N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Instrumentation
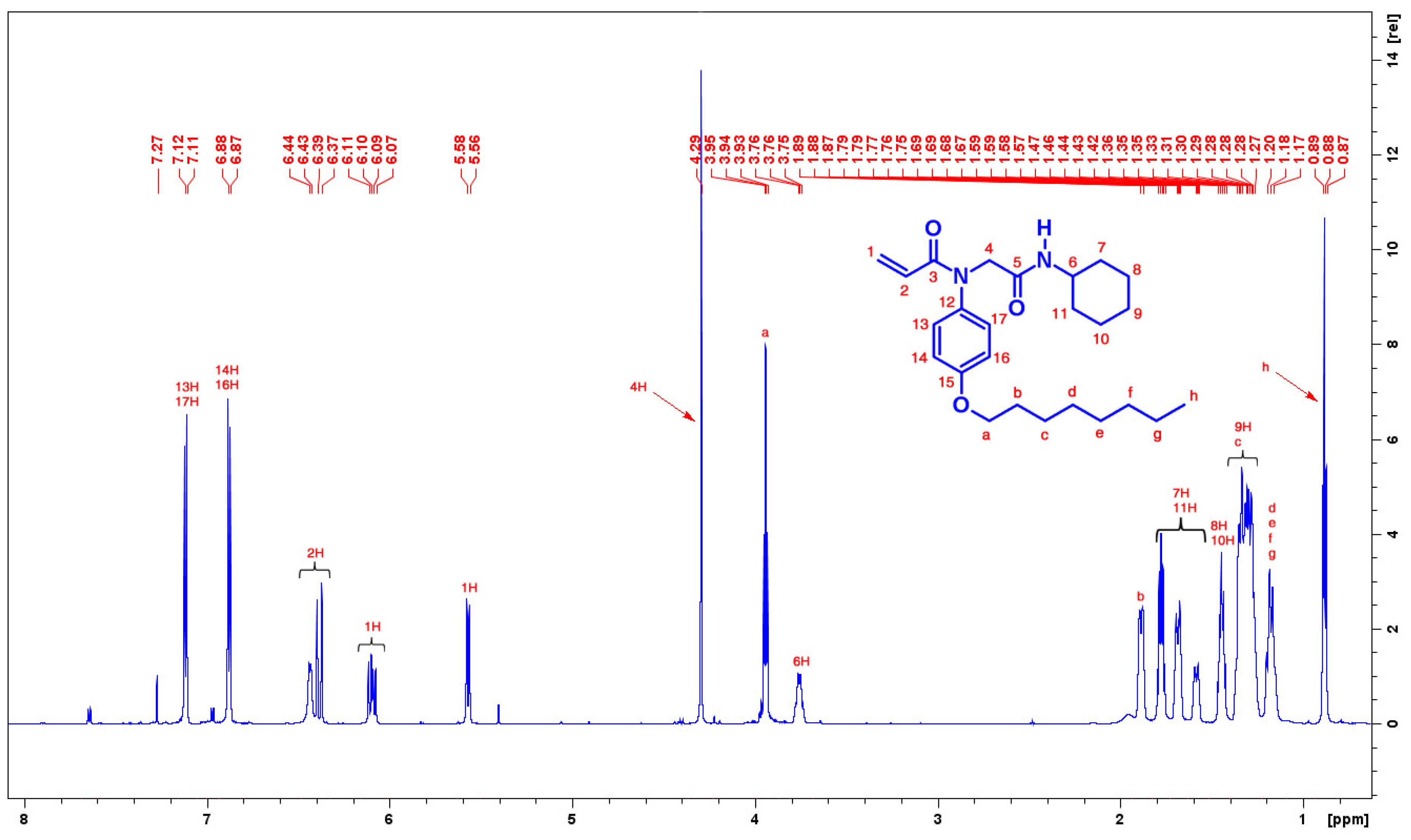
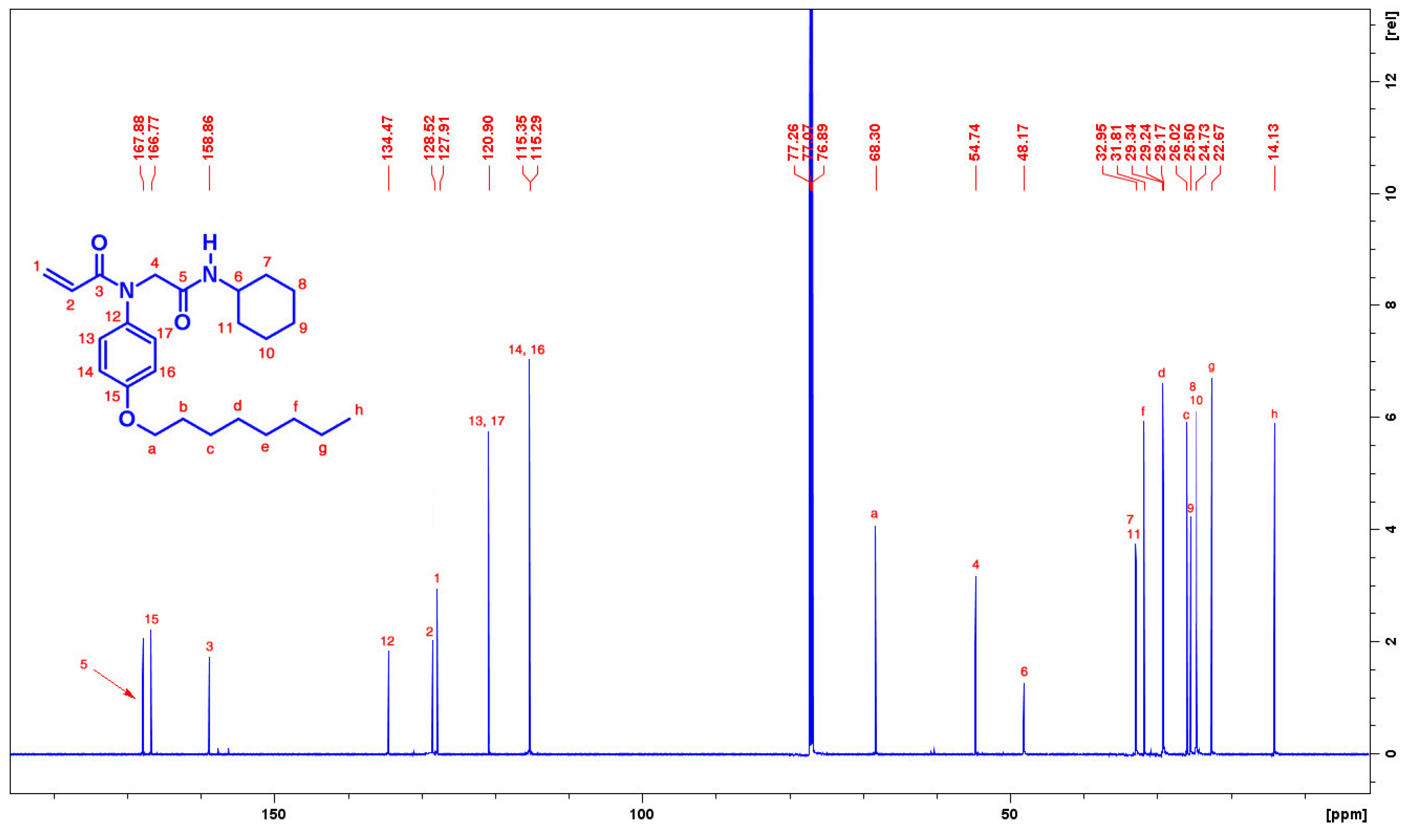
3.3. Synthesis of N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Simon, R.J.; Kania, R.S.; Zuckermann, R.N.; Huebner, V.D.; Jewell, D.A.; Banville, S.; Ng, S.; Wang, L.; Rosenberg, S.; Marlowe, C.K.; et al. Peptoids: A modular approach to drug discovery. Proc. Natl. Acad. Sci. USA 1992, 89, 9367–9371. [Google Scholar] [CrossRef]
- Zuckermann, R.N. Peptoid Origins. Biopolymers (PeptSci) 2011, 96, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Zuckermann, R.N.; Kodadek, T. Peptoids as potential therapeutics. Curr. Opin. Mol. Ther. 2009, 11, 299–307. [Google Scholar] [PubMed]
- Váradi, A.; Palmer, T.C.; Dardashti, R.N.; Majumdar, S. Isocyanide-Based Multicomponent Reactions for the Synthesis of Heterocycles. Molecules 2016, 21, 19. [Google Scholar] [CrossRef]
- Koopmanschap, G.; Ruijter, E.; Orru, R.V.A. Isocyanide-based multicomponent reactions towards cyclic constrained peptidomimetics. Beilstein J. Org. Chem. 2014, 10, 544–598. [Google Scholar] [CrossRef] [PubMed]
- El Kaim, L.; Grimaud, L.; Oble, J. Phenol Ugi-Smiles Systems: Strategies for the Multicomponent N-Arylation of Primary Amines with Isocyanides, Aldehydes, and Phenols. Angew. Chem. Int. Ed. 2005, 44, 7961–7964. [Google Scholar] [CrossRef] [PubMed]
- Tye, H.; Whittaker, M. Use of a Design of Experiments approach for the optimisation of a microwave assisted Ugi reaction. Org. Biomol. Chem. 2004, 2, 813–815. [Google Scholar] [CrossRef] [PubMed]
- Sung, K.; Chen, F.-L.; Huang, P.-C. A Modified U-4CR Reaction with 2-Nitrobenzylamine as an Ammonia Equivalent. Synlett 2006, 2667–2669. [Google Scholar] [CrossRef]
- Galetti, M.D.; Cirigliano, A.M.; Cabrera, G.M.; Ramírez, J.A. Multicomponent synthesis of acylated short peptoids with antifungal activity against plant pathogens. Mol. Divers. 2012, 16, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, S.; Liu, J.; Xie, Z.; Luan, S.; Xiao, C.; Tao, Y.; Wang, X. Ugi Reaction of Natural Amino Acids: A General Route toward Facile Synthesis of Polypeptoids for Bioapplications. ACS Macro Lett. 2016, 5, 1049–1054. [Google Scholar] [CrossRef]
- Ghasemi, E.; Shahvelayati, A.S.; Yavari, I. Ugi reaction of thiouridocarboxylic acids: A synthesis of thiourea-peptoids. Phosphorus Sulfur Silicon Relat. Elem. 2016, 191, 746–750. [Google Scholar] [CrossRef]
- Silva, E.H.B.; Emery, F.S.; Ponte, G.D.; Donate, P.M. Synthesis of Some Functionalized Peptomers via Ugi Four-Component Reaction. Synth. Commun. 2015, 45, 1761–1767. [Google Scholar] [CrossRef]
- Savithri, A.; Thulasi, S.; Varma, R.L. Narrow-rim functionalization of calix[4]arene through Ugi-4CR: Synthesis of a series of calix[4]arene peptoids. J. Org. Chem. 2014, 79, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Mangunuru, H.P.R.; Yang, H.; Wang, G. Synthesis of peptoid based small molecular gelators by a multiple component reaction. Chem. Commun. 2013, 49, 4489–4491. [Google Scholar] [CrossRef] [PubMed]
- Brauer, M.C.N.; Neves, R.A.W.; Westermann, B.; Heinke, R.; Wessjohann, L.A. Synthesis of antibacterial 1,3-diyne-linked peptoids from an Ugi-4CR/Glaser coupling approach. Beilstein J. Org. Chem. 2015, 11, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.; Joo, M.K.; Sohn, Y.S.; Jeong, B. Reverse thermal organogel. Adv. Mater. 2007, 19, 3947–3950. [Google Scholar] [CrossRef]
- Wang, G.; Cheuk, S.; Yang, H.; Goyal, N.; Reddy, P.V.N.; Hopkinson, B. Synthesis and Characterization of Monosaccharide-Derived Carbamates as Low-Molecular-Weight Gelators. Langmuir 2009, 25, 8696–8705. [Google Scholar] [CrossRef] [PubMed]
- Biswas, G.; Moon, H.J.; Boratyński, P.; Jeong, B.; Kwon, Y.-U. Structural sensitivity of peptoid-based low molecular mass organogelator. Mater. Des. 2016, 108, 659–665. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganesh, S.D.; Saha, N.; Kucharczyk, P.; Sáha, P. N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide. Molbank 2016, 2016, M921. https://doi.org/10.3390/M921
Ganesh SD, Saha N, Kucharczyk P, Sáha P. N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide. Molbank. 2016; 2016(4):M921. https://doi.org/10.3390/M921
Chicago/Turabian StyleGanesh, Shimoga D., Nabanita Saha, Pavel Kucharczyk, and Petr Sáha. 2016. "N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide" Molbank 2016, no. 4: M921. https://doi.org/10.3390/M921
APA StyleGanesh, S. D., Saha, N., Kucharczyk, P., & Sáha, P. (2016). N-[2-(Cyclohexylamino)-2-oxoethyl]-N-(4-octyloxy)phenyl-prop-2-enamide. Molbank, 2016(4), M921. https://doi.org/10.3390/M921




