Abstract
The value of gadoxetic acid in the diagnosis of hepatocellular carcinoma (HCC), based on perfusion criteria, is under dispute. This post-hoc analysis of the prospective, phase II, randomized, controlled SORAMIC study compared the accuracy of gadoxetic acid-enhanced dynamic magnetic resonance imaging (MRI) (arterial, portovenous, and venous phase only) versus contrast-enhanced computed tomography (CT) for stratifying patients with HCC to curative ablation or palliative treatment. Two reader groups (radiologists, R1 and R2) performed blind reads of CT and gadoxetic acid-enhanced MRI (contrast dynamics only). A truth panel, with access to clinical and imaging follow-up data, served as reference. Primary endpoint was non-inferiority (margin: 5% points) of MRI vs. CT (lower 95% confidence interval [CI] > 0.75) in a first step and superiority (complete 95% CI > 1) in a second step. The intent-to-treat population comprised 538 patients. Accuracy of treatment decisions was 73.4% and 70.8% for CT (R1 and R2, respectively) and 75.1% and 70.3% for gadoxetic acid-enhanced dynamic MRI. Non-inferiority but not superiority of gadoxetic acid-enhanced dynamic MRI versus CT was demonstrated (odds ratio 1.01; CI 0.97–1.05). Despite a theoretical disadvantage in wash-out depiction, gadoxetic acid-enhanced dynamic MRI is non-inferior to CT in accuracy of treatment decisions for curative ablation versus palliative strategies. This outcome was not subject to the use of additional MR standard sequences.
1. Introduction
Hepatocellular carcinoma (HCC) represents approximately 80–90% of all liver cancers [1]. In 2020, there were an estimated 52,450 new cases and 32,750 deaths from liver, intrahepatic bile duct, gallbladder, and other biliary cancer, combined, in the USA [2].
Guidelines on the recommended imaging techniques to diagnose HCC show geographical variations. For the radiological work-up of patients with suspected HCC, the Asia-Pacific guidelines recommend use of gadoxetic acid-enhanced hepatobiliary MRI (HBI) combined with dynamic imaging in the first line [3]. North American and European guidelines, by contrast, do not incorporate HBI features and focus exclusively on dynamic criteria: namely, contrast-medium enhancement in the arterial phase, with wash-out in the portovenous/venous phase [4,5]. The theoretical rationale for excluding HBI criteria in Western guidelines is that uptake of gadoxetic acid early in the venous phase (resulting in a so-called transitional phase), in the surrounding liver parenchyma, may lead to a wash-out characterization of lesions besides perfusion-related factors only and, therefore, decrease specificity [4,5]. This technical consideration has not been reproduced, so far, in a prospective trial incorporating a clinically meaningful study endpoint, such as treatment decision-making.
The prospective, phase II, randomized, controlled SORAMIC study investigated sorafenib, in combination with microtherapy, in HCC patients (EudraCT 2009-012576-27, NCT01126645). A SORAMIC substudy compared gadoxetic acid-enhanced MRI against contrast-enhanced multislice CT for the accurate stratification of patients to local ablation (i.e., curative treatment) versus palliative treatment [6]. An analysis from the SORAMIC diagnostic cohort showed that gadoxetic acid-enhanced HBI MRI, including dynamic parameters as well as hepatobiliary phase (HBP), was superior to contrast-enhanced CT for treatment decision-making [7].
The purpose of the current analyses from the SORAMIC diagnostic cohort was to compare the accuracy of gadoxetic acid-enhanced dynamic MRI against contrast-enhanced CT when applying perfusion criteria only, i.e., following current Western guidelines. We sought to determine whether the theoretical limitation of obscured wash-out, employing gadoxetic acid for dynamic MRI, has a negative impact on treatment decisions in patients with HCC employing standard criteria with arterial wash-in and portovenous/venous wash-out. Eventually, this could support the clinical use of gadoxetic acid-enhanced contrast dynamics as part of multiparametric hepatobiliary liver MRI.
2. Materials and Methods
SORAMIC is a prospective, phase II, open-label, multicentre, randomized trial conducted at 38 sites in 12 European countries. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all patients for being included in the study.
The SORAMIC diagnostic study had the primary objective to confirm, by a two-step procedure, that gadoxetic acid-enhanced HBP MRI is: (1) non-inferior or (2) superior to contrast-enhanced multislice CT for stratifying patients to palliative or local ablation strategies [7]. In this post-hoc analysis, we report the same endpoint, including identical biostatistical considerations, for the comparison of gadoxetic acid-enhanced dynamic MRI (i.e., a subset of the full HBI MRI) to contrast-enhanced CT, applying perfusion criteria only. Study methods for the SORAMIC diagnostic study are detailed in Ricke et al. [7].
Patients with confirmed HCC in Barcelona Clinic Liver Cancer (BCLC) stages A, B, and C, as well as Child–Pugh A through B7, were eligible for investigation by gadoxetic acid-enhanced MRI and contrast-enhanced CT. Allocation to the treatment strategy within the therapeutic study (curative versus palliative) was done independently at initial investigator assignment. The intent-to-treat (ITT) population included all patients undergoing both CT and MRI. The per-protocol population (PP) was defined by the absence of major image artefacts.
At baseline, patients underwent both CT and MRI within two weeks. At follow-up, patients assigned to the curative intent arm (independently at initial investigator assignment, see above) subsequently underwent MRI and CT every two months; patients in the palliative cohort did not follow a fixed imaging protocol.
The CT protocol included pre-contrast, arterial, and portovenous phases of the upper abdomen, together with venous phase, of the whole abdomen. Contrast injection speed was 4 mL/s. The trigger delay for arterial and portovenous phases was 15 s and 50 s, respectively, after the bolus attained 100 HU in the descending aorta, while venous phase assessment was 120 s after contrast medium injection. The maximum accepted slice thickness was 5 mm.
The analysed MR protocol consisted of T1-weighted gradient echo sequences (T1-w GRE) and 3D (axial, slice thickness ≤ 5 mm) pre-contrast sequences. Following that, injection of gadoxetic acid was performed via rapid hand or power injector (1.5 mL/s) at a dose of 0.025 mmol/kg body weight, followed by a 30 mL saline flush. After injection, dynamic T1-w GRE 3D sequences were acquired in the late arterial phase (start of sequence via bolus tracking, intended start of central k-space readout 15 s after bolus detected in the descending aorta), portovenous phase (start 60–70 s after contrast injection), and venous phase (120 s after contrast injection); axial, slice thickness ≤ 5 mm (Supplementary Material File S1). In order to compare the diagnostic capacity of the dynamic criteria only, any additional MR sequence, such as T2 or diffusion-weighted imaging (DWI), was not the subject of analysis.
The CT and gadoxetic acid-enhanced dynamic MRI images were assessed in blinded fashion by two reader groups with >7 years of experience in abdominal diagnostic imaging: reader group 1 (R1), comprising 1 radiologist, and reader group 2 (R2), consisting of 6 radiologists.
The truth panel consisted of a hepatologist and a radiologist at the same tertiary HCC and liver transplantation centre, both with >10 years of experience. The truth panel had access to baseline clinical data, as well as all CT and MRI images at baseline and during the first year after study inclusion. Decisions by the truth panel were made by consensus, with the inclusion of a second experienced radiologist in case no consensus was reached.
Imaging criteria for HCC diagnosis included: lesion diameter > 1 cm with the presence of arterial enhancement and wash-out (‘typical HCC’) in the dynamic image data sets. The imaging criteria for performing a local ablation were: up to 4 lesions < 5 cm and an absence of macrovascular invasion (i.e., the criteria reported in the SORAMIC study [7]). Diagnostic confidence was assessed by a four-point scale.
Statistical analysis used SAS (version 9.4, SAS Institute, Cary, NY, USA) and the R-system for statistical computing (version 3. 5.1, R Foundation, Vienna, Austria).
For determination of sample size, it was assumed that MRI and CT had accuracies of 80–85% and 80%, respectively. The non-inferiority margin of −5% points was equivalent to an odds ratio (OR) of 0.75. MRI was concluded to be non-inferior to CT, if the lower limit of the 95% confidence interval (CI) for the OR of the accuracies of MRI and CT was >0.75%, and MRI was considered to be superior to CT if the 95% CI was >1. The main efficacy variable was analysed with generalised estimated equations (GEEs) (SAS: proc genmod) and an independent working correlation matrix. A simulation study showed the non-inferiority endpoint had a power of 99.9% [7].
Quantitative variables were reported by descriptive statistics, and categorical data were expressed as absolute and relative frequencies. Forest plots depicted the ORs of accuracies in the data sets in association with confounding parameters. The Cis for ORs are two-sided and provide 95% confidence.
3. Results
Patients were recruited into the SORAMIC diagnostic study between 5 January 2011 and 19 April 2016. The ITT and PP populations included 538 and 363 patients, respectively, for the diagnostic study, as well as for the post-hoc analysis described herein (Figure 1). The majority of patients were male (87%) and Caucasian (94%), with a median age of 66 years. Baseline characteristics are described in detail in Table 1.
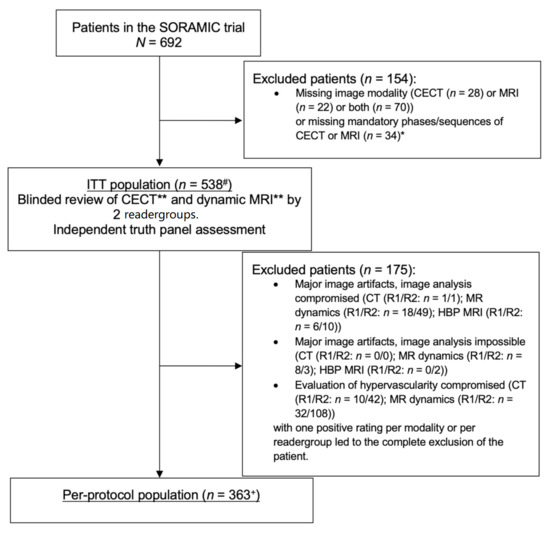
Figure 1.
Flow diagram. * Mandatory phases/sequences were arterial, portovenous, and venous phases for contrast-enhanced CT (CECT) and axial T1 3D pre-contrast, arterial phase, portovenous phase, venous phase, and hepatobiliary phase (HBP), coronal T1 3D HBP, and axial T2 turbo spin echo (TSE) with or without fat saturation for MRI. ** Imaging criteria for HCC in CECT and MRI without HBP: wash-in and wash-out. # including 91 screening failures in the therapeutic study arms of the SORAMIC trial. + including 60 screening failures in the therapeutic study arms of the SORAMIC trial.

Table 1.
Demographics and baseline characteristics.
3.1. Accuracy of Treatment Decisions
The accuracy of treatment decisions in the ITT was 75.1% and 70.3% (R1 and R2) for gadoxetic acid-enhanced dynamic MRI and 73.4% and 70.8%, respectively, for CT (Table 2). The OR for gadoxetic acid-enhanced dynamic MRI versus dynamic CT was 1.01 (0.97–1.05), therefore demonstrating non-inferiority, but not superiority, between the techniques. In the PP population, the accuracies of treatment decisions were 79.1% and 72.2% for gadoxetic acid-enhanced dynamic MRI and 76.6% and 71.6% employing CT. Relevant imaging artefacts, therefore, had no impact on the accuracy of treatment decisions. Subgroup analysis of patients with histological verification of the disease (ITT population) again revealed no difference in the accuracy of treatment decisions between gadoxetic acid-enhanced MRI (78.5% and 74.0%, R1 and R2) and CT (76.2% and 75.3%); OR 0.99 (95% CI 0.94–1.05).

Table 2.
Accuracy of treatment decision and comparison of modalities.
Results of GEE analysis, including factors with a potential influence on the accuracy of treatment decisions, are shown in Figure 2. The non-inferiority of gadoxetic acid-enhanced dynamic MRI, compared to dynamic CT, was confirmed.
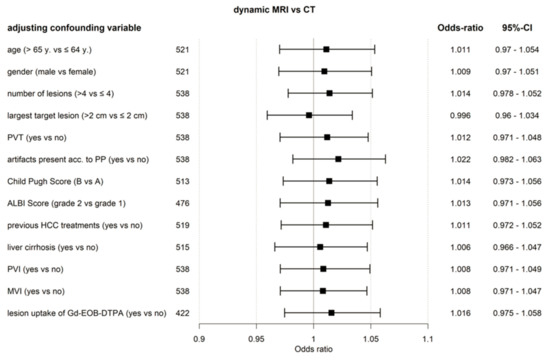
Figure 2.
Forest plot. Accuracy of the treatment decision, dynamic MRI versus CT (ITT population) based on GEE model including confounding factors. ALBI, assessment of albumin-bilirubin; CT, computed tomography; Gd-EOB-DTPA, gadoxetic acid; HCC, hepatocellular carcinoma; MRI, magnetic resonance imaging; MVI, macrovascular invasion; PP, per protocol; PVI, portal vein infiltration; PVT, portal vein thrombosis; y, year.
Interreader agreement between the reader groups was moderate for CT (Cohen’s kappa: 0.58 [95% CI 0.51–0.66], R1 vs. R2, ITT) and substantial for dynamic MRI (0.67 [95% CI 0.61–0.74]). Results for the PP population were comparable between dynamic MRI and CT at 0.73 (95% CI 0.66–0.81) and 0.61 (95% CI 0.53–0.7), respectively.
3.2. Diagnostic Confidence
Diagnostic confidence (i.e., combined percentages of ‘very confident’ and ‘confident’, ITT population) was inferior for gadoxetic acid-enhanced dynamic MRI, compared to CT for R1, and was similar for R2 (MRI: R1: 88.3%, R2: 86.9%; CT: R1: 95.7%, R2: 87.9%; OR R1: 0.3 [CI 0.2–0.6]; OR R2: 0.9 [CI 0.6–1.3]).
3.3. Detection Rate of Lesions
The dichotomized assessment of lesion number showed that CT identified 0–4 lesions in 65.8% (R1) and 62.6% (R2) of patients, compared to 67.7% (R1) and 66.5% (R2), respectively, by gadoxetic acid-enhanced dynamic MRI. Comparing gadoxetic acid-enhanced dynamic MRI and CT, the OR for correct assessment of lesion number 0–4 versus >4 was 0.9 (CI 0.7–1.1) for R1 and 0.8 (CI 0.7–1.1) for R2. Details on lesion detection rate and maximum lesion size are outlined in Table 3.

Table 3.
Lesion characteristics.
3.4. Artefacts
Image quality of CT (good or average versus poor) was statistically superior, in both reader groups, to gadoxetic acid-enhanced dynamic MRI, with 99.8/99.4% (R1/R2) assessed as good in CT and 89.2/91.9% (R1/R2) in gadoxetic acid-enhanced dynamic MRI. Comparing the presence of relevant artefacts (major artefacts compromising the analysis or making analysis impossible versus no and minor artefacts) revealed statistically fewer cases for CT, compared to gadoxetic acid-enhanced dynamic MRI, in both reader groups (0.2/0.2% [R1/R2] vs. 4.9/9.7% [R1/R2]). CT showed significantly fewer cases of incorrect timing of the arterial phase compared to gadoxetic acid-enhanced dynamic MRI in both reader groups (3.3/14.9% [R1/R2] vs. 7.8/26.8% [R1/R2]). CT delivered significantly fewer cases in which the assessment of tumour hypervascularity was compromised (1.9/7.8% [R1/R2] vs. 5.9/20.1% [R1/R2]).
The PP population was defined by the absence of major artefacts compromising the analysis. In both reader groups, CT showed significantly fewer cases meeting this definition as compared to gadoxetic acid-enhanced dynamic MRI (1.9/8.0% [R1/R2] vs. 7.6/22.1% [R1/R2]). Details of the imaging artefacts are outlined in Table 4.

Table 4.
Imaging artefacts.
3.5. Portal Vein Thrombosis and Portal Vein or Other Macrovascular Invasion
Presence of portal vein thrombosis was identified in 34.4% (R1) and 41.5% (R2) of cases by CT and in 32.3% (R1) and 42.0% (R2) by gadoxetic acid-enhanced dynamic MRI (OR 0.9 [95% CI 0.7–1.2] for R1 and 1.0 [95% CI 0.8–1.3] for R2). Portal vein invasion/macrovascular invasion were identified in 31.6/32.7% (R1) and 30.9/35.3% (R2) by CT and in 29.4/30.5% (R1) and 30.5/35.5% (R2) by gadoxetic acid-enhanced dynamic MRI (OR 0.9 [95% CI 0.7–1.2] for R1 and 1.0 [95% CI 0.8–1.3] for R2).
4. Discussion
In this post-hoc analysis of the SORAMIC diagnostic study, we describe the non-inferiority of gadoxetic acid-enhanced dynamic MRI, compared to contrast-enhanced CT for the accuracy of treatment decision-making, in patients with HCC. HCC diagnosis was based on perfusion criteria only, as determined in current Western guidelines. All assessments, therefore, were based on dynamic MR and CT imaging (for arterial, portovenous, and venous phase only). A previously reported analysis from the SORAMIC diagnostic study concluded that hepatobiliary imaging (in addition to perfusion criteria) with gadoxetic acid-enhanced MRI was superior to CT in the accuracy of treatment decisions [7]. In addition to these former results, the post hoc analysis described herein confirms the non-inferiority of gadoxetic acid-based dynamic MRI versus contrast-enhanced CT for HCC perfusion imaging.
Current western guidelines use exclusively arterial and portovenous/venous criteria on contrast-enhanced MRI, with the aim to enhance the specificity of the MR technique [4,5]. Our study considered a signal decrease of a lesion, during the portovenous or venous phase (defined as 60–70 s and 120 s after gadoxetic acid injection, respectively), as positive for wash-out, when assessing the dynamic image data set of gadoxetic acid, and is in line with the recommendations in Western guidelines when applying extracellular MR contrast media [5,8]. However, the value of gadoxetic acid for HCC diagnosis based on perfusion criteria, and the criteria to be included, are under dispute, since early liver uptake of gadoxetic acid may obscure the depiction of true lesion wash-out, which led to a renaming of the venous phase to the transitional phase in gadoxetic acid liver MRI [4,9]. Retrospective studies have reported high sensitivity but a reduction in specificity when wash-out in the portovenous phase is combined with hypointensity in the transitional phase, measured 3 min after injection (decrease in sensitivity: from 97.9% to 86.3% [10], 92.9% to 78.6% [11], and 100% to 94.9% [12], while two other retrospective studies have interpreted that extending the wash-out appearance to transitional phase or HBP, rather than portovenous phase alone, allows high sensitivity without significant reduction in specificity (decrease in specificity: from 94.1% to 82.0% [13] and 90.9% to 84.8% [14]), in support of our findings.
Numerous studies have compared gadoxetic acid-enhanced HBI MRI versus contrast-enhanced CT in HCC, with the conclusion that the former technique consistently provides superior lesion detection [13,15,16,17,18]. There is, however, a paucity of data that directly compare gadoxetic acid-enhanced dynamic MRI against CT, without addition of further MR sequences such as T2 and DWI. A recent study by Semaan et al. reported per-lesion sensitivities for HCC detection of 59.5% for CT versus 69.7% for gadoxetic acid-enhanced dynamic MRI, which were both lower than 76.8% for gadoxetic acid-enhanced MRI, including HBI [19]. To the best of our knowledge, our study is the first to relate dynamic contrast-enhanced MRI to a clinical decision endpoint.
Uptake of gadoxetic acid in the late phase is seen in approximately 10% of HCC lesions [20]. Theoretically, this uptake, potentially already starting in the transitional phase, can pose a bias in the evaluation of venous wash-out in gadoxetic acid-enhanced liver MRI. However, lesion uptake of gadoxetic acid in the late phase was not a significant confounding factor in our analysis (Figure 2).
Gadoxetic acid-enhanced dynamic MRI appeared to perform independently of a number of influencing factors (Figure 2). Compromised imaging in the arterial phase was reported in up to 20.1% patients (reader group 2) in the gadoxetic acid group (a sum of incorrect timing and movement artefacts), in the majority of cases most probably related to transient severe motion (TSM). Similar or lower rates of TSM have been reported in the literature [21,22]. Although patients showed significantly more imaging artefacts with gadoxetic acid-enhanced MRI than CT, treatment decision-making was not impaired in both the ITT and PP populations.
Trial limitations include the lack of histopathology to confirm the truth panel assessment in approximately one-half of patients. We consider, however, that potential bias should not be a concern, in view of the large data sets available before and after treatment. Secondly, more advanced tumour stages were more frequent than earlier tumour stages in our trial. However, a higher rate of advanced tumour stages reflects real life in HCC diagnosis [5]. Thirdly, the inclusion criteria in SORAMIC do not reflect the current guidelines for local ablation. However, we propose that these expanded criteria for ablation, regarding size and number of lesions, do not affect the study outcomes, which result from appropriate lesion identification. Our study does not include the LI-RADS classification. LI-RADS was adopted in the AASLD guidelines in 2018 [8], and further validation is ongoing, most likely leading to a powerful tool for standardized HCC diagnosis. However, at protocol development and Statistical Analysis Plan generation, LI-RADS had not yet been established. LI-RADS, therefore, was not included in our blind read. We suggest LI-RADS would not add benefit to our analysis since it comprises perfusion criteria only. T2 sequences, as well as DWI today, are part of the recommended MR protocols [4,8]. These sequences were not part of the study hypothesis. Inclusion of T2 sequences and DWI would likely have improved the performance of MR in our study.
5. Conclusions
Gadoxetic acid-enhanced dynamic MRI (employing arterial, portovenous, and venous phase) was shown to be non-inferior to CT in the accuracy of treatment decisions for curative ablation versus palliative strategies. Information on gadoxetic acid-enhanced MRI contrast dynamics may, therefore, be used for HCC diagnosis, based on perfusion criteria, supporting the use of gadoxetic acid-enhanced multiparametric liver MRI in the diagnosis and surveillance of HCC.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/curroncol29020051/s1, Supplementary File S1: Gadoxetic acid MRI protocol.
Author Contributions
Conceptualization, M.S. and J.R.; methodology, M.S., I.G.S. and J.R.; formal analysis and investigation, M.S., I.G.S. and J.R.; writing—original draft preparation, M.S. and J.R.; writing—review and editing, I.G.S., I.B., T.B., A.B., B.G., R.I., C.L., M.K., M.P., C.S., O.v.D., V.V. and C.J.Z.; supervision, M.S. and J.R.; funding acquisition, J.R. All authors have read and agreed to the published version of the manuscript.
Funding
SORAMIC is an investigator-initiated trial sponsored by the University of Magdeburg. Financial support was granted by Sirtex Medical and Bayer Healthcare.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Boards of all participating centers.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data are available from the corresponding author on reasonable request.
Conflicts of Interest
Max Seidensticker reports grants and personal fees from Sirtex, grants and personal fees from Bayer Healthcare, personal fees from Siemens, Cook, Boston Scientific, Astra Zeneca. Ingo G. Steffen reports nothing to disclose. Irene Bargellini reports grants from BTG Ltd, personal fees from BTG Ltd, Sirtex Medical, Terumo Europe, Ge Healthcare, Boston Scientific, EISAI, Guerbet, SOBI and Bayer HealthcareThomas Berg currently acts as an advisor to Abbvie, Alexion, Bayer, BMS, Gilead, Intercept, Janssen, MSD/Merck, Merz, Novartis, and Sequana Medical. He has received speaking honoraria from Abbvie, Alexion, Bayer, BMS, Eisai, Gilead, Intercept, Ipsen, Janssen, MSD/Merck, Merz, Novartis, Sirtex and Sequana Medical in the past 2 years. He has received grant support from Abbvie, BMS, Gilead, Humedics, Intercept, Janssen, MSD/Merck, Merz, Novartis, and Sequana MedicalJosé. Alberto Benito reports nothing to disclose. Bernhard Gebauer reports personal fees and non-financial support from Sirtex Medical, personal fees and non-financial support from Bayer Healthcare; personal fees from Sirtex Medical, COOK, Pfizer, Merck, MSD, ICON, PAREXEL, BD/CR BARD, Ewimed, Pharmcept, ROCHE, Terumo, Guerbet, INARI, Elsai. Roberto Iezzi reports grants and personal fees from Sirtex Medical, Terumo Europe and Boston Scientific. Christian Loewe reports nothing to disclose. Musturay Karçaaltincaba reports personal fees from Bayer, personal fees from GE Healthcare, personal fees from Pfizer. Maciej Pech reports grants from Bayer Healthcare, grants and personal fees from Sirtex. Christian Sengel reports nothing to disclose. Otto van Delden reports nothing to disclose. Vincent Vandecaveye reports nothing to disclose. Christoph J. Zech reports grants and personal fees from Bayer Healthcare. Jens Ricke reports grants and personal fees from Sirtex Medical, grants and personal fees from Bayer Heathcare.
References
- Perz, J.F.; Armstrong, G.L.; Farrington, L.A.; Hutin, Y.J.; Bell, B.P. The Contributions of Hepatitis B Virus and Hepatitis C Virus Infections to Cirrhosis and Primary Liver Cancer Worldwide. J. Hepatol. 2006, 45, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Recio-Boiles, A.; Hoilat, G.J.; Smith, M.; Babiker, H.M. Hepatobiliary Tract Cancer; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Omata, M.; Cheng, A.L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific Clinical Practice Guidelines on the Management of Hepatocellular Carcinoma: A 2017 Update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LI-RADS. CT/MRI LI-RADS v2018. Available online: https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS/CT-MRI-LI-RADS-v2018 (accessed on 5 August 2021).
- European Association for the Study of the Liver EASL Clinical Practice Guidelines: Management of Hepatocellular Carcinoma. J. Hepatol. 2018, 69, 182–236. [CrossRef] [PubMed] [Green Version]
- Ricke, J.; Klümpen, H.J.; Amthauer, H.; Bargellini, I.; Bartenstein, P.; de Toni, E.N.; Gasbarrini, A.; Pech, M.; Peck-Radosavljevic, M.; Popovič, P.; et al. Impact of Combined Selective Internal Radiation Therapy and Sorafenib on Survival in Advanced Hepatocellular Carcinoma. J. Hepatol. 2019, 71, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Ricke, J.; Steffen, I.G.; Bargellini, I.; Berg, T.; Bilbao Jaureguizar, J.I.; Gebauer, B.; Iezzi, R.; Loewe, C.; Karçaaltincaba, M.; Pech, M.; et al. Gadoxetic Acid-Based Hepatobiliary MRI in Hepatocellular Carcinoma. JHEP Rep. Innov. Hepatol. 2020, 2, 100173. [Google Scholar] [CrossRef] [PubMed]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD Guidelines for the Treatment of Hepatocellular Carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fowler, K.J.; Sirlin, C.B. Is It Time to Expand the Definition of Washout Appearance in LI-RADS? Radiology 2019, 291, 658–659. [Google Scholar] [CrossRef] [PubMed]
- Joo, I.; Lee, J.M.; Lee, D.H.; Jeon, J.H.; Han, J.K.; Choi, B.I. Noninvasive Diagnosis of Hepatocellular Carcinoma on Gadoxetic Acid-Enhanced MRI: Can Hypointensity on the Hepatobiliary Phase Be Used as an Alternative to Washout? Eur. Radiol. 2015, 25, 2859–2868. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.; Lee, J.M.; Shin, C.I.; Lee, E.S.; Yoon, J.H.; Joo, I.; Kim, S.H.; Hwang, I.; Han, J.K.; Choi, B.I. Differentiation of Intrahepatic Mass-Forming Cholangiocarcinoma from Hepatocellular Carcinoma on Gadoxetic Acid-Enhanced Liver MR Imaging. Eur. Radiol. 2016, 26, 1808–1817. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Lee, S.S.; Kim, S.Y.; Park, S.H.; Park, S.H.; Kim, K.M.; Hong, S.M.; Yu, E.; Lee, M.G. Intrahepatic Cholangiocarcinoma in Patients with Cirrhosis: Differentiation from Hepatocellular Carcinoma by Using Gadoxetic Acid-Enhanced MR Imaging and Dynamic CT. Radiology 2017, 282, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Choi, S.H.; Kim, S.Y.; Kim, M.J.; Lee, S.S.; Byun, J.H. Gadoxetic Acid-Enhanced MRI of Hepatocellular Carcinoma: Value of Washout in Transitional and Hepatobiliary Phases. Radiology 2019, 292, 270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, K.A.; Kim, S.S.; Shin, H.C.; Hwang, J.A.; Choi, S.Y.; Lee, W.H.; Park, C.H.; Lee, H.N.; Heo, N.H. Gadoxetic Acid-Enhanced MRI for Diagnosis of Hepatocellular Carcinoma in Patients with Chronic Liver Disease: Can Hypointensity on the Late Portal Venous Phase Be Used as an Alternative to Washout? Abdom. Radiol. N. Y. 2020, 45, 2705–2716. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Lee, J.M.; Lee, J.S.; Lee, H.Y.; Park, B.H.; Kim, Y.H.; Han, J.K.; Choi, B.I. Hepatocellular Carcinoma: Diagnostic Performance of Multidetector CT and MR Imaging-a Systematic Review and Meta-Analysis. Radiology 2015, 275, 97–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Zou, L.; Liu, F.; Zhou, Y.; Song, B. Gadoxetic Acid Disodium-Enhanced Magnetic Resonance Imaging for the Detection of Hepatocellular Carcinoma: A Meta-Analysis. PLoS ONE 2013, 8, e70896. [Google Scholar] [CrossRef] [Green Version]
- Haradome, H.; Grazioli, L.; Tinti, R.; Morone, M.; Motosugi, U.; Sano, K.; Ichikawa, T.; Kwee, T.C.; Colagrande, S. Additional Value of Gadoxetic Acid-DTPA-Enhanced Hepatobiliary Phase MR Imaging in the Diagnosis of Early-Stage Hepatocellular Carcinoma: Comparison with Dynamic Triple-Phase Multidetector CT Imaging. J. Magn. Reson. Imaging 2011, 34, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, M.; Marin, D.; Guerrisi, A.; Baski, M.; Galati, F.; Rossi, M.; Brozzetti, S.; Masciangelo, R.; Passariello, R.; Catalano, C. Intraindividual Comparison of Gadoxetate Disodium-Enhanced MR Imaging and 64-Section Multidetector CT in the Detection of Hepatocellular Carcinoma in Patients with Cirrhosis. Radiology 2010, 256, 806–816. [Google Scholar] [CrossRef] [PubMed]
- Semaan, S.; Vietti Violi, N.; Lewis, S.; Chatterji, M.; Song, C.; Besa, C.; Babb, J.S.; Fiel, M.I.; Schwartz, M.; Thung, S.; et al. Hepatocellular Carcinoma Detection in Liver Cirrhosis: Diagnostic Performance of Contrast-Enhanced CT vs. MRI with Extracellular Contrast vs. Gadoxetic Acid. Eur. Radiol. 2020, 30, 1020–1030. [Google Scholar] [CrossRef] [PubMed]
- Erra, P.; Puglia, M.; Ragozzino, A.; Maurea, S.; Liuzzi, R.; Sabino, G.; Barbuto, L.; Cuocolo, A.; Imbriaco, M. Appearance of Hepatocellular Carcinoma on Gadoxetic Acid-enhanced Hepato-biliary Phase MR Imaging: A Systematic Review. Radiol. Med. 2015, 120, 1002–1011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davenport, M.S.; Caoili, E.M.; Kaza, R.K.; Hussain, H.K. Matched Within-Patient Cohort Study of Transient Arterial Phase Respiratory Motion-Related Artifact in MR Imaging of the Liver: Gadoxetate Disodium versus Gadobenate Dimeglumine. Radiology 2014, 272, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Well, L.; Weinrich, J.M.; Adam, G.; Bannas, P. Transient Severe Respiratory Motion Artifacts After Application of Gadoxetate Disodium: What We Currently Know. Rofo 2018, 190, 20–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).