Neuroprotective Properties of Chitosan and Its Derivatives
Abstract
:1. Introduction
2. Suppressing Effect on β-Amyloid Formation
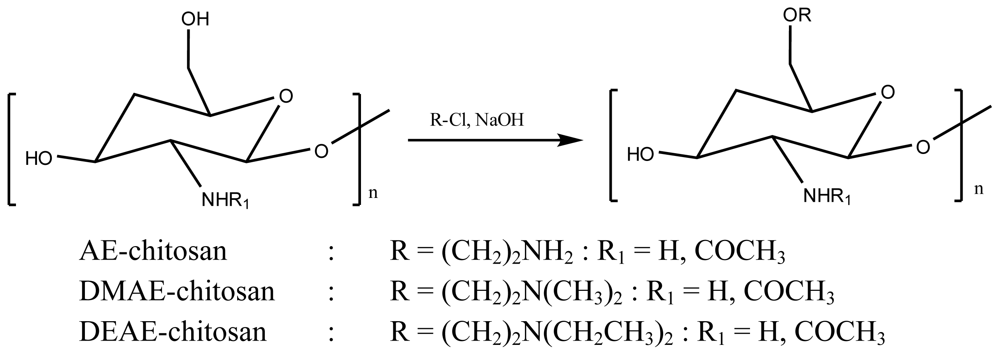
3. Acetylcholinesterase Inhibitory Activity
4. Anti-Neuroinflammatory
5. Apoptosis Inhibitors
6. Other Activities
7. Conclusions
Acknowledgement
- Samples Availability: Available from the authors.
References
- Mundkur, N. Neuroplasticity in children. Indian J. Pediatr 2005, 72, 855–857. [Google Scholar]
- Montgomery, PC; Connolly, BH; Bohmert, JA. Impaired motor function and sensory integrity associated with nonprogressive disorders of the central nervous system–congenital origin or acquired in infancy or childhood (Pattern C). In Neuromuscular Essentials Applying the Preferred Physical Therapist Practice Patterns; SLACK Incorporated: Thorofare, NJ, USA, 2008; pp. 43–70. [Google Scholar]
- Hosoi, R; Kashiwagi, Y; Hatazawa, J; Gee, A; Inoue, O. Glial metabolic dysfunction caused neural damage by short-term ischemia in brain. Ann. Nucl. Med 2006, 20, 377–380. [Google Scholar]
- Allen, NJ; Barres, BA. Neuroscience: Glia more than just brain glue. Nature 2009, 457, 675–677. [Google Scholar]
- Yuan, J; Yankner, BA. Apoptosis in the nervous system. Nature 2000, 407, 802–809. [Google Scholar]
- Bjorklund, A; Lindvall, O. Cell replacement therapies for central nervous system disorders. Nat. Neurosci 2000, 3, 537–544. [Google Scholar]
- Nakatomi, H; Kuriu, T; Okabe, S; Yamamoto, S; Hatano, O; Kawahara, N; Tamura, A; Kirino, T; Nakafuku, M. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell 2002, 110, 429–441. [Google Scholar]
- Zarros, A. In which cases is neuroprotection useful? Adv. Altern. Thinking Neurosci 2009, 1, 3–5. [Google Scholar]
- Tucci, P; Bagetta, G. How to study neuroprotection? Cell Death Differ 2008, 15, 1084–1085. [Google Scholar]
- Pellicciari, R; Costantino, G; Marinozzi, M; Natalini, B. Modulation of glutamate receptor pathways in the search for new neuroprotective agents. Il Farmaco 1998, 53, 255–261. [Google Scholar]
- Behl, C; Moosmann, B. Antioxidant neuroprotection in Alzheimer’s disease as preventive and therapeutic approach. Free Radic. Biol. Med 2002, 33, 182–191. [Google Scholar]
- Agnello, D; Bigini, P; Villa, P; Mennini, T; Cerami, A; Brines, ML; Ghezzi, P. Erythropoietin exerts an anti-inflammatory effect on the CNS in a model of experimental autoimmune encephalomyelitis. Brain Res 2002, 952, 128–134. [Google Scholar]
- Gao, H-M; Liu, B; Zhang, W; Hong, J-S. Novel anti-inflammatory therapy for Parkinson’s disease. Trends Pharmacol. Sci 2003, 24, 395–401. [Google Scholar]
- Volbracht, C; van Beek, J; Zhu, C; Blomgren, K; Leist, M. Neuroprotective properties of memantine in different in vitro and in vivo models of excitotoxicity. Eur. J. Neurosci 2006, 23, 2611–2622. [Google Scholar]
- Yu, X; An, L; Wang, Y; Zhao, H; Gao, C. Neuroprotective effect of Alpinia oxyphylla Miq. fruits against glutamate-induced apoptosis in cortical neurons. Toxicol. Lett 2003, 144, 205–212. [Google Scholar]
- Kietzmann, T; Knabe, W; Schmidt-Kastner, R. Hypoxia and hypoxia-inducible factor modulated gene expression in brain: involvement in neuroprotection and cell death. Eur. Arch. Psychiatry Clin. Neurosci 2001, 251, 170–178. [Google Scholar]
- Heurteaux, C; Guy, N; Laigle, C; Blondeau, N; Duprat, F; Mazzuca, M; Lang-Lazdunski, L; Widmann, C; Zanzouri, M; Romey, G; Lazdunski, M. TREK-1, a K+ channel involved in neuroprotection and general anesthesia. EMBO J 2004, 23, 2684–2695. [Google Scholar]
- Schwartz, G; Fehlings, MG. Evaluation of the neuroprotective effects of sodium channel blockers after spinal cord injury: improved behavioral and neuroanatomical recovery with riluzole. J. Neurosurg.: Spine 2001, 94, 245–256. [Google Scholar]
- Youdim, MBH; Fridkin, M; Zheng, H. Novel bifunctional drugs targeting monoamine oxidase inhibition and iron chelation as an approach to neuroprotection in Parkinson’s disease and other neurodegenerative diseases. J. Neural Transm 2004, 111, 1455–1471. [Google Scholar]
- Gaeta, A; Hider, RC. The crucial role of metal ions in neurodegeneration: the basis for a promising therapeutic strategy. Br. J. Pharmacol 2005, 146, 1041–1059. [Google Scholar]
- Tremblay, R; Hewitt, K; Lesiuk, H; Mealing, G; Morley, P; Durkin, JP. Evidence that Brain-Derived Neurotrophic Factor Neuroprotection Is Linked to Its Ability to Reverse the NMDA-Induced Inactivation of Protein Kinase C in Cortical Neurons. J. Neurochem 1999, 72, 102–111. [Google Scholar]
- Moalem, G; Gdalyahu, A; Shani, Y; Otten, U; Lazarovici, P; Cohen, IR; Schwartz, M. Production of Neurotrophins by Activated T Cells: Implications for Neuroprotective Autoimmunity. J. Autoimmun 2000, 15, 331–345. [Google Scholar]
- Akerud, P; Canals, JM; Snyder, EY; Arenas, E. Neuroprotection through delivery of glial cell line-derived neurotrophic factor by neural stem cells in a mouse model of Parkinson’s disease. J. Neurosci 2001, 21, 8108–8118. [Google Scholar]
- Woo, M-S; Park, J-S; Choi, I-Y; Kim, W-K; Kim, H-S. Inhibition of MMP-3 or -9 suppresses lipopolysaccharide-induced expression of proinflammatory cytokines and iNOS in microglia. J. Neurochem 2008, 106, 770–780. [Google Scholar]
- Chandrasekaran, K; Mehrabian, Z; Spinnewyn, B; Chinopoulos, C; Drieu, K; Fiskum, G. Neuroprotective Effects of Bilobalide, a Component of Ginkgo biloba Extract (EGb 761®) in Global Brain Ischemia and in Excitotoxicity-induced Neuronal Death. Pharmacopsychiatry 2003, 36, 89–94. [Google Scholar]
- Narang, S; Gibson, D; Wasan, AD; Ross, EL; Michna, E; Nedeljkovic, SS; Jamison, RN. Efficacy of Dronabinol as an Adjuvant Treatment for Chronic Pain Patients on Opioid Therapy. J. Pain 2008, 9, 254–264. [Google Scholar]
- Alonso, D; Castro, A; Martinez, A. Marine compounds for the therapeutic treatment of neurological disorders. Expert Opin. Ther. Patents 2005, 15, 1377–1386. [Google Scholar]
- Je, J-Y; Kim, S-K. Water-soluble chitosan derivatives as a BACE1 inhibitor. Bioorg. Med. Chem 2005, 13, 6551–6555. [Google Scholar]
- Kim, S-K; Rajapakse, N. Enzymatic production and biological activities of chitosan oligosaccharides (COS): A review. Carbohydr. Polym 2005, 62, 357–368. [Google Scholar]
- Ravi Kumar, MNV. A review of chitin and chitosan applications. React. Funct. Polym 2000, 46, 1–27. [Google Scholar]
- Jeon, Y-J; Park, P-J; Kim, S-K. Antimicrobial effect of chitooligosaccharides produced by bioreactor. Carbohydr. Polym 2001, 44, 71–76. [Google Scholar]
- Jeon, Y-J; Shahidi, F; Kim, S-K. Preparation of chitin and chitosan oligomers and their applications in physiological functional foods. Food Rev. Int 2000, 16, 159–176. [Google Scholar]
- Turan, K; Nagata, K. Chitosan-DNA nanoparticles: the effect of cell type and hydrolysis of chitosan on in vitro DNA transfection. Pharm. Dev. Technol 2006, 11, 503–512. [Google Scholar]
- Prabaharan, M. Review paper: chitosan derivatives as promising materials for controlled drug delivery. J. Biomater. Appl 2008, 23, 5–36. [Google Scholar]
- Jeon, Y-J; Kim, S-K. Production of chitooligosaccharides using an ultrafiltration membrane reactor and their antibacterial activity. Carbohydr. Polym 2000, 41, 133–141. [Google Scholar]
- Suzuki, K; Mikami, T; Okawa, Y; Tokoro, A; Suzuki, S; Suzuki, M. Antitumor effect of hexa-N-acetylchitohexaose and chitohexaose. Carbohydr. Res 1986, 151, 403–408. [Google Scholar]
- Je, J-Y; Park, P-J; Kim, S-K. Free radical scavenging properties of hetero-chitooligosaccharides using an ESR spectroscopy. Food Chem. Toxicol 2004, 42, 381–387. [Google Scholar]
- Rajapakse, N; Kim, M-M; Mendis, E; Huang, R; Kim, S-K. Carboxylated chitooligosaccharides (CCOS) inhibit MMP-9 expression in human fibrosarcoma cells via down-regulation of AP-1. Biochimica et Biophysica Acta (BBA)-General Subjects 2006, 1760, 1780–1788. [Google Scholar]
- Kim, M-M; Kim, S-K. Chitooligosaccharides inhibit activation and expression of matrix metalloproteinase-2 in human dermal fibroblasts. FEBS Lett 2006, 580, 2661–2666. [Google Scholar]
- Van Ta, Q; Kim, M-M; Kim, S-K. Inhibitory Effect of Chitooligosaccharides on Matrix Metalloproteinase-9 in Human Fibrosarcoma Cells (HT1080). Mar. Biotechnol 2006, 8, 593–599. [Google Scholar]
- Liu, B; Liu, W; Han, B; Sun, Y. Antidiabetic effects of chitooligosaccharides on pancreatic islet cells in streptozotocin-induced diabetic rats. World J. Gastroenterol 2007, 13, 725. [Google Scholar]
- Artan, M; Karadeniz, F; Karagozlu, MZ; Kim, M-M; Kim, S-K. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr. Res 2010, 345, 656–662. [Google Scholar]
- Yang, E-J; Kim, J-G; Kim, J-Y; Kim, S; Lee, N; Hyun, C-G. Anti-inflammatory effect of chitosan oligosaccharides in RAW 264.7 cells. Cent. Eur. J. Biol 2010, 5, 95–102. [Google Scholar]
- Liu, D; Hsieh, J; Fan, X; Yang, J; Chung, T. Synthesis, characterization and drug delivery behaviors of new PCP polymeric micelles. Carbohydr. Polym 2007, 68, 544–554. [Google Scholar]
- Jayakumar, R; Nagahama, H; Furuike, T; Tamura, H. Synthesis of phosphorylated chitosan by novel method and its characterization. Int. J. Biol. Macromol 2008, 42, 335–339. [Google Scholar]
- LaFerla, FM; Green, KN; Oddo, S. Intracellular amyloid-[beta] in Alzheimer’s disease. Nat. Rev. Neurosci 2007, 8, 499–509. [Google Scholar]
- Agdeppa, ED; Kepe, V; Liu, J; Flores-Torres, S; Satyamurthy, N; Petric, A; Cole, GM; Small, GW; Huang, SC; Barrio, JR. Binding characteristics of radiofluorinated 6-dialkylamino-2-naphthylethylidene derivatives as positron emission tomography imaging probes for beta-amyloid plaques in Alzheimer’s disease. J. Neurosci 2001, 21, RC189. [Google Scholar]
- Lukiw, WJ. Emerging amyloid beta (Ab) peptide modulators for the treatment of Alzheimer’s disease (AD). Expert Opin. Emerging Drugs 2008, 13, 255–271. [Google Scholar]
- Okamura, N; Suemoto, T; Shiomitsu, T; Suzuki, M; Shimadzu, H; Akatsu, H; Yamamoto, T; Arai, H; Sasaki, H; Yanai, K; Staufenbiel, M; Kudo, Y; Sawada, T. A novel imaging probe for in vivo detection of neuritic and diffuse amyloid plaques in the brain. J. Mol. Neurosci 2004, 24, 247–255. [Google Scholar]
- Vassar, R. [beta]-Secretase (BACE) as a drug target for alzheimer’s disease. Adv. Drug Delivery Rev 2002, 54, 1589–1602. [Google Scholar]
- Hampel, H; Shen, Y. Beta-site amyloid precursor protein cleaving enzyme 1 (BACE1) as a biological candidate marker of Alzheimer’s disease. Scand. J. Clin. Lab. Invest 2009, 69, 8–12. [Google Scholar]
- Tang, K; Hynan, LS; Baskin, F; Rosenberg, RN. Platelet amyloid precursor protein processing: A bio-marker for Alzheimer’s disease. J. Neurol. Sci 2006, 240, 53–58. [Google Scholar]
- Byun, H-G; Kim, Y-T; Park, P-J; Lin, X; Kim, S-K. Chitooligosaccharides as a novel [beta]-secretase inhibitor. Carbohydr. Polym 2005, 61, 198–202. [Google Scholar]
- Khodagholi, F; Eftekharzadeh, B; Maghsoudi, N; Rezaei, P. Chitosan prevents oxidative stress-induced amyloid β formation and cytotoxicity in NT2 neurons: involvement of transcription factors Nrf2 and NF-κB. Mol. Cell. Biochem 2010, 337, 39–51. [Google Scholar]
- Tamagno, E; Bardini, P; Obbili, A; Vitali, A; Borghi, R; Zaccheo, D; Pronzato, MA; Danni, O; Smith, MA; Perry, G; Tabaton, M. Oxidative Stress Increases Expression and Activity of BACE in NT2 Neurons. Neurobiol. Dis 2002, 10, 279–288. [Google Scholar]
- Tabet, N. Acetylcholinesterase inhibitors for Alzheimer’s disease: anti-inflammatories in acetylcholine clothing! Age Ageing 2006, 35, 336–338. [Google Scholar]
- Terry, AV, Jr; Buccafusco, JJ. The cholinergic hypothesis of age and Alzheimer’s disease-related cognitive deficits: recent challenges and their implications for novel drug development. J. Pharmacol. Exp. Ther 2003, 306, 821–827. [Google Scholar]
- Martinez, A; Castro, A. Novel cholinesterase inhibitors as future effective drugs for the treatment of Alzheimer’s disease. Expert Opin. Invest. Drugs 2005, 15, 1–12. [Google Scholar]
- Ibrahim, F; André, C; Thomassin, M; Guillaume, Y-C. Association mechanism of four acetylcholinesterase inhibitors (AChEIs) with human serum albumin: A biochromatographic approach. J. Pharm. Biomed. Anal 2008, 48, 1345–1350. [Google Scholar]
- Lee, S-H; Park, J-S; Kim, S-K; Ahn, C-B; Je, J-Y. Chitooligosaccharides suppress the level of protein expression and acetylcholinesterase activity induced by A[beta]25–35 in PC12 cells. Bioorg. Med. Chem. Lett 2009, 19, 860–862. [Google Scholar]
- Yoon, NY; Ngo, D-N; Kim, S-K. Acetylcholinesterase inhibitory activity of novel chitooligosaccharide derivatives. Carbohydr. Polym 2009, 78, 869–872. [Google Scholar]
- Kim, YS; Joh, TH. Microglia, major player in the brain inflammation: their roles in the pathogenesis of Parkinson’s disease. Exp. Mol. Med 2006, 38, 333–347. [Google Scholar]
- Aktas, O; Ullrich, O; Infante-Duarte, C; Nitsch, R; Zipp, F. Neuronal Damage in Brain Inflammation. Arch. Neurol 2007, 64, 185–189. [Google Scholar]
- Kim, M-S; Sung, M-J; Seo, S-B; Yoo, S-J; Lim, W-K; Kim, H-M. Water-soluble chitosan inhibits the production of pro-inflammatory cytokine in human astrocytoma cells activated by amyloid [beta] peptide and interleukin-1[beta]. Neurosci. Lett 2002, 321, 105–109. [Google Scholar]
- Kalmar, B; Greensmith, L. Induction of heat shock proteins for protection against oxidative stress. Adv. Drug Deliv. Rev 2009, 61, 310–318. [Google Scholar]
- Twomey, C; McCarthy, J. Pathways of apoptosis and importance in development. J. Cell. Mol. Med 2005, 9, 345. [Google Scholar]
- Fadeel, B; Orrenius, S. Apoptosis: a basic biological phenomenon with wide-ranging implications in human disease. Apoptosis 2005, 479, 517. [Google Scholar]
- Vila, M; Przedborski, S. Targeting programmed cell death in neurodegenerative diseases. Nat. Rev. Neurosci 2003, 4, 365–375. [Google Scholar]
- Koo, HN; Jeong, HJ; Hong, SH; Choi, JH; An, NH; Kim, HM. High molecular weight water-soluble chitosan protects against apoptosis induced by serum starvation in human astrocytes. J. Nutr. Biochem 2002, 13, 245–249. [Google Scholar]
- Zhou, S; Yang, Y; Gu, X; Ding, F. Chitooligosaccharides protect cultured hippocampal neurons against glutamate-induced neurotoxicity. Neurosci. Lett 2008, 444, 270–274. [Google Scholar]
- Cho, Y; Shi, R; Ben Borgens, R. Chitosan nanoparticle-based neuronal membrane sealing and neuroprotection following acrolein-induced cell injury. J. Biol. Eng 2010, 4, 2. [Google Scholar]
- Aktas, Y; Yemisci, M; Andrieux, K; Gursoy, RN; Alonso, MJ; Fernandez-Megia, E; Novoa-Carballal, R; Quinoa, E; Riguera, R; Sargon, MF; Celik, HH; Demir, AS; Hincal, AA; Dalkara, T; Capan, Y; Couvreur, P. Development and brain delivery of chitosan-PEG nanoparticles functionalized with the monoclonal antibody OX26. Bioconjug. Chem 2005, 16, 1503–1511. [Google Scholar]
| COS | IC50 | Ref |
|---|---|---|
| 90-HMWCOS | 2.59 mg/mL | [60] |
| 90-MMWCOS | 1.67 mg/mL | [60] |
| 90-LMWCOS | 3.52 mg/mL | [60] |
| 50-HMWCOS | 1.98 mg/mL | [60] |
| 50-MMWCOS | 2.93 mg/mL | [60] |
| 50-LMWCOS | >4.00 mg/mL | [60] |
| AE-COS | 56.5 ± 0.26 μg/mL | [61] |
| DMAE-COS | 24.1 ± 0.39 μg/mL | [61] |
| DEAE-COS | 9.2 ± 0.33 μg/mL | [61] |
| Eserinea | 0.0089 ± 0.00005 μg/mL | [61] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pangestuti, R.; Kim, S.-K. Neuroprotective Properties of Chitosan and Its Derivatives. Mar. Drugs 2010, 8, 2117-2128. https://doi.org/10.3390/md8072117
Pangestuti R, Kim S-K. Neuroprotective Properties of Chitosan and Its Derivatives. Marine Drugs. 2010; 8(7):2117-2128. https://doi.org/10.3390/md8072117
Chicago/Turabian StylePangestuti, Ratih, and Se-Kwon Kim. 2010. "Neuroprotective Properties of Chitosan and Its Derivatives" Marine Drugs 8, no. 7: 2117-2128. https://doi.org/10.3390/md8072117
APA StylePangestuti, R., & Kim, S.-K. (2010). Neuroprotective Properties of Chitosan and Its Derivatives. Marine Drugs, 8(7), 2117-2128. https://doi.org/10.3390/md8072117





