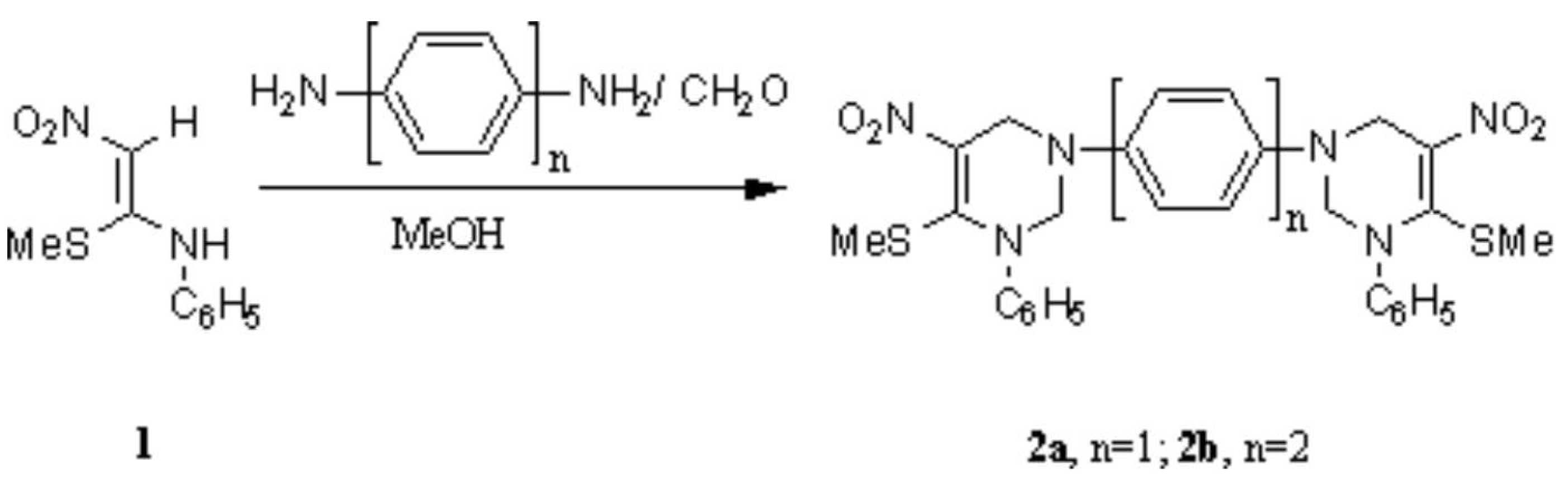
In continuation with our on going program on the synthesis of tetrahydropyrimidines [1,2], we have recently reported the synthesis of Bis-tetrahydropyrimidines [3] in which the rings are linked through flexible aliphatic chains. We now report the synthesis of the title compounds in which the rings are bonded through rigid aromatic systems. A mixture of p-phenylenediamine (54 mg, 0.5 mmol) and formaldehyde (60 mg, 2 mmol, 40% solution) was stirred in methanol (3 mL) for ten minutes and to this a solution of 1-nitro-2-anilino-2-methylthioethene 1 [4] (210 mg, 1 mmol) in 6 mL methanol was added and the mixture was stirred at room temperature for 5 hours, when a yellow solid precipitated out. After the completion of the reaction (monitored by tlc) the reaction mixture was cooled in ice water and the solid filtered, washed with methanol (2X2 mL) to give pure 2a (220 mg, 76%), which was recrystallized from methanol. The reaction of 1 with benzidine was carried out in refluxing methanol to give 2b in 63 % yield, which was recrystallized from benzene.
1,4-Bis-(1-phenyl-5-nitro-6-methylthio-1,2,3,4-tetrahydropyrimidinyl)benzene (2a)
Melting point: 168-169 °C (methanol, uncorrected).
IR (KBr, cm-1): 1456; 1503; 1544; 1611.
1H-NMR (300 MHz, CDCl3): δ= 1.94 (s, 6H, SCH3); 4.49(s, 4H, N-CH2C=); 4.87 (s, 4H, N-CH2-N); 6.70-6.73 (m, 4H); 7.06-7.33 (m, 10H).
13C-NMR(75 MHz, CDCl3): δ= 16.5; 48.8; 72.1; 116.5; 124.8; 126.4; 127.3; 129.8; 133.2; 145.5; 146.0; 160.2.
MS (m/z): 577 (M+).
4,4′-Bis-(1-phenyl-5-nitro-6-methylthio-1,2,3,4-tetrahydropyrimidinyl)diphenyl (2b)
Melting point: 167-169 °C (benzene, uncorrected).
IR (KBr, cm−1): 1430; 1514; 1540; 1591.
1H-NMR (300 MHz, CDCl3): δ= 1.94 (s, 6H, SCH3); 4.49 (s, 4H, N-CH2C=); 4.87 (s, 4H, N-CH2-N); 6.70-6.73 (m, 4H); 7.14-7.36 (m, 14H).
13C-NMR (75 MHz, CDCl3): δ= 16.5; 48.8; 72.1; 116.5; 124.8; 126.4; 127.3; 128.3; 129.8; 133.2; 145.5; 146.0; 160.2.
MS (m/z): 653 (M+).
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Supplementary File 5Supplementary File 6Acknowledgements
The authors wish to thank Fr. I. Warpakma for facilities and Fr. Stephan Mavely & Fr. J. Nellanatt for encouragement. Financial assistance from ICAR-NATP is gratefully acknowledged. The authors also wish to express their gratitude to Dr. Anubrata Das of ICAR for his keen interest in this investigation.
References
- Vishwakarma, J.N.; Mofizuddin, M.; Ila, H.; Junjappa, H. J. Heterocyclic Chem. 1988, 25, 1387.
- Karim, E.; Kishore, K.; Vishwakarma, J.N. J. Heterocyclic Chem. 2003, 40, 901-03.
- Chanda, K.; Dutta, M.C.; Kishore, K.; Vishwakarma, J.N. Molbank 2004, M367.
- Gompper, R.; Schaefer, H. Chem. Ber. 1967, 100, 591–604.
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
