Detection of Acetaminophen in Groundwater by Laccase-Based Amperometric Biosensors Using MoS2 Modified Carbon Paper Electrodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Laccase Enzymes
2.3. Synthesis of MoS2 Nanostructured Material
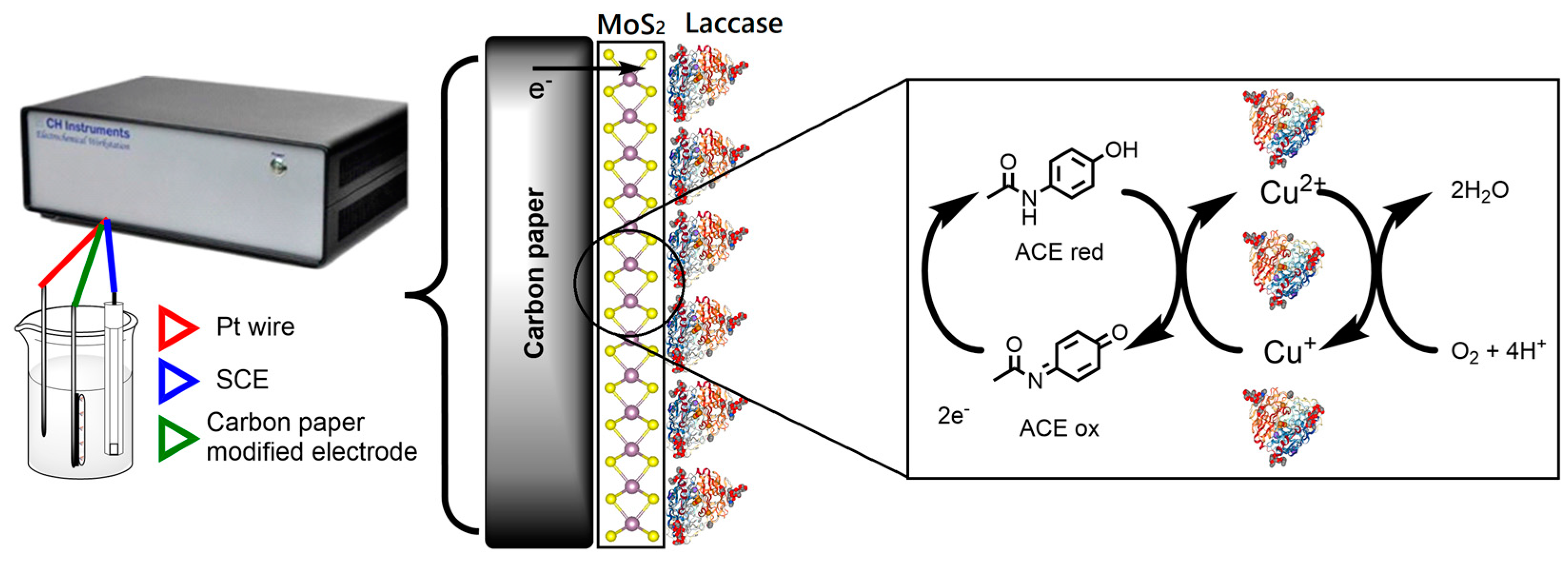
2.4. Immobilization of Laccase onto MoS2 Modified Electrodes
2.5. Electrochemical Measurements in the Optimization of Acetaminophen Detection
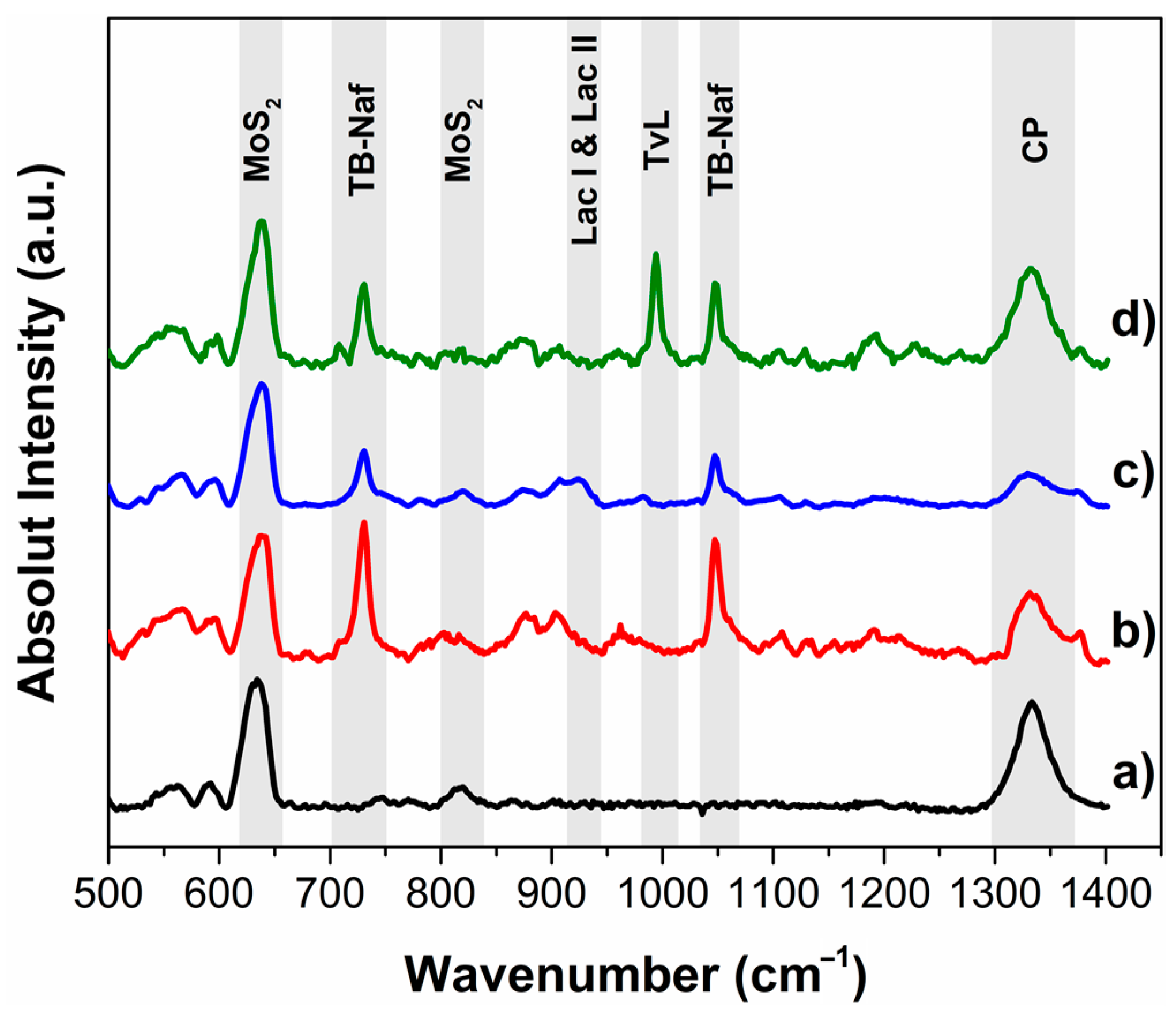
2.6. Characterization Techniques
2.7. Application of Optimized Electrodes for the Detection of Acetaminophen in Groundwater Samples
3. Results and Discussion
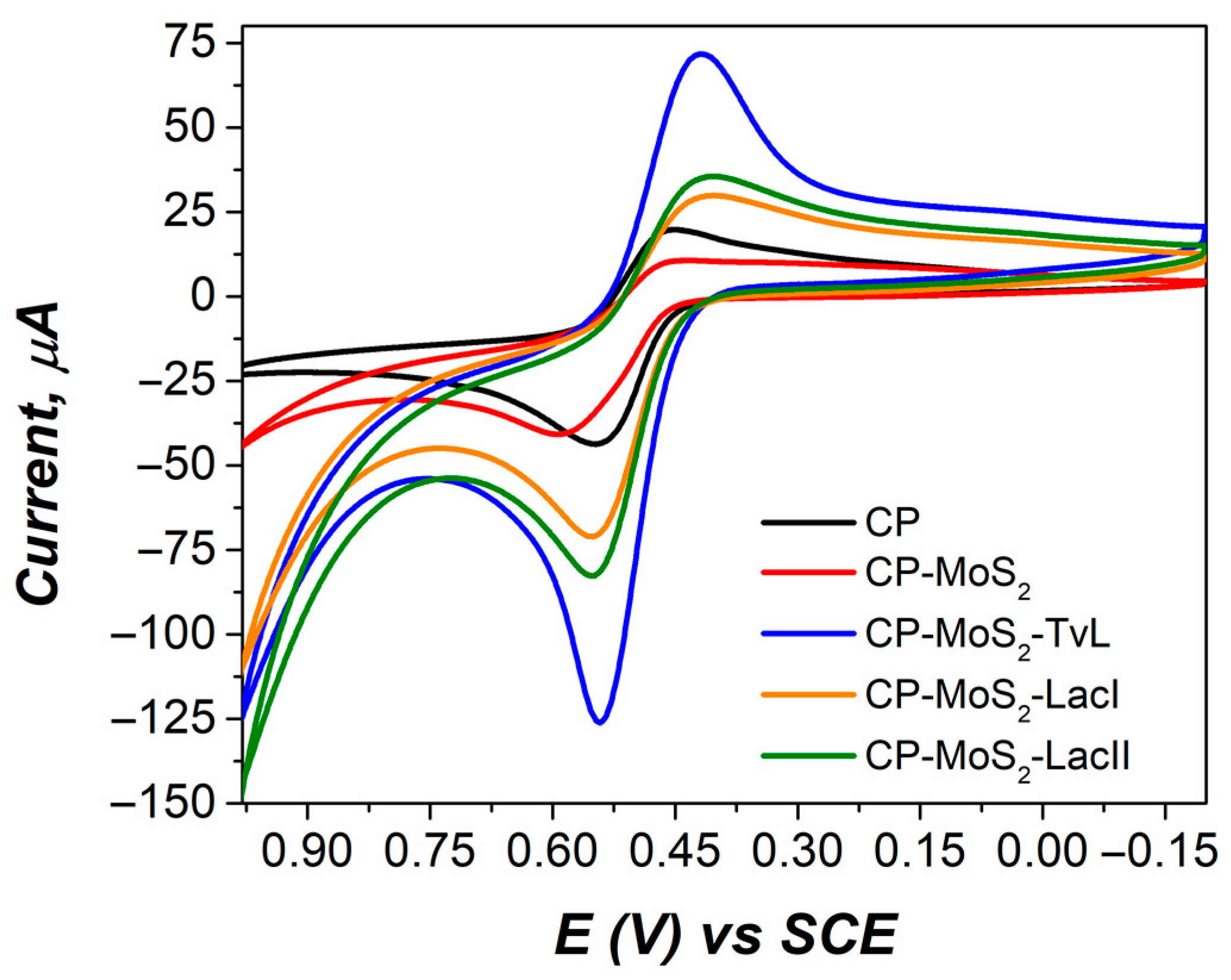
3.1. Immobilization of Laccases onto MoS2 Modified Electrodes
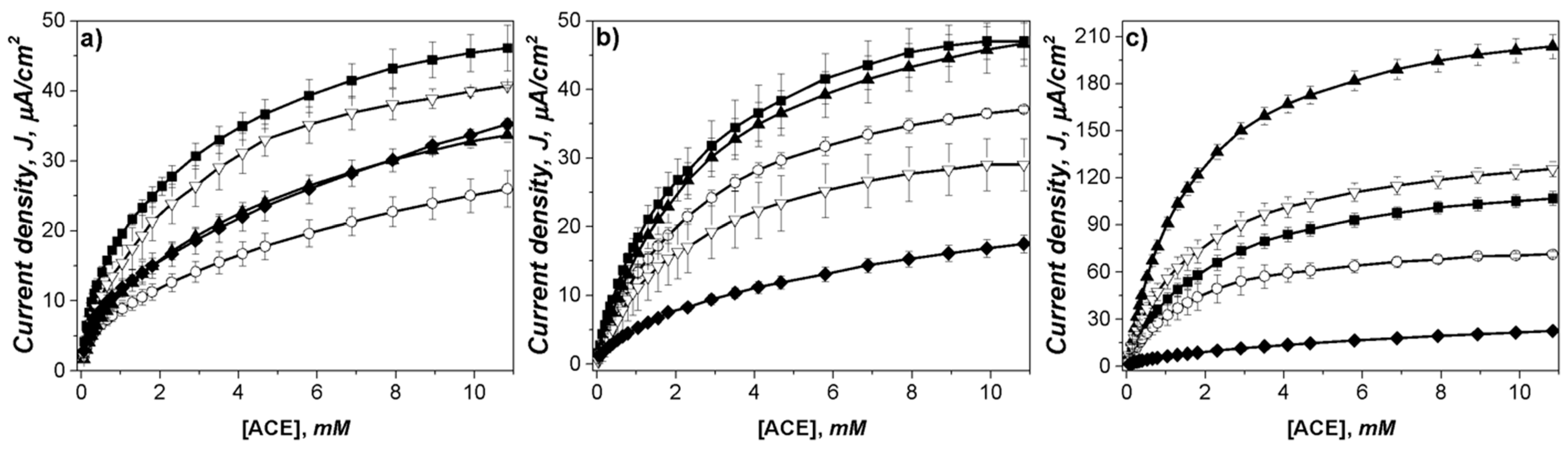
3.2. Optimum MoS2 Concentration for Laccase Bioelectrode Modification
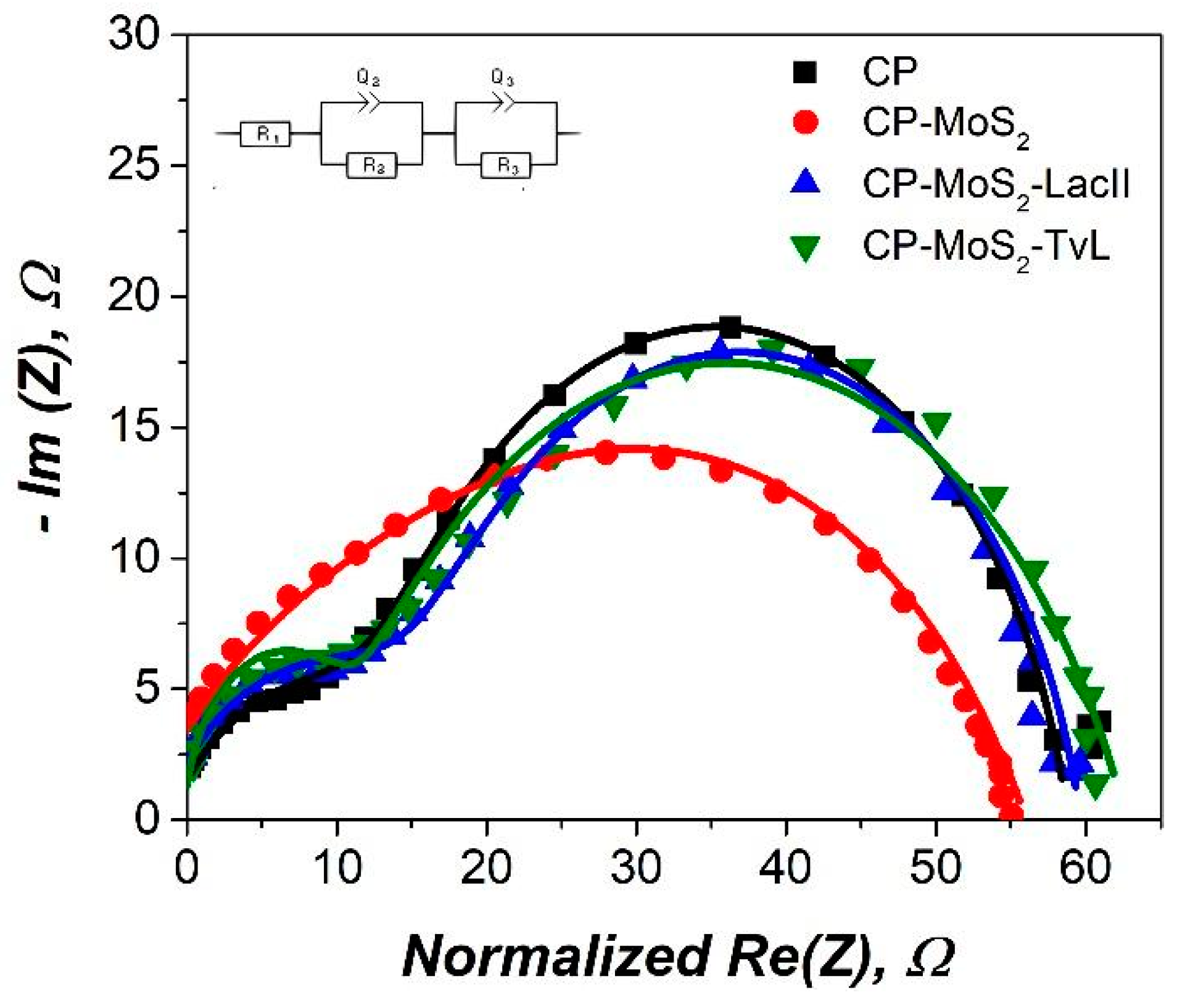
3.3. Characterization of Optimum MoS2 Modified Electrodes
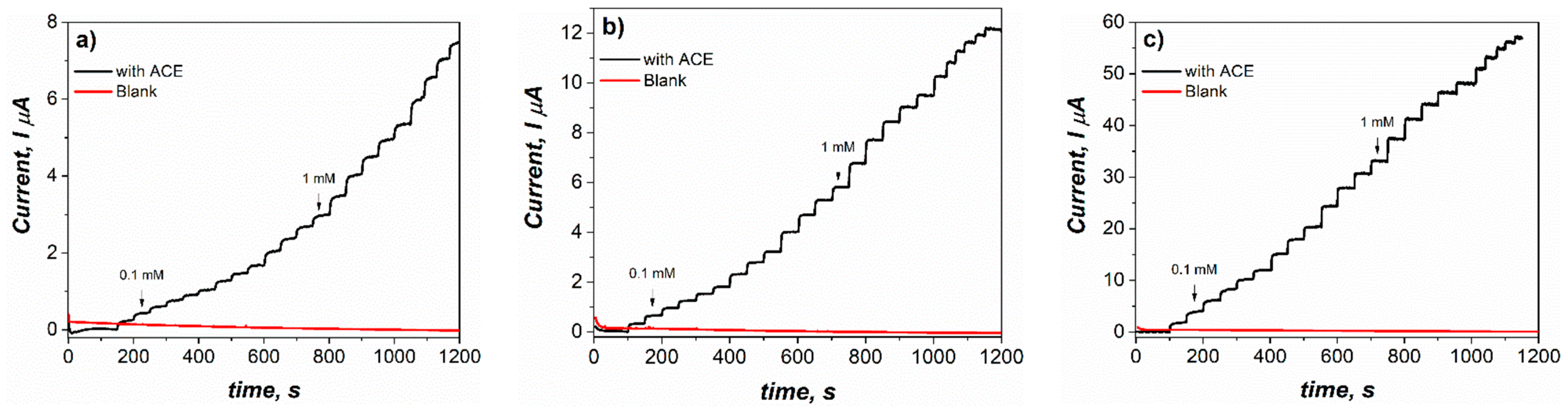
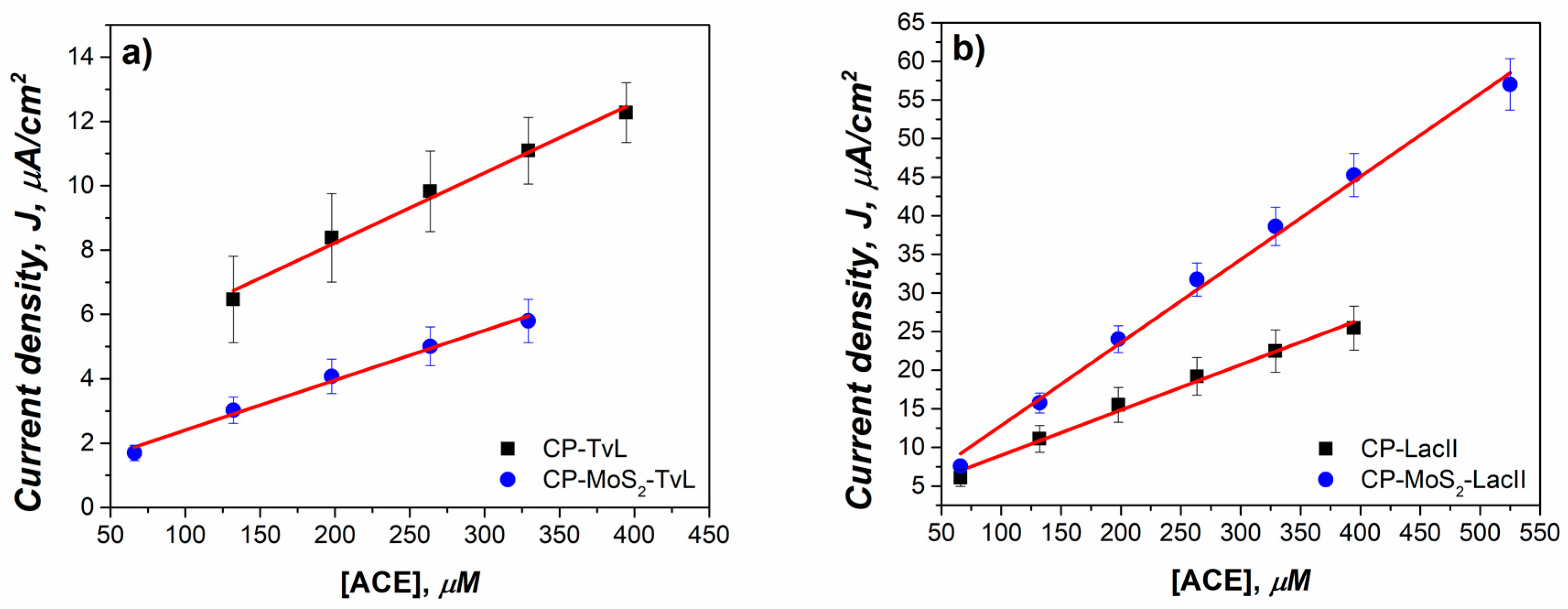
3.4. Application of Modified Electrodes in the Detection of Acetaminophen
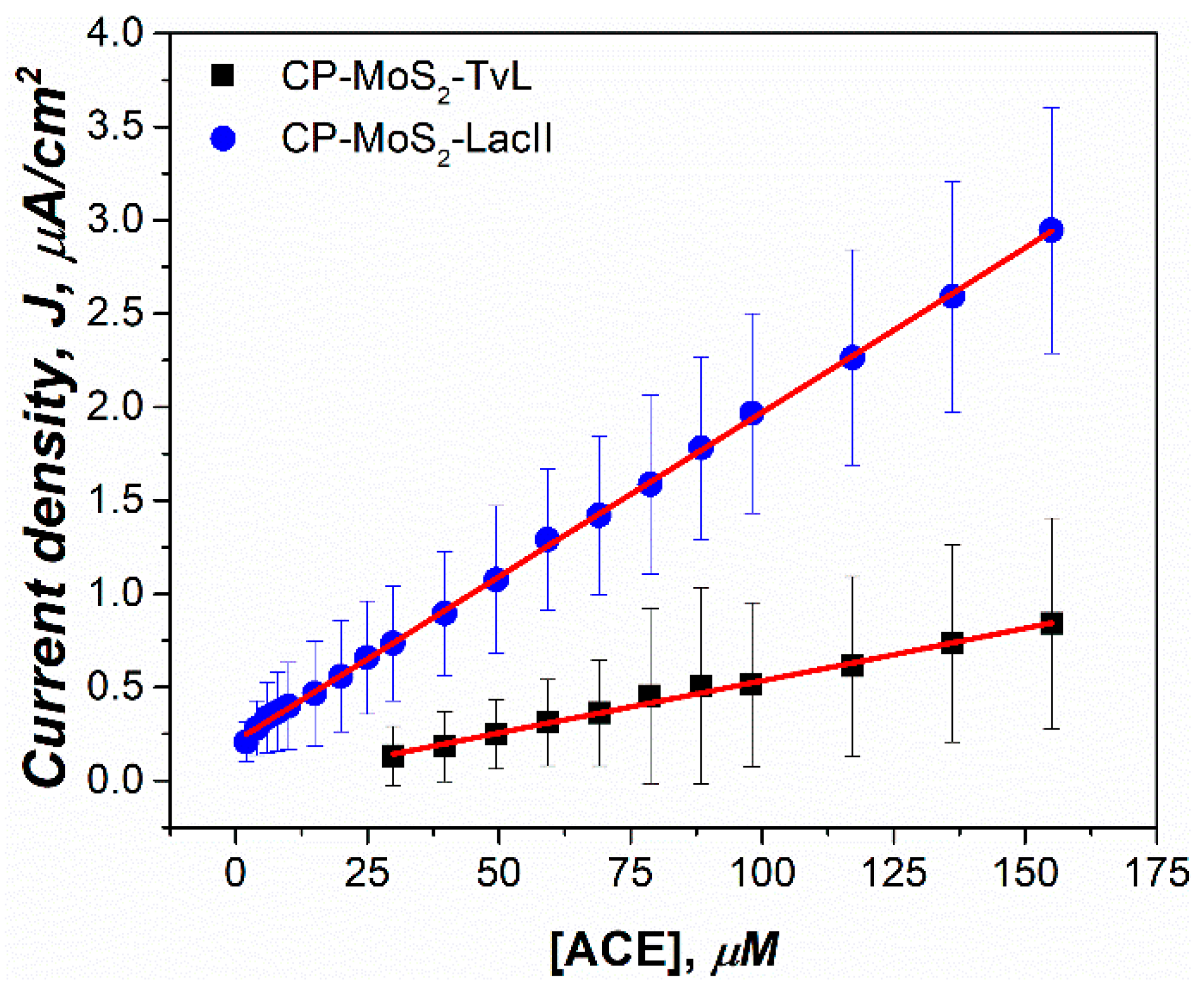
3.5. Application of Modified Electrodes in the Amperometric Detection of Acetaminophen in a Groundwater Sample from a City in Northeastern Mexico
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Butkovskyi, A.; Leal, L.H.; Zeeman, G.; Rijnaarts, H.H.M. Micropollutants in Source Separated Wastewater Streams and Recovered Resources of Source Separated Sanitation. Environ. Res. 2017, 156, 434–442. [Google Scholar] [CrossRef]
- Sousa, J.C.G.; Ribeiro, A.R.; Barbosa, M.O.; Pereira, M.F.R.; Silva, A.M.T. A Review on Environmental Monitoring of Water Organic Pollutants Identified by EU Guidelines. J. Hazard. Mater. 2018, 344, 146–162. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.; Becerril-Bravo, E.; Silva-Castro, V.; Jiménez, B. Determination of Acidic Pharmaceuticals and Potential Endocrine Disrupting Compounds in Wastewaters and Spring Waters by Selective Elution and Analysis by Gas Chromatography-Mass Spectrometry. J. Chromatogr. A 2007, 1169, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B.; Pinto, G.; Martins, L.; Gonçalves, F.; Antunes, S.C. Biochemical and Standard Toxic Effects of Acetaminophen on the Macrophyte Species Lemna minor and Lemna gibba. Environ. Sci. Pollut. Res. 2014, 21, 10815–10822. [Google Scholar] [CrossRef] [PubMed]
- Anekar, A.A.; Cascella, M. WHO Analgesic Ladder. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554435/ (accessed on 12 October 2022).
- Yoon, E.; Babar, A.; Choudhary, M.; Kutner, M.; Pyrsopoulos, N. Acetaminophen-Induced Hepatotoxicity: A Comprehensive Update. J. Clin. Transl. Hepatol. 2016, 4, 131. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Zhang, M.; Yuan, S.; Feng, J.; Wang, Q.; Wang, W.; Hu, Z. Transformation of Acetaminophen during Water Chlorination Treatment: Kinetics and Transformation Products Identification. Environ. Sci. Pollut. Res. 2016, 23, 12303–12311. [Google Scholar] [CrossRef]
- aus der Beek, T.; Weber, F.A.; Bergmann, A.; Hickmann, S.; Ebert, I.; Hein, A.; Küster, A. Pharmaceuticals in the Environment-Global Occurrences and Perspectives. Environ. Toxicol. Chem. 2016, 35, 823–835. [Google Scholar] [CrossRef]
- Montaseri, H.; Forbes, P.B.C. Analytical Techniques for the Determination of Acetaminophen: A Review. TrAC-Trends Anal. Chem. 2018, 108, 122–134. [Google Scholar] [CrossRef]
- Anthérieu, S.; Le Guillou, D.; Coulouarn, C.; Begriche, K.; Trak-Smayra, V.; Martinais, S.; Porceddu, M.; Robin, M.-A.; Fromenty, B. Chronic Exposure to Low Doses of Pharmaceuticals Disturbs the Hepatic Expression of Circadian Genes in Lean and Obese Mice. Toxicol. Appl. Pharmacol. 2014, 276, 63–72. [Google Scholar] [CrossRef]
- Togola, A.; Budzinski, H. Multi-Residue Analysis of Pharmaceutical Compounds in Aqueous Samples. J. Chromatogr. A 2008, 1177, 150–158. [Google Scholar] [CrossRef]
- Environmental Health Division. Acetaminophen in Drinking Water; Environ. Heal. Minnesota Department of Health: Saint Paul, MN, USA, 2014; Available online: www.health.state.mn.us/eh (accessed on 4 May 2023).
- González-Sánchez, M.I.; Rubio-Retama, J.; López-Cabarcos, E.; Valero, E. Development of an Acetaminophen Amperometric Biosensor Based on Peroxidase Entrapped in Polyacrylamide Microgels. Biosens. Bioelectron. 2011, 26, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Thévenot, D.R.; Toth, K.; Durst, R.A.; Wilson, G.S. Electrochemical Biosensors: Recommended Definitions and Classification. Biosens. Bioelectron. 2001, 16, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Calas-Blanchard, C.; Istamboulié, G.; Bontoux, M.; Plantard, G.; Goetz, V.; Noguer, T. Biosensor-Based Real-Time Monitoring of Paracetamol Photocatalytic Degradation. Chemosphere 2015, 131, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Ronkainen, N.J.; Halsall, H.B.; Heineman, W.R. Electrochemical Biosensors. Chem. Soc. Rev. 2010, 39, 1747. [Google Scholar] [CrossRef]
- Barsan, M.M.; Emilia Ghica, M.; Brett, C.M.A. Electrochemical Biosensors. In Portable Biosensing of Food Toxicants and Environmental Pollutants; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2013; Volume 39, pp. 33–69. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, G.; Li, H.; Du, D.; Lin, Y. Electrochemical Sensors and Biosensors Based on Nanomaterials and Nanostructures. Anal. Chem. 2015, 87, 230–249. [Google Scholar] [CrossRef] [PubMed]
- Perumal, V.; Hashim, U. Advances in Biosensors: Principle, Architecture and Applications. J. Appl. Biomed. 2014, 12, 1–15. [Google Scholar] [CrossRef]
- Mayer, A.M.; Staples, R.C. Laccase: New Functions for an Old Enzyme. ChemInform 2002, 33, 256. [Google Scholar] [CrossRef]
- Yaropolov, A.I.; Skorobogat’ko, O.V.; Vartanov, S.S.; Varfolomeyev, S.D. Laccase—Properties, Catalytic Mechanism, and Applicability. Appl. Biochem. Biotechnol. 1994, 49, 257–280. [Google Scholar] [CrossRef]
- Madhavi, V.; Lele, S.S. Laccase: Properties and Applications. BioResources 2009, 4, 1694–1717. [Google Scholar]
- Rodríguez-Delgado, M.M.; Alemán-Nava, G.S.; Rodríguez-Delgado, J.M.; Dieck-Assad, G.; Martínez-Chapa, S.O.; Barceló, D.; Parra, R. Laccase-Based Biosensors for Detection of Phenolic Compounds. TrAC-Trends Anal. Chem. 2015, 74, 21–45. [Google Scholar] [CrossRef]
- Ramírez-Cavazos, L.I.; Junghanns, C.; Ornelas-Soto, N.; Cárdenas-Chávez, D.L.; Hernández-Luna, C.; Demarche, P.; Enaud, E.; García-Morales, R.; Agathos, S.N.; Parra, R. Purification and Characterization of Two Thermostable Laccases from Pycnoporus Sanguineus and Potential Role in Degradation of Endocrine Disrupting Chemicals. J. Mol. Catal. B Enzym. 2014, 108, 32–42. [Google Scholar] [CrossRef]
- Holmberg, S.; Rodriguez-Delgado, M.; Milton, R.D.; Ornelas-Soto, N.; Minteer, S.D.; Parra, R.; Madou, M.J. Bioelectrochemical Study of Thermostable Pycnoporus sanguineus CS43 Laccase Bioelectrodes Based on Pyrolytic Carbon Nanofibers for Bioelectrocatalytic O2 Reduction. ACS Catal. 2015, 5, 7507–7518. [Google Scholar] [CrossRef]
- Kolev, S.K.; Aleksandrov, H.A.; Atanasov, V.A.; Popov, V.N.; Milenov, T.I. Interaction of Graphene with Out-of-Plane Aromatic Hydrocarbons. J. Phys. Chem. C 2019, 123, 21448–21456. [Google Scholar] [CrossRef]
- Huang, K.J.; Liu, Y.J.; Liu, Y.M.; Wang, L.L. Molybdenum Disulfide Nanoflower-Chitosan-Au Nanoparticles Composites Based Electrochemical Sensing Platform for Bisphenol a Determination. J. Hazard. Mater. 2014, 276, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Bollella, P.; Fusco, G.; Tortolini, C.; Sanzò, G.; Favero, G.; Gorton, L.; Antiochia, R. Beyond Graphene: Electrochemical Sensors and Biosensors for Biomarkers Detection. Biosens. Bioelectron. 2017, 89, 152–166. [Google Scholar] [CrossRef]
- Shavanova, K.; Bakakina, Y.; Burkova, I.; Shtepliuk, I.; Viter, R.; Ubelis, A.; Beni, V.; Starodub, N.; Yakimova, R.; Khranovskyy, V. Application of 2D Non-Graphene Materials and 2D Oxide Nanostructures for Biosensing Technology. Sensors 2016, 16, 223. [Google Scholar] [CrossRef]
- Wang, Y.H.; Huang, K.J.; Wu, X. Recent Advances in Transition-Metal Dichalcogenides Based Electrochemical Biosensors: A Review. Biosens. Bioelectron. 2017, 97, 305–316. [Google Scholar] [CrossRef]
- Zhang, H. Ultrathin Two-Dimensional Nanomaterials. ACS Nano 2015, 9, 9451–9469. [Google Scholar] [CrossRef]
- Li, H.; Yin, Z.; He, Q.; Li, H.; Huang, X.; Lu, G.; Fam, D.W.H.; Tok, A.I.Y.; Zhang, Q.; Zhang, H. Fabrication of Single- and Multilayer MoS2 Film-Based Field-Effect Transistors for Sensing NO at Room Temperature. Small 2012, 8, 63–67. [Google Scholar] [CrossRef]
- Pramoda, K.; Moses, K.; Maitra, U.; Rao, C.N.R. Superior Performance of a MoS2 -RGO Composite and a Borocarbonitride in the Electrochemical Detection of Dopamine, Uric Acid and Adenine. Electroanalysis 2015, 27, 1892–1898. [Google Scholar] [CrossRef]
- Mani, V.; Govindasamy, M.; Chen, S.M.; Karthik, R.; Huang, S.T. Determination of Dopamine Using a Glassy Carbon Electrode Modified with a Graphene and Carbon Nanotube Hybrid Decorated with Molybdenum Disulfide Flowers. Microchim. Acta 2016, 183, 2267–2275. [Google Scholar] [CrossRef]
- Jeong, J.M.; Yang, M.H.; Kim, D.S.; Lee, T.J.; Choi, B.G.; Kim, D.H. High Performance Electrochemical Glucose Sensor Based on Three-Dimensional MoS2/Graphene Aerogel. J. Colloid Interface Sci. 2017, 506, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Nan, F.; Wang, X.; Jiao, K.; Ge, T.; Zhao, J.; Yang, T. A Label-Free Ultrasensitive Electrochemical DNA Sensor Based on Thin-Layer MoS2 Nanosheets with High Electrochemical Activity. Biosens. Bioelectron. 2014, 64, 386–391. [Google Scholar] [CrossRef]
- Hickey, D.P.; Lim, K.; Cai, R.; Patterson, A.R.; Yuan, M.; Sahin, S.; Abdellaoui, S.; Minteer, S.D. Pyrene Hydrogel for Promoting Direct Bioelectrochemistry: ATP-Independent Electroenzymatic Reduction of N2. Chem. Sci. 2018, 9, 5172–5177. [Google Scholar] [CrossRef]
- Najmaei, S.; Liu, Z.; Zhou, W.; Zou, X.; Shi, G.; Lei, S.; Yakobson, B.I.; Idrobo, J.-C.; Ajayan, P.M.; Lou, J. Vapor Phase Growth and Grain Boundary Structure of Molybdenum Disulfide Atomic Layers Vapour Phase Growth and Grain Boundary Structure of Molybdenum Disulphide Atomic Layers. Nat. Mater. 2013, 12, 754–759. [Google Scholar] [CrossRef]
- Dominguez-Rovira, M.A.; Garcia-Garcia, A.; Toxqui-Teran, A. Metodo Para La Obtencion de Cintas de Disulfuro de Molibdeno En Fase Gaseosa, a Partir de La Sulfuracion de Trioxido de Molibdeno. Patent Process Number MX-a-2017-016742, 2017. [Google Scholar]
- Moore, C.M.; Akers, N.L.; Hill, A.D.; Johnson, Z.C.; Minteer, S.D. Improving the Environment for Immobilized Dehydrogenase Enzymes by Modifying Nafion with Tetraalkylammonium Bromides. Biomacromolecules 2004, 5, 1241–1247. [Google Scholar] [CrossRef]
- Meredith, S.; Xu, S.; Meredith, M.T.; Minteer, S.D. Hydrophobic Salt-Modified Nafion for Enzyme Immobilization and Stabilization. J. Vis. Exp. 2012, 65, e3949. [Google Scholar] [CrossRef]
- Mora, A.; Mahlknecht, J.; Rosales-Lagarde, L.; Hernández-Antonio, A. Assessment of Major Ions and Trace Elements in Groundwater Supplied to the Monterrey Metropolitan Area, Nuevo León, Mexico. Environ. Monit. Assess. 2017, 189, 394. [Google Scholar] [CrossRef] [PubMed]
- Spinelli, D.; Fatarella, E.; Di Michele, A.; Pogni, R. Immobilization of Fungal (Trametes versicolor) Laccase onto Amberlite IR-120 H Beads: Optimization and Characterization. Process Biochem. 2013, 48, 218–223. [Google Scholar] [CrossRef]
- Fatarella, E.; Spinelli, D.; Ruzzante, M.; Pogni, R. Nylon 6 Film and Nanofiber Carriers: Preparation and Laccase Immobilization Performance. J. Mol. Catal. B Enzym. 2014, 102, 41–47. [Google Scholar] [CrossRef]
- Batra, B.; Yadav, M.; Pundir, C.S. L-Glutamate Biosensor Based on l-Glutamate Oxidase Immobilized onto ZnO Nanorods/Polypyrrole Modified Pencil Graphite Electrode. Biochem. Eng. J. 2016, 105, 428–436. [Google Scholar] [CrossRef]
- Pereira, A.S.; Tavares, P.; Limão-Vieira, P. Radiation in Bioanalysis; Springer: Cham, Switzerland, 2019; ISBN 9783030282462. [Google Scholar]
- Qian, X.; Wang, X.; Zhong, J.; Zhi, J.; Heng, F.; Zhang, Y.; Song, S. Effect of Fiber Microstructure Studied by Raman Spectroscopy upon the Mechanical Properties of Carbon Fibers. J. Raman Spectrosc. 2019, 50, 665–673. [Google Scholar] [CrossRef]
- Castellanos-Gomez, A.; Barkelid, M.; Goossens, A.M.; Calado, V.E.; Van Der Zant, H.S.J.; Steele, G.A. Laser-Thinning of MoS2: On Demand Generation of a Single-Layer Semiconductor. Nano Lett. 2012, 12, 3187–3192. [Google Scholar] [CrossRef] [PubMed]
- Windom, B.C.; Sawyer, W.G.; Hahn, D.W. A Raman Spectroscopic Study of MoS2 and MoO3: Applications to Tribological Systems. Tribol. Lett. 2011, 42, 301–310. [Google Scholar] [CrossRef]
- Zeng, J.; Jean, D.I.; Ji, C.; Zou, S. In Situ Surface-Enhanced Raman Spectroscopic Studies of Nafion Adsorption on Au and Pt Electrodes. Langmuir 2012, 28, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Abdellaoui, S.; Kitt, J.P.; Irvine, C.; Harris, J.M.; Minteer, S.D.; Korzeniewski, C. Confocal Raman Microscopy for the Determination of Protein and Quaternary Ammonium Ion Loadings in Biocatalytic Membranes for Electrochemical Energy Conversion and Storage. Anal. Chem. 2017, 89, 13290–13298. [Google Scholar] [CrossRef]
- Mailley, P.; Cummings, E.A.; Mailley, S.; Cosnier, S.; Eggins, B.R.; McAdams, E. Amperometric Detection of Phenolic Compounds by Polypyrrole-Based Composite Carbon Paste Electrodes. In Proceedings of the Bioelectrochemistry; Elsevier: Amsterdam, The Netherlands, 2004; Volume 63, pp. 291–296. [Google Scholar]
- Sumayya, A.; Yohannan Panicker, C.; Varghese, H.T.; Harikumar, B. Vibrational Spectroscopic Studies and AB Initio Calculations of L-Glutamic Acid 5-Amide. Rasayan J. Chem. 2008, 1, 548–555. [Google Scholar]
- Baker, M.J.; Hussain, S.R.; Lovergne, L.; Untereiner, V.; Hughes, C.; Lukaszewski, R.A.; Thiéfin, G.; Sockalingum, G.D. Developing and Understanding Biofluid Vibrational Spectroscopy: A Critical Review. Chem. Soc. Rev. 2016, 45, 1803–1818. [Google Scholar] [CrossRef]
- Movasaghi, Z.; Rehman, S.; Rehman, I.U. Raman Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2007, 42, 493–541. [Google Scholar] [CrossRef]
- Yang, S.; Hai, F.I.; Nghiem, L.D.; Price, W.E.; Roddick, F.; Moreira, M.T.; Magram, S.F. Understanding the Factors Controlling the Removal of Trace Organic Contaminants by White-Rot Fungi and Their Lignin Modifying Enzymes: A Critical Review. Bioresour. Technol. 2013, 141, 97–108. [Google Scholar] [CrossRef]
- Donarelli, M.; Ottaviano, L. 2D Materials for Gas Sensing Applications: A Review on Graphene Oxide, MoS2, WS2 and Phosphorene. Sensors 2018, 18, 3638. [Google Scholar] [CrossRef]
- Saraf, M.; Natarajan, K.; Saini, A.K.; Mobin, S.M. Small Biomolecule Sensors Based on an Innovative MoS2–RGO Heterostructure Modified Electrode Platform: A Binder-Free Approach. Dalt. Trans. 2017, 46, 15848–15858. [Google Scholar] [CrossRef]
- Barua, S.; Dutta, H.S.; Gogoi, S.; Devi, R.; Khan, R. Nanostructured MoS2-Based Advanced Biosensors: A Review. ACS Appl. Nano Mater. 2018, 1, 2–25. [Google Scholar] [CrossRef]
- Rodríguez, M.C.; Rivas, G.A. Glassy Carbon Paste Electrodes Modified with Polyphenol Oxidase: Analytical Applications. Anal. Chim. Acta 2002, 459, 43–51. [Google Scholar] [CrossRef]
- Economou, A. Enzymatic Biosensors. In Portable Biosensing of Food Toxicants and Environmental Pollutants; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2013; pp. 123–160. [Google Scholar]
- Janata, J. Principles of Chemical Sensors, 2nd ed.; Springer: New York, NY, USA, 2009; ISBN 9780387699301. [Google Scholar]
- Mross, S.; Pierrat, S.; Zimmermann, T.; Kraft, M. Microfluidic Enzymatic Biosensing Systems: A Review. Biosens. Bioelectron. 2015, 70, 376–391. [Google Scholar] [CrossRef]
- Milton, R.D.; Wu, F.; Lim, K.; Abdellaoui, S.; Hickey, D.P.; Minteer, S.D. Promiscuous Glucose Oxidase: Electrical Energy Conversion of Multiple Polysaccharides Spanning Starch and Dairy Milk. ACS Catal. 2015, 5, 7218–7225. [Google Scholar] [CrossRef]
- Ranjbakhsh, E.; Bordbar, A.K.; Abbasi, M.; Khosropour, A.R.; Shams, E. Enhancement of Stability and Catalytic Activity of Immobilized Lipase on Silica-Coated Modified Magnetite Nanoparticles. Chem. Eng. J. 2012, 179, 272–276. [Google Scholar] [CrossRef]
- De Lima Citolino, L.V.; Braunger, M.L.; Oliveira, V.J.R.; Olivati, C.A. Study of the Nanostructure Effect on Polyalkylthiophene Derivatives Films Using Impedance Spectroscopy. Mater. Res. 2017, 20, 874–881. [Google Scholar] [CrossRef]
- RoyChoudhury, S.; Umasankar, Y.; Hutcheson, J.D.; Lev-Tov, H.A.; Kirsner, R.S.; Bhansali, S. Uricase Based Enzymatic Biosensor for Non-Invasive Detection of Uric Acid by Entrapment in PVA-SbQ Polymer Matrix. Electroanalysis 2018, 30, 2374–2385. [Google Scholar] [CrossRef]
- Banica, F.-G. Chemical Sensors and Biosensors: Fundamentals and Applications; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2012; ISBN 0470710667. [Google Scholar]
- Cernat, A.; Tertis, M.; Griveau, S.; Bedioui, F.; Sandulescu, R. New Modified Electrodes with HRP Immobilized in Polymeric Films for Paracetamol Analysis. Farmacia 2012, 60, 1–12. [Google Scholar]
- Cernat, A.; Griveau, S.; Richard, C.; Bedioui, F.; Sǎndulescu, R. Horseradish Peroxidase Nanopatterned Electrodes by Click Chemistry: Application to the Electrochemical Detection of Paracetamol. Electroanalysis 2013, 25, 1369–1372. [Google Scholar] [CrossRef]
- Maghear, A.; Cristea, C.; Marian, A.; Marian, I.O.; Sǎndulescu, R. A Novel Biosensor for Acetaminophen Detection with Romanian Clays and Conductive Polymeric Films. Farmacia 2013, 61, 1–11. [Google Scholar]
- Garcia, L.F.; Benjamin, S.R.; Antunes, R.S.; Lopes, F.M.; Somerset, V.S.; Gil, E.d.S. Solanum Melongena Polyphenol Oxidase Biosensor for the Electrochemical Analysis of Paracetamol. Prep. Biochem. Biotechnol. 2016, 46, 850–855. [Google Scholar] [CrossRef]
- Antunes, R.S.; Garcia, L.F.; Somerset, V.S.; de Souza Gil, E.; Lopes, F.M. The Use of a Polyphenoloxidase Biosensor Obtained from the Fruit of Jurubeba (Solanum paniculatum L.) in the Determination of Paracetamol and Other Phenolic Drugs. Biosensors 2018, 8, 36. [Google Scholar] [CrossRef]
- Brack, W.; Dulio, V.; Slobodnik, J. The NORMAN Network and Its Activities on Emerging Environmental Substances with a Focus on Effect-Directed Analysis of Complex Environmental Contamination. Environ. Sci. Eur. 2012, 24, 29. [Google Scholar] [CrossRef]
- Fernández-Fernández, M.; Sanromán, M.Á.; Moldes, D. Recent Developments and Applications of Immobilized Laccase. Biotechnol. Adv. 2013, 31, 1808–1825. [Google Scholar] [CrossRef] [PubMed]

| Sample | Conc. (mg/mL) | Activity (U/mL) | Specific Activity (U/mg) |
|---|---|---|---|
| TvL | 1.32 ± 0.64 | 81.00 ± 30 | 61.50 ± 3.29 |
| LacI | 0.31 ± 0.48 | 33.87 ± 1.38 | 108.17 ± 7.35 |
| LacII | 0.48 ± 0.17 | 74.49 ± 0.97 | 155.11 ± 1.50 |
| Laccase | KMapp | |
|---|---|---|
| Free | Immobilized | |
| TvL | 42.07 ± 3.136 | 123 ± 26.96 |
| LacI | 16.64 ± 2.548 | 58.68 ± 10.12 |
| LacII | 34.51 ± 5.857 | 35.57 ± 4.79 |
| MoS2 | TvL | LacI | LacII | |||
|---|---|---|---|---|---|---|
| Concentration | KMapp (µM) | Jmax (µA/cm2) | KMapp (µM) | Jmax (µA/cm2) | KMapp (µM) | Jmax (µA/cm2) |
| Unmodified | ||||||
| - | 1659 ± 250 | 51.11 ± 2.59 | 2164 ± 151 | 53.68 ± 1.43 | 1908 ± 77 | 129.3 ± 1.76 |
| Modified with MoS2 | ||||||
| 0.5mg/mL | 2613 ± 254 | 29.80 ± 1.09 | 2474 ± 65 | 45.39 ± 0.44 | 1501 ± 99 | 81.05 ± 1.68 |
| 1 mg/mL | 2952 ± 155 | 41.03 ± 0.85 | 2702 ± 143 | 58.01 ± 1.17 | 1656 ± 37 | 234.7± 1.65 |
| 2 mg/mL | 1791 ± 99 | 49.84 ± 0.91 | 2429 ± 232 | 34.34 ± 1.30 | 1604 ± 66 | 142.2± 1.81 |
| 5 mg/mL | 2733 ± 253 | 40.33 ± 1.43 | 3535 ± 250 | 22.07 ± 0.65 | 4176 ± 331 | 29.54 ± 1.04 |
| Bioelectrode | Linear Range (µM) | Sensitivity (µA/µM cm2) | R2 |
|---|---|---|---|
| CP-LacII | 66–395 | 0.058 | 0.9901 |
| CP-MoS2-LacII | 66–525 | 0.108 | 0.9948 |
| CP-TvL | 132–395 | 0.021 | 0.9901 |
| CP-MoS2-TvL | 66–329 | 0.015 | 0.9901 |
| Electrode * | Detection Method | Sample | Linear Range (µM) | Sensitivity (µA/µM cm2) | LOD (µM) | Ref. |
|---|---|---|---|---|---|---|
| GCPE-PPO | Amp (−0.1 V) | 50 M phosphate buffer solution pH 7.4. | Up to70 | 0.015 µA/µM | 7.8 | [60] |
| GCE-HRP @ PAA-BIS | Amp (−0.1 V) | 50 mM sodium phosphate buffer pH 7.0 with 100 mM KCl | 5.6–331.1 | 0.069 | 1.7 | [13] |
| GCE-HRP-PPY SPE-HRP-PPY | Amp (−0.175 V) | 0.2 mM H2O2 in phosphate buffer pH = 7.4 | 9.3–83.7 3.1–55.9 | - - | 6.5 1.52 | [69] |
| GCE-nano PPY-HRP GCE-flat PPY-HRP | Amp (−0.2 V) | Phosphate buffer solution (pH 7.4) | 5–60 5–300 | 0.050 0.002 | 0.1 4.1 | [70] |
| GCE-clay-PEI-HRP | Amp (0 V) | 100 mM phosphate buffer saline (pH 7.4) | 5.25–49.5 | 0.013 µA/µM | 0.63 | [71] |
| SPE-CoPC/Tyr | CV | 100 m M phosphate-buffer pH 6/100 mM KCl | up to 40 | −0.088 | 0.5 | [15] |
| CPE-EP-PPO | DPV 10 mV/s | 100 mM phosphate buffer (pH 6.0) | 600–1150 | - | 5.0 | [72] |
| CPE-PPO | DPV 10 mV/s | 100 mM phosphate buffer pH 7.0 | 5–245 | - | 3.0 | [73] |
| CP-LacII | Amp −0.1 V | 100 mM citric acid/2 M KOH buffer (pH 4) | 66–395 | 0.058 | - | Present study |
| CP-TvL | 132–395 | 0.021 | - | |||
| CP-MoS2-LacII | 66–525 | 0.108 | 0.2 | |||
| CP-MoS2-TvL | 66–329 | 0.015 | 2.0 | |||
| CP-MoS2-LacII | Amp −0.1 V | Groundwater (50 mM citric acid/2 M KOH) | 1–155.1 | 0.017 | 0.50 | Present study |
| CP-MoS2-TvL | 29.82–155.1 | 0.005 | 24.88 |
| Electrode | Linear Range (µM) | Linear Range (µg/L) | Sensitivity (nA/µMcm2) | R2 | LOD (µM) | LOD (µg/L) |
|---|---|---|---|---|---|---|
| CP-MoS2-Lac | 1–155 | 151.13–23,436.1 | 17.7 | 0.9992 | 0.50 | 75.57 |
| CP-MoS2-TvL | 29.82–155 | 4507.84–23,436.1 | 5.6 | 0.9933 | 24.88 | 3760.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera-Domínguez, M.; Lim, K.; Aguilar-Hernández, I.; García-García, A.; Minteer, S.D.; Ornelas-Soto, N.; Garcia-Morales, R. Detection of Acetaminophen in Groundwater by Laccase-Based Amperometric Biosensors Using MoS2 Modified Carbon Paper Electrodes. Sensors 2023, 23, 4633. https://doi.org/10.3390/s23104633
Herrera-Domínguez M, Lim K, Aguilar-Hernández I, García-García A, Minteer SD, Ornelas-Soto N, Garcia-Morales R. Detection of Acetaminophen in Groundwater by Laccase-Based Amperometric Biosensors Using MoS2 Modified Carbon Paper Electrodes. Sensors. 2023; 23(10):4633. https://doi.org/10.3390/s23104633
Chicago/Turabian StyleHerrera-Domínguez, Marcela, Koun Lim, Iris Aguilar-Hernández, Alejandra García-García, Shelley D. Minteer, Nancy Ornelas-Soto, and Raúl Garcia-Morales. 2023. "Detection of Acetaminophen in Groundwater by Laccase-Based Amperometric Biosensors Using MoS2 Modified Carbon Paper Electrodes" Sensors 23, no. 10: 4633. https://doi.org/10.3390/s23104633
APA StyleHerrera-Domínguez, M., Lim, K., Aguilar-Hernández, I., García-García, A., Minteer, S. D., Ornelas-Soto, N., & Garcia-Morales, R. (2023). Detection of Acetaminophen in Groundwater by Laccase-Based Amperometric Biosensors Using MoS2 Modified Carbon Paper Electrodes. Sensors, 23(10), 4633. https://doi.org/10.3390/s23104633






