Abstract
The reaction of 3-cyclohexyl-2-(cyclohexylimino)-6-(4-ethoxyphenyl)-5-(4-methyl-3-oxo-3,4-dihydroquinoxalin-2-yl)-2,3-dihydro-4H-1,3-oxazin-4-one with ammonium acetate afforded 3-(1-cyclohexyl-2-(cyclohexylamino)-4-(4-ethoxyphenyl)-6-oxo-1,6-dihydropyrimidin-5-yl)-1-methylquinoxalin-2(1H)-one in a good yield. The compound was fully characterized.
1. Introduction
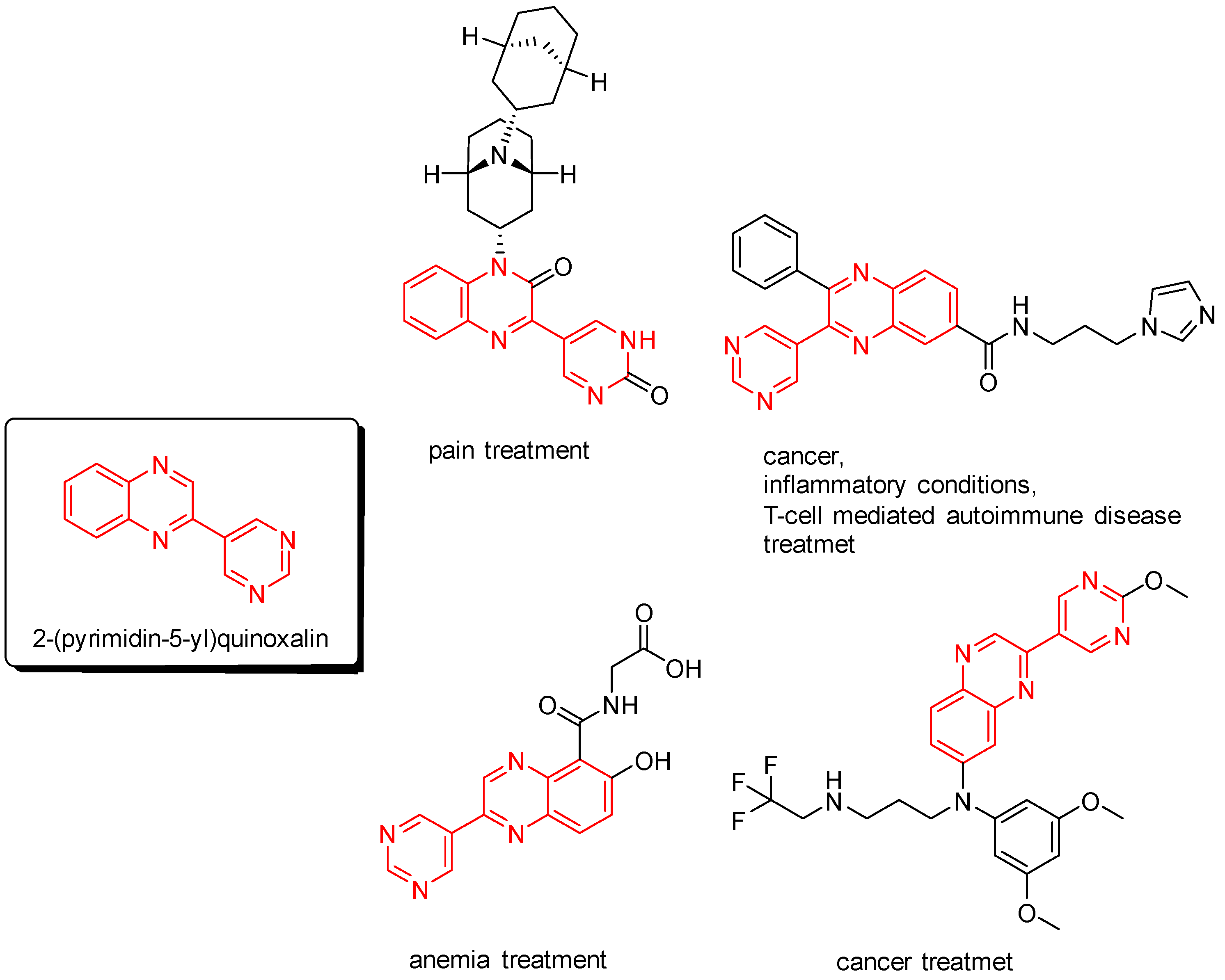
2-(Pyrimidin-5-yl)quinoxalines are interesting nitrogen heterocycles for medical applications. For example, potential pharmaceutical substances, bearing a 2-(pyrimidin-5-yl)quinoxaline core, were developed for treating pain [1], inflammatory conditions [2], T-cell-mediated autoimmune disease [2], anemia [3], and cancer [2,4] (Figure 1).
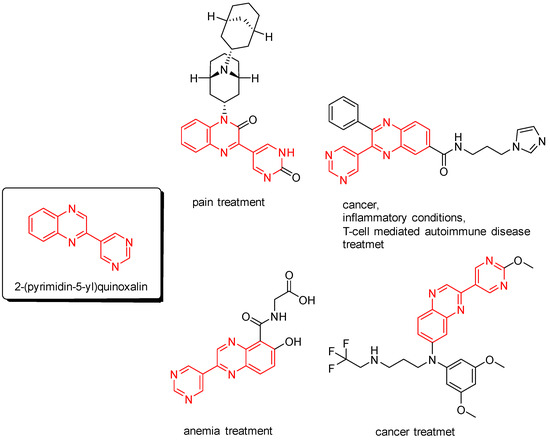
Figure 1.
Potential pharmaceutical substances, bearing a 2-(pyrimidin-5-yl)quinoxaline core.
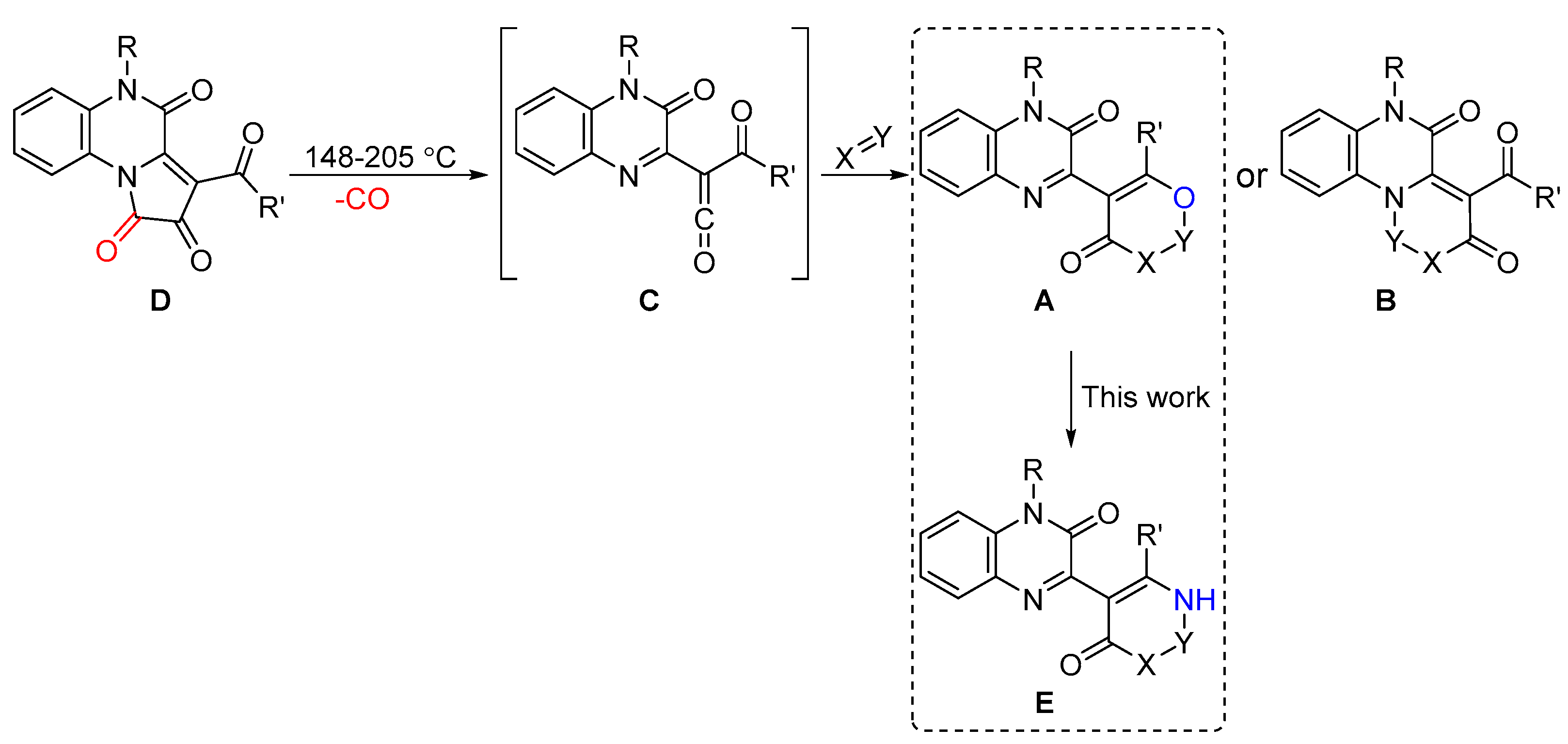
As a continuation of our study on the syntheses of quinoxaline-based heterocycles A and B, via reactions of acyl(quinoxalin-2-yl)ketenes C, generated by the thermolysis of [e]-fused 1H-pyrrole-2,3-diones D [5], we investigated the capacity of the conversion of heterocycles A to their aza-analogs E, e.g., pyrimidinylquinoxalines (X = NR″; Y = CR‴2) (Scheme 1).
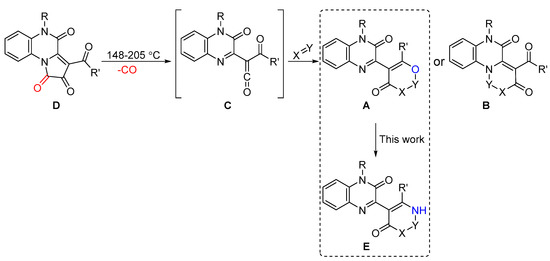
Scheme 1.
Syntheses of quinoxaline derivatives A and B via reactions of acyl(quinoxalin-2-yl)ketenes C.
2. Results and Discussion
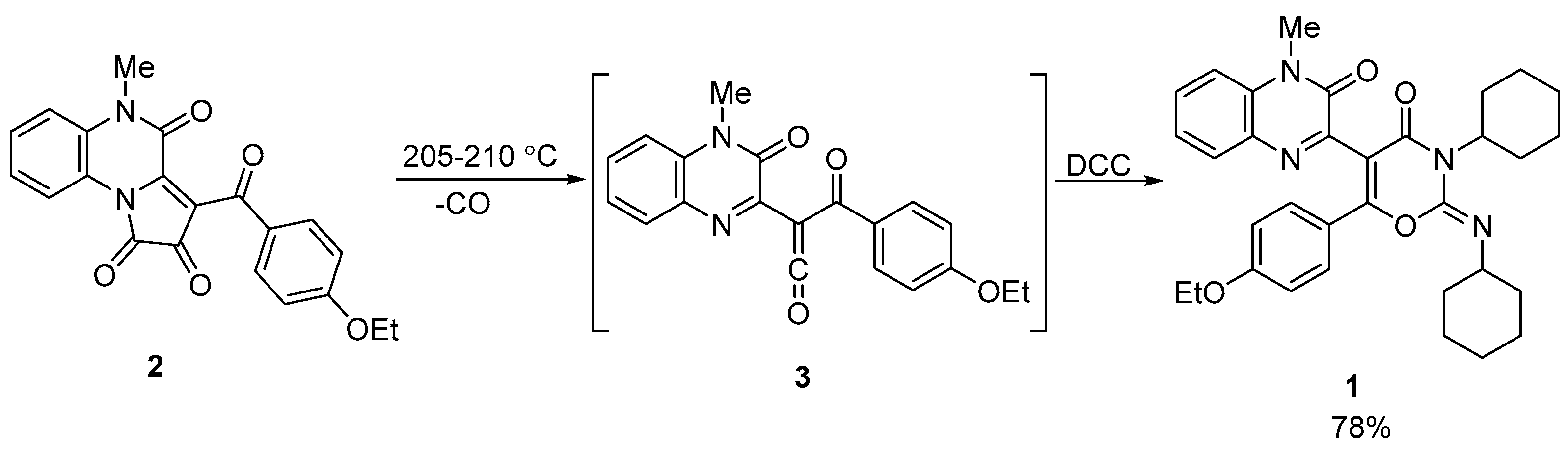
The starting quinoxaline derivative 1 was synthesized according to the previously reported procedure [6] using the solvent-free reaction of [e]-fused 1H-pyrrole-2,3-dione 2 with dicyclohexylcarbodiimide (DCC) (Scheme 2). This transformation was completed in two steps through a one-pot procedure. First, pyrrole-2,3-dione 2 underwent thermal decomposition to form a highly reactive acyl(quinoxalin-2-yl)ketene 3 in situ. Second, ketene 3 was involved in a [4+2] cycloaddition reaction with DCC to afford the desired compound 1.
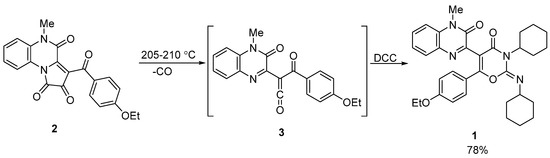
Scheme 2.
Synthesis of the starting quinoxaline derivative 1 [6].
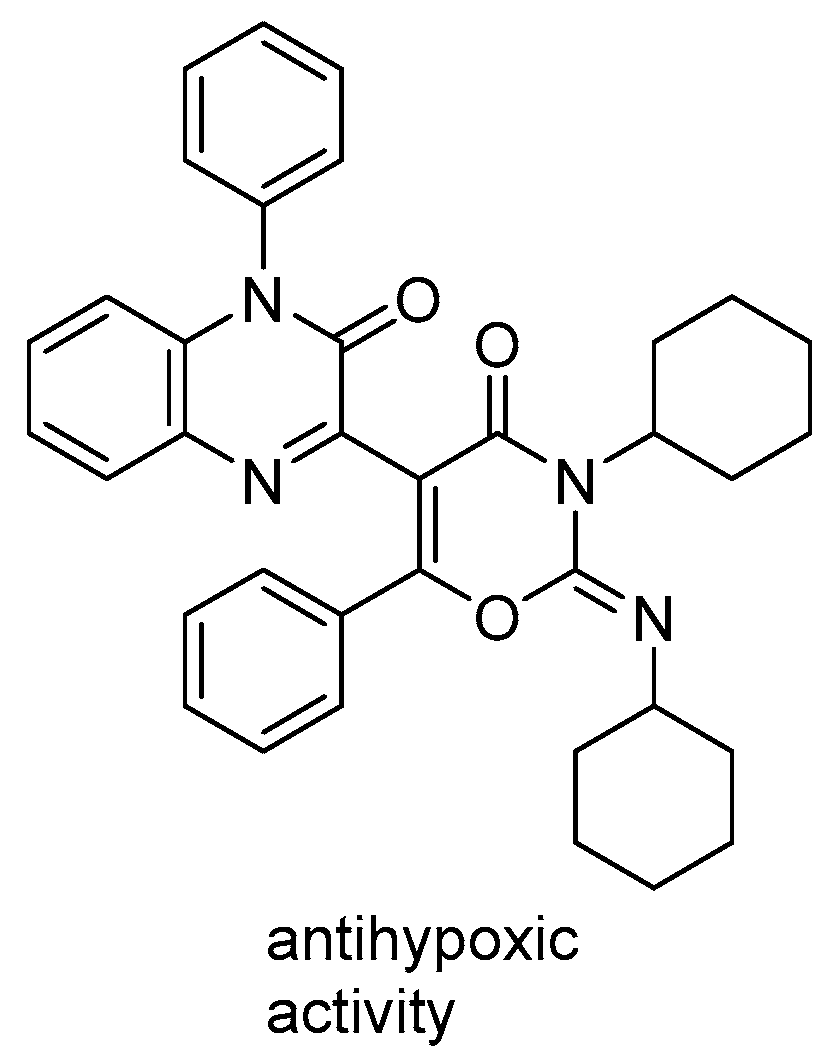
It should be mentioned that compound 1 was chosen for this study, since its closest analogs had shown significant antihypoxic effects in vivo [7] (Figure 2).
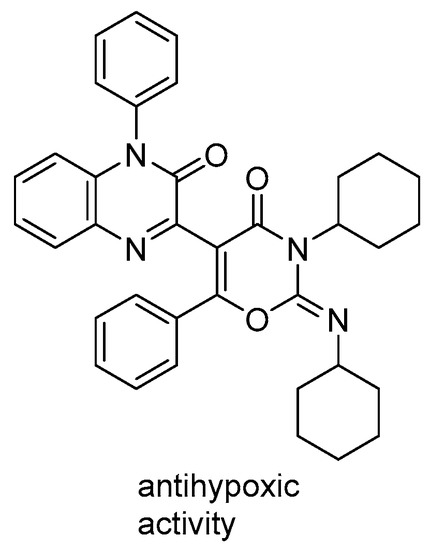
Figure 2.
Close analog of quinoxaline derivative 1 [7].
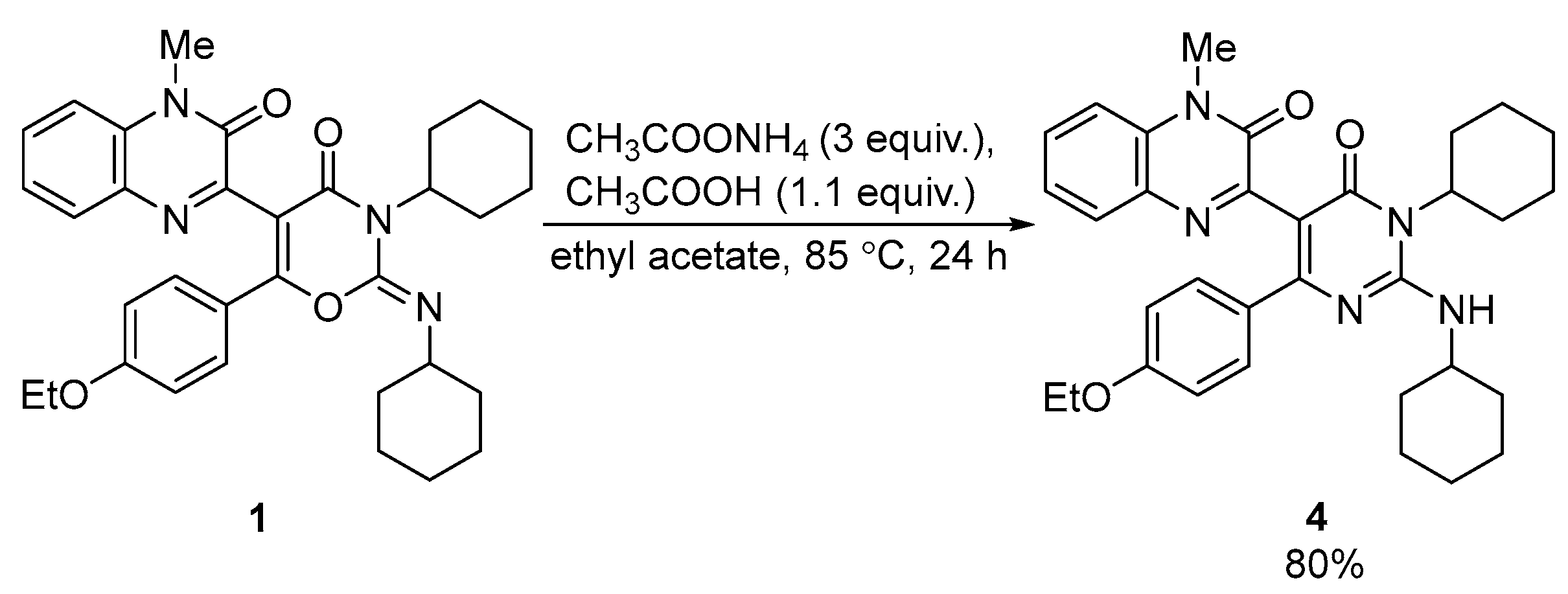
The reaction of compound 1 with ammonium acetate afforded pyrimidinylquinoxaline derivative 4 in a good yield (80%) (Scheme 3). No other products were observed through the UPLC-UV-MS of the reaction mixture. The structure of compound 4 was unambiguously confirmed through single crystal X-ray analysis (CCDC Refcode = ZUNYIT, CCDC 2010489 [8]).
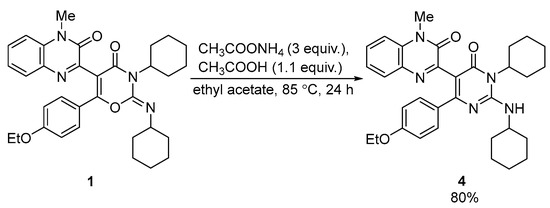
Scheme 3.
Synthesis of pyrimidinylquinoxaline 4 from quinoxalinyl-1,3-oxazine 1.
3. Materials and Methods
3.1. General Information
1H and 13C NMR spectra (Supplementary Materials) were acquired using a Bruker Avance III 400 HD spectrometer (at 400 and 100 MHz, respectively) in CDCl3 (stab. with Ag) using the HMDS signal (in 1H NMR) or solvent residual signal (in 13C NMR, 77.00 for CDCl3) as internal standards. The IR spectrum was recorded using a Perkin–Elmer Spectrum Two spectrometer from a mull in mineral oil. The melting point was measured on a Khimlabpribor PTP apparatus. Elemental analysis was carried out on a Vario MICRO Cube analyzer. The reaction conditions were optimized using UPLC-UV-MS (Waters ACQUITY UPLC I-Class system; Acquity UPLC BEH C18 column; grain size of 1.7 μm; acetonitrile–water as eluents; flow rate of 0.6 mL/min; ACQUITY UPLC PDA eλ Detector (wavelength range of 230–780 nm); Xevo TQD mass detector; electrospray ionization (ESI); positive and negative ion detection; ion source temperature of 150 °C; capillary voltage of 3500–4000 V; cone voltage of 20–70 V; vaporizer temperature of 200 °C). The single crystal X-ray analysis of compound 4 was performed on an Xcalibur Ruby diffractometer (Agilent Technologies). The empirical absorption correction was introduced by the multi-scan method using the SCALE3 ABSPACK algorithm [9]. Using OLEX2 [10], the structure was solved using the SUPERFLIP [11] program and refined by the full-matrix least-squares minimization in the anisotropic approximation for all non-hydrogen atoms with the SHELXL [12] program. Hydrogen atoms bound to carbon were positioned geometrically and refined using a riding model. The hydrogen atom of the NH group was refined freely with isotropic displacement parameters. Starting compound 1 was obtained according to the reported procedure [6] from DCC (purchased from commercial vendors) and 1H-pyrrole-2,3-dione 2 (obtained according to the reported procedure [6] from commercially available reagents). All other solvents and reagents were purchased from commercial vendors and used as received.
3.2. 3-(1-Cyclohexyl-2-(cyclohexylamino)-4-(4-ethoxyphenyl)-6-oxo-1,6-dihydropyrimidin-5-yl)-1-methylquinoxalin-2(1H)-one 4
A mixture of 50 mg (90 µmol) of compound 1, 21 mg (270 µmol) of ammonium acetate, and 6 mg (100 µmol) of acetic acid in 1 mL of ethyl acetate were stirred in a screw-capped vial at 85 °C for 24 h. Then, the reaction mixture was cooled to room temperature. The formed precipitate was filtered off and recrystallized from ethyl acetate to yield the title compound 4 (yield: 40 mg (80%); pale yellow solid; mp 289–291 °C). 1H NMR (CDCl3, 400 MHz): δ = 7.77 (m, 1 H), 7.50 (m, 3 H), 7.28 (m, 2 H), 6.73 (m, 2 H), 5.16 (br.s, 1 H), 4.92 (br.s, 1 H), 4.18 (m, 1 H), 3.97 (q, J 6.8 Hz, 2 H), 3.70 (s, 3 H), 2.16–1.12 (m, 25 H) ppm. 13C NMR (CDCl3, 100 MHz): δ = 162.8, 159.8, 156.9, 154.8, 152.0, 133.6, 133.1, 133.1, 130.4, 130.3, 130.0, 130.0, 123.2, 123.1, 113.9, 113.9, 113.4, 63.3, 50.4, 32.9, 29.8, 29.3, 26.5, 25.7, 24.6, 14.7 ppm. IR (mineral oil): 3366, 1654 cm−1. Anal. Calcd. (%) for C33H39N5O3: C 71.58; H 7.10; N 12.65. Found: C 71.92; H 7.13; N 12.51. MS (ESI+): m/z calcd. for C33H39N5O3+H+: 554.31 [M+H+]; found: 554.30. The crystal structure of compound 4 was deposited at the Cambridge Crystallographic Data Centre with the deposition number CCDC 2010489, CCDC Refcode = ZUNYIT [8].
Supplementary Materials
The following are available online: copies of NMR spectra for new compound.
Author Contributions
Conceptualization, E.E.K.; methodology, E.E.K.; validation, E.E.K.; investigation, E.E.K., S.O.K. (synthetic chemistry) and M.V.D. (X-ray analysis); writing—original draft preparation, E.E.K.; writing—review and editing, E.E.K. and M.V.D.; visualization, E.E.K.; supervision, E.E.K. and A.N.M.; project administration, E.E.K.; funding acquisition, A.N.M. All authors have read and agreed to the published version of the manuscript.
Funding
This study was performed under financial support by the Perm Research and Educational Center “Rational subsoil use”, 2022.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The presented data are available in this article.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of all compounds are available from the authors.
References
- Ueno, T.; Tsuno, N.; Zhou, X. Cyclic Urea- or Lactam-Substituted Quinoxaline-Type Piperidine Compounds and the Uses Thereof. WO Patent WO2014020405A1, 6 February 2014. [Google Scholar]
- Gigstad, K.M.; Cardin, D.P.; Hirayama, T.; Hirose, M.; Hu, Y.; Kakei, H.; Lee, H.M.; Motoyaji, T.; Nii, N.; Shi, Z.; et al. Quinoxaline Compounds and Uses Thereof. WO Patent WO2015161142A1, 22 October 2015. [Google Scholar]
- Colon, M.; Fitch, D.M. Prolyl Hydroxylase Inhibitors. US Patent US2010305133A1, 2 December 2010. [Google Scholar]
- Woodhead, S.J.; Murray, C.W.; Berdini, V.; Saxty, G.; Besong, G.E.; Meerpoel, L.; Querolle, O.A.G.; Poncelet, V.S. Anticancer Benzopyrazines via the Inhibition of FGFR Kinases. WO Patent WO2013061081A1, 2 May 2013. [Google Scholar]
- Lystsova, E.A.; Khramtsova, E.E.; Maslivets, A.N. Acyl(imidoyl)ketenes: Reactive Bidentate Oxa/Aza-Dienes for Organic Synthesis. Symmetry 2021, 13, 1509. [Google Scholar] [CrossRef]
- Kasatkina, S.; Stepanova, E.; Dmitriev, M.; Mokrushin, I.; Maslivets, A. Divergent synthesis of (quinoxalin-2-yl)-1,3-oxazines and pyrimido[1,6-a]quinoxalines via the cycloaddition reaction of acyl(quinoxalinyl)ketenes. Tetrahedron Lett. 2019, 60, 151088. [Google Scholar] [CrossRef]
- Maslivets, A.N.; Kasatkina, S.O.; Stepanova, E.E.; Makhmudov, R.R. Method of Producing 5-(3-oxo-3,4-dihydroquinoxalin-2-yl)-2,3-dihydro-4H-1,3-oxazin-4-ones. RU Patent RU2707194C1, 25 November 2019. [Google Scholar]
- Kasatkina, S.O.; Stepanova, E.E.; Dmitriev, M.V.; Maslivets, A.N. CCDC 2010489: Experimental Crystal Structure Determination; Cambridge Crystallographic Data Centre: Cambridge, UK, 2020. [Google Scholar] [CrossRef]
- CrysAlisPro, Oxford Diffraction/Agilent Technologies UK Ltd., Yarnton, England, Version 1.171.37.33 (Release 27-03-2014 CrysAlis171.NET). Available online: https://www.rigaku.com/products/crystallography/crysalis (accessed on 7 June 2022).
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Palatinus, L.; Chapuis, G. SUPERFLIP—A computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Cryst. 2007, 40, 786–790. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).