A Novel VPS13A Deletion in VPS13A Disease (Chorea-Acanthocytosis): A Case Report with Brief Literature Summary
Abstract
1. Introduction
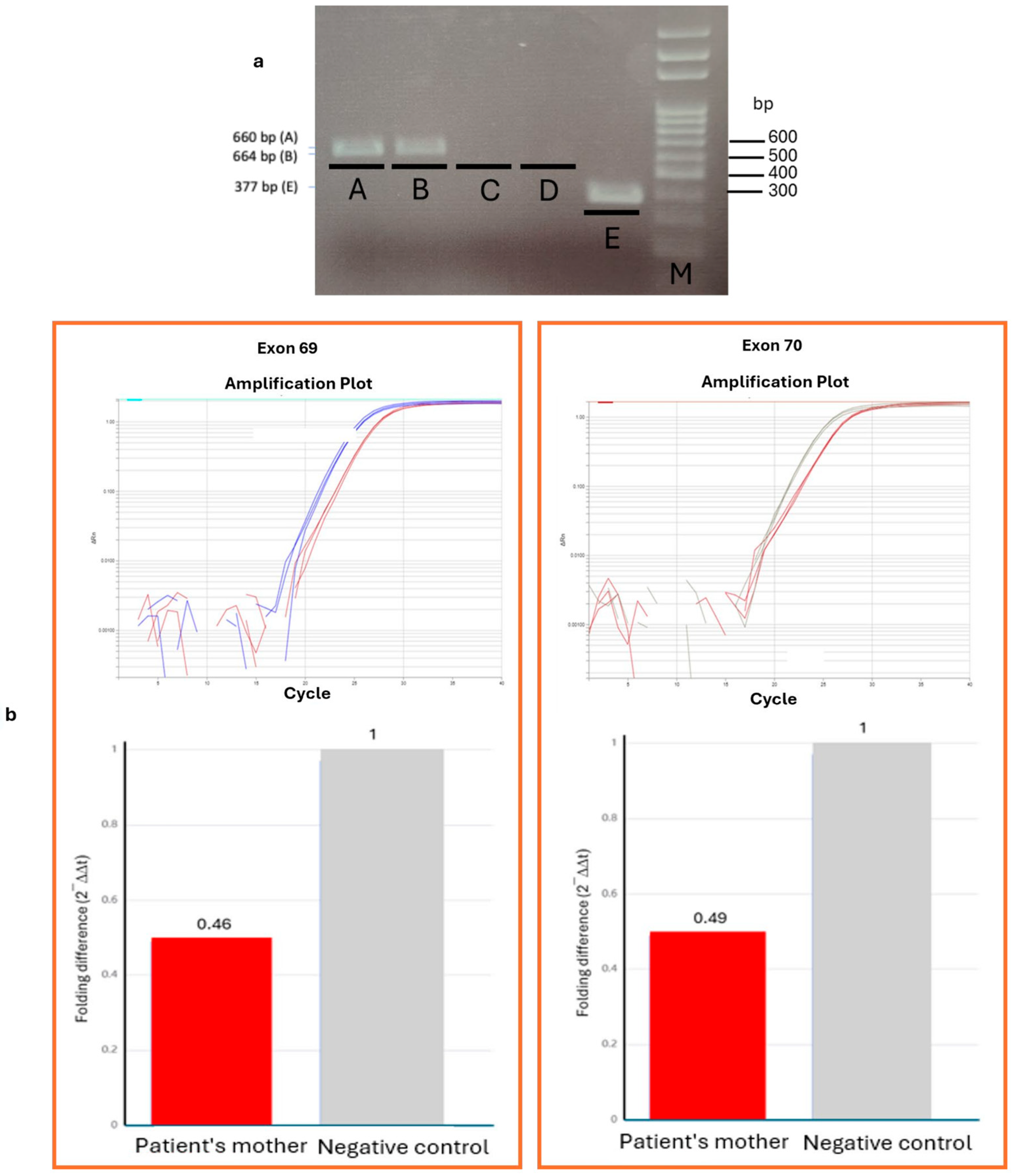
2. Case Description
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACMG | American College of Medical Genetics and Genomics |
| APT1 | Aberrant pollen transcription 1 |
| ATG_C | C-terminal ATG2 domain |
| BLTPs | Bridge-like lipid transfer proteins |
| ChAc | Chorea-acanthocytosis |
| CK | Creatine phosphokinase |
| CNV | Copy number variant |
| CS | Calf serum |
| DMEM | Dulbecco’s modified Eagle’s medium |
| EMG | Electromyography |
| ER | Endoplasmic reticulum |
| FFAT | Two phenylalanines in an acidic tract |
| HGMD | Human Gene Mutation Database |
| IF | Immunofluorescence |
| MCS | Membrane contact sites |
| MTTP | Microsomal triglyceride transfer protein |
| PANK2 | Pantothenate kinase 2 |
| PCR | Polymerase chain reaction |
| PH-like domain | Pleckstrin homology-like domain |
| VAB | VPS13 adaptor binding |
| VAMP | Vesicle-associated membrane protein |
| VAP | VAMP-associated protein |
| VPS13A | Vacuolar protein sorting 13 homolog A |
| WB | Western blot |
| XK | X-linked Kx blood group antigen, Kell and VPS13A binding protein |
References
- Peikert, K.; Dobson-Stone, C.; Rampoldi, L.; Miltenberger-Miltenyi, G.; Neiman, A.; De Camilli, P.; Hermann, A.; Walker, R.H.; Monaco, A.P.; Danek, A. VPS13A Disease. In GeneReviews(®); Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Liu, Y.; Liu, Z.Y.; Wan, X.H.; Guo, Y. Progress in the Diagnosis and Management of Chorea-acanthocytosis. Chin. Med. Sci. J. Chung-Kuo I Hsueh K’o Hsueh Tsa Chih 2018, 33, 53–59. [Google Scholar] [CrossRef]
- Ueno, S.; Maruki, Y.; Nakamura, M.; Tomemori, Y.; Kamae, K.; Tanabe, H.; Yamashita, Y.; Matsuda, S.; Kaneko, S.; Sano, A. The gene encoding a newly discovered protein, chorein, is mutated in chorea-acanthocytosis. Nat. Genet. 2001, 28, 121–122. [Google Scholar] [CrossRef]
- Dobson-Stone, C.; Danek, A.; Rampoldi, L.; Hardie, R.J.; Chalmers, R.M.; Wood, N.W.; Bohlega, S.; Dotti, M.T.; Federico, A.; Shizuka, M.; et al. Mutational spectrum of the CHAC gene in patients with chorea-acanthocytosis. Eur. J. Hum. Genet. 2002, 10, 773–781. [Google Scholar] [CrossRef]
- Peikert, K.; Danek, A.; Hermann, A. Current state of knowledge in Chorea-Acanthocytosis as core Neuroacanthocytosis syndrome. Eur. J. Med. Genet. 2018, 61, 699–705. [Google Scholar] [CrossRef]
- Zamora, J.G.; Rivera, C.; Utsman, R.; Klasser, G.D. Orofacial manifestations of chorea-acanthocytosis: Case presentation and literature review. Quintessence Int. 2022, 53, 270–276. [Google Scholar] [CrossRef]
- Jung, H.H.; Danek, A.; Walker, R.H. Neuroacanthocytosis syndromes. Orphanet J. Rare Dis. 2011, 6, 68. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, J.; Tao, M.; Lv, Y.; Xu, L.; Liang, Z. Two case reports of chorea-acanthocytosis and review of literature. Eur. J. Med. Res. 2022, 27, 22. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Meng, H.; Shafeng, N.; Li, J.; Sun, H.; Yang, X.; Chen, Z.; Hou, S. Exploring the pathophysiological mechanisms and wet biomarkers of VPS13A disease. Front. Neurol. 2024, 15, 1482936. [Google Scholar] [CrossRef] [PubMed]
- Hanna, M.; Guillén-Samander, A.; De Camilli, P. RBG Motif Bridge-Like Lipid Transport Proteins: Structure, Functions, and Open Questions. Annu. Rev. Cell Dev. Biol. 2023, 39, 409–434. [Google Scholar] [CrossRef] [PubMed]
- Swan, L.E. VPS13 and bridge-like lipid transporters, mechanisms, and mysteries. Front. Neurosci. 2025, 19, 1534061. [Google Scholar] [CrossRef]
- Kumar, N.; Leonzino, M.; Hancock-Cerutti, W.; Horenkamp, F.A.; Li, P.; Lees, J.A.; Wheeler, H.; Reinisch, K.M.; De Camilli, P. VPS13A and VPS13C are lipid transport proteins differentially localized at ER contact sites. J. Cell Biol. 2018, 217, 3625–3639. [Google Scholar] [CrossRef]
- Neuman, S.D.; Thakur, R.S.; Gratz, S.J.; O’Connor-Giles, K.M.; Bashirullah, A. Neurodegenerative and Neurodevelopmental Roles for Bulk Lipid Transporters VPS13A and BLTP2. Mov. Disord. Off. J. Mov. Disord. Soc. 2025, 40, 1356–1368. [Google Scholar] [CrossRef] [PubMed]
- Kaminska, J.; Soczewka, P.; Rzepnikowska, W.; Zoladek, T. Yeast as a Model to Find New Drugs and Drug Targets for VPS13-Dependent Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 5106. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.H.; Xiao, B.; Chen, R.K.; Chen, J.Y.; Cai, N.Q.; Cao, C.Y.; Zhan, L.Q. Novel loss-of-function mutations in VPS13A cause chorea-acanthocytosis in two families. Front. Neurol. 2025, 16, 1643889. [Google Scholar] [CrossRef] [PubMed]
- Ghodsinezhad, V.; Ghoreishi, A.; Rohani, M.; Dadfar, M.; Mohammadzadeh, A.; Rostami, A.; Rahimi, H. Identification of four novel mutations in VSP13A in Iranian patients with Chorea-acanthocytosis (ChAc). Mol. Genet. Genomics 2024, 299, 39. [Google Scholar] [CrossRef]
- Kim, A.; Chae, H.Y.; Park, H.S. Compound Heterozygous VPS13A Variants in a Patient with Neuroacanthocytosis: A Case Report and Review of the Literature. Lab. Med. 2022, 53, 433–435. [Google Scholar] [CrossRef]
- Nishida, Y.; Nakamura, M.; Urata, Y.; Kasamo, K.; Hiwatashi, H.; Yokoyama, I.; Mizobuchi, M.; Sakurai, K.; Osaki, Y.; Morita, Y.; et al. Novel pathogenic VPS13A gene mutations in Japanese patients with chorea-acanthocytosis. Neurol. Genet. 2019, 5, e332. [Google Scholar] [CrossRef]
- Shin, H.; Ki, C.S.; Cho, A.R.; Lee, J.I.; Ahn, J.Y.; Lee, J.H.; Cho, J.W. Globus pallidus interna deep brain stimulation improves chorea and functional status in a patient with chorea-acanthocytosis. Stereotact. Funct. Neurosurg. 2012, 90, 273–277. [Google Scholar] [CrossRef]
- Tomiyasu, A.; Nakamura, M.; Ichiba, M.; Ueno, S.; Saiki, S.; Morimoto, M.; Kobal, J.; Kageyama, Y.; Inui, T.; Wakabayashi, K.; et al. Novel pathogenic mutations and copy number variations in the VPS13A gene in patients with chorea-acanthocytosis. Am. J. Med. Genet. Part B Neuropsychiatr Genet. Off. Publ. Int. Soc. Psychiatr. Genet. 2011, 156b, 620–631. [Google Scholar] [CrossRef]
- Nagy, A.; Noyce, A.; Velayos-Baeza, A.; Lees, A.J.; Warner, T.T.; Ling, H. Late Emergence of Parkinsonian Phenotype and Abnormal Dopamine Transporter Scan in Chorea-Acanthocytosis. Mov. Disord. Clin. Pract. 2015, 2, 182–186. [Google Scholar] [CrossRef]
- Rampoldi, L.; Dobson-Stone, C.; Rubio, J.P.; Danek, A.; Chalmers, R.M.; Wood, N.W.; Verellen, C.; Ferrer, X.; Malandrini, A.; Fabrizi, G.M.; et al. A conserved sorting-associated protein is mutant in chorea-acanthocytosis. Nat. Genet. 2001, 28, 119–120. [Google Scholar] [CrossRef]
- Buchberger, A.; Riedel, E.; Hackenberg, M.; Mensch, A.; Beck-Woedl, S.; Park, J.; Haack, T.B.; Haslinger, B.; Kirschke, J.; Prokisch, H.; et al. The Diverse Neuromuscular Spectrum of VPS13A Disease. Ann. Clin. Transl. Neurol. 2025; in press. [Google Scholar] [CrossRef]
- Ouchkat, F.; Regragui, W.; Smaili, I.; Naciri Darai, H.; Bouslam, N.; Rahmani, M.; Melhaoui, A.; Arkha, Y.; El Fahime, E.; Bouhouche, A. Novel pathogenic VPS13A mutation in Moroccan family with Choreoacanthocytosis: A case report. BMC Med. Genet. 2020, 21, 47. [Google Scholar] [CrossRef]
- Connolly, B.S.; Hazrati, L.N.; Lang, A.E. Neuropathological findings in chorea-acanthocytosis: New insights into mechanisms underlying parkinsonism and seizures. Acta Neuropathol. 2014, 127, 613–615. [Google Scholar] [CrossRef]
- Chaudhari, S.; Ware, A.P.; Jasti, D.B.; Gorthi, S.P.; Acharya, L.P.; Bhat, M.; Mallya, S.; Satyamoorthy, K. Exome sequencing of choreoacanthocytosis reveals novel mutations in VPS13A and co-mutation in modifier gene(s). Mol. Genet. Genomics 2023, 298, 965–976. [Google Scholar] [CrossRef]
- Vaisfeld, A.; Bruno, G.; Petracca, M.; Bentivoglio, A.R.; Servidei, S.; Vita, M.G.; Bove, F.; Straccia, G.; Dato, C.; Di Iorio, G.; et al. Neuroacanthocytosis Syndromes in an Italian Cohort: Clinical Spectrum, High Genetic Variability and Muscle Involvement. Genes 2021, 12, 344. [Google Scholar] [CrossRef] [PubMed]
- Benninger, F.; Afawi, Z.; Korczyn, A.D.; Oliver, K.L.; Pendziwiat, M.; Nakamura, M.; Sano, A.; Helbig, I.; Berkovic, S.F.; Blatt, I. Seizures as presenting and prominent symptom in chorea-acanthocytosis with c.2343del VPS13A gene mutation. Epilepsia 2016, 57, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, P.; Wang, L.; Zhang, Y. Novel heterozygous VPS13A pathogenic variants in chorea-neuroacanthocytosis: A case report. BMC Neurol. 2023, 23, 350. [Google Scholar] [CrossRef] [PubMed]
- Miki, Y.; Nishie, M.; Ichiba, M.; Nakamura, M.; Mori, F.; Ogawa, M.; Kaimori, M.; Sano, A.; Wakabayashi, K. Chorea-acanthocytosis with upper motor neuron degeneration and 3419_3420 delCA and 3970_3973 delAGTC VPS13A mutations. Acta Neuropathol. 2010, 119, 271–273. [Google Scholar] [CrossRef]
- Ruiz-Sandoval, J.L.; García-Navarro, V.; Chiquete, E.; Dobson-Stone, C.; Monaco, A.P.; Alvarez-Palazuelos, L.E.; Padilla-Martínez, J.J.; Barrera-Chairez, E.; Rodríguez-Figueroa, E.I.; Pérez-García, G. Choreoacanthocytosis in a Mexican family. Arch. Neurol. 2007, 64, 1661–1664. [Google Scholar] [CrossRef][Green Version]
- Hadzsiev, K.; Szőts, M.; Fekete, A.; Balikó, L.; Boycott, K.; Nagy, F.; Melegh, B. Neuroacanthocytosis diagnosis with new generation whole exome sequencing. Orvosi Hetil. 2017, 158, 1681–1684. [Google Scholar] [CrossRef]
- Dobson-Stone, C.; Velayos-Baeza, A.; Jansen, A.; Andermann, F.; Dubeau, F.; Robert, F.; Summers, A.; Lang, A.E.; Chouinard, S.; Danek, A.; et al. Identification of a VPS13A founder mutation in French Canadian families with chorea-acanthocytosis. Neurogenetics 2005, 6, 151–158. [Google Scholar] [CrossRef]
- Ogawa, I.; Saigoh, K.; Hirano, M.; Mtsui, Y.; Sugioka, K.; Takahashi, J.; Shimomura, Y.; Tani, Y.; Nakamura, Y.; Kusunoki, S. Ophthalmologic involvement in Japanese siblings with chorea-acanthocytosis caused by a novel chorein mutation. Park. Relat. Disord. 2013, 19, 913–915. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, Y.; Matsumoto, K.; Ichikawa, K.; Ueno, S.; Ichiba, M.; Nakamura, M.; Sano, A. A new phenotype of chorea-acanthocytosis with dilated cardiomyopathy and myopathy. Mov. Disord. Off. J. Mov. Disord. Soc. 2007, 22, 1669–1670. [Google Scholar] [CrossRef] [PubMed]
- Merwick, Á.; Mok, T.; McNamara, B.; Parfrey, N.A.; Moore, H.; Sweeney, B.J.; Hand, C.K.; Ryan, A.M. Phenotypic Variation in a Caucasian Kindred with Chorea-Acanthocytosis. Mov. Disord. Clin. Pract. 2015, 2, 86–89. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. Off. J. Am. Coll. Med. Genet. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Lo Bello, M.; Di Fini, F.; Notaro, A.; Spataro, R.; Conforti, F.L.; La Bella, V. ALS-Related Mutant FUS Protein Is Mislocalized to Cytoplasm and Is Recruited into Stress Granules of Fibroblasts from Asymptomatic FUS P525L Mutation Carriers. Neurodegener. Dis. 2017, 17, 292–303. [Google Scholar] [CrossRef]
- La Bella, V.; Cisterni, C.; Salaün, D.; Pettmann, B. Survival motor neuron (SMN) protein in rat is expressed as different molecular forms and is developmentally regulated. Eur. J. Neurosci. 1998, 10, 2913–2923. [Google Scholar] [CrossRef]
- Park, J.S.; Hu, Y.; Hollingsworth, N.M.; Miltenberger-Miltenyi, G.; Neiman, A.M. Interaction between VPS13A and the XK scramblase is important for VPS13A function in humans. J. Cell Sci. 2022, 135, jcs260227. [Google Scholar] [CrossRef]
- Guillén-Samander, A.; Wu, Y.; Pineda, S.S.; García, F.J.; Eisen, J.N.; Leonzino, M.; Ugur, B.; Kellis, M.; Heiman, M.; De Camilli, P. A partnership between the lipid scramblase XK and the lipid transfer protein VPS13A at the plasma membrane. Proc. Natl. Acad. Sci. USA 2022, 119, e2205425119. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrone, B.; Mosca, V.; Pecoraro, M.; Ruffo, P.; Giudice, E.D.; Leon, A.; Maino, M.; La Bella, V.; Spataro, R.; Conforti, F.L. A Novel VPS13A Deletion in VPS13A Disease (Chorea-Acanthocytosis): A Case Report with Brief Literature Summary. Int. J. Mol. Sci. 2025, 26, 11521. https://doi.org/10.3390/ijms262311521
Perrone B, Mosca V, Pecoraro M, Ruffo P, Giudice ED, Leon A, Maino M, La Bella V, Spataro R, Conforti FL. A Novel VPS13A Deletion in VPS13A Disease (Chorea-Acanthocytosis): A Case Report with Brief Literature Summary. International Journal of Molecular Sciences. 2025; 26(23):11521. https://doi.org/10.3390/ijms262311521
Chicago/Turabian StylePerrone, Benedetta, Viviana Mosca, Martina Pecoraro, Paola Ruffo, Elda Del Giudice, Alberta Leon, Martina Maino, Vincenzo La Bella, Rossella Spataro, and Francesca Luisa Conforti. 2025. "A Novel VPS13A Deletion in VPS13A Disease (Chorea-Acanthocytosis): A Case Report with Brief Literature Summary" International Journal of Molecular Sciences 26, no. 23: 11521. https://doi.org/10.3390/ijms262311521
APA StylePerrone, B., Mosca, V., Pecoraro, M., Ruffo, P., Giudice, E. D., Leon, A., Maino, M., La Bella, V., Spataro, R., & Conforti, F. L. (2025). A Novel VPS13A Deletion in VPS13A Disease (Chorea-Acanthocytosis): A Case Report with Brief Literature Summary. International Journal of Molecular Sciences, 26(23), 11521. https://doi.org/10.3390/ijms262311521










