Abstract
Sponges are the most prolific marine organisms with respect to their arsenal of bioactive compounds including antimicrobials. However, the majority of these substances are probably not produced by the sponge itself, but rather by bacteria or fungi that are associated with their host. This review for the first time provides a comprehensive overview of antimicrobial compounds that are known to be produced by sponge-associated microbes. We discuss the current state-of-the-art by grouping the bioactive compounds produced by sponge-associated microorganisms in four categories: antiviral, antibacterial, antifungal and antiprotozoal compounds. Based on in vitro activity tests, identified targets of potent antimicrobial substances derived from sponge-associated microbes include: human immunodeficiency virus 1 (HIV-1) (2-undecyl-4-quinolone, sorbicillactone A and chartarutine B); influenza A (H1N1) virus (truncateol M); nosocomial Gram positive bacteria (thiopeptide YM-266183, YM-266184, mayamycin and kocurin); Escherichia coli (sydonic acid), Chlamydia trachomatis (naphthacene glycoside SF2446A2); Plasmodium spp. (manzamine A and quinolone 1); Leishmania donovani (manzamine A and valinomycin); Trypanosoma brucei (valinomycin and staurosporine); Candida albicans and dermatophytic fungi (saadamycin, 5,7-dimethoxy-4-p-methoxylphenylcoumarin and YM-202204). Thirty-five bacterial and 12 fungal genera associated with sponges that produce antimicrobials were identified, with Streptomyces, Pseudovibrio, Bacillus, Aspergillus and Penicillium as the prominent producers of antimicrobial compounds. Furthemore culture-independent approaches to more comprehensively exploit the genetic richness of antimicrobial compound-producing pathways from sponge-associated bacteria are addressed.
1. Introduction
Antimicrobial resistance (AMR) is an emerging global threat, decreasing the possibilities for prevention and treatment of infectious diseases caused by viruses, bacteria, parasites and fungi [1,2]. A global surveillance report by the World Health Organization (WHO) [2] indicated an increase of morbidity and mortality of infectious diseases due to AMR, which could lead to a world-wide economic loss of up to 100 trillion US dollars (USD) in 2050 as the result of a 2%–3% reduction in the gross domestic product (GDP) [1]. A conservative estimation is that AMR now annually attributes to 700,000 deaths globally, with a potential leap to 10 million in 2050 [1]. AMR is a response of microorganisms against antimicrobial compounds, which can arise via several mechanisms such as chromosomal mutations [1], binding site modifications [2] or horizontal transfer of genes conferring resistance [3]. For several pathogenic bacteria such as Staphylococcus aureus [4], Pseudomonas aeruginosa [1,5], and Mycobacterium tuberculosis [6], the emergence of multi drug resistant (MDR) strains has been reported, which make infections with these strains increasingly difficult to treat with currently available antibiotics [3].
In the context of the arms race between humans and infectious agents, the discovery and development of new types of antimicrobial compounds with pronounced bioactivity and clinical significance are urgent [4,5]. The efforts to modify existing drugs are often not effective to overcome the mutation rate of pathogens and do not lead to the introduction of new classes of antimicrobial compounds [6]. The terrestrial environment has been the main focus of microbial-derived drug discovery since the first report on Penicillin in 1929 [7], followed by the booming of new classes of antibiotics in 1960s [8]. Although novel antimicrobials are still being discovered from the soil niche, e.g., turbomycin A and B [9] and teixobactin [10], there are issues with de-replication, which significantly reduces the discovery rate of new compounds from heavily screened environments [11].
In comparison with soils, the marine environment has been largely neglected for discovery of antibiotics until recently, mainly because of accessibility issues, but yet hold a huge biodiversity and potential novelty of antimicrobial compounds [12]. Of many marine organisms, sponges (phylum Porifera) are considered as the most prolific source of therapeutic compounds as these animals harbour a large variety of secondary metabolites, many of which are beneficial for human health purposes [13,14,15,16,17]. The “Supply Issue” is the main obstacle to exploit the biological activity of sponges’ metabolites since a large quantity of biomaterial is required for experimental purposes [13]. Interestingly, in recent years an increasing number of studies highlighted that many active substances from sponges are of bacterial origin due to similarity to chemical structures found in terrestrial microorganisms [13,14,15]. Furthermore, several studies have reported a wide diversity of antimicrobial activities from sponge-associated microbes, which make these microbial communities a valuable source for novel antimicrobials [14,16,17,18,19,20].
This review highlights the current knowledge of antimicrobial compounds produced by sponge-associated microbes. Our definition of “antimicrobial” is not limited to antibacterial agents, but also includes compounds active against viruses, fungi and infectious protozoa. For each of the four biological activities, a few substances are highlighted because of their high activity, along with the most complete overview to date of other known compounds with antimicrobial activity from sponge-associated microorganisms. To compare different bioactive compounds and crude extracts, inhibitory concentrations of substances reviewed have been as much as possible expressed in the same unit (µg/mL). Original articles use minimum inhibitory concentrations (MIC), half maximum inhibitory concentrations (IC50) and the concentration of a drug that give the half-maximal response (EC50). As they are not easily converted, we sticked to the original measures.
Moreover, we analyzed the distribution of bacterial and fungal genera associated with sponges that have been reported to produce antimicrobial compounds to identify the most prolific genera. In addition, the potential for application of metagenomics to complement culture-dependent antimicrobial screening strategies is also discussed.
3. Antibacterial Compounds
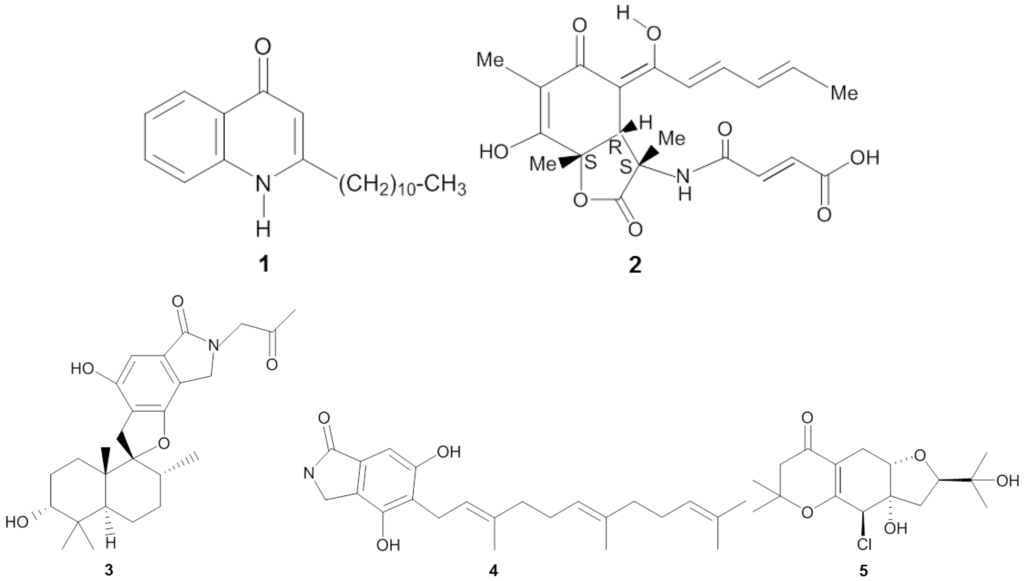
The screening procedure for antibacterial activity often includes both Gram positive and Gram negative target strains, including, e.g., Staphylococcus spp., Streptococcus spp., Bacillus spp., Clostridium spp., Escherichia spp., and Pseudomonas spp. From a medical point of view, these genera receive attention because they are well represented among the causative agents for human infectious diseases, such as pneumonia, urinary tract and blood stream infections [38,39]. Microbial isolates from marine sponges have been shown to exhibit bioactivity against a wide spectrum of pathogenic bacteria (Table 2). The novel thiopeptide antibiotics YM-266183 (6) and YM-266184 (7) (Figure 2), which were isolated from the sponge-associated bacterium Bacillus cereus QN03323, showed antibacterial activity against nosocomial infectious Gram positive bacteria in vitro [40,41]. Both YM-266183 and YM-266184 effectively inhibited Staphylococcus aureus and vancomycin-resistant Enterococcus faecium as indicated by minimal inhibition concentration (MIC) values as low as 0.025 µg/mL. In addition, compound YM-266184 was found particularly active against methicillin resistant Staphylococcus aureus (MRSA) with a MIC of 0.39 µg/mL. Compound YM-266183 also inhibited MRSA but required a two-fold higher concentration of the pure compound. Bioactivity of these thiopeptides was also observed against Streptococcus epidermidis and Enterococcus spp. (Table 2). The compound kocurin (8) was identified from three sponge-associated actinobacteria: Kocuria marina F-276,310, Kocuria palustris F-276,345, and Micrococcus yunnanensis F-256,446 [42,43]. Kocurin is a new member of the thiazolyl peptide family and exhibited anti-MRSA activity with an MIC of 0.25 µg/mL, which to date is the most potent anti-MRSA compound reported from sponge-associated microbes. Scheenemaan et al. [44] isolated Streptomyces sp. HB202 from the sponge Haliclona simulans, which lead to discovery of the polyketide mayamycin. In vitro assays with mayamycin (9) showed bioactivity against S. aureus and MRSA with IC50 values of 1.16 µg/mL and 0.58 µg/mL respectively, along with an IC50 of 0.14 µg/mL against Staphylococcus epidermidis [45].

Table 2.
Bioactive compounds with antibacterial activity from sponge-associated microbes.
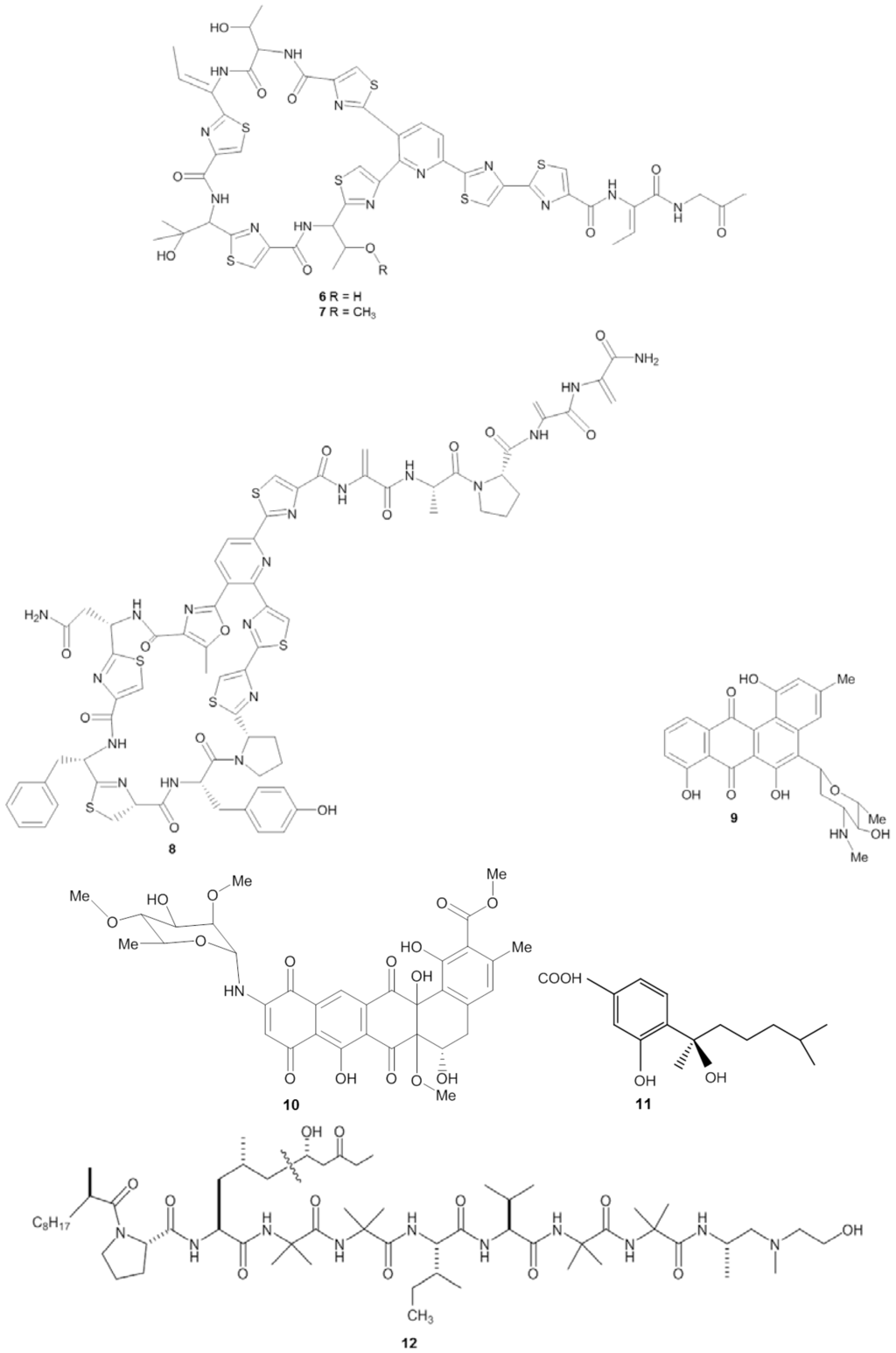
Figure 2.
Chemical structures of the antibacterial compounds YM-266183 (6), YM-266184 (7), kocurin (8), mayamycin (9), naphthacene glycoside SF2446A2 (10), sydonic acid (11) and trichoderin A (12).
Although many studies on antibacterial activity from sponge-associated microbes included Gram negative strains (Table 2), reports on pronounced antibacterial compounds active against Gram negative bacteria are limited in comparison to those that inhibit Gram positive strains. One of the examples of an inhibitor of a Gram negative bacterium is the compound naphthacene glycoside SF2446A2 (10) isolated from Streptomyces sp. RV15 that was originally obtained from the marine sponge Dysidea tupha [46]. Naphthacene glycoside SF2446A2 (10) inhibited the Gram-negative bacterium Chlamydia trachomatis at an IC50 value of 2.81 ± 0.24 µg/mL. Reimer et al. [46] underlined that compound 10 not only effectively inhibited the formation of chlamydial inclusion bodies during the primary infection but also affected the ability of C. trachomatis in producing viable progeny during the developmental cycle. Chlamydia trachomatis is an obligate intracellular Gram negative bacterium which is a leading cause of sexually transmitted diseases, and currently no methods are available to treat this infectious microorganism [46,47]. Li et al. [48] isolated four new bisabolane-typesesquiterpenoids: aspergiterpenoid A, (−)-sydonol, (−)-sydonic acid, (−)-5-(hydroxymethyl)-2-(2′,6′,6′-trimethyltetrahydro-2H-pyran-2-yl)phenol and a known compound (Z)-5-(Hydroxymethyl)-2-(6′-methylhept-2′-en-2′-yl)phenol from a sponge-associated Aspergillus sp. (Table 2). Of these five substances, the compound sydonic acid (11) exhibited the lowest MIC value against Escherichia coli at 1.33 µg/mL. This is the lowest inhibition concentration against E.coli reported from a compound produced by sponge-associated microbes although the inhibition concentration is still higher than the positive control ciprofloxacin (0.21 µg/mL) (Table 2).
Pruksakorn et al. [49] reported three prospective anti-tuberculosis compounds: trichoderin A (12), A1 and B from the sponge-associated fungus Trichoderma sp. 05FI48. Both under standard aerobic growth and dormancy-inducing hypoxic conditions, these three compounds inhibited Mycobacterium smegmatis, M. bovis BCG, and M. tuberculosis H37Rv with MIC values in the range of 0.02–2.0 µg/mL. Of these three compounds, trichoderin A was the most potent compound indicated by the lowest MIC values against those Mycobacterium strains. Additional analysis revealed that bioactivity of trichoderin A is based on its ability to inhibit adenosine triphosphate (ATP) synthesis of mycobacteria [50]. Compounds such as trichoderin A are particularly important because in many cases, pathogens such as Campylobacter spp., Helicobacter pylori, and Legionella pneumophila are difficult to treat due to the fact that they are present in a dormant state [51]. Such physiologically inactive cells highly contribute to the need for prolongued antibiotic treatments, which may lead to the emergence of resistant strains [52,53].
4. Antifungal Activity
The incidence rate of fungal infections has increased significantly over the past decades. This is mainly caused by clinical use of antibacterial drugs and immunosuppressive agents after organ transplantation, cancer chemotherapy, and advances in surgery [102,103]. Several fungal species that often cause human infections include Candida albicans, Candida glabrata, Cryptococcus neoformans and Aspergillus fumigatus [102,104,105]. The story becomes more complex as many of these pathogenic fungi develop resistance against available antifungal drugs, which will prolong duration of treatments [106].
Screening for antifungals is often focused on finding compounds active against Candida albicans, the prominent agent for candidiasis (Table 3). Invasive candidiasis is accounted as the most common nosocomial fungal infection resulting in an average mortality rate between 25%–38% [103]. El-Gendy et al. [107] isolated Streptomyces sp. Hedaya 48 from the sponge Aplysina fistularis and identified two compounds: the novel compound saadamycin (13) and the known compound 5,7-dimethoxy-4-p-methoxylphenylcoumarin (14) (Figure 3). Bioassays indicated that both saadamycin and 5,7-dimethoxy-4-p-methoxylphenylcoumarin displayed pronounced antifungal activity against Candida albicans with MIC values of 2.22 µg/mL and 15 µg/mL, respectively. In addition, both compounds displayed bioactivity against some pathogenic dermatophytes (skin-infecting fungi), such as Epidermophyton floccosum, Trichophyton rubrum, Trichophyton mentagrophytes, Microsporum gypseum, Aspergillus niger, Aspergillus fumigatus, Fusarium oxysporum, and Cryptococcus humicolus (Table 3). Further analysis showed that saadamycin displayed a more potent bioactivity indicated by a 3875 fold lower MIC than that of the reference compound, miconazole, whereas 5,7-dimethoxy-4-p-methoxylphenylcoumarin was around a 200 fold more potent than miconazole.

Table 3.
Bioactive compounds with antifungal activity from sponge-associated microbes.
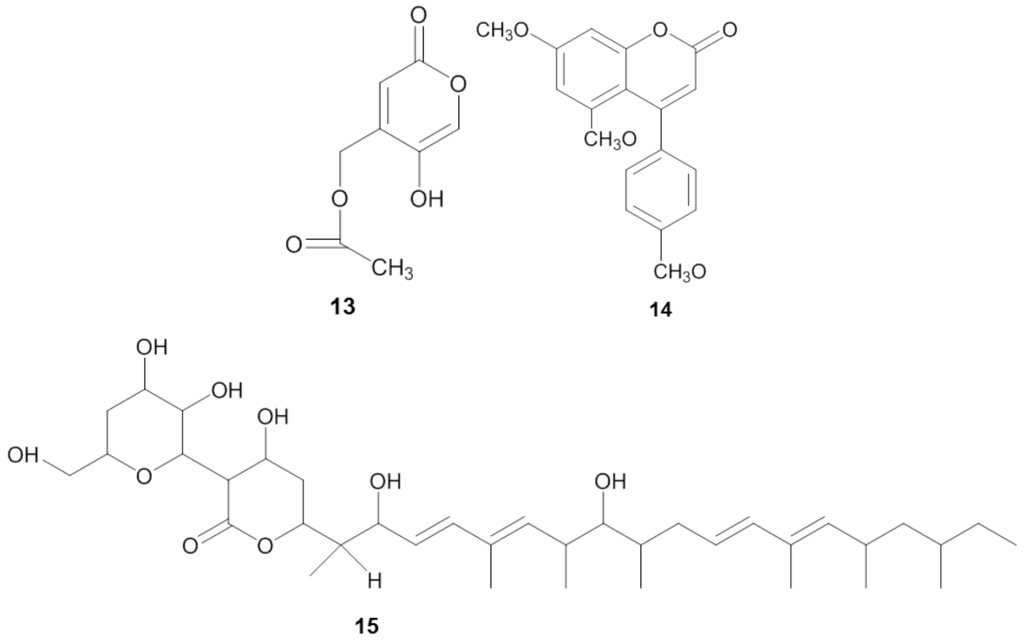
Figure 3.
Chemical structures of the antifungal compounds saadamycin (13), 5,7-dimethoxy-4-p-methoxylphenylcoumarin (14) and YM-202204 (15).
Antifungal activity was also detected from the sponge-associated fungus Phoma sp. Q60596. The sponge-derived fungus produced a new lactone compound, YM-202204 (15) [108], which was effective against C. albicans (IC80 of 6.25 µg/mL), along with Cryptococcus neoformans (IC80 of 1.56 µg/mL), Saccharomyces cerevisiae (IC80 of 1.56 µg/mL) and Aspergillus fumigatus (IC80 of 12.5 µg/mL). Furthermore, Nagai et al. [108] showed that YM-202204 was able to block the glycophosphatidylinositol (GPI) anchor, an important structure for protein attachment in the membrane of eukaryotic cells and one of the targets in developing antifungal drugs [109,110].
5. Antiprotozoal Activity
Malaria, caused by Plasmodium spp. infections, represents the most devastating protozoal disease worldwide, and results in both mortality and economic loss, mainly in developing countries [116]. Developing drugs with a better therapeutic profile against the parasite is one of the key aims of current malaria research, which includes screening for antimalarial substances from marine organisms [117,118].
Manzamine A (16) (Figure 4), first reported by Sakai and co-workers [119] from the sponge Haliclona sp., is a promising substance against Plasmodium spp. Initially, its antitumor property was of main interest, but subsequently diverse antimicrobial activities such as: anti-HIV, antibacterial, and antifungal were identified from the compound [120]. Currently the antimalaria properties of manzamine A are considered its most promising bioactivity. Manzamine A was shown to inhibit P. falciparum D6 and W3 clonal cell lines that are sensitive and resistant against the antimalarial chloroquine [121], with IC50 values of 0.0045 and 0.008 µg/mL, respectively [122]. Furthermore, in vivo screening by Ang et al. [116] showed that manzamine A at concentration of 0.008 µg/mL inhibited 90% growth of the parasite Plasmodium berghei that causes malaria in rodents. In addition, Rao et al. reported [122] that manzamine A displayed anti-Leishmania activity, indicated by IC50 and IC90 values of 0.9 µg/mL and 1.8 µg/mL, respectively, against Leishmania donovani.
Figure 4.
Chemical structures of the antiprotozoal compounds manzamine A (16), valinomycin (17), staurosporine (18) and butenolide (19).
Isolation of manzamine A from several other sponge species [120] raised the hypothesis that it was of microbial origin [123,124]. Hill et al. [125] confirmed this hypothesis by isolating Micromonospora sp. M42 as the microbial producer of manzamine A from the Indonesian sponge Acanthostrongylophora ingens. A series of analyses using molecular-microbial community analysis, and Matrix Assisted Laser Desorption Ionization-Mass Spectrometry (MALDI-MS) corroborated that indeed the strain Micromonospora sp. M42 synthesizes manzamine A [126,127]. Considering the therapeutic potential of manzamine A for treating malaria and leishmaniasis, Micromonospora sp. M42 could be a sustainable provider of the substance, because the “Sponge Supply Problem” has been overcome [127]. Moreover, identification of several manzamine-derivatives e.g. manzamine E, F, J, and 8-hydroxymanzamine A, from marine sponges which displayed antibacterial, antifungal and antiprotozoal activity [122,124], could also lead to isolation of associated microbial producers in the future.
Pimentel-Elardo et al. [128] identified three compounds with anti-Leishmania and anti-Trypanosoma activity from a sponge-associated Streptomyces sp, namely the cyclic depsipeptide valinomycin (17), the indolocarbazole alkaloid staurosporine (18) and butenolide (19) (Table 4). Valinomycin and staurosporine inhibited the growth of L. major with IC50 values of 0.12 µg/mL and 1.24 µg/mL, respectively. In addition, the three compounds displayed bioactivity against Trypanosoma brucei with IC50 values of 0.0036 µg/mL for valinomycin, 0.0051 µg/mL for staurosporine and 7.92 µg/mL for butenolide.

Table 4.
Bioactive compounds with antiprotozoal activity from sponge-associated microbes.
Scopel et al. [129] isolated two sponge-associated fungi, namely Hypocrea lixii F02 and Penicillium citrinum F40 (Table 4) that were active against the protozoal parasite Trichomonas vaginalis, which causes trichomoniasis, a sexually transmitted disease [130]. Culture filtrates of both isolates inhibited T. vaginalis ATCC 30236 and fresh clinical isolates, including the metronidazole-resistant TV-LACM2, with MIC values of 2.5 mg/mL. Further observation indicated that culture filtrates of these two fungi had no haemolytic effect against mammalian cells, which is one of the important criteria to further develop anti-protozoal drugs [129].
6. Dicussion
6.1. Antimicrobial Compounds from Sponge-Associated Microbes: What We Learned So Far
Bioprospecting is the effort to discover natural compounds with therapeutic and biological applications [140]. In line with this definition, sponge-associated microbes offer a huge potential as the source of antimicrobial substances as shown by many microbial isolates being reported to inhibit pathogenic reference strains in vitro and to synthesize active substances against one or several groups of infectious agents. Based on our review, antimicrobial compounds produced by sponge-associated microbes with the most pronounced bioactivity include: 2-undecyl-4-quinolone, sorbicillactone A, stachybotrin D and chartarutine B against HIV-1; truncateol M against H1N1 M; YM-266183, YM-266184, kocurin, mayamycin, sydonic acid, naphthacene glycoside SF2446A2 and trichoderin A against a variety of bacterial strains; saadamycin and YM-202204 against fungi; manzamine-A against malaria; and valinomycin against Trypanosoma. In this case the most pronounced activity is solely based on reported inhibition data and does not yet take potential side effects into account. Therefore the most promising compounds may be ones that have higher IC50 values, but cause less side effects. As these data are not available for the majority of the reported compounds, we have focused on the most potent compounds.
Sponge-associated bacteria and fungi are the two groups of microorganisms that have been found to produce antimicrobial compounds (Figure 5). The large majority of the antimicrobial compounds found in sponge-associated microbiota is produced by bacteria (90%), while fungi account for approximately 10% of the compounds reported. Sponge-associated bacteria derived antimicrobial compounds were found from 35 genera (Figure 5B). At a higher taxonomic level, these 35 bacterial genera can be classified into the four phyla Actinobacteria, Proteobacteria, Firmicutes and Cyanobacteria with percentages of 48.8%, 36.6%, 11.4% and 0.4% respectively. In contrast, sponge-associated fungi that have been found to produce antimicrobials are affiliated solely to the phylum Ascomycota.
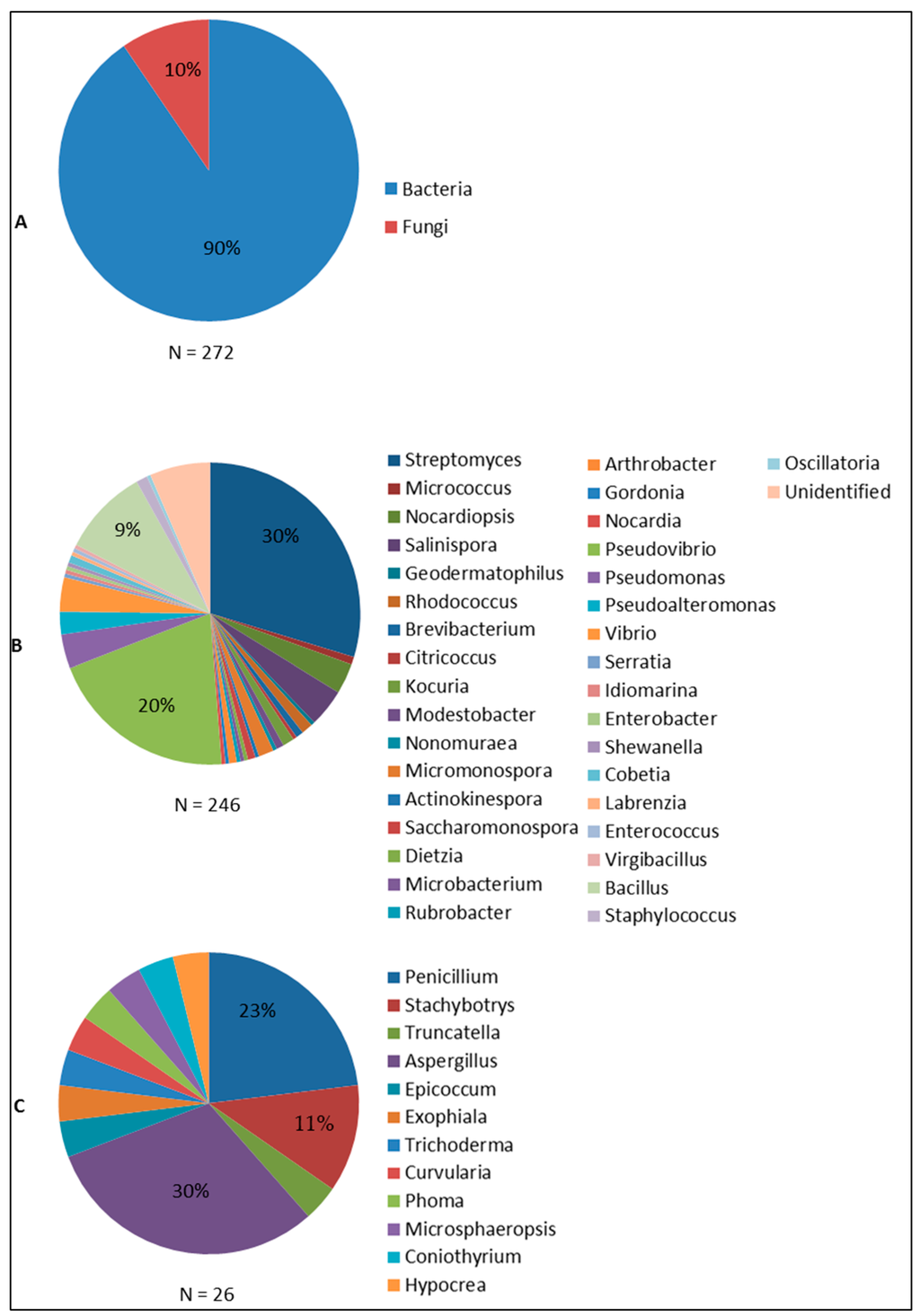
Figure 5.
Distribution of sponge-associated microorganisms found to produce antimicrobial compounds: (A) Bacteria and Fungi; (B) Bacterial genera; and (C) Fungal genera. Figure 5 was made based on the summary of the taxonomic affiliations of sponge-associated microbes (N = 272) that were found to produce antimicrobials.
Streptomyces is the most prominent genus as indicated by 30% of sponge bacteria-derived compounds. Streptomyces has become a main target for screening for bioactive compounds both from terrestrial and marine environments due to the high diversity of secondary metabolites they produce [141,142]. Of the many sponge-associated Streptomyces isolates reported, Streptomyces sp. HB202 and Streptomyces sp. RV15 are of particular interest in term of producing antibacterial compounds. Streptomyces sp. HB202, isolated from the sponge Halichondria panicea has been documented to produce three antibacterial substances: mayamycin, streptophenazine G and K, which are mainly active against Gram positive pathogenic bacteria (Table 2). Streptomyces sp. RV15, on the other hand, produces the compound naphthacene glycoside which up to now is the only anti-Chlamydia reported from sponge-associated microbes [46]. In addition, the report on crude extract inhibition of Streptomyces sp. RV15 against S. aureus and E. faecalis [82] may give a hint to discover other antibacterial substances from this strain. Streptomyces sp. Hedaya48 is currently the most potent sponge-associated bacterial isolate for antifungal activities with the production of saadamycin and 5,7-dimethoxy-4-p-methoxylphenylcoumarin [107]. In addition, isolation of the anti-Trypanosoma and anti-Leishmania compounds valinomycin, staurosporine and butenolide from Streptomyces sp. 43, 21 and 11 [128], affirms Streptomyces as the currently most prominent producer of antimicrobial substances from sponges.
Pseudovibrio follows as the second most prolific bacterial genus isolated from sponges (20%) with respect to antimicrobial activities. Reports on Pseudovibrio spp. are concentrated on antibacterial activity and are mainly based on screening of crude extracts. Up to now, tropodithietic acid is the only antibacterial compound that has been identified from Pseudovibrio [72]. Although representing a lower percentage of the sponge-associated bacteria found to produce antimicrobials than Streptomyces and Pseudovibrio, 9% of the currently known bioactives was found to be produced by sponge-associated Bacillus spp., with activities against viruses, bacteria and fungi. Bacillus cereus QNO3323 is currently the most prominent antimicrobial producer from this genus with the very potent thiopeptides YM-266183 and YM-266184 that are active against Gram positive bacteria.
Sponge-associated Ascomycota found to produce antimicrobials can be further classified into 12 genera. Of these 12 fungal genera, Aspergillus (30%) and Penicillium (23%) are currently the two most prominent groups of sponge-associated fungi reported as antimicrobial producers. This finding is not suprising since both Aspergillus and Penicillium are known prolific producers of secondary metabolites from other sources [143]. Aspergillus versicolor [58] and an unidentified Aspergillus sp. isolated from the sponge Xestospongia testudinaria [48] showed a strong antibacterial activity as indicated by potent inhibition of pathogenic bacteria. The antimicrobial activities found from sponge-associated Penicillium spp. are particular remarkable as it is the only fungal genus that is found to produce antivirals, antibacterials antifungals and antiprotozoals. Penicillium chrysogenum [26] and Penicillium sp. FF01 [57] are to date the most promising sponge-associated Penicillium isolates for which anti-HIV activity (sorbicillactone) and antibacterial activity (citrinin) were reported, respectively. Sponge-derived Stachybotrys spp. are only known for antiviral activity, particularly against HIV and enterovirus 71 (EV71), and there are no reports of other antimicrobial activities. Generally, although the number of produced antimicrobials is outnumbered by those of sponge-associated bacteria, sponge-associated fungi should be considered as an important reservoir of antimicrobial compounds.
When the chemical structures of sponge-microbe-derived compounds are considered, a rather diverse array of structures is observed, including peptides, terpenoids, phenazines, indoles, phenoles and polyketides. Sixty percent of the antivirals from sponge-associated microbes are ketone derivatives (quinolone, sorbicillactone, isoindolinone, butyrolactone, furanone, xanthone, methanone, phenone). Peptide derivatives constitute 19% of the total identified antibacterial substances and roughly 12.5% from the total antimicrobial compounds reviewed here. Phenazine derivatives are the second most frequently isolated class of antibacterial compounds from sponge-associated microbes (15%) as exemplified in this review by the antibacterial compounds streptophenazine [89], phenazine alkaloid antibiotics [55], 6-hydroxymethyl-1-phenazine-carboxamide and 1,6-phenazinedimethanol [94]. Phenazine is a nitrogen-containing heterocyclic compound with a wide range of biological activities [67,144], and several studies from terrestrial environments and chemically synthesized phenazines have been reported as antiviral [145], antibacterial [146], and antimalaria [147]. Moreover, this group of compounds is attractive for therapeutic application since their structures are relatively small and hence can easily reach tissues and organs [67,148].
6.2. Discovering Antimicrobial Compounds from Sponge-Associated Microbes: From Culture-Dependent to Culture-Independent Methods
Isolation of antimicrobial producers provides a valuable basis for assessing the biotechnological potential of sponge-associated microbes. In a wider perspective, however, only a small fraction of this sponge-microbial community has been isolated under laboratory conditions leaving the majority resistant to in vitro growth with current cultivation approaches [15,149,150]. Several studies have focused on improving cultivability of sponge-associated microbes. Some of the approaches include using low nutrient media [151], floating filter cultures [152], employing different carbon sources, e.g., lectin [153], sponge extracts [152], and in situ cultivation using a diffusion growth chamber [154]. Furthermore, flow-cytometry and density gradient centrifugation have been applied to separate sponge cells from their associated bacteria to enrich the inoculum [155,156]. Additionally, co-cultivation through mixing of two or more microbial isolates in vitro [157] is an approach proposed to discover more natural compounds from sponge-associated microbes. The idea behind co-culture lies in the fact that many biosynthetic gene clusters found in microorganisms remain cryptic under standard laboratory conditions, and co-cultivation might provide a possibility to activate these silent genes [158,159]. As an example, the co-culture by Dashti et al. [98] of the sponge-associated Actinobacteria, Actinokinespora sp. EG49 and Nocardiopsis sp. RV163, resulted in isolation of the antibacterial compound 1,6-dihydroxyphenazine, which was not found from the individual isolates. However, even if the cultivability of sponge-associated microbes is improved, there is a long way ahead to reach a point that we will be able to isolate and routinely cultivate 50% of the microbes that are found in sponges. At the same time, the advance of genetic and molecular studies has resulted in the development of tools to study genes, transcripts and proteins by directly analyzing environmental DNA, RNA and proteins, thus bypassing cultivation procedures [157]. In relation to screening for antimicrobial activity, metagenomics has been applied to identify antimicrobials of uncultivated microorganisms from terrestrial environments, such as the antimycobacterial nocardamine, the putative antibacterial activity of terragines A–E [160], violacein that is active against S. aureus, Bacillus sp. and Streptococcus sp. [161] and a polyketide with activity against the yeast Saccharomyces cerevisiae [162].
Two main metagenomic approaches, functional screening and sequence homology-based methods, are generally distinguished [163]. Functional screening relies on detection of the metabolic activities of metagenomic library clones without requiring any prior sequence information [163,164,165]. Gillespie et al. [9] applied function-based metagenomics with E. coli as expression host, to identify the antibiotics turbomycin A and B from a soil sample. MacNeil et al. [166] identified the antimicrobial indirubin by constructing a BAC (bacterial artificial chromosome) library in E.coli. Yung et al. [167] reported two hydrolytic enzymes from fosmid clones CcAb1 and CcAb2, which were derived from a metagenome of the sponge Cymbastela concentrica using E. coli as the host. Both fosmid clones inhibited the growth of Bacillus sp. with an inhibition diameter of 20 mm, and clone CcAb1 showed additional inhibition of S. aureus and an Alteromonas sp. with diameters of inhibition of 50 mm and 60 mm, respectively. Further phylogenetic analysis showed that active genes encoding for these enzymes were of microbial origin [167]. He et al. [168] constructed a fosmid library of the sponge Discodermia calyx using E. coli as the host and identified antimicrobial activity of the enzyme 3-hydroxypalmitic acid against B. cereus and C. albicans. In addition, using the same approach He et al. [169] observed an active clone, pDC113, that displayed a clear inhibition zone against B. cereus. Subsequently, 11 cyclodipeptides were identified from this clone. Generally, it can be stated that although a number of antimicrobials have been discovered through functional screening of metagenomic libraries from sponges, the expression of large gene clusters such as those encoding (polyketide synthase( PKS) and (non-ribosomal peptide synthetase (NRPS) is still a difficult hurdle to take. Several key elements need to be considered to achieve successful expression of biosynthetic gene clusters; namely mobilizing the biosynthetic pathway into a suitable vector, selecting an appropriate heterologous host and stably maintaining the gene clusters in the host [170]. The size of many of these gene clusters requires the use of cloning vectors that can accept large inserts, such as fosmids, or BACs if the required insert size is over 100 kb [171]. Selection of heterologous expression systems in particular is a crucial factor before applying functional metagenomics to identify antimicrobials, because expression hosts are microbes as well and especially clones that express genes encoding for enzymes involved in production of antimicrobials may therefore be non-viable. Ongley et al. [170] pointed out some considerations in selecting an expression host such as relatedness to the native producer, availability of genetic tools and precursors, a high growth rate, and suitability for fermentation at a large scale. E. coli, the most commonly used expression host, has limitations for expressing parts of metagenomes because, e.g., of the sheer size of some gene clusters, genes with deviating codon usage, incompatible regulatory elements, lack of biosynthesis precursors or unavailability of posttranslational modifications [165,172]. Therefore, in order to make screening for antimicrobials through metagenomic libraries more efficient, it is of utmost importance to diversify the suite of expression hosts used. Several non-E.coli hosts, such as Agrobacterium tumefaciens, Bacillus subtilis, Burkholderia graminis, Caulobacter vibrioides, Pseudoalteromonas haloplanktis, Pseudomonas putida, Ralstonia metallidurans, Rhizobium leguminosarum, Streptomyces avermitilis, S. albus, Pseudomonas putida, Sulfolobus solfataricus, Thermus thermophilus, Thiocapsa roseopersicina and Saccharopolyspora sp. have been developed and should be more seriously considered as expression hosts when performing metagenomic screenings for antimicrobials [165,172,173].
Sequence-based screening, on the other hand, requires information on the sequence of genes involved in the production of a natural product as guidance to search for similar sequences in a sequenced metagenomic library or scaffolds reconstructed from direct metagenomic sequencing [165]. Homology-based screening is suitable to identify a compound with highly conserved biosynthesis pathways, e.g., those mediated by PKS and NRPS [174]. Piel and colleagues [175,176,177,178,179] applied this method, and identified the antitumor polyketide onnamide from uncultivated bacteria of the sponge T. swinhoei. Sequence-based screening was applied by Fisch [180] to unravel the complete pathway of the polyketide psymberin that was found to possess a potent antitumor activity, from uncultivated sponge-associated microbes. By sequence-based screening of metagenomic libraries, Schirmer et al. [181] reported diverse polyketide gene clusters in microorganisms from the sponge Discodermia dissoluta. The development of techniques that yield longer read lengths, such as Pacific Biosciences (PacBio) RS II SMRT (Single Molecule Real-Time) sequencing technology, in which a single read can be extended over 10 kbp [182], can be instrumental in increasing the accuracy in assembling large gene clusters. Application of PacBio for secondary metabolite gene clusters has been reported by Alt and Wilkinson [183], who identified the 53,253 bp genomic fragment encoding the transacyltransferase (trans-AT) polyketide synthase (PKS) from a marine Streptomyces sp responsible for the production of the antibiotic anthracimycin (atc). Furthermore, using Streptomyces coelicolor as heterologous expression host, the authors confirmed production of anthracimycin [183]. Furthermore, single cell analysis by combining cell separation and fluorescence-assisted cell sorting (FACS) could be a strategy to overcome the complexity of the microbial community in sponges since this method can be used to select for genomes from microbes that are present in low abundance in the sponge leading to a simplified reconstruction of secondary metabolite gene clusters present in these bacteria [184]. This strategy has been applied by Wilson et al. [185] for resolving the gene clusters encoding the machinery needed for the production of the polytheonamides produced by the candidate genus Entotheonella from the sponge Theonella swinhoei.
Inspired by these examples, homology-based screening could be further exploited to identify biosynthesis gene sequences that could lead to the identification of novel antimicrobial substances from Nature’s excessive diversity. Moreover, application of homology-based screening can benefit from publicly available metagenomic sequencing data and prediction tools for analyzing biosynthesis gene clusters, e.g., AntiSMASH (Antibiotics and Secondary Metabolite Analysis Shell) [186,187]. Application of sequence-based screening, however, is limited by the fact that the found sequences need to be related to known compounds, inherently limiting the potential for novelty. Furthermore, information on gene sequences is no guarantee that the acquisition of a complete gene pathway has been obtained [188]. Therefore, sequence-based methagenomics should ideally be complemented by chemical analysis to confirm whether the predicted compound exists and is fully functional (Figure 6).
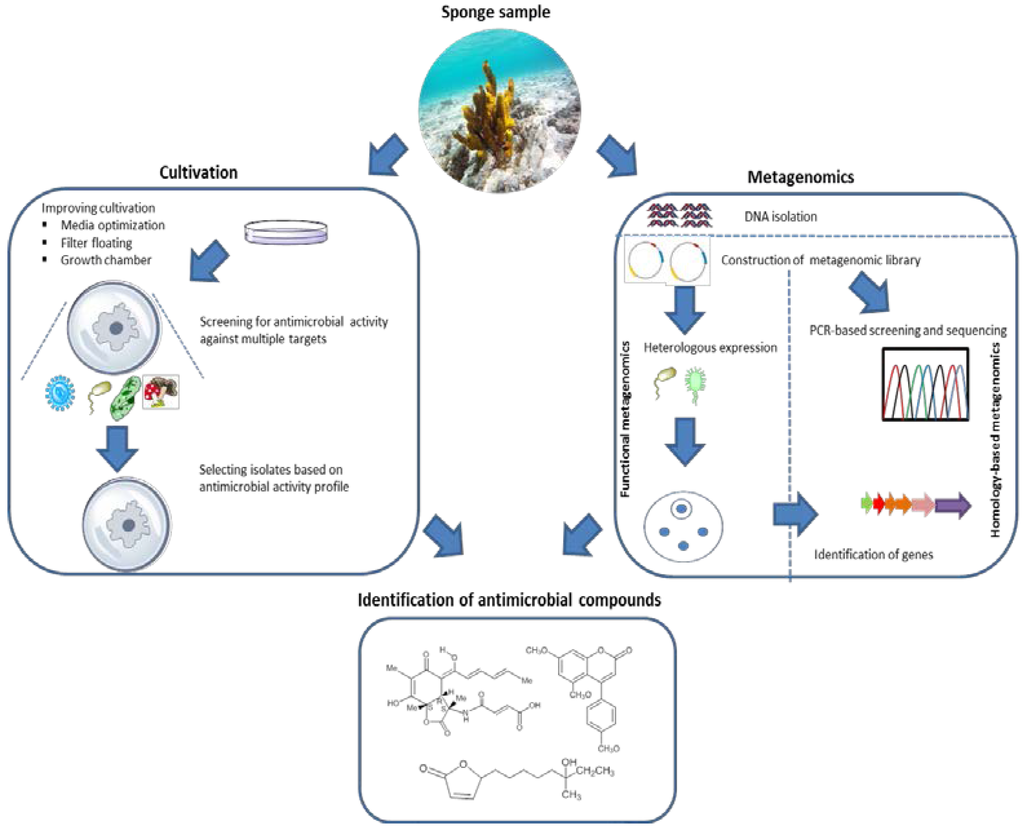
Figure 6.
General overview of the strategies used to discover antimicrobial compounds from sponge-associated microorganisms.
7. Conclusions and Outlook
Sponge-associated microbes already offer a rich source of potent antimicrobial compounds against viruses, bacteria, protozoa and fungi, and currently available compounds are predominantly active against HIV-1, H1N1, nosocomial Gram positive bacteria, Escherichia coli, Plasmodium spp, Leishmania donovani, Trypanosoma brucei, Candida albicans and dermatophytic fungi. Streptomyces, Pseudovibrio, Bacillus, Aspergillus and Penicillium are the microbial genera associated with sponges from which potent antimicrobial compounds are most frequently isolated. However, none of the antimicrobial compounds highlighted in this review have been succcesfully marketed as pharmaceuticals. To clearly translate bioactivity of these important compounds it is crucial to further unravel their mode of actions and measure their level of toxicity, since the majority of these studies has been focused on in vitro bioassays and elucidation of the chemical structures only.
The known versatility of antimicrobial activities found in sponge-associated microorganims could easily be expanded even without considering additional sponge sampling campaigns. Bioactivity screens of identified compounds or undefined sponge extracts is often restricted to a specific antimicrobial activity. The selection, for instance, relies on the specific research activities of the groups involved in isolating the microbes [117]. Consequently, it is probably safe to assume that other potent antimicrobial properties from many sponge isolates and their bioactive compounds remain undetected. Therefore, known antimicrobial compounds and producer strains are a valuable source for additional antimicrobial activities screenings using different target types (viruses, bacteria, fungi, protozoa and beyond). In addition, sponge-derived strain collections that comprise isolates that tested negative for antimicrobial activity at first may have done so, because the compound of interest is not produced under standard laboratory conditions. Exposure of these strains to potential microbial targets may lead to recovery of bioactivity that would otherwise go unnoticed.
Ideally, researchers who isolate microbes from sponges will deposit them to publicly available culture collections so that laboratories with complementary expertise and interests could benefit and screen the deposited isolates for different antimicrobial activities. This will make exchange of materials and knowledge that can be obtained much more efficient. Importantly, a fair agreement on intellectual property rights needs to be established for translating this into reality. Lastly, the revolutionary advance of next generation sequencing technologies combined with more diversified heterologous expression systems (Figure 6) are expected to open up the large unexplored reservoir of antimicrobials produced by yet uncultivated sponge-associated microbes.
Acknowledgments
Anak Agung Gede Indraningrat receives a PhD fellowship from the Indonesia Endowment Fund for Education (LPDP), grant number 20140812021557. This work was also supported by the EC grant “BluePharmTrain” (grant agreement no. 607786).
Author Contributions
Anak Agung Gede Indraningrat and Detmer Sipkema conceived the idea for the review, Anak Agung Gede Indraningrat compiled literatures, drew figures, and wrote the manuscript. Detmer Sipkema and Hauke Smidt checked and improved the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. Review on Antimicrobial Resistance: Chaired by Jim O’Neill. 2014. Available online: http://amr-review.org/Publications (accessed on 1 September 2015).
- Antimicrobial Resistance Global Report on Surveillance. World Health Organization, Geneva, Switzerland, 2014. Available online: http://www.who.int/drugresistance/documents/surveillancereport/en/ (accessed on 1 September 2015).
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Aminov, R.I. A brief history of the antibiotic era: Lessons learned and challenges for the future. Front. Microbiol. 2010, 1, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Moellering, R.C., Jr. Discovering new antimicrobial agents. Int. J. Antimicrob. Agents 2011, 37, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Projan, S.J. Why is big Pharma getting out of antibacterial drug discovery? Curr. Opin. Microbiol. 2003, 6, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A. On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzæ. Br. J. Exp. Pathol. 1929, 10, 226–236. [Google Scholar] [CrossRef]
- White, R.J. The Early History of Antibiotic Discovery: Empiricism Ruled. In Antibiotic Discovery and Development; Dougherty, T.J., Pucci, M.J., Eds.; Springer: New York, NY, USA, 2012. [Google Scholar]
- Gillespie, D.E.; Brady, S.F.; Bettermann, A.D.; Cianciotto, N.P.; Liles, M.R.; Rondon, M.R.; Clardy, J.; Goodman, R.M.; Handelsman, J. Isolation of Antibiotics Turbomycin A and B from a Metagenomic Library of Soil Microbial DNA. Appl. Environ. Microbiol. 2002, 68, 4301–4306. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schaberle, T.F.; Hughes, D.E.; Epstein, S.; et al. A new antibiotic kills pathogens without detectable resistance. Br. Dent. J. 2015, 517, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.L.; Wright, G.D. Novel approaches to discovery of antibacterial agents. Anim. Health Res. Rev. 2008, 9, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Hughes, C.C.; Fenical, W. Antibacterials from the Sea. Chem. Eur. J. 2010, 16, 12512–12525. [Google Scholar] [CrossRef] [PubMed]
- Thoms, C.; Schupp, P. Biotechnological potential of marine sponges and their associated bacteria as producers of new pharmaceuticals (part II). J. Int. Biotechnol. Law 2005, 2, 257–264. [Google Scholar] [CrossRef]
- Fuerst, J.A. Diversity and biotechnological potential of microorganisms associated with marine sponges. Appl. Microbiol. Biot. 2014, 98, 7331–7347. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.W.; Radax, R.; Steger, D.; Wagner, M. Sponge-associated microorganisms: Evolution, ecology, and biotechnological potential. Microbiol. Mol. Biol. R. 2007, 71, 295–347. [Google Scholar] [CrossRef] [PubMed]
- Laport, M.S.; Santos, O.C.S.; Muricy, G. Marine Sponges: Potential Sources of New Antimicrobial Drugs. Curr. Pharm. Biotechnol. 2009, 10, 86–105. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.R.A.; Kavlekar, D.P.; LokaBharathi, P.A. Marine drugs from sponge-microbe association—A review. Mar. Drugs 2010, 8, 1417–1468. [Google Scholar] [CrossRef] [PubMed]
- Santos-Gandelman, J.F.; Giambiagi-deMarval, M.; Oelemann, W.M.R.; Laport, M.S. Biotechnological Potential of Sponge-Associated Bacteria. Curr. Pharm. Biotechnol. 2014, 15, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Graça, A.P.; Viana, F.; Bondoso, J.; Correia, M.I.; Gomes, L.A.G.R.; Humanes, M.; Reis, A.; Xavier, J.; Gaspar, H.; Lage, O. The antimicrobial activity of heterotrophic bacteria isolated from the marine sponge Erylus deficiens (Astrophorida, Geodiidae). Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Hoppers, A.; Stoudenmire, J.; Wu, S.; Lopanik, N.B. Antibiotic activity and microbial community of the temperate sponge, Haliclona sp. J. Appl. Microbiol. 2015, 118, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Sagar, S.; Kaur, M.; Minneman, K.P. Antiviral Lead Compounds from Marine Sponges. Mar. Drugs 2010, 8, 2619–2638. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, W.; Feeney, R.J. The isolation of a new thymine pentoside from sponges. J. Am. Chem. Soc. 1950, 72, 2809–2810. [Google Scholar] [CrossRef]
- Bergmann, W.; Feeney, R.J. Contributions to the Study of Marine Products. XXXII. The nucleosides of sponges. I. J. Organ. Chem. 1951, 16, 981–987. [Google Scholar] [CrossRef]
- Yasuhara-Bell, J.; Lu, Y. Marine compounds and their antiviral activities. Antiviral Res. 2010, 86, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Bultel-Poncé, V.; Berge, J.-P.; Debitus, C.; Nicolas, J.-L.; Guyot, M. Metabolites from the sponge-associated bacterium Pseudomonas species. Mar. Biotechnol. 1999, 1, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Bringmann, G.; Lang, G.; Muhlbacher, J.; Schaumann, K.; Steffens, S.; Rytik, P.G.; Hentschel, U.; Morschhauser, J.; Müller, W.E.G. Sorbicillactone A: A structurally unprecedented bioactive novel-type alkaloid from a sponge-derived fungus. Prog. Mol. Subcell. Biol. 2003, 37, 231–253. [Google Scholar] [PubMed]
- Ma, X.H.; Lo, L.T.; Zhu, T.J.; Ba, M.Y.; Li, G.Q.; Gu, Q.Q.; Guo, Y.; Li, D.H. Phenylspirodrimanes with Anti-HIV activity from the sponge-derived fungus Stachybotrys chartarum MXH-X73. J. Nat. Prod. 2013, 76, 2298–2306. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, D.; Cen, S.; Proksch, P.; Lin, W. Isoindolinone-type alkaloids from the sponge-derived fungus Stachybotrys chartarum. Tetrahedron 2014, 70, 7010–7015. [Google Scholar] [CrossRef]
- Zhao, Y.; Si, L.; Liu, D.; Proksch, P.; Zhou, D.; Lin, W. Truncateols A-N, new isoprenylated cyclohexanols from the sponge-associated fungus Truncatella angustata with anti-H1N1 virus activities. Tetrahedron 2015, 71, 2708–2718. [Google Scholar] [CrossRef]
- Krol, E.; Rychowska, M.; Szewczyk, B. Antivirals—Current trends in fighting influenza. Acta Biochim. Pol. 2014, 61, 495–504. [Google Scholar] [PubMed]
- Pauletti, P.M.; Cintra, L.S.; Braguine, C.G.; da Silva Filho, A.A.; Silva, M.L.A.E.; Cunha, W.R.; Januário, A.H. Halogenated Indole Alkaloids from Marine Invertebrates. Mar. Drugs 2010, 8, 1526–1549. [Google Scholar] [CrossRef] [PubMed]
- Neumann, C.S.; Fujimori, D.G.; Walsh, C.T. Halogenation Strategies In Natural Product Biosynthesis. Chem. Biol. 2008, 15, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.X.; Jiao, J.Y.; Li, J.; Wang, W.; Gu, Q.Q.; Zhu, T.J.; Li, D.H. Pyronepolyene C-glucosides with NF-kappa B inhibitory and anti-influenza A viral (H1N1) activities from the sponge-associated fungus Epicoccum sp. JJY40. Bioorg. Med. Chem. Lett. 2012, 22, 3188–3190. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.H.; Zhu, T.J.; Gu, Q.Q.; Xi, R.; Wang, W.; Li, D.H. Structures and antiviral activities of butyrolactone derivatives isolated from Aspergillus terreus MXH-23. J. Ocean Univ. China 2014, 13, 1067–1070. [Google Scholar] [CrossRef]
- Wang, J.F.; Lin, X.P.; Qin, C.; Liao, S.R.; Wan, J.T.; Zhang, T.Y.; Liu, J.; Fredimoses, M.; Chen, H.; Yang, B.; et al. Antimicrobial and antiviral sesquiterpenoids from sponge-associated fungus, Aspergillus sydowii ZSDS1-F6. J. Antibiot. 2014, 67, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Lin, X.P.; Lu, X.; Wan, J.T.; Zhou, X.F.; Liao, S.R.; Tu, Z.C.; Xu, S.H.; Liu, Y.H. Sesquiterpenoids and xanthones derivatives produced by sponge-derived fungus Stachybotry sp. HH1 ZSDS1F1–2. J. Antibiot. 2015, 68, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Bastos, J.C.S.; Kohn, L.K.; Fantinatti-Garboggini, F.; Padilla, M.A.; Flores, E.F.; da Silva, B.P.; de Menezes, C.B.A.; Arns, C.W. Antiviral Activity of Bacillus sp. Isolated from the Marine Sponge Petromica citrina against Bovine Viral Diarrhea Virus, a Surrogate Model of the Hepatitis C Virus. Viruses 2013, 5, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Inweregbu, K.; Dave, J.; Pittard, A. Nosocomial infections. Cont. Educ. Anaesth. Crit. Care Pain 2005, 5, 14–17. [Google Scholar] [CrossRef]
- Weinstein, R.A.; Gaynes, R.; Edwards, J.R.; System, N.N.I.S. Overview of Nosocomial Infections Caused by Gram-Negative Bacilli. Clin. Infect. Dis. 2005, 41, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Kamigiri, K.; Arao, N.; Suzumura, K.; Kawano, Y.; Yamaoka, M.; Zhang, H.P.; Watanabe, M.; Suzuki, K. YM-266183 and YM-266184, novel thiopeptide antibiotics produced by Bacillus cereus isolated from a marine sponge—I. Taxonomy, fermentation, isolation, physico-chemical properties and biological properties. J. Antibiot. 2003, 56, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Suzumura, K.; Yokoi, T.; Funatsu, M.; Nagai, K.; Tanaka, K.; Zhang, H.P.; Suzuki, K. YM-266183 and YM-266184, novel thiopeptide antibiotics produced by Bacillus cereus isolated from a marine sponge—II. Structure elucidation. J. Antibiot. 2003, 56, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Palomo, S.; Gonzalez, I.; de la Cruz, M.; Martin, J.; Tormo, J.R.; Anderson, M.; Hill, R.T.; Vicente, F.; Reyes, F.; Genilloud, O. Sponge-derived Kocuria and Micrococcus spp. as sources of the new thiazolyl peptide antibiotic kocurin. Mar. Drugs 2013, 11, 1071–1086. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.; da S. Sousa, T.; Crespo, G.; Palomo, S.; González, I.; Tormo, J.R.; de la Cruz, M.; Anderson, M.; Hill, R.T.; Vicente, F.; et al. Kocurin, the true structure of PM181104, an Anti-Methicillin-Resistant Staphylococcus aureus (MRSA) thiazolyl peptide from the marine-derived bacterium Kocuria palustris. Mar. Drugs 2013, 11, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Schneemann, I.; Nagel, K.; Kajahn, I.; Labes, A.; Wiese, J.; Imhoff, J.F. Comprehensive investigation of marine Actinobacteria associated with the sponge Halichondria panicea. Appl. Environ. Microbiol. 2010, 76, 3702–3714. [Google Scholar] [CrossRef] [PubMed]
- Schneemann, I.; Kajahn, I.; Ohlendorf, B.; Zinecker, H.; Erhard, A.; Nagel, K.; Wiese, J.; Imhoff, J.F. Mayamycin, a cytotoxic polyketide from a Streptomyces strain isolated from the marine sponge Halichondria panicea. J. Natl. Prod. 2010, 73, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Reimer, A.; Blohm, A.; Quack, T.; Grevelding, C.G.; Kozjak-Pavlovic, V.; Rudel, T.; Hentschel, U.; Abdelmohsen, U.R. Inhibitory activities of the marine streptomycete-derived compound SF2446A2 against Chlamydia trachomatis and Schistosoma mansoni. J. Antibiot. 2015. [Google Scholar] [CrossRef] [PubMed]
- Vasilevsky, S.; Greub, G.; Nardelli-Haefliger, D.; Baud, D. Genital Chlamydia trachomatis: Understanding the Roles of Innate and Adaptive Immunity in Vaccine Research. Clin. Microbiol. Rev. 2014, 27, 346–370. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, Y.; Shao, C.-L.; Yang, R.-Y.; Zheng, C.-J.; Chen, Y.-Y.; Fu, X.-M.; Qian, P.-Y.; She, Z.-G.; de Voogd, N.J.; et al. Antibacterial Bisabolane-Type Sesquiterpenoids from the Sponge-Derived Fungus Aspergillus sp. Mar. Drugs 2012, 10, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Pruksakorn, P.; Arai, M.; Kotoku, N.; Vilchèze, C.; Baughn, A.D.; Moodley, P.; Jacobs, W.R., Jr.; Kobayashi, M. Trichoderins, novel aminolipopeptides from a marine sponge-derived Trichoderma sp., are active against dormant mycobacteria. Bioorg. Med. Chem. Lett. 2010, 20, 3658–3663. [Google Scholar] [CrossRef] [PubMed]
- Pruksakorn, P.; Arai, M.; Liu, L.; Moodley, P.; Jacobs, W.R., Jr.; Kobayashi, M. Action-Mechanism of Trichoderin A, an Anti-dormant Mycobacterial Aminolipopeptide from Marine Sponge-Derived Trichoderma sp. Biol. Pharm. Bull. 2011, 34, 1287–1290. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.D. Recent findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microbiol. Rev. 2010, 34, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.R.M.; Hu, Y. Novel approaches to developing new antibiotics for bacterial infections. Br. J. Pharmacol. 2007, 152, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.R.M.; Hu, Y. Targeting non-multiplying organisms as a way to develop novel antimicrobials. Trends Pharmacol. Sci. 2008, 29, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Eltamany, E.E.; Abdelmohsen, U.R.; Ibrahim, A.K.; Hassanean, H.A.; Hentschel, U.; Ahmed, S.A. New antibacterial xanthone from the marine sponge-derived Micrococcus sp. EG45. Bioorg. Med. Chem. Lett. 2014, 24, 4939–4942. [Google Scholar] [CrossRef] [PubMed]
- Jayatilake, G.S.; Thornton, M.P.; Leonard, A.C.; Grimwade, J.E.; Baker, B.J. Metabolites from an Antarctic sponge-associated bacterium, Pseudomonas aeruginosa. J. Natl. Prod. 1996, 59, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Song, F.H.; Ren, B.; Chen, C.X.; Yu, K.; Liu, X.R.; Zhang, Y.H.; Yang, N.; He, H.T.; Liu, X.T.; Dai, H.Q.; et al. Three new sterigmatocystin analogues from marine-derived fungus Aspergillus versicolor MF359. Appl. Microbiol. Biot. 2014, 98, 3753–3758. [Google Scholar] [CrossRef] [PubMed]
- Subramani, R.; Kumar, R.; Prasad, P.; Aalbersberg, W. Cytotoxic and antibacterial substances against multi-drug resistant pathogens from marine sponge symbiont: Citrinin, a secondary metabolite of Penicillium sp. Asian Pac. J. Trop. Biomed. 2013, 3, 291–296. [Google Scholar] [CrossRef]
- Lee, Y.; Li, H.; Hong, J.; Cho, H.; Bae, K.; Kim, M.; Kim, D.-K.; Jung, J. Bioactive metabolites from the sponge-derived fungus Aspergillus versicolor. Arch. Pharm. Res. 2010, 33, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Chen, H.; Han, X.; Lin, W.; Yan, X. Antimicrobial screening and active compound isolation from marine bacterium NJ6-3-1 associated with the sponge Hymeniacidon perleve. World J. Microbiol. Biotechnol. 2005, 21, 201–206. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, X.; Kang, J.S.; Choi, H.D.; Son, B.W. Chlorohydroaspyrones A and B, Antibacterial Aspyrone Derivatives from the Marine-Derived Fungus Exophiala sp. J. Nat. Prod. 2008, 71, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Ouyang, Y.; Zou, K.; Wang, G.; Chen, M.; Sun, H.; Dai, S.; Li, X. Isolation and Difference in Anti-Staphylococcus aureus Bioactivity of Curvularin Derivates from Fungus Eupenicillium sp. Appl. Biochem. Biotechnol. 2009, 159, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Santos, O.C.S.; Soares, A.R.; Machado, F.L.S.; Romanos, M.T.V.; Muricy, G.; Giambiagi-deMarval, M.; Laport, M.S. Investigation of biotechnological potential of sponge-associated bacteria collected in Brazilian coast. Lett. Appl. Microbiol. 2015, 60, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Sathiyanarayanan, G.; Gandhimathi, R.; Sabarathnam, B.; Seghal Kiran, G.; Selvin, J. Optimization and production of pyrrolidone antimicrobial agent from marine sponge-associated Streptomyces sp. MAPS15. Bioprocess. Biosyst. Eng. 2014, 37, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Unson, M.D.; Holland, N.D.; Faulkner, D.J. A brominated secondary metabolite synthesized by the cyanobacterial symbiont of a marine sponge and accumulation of the crystalline metabolite in the sponge tissue. Mar. Biol. 1994, 119, 1–11. [Google Scholar] [CrossRef]
- Kobayashi, M.; Aoki, S.; Gato, K.; Matsunami, K.; Kurosu, M.; Kitagawa, I. Marine Natural-Products. XXXIV. Trisindoline, a New Antibiotic Indole Trimer, Produced by a Bacterium of Vibrio sp. Separated from the Marine Sponge Hyrtios-Altum. Chem. Pharm. Bull. 1994, 42, 2449–2451. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, D.; Nazari, T.F.; Kassim, J.; Lim, S.-H. Prodigiosin—An antibacterial red pigment produced by Serratia marcescens IBRL USM 84 associated with a marine sponge Xestospongia testudinaria. J. Appl. Pharm. Sci. 2014, 4, 1–6. [Google Scholar]
- Karuppiah, V.; Li, Y.; Sun, W.; Feng, G.; Li, Z. Functional gene-based discovery of phenazines from the actinobacteria associated with marine sponges in the South China Sea. Appl. Microbiol. Biotechnol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Pabel, C.T.; Vater, J.; Wilde, C.; Franke, P.; Hofemeister, J.; Adler, B.; Bringmann, G.; Hacker, J.; Hentschel, U. Antimicrobial activities and matrix-assisted laser desorption/ionization mass spectrometry of Bacillus isolates from the marine sponge Aplysina aerophoba. Mar. Biotechnol. 2003, 5, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Devi, P.; Wahidullah, S.; Rodrigues, C.; Souza, L.D. The Sponge-associated Bacterium Bacillus licheniformis SAB1: A Source of Antimicrobial Compounds. Mar. Drugs 2010, 8, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Jadulco, R.; Brauers, G.; Edrada, R.A.; Ebel, R.; Wray, V.; Proksch, P. New Metabolites from Sponge-Derived Fungi Curvularia lunata and Cladosporium herbarum II. J. Natl. Prod. 2002, 65, 730–733. [Google Scholar] [CrossRef]
- Phelan, R.W.; Barret, M.; Cotter, P.D.; O’Connor, P.M.; Chen, R.; Morrissey, J.P.; Dobson, A.D.W.; O’Gara, F.; Barbosa, T.M. Subtilomycin: A New Lantibiotic from Bacillus subtilis Strain MMA7 Isolated from the Marine Sponge Haliclona simulans. Mar. Drugs 2013, 11, 1878–1898. [Google Scholar] [CrossRef] [PubMed]
- Harrington, C.; Reen, F.; Mooij, M.; Stewart, F.; Chabot, J.-B.; Guerra, A.; Glöckner, F.; Nielsen, K.; Gram, L.; Dobson, A.; et al. Characterisation of Non-Autoinducing Tropodithietic Acid (TDA) Production from Marine Sponge Pseudovibrio Species. Mar. Drugs 2014, 12, 5960–5978. [Google Scholar] [CrossRef] [PubMed]
- Selvin, J. Exploring the Antagonistic Producer Streptomyces MSI051: Implications of Polyketide Synthase Gene Type II and a Ubiquitous Defense Enzyme Phospholipase A2 in the Host Sponge Dendrilla nigra. Curr. Microbiol. 2009, 58, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Yan, X.; Xu, J.; Chen, H.; Lin, W. Hymeniacidon perleve associated bioactive bacterium Pseudomonas sp. NJ6–3-1. Appl. Biochem. Microbiol. 2005, 41, 29–33. [Google Scholar] [CrossRef]
- Meenupriya, J.; Thangaraj, M. Isolation and molecular characterization of bioactive secondary metabolites from Callyspongia spp. associated fungi. Asian Pac. J. Trop. Med. 2010, 3, 738–740. [Google Scholar] [CrossRef]
- Ye, L.; Santos-Gandelman, J.; Hardoim, C.P.; George, I.; Cornelis, P.; Laport, M. Antibacterial activity and mutagenesis of sponge-associated Pseudomonas fluorescens H41. Antonie Leeuwenhoek 2015, 108, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Santos, O.C.S.; Pontes, P.V.M.L.; Santos, J.F.M.; Muricy, G.; Giambiagi-deMarval, M.; Laport, M.S. Isolation, characterization and phylogeny of sponge-associated bacteria with antimicrobial activities from Brazil. Res. Microbiol. 2010, 161, 604–612. [Google Scholar] [CrossRef] [PubMed]
- O’ Halloran, J.A.; Barbosa, T.M.; Morrissey, J.P.; Kennedy, J.; O’ Gara, F.; Dobson, A.D.W. Diversity and antimicrobial activity of Pseudovibrio spp. from Irish marine sponges. J. Appl. Microbiol. 2011, 110, 1495–1508. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Garson, M.J.; Fuerst, J.A. Marine actinomycetes related to the “Salinospora“ group from the Great Barrier Reef sponge Pseudoceratina clavata. Environ. Microbiol. 2005, 7, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Selvin, J.; Joseph, S.; Asha, K.R.T.; Manjusha, W.A.; Sangeetha, V.S.; Jayaseema, D.M.; Antony, M.C.; Vinitha, A.J.D. Antibacterial potential of antagonistic Streptomyces sp. isolated from marine sponge Dendrilla nigra. FEMS Microbiol. Ecol. 2004, 50, 117–122. [Google Scholar] [PubMed]
- Su, P.; Wang, D.-X.; Ding, S.-X.; Zhao, J. Isolation and diversity of natural product biosynthetic genes of cultivable bacteria associated with marine sponge Mycale sp. from the coast of Fujian, China. Can. J. Microbiol. 2014, 60, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, U.R.; Pimentel-Elardo, S.M.; Hanora, A.; Radwan, M.; Abou-El-Ela, S.H.; Ahmed, S.; Hentschel, U. Isolation, Phylogenetic Analysis and Anti-infective Activity Screening of Marine Sponge-Associated Actinomycetes. Mar. Drugs 2010, 8, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Flemer, B.; Kennedy, J.; Margassery, L.M.; Morrissey, J.P.; O’Gara, F.; Dobson, A.D.W. Diversity and antimicrobial activities of microbes from two Irish marine sponges, Suberites carnosus and Leucosolenia sp. J. Appl. Microbiol. 2012, 112, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Hentschel, U.; Schmid, M.; Wagner, M.; Fieseler, L.; Gernert, C.; Hacker, J. Isolation and phylogenetic analysis of bacteria with antimicrobial activities from the Mediterranean sponges Aplysina aerophoba and Aplysina cavernicola. FEMS Microbiol. Ecol. 2001, 35, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Gopi, M.; Kumaran, S.; Kumar, T.T.A.; Deivasigamani, B.; Alagappan, K.; Prasad, S.G. Antibacterial potential of sponge endosymbiont marine Enterobacter sp. at Kavaratti Island, Lakshadweep archipelago. Asian Pac. J. Trop. Med. 2012, 5, 142–146. [Google Scholar] [CrossRef]
- Chelossi, E.; Milanese, M.; Milano, A.; Pronzato, R.; Riccardi, G. Characterisation and antimicrobial activity of epibiotic bacteria from Petrosia ficiformis (Porifera, Demospongiae). J. Exp. Mar. Biol. Ecol. 2004, 309, 21–33. [Google Scholar] [CrossRef]
- Skariyachan, S.; Rao, A.G.; Patil, M.R.; Saikia, B.; Bharadwaj Kn, V.; Rao Gs, J. Antimicrobial potential of metabolites extracted from bacterial symbionts associated with marine sponges in coastal area of Gulf of Mannar Biosphere, India. Lett. Appl. Microbiol. 2014, 58, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.; Baker, P.; Piper, C.; Cotter, P.; Walsh, M.; Mooij, M.; Bourke, M.; Rea, M.; O’Connor, P.; Ross, R.P.; et al. Isolation and Analysis of Bacteria with Antimicrobial Activities from the Marine Sponge Haliclona simulans Collected from Irish Waters. Mar. Biotechnol. 2009, 11, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Kunz, A.L.; Labes, A.; Wiese, J.; Bruhn, T.; Bringmann, G.; Imhoff, J.F. Nature’s Lab for Derivatization: New and Revised Structures of a Variety of Streptophenazines Produced by a Sponge-Derived Streptomyces Strain. Mar. Drugs 2014, 12, 1699–1714. [Google Scholar] [CrossRef] [PubMed]
- Scopel, M.; Abraham, W.-R.; Henriques, A.T.; Macedo, A.J. Dipeptide cis-cyclo(Leucyl-Tyrosyl) produced by sponge associated Penicillium sp. F37 inhibits biofilm formation of the pathogenic Staphylococcus epidermidis. Bioorg. Med. Chem. Lett. 2013, 23, 624–626. [Google Scholar] [CrossRef] [PubMed]
- Manilal, A.; Sabarathnam, B.; Kiran, G.S.; Sujith, S.; Shakir, C.; Selvin, J. Antagonistic Potentials of Marine Sponge Associated Fungi Aspergillus clavatus MFD15. Asian J. Med. Sci. 2010, 2, 195–200. [Google Scholar]
- Gandhimathi, R.; Arunkumar, M.; Selvin, J.; Thangavelu, T.; Sivaramakrishnan, S.; Kiran, G.S.; Shanmughapriya, S.; Natarajaseenivasan, K. Antimicrobial potential of sponge associated marine actinomycetes. J. Med. Mycol. 2008, 18, 16–22. [Google Scholar] [CrossRef]
- Selvin, J.; Shanmughapriya, S.; Gandhimathi, R.; Seghal Kiran, G.; Rajeetha Ravji, T.; Natarajaseenivasan, K.; Hema, T.A. Optimization and production of novel antimicrobial agents from sponge associated marine actinomycetes Nocardiopsis dassonvillei MAD08. Appl. Microbiol. Biotechnol. 2009, 83, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.J.; Kwon, H.C.; Ham, J.; Yang, H.O. 6-Hydroxymethyl-1-phenazine-carboxamide and 1,6-phenazinedimethanol from a marine bacterium, Brevibacterium sp. KMD 003, associated with marine purple vase sponge. J. Antibiot. 2009, 62, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Viegelmann, C.; Margassery, L.M.; Kennedy, J.; Zhang, T.; O’Brien, C.; O’Gara, F.; Morrissey, J.P.; Dobson, A.D.W.; Edrada-Ebel, R. Metabolomic Profiling and Genomic Study of a Marine Sponge-Associated Streptomyces sp. Mar. Drugs 2014, 12, 3323–3351. [Google Scholar] [CrossRef] [PubMed]
- Anand, T.P.; Bhat, A.W.; Shouche, Y.S.; Roy, U.; Siddharth, J.; Sarma, S.P. Antimicrobial activity of marine bacteria associated with sponges from the waters off the coast of South East India. Microbiol. Res. 2006, 161, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Margassery, L.M.; Kennedy, J.; O’Gara, F.; Dobson, A.D.; Morrissey, J.P. Diversity and antibacterial activity of bacteria isolated from the coastal marine sponges Amphilectus fucorum and Eurypon major. Lett. Appl. Microbiol. 2012, 55, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Dashti, Y.; Grkovic, T.; Abdelmohsen, U.R.; Hentschel, U.; Quinn, R.J. Production of Induced Secondary Metabolites by a Co-Culture of Sponge-Associated Actinomycetes, Actinokineospora sp. EG49 and Nocardiopsis sp. RV163. Mar. Drugs 2014, 12, 3046–3059. [Google Scholar] [CrossRef] [PubMed]
- Rachanamol, R.S.; Lipton, A.P.; Thankamani, V.; Sarika, A.R.; Selvin, J. Molecular characterization and bioactivity profile of the tropical sponge-associated bacterium Shewanella algae VCDB. Helgol. Mar. Res. 2014, 68, 263–269. [Google Scholar] [CrossRef]
- Dupont, S.; Carre-Mlouka, A.; Domart-Coulon, I.; Vacelet, J.; Bourguet-Kondracki, M.-L. Exploring cultivable Bacteria from the prokaryotic community associated with the carnivorous sponge Asbestopluma hypogea. FEMS Microbiol. Ecol. 2014, 88, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Carré-Mlouka, A.; Descarrega, F.; Ereskovsky, A.; Longeon, A.; Mouray, E.; Florent, I.; Bourguet-Kondracki, M.L. Diversity and biological activities of the bacterial community associated with the marine sponge Phorbas tenacior (Porifera, Demospongiae). Lett. Appl. Microbiol. 2014, 58, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Karkowska-Kuleta, J.; Rapala-Kozik, M.; Kozik, A. Fungi pathogenic to humans: Molecular bases of virulence of Candida albicans, Cryptococcus neoformans and Aspergillus fumigatus. Acta Biochim. Pol. 2009, 56, 211–224. [Google Scholar] [PubMed]
- Van Thiel, D.H.; George, M.; Moore, C.M. Fungal Infections: Their Diagnosis and Treatment in Transplant Recipients. Int. J. Hepatol. 2012, 2012, 106923. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.D. Changing patterns and trends in systemic fungal infections. J. Antimicrob. Chemoth. 2005, 56, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Fidel, P.L.; Vazquez, J.A.; Sobel, J.D. Candida glabrata: Review of Epidemiology, Pathogenesis, and Clinical Disease with Comparison to C. albicans. Clin. Microbiol. Rev. 1999, 12, 80–96. [Google Scholar] [PubMed]
- Galimberti, R.; Torre, A.C.; Baztán, M.C.; Rodriguez-Chiappetta, F. Emerging systemic fungal infections. Clin. Dermatol. 2012, 30, 633–650. [Google Scholar] [CrossRef] [PubMed]
- El-Gendy, M.A.; El-Bondkly, A.A. Production and genetic improvement of a novel antimycotic agent, Saadamycin, against Dermatophytes and other clinical fungi from Endophytic Streptomyces sp. Hedaya48. J. Ind. Microbiol. Biotechnol. 2010, 37, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Kamigiri, K.; Matsumoto, H.; Kawano, Y.; Yamaoka, M.; Shimoi, H.; Watanabe, M.; Suzuki, K. YM-202204, a new antifungal antibiotic produced by marine fungus Phoma sp. J. Antibiot. 2002, 55, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- McLellan, C.A.; Whitesell, L.; King, O.D.; Lancaster, A.K.; Mazitschek, R.; Lindquist, S. Inhibiting GPI Anchor Biosynthesis in Fungi Stresses the Endoplasmic Reticulum and Enhances Immunogenicity. ACS Chem. Biol. 2012, 7, 1520–1528. [Google Scholar] [CrossRef] [PubMed]
- Butts, A.; Krysan, D.J. Antifungal Drug Discovery: Something Old and Something New. PLoS Pathog. 2012, 8. [Google Scholar] [CrossRef] [PubMed]
- Edrada, R.A.; Heubes, M.; Brauers, G.; Wray, V.; Berg, A.; Grafe, U.; Wohlfarth, M.; Muhlbacher, J.; Schaumann, K.; Sudarsono, S.; et al. Online analysis of xestodecalactones A–C, novel bioactive metabolites from the fungus Penicillium cf. montanense and their subsequent isolation from the sponge Xestospongia exigua. J. Natl. Prod. 2002, 65, 1598–1604. [Google Scholar] [CrossRef]
- Imamura, N.; Nishijima, M.; Adachi, K.; Sano, H. Novel Antimycin Antibiotics, Urauchimycin-a and Urauchimycin-B, Produced by Marine Actinomycete. J. Antibiot. 1993, 46, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.T.; Takagi, M.; Shin-ya, K. Diversity, Salt Requirement, and Antibiotic Production of Actinobacteria Isolated from Marine Sponges. Actinomycetologica 2010, 24, 18–23. [Google Scholar] [CrossRef]
- Cohen, E.; Koch, L.; Thu, K.M.; Rahamim, Y.; Aluma, Y.; Ilan, M.; Yarden, O.; Carmeli, S. Novel terpenoids of the fungus Aspergillus insuetus isolated from the Mediterranean sponge Psammocinia sp. collected along the coast of Israel. Bioorg. Med. Chem. 2011, 19, 6587–6593. [Google Scholar] [CrossRef] [PubMed]
- Holler, U.; Konig, G.M.; Wright, A.D. Three new metabolites from marine-derived fungi of the genera Coniothyrium and Microsphaeropsis. J. Natl. Prod. 1999, 62, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.K.H.; Holmes, M.J.; Higa, T.; Hamann, M.T.; Kara, U.A.K. In vivo antimalarial activity of the beta-carboline alkaloid manzamine A. Antimicrob. Agents Chim. 2000, 44, 1645–1649. [Google Scholar] [CrossRef]
- Sipkema, D.; Franssen, M.C.R.; Osinga, R.; Tramper, J.; Wijffels, R.H. Marine sponges as pharmacy. Mar. Biotechnol. 2005, 7, 142–162. [Google Scholar] [CrossRef] [PubMed]
- Fattorusso, E.; Taglialatela-Scafati, O. Marine Antimalarials. Mar. Drugs 2009, 7, 130–152. [Google Scholar] [CrossRef] [PubMed]
- Sakai, R.; Higa, T.; Jefford, C.W.; Bernardinelli, G. Manzamine A, a novel antitumor alkaloid from a sponge. J. Am. Chem. Soc. 1986, 108, 6404–6405. [Google Scholar] [CrossRef]
- Radwan, M.; Hanora, A.; Khalifa, S.; Abou-El-Ela, S.H. Manzamines. Cell Cycle 2012, 11, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Eyase, F.L.; Akala, H.M.; Johnson, J.D.; Walsh, D.S. Inhibitory Activity of Ferroquine, versus Chloroquine, against Western Kenya Plasmodium falciparum Field Isolates Determined by a SYBR Green I in Vitro Assay. Am. J. Trop. Med. Hyg. 2011, 85, 984–988. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.V.; Santarsiero, B.D.; Mesecar, A.D.; Schinazi, R.F.; Tekwani, B.L.; Hamann, M.T. New manzamine alkaloids with activity against infectious and tropical parasitic diseases from an Indonesian sponge. J. Natl. Prod. 2003, 66, 823–828. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Chen, Y.-J.; Aoki, S.; In, Y.; Ishida, T.; Kitagawa, I. Four new β-carboline alkaloids isolated from two Okinawan marine sponges of Xestospongia sp. and Haliclona sp. Tetrahedron 1995, 51, 3727–3736. [Google Scholar] [CrossRef]
- Rao, K.V.; Kasanah, N.; Wahyuono, S.U.; Tekwani, B.L.; Schinazi, R.F.; Hamann, M.T. Three new manzamine alkaloids from a common Indonesian sponge and their activity against infectious and tropical parasitic diseases. J. Natl. Prod. 2004, 67, 1314–1318. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.T.; Hamann, M.; Peraud, O.T.; Kasanah, N. Manzamine Producing Actinomycetes. U.S. Patent 20050244938 A1, 3 November 2005. [Google Scholar]
- Peraud, O. Isolation and Characterization of a Sponge-Associated Actinomycete that Produces Manzamines. University of Maryland, 2006. Available online: http://drum.lib.umd.edu/handle/1903/4114 (accessed on 1 September 2015).
- Waters, A.L.; Peraud, O.; Kasanah, N.; Sims, J.; Kothalawala, N.; Anderson, M.A.; Abbas, S.H.; Rao, K.V.; Jupally, V.R.; Kelly, M.; et al. An analysis of the sponge Acanthostrongylophora igens’ microbiome yields an actinomycete that produces the natural product manzamine A. Front. Mar. Sci. 2014, 1. [Google Scholar] [CrossRef]
- Pimentel-Elardo, S.M.; Kozytska, S.; Bugni, T.S.; Ireland, C.M.; Moll, H.; Hentschel, U. Anti-Parasitic Compounds from Streptomyces sp. Strains Isolated from Mediterranean Sponges. Mar. Drugs 2010, 8, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Scopel, M.; dos Santos, O.; Frasson, A.P.; Abraham, W.-R.; Tasca, T.; Henriques, A.T.; Macedo, A.J. Anti-Trichomonas vaginalis activity of marine-associated fungi from the South Brazilian Coast. Exp. Parasitol. 2013, 133, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Petrin, D.; Delgaty, K.; Bhatt, R.; Garber, G. Clinical and microbiological aspects of Trichomonas vaginalis. Clin. Microbiol Rev. 1998, 11, 300. [Google Scholar] [PubMed]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of marine sponge Hyattella intestinalis associated bacteria against Plasmodium falciparum. Asian Pac. J. Trop. Biomed. 2011, 1, S100–S104. [Google Scholar] [CrossRef]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of marine sponge Stylissa carteri associated bacteria against Plasmodium falciparum. Asian Pac. J. Trop. Dis. 2012, 2, 370–374. [Google Scholar] [CrossRef]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of marine sponge Clathria indica associated bacteria against Plasmodium falciparum. Asian Pac. J. Trop. Biomed. 2012, 2, S1090–S1095. [Google Scholar] [CrossRef]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of Clathria vulpina sponge associated bacteria against Plasmodium falciparum. Asian Pac. J. Trop. Dis. 2012, 2, 319–323. [Google Scholar] [CrossRef]
- Inbaneson, S.J.; Ravikumar, S. In vitro antiplasmodial activity of bacterium RJAUTHB 14 associated with marine sponge Haliclona Grant against Plasmodium falciparum. Parasitol. Res. 2012, 110, 2255–2262. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, U.R.; Szesny, M.; Othman, E.M.; Schirmeister, T.; Grond, S.; Stopper, H.; Hentschel, U. Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115. Mar. Drugs 2012, 10, 2208–2221. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, U.R.; Cheng, C.; Viegelmann, C.; Zhang, T.; Grkovic, T.; Ahmed, S.; Quinn, R.J.; Hentschel, U.; Edrada-Ebel, R. Dereplication Strategies for Targeted Isolation of New Antitrypanosomal Actinosporins A and B from a Marine Sponge Associated-Actinokineospora sp. EG49. Mar. Drugs 2014, 12, 1220–1244. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; MacIntyre, L.; Abdelmohsen, U.R.; Horn, H.; Polymenakou, P.N.; Edrada-Ebel, R.; Hentschel, U. Biodiversity, Anti-Trypanosomal Activity Screening, and Metabolomic Profiling of Actinomycetes Isolated from Mediterranean Sponges. PLoS ONE 2015, 10, e0138528. [Google Scholar] [CrossRef] [PubMed]
- Pimentel-Elardo, S.M.; Buback, V.; Gulder, T.A.M.; Bugni, T.S.; Reppart, J.; Bringmann, G.; Ireland, C.M.; Schirmeister, T.; Hentschel, U. New Tetromycin Derivatives with Anti-Trypanosomal and Protease Inhibitory Activities. Mar. Drugs 2011, 9, 1682–1697. [Google Scholar] [CrossRef] [PubMed]
- Ashforth, E.J.; Fu, C.Z.; Liu, X.Y.; Dai, H.Q.; Song, F.H.; Guo, H.; Zhang, L.X. Bioprospecting for antituberculosis leads from microbial metabolites. Natl. Prod. Rep. 2010, 27, 1709–1719. [Google Scholar] [CrossRef] [PubMed]
- Seipke, R.F.; Kaltenpoth, M.; Hutchings, M.I. Streptomyces as symbionts: An emerging and widespread theme? FEMS Microbiol. Rev. 2012, 36, 862–876. [Google Scholar] [CrossRef] [PubMed]
- Traxler, M.F.; Kolter, R. Natural products in soil microbe interactions and evolution. Natl. Prod. Rep. 2015, 32, 956–970. [Google Scholar] [CrossRef] [PubMed]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism—From biochemistry to genomics. Nat. Rev. Microb. 2005, 3, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Laursen, J.B.; Nielsen, J. Phenazine Natural Products: Biosynthesis, Synthetic Analogues, and Biological Activity. Chem. Rev. 2004, 104, 1663–1686. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Préville, P.; Morin, N.; Mounir, S.; Cai, W.; Siddiqui, M.A. Hepatitis C viral IRES inhibition by phenazine and phenazine-like molecules. Bioorg. Med.Chem. Lett. 2000, 10, 1151–1154. [Google Scholar] [CrossRef]
- Mavrodi, D.V.; Mavrodi, O.V.; Parejko, J.A.; Bonsall, R.F.; Kwak, Y.S.; Paulitz, T.C.; Thomashow, L.S.; Weller, D.M. Accumulation of the Antibiotic Phenazine-1-Carboxylic Acid in the Rhizosphere of Dryland Cereals. Appl. Environ. Microb. 2012, 78, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Makgatho, M.E.; Anderson, R.; O’Sullivan, J.F.; Egan, T.J.; Freese, J.A.; Cornelius, N.; van Rensburg, C.E.J. Tetramethylpiperidine-substituted phenazines as novel anti-plasmodial agents. Drug Dev. Res. 2000, 50, 195–202. [Google Scholar] [CrossRef]
- Gao, X.; Lu, Y.; Xing, Y.; Ma, Y.; Lu, J.; Bao, W.; Wang, Y.; Xi, T. A novel anticancer and antifungus phenazine derivative from a marine actinomycete BM-17. Microbiol. Res. 2012, 167, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Hentschel, U.; Piel, J.; Degnan, S.M.; Taylor, M.W. Genomic insights into the marine sponge microbiome. Nat. Rev. Microbiol. 2012, 10, 641–675. [Google Scholar] [CrossRef] [PubMed]
- Schippers, K.J.; Sipkema, D.; Osinga, R.; Smidt, H.; Pomponi, S.A.; Martens, D.E.; Wijffels, R.H. Cultivation of sponges, sponge cells and symbionts: Achievements and future prospects. Adv. Mar. Biol. 2012, 62, 273–337. [Google Scholar] [PubMed]
- Ozturk, B.; de Jaeger, L.; Smidt, H.; Sipkema, D. Culture-dependent and independent approaches for identifying novel halogenases encoded by Crambe crambe (marine sponge) microbiota. Sci. Rep. UK 2013, 3. [Google Scholar] [CrossRef]
- Sipkema, D.; Schippers, K.; Maalcke, W.J.; Yang, Y.; Salim, S.; Blanch, H.W. Multiple Approaches To Enhance the Cultivability of Bacteria Associated with the Marine Sponge Haliclona (gellius) sp. Appl. Environ. Microb. 2011, 77, 2130–2140. [Google Scholar] [CrossRef] [PubMed]
- Muller, W.E.G.; Zahn, R.K.; Kurelec, B.; Lucu, C.; Muller, I.; Uhlenbruck, G. Lectin, a Possible Basis for Symbiosis between Bacteria and Sponges. J. Bacteriol. 1981, 145, 548–558. [Google Scholar] [PubMed]
- Steinert, G.; Whitfield, S.; Taylor, M.; Thoms, C.; Schupp, P. Application of Diffusion Growth Chambers for the Cultivation of Marine Sponge-Associated Bacteria. Mar. Biotechnol. 2014, 16, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Blasiak, L.C.; Karolin, J.O.; Powell, R.J.; Geddes, C.D.; Hill, R.T. Phosphorus sequestration in the form of polyphosphate by microbial symbionts in marine sponges. Proc. Natl. Acad.Sci. USA 2015, 112, 4381–4386. [Google Scholar] [CrossRef] [PubMed]
- Unson, M.D.; Faulkner, D.J. Cyanobacterial Symbiont Biosynthesis of Chlorinated Metabolites from Dysidea-Herbacea (Porifera). Experientia 1993, 49, 349–353. [Google Scholar] [CrossRef]
- Milshteyn, A.; Schneider, J.S.; Brady, S.F. Mining the Metabiome: Identifying Novel Natural Products from Microbial Communities. Chem. Biol. 2014, 21, 1211–1223. [Google Scholar] [CrossRef] [PubMed]
- Marmann, A.; Aly, A.; Lin, W.; Wang, B.; Proksch, P. Co-Cultivation—A Powerful Emerging Tool for Enhancing the Chemical Diversity of Microorganisms. Mar. Drugs 2014, 12, 1043. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, S.; Bohni, N.; Schnee, S.; Schumpp, O.; Gindro, K.; Wolfender, J.-L. Metabolite induction via microorganism co-culture: A potential way to enhance chemical diversity for drug discovery. Biotechnol. Adv. 2014, 32, 1180–1204. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-Y.-S.; Graziani, E.; Waters, B.; Pan, W.; Li, X.; McDermott, J.; Meurer, G.; Saxena, G.; Andersen, R.J.; Davies, J. Novel Natural Products from Soil DNA Libraries in a Streptomycete Host. Organ. Lett. 2000, 2, 2401–2404. [Google Scholar] [CrossRef]
- Brady, S.F.; Chao, C.J.; Handelsman, J.; Clardy, J. Cloning and heterologous expression of a natural product biosynthetic gene cluster from eDNA. Organ. Lett. 2001, 3, 1981–1984. [Google Scholar] [CrossRef]
- Chung, E.J.; Lim, H.K.; Kim, J.C.; Choi, G.J.; Park, E.J.; Lee, M.H.; Chung, Y.R.; Lee, S.W. Forest soil metagenome gene cluster involved in antifungal activity expression in Escherichia coli. Appl. Environ. Microb. 2008, 74, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.; Daniel, R. Metagenomic Analyses: Past and Future Trends. Appl. Environ. Microb. 2011, 77, 1153–1161. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.C.; Piel, J. Metagenomic Approaches for Exploiting Uncultivated Bacteria as a Resource for Novel Biosynthetic Enzymology. Chem. Biol. 2013, 20, 636–647. [Google Scholar] [CrossRef] [PubMed]
- Piel, J. Approaches to Capturing and Designing Biologically Active Small Molecules Produced by Uncultured Microbes. Annu. Rev. Microbiol. 2011, 65, 431–453. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, I.A.; Tiong, C.L.; Minor, C.; August, P.R.; Grossman, T.H.; Loiacono, K.A.; Lynch, B.A.; Phillips, T.; Narula, S.; Sundaramoorthi, R.; et al. Expression and isolation of antimicrobial small molecules from soil DNA libraries. J. Mol. Microb. Biotech. 2001, 3, 301–308. [Google Scholar]
- Yung, P.Y.; Burke, C.; Lewis, M.; Kjelleberg, S.; Thomas, T. Novel Antibacterial Proteins from the Microbial Communities Associated with the Sponge Cymbastela concentrica and the Green Alga Ulva australis. Appl. Environ. Microb. 2011, 77, 1512–1515. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Wakimoto, T.; Egami, Y.; Kenmoku, H.; Ito, T.; Asakawa, Y.; Abe, I. Heterologously expressed β-hydroxyl fatty acids from a metagenomic library of a marine sponge. Bioorg. Med. Chem. Lett. 2012, 22, 7322–7325. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Wang, B.C.; Wakimoto, T.; Wang, M.Y.; Zhu, L.C.; Abe, I. Cyclodipeptides from Metagenomic Library of a Japanese Marine Sponge. J. Brazil. Chem. Soc. 2013, 24, 1926. [Google Scholar] [CrossRef]
- Ongley, S.E.; Bian, X.Y.; Neilan, B.A.; Muller, R. Recent advances in the heterologous expression of microbial natural product biosynthetic pathways. Natl. Prod. Rep. 2013, 30, 1121–1138. [Google Scholar] [CrossRef] [PubMed]
- Baltz, R.H. Molecular engineering approaches to peptide, polyketide and other antibiotics. Nat. Biotechnol. 2006, 24, 1533–1540. [Google Scholar] [CrossRef] [PubMed]
- Dobson, A.W.; Jackson, S.; Kennedy, J.; Margassery, L.; Flemer, B.; O’Leary, N.; Morrissey, J.; O’Gara, F. Marine Sponges—Molecular Biology and Biotechnology. In Springer Handbook of Marine Biotechnology; Kim, S.-K., Ed.; Springer Berlin Heidelberg: Heidelberg, Germany, 2015; pp. 219–254. [Google Scholar]
- Baltz, R.H. Streptomyces and Saccharopolyspora hosts for heterologous expression of secondary metabolite gene clusters. J. Ind. Microbiol. Biot. 2010, 37, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.; Marchesi, J.R.; Dobson, A.D.W. Metagenomic approaches to exploit the biotechnological potential of the microbial consortia of marine sponges. Appl. Microbiol. Biotechnol. 2007, 75, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Piel, J. A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc. Natl. Acad. Sci. USA 2002, 99, 14002–14007. [Google Scholar] [CrossRef] [PubMed]
- Piel, J.; Hui, D.; Wen, G.; Butzke, D.; Platzer, M.; Fusetani, N.; Matsunaga, S. Antitumor polyketide biosynthesis by an uncultivated bacterial symbiont of the marine sponge Theonella swinhoei. Proc. Natl. Acad. Sci. USA 2004, 101, 16222–16227. [Google Scholar] [CrossRef] [PubMed]
- Piel, J.; Hui, D.Q.; Fusetani, N.; Matsunaga, S. Targeting modular polyketide synthases with iteratively acting acyltransferases from metagenomes of uncultured bacterial consortia. Environ. Microbiol. 2004, 6, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Piel, J.; Wen, G.; Platzer, M.; Hui, D. Unprecedented Diversity of Catalytic Domains in the First Four Modules of the Putative Pederin Polyketide Synthase. ChemBioChem. 2004, 5, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Piel, J.; Butzke, D.; Fusetani, N.; Hui, D.; Platzer, M.; Wen, G.; Matsunaga, S. Exploring the Chemistry of Uncultivated Bacterial Symbionts: Antitumor Polyketides of the Pederin Family. J. Natl. Prod. 2005, 68, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Fisch, K.M.; Gurgui, C.; Heycke, N.; van der Sar, S.A.; Anderson, S.A.; Webb, V.L.; Taudien, S.; Platzer, M.; Rubio, B.K.; Robinson, S.J.; et al. Polyketide assembly lines of uncultivated sponge symbionts from structure-based gene targeting. Nat. Chem. Biol. 2009, 5, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, A.; Gadkari, R.; Reeves, C.D.; Ibrahim, F.; DeLong, E.F.; Hutchinson, C.R. Metagenomic analysis reveals diverse polyketide synthase gene clusters in microorganisms associated with the marine sponge Discodermia dissoluta. Appl. Environ. Microb. 2005, 71, 4840–4849. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, A.; Au, K.F. PacBio Sequencing and Its Applications. Genomics Proteom. Bioinform. 2015, 13, 278–289. [Google Scholar] [CrossRef] [PubMed]
- Alt, S.; Wilkinson, B. Biosynthesis of the Novel Macrolide Antibiotic Anthracimycin. Acs Chem. Biol. 2015, 10, 2468–2479. [Google Scholar] [CrossRef] [PubMed]
- Podar, M.; Abulencia, C.B.; Walcher, M.; Hutchison, D.; Zengler, K.; Garcia, J.A.; Holland, T.; Cotton, D.; Hauser, L.; Keller, M. Targeted access to the genomes of low-abundance organisms in complex microbial communities. Appl. Environ. Microb. 2007, 73, 3205–3214. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.C.; Mori, T.; Ruckert, C.; Uria, A.R.; Helf, M.J.; Takada, K.; Gernert, C.; Steffens, U.A.E.; Heycke, N.; Schmitt, S.; et al. An environmental bacterial taxon with a large and distinct metabolic repertoire. Nature 2014, 506, 58. [Google Scholar] [CrossRef] [PubMed]
- Banik, J.J.; Brady, S.F. Recent application of metagenomic approaches toward the discovery of antimicrobials and other bioactive small molecules. Curr. Opin. Microbiol. 2010, 13, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Medema, M.H.; Blin, K.; Cimermancic, P.; de Jager, V.; Zakrzewski, P.; Fischbach, M.A.; Weber, T.; Takano, E.; Breitling, R. antiSMASH: Rapid identification, annotation and analysis of secondary metabolite biosynthesis gene clusters in bacterial and fungal genome sequences. Nucleic Acids Res. 2011, 39, W339–W346. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.; Ryu, S. Screening for novel enzymes from metagenome and SIGEX, as a way to improve it. Microb. Cell Fact. 2005, 4, 8. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).