Review on New-Generation Batteries Technologies: Trends and Future Directions
Abstract
:1. Introduction
2. New Generation Batteries
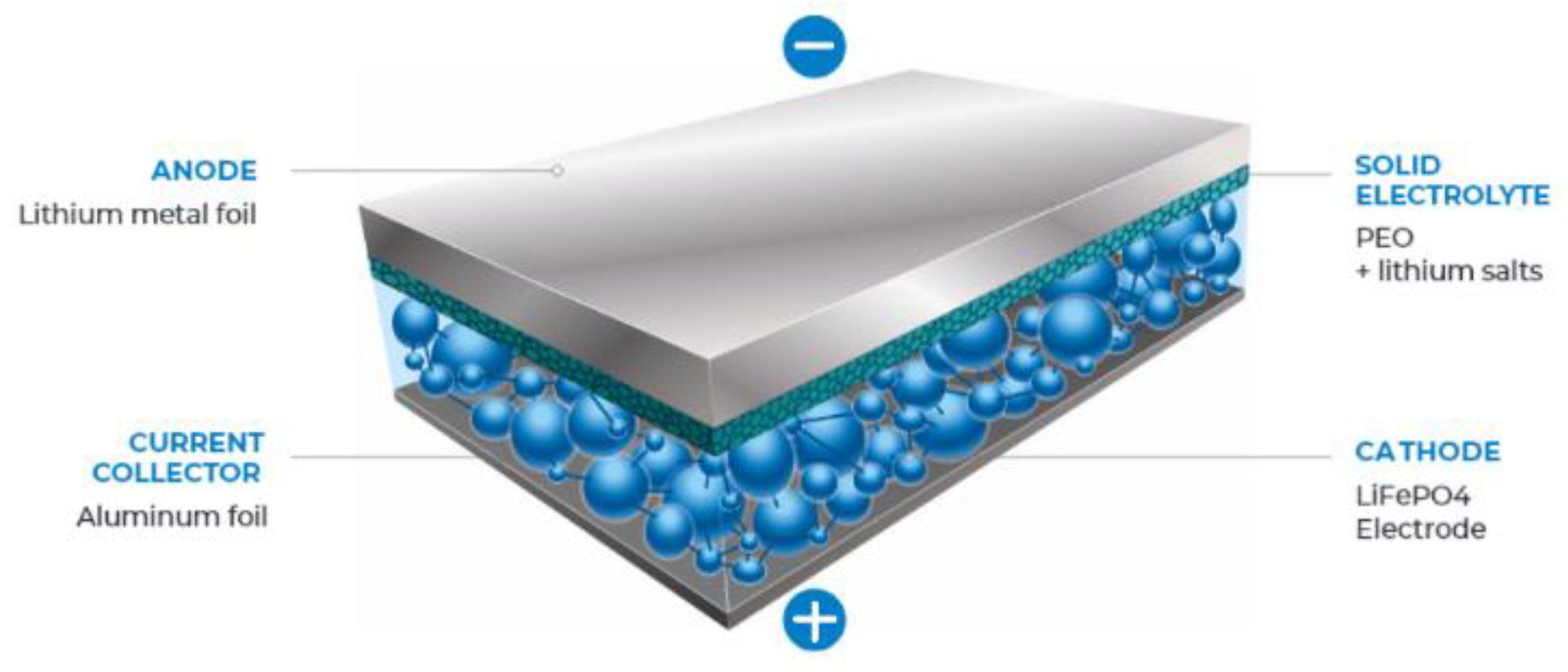
2.1. Components of a Lithium Battery
2.1.1. Positive Electrode (Cathode)
2.1.2. Negative Electrode (Anode)
2.1.3. Electrolytes
2.1.4. Separator
2.1.5. Current Collectors
2.1.6. Anode and Cathode Coatings
2.2. Before the Lithium
- lead-acid based: affordable, safe, and sustainable;
- lithium-based: high energy density, low weight;
- nickel-based: long life, reliable (NiMH, NiCd);
- sodium-based: relative low cost; and
- flow batteries [73].
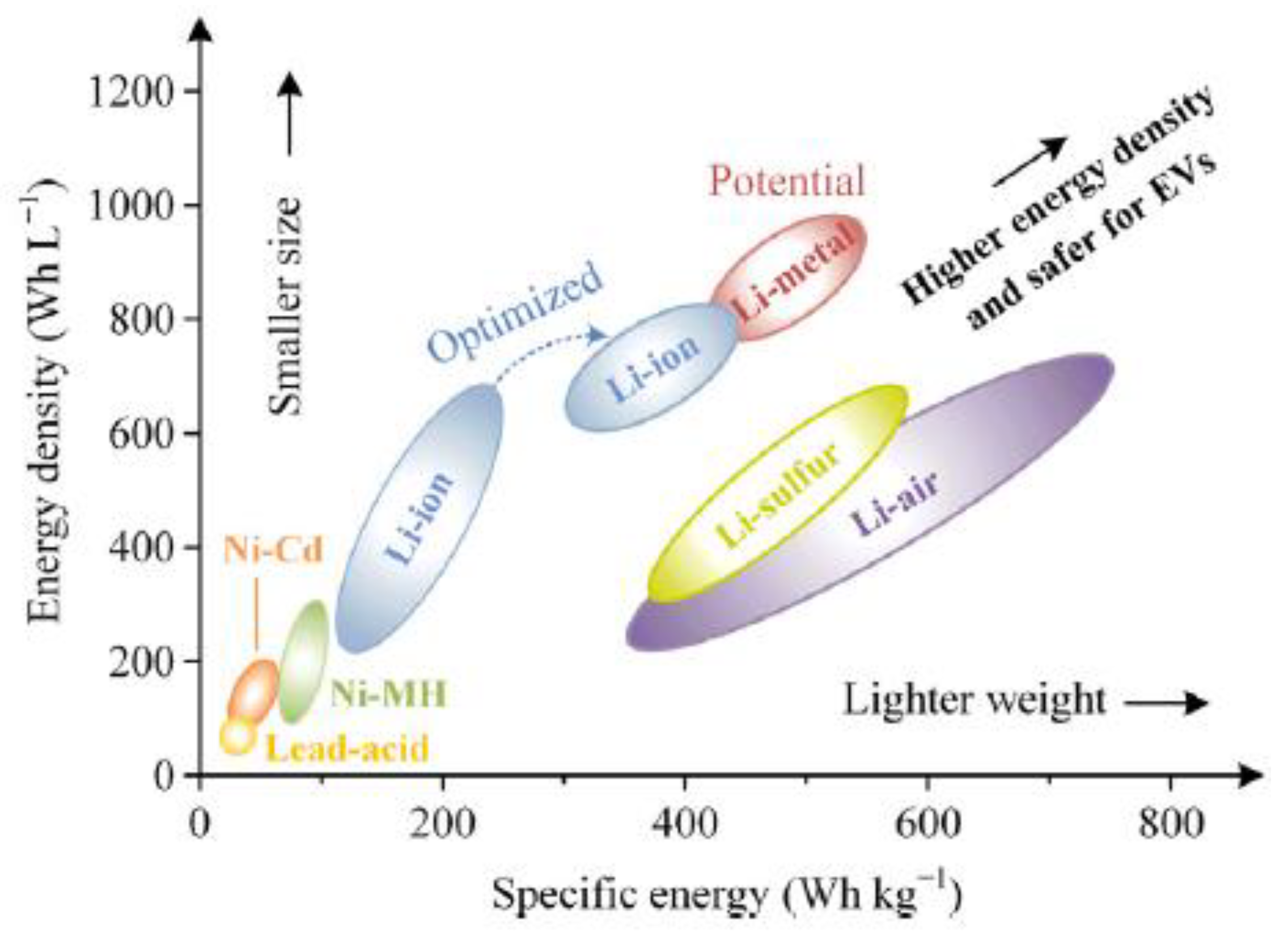
2.3. The Lithium-Ion Battery
2.4. Current and Future Promising Technologies
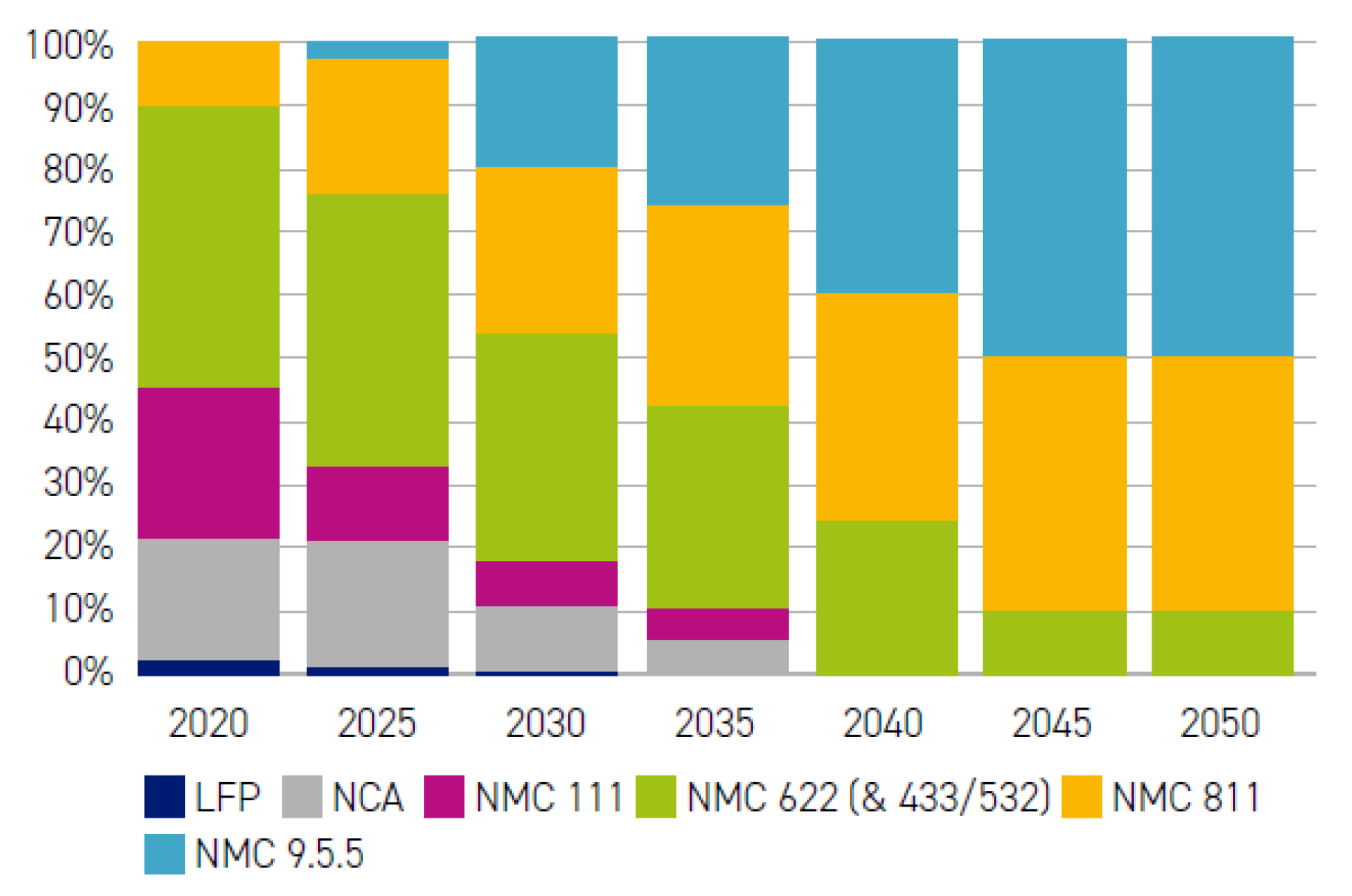
2.4.1. Generation 3
2.4.2. Generation 4
2.4.3. Generation 5
- Metal-Air Battery
- b.
- Lithium Sulfur Battery
- c.
- Batteries beyond Lithium
- d.
- Solid-State Li-Ion Micro-Batteries
3. Analyses and Discussions on Future Batteries Challenges
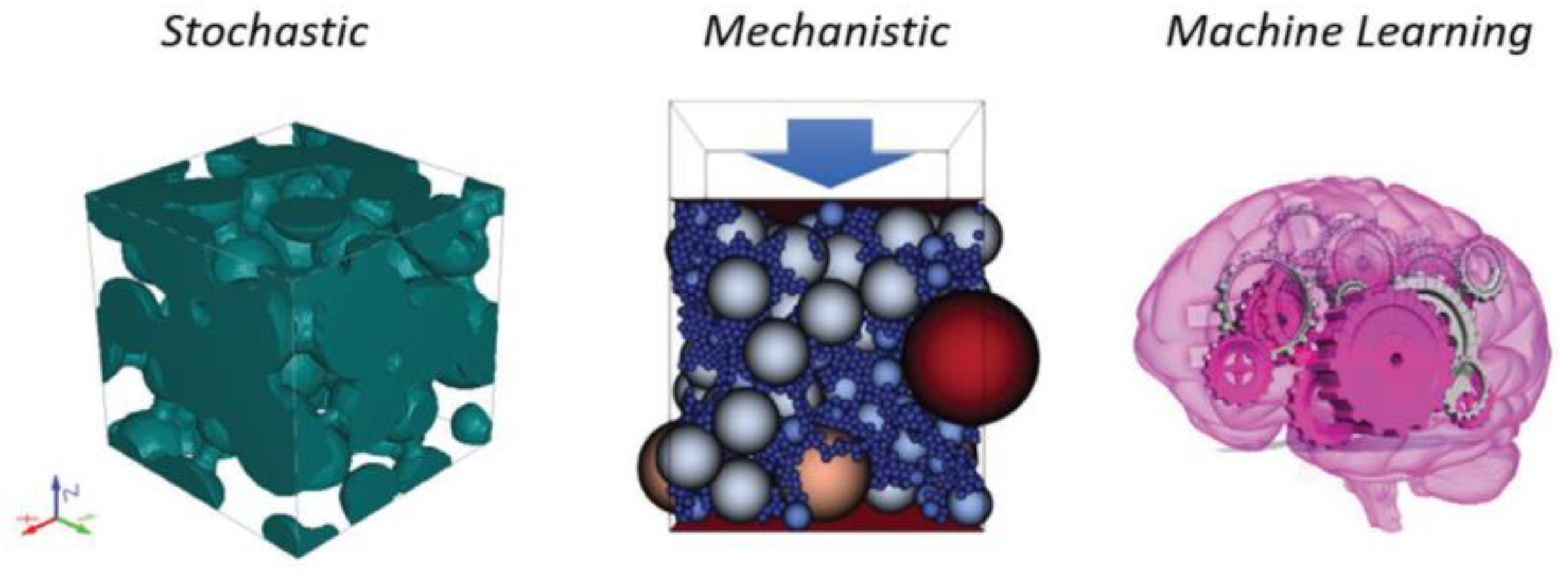
3.1. Modeling and Components
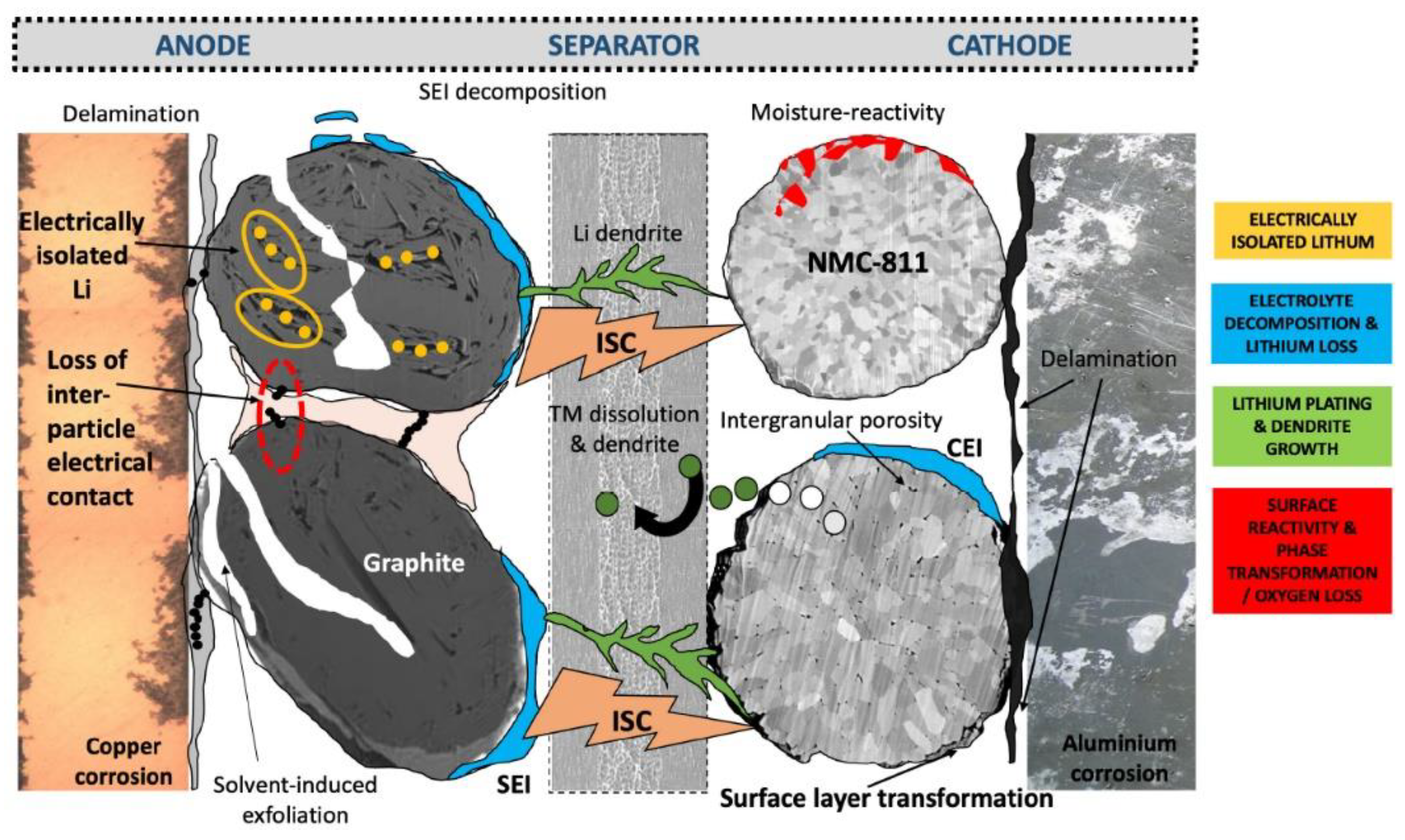
3.2. Battery Degradation
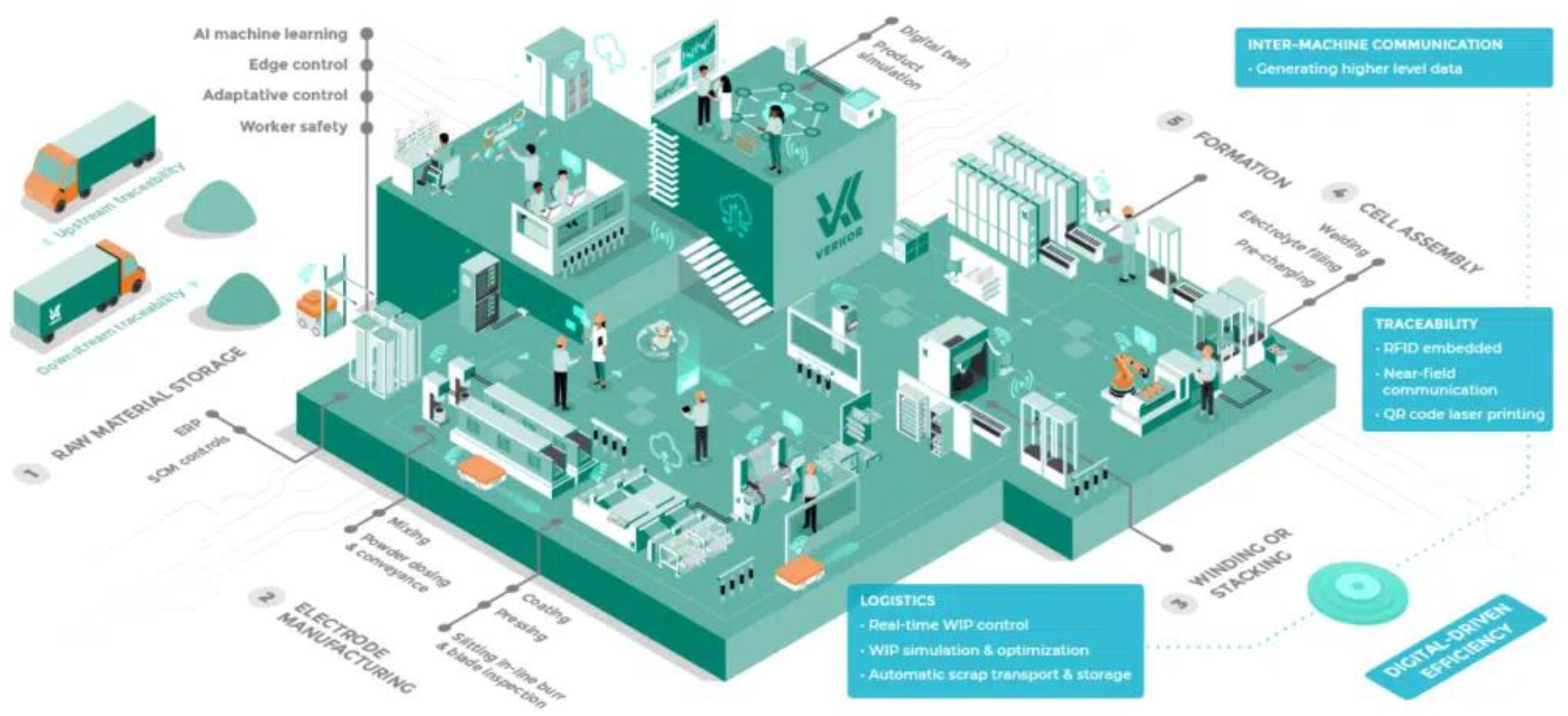
3.3. Lithium-Ion Battery Manufacturing
3.4. Self-Healing
3.5. Battery Passport
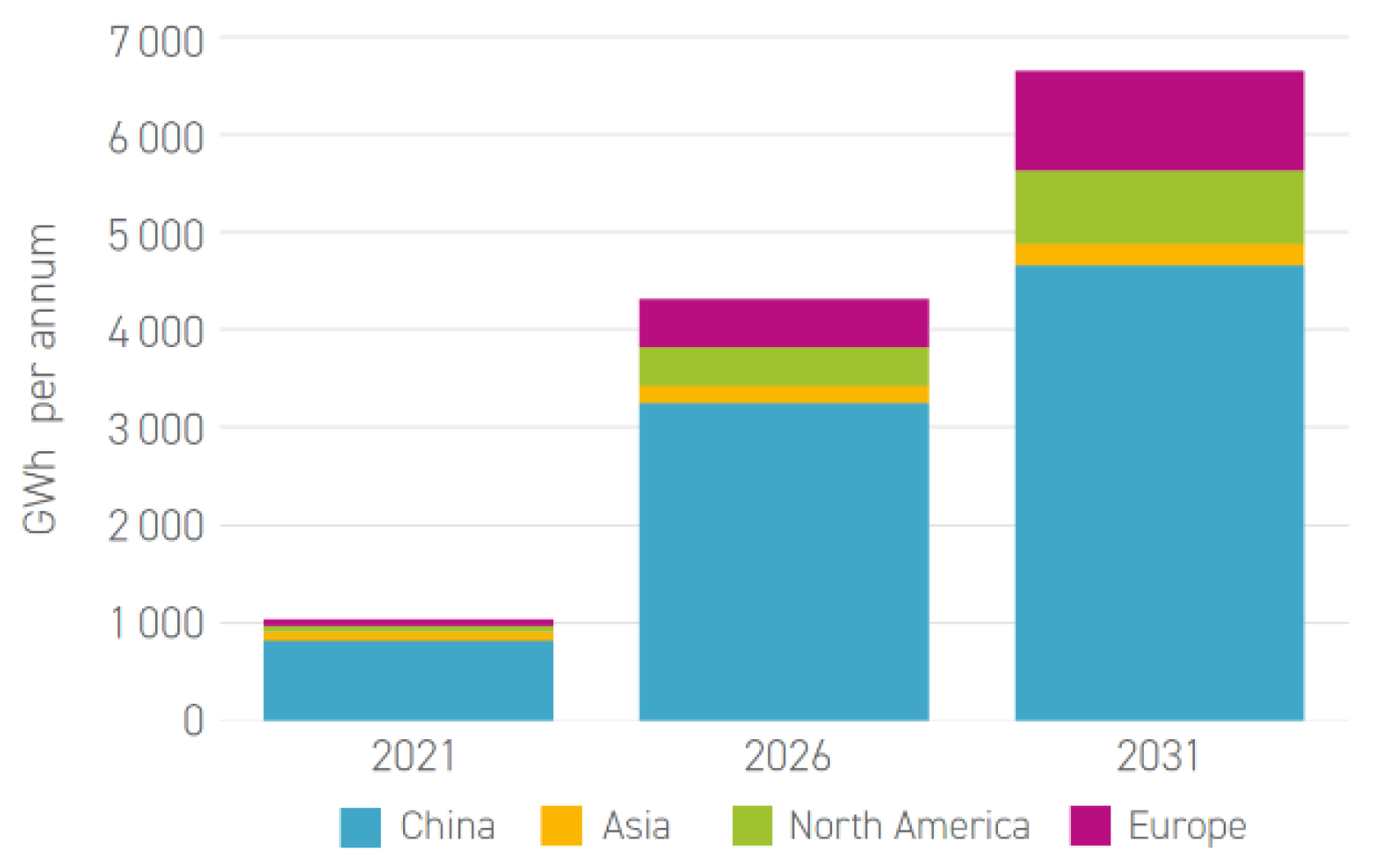
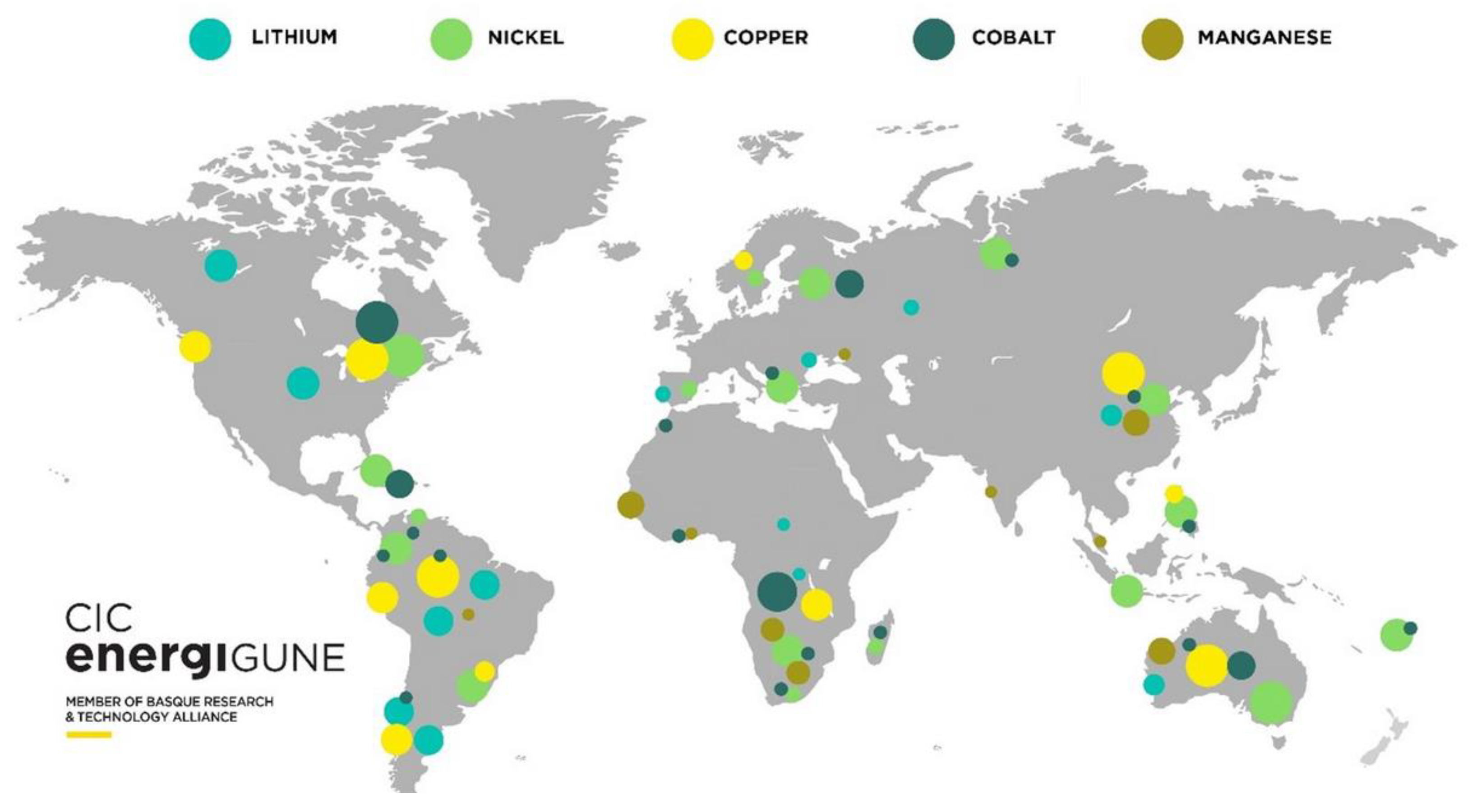
3.6. Mining of Critical Materials for Battery Applications
3.7. Second Life—Remaining Useful Life—Recycling
4. Conclusions
- -
- Technological limitations: The review is influenced by the current technological landscape in battery materials, including constraints related to the reliability, efficiency, and autonomy of battery systems.
- -
- Societal and policy limitations: Societal acceptability, economic factors, and political decisions, such as the governmental decisions to implement gigafactories, play a significant role in shaping the future of batteries and their adoption.
- -
- Cost, material, and environmental limitations: The study is also bounded by the limitations associated with the cost of materials, considerations related to the circular economy, and the environmental footprint of battery technologies.
Author Contributions
Funding
Conflicts of Interest
References
- Schleussner, C.; Rogelj, J.; Schaeffer, M.; Lissner, T.; Licker, R.; Fischer, E.; Knutti, R.; Levermann, A.; Frieler, K.; Hare, W. Science and policy characteristics of the Paris Agreement temperature goal. Nat. Clim. Chang. 2016, 6, 827–835. [Google Scholar] [CrossRef]
- Sikora, A. European Green Deal—Legal and financial challenges of the climate change. In Era Forum; Springer: Berlin/Heidelberg, Germany, 2021. [Google Scholar]
- Mathieu, C. The European battery alliance is moving up a gear. The European Battery Alliance is Moving up a Gear; Edito de l’Ifri: Paris, France, 2019. [Google Scholar]
- Shale-Hester, T. UK 2030 Petrol and Diesel Ban: What Is It and Which Cars Are Affected? Available online: https://www.autoexpress.co.uk/news/108960/uk-2030-petrol-and-diesel-ban-what-it-and-which-cars-are-affected (accessed on 8 June 2023).
- Register, F. Tackling the Climate Crisis at Home and Abroad. Presidential Documents, United States of America, Federal Register/Vol. 86, No. 19, Monday. 1 February 2021. Available online: https://downloads.regulations.gov/EPA-HQ-OPPT-2021-0202-0012/content.pdf (accessed on 1 July 2023).
- BloombergNEF; BloombergPhilanthropies. Energy Transition Factbook; Clean Energy Ministerial: Paris, France, 2021. [Google Scholar]
- Granholm, J. National Blueprint for Lithium Batteries 2021–2030; U.S. Department of Energy: Washington, DC, USA, 2021. [Google Scholar]
- Van Mierlo, J.; Messagie, M.; Rangaraju, S. Comparative environmental assessment of alternative fueled vehicles using a life cycle assessment. Transp. Res. Procedia 2017, 25, 3435–3445. [Google Scholar] [CrossRef]
- Field, K. BloombergNEF: Lithium-Ion Battery Cell Densities Have Almost Tripled Since 2010. 19 February 2020. Available online: https://cleantechnica.com/2020/02/19/bloombergnef-lithium-ion-battery-cell-densities-have-almost-tripled-since-2010/ (accessed on 1 June 2023).
- Vehicle Technologies Office. Volumetric Energy Density of Lithium-Ion Batteries Increased by More than Eight Times between 2008 and 2020. 18 April 2022. Available online: https://www.energy.gov/eere/vehicles/articles/fotw-1234-april-18-2022-volumetric-energy-density-lithium-ion-batteries (accessed on 1 July 2023).
- Cao, W.; Zhang, J.; Li, H. Batteries with high theoretical energy densities. Energy Storage Mater. 2020, 26, 46–55. [Google Scholar] [CrossRef]
- Gordon, D. Battery Market Forecast to 2030: Pricing, Capacity, and Supply and Demand. 15 March 2022. Available online: https://www.esource.com/report/130221hvfd/battery-market-forecast-2030-pricing-capacity-and-supply-and-demand (accessed on 1 June 2023).
- Yue, Q.; He, C.; Wu, M.; Zhao, T. Advances in thermal management systems for next-generation power batteries. Int. J. Heat Mass Transf. 2021, 181, 121853. [Google Scholar] [CrossRef]
- Liu, P.; Chen, T.; Yang, H. A Li-Ion Battery Charger With Variable Charging Current and Automatic Voltage-Compensation Controls for Parallel Charging. IEEE J. Emerg. Sel. Top. Power Electron. 2021, 10, 997–1006. [Google Scholar] [CrossRef]
- Collin, R.; Miao, Y.; Yokochi, A.; Enjeti, P.; Von Jouanne, A. Advanced electric vehicle fast-charging technologies. Energies 2019, 12, 1839. [Google Scholar] [CrossRef]
- Afonso, J.; Cardoso, L.; Pedrosa, D.; Sousa, T.; Machado, L.; Tanta, M.; Monteiro, V. A review on power electronics technologies for electric mobility. Energies 2020, 13, 6343. [Google Scholar] [CrossRef]
- Amry, Y.; Elbouchikhi, E.; Le Gall, F.; Ghogho, M.; El Hani, S. Electric Vehicle Traction Drives and Charging Station Power Electronics: Current Status and Challenges. Energies 2022, 15, 6037. [Google Scholar] [CrossRef]
- Habib, S.; Ehsan, F.; Liu, H.; Nadeem, M.; Abbas, F.; Numan, M. A Comprehensive Topological Assessment of Power Electronics Converters for Charging of Electric Vehicles. In Flexible Resources for Smart Cities; Springer: Cham, Switzerland, 2021; pp. 133–183. [Google Scholar]
- Ronanki, D.; Kelkar, A.; Williamson, S. Extreme fast charging technology—Prospects to enhance sustainable electric transportation. Energies 2019, 12, 3721. [Google Scholar] [CrossRef]
- Al-Saadi, M.; Olmos, J.; Saez-de-Ibarra, A.; Van Mierlo, J.; Berecibar, M. Fast Charging Impact on the Lithium-Ion Batteries’ Lifetime and Cost-Effective Battery Sizing in Heavy-Duty Electric Vehicles Applications. Energies 2022, 15, 1278. [Google Scholar] [CrossRef]
- Komsiyska, L.; Buchberger, T.; Diehl, S.; Ehrensberger, M.; Hanzl, C.; Hartmann, C.; Hölzle, M.; Kleiner, J.; Lewerenz, M.; Liebhart, B.; et al. Critical review of intelligent battery systems: Challenges, implementation, and potential for electric vehicles. Energies 2021, 14, 5989. [Google Scholar] [CrossRef]
- Polater, N.; Tricoli, P. Technical Review of Traction Drive Systems for Light Railways. Energies 2022, 15, 3187. [Google Scholar] [CrossRef]
- Abdelaal, A.; Mukhopadhyay, S.; Rehman, H. Battery Energy Management Techniques for an Electric Vehicle Traction System. IEEE Access 2021, 10, 84015–84037. [Google Scholar] [CrossRef]
- Mahamud, R.; Park, C. Theory and Practices of Li-Ion Battery Thermal Management for Electric and Hybrid Electric Vehicles. Energies 2022, 15, 3930. [Google Scholar] [CrossRef]
- Yamamoto, M.; Kakisaka, T.; Imaoka, J. Technical trend of power electronics systems for automotive applications. Jpn. J. Appl. Phys. 2020, 59, SG0805. [Google Scholar] [CrossRef]
- Shi, B.; Ramones, A.; Liu, Y.; Wang, H.; Li, Y.; Pischinger, S.; Andert, J. A review of silicon carbide MOSFETs in electrified vehicles: Application, challenges, and future development. IET Power Electron. 2023. [Google Scholar] [CrossRef]
- AEHR Test System. Aehr Comments on Recent Tesla Statements on Silicon Carbide and Reiterates its Annual Guidance. Press Release. 2 March 2023. Available online: https://www.globenewswire.com/news-release/2023/03/02/2619726/11245/en/Aehr-Comments-on-Recent-Tesla-Statements-on-Silicon-Carbide-and-Reiterates-its-Annual-Guidance.html (accessed on 11 June 2023).
- Maroti, P.; Padmanaban, S.; Bhaskar, M.; Ramachandaramurthy, V.; Blaabjerg, F. The state-of-the-art of power electronics converters configurations in electric vehicle technologies. Power Electron. Devices Compon. 2022, 1, 100001. [Google Scholar] [CrossRef]
- Kumar, S.; Adil, U. A review of converter topologies for battery charging applications in plug-in hybrid electric vehicles. In Proceedings of the 2018 IEEE Industry Applications Society Annual Meeting (IAS), Portland, OR, USA, 23–27 September 2018; pp. 1–9. [Google Scholar]
- Wen, H.; Xiao, W.; Wen, X.; Armstrong, P. Analysis and Evaluation of DC-Link Capacitors for High-Power-Density Electric Vehicle Drive Systems. IEEE Trans. Veh. Technol. 2012, 61, 2950–2964. [Google Scholar]
- Zhang, C.; Srdic, S.; Lukic, S.; Sun, K.; Wang, J.; Burgos, R. A SiC-Based Liquid-Cooled Electric Vehicle Traction Inverter Operating at High Ambient Temperature. CPSS Trans. Power Electron. Appl. 2022, 7, 160–175. [Google Scholar] [CrossRef]
- Ding, C.; Lu, S.; Zhang, Z.; Zhang, K.; Nguyen, T.; Ngo, K.; Burgos, R.; Lu, G. Double-Side Cooled SiC MOSFET Power Modules with Sintered-Silver Interposers for a 100 kW/L Traction Inverter. IEEE Trans. Power Electron. 2023, 38, 9685–9694. [Google Scholar] [CrossRef]
- Das, S.; Marlino, L.; Armstrong, K. Wide Bandgap Semiconductor Opportunities in Power Electronics; Oak Ridge National Lab.(ORNL): Oak Ridge, TN, USA, 2018. [Google Scholar]
- Liu, W.; Placke, T.; Chau, K. Overview of batteries and battery management for electric vehicles. Energy Rep. 2022, 8, 4058–4084. [Google Scholar] [CrossRef]
- Gifford, S. The Gigafactory Boom: The Demand for Battery Manufacturing in the UK; FARADAY INSIGHTS; The Faraday Institution: Cambridge, UK, 2022. [Google Scholar]
- CIC energiGUNe. World Map of Gigafactories. 21 October 2021. Available online: https://cicenergigune.com/en/blog/world-map-gigafactories (accessed on 7 January 2023).
- Ortiz, S.; Careaga, I. Gigafactories: Europe’s Major Commitment to Economic Recovery through the Development of Battery Factories. 7 April 2021. Available online: https://cicenergigune.com/en/blog/gigafactories-europe-commitment-economic-recovery-battery-factories (accessed on 7 January 2023).
- Itani, K.; De Bernardinis, A. Management of the Energy Storage Hybridization in Electric Vehicles. In Encyclopedia of Electrical and Electronic Power Engineering; Elsevier: Amsterdam, The Netherlands, 2023; pp. 542–562. [Google Scholar]
- Sutcliffe, E. Electric Vehicle Fires on Ships & Ferries—EV Fire Safe. 14 September 2022. Available online: https://www.evfiresafe.com/post/electric-vehicle-fires-on-ships-ferries (accessed on 7 January 2023).
- Booth, S.; Nedoma, A.; Anthonisamy, N.; Baker, P.; Boston, R.; Bronstein, H.; Clarke, S.; Cussen, E.J.; Daramalla, V.; De Volder, M.; et al. Perspectives for next generation lithium-ion battery cathode materials. Appl. Mater. 2021, 9, 109201. [Google Scholar] [CrossRef]
- Gifford, S. Lithium, Cobalt and Nickel: The Gold Rush of the 21st Century. Changes 2022, 2050, 6–500. [Google Scholar]
- Berckmans, G.; Messagie, M.; Smekens, J.; Omar, N.; Vanhaverbeke, L.; Van Mierlo, J. Cost projection of state of the art lithium-ion batteries for electric vehicles up to 2030. Energies 2017, 10, 1314. [Google Scholar] [CrossRef]
- Barbosa, J.; Gonçalves, R.; Costa, C.; Lanceros-Mendez, S. Recent advances on materials for lithium-ion batteries. Energies 2021, 14, 3145. [Google Scholar] [CrossRef]
- Van Mierlo, J.; Berecibar, M.; El Baghdadi, M.; De Cauwer, C.; Messagie, M.; Coosemans, T.; Jacobs, V.; Hegazy, O. Beyond the state of the art of electric vehicles: A fact-based paper of the current and prospective electric vehicle technologies. World Electr. Veh. J. 2021, 12, 20. [Google Scholar] [CrossRef]
- Kim, Y.; Seong, W.; Manthiram, A. Cobalt-free, high-nickel layered oxide cathodes for lithium-ion batteries: Progress, challenges, and perspectives. Energy Storage Mater. 2021, 34, 250–259. [Google Scholar] [CrossRef]
- Lee, S.; Manthiram, A. Can cobalt be eliminated from lithium-ion batteries? ACS Energy Lett. 2022, 7, 3058–3063. [Google Scholar] [CrossRef]
- Lima, P. CATL’s NCM 811 Battery Cells Are Problematic. 28 September 2020. Available online: https://pushevs.com/2020/09/28/catl-ncm-811-battery-cells-are-problematic/ (accessed on 9 June 2023).
- SMM. Tragedy of Electric Car Catching Fire Immediately After Collision in China’s Hangzhou City Renews Concerns Over Safety of Nickel-Manganese-Cobalt (NMC 811) Battery. 7 June 2023. Available online: https://news.metal.com/newscontent/102243830/tragedy-of-electric-car-catching-fire-immediately-after-collision-in-china%E2%80%99s-hangzhou-city-renews-concerns-over-safety-of-nickel-manganese-cobalt-nmc-811-battery (accessed on 9 June 2023).
- Xia, M.; Lin, M.; Liu, G.; Cheng, Y.; Jiao, T.; Fu, A.; Yang, Y.; Wang, M.; Zheng, J. Stable cycling and fast charging of high-voltage lithium metal batteries enabled by functional solvation chemistry. Chem. Eng. J. 2022, 442, 136351. [Google Scholar] [CrossRef]
- Mao, N.; Gadkari, S.; Wang, Z.; Zhang, T.; Bai, J.; Cai, Q. A comparative analysis of lithium-ion batteries with different cathodes under overheating and nail penetration conditions. Energy 2023, 278, 128027. [Google Scholar] [CrossRef]
- Adham, S.; Jackson, M.D.C.; Laugharne, A. Navigating the Rapidly Evolving EV Battery Chemistry Mix. CRU Group. February 2023. Available online: https://www.crugroup.com/knowledge-and-insights/insights/2023/navigating-the-rapidly-evolving-ev-battery-chemistry-mix/ (accessed on 22 October 2023).
- Adham, S. The Next-Generation Cobalt-Free EV Battery Is Just Around the Corner. 27 May 2021. Available online: https://lmc-auto.com/wp-content/uploads/2021/05/The-next-generation-cobalt-free-EV-battery-is-just-around-the-corner.pdf (accessed on 22 October 2023).
- Müller, J.; Abdollahifar, M.; Doose, S.; Michalowski, P.; Wu, N.; Kwade, A. Effects of carbon coating on calendered nano-silicon graphite composite anodes of LiB. J. Power Sources 2022, 548, 232000. [Google Scholar] [CrossRef]
- Yi, S.; Wang, B.; Chen, Z.; Wang, R.; Wang, D. A study on LiFePO 4/graphite cells with built-in Li4 Ti5 O12 reference electrodes. RSC Adv. 2018, 8, 18597–18603. [Google Scholar] [CrossRef] [PubMed]
- da Silva Lima, L.; Wu, J.; Cadena, E.; Groombridge, A.; Dewulf, J. Towards environmentally sustainable battery anode materials: Life cycle assessment of mixed niobium oxide (XNO™) and lithium titanium-oxide (LTO). Sustain. Mater. Technol. 2023, 37, e00654. [Google Scholar]
- Tian, T.; Lu, L.; Yin, Y.; Li, F.; Zhang, T.; Song, Y.; Tan, Y.; Yao, H. Multiscale designed niobium titanium oxide anode for fast charging lithium ion batteries. Adv. Funct. Mater. 2021, 31, 2007419. [Google Scholar] [CrossRef]
- Mehta, J. A Review of Temperature Effects on Varying Li-Ion Cell Chemistries for Electric Vehicle Applications. SSRN 4254130. 16 September 2022. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=4254130 (accessed on 1 July 2023).
- Kurzweil, P.; Garche, J. Overview of Batteries for Future Automobiles. In Lead-Acid Batteries for Future Automobiles; Elsevier: Amsterdam, The Netherlands, 2017; pp. 27–96. [Google Scholar]
- Zheng, F.; Kotobuki, M.; Song, S.; Lai, M.; Lu, L. Review on solid electrolytes for all-solid-state lithium-ion batteries. J. Power Sources 2018, 389, 198–213. [Google Scholar] [CrossRef]
- Costa, C.; Lanceros-Mendez, S. Recent advances on battery separators based on poly (vinylidene fluoride) and its copolymers for lithium-ion battery applications. Curr. Opin. Electrochem. 2021, 29, 100752. [Google Scholar] [CrossRef]
- Chen, X.; Chen, S.; Lin, Y.; Wu, K.; Lu, S. Multi-functional ceramic-coated separator for lithium-ion batteries safety tolerance improvement. Ceram. Int. 2020, 46, 24689–24697. [Google Scholar] [CrossRef]
- Kim, S.; Song, Y.; Wee, J.; Kim, C.; Ahn, B.; Lee, J.; Shu, S.; Terrones, M.; Kim, Y.; Yang, C. Few-layer graphene coated current collectors for safe and powerful lithium ion batteries. Carbon 2019, 153, 495–503. [Google Scholar] [CrossRef]
- Ashuri, M.; He, Q.; Shaw, L. Silicon as a potential anode material for Li-ion batteries: Where size, geometry and structure matter. Nanoscale 2016, 8, 74–103. [Google Scholar] [CrossRef]
- Gonzalez, A.F.; Yang, N.; Liu, R. Silicon anode design for lithium-ion batteries: Progress and perspectives. J. Phys. Chem. C 2017, 121, 27775–27787. [Google Scholar] [CrossRef]
- Ashuri, M.; He, Q.; Shaw, L. Silicon oxides for Li-ion battery anode applications: Toward long-term cycling stability. J. Power Sources 2023, 559, 232660. [Google Scholar] [CrossRef]
- Peng, C.; Chen, H.; Li, Q.; Cai, W.; Yao, Q.; Wu, Q.; Yang, J.; Yang, Y. Synergistically reinforced lithium storage performance of in situ chemically grown silicon@ silicon oxide core—Shell nanowires on three-dimensional conductive graphitic scaffolds. J. Mater. Chem. A 2014, 2, 13859–13867. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Q.; Zhao, Y.; He, R.; Xu, M.; Feng, S.; Li, S.; Zhou, L.; Mai, L. Silicon oxides: A promising family of anode materials for lithium-ion batteries. Chem. Soc. Rev. 2019, 1, 48. [Google Scholar] [CrossRef]
- Shaw, L.; Ashuri, M. Coating-A potent method to enhance electrochemical performance of Li (NixMnyCoz) O2 cathodes for Li-ion batteries. Adv. Mater. Lett. 2019, 10, 369–380. [Google Scholar] [CrossRef]
- Hassan, E.; Amiriyan, M.; Frisone, D.; Dunham, J.; Farahati, R.; Farhad, S. Effects of Coating on the Electrochemical Performance of a Nickel-Rich Cathode Active Material. Energies 2022, 15, 4886. [Google Scholar] [CrossRef]
- Kaur, G.; Gates, B. Surface Coatings for Cathodes in Lithium Ion Batteries: From Crystal Structures to Electrochemical Performance. J. Electrochem. Soc. 2022, 169, 043504. [Google Scholar] [CrossRef]
- Emani, S.; Liu, C.; Ashuri, M.; Sahni, K.; Wu, J.; Yang, W.; Németh, K.; Shaw, L. Li3BN2 as a Transition Metal Free, High Capacity Cathode for Li-ion Batteries. ChemElectroChem 2019, 6, 320–325. [Google Scholar] [CrossRef]
- Rodrigues, M.; Kalaga, K.; Gullapalli, H.; Babu, G.; Reddy, A.; Ajayan, P. Hexagonal boron nitride-based electrolyte composite for Li-ion battery operation from room temperature to 150 C. Adv. Energy Mater. 2016, 6, 1600218. [Google Scholar] [CrossRef]
- Soloveichik, G. Flow batteries: Current status and trends. Chem. Rev. 2015, 115, 11533–11558. [Google Scholar] [CrossRef]
- Lewis, G.N.; Keyes, F.G. The potential of Lithium Electrode. J. Am. Chem. Soc. 1913, 35, 340–344. [Google Scholar] [CrossRef]
- Hanusa, T.P. The Lightest Metals: Science and Technology from Lithium to Calcium; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Diouf, B.; Pode, R. Potential of lithium-ion batteries in renewable energy. Renew. Energy 2015, 76, 375–380. [Google Scholar] [CrossRef]
- Gao, J.; Shi, S.-Q.; Li, H. Brief overview of electrochemical potential in lithium ion batteries. Chin. Phys. B 2015, 25, 018210. [Google Scholar] [CrossRef]
- Jarrat, E. New Lessons from the Epic Story of Moli Energy, the Canadian Pioneer of Rechargeable Lithium Battery Technology. September 2020. Available online: https://electricautonomy.ca/2020/09/18/moli-energy-lithium-battery-technology/ (accessed on 5 January 2023).
- Zhu, K.; Wang, C.; Chi, Z.; Ke, F.; Yang, Y.; Wang, A.; Wang, W.; Miao, L. How far away are lithium-sulfur batteries from commercialization? Front. Energy Res. 2019, 15, 123. [Google Scholar] [CrossRef]
- Wu, F.; Chu, F.; Xue, Z. Lithium-Ion Batteries. In Encyclopedia of Energy Storage; Cabeza, L.F., Ed.; Elsevier: Oxford, UK, 2022; pp. 5–13. [Google Scholar]
- Xu, W.; Wang, J.; Ding, F.; Chen, X.; Nasybulin, E.; Zhang, Y.; Zhang, J. Lithium metal anodes for rechargeable batteries. Energy Environ. Sci. 2014, 7, 513–537. [Google Scholar] [CrossRef]
- Jennifer, L. Lithium-ion Battery Capacity to Reach 6.5 TWh in 2030, Says S&P Global. Carbon Credits. 18 July 2023. Available online: https://carboncredits.com/lithium-ion-battery-capacity-to-reach-6-5-twh-in-2030-says-sp-global/ (accessed on 1 July 2023).
- Li, Q.; Yang, Y.; Yu, X.; Li, H. A 700 W⋅ h⋅ kg− 1 Rechargeable Pouch Type Lithium Battery. Chin. Phys. Lett. 2023, 40, 048201. [Google Scholar] [CrossRef]
- Gipson, L. NASA’s Solid-State Battery Research Exceeds Initial Goals, Draws Interest. 7 October 2022. Available online: https://www.nasa.gov/aeroresearch/nasa-solid-state-battery-research-exceeds-initial-goals-draws-interest (accessed on 5 June 2023).
- Walters, I.; Hull, R. Weight of Electric Vehicles Could Cause ‘Catastrophic’ Damage and ‘Lead to Car Parks Collapsing’: Engineers Warn Britain’s Parking Facilities Were Not Designed for Hulking Battery Cars. 19 December 2022. Available online: https://www.dailymail.co.uk/news/article-11550269/Weight-electric-vehicles-cause-catastrophic-damage-lead-car-parks-collapsing.html (accessed on 27 December 2022).
- Zhao, M.; Li, B.; Zhang, X.; Huang, J.; Zhang, Q. A perspective toward practical lithium–sulfur batteries. ACS Cent. Sci. 2020, 6, 1095–1104. [Google Scholar] [CrossRef]
- Aurbach, D.; McCloskey, B.; Nazar, L.; Bruce, P. Advances in understanding mechanisms underpinning lithium–air batteries. Nat. Energy 2016, 1, 16128. [Google Scholar] [CrossRef]
- Lim, H.; Park, J.; Shin, H.; Jeong, J.; Kim, J.; Nam, K.; Jung, H.; Chung, K. A review of challenges and issues concerning interfaces for all-solid-state batteries. Energy Storage Mater. 2020, 25, 224–250. [Google Scholar] [CrossRef]
- Salgado, R.M.; Oliveira, J.C.R.E.; Braga, H. Intelligence in Mobile Battery Applications Desk Research Report. Alliance for Batteries Technology, Training and Skills; The European Commission: Brussels, Belgium, 2020. [Google Scholar]
- Kalhoff, J.; Eshetu, G.; Bresser, D.; Passerini, S. Safer electrolytes for lithium-ion batteries: State of the art and perspectives. ChemSusChem 2015, 8, 2154–2175. [Google Scholar] [CrossRef]
- European Commission. Batteries Europe. December 2021. Available online: https://energy.ec.europa.eu/system/files/2021-12/vol-3-008-2.pdf (accessed on 6 June 2023).
- Wu, X.; Chen, K.; Yao, Z.; Hu, J.; Huang, M.; Meng, J.; Ma, S.; Wu, T.; Cui, Y.; Li, C. Metal organic framework reinforced polymer electrolyte with high cation transference number to enable dendrite-free solid state Li metal conversion batteries. J. Power Sources 2021, 501, 229946. [Google Scholar] [CrossRef]
- Xu, H.; Ye, W.; Wang, Q.; Han, B.; Wang, J.; Wang, C.; Deng, Y. An in situ photopolymerized composite solid electrolyte from halloysite nanotubes and comb-like polycaprolactone for high voltage lithium metal batteries. J. Mater. Chem. A 2021, 9, 9826–9836. [Google Scholar] [CrossRef]
- Park, J.; Park, D.; Go, S.; Nam, D.; Oh, J.; Han, Y.; Lee, H. Malonatophosphate as an SEI-and CEI-forming additive that outperforms malonatoborate for thermally robust lithium-ion batteries. Energy Storage Mater. 2022, 50, 75–85. [Google Scholar] [CrossRef]
- B.S.B. Group. Battery Technology. 2023. Available online: https://www.blue-solutions.com/en/battery-technology/ (accessed on 20 August 2023).
- Trofimov, B.; Myachina, G.; Oparina, L.; Korzhova, S.; Doo, G.N.S.; Cho, M.; Kim, H. Triethyl 2-(1, 3-oxazolidin-3-yl) ethyl orthosilicate as a new type electrolyte additive for lithium-ion batteries with graphite anodes. J. Power Sources 2005, 147, 260–263. [Google Scholar] [CrossRef]
- Aziam, H.; Larhrib, B.; Hakim, C.; Sabi, N.; Youcef, H.; Saadoune, I. Solid-state electrolytes for beyond lithium-ion batteries: A review. Renew. Sustain. Energy Rev. 2022, 167, 112694. [Google Scholar] [CrossRef]
- Yamauchi, H.; Ikejiri, J.; Tsunoda, K.; Tanaka, A.; Sato, F.; Honma, T.; Komatsu, T. Enhanced rate capabilities in a glass-ceramic-derived sodium all-solid-state battery. Sci. Rep. 2020, 10, 9453. [Google Scholar] [CrossRef]
- Bhat, M.; Hashmi, S. Mixture of non-ionic and organic ionic plastic crystals immobilized in poly (vinylidene fluoride-co-hexafluoropropylene): A flexible gel polymer electrolyte composition for high performance carbon supercapacitors. J. Energy Storage 2022, 51, 104514. [Google Scholar] [CrossRef]
- Fan, L.; Wei, S.; Li, S.; Li, Q.; Lu, Y. Recent progress of the solid-state electrolytes for high-energy metal-based batteries. Adv. Energy Mater. 2018, 8, 1702657. [Google Scholar] [CrossRef]
- Alliance for Batteries Technology, Training and Skills. In Desk Research and Data Analysis for Sub-Sector IMBA—ALBATT; The European Commission: Brussels, Belgium, 2021.
- Li, M.; Jafta, C.; Belharouak, I. Progress of nanotechnology for lithium-sulfur batteries. Front. Nanosci. 2021, 19, 137–164. [Google Scholar]
- Kim, S.; Chart, Y.; Narayanan, S.; Pasta, M. Thin Solid Electrolyte Separators for Solid-State Lithium–Sulfur Batteries. Nano Lett. 2022, 22, 10176–10183. [Google Scholar] [CrossRef]
- McAlpine, K. 1,000-Cycle Lithium-Sulfur Battery Could Quintuple Electric Vehicle Ranges. 12 January 2022. Available online: https://news.umich.edu/1000-cycle-lithium-sulfur-battery-could-quintuple-electric-vehicle-ranges/ (accessed on 6 June 2023).
- Bi, C.; Zhao, M.; Hou, L.; Chen, Z.; Zhang, X.; Li, B.; Yuan, H.; Huang, J. Anode material options toward 500 Wh kg−1 lithium–sulfur batteries. Adv. Sci. 2022, 9, 2103910. [Google Scholar] [CrossRef]
- Robinson, J.; Xi, K.; Kumar, R.; Ferrari, A.; Au, H.; Titirici, M.; Parra-Puerto, A.; Kucernak, A.; Fitch, S.; Garcia-Araez, N.; et al. 2021 roadmap on lithium sulfur batteries. J. Phys. Energy 2021, 3, 031501. [Google Scholar] [CrossRef]
- Abert, M. Analysis of gases emitted in safety events. Electrochemical Powers Sources: Fundamentals, Systems, and Applications. In Li-Battery Safety; Elsevier: Amsterdam, The Netherlands, 2018; pp. 196–215. [Google Scholar]
- Seh, Z.; Sun, Y.; Zhang, Q.; Cui, Y. Designing high-energy lithium–sulfur batteries. Chem. Soc. Rev. 2016, 45, 5605–5634. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, T.; Gao, Y.; Pan, Z.; Hu, R.; Wang, J. Will lithium-sulfur batteries be the next beyond-lithium ion batteries and even much better? InfoMat 2022, 4, e12359. [Google Scholar] [CrossRef]
- Nayak, P.; Yang, L.; Brehm, W.; Adelhelm, P. From lithium-ion to sodium-ion batteries: Advantages, challenges, and surprises. Angew. Chem. Int. Ed. 2018, 57, 102–120. [Google Scholar] [CrossRef]
- Abraham, K. How comparable are sodium-ion batteries to lithium-ion counterparts? ACS Energy Lett. 2020, 5, 3544–3547. [Google Scholar] [CrossRef]
- Hu, Y.; Li, Y. Unlocking sustainable Na-ion batteries into industry. ACS Energy Lett. 2021, 6, 4115–4117. [Google Scholar] [CrossRef]
- Zhao, C.; Lu, Y.; Li, Y.; Jiang, L.; Rong, X.; Hu, Y.; Li, H.; Chen, L. Novel Methods for Sodium-Ion Battery Materials. Small Methods 2017, 1, 1600063. [Google Scholar] [CrossRef]
- Guo, Q.; Zeng, W.; Liu, S.; Li, Y.; Xu, J.; Wang, J.; Wang, Y. Recent developments on anode materials for magnesium-ion batteries: A review. Rare Met. 2021, 40, 290–308. [Google Scholar] [CrossRef]
- Kyeremateng, N.; Hahn, R. Attainable energy density of microbatteries. ACS Energy Lett. 2018, 3, 1172–1175. [Google Scholar] [CrossRef]
- MARKETS AND MARKETS. Micro Battery Market. July 2023. Available online: https://www.marketsandmarkets.com/Market-Reports/micro-battery-market-25755945.html?gclid=EAIaIQobChMIg670_fqugQMV4BkGAB3hIAF6EAAYASAAEgJ0VfD_BwE (accessed on 5 September 2023).
- Xia, Q.; Zan, F.; Zhang, Q.; Liu, W.; Li, Q.; He, Y.; Hua, J.; Liu, J.; Xu, J.; Wang, J.; et al. All-Solid-State Thin Film Lithium/Lithium-Ion Microbatteries for Powering the Internet of Things. Adv. Mater. 2023, 35, 2200538. [Google Scholar] [CrossRef]
- Jafferis, N.; Helbling, E.; Karpelson, M.; Wood, R. Untethered flight of an insect-sized flapping-wing microscale aerial vehicle. Nature 2019, 570, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, O. RoboBee breaks free. Nat. Electron. 2019, 2, 265. [Google Scholar] [CrossRef]
- Hur, J.; Smith, L.; Dunn, B. High areal energy density 3D lithium-ion microbatteries. Joule 2018, 2, 1187–1201. [Google Scholar] [CrossRef]
- Han, Y.; Dai, C.L.J.; Liu, F.; Ma, H.; Wang, Y.; Lu, B.; Shao, C.; Guo, Q.; Jin, X.; Zhang, X. A compact aqueous K-ion Micro-battery by a Self-shrinkage assembly strategy. Chem. Eng. J. 2022, 429, 13229. [Google Scholar] [CrossRef]
- ITEN. Available online: https://www.iten.com/en/micro-batteries (accessed on 5 September 2023).
- ST. EFL700A39. June 2014. Available online: https://www.st.com/resource/en/datasheet/efl700a39.pdf (accessed on 5 September 2023).
- MURATA. CR1216. Available online: https://www.murata.com/products/productdata/8808560951326/CR1216-DATASHEET.pdf?1617161448000 (accessed on 6 September 2023).
- Ayerbe, E.; Berecibar, M.; Clark, S.; AA, F.; Ruhland, J. Digitalization of battery manufacturing: Current status, challenges, and opportunities. Adv. Energy Mater. 2022, 12, 2102696. [Google Scholar] [CrossRef]
- Loveridge, M.; Dowson, M. Why Batteries Fail and How to Improve Them: Understanding Degradation to Advance Lithium-Ion Battery Performance; The Faraday Institution, Warwick Manufacturing Group, University of Warwick: Coventry, UK, 2021. [Google Scholar]
- International Energy Agency. Global EV Outlook 2023—Catching up with Climate Ambitions; IEA: Paris, France, 2023. [Google Scholar]
- Liu, Y.; Zhang, R.; Wang, J.; Wang, Y. Current and future lithium-ion battery manufacturing. IScience 2021, 24, 102332. [Google Scholar] [CrossRef] [PubMed]
- Jinasena, A.; Burheim, O.; AH, S. A flexible model for benchmarking the energy usage of automotive lithium-ion battery cell manufacturing. Batteries 2021, 7, 14. [Google Scholar] [CrossRef]
- Yuan, C.; Deng, Y.; Li, T.; Yang, F. Manufacturing energy analysis of lithium ion battery pack for electric vehicles. CIRP Ann. 2017, 66, 53–56. [Google Scholar] [CrossRef]
- Hatzell, K.; Zheng, Y. Prospects on large-scale manufacturing of solid state batteries. MRS Energy Sustain. 2021, 8, 33–39. [Google Scholar] [CrossRef]
- Heimes, H.; Kampfer, A.; Hemdt, A.; Schön, C.; Michaelis, S.; Rahimzei, E. Production of All Solid-State Battery Cells; German Mechanical Engineering Industry Association—RWTH Aachen University: Aachen, Germany, 2018. [Google Scholar]
- European Commission. Questions and Answers on Revised EU Rules on Industrial Emissions; European Commission: Brussels, Belgium, 2022. [Google Scholar]
- Cooke, P. Gigafactory logistics in space and time: Tesla’s fourth gigafactory and its rivals. Sustainability 2020, 12, 2044. [Google Scholar] [CrossRef]
- Brückner, L.; Frank, J.; Elwert, T. Industrial recycling of lithium-ion batteries—A critical review of metallurgical process routes. Metals 2020, 10, 1107. [Google Scholar] [CrossRef]
- VERKOR. Everything You Need to Know about Lithium-Ion Batteries. 14 December 2021. Available online: https://verkor.com/en/everything-you-need-to-know-about-lithium-ion-batteries/ (accessed on 5 June 2022).
- Xu, J.; Ding, C.; Chen, P.; Tan, L.; Chen, C.; Fu, J. Intrinsic self-healing polymers for advanced lithium-based batteries: Advances and strategies. Appl. Phys. Rev. 2020, 7, 031304. [Google Scholar] [CrossRef]
- Narayan, R.; Laberty-Robert, C.; Pelta, J.; Tarascon, J.; Dominko, R. Self-Healing: An Emerging Technology for Next-Generation Smart Batteries. Adv. Energy Mater. 2022, 12, 2102652. [Google Scholar] [CrossRef]
- European Commission. Regulation of the European Parliament and of the Council Concerning Batteries and Waste Batteries, Repealing Directive 2006/66/EC and Amending Regulation (EU) No 2019/1020; European Commission: Brussels, Belgium, 2020. [Google Scholar]
- Global Battery Alliance. A Vision for a Sustainable Battery Value Chain in 2030: Unlocking the Full Potential to Power Sustainable Development and Climate Change Mitigation; World Economic Forum: Geneva, Switzerland, 2019. [Google Scholar]
- Zhengh, M. The Environmental Impacts of Lithium and Cobalt Mining. EARTH.ORG. 31 March 2023. Available online: https://earth.org/lithium-and-cobalt-mining/ (accessed on 21 October 2023).
- Mancini, L.; Eslava, N.; Traverso, M.; Mathieux, F. Responsible and Sustainable Sourcing of Battery Raw Materials; Publications Office of the European Union: Luxembourg, 2020. [Google Scholar]
- Campbell, M. In pictures: South America’s ‘Lithium Fields’ Reveal the Dark Side of Our Electric Future. Euronews. Green. 1 February 2022. Available online: https://www.euronews.com/green/2022/02/01/south-america-s-lithium-fields-reveal-the-dark-side-of-our-electric-future (accessed on 21 October 2023).
- Luong, J.; Tran, C.; Ton-That, D. A Paradox over Electric Vehicles, Mining of Lithium for Car Batteries. Energies 2022, 15, 7997. [Google Scholar] [CrossRef]
- Tsuji, K. Global Value Chains: Graphite in Lithium-ion Batteries for Electric Vehicles; Office of Industries, US International Trade Commission: Washington, DC, USA, 2022. [Google Scholar]
- Nate, S.; Bilan, Y.; Kurylo, M.; Lyashenko, O.; Napieralski, P.; Kharlamova, G. Mineral policy within the framework of limited critical resources and a green energy transition. Energies 2021, 14, 2688. [Google Scholar] [CrossRef]
- Careaga, I. Raw Material Shortage: The New Big Challenge for the Battery Industry. CIC energiGUNE. 29 June 2022. Available online: https://cicenergigune.com/en/blog/raw-material-shortage-new-big-challenge-battery-industry (accessed on 5 January 2023).
- Jiao, N. Second-Life Electric Vehicle Batteries 2020–2030; IDTechx: Tokyo, Japan, 2020. [Google Scholar]
- Engel, H.; Hertzke, P.; Siccardo, G. Second-Life EV Batteries: The Newest Value Pool in Energy Storage. 30 April 2019. Available online: https://www.mckinsey.com/industries/automotive-and-assembly/our-insights/second-life-ev-batteries-the-newest-value-pool-in-energy-storage (accessed on 20 May 2023).
- Huang, S.; Tseng, K.; Liang, J.; Chang, C.; Pecht, M. An online SOC and SOH estimation model for lithium-ion batteries. Energies 2017, 10, 512. [Google Scholar] [CrossRef]
- Oji, T.; Zhou, Y.; Ci, S.; Kang, F.; Chen, X.; Liu, X. Data-driven methods for battery soh estimation: Survey and a critical analysis. IEEE Access 2021, 10, 126903–126916. [Google Scholar] [CrossRef]
- Illa Font, C.H.; Siqueira, H.V.; Machado Neto, J.E.; Santos, J.L.F.D.; Stevan, S.L., Jr.; Converti, A.; Corrêa, F.C. Second Life of Lithium-Ion Batteries of Electric Vehicles: A Short Review and Perspectives. Energies 2023, 16, 953. [Google Scholar] [CrossRef]
- Hossain, E.; Murtaugh, D.; Mody, J.; Faruque, H.; Sunny, M.; Mohammad, N. A comprehensive review on second-life batteries: Current state, manufacturing considerations, applications, impacts, barriers & potential solutions, business strategies, and policies. IEEE Access 2019, 7, 73215–73252. [Google Scholar]
- Wei, Z.; Zhao, J.; He, H.; Ding, G.; Cui, H.; Liu, L. Future smart battery and management: Advanced sensing from external to embedded multi-dimensional measurement. J. Power Sources 2021, 489, 229462. [Google Scholar] [CrossRef]
- Wei, Z.; Hu, J.; He, H.; Yu, Y.; Marco, J. Embedded distributed temperature sensing enabled multistate joint observation of smart lithium-ion battery. IEEE Trans. Ind. Electron. 2022, 70, 555–565. [Google Scholar] [CrossRef]
- European Commission. Green Deal: EU Agrees New Law on More Sustainable and Circular Batteries to Support EU’s Energy Transition and Competitive Industry. 9 December 2022. Available online: https://ec.europa.eu/commission/presscorner/detail/en/ip_22_7588 (accessed on 25 May 2023).
- Halleux, V. New EU Regulatory Framework for Batteries; European Parliamentary Research Service: Brussels, Belgium, 2023. [Google Scholar]
- Gaines, L. Lithium-ion battery recycling processes: Research towards a sustainable course. Sustain. Mater. Technol. 2018, 17, e00068. [Google Scholar] [CrossRef]
- Duan, L.; Cui, Y.; Li, Q.; Wang, J.; Man, C.; Wang, X. Recycling and direct-regeneration of cathode materials from spent ternary lithium-ion batteries by hydrometallurgy: Status quo and recent developments: Economic recovery methods for lithium nickel cobalt manganese oxide cathode materials. Johns. Matthey Technol. Rev. 2021, 65, 431–452. [Google Scholar] [CrossRef]
- EUROBAT. Battery Innovation—Roadmap 2030; Version 2.0; EUROBAT: Brussels, Belgium, 2022. [Google Scholar]




| Year | |||
|---|---|---|---|
| 2020 | 2025 | 2030 | |
| Electric Mobility | 81.21% | 83.21% | 88.94% |
| Energy Storage | 3.55% | 10.81% | 8.43% |
| Portable Electronics | 15.25% | 5.97% | 2.63% |
| Battery Generation | Technology/Electrode Active Materials | Cell Chemistry/Type | Implementation Date/Forecast Market Deployment |
|---|---|---|---|
| Gen 1 | Cathode: NFP, NCA, LCO | Lithium-Ion | 1991 |
| Anode: Carbone/Graphite | |||
| Gen 2a | Cathode: NMC111, LMO | 1994 | |
| Anode: Carbone/Graphite | |||
| Gen 2b | Cathode: NMC532, NMC622 | 2005 | |
| Anode: Carbone/Graphite | |||
| Gen 3a | Cathode: NMC622, NMC 811 | 2020 | |
| Anode: Graphite + 5/10% Si | |||
| Gen 3b | Cathode: High Energy NMC, High Voltage Spinel—5 V | Optimized Lithium-Ion | 2025 |
| Anode: Silicon/Carbon | |||
| Gen 4a | Cathode: NMC | Solid State Lithium-Ion | 2025 |
| Anode: Silicon/Carbon | |||
| Solid Electrolyte | |||
| Gen 4b | Cathode: NMC | Solid State Lithium-Metal | >2025 |
| Anode: Lithium metal | |||
| Solid Electrolyte | |||
| Gen 4c | Cathode: High Energy NMC, High Voltage Spinel | Advanced Solid State | 2030 |
| Anode: Lithium metal | |||
| Solid Electrolyte | |||
| Gen 5 | LiO2 Li-Air/Metal-Air | Metal-Air | >2030 |
| Li-Sulphur | LiS | ||
| New ion-based systems (Na, Mg, Zn or Al) | New ion-based insertion chemistries |
| Manufacturer | ITEN | ST Micro | MURATA |
| Product number | ITX121005B | EFL700A39 | CR1216 |
| Type | thin-film solid-state | thin-film solid-state | coin cell |
| Footprint | 3.2 mm × 2.5 mm | 25.7 × 25.7 mm | 12.5 mm |
| Thickness (µm) | 600 mm | 220 mm | 1600 mm |
| Capacity (µAh) | 50 µAh | 700 µAh | 30,000 µAh |
| Voltage (V) | 2.5 V | 3.9 V | 3.0V |
| Operating temperature range | −40 °C/+85 °C | −20 °C to 60 °C | −30 °C to 70 °C |
| Reference | [122] | [123] | [124] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Itani, K.; De Bernardinis, A. Review on New-Generation Batteries Technologies: Trends and Future Directions. Energies 2023, 16, 7530. https://doi.org/10.3390/en16227530
Itani K, De Bernardinis A. Review on New-Generation Batteries Technologies: Trends and Future Directions. Energies. 2023; 16(22):7530. https://doi.org/10.3390/en16227530
Chicago/Turabian StyleItani, Khaled, and Alexandre De Bernardinis. 2023. "Review on New-Generation Batteries Technologies: Trends and Future Directions" Energies 16, no. 22: 7530. https://doi.org/10.3390/en16227530
APA StyleItani, K., & De Bernardinis, A. (2023). Review on New-Generation Batteries Technologies: Trends and Future Directions. Energies, 16(22), 7530. https://doi.org/10.3390/en16227530







