russ. Phys. J. 2023, 66, 740–748. [Google Scholar] [CrossRef]
- Butt, M.S.; Bai, J.; Wan, X.; Chu, C.; Xue, F.; Ding, H.; Zhou, G. Mechanical and Degradation Properties of Biodegradable Mg Strengthened Poly-Lactic Acid Composite through Plastic Injection Molding. Mater. Sci. Eng. C 2017, 70, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Butt, M.S.; Maqbool, A.; Umer, M.A.; Saleem, M.; Malik, R.A.; Alarifi, I.M.; Alrobei, H. Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites. Crystals 2020, 10, 381. [Google Scholar] [CrossRef]
- Zeng, R.-C.; Cui, L.; Jiang, K.; Liu, R.; Zhao, B.-D.; Zheng, Y.-F. In Vitro Corrosion and Cytocompatibility of a Microarc Oxidation Coating and Poly(L-Lactic Acid) Composite Coating on Mg-1Li-1Ca Alloy for Orthopedic Implants. ACS Appl. Mater. Interfaces 2016, 8, 10014–10028. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Ebrahimi-Kahrizsangi, R.; Daroonparvar, M.; Medraj, M. Fabrication and Characterization of Hydrophobic Microarc Oxidation/Poly-Lactic Acid Duplex Coating on Biodegradable Mg–Ca Alloy for Corrosion Protection. Vacuum 2016, 125, 185–188. [Google Scholar] [CrossRef]
- Gao, J.H.; Shi, X.Y.; Yang, B.; Hou, S.S.; Meng, E.C.; Guan, F.X.; Guan, S.K. Fabrication and Characterization of Bioactive Composite Coatings on Mg–Zn–Ca Alloy by MAO/Sol–Gel. J. Mater. Sci. Mater. Med. 2011, 22, 1681–1687. [Google Scholar] [CrossRef]
- Calejo, M.T.; Ilmarinen, T.; Skottman, H.; Kellomäki, M. Breath Figures in Tissue Engineering and Drug Delivery: State-of-the-Art and Future Perspectives. Acta Biomater. 2018, 66, 44–66. [Google Scholar] [CrossRef]
- Yabu, H. Fabrication of Honeycomb Films by the Breath Figure Technique and Their Applications. Sci. Technol. Adv. Mater. 2018, 19, 802–822. [Google Scholar] [CrossRef]
- Santos-Coquillat, A.; Martínez-Campos, E.; Vargas-Alfredo, N.; Arrabal, R.; Rodríguez-Hernández, J.; Matykina, E. Hierarchical Functionalized Polymeric-Ceramic Coatings on Mg-Ca Alloys for Biodegradable Implant Applications. Macromol. Biosci. 2019, 19, 1900179. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Tang, H.; Liu, L.; He, Q.; Zhao, L.; Huang, Z.; Yang, J.; Cao, C.; Chen, J.; Wang, A. Biomimetic Titanium Implant Coated with Extracellular Matrix Enhances and Accelerates Osteogenesis. Nanomedicine 2020, 15, 1779–1793. [Google Scholar] [CrossRef]
- Shen, X.; Zhang, F.; Li, K.; Qin, C.; Ma, P.; Dai, L.; Cai, K. Cecropin B Loaded TiO2 Nanotubes Coated with Hyaluronidase Sensitive Multilayers for Reducing Bacterial Adhesion. Mater. Des. 2016, 92, 1007–1017. [Google Scholar] [CrossRef]
- Jia, H.; Kerr, L.L. Sustained Ibuprofen Release Using Composite Poly(Lactic-Co-Glycolic Acid)/Titanium Dioxide Nanotubes from Ti Implant Surface. J. Pharm. Sci. 2013, 102, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xie, C.; Xiao, X. Polydopamine Modified TiO2 Nanotube Arrays as a Local Drug Delivery System for Ibuprofen. J. Drug Deliv. Sci. Technol. 2020, 56, 101537. [Google Scholar] [CrossRef]
- Lee, J.-H.; Moon, S.-K.; Kim, K.-M.; Kim, K.-N. Modification of TiO2 Nanotube Surfaces by Electro-Spray Deposition of Amoxicillin Combined with PLGA for Bactericidal Effects at Surgical Implantation Sites. Acta Odontol. Scand. 2013, 71, 168–174. [Google Scholar] [CrossRef]
- Ji, X.-J.; Gao, L.; Liu, J.-C.; Wang, J.; Cheng, Q.; Li, J.-P.; Li, S.-Q.; Zhi, K.-Q.; Zeng, R.-C.; Wang, Z.-L. Corrosion Resistance and Antibacterial Properties of Hydroxyapatite Coating Induced by Gentamicin-Loaded Polymeric Multilayers on Magnesium Alloys. Colloids Surf. B Biointerfaces 2019, 179, 429–436. [Google Scholar] [CrossRef]
- Xu, X.; Lu, P.; Guo, M.; Fang, M. Cross-Linked Gelatin/Nanoparticles Composite Coating on Micro-Arc Oxidation Film for Corrosion and Drug Release. Appl. Surf. Sci. 2010, 256, 2367–2371. [Google Scholar] [CrossRef]
- Lu, P.; Fan, H.; Liu, Y.; Cao, L.; Wu, X.; Xu, X. Controllable Biodegradability, Drug Release Behavior and Hemocompatibility of PTX-Eluting Magnesium Stents. Colloids Surf. B Biointerfaces 2011, 83, 23–28. [Google Scholar] [CrossRef]
- Lu, P.; Liu, Y.; Guo, M.; Fang, H.; Xu, X. Corrosion and Drug Release Properties of EN-Plating/PLGA Composite Coating on MAO Film. Mater. Sci. Eng. C 2011, 31, 1285–1289. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, Y.; Li, R.; Li, J.; Lu, X.; Zhang, Y. The Efficacy and Safety Comparison of First-Line Chemotherapeutic Agents (High-Dose Methotrexate, Doxorubicin, Cisplatin, and Ifosfamide) for Osteosarcoma: A Network Meta-Analysis. J. Orthop. Surg. Res. 2020, 15, 51. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, Y.; Qiang, L.; Ren, Y.; Lin, Y.; Li, H.; Chen, Q.; Gao, S.; Yang, X.; Zhang, C.; et al. Multifunctional 3D-Printed Bioceramic Scaffolds: Recent Strategies for Osteosarcoma Treatment. J. Tissue Eng. 2023, 14, 20417314231170371. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liao, R.; Sheng, X.; Luo, X.; Zhang, X.; Wen, X.; Zhou, J.; Peng, K. Hydrogen Gas in Cancer Treatment. Front. Oncol. 2019, 9, 696. [Google Scholar] [CrossRef]
- Cheng, X.; Wei, J.; Ge, Q.; Xing, D.; Zhou, X.; Qian, Y.; Jiang, G. The Optimized Drug Delivery Systems of Treating Cancer Bone Metastatic Osteolysis with Nanomaterials. Drug Deliv. 2021, 28, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Bordbar-Khiabani, A.; Yarmand, B.; Sharifi-Asl, S.; Mozafari, M. Improved Corrosion Performance of Biodegradable Magnesium in Simulated Inflammatory Condition via Drug-Loaded Plasma Electrolytic Oxidation Coatings. Mater. Chem. Phys. 2020, 239, 122003. [Google Scholar] [CrossRef]
- Moreno Turiégano, L. Surface Functionalisation of Magnesium Alloys for Biodegradable Implants; Universidad Complutense de Madrid: Madrid, Spain, 2022. [Google Scholar]
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012, 2012, 195727. [Google Scholar] [CrossRef]
- Ponnusamy, T.; Lawson, L.B.; Freytag, L.C.; Blake, D.A.; Ayyala, R.S.; John, V.T. In Vitro Degradation and Release Characteristics of Spin Coated Thin Films of PLGA with a “Breath Figure” Morphology. Biomatter 2012, 2, 77–86. [Google Scholar] [CrossRef]
- Moreno, L.; Mohedano, M.; Arrabal, R.; Rodríguez-Hernández, J.; Matykina, E. Development of Hybrid Hierarchical Coatings on Mg3Zn0.4Ca Alloy for Orthopaedic Implants. J. Mater. Res. Technol. 2023, 24, 5823–5838. [Google Scholar] [CrossRef]
- Moreno, L.; Wang, C.; Lamaka, S.V.; Zheludkevich, M.L.; Rodríguez-Hernández, J.; Arrabal, R.; Matykina, E. Ciprofloxacin Release and Corrosion Behaviour of a Hybrid PEO/PCL Coating on Mg3Zn0.4Ca Alloy. J. Funct. Biomater. 2023, 14, 65. [Google Scholar] [CrossRef]
- Martínez-Campos, E.; Elzein, T.; Bejjani, A.; García-Granda, M.J.; Santos-Coquillat, A.; Ramos, V.; Muñoz-Bonilla, A.; Rodríguez-Hernández, J. Toward Cell Selective Surfaces: Cell Adhesion and Proliferation on Breath Figures with Antifouling Surface Chemistry. ACS Appl. Mater. Interfaces 2016, 8, 6344–6353. [Google Scholar] [CrossRef]
- Bridle, M.J.; Janesko, B.G. Computational Study of Fluoroquinolone Binding to and Its Applicability to Future Drug Design. Int. J. Quantum Chem. 2017, 117, e25428. [Google Scholar] [CrossRef]
- Chandrathilaka, A.; Ileperuma, O.; Hettiarachchi, C. Spectrophotometric and pH-Metric Studies on Pb(II), Cd(II), Al(III) and Cu(II) Complexes of Paracetamol and Ascorbic Acid. J. Natl. Sci. Found. Sri Lanka 2013, 41, 337–344. [Google Scholar] [CrossRef]
- Sultan, M.A.; Karim, A.; Kandory, A.; Al-metwali, A. Synthesis and Characterization of Al(III) Complex with Paracetamol. Int. J. Drug Deliv. Technol. 2020, 10, 156–159. [Google Scholar] [CrossRef]
- Sasireka, A.; Rajendran, R.; Priya, P.; Raj, V. Ciprofloxacin-Loaded Ceramic/Polymer Composite Coatings on Ti with Improved Antibacterial and Corrosion Resistance Properties for Orthopedic Applications. ChemistrySelect 2019, 4, 1166–1175. [Google Scholar] [CrossRef]
- Walden, D.M.; Khotimchenko, M.; Hou, H.; Chakravarty, K.; Varshney, J. Effects of Magnesium, Calcium, and Aluminum Chelation on Fluoroquinolone Absorption Rate and Bioavailability: A Computational Study. Pharmaceutics 2021, 13, 594. [Google Scholar] [CrossRef]
- Refat, M.S.; Mohamed, G.G.; El-Sayed, M.Y.; Killa, H.M.A.; Fetooh, H. Spectroscopic and Thermal Degradation Behavior of Mg(II), Ca(II), Ba(II) and Sr(II) Complexes with Paracetamol Drug. Arab. J. Chem. 2017, 10, S2376–S2387. [Google Scholar] [CrossRef]
- Fekry, A.M.; Fatayerji, M.Z. Electrochemical Corrosion Behavior of AZ91D Alloy in Ethylene Glycol. Electrochim. Acta 2009, 54, 6522–6528. [Google Scholar] [CrossRef]
- Pouzaud, F.; Bernard-Beaubois, K.; Thevenin, M.; Warnet, J.-M.; Hayem, G.; Rat, P. In Vitro Discrimination of Fluoroquinolones Toxicity on Tendon Cells: Involvement of Oxidative Stress. J. Pharmacol. Exp. Ther. 2004, 308, 394–402. [Google Scholar] [CrossRef]
- Jeong, H.; Rho, J.; Shin, J.-Y.; Lee, D.Y.; Hwang, T.; Kim, K.J. Mechanical Properties and Cytotoxicity of PLA/PCL Films. Biomed. Eng. Lett. 2018, 8, 267–272. [Google Scholar] [CrossRef]
- Moreno, L.; Mohedano, M.; Arrabal, R.; Matykina, E. Development and Screening of (Ca-P-Si-F)-PEO Coatings for Biodegradability Control of Mg-Zn-Ca Alloys. J. Magnes. Alloys 2022, 10, 2220–2237. [Google Scholar] [CrossRef]
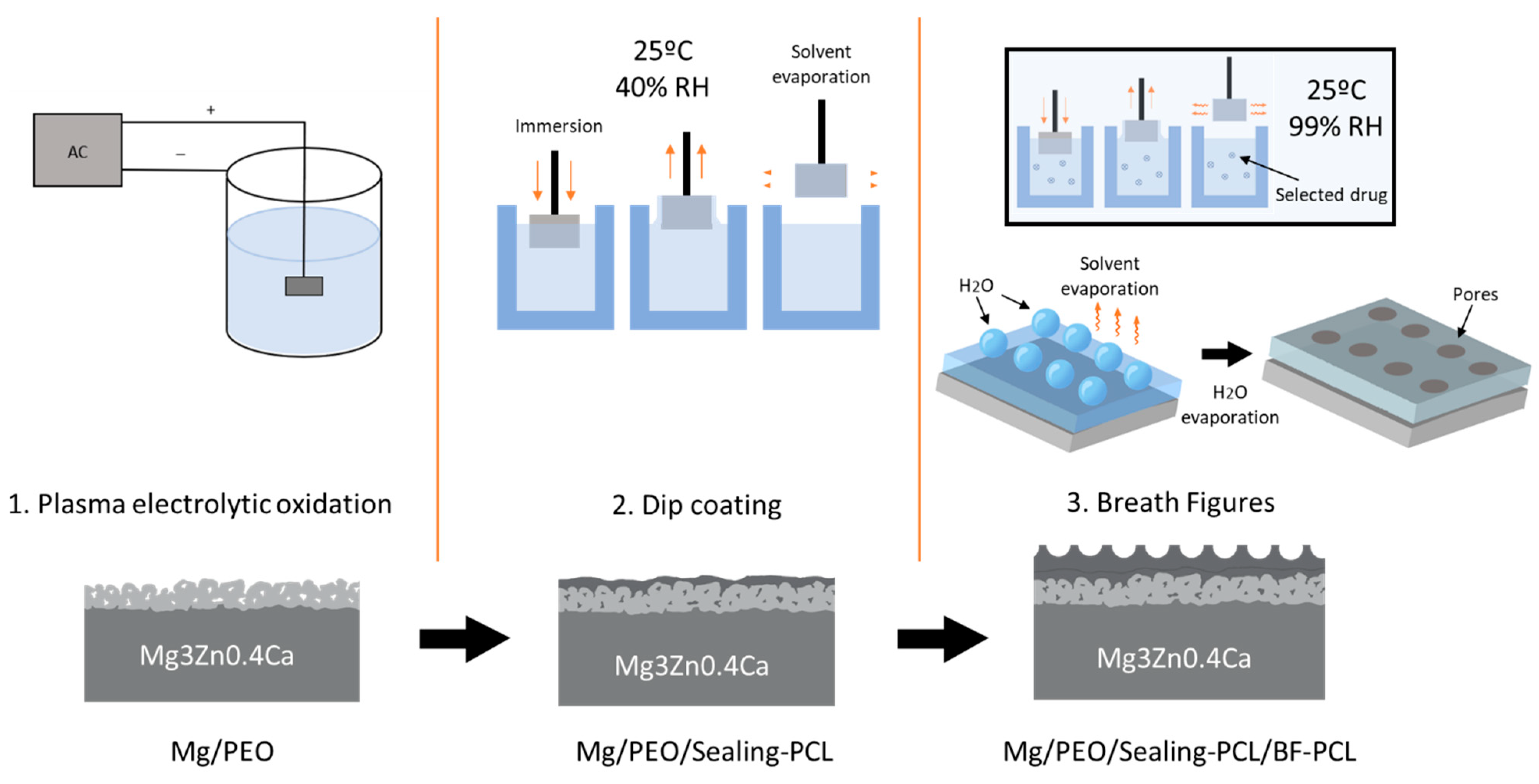







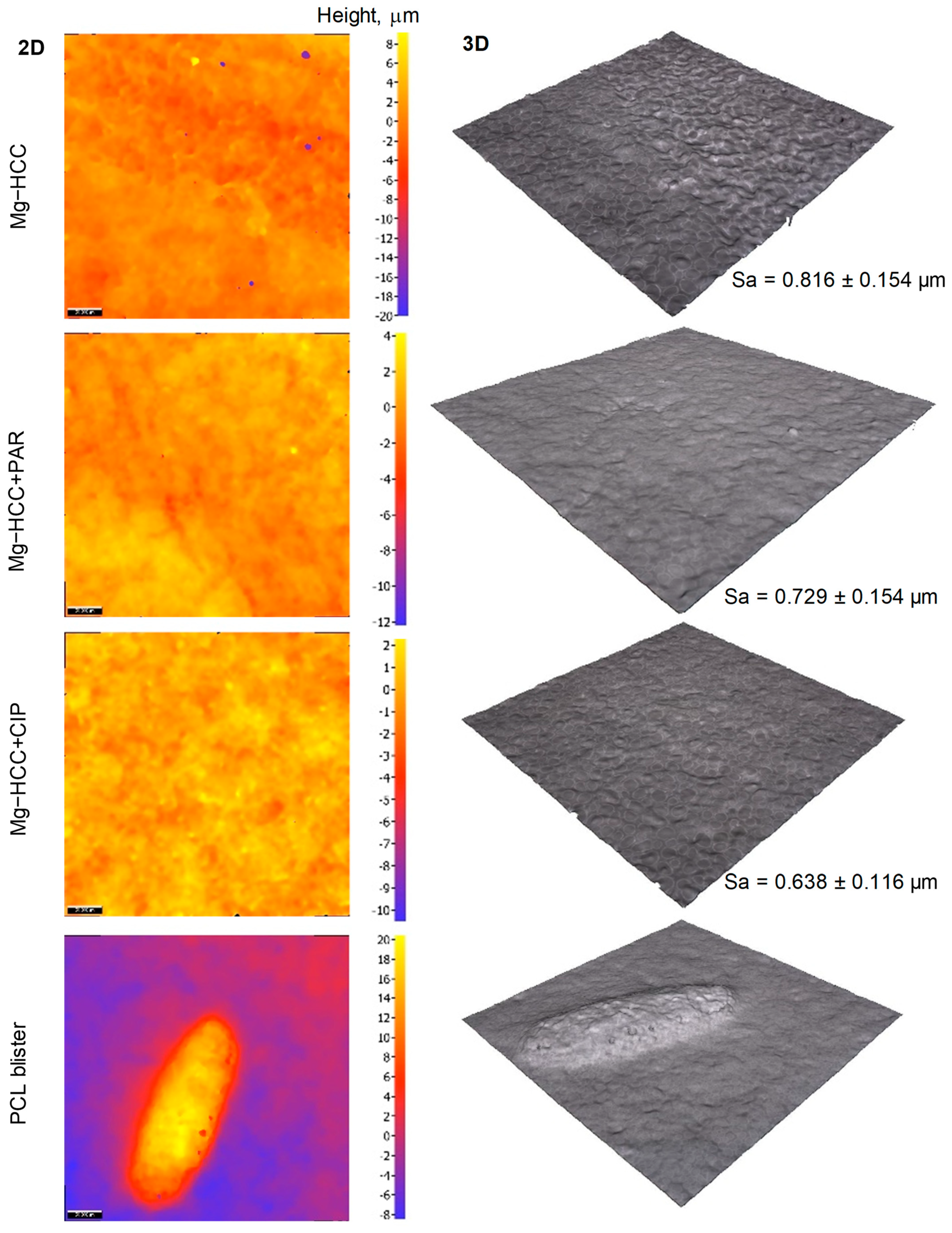
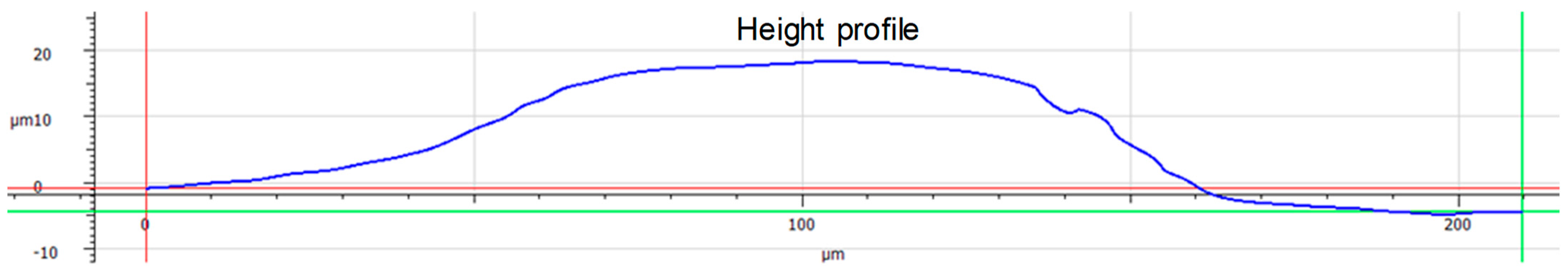
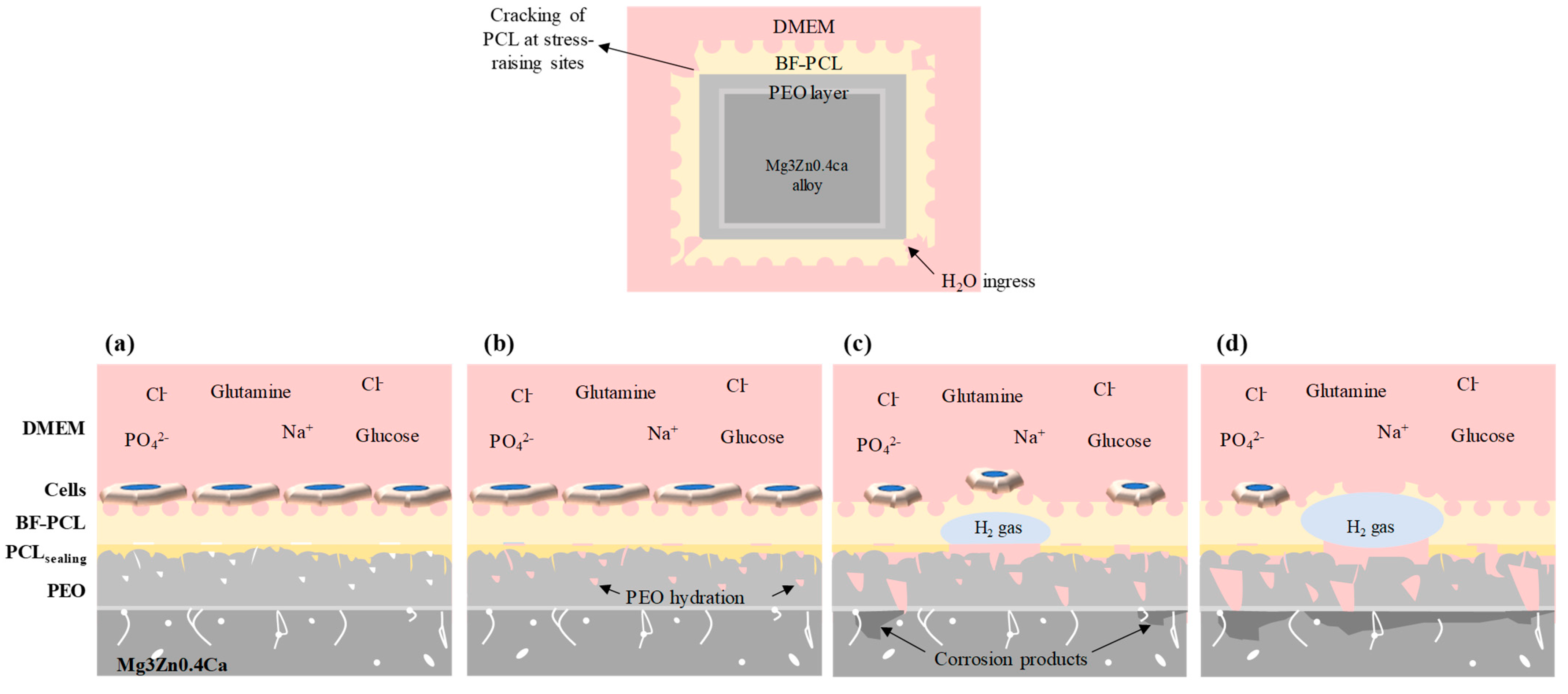
| Coating Layer | Withdrawal Speed | No. of Immersion Cycles |
|---|---|---|
| Sealing PCL | 0.3 mm/s | 1 |
| BF PCL | 2 mm/s | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolao-Gómez, A.; Martínez-Campos, E.; Moreno, L.; Rodríguez-Hernández, J.; Matykina, E. Hierarchical Hybrid Coatings with Drug-Eluting Capacity for Mg Alloy Biomaterials. Materials 2023, 16, 7688. https://doi.org/10.3390/ma16247688
Nicolao-Gómez A, Martínez-Campos E, Moreno L, Rodríguez-Hernández J, Matykina E. Hierarchical Hybrid Coatings with Drug-Eluting Capacity for Mg Alloy Biomaterials. Materials. 2023; 16(24):7688. https://doi.org/10.3390/ma16247688
Chicago/Turabian StyleNicolao-Gómez, Ana, Enrique Martínez-Campos, Lara Moreno, Juan Rodríguez-Hernández, and Endzhe Matykina. 2023. "Hierarchical Hybrid Coatings with Drug-Eluting Capacity for Mg Alloy Biomaterials" Materials 16, no. 24: 7688. https://doi.org/10.3390/ma16247688
APA StyleNicolao-Gómez, A., Martínez-Campos, E., Moreno, L., Rodríguez-Hernández, J., & Matykina, E. (2023). Hierarchical Hybrid Coatings with Drug-Eluting Capacity for Mg Alloy Biomaterials. Materials, 16(24), 7688. https://doi.org/10.3390/ma16247688








