Abstract
Vasculitides are a heterogeneous group of disorders characterized by inflammation of blood vessel walls, leading to tissue ischemia and organ injury. Traditional inflammatory markers such as the erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are widely used but lack diagnostic specificity. This has driven the search for more informative biomarkers across vasculitis subtypes. This review summarizes current evidence for validated and emerging biomarkers in large-, medium-, small-, and variable-vessel vasculitis, as well as single-organ vasculitis. Key analytes reflect systemic inflammation, such as serum amyloid A (SAA) and interleukin-6 (IL-6), as well as endothelial activation, complement pathways, neutrophil and macrophage activation, and organ-specific damage. Promising candidates include pentraxin-3 (PTX3) and matrix metalloproteinase-9 (MMP-9) in large-vessel vasculitis; N-terminal pro-B-type natriuretic peptide (NT-proBNP) and S100 proteins in Kawasaki disease; galactose-deficient immunoglobulin A1 (Gd-IgA1) and urinary angiotensinogen (AGT) in IgA vasculitis; and tissue inhibitor of metalloproteinases-1 (TIMP-1), S100 proteins, complement C3, and PTX3 in antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis. Although these biomarkers provide mechanistic insight, most lack disease-specificity, external validation, or standardized assays. Future progress will require multicenter studies, harmonized testing, and integrated biomarker panels combined with imaging modalities to improve diagnosis, activity assessment, and monitoring.
1. Introduction
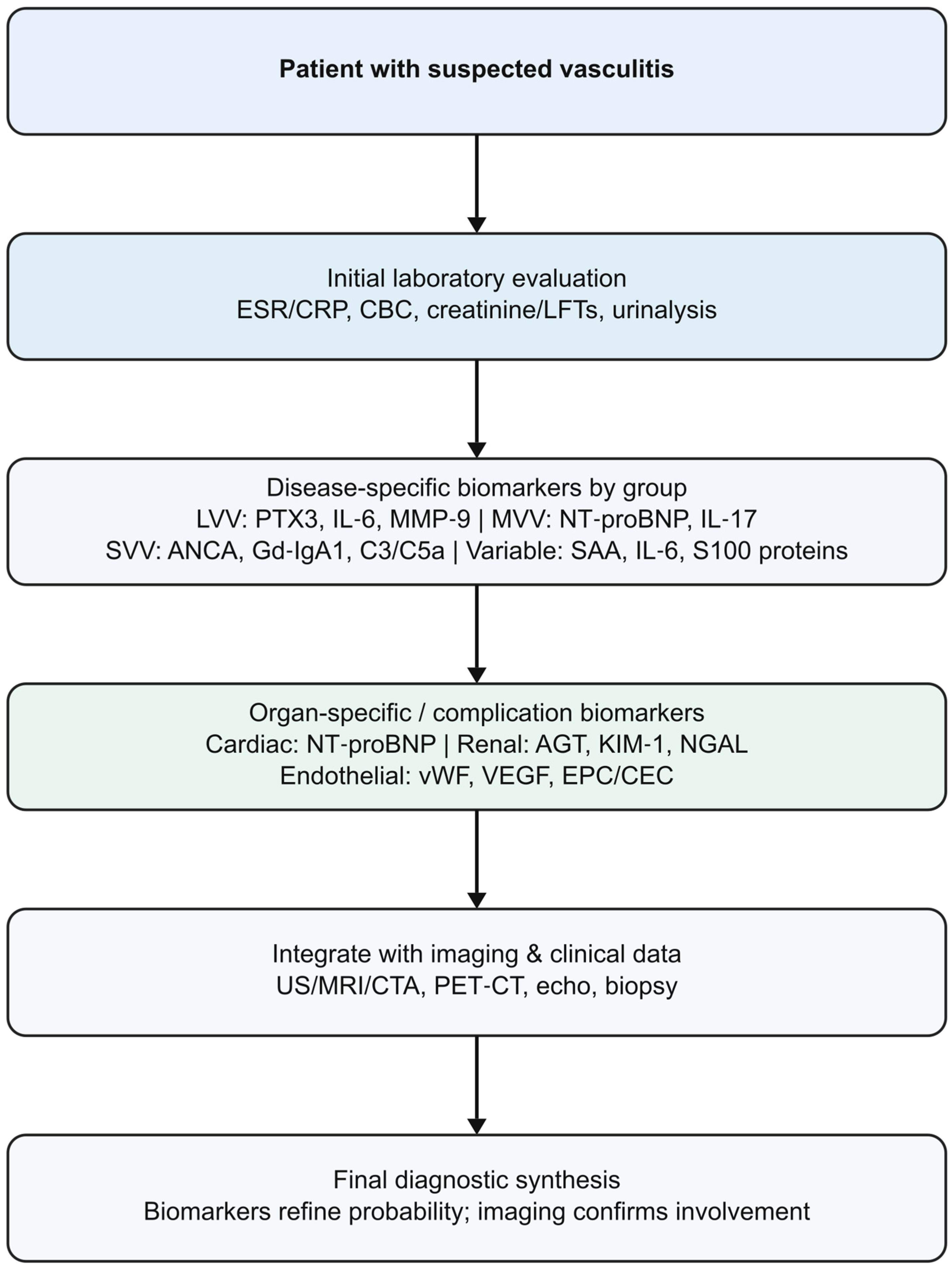
Vasculitides represent a heterogeneous group of disorders characterized by inflammation of blood vessel walls, leading to tissue ischemia and organ damage. They are classified according to the size of predominantly affected vessels as small-, medium-, or large-vessel vasculitis, with additional categories for variable-vessel vasculitis, single-organ vasculitis, vasculitis associated with systemic diseases, and vasculitis associated with probable etiology [1,2]. Although the etiology and pathogenesis differ among entities, early diagnosis remains a challenge because clinical presentations are often nonspecific and conventional laboratory markers such as ESR and CRP lack disease specificity (Figure 1). The increasing availability of advanced serologic, genetic, imaging and omics-based technologies has accelerated biomarker research in vasculitis, yet the translation of candidate biomarkers into routine practice remains limited [3,4,5,6,7,8].
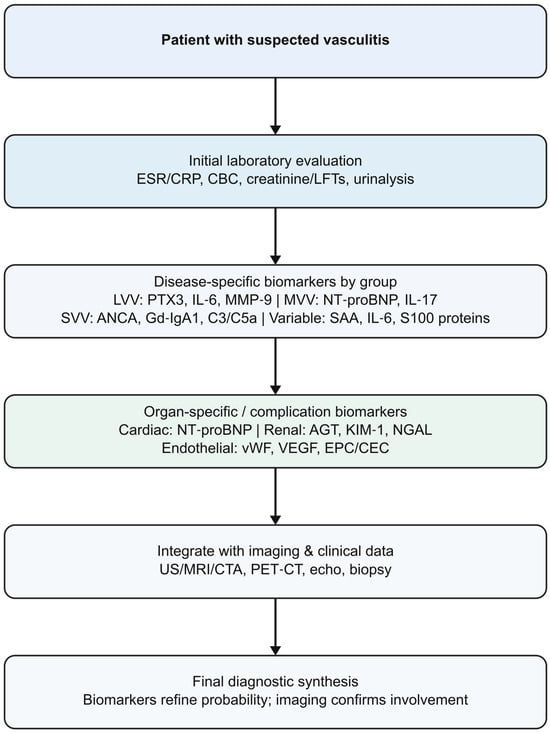
Figure 1.
Diagnostic role of biomarkers in vasculitis. List of abbreviations: AGT: angiotensinogen; ANCA: antineutrophil cytoplasmic antibodies; C3: complement component 3; C5a: complement component C5a; CBC: complete blood count; CEC: circulating endothelial cells; CRP: C-reactive protein; CTA: computed tomography angiography; EPC: endothelial progenitor cells; ESR: erythrocyte sedimentation rate; Gd-IgA1: galactose-deficient immunoglobulin A1; IL-6: interleukin-6; IL-17: interleukin-17; KIM-1: kidney injury molecule-1; LFTs: liver function tests; LVV: large-vessel vasculitis; MMP-9: Matrix metalloproteinase-9; MRI: magnetic resonance imaging; NGAL: neutrophil gelatinase-associated lipocalin; NT-proBNP: N-terminal pro–B-type natriuretic peptide; PTX3: pentraxin-3; SVV: small-vessel vasculitis; US: ultrasound; VEGF: vascular endothelial growth factor; vWF: von Willebrand factor.
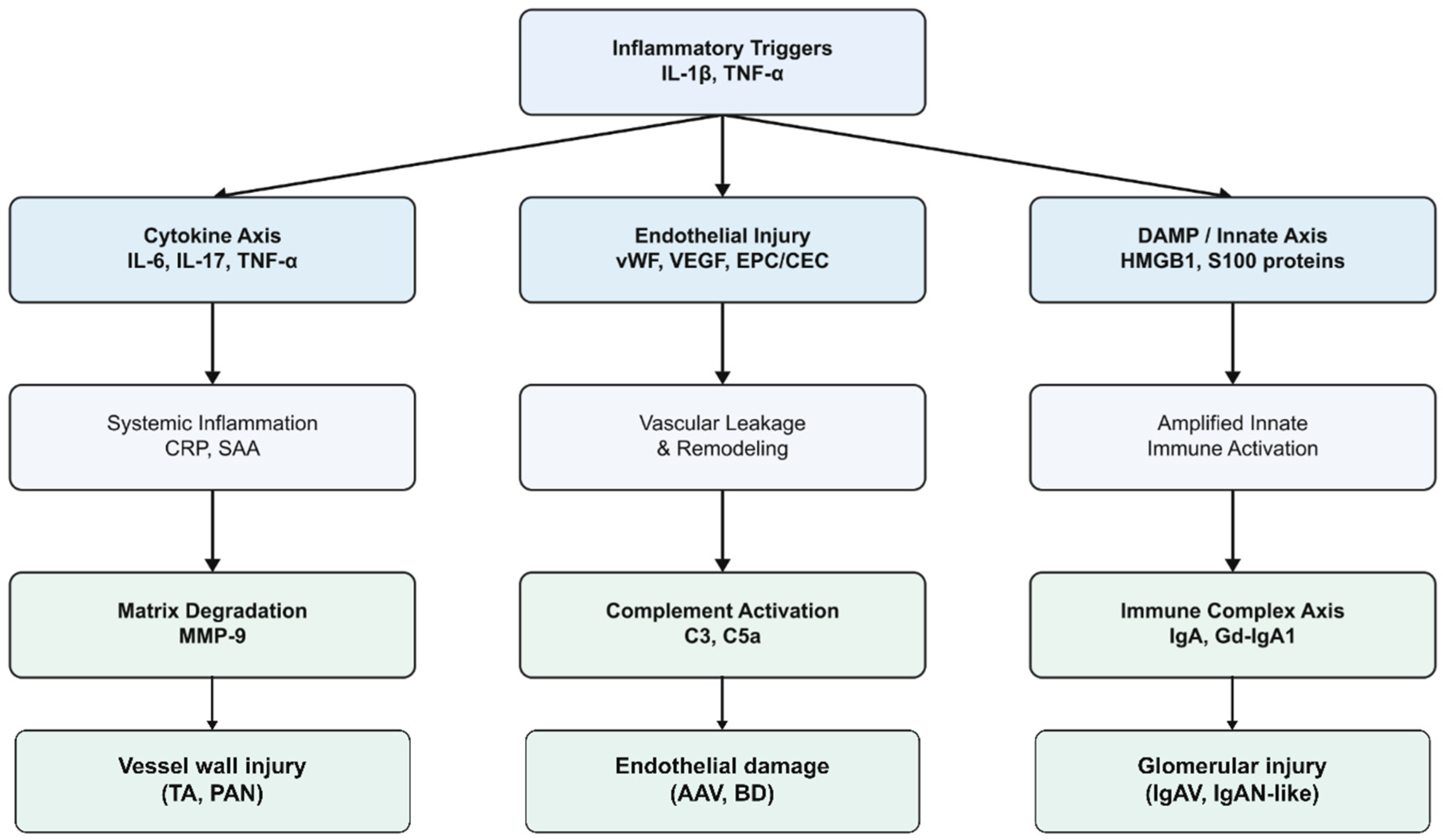
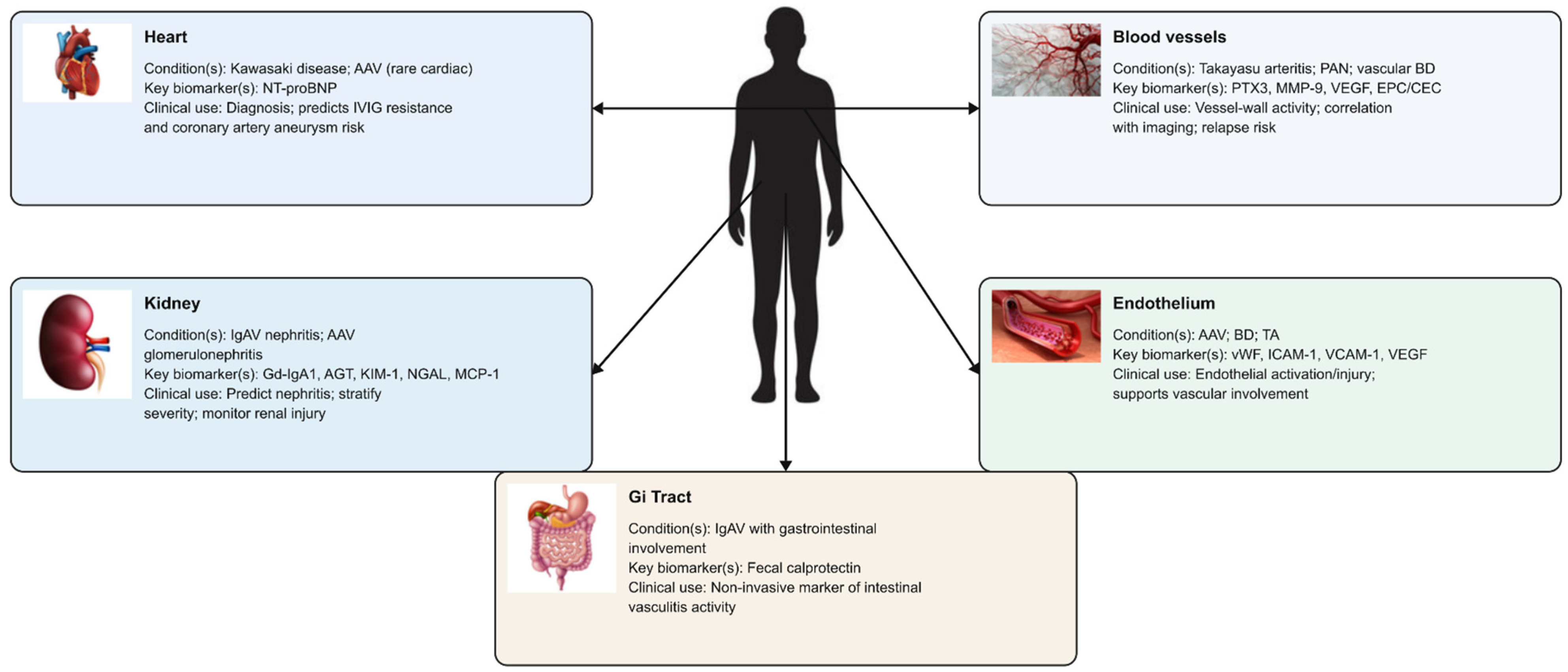
An ideal biomarker in vasculitis would improve diagnostic accuracy, differentiate disease subsets, predict disease activity or relapse, monitor treatment response, and forecast long-term outcomes. Numerous molecules reflecting inflammation, endothelial injury, immune activation, complement activation, tissue damage and genetic or epigenetic alterations have been proposed (Figure 2). However, only a few biomarkers have consistently demonstrated useful performance across cohorts, and even these require careful interpretation in the clinical context (Figure 3).
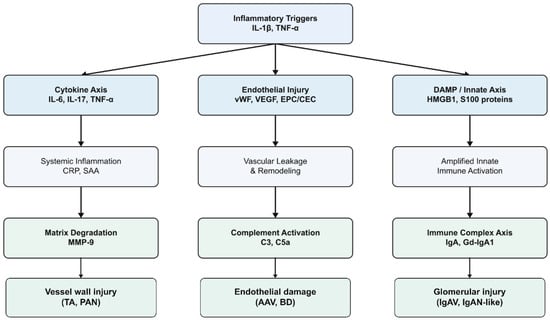
Figure 2.
Pathogenic pathways and biomarker origins. List of abbreviations: AAV: ANCA-associated vasculitis; BD: Behçet’s disease; C3: complement component 3; C5a: complement component C5a; CEC: circulating endothelial cells; CRP: C-reactive protein; DAMP: damage-associated molecular pattern; EPC: endothelial progenitor cells; Gd-IgA1: galactose-deficient immunoglobulin A1; HMGB1: high-mobility group box-1 protein; IgA: immunoglobulin A; IgAN: IgA nephropathy; IgAV: IgA vasculitis; IL-1β: inteleukin-1β; IL-6: interleukin-6; IL-17: interleukin-17; MMP-9: Matrix metalloproteinase-9; PAN: polyarteritis nodosa; S100 proteins: S100 calcium-binding proteins; SAA: serum amyloid A; TA: Takayasu arteritis; TNF-α: tumor necrosis factor-alpha; VEGF: vascular endothelial growth factor; vWF: von Willebrand factor.
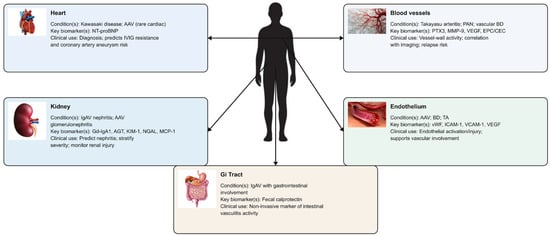
Figure 3.
Organ-specific damage biomarkers in vasculitis. List of abbreviations: AAV: antineutrophil cytoplasmic antibody-associated vasculitis; AGT: angiotensinogen; BD: Behçet’s disease; CEC: circulating endothelial cells; EPC: endothelial progenitor cells; Gd-IgA1: galactose-deficient immunoglobulin A1; Gi: gastrointestinal; IgAV: IgA vasculitis; ICAM-1: intercellular adhesion molecule-1; IVIG: intravenous immunoglobulin; KIM-1: kidney injury molecule-1; MCP-1: monocyte chemoattractant protein-1; MMP-9: Matrix metalloproteinase-9; NGAL: neutrophil gelatinase-associated lipocalin; NT-proBNP: N-terminal pro–B-type natriuretic peptide; PAN: polyarteritis nodosa; PTX3: pentraxin-3; TA: Takayasu arteritis; VCAM-1: vascular cell adhesion molecule-1; VEGF: vascular endothelial growth factor; vWF: von Willebrand factor.
Recent years have seen a growing number of narrative and time-restricted reviews summarizing advances in systemic vasculitis, including annual overviews and thematic syntheses addressing disease classification, pathogenesis, and therapeutic developments [2,3,4,6,7,8]. In parallel with these efforts, increasing attention has been directed toward the identification of biomarkers that capture vascular inflammation, immune activation, endothelial injury, and organ-specific damage across vasculitis subtypes. The present review focuses on this biomarker-oriented perspective, integrating data from large-, medium-, small-, variable-vessel, and single-organ vasculitides, and highlighting both validated and emerging candidates in relation to their biological pathways and potential clinical utility, emphasizing their limitations and highlighting areas requiring further investigation (Table 1 and Table 2).

Table 1.
Biomarker landscape across vasculitis types.

Table 2.
Biomarkers by disease and clinical use.
This narrative review is based on a focused and iterative appraisal of the published literature on biomarkers in primary systemic vasculitides. Relevant publications were identified through targeted searches of major biomedical databases, including PubMed, Web of Science, and Scopus, complemented by manual screening of reference lists from key reviews and original articles. Emphasis was placed on studies providing mechanistic insight, clinical relevance, or translational potential of biomarkers across different vasculitis subtypes. Rather than applying rigid inclusion or exclusion criteria, the literature was selected to reflect seminal work, recent advances, and clinically informative biomarker candidates, in line with the objectives of a narrative review.
2. Biomarkers in Large-Vessel Vasculitis
Large-vessel vasculitis (LVV) includes giant cell arteritis (GCA) and Takayasu arteritis (TA), two granulomatous vasculitides affecting the aorta and its major branches [1]. While sharing histopathologic similarities, they differ in age distribution, epidemiology, and vascular territories. GCA is the most common primary systemic vasculitis in adults, occurring almost exclusively after age 50, with the highest prevalence in Northern European descent [2,9]. TA predominantly affects young women under 40 years, with the highest incidence in Asian populations [2,10]. Both diseases may present with systemic inflammation and vascular complications, including stenosis, occlusion, aneurysm formation, and ischemic events involving the eyes, brain, and other organs.
2.1. Current Clinical Biomarkers
In GCA, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) remain the most widely used biomarkers of systemic inflammation. Elevated ESR (>50 mm/h) is included in the 1990 American College of Rheumatology classification criteria [11]. In a cohort of biopsy-proven GCA, CRP showed slightly higher sensitivity than ESR (86.4% vs. 84.2%), while the specificity of either marker alone remained low (approximately 30%) and increased only modestly when used together (41%) [12]. Approximately 10% of biopsy-proven GCA cases present with normal ESR and CRP, illustrating their limited sensitivity [12].
Serum amyloid A (SAA), another hepatic acute phase protein induced by interleukin (IL)-1, IL-6, and tumor necrosis factor-alpha (TNF-α), is markedly elevated in untreated GCA, up to 80-fold higher than in controls, and correlates strongly with IL-6 levels [13]. Higher baseline ESR, CRP, SAA, and other acute-phase reactants have been linked to increased relapse risk [14].
In TA, ESR and CRP are similarly used to assess systemic inflammation. Between 10 and 30% of patients with clinically active TA have normal levels of these markers [15]. In TA patients with coronary artery involvement, baseline high-sensitivity CRP independently predicted major cardiovascular events [16]. Elevated ESR and CRP have also been associated with prolonged time to remission and higher relapse risk [17]. SAA in TA has shown higher concentrations in active disease compared with inactive disease and healthy controls, decreasing with treatment, although overlap limits accuracy [18].
Complete blood count-derived indices such as CRP/albumin ratio, neutrophil–lymphocyte ratio (NLR), platelet–lymphocyte ratio (PLR), monocyte–lymphocyte ratio (MLR), and red cell distribution width (RDW) are increased in TA compared with controls and correlate with CRP and ESR [19]. These indices reflect systemic inflammation but lack specificity.
2.2. Emerging Biomarkers
Pentraxin-3 (PTX3) is produced locally by endothelial cells, macrophages, and dendritic cells and stored in neutrophil granules. In TA, PTX3 levels are elevated compared with controls and outperform CRP and ESR in distinguishing active from inactive disease in some cohorts [20,21]. PTX3 also correlates with vascular enhancement and angiographic progression, reflecting local vessel-wall inflammation rather than hepatic acute-phase responses [22].
Matrix metalloproteinase-9 (MMP-9), a mediator of extracellular matrix degradation, is significantly increased in active TA and decreases with treatment, correlating with disease activity and inflammatory markers [23]. Together, PTX3 and MMP-9 provide insight into vessel-wall injury and may be less susceptible to glucocorticoid suppression.
Elevated IL-6, TNF-α, IL-18, and other cytokines are consistently reported in active TA, though correlations with clinical or imaging activity vary [17,24,25,26,27]. The T helper 17 cells (Th17)/IL-23 axis is particularly relevant; active TA demonstrates increased IL-17 and IL-23, and Th17 cell expansion tracks with disease activity [28].
Among chemokines, monocyte chemoattractant protein-1 (MCP-1) is regularly elevated in TA and correlates with disease activity scores and inflammatory markers such as ESR, CRP, and IL-6 [29].
Endothelial injury and repair biomarkers such as vascular endothelial growth factor (VEGF), endothelial progenitor cells (EPCs), and circulating endothelial cells (CECs) are increased in TA and correlate with disease activity in several studies [30,31].
Adipokines such as leptin have been associated with active TA in some cohorts, whereas adiponectin findings are inconsistent [32].
2.3. Clinical Utility and Limitations
ESR, CRP, and SAA remain the primary laboratory tools for assessing disease activity in LVV; however, their clinical utility is limited by poor specificity and suboptimal sensitivity, with normal values occurring in up to 10% of GCA and 10–30% of TA patients despite active disease [12,15], and by their rapid suppression following glucocorticoid therapy. Among emerging biomarkers, PTX3 shows the greatest promise because of its vascular-wall origin and its correlations with ischemic complications, imaging progression, and disease activity, while being less susceptible to glucocorticoid suppression [20,21,22]. MMP-9 provides complementary information about extracellular matrix remodeling and arterial injury [23], though its use remains constrained by assay variability and lack of standardization.
Cytokines such as IL-6 and those within the Th17/IL-17/IL-23 axis correlate with activity in both GCA and TA [24,25,26,27,28], but their measurement is limited by technical complexity and cost. S100 proteins (S100A8/A9 and S100A12) and myeloid-related protein (MRP8/14) may help differentiate active from inactive disease, especially when combined with ESR or CRP [33,34], although further validation is necessary. Endothelial biomarkers, including VEGF, ECPs, and CECs, have demonstrated associations with disease activity but inconsistent performance across studies [30,31]. Adipokines continue to be exploratory markers without confirmed diagnostic or prognostic utility [32].
Taken together, biomarker research in LVV underscores the limitations of traditional acute-phase reactants and highlights PTX3 and MMP-9 as the most promising emerging tools for assessing vascular-wall inflammation and matrix remodeling. Cytokines, chemokines, and endothelial injury markers contribute to mechanistic insight but are not ready for routine clinical use. Progress will depend on multicenter studies, harmonized assay protocols, and incorporation of biomarker data alongside imaging to improve diagnostic and monitoring strategies.
3. Biomarkers in Medium-Vessel Vasculitis
Medium-sized vessel vasculitides (MVV) primarily affect muscular arteries and include Kawasaki disease (KD) and polyarteritis nodosa (PAN), which differ in epidemiology, pathogenesis, and clinical presentation [1,9]. KD is an acute pediatric vasculitis predominantly affecting the coronary arteries and remains the leading cause of acquired heart disease in children worldwide [35]. PAN is a necrotizing arteritis of medium and small arteries characterized by aneurysm formation and multisystem ischemia [1]. Although numerous biomarkers have been investigated, reliable tools that enable early diagnosis, predict disease course, and stratify vascular risk remain limited.
3.1. Current Clinical Biomarkers in KD
Conventional inflammatory markers are widely used but lack specificity. CRP and ESR are typically elevated in acute KD, with CRP increasing early and ESR remaining high into the subacute phase, especially after IVIG administration [36]. Additional abnormalities include leukocytosis, anemia of inflammation, thrombocytosis, hypoalbuminemia, and hyponatremia.
Serum albumin is a reproducible prognostic marker. Hypoalbuminemia reflects systemic inflammation and vascular permeability and is consistently associated with IVIG resistance and coronary artery abnormalities (CAA) across multiple cohorts [35]. Complete blood count-derived indices, such as the NLR, have also shown predictive value for intravenous immunoglobulins (IVIG) resistance and early CAA formation.
NT-proBNP is the most extensively validated organ-specific biomarker in KD. Levels are significantly higher in KD than in febrile controls, including incomplete presentations, and correlate with myocardial inflammation, early coronary changes, and likelihood of IVIG non-response [37]. Despite variability in cutoff values, NT-proBNP consistently outperforms standard inflammatory markers in diagnostic and prognostic assessment.
3.2. Emerging Biomarkers in KD
Acute KD is characterized by increased concentrations of cytokines and other immune mediators, including IL-6, TNF-α, IL-1β, IL-10, and IL-17A, which together reflect activation of both innate and adaptive immunity [38,39]. IL-1β shows a strong association with the development of coronary arteritis and correlates with clinical severity [38], while IL-17A and the broader Th17 axis are significantly upregulated compared with febrile controls and link systemic inflammation to the risk of coronary artery abnormalities [39,40]. Soluble IL-2 receptor, a marker of T-cell activation, is also elevated in acute KD and decreases after intravenous immunoglobulin treatment, although it adds limited diagnostic value [41].
Endothelial perturbation is a prominent feature of KD, as demonstrated by elevated levels of vascular endothelial growth factor and soluble adhesion molecules such as vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule-1 (ICAM-1), and E-selectin, which reflect increased vascular permeability and endothelial activation [42,43]. Circulating endothelial cells rise during acute illness and fall after treatment, indicating direct injury to the vascular endothelium [44].
Matrix-remodeling pathways contribute to coronary artery changes, with MMP-9 consistently elevated in acute KD and correlating with coronary dilation and aneurysm formation [45].
Additional biomarkers of innate immune activation include high-mobility group box 1 protein (HMGB1) and S100 proteins such as S100A8/A9 and S100A12, which are markedly increased during acute disease. S100A8/A9 helps distinguish KD from other febrile illnesses and may identify patients at risk of resistance to IVIG [46], while S100A12 reflects neutrophil-mediated vascular inflammation and is associated with early coronary involvement [47].
Fibrotic and vascular-remodeling pathways are reflected by increased galectin-3 concentrations, which correlate with the development of CAA [48]. Ferritin provides information on macrophage activation and helps identify a subset of children with macrophage activation syndrome, a hyperinflammatory phenotype associated with treatment resistance and higher risk of adverse outcomes [49].
3.3. Clinical Utility and Limitations of Biomarkers in KD
KD demonstrates a broad biomarker profile reflecting activation of innate and adaptive immunity, endothelial injury, extracellular-matrix degradation, and fibrotic remodelling. Despite this, no single biomarker independently confirms the diagnosis or reliably predicts coronary outcomes, and most markers are limited by nonspecificity, assay variability, or insufficient validation. CRP and ESR remain useful general inflammatory markers but are influenced by intravenous immunoglobulin treatment. Albumin and the NLR offer accessible prognostic information, while galectin-3 and ferritin highlight more severe inflammatory phenotypes but are not routinely applied.
NT-proBNP remains the most clinically useful biomarker in KD due to its ability to aid the diagnosis of incomplete KD and predict intravenous immunoglobulin resistance and early coronary artery changes. Emerging biomarkers, including IL-1β, IL-17A, VEGF, CECs, MMP-9, S100 proteins, and galectin-3, provide important mechanistic insights but require methodological standardization and multicenter evaluation before they can be incorporated into routine practice. Biomarkers, therefore, support but do not replace clinical evaluation and imaging in KD management.
3.4. Current Clinical Biomarkers in PAN
In PAN, ESR and CRP are frequently elevated during active disease but lack specificity and do not reliably distinguish PAN from other vasculitides [50]. Additional abnormalities—leukocytosis, anemia of inflammation, renal dysfunction, and proteinuria—reflect systemic inflammation or organ damage but do not provide disease-specific information.
3.5. Emerging Biomarkers in PAN
Serum HMGB1 levels are significantly elevated in PAN and may exceed those seen in other vasculitides [51]. HMGB1 correlates with CRP and markers of renal involvement.
Anti-phosphatidylserine–prothrombin antibodies (anti-PSPT) are elevated in PAN and decline with treatment [52]. Anti-moesin antibodies correlate with disease activity scores and vascular damage indices [52].
Serum lysosome-associated membrane protein 2 (LAMP-2) concentrations distinguish PAN from antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis at proposed cutoff values and correlate with disease activity [53]. Anti-LAMP-2 antibodies are also increased and show diagnostic potential [54].
Endothelial injury biomarkers, such as soluble VCAM-1, are elevated in small PAN cohorts and reflect endothelial injury or activation [55]. Anti-endothelial cell antibodies (AECA) support an immune-mediated mechanism but lack specificity.
Matrix metalloproteinases, such as MMP-3, may contribute to arterial wall destruction in PAN, though direct clinical evidence is limited [56].
3.6. Clinical Utility and Limitations of Biomarkers in PAN
No available biomarker reliably establishes the diagnosis of PAN or consistently distinguishes it from mimics such as deficiency of adenosine deaminase 2 (DADA2) or hepatitis-associated arteritis. Traditional markers such as ESR and CRP reflect systemic inflammation but lack specificity. HMGB1 may help differentiate PAN from other vasculitides and correlates with markers of renal involvement, though studies remain small. Anti-phosphatidylserine–prothrombin and anti-moesin antibodies correlate with disease activity and vascular damage but require larger validation.
LAMP-2 and anti-LAMP-2 antibodies show the greatest promise for diagnostic use, as proposed concentrations may distinguish PAN from ANCA-associated vasculitis, though methodological variability limits their application. Endothelial injury markers such as soluble VCAM-1, thrombomodulin, and anti-endothelial cell antibodies offer mechanistic insight but insufficient diagnostic discrimination. Overall, PAN currently lacks validated disease-specific biomarkers, and multicenter studies are needed to clarify which emerging candidates may ultimately support diagnosis or prognosis.
4. Biomarkers in Small-Vessel Vasculitis
Small-vessel vasculitides include ANCA-associated vasculitis (AAV) and immune complex–mediated diseases such as IgA vasculitis (IgAV), cryoglobulinemic vasculitis, and hypocomplementemic urticarial vasculitis [1]. IgAV is the most common systemic vasculitis in childhood, although it also affects adults, and is characterized by IgA-containing immune complex deposition with predominant skin, gastrointestinal, and renal involvement [57,58]. Biomarker research is particularly active in IgAV, especially regarding early prediction of nephritis.
4.1. Current Clinical Biomarkers in IgAV
Conventional inflammatory markers such as leukocyte count, CRP, IL-6, and SAA are elevated in children with IgAV compared with controls, and composite biomarker indices combining SAA, IgA, IgM, and CRP have shown diagnostic potential [59]. Adults similarly demonstrate increased IL-6 and SAA levels [60]. CRP, however, reflects nonspecific inflammation and does not reliably predict organ involvement [61].
Pro-inflammatory cytokines, including IL-17A, IL-18, and IL-23, are elevated in the acute phase of IgAV, supporting a role for Th17-skewed immunity [62,63]. TNF-α is increased in both pediatric and adult IgAV and correlates with renal impairment and disease severity [64,65]. Serum calprotectin (S100A8/A9) correlates with disease activity indices such as the Paediatric Vasculitis Activity Score (PVAS) and may offer greater specificity for vasculitic inflammation than CRP [66].
HMGB1 is also elevated in IgAV and is strongly expressed in lesional endothelium, inducing IL-6 and TNF-α release [67]. Higher serum HMGB1 levels are associated with nephritis in both children and adults and correlate with inflammatory and coagulation markers [68,69]. Urinary HMGB1 remains elevated in children with IgAV nephritis (IgAVN) during follow-up and correlates with hematuria and albuminuria [70].
4.2. Emerging Serum Biomarkers for Nephritis (IgAVN)
Galactose-deficient IgA1 (Gd-IgA1) is the most consistently validated serum biomarker for IgAVN. Pediatric and adult cohorts demonstrate higher Gd-IgA1 levels in patients with nephritis than in those without renal involvement, reflecting pathogenic overlap with IgA nephropathy [71,72,73,74,75,76,77]. A recent mechanistic study further confirmed interactions between Gd-IgA1, HMGB1, receptor for advanced glycation end-products (RAGE), and epithelial barrier molecules in IgAV [70].
Other serum markers linked to nephritis include PTX3, which predicts early IgAVN in children [78], and α-smooth muscle actin and c-Met, which correlate with histologic severity and may reflect mesangial and tubular injury [79]. Lower apolipoprotein M levels have been described in pediatric IgAV and IgAVN, suggesting impaired endothelial protective pathways [80]. Proteomic studies identified elevated serum angiotensinogen (AGT) as a marker of IgAVN, possibly reflecting activation of the intrarenal renin–angiotensin system [81].
In adult IgAVN, TNF-α levels associate with renal function impairment and biopsy severity [82].
4.3. Emerging Urinary and Renal Biomarkers in IgAV
Urinary biomarkers show strong potential for early identification of nephritis. Complement components, including urinary C3, C4, C5, and C5a, are increased in children with IgAVN, indicating complement activation within the kidney [83]. A meta-analysis found serum and urinary MMP-9 levels associated with nephritis risk [84].
Urinary AGT is one of the most informative markers. Children with IgAVN have significantly higher urinary AGT at onset, with greater levels in those presenting with proteinuria; values correlate with proteinuria and serum creatinine and often remain elevated during convalescence [85]. Urinary macrophage migration inhibitory factor (MIF) is similarly increased and correlates with microalbuminuria [86].
A systematic review identified kidney injury molecule-1 (KIM-1), MCP-1, N-acetyl-β-D-glucosaminidase (NAG), and AGT as the most promising urinary biomarkers in pediatric IgAVN [87]. Erythrocyte glutathione S-transferase (e-GST) is increased in children with IgAVN at onset and remains elevated for months, suggesting persistent tubular dysfunction [88].
In adult IgAVN, urinary IL-1β, IL-6, IL-8, and neutrophil gelatinase-associated lipocalin (NGAL) distinguish renal involvement from isolated extrarenal disease, and urinary IgA is associated with poor renal outcomes [77].
4.4. Biomarkers of Gastrointestinal Involvement in IgAV
Markers predicting gastrointestinal (GI) involvement include increased platelet and neutrophil counts, higher NLR and PLR, and lower lymphocyte count, mean platelet volume (MPV), and MPV-to-platelet ratio (MPR) in children with significant GI disease [89,90,91,92]. Elevated NLR predicts GI bleeding in adults as well [60]. Serum procalcitonin correlates with GI bleeding risk in pediatric IgAV [61].
Fecal calprotectin is the most validated biomarker of GI involvement. Levels are markedly elevated in children with intestinal symptoms and outperform CRP and blood counts for detecting mucosal inflammation [93,94]. GI involvement is itself associated with increased nephritis risk [58].
4.5. Clinical Utility and Limitations of Biomarkers in IgAV
IgAV is associated with a wide range of biomarkers reflecting systemic inflammation, immune dysregulation, complement activation, endothelial injury, and renal tubular damage. Although commonly used, conventional inflammatory markers do not reliably predict organ involvement. Gd-IgA1 is the most consistently validated serum biomarker for identifying patients at higher risk of nephritis, yet standardized cutoffs and broadly available assays are still lacking.
Urinary biomarkers, such as ATG, KIM-1, MCP-1, NAG, NGAL, complement fragments, and e-GST, show strong potential for early detection and monitoring of renal involvement, although most studies remain limited by small cohorts. Damage-associated molecular patterns, including HMGB1 and S100A8/A9, correlate with systemic inflammation and renal involvement. Fecal calprotectin is the most practical and validated biomarker for detecting gastrointestinal disease.
Although promising, most biomarkers require greater standardization and multicenter validation before routine clinical use.
4.6. Current Clinical Biomarkers in AAV
ESR, CRP, neutrophil counts, and ANCA serology remain the most widely used biomarkers in AAV. CRP and ESR increase during active disease but lack specificity. Anti-myeloperoxidase (anti-MPO) and anti-proteinase 3 (anti-PR3) titers are essential for diagnosis and classification; rising ANCA levels, particularly reappearance of MPO-ANCA, may precede relapse in a subset of patients [95], although the results are not unequivocal [96].
Ratios integrating inflammation and nutrition have been explored: the CRP-to-albumin ratio (CAR) is an independent predictor of all-cause mortality in AAV, comparable in prognostic value to major clinical risk factors [97].
Complement activation has emerged as a key pathogenic pathway. Lower serum C3 at diagnosis is associated with poor renal prognosis, higher chronicity indices, and reduced treatment responsiveness [98]. In pediatric AAV, elevated von Willebrand factor (vWF) antigen correlates with active disease and endothelial activation [99].
Delta neutrophil index (DNI), representing immature granulocyte proportion, correlates with disease activity (BVAS) and predicts relapse risk, especially in granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA) [100].
4.7. Emerging Serum Biomarkers in AAV
Targeted proteomics identified tissue inhibitor of metalloproteinase-1 (TIMP-1) as a strong discriminator between active AAV and remission, and between mild and severe disease [101].
Comprehensive cytokine profiling revealed that IL-8, IL-15, and IL-18 binding proteins, nerve growth factor (NGF)-β, thymic and activating regulatory chemokine (TARC), osteopontin, soluble ICAM-1, and KIM-1 distinguish patients more effectively when grouped by ANCA serotype than by clinical subtype, supporting the relevance of ANCA-specific immunopathologic signatures [102].
PTX3 is highly promising: serum and urine PTX3 are elevated in active AAV, correlate with BVAS, and outperform CRP for detecting renal involvement [103]. Anti-PTX3 autoantibodies may help identify ANCA-negative AAV [104].
HMGB1 and calgranulins (S100A8/A9, S100A12) are elevated in AAV and reflect damage-associated molecular pattern (DAMP)-driven inflammation. HMGB1 is particularly associated with renal involvement and granulomatous burden, though findings vary among studies [105,106,107,108,109,110]. S100A8/A9 and S100A12 are elevated in MPO-AAV and predict relapse in PR3-AAV patients treated with rituximab [111,112,113].
Low soluble RAGE (sRAGE) levels support enhanced DAMP-mediated inflammation [110].
4.8. Emerging Urinary and Renal Biomarkers in AAV
Urinary PTX3 correlates with active renal vasculitis and reflects albuminuria and estimated glomerular filtration rate (eGFR) decline [103]. Urinary HMGB1 correlates with ESR, CRP, and BVAS and indicates intrarenal inflammation [114].
Urinary KIM-1 and other renal injury molecules have shown discriminatory potential, particularly across ANCA serotypes, though validation remains limited [102].
4.9. Clinical Utility and Limitations in AAV
AAV is associated with numerous circulating and urinary biomarkers reflecting inflammation, neutrophil activation, complement consumption, endothelial injury, and DAMP-mediated pathways. Traditional inflammatory markers remain widely used but lack specificity, and ANCA titers, while essential for diagnosis and classification, do not reliably predict relapse across all patients. Low C3 at diagnosis consistently predicts worse renal outcomes.
Among emerging biomarkers, PTX-3 is particularly promising due to its vascular-wall origin, correlation with disease activity, and reduced susceptibility to glucocorticoid suppression. HMGB1 and S100 proteins reflect DAMP-mediated inflammation and may aid relapse prediction. TIMP-1 discriminates active from quiescent disease and stratifies severity. Urinary biomarkers, including HMGB1, PTX3, and KIM-1, reflect intrarenal inflammation and may improve assessment of renal involvement, although larger studies are required. Overall, most biomarkers remain investigational and require robust validation before integration into routine practice.
4.10. Cryoglobulinemic Vasculitis
Biomarker studies in cryoglobulinemic vasculitis (CV) are scarce. IL-6 is higher in active CV than in remission [115]. Interferon-related chemokines, particularly C-X-C motif chemokine ligand 10 (CXCL10/IP-10), are elevated in hepatitis C-associated mixed cryoglobulinemia [116,117]. However, their diagnostic and prognostic utility remains unproven.
4.11. Hypocomplementemic Urticarial Vasculitis
Validated biomarkers are essentially absent. Persistent hypocomplementemia, particularly low C1q, C3, and C4, is the hallmark laboratory abnormality and part of the diagnostic criteria. Anti-C1q antibodies support diagnosis but do not track activity. No cytokine, chemokine, or endothelial marker has demonstrated reproducible clinical utility [1].
5. Biomarkers in Variable-Vessel Vasculitis
Variable-vessel vasculitis comprises disorders affecting arteries, veins, and capillaries of multiple sizes. Behçet’s disease (BD) and Cogan’s syndrome are the principal conditions in this category, although biomarker research is almost entirely concentrated on BD. Cogan’s syndrome remains limited to anecdotal reports and lacks validated biomarkers [118].
5.1. Current Clinical and Genetic Markers in BD
BD demonstrates a strong association with HLA-B51, which contributes to genetic susceptibility and varies across phenotypes, with higher prevalence in males and in patients presenting with ocular, genital, and cutaneous disease [119,120]. Nevertheless, HLA-B51 lacks diagnostic specificity and is not included in classification criteria [121].
Routine inflammatory markers such as ESR and CRP often increase during active BD but lack specificity [122,123]. Hematologic indices, especially increased NLR and reduced MPV, correlate with disease severity and vascular involvement [124,125,126,127]. Higher NLR has been observed in neuro-Behçet and in patients with active genital ulcers [127]. Composite inflammation–nutrition indices, such as the CAR, also reflect systemic inflammatory burden but remain nonspecific [120].
5.2. Serum Inflammatory Biomarkers and Cytokines in BD
SAA is frequently elevated in BD and is associated with oral ulcers, neurologic involvement, and ocular disease. Persistently high SAA levels may correlate with thrombotic risk and ocular relapse [128,129,130]. SAA also shows an association with IgD levels in mucocutaneous BD [131].
Markers of endothelial dysfunction, including vWF, soluble ICAM-1, VCAM-1, and endothelial microparticles, are increased particularly in vasculo-BD, although their diagnostic discrimination is limited [132,133]. Homocysteine is often elevated in BD and may contribute to thrombosis risk.
Numerous cytokines show consistent elevation in BD, including TNF-α, IL-1β, IL-2, IL-6, IL-8, IL-17, IL-18, and IL-23 [134,135,136,137,138]. IL-6 correlates with arthritic symptoms and major organ inflammatory events [135]. IL-18 increases during ocular flares [137]. IL-23 is elevated in uveitis [139]. IL-8 is strongly associated with mucocutaneous and vascular involvement and may be more sensitive than ESR or CRP for detecting active disease [135,136].
Salivary cytokines mirror mucosal inflammation. IL-1β, IL-8, TNF-α, and IL-6 are increased in BD patients with oral ulcers and vary with disease activity, supporting potential for noninvasive monitoring [134].
Alarmins such as HMGB1 and S100 proteins (S100A8/A9, S100A12) are elevated in BD, but correlations with disease activity are inconsistent, limiting their value as clinical biomarkers [140,141,142,143,144].
Circulating microRNAs may differentiate BD from controls and reflect thrombo-inflammatory pathways, but their use is still investigational.
5.3. Urinary Metabolomic and Proteomic Biomarkers in BD
Urine-based metabolomics has identified panels of discriminatory metabolites separating BD patients from healthy controls, although findings require external validation [145]. Urinary proteomic profiling in BD-associated uveitis has identified differentially expressed proteins such as CD38, dipeptidyl peptidase-4 (DPP4), creatine kinase B-type, and S100A8/A9, suggesting potential relevance for monitoring ocular inflammation [146].
5.4. Clinical Utility and Limitations of Biomarkers in BD
Despite extensive investigation, no biomarker has demonstrated adequate sensitivity, specificity, or reproducibility for routine diagnostic or monitoring use in BD. HLA-B51 reflects genetic susceptibility but lacks diagnostic power. ESR, CRP, NLR, and MPV provide general inflammatory assessment but are nonspecific. Serum amyloid A and cytokines such as IL-6, IL-8, IL-18, and IL-23 show consistent associations with disease activity and specific organ involvement, yet standardized thresholds and multicenter validation are lacking.
Markers of endothelial dysfunction and alarmins such as HMGB1 and S100 proteins offer mechanistic relevance but inconsistent clinical utility. MicroRNAs and proteomic or metabolomic urinary signatures remain exploratory. Overall, biomarker development in BD remains limited by heterogeneous study designs and small cohorts.
6. Single-Organ Vasculitis
Primary angiitis of the central nervous system (PACNS) is a rare vasculitis restricted to the brain and spinal cord. It presents with acute or subacute neurologic or psychiatric symptoms that frequently resemble alternative neurologic disorders, complicating diagnosis [147,148,149]. Biomarker research in PACNS remains limited, especially in children. The only pediatric study to date reported increased levels of vWF antigen in 39 children with childhood-onset PACNS, with levels declining in parallel with clinical improvement. vWF correlated more closely with disease activity than ESR or CRP, suggesting value as a marker of vascular inflammation in this population [150].
In adults, additional biomarker candidates have been proposed. Soluble triggering receptor expressed on myeloid cells 2 (sTREM2) is elevated in both serum and cerebrospinal fluid (CSF), particularly in patients with poorer outcomes. sTREM2 levels correlate with lesion volume and inflammatory mediators such as TNF-α, IL-8, IL-6, and complement components, indicating involvement of myeloid activation in PACNS pathobiology [151].
CECs, reflecting endothelial injury, are increased in active PACNS compared with remission, healthy controls, and non-inflammatory vasculopathies, including reversible cerebral vasoconstriction syndrome and moyamoya disease. These findings suggest that CEC may aid both differential diagnosis and assessment of ongoing inflammatory activity [152].
Persistently elevated CSF IL-17 production has been demonstrated in patients with PACNS compared with non-inflammatory neurological controls. In stroke presentations, increased IL-17 production distinguished PACNS with high specificity, supporting its potential use in select diagnostic contexts [153].
A mass spectrometry-based CSF proteomic study identified 14 proteins, including the amyloid-β A4 protein, present at lower concentrations in PACNS than in controls. These findings suggest neuroaxonal injury and support the potential of CSF proteomics for the discovery of new biomarkers, though all candidates require validation in larger cohorts [154].
7. Perspectives on Novel Biomarker Discovery in Vasculitis
While numerous inflammatory biomarkers have been described in vasculitis, recent research increasingly focuses on identifying biomarkers that reflect underlying molecular and genetic mechanisms of disease. Genetic studies have provided important insights into disease susceptibility and heterogeneity across vasculitis subtypes. Genome-wide association studies have identified associations between specific HLA alleles and immune-regulatory genes with diseases such as GCA, TA, AAV, and BD, supporting a role for genetically driven immune dysregulation and offering a framework for genetically informed biomarker discovery [119,120,155,156,157].
Molecular profiling approaches have further expanded biomarker research beyond traditional serum inflammatory markers. Transcriptomic analyses of blood and tissue samples have identified disease- and activity-associated gene expression signatures, particularly involving interferon pathways, Th1/Th17 polarization, neutrophil activation, and complement-related genes in AAV and LVV [28,97,98,101,102,111,112].
Proteomic and metabolomic studies have enabled the identification of circulating and urinary biomarkers linked to endothelial injury, extracellular matrix remodeling, and organ-specific damage, such as urinary biomarkers reflecting intrarenal inflammation in IgAV and AAV [74,83,87,101,114,145,146].
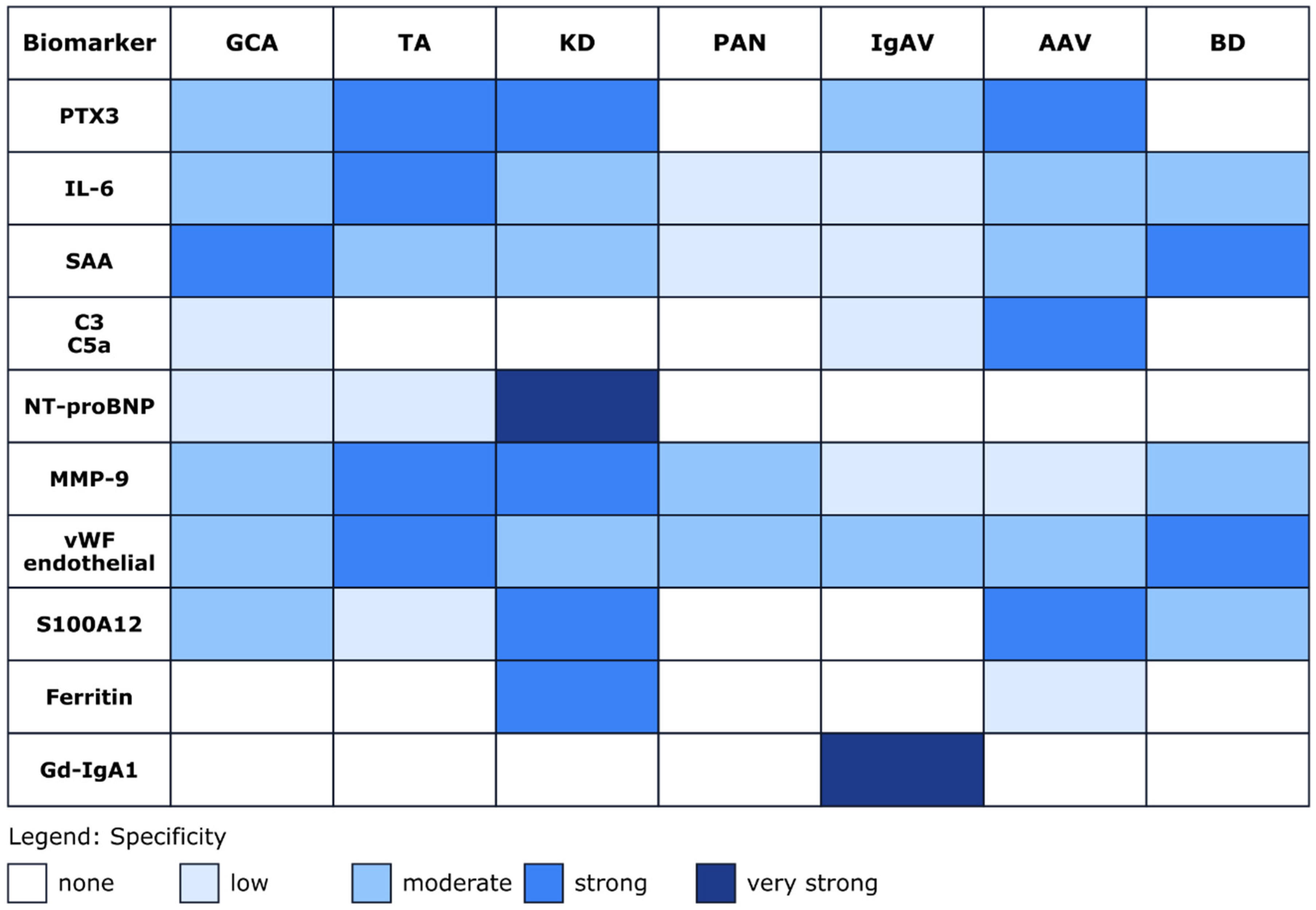
Importantly, several of the most promising emerging biomarkers discussed in this review (Figure 4), including PTX3, S100 proteins, complement components, Gd-IgA1, and urinary kidney injury markers, have been identified through mechanistic or molecularly guided approaches rather than unbiased screening alone [20,22,33,66,71,74,87,114]. Future biomarker discovery is therefore likely to benefit from integrative strategies combining genetic susceptibility data, molecular pathway analysis, and longitudinal clinical phenotyping, with the goal of identifying biomarkers that improve disease stratification, activity assessment, and prediction of organ involvement.
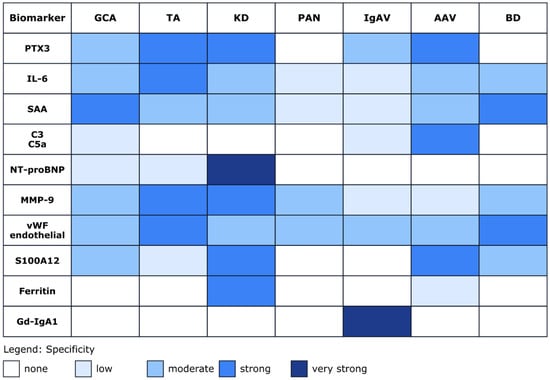
Figure 4.
Biomarker specificity heatmap across vasculitis types. The heatmap illustrates the relative association and reported specificity of selected biomarkers across different vasculitis subtypes. The color intensity reflects the strength and consistency of associations reported in the literature. Color gradients indicate relative trends reported across studies and should not be interpreted as quantitative effect sizes. The heatmap was compiled by the authors based on a qualitative synthesis of published studies and expert interpretation of the available evidence, rather than derived from a single dataset or quantitative meta-analysis. Where evidence was inconsistent across studies, we assigned a lower intensity to reflect uncertainty. The selected references for individual biomarkers are as follows. PTX3: [20,21,22,78,103,158,159,160]; IL-6: [17,23,24,26,27,38,59,60,161,162,163]; SAA: [13,18,59,128,129,130,164,165]; C3, C5a: [83,98,166]; NT-proBNP: [37,167]; MMP-9: [23,45,56,168,169]; vWF/endothelial markers: [99,170,171,172,173]; S100A12: [34,46,47,111,112,113,144]; Ferritin: [49,174]; and Gd-IgA1: [72,73,74,75]. The list of abbreviations is as follows: AAV: ANCA-associated vasculitis; BD: Behçet’s disease; C3: complement component 3; C5a: complement component C5a; GCA: giant cell arteritis; Gd-IgA1: galactose-deficient immunoglobulin A1; IgAV: IgA vasculitis; IL-6: interleukin-6; KD: Kawasaki disease; MMP-9: Matrix metalloproteinase-9; NT-proBNP: N-terminal pro–B-type natriuretic peptide; PAN: polyarteritis nodosa; PTX3: pentraxin-3; S100A12: S100 calcium-binding protein A12; SAA: serum amyloid A; TA: Takayasu arteritis; vWF: von Willebrand factor.
8. Conclusions
Across vasculitides, biomarker discovery is hindered by small cohort sizes, treatment heterogeneity, lack of standardized outcome measures, and variability in assay methodology. Most candidate biomarkers lack external validation, disease specificity, or incremental predictive performance compared with established clinical and imaging tools. Future progress will require large, prospective, multi-omic studies with harmonized sample processing, integration of vascular imaging endpoints, and evaluation of biomarker panels rather than single analytes.
Author Contributions
Conceptualization, M.S. and M.J.; validation, M.S. and M.H.; investigation, M.S. and M.H.; data curation, M.S. and M.H.; writing—original draft preparation, M.S. and M.H.; writing—review and editing, M.J.; visualization, M.S. and M.H.; supervision, M.J.; project administration, M.J.; funding acquisition, M.J. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Croatian Science Foundation under the project number HRZZ-IP-2024-05-6848 and National Recovery and Resilience Plan (NPOO) 10106-25-2897 (GUT-VAS)—funded by the European Union–NextGenerationEU.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Jennette, J.C.; Falk, R.J.; Bacon, P.A.; Basu, N.; Cid, M.C.; Ferrario, F.; Flores-Suarez, L.F.; Gross, W.L.; Guillevin, L.; Hagen, E.C.; et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013, 65, 1–11. [Google Scholar] [CrossRef]
- Watts, R.; Hatemi, G.; Burns, J.; Mohammad, A. Global epidemiology of vasculitis. Nat. Rev. Rheumatol. 2022, 18, 22–34. [Google Scholar] [CrossRef]
- Monach, P. Biomarkers in vasculitis. Curr. Opin. Rheumatol. 2014, 26, 24–30. [Google Scholar] [CrossRef]
- Csernok, E.; Bossuyt, X. Investigations in systemic vasculitis. The role of the laboratory. Best Pract. Res. Clin. Rheumatol. 2018, 32, 52–62. [Google Scholar] [CrossRef]
- Grayson, P.C.; Kaplan, M.J. Diseases of blood vessels: Immune system involvement in vasculitis and vasculopathy. Semin. Immunopathol. 2022, 44, 255–258. [Google Scholar] [CrossRef]
- Renson, T.; Kelly, M.M.; Benediktsson, H.; Grundhoefer, N.; Luca, N.; Miettunen, P.; Twilt, M.; Grisaru, S.; Wade, A.; Banks, A.; et al. Non-invasive biomarkers of disease activity and organ damage in ANCA-associated vasculitis: A systematic review. RMD Open 2024, 10, e003579. [Google Scholar] [CrossRef] [PubMed]
- Delvino, P.; Baldini, C.; Bonacini, M.; Croci, S.; Di Cianni, F.; Ferro, F.; Marvisi, C.; Monti, S.; Moretti, M.; Muratore, F.; et al. Systemic vasculitis: One year in review 2025. Clin. Exp. Rheumatol. 2025, 43, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Shumnalieva, R.; Ermencheva, P.; Kotov, G.; Parvova-Hristova, I.; Bakopoulou, K.; Kaouri, I.E.; Mileva, N.; Velikova, T. New Biomarkers for Systemic Necrotizing Vasculitides. J. Clin. Med. 2024, 13, 2264. [Google Scholar] [CrossRef] [PubMed]
- Cid, M.C.; Prieto-González, S.; Arguis, P.; Espígol-Frigolé, G.; Butjosa, M.; Hernández-Rodríguez, J.; Segarra, M.; Lozano, E.; García-Martínez, A. The spectrum of vascular involvement in giant-cell arteritis: Clinical consequences of detrimental vascular remodelling at different sites. APMIS 2009, 117, 10–20. [Google Scholar] [CrossRef]
- Numano, F.; Okawara, M.; Inomata, H.; Kobayashi, Y. Takayasu’s arteritis. Lancet 2000, 356, 1023–1025. [Google Scholar] [CrossRef]
- Hunder, G.G.; Bloch, D.A.; Michel, B.A.; Stevens, M.B.; Arend, W.P.; Calabrese, L.H.; Edworthy, S.M.; Fauci, A.S.; Leavitt, R.Y.; Lie, J.T.; et al. The American College of Rheumatology 1990 criteria for the classification of giant cell arteritis. Arthritis Rheum. 1990, 33, 1122–1128. [Google Scholar] [CrossRef]
- Kermani, T.A.; Schmidt, J.; Crowson, C.S.; Ytterberg, S.R.; Hunder, G.G.; Matteson, E.L.; Warrington, K.J. Utility of erythrocyte sedimentation rate and C-reactive protein for the diagnosis of giant cell arteritis. Semin. Arthritis Rheum. 2012, 41, 866–871. [Google Scholar] [CrossRef]
- Burja, B.; Feichtinger, J.; Lakota, K.; Thallinger, G.G.; Sodin-Semrl, S.; Kuret, T.; Rotar, Ž.; Ješe, R.; Žigon, P.; Čučnik, S.; et al. Utility of serological biomarkers for giant cell arteritis in a large cohort of treatment-naïve patients. Clin. Rheumatol. 2019, 38, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Hocevar, A.; Rotar, Z.; Jese, R.; Semrl, S.S.; Pizem, J.; Hawlina, M.; Tomšič, M. Do Early Diagnosis and Glucocorticoid Treatment Decrease the Risk of Permanent Visual Loss and Early Relapses in Giant Cell Arteritis: A Prospective Longitudinal Study. Medicine 2016, 95, e3210. [Google Scholar] [CrossRef]
- Quinn, K.A.; Gribbons, K.B.; Carette, S.; Cuthbertson, D.; Khalidi, N.A.; Koening, C.L.; Langford, C.A.; McAlear, C.A.; Monach, P.A.; Moreland, L.W.; et al. Patterns of clinical presentation in Takayasu’s arteritis. Semin. Arthritis Rheum. 2020, 50, 576–581. [Google Scholar] [CrossRef]
- Wang, X.; Dang, A.; Lv, N.; Liu, Q.; Chen, B. High-sensitivity C-reactive protein predicts adverse cardiovascular events in patients with Takayasu arteritis with coronary artery involvement. Clin. Rheumatol. 2016, 35, 679–684. [Google Scholar] [CrossRef]
- Li, J.; Wang, Y.; Wang, Y.; Wang, Y.; Yang, Y.; Zhao, J.; Li, M.; Tian, X.; Zeng, X. Association between acute phase reactants, interleukin-6, tumor necrosis factor-α, and disease activity in Takayasu’s arteritis patients. Arthritis Res. Ther. 2020, 22, 285. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.M.; Goel, R.; Hindhumati, M.; Jayakanthan, K.; Visalakshi, J.; Joseph, G.; Danda, S.; Danda, D. Serum amyloid A as a marker of disease activity and treatment response in Takayasu arteritis. Rheumatol. Int. 2017, 37, 1643–1649. [Google Scholar] [CrossRef] [PubMed]
- Seringec Akkececi, N.; Yildirim Cetin, G.; Gogebakan, H.; Acipayam, C. The C-Reactive Protein/Albumin Ratio and Complete Blood Count Parameters as Indicators of Disease Activity in Patients with Takayasu Arteritis. Med. Sci. Monit. 2019, 25, 1401–1409. [Google Scholar] [CrossRef]
- Dagna, L.; Salvo, F.; Tiraboschi, M.; Bozzolo, E.P.; Franchini, S.; Doglioni, C.; Manfredi, A.A.; Baldissera, E.; Sabbadini, M.G. Pentraxin-3 as a marker of disease activity in Takayasu arteritis. Ann. Intern. Med. 2011, 155, 425–433. [Google Scholar] [CrossRef]
- Alibaz-Oner, F.; Aksu, K.; Yentur, S.P.; Keser, G.; Saruhan-Direskeneli, G.; Direskeneli, H. Plasma pentraxin-3 levels in patients with Takayasu’s arteritis during routine follow-up. Clin. Exp. Rheumatol. 2016, 34, S73–S76. [Google Scholar]
- Tombetti, E.; Di Chio, M.C.; Sartorelli, S.; Papa, M.; Salerno, A.; Bottazzi, B.; Bozzolo, E.P.; Greco, M.; Rovere-Querini, P.; Baldissera, E.; et al. Systemic pentraxin-3 levels reflect vascular enhancement and progression in Takayasu arteritis. Arthritis Res. Ther. 2014, 16, 479. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ma, L.; Yan, F.; Liu, H.; Ding, Y.; Hou, J.; Jiang, L. MMP-9 and IL-6 are potential biomarkers for disease activity in Takayasu’s arteritis. Int. J. Cardiol. 2012, 156, 236–238. [Google Scholar] [CrossRef]
- Park, M.C.; Lee, S.W.; Park, Y.B.; Lee, S.K. Serum cytokine profiles and their correlations with disease activity in Takayasu’s arteritis. Rheumatology 2006, 45, 545–548. [Google Scholar] [CrossRef]
- Arraes, A.E.D.; de Souza, A.W.S.; Mariz, H.A.; Silva, N.P.; Torres, I.C.G.; Pinto, P.N.V.; Lima, E.N.; Sato, E.I. 18F-Fluorodeoxyglucose positron emission tomography and serum cytokines and matrix metalloproteinases in the assessment of disease activity in Takayasu’s arteritis. Rev. Bras. Reumatol. Engl. Ed. 2016, 56, 299–308. [Google Scholar] [CrossRef]
- Noris, M.; Daina, E.; Gamba, S.; Bonazzola, S.; Remuzzi, G. Interleukin-6 and RANTES in Takayasu arteritis: A guide for therapeutic decisions? Circulation 1999, 100, 55–60. [Google Scholar] [CrossRef]
- Savioli, B.; Abdulahad, W.H.; Brouwer, E.; Kallenberg, C.G.M.; de Souza, A.W.S. Are cytokines and chemokines suitable biomarkers for Takayasu arteritis? Autoimmun. Rev. 2017, 16, 1071–1078. [Google Scholar] [CrossRef]
- Saadoun, D.; Garrido, M.; Comarmond, C.; Desbois, A.C.; Domont, F.; Savey, L.; Terrier, B.; Geri, G.; Rosenzwajg, M.; Klatzmann, D.; et al. Th1 and Th17 cytokines drive inflammation in Takayasu arteritis. Arthritis Rheumatol. 2015, 67, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Xu, M.; Cui, X.; Ma, L.; Cheng, H.; Hou, J.; Sun, X.; Ma, L.; Jiang, L. Potential Role of Macrophage Phenotypes and CCL2 in the Pathogenesis of Takayasu Arteritis. Front. Immunol. 2021, 12, 646516. [Google Scholar] [CrossRef]
- Dogan, S.; Piskin, O.; Solmaz, D.; Akar, S.; Gulcu, A.; Yuksel, F.; Cakır, V.; Sari, I.; Akkoc, N.; Onen, F. Markers of endothelial damage and repair in Takayasu arteritis: Are they associated with disease activity? Rheumatol. Int. 2014, 34, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Keşkek, Ş.Ö.; Bozkırlı-Ersözlü, E.D.; Kozanoglu, I.; Yücel, A.E. High Levels of Circulating Endothelial Progenitor Cells Are Associated with Acrotism in Patients with Takayasu Arteritis. Med. Princ. Pract. 2017, 26, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, H.; Gerdan, V.; Kozaci, D.; Solmaz, D.; Akar, S.; Can, G.; Gulcu, A.; Goktay, Y.; Sari, I.; Birlik, M.; et al. Ghrelin and adipokines as circulating markers of disease activity in patients with Takayasu arteritis. Arthritis Res. Ther. 2012, 14, R272. [Google Scholar] [CrossRef] [PubMed]
- Goel, R.; Nair, A.; Kabeerdoss, J.; Mohan, H.; Jeyaseelan, V.; Joseph, G.; Danda, D. Study of serial serum myeloid-related protein 8/14 as a sensitive biomarker in Takayasu arteritis: A single centre study. Rheumatol. Int. 2018, 38, 623–630. [Google Scholar] [CrossRef]
- Springer, J.M.; Monach, P.; Cuthbertson, D.; Carette, S.; Khalidi, N.A.; McAlear, C.A.; Pagnoux, C.; Seo, P.; Warrington, K.J.; Ytterberg, S.R.; et al. Serum S100 Proteins as a Marker of Disease Activity in Large Vessel Vasculitis. J. Clin. Rheumatol. 2018, 24, 393–395. [Google Scholar] [CrossRef]
- Burns, J.C.; Glodé, M.P. Kawasaki syndrome. Lancet 2004, 364, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Newburger, J.W.; Takahashi, M.; Gerber, M.A.; Gewitz, M.; Tani, L.Y.; Burns, J.C.; Bolger, A.F.; Gewitz, M.; Baker, A.L.; Jackson, M.A.; et al. Diagnosis, treatment, and long-term management of Kawasaki disease. Circulation 2004, 110, 2747–2771. [Google Scholar] [CrossRef]
- Dahdah, N.; Siles, A.; Fournier, A.; Cousineau, J.; Delvin, E.; Saint-Cyr, C.; Spiegelblatt, L.; Bonny, Y.; Vartian, M.; Montigny, M. Natriuretic peptide as an adjunctive diagnostic test in the acute phase of Kawasaki disease. Pediatr. Cardiol. 2009, 30, 810–817. [Google Scholar] [CrossRef]
- Zhang, H.; Song, H.B.; Wang, D.X.; Deng, H.Y.; Sun, W.L. Correlation between the Level of Inflammatory Cytokines and Prognosis in Children with IVIG-sensitive Kawasaki Disease and IVIG-resistant Kawasaki Disease. Pak. J. Med. Sci. 2022, 38, 1165–1169. [Google Scholar] [CrossRef]
- Lu, Y.; Hu, F.Q. Elevated Serum IL-17A in Kawasaki Disease Patients Predicts Responsiveness to Intravenous Immunoglobulin Therapy. Int. Arch. Allergy Immunol. 2025, 186, 159–165. [Google Scholar] [CrossRef]
- Brodeur, K.E.; Liu, M.; Ibanez, D.; de Groot, M.J.; Chen, L.; Du, Y.; Seyal, E.; Laza-Briviesca, R.; Baker, A.; Chang, J.C.; et al. Elevation of IL-17 Cytokines Distinguishes Kawasaki Disease From Other Pediatric Inflammatory Disorders. Arthritis Rheumatol. 2024, 76, 285–292. [Google Scholar] [CrossRef]
- Teraura, H.; Kotani, K.; Minami, T.; Takeshima, T.; Shimooki, O.; Kajii, E. The serum concentration of soluble interleukin-2 receptor in patients with Kawasaki disease. Ann. Clin. Biochem. 2017, 54, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Nash, M.C.; Shah, V.; Dillon, M.J. Soluble cell adhesion molecules and von Willebrand factor in children with Kawasaki disease. Clin. Exp. Immunol. 1995, 101, 13–17. [Google Scholar] [CrossRef]
- Yu, X.; Wu, D.; Song, G. Assessment of Endothelial Dysfunction in Patients with Kawasaki Disease: A Meta-Analysis. Rev. Cardiovasc. Med. 2022, 23, 260. [Google Scholar] [CrossRef]
- Nakatani, K.; Takeshita, S.; Tsujimoto, H.; Kawamura, Y.; Tokutomi, T.; Sekine, I. Circulating endothelial cells in Kawasaki disease. Clin. Exp. Immunol. 2003, 131, 536–540. [Google Scholar] [CrossRef]
- Tian, F.; Ma, L.; Zhao, R.; Ji, L.; Wang, X.; Sun, W.; Jiang, Y. Correlation Between Matrix Metalloproteinases With Coronary Artery Lesion Caused by Kawasaki Disease. Front. Pediatr. 2022, 10, 802217. [Google Scholar] [CrossRef]
- Feng, C.; Li, Q.; Yang, M.; Chen, Y.; Zhang, M.; Wang, H.; Li, X. Single-cell transcriptomic analysis reveals a systemic immune dysregulation in intravenous immunoglobulin non-responsive Kawasaki disease. Front. Immunol. 2025, 16, 1702290. [Google Scholar] [CrossRef] [PubMed]
- Agrafiotou, A.; Sapountzi, E.; Margoni, A.; Fotis, L. Immunophenotype of Kawasaki Disease: Insights into Pathogenesis and Treatment Response. Life 2025, 15, 1012. [Google Scholar] [CrossRef]
- Numano, F.; Shimizu, C.; Jimenez-Fernandez, S.; Vejar, M.; Oharaseki, T.; Takahashi, K.; Salgado, A.; Tremoulet, A.H.; Gordon, J.B.; Burns, J.C.; et al. Galectin-3 is a marker of myocardial and vascular fibrosis in Kawasaki disease patients with giant aneurysms. Int. J. Cardiol. 2015, 201, 429–437. [Google Scholar] [CrossRef]
- Wen, H.; Hun, M.; Zhao, M.; Han, P.; He, Q. Serum ferritin as a crucial biomarker in the diagnosis and prognosis of intravenous immunoglobulin resistance and coronary artery lesions in Kawasaki disease: A systematic review and meta-analysis. Front. Med. 2022, 9, 941739. [Google Scholar] [CrossRef]
- Hernández-Rodríguez, J.; Alba, M.A.; Prieto-González, S.; Cid, M.C. Diagnosis and classification of polyarteritis nodosa. J. Autoimmun. 2014, 48–49, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Li, N.; Zhu, Q.; Wu, T.; Heizati, M.; Wang, G.; Yao, X.; Luo, Q.; Liu, S.; Liu, S.; et al. Association of serum high mobility group box 1 levels with disease activity and renal involvement in patients with systemic vasculitis. Medicine 2019, 98, e14493. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Takeuchi, S.; Soma, Y.; Suzuki, K.; Tsukita, S.; Ishizu, A.; Suzuki, K.; Kawakami, T. Presence of anti-phosphatidylserine-prothrombin complex antibodies and anti-moesin antibodies in patients with polyarteritis nodosa. J. Dermatol. 2017, 44, 18–22. [Google Scholar] [CrossRef]
- Li, N.; Zhu, B.; Zhu, Q.; Heizati, M.; Wu, T.; Wang, G.; Yao, X.; Luo, Q.; Liu, S.; Liu, S. Serum lysosomal-associated membrane protein-2 levels are increased in small and medium-vessel vasculitis, especially in polyarteritis nodosa. Clin. Exp. Rheumatol. 2019, 37, 79–85. [Google Scholar]
- Gibson, K.M.; Kain, R.; Luqmani, R.A.; Ross, C.J.; Cabral, D.A.; Brown, K.L. Autoantibodies Against Lysosome Associated Membrane Protein-2 (LAMP-2) in Pediatric Chronic Primary Systemic Vasculitis. Front. Immunol. 2021, 11, 624758. [Google Scholar] [CrossRef]
- Coll-Vinent, B.; Grau, J.M.; López-Soto, A.; Oristrell, J.; Font, C.; Bosch, X.; Mirapeix, E.; Urbano-Márquez, A.; Cid, M.C. Circulating soluble adhesion molecules in patients with classical polyarteritis nodosa. Br. J. Rheumatol. 1997, 36, 1178–1183. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Pla, A.; Warner, R.L.; Cuthbertson, D.; Carette, S.; Khalidi, N.A.; Koening, C.L.; Langford, C.A.; McAlear, C.A.; Moreland, L.W.; Pagnoux, C.; et al. Evaluation of Potential Serum Biomarkers of Disease Activity in Diverse Forms of Vasculitis. J. Rheumatol. 2020, 47, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Jelusic, M.; Sestan, M.; Giani, T.; Cimaz, R. New Insights and Challenges Associated With IgA Vasculitis and IgA Vasculitis With Nephritis-Is It Time to Change the Paradigm of the Most Common Systemic Vasculitis in Childhood? Front. Pediatr. 2022, 10, 853724. [Google Scholar] [CrossRef]
- Sestan, M.; Kifer, N.; Frkovic, M.; Sapina, M.; Srsen, S.; Batnozic Varga, M.; Ovuka, A.; Held, M.; Gudelj Gracanin, A.; Kozmar, A.; et al. Gastrointestinal involvement and its association with the risk for nephritis in IgA vasculitis. Ther. Adv. Musculoskelet. Dis. 2021, 13, 1759720X211024828. [Google Scholar] [CrossRef]
- Purevdorj, N.; Mu, Y.; Gu, Y.; Zheng, F.; Wang, R.; Yu, J.; Sun, X. Clinical significance of the serum biomarker index detection in children with Henoch-Schönlein purpura. Clin. Biochem. 2018, 52, 167–170. [Google Scholar] [CrossRef]
- Kuret, T.; Lakota, K.; Žigon, P.; Ogrič, M.; Sodin-Šemrl, S.; Čučnik, S.; Tomšič, M.; Hočevar, A. Insight into inflammatory cell and cytokine profiles in adult IgA vasculitis. Clin. Rheumatol. 2019, 38, 331–338. [Google Scholar] [CrossRef]
- Teng, X.; Wang, Y.; Lin, N.; Sun, M.; Wu, J. Evaluation of serum procalcitonin and C-reactive protein levels as biomarkers of Henoch-Schönlein purpura in pediatric patients. Clin. Rheumatol. 2016, 35, 667–671. [Google Scholar] [CrossRef]
- Jaszczura, M.; Mizgała-Izworska, E.; Świętochowska, E.; Machura, E. Serum levels of selected cytokines [interleukin (IL)-17A, IL-18, IL-23] and chemokines (RANTES, IP10) in the acute phase of immunoglobulin A vasculitis in children. Rheumatol. Int. 2019, 39, 1945–1953. [Google Scholar] [CrossRef] [PubMed]
- Kisaoglu, H.; Misir, S.; Aliyazicioglu, Y.; Kalyoncu, M. Interleukin -17 and oxidative stress in children with immunoglobulin A vasculitis. Scand. J. Rheumatol. 2022, 51, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Wang, Q.; Zhang, S.; Zhang, L. Correlation between serum inflammatory factors TNF-α, IL-8, IL-10 and Henoch-Schonlein purpura with renal function impairment. Exp. Ther. Med. 2018, 15, 3924–3928. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.S. The role of tumor necrosis factor-alpha in Henoch-Schonlein purpura. Pediatr. Nephrol. 2005, 20, 149–153. [Google Scholar] [CrossRef]
- Srsen, S.; Held, M.; Sestan, M.; Kifer, N.; Kozmar, A.; Supe Domic, D.; Benzon, B.; Gagro, A.; Frkovic, M.; Jelusic, M. Serum Levels of S100A8/A9 as a Biomarker of Disease Activity in Patients with IgA Vasculitis. Biomedicines 2024, 12, 750. [Google Scholar] [CrossRef]
- Chen, T.; Guo, Z.P.; Wang, W.J.; Qin, S.; Cao, N.; Li, M.M. Increased serum HMGB1 levels in patients with Henoch-Schönlein purpura. Exp. Dermatol. 2014, 23, 419–423. [Google Scholar] [CrossRef]
- Wang, F.Y.; Jiang, X.M.; Lu, M. Expression and clinical significance of serum high-mobility group protein box 1 in children with Henoch-Schönlein purpura. Zhongguo Dang Dai Er Ke Za Zhi 2015, 17, 792–795. [Google Scholar]
- Sato, F.; Maruyama, S.; Hayashi, H.; Sakamoto, I.; Yamada, S.; Uchimura, T.; Morita, Y.; Ito, Y.; Yuzawa, Y.; Maruyama, I.; et al. High mobility group box chromosomal protein 1 in patients with renal diseases. Nephron Clin. Pract. 2008, 108, c194–c201. [Google Scholar] [CrossRef]
- Held, M.; Kozmar, A.; Sestan, M.; Turudic, D.; Kifer, N.; Srsen, S.; Gagro, A.; Frkovic, M.; Jelusic, M. Insight into the Interplay of Gd-IgA1, HMGB1, RAGE and PCDH1 in IgA Vasculitis (IgAV). Int. J. Mol. Sci. 2024, 25, 4383. [Google Scholar] [CrossRef]
- Suzuki, H.; Yasutake, J.; Makita, Y.; Tanbo, Y.; Yamasaki, K.; Sofue, T.; Kano, T.; Suzuki, Y. IgA nephropathy and IgA vasculitis with nephritis have a shared feature involving galactose-deficient IgA1-oriented pathogenesis. Kidney Int. 2018, 93, 700–705. [Google Scholar] [CrossRef]
- Tang, M.; Zhang, X.; Li, X.; Lei, L.; Zhang, H.; Ling, C.; Ni, J.; Lv, J.; Liu, X.; Chen, X. Serum levels of galactose-deficient IgA1 in Chinese children with IgA nephropathy, IgA vasculitis with nephritis, and IgA vasculitis. Clin. Exp. Nephrol. 2021, 25, 37–43. [Google Scholar] [CrossRef]
- Mizerska-Wasiak, M.; Gajewski, Ł.; Cichoń-Kawa, K.; Siejko, A.; Małdyk, J.; Spława-Neyman, A.; Zachwieja, J.; Firszt-Adamczyk, A.; Stankiewicz, R.; Drożyńska-Duklas, M.; et al. Relationship between Gd-IgA1 and TNFR1 in IgA nephropathy and IgA vasculitis nephritis in children—Multicenter study. Cent. Eur. J. Immunol. 2021, 46, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Pillebout, E.; Jamin, A.; Ayari, H.; Housset, P.; Pierre, M.; Sauvaget, V.; Viglietti, D.; Deschenes, G.; Monteiro, R.C.; Berthelot, L. Biomarkers of IgA vasculitis nephritis in children. PLoS ONE 2017, 12, e0188718. [Google Scholar] [CrossRef] [PubMed]
- Mizerska-Wasiak, M.; Gajewski, Ł.; Cichoń-Kawa, K.; Małdyk, J.; Dziedzic-Jankowska, K.; Leszczyńska, B.; Rybi-Szumińska, A.; Wasilewska, A.; Pukajło-Marczyk, A.; Zwolińska, D.; et al. Serum GDIgA1 levels in children with IgA nephropathy and Henoch-Schönlein nephritis. Cent. Eur. J. Immunol. 2018, 43, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Hastings, M.C.; Rizk, D.V.; Kiryluk, K.; Nelson, R.; Zahr, R.S.; Novak, J.; Wyatt, R.J. IgA vasculitis with nephritis: Update of pathogenesis with clinical implications. Pediatr. Nephrol. 2022, 37, 719–733. [Google Scholar] [CrossRef]
- Berthelot, L.; Jamin, A.; Viglietti, D.; Chemouny, J.M.; Ayari, H.; Pierre, M.; Housset, P.; Sauvaget, V.; Hurtado-Nedelec, M.; Vrtovsnik, F.; et al. Value of biomarkers for predicting immunoglobulin A vasculitis nephritis outcome in an adult prospective cohort. Nephrol. Dial. Transplant. 2018, 33, 1579–1590. [Google Scholar] [CrossRef]
- Ge, W.; Wang, H.L.; Sun, R.P. Pentraxin 3 as a novel early biomarker for the prediction of Henoch-Schönlein purpura nephritis in children. Eur. J. Pediatr. 2014, 173, 213–218. [Google Scholar] [CrossRef]
- Zhang, L.; Han, C.; Sun, C.; Meng, H.; Ye, F.; Na, S.; Chen, F.; Zhang, D.; Jin, X. Serum levels of alpha-smooth muscle actin and c-Met as biomarkers of the degree of severity of Henoch-Schonlein purpura nephritis. Transl. Res. 2013, 161, 26–36. [Google Scholar] [CrossRef]
- Wu, J.; He, L.; Bai, L.; Tan, L.; Hu, M. Apolipoprotein M Serum Levels Correlate with IgA Vasculitis and IgA Vasculitis Nephritis. Dis. Markers 2019, 2019, 1825849. [Google Scholar] [CrossRef]
- He, X.; Yin, W.; Ding, Y.; Cui, S.J.; Luan, J.; Zhao, P.; Yue, X.; Yu, C.; Laing, X.; Zhao, Y. Higher Serum Angiotensinogen Is an Indicator of IgA Vasculitis with Nephritis Revealed by Comparative Proteomes Analysis. PLoS ONE 2015, 10, e0130536. [Google Scholar] [CrossRef][Green Version]
- Wu, H.; Wen, Y.; Yue, C.; Li, X.; Gao, R. Serum TNF-α Level Is Associated with Disease Severity in Adult Patients with Immunoglobulin A Vasculitis Nephritis. Dis. Markers 2020, 2020, 5514145. [Google Scholar] [CrossRef]
- Wright, R.D.; Marro, J.; Northey, S.J.; Corkhill, R.; Beresford, M.W.; Oni, L. Urinary complement proteins are increased in children with IgA vasculitis (Henoch-Schönlein purpura) nephritis. Pediatr. Nephrol. 2023, 38, 1491–1498. [Google Scholar] [CrossRef] [PubMed]
- Erol, M.; Yigit, O.; Tasdemir, M.; Bostan Gayret, O.; Buke, O.; Gunes, A.; Hamilcikan, S.; Kasapcopur, O. Potential of Serum and Urinary Matrix Metalloproteinase-9 Levels for the Early Detection of Renal Involvement in Children With Henoch-Schönlein Purpura. Iran. J. Pediatr. 2016, 26, e6129. [Google Scholar] [CrossRef]
- Mao, Y.N.; Liu, W.; Li, Y.G.; Jia, G.C.; Zhang, Z.; Guan, Y.J.; Zhou, X.F.; Liu, Y.F. Urinary angiotensinogen levels in relation to renal involvement of Henoch-Schonlein purpura in children. Nephrology 2012, 17, 53–57. [Google Scholar] [CrossRef]
- Wang, J.; Ying, Q.; Zhong, S.; Chen, Y.; Di, Y.; Dai, X.; Zheng, J.; Shen, M. Elevated urinary monocyte chemoattractant protein-1 levels in children with Henoch-Schonlein purpura nephritis. Pediatr. Neonatol. 2018, 59, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.E.C.; Toner, A.; Wright, R.D.; Oni, L. A systematic review of urine biomarkers in children with IgA vasculitis nephritis. Pediatr. Nephrol. 2021, 36, 3033–3044. [Google Scholar] [CrossRef]
- Frkovic, M.; Turcic, A.; Gagro, A.; Srsen, S.; Frkovic, S.H.; Rogic, D.; Jelusic, M. Erythrocyte Glutathione S-Transferase Activity as a Sensitive Marker of Kidney Function Impairment in Children with IgA Vasculitis. Int. J. Mol. Sci. 2024, 25, 3795. [Google Scholar] [CrossRef]
- Karadağ, Ş.G.; Çakmak, F.; Çil, B.; Tanatar, A.; Sönmez, H.E.; Kıyak, A.; Yavuz, S.; Çakan, M.; Aktay Ayaz, N. The relevance of practical laboratory markers in predicting gastrointestinal and renal involvement in children with Henoch-Schönlein Purpura. Postgrad. Med. 2021, 133, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.H.; Kim, C.J.; Yang, E.M. Neutrophil-to-lymphocyte ratio to predict gastrointestinal bleeding in Henoch: Schönlein purpura. Pediatr. Int. 2018, 60, 791–795. [Google Scholar] [CrossRef]
- Makay, B.; Gücenmez, Ö.A.; Duman, M.; Ünsal, E. The relationship of neutrophil-to-lymphocyte ratio with gastrointestinal bleeding in Henoch-Schonlein purpura. Rheumatol. Int. 2014, 34, 1323–1327. [Google Scholar] [CrossRef]
- Li, B.; Ren, Q.; Ling, J.; Tao, Z.; Yang, X.; Li, Y. Clinical relevance of neutrophil-to-lymphocyte ratio and mean platelet volume in pediatric Henoch-Schonlein Purpura: A meta-analysis. Bioengineered 2021, 12, 286–295. [Google Scholar] [CrossRef]
- Paek, E.Y.; Yi, D.Y.; Kang, B.; Choe, B.H. Fecal calprotectin as a marker of gastrointestinal involvement in pediatric Henoch-Schönlein purpura patients: A retrospective analysis. BMC Pediatr. 2020, 20, 374. [Google Scholar] [CrossRef]
- Teng, X.; Gao, C.; Sun, M.; Wu, J. Clinical significance of fecal calprotectin for the early diagnosis of abdominal type of Henoch-Schonlein purpura in children. Clin. Rheumatol. 2018, 37, 1667–1673. [Google Scholar] [CrossRef]
- Watanabe, H.; Sada, K.; Matsumoto, Y.; Harigai, M.; Amano, K.; Dobashi, H.; Fujimoto, S.; Usui, J.; Yamagata, K.; Atsumi, T.; et al. Association between reappearance of myeloperoxidase-antineutrophil cytoplasmic antibody and relapse in antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheumatol. 2018, 70, 1626–1633. [Google Scholar] [CrossRef] [PubMed]
- Tomasson, G.; Grayson, P.C.; Mahr, A.D.; Lavalley, M.; Merkel, P.A. Value of ANCA measurements during remission to predict a relapse of ANCA-associated vasculitis--a meta-analysis. Rheumatology 2012, 51, 100–109. [Google Scholar] [CrossRef]
- Moon, J.S.; Ahn, S.S.; Park, Y.B.; Lee, S.K.; Lee, S.W. C-Reactive Protein to Serum Albumin Ratio Is an Independent Predictor of All-Cause Mortality in Patients with ANCA-Associated Vasculitis. Yonsei Med. J. 2018, 59, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Scurt, F.G.; Hirschfeld, V.; Ganz, M.; Mertens, P.R.; Chatzikyrkou, C. Low levels of complement factor C3 at diagnosis can predict outcome in antineutrophil antibody associated vasculitis. J. Nephrol. 2023, 36, 2281–2293. [Google Scholar] [CrossRef] [PubMed]
- Go, E.; Aeschlimann, F.A.; Lu, H.; Larry, J.R.; Hebert, D.; Yeung, R.S.M.; Noone, D. Von Willebrand factor antigen as a marker of disease activity in childhood-onset antineutrophil cytoplasmic antibody-associated vasculitis. Rheumatology 2024, 63, SI228–SI232. [Google Scholar] [CrossRef]
- Yoo, J.; Ahn, S.S.; Jung, S.M.; Song, J.J.; Park, Y.B.; Lee, S.W. Delta Neutrophil Index Is Associated with Vasculitis Activity and Risk of Relapse in ANCA-Associated Vasculitis. Yonsei Med. J. 2018, 59, 397–405. [Google Scholar] [CrossRef]
- Ishizaki, J.; Takemori, A.; Suemori, K.; Matsumoto, T.; Akita, Y.; Sada, K.E.; Yuzawa, Y.; Amano, K.; Takasaki, Y.; Harigai, M.; et al. Targeted proteomics reveals promising biomarkers of disease activity and organ involvement in antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Res. Ther. 2017, 19, 218. [Google Scholar] [CrossRef]
- Berti, A.; Warner, R.; Johnson, K.; Cornec, D.; Schroeder, D.; Kabat, B.; Langford, C.A.; Hoffman, G.S.; Fervenza, F.C.; Kallenberg, C.G.M.; et al. Brief Report: Circulating Cytokine Profiles and Antineutrophil Cytoplasmic Antibody Specificity in Patients With Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. Arthritis Rheumatol. 2018, 70, 1114–1121. [Google Scholar] [CrossRef] [PubMed]
- Jonasdottir, A.D.; Antovic, A.; Qureshi, A.R.; Nordin, A.; Malmström, V.; Gunnarsson, I.; Bruchfeld, A. Pentraxin-3—A potential biomarker in ANCA-associated vasculitis. Scand. J. Rheumatol. 2023, 52, 293–301. [Google Scholar] [CrossRef]
- Simon, A.; Subra, J.F.; Guilpain, P.; Jeannin, P.; Pignon, P.; Blanchard, S.; Garo, E.; Jaillon, S.; Chevailler, A.; Renier, G.; et al. Detection of Anti-Pentraxin-3 Autoantibodies in ANCA-Associated Vasculitis. PLoS ONE 2016, 11, e0147091. [Google Scholar] [CrossRef]
- Wang, C.; Gou, S.J.; Chang, D.Y.; Yu, F.; Zhao, M.H.; Chen, M. Association of circulating level of high mobility group box 1 with disease activity in antineutrophil cytoplasmic autoantibody-associated vasculitis. Arthritis Care Res. 2013, 65, 1828–1834. [Google Scholar] [CrossRef]
- Bruchfeld, A.; Wendt, M.; Bratt, J.; Qureshi, A.R.; Chavan, S.; Tracey, K.J.; Palmblad, K.; Gunnarsson, I. High-mobility group box-1 protein (HMGB1) is increased in antineutrophilic cytoplasmatic antibody (ANCA)-associated vasculitis with renal manifestations. Mol. Med. 2011, 17, 29–35. [Google Scholar] [CrossRef]
- de Souza, A.; Westra, J.; Bijzet, J.; Limburg, P.C.; Stegeman, C.A.; Bijl, M.; Kallenberg, C.G. Is serum HMGB1 a biomarker in ANCA-associated vasculitis? Arthritis Res. Ther. 2013, 15, R104. [Google Scholar] [CrossRef] [PubMed]
- Wibisono, D.; Csernok, E.; Lamprecht, P.; Holle, J.U.; Gross, W.L.; Moosig, F. Serum HMGB1 levels are increased in active Wegener’s granulomatosis and differentiate between active forms of ANCA-associated vasculitis. Ann. Rheum. Dis. 2010, 69, 1888–1889. [Google Scholar] [CrossRef] [PubMed]
- Henes, F.O.; Chen, Y.; Bley, T.A.; Fabel, M.; Both, M.; Herrmann, K.; Csernok, E.; Gross, W.L.; Moosig, F. Correlation of serum level of high mobility group box 1 with the burden of granulomatous inflammation in granulomatosis with polyangiitis (Wegener’s). Ann. Rheum. Dis. 2011, 70, 1926–1929. [Google Scholar] [CrossRef]
- Souza, A.W.; de Leeuw, K.; van Timmeren, M.M.; Limburg, P.C.; Stegeman, C.A.; Bijl, M.; Westra, J.; Kallenberg, C.G. Impact of serum high mobility group box 1 and soluble receptor for advanced glycation end-products on subclinical atherosclerosis in patients with granulomatosis with polyangiitis. PLoS ONE 2014, 9, e96067. [Google Scholar] [CrossRef][Green Version]
- Bai, X.; Xu, P.C.; Chen, T.; Zhang, H.M.; Wu, S.J.; Yang, X.; Gao, S.; Jia, J.Y.; Jiang, J.Q.; Yan, T.K. The potential pathogenic roles of S100A8/A9 and S100A12 in patients with MPO-ANCA-positive vasculitis. BMC Immunol. 2022, 23, 42. [Google Scholar] [CrossRef]
- Pepper, R.J.; Draibe, J.B.; Caplin, B.; Fervenza, F.C.; Hoffman, G.S.; Kallenberg, C.G.; Langford, C.A.; Monach, P.A.; Seo, P.; Spiera, R.; et al. Association of Serum Calprotectin (S100A8/A9) Level With Disease Relapse in Proteinase 3-Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. Arthritis Rheumatol. 2017, 69, 185–193. [Google Scholar] [CrossRef]
- Komatsuda, A.; Ohtani, H.; Wakui, H.; Chyzh, K.A.; Hatakeyama, T.; Iwamoto, K.; Maki, N.; Kimura, T.; Hitomi, J.; Sawada, K. Increased serum levels of S100A12 in patients with MPO-ANCA-associated glomerulonephritis. Clin. Nephrol. 2006, 66, 315–321. [Google Scholar] [CrossRef] [PubMed]
- de Souza, A.W.; Abdulahad, W.H.; Sosicka, P.; Bijzet, J.; Limburg, P.C.; Stegeman, C.A.; Bijl, M.; Westra, J.; Kallenberg, C.G. Are urinary levels of high mobility group box 1 markers of active nephritis in anti-neutrophil cytoplasmic antibody-associated vasculitis? Clin. Exp. Immunol. 2014, 178, 270–278. [Google Scholar] [CrossRef]
- Antonelli, A.; Ferri, C.; Fallahi, P.; Ferrari, S.M.; Sebastiani, M.; Ferrari, D.; Giunti, M.; Frascerra, S.; Tolari, S.; Franzoni, F.; et al. High values of CXCL10 serum levels in mixed cryoglobulinemia associated with hepatitis C infection. Am. J. Gastroenterol. 2008, 103, 2488–2494. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, A.; Ferri, C.; Ferrari, S.M.; Ghiri, E.; Marchi, S.; Sebastiani, M.; Fallahi, P. Serum concentrations of interleukin 1beta, CXCL10, and interferon-gamma in mixed cryoglobulinemia associated with hepatitis C infection. J. Rheumatol. 2010, 37, 91–97. [Google Scholar] [CrossRef]
- Ferri, C.; Mascia, M.T. Cryoglobulinemic vasculitis. Curr. Opin. Rheumatol. 2006, 18, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Gallo, A.; Fusconi, M.; Magliulo, G.; Turchetta, R.; Marinelli, C.; Macri, G.F.; De Virgilio, A.; de Vincentiis, M. Cogan’s syndrome: An autoimmune inner ear disease. Autoimmun. Rev. 2013, 12, 396–400. [Google Scholar] [CrossRef]
- Gül, A. Genetics of Behçet’s disease: Lessons learned from genomewide association studies. Curr. Opin. Rheumatol. 2014, 26, 56–63. [Google Scholar] [CrossRef]
- Maldini, C.; Lavalley, M.P.; Cheminant, M.; de Menthon, M.; Mahr, A. Relationships of HLA-B51 or B5 genotype with Behcet’s disease clinical characteristics: Systematic review and meta-analyses of observational studies. Rheumatology 2012, 51, 887–900. [Google Scholar] [CrossRef]
- International Team for the Revision of the International Criteria for Behçet’s Disease (ITR-ICBD). The International Criteria for Behçet’s Disease (ICBD): A collaborative study of 27 countries on the sensitivity and specificity of the new criteria. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 338–347. [CrossRef]
- Pala, E.; Bayraktar, M. Relationship between C-reactive protein/albumin ratio and mucocutaneous symptom frequency and disease severity in Behçet’s disease. Turk. J. Med. Sci. 2024, 54, 384–390. [Google Scholar] [CrossRef]
- Cheng, L.; Li, L.; Liu, C.; Yan, S.; Chen, H.; Li, H.; Zhang, F.; Chen, H.; Li, Y. Variation of red blood cell parameters in Behcet’s disease: Association with disease severity and vascular involvement. Clin. Rheumatol. 2021, 40, 1457–1464. [Google Scholar] [CrossRef]
- Balkarli, A.; Kucuk, A.; Babur, H.; Erbasan, F. Neutrophil/lymphocyte ratio and mean platelet volume in Behçet’s disease. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 3045–3050. [Google Scholar] [CrossRef]
- Arbrile, M.; Radin, M.; Medica, D.; Miraglia, P.; Rilat, L.; Cecchi, I.; Cecchi, I.; Foddai, S.G.; Barinotti, A.; Menegatti, E.; et al. Finding the Needle in the Haystack: Serological and Urinary Biomarkers in Behçet’s Disease: A Systematic Review. Int. J. Mol. Sci. 2023, 24, 3041. [Google Scholar] [CrossRef]
- Ozturk, C.; Balta, S.; Balta, I.; Demirkol, S.; Celik, T.; Turker, T.; Iyisoy, A.; Eksioglu, M. Neutrophil-lymphocyte ratio and carotid-intima media thickness in patients with Behçet disease without cardiovascular involvement. Angiology 2015, 66, 291–296. [Google Scholar] [CrossRef]
- Yolbas, S.; Yildirim, A.; Gozel, N.; Uz, B.; Koca, S.S. Hematological Indices May Be Useful in the Diagnosis of Systemic Lupus Erythematosus and in Determining Disease Activity in Behçet’s Disease. Med. Princ. Pract. 2016, 25, 510–516. [Google Scholar] [CrossRef]
- Vitale, A.; Rigante, D.; Lopalco, G.; Brizi, M.G.; Caso, F.; Franceschini, R.; Denaro, R.; Galeazzi, M.; Punzi, L.; Iannone, F.; et al. Serum amyloid-A in Behçet’s disease. Clin. Rheumatol. 2014, 33, 1165–1167. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lopalco, G.; Lucherini, O.M.; Vitale, A.; Talarico, R.; Lopalco, A.; Galeazzi, M.; Lapadula, G.; Cantarini, L.; Iannone, F. Putative Role of Serum Amyloid-A and Proinflammatory Cytokines as Biomarkers for Behcet’s Disease. Medicine 2015, 94, e1858. [Google Scholar] [CrossRef] [PubMed]
- Sota, J.; Vitale, A.; Rigante, D.; Orlando, I.; Lucherini, O.M.; Simpatico, A.; Lopalco, G.; Franceschini, R.; Galeazzi, M.; Frediani, B.; et al. Correlation of Serum Amyloid-A Levels, Clinical Manifestations, Treatment, and Disease Activity in Patients with Behçet’s Disease. Isr. Med. Assoc. J. 2018, 20, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Lucherini, O.M.; Vitale, A.; Orlando, I.; Sota, J.; Fabiani, C.; Franceschini, R.; Simpatico, A.; Frediani, B.; Galeazzi, M.; Tosi, G.M.; et al. Serum immunoglobulin D levels in patients with Behçet’s disease according to different clinical manifestations. Clin. Exp. Rheumatol. 2018, 36, 110–115. [Google Scholar] [PubMed]
- Mejía, J.C.; Espinosa, G.; Tàssies, D.; Reverter, J.C.; Cervera, R. Endogenous thrombin potential in Behçet’s disease: Relationship with thrombosis and anticoagulant therapy. Clin. Exp. Rheumatol. 2014, 32, S67–S71. [Google Scholar]
- Direskeneli, H.; Keser, G.; D’Cruz, D.; Khamashta, M.A.; Akoğlu, T.; Yazici, H.; Yurdakul, S.; Hamuryudan, V.; Ozgün, S.; Goral, A.J.; et al. Anti-endothelial cell antibodies, endothelial proliferation and von Willebrand factor antigen in Behçet’s disease. Clin. Rheumatol. 1995, 14, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Novak, T.; Hamedi, M.; Bergmeier, L.A.; Fortune, F.; Hagi-Pavli, E. Saliva and Serum Cytokine Profiles During Oral Ulceration in Behçet’s Disease. Front. Immunol. 2021, 12, 724900. [Google Scholar] [CrossRef] [PubMed]
- Hirahara, L.; Kirino, Y.; Soejima, Y.; Iizuka, Y.; Yoshimi, R.; Fujieda, Y.; Atsumi, T.; Tono, T.; Kobayashi, D.; Meguro, A.; et al. Association of high disease activity and serum IL-6 levels with the incidence of inflammatory major organ events in Behçet disease: A prospective registry study. Front. Immunol. 2024, 15, 1354969. [Google Scholar] [CrossRef]
- Gür-Toy, G.; Lenk, N.; Yalcin, B.; Aksaray, S.; Alli, N. Serum interleukin-8 as a serologic marker of activity in Behçet’s disease. Int. J. Dermatol. 2005, 44, 657–660. [Google Scholar] [CrossRef]
- Durmazlar, S.P.; Ulkar, G.B.; Eskioglu, F.; Tatlican, S.; Mert, A.; Akgul, A. Significance of serum interleukin-8 levels in patients with Behcet’s disease: High levels may indicate vascular involvement. Int. J. Dermatol. 2009, 48, 259–264. [Google Scholar] [CrossRef]
- Chekaoui, A.; Lahmar, K.; Belguendouz, H.; Mazari, F.; Terahi, M.; Hakem, D.; Youinou, P.; Touil-Boukoffa, C. Increased IL-1β levels are associated with an imbalance of “oxidant/antioxidant” status during Behçet’s disease. Eur. Cytokine Netw. 2018, 29, 95–102. [Google Scholar] [CrossRef]
- Habibagahi, Z.; Habibagahi, M.; Heidari, M. Raised concentration of soluble form of vascular endothelial cadherin and IL-23 in sera of patients with Behçet’s disease. Mod. Rheumatol. 2010, 20, 154–159. [Google Scholar] [CrossRef]
- de Souza, A.W.; Perazzio, S.F.; de França, N.R.; Andrade, L.E.; Bijl, M.; Westra, J.; Kallenberg, C.G. High mobility group box 1 serum levels are increased in Behçet’s disease, but not associated with disease activity or disease manifestations. Rheumatology 2015, 54, 2151–2155. [Google Scholar] [CrossRef]
- Omma, A.; Sandikci, S.C.; Colak, S.; Tecer, D.; Yucel, C.; Ozbalkan, Z. Serum calprotectin and ischemia modified albumin levels as markers of disease activity in Behçet’s disease. Postepy Dermatol. Alergol. 2018, 35, 609–613. [Google Scholar] [CrossRef]
- Esatoglu, S.N.; Hatemi, I.; Ozguler, Y.; Hatemi, G.; Uzun, H.; Celik, A.F.; Yazici, H. Faecal but not serum calprotectin levels look promising in predicting active disease in Behçet’s syndrome patients with gastrointestinal involvement. Clin. Exp. Rheumatol. 2018, 36, 90–96. [Google Scholar]
- Mumcu, G.; Cimilli, H.; Karacayli, Ü.; Inanc, N.; Türe-Özdemir, F.; Eksioglu-Demiralp, E.; Ergun, T.; Direskeneli, H. Salivary levels of HNP 1-3 are related to oral ulcer activity in Behçet’s disease. Int. J. Dermatol. 2013, 52, 1198–1201. [Google Scholar] [CrossRef]
- Han, E.C.; Cho, S.B.; Ahn, K.J.; Oh, S.H.; Kim, J.; Kim, D.S.; Lee, K.H.; Bang, D. Expression of Pro-inflammatory Protein S100A12 (EN-RAGE) in Behçet’s Disease and Its Association with Disease Activity: A Pilot Study. Ann. Dermatol. 2011, 23, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.K.; Kim, J.; Hwang, J.; Song, J.; Kim, K.H.; Cha, H.S. Urinary Metabolomic Profiling to Identify Potential Biomarkers for the Diagnosis of Behcet’s Disease by Gas Chromatography/Time-of-Flight-Mass Spectrometry. Int. J. Mol. Sci. 2017, 18, 2309. [Google Scholar] [CrossRef]
- Qin, W.; Liang, A.; Han, X.; Zhang, M.; Gao, Y.; Zhao, C. Quantitative urinary proteome analysis reveals potential biomarkers for disease activity of Behcet’s disease uveitis. BMC Ophthalmol. 2024, 24, 277. [Google Scholar] [CrossRef] [PubMed]
- Salvarani, C.; Brown, R.D., Jr.; Calamia, K.T.; Christianson, T.J.; Weigand, S.D.; Miller, D.V.; Giannini, C.; Meschia, J.F.; Huston, J., 3rd; Hunder, G.G. Primary central nervous system vasculitis: Analysis of 101 patients. Ann. Neurol. 2007, 62, 442–451. [Google Scholar] [CrossRef]
- Calabrese, L.H.; Furlan, A.J.; Gragg, L.A.; Ropos, T.J. Primary angiitis of the central nervous system: Diagnostic criteria and clinical approach. Clevel. Clin. J. Med. 1992, 59, 293–306. [Google Scholar] [CrossRef]
- Benseler, S.M.; Silverman, E.; Aviv, R.I.; Schneider, R.; Armstrong, D.; Tyrrell, P.N.; deVeber, G. Primary central nervous system vasculitis in children. Arthritis Rheum. 2006, 54, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Cellucci, T.; Tyrrell, P.N.; Pullenayegum, E.; Benseler, S.M. von Willebrand factor antigen--a possible biomarker of disease activity in childhood central nervous system vasculitis? Rheumatology 2012, 51, 1838–1845. [Google Scholar] [CrossRef][Green Version]
- Guo, T.; Ma, J.; Sun, J.; Xu, W.; Cong, H.; Wei, Y.; Ma, Y.; Dong, Q.; Kou, Y.; Yin, L.; et al. Soluble TREM2 is a potential biomarker for the severity of primary angiitis of the CNS. Front. Immunol. 2022, 13, 963373. [Google Scholar] [CrossRef] [PubMed]
- Deb-Chatterji, M.; Pinnschmidt, H.O.; Duan, Y.; Haeussler, V.; Rissiek, B.; Gerloff, C.; Thomalla, G.; Magnus, T. Circulating Endothelial Cells as Promising Biomarkers in the Differential Diagnosis of Primary Angiitis of the Central Nervous System. Front. Neurol. 2020, 11, 205. [Google Scholar] [CrossRef] [PubMed]
- Thom, V.; Schmid, S.; Gelderblom, M.; Hackbusch, R.; Kolster, M.; Schuster, S.; Thomalla, G.; Keminer, O.; Pleß, O.; Bernreuther, C.; et al. IL-17 production by CSF lymphocytes as a biomarker for cerebral vasculitis. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e214. [Google Scholar] [CrossRef]
- Ruland, T.; Wolbert, J.; Gottschalk, M.G.; König, S.; Schulte-Mecklenbeck, A.; Minnerup, J.; Meuth, S.G.; Groß, C.C.; Wiendl, H.; Meyer Zu Hörste, G. Cerebrospinal Fluid Concentrations of Neuronal Proteins Are Reduced in Primary Angiitis of the Central Nervous System. Front. Neurol. 2018, 9, 407. [Google Scholar] [CrossRef] [PubMed]
- Carmona, F.D.; Mackie, S.L.; Martín, J.E.; Taylor, J.C.; Vaglio, A.; Eyre, S.; Bossini-Castillo, L.; Castañeda, S.; Cid, M.C.; Hernández-Rodríguez, J.; et al. A large-scale genetic analysis reveals a strong contribution of the HLA class II region to giant cell arteritis susceptibility. Am. J. Hum. Genet. 2015, 96, 565–580. [Google Scholar] [CrossRef]
- Saruhan-Direskeneli, G.; Hughes, T.; Aksu, K.; Keser, G.; Coit, P.; Aydin, S.Z.; Alibaz-Oner, F.; Kamalı, S.; Inanc, M.; Carette, S.; et al. Identification of multiple genetic susceptibility loci in Takayasu arteritis. Am. J. Hum. Genet. 2013, 93, 298–305. [Google Scholar] [CrossRef]
- Lyons, P.A.; Rayner, T.F.; Trivedi, S.; Holle, J.U.; Watts, R.A.; Jayne, D.R.; Baslund, B.; Brenchley, P.; Bruchfeld, A.; Chaudhry, A.N.; et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 2012, 367, 214–223. [Google Scholar] [CrossRef]
- Ching, L.L.; Nerurkar, V.R.; Lim, E.; Shohet, R.V.; Melish, M.E.; Bratincsak, A. Elevated Levels of Pen-traxin 3 Correlate With Neutrophilia and Coronary Artery Dilation During Acute Kawasaki Disease. Front. Pediatr. 2020, 8, 295. [Google Scholar] [CrossRef]
- Kitoh, T.; Ohara, T.; Muto, T.; Okumura, A.; Baba, R.; Koizumi, Y.; Yamagishi, Y.; Mikamo, H.; Daigo, K.; Hamakubo, T. Increased Pentraxin 3 Levels Correlate With IVIG Responsiveness and Coronary Artery Aneurysm Formation in Kawasaki Disease. Front. Immunol. 2021, 12, 624802. [Google Scholar] [CrossRef]
- Yazmalar, L.; Batmaz, İ.; Sula, B.; Alpaycı, M.; Aydın, F.; Türkçü, F.; Yıldız, M.; Kaplan, İ.; Bozkurt, M.; Dağlı, A.Z.; et al. Serum levels of alpha-1 acid glycoprotein and pentraxin 3 in patients with Behçet’s disease and relationship with disease activity. Int. J. Dermatol. 2015, 54, e394–e400. [Google Scholar] [CrossRef]
- Kawakami, T.; Takeuchi, S.; Soma, Y. Serum levels of interleukin-6 in patients with cutaneous polyarteritis nodosa. Acta Derm. Venereol. 2012, 92, 322–323. [Google Scholar] [CrossRef]
- Berti, A.; Warner, R.; Johnson, K.; Cornec, D.; Schroeder, D.R.; Kabat, B.F.; Langford, C.A.; Kallenberg, C.G.M.; Seo, P.; Spiera, R.F.; et al. The association of serum interleukin-6 levels with clinical outcomes in antineutrophil cytoplasmic antibody-associated vasculitis. J. Autoimmun. 2019, 105, 102302. [Google Scholar] [CrossRef]
- Adam, B.; Calikoglu, E. Serum interleukin-6, procalcitonin and C-reactive protein levels in subjects with active Behçet’s disease. J. Eur. Acad. Dermatol. Venereol. 2004, 18, 318–320. [Google Scholar] [CrossRef]
- Whitin, J.C.; Yu, T.T.; Ling, X.B.; Kanegaye, J.T.; Burns, J.C.; Cohen, H.J. A Novel Truncated Form of Serum Amyloid A in Kawasaki Disease. PLoS ONE 2016, 11, e0157024. [Google Scholar] [CrossRef]
- Yoon, T.; Ahn, S.S.; Yoo, J.; Song, J.J.; Park, Y.B.; Lee, S.W. Serum Amyloid A Is a Biomarker of Disease Activity and Health-Related Quality-of-Life in Patients with Antineutrophil Cytoplasmic Anti-body-Associated Vasculitis. Dis. Markers 2020, 2020, 8847306. [Google Scholar] [CrossRef] [PubMed]
- Corbera-Bellalta, M.; Farran-Centelles, N.; Visocnik, N.; Prieto-Gonzalez, S.; Hernandez-Rodriguez, J.; Marco-Hernandez, J.; Espigol-Frigolé, G.; Cid, M. Does complement product C5a and its receptor C5a receptor 1 play a role in giant-cell arteritis? Arthritis Rheumatol. 2025, 77, 0896. [Google Scholar]
- Tombetti, E.; Hysa, E.; Mason, J.C.; Cimmino, M.A.; Camellino, D. Blood Biomarkers for Monitoring and Prognosis of Large Vessel Vasculitides. Curr. Rheumatol. Rep. 2021, 23, 17. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Suzuki, K.; Magi, M.; Onishi, S.; Yoshida, H.; Takeshita, M.; Kuramoto, J.; Yazawa, M.; Kato, T.; Shimizu, H.; et al. Trans-omics landscape of systemic vasculitis identified matrix metalloproteinase 12 as a novel signature molecule. Rheumatology 2025, 64, 4766–4775. [Google Scholar] [CrossRef] [PubMed]
- Pay, S.; Abbasov, T.; Erdem, H.; Musabak, U.; Simsek, I.; Pekel, A.; Akdogan, A.; Sengul, A.; Dinc, A. Serum MMP-2 and MMP-9 in patients with Behçet’s disease: Do their higher levels correlate to vasculo-Behçet’s disease associated with aneurysm formation? Clin. Exp. Rheumatol. 2007, 25, S70–S75. [Google Scholar]
- Cheng, X.; Dang, A.; Lv, N.; Zhao, T. Microparticles from Endothelial Cells and Immune Cells in Patients with Takayasu Arteritis. J. Atheroscler. Thromb. 2019, 26, 547–558. [Google Scholar] [CrossRef]
- Jakob, A.; Schachinger, E.; Klau, S.; Lehner, A.; Ulrich, S.; Stiller, B.; Zieger, B. Von Willebrand factor para-meters as potential biomarkers for disease activity and coronary artery lesion in patients with Kawasaki disease. Eur. J. Pediatr. 2020, 179, 377–384. [Google Scholar] [CrossRef] [PubMed]
- De Mattia, D.; Penza, R.; Giordano, P.; Del Vecchio, G.C.; Aceto, G.; Altomare, M.; Schettini, F. von Willebrand factor and factor XIII in children with Henoch-Schonlein purpura. Pediatr. Nephrol. 1995, 9, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Beyan, E.; Sadikoğlu, B.; Ertuğrul, E.; Beyan, C. Von Willebrand factor antigen levels in Behçet disease. Am. J. Hematol. 2005, 79, 70–72. [Google Scholar] [CrossRef]
- Xie, L.Y.; Qiu, X.Y.; Li, Y.N.; Zhang, H.M.; Chen, H.S.; Gu, Q.H.; Yan, T.K.; Jia, J.Y.; Xu, P.C. Serum ferritin is a superior biomarker for evaluating disease activity and kidney injury compared with C-reactive protein in an-ti-neutrophil cytoplasmic antibody-associated vasculitis. Clin. Rheumatol. 2025, 44, 2009–2021. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.