Zileuton Attenuates Acute Kidney Injury in Glycerol-Induced Rhabdomyolysis by Regulating Myeloid-Derived Suppressor Cells in Mice
Abstract
1. Introduction
2. Results
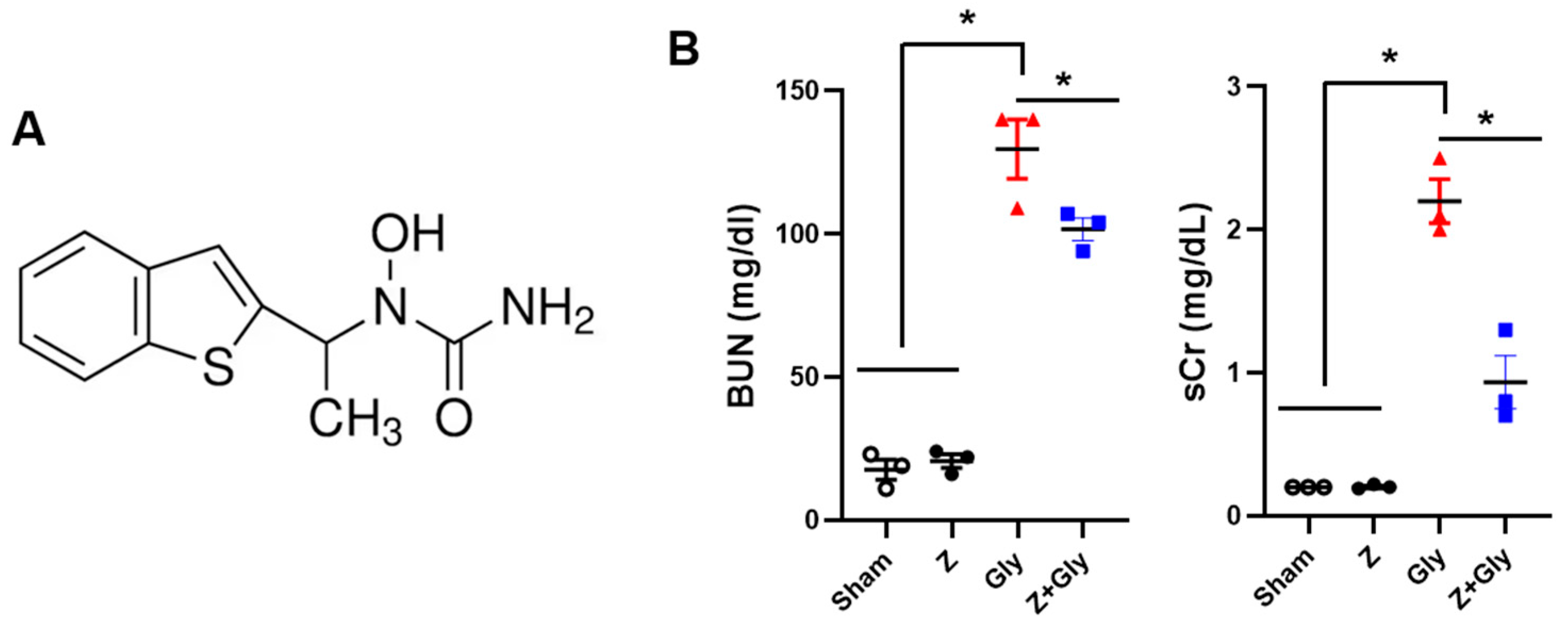
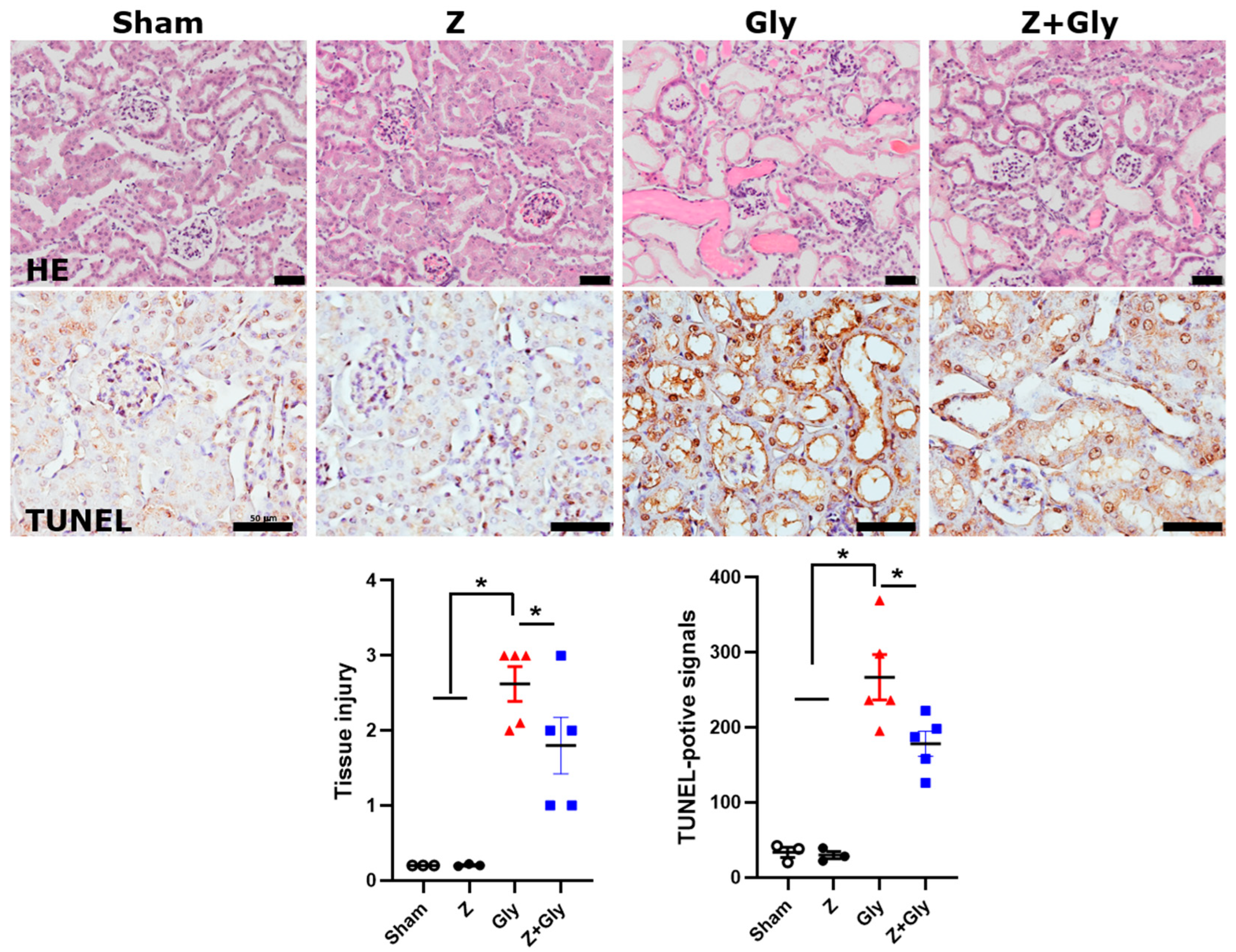
2.1. Zileuton Ameliorates Gly-Induced Renal Dysfunction and Tissue Damage
2.2. Zileuton Reduces Macrophage Infiltration
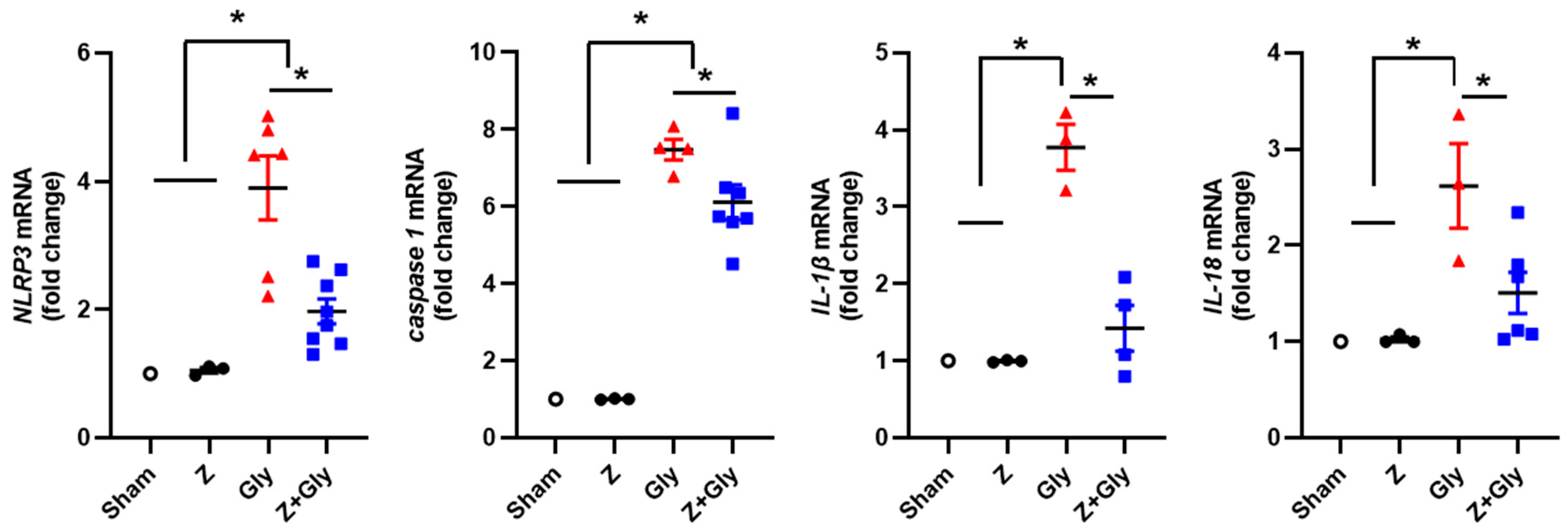
2.3. Zileuton Decreases the Activation of Inflammasomes
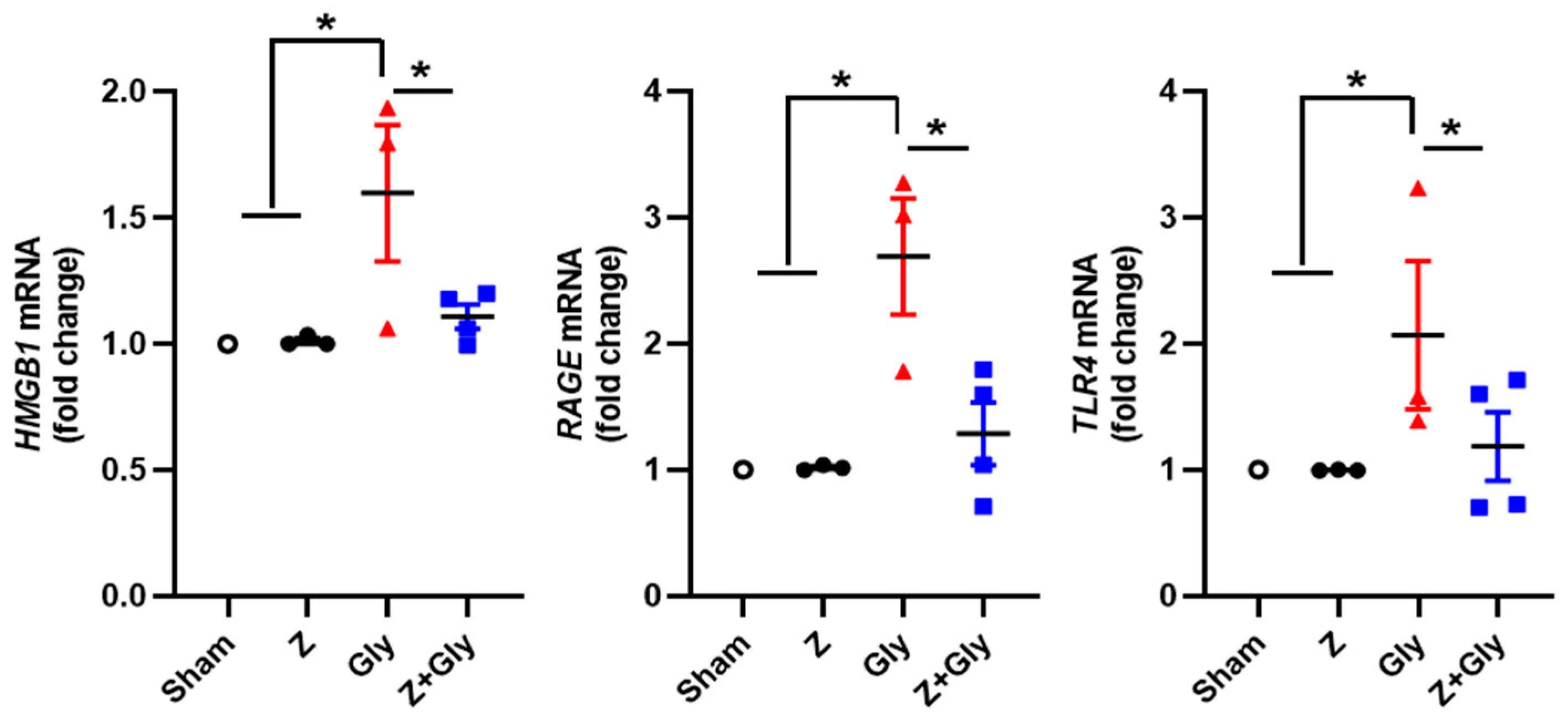
2.4. Zileuton Decreases the Activation of the HMGB1 Pathway
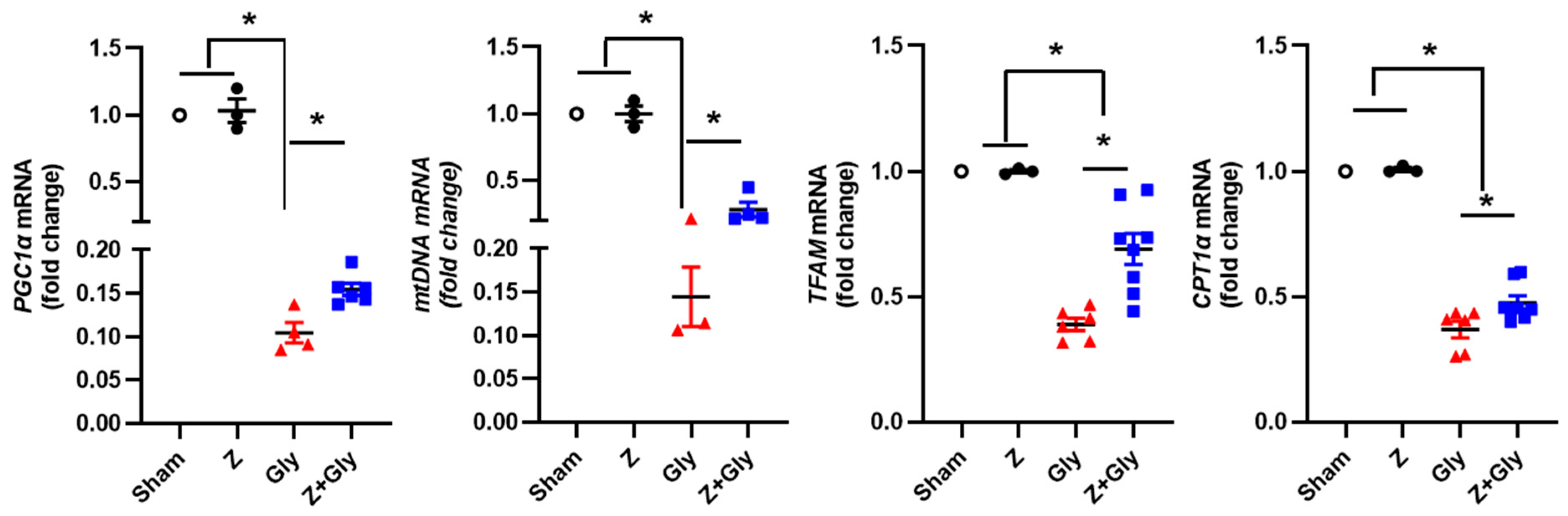
2.5. Effects of Zileuton on Renal Mitochondrial Quality Control
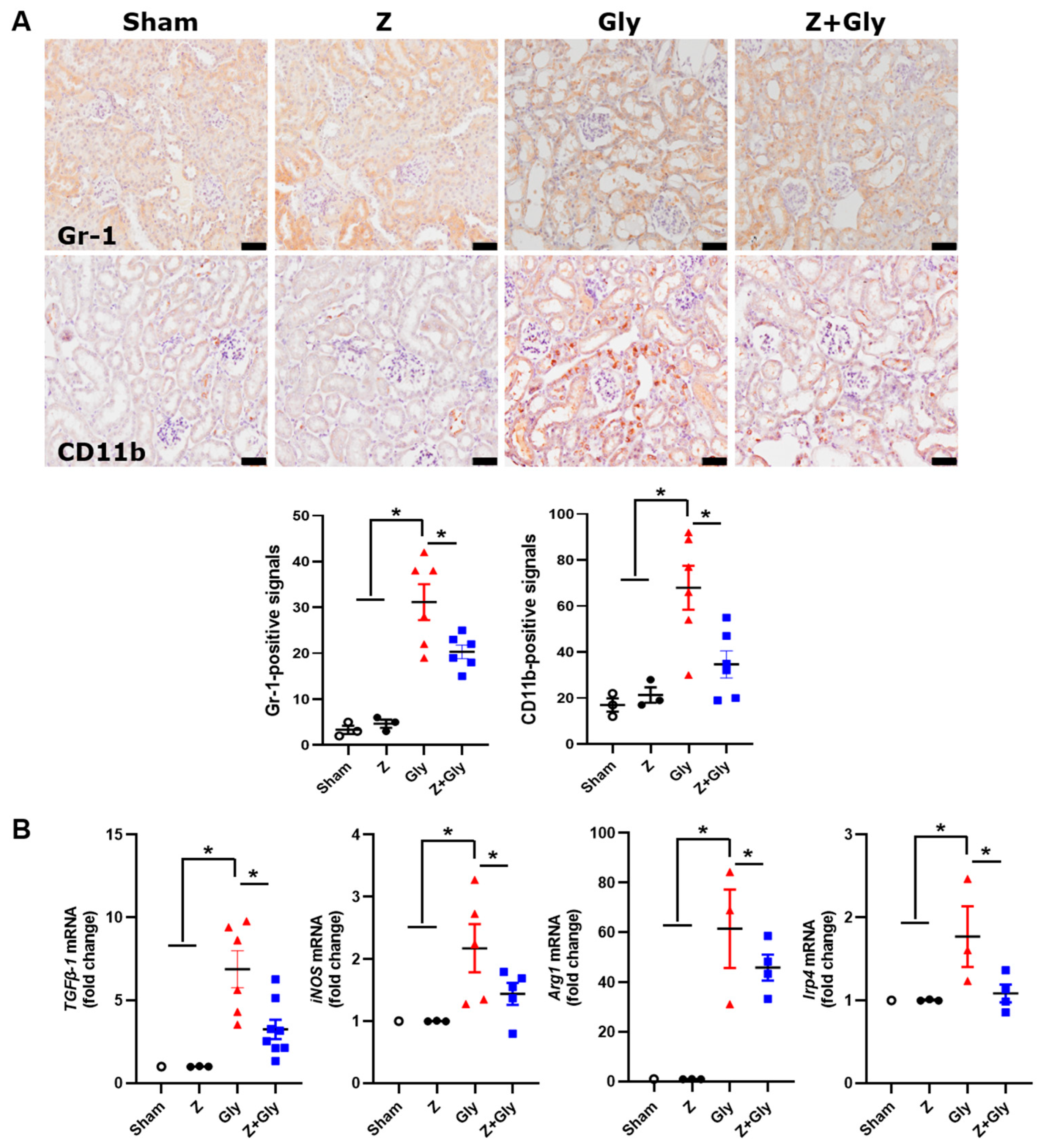
2.6. Zileuton Reduces MDSC Infiltration
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Animal Housing
4.3. Animal Model and Experimental Design
4.4. Histological Analysis
4.5. Renal Function Assessment
4.6. TUNEL Assay
4.7. Quantitative Real-Time PCR (qPCR)
4.8. Western Blot Analysis
4.9. Immunohistochemistry
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bagley, W.H.; Yang, H.; Shah, K.H. Rhabdomyolysis. Intern. Emerg. Med. 2007, 2, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Bosch, X.; Poch, E.; Grau, J.M. Rhabdomyolysis and acute kidney injury. N. Engl. J. Med. 2009, 361, 62–72. [Google Scholar] [CrossRef]
- Chatzizisis, Y.S.; Misirli, G.; Hatzitolios, A.I.; Giannoglou, G.D. The syndrome of rhabdomyolysis: Complications and treatment. Eur. J. Intern. Med. 2008, 19, 568–574. [Google Scholar] [CrossRef]
- Chander, V.; Chopra, K. Molsidomine, a nitric oxide donor and l-arginine protects against rhabdomyolysis-induced myoglobinuric acute renal failure. Biochim. Biophys. Acta 2005, 1723, 208–214. [Google Scholar] [CrossRef]
- Melli, G.; Chaudhry, V.; Cornblath, D.R. Rhabdomyolysis: An evaluation of 475 hospitalized patients. Medicine 2005, 84, 377–385. [Google Scholar] [CrossRef]
- Polderman, K.H. Acute renal failure and rhabdomyolysis. Int. J. Artif. Organs 2004, 27, 1030–1033. [Google Scholar] [CrossRef]
- Atkins, R.C. Macrophages in renal injury. Am. J. Kidney Dis. 1998, 31, xlv–xlvii. [Google Scholar] [CrossRef]
- Homsi, E.; Janino, P.; de Faria, J. Role of caspases on cell death, inflammation, and cell cycle in glycerol-induced acute renal failure. Kidney Int. 2006, 69, 1385–1392. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, D.-W.; Jung, M.H.; Cho, H.-S.; Jeon, D.-H.; Chang, S.-H.; Park, D.J. Macrophage depletion ameliorates glycerol-induced acute kidney injury in mice. Nephron Exp. Nephrol. 2014, 128, 21–29. [Google Scholar] [CrossRef]
- Komada, T.; Usui, F.; Kawashima, A.; Kimura, H.; Karasawa, T.; Inoue, Y.; Kobayashi, M.; Mizushina, Y.; Kasahara, T.; Taniguchi, S.; et al. Role of NLRP3 Inflammasomes for Rhabdomyolysis-induced Acute Kidney Injury. Sci. Rep. 2015, 5, 10901. [Google Scholar] [CrossRef] [PubMed]
- Belliere, J.; Casemayou, A.; Ducasse, L.; Zakaroff-Girard, A.; Martins, F.; Iacovoni, J.S.; Guilbeau-Frugier, C.; Buffin-Meyer, B.; Pipy, B.; Chauveau, D.; et al. Specific Macrophage Subtypes Influence the Progression of Rhabdomyolysis-Induced Kidney Injury. J. Am. Soc. Nephrol. 2015, 26, 1363–1377. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Bronte, V.; Chen, S.-H.; Colombo, M.P.; Ochoa, A.; Ostrand-Rosenberg, S.; Schreiber, H. The terminology issue for myeloid derived suppressor cells. Cancer Res. 2007, 67, 425; author reply 426. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009, 9, 162–174. [Google Scholar] [CrossRef]
- Cripps, J.G.; Gorham, J.D. MDSC in autoimmunity. Int. Immunopharmacol. 2011, 11, 789–793. [Google Scholar] [CrossRef] [PubMed]
- LaFace, D.; Talmadge, J. Meeting report: Regulatory myeloid cells. Int. Immunopharmacol. 2011, 11, 780–782. [Google Scholar] [CrossRef]
- Golden, P.M.; Henderson, W.R. Leukotrienes. N. Engl. J. Med. 2007, 357, 1841–1854. [Google Scholar] [CrossRef] [PubMed]
- McGill, K.A.; Busse, W.W. Zileuton. Lancet 1996, 348, 519–524. [Google Scholar] [CrossRef]
- Helmy, M.M.; Hashim, A.A.; Mouneir, S.M. Zileuton alleviates acute cisplatin nephrotoxicity: Inhibition of lipoxygenase pathway favorably modulates the renal oxidative/inflammatory/caspase-3 axis. Prostaglandins Other Lipid Mediat. 2018, 135, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sultan, A.M.; Sahib, H.H.; Saheb, H.A.; Mohammad, B.I. Comparative protective effect of zileuton and MK-886 against acute kidney injury induced by doxorubicin. Prensa Med. Argent. 2019, 105, 155–159. [Google Scholar] [CrossRef]
- Hudson, C.S.; Smith, J.E.; Eales, B.M.; Kajiji, S.; Liu, X.; Truong, L.D.; Tam, V.H. Zileuton ameliorates aminoglycoside and polymyxin-associated acute kidney injury in an animal model. J. Antimicrob. Chemother. 2023, 78, 2435–2441. [Google Scholar] [CrossRef]
- Ha, M.H.; An, H.J.; Sung, M.J.; Jeong, H.; Baek, J.; Lee, S.Y.; Lee, Y.H. 5-lipoxygenase inhibition ameliorates diabetic kidney disease by attenuating renal tubular epithelial cell ferroptosis. J. Am. Soc. Nephrol. 2024, 35, SA-PO274. [Google Scholar] [CrossRef]
- Jung, K.H.; Kim, S.E.; Go, H.G.; Lee, Y.J.; Park, M.S.; Ko, S.; Han, B.S.; Yoon, Y.-C.; Cho, Y.J.; Lee, P.; et al. Synergistic Renoprotective Effect of Melatonin and Zileuton by Inhibition of Ferroptosis via the AKT/mTOR/NRF2 Signaling in Kidney Injury and Fibrosis. Biomol. Ther. 2023, 31, 599–610. [Google Scholar] [CrossRef]
- Matsumoto, M.; Tanaka, T.; Kaisho, T.; Sanjo, H.; Copeland, N.G.; Gilbert, D.J.; Jenkins, N.A.; Akira, S. A novel LPS-inducible C-type lectin Is a transcriptional target of NF-IL6 in macrophages. J. Immunol. 1999, 163, 5039–5048. [Google Scholar] [CrossRef]
- Lv, L.L.; Tang, P.M.-K.; Li, C.J.; You, Y.K.; Li, J.; Huang, X.-R.; Ni, J.; Feng, M.; Liu, B.C.; Lan, H.-Y. The pattern recognition receptor, Mincle, is essential for maintaining the M1 macrophage phenotype is acute renal inflammation. Kidney Int. 2017, 91, 587–602. [Google Scholar] [CrossRef]
- Lv, L.-L.; Wang, C.; Li, Z.-L.; Cao, J.-Y.; Zhong, X.; Feng, Y.; Chen, J.; Tang, T.-T.; Ni, H.-F.; Wu, Q.-L.; et al. SAP130 released by damaged tubule drives necroinflammation via miRNA-219c/Mincle signaling in acute kidney injury. Cell Death Dis. 2021, 12, 866. [Google Scholar] [CrossRef]
- Chen, R.; Kang, R.; Tang, D. The mechanism of HMGB1 secretion and release. Exp. Mol. Med. 2022, 54, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Qin, Y.; Liu, B.; Gao, M.; Li, A.; Li, X.; Gong, G. PGC-1α-mediated mitochondrial quality control: Molecular mechanisms and implications for heart failure. Front. Cell Dev. Biol. 2022, 10, 871357. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.K.; Sung, S.A.; Cho, W.Y.; Go, K.J.; Kim, H.K. Macrophages contribute to the initiation of ischemic acute renal failure in rats. Nephrol. Dial. Transplant. 2006, 21, 1231–1239. [Google Scholar] [CrossRef]
- Ricardo, S.D.; van Goor, H.; Eddy, A.A. Macrophage diversity in renal injury and repair. J. Clin. Investig. 2008, 118, 3522–3530. [Google Scholar] [CrossRef]
- Ferenbach, D.A.; Sheldrake, T.A.; Dhaliwal, K.; Kipari, T.M.; Marson, L.P.; Kluth, D.C.; Hughes, J. Macrophage/monocyte depletion by clodronate, but not diphtheria toxin, improves renal ischemia/reperfusion injury in mice. Kidney Int. 2012, 82, 928–933. [Google Scholar] [CrossRef]
- Richardson, M.B.; Williams, S.J. MCL and mincle: C-type lectin receptors that sense damaged self and pathogen-associated molecular patterns. Front. Immunol. 2014, 5, 288. [Google Scholar] [CrossRef]
- Yamasaki, S.; Ishikawa, E.; Sakuma, M.; Hara, H.; Ogata, K.; Saito, T. Mincle is an ITAM-coupled activating receptor that senses damaged cells. Nat. Immunol. 2008, 9, 1179–1188. [Google Scholar] [CrossRef]
- Shigeoka, A.A.; Mueller, J.L.; Kambo, A.; Mathison, J.C.; King, A.J.; Hall, W.F.; da Silva Correia, J.; Ulevitch, R.J.; Hoffman, H.M.; McKay, D.B. An inflammasome-independent role for epithelial-expressed NLRP3 in renal ischemia-reperfusion injury. J. Immunol. 2010, 185, 6277–6285. [Google Scholar] [CrossRef]
- Shen, J.; Wang, L.; Jiang, N.; Mou, S.; Zhang, M.; Gu, L.; Shao, X.; Wang, Q.; Qi, C.; Li, S.; et al. NLRP3 inflammasome mediates contrast media-induced acute kidney injury by regulating cell apoptosis. Sci. Rep. 2016, 6, 34682. [Google Scholar] [CrossRef]
- Gong, W.; Mao, S.; Yu, J.; Song, J.; Jia, Z.; Huang, S.; Zhang, A. NLRP3 deletion protects against renal fibrosis and attenuates mitochondrial abnormality in mouse with 5/6 nephrectomy. Am. J. Physiol. Renal Physiol. 2016, 310, F1081–F1088. [Google Scholar] [CrossRef] [PubMed]
- Ludwig-Portugall, I.; Bartok, E.; Dhana, E.; Evers, B.D.; Primiano, M.J.; Hall, J.P.; Franklin, B.S.; Knolle, P.A.; Hornung, V.; Hartmann, G.; et al. An NLRP3-specific inflammasome inhibitor attenuates crystal-induced kidney fibrosis in mice. Kidney Int. 2016, 90, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, K.; Bock, F.; Dong, W.; Wang, H.; Kopf, S.; Kohli, S.; Al-Dabet, M.M.; Ranjan, S.; Wolter, J.; Wacker, C.; et al. Nlrp3-inflammasome activation in non-myeloid-derived cells aggravates diabetic nephropathy. Kidney Int. 2015, 87, 74–84. [Google Scholar] [CrossRef]
- Homsi, E.; Ribeiro-Alves, M.A.; Lopes de Faria, J.B.; Dias, E.P. Interleukin-6 stimulates tubular regeneration in rats with glycerol-induced acute renal failure. Nephron 2002, 92, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Song, S.J.; Kim, S.-M.; Lee, S.-H.; Moon, J.-Y.; Hwang, H.S.; Kim, J.S.; Park, S.-H.; Jeong, K.H.; Kim, Y.G. Rhabdomyolysis-induced AKI was ameliorated in NLRP3 KO mice via alleviation of mitochondrial lipid peroxidation in renal tubular cells. Int. J. Mol. Sci. 2020, 21, 8564. [Google Scholar] [CrossRef]
- Pellegrini, L.; Foglio, E.; Pontemezzo, E.; Germani, A.; Russo, M.A.; Limana, F. HMGB1 and repair: Focus on the heart. Pharmacol. Ther. 2019, 196, 160–182. [Google Scholar] [CrossRef]
- Chen, R.; Huang, Y.; Quan, J.; Liu, J.; Wang, H.; Billiar, T.R.; Lotze, M.T.; Zeh, H.J.; Kang, R.; Tang, D. HMGB1 as a potential biomarker and therapeutic target for severe COVID-19. Heliyon 2020, 6, e05672. [Google Scholar] [CrossRef] [PubMed]
- Lamkanfi, M.; Sarkar, A.; Walle, L.V.; Vitari, A.C.; Amer, A.O.; Wewers, M.D.; Tracey, K.J.; Kanneganti, T.-D.; Dixit, V.M. Inflammasome-dependent release of the alarmin HMGB1 in endotoxemia. J. Immunol. 2010, 185, 4385–4392. [Google Scholar] [CrossRef]
- Barlan, A.U.; Griffin, T.M.; Mcguire, K.A.; Wiethoff, C.M. Adenovirus membrane penetration activates the NLRP3 inflammasome. J. Virol. 2011, 85, 146–155. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.S.; Jung, M.H.; Yeo, H.D.; Kim, H.-J.; Yang, J.I.; Roh, G.S.; Chang, S.H.; Park, D.J. N-acetylcysteine attenuates glycerol-induced acute kidney injury by regulating MAPKs and Bcl-2 family proteins. Nephrol. Dial. Transplant. 2010, 25, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Panizo, N.; Rubio-Navarro, A.; Amaro-Villalobos, J.M.; Egido, J.; Moreno, J.A. Molecular mechanisms and novel therapeutic approaches to rhabdomyolysis-induced acute kidney injury. Kidney Blood Press. Res. 2015, 40, 520–532. [Google Scholar] [CrossRef]
- Ulusoy, S.; Ozkan, G.; Alkanat, M.; Mungan, S.; Yuluğ, E.; Orem, A. Perspective on rhabdomyolysis-induced acute kidney injury and new treatment options. Am. J. Nephrol. 2013, 38, 368–378. [Google Scholar] [CrossRef]
- Kim, S.-M.; Kim, Y.G.; Kim, D.-J.; Park, S.H.; Jeong, K.-H.; Lee, Y.H.; Lim, S.J.; Lee, S.-H.; Moon, J.-Y. Inflammasome-Independent Role of NLRP3 Mediates Mitochondrial Regulation in Renal Injury. Front. Immunol. 2018, 9, 2563. [Google Scholar] [CrossRef]
- Chung, H.; Vilaysane, A.; Lau, A.; Stahl, M.; Morampudi, V.; Bondzi-Simpson, A.; Platnich, J.M.; Bracey, N.A.; French, M.-C.; Beck, P.L.; et al. NLRP3 regulates a non-canonical platform for caspase-8 activation during epithelial cell apoptosis. Cell Death Differ. 2016, 23, 1331–1346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, S.; Li, J.; Zhang, W.; Zheng, L.; Yang, C.; Zhu, T.; Rong, R. The mTOR signal regulates myeloid-suppressor cells differentiation and immunosuppressive function in acute kidney injury. Cell Death Dis. 2017, 8, e2695. [Google Scholar] [CrossRef]
- Qiu, Y.; Cao, Y.; Tu, G.; Li, J.; Su, Y.; Fang, F.; Zhang, X.; Cang, J.; Rong, R.; Luo, Z. Myeloid-derived suppressor cells alleviate renal fibrosis progression via regulation of CCL5-CCR5 axis. Front Immunol 2021, 12, 698894. [Google Scholar] [CrossRef]
- Hsieh, C.-C.; Chang, C.-C.; Hsu, Y.-C.; Lin, C.-L. Immune modulation by myeloid-derived suppressor cells in diabetic kidney disease. Int. J. Mol. Sci. 2022, 23, 13263. [Google Scholar] [CrossRef]
- Li, D.; Shi, G.; Wang, J.; Zhang, D.; Pan, Y.; Dou, H.; Hou, Y. Baicalein ameliorates pristane-induced lupus nephritis via activating Nrf2/HO-1 in myeloid-derived suppressor cells. Arthritis Res. Ther. 2019, 21, 105. [Google Scholar] [CrossRef]
- Popov, L.D. Mitochondrial Biogenesis: An Update. J. Cell Mol. Med. 2020, 24, 4892–4899. [Google Scholar] [CrossRef]
- Li, P.A.; Hou, X.; Hao, S. Mitochondrial Biogenesis in Neurodegeneration. J. Neurosci. Res. 2017, 95, 2025–2029. [Google Scholar] [CrossRef] [PubMed]
- Uittenbogaard, M.; Chiaramello, A. Mitochondrial Biogenesis: A Therapeutic Target for Neurodevelopmental Disorders and Neurodegenerative Diseases. Curr. Pharm. Des. 2014, 20, 5574–5593. [Google Scholar] [CrossRef]
- Kelly, D.P.; Scarpulla, R.C. Transcriptional Regulatory Circuits Controlling Mitochondrial Biogenesis and Function. Genes Dev. 2004, 18, 357–368. [Google Scholar] [CrossRef]
- Gleyzer, N.; Vercauteren, K.; Scarpulla, R.C. Control of Mitochondrial Transcription Specificity Factors (TFB1M and TFB2M) by Nuclear Respiratory Factors (NRF-1 and NRF-2) and PGC-1 Family Coactivators. Mol. Cell. Biol. 2005, 25, 1354–1366. [Google Scholar] [CrossRef]
- Viña, J.; Gomez-Cabrera, M.C.; Borras, C.; Froio, T.; Sanchis-Gomar, F.; Martinez-Bello, V.E.; Pallardo, F.V. Mitochondrial Biogenesis in Exercise and in Ageing. Adv. Drug Deliv. Rev. 2009, 61, 1369–1374. [Google Scholar] [CrossRef]
- Choi, J.; Avinash, R.; Vamshi, N.; Manfred, S.; Rudolph, J.C.; James, W.R. Potential Roles of PINK1 for Increased PGC-1alpha-Mediated Mitochondrial Fatty Acid Oxidation and Their Associations with Alzheimer Disease and Diabetes. Mitochondrion 2014, 18, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Lei, Y.H.; Zhou, J.P.; Hou, Y.Y.; Wan, Z.; Wang, H.L.; Meng, H. Role of PGC-1alpha in Mitochondrial Quality Control in Neurodegenerative Diseases. Neurochem. Res. 2019, 44, 2031–2043. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.Y.; Cong, L.; Liu, Z.Q.; Liu, Z.F.; Ma, Z.; Liu, K.; Li, J.; Deng, Y.; Liu, W.; Xu, B. Resveratrol Reduces DRP1-Mediated Mitochondrial Dysfunction via the SIRT1-PGC-1αlpha Signaling Pathway in Manganese-Induced Nerve Damage in Mice. Environ. Toxicol. 2021, 37, 282–298. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Song, J.; Jin, E.J. BNIP3-Dependent Mitophagy via PGC-1α Promotes Cartilage Degradation. Cells 2021, 10, 1839. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B. Amelioration of aminoglycoside nephrotoxicity requires protection of renal mitochondria. Kidney Int. 2010, 77, 841–843. [Google Scholar] [CrossRef]
- Azad, M.A.K.; Akter, J.; Rogers, K.L.; Nation, R.L.; Velkov, T.; Li, J. Major pathways of polymyxin-induced apoptosis in rat kidney proximal tubular cells. Antimicrob. Agents Chemother. 2015, 59, 2136–2143. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.; Kim, J.H.; Jung, M.H.; Jang, S.J.; Lee, T.W.; Jung, S.; Lee, S.; Jang, H.N.; Chang, S.-H.; Park, D.J.; et al. Paricalcitol Attenuates Contrast-Induced Acute Kidney Injury by Regulating Mitophagy and Senescence. Oxidative Med. Cell. Longev. 2020, 2020, 7627934. [Google Scholar] [CrossRef]
- Lee, T.W.; Bae, E.; Kim, J.H.; Jung, M.H.; Park, D.J. Psoralen Alleviates Renal Fibrosis by Attenuating Inflammasome-Dependent NLRP3 Activation and Epithelial–Mesenchymal Transition in a Mouse Unilateral Ureteral Obstruction Model. Int. J. Mol. Sci. 2023, 24, 13171. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, T.W.; Bae, E.; Kim, J.H.; Jung, M.H.; Park, D.J. Zileuton Attenuates Acute Kidney Injury in Glycerol-Induced Rhabdomyolysis by Regulating Myeloid-Derived Suppressor Cells in Mice. Int. J. Mol. Sci. 2025, 26, 8353. https://doi.org/10.3390/ijms26178353
Lee TW, Bae E, Kim JH, Jung MH, Park DJ. Zileuton Attenuates Acute Kidney Injury in Glycerol-Induced Rhabdomyolysis by Regulating Myeloid-Derived Suppressor Cells in Mice. International Journal of Molecular Sciences. 2025; 26(17):8353. https://doi.org/10.3390/ijms26178353
Chicago/Turabian StyleLee, Tae Won, Eunjin Bae, Jin Hyun Kim, Myeong Hee Jung, and Dong Jun Park. 2025. "Zileuton Attenuates Acute Kidney Injury in Glycerol-Induced Rhabdomyolysis by Regulating Myeloid-Derived Suppressor Cells in Mice" International Journal of Molecular Sciences 26, no. 17: 8353. https://doi.org/10.3390/ijms26178353
APA StyleLee, T. W., Bae, E., Kim, J. H., Jung, M. H., & Park, D. J. (2025). Zileuton Attenuates Acute Kidney Injury in Glycerol-Induced Rhabdomyolysis by Regulating Myeloid-Derived Suppressor Cells in Mice. International Journal of Molecular Sciences, 26(17), 8353. https://doi.org/10.3390/ijms26178353




