Hydrogen Generation from a Small-Scale Solar Photovoltaic Thermal (PV/T) Electrolyzer System: Numerical Model and Experimental Verification
Abstract
1. Introduction
2. Materials and Methods
2.1. System Design
2.2. Mathematical Model of the System
2.3. PEM Mathematical Model
2.4. Economic Analysis
- The economic evaluation (N) period is 25 years for the PV/T panels, the DC/DC converter, and the storage systems, and is 15 years for the PEM electrolyzer.
- The economic indicators of nominal discount rate and inflation rate were used as 9% and 12%, respectively, for 2017 [66].
- The installation, operation and maintenance are not included in cost calculation.
- The capital costs per watt for the PV, the PV/T, and the PEM electrolyzer were taken as 0.82, 2, and 0.9 USD/W, respectively.
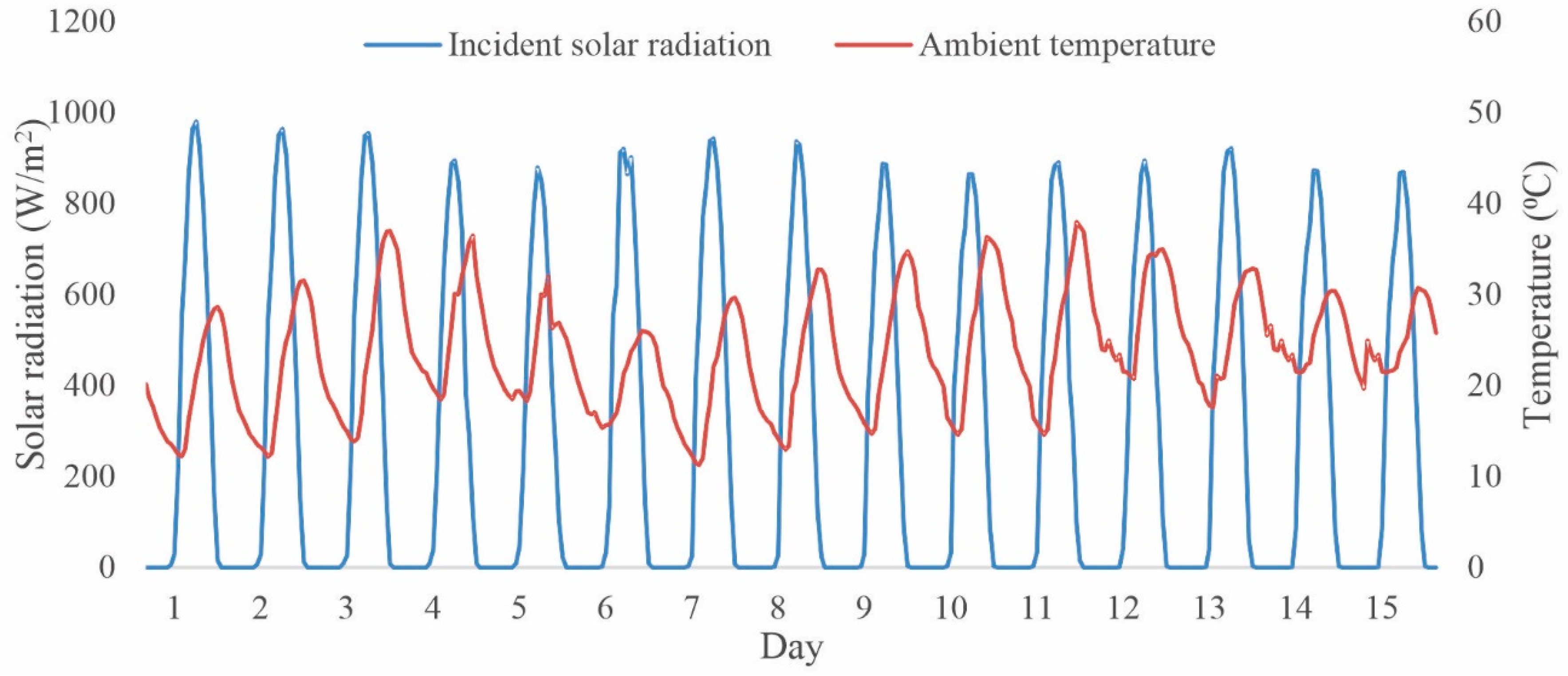
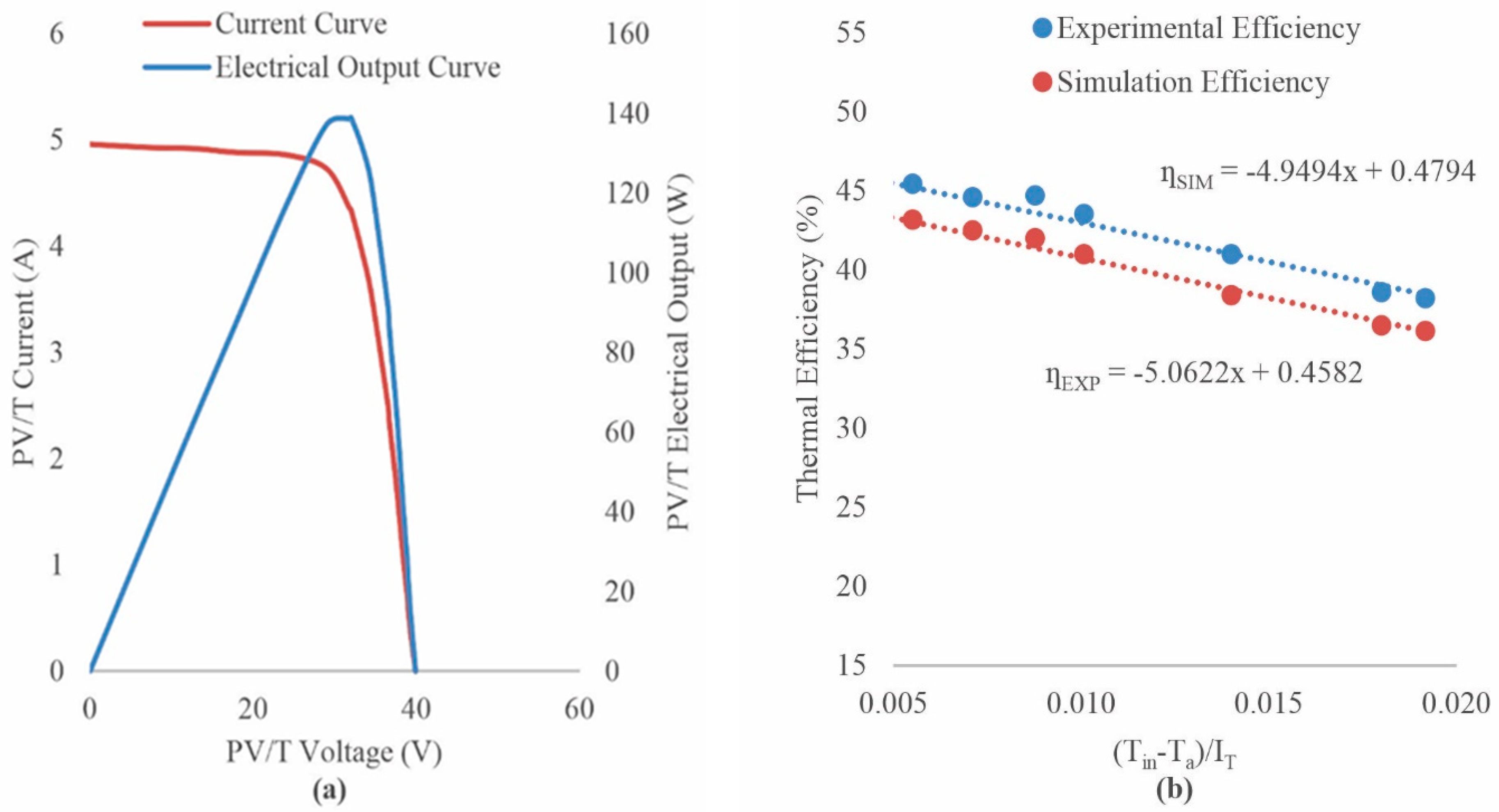
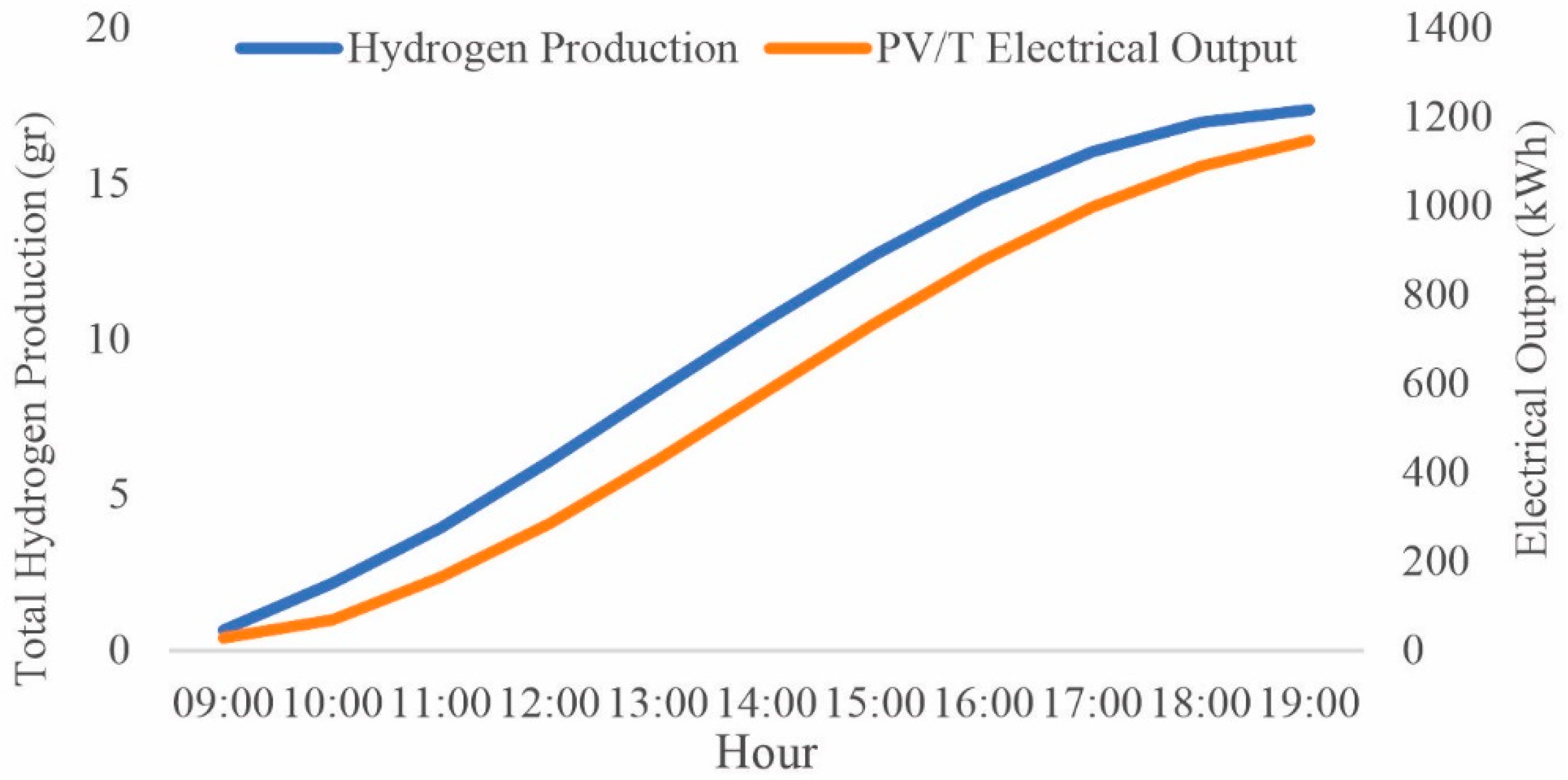
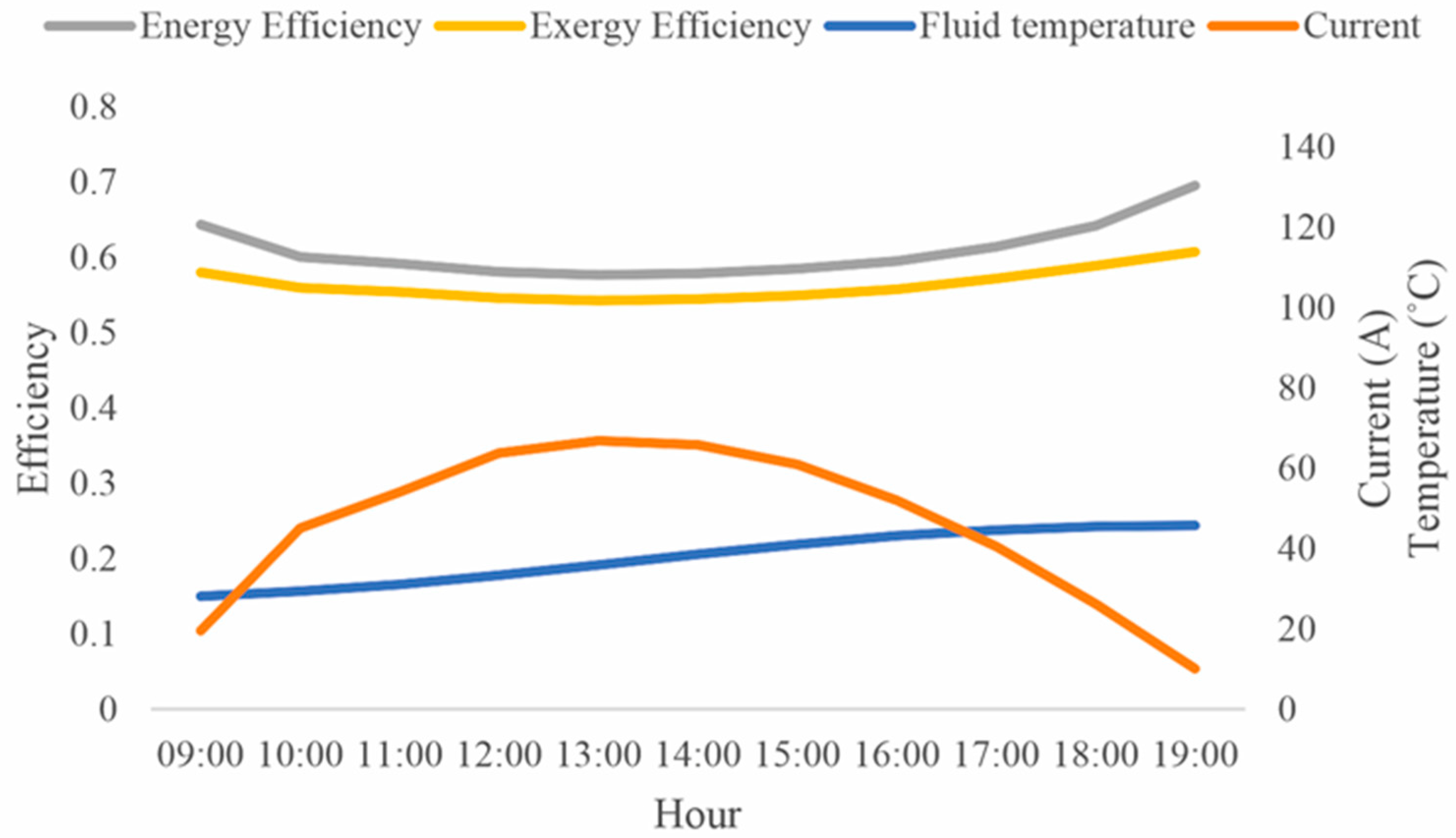
3. Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature and Subscripts
| Nomenclature | Subscripts | ||
| IT | Inclined solar radiation (W/m2) | A | Ambient |
| Qu | Useful energy (w) | in | Inlet |
| UL | Overall heat loss coefficient (w/m2 °C) | o | Outlet |
| W | Width of the tube spacing (m) | a | Anode |
| D | Diameter of copper tube (m) | c | Cathode |
| τ | Transmissivity of glass | rev | Reversible voltage |
| αg | Absorptivity of glass | th | Thermal |
| Cp | Water specific heat, j kg−1 k−1 | e | Electrical |
| k | Specific heat rate | t | Total |
| F’ | Collector efficiency factor | m | Membrane |
| FR | Collector flow rate factor | H2O | Water |
| n | Number of electron | O2 | Oxygen |
| p | Partial pressure | H2 | Hydrogen |
| z | Stoichiometric coefficient of electron | ph | Physical |
| α | Transfer coefficient | ch | Chemical |
| h | Overpotential | elc | Electrolyzer |
| δ | Membrane thickness | ohm | Ohmic |
| s | Membrane conductivity | act | Activation |
| r | Material resistivity | LHV | Lower heat value |
| i | Current density | η | Efficiency |
| io | Exchange current density | A | Surface area (m2) |
| P | Operating pressure | PEM | Polymer electrolyte membrane |
| ΔH | Enthalpy change | PV | Photovoltaic |
| ΔS | Entropy change | PV/T | Photovoltaic/Thermal |
| R | Universal gas constant | sim | Simulation |
| F | Faraday’s constant | Exp | Experimental |
| z | Stoichiometric coefficient | TLCC | Total life-cycle cost |
| T | Temperature | f’ | Inflation |
| λ | Degree of humidification | CoE | Cost of energy |
| ṁ | Mass flow rate (kg/s) | N | Lifetime (year) |
| Er | Reversible voltage | R’ | Real discount rate |
| Ex | Exergy | i’ | Nominal discount rate |
| ΔG | Gibbs free energy | CRF | Capital recovery factor |
| E0 | Reversible potential of water splitting | TPV | Total present value |
| δ | Thickness | r | Reactant |
| Ė | Corresponding rate of exergy input | p | Product |
| HOMER | Hybrid optimization model for electric renewable |
References
- Li, W.; Liu, T.; Liu, X.; Hu, X. Thermodynamic analysis and optimization of photovoltaic/thermal hybrid hydrogen generation system based on complementary combination of photovoltaic cells and proton exchange membrane electrolyzer. Energy Convers. Manag. 2019, 183, 97–108. [Google Scholar]
- Ramzy, K.; El Elnaghi, B.; Ismail, T.; Abd El-Salam, M.; Abewahab, M.N. Using MATLAB to model and simulate a photovoltaic system to produce hydrogen. Energy Convers. Manag. 2019, 185, 101–129. [Google Scholar]
- International Energy Agency, IEA. Energy Climateand Change-World Energy Outlook Special Report, Special Report on Energy and Climate Change; IEA: Paris, France, 2015. [Google Scholar]
- Nazri, N.S.; Fudholi, A.; Bakhtyar, B.; Yen, C.H.; Ibrahim, A.; Ruslan, M.H.; Mat, S. Energy economic analysis of photovoltaicethermal-thermoelectric (PVT-TE) air collectors. Renew. Sustain. Energy Rev. 2018, 92, 187–197. [Google Scholar] [CrossRef]
- Tripanagnostopoulos, Y. Aspects and improvements of hybrid photovoltaic/thermal solar energy systems. Sol. Energy 2007, 81, 1117–1131. [Google Scholar] [CrossRef]
- Tripanagnostopoulos, Y.; Nousia, T.; Souliotis, M.; Yianoulis, P. Hybrid photovoltaic/thermal solar system. Sol. Energy 2002, 72, 217–234. [Google Scholar] [CrossRef]
- Huang, B.J.; Lin, T.H.; Hung, W.C.; Sun, F.S. Performance evaluation of solar photovoltaic/thermal systems. Sol. Energy 2001, 70, 443–448. [Google Scholar] [CrossRef]
- Tiwari, A.; Sodha, M.S. Performance evaluation of solar PV/T system: An experimental validation. Sol. Energy 2006, 80, 751–759. [Google Scholar] [CrossRef]
- Chow, T.T. A review on photovoltaic/thermal hybrid solar technology. Appl. Energy 2010, 87, 365–379. [Google Scholar] [CrossRef]
- Arif, H.M.; Sumathy, K. Photovoltaic thermal module concepts and their performance analysis: A review. Renew. Sustain. Energy 2010, 14, 1845–1859. [Google Scholar]
- Tiwari, G.N.; Mishra, R.K.; Solanki, S.C. Photovoltaic modules and their applications: A review on thermal modelling. Appl. Energy 2011, 88, 2287–2304. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Kaushik, S.C.; Tyagi, S.K. Avancement in solar photovoltaic/thermal (PV/T) hybrid collector technology. Renew Sustain. Energy 2012, 16, 1383–1398. [Google Scholar] [CrossRef]
- Wolf, M. Performance analysis of combined heating and photovoltaic power systems for residences. Energy Convers. Manag. 1976, 16, 79–90. [Google Scholar] [CrossRef]
- Khelifa, A.; Touafek, K.; Ben Moussa, H. Approach for the modelling of hybrid photovoltaic–thermal solar collector. IET Renew. Power Gener. 2015, 9, 207–217. [Google Scholar] [CrossRef]
- Bergene, T.; Løvvik, O.M. Model calculations on a flat-plate solar heat collector with integrated solar cells. Sol. Energy 1995, 55, 453–462. [Google Scholar] [CrossRef]
- Matuska, T. Performance and economic analysis of hybrid PVT collectors in solar DHW system. Energy Proc. 2014, 48, 150–156. [Google Scholar] [CrossRef]
- Singh, H.; Jain, A.; Singh, A.; Arora, S. Influence of absorber plate shape factor and mass flow rate on the performance of the PVT system. Appl. Therm. Eng. 2019, 156, 1016. [Google Scholar]
- Touafek, K.; Khelifa, A.; Adouane, M. Theoretical and experimental study of sheet and tubes hybrid PVT collector. Energy Convers. Manag. 2014, 80, 71–77. [Google Scholar] [CrossRef]
- Ibrahim, A.; Othman, M.Y.; Ruslan, M.H.; Mat, S.; Sopian, K. Recent advances in flat plate photovoltaic/thermal (PV/T) solar collectors. Renew. Sustain. Energy 2011, 15, 352–365. [Google Scholar] [CrossRef]
- Choi, H.; Choi, K.H. Performance Evaluation of PV/T Air Collector Having a Single-Pass Double-Flow Air Channel and Non-Uniform Cross-Section Transverse Rib. Energies 2020, 13, 2203. [Google Scholar] [CrossRef]
- Kim, J.; Ahn, J.G.; Kim, J.T. Demonstration of the Performance of an Air-Type Photovoltaic Thermal (PVT) System Coupled with a Heat-Recovery Ventilator. Energies 2016, 9, 728. [Google Scholar] [CrossRef]
- Calise, F.; Figaj, R.; Vanoli, L. Experimental and Numerical Analyses of a Flat Plate Photovoltaic/Thermal Solar Collector. Energies 2017, 10, 491. [Google Scholar] [CrossRef]
- Sakhr, M.; Ervina, E.; Saqaff, A. Theoretical study of new configuration of PVT system design. J. Adv. Mater. Res. 2013, 681–687. [Google Scholar]
- Fudholi, A.; Sopian, K.; Yazdı, M.; Ruslan, I.; Kazem, H. Performance Analysis of Photovoltaic Thermal (PVT) Water Collectors. Energy Convers. Manag. 2014, 78, 641–651. [Google Scholar] [CrossRef]
- Ibrahim, A.; Fudholi, A.; Sopian, K.; Othman, M.; Ruslan, M.H. Efficiencies and improvement potential of building integrated photovoltaic thermal (BIPVT) system. Energy Convers. Manag. 2014, 77, 527–534. [Google Scholar] [CrossRef]
- Nualboonrueng, T.; Tuenpusa, P.; Ueda, Y.; Akisawa, A. Field Experiments of PV-Thermal Collectors for Residential Application in Bangkok. Energies 2012, 5, 12–29. [Google Scholar] [CrossRef]
- Guarracino, I.; Freeman, J.; Ekins-Daukes, N.; Markides, C.N. Performance assessment and comparison of solar orc and hybrid pvt systems for the combined distributed generation of domestic heat and power. In Proceedings of the 12th International Conference on Heat Transfer, Fluid Mechanics and Thermodynamics 2016, Costa del Sol, Spain, 11–13 July 2016. [Google Scholar]
- Shekardasht, S.; Canli, E.; Ateş, A. Solar energy supported hydrogen production: A theoretical case study. Selcuk Univ. J. Eng. Sci. Technol. 2017, 5, 536–554. [Google Scholar]
- Chong, L. Intelligent Renewable Energy Storage and Management System for Rural Household. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2020. [Google Scholar]
- Yan, T.; Li, T.X.; Wang, R.Z. Thermochemical heat storage for solar heating and cooling systems. Adv. Sol. Heat. Cool. 2016, 10, 1016. [Google Scholar]
- Jain, S.; Agrawal, N. Storage of Renewable Energy Using Phase Change Materials (PCMs); WAITRO: New Delhi, India, 2012. [Google Scholar]
- Ineneji, C.; Bamisile, O.; Kuşaf, M. Super-Capacitors as an Alternative for Renewable Energy Unstable Supply. Acad. Perspect. Procedia 2018, 1, 11–20. [Google Scholar] [CrossRef]
- Arias, P.L.; Cambra, J.; Bilbao, M.; Barrio, V.L.; Requies, J. Hydrogen as a new energy vector. Dyna Bilbao 2008, 83, 291–295. [Google Scholar]
- Bozoglan, E.; Midilli, A.; Hepbasli, A. Sustainable assessment of solar hydrogen production techniques. Energy 2012, 46, 85–93. [Google Scholar] [CrossRef]
- Midilli, A.; Dincer, I. Hydrogen as a renewable and sustainable solution in reducing global fossil fuel consumption. Int. J. Hydrogen Energy 2008, 33, 4209–4222. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C. A review and recent developments in photo catalytic water splitting using TiO2 for hydrogen production. Renew. Sustain. Energy Rev. 2007, 11, 401–425. [Google Scholar] [CrossRef]
- Barone, G.; Buonomano, A.; Forzano, C.; Palombo, A.; Panagopoulos, O. Photovoltaic thermal collectors: Experimental analysis and simulation model of an innovative low-cost water-based prototype. Energy 2019, 179, 502–516. [Google Scholar] [CrossRef]
- Di Blasi, A.; Durso, C.; Baglio, V.; Antonucci, V.; Aricò, A.; Ornelas, R.; Matteucci, F.; Orozco, G.; Beltran, D.; Meas, Y.; et al. Preparation and evaluation of RuO2-IrO2, IrO 2-Pt and IrO2-Ta2O5 catalysts for the oxygen evolution reaction in an SPE electrolyzer. J. Appl. Electrochem. 2009, 39, 191–196. [Google Scholar] [CrossRef]
- Siracusano, S.; Baglio, V.; Di Blasi, A.; Briguglio, N.; Stassi, A.; Ornelas, R. Electrochemical characterization of single cell and short stack PEM electrolyzers based on a nanosized IrO2 anode electrocatalyst. Int. J. Hydrogen Energy 2010, 35, 5558–5568. [Google Scholar] [CrossRef]
- Millet, P.; Ngameni, R.; Grigoriev, S.A.; Mbemba, N.; Brisset, F.; Ranjbari, A. PEM water electrolyzers: From electrocatalysis to stack development. Int. J. Hydrogen Energy 2010, 35, 5043–5052. [Google Scholar] [CrossRef]
- Millet, P.; Mbemba, N.; Grigoriev, S.A.; Fateev, V.N.; Aukauloo, A.; Etie’vant, C. Electrochemical performances of PEM water electrolysis cells and perspectives. Int. J. Hydrogen Energy 2011, 36, 4134–4142. [Google Scholar] [CrossRef]
- Decourt, B.; Lajoie, B.; Debarre, R.; Soupa, O. The Hydrogen-Based Energy Conversion Fact Book; The SBC Energy Institute: Gravenhage, The Netherlands, 2014. [Google Scholar]
- Carmo, M.; Fritz, D.; Mergel, J.; Stolten, D. A Comprehensive review on PEM water electrolysis. Int. J. Hydrogen Energy 2013, 38, 4901–4934. [Google Scholar] [CrossRef]
- Bertuccioli, L.; Chan, A.; Hart, D.; Lehner, F.; Madden, B.; Standen, E. Study on Development of Water Electrolysis in the EU; Final Report in Fuel Cells and Hydrogen Joint Undertaking; FCH JU: Saint-Gilles, Belgium, 2014. [Google Scholar]
- Demir, M.; Dincer, I. Development of a hybrid solar thermal system with TEG and PEM electrolyzer for hydrogen and power production. Int. J. Hydrogen Energy 2017, 42, 30044–30056. [Google Scholar] [CrossRef]
- Barbir, F. PEM electrolysis for production of hydrogen from renewable energy sources. Sol. Energy 2005, 78, 661. [Google Scholar] [CrossRef]
- Lodhi, M.A. Photovoltaics and hydrogen: Future energy options. Energy Convers. Manag. 1997, 38, 1881–1893. [Google Scholar] [CrossRef]
- Yunez-Cano, A.; González-Huerta, R.D.; Tufino-Velazquez, M.; Barbosa, R.; Escobar, B. Solar-hydrogen hybrid system integrated to a sustainable house in Mexico. Int. J. Hydrogen Energy 2016, 41, 19539–19545. [Google Scholar] [CrossRef]
- Corumlu, V.; Ozsoy, A.; Ozturk, M. Thermodynamic studies of a novel heat pipe evacuated tube solar collectors based integrated process for hydrogen production. Int. J. Hydrogen Energy 2017, 43, 1060–1070. [Google Scholar] [CrossRef]
- Omar, M.; Altinisik, K. Simulation of hydrogen production system with hybrid solar collector. Int. J. Hydrogen Energy 2016, 41, 12836–12841. [Google Scholar] [CrossRef]
- Zafar, S.; Dincer, I. Thermodynamic analysis of a combined PV/T–fuel cell system for power, heat, fresh water and hydrogen production. Int. J. Hydrogen Energy 2014, 39, 9962–9972. [Google Scholar] [CrossRef]
- Oruc, M.E.; Desai, A.V.; Kenis, P.J.A.; Nuzzo, R.G. Comprehensive energy analysis of a photovoltaic thermal water electrolyzer. Appl. Energy 2016, 164, 294–302. [Google Scholar] [CrossRef]
- Ratlamwala, T.A.H.; Gadalla, M.A.; Dincer, I. Performance assessment of an integrated PV/T and triple effect cooling system for hydrogen and cooling production. Int. J. Hydrogen Energy 2011, 36, 11282–11291. [Google Scholar] [CrossRef]
- Çiloğullari, M.; Erden, M.; Karakilcik, M.; Dincer, I. Investigation of hydrogen production performance of a Photovoltaic and Thermal System. Int. J. Hydrogen Energy 2016, 42, 25447–25452. [Google Scholar]
- Hosseini, S.E.; Butler, B. Design and Analysis of a Hybrid Concentrated Photovoltaic Thermal System Integrated with an Organic Rankine Cycle for Hydrogen Production. J. Therm. Anal. Calorim. 2020, 20. [Google Scholar] [CrossRef]
- Simulink Index. Available online: https://www.mathworks.com/help/simulink/index.html (accessed on 5 April 2020).
- Duffie, J.A.; Beckman, W.A. Solar Engineering of Thermal Processes; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Radziemska, E. Thermal performance of Si and GaAs based solar cells and modules: A review. Prog. Energy Combust. Sci. 2003, 29, 407–424. [Google Scholar] [CrossRef]
- Zhang, H.; Lin, G.; Chen, J. Evaluation and calculation on the efficiency of a water electrolysis system for hydrogen production. Int. J. Hydrogen Energy 2010, 35, 10851–10858. [Google Scholar] [CrossRef]
- Siracusano, S.; Baglio, V.; Briguglio, N. An electrochemical study of a PEM stack for water electrolysis. Int. J. Hydrogen Energy 2012, 37, 1939–1946. [Google Scholar] [CrossRef]
- Singh, P.; Ravindra, N. Temperature dependence of solar cell performance—An analysis. Sol. Energy Mater. Sol. Cells 2012, 101, 36–45. [Google Scholar] [CrossRef]
- Spiegel, C. PEM Fuel Cell Modeling and Simulation Using MATLAB; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar]
- Kazim, A. Exergoeconomic analysis of a PEM electrolyser at various operating temperatures and pressures. Int. J. Energy Res. 2005, 29, 539–548. [Google Scholar] [CrossRef]
- Çengel, Y.A.; Boles, M.A. Thermodynamics: An Engineering Approach, 5th ed.; McGraw-Hill: New York, NY, USA, 2008. [Google Scholar]
- Akyuz, E.; Coskun, C.; Oktay, Z.; Dincer, I. Hydrogen production probability distributions for a PV-electrolyser system. Int. J. Hydrogen Energy 2011, 36, 292–299. [Google Scholar] [CrossRef]
- Ceicdata. Available online: https://www.ceicdata.com/en/turkey/saving-discount-rate-and-interbank-rate/central-bank-discount-rate (accessed on 21 April 2020).
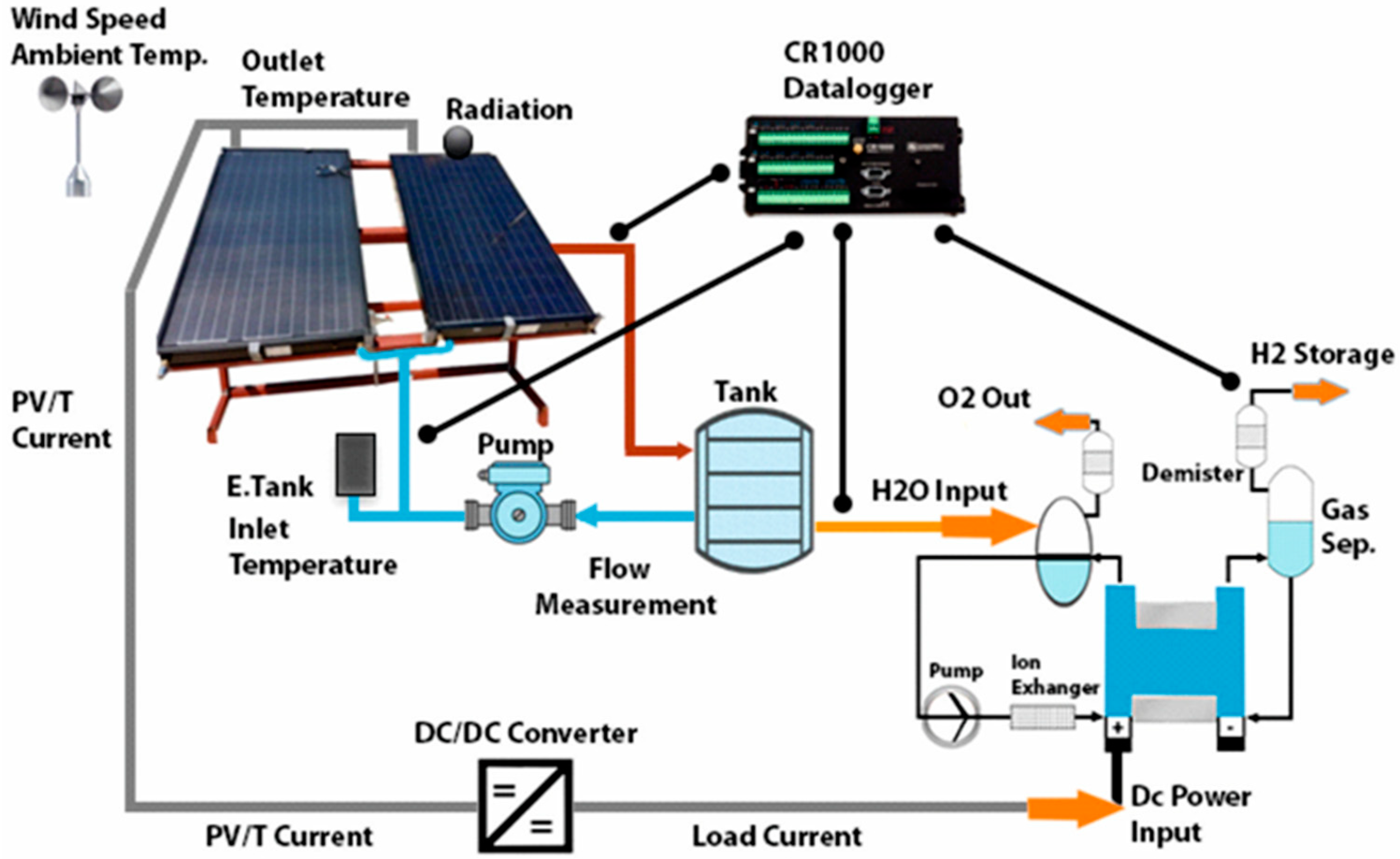








| Dimensions | 1640 × 870 × 105 mm |
|---|---|
| Maximum current (A) | 5.43 |
| Short circuit current (A) | 5.67 |
| Maximum voltage (V) | 36.8 |
| Open circuit voltage (V) | 45.43 |
| Glass | Extra Solar Glass |
| Cell number | 72 |
| Type | Monocrystal |
| Thermal power (W) | 680 |
| Active cell area (m2) | 1.222 |
| Material | Feature | Measured Variable | Accuracy |
|---|---|---|---|
| Pyranometer | Kipp and Zonen CMP11 | Global irradiance | ±2% |
| Anemometer | NRG 40 | Wind speed | ±1% |
| Temperature sensor | PT100, J type | Ambient & collector | ±0.8% |
| Current Sensor | ASC712 (0–75 A) | Current | ±0.15% |
| Flowmeter | LZB-15 SL (0–250 L/h) | Mass flow rate | ±1.5% |
| Symbols—Parameters | Values |
|---|---|
| Diameter of the copper tube (D) | 0.01 m |
| Width of the tube spacing (W) | 0.092 m |
| PV thermal conductivity (kpv) | 149 W/m K |
| PV laminate thickness (lpv) | 0.0002 m |
| Absorber thermal conductivity (kabs) | 385 W/m K |
| Absorber laminate thickness (labs) | 0.00012 m |
| Insulation material thermal conductivity (ki) | 0.04 W/m K |
| Insulation material thickness (li) | 0.05 m |
| Transmission–absorption coefficient (ταg) | 0.8 W/m K |
| Symbols—Parameters | Values |
|---|---|
| Transfer coefficient anode–cathode (αa, αc) | 0.5 |
| Anode–cathode electrode thickness (δa, δc) | 5 × 10−6 m |
| Anode exchange current densities (i0,a) | 0.0042 A/cm2 |
| Cathode exchange current density (i0,c) | 0.001 A/cm2 |
| Current density (i) | 1.6 A/cm2 |
| Operating pressure (P) | 1 bar |
| Faraday number (F) | 96,485.3 A/mol |
| Operating temperature (T) | 303 K |
| Degree of humidification (λ) | 24 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gül, M.; Akyüz, E. Hydrogen Generation from a Small-Scale Solar Photovoltaic Thermal (PV/T) Electrolyzer System: Numerical Model and Experimental Verification. Energies 2020, 13, 2997. https://doi.org/10.3390/en13112997
Gül M, Akyüz E. Hydrogen Generation from a Small-Scale Solar Photovoltaic Thermal (PV/T) Electrolyzer System: Numerical Model and Experimental Verification. Energies. 2020; 13(11):2997. https://doi.org/10.3390/en13112997
Chicago/Turabian StyleGül, Metin, and Ersin Akyüz. 2020. "Hydrogen Generation from a Small-Scale Solar Photovoltaic Thermal (PV/T) Electrolyzer System: Numerical Model and Experimental Verification" Energies 13, no. 11: 2997. https://doi.org/10.3390/en13112997
APA StyleGül, M., & Akyüz, E. (2020). Hydrogen Generation from a Small-Scale Solar Photovoltaic Thermal (PV/T) Electrolyzer System: Numerical Model and Experimental Verification. Energies, 13(11), 2997. https://doi.org/10.3390/en13112997






