3.1. Voltammetric Studies
Taking into account the electrochemical behaviour of the BDD electrode in the presence of sulfide and nitrite alone in 0.1 M Na
2SO
4 supporting electrolyte (results not shown here) in relation with the oxidation potential value (+0.9 V/SCE for S
2− and +1.4 V/SCE for NO
2−) and the sensitivity (55 µ·Am·M
−1 for S
2− and 67 µ·Am·M
−1 for NO
2−), the question that was raised is whether nitrite can be detected in the presence of sulfide envisaging their simultaneous detection. In
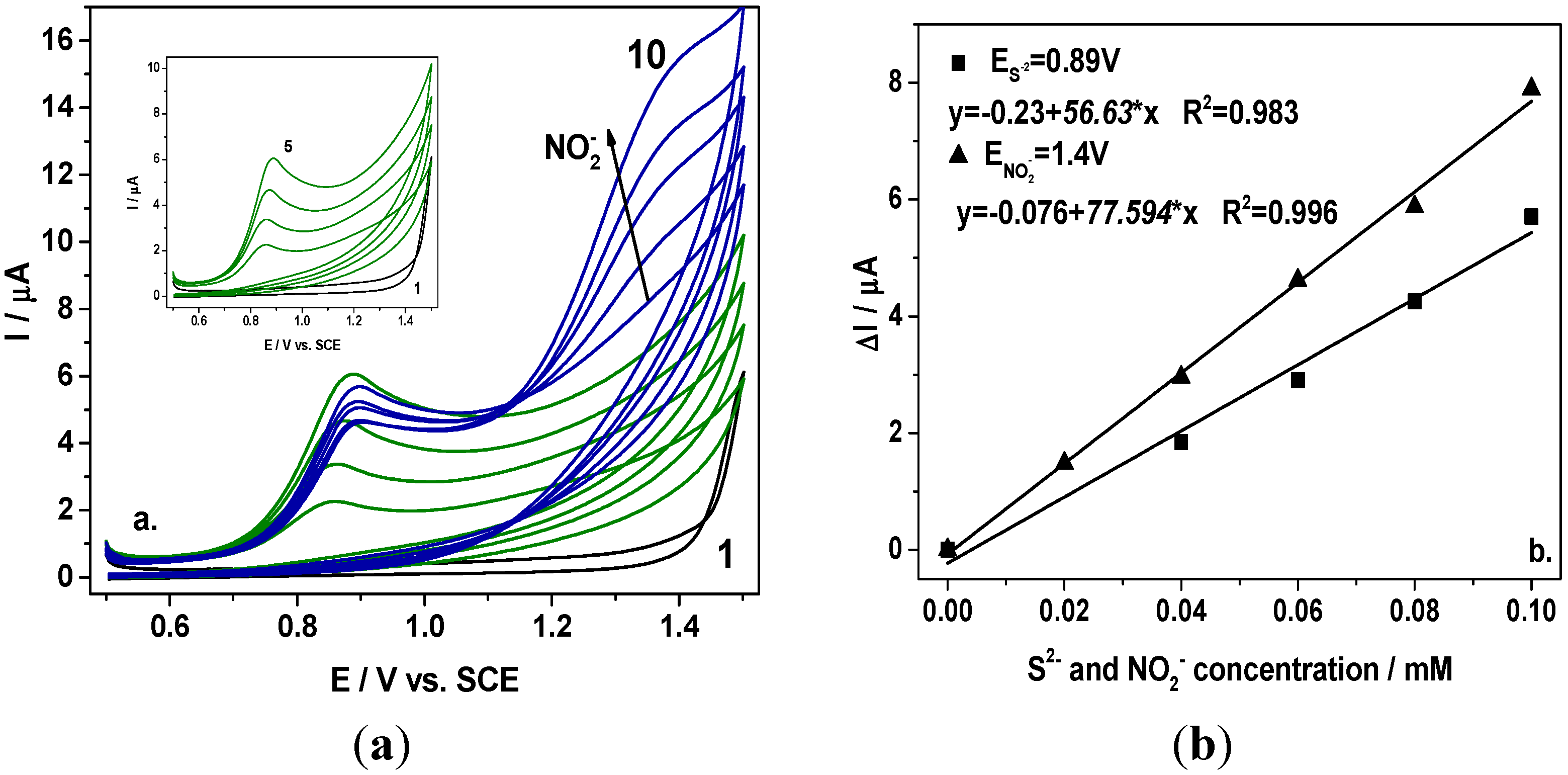
Figure 1 are presented the cyclic voltammograms recorded at the BDD electrode in the presence of sulfide within the concentration range from 0.02 mM to 0.1 mM and further, after adding nitrite within the same concentration range. It can be noticed that nitrite oxidation started at +1.15 V/SCE and the oxidation peaks are well-separated for each anion. In comparison with the results obtained for individual detection of nitrite, the presence of sulfide led to a shifting of the potential value at which the nitrite oxidation started to a more positive potential. However, the detection potential value was not influenced, and the nitrite detection was recorded at +1.4 V/SCE.
Figure 1.
(a) Cyclic voltammograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM sulfide (curves 2–5; Inset of figure), and 0.02–0.1 mM nitrite (curves 6–10) at a potential scan rate of 0.05 Vs−1 in a potential range from +0.5 to +1.5 V/SCE; (b) Calibration plots of the current vs. anion concentration recorded at E = +0.89 V vs. SCE for sulfide and E = +1.4 V vs. SCE for nitrite.
Figure 1.
(a) Cyclic voltammograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM sulfide (curves 2–5; Inset of figure), and 0.02–0.1 mM nitrite (curves 6–10) at a potential scan rate of 0.05 Vs−1 in a potential range from +0.5 to +1.5 V/SCE; (b) Calibration plots of the current vs. anion concentration recorded at E = +0.89 V vs. SCE for sulfide and E = +1.4 V vs. SCE for nitrite.
A linear dependence between oxidation peak height and anion concentration was found with good correlation coefficient. The sensitivities determined under these conditions were almost similarly with those obtained for the detection of individual anion. It should be mentioned that within the framework of this scheme, the sulfide is detected individually and nitrite is detected in the presence of sulfide.
These results are very promising and further experiments were conducted to improve the sensitivity for simultaneous voltammetric detection of nitrite and sulfide. Thus, the differential-pulsed technique (DPV) was applied under the operating conditions of 0.2 V modulation amplitude and 0.01 V step potential by alternative adding of each sulfide and nitrite species.
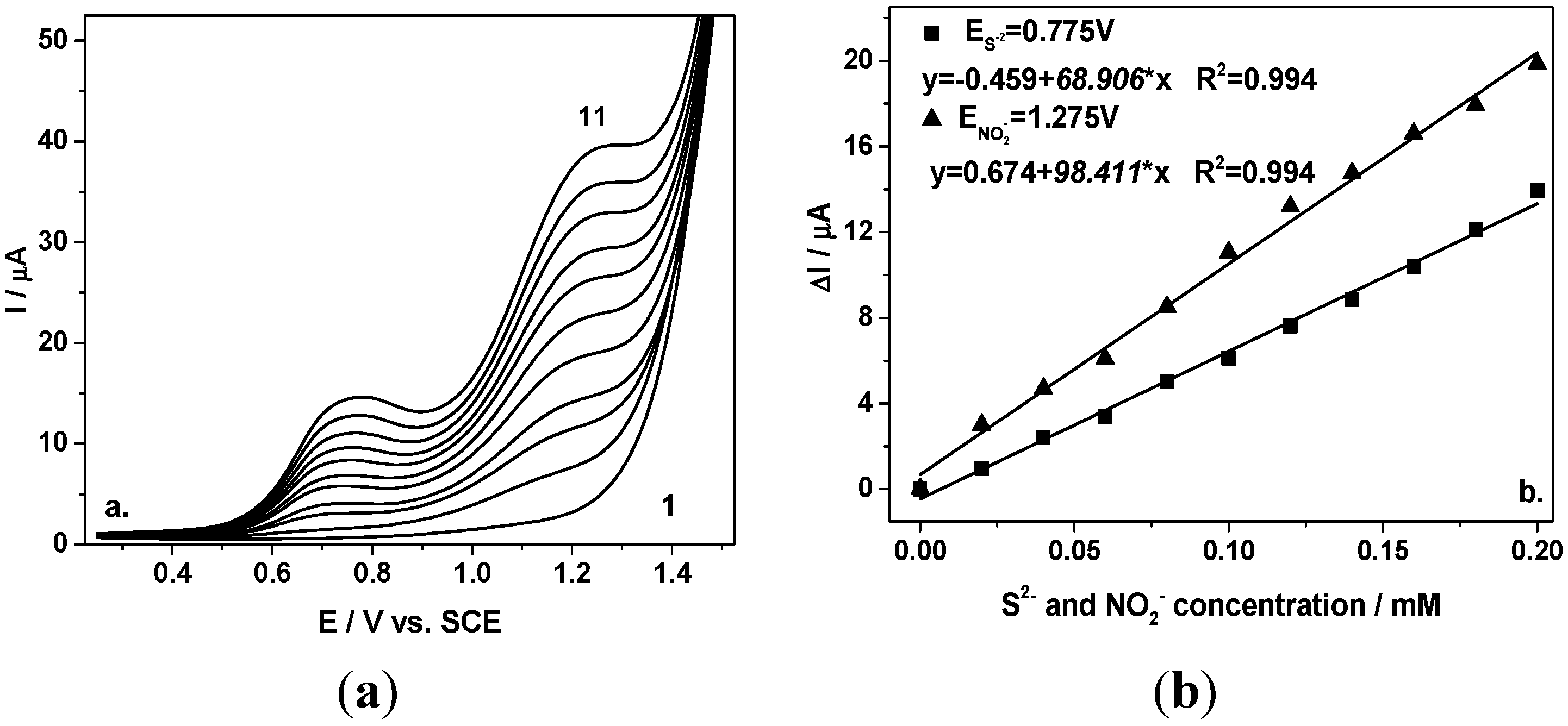
Figure 2a depicts the DPVs recorded with the BDD electrode under the conditions presented above. Linear dependences of peak current versus sulfide and nitrite concentration, respectively, with good correlation coefficients were achieved.
In comparison with CV, the electroanalytical performance related to the sensitivity and the detection potential was improved. Better sensitivity was achieved for both anions, 68.9 vs. 56.63 µ·Am·M−1 for S2− and 98.41 vs. 77.59 µ·Am·M−1 for NO2−. Also, the detection potentials were shifted to less positive potential, maintaining the same about 500 mV potential separation between the two detection potentials.
Figure 2.
(a) Differential-pulsed voltammograms recorded at the BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in a mixture of 0.02–0.2 mM sulfide and nitrite (curves 2–11), under 0.2 V modulation amplitude, 0.01 V step potential, and 0.1 V·s−1 scan rate in a potential range from +0.25 to +1.5 V/SCE; (b) Calibration plot of the current vs. anions concentration recorded at E= +0.775 V vs. SCE for sulfide, respectively, and at E = +1.275 V vs. SCE for nitrite.
Figure 2.
(a) Differential-pulsed voltammograms recorded at the BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in a mixture of 0.02–0.2 mM sulfide and nitrite (curves 2–11), under 0.2 V modulation amplitude, 0.01 V step potential, and 0.1 V·s−1 scan rate in a potential range from +0.25 to +1.5 V/SCE; (b) Calibration plot of the current vs. anions concentration recorded at E= +0.775 V vs. SCE for sulfide, respectively, and at E = +1.275 V vs. SCE for nitrite.
In order to further improve the electroanalytical performance of BDD for simultaneous detection of sulfide and nitrite, the square-wave voltammetry technique (SWV) was applied under various operating conditions in order to optimize the results with the best sensitivities and detection potential. A similar working protocol to that described previously was used for the simultaneous detection of both anions. An optimization procedure was performed under the operating conditions gathered in
Table 1, which also presents the sensitivity and the detection potential for each anion.
To check the reproducibility and the interference of sulfide on nitrite detection, two working protocols related to anion addition were proposed. The first representing the detection of nitrite in the presence of sulfide consisted of adding a sulfide concentration up to desired maximum level followed by adding nitrite concentrations (
Figure 3a). The second working protocol consisted of alternative additions of each anion (
Figure 4a). For each working protocol, the linear calibration plots of current
versus each anion concentration showed good correlation coefficients (
Figure 3b and
Figure 4b). Based on these results, it can be observed that the sensitivities for each anion under both working protocols were similar, which shows that the results are reproducible.
Table 1.
Operating conditions for SWV and electroanalytical performance recorded at BDD electrode for simultaneous detection of sulfide and nitrite.
Table 1.
Operating conditions for SWV and electroanalytical performance recorded at BDD electrode for simultaneous detection of sulfide and nitrite.
| a (V) | ΔEs (V) | f (Hz) | Anion | Eox (V/SCE) | Sensitivity (µA/mM) | R2 |
|---|
| 0.01 | 0.001 | 50 | S2− | 0.92 | 11.4 | 0.996 |
| NO2− | 1.355 | 7.7 | 0.975 |
| 100 | S2− | 0.918 | 14.6 | 0.998 |
| NO2− | 1.385 | 11.0 | 0.970 |
| 0.1 | 0.01 | 10 | S2− | 0.85 | 150.4 | 0.997 |
| NO2− | 1.35 | 135.7 | 0.992 |
| 0.2 | 0.01 | 10 | S2− | 0.8 | 314.0 | 0.998 |
| NO2− | 1.3 | 232.0 | 0.993 |
| 0.02 | 10 | S2− | 0.805 | 280.4 | 0.990 |
| NO2− | 1.3 | 183.7 | 0.966 |
| 0.5 | 0.01 | 10 | S2− | 0.63 | 292.0 | 0.997 |
| NO2− | 1.02 | 198.0 | 0.973 |
| 0.02 | 10 | S2− | 0.63 | 396.0 | 0.997 |
| NO2− | 1.02 | 300.0 | 0.981 |
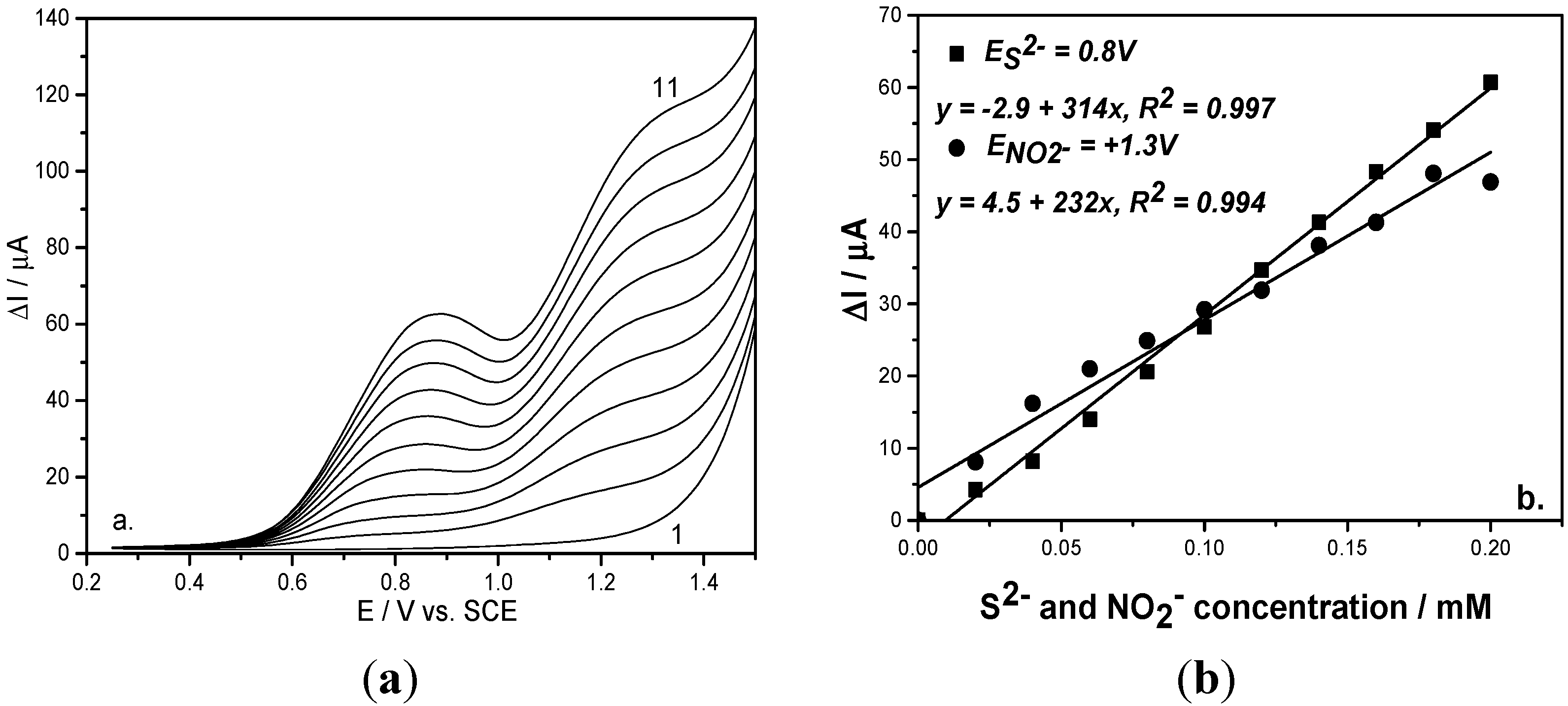
Figure 3.
(a) Square-wave voltammograms recorded on a BDD electrode under 0.01 V step potential and 0.2 V modulation amplitude, 10 Hz frequency, 0.1 V·s−1 scan rate, between −0.25 and +1.5 V vs. SCE in 0.1 M Na2SO4 supporting electrolyte (curve 1) and in the presence of mixtures of 0.02–0.2 mM sulfide and nitrite concentrations (curves 2–11); (b) Calibration plots of the current vs. anion concentrations recorded at: E = +0.8 V/SCE for sulfide concentration and E = +1.3 V/SCE for nitrite concentration.
Figure 3.
(a) Square-wave voltammograms recorded on a BDD electrode under 0.01 V step potential and 0.2 V modulation amplitude, 10 Hz frequency, 0.1 V·s−1 scan rate, between −0.25 and +1.5 V vs. SCE in 0.1 M Na2SO4 supporting electrolyte (curve 1) and in the presence of mixtures of 0.02–0.2 mM sulfide and nitrite concentrations (curves 2–11); (b) Calibration plots of the current vs. anion concentrations recorded at: E = +0.8 V/SCE for sulfide concentration and E = +1.3 V/SCE for nitrite concentration.
In relation with the best sensitivity and less positive detection potential values, the optimum operating conditions for SWV were determined,
i.e., 0.5 V modulation amplitude (a), 0.02 V step potential (ΔE
s) and 10 Hz frequency (f). Also, comparable results in relation with the sensitivity were achieved for 0.2 V modulation amplitude, 0.01 V step potential and 10 Hz frequency and the difference consisted of a more positive detection potential value. However, it must be mentioned that the better potential separation was reached for these last operating conditions. The choice of operating conditions will be based on the concrete objective related to the final application. In comparison with the voltammetric techniques presented above, CV and DPV, the SWV technique allowed the best analytical performance for simultaneous sulfide and nitrite detection (see
Table 1).
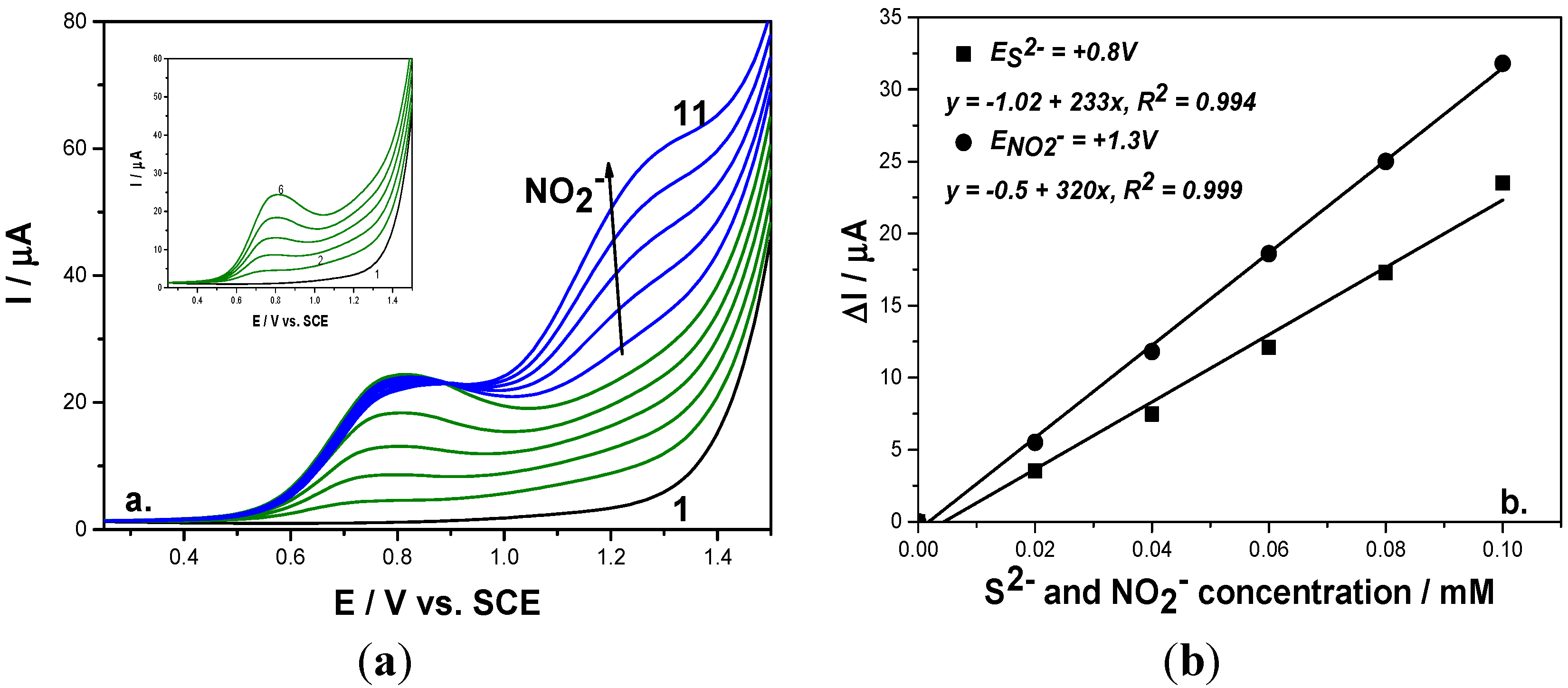
Figure 4.
(a) Square-wave voltammograms recorded on a BDD electrode under 0.01 V step potential and 0.2 V modulation amplitude, 10 Hz frequency, scan rate 0.1 V·s−1, between −0.25 and +1.5 V vs. SCE in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM mixtures of sulfide (curves 2–6; Inset of figure), and 0.02–0.1 mM nitrite (curves 7–11). (b) Calibration plots of the current vs. anion concentration recorded at: E = 0.8 V/SCE vs. sulfide concentration and E = +1.3 V/SCE vs. nitrite concentration.
Figure 4.
(a) Square-wave voltammograms recorded on a BDD electrode under 0.01 V step potential and 0.2 V modulation amplitude, 10 Hz frequency, scan rate 0.1 V·s−1, between −0.25 and +1.5 V vs. SCE in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM mixtures of sulfide (curves 2–6; Inset of figure), and 0.02–0.1 mM nitrite (curves 7–11). (b) Calibration plots of the current vs. anion concentration recorded at: E = 0.8 V/SCE vs. sulfide concentration and E = +1.3 V/SCE vs. nitrite concentration.
3.2. Amperometric Studies
It is well-known that chronoamperometry is the easiest electrochemical technique for detection applications. Having CV results as a reference basis, the operating conditions for CA were set up to detect each anion by applying two potential levels characteristic of the detection potential corresponding to sulfide (+0.85 V/SCE) and nitrite (+1.25 V/SCE) oxidation. It should be mentioned that under CA conditions, by applying the second detection potential level of +1.4 V/SCE no reproducible results were obtained. A possible reason for this situation could be the initiation of oxygen evolution that overlapped with the oxidation process. The current corresponding to the oxygen evolution cannot be controlled and thus, no reproducible results were achieved at the potential value of +1.4 V/SCE.
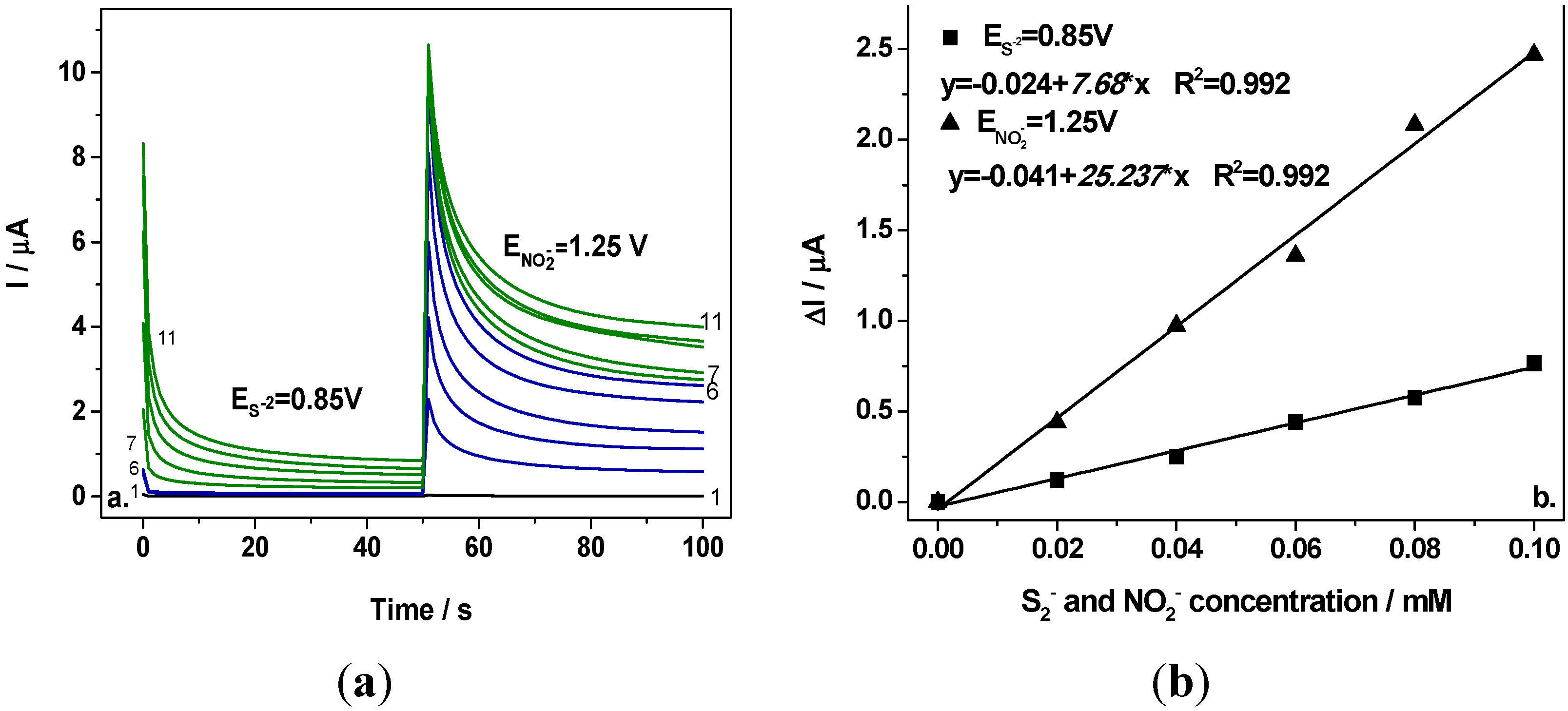
Figure 5a shows two-level chronoamperograms recorded at the first potential level of +0.85 V/SCE and the second potential level of +1.25 V/SCE in 0.1 M Na
2SO
4 supporting electrolyte (curve 1) and in the presence of various nitrite concentrations (curves 2–6) and the mixture of 0.1 mM nitrite and various concentrations of sulfide (curves 7–11). It can be easily noticed that at the first potential level corresponding to the sulfide oxidation no signal corresponding to nitrite detection was recorded (curves 2–6 at E = +0.85 V/SCE) and the signal was recorded only by adding sulfide, which increased linearly with sulfide concentration (see
Figure 5b, curve 1). At the second potential level operated at E = +1.25 V/SCE, the useful currents recorded after running for 100 s (
Figure 5a, curves 2–6) depended linearly on the nitrite concentration (see
Figure 5b, curve 2). For each anion detection a good correlation coefficient was achieved. The sensitivities determined under CA operating conditions were lower in comparison with CV, which could be explained by a possible electrode fouling that is characteristic when applying chronoamperometry by current measurements at the detection potential for long time. Analyte and the oxidation products should be responsible for the electrode fouling. Moreover, the presence of sulfide is detected also, at the potential level corresponding to nitrite detection (+1.25 V/SCE) that means that sulfide should interfere with nitrite detection. At this potential level, a cumulative signal corresponding to the presence of the sum of both anions will be detected. The main problem in this situation is that the useful current corresponding to the presence of sulfide did not depend linearly on the sulfide concentration at this potential level and no quantitative assessment of nitrite can be achieved under these conditions.
Figure 5.
(a) Chronoamperograms recorded at BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM nitrite (curves 2–6) in a mixture of 0.02–0.1 mM sulfide and 0.1 mM nitrite (curves 7–11), at the potential values of E1 = +0.85 V/SCE for sulfide and E2 = +1.25 V/SCE for nitrite; (b) Calibration plot of the current vs. anion concentration recorded at E1 = +0.85 V/SCE for sulfide (curves 7–11), respectively E2 = +1.25 V/SCE (curves 2–6) for nitrite.
Figure 5.
(a) Chronoamperograms recorded at BDD electrode in 0.1 M Na2SO4 supporting electrolyte (curve 1) in the presence of 0.02–0.1 mM nitrite (curves 2–6) in a mixture of 0.02–0.1 mM sulfide and 0.1 mM nitrite (curves 7–11), at the potential values of E1 = +0.85 V/SCE for sulfide and E2 = +1.25 V/SCE for nitrite; (b) Calibration plot of the current vs. anion concentration recorded at E1 = +0.85 V/SCE for sulfide (curves 7–11), respectively E2 = +1.25 V/SCE (curves 2–6) for nitrite.
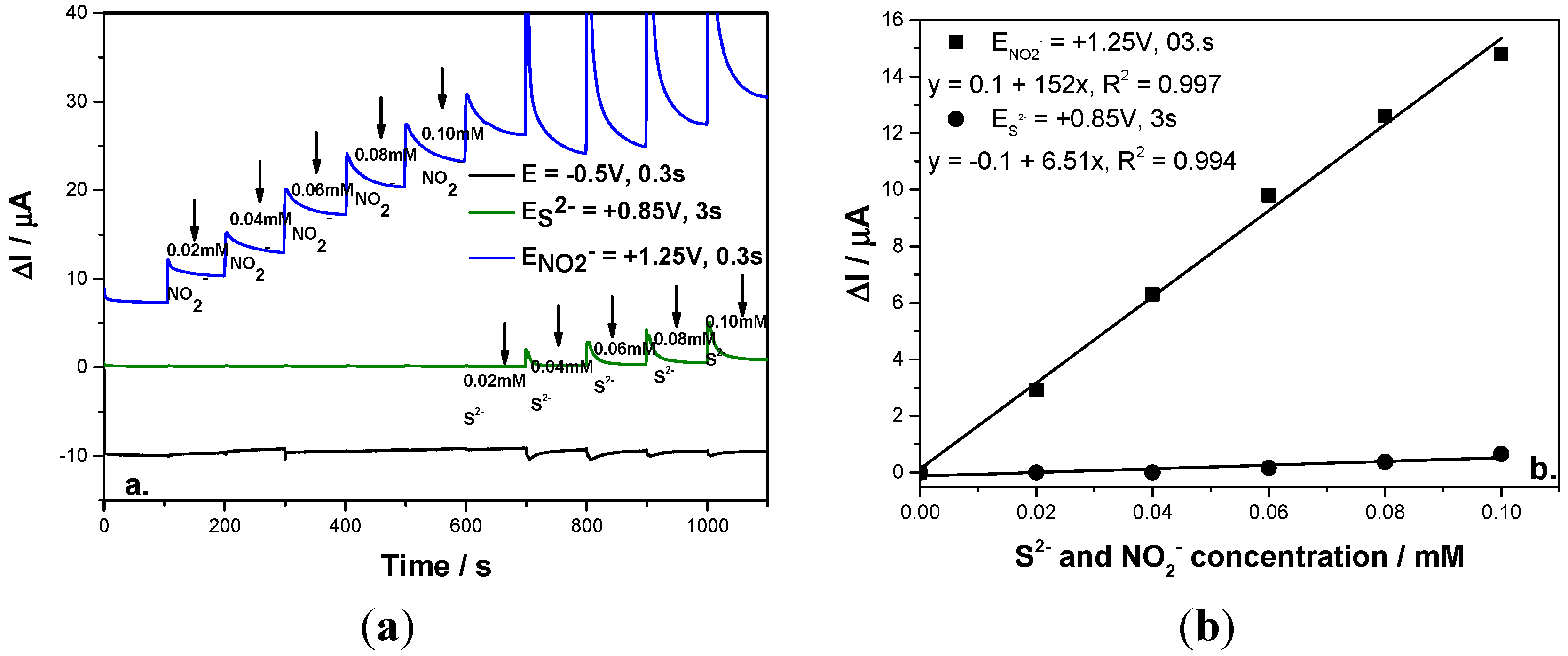
In order to simultaneously detect each anion without the interference of the other as an amperometric detector, the multiple-pulsed amperometry technique was explored and the detection scheme was elaborated based on the potential levels number, values and operating time. In the first stage, the multiple-pulsed amperometry technique was operated under similar conditions to the CA and even if for nitrite the sensitivity was better, no signal for sulfide detection was found (results are not shown here). Under these conditions, the reference points for the potential values for three levels based multiple-pulsed amperometry applying were selected based on the curve CV shape. The two detection potential values were chosen based on the oxidation potential value for each anion, i.e., +0.85 V /SCE for sulfide and +1.25 V/SCE for nitrite, the last one being considered also as a cleaning step to avoid fouling the electrode and the third one maintained at −0.5 V/SCE as a conditioning step. This scheme was applied for the continuous addition of nitrite for concentrations ranging from 0.02 mM to 0.1 mM followed by continuous addition of sulfide at the same concentration range to be able to follow the signals recorded for each anion.
To avoid the interference of sulfide in nitrite detection, a very important parameter in this scheme was found to be the time at which each potential level is maintained. This parameter was optimized in relation with the time allocated to the oxidation of both anions. It was found that by applying the same time for sulfide and nitrite oxidation, only the signal for nitrite detection was recorded and sulfide was not detected, probably due to the fact the sulfide oxidation process is slower in comparison with the nitrite oxidation process in terms of the mass transport. By increasing the time corresponding to the sulfide oxidation to assure a 10:1:1 ratio between sulfide oxidation time:nitrite oxidation time:cleaning time, sulfide detection was possible with the similar sensitivity to that achieved by the CA technique and no influence on nitrite detection was found by applying the above-described MPA (see
Figure 6a). The concentration step for both anions was 0.02 mM that was added continuously into the supporting electrolyte and both concentrations ranged from 0.02 mM to 0.1 mM. Moreover, the sensitivity for the nitrite detection was much improved in comparison with CA (
Figure 6b).
Figure 6.
(a) Multiple-pulsed amperograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte and adding consecutively and continuously each of 0.02 mM nitrite and respective, sulfide, recorded at E1 = −0.5 V/SCE, E2 = +0.85 V/SCE, E3 = +1.25 V/SCE; (b) Calibration plot of the current vs. anion concentration recorded at: E2 = +0.85 V/SCE vs. sulfide concentration and E3 = +1.3 V/SCE vs. nitrite concentration.
Figure 6.
(a) Multiple-pulsed amperograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte and adding consecutively and continuously each of 0.02 mM nitrite and respective, sulfide, recorded at E1 = −0.5 V/SCE, E2 = +0.85 V/SCE, E3 = +1.25 V/SCE; (b) Calibration plot of the current vs. anion concentration recorded at: E2 = +0.85 V/SCE vs. sulfide concentration and E3 = +1.3 V/SCE vs. nitrite concentration.
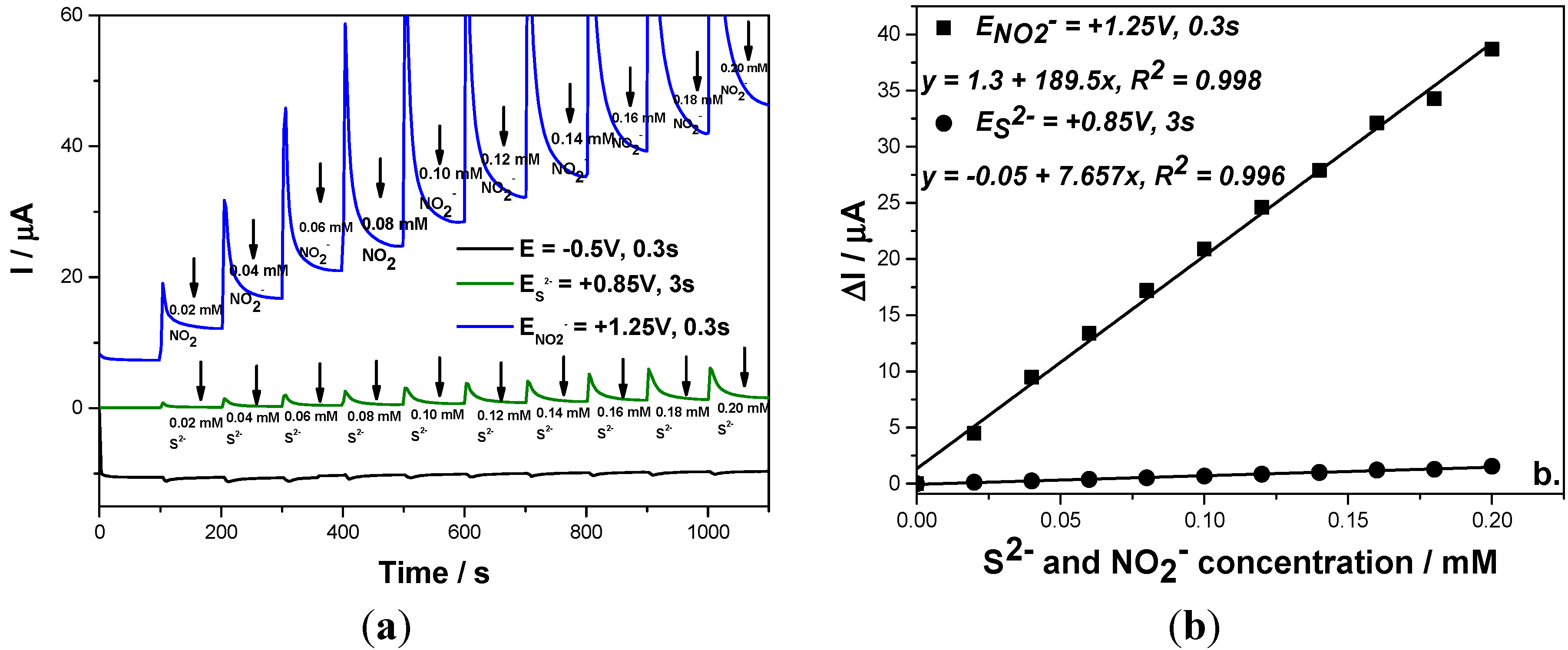
This detection scheme was also applied for the simultaneous amperometric detection of both anions in their mixtures and the results were reproducible (
Figure 7). Based on these results, it can be concluded that the simultaneous detection of sulfide and nitrite without interference with each other to other was achieved using a three levels-MPA detection procedure for which the pulses were applied continuously based on the following scheme:
- (a)
−0.5 V/SCE for a duration of 0.3 s, as conditioning step.
- (b)
+0.85 V/SCE for a duration of 3 s, which assure the sulfide oxidation.
- (c)
+1.25 V/SCE for a duration of 0.3 s, which assure the nitrite oxidation.
The electroanalytical parameters established for the simultaneous detection of sulfide and nitrite under the optimum operating conditions for each voltammetric/amperometric technique used are gathered in
Table 2. It can be noticed that the best electroanalytical parameters for the simultaneous voltammetric detection were acheived by the SWV technique, while for the simultaneous amperometric detection the three levels-MPA technique led to very good results, this technique being more suitable for
in-field practical applications.
Figure 7.
(a) Multiple-pulsed amperograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte and adding continuously a mixture of 0.02 mM NO2− and 0.02 mM S2−, recorded at E1 = −0.5 V/SCE, E2 = +0.85 V/SCE, E3 = +1.25 V/SCE; (b) Calibration plot of the current vs. anion concentration recorded at: E2 = +0.85 V/SCE vs. sulfide concentration and E3 = +1.3 V/SCE vs. nitrite concentration.
Figure 7.
(a) Multiple-pulsed amperograms recorded at a BDD electrode in 0.1 M Na2SO4 supporting electrolyte and adding continuously a mixture of 0.02 mM NO2− and 0.02 mM S2−, recorded at E1 = −0.5 V/SCE, E2 = +0.85 V/SCE, E3 = +1.25 V/SCE; (b) Calibration plot of the current vs. anion concentration recorded at: E2 = +0.85 V/SCE vs. sulfide concentration and E3 = +1.3 V/SCE vs. nitrite concentration.
Table 2.
The electroanalytical parameters of simultaneous detection of voltammetric/amperometric detection of sulfide and nitrite at a BDD electrode using different techniques.
Table 2.
The electroanalytical parameters of simultaneous detection of voltammetric/amperometric detection of sulfide and nitrite at a BDD electrode using different techniques.
| Tehnique | E,V/SCE | Sensitivity, µA/mM | Correlation Coefficient, R2 | LOD, mM |
|---|
| S2− | NO2− | S2− | NO2− | S2− | NO2− | S2− | NO2− |
|---|
| CV | 0.9 | 1.35 | 56.63 | 77.94 | 0.998 | 0.994 | 8.66 × 10−4 | 2.88 × 10−3 |
| DPV | 0.77 | 1.27 | 68.90 | 12.75 | 0.997 | 0.995 | 1.83 × 10−4 | 6.09 × 10−4 |
| SWV/ Step potential: 0.01 V; Modulation amplitude: 0.2 V; Frequency: 10 Hz | 0.90 | 1.3 | 314 | 232 | 0.997 | 0.987 | 5.55 × 10−5 | 1.84 × 10−4 |
| SWV/ Step potential: 0.01 V; Modulation amplitude: 0.5 V; Frequency: 10 Hz | 0.66 | 1.04 | 396 | 300 | 0.997 | 0.981 | 1.16 × 10−5 | 3.86 × 10−4 |
| CA | 0.85 | 1.25 | 7.68 | 25.23 * | 0.992 | 0.992 | 9.83 × 10−4 | 7.14 × 10−3 |
| MPA | 0.85 | 1.25 | 7.66 | 189 | 0.996 | 0.998 | 7.17 × 10−4 | 2.39 × 10−3 |
A recovery test was performed by analyzing three parallel tapwater samples, which contained 0.05 mM sulfide and 0.05 mM nitrite. This test was run in 0.1 M Na
2SO
4 as supporting electrolyte and a recovery of 94% with a relative standard deviation of 3.8% was found for sulfide and a recovery of 96% with a relative standard deviation of 2.8% was found for nitrite using the three potentials-MPA procedure presented above. Finally, the results obtained by this method were compared with those obtained by means of the standardized methods for nitrite and sulfide determination [
22]. Based on the results obtained, it can be concluded that the two methods lead to very close results and that the accuracy of the proposed MPA method is good. The interference effect of various anion and cation species that are common water matrix components was investigated in the presence of 0.1 mM Na
2S and 0.1 mM NaNO
2. These species were added at the concentrations 100 times higher. No interference effect was noticed in the presence of Mg
2+, Ca
2+, Mn
2+, Fe
3+, SO
42−, NO
3−, CO
32−, I
−, Cl
−, F
−.