Human Synovial Mesenchymal Stem Cells Expressed Immunoregulatory Factors IDO and TSG6 in a Context of Arthritis Mediated by Alphaviruses
Abstract
:1. Introduction
2. Results
2.1. ONNV Can Infect Human Synovial Tissue-Derived MSCs
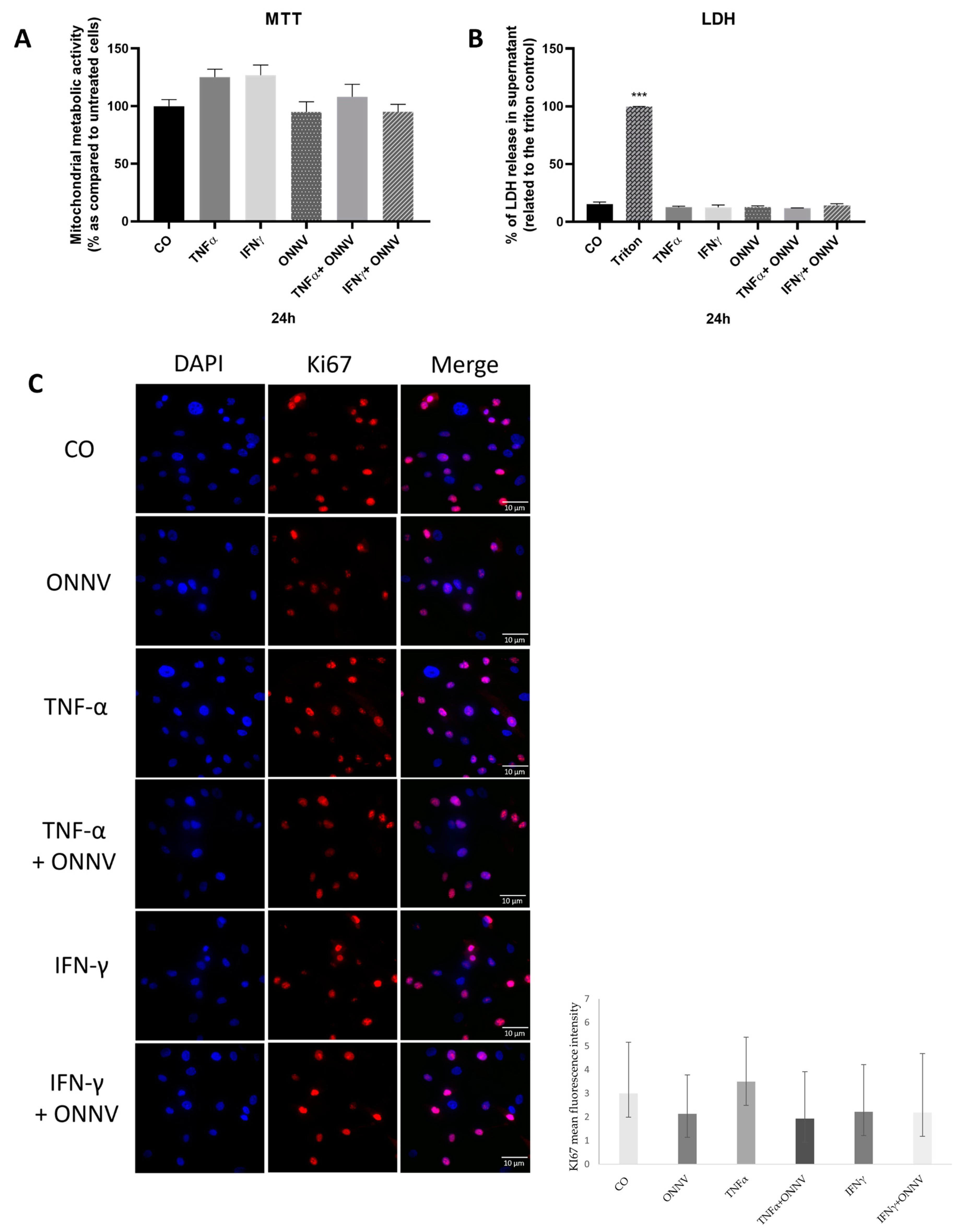
2.2. ONNV (MOI of 1) Alone or in Combination with Pro-Inflammatory Mediators Was Not Cytotoxic to Human Synovial MSCs and Did Not Affect Cell Proliferation
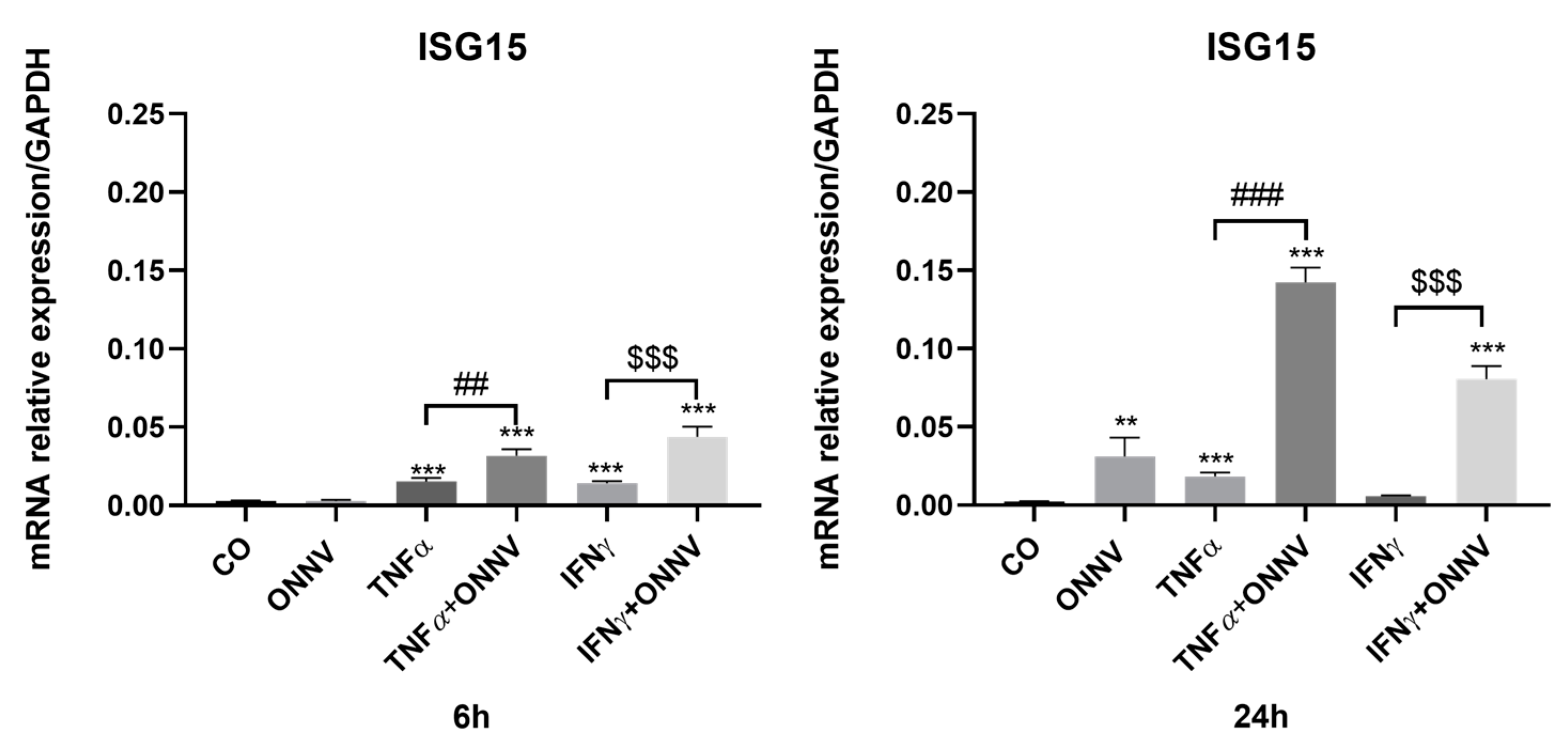
2.3. ONNV (MOI of 1) Alone or in Combination with Pro-Inflammatory Cytokines Promoted the Expression of the Antiviral ISG15 mRNA
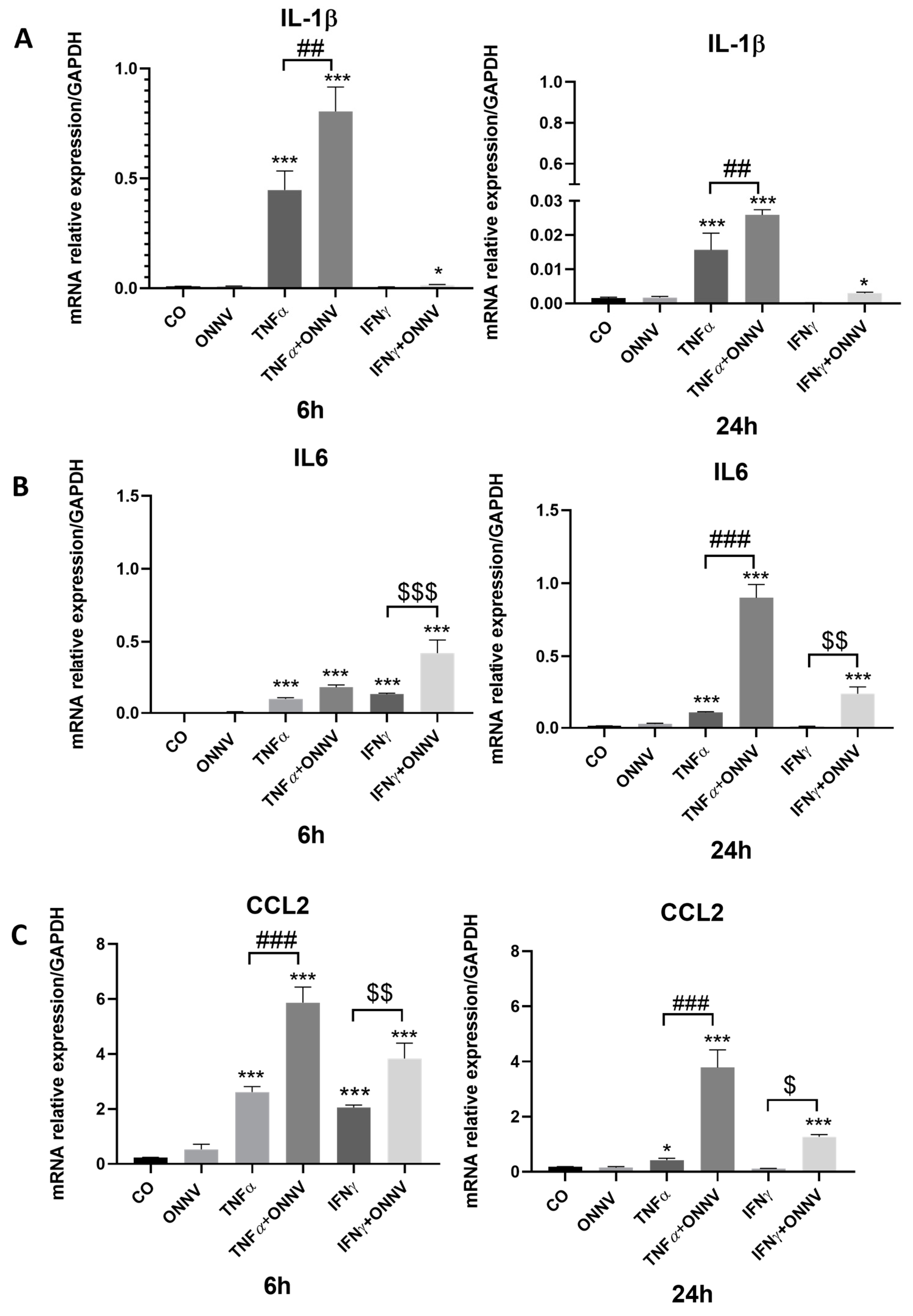
2.4. ONNV (MOI of 1) Alone Failed to Upregulate the Expression of Pro-Inflammatory Factors (IL1β, IL6, CCL2)
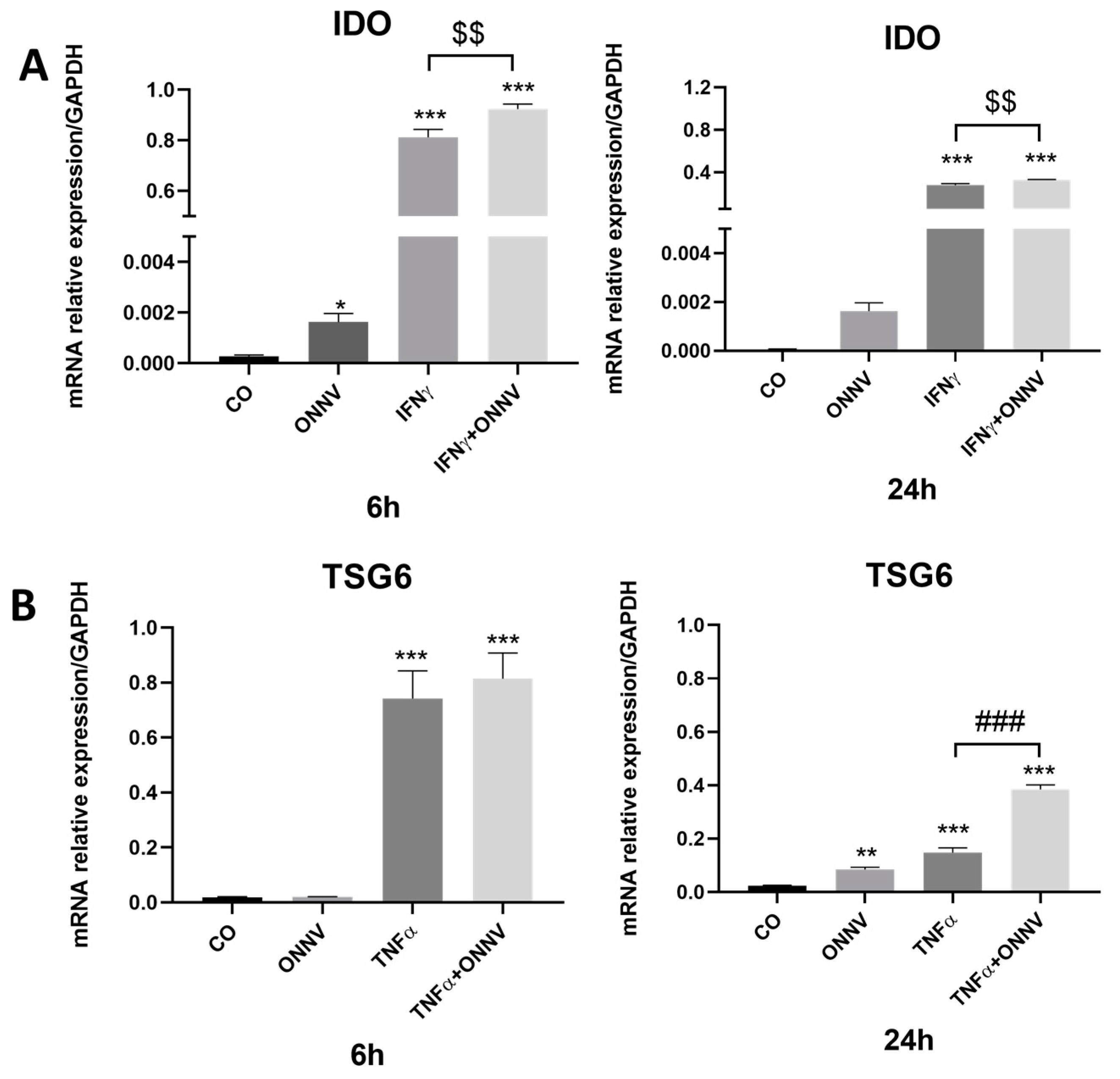
2.5. ONNV Alone and/or in the Presence of Pro-Inflammatory Mediators Can Upregulate the Expression of Immunoregulatory Factors IDO and TSG6 by Human Synovial MSCs
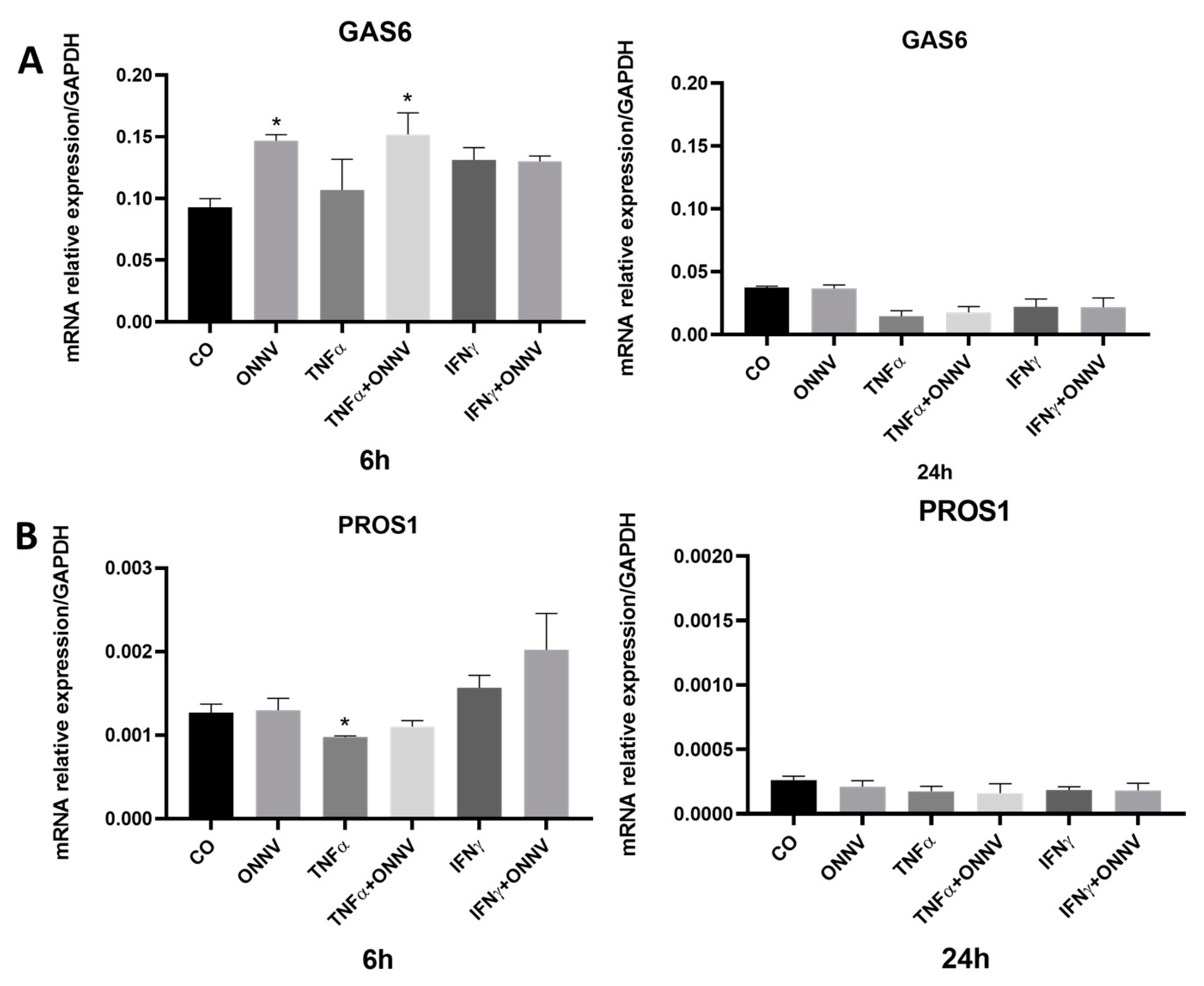
2.6. ONNV Infection Increases GAS6 Expression but Has No Effect on PROS1 Expression in Human Synovial Tissue-Derived MSCs
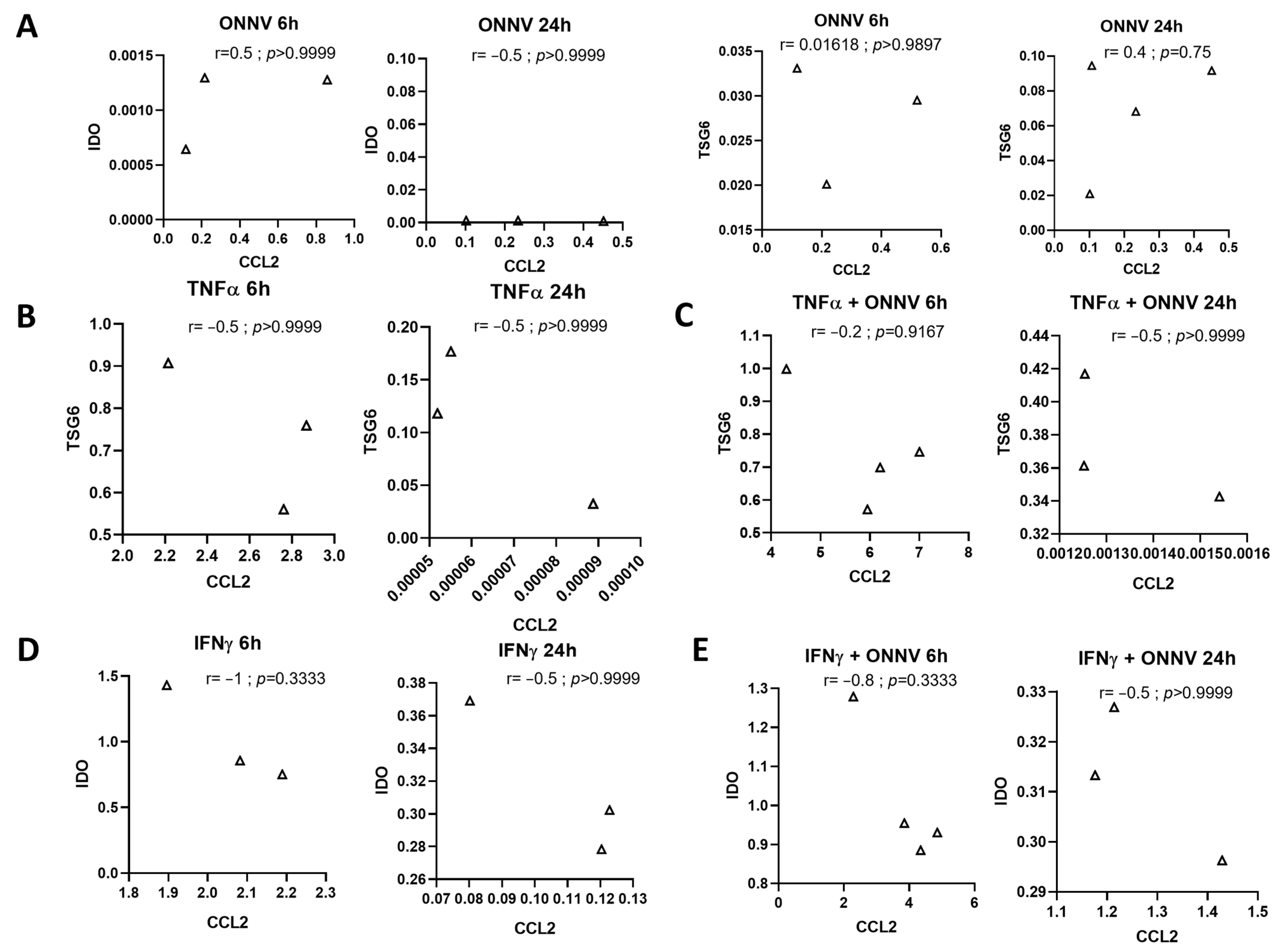
2.7. There Was No Correlation between Pro- and Anti-Inflammatory Responses
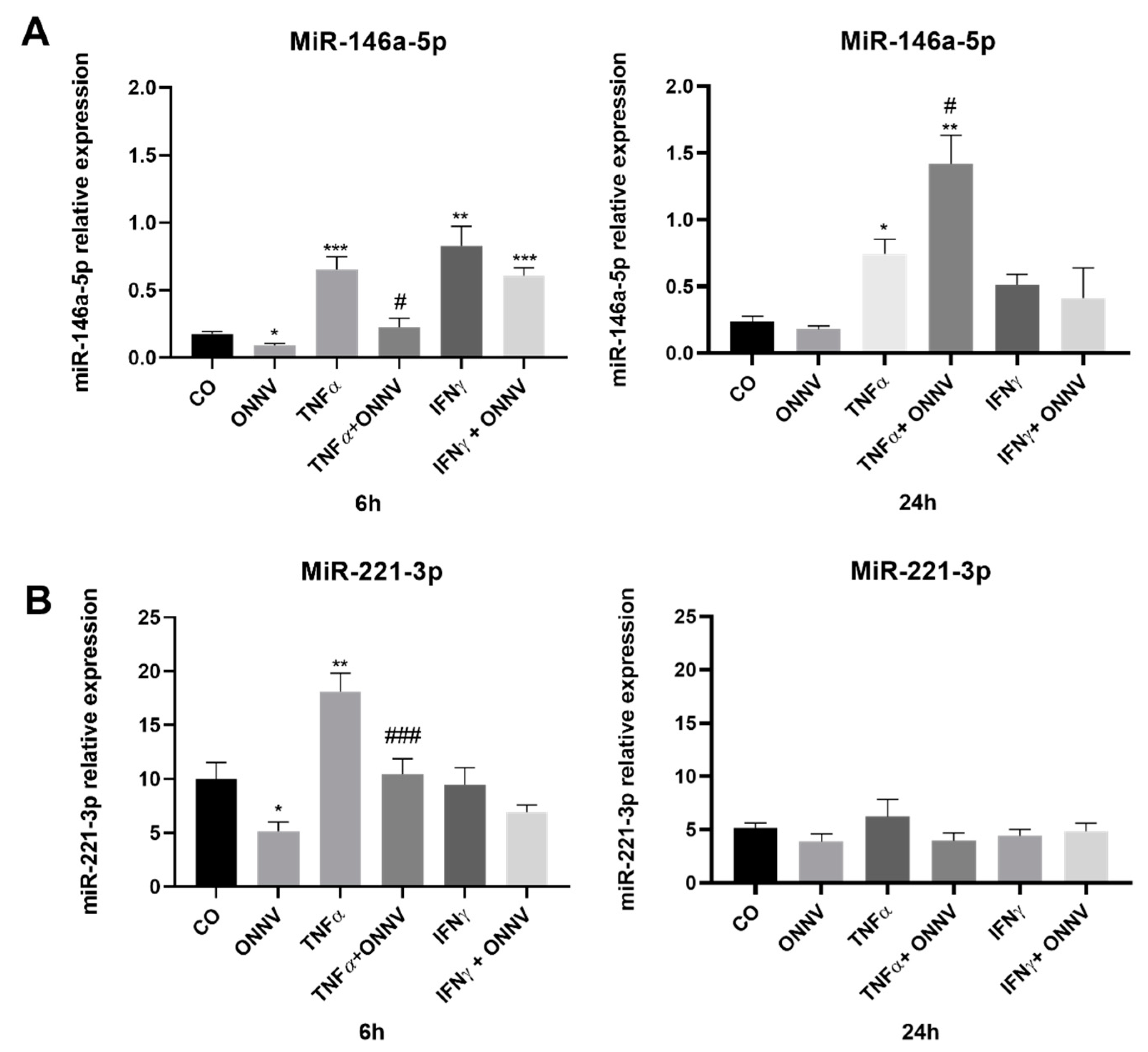
2.8. ONNV Infection Downregulated the Expression of Immunoregulatory miR-146a-5p in Human Synovial MSCs Treated with TNF-α
3. Discussion
4. Materials and Methods
4.1. Cell Culture Reagents and Viruses
4.2. Cell Treatments
4.3. Cytotoxicity Assay
4.4. Mitochondrial Metabolic Activity Measurements
4.5. RNA Extraction and qPCR
4.6. mi-RNA Extraction and RT-PCR
4.7. Immunofluorescence
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mostafavi, H.; Abeyratne, E.; Zaid, A.; Taylor, A. Arthritogenic Alphavirus-Induced Immunopathology and Targeting Host Inflammation as a Therapeutic Strategy for Alphaviral Disease. Viruses 2019, 11, 290. [Google Scholar] [CrossRef] [PubMed]
- Bartholomeeusen, K.; Daniel, M.; LaBeaud, D.A.; Gasque, P.; Peeling, R.W.; Stephenson, K.E.; Ng, L.F.P.; Ariën, K.K. Chikungunya Fever. Nat. Rev. Dis. Primers 2023, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Toivanen, A. Alphaviruses: An Emerging Cause of Arthritis? Curr. Opin. Rheumatol. 2008, 20, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Suhrbier, A.; La Linn, M. Clinical and Pathologic Aspects of Arthritis Due to Ross River Virus and Other Alphaviruses. Curr. Opin. Rheumatol. 2004, 16, 374–379. [Google Scholar] [CrossRef]
- Suhrbier, A.; Jaffar-Bandjee, M.C.; Gasque, P. Arthritogenic Alphaviruses—An Overview. Nat. Rev. Rheumatol. 2012, 8, 420–429. [Google Scholar] [CrossRef]
- Katze, M.G.; He, Y.; Gale, M. Viruses and Interferon: A Fight for Supremacy. Nat. Rev. Immunol. 2002, 2, 675–687. [Google Scholar] [CrossRef]
- Couderc, T.; Chretien, F.; Schilte, C.; Disson, O.; Brigitte, M.; Guivel-Benhassine, F.; Touret, Y.; Barau, G.; Cayet, N.; Schuffenecker, I.; et al. A Mouse Model for Chikungunya: Young Age and Inefficient Type-I Interferon Signaling Are Risk Factors for Severe Disease. PLoS Pathog. 2008, 4, e29. [Google Scholar] [CrossRef]
- Gardner, J.; Anraku, I.; Le, T.T.; Larcher, T.; Major, L.; Roques, P.; Schroder, W.A.; Higgs, S.; Suhrbier, A. Chikungunya Virus Arthritis in Adult Wild-Type Mice. J. Virol. 2010, 84, 8021–8032. [Google Scholar] [CrossRef]
- Simon, F.; Javelle, E.; Gasque, P. Chikungunya Virus Infections. N. Engl. J. Med. 2015, 373, 93–94. [Google Scholar] [CrossRef]
- Her, Z.; Malleret, B.; Chan, M.; Ong, E.K.; Wong, S.C.; Kwek, D.J.; Tolou, H.; Lin, R.T.; Tambyah, P.A.; Renia, L.; et al. Active Infection of Human Blood Monocytes by Chikungunya Virus Triggers an Innate Immune Response. J. Immunol. 2010, 184, 5903–5913. [Google Scholar] [CrossRef]
- Mateo, L.; La Linn, M.; McColl, S.R.; Cross, S.; Gardner, J.; Suhrbier, A. An Arthrogenic Alphavirus Induces Monocyte Chemoattractant Protein-1 and Interleukin-8. Intervirology 2000, 43, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Chow, A.; Her, Z.; Ong, E.K.S.; Chen, J.; Dimatatac, F.; Kwek, D.J.C.; Barkham, T.; Yang, H.; Rénia, L.; Leo, Y.-S.; et al. Persistent Arthralgia Induced by Chikungunya Virus Infection Is Associated with Interleukin-6 and Granulocyte Macrophage Colony-Stimulating Factor. J. Infect. Dis. 2011, 203, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Kelvin, A.A.; Banner, D.; Silvi, G.; Moro, M.L.; Spataro, N.; Gaibani, P.; Cavrini, F.; Pierro, A.; Rossini, G.; Cameron, M.J.; et al. Inflammatory Cytokine Expression Is Associated with Chikungunya Virus Resolution and Symptom Severity. PLoS Negl. Trop. Dis. 2011, 5, e1279. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Foo, S.S.; Sims, N.A.; Herrero, L.J.; Walsh, N.C.; Mahalingam, S. Arthritogenic Alphaviruses: New Insights into Arthritis and Bone Pathology. Trends Microbiol. 2014, 23, 35–43. [Google Scholar] [CrossRef]
- Jaffar-Bandjee, M.C.; Das, T.; Hoarau, J.J.; Krejbich Trotot, P.; Denizot, M.; Ribera, A.; Roques, P.; Gasque, P. Chikungunya Virus Takes Centre Stage in Virally Induced Arthritis: Possible Cellular and Molecular Mechanisms to Pathogenesis. Microbes Infect. 2009, 11, 1206–1218. [Google Scholar] [CrossRef]
- Zaid, A.; Rulli, N.E.; Rolph, M.S.; Suhrbier, A.; Mahalingam, S. Disease Exacerbation by Etanercept in a Mouse Model of Alphaviral Arthritis and Myositis. Arthritis Rheum. 2011, 63, 488–491. [Google Scholar] [CrossRef]
- Fros, J.J.; Liu, W.J.; Prow, N.A.; Geertsema, C.; Ligtenberg, M.; Vanlandingham, D.L.; Schnettler, E.; Vlak, J.M.; Suhrbier, A.; Khromykh, A.A.; et al. Chikungunya Virus Nonstructural Protein 2 Inhibits Type I/II Interferon-Stimulated JAK-STAT Signaling. J. Virol. 2010, 84, 10877–10887. [Google Scholar] [CrossRef]
- Thon-Hon, V.G.; Denizot, M.; Li-Pat-Yuen, G.; Giry, C.; Jaffar-Bandjee, M.-C.; Gasque, P. Deciphering the Differential Response of Two Human Fibroblast Cell Lines Following Chikungunya Virus Infection. Virol. J. 2012, 9, 213. [Google Scholar] [CrossRef]
- Krejbich-Trotot, P.; Gay, B.; Li-Pat-Yuen, G.; Hoarau, J.J.; Jaffar-Bandjee, M.C.; Briant, L.; Gasque, P.; Denizot, M. Chikungunya Triggers an Autophagic Process Which Promotes Viral Replication. Virol. J. 2011, 8, 432. [Google Scholar] [CrossRef]
- Pott, F.; Postmus, D.; Brown, R.J.P.; Wyler, E.; Neumann, E.; Landthaler, M.; Goffinet, C. Single-Cell Analysis of Arthritogenic Alphavirus-Infected Human Synovial Fibroblasts Links Low Abundance of Viral RNA to Induction of Innate Immunity and Arthralgia-Associated Gene Expression. Emerg. Microbes Infect. 2021, 10, 2151–2168. [Google Scholar] [CrossRef]
- Zhang, R.; Kim, A.S.; Fox, J.M.; Nair, S.; Basore, K.; Klimstra, W.B.; Rimkunas, R.; Fong, R.H.; Lin, H.; Poddar, S.; et al. Mxra8 Is a Receptor for Multiple Arthritogenic Alphaviruses. Nature 2018, 557, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Bačenková, D.; Trebuňová, M.; Morochovič, R.; Dosedla, E.; Findrik Balogová, A.; Gašparová, P.; Živčák, J. Interaction between Mesenchymal Stem Cells and the Immune System in Rheumatoid Arthritis. Pharmaceuticals 2022, 15, 941. [Google Scholar] [CrossRef] [PubMed]
- Milner, C.M.; Day, A.J. TSG-6: A Multifunctional Protein Associated with Inflammation. J. Cell Sci. 2003, 116, 1863–1873. [Google Scholar] [CrossRef] [PubMed]
- Munn, D.H.; Mellor, A.L. Indoleamine 2,3 Dioxygenase and Metabolic Control of Immune Responses. Trends Immunol. 2013, 34, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Cao, K.; Liu, K.; Xue, Y.; Roberts, A.I.; Li, F.; Han, Y.; Rabson, A.B.; Wang, Y.; Shi, Y. Kynurenic Acid, an IDO Metabolite, Controls TSG-6-Mediated Immunosuppression of Human Mesenchymal Stem Cells. Cell Death Differ. 2018, 25, 1209–1223. [Google Scholar] [CrossRef]
- Mittal, M.; Tiruppathi, C.; Nepal, S.; Zhao, Y.-Y.; Grzych, D.; Soni, D.; Prockop, D.J.; Malik, A.B. TNFα-Stimulated Gene-6 (TSG6) Activates Macrophage Phenotype Transition to Prevent Inflammatory Lung Injury. Proc. Natl. Acad. Sci. USA 2016, 113, E8151–E8158. [Google Scholar] [CrossRef]
- Dyer, D.P.; Salanga, C.L.; Johns, S.C.; Valdambrini, E.; Fuster, M.M.; Milner, C.M.; Day, A.J.; Handel, T.M. The Anti-Inflammatory Protein TSG-6 Regulates Chemokine Function by Inhibiting Chemokine/Glycosaminoglycan Interactions. J. Biol. Chem. 2016, 291, 12627–12640. [Google Scholar] [CrossRef]
- Lee, R.H.; Yu, J.M.; Foskett, A.M.; Peltier, G.; Reneau, J.C.; Bazhanov, N.; Oh, J.Y.; Prockop, D.J. TSG-6 as a Biomarker to Predict Efficacy of Human Mesenchymal Stem/Progenitor Cells (hMSCs) in Modulating Sterile Inflammation in Vivo. Proc. Natl. Acad. Sci. USA 2014, 111, 16766–16771. [Google Scholar] [CrossRef]
- van der Meer, J.H.M.; van der Poll, T.; van ’t Veer, C. TAM Receptors, Gas6, and Protein S: Roles in Inflammation and Hemostasis. Blood 2014, 123, 2460–2469. [Google Scholar] [CrossRef]
- Camenisch, T.D.; Koller, B.H.; Earp, H.S.; Matsushima, G.K. A Novel Receptor Tyrosine Kinase, Mer, Inhibits TNF-Alpha Production and Lipopolysaccharide-Induced Endotoxic Shock. J. Immunol. 1999, 162, 3498–3503. [Google Scholar] [CrossRef]
- Rothlin, C.V.; Ghosh, S.; Zuniga, E.I.; Oldstone, M.B.A.; Lemke, G. TAM Receptors Are Pleiotropic Inhibitors of the Innate Immune Response. Cell 2007, 131, 1124–1136. [Google Scholar] [CrossRef] [PubMed]
- van den Brand, B.T.; Abdollahi-Roodsaz, S.; Vermeij, E.A.; Bennink, M.B.; Arntz, O.J.; Rothlin, C.V.; van den Berg, W.B.; van de Loo, F.A.J. Therapeutic Efficacy of Tyro3, Axl, and MerTK Agonists in Collagen-Induced Arthritis. Arthritis Rheum. 2013, 65, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Payet, M.; Dargai, F.; Gasque, P.; Guillot, X. Epigenetic Regulation (Including Micro-RNAs, DNA Methylation and Histone Modifications) of Rheumatoid Arthritis: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 12170. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Pan, W.; Song, X.; Liu, Y.; Shao, X.; Tang, Y.; Liang, D.; He, D.; Wang, H.; Liu, W.; et al. The microRNA miR-23b Suppresses IL-17-Associated Autoimmune Inflammation by Targeting TAB2, TAB3 and IKK-α. Nat. Med. 2012, 18, 1077–1086. [Google Scholar] [CrossRef]
- Yang, S.; Yang, Y. Downregulation of microRNA-221 Decreases Migration and Invasion in Fibroblast-like Synoviocytes in Rheumatoid Arthritis. Mol. Med. Rep. 2015, 12, 2395–2401. [Google Scholar] [CrossRef]
- Quero, L.; Tiaden, A.N.; Hanser, E.; Roux, J.; Laski, A.; Hall, J.; Kyburz, D. miR-221-3p Drives the Shift of M2-Macrophages to a Pro-Inflammatory Function by Suppressing JAK3/STAT3 Activation. Front. Immunol. 2019, 10, 3087. [Google Scholar] [CrossRef]
- Cuffy, M.C.; Silverio, A.M.; Qin, L.; Wang, Y.; Eid, R.; Brandacher, G.; Lakkis, F.G.; Fuchs, D.; Pober, J.S.; Tellides, G. Induction of Indoleamine 2,3-Dioxygenase in Vascular Smooth Muscle Cells by Interferon-Gamma Contributes to Medial Immunoprivilege. J. Immunol. 2007, 179, 5246–5254. [Google Scholar] [CrossRef]
- Bernardo, M.E.; Fibbe, W.E. Mesenchymal Stromal Cells: Sensors and Switchers of Inflammation. Cell Stem Cell 2013, 13, 392–402. [Google Scholar] [CrossRef]
- English, K. Mechanisms of Mesenchymal Stromal Cell Immunomodulation. Immunol. Cell Biol. 2013, 91, 19–26. [Google Scholar] [CrossRef]
- Ren, G.; Zhang, L.; Zhao, X.; Xu, G.; Zhang, Y.; Roberts, A.I.; Zhao, R.C.; Shi, Y. Mesenchymal Stem Cell-Mediated Immunosuppression Occurs via Concerted Action of Chemokines and Nitric Oxide. Cell Stem Cell 2008, 2, 141–150. [Google Scholar] [CrossRef]
- Ren, G.; Su, J.; Zhang, L.; Zhao, X.; Ling, W.; L’huillie, A.; Zhang, J.; Lu, Y.; Roberts, A.I.; Ji, W.; et al. Species Variation in the Mechanisms of Mesenchymal Stem Cell-Mediated Immunosuppression. Stem Cells 2009, 27, 1954–1962. [Google Scholar] [CrossRef] [PubMed]
- Tas, S.W.; Vervoordeldonk, M.J.; Hajji, N.; Schuitemaker, J.H.N.; van der Sluijs, K.F.; May, M.J.; Ghosh, S.; Kapsenberg, M.L.; Tak, P.P.; de Jong, E.C. Noncanonical NF-kappaB Signaling in Dendritic Cells Is Required for Indoleamine 2,3-Dioxygenase (IDO) Induction and Immune Regulation. Blood 2007, 110, 1540–1549. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Yang, X.; Wu, J.; Tang, S.; Yang, L.; Fei, X.; Wang, M. Exosome from Indoleamine 2,3-Dioxygenase-Overexpressing Bone Marrow Mesenchymal Stem Cells Accelerates Repair Process of Ischemia/Reperfusion-Induced Acute Kidney Injury by Regulating Macrophages Polarization. Stem Cell Res. Ther. 2022, 13, 367. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yuan, Y.; Chen, H.; Dai, H.; Li, J. Indoleamine 2,3-Dioxygenase Mediates the Therapeutic Effects of Adipose-Derived Stromal/Stem Cells in Experimental Periodontitis by Modulating Macrophages through the Kynurenine-AhR-NRF2 Pathway. Mol. Metab. 2022, 66, 101617. [Google Scholar] [CrossRef]
- Mehraj, V.; Routy, J.-P. Tryptophan Catabolism in Chronic Viral Infections: Handling Uninvited Guests. Int. J. Tryptophan Res. 2015, 8, 41–48. [Google Scholar] [CrossRef]
- Hoarau, J.J.; Jaffar Bandjee, M.C.; Krejbich Trotot, P.; Das, T.; Li-Pat-Yuen, G.; Dassa, B.; Denizot, M.; Guichard, E.; Ribera, A.; Henni, T.; et al. Persistent Chronic Inflammation and Infection by Chikungunya Arthritogenic Alphavirus in Spite of a Robust Host Immune Response. J. Immunol. 2010, 184, 5914–5927. [Google Scholar] [CrossRef]
- Lee, T.H.; Wisniewski, H.-G.; Vilcek, J. A Novel Secretory Tumor Necrosis Factor-Inducible Protein (TSG-6) Is a Member of the Family of Hyaluronate Binding Proteins, Closely Related to the Adhesion Receptor CD44. J. Cell Biol. 1992, 116, 545–557. [Google Scholar] [CrossRef]
- Wisniewski, H.G.; Maier, R.; Lotz, M.; Lee, S.; Klampfer, L.; Lee, T.H.; Vilcek, J. TSG-6: A TNF-, IL-1-, and LPS-Inducible Secreted Glycoprotein Associated with Arthritis. J. Immunol. 1993, 151, 6593–6601. [Google Scholar] [CrossRef]
- Bayliss, M.T.; Howat, S.L.; Dudhia, J.; Murphy, J.M.; Barry, F.P.; Edwards, J.C.; Day, A.J. Up-Regulation and Differential Expression of the Hyaluronan-Binding Protein TSG-6 in Cartilage and Synovium in Rheumatoid Arthritis and Osteoarthritis. Osteoarthr. Cartil. 2001, 9, 42–48. [Google Scholar] [CrossRef]
- Mindrescu, C.; Dias, A.A.M.; Olszewski, R.J.; Klein, M.J.; Reis, L.F.L.; Wisniewski, H.-G. Reduced Susceptibility to Collagen-Induced Arthritis in DBA/1J Mice Expressing the TSG-6 Transgene. Arthritis Rheum. 2002, 46, 2453–2464. [Google Scholar] [CrossRef]
- Bárdos, T.; Kamath, R.V.; Mikecz, K.; Glant, T.T. Anti-Inflammatory and Chondroprotective Effect of TSG-6 (Tumor Necrosis Factor-α-Stimulated Gene-6) in Murine Models of Experimental Arthritis. Am. J. Pathol. 2001, 159, 1711–1721. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wu, Y.-H.; Zhang, L.; Xue, B.; Wang, Y.; Liu, B.; Liu, X.-Y.; Zuo, F.; Yang, X.-Y.; Chen, F.-Y.; et al. MicroRNA-146a Suppresses Rheumatoid Arthritis Fibroblast-like Synoviocytes Proliferation and Inflammatory Responses by Inhibiting the TLR4/NF-kB Signaling. Oncotarget 2018, 9, 23944–23959. [Google Scholar] [CrossRef] [PubMed]
- Selvamani, S.P.; Mishra, R.; Singh, S.K. Chikungunya Virus Exploits miR-146a to Regulate NF-κB Pathway in Human Synovial Fibroblasts. PLoS ONE 2014, 9, e103624. [Google Scholar] [CrossRef] [PubMed]
- Stanczyk, J.; Pedrioli, D.M.L.; Brentano, F.; Sanchez-Pernaute, O.; Kolling, C.; Gay, R.E.; Detmar, M.; Gay, S.; Kyburz, D. Altered Expression of MicroRNA in Synovial Fibroblasts and Synovial Tissue in Rheumatoid Arthritis. Arthritis Rheum. 2008, 58, 1001–1009. [Google Scholar] [CrossRef]


| Target Gene | Forward Sequence (5′–3′) | Reverse Sequence (3′–5′) |
|---|---|---|
| GAPDH | TGCGTCGCCAGCCGAG | AGTTAAAAGCAGCCCTGGTGA |
| GAS6 | ACGACCCCGAGACGGATTAT | CTTCCTATCGCAGGGGTTGG |
| IDO | AGCCCCTGACTTATGAGAACA | AGCTATTTCCAACAGCGCCT |
| MXRA8 | TACTGTGGCCTGCACGAAC | CTCTCGGGGACGATGACATT |
| ONNV Capsid | TAGAACACGCCCGTCGTATG | GCAACGCCTTCAGAAACGC |
| ONNV E1 | CACCGTCCCCGTACGTAAAA | GGCTCTGTAGGCTGATGCAA |
| ONNV E2 | CCCCTGACTACACGCTGATG | CCTTCATTGGAGCCGTCACA |
| PROS1 | GTCTCAGAGGCAAACTTTTGTT | AGAATTTGCACGACGCTTCC |
| TSG6 | TGGCTTTGTGGGAAGATACYGT | TGGAAACCTCCAGCTGTCAC |
| Target mi-RNAs | Forward (5′–3′) |
|---|---|
| hsa-miR-146a-5p | GCAGTGAGAACTGAATTCCATG |
| hsa-miR-221-3p | GCAGAGCTACATTGTCTGCT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Payet, M.; Septembre-Malaterre, A.; Gasque, P.; Guillot, X. Human Synovial Mesenchymal Stem Cells Expressed Immunoregulatory Factors IDO and TSG6 in a Context of Arthritis Mediated by Alphaviruses. Int. J. Mol. Sci. 2023, 24, 15932. https://doi.org/10.3390/ijms242115932
Payet M, Septembre-Malaterre A, Gasque P, Guillot X. Human Synovial Mesenchymal Stem Cells Expressed Immunoregulatory Factors IDO and TSG6 in a Context of Arthritis Mediated by Alphaviruses. International Journal of Molecular Sciences. 2023; 24(21):15932. https://doi.org/10.3390/ijms242115932
Chicago/Turabian StylePayet, Melissa, Axelle Septembre-Malaterre, Philippe Gasque, and Xavier Guillot. 2023. "Human Synovial Mesenchymal Stem Cells Expressed Immunoregulatory Factors IDO and TSG6 in a Context of Arthritis Mediated by Alphaviruses" International Journal of Molecular Sciences 24, no. 21: 15932. https://doi.org/10.3390/ijms242115932
APA StylePayet, M., Septembre-Malaterre, A., Gasque, P., & Guillot, X. (2023). Human Synovial Mesenchymal Stem Cells Expressed Immunoregulatory Factors IDO and TSG6 in a Context of Arthritis Mediated by Alphaviruses. International Journal of Molecular Sciences, 24(21), 15932. https://doi.org/10.3390/ijms242115932






