Elevated Epicardial Adipose Tissue and Ischemic Stroke Risk: A Systematic Review and Meta-Analysis †
Abstract
1. Introduction
2. Materials and Methods
2.1. EAT Measurement and Imaging Modalities
2.2. Search Strategy and Data Sources
2.3. Study Selection and Inclusion Criteria
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
3.1. Systematic Search and Quality Assessment
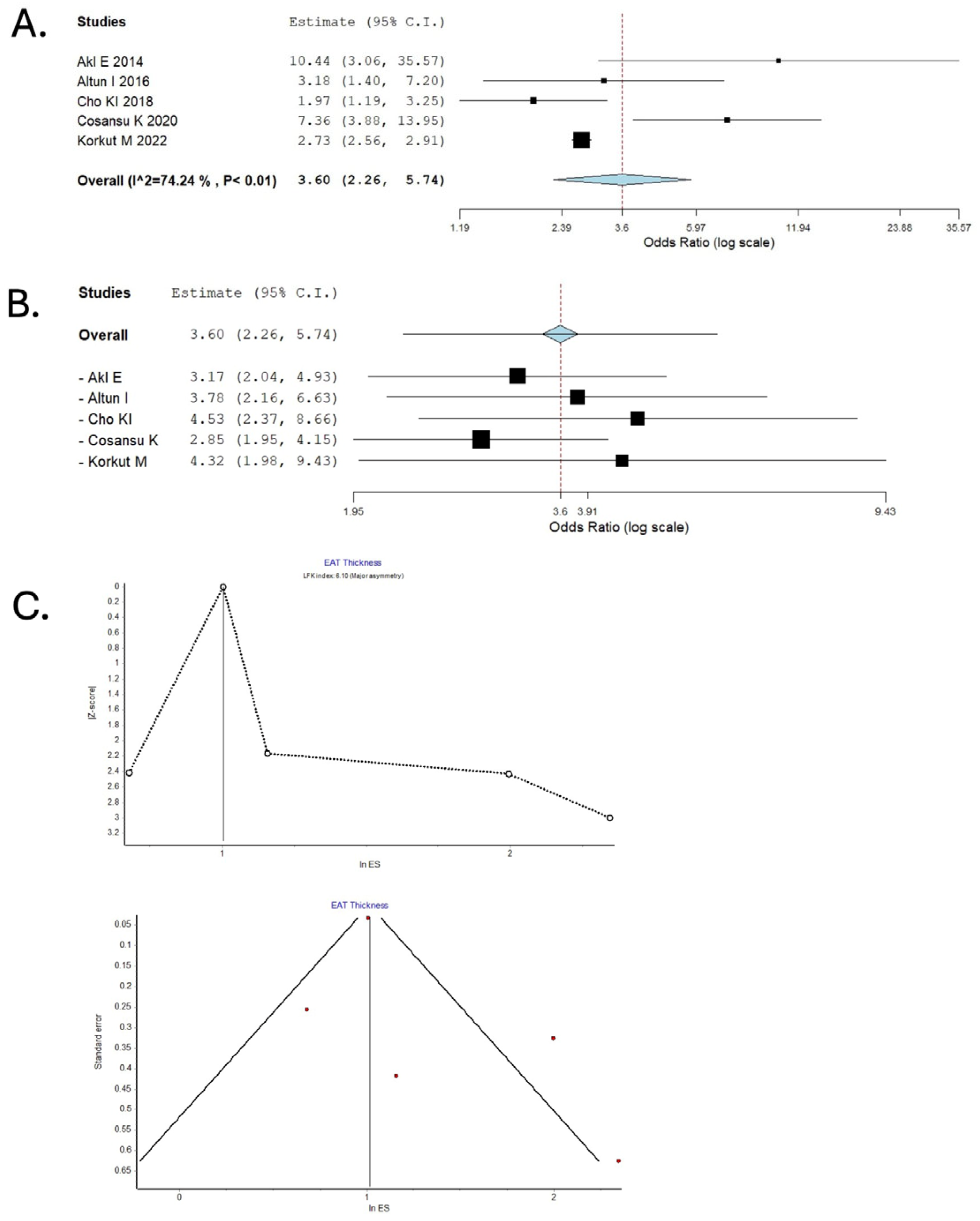
3.2. Epicardial Adipose Thickness and Acute Ischemic Stroke
3.3. Epicardial Adipose Volume and Acute Ischemic Stroke
4. Discussion
4.1. Clinical Implications
4.2. Strengths and Limitations
4.3. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Collaborators GBDS. Global, regional, and national burden of stroke, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 439–458. [Google Scholar] [CrossRef]
- Rodriguez-Granillo, G.A.; Cirio, J.J.; Ciardi, C.; Caballero, M.L.; Fontana, L.; Perez, N.; Ingino, C.A.; Lylyk, P. Epicardial and periaortic fat characteristics in ischemic stroke: Relationship with stroke etiology and calcification burden. Eur. J. Radiol. 2022, 146, 110102. [Google Scholar] [CrossRef]
- Chong, B.; Jayabaskaran, J.; Ruban, J.; Goh, R.; Chin, Y.H.; Kong, G.; Ng, C.H.; Lin, C.; Loong, S.; Muthiah, M.D.; et al. Epicardial Adipose Tissue Assessed by Computed Tomography and Echocardiography Are Associated with Adverse Cardiovascular Outcomes: A Systematic Review and Meta-Analysis. Circ. Cardiovasc. Imaging 2023, 16, e015159. [Google Scholar] [CrossRef]
- Iacobellis, G.; Lonn, E.; Lamy, A.; Singh, N.; Sharma, A.M. Epicardial fat thickness and coronary artery disease correlate independently of obesity. Int. J. Cardiol. 2011, 146, 452–454. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.W.; Tsai, I.C.; Lee, W.J.; Lin, S.Y.; Lee, W.L.; Lee, I.T.; Fu, C.-P.; Wang, J.-S.; Sheu, W.H.-H. Correlation between reduction of superior interventricular groove epicardial fat thickness and improvement of insulin resistance after weight loss in obese men. Diabetol. Metab. Syndr. 2014, 6, 115. [Google Scholar] [CrossRef] [PubMed]
- Kaya, H.; Ertas, F.; Oylumlu, M.; Bilik, M.Z.; Yildiz, A.; Yuksel, M.; Polat, N.; Acet, H.; Işık, F.; Ülgen, M.S. Relation of epicardial fat thickness and brachial flow-mediated vasodilation with coronary artery disease. J. Cardiol. 2013, 62, 343–347. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ouwens, D.M.; Sell, H.; Greulich, S.; Eckel, J. The role of epicardial and perivascular adipose tissue in the pathophysiology of cardiovascular disease. J. Cell. Mol. Med. 2010, 14, 2223–2234. [Google Scholar] [CrossRef]
- Chu, C.Y.; Lee, W.H.; Hsu, P.C.; Lee, M.K.; Lee, H.H.; Chiu, C.A.; Lin, T.-H.; Lee, C.-S.; Yen, H.-W.; Voon, W.-C.; et al. Association of Increased Epicardial Adipose Tissue Thickness with Adverse Cardiovascular Outcomes in Patients with Atrial Fibrillation. Medicine 2016, 95, e2874. [Google Scholar] [CrossRef]
- Ahn, J.; Shin, S.Y.; Shim, J.; Kim, Y.H.; Han, S.J.; Choi, E.K.; Oh, S.; Shin, J.Y.; Choe, J.C.; Park, J.S.; et al. Association between epicardial adipose tissue and embolic stroke after catheter ablation of atrial fibrillation. J. Cardiovasc. Electrophysiol. 2019, 30, 2209–2216. [Google Scholar] [CrossRef]
- Hendricks, S.; Dykun, I.; Balcer, B.; Totzeck, M.; Rassaf, T.; Mahabadi, A.A. Epicardial adipose tissue is a robust measure of increased risk of myocardial infarction—A meta-analysis on over 6600 patients and rationale for the EPIC-ACS study. Medicine 2021, 100, e28060. [Google Scholar] [CrossRef]
- Requena-Ibanez, J.A.; Santos-Gallego, C.G.; Rodriguez Cordero, A.J.; Fardman, B.; Sartori, S.; Sanz, J.; Fuster, V.; Badimon, J.J. Not only how much, but also how to, when measuring epicardial adipose tissue. Magn. Reson. Imaging 2022, 86, 149–151. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Grainger, M.J.; Gray, C.T. Citationchaser: A tool for transparent and efficient forward and backward citation chasing in systematic searching. Res. Synth. Methods 2022, 13, 533–545. [Google Scholar] [CrossRef]
- Akil, E.; Akil, M.A.; Varol, S.; Ozdemir, H.H.; Yucel, Y.; Arslan, D.; Akyüz, A.; Alan, S. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J. Stroke Cerebrovasc. Dis. 2014, 23, 2328–2334. [Google Scholar] [CrossRef] [PubMed]
- Altun, I.; Unal, Y.; Basaran, O.; Akin, F.; Emir, G.K.; Kutlu, G.; Biteker, M. Increased Epicardial Fat Thickness Correlates with Aortic Stiffness and N-Terminal Pro-Brain Natriuretic Peptide Levels in Acute Ischemic Stroke Patients. Tex. Heart Inst. J. 2016, 43, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Tsao, H.M.; Hu, W.C.; Tsai, P.H.; Lee, C.L.; Liu, F.C.; Wang, H.H.; Lo, L.-W.; Chang, S.-L.; Chao, T.-F.; Chen, S.-A. The Abundance of Epicardial Adipose Tissue Surrounding Left Atrium Is Associated with the Occurrence of Stroke in Patients with Atrial Fibrillation. Medicine 2016, 95, e3260. [Google Scholar] [CrossRef]
- Shah, R.V.; Anderson, A.; Ding, J.; Budoff, M.; Rider, O.; Petersen, S.E.; Jensen, M.K.; Koch, M.; Allison, M.; Kawel-Boehm, N.; et al. Pericardial, But Not Hepatic, Fat by CT Is Associated with CV Outcomes and Structure: The Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc. Imaging 2017, 10, 1016–1027. [Google Scholar] [CrossRef]
- Cho, K.I.; Kim, B.J.; Cho, S.H.; Lee, J.H.; Kim, M.K.; Yoo, B.G. Epicardial Fat Thickness and Free Fatty Acid Level are Predictors of Acute Ischemic Stroke with Atrial Fibrillation. J. Cardiovasc. Imaging 2018, 26, 65–74. [Google Scholar] [CrossRef]
- Cosansu, K.; Yilmaz, S. Is epicardial fat thickness associated with acute ischemic stroke in patients with atrial fibrillation? J. Stroke Cerebrovasc. Dis. 2020, 29, 104900. [Google Scholar] [CrossRef]
- Korkut, M.; Selvi, F.; Bedel, C. Echocardiographic epicardial fat thickness and immature granulocyte are novel inflammatory predictors of acute ischemic stroke: A prospective study. Sao Paulo Med. J. 2022, 140, 384–389. [Google Scholar] [CrossRef]
- West, H.W.; Siddique, M.; Williams, M.C.; Volpe, L.; Desai, R.; Lyasheva, M.; Thomas, S.; Dangas, K.; Kotanidis, C.P.; Tomlins, P.; et al. Deep-Learning for Epicardial Adipose Tissue Assessment with Computed Tomography: Implications for Cardiovascular Risk Prediction. JACC Cardiovasc. Imaging 2023, 16, 800–816. [Google Scholar] [CrossRef]
- Furuya-Kanamori, L.; Barendregt, J.J.; Doi, S.A.R. A new improved graphical and quantitative method for detecting bias in meta-analysis. Int. J. Evid. Based Healthc. 2018, 16, 195–203. [Google Scholar] [CrossRef]
- Ansaldo, A.M.; Montecucco, F.; Sahebkar, A.; Dallegri, F.; Carbone, F. Epicardial adipose tissue and cardiovascular diseases. Int. J. Cardiol. 2019, 278, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, N.; Moreno-Villegas, Z.; Gonzalez-Bris, A.; Egido, J.; Lorenzo, O. Regulation of visceral and epicardial adipose tissue for preventing cardiovascular injuries associated to obesity and diabetes. Cardiovasc. Diabetol. 2017, 16, 44. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Choi, S.Y.; Zheng, M.; Yang, H.M.; Lim, H.S.; Choi, B.J.; Yoon, M.-H.; Hwang, G.-S.; Tahk, S.-J.; Shin, J.-H. Epicardial adipose tissue thickness is a predictor for plaque vulnerability in patients with significant coronary artery disease. Atherosclerosis 2013, 226, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G.; Zaki, M.C.; Garcia, D.; Willens, H.J. Epicardial fat in atrial fibrillation and heart failure. Horm. Metab. Res. 2014, 46, 587–590. [Google Scholar] [CrossRef]
- Wang, T.; Liu, Q.; Liu, C.; Sun, L.; Li, D.; Liu, A.; Jia, R. Correlation of echocardiographic epicardial fat thickness with severity of coronary artery disease in patients with acute myocardial infarction. Echocardiography 2014, 31, 1177–1181. [Google Scholar] [CrossRef]
- Tanindi, A.; Erkan, A.F.; Ekici, B. Epicardial adipose tissue thickness can be used to predict major adverse cardiac events. Coron. Artery Dis. 2015, 26, 686–691. [Google Scholar] [CrossRef]
- Thanassoulis, G.; Massaro, J.M.; O’Donnell, C.J.; Hoffmann, U.; Levy, D.; Ellinor, P.T.; Wang, T.J.; Schnabel, R.B.; Vasan, R.S.; Fox, C.S.; et al. Pericardial fat is associated with prevalent atrial fibrillation: The Framingham Heart Study. Circ. Arrhythmia Electrophysiol. 2010, 3, 345–350. [Google Scholar] [CrossRef]
- Al Chekakie, M.O.; Welles, C.C.; Metoyer, R.; Ibrahim, A.; Shapira, A.R.; Cytron, J.; Santucci, P.; Wilber, D.J.; Akar, J.G. Pericardial fat is independently associated with human atrial fibrillation. J. Am. Coll. Cardiol. 2010, 56, 784–788. [Google Scholar] [CrossRef]
- Gurdal, A.; Keskin, K.; Orken, D.N.; Baran, G.; Kilickesmez, K. Evaluation of Epicardial Fat Thickness in Young Patients with Embolic Stroke of Undetermined Source. Neurologist 2018, 23, 113–117. [Google Scholar] [CrossRef]
- Chahine, Y.; Askari-Atapour, B.; Kwan, K.T.; Anderson, C.A.; Macheret, F.; Afroze, T.; Bifulco, S.F.; Cham, M.D.; Ordovas, K.; Boyle, P.M.; et al. Epicardial adipose tissue is associated with left atrial volume and fibrosis in patients with atrial fibrillation. Front. Cardiovasc. Med. 2022, 9, 1045730. [Google Scholar] [CrossRef]
- Zghaib, T.; Ipek, E.G.; Zahid, S.; Balouch, M.A.; Misra, S.; Ashikaga, H.; Berger, R.D.; Marine, J.E.; Spragg, D.D.; Zimmerman, S.L.; et al. Association of left atrial epicardial adipose tissue with electrogram bipolar voltage and fractionation: Electrophysiologic substrates for atrial fibrillation. Heart Rhythm 2016, 13, 2333–2339. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G. Epicardial adipose tissue in contemporary cardiology. Nat. Rev. Cardiol. 2022, 19, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Bakirci, E.M.; Degirmenci, H.; Duman, H.; Inci, S.; Hamur, H.; Buyuklu, M.; Ceyhun, G.; Topal, E. Increased Epicardial Adipose Tissue Thickness is Associated with Angiographic Thrombus Burden in the Patients with Non-ST-Segment Elevation Myocardial Infarction. Clin. Appl. Thromb. Hemost. 2015, 21, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Uslu, A.; Kup, A.; Dogan, C.; Sari, M.; Cersit, S.; Aksu, U.; Kanat, S.; Demir, M.; Tenekecioglu, E. Relationship between epicardial adipose tissue thickness and coronary thrombus burden in patients with ST-elevation myocardial infarction. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2020, 164, 141–146. [Google Scholar] [CrossRef]
- Fox, C.S.; Gona, P.; Hoffmann, U.; Porter, S.A.; Salton, C.J.; Massaro, J.M.; Levy, D.; Larson, M.G.; D’Agostino, R.B., Sr.; O’Donnell, C.J.; et al. Pericardial fat, intrathoracic fat, and measures of left ventricular structure and function: The Framingham Heart Study. Circulation 2009, 119, 1586–1591. [Google Scholar] [CrossRef]
- Akdag, S.; Simsek, H.; Sahin, M.; Akyol, A.; Duz, R.; Babat, N. Association of epicardial adipose tissue thickness and inflammation parameters with CHA2DS2-VASASc score in patients with nonvalvular atrial fibrillation. Ther. Clin. Risk Manag. 2015, 11, 1675–1681. [Google Scholar] [CrossRef]
- Zehir, R.; Güner, A.; Hayiroglu, M.I.; Oz, T.K.; Osken, A.; Aksu, H.; Dayi, S.U.; Hobikoğlu, G.F. Clinical usefulness of epicardial adipose tissue in patients with high-intermediate pre-test probability for coronary artery disease. Kardiol. Pol. 2018, 76, 1002–1008. [Google Scholar] [CrossRef]
- STARSurg Collaborative and COVIDSurg Collaborative. Death following pulmonary complications of surgery before and during the SARS-CoV-2 pandemic. Br. J. Surg. 2021, 108, 1448–1464. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
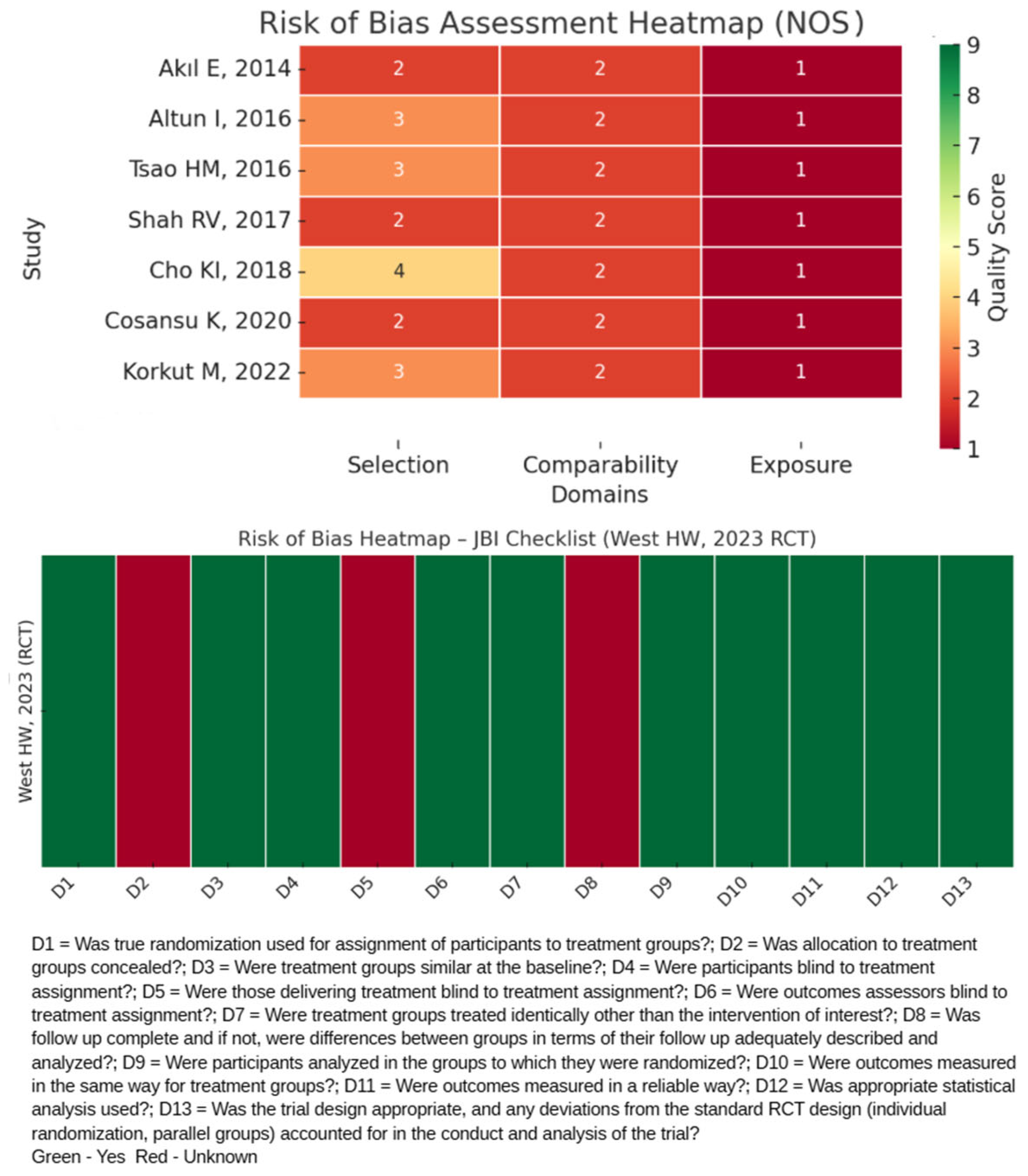



| Study | Number of Stars | Overall | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Selection † | Comparability # | Exposure $ | |||||||||||
| Akıl E, 2014, Turkey [13] | ** | ** | * | 5/9 | |||||||||
| Altun I, 2016, Turkey [14] | *** | ** | * | 6/9 | |||||||||
| Tsao HM, 2016, Taiwan [15] | *** | ** | * | 6/9 | |||||||||
| Shah RV, 2017, USA [16] | ** | ** | * | 5/9 | |||||||||
| Cho KI, 2018, South Korea [17] | **** | ** | * | 7/9 | |||||||||
| Cosansu K, 2020, Turkey [18] | ** | ** | * | 5/9 | |||||||||
| Korkut M, 2022, Turkey [19] | *** | ** | * | 6/9 | |||||||||
| JBI-RCT Tool/Domains | D1 | D2 | D3 | D4 | D5 | D6 | D7 | D8 | D9 | D10 | D11 | D12 | D13 |
| West HW, 2023, Scotland (RCT) [20] | Yes | UN | Yes | Yes | UN | Yes | Yes | UN | Yes | Yes | Yes | Yes | Yes |
| Author, Year, Country | Design | Mean Age Case/Control | Number of Cases (n) (F/M) | Number of Control (n) (F/M) | Definition of Exposure | Measurement of Exposure | Stroke Diagnosis | Size EAT | Size EAT |
|---|---|---|---|---|---|---|---|---|---|
| Case | Control | ||||||||
| Akıl E, 2014, Turkey [13] | Cross-sectional study | 50.5 ± 13.9/ 53.7 ± 9.0 | 38 (15/23) | 47 (20/27) | Epicardial Fat Thickness in mm | Echo TTE; Two measurements by two blinded cardiologists; Echo-free space between two layers of pericardium | CT and MRI | 5.95 ± 1.14 mm | 4.86 ± 0.68 mm |
| Altun I, 2016, Turkey [14] | Case–control study | 71.4 ± 11/ 68.6 ± 8 | 61 (34/27) | 82 (40/42) | Epicardial Fat Thickness in mm | Echo TTE; Echo-free space between the outer wall, myocardium and visceral pericardium | CT and MRI | 4.8 ± 0.9 mm | 3.8 ± 0.7 mm |
| Tsao HM, 2016, Taiwan [15] | Cross-sectional study | 64.11 ± 11.43/ 63.25 ± 7.56 | 27 (8/19) | 20 (5/15) | Epicardial Fat Volume | Contrast CT and EAT Volume | Clinical Signs | 53.07 ± 14.67 cm3 | 21.46 ± 5.81 cm3 |
| Shah RV, 2017, USA [16] | Prospective cohort | NR | NR | NR | Pericardial Fat Volume = Pericardial Fat Volume and Epicardial Fat Volume | CT and CMRI; Includes both Epicardial Fat and Pericardial Fat | NR | NR | NR |
| Cho KI, 2018, South Korea [17] | Retrospective cohort | Overall 65.4 ± 12.1/75.0 ± 10.6 | Epicardial Fat Thickness in mm | Echo TTE; Two measurements by two blinded cardiologists; Echo-free space between the outer wall, myocardium and visceral pericardium | CT, MRI, CT-A, and MR-A | 6.5 ± 1.2 mm | 5.3 ± 1.2 mm | ||
| Cosansu K, 2020, Turkey [18] | Cross-sectional study | 75.19 ± 9.39/ 73.72 ± 8.60 | 80 (50/30) | 80 (46/34) | Epicardial Thickness in mm | Echo TTE; Measured twice by two cardiologists; Echo-free space between the outer wall, myocardium and visceral pericardium | CT and MRI | 8.55 ± 1.08 mm | 5.90 ± 1.35 mm |
| Korkut M, 2022, Turkey [19] | Case–control study | 71.15 ± 12.32/ 69.78 ± 10.31 | 53 (25/28) | 41 (22/19) | Epicardial Thickness in mm | Echo TTE; Three measurements, single-blinded physician; Echo: Free space between the RV and the inner sheet of the pericardium | CT and MRI | 6.33 ± 1.47 mm | 3.74 ± 0.61 mm |
| West HW, 2023, Scotland [20] | SCOT-HEART Randomized Control Trial | NR | NR | NR | Epicardial Fat Volume | CCTA Scans with DLN for Automated Estimation of EAT Volume | NR | NR | NR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahadevan, A.; Pinnamaneni, M.; Krishnakumar, M.; Mishra, T.; Adrejiya, P.; Sanjeevi, A.; Patel, B.M.; Patel, S.; Patel, R.; Jena, N.; et al. Elevated Epicardial Adipose Tissue and Ischemic Stroke Risk: A Systematic Review and Meta-Analysis. Medicina 2025, 61, 2128. https://doi.org/10.3390/medicina61122128
Mahadevan A, Pinnamaneni M, Krishnakumar M, Mishra T, Adrejiya P, Sanjeevi A, Patel BM, Patel S, Patel R, Jena N, et al. Elevated Epicardial Adipose Tissue and Ischemic Stroke Risk: A Systematic Review and Meta-Analysis. Medicina. 2025; 61(12):2128. https://doi.org/10.3390/medicina61122128
Chicago/Turabian StyleMahadevan, Arankesh, Monitha Pinnamaneni, Manaswini Krishnakumar, Tanisha Mishra, Parth Adrejiya, Aditya Sanjeevi, Bhaumikkumar Mukeshbhai Patel, Sneh Patel, Rahul Patel, Nihar Jena, and et al. 2025. "Elevated Epicardial Adipose Tissue and Ischemic Stroke Risk: A Systematic Review and Meta-Analysis" Medicina 61, no. 12: 2128. https://doi.org/10.3390/medicina61122128
APA StyleMahadevan, A., Pinnamaneni, M., Krishnakumar, M., Mishra, T., Adrejiya, P., Sanjeevi, A., Patel, B. M., Patel, S., Patel, R., Jena, N., Vyas, A., & Desai, R. (2025). Elevated Epicardial Adipose Tissue and Ischemic Stroke Risk: A Systematic Review and Meta-Analysis. Medicina, 61(12), 2128. https://doi.org/10.3390/medicina61122128







