Abstract
In the hexaploid wheat genome, there are three Gα genes, three Gβ and twelve Gγ genes, but the function of Gβ in wheat has not been explored. In this study, we obtained the overexpression of TaGB1 Arabidopsis plants through inflorescence infection, and the overexpression of wheat lines was obtained by gene bombardment. The results showed that under drought and NaCl treatment, the survival rate of Arabidopsis seedlings’ overexpression of TaGB1-B was higher than that of the wild type, while the survival rate of the related mutant agb1-2 was lower than that of the wild type. The survival rate of wheat seedlings with TaGB1-B overexpression was higher than that of the control. In addition, under drought and salt stress, the levels of superoxide dismutase (SOD) and proline (Pro) in the wheat overexpression of TaGB1-B were higher than that of the control, and the concentration of malondialdehyde (MDA) was lower than that of the control. This indicates that TaGB1-B could improve the drought resistance and salt tolerance of Arabidopsis and wheat by scavenging active oxygen. Overall, this work provides a theoretical basis for wheat G-protein β-subunits in a further study, and new genetic resources for the cultivation of drought-tolerant and salt-tolerant wheat varieties.
1. Introduction
Wheat (Triticum aestivum L.) is affected by various abiotic stresses during its growth and development, such as drought, salinity, heat, cold, heavy metals and the ozone [1]. Among them, droughts and soil salinization are the main factors limiting the increase in wheat yield and quality [2]. When plants were subjected to abiotic stresses such as drought, saline–alkali, extreme temperature, etc., the stimulation of external stress factors recognizes, transforms and transmits these stimulation signals through a series of complex signal transduction networks, and finally induces the expression of specific functional genes, and then regulates the biochemical and physiological reactions to respond to these stimuli [3]. Studies have shown that the G-protein is a sensor involved in various important biological pathways. It plays an essential role in the response of plants to abiotic stress by participating in various signal pathways and different signal transduction levels.
Heterotrimeric G-proteins (G-proteins) are composed of Gα (40–46 kDa), Gβ (37–44 kDa) and Gγ (6–9 kDa) subunits [4], the latter two of which are referred to as the βγ complex. In general, the Gα subunit contains a Ras-like domain and a helical domain. The Gβ subunit contains a coiled-coil domain in the N-terminal region and has seven WD40 repeats. The Gγ proteins are subdivided into three types and the A-type is defined by a CaaX motif, whereas the B-type is similar to the A-type, but it lacks the CaaX motif. C-types contain enriched cysteine residues at the C-terminal and N-terminal, similar to the canonical Gγ subunits [5,6]. As crucial signal transducers, G-proteins have attracted increasing attention in the field of signal transduction. According to the classical paradigm, G-protein signaling is initiated by the association of extracellular ligands with GPCR (or RGS) and subsequently inducing a conformational change, which promotes the exchange of GDP for GTP associated with the Gα subunit. GTP-bound Gαβγ heterotrimeric complex tends to dissociate into a Gα-GTP monomer and Gβγ heterodimer [7]. The dissociated units of Gα-GTP monomer and Gβγ heterodimer can interact with a wide array of specific downstream effectors of distinct signaling pathways [8].
Plant G-proteins are involved in multiple aspects of plant growth, including seed germination, seedling development, and biotic and abiotic stresses. For instance, Arabidopsis GPA1 plays a positive role in the signal of three ion channels (K+, Ca2+, anion) that ABA inhibits in the stomatal opening [9,10], and is also a positive regulator of plant cell division [11,12]. AGB1 is involved in the modulation of cell proliferation in hypocotyls [12,13], as well as organ differentiation [14]. It positively regulates the salt tolerance of plants [15] and the defense responses to fungal infection [16,17], and modulates apoptosis caused by tunicamycin [18]. AGG3 is a positive regulator of stress responses [19]. Maize Gα subunits (CT2) control meristem development [20], and the Gβ subunit controls shoot meristem development and immune responses [21]. Cucumber CsGPA1 regulates seed germination and early seedling development, including hypocotyl elongation and root growth [22]. CsGG3.2 positively regulates the chilling tolerance of cucumber [23]. In rice, RGA1 participates in the regulation of plant growth and development. The disruption of RGA1 leads to the dwarfing of upright leaves [24,25,26]. RGB1 cooperates with RGA1 in cellular proliferation and seed fertility [27]. RGB1 is also a positive regulator of drought adaption, whereas qPE9-1 is modulated by RGB1 and functions as a negative regulator in the drought stress response [28]. The overexpression of RGG1 can enhance the salt tolerance of plants [29]. In pea, the G-protein members, PsGα and PsGβ, were expressed in large amounts under drought, high temperature and high salt treatment. The Gβ subunit has a role in the nitric-oxide-induced stomatal closure in response to heat and drought stress [30,31]. The Gγ subunits, GW2 homologues, exist in other grain crops (such as corn, wheat, and sorghum), and increase the cell number of spikelet shells to participate in regulating the seed weight [32,33,34].
At present, many related research results of G-protein have been reported in succession, among which the study of Arabidopsis G-protein is more thorough. However, in wheat, the function of wheat G-protein β-subunits has not been determined, especially since they participate in the regulation of abiotic stress response. The homology between TaGB1 and AGB1 was as high as 93.46%, indicating that TaGB1 and AGB1 may have some similarities in function [35]. Therefore, we will further analyze the functions of TaGB1. Arabidopsis plants overexpressing TaGB1-B were obtained by inflorescence infection, and wheat lines overexpressing TaGB1-B were obtained by gene bombardment. The results showed that the overexpression of TaGB1-B could enhance the tolerance of Arabidopsis and wheat to drought and salt stress. This study provides evidence for the involvement of TaGB1-B in the response of plants to drought and salt stress, and provides useful information for the genetic improvement of drought and salt resistance of wheat.
2. Results
2.1. Expression Profile Analysis of Wheat TaGB1 Homologs
An Ensembl Plants database search showed that TaGB1 exists in three chromosomal groups of A, B and D. Therefore, we named the three homologs TaGB1-A, -B and -D. The homologies of the three homologs in the coding sequence are 99.33%, and the homologies of the amino acid sequence are 99.91%. In order to further analyze the difference of expression of these three homologs under stress treatment, we also downloaded the relative expression abundance of TaGB1 homologues in the leaves of 7-day-old seedlings under drought-, heat- and PEG-simulated drought stress from the wheat expression database (Table S1). Then, we analyzed the expression levels of TaGB1 genes under different stress conditions. From the overall trend, the expression level of all three homologs is similar (Figure 1), but due to the large difference in the expression levels of TaGB1-B under different stress treatments, we finally decided to select TaGB1-B for follow-up research.
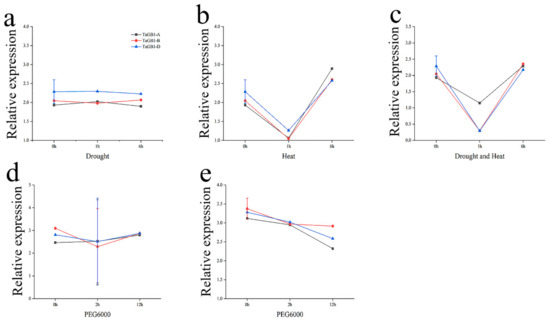
Figure 1.
The expression of TaGB1 homologs under different stress treatments in one-week-old wheat seedling (based on the wheat expression database). (a) Expression of TaGB1 homologs under drought stress in wheat variety TAM107. (b) Expression of TaGB1 homologs under heat stress in wheat variety TAM107. (c) Expression of TaGB1 homologs under drought and heat stress in wheat variety TAM107. (d) Expression of TaGB1 homologs under PEG6000 stress in wheat variety Gemmiza 10. (e) Expression of TaGB1 homologs under PEG6000 stress in wheat variety Giza 168.
2.2. Expression Patterns of TaGB1-B in Different Tissues under Stress
The expression level of TaGB1-B in different stress and hormone treatments was detected by qRT-PCR. The results showed that the expression of TaGB1-B changed under stress and hormones. We found that the expression of TaGB1-B changed under the stress and hormone treatments, it shows a trend of rising first and then falling as a whole (Figure 2a–i). The results showed that TaGB1-B was involved in abiotic stress response. Then, we continued to evaluate the expression level of TaGB1-B in wheat seedling tissue under drought and salt treatment. The results showed that TaGB1-B was differentially expressed in roots, stems and leaves (Figure 2j,k). Drought treatment upregulated TaGB1-B in roots and leaves, and its expression was the highest after 6 h of stress, showing about 3.5 times the upregulation. Under salt stress, the expression of TaGB1-B in roots, stems and leaves showed little difference, and the expression showed an upward trend, reaching the peak at 12 h, and slowly returning to the initial expression level after 24 h.
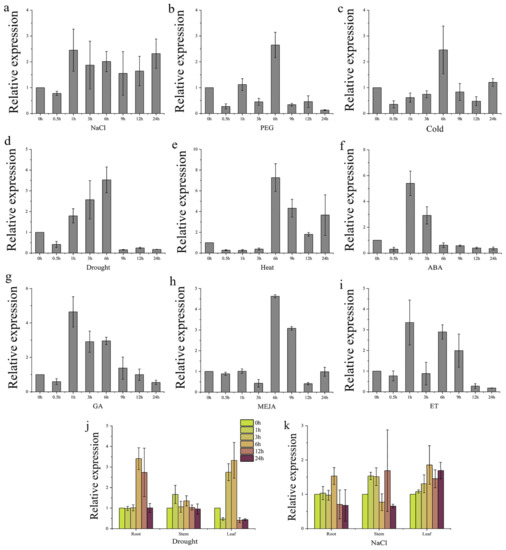
Figure 2.
The expression of TaGB1-B under different stress treatments and tissue-specific expression of TaGB1-B under drought and salt treatments in one-week-old wheat seedling. (a) The expression of TaGB1-B under NaCl treatment; (b) PEG6000; (c) cold; (d) drought; (e) heat; (f) ABA; (g) GA; (h) MeJA; and (i) ET. (j) Tissue-specific expression of TaGB1-B under drought treatment. (k) Tissue-specific expression of TaGB1-B under salt treatment. Error bars show standard deviations (mean ± SD and n = 3).
2.3. Overexpression of TaGB1-B Enhances Drought and Salt Resistance of Arabidopsis
To study the effects of TaGB1-B on plants under drought or salt stress, we selected a homozygous T3 generation, TaGB1-B, overexpressing the Arabidopsis line (OE) with the highest expression level, agb1-2 mutant, and the restorer line of agb1-2 mutant (Res) for follow-up experiments (Figure S1). The seeds from wild-type Arabidopsis (WT), OE, Res, and agb1-2 lines, were evenly seeded in the MS medium, without additives or containing 9% PEG6000, 12% PEG6000, 100 mM NaCl and 150 mM NaCl, respectively. We found that the seed germination rates of WT, overexpression TaGB1-B Arabidopsis line (OE), mutant Arabidopsis(agb1-2), and the restorer line of agb1-2 mutant (Res) on the MS medium were not significantly different (Figure 3). In MS supplemented with PEG6000, the seed germination rates of several lines were inhibited, and the germination rate of all lines was significantly inhibited on the MS medium containing NaCl (Figure 4). In general, the germination rate of OE and Res lines is higher than that of WT and agb1-2. The germination rate of OE is the highest overall, and agb1-2 is the lowest (Figure 3 and Figure 4).
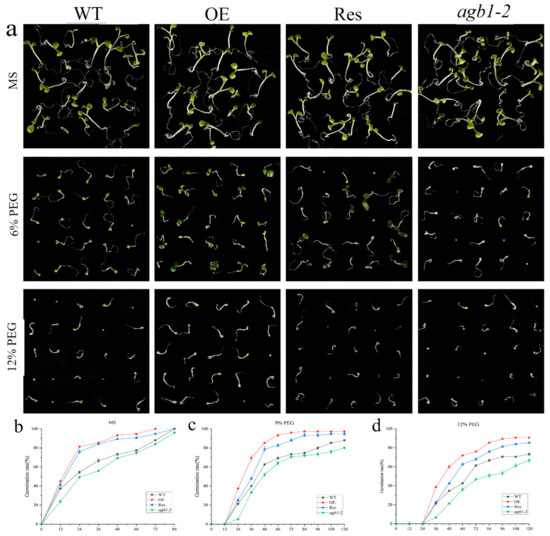
Figure 3.
Germination assay of wild-type (WT), TaGB1-B overexpression Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant (Res) seeds under PEG6000 treatment. (a) The phenotypes of WT, TaGB1-B overexpression Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant seeds under 9% and 12% PEG6000 treatments. (b) The germination rates of WT, TaGB1-B overexpression Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant seeds at different time points on the MS medium. (c) The germination rates under 9% PEG6000 treatment. (d) The germination rates under 12% PEG6000 treatment.
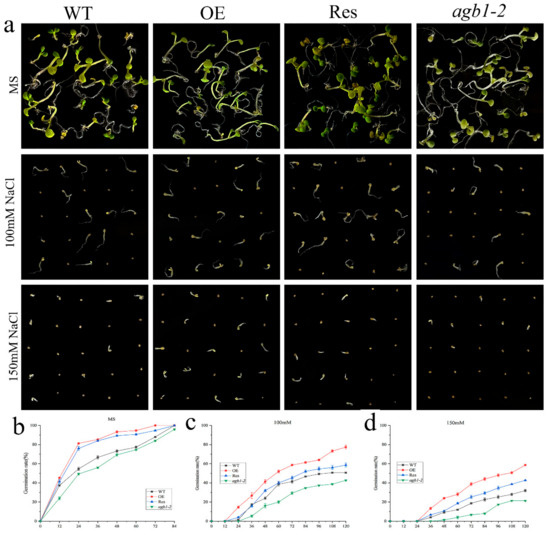
Figure 4.
Germination assay of wild-type (WT), TaGB1-B overexpression Arabidopsis, Arabidopsis mutants (agb1-2) and complementary mutant (Res) seeds under NaCl treatment. (a) The phenotypes of WT, TaGB1-B overexpression of Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant seeds under 100 mM and 150 mM NaCl treatments. (b) The germination rates of WT, TaGB1-B overexpression of Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant seeds at different time points on the MS medium. (c) The germination rates under 100 mM NaCl treatment. (d) The germination rates under 150 mM NaCl treatment.
After that, we conducted root length experiments and evaluated the phenotypic changes of these lines. For samples cultured on the MS medium, the root length of Arabidopsis lines did not change significantly (Figure 5). However, the growth of each line was significantly limited in the MS medium supplemented with PEG6000 or NaCl, and fresh weight also decreased, and the root length and fresh weight of OE and Res lines were significantly higher than those of WT and agb1-2. Therefore, the TaGB1-B gene may affect the ability of Arabidopsis to resist drought and salt stress.
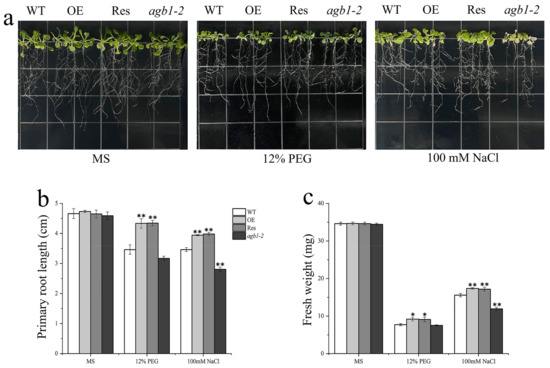
Figure 5.
The overexpression of TaGB1-B enhanced the tolerance to drought and salt stresses in Arabidopsis. (a) Morphological differences between wild-type (WT), TaGB1-B overexpression Arabidopsis, mutant Arabidopsis (agb1-2) and complementary mutant seeds at seedling stages under PEG6000 treatment and NaCl. (b) Statistical analysis of root length. (c) Fresh weight. The error column represents the standard deviation (mean ± SD and n = 3), * and ** above each column indicate a significant difference compared with WT plants (* p < 0.05; ** p < 0.01).
To explore whether TaGB1-B is involved in plant stress resistance at the seedling stage, we conducted drought and salt treatments in the Arabidopsis seedling stage. We counted the survival rate of the four lines and measured the corresponding proline (Pro) and malondialdehyde (MDA) contents. We found that there was no difference among the lines before treatment (Figure 6a and Figure 7a). After treatment, all Arabidopsis lines were stressed. After two weeks of drought stress, agb1-2 withered seriously. However, the effect of drought stress on OE and Res is relatively little effect. After 3 days of re-watering, OE and Res continued to grow, while agb1-2 died as the leaves turned green. The survival rates of OE and Res were 88% and 86%, much higher than WT (68%) and agb1-2 (45%) (Figure 6b). The MDA content of OE was significantly lower than WT, while the Pro content was higher than WT (Figure 6c,d). These results showed that overexpression of TaGB1-B enhanced the drought tolerance of Arabidopsis. After salt stress, WT and agb1-2 were undergoing serious chlorosis, while OE and Res were slightly withered. The survival rate of OE (66%) and Res (66%) was higher than that of WT (38%) and agb1-2 (22%) (Figure 7b). After stress, the content of Pro in OE and Res was significantly higher than WT and agb1-2, and the content of MDA was significantly lower than WT and agb1-2 (Figure 7c,d). The above results showed that the overexpression of TaGB1-B enhanced the salt tolerance of Arabidopsis.
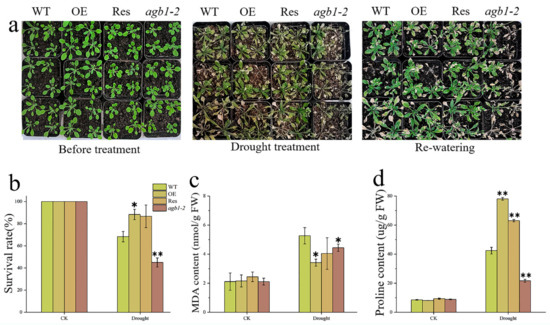
Figure 6.
The functional characteristics of drought tolerance of transgenic Arabidopsis with TaGB1-B gene under drought stress. (a) Phenotypic analysis of each plant line before treatment, 14 days after drought treatment, and 3 days after rehydration. (b) Survival rate of each line after drought stress. (c) The content of malondialdehyde (MDA) in Arabidopsis treated by drought. (d) Proline content. The data are expressed as the mean ± SDs (n = 3) of three experiments. * and ** above each column indicate a significant difference compared with WT plants (* p < 0.05; ** p < 0.01).
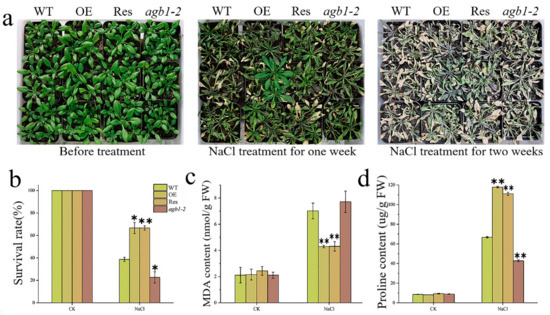
Figure 7.
The functional characteristics of drought tolerance of transgenic Arabidopsis with TaGB1-B gene under NaCl stress. (a) Phenotypic analysis of each plant line before treatment, one week after NaCl treatment and two weeks after NaCl. (b) Survival rate of each line after NaCl stress. (c) The content of malondialdehyde (MDA) in Arabidopsis treated by NaCl. (d) Proline content. The data are expressed as the mean ± SDs (n = 3) of three experiments. * and ** above each column indicate a significant difference compared with WT plants (* p < 0.05; ** p < 0.01).
2.4. Overexpression of TaGB1-B Enhances the Ability of Wheat to Resist Drought and Salt Stress
In order to determine the role of TaGB1-B in drought and salt tolerance of wheat, transgenic lines of wheat variety KeNong 199 (KN199) overexpressing TaGB1-B and an empty vector gene were produced, and T3 transgenic homozygous lines were obtained and detected by qRT-PCR. We selected three T3 generation TaGB1-B overexpression wheat transgenic lines (TaOE3, TaOE4, TaOE5) with high expression levels, and one T3 generation transgenic wheat line with an empty vector (EV) for follow-up experiments (Figure S2). We evaluated the expression level of TaGB1-B under drought and salt stress in one-week-old overexpression wheat seedlings. The data indicated that under stress conditions, the expression levels of TaGB1-B genes were upregulated in WT and transgenic plants, but the expression levels were higher in the TaGB1-B overexpressing plants than in the control plants (Figure 8). Under drought stress, the expression of TaGB1-B reached its peak at 24 h, and then showed a downward trend. The expression of TaGB1-B in TaOE3 was the highest at 24 h, being about 6.5 times higher (Figure 8a). Under salt stress, the expression of TaGB1-B decreased first and then increased. The expression of TaGB1-B in TaOE3 was the highest at 48 h, being about 3.4 times higher (Figure 8b).
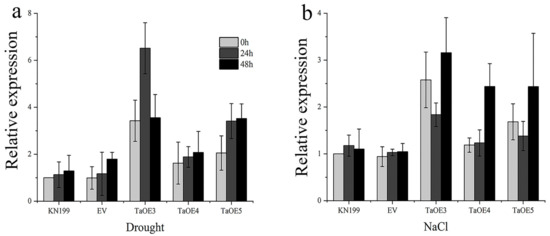
Figure 8.
Transcription spectrum analysis of TaGB1-B transgenic wheat under drought and salt stress. (a) The expression level of TaGB1-B in each line under drought treatment. (b) The expression level of TaGB1-B of each line under NaCl treatment. Error bars show standard deviations (mean ± SD and n = 3).
Considering that the root system is related to the abiotic stress resistance of plants, we also examined the morphology of the roots and found that there are no obvious differences in root length and lateral root number between the KN199 and transgenic wheat with the empty vector gene (EV) under various growth conditions (Figure 9). This indicates that the empty vector gene transferred into wheat has no effect on the root growth of wheat. Under normal growth conditions, there was no significant difference in the root length between KN199 and TaGB1-B overexpressing wheat transgenic lines (TaOE3, TaOE4, TaOE5) (Figure 9b), but the number of lateral roots of KN199 was significantly higher than that of overexpression wheat (Figure 9c). After the treatment of drought and salt stress, the root length and lateral root number of the overexpression line were significantly higher than that of KN199, and the root length and lateral root number of TaOE3 were the highest as a whole. Taken together, these results indicated that the overexpression of TaGB1-B increased the root length and lateral root number of wheat under drought and salt stress, and improved the ability of wheat to resist drought and salt stress.
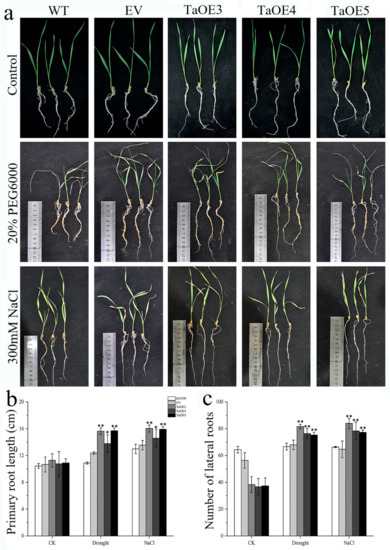
Figure 9.
TaGB1-B enhanced the drought and salt tolerance of wheat. (a) Drought and salt stress tolerance responses of TaGB1-B overexpression transgenic KN199 and the empty vector transgenic plants. (b) Primary root length. (c) Number of lateral roots. Data are presented as the mean ± standard deviation calculated from triplicates. * and ** above each column indicate a significant difference compared with WT plants (* p < 0.05; ** p < 0.01).
In order to explore whether TaGB1-B is involved in the stress resistance of wheat seedlings, we conducted drought and salt treatment on two-week-old wheat seedlings. We measured several physiological indexes and counted the survival rate of five lines after rehydration. Under normal conditions, no visible phenotypic difference was observed between the OE lines, EV and WT, and the phenotypes and physiological indexes of EV and WT had no significant difference under stress treatments (Figure 10 and Figure 11). Drought stress had little effect on the overexpression lines, and the growth of OE lines was better than that of WT and EV controls (Figure 10a). After drought treatment, physiological indicators had an increasing trend, but the trend is different. The MDA content of several lines decreased, and the PRO content and the activities of superoxide dismutase (SOD) and peroxidase (POD) increased, but the MDA content in OE was lower than WT, and the PRO content and SOD content were higher than WT (Figure 10c–f). In addition, the survival rate of OE after rehydration was higher than that of KN199 (Figure 10b). After salt stress treatment, both KN199 and overexpression plants showed some degree of wilting and inhibited growth, but the wilting degree of overexpression plants was significantly lower than that of KN199 (Figure 11a). The changes in several physiological indicators were basically consistent with the drought treatment (Figure 11). These results showed that the overexpression of TaGB1-B increased the tolerance of wheat variety KN199 to drought and salt at the seedling stage.
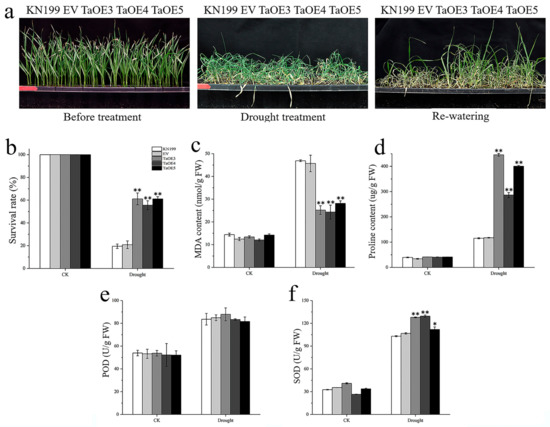
Figure 10.
The functional characteristics of TaGB1-B overexpression wheat plants under drought stress. (a) Phenotypic analysis of TaGB1-B overexpression plants under normal treatment, drought stress treatment for 14 days and rehydration for 3 days. (b) The survival rate of each line after drought stress. (c–f). Physiological index of wheat plant. (c) MDA content. (d) Proline content. (e) POD. (f) SOD. The error column represents the standard deviation (mean ± SD and n = 3), and * and ** above each column indicate a significant difference compared with WT plants (* p < 0.05; ** p < 0.01).
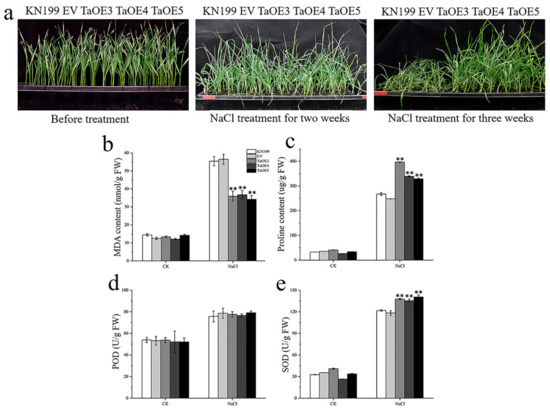
Figure 11.
The functional characteristics of TaGB1-B overexpression wheat plants under NaCl stress. (a) Phenotypic analysis of TaGB1-B overexpression plants under normal treatment, NaCl stress treatment for two weeks and NaCl stress treatment for three weeks. (b–e) Physiological index of wheat plant. (b) MDA content. (c) Proline content. (d) POD. (e) SOD. The error column represents the standard deviation (mean ± SD and n = 3), and and ** above each column indicate a significant difference compared with WT plants (** p < 0.01).
3. Discussion
The hexaploid wheat genome (2n = 6x = 42, AABBDD) is composed of three subgenomes, A, B and D, and is extremely complex. More than 80% of the sequences in the three subgenomes are similar [36]. Gawande used the dataset from the expression database to compare the expression of TaGB1 homologs in all 71 tissues and the development time points, and found that the level of expression for the A, B and D homologs was similar. In 71 tissues and development stages, all three homologs showed relatively similar levels of expression [37]. In this study, TaGB1 showed a high level of homology across the A, B and D chromosome groups, and its expression levels are similar under different stress conditions (Figure 1). This suggests that TaGB1 is a highly conserved gene across the wheat chromosome groups.
TaGB1 contains seven WD40-conserved domains and is a member of the WD40 protein family. WD40 proteins are involved in various processes, such as growth and development, metabolite biosynthesis, and immune and stress responses [13,21,38,39,40,41,42,43,44,45]. Furthermore, it has been shown in Arabidopsis [15,46,47], rice [29,48,49,50], maize [51,52,53], and rapeseed [54,55] that G-proteins are involved in abiotic stresses. In this study, the qRT-PCR analysis showed that the expression of TaGB1-B changed under stress and hormone treatments (Figure 2a–i). This finding is consistent with previous studies. Further analysis of TaGB1-B expression under drought and salt treatment showed that its expression was regulated differently in different tissues. Specifically, drought treatment upregulated the expression of TaGB1-B in roots and leaves, while TaGB1-B expression in roots, stems, and leaves showed almost no difference under salt stress, but showed an upward trend instead (Figure 2j,k). These results indicate that TaGB1-B can regulate the response of wheat to abiotic stress.
Seed germination rate is one of the important indicators for measuring the quality of crop seeds, which directly affects the yield and quality of crops. A high seed germination rate means more seeds can successfully germinate into seedlings, thereby increasing the crop emergence rate and growth speed, and improving crop yield and quality. In addition, the seed germination rate also affects the entire growth season and the maturity time of crops, thereby affecting the harvest and efficiency of agricultural production. Therefore, ensuring a high seed germination rate is one of the important measures to improve crop yield and quality [56,57,58,59]. The root system is the main organ for nutrient uptake in plants, capable of absorbing water, nutrients, and other essential substances. A well-structured and functional root system can enhance plant growth rate and yield, as well as increase the plant’s ability to withstand adversity. Environmental disruptions are closely related to changes in the root structure. In order to adapt to adversity, plants can regulate the growth of primary roots, lateral roots, or adventitious roots, as well as the length and distribution of root hairs [60,61]. Existing research has shown that under non-biological stress, Arabidopsis thaliana strains with transferred related genes have higher germination rates and longer roots than wild-type plants [62,63,64]. This situation has also been observed in wheat overexpressing TaSNAC8-6A [65], transgenic Arabidopsis overexpressing AtNAC2 [15,66], and transgenic rice overexpressing OsNAC6 [67]. In our experiment, the overexpression of TaGB1-B in Arabidopsis resulted in higher germination rates and longer primary roots under drought and salt stress compared to WT and agb1-2 mutant lines (Figure 3, Figure 4 and Figure 5). The situation in wheat is similar to that in Arabidopsis, with overexpression leading to a longer root length and more lateral roots than the KN199 (Figure 9). Our findings suggest that TaGB1-B plays an important role in regulating root development and stress responses, and may be a potential target for improving plant growth and stress tolerance.
Malondialdehyde (MDA) is one of the commonly used indicators to measure the degree of oxidative stress and reflects the degree of membrane lipid peroxidation in plants. Proline (Pro) is one of the components of plant proteins and can exist widely in a free state in the plant body, and accumulated Pro acts as an osmoregulatory substance in plant cytoplasm [44,68,69]. When plants are subjected to abiotic stress, the higher the oxidative stress response, the higher the MDA content. Conversely, for the soluble substance Pro in the cells, the higher the content, the stronger the protective effect on plants [62,64,70]. Our research results indicate that under drought and salt stress, the MDA and Pro contents of all lines increased significantly, but the MDA content of the transgenic Arabidopsis and wheat was lower than that of the WT, and the Pro content was higher than that of the WT (Figure 6 and Figure 7). This indicates that transgenic plants overexpressing TaGB1-B have stronger drought and salt tolerance.
Among the physiological issues that accompany abiotic stresses in plants, the excessive accumulation of ROS (particularly O2− and H2O2) and high concentrations of ROS lead to the damage of proteins, lipids and nucleic acid, and eventually lead to cell damage or death [71]. A large amount of evidence shows that drought and salt stress change the number and activity of enzymes involved in scavenging oxygen free radicals. G-protein can reduce the damage of reactive oxygen species to cells by limiting the production of ROS or enhancing the detoxification mechanism, thus enhancing the adaptation to abiotic stress. Plants have evolved a suite of corresponding protection strategies (i.e., enzymatic and non-enzymatic anti-oxidative systems) to protect themselves from oxidative damage [72]. Antioxidant enzymes (AE), such as SOD, CAT and POD, are critical mediators of ROS detoxification caused by environmental stressors. In recent years, it has been confirmed that the transcription of different AE genes is regulated by G-protein subunits. For example, the transcripts of the CAT gene in rice are regulated by RGG1, a γ subunit of the G-protein. An overexpression of RGG1 can enhance CAT activity and salt tolerance of plants [29]. The ectopic expression of rice RGB1 in transgenic lines enhanced the salt tolerance of rice and the expression level of enzymes involved in antioxidant stress defense (such as superoxide dismutase (SOD) and ascorbic acid peroxidase (APX)) increased in the overexpression lines [73]. These findings suggest a relationship between the G-protein subunit and ROS homeostasis in plant cells. Our results showed that after drought treatment, the activities of superoxide dismutase (SOD) and peroxidase (POD) increased in several wheat lines, but the SOD content was higher in OE than in WT (Figure 10). After salt stress treatment, the changes in several physiological indicators were similar to those observed under drought treatment (Figure 11). These results suggest that TaGB1-B may play an important role in conferring drought and salt tolerance to wheat.
In summary, our study provides evidence for the involvement of the TaGB1-B gene in enhancing drought and salt tolerance in Arabidopsis and wheat. Our results suggest that TaGB1-B may be a promising candidate gene for improving drought and salt tolerance in wheat. Further research is needed to elucidate the molecular mechanisms of TaGB1-B gene expression regulation, and explore its potential applications in crop improvement programs.
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
Wheat seeds (“KN199” and “Chinese Spring”) and wild-type Arabidopsis (Col-0) were from our laboratory (the laboratory for genetic improvement of wheat stress resistance, Northwest Agricultural and Forestry University). Both wheat and Arabidopsis were cultured at 20–25 °C, with a light cycle of 16 h of light/8 h of dark, light intensity of 15000 Lux, and humidity of about 70%.
4.2. Sequence Analysis and Expression of TaGB1 Homologous Gene
We used transcriptome data from the WheatEXP website (https://wheat.pw.usda.gov/WheatExp/, accessed on 24 March 2021) to study the expression pattern of TaGB1 homologues in response to abiotic stress. The RNA-seq data comes from nine different growth stages and tissues of Chinese Spring under normal conditions, as well as the following six kinds of stress and normal control (heat stress 1 h, drought stress 1 h, heat stress 6 h, drought stress 6 h, combined drought and heat stress 1 h, combined drought and heat stress 6 h, no threat control) (Table S1). The cDNA-coding region of TaGB1-B was isolated from CS. The primers used are shown in Table S2.
4.3. RNA Extraction and qRT-PCR Analysis
The Chinese Spring was placed evenly in the culture dish, and when it was cultivated to the trefoil stage (about 10 days), drought, NaCl (300 mmol/L), PEG6000 (20%), cold, heat (38 °C), ABA (100 μmol), GA (100 μmol), MEJA (100 μmol) and ET (100 μmol) treatments were carried out. Leaves were taken at 0 h (CK), 0.5 h, 1 h, 3 h, 6 h, 9 h, 12 h and 24 h, and the roots, stems and leaves after drought and salt treatment at 0 h (CK), 1 h, 3 h, 6 h, 12 h and 24 h. Then, they were quickly frozen with liquid nitrogen and stored in a refrigerator at −80 °C for standby. The total RNA was extracted with the GenStar (GenStar, Beijing, China) Trizol kit, and cDNA was synthesized with the TransGen (TransGen, Beijing, China) reverse-transcription kit. The cDNA of wheat under two stress treatments was used as the template, and β-Actin was used as the internal reference gene. qRT-PCR was performed with the SYBR Green dye method to analyze the expression of the TaGB1 gene. Three replicates were set for each sample. The relative expression of each gene was calculated according to the 2−∆∆CT value. The primers used in this experiment are shown in Table S2.
4.4. Arabidopsis Transformation and Stress Tolerance Assays
In order to obtain the overexpressed plant of Arabidopsis, we connected the sequence of TaGB1-B to the pCAMBIA1302 vector. The recombinant plasmid with the correct sequence was transformed into Agrobacterium line GV3101, and then transformed into the Arabidopsis Col-0 type using the inflorescence immersion method. The mutant and overexpression information of Arabidopsis are shown in Figure S1. Then, the TaGB1-B overexpression seeds were cultured to the T3 generation. The growth conditions of Arabidopsis were the same as for the wheat. We cultured the seeds of Arabidopsis WT, TaGB1-B overexpression, agb1-2 mutant and complementary agb1-2 mutant lines on the MS medium containing PEG6000 (9% and 12%), NaCl (100 mM and 150 mM), and MS with no additives, and recorded the germination rate every 12 h.
The same growth state Arabidopsis seedlings were cultured vertically on the MS medium containing 12% PEG6000 or 100 mM NaCl, and the root length was recorded after seven days of growth. In order to test the drought tolerance of Arabidopsis, we transferred Arabidopsis seedlings with the same growth state on the MS into the soil. After three weeks of culture, the seedlings were subjected to drought stress (no water supply) for two weeks, and then rehydrated for three days. The survival rate and the contents of MDA and Pro were recorded. In order to investigate the salt tolerance, after three weeks of culture, plants were irrigated with 200 mM NaCl for two weeks, and the survival rate and the contents of MDA and Pro were recorded. We performed three independent biological replications.
4.5. Wheat Transformation and Stress Tolerance Assays
Callus was induced from the young embryos of KeNong 199 which were pollinated for about 14 days. The constructed pWMB003-TaGB1 and pWMB003 empty vector plasmid were bombarded into the calli by gene gun bombardment, and then differentiated and screened. Transgenic-positive and empty vector (EV) transgenic line plants in each generation were screened by PCR. The relative expression of TaGB1-B in transgenic plants was detected by using the methods of RNA extraction, cDNA synthesis and qRT-PCR, described above.
In order to determine the effect of drought and salt stress on the root at the wheat seedling stage, the wheat seedlings of WT, EV and OE lines, at the age of one week, were cultured with 1/2 Hoagland nutrient solution plus 20% (M/V) polyethylene glycol (PEG6000) and 1/2 Hoagland nutrient solution with 300 mM NaCl, respectively. After one week of treatment, the morphological and physiological parameters of the plant roots were measured.
In order to analyze the drought tolerance of wheat at the seedling stage, WT, EV and OE seeds were germinated in Petri dishes and transplanted into the greenhouse for 16 h of light/8 h of darkness for two weeks. When the relative water content of the soil is about 60%, the drought stress is carried out (no watering for two weeks), and then re-watering takes place for 3 days. The phenotype and survival rate were recorded and the physiological indexes of each wheat line under drought stress were determined. In order to analyze the salt tolerance of wheat seedlings, the salt tolerance experiment was carried out on the wheat growing for three weeks. The wheat was irrigated with 300 mM NaCl for three weeks. The plant survival rate and physiological indexes of each wheat line were recorded. Three independent biological replications were carried out in the above experiments.
4.6. Statistical Analysis
We used the SPSS (Chicago, Illinois, USA) software for statistical analysis. The data were calculated with the mean ± standard deviation (SD) and an analysis of variance (ANOVA), and the significance level was defined as * (p < 0.05) and ** (p < 0.01).
5. Conclusions
In this study, we selected TaGB1-B for subsequent functional verification. The results showed that TaGB1-B improved the tolerance of Arabidopsis and wheat plants to drought and salt stress. This work provides a theoretical basis for wheat G-protein β-subunits in a further study, and new genetic resources for the cultivation of drought-tolerant and salt-tolerant wheat varieties. This work has a strong future application value, especially considering the expected crop losses related to climate change.
Supplementary Materials
The supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms24087337/s1.
Author Contributions
D.-H.M. and X.-H.Z. conceived and designed the experiments. X.-X.X. performed the experiments and wrote the paper. Y.L. and L.-L.Z. helped in the analysis of the data. X.-J.L. collected the previous studies. Y.Z. (Yue Zhao), Y.Z. (Yan Zheng), Q.-H.Y. and Y.Y. carried out some experiments. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by the National Key Research and Development Plan Project (2022YFD1200205), the Agricultural Science and Technology Innovation Program of the Shaanxi Province (NYKJ-2002-YL(XN)25) and the Key Research and Development Program of the Shaanxi Province (2022NY-18).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Abdel Latef, A.A.H.; Kordrostami, M.; Zakir, A.; Zaki, H.; Saleh, O.M. Eustress with H(2)O(2) Facilitates Plant Growth by Improving Tolerance to Salt Stress in Two Wheat Cultivars. Plants 2019, 8, 303. [Google Scholar] [CrossRef]
- Zhu, J.-K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef]
- Shao, H.; Liang, Z.; Shao, M. Adaptation of higher plants to environmental stresses and stress signal transduction. Acta Ecol. Sin. 2005, 25, 1772–1781. [Google Scholar]
- Gilman, A.G. G proteins: Transducers of receptor-generated signals. Annu. Rev. Biochem. 1987, 56, 615–649. [Google Scholar] [CrossRef]
- Sprang, S.R. G protein mechanisms: Insights from structural analysis. Annu. Rev. Biochem. 1997, 66, 639–678. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Assmann, S.M. Plants: The latest model system for G-protein research. EMBO Rep. 2004, 5, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xie, P.; Xu, X.; Xie, Q.; Yu, F. Heterotrimeric G protein signalling in plant biotic and abiotic stress response. Plant Biol. 2021, 23, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Oldham, W.M.; Hamm, H.E. Heterotrimeric G protein activation by G-protein-coupled receptors. Nat. Rev. Mol. Cell Biol. 2008, 9, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Song, C.P. Guard-cell signalling for hydrogen peroxide and abscisic acid. New Phytol. 2008, 178, 703–718. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jeon, B.W.; Assmann, S.M. Heterotrimeric G-protein regulation of ROS signalling and calcium currents in Arabidopsis guard cells. J. Exp. Bot. 2011, 62, 2371–2379. [Google Scholar] [CrossRef]
- Ullah, H.; Chen, J.-G.; Young, J.C.; Im, K.-H.; Sussman, M.R.; Jones, A.M. Modulation of cell proliferation by heterotrimeric G protein in Arabidopsis. Science 2001, 292, 2066–2069. [Google Scholar] [CrossRef]
- Chen, J.-G.; Gao, Y.; Jones, A.M. Differential roles of Arabidopsis heterotrimeric G-protein subunits in modulating cell division in roots. Plant Physiol. 2006, 141, 887–897. [Google Scholar] [CrossRef]
- Lease, K.A.; Wen, J.; Li, J.; Doke, J.T.; Liscum, E.; Walker, J.C. A mutant Arabidopsis heterotrimeric G-protein β subunit affects leaf, flower, and fruit development. Plant Cell 2001, 13, 2631–2641. [Google Scholar] [PubMed]
- Ullah, H.; Chen, J.G.; Temple, B.; Boyes, D.C.; Alonso, J.M.; Davis, K.R.; Ecker, J.R.; Jones, A.M. The beta-subunit of the Arabidopsis G protein negatively regulates auxin-induced cell division and affects multiple developmental processes. Plant Cell 2003, 15, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-n.; Chen, M.; Xu, D.-b.; Fang, G.-n.; Wang, E.-h.; Gao, S.-q.; Xu, Z.-s.; Li, L.-c.; Zhang, X.-h.; Min, D.-h.; et al. G-protein β subunit AGB1 positively regulates salt stress tolerance in Arabidopsis. J. Integr. Agric. 2015, 14, 314–325. [Google Scholar] [CrossRef]
- Trusov, Y.; Rookes, J.E.; Chakravorty, D.; Armour, D.; Schenk, P.M.; Botella, J.R. Heterotrimeric G proteins facilitate Arabidopsis resistance to necrotrophic pathogens and are involved in jasmonate signaling. Plant Physiol. 2006, 140, 210–220. [Google Scholar] [CrossRef]
- Delgado-Cerezo, M.; Sánchez-Rodríguez, C.; Escudero, V.; Miedes, E.; Fernández, P.V.; Jordá, L.; Hernández-Blanco, C.; Sánchez-Vallet, A.; Bednarek, P.; Schulze-Lefert, P.; et al. Arabidopsis heterotrimeric G-protein regulates cell wall defense and resistance to necrotrophic fungi. Mol. Plant 2012, 5, 98–114. [Google Scholar] [CrossRef]
- Wang, S.; Narendra, S.; Fedoroff, N. Heterotrimeric G protein signaling in the Arabidopsis unfolded protein response. Proc. Natl. Acad. Sci. USA 2007, 104, 3817–3822. [Google Scholar] [CrossRef]
- Kaur, J.; Roy Choudhury, S.; Vijayakumar, A.; Hovis, L.; Rhodes, Z.; Polzin, R.; Blumenthal, D.; Pandey, S. Arabidopsis Type III Gγ Protein AGG3 Is a Positive Regulator of Yield and Stress Responses in the Model Monocot Setaria viridis. Front. Plant Sci. 2018, 9, 109. [Google Scholar] [CrossRef]
- Bommert, P.; Je, B.I.; Goldshmidt, A.; Jackson, D. The maize Gα gene COMPACT PLANT2 functions in CLAVATA signalling to control shoot meristem size. Nature 2013, 502, 555–558. [Google Scholar] [CrossRef]
- Wu, Q.; Xu, F.; Liu, L.; Char, S.N.; Ding, Y.; Je, B.I.; Schmelz, E.; Yang, B.; Jackson, D. The maize heterotrimeric G protein β subunit controls shoot meristem development and immune responses. Proc. Natl. Acad. Sci. USA 2020, 117, 1799–1805. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Zhang, W.; Li, Y.; He, C.; Gao, L.; Yu, X. Functions of CsGPA1 on the hypocotyl elongation and root growth of cucumbers. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Liu, Y.; Mu, Y.; Anwar, A.; He, C.; Yan, Y.; Li, Y.; Yu, X. Heterotrimeric G-Protein γ Subunit CsGG3.2 Positively Regulates the Expression of CBF Genes and Chilling Tolerance in Cucumber. Front. Plant Sci. 2018, 9, 488. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, Y.; Kato, T.; Ohki, S.; Ishikawa, A.; Kitano, H.; Sasaki, T.; Asahi, T.; Iwasaki, Y. Suppression of the heterotrimeric G protein causes abnormal morphology, including dwarfism, in rice. Proc. Natl. Acad. Sci. USA 1999, 96, 7575–7580. [Google Scholar] [CrossRef] [PubMed]
- Ashikari, M.; Wu, J.; Yano, M.; Sasaki, T.; Yoshimura, A. Rice gibberellin-insensitive dwarf mutant gene Dwarf 1 encodes the α-subunit of GTP-binding protein. Proc. Natl. Acad. Sci. USA 1999, 96, 10284–10289. [Google Scholar] [CrossRef] [PubMed]
- Ueguchi-Tanaka, M.; Fujisawa, Y.; Kobayashi, M.; Ashikari, M.; Iwasaki, Y.; Kitano, H.; Matsuoka, M. Rice dwarf mutant d1, which is defective in the α subunit of the heterotrimeric G protein, affects gibberellin signal transduction. Proc. Natl. Acad. Sci. USA 2000, 97, 11638–11643. [Google Scholar] [CrossRef]
- Utsunomiya, Y.; Samejima, C.; Takayanagi, Y.; Izawa, Y.; Yoshida, T.; Sawada, Y.; Fujisawa, Y.; Kato, H.; Iwasaki, Y. Suppression of the rice heterotrimeric G protein β-subunit gene, RGB1, causes dwarfism and browning of internodes and lamina joint regions. Plant J. 2011, 67, 907–916. [Google Scholar] [CrossRef]
- Zhang, D.P.; Zhou, Y.; Yin, J.F.; Yan, X.J.; Lin, S.; Xu, W.F.; Baluška, F.; Wang, Y.P.; Xia, Y.J.; Liang, G.H.; et al. Rice G-protein subunits qPE9-1 and RGB1 play distinct roles in abscisic acid responses and drought adaptation. J. Exp. Bot. 2015, 66, 6371–6384. [Google Scholar] [CrossRef]
- Swain, D.M.; Sahoo, R.K.; Srivastava, V.K.; Tripathy, B.C.; Tuteja, R.; Tuteja, N. Function of heterotrimeric G-protein γ subunit RGG1 in providing salinity stress tolerance in rice by elevating detoxification of ROS. Planta 2017, 245, 367–383. [Google Scholar] [CrossRef]
- Misra, S.; Wu, Y.; Venkataraman, G.; Sopory, S.K.; Tuteja, N. Heterotrimeric G-protein complex and G-protein-coupled receptor from a legume (Pisum sativum): Role in salinity and heat stress and cross-talk with phospholipase C. Plant J. 2007, 51, 656–669. [Google Scholar] [CrossRef]
- Bhardwaj, D.; Sahoo, R.K.; Naqvi, A.R.; Lakhanpaul, S.; Tuteja, N. Pea Gβ subunit of G proteins has a role in nitric oxide-induced stomatal closure in response to heat and drought stress. Protoplasma 2020, 257, 1639–1654. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Mace, E.S.; Tai, S.; Cruickshank, A.; Campbell, B.C.; Zhao, X.; Van Oosterom, E.J.; Godwin, I.D.; Botella, J.R.; Jordan, D.R. Whole-Genome Analysis of Candidate genes Associated with Seed Size and Weight in Sorghum bicolor Reveals Signatures of Artificial Selection and Insights into Parallel Domestication in Cereal Crops. Front. Plant Sci. 2017, 8, 1237. [Google Scholar] [CrossRef]
- Su, Z.; Hao, C.; Wang, L.; Dong, Y.; Zhang, X. Identification and development of a functional marker of TaGW2 associated with grain weight in bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2011, 122, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Li, L.; Yang, X.; Warburton, M.L.; Bai, G.; Dai, J.; Li, J.; Yan, J. Relationship, evolutionary fate and function of two maize co-orthologs of rice GW2 associated with kernel size and weight. BMC Plant Biol. 2010, 10, 143. [Google Scholar] [CrossRef]
- Wang, X.T.; Wang, W.J.; Li, B.; Yang, W.J.; Hu, W.G. Cloning and Expression Analysis of Resistance-Related Gene TaGB1 in Wheat. Triticeae Crops 2018, 38, 1011–1019. [Google Scholar]
- Walkowiak, S.; Gao, L.; Monat, C.; Haberer, G.; Kassa, M.T.; Brinton, J.; Ramirez-Gonzalez, R.H.; Kolodziej, M.C.; Delorean, E.; Thambugala, D.; et al. Multiple wheat genomes reveal global variation in modern breeding. Nature 2020, 588, 277–283. [Google Scholar] [CrossRef]
- Gawande, N.D.; Hamiditabar, Z.; Brunetti, S.C.; Gulick, P.J. Characterization of the heterotrimeric G protein gene families in Triticum aestivum and related species. 3 Biotech. 2022, 12, 99. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, Y.; Shen, J.; Yin, J.; Li, D.; Gao, Y.; Xu, W.; Liang, J. OsRACK1A, encodes a circadian clock-regulated WD40 protein, negatively affect salt tolerance in rice. Rice 2018, 11, 45. [Google Scholar] [CrossRef]
- Kong, D.; Li, M.; Dong, Z.; Ji, H.; Li, X. Identification of TaWD40D, a wheat WD40 repeat-containing protein that is associated with plant tolerance to abiotic stresses. Plant Cell Rep. 2015, 34, 395–410. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Li, Y.H.; Yuan, H.M.; Zhang, B.L.; Zhai, S.; Lu, Y.T. WD40-REPEAT 5a functions in drought stress tolerance by regulating nitric oxide accumulation in Arabidopsis. Plant Cell Environ. 2017, 40, 543–552. [Google Scholar] [CrossRef]
- Tian, G.; Wang, S.; Wu, J.; Wang, Y.; Wang, X.; Liu, S.; Han, D.; Xia, G.; Wang, M. Allelic variation of TaWD40-4B.1 contributes to drought tolerance by modulating catalase activity in wheat. Nat. Commun. 2023, 14, 1200. [Google Scholar] [CrossRef]
- Hu, R.; Xiao, J.; Gu, T.; Yu, X.; Zhang, Y.; Chang, J.; Yang, G.; He, G. Genome-wide identification and analysis of WD40 proteins in wheat (Triticum aestivum L.). BMC Genom. 2018, 19, 1–13. [Google Scholar]
- Jain, B.P.; Pandey, S. WD40 Repeat Proteins: Signalling Scaffold with Diverse Functions. Protein J. 2018, 37, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Feyissa, B.A.; Arshad, M.; Gruber, M.Y.; Kohalmi, S.E.; Hannoufa, A. The interplay between miR156/SPL13 and DFR/WD40-1 regulate drought tolerance in alfalfa. BMC Plant Biol. 2019, 19, 434. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yu, F.; Xie, P.; Sun, S.; Qiao, X.; Tang, S.; Chen, C.; Yang, S.; Mei, C.; Yang, D.; et al. A Gγ protein regulates alkaline sensitivity in crops. Science 2023, 379, eade8416. [Google Scholar] [CrossRef]
- Colaneri, A.C.; Tunc-Ozdemir, M.; Huang, J.P.; Jones, A.M. Growth attenuation under saline stress is mediated by the heterotrimeric G protein complex. BMC Plant Biol. 2014, 14, 129. [Google Scholar] [CrossRef]
- Mason, M.G.; Botella, J.R. Completing the heterotrimer: Isolation and characterization of an Arabidopsis thaliana G protein γ-subunit cDNA. Proc. Natl. Acad. Sci. USA 2000, 97, 14784–14788. [Google Scholar] [CrossRef]
- Yadav, D.K.; Shukla, D.; Tuteja, N. Rice heterotrimeric G-protein alpha subunit (RGA1): In silico analysis of the gene and promoter and its upregulation under abiotic stress. Plant Physiol. Biochem. 2013, 63, 262–271. [Google Scholar] [CrossRef]
- Matsuta, S.; Nishiyama, A.; Chaya, G.; Itoh, T.; Miura, K.; Iwasaki, Y. Characterization of heterotrimeric G protein γ4 subunit in rice. Int. J. Mol. Sci. 2018, 19, 3596. [Google Scholar] [CrossRef]
- Yadav, D.K.; Islam, S.S.; Tuteja, N. Rice heterotrimeric G-protein gamma subunits (RGG1 and RGG2) are differentially regulated under abiotic stress. Plant Signal. Behav. 2012, 7, 733–740. [Google Scholar] [CrossRef]
- Urano, D.; Jones, A.M. Heterotrimeric G protein-coupled signaling in plants. Annu. Rev. Plant Biol. 2014, 65, 365–384. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, S.; Du, W.; Wang, Y.; Wu, Y.; Li, W.; Ding, Y.; Wang, Y. G-protein couples MAPK cascade through maize heterotrimeric Gβ subunit. Plant Cell Rep. 2022, 41, 1763–1774. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Regan, M.; Furukawa, H.; Jackson, D. Role of heterotrimeric Gα proteins in maize development and enhancement of agronomic traits. PLoS Genet. 2018, 14, e1007374. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, T.; Liu, Y.; Ren, C.; Zhao, Y.; Wang, M. Isolation and characterization of gene encoding G protein α subunit protein responsive to plant hormones and abiotic stresses in Brassica napus. Mol. Biol. Rep. 2010, 37, 3957–3965. [Google Scholar] [CrossRef]
- Xie, Y.; Nan, Y.; Atif, A.; Hu, W.; Zhang, Y.; Tian, H.; Gao, Y. Molecular Identification of the G-Protein Genes and Their Expression Profiles in Response to Nitrogen Deprivation in Brassica napus. Int. J. Mol. Sci. 2022, 23, 8151. [Google Scholar] [CrossRef]
- Pham, V.N.; Kathare, P.K.; Huq, E. Phytochromes and Phytochrome Interacting Factors. Plant Physiol. 2018, 176, 1025–1038. [Google Scholar] [CrossRef]
- Yang, L.; Liu, S.; Lin, R. The role of light in regulating seed dormancy and germination. J. Integr. Plant Biol. 2020, 62, 1310–1326. [Google Scholar] [CrossRef]
- Klupczyńska, E.A.; Pawłowski, T.A. Regulation of Seed Dormancy and Germination Mechanisms in a Changing Environment. Int. J. Mol. Sci. 2021, 22, 1357. [Google Scholar] [CrossRef]
- Shu, K.; Liu, X.D.; Xie, Q.; He, Z.H. Two Faces of One Seed: Hormonal Regulation of Dormancy and Germination. Mol. Plant 2016, 9, 34–45. [Google Scholar] [CrossRef]
- Malamy, J.E. Intrinsic and environmental response pathways that regulate root system architecture. Plant Cell Environ. 2005, 28, 67–77. [Google Scholar] [CrossRef]
- Karlova, R.; Boer, D.; Hayes, S.; Testerink, C. Root plasticity under abiotic stress. Plant Physiol. 2021, 187, 1057–1070. [Google Scholar] [CrossRef]
- Ma, Q.; Xia, Z.; Cai, Z.; Li, L.; Cheng, Y.; Liu, J.; Nian, H. GmWRKY16 Enhances Drought and Salt Tolerance Through an ABA-Mediated Pathway in Arabidopsis thaliana. Front. Plant Sci. 2018, 9, 1979. [Google Scholar] [CrossRef] [PubMed]
- Zang, X.; Geng, X.; Liu, K.; Wang, F.; Liu, Z.; Zhang, L.; Zhao, Y.; Tian, X.; Hu, Z.; Yao, Y.; et al. Ectopic expression of TaOEP16-2-5B, a wheat plastid outer envelope protein gene, enhances heat and drought stress tolerance in transgenic Arabidopsis plants. Plant Sci. 2017, 258, 1–11. [Google Scholar] [CrossRef]
- Ju, Y.L.; Yue, X.F.; Min, Z.; Wang, X.H.; Fang, Y.L.; Zhang, J.X. VvNAC17, a novel stress-responsive grapevine (Vitis vinifera L.) NAC transcription factor, increases sensitivity to abscisic acid and enhances salinity, freezing, and drought tolerance in transgenic Arabidopsis. Plant Physiol. Biochem. 2020, 146, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.; Li, S.; Wang, Z.; Cheng, X.; Li, F.; Mei, F.; Chen, N.; Kang, Z. Regulatory changes in TaSNAC8-6A are associated with drought tolerance in wheat seedlings. Plant Biotechnol. J. 2020, 18, 1078–1092. [Google Scholar] [CrossRef] [PubMed]
- He, X.J.; Mu, R.L.; Cao, W.H.; Zhang, Z.G.; Zhang, J.S.; Chen, S.Y. AtNAC2, a transcription factor downstream of ethylene and auxin signaling pathways, is involved in salt stress response and lateral root development. Plant J. 2005, 44, 903–916. [Google Scholar] [CrossRef]
- Lee, D.K.; Chung, P.J.; Jeong, J.S.; Jang, G.; Bang, S.W.; Jung, H.; Kim, Y.S.; Ha, S.H.; Choi, Y.D.; Kim, J.K. The rice OsNAC6 transcription factor orchestrates multiple molecular mechanisms involving root structural adaptions and nicotianamine biosynthesis for drought tolerance. Plant Biotechnol. J. 2017, 15, 754–764. [Google Scholar] [CrossRef]
- Abid, M.; Tian, Z.; Ata-Ul-Karim, S.T.; Cui, Y.; Liu, Y.; Zahoor, R.; Jiang, D.; Dai, T. Nitrogen Nutrition Improves the Potential of Wheat (Triticum aestivum L.) to Alleviate the Effects of Drought Stress during Vegetative Growth Periods. Front. Plant Sci. 2016, 7, 981. [Google Scholar] [CrossRef]
- Nazarli, H.; Faraji, F. Response of proline, soluble sugars and antioxidant enzymes in wheat (Triticum aestivum L.) to different irrigation regimes in greenhouse condition. Cercet. Agron. Mold. (Agron. Res. Mold.) 2011, 44. [Google Scholar] [CrossRef]
- Cheng, X.; Yao, H.; Cheng, Z.; Tian, B.; Gao, C.; Gao, W.; Yan, S.; Cao, J.; Pan, X.; Lu, J.; et al. The Wheat Gene TaVQ14 Confers Salt and Drought Tolerance in Transgenic Arabidopsis thaliana Plants. Front. Plant Sci. 2022, 13, 870586. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Chan, Z. ROS Regulation During Abiotic Stress Responses in Crop Plants. Front. Plant Sci. 2015, 6, 1092. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Islam, M.N.; Sarker, S.; Tuteja, N.; Seraj, Z.I. Overexpression of heterotrimeric G protein beta subunit gene (OsRGB1) confers both heat and salinity stress tolerance in rice. Plant Physiol. Biochem. 2019, 144, 334–344. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).