cDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis)
Abstract
:1. Introduction
2. Results and Discussion
2.1. cDNA Sequence of Mytilec-1 and Virtual Translation of the Polypeptide
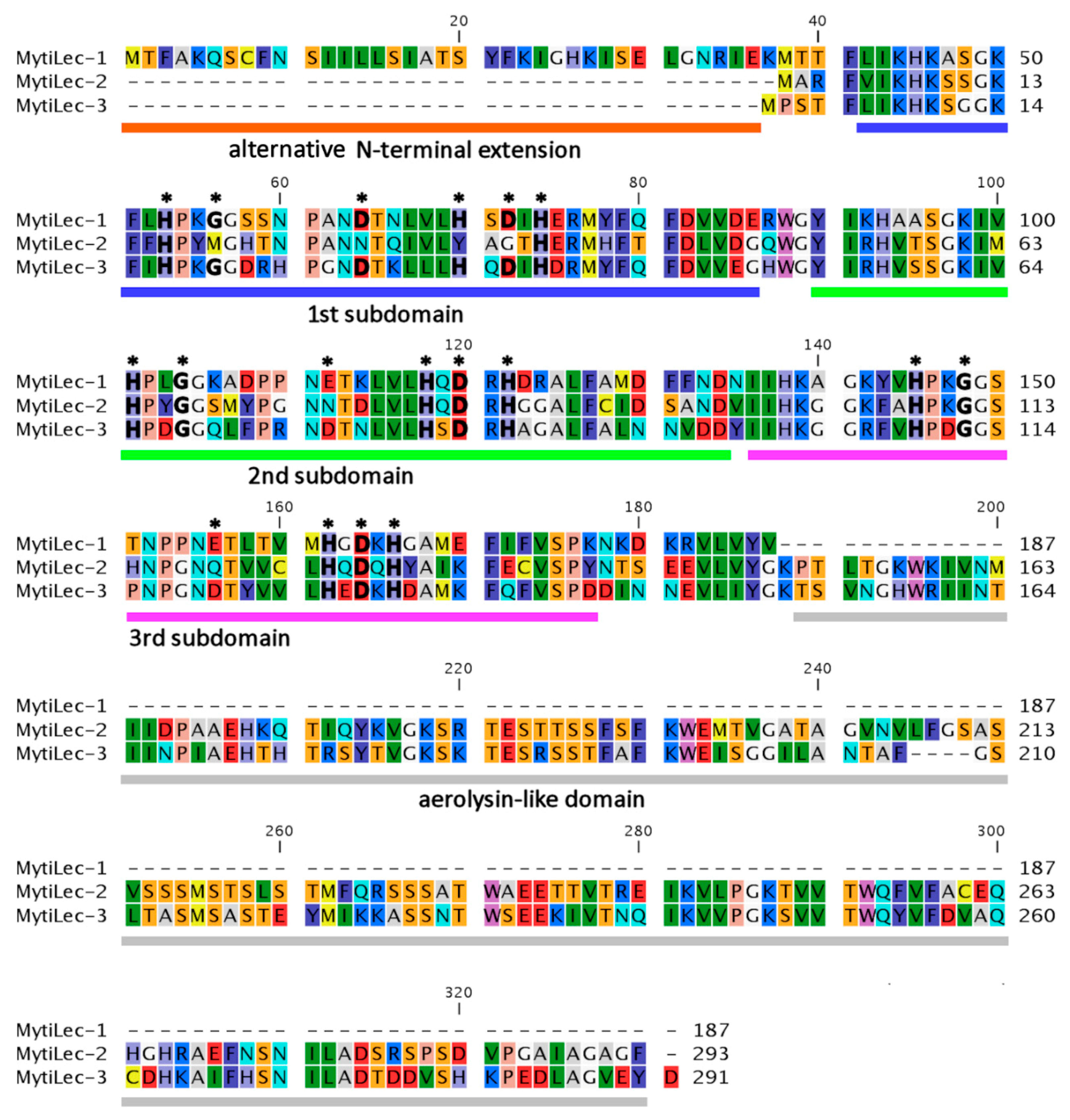
2.2. Comparison of Deduced Primary Structures among M. galloprovincialis Mytilectins
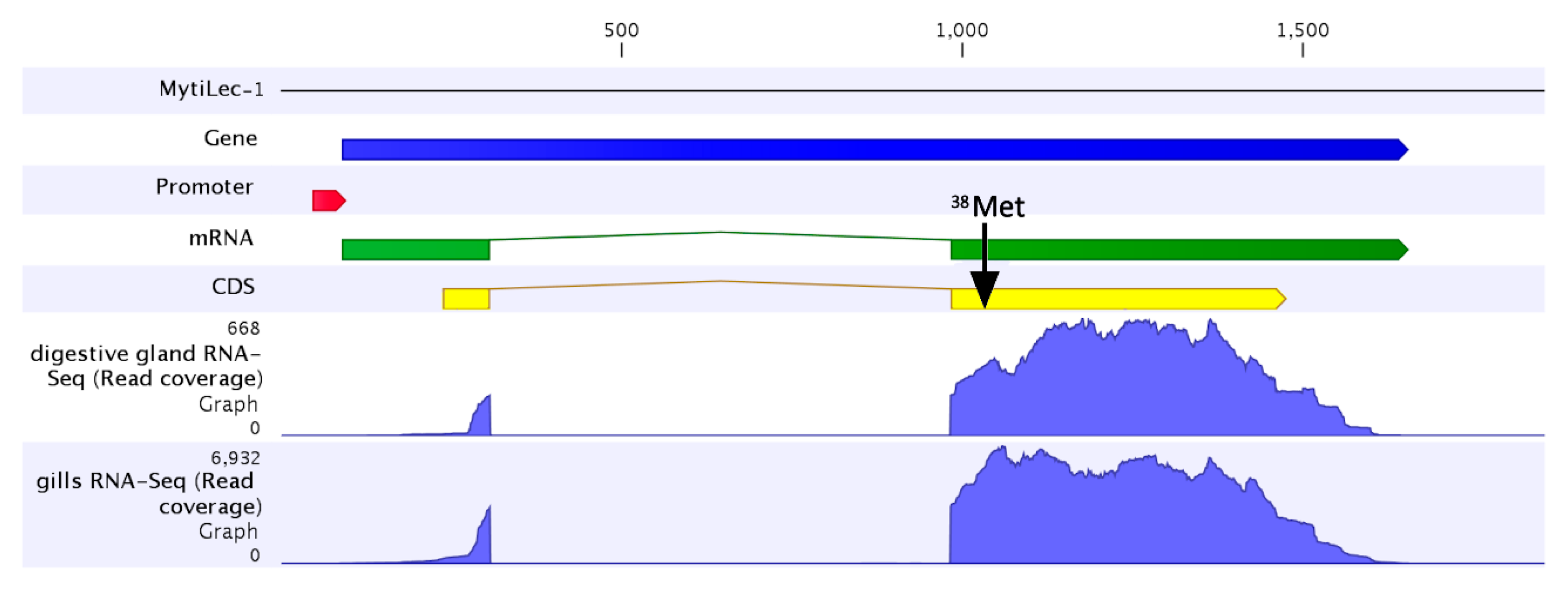
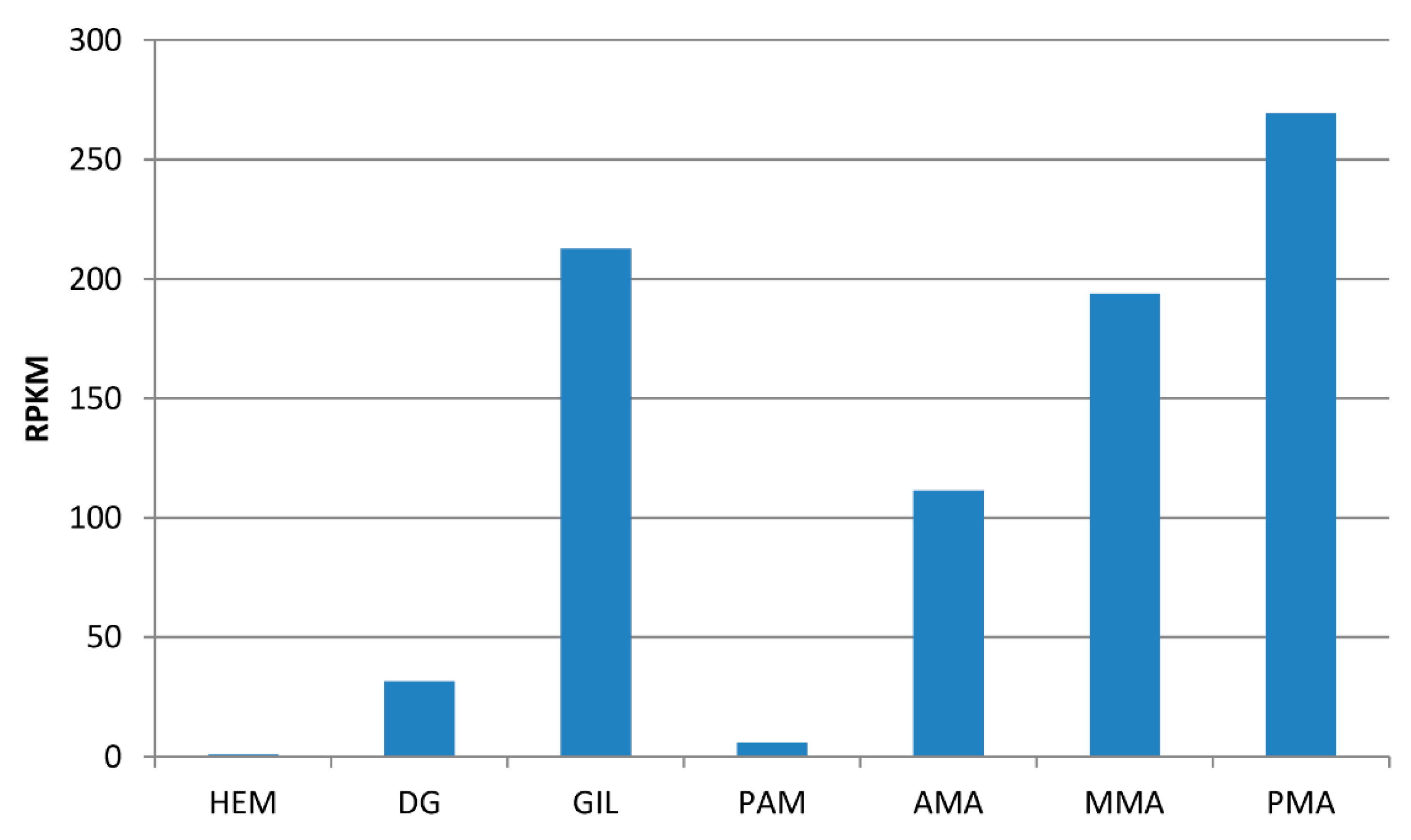
2.3. The MytiLec-1 Gene Comprises Two Exons
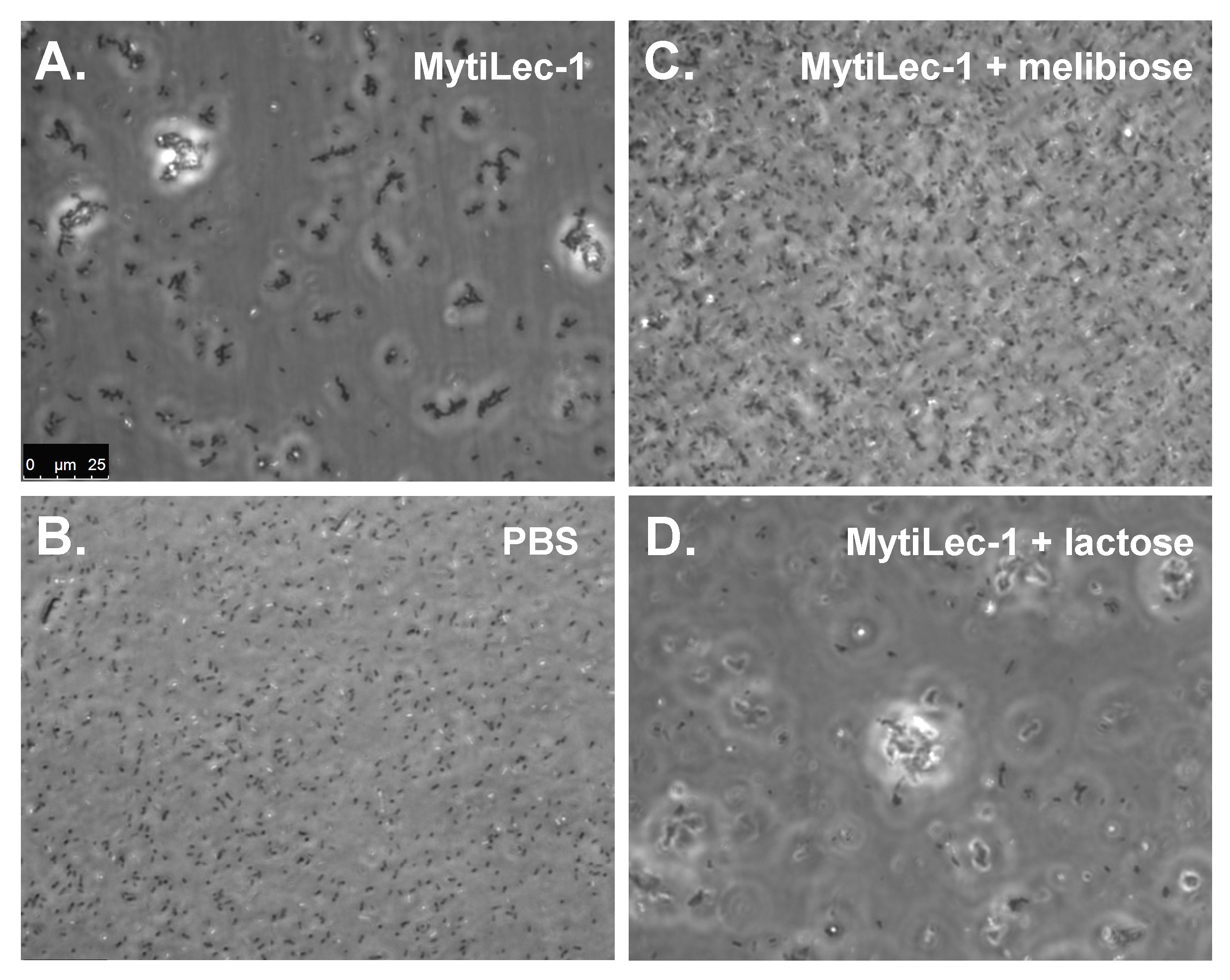
2.4. Agglutination of Bacteria by MytiLec-1 Signifies Its Bacteriostatic Activity
3. Experimental Design
3.1. cDNA Cloning and Sequencing of MytiLec-1
3.2. Transcriptome Analysis of Full Length of MytiLec-1 mRNA
3.3. Gene Structure Analysis of MytiLec-1
3.4. Purification of MytiLec-1
3.5. Bacteriostatic Assay of MytiLec-1
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Suárez-Ulloa, V.; Fernández-Tajes, J.; Manfrin, C.; Gerdol, M.; Venier, P.; Eirín-López, J. Bivalve omics: State of the art and potential applications for the biomonitoring of harmful marine compounds. Mar. Drugs 2013, 11, 4370–4389. [Google Scholar] [CrossRef] [PubMed]
- Venier, P.; De Pittà, C.; Bernante, F.; Varotto, L.; De Nardi, B.; Bovo, G.; Roch, P.; Novoa, B.; Figueras, A.; Pallavicini, A.; et al. MytiBase: A knowledgebase of mussel (M. galloprovincialis) transcribed sequences. BMC Genom. 2009, 10, 72. [Google Scholar] [CrossRef] [PubMed]
- Venier, P.; Varotto, L.; Rosani, U.; Millino, C.; Celegato, B.; Bernante, F.; Lanfranchi, G.; Novoa, B.; Roch, P.; Figueras, A.; et al. Insights into the innate immunity of the Mediterranean mussel Mytilus galloprovincialis. BMC Genom. 2011, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Gerdol, M.; Venier, P. An updated molecular basis for mussel immunity. Fish Shellfish Immunol. 2015, 46, 17–38. [Google Scholar] [CrossRef] [PubMed]
- Buchmann, K. Evolution of innate immunity: Clues from Invertebrates via fish to mammals. Front. Immunol. 2014, 5, 459. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, H.; Jian, S.; Wang, W.; Xin, L.; Wang, H.; Wang, L.; Song, L. A single-CRD C-type lectin from oyster Crassostrea gigas mediates immune recognition and pathogen elimination with a potential role in the activation of complement system. Fish Shellfish Immunol. 2015, 44, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Toubiana, M.; Gerdol, M.; Rosani, U.; Pallavicini, A.; Venier, P.; Roch, P. Toll-like receptors and MyD88 adaptors in Mytilus: Complete cds and gene expression levels. Dev. Comp. Immunol. 2013, 40, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Dohmae, N.; Takio, K.; Kawsar, S.M.; Matsumoto, R.; Hasan, I.; Koide, Y.; Kanaly, R.A.; Yasumitsu, H.; Ogawa, Y.; et al. A lectin from the mussel Mytilus galloprovincialis has a highly novel primary structure and induces glycan-mediated cytotoxicity of globotriaosylceramide-expressing lymphoma cells. J. Biol. Chem. 2012, 287, 44772–44783. [Google Scholar] [CrossRef] [PubMed]
- UniProtKB-B3EWR1 (LEC_MYTGA). Available online: http://www.uniprot.org/uniprot/B3EWR1 (accessed on 1 March 2016).
- PDBe > 3wmv. Available online: http://www.ebi.ac.uk/pdbe/entry/pdb/3WMV (accessed on 1 March 2016).
- Tame, J.; (Yokohama City University, Yokohama, Japan). Personal communication, 2015.
- Kovalchuk, S.N.; Chikalovets, I.V.; Chernikov, O.V.; Molchanova, V.I.; Li, W.; Rasskazov, V.A.; Lukyanov, P.A. cDNA cloning and structural characterization of a lectin from the mussel Crenomytilus. grayanus with a unique amino acid sequence and antibacterial activity. Fish Shellfish Immunol. 2013, 35, 1320–1324. [Google Scholar] [CrossRef] [PubMed]
- Chikalovets, I.V.; Kovalchuk, S.N.; Litovchenko, A.P.; Molchanova, V.I.; Pivkin, M.V.; Chernikov, O.V. A new Gal/GalNAc-specific lectin from the mussel Mytilus trossulus: Structure, tissue specificity, antimicrobial and antifungal activity. Fish Shellfish Immunol. 2016, 50, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Ballard, J.; Crabtree, J.; Roe, B.A.; Tweten, R.K. The primary structure of Clostridium septicum alpha-toxin exhibits similarity with that of Aeromonas hydrophila aerolysin. Infect. Immun. 1995, 63, 340–344. [Google Scholar] [PubMed]
- Szczesny, P.; Iacovache, I.; Muszewska, A.; Ginalski, K.; van der Goot, F.G.; Grynberg, M. Extending the aerolysin family: From bacteria to vertebrates. PLoS ONE 2011, 6, e20349. [Google Scholar] [CrossRef] [PubMed]
- Hasan, I.; Sugawara, S.; Fujii, Y.; Koide, Y.; Terada, D.; Iimura, N.; Fujiwara, T.; Takahashi, K.G.; Kojima, N.; Rajia, S.; et al. MytiLec, a mussel R-type lectin, interacts with surface glycan Gb3 on Burkitt’s lymphoma cells to trigger apoptosis through multiple pathways. Mar. Drugs 2015, 13, 7377–7389. [Google Scholar] [CrossRef] [PubMed]
- Mg_Nor01_51P18 Nor01 Mytilus galloprovincialis cDNA 3-, mRNA Sequence. Available online: http://www.ncbi.nlm.nih.gov/nucest/223022238 (accessed on 1 March 2016).
- Mytilus galloprovincialis mRNA for alpha-galactose-binding Lectin, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/LC125182 (accessed on 1 March 2016).
- Mytilus californianus Lectin mRNA, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/KT695159.1 (accessed on 1 March 2016).
- Mytilus trossulus Isolate S14-Myt GalNAc/Gal-Specific Lectin mRNA, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/KR019779.1?report=genbank&log$=seqview (accessed on 1 March 2016).
- Crenomytilus grayanus GalNAc/Gal-specific Lectin mRNA, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/JQ314213.1 (accessed on 1 March 2016).
- Pérez-García, C.; Morán, P.; Pasantes, J.J. Karyotypic diversification in Mytilus mussels (Bivalvia: Mytilidae) inferred from chromosomal mapping of rRNA and histone gene clusters. BMC Genet. 2014, 15, 84. [Google Scholar] [CrossRef] [PubMed]
- Fraïsse, C.; Belkhir, K.; Welch, J.J.; Bierne, N. Local interspecies introgression is the main cause of extreme levels of intraspecific differentiation in mussels. Mol. Ecol. 2016, 25, 269–286. [Google Scholar] [CrossRef] [PubMed]
- Gerdol, M.; (University of Trieste, Trieste, Italy). Personal communication, 2015.
- Kozak, M. Initiation of translation in prokaryotes and eukaryotes. Gene 1999, 234, 187–208. [Google Scholar] [CrossRef]
- Mytilus galloprovincialis MytiLec2 mRNA, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/KP125931.1 (accessed on 1 March 2016).
- Mytilus galloprovincialis MytiLec3 mRNA, Complete cds. Available online: http://www.ncbi.nlm.nih.gov/nuccore/KP125932.1 (accessed on 1 March 2016).
- Gaudet, R.G.; Sintsova, A.; Buckwalter, C.M.; Leung, N.; Cochrane, A.; Li, J.; Cox, A.D.; Moffat, J.; Gray-Owen, S.D. Cytosolic detection of the bacterial metabolite HBP activates TIFA-dependent innate immunity. Science 2015, 348, 1251–1255. [Google Scholar] [CrossRef] [PubMed]
- Wollin, R.; Creeger, E.S.; Rothfield, L.I.; Stocker, B.A.; Lindberg, A.A. Salmonella typhimurium mutants defective in UDP-d-galactose: Lipopolysaccharide α1,6-d-galactosyltransferase. J. Biol. Chem. 1983, 258, 3769–3774. [Google Scholar] [PubMed]
- Klein, R.A.; Hartmann, R.; Egge, H.; Behr, T.; Fisher, W. The aqueous solution structure of a lipoteichoic acid from Streptococcus pneumoniae strain R6 containing 2,4-diamino-2,4,6-trideoxy-galactose: Evidence for conformational mobility of the galactopyranose ring. Carbohydr. Res. 1998, 281, 79–98. [Google Scholar] [CrossRef]
- Rutenber, E.; Robertus, J.D. Structure of ricin B-chain at 2.5 Å resolution. Proteins 1991, 10, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Matsushima-Hibiya, Y.; Watanabe, M.; Hidari, K.I.; Miyamoto, D.; Suzuki, Y.; Kasama, T.; Kasama, T.; Koyama, K.; Sugimura, T.; Wakabayashi, K. Identification of glycosphingolipid receptors for pierisin-1, a guanine-specific ADP-ribosylating toxin from the cabbage butterfly. J. Biol. Chem. 2003, 278, 9972–9978. [Google Scholar] [CrossRef] [PubMed]
- Uchida, T.; Yamasaki, T.; Eto, S.; Sugawara, H.; Kurisu, G.; Nakagawa, A.; Kusunoki, M.; Hatakeyama, T. Crystal structure of the hemolytic lectin CEL-III isolated from the marine invertebrate Cucumaria echinata: Implications of domain structure for its membrane pore-formation mechanism. J. Biol. Chem. 2004, 279, 37133–37141. [Google Scholar] [CrossRef] [PubMed]
- Jakób, M.; Lubkowski, J.; O’Keefe, B.R.; Wlodawer, A. Structure of a lectin from the sea mussel Crenomytilus grayanus (CGL). Acta Crystallogr. Sect. F Struct. Biol. Commun. 2015, 71, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Betti, M.; Canonico, B.; Citterio, B.; Roch, P.; Canesi, L. Specificity of anti-Vibrio immune response through p38 MAPK and PKC activation in the hemocytes of the mussel Mytilus galloprovincialis. J. Invertebr. Pathol. 2010, 105, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Toubiana, M.; Rosani, U.; Giambelluca, S.; Cammarata, M.; Gerdol, M.; Pallavicini, A.; Venier, P.; Roch, P. Toll signal transduction pathway in bivalves: Complete cds of intermediate elements and related gene transcription levels in hemocytes of immune stimulated Mytilus galloprovincialis. Dev. Comp. Immunol. 2014, 45, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Gourgou, E.; Aggeli, I.K.; Beis, I.; Gaitanaki, C. Hyperthermia-induced Hsp70 and MT20 transcriptional upregulation are mediated by p38-MAPK and JNKs in Mytilus galloprovincialis (Lamarck); a pro-survival response. J. Exp. Biol. 2010, 213, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Kubler-Kielb, J.; Lai, W.T.; Schneerson, R.; Vinogradov, E. The structure of the Escherichia coli O148 lipopolysaccharide core region and its linkage to the O-specific polysaccharide. Carbohydr. Res. 2011, 346, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Valiente, E.; Jiménez, N.; Merino, S.; Tomás, J.M.; Amaro, C. Vibrio vulnificus biotype 2 serovar E gne but not galE is essential for lipopolysaccharide biosynthesis and virulence. Infect. Immun. 2008, 76, 1628–1638. [Google Scholar] [CrossRef] [PubMed]
- Gerdol, M.; De Moro, G.; Manfrin, C.; Milandri, A.; Riccardi, E.; Beran, A.; Venier, P.; Pallavicini, A. RNA sequencing and de novo assembly of the digestive gland transcriptome in Mytilus galloprovincialis fed with toxinogenic and non-toxic strains of Alexandrium minutum. BMC Res. Notes 2014, 7, 722. [Google Scholar] [CrossRef] [PubMed]
- Romiguier, J.; Gayral, P.; Ballenghien, M.; Bernard, A.; Cahais, V.; Chenuil, A.; Chiari, Y.; Dernat, R.; Duret, L.; Faivre, N.; et al. Comparative population genomics in animals uncovers the determinants of genetic diversity. Nature 2014, 515, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Moreira, R.; Pereiro, P.; Canchaya, C.; Posada, D.; Figueras, A.; Novoa, B. RNA-Seq in Mytilus galloprovincialis: Comparative transcriptomics and expression profiles among different tissues. BMC Genom. 2015, 16, 728. [Google Scholar]
- Bjärnmark, N.A.; Yarra, T.; Churcher, A.M.; Felix, R.C.; Clark, M.S.; Power, D.M. Transcriptomics provides insight into Mytilus galloprovincialis (Mollusca: Bivalvia) mantle function and its role in biomineralisation. Mar. Genom. 2016. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucl. Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Reese, M.G.; Eeckman, F.H.; Kulp, D.; Haussler, D. Improved splice site detection in Genie. J. Comput. Biol. 1997, 4, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Reese, M.G. Application of a time-delay neural network to promoter annotation in the Drosophila melanogaster genome. Comput. Chem. 2001, 26, 51–56. [Google Scholar] [CrossRef]
- Naganuma, T.; Ogawa, T.; Hirabayashi, J.; Kasai, K.; Kamiya, H.; Muramoto, K. Isolation, characterization and molecular evolution of a novel pearl shell lectin from a marine bivalve, Pteria penguin. Mol. Divers. 2006, 10, 607–618. [Google Scholar] [CrossRef] [PubMed]

| Bacteria | Growth Suppressive Activity (%) | Agglutination b |
|---|---|---|
| Gram-positive | ||
| Bacillus subtilis | 74 ± 8 | ++ |
| Staphylococcus aureus | 61 ± 3 | + |
| Gram-negative | ||
| Escherichia coli | 58 ± 5 | ++ |
| Vibrio parahaemolyticus | 67 ± 1 | ++ |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, I.; Gerdol, M.; Fujii, Y.; Rajia, S.; Koide, Y.; Yamamoto, D.; Kawsar, S.M.A.; Ozeki, Y. cDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis). Mar. Drugs 2016, 14, 92. https://doi.org/10.3390/md14050092
Hasan I, Gerdol M, Fujii Y, Rajia S, Koide Y, Yamamoto D, Kawsar SMA, Ozeki Y. cDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis). Marine Drugs. 2016; 14(5):92. https://doi.org/10.3390/md14050092
Chicago/Turabian StyleHasan, Imtiaj, Marco Gerdol, Yuki Fujii, Sultana Rajia, Yasuhiro Koide, Daiki Yamamoto, Sarkar M. A. Kawsar, and Yasuhiro Ozeki. 2016. "cDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis)" Marine Drugs 14, no. 5: 92. https://doi.org/10.3390/md14050092
APA StyleHasan, I., Gerdol, M., Fujii, Y., Rajia, S., Koide, Y., Yamamoto, D., Kawsar, S. M. A., & Ozeki, Y. (2016). cDNA and Gene Structure of MytiLec-1, A Bacteriostatic R-Type Lectin from the Mediterranean Mussel (Mytilus galloprovincialis). Marine Drugs, 14(5), 92. https://doi.org/10.3390/md14050092












