Evaluation of [18F]Favipiravir in Rodents and Nonhuman Primates (NHP) with Positron Emission Tomography
Abstract
1. Introduction
2. Results
2.1. Radiochemistry
2.2. PET Imaging in Naive Mice with [18F]Favipiravir
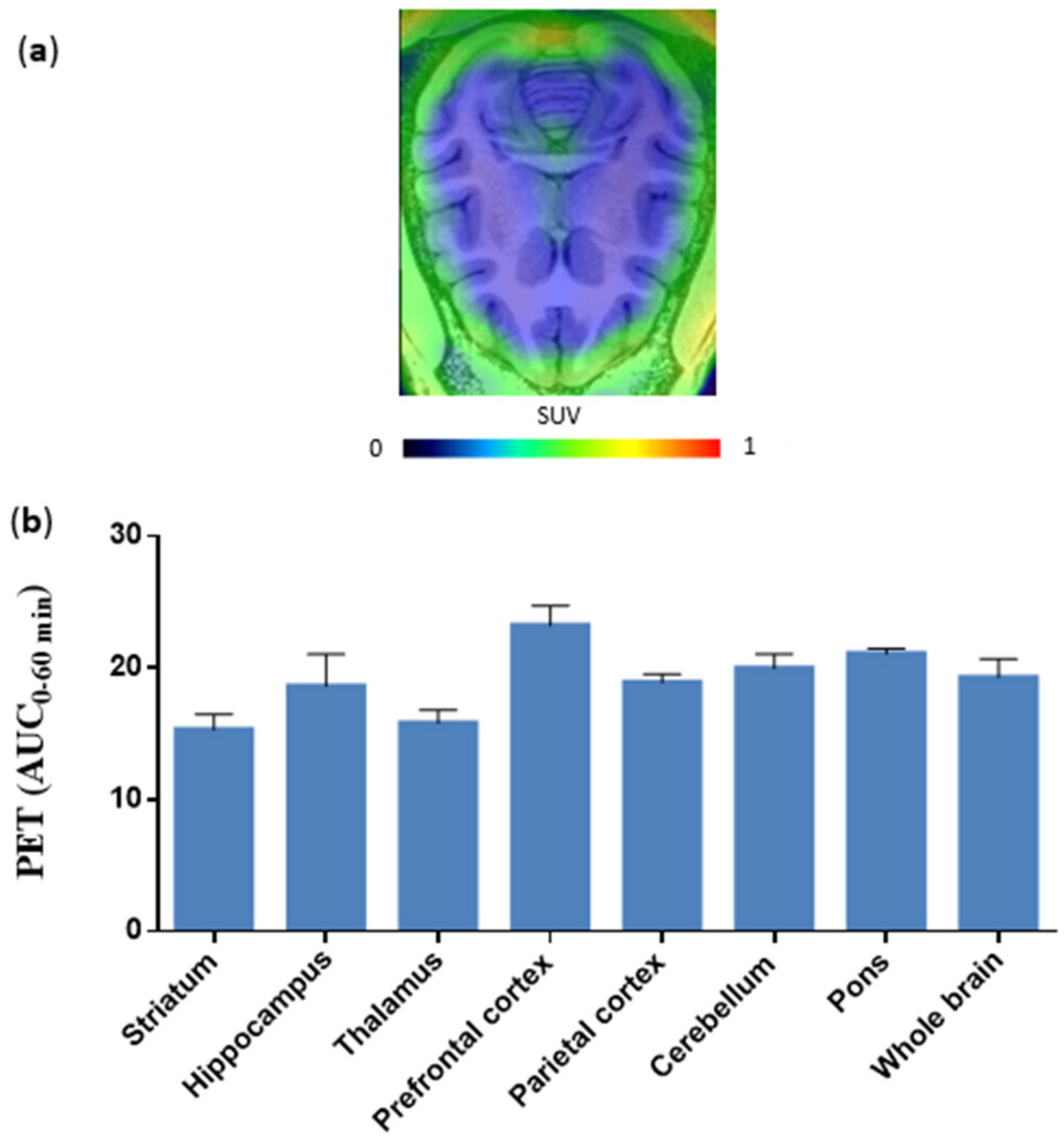
2.3. PET Imaging in Transgenic AD and WT Mice with [18F]Favipiravir
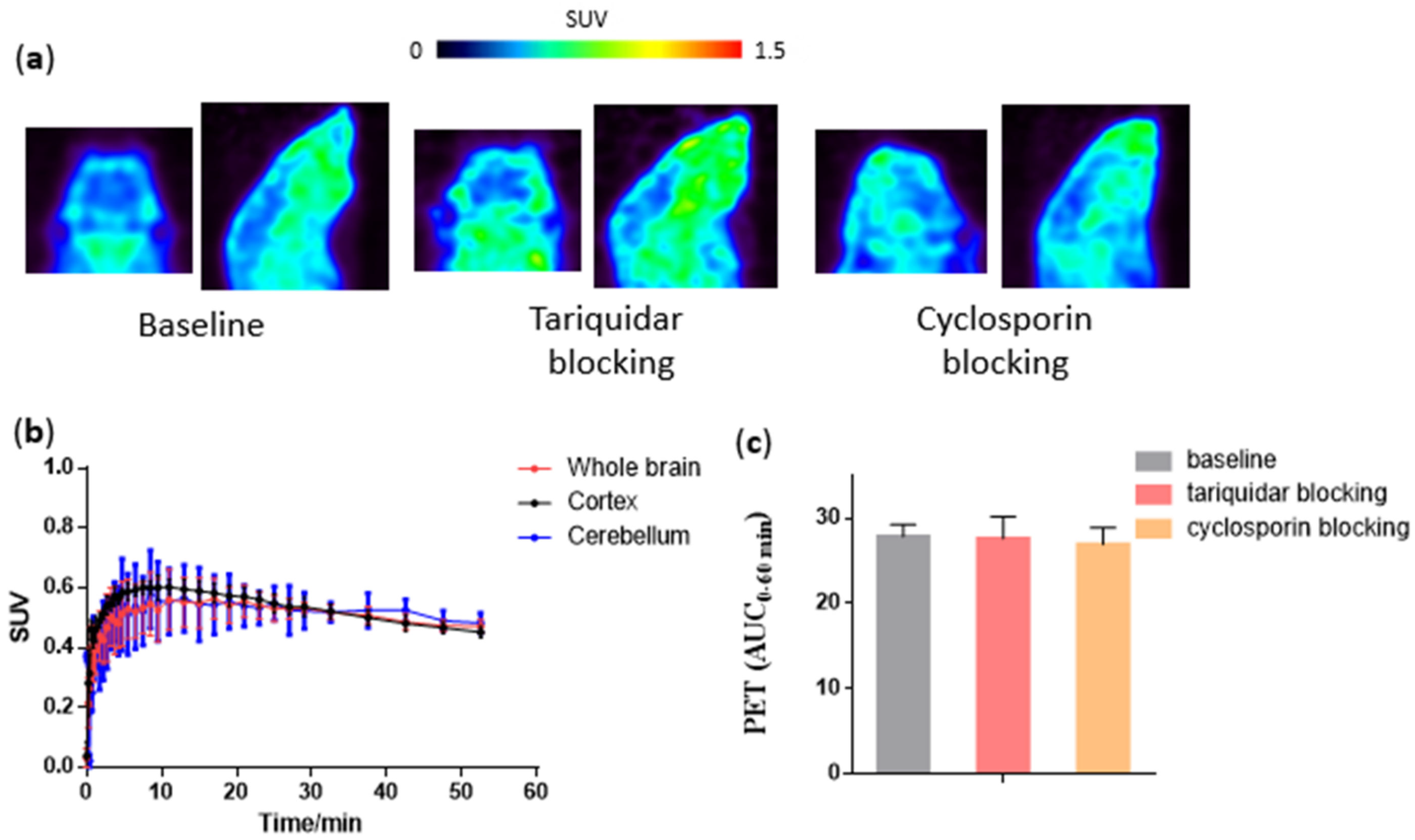
2.4. PET Imaging in NHP with [18F]Favipiravir
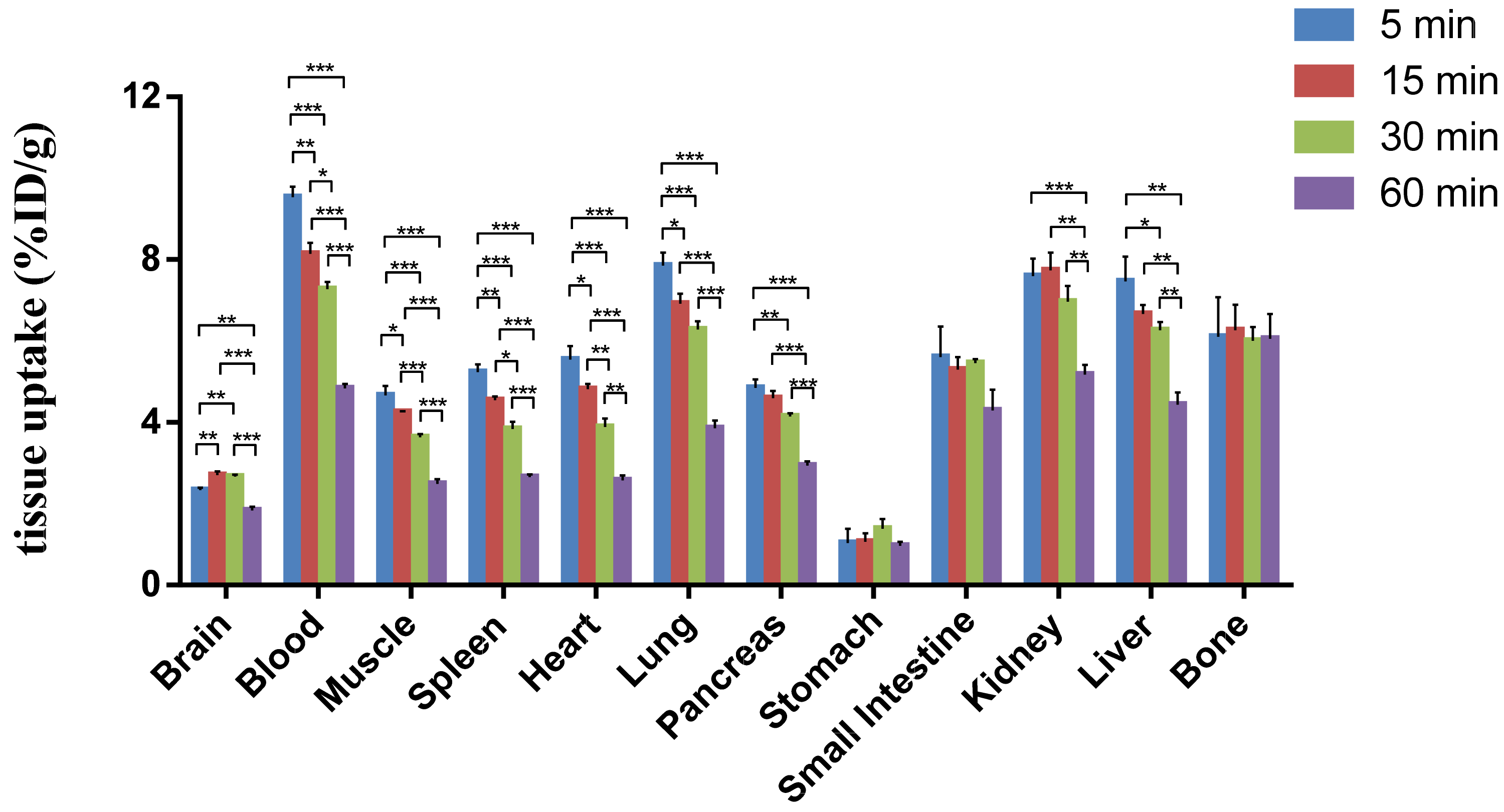
2.5. Ex Vivo Whole-Body Biodistribution in Mice with [18F]Favipiravir
2.6. Metabolic Analysis of [18F]Favipiravir in Mice
3. Discussion
4. Materials and Methods
4.1. Labeling of [18F]Favipiravir
4.2. PET Imaging in Mice with [18F]Favipiravir
4.3. PET/MRI Imaging in NHP with [18F]Favipiravir
4.4. Ex Vivo Whole-Body Biodistribution in Mice with [18F]Favipiravir
4.5. Radiometabolite Analysis with [18F]Favipiravir in Mice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Data from COVID-19 Map-Johns Hopkins Coronavirus Resource Center. Available online: https://coronavirus.jhu.edu/map.html (accessed on 10 February 2023).
- Sleeman, K.; Mishin Vasiliy, P.; Deyde Varough, M.; Furuta, Y.; Klimov Alexander, I.; Gubareva Larisa, V. In Vitro Antiviral Activity of Favipiravir (T-705) against Drug-Resistant Influenza and 2009 A(H1N1) Viruses. Antimicrob. Agents Chemother. 2010, 54, 2517–2524. [Google Scholar] [CrossRef]
- Furuta, Y.; Komeno, T.; Nakamura, T. Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc. Jpn. Acad. Ser. B 2017, 93, 449–463. [Google Scholar] [CrossRef]
- Du, Y.-X.; Chen, X.-P. Favipiravir: Pharmacokinetics and Concerns About Clinical Trials for 2019-nCoV Infection. Clin. Pharmacol. Ther. 2020, 108, 242–247. [Google Scholar] [CrossRef]
- Sangawa, H.; Komeno, T.; Nishikawa, H.; Yoshida, A.; Takahashi, K.; Nomura, N.; Furuta, Y. Mechanism of Action of T-705 Ribosyl Triphosphate against Influenza Virus RNA Polymerase. Antimicrob. Agents Chemother. 2013, 57, 5202–5208. [Google Scholar] [CrossRef]
- Kiso, M.; Takahashi, K.; Sakai-Tagawa, Y.; Shinya, K.; Sakabe, S.; Le, Q.M.; Ozawa, M.; Furuta, Y.; Kawaoka, Y. T-705 (favipiravir) activity against lethal H5N1 influenza A viruses. Proc. Natl. Acad. Sci. USA 2010, 107, 882–887. [Google Scholar] [CrossRef]
- Oestereich, L.; Lüdtke, A.; Wurr, S.; Rieger, T.; Muñoz-Fontela, C.; Günther, S. Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model. Antivir. Res. 2014, 105, 17–21. [Google Scholar] [CrossRef]
- Ko, W.-C.; Rolain, J.-M.; Lee, N.-Y.; Chen, P.-L.; Huang, C.-T.; Lee, P.-I.; Hsueh, P.-R. Arguments in favour of remdesivir for treating SARS-CoV-2 infections. Int. J. Antimicrob. Agents 2020, 55, 105933. [Google Scholar] [CrossRef]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef]
- Touret, F.; de Lamballerie, X. Of chloroquine and COVID-19. Antivir. Res. 2020, 177, 104762. [Google Scholar] [CrossRef]
- Lumby, C.K.; Zhao, L.; Oporto, M.; Best, T.; Tutill, H.; Shah, D.; Veys, P.; Williams, R.; Worth, A.; Illingworth, C.J.R.; et al. Favipiravir and Zanamivir Cleared Infection with Influenza B in a Severely Immunocompromised Child. Clin. Infect. Dis. 2020, 71, e191–e194. [Google Scholar] [CrossRef]
- Li, G.; De Clercq, E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nat. Rev. Drug Discov. 2020, 19, 149–150. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Hu, S.; Gao, J. Discovering drugs to treat coronavirus disease 2019 (COVID-19). Drug Discov. Ther. 2020, 14, 58–60. [Google Scholar] [CrossRef] [PubMed]
- NCT04310228; Favipiravir Combined with Tocilizumab in the Treatment of Corona Virus Disease 2019. Peking University First Hospital: Beijing, China, 2019.
- NCT04319900; Clinical Trial of Favipiravir Tablets Combine with Chloroquine Phosphate in the Treatment of Novel Coronavirus Pneumonia. Beijing Chao Yang Hospital: Beijing, China, 2020.
- NCT04333589; Corona Virus Disease 2019 Patients Whose Nucleic Acids Changed from Negative to Positive. Peking University First Hospital: Beijing, China, 2019.
- Note, Fujifilm tests favipiravir as a COVID-19 treatment. Available online: https://cen.acs.org/pharmaceuticals/drug-development/Fujifilm-tests-favipiravir-COVID-19/98/i15 (accessed on 16 April 2020).
- NCT04336904; Clinical Study To Evaluate The Performance And Safety of Favipiravir in COVID-19. ASST Fatebenefratelli Sacco: Milan, Italy, 2020.
- NCT04358549; Study of the Use of Favipiravir in Hospitalized Subjects with COVID-19. Fujifilm Pharmaceuticals U.S.A., Inc: Cambridge, MA, USA, 2020.
- NCT04346628; Oral Favipiravir Compared to Standard Supportive Care in Subjects with Mild COVID-19. Stanford University: Stanford, CA, USA, 2020.
- Deng, X.; Rong, J.; Wang, L.; Vasdev, N.; Zhang, L.; Josephson, L.; Liang, S.H. Chemistry for Positron Emission Tomography: Recent Advances in 11C-, 18F-, 13N-, and 15O-Labeling Reactions. Angew. Chem. Int. Ed. 2019, 58, 2580–2605. [Google Scholar] [CrossRef] [PubMed]
- Bocan, T.M.; Basuli, F.; Stafford, R.G.; Brown, J.L.; Zhang, X.; Duplantier, A.J.; Swenson, R.E. Synthesis of [18F]Favipiravir and Biodistribution in C3H/HeN Mice as Assessed by Positron Emission Tomography. Sci. Rep. 2019, 9, 1785. [Google Scholar] [CrossRef]
- Verbeek, J.; Eriksson, J.; Syvanen, S.; Huisman, M.; Schuit, R.C.; Molthoff, C.F.M.; Voskuyl, R.A.; de Lange, E.C.; Lammertsma, A.A.; Windhorst, A.D. Synthesis and preliminary preclinical evaluation of fluorine-18 labelled isatin-4-(4-methoxyphenyl)-3-thiosemicarbazone ([(18)F]4FIMPTC) as a novel PET tracer of P-glycoprotein expression. EJNMMI Radiopharm. Chem. 2018, 3, 11. [Google Scholar] [CrossRef]
- Tu, Z.; Li, S.; Xu, J.; Chu, W.; Jones, L.A.; Luedtke, R.R.; Mach, R.H. Effect of cyclosporin A on the uptake of D3-selective PET radiotracers in rat brain. Nucl. Med. Biol. 2011, 38, 725–739. [Google Scholar] [CrossRef]
- Zareifopoulos, N.; Lagadinou, M.; Karela, A.; Kyriakopoulou, O.; Velissaris, D. Neuropsychiatric Effects of Antiviral Drugs. Cureus 2020, 12, e9536. [Google Scholar] [CrossRef]
- Rong, J.; Mori, W.; Xia, X.; Schafroth, M.A.; Zhao, C.; Van, R.S.; Yamasaki, T.; Chen, J.; Xiao, Z.; Haider, A.; et al. Novel Reversible-Binding PET Ligands for Imaging Monoacylglycerol Lipase Based on the Piperazinyl Azetidine Scaffold. J. Med. Chem. 2021, 64, 14283–14298. [Google Scholar] [CrossRef]
- Cheng, R.; Mori, W.; Ma, L.; Alhouayek, M.; Hatori, A.; Zhang, Y.; Ogasawara, D.; Yuan, G.; Chen, Z.; Zhang, X.; et al. In Vitro and in Vivo Evaluation of 11C-Labeled Azetidinecarboxylates for Imaging Monoacylglycerol Lipase by PET Imaging Studies. J. Med. Chem. 2018, 61, 2278–2291. [Google Scholar] [CrossRef]
- Wang, L.; Mori, W.; Cheng, R.; Yui, J.; Hatori, A.; Ma, L.; Zhang, Y.; Rotstein, B.H.; Fujinaga, M.; Shimoda, Y.; et al. Synthesis and Preclinical Evaluation of Sulfonamido-based [11C-Carbonyl]-Carbamates and Ureas for Imaging Monoacylglycerol Lipase. Theranostics 2016, 6, 1145–1159. [Google Scholar] [CrossRef]
- Chen, Z.; Mori, W.; Fu, H.; Schafroth, M.A.; Hatori, A.; Shao, T.; Zhang, G.; Van, R.S.; Zhang, Y.; Hu, K.; et al. Design, Synthesis, and Evaluation of 18F-Labeled Monoacylglycerol Lipase Inhibitors as Novel Positron Emission Tomography Probes. J. Med. Chem. 2019, 62, 8866–8872. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rong, J.; Zhao, C.; Xia, X.; Li, G.; Haider, A.; Wei, H.; Chen, J.; Xiao, Z.; Li, Y.; Zhou, X.; et al. Evaluation of [18F]Favipiravir in Rodents and Nonhuman Primates (NHP) with Positron Emission Tomography. Pharmaceuticals 2023, 16, 524. https://doi.org/10.3390/ph16040524
Rong J, Zhao C, Xia X, Li G, Haider A, Wei H, Chen J, Xiao Z, Li Y, Zhou X, et al. Evaluation of [18F]Favipiravir in Rodents and Nonhuman Primates (NHP) with Positron Emission Tomography. Pharmaceuticals. 2023; 16(4):524. https://doi.org/10.3390/ph16040524
Chicago/Turabian StyleRong, Jian, Chunyu Zhao, Xiaotian Xia, Guocong Li, Achi Haider, Huiyi Wei, Jiahui Chen, Zhiwei Xiao, Yinlong Li, Xin Zhou, and et al. 2023. "Evaluation of [18F]Favipiravir in Rodents and Nonhuman Primates (NHP) with Positron Emission Tomography" Pharmaceuticals 16, no. 4: 524. https://doi.org/10.3390/ph16040524
APA StyleRong, J., Zhao, C., Xia, X., Li, G., Haider, A., Wei, H., Chen, J., Xiao, Z., Li, Y., Zhou, X., Xu, H., Collier, T. L., Wang, L., & Liang, S. H. (2023). Evaluation of [18F]Favipiravir in Rodents and Nonhuman Primates (NHP) with Positron Emission Tomography. Pharmaceuticals, 16(4), 524. https://doi.org/10.3390/ph16040524






