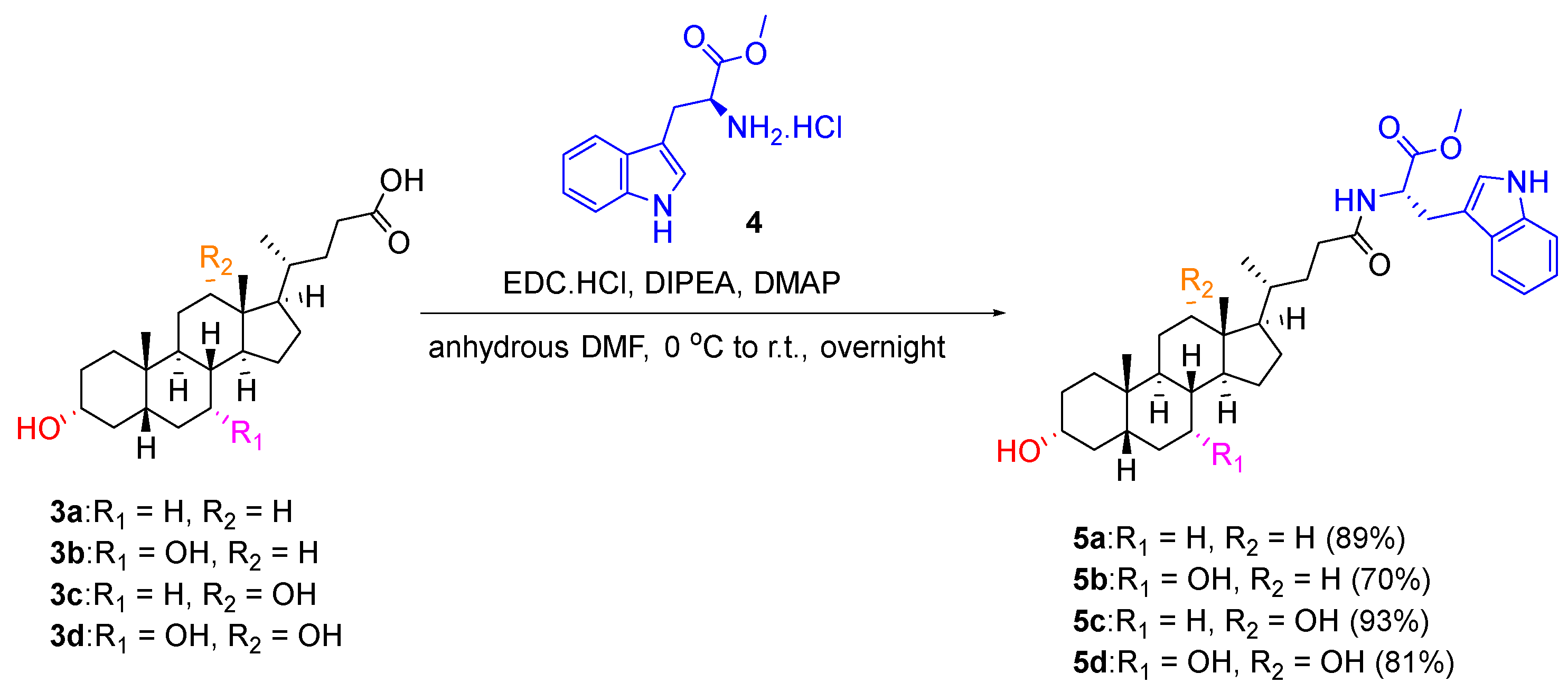
3.1. Synthesis of Analogues
All commercially available reagents were purchased from standard suppliers (Sigma Aldrich, St Louis, MO, USA and Alfa-Aesar, Ward Hill, MA, USA) and were used without further purification. All reactions were performed under anhydrous conditions with an atmosphere of nitrogen and anhydrous solvent (as required). Anhydrous solvents were obtained using PureSolv MD Solvent Purification System. Reactions were monitored by thin-layer chromatography precoated with Merck silica gel 60 F254 and visualization was performed by using short or long wavelength of ultraviolet light. Flash chromatography was performed using Grace Davisil LC60A silica.
Melting points were measured using an OptiMelt melting point apparatus and were uncorrected. 1H and 13C NMR spectra were obtained in the specified solvents on a Bruker Avance III 300, Bruker Avance III HD 400 or Bruker Avance III 600 Cryo spectrometer. Chemical shifts (δ) are in parts per million (ppm), internally referenced to the solvent nuclei. Multiplicities are assigned as singlet (s), broad singlet (bs), doublet (d), triplet (t), quartet (q), quintet (quint), sextet (sext), septet (sept), multiplet (m) or a combination of these (e.g., dd, dt, td), and coupling constants (J) are described in Hertz (Hz). Infrared (IR) spectra were recorded using a Cary 630 FTIR spectrometer fitted with a diamond attenuated total reflectance (ATR) sample interface. Low-resolution mass spectrometry was performed using a Thermo Fisher LCQ Mass Spectrometer while high-resolution mass spectrometry (HRMS) was performed by the UNSW Bioanalytical Mass Spectrometry Facility using a Thermo LTQ Orbitrap XL instrument.
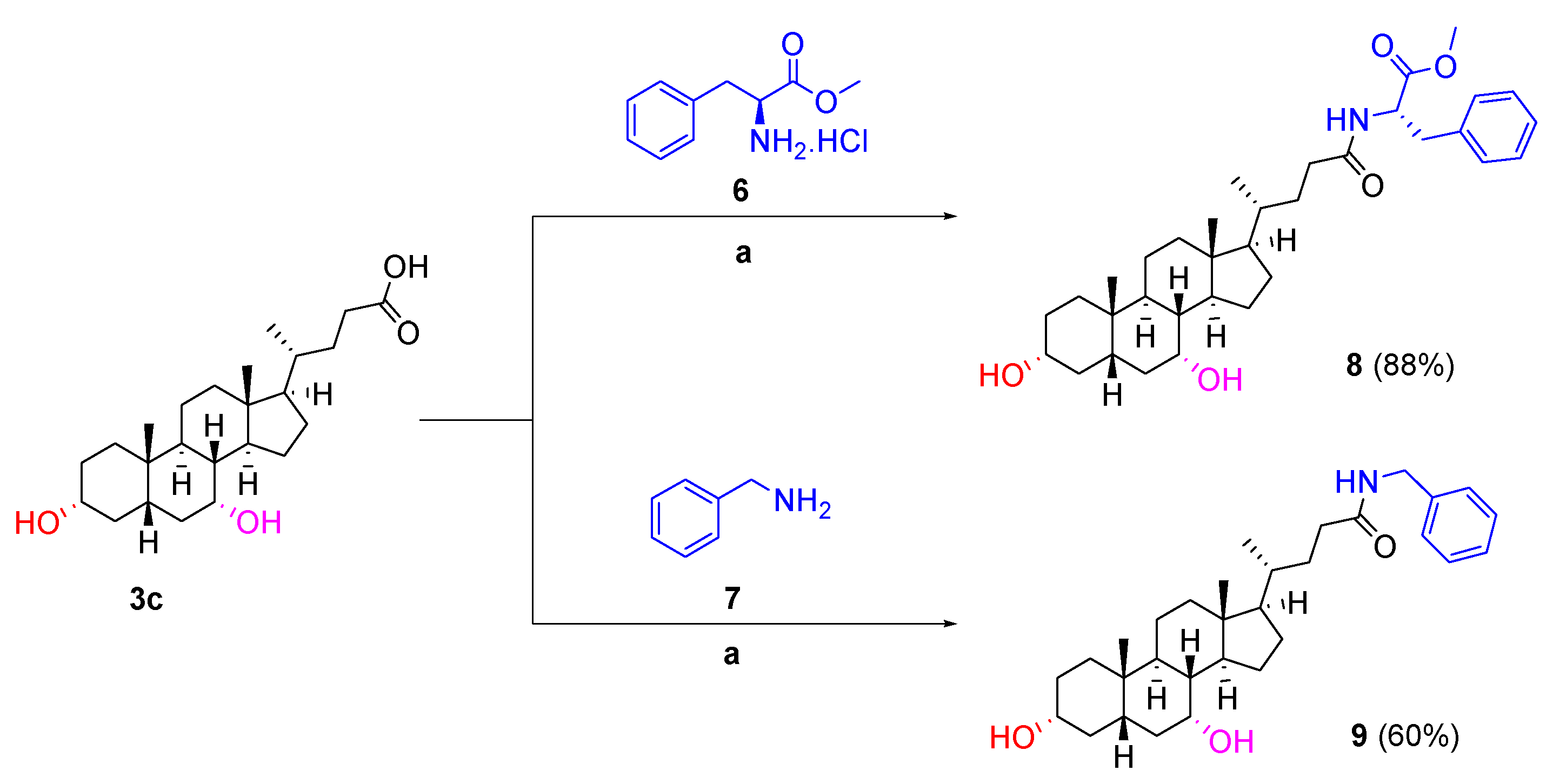
3.1.1. General Synthetic Procedure A for Coupling of Hydrophobic Groups
To a solution of appropriate cholic acid derivative (1.0 equivalent) in DMF (3 mL), EDC.HCl (1.1 equivalent) and DIPEA (2.0 equivalents) were added portion-wise at room temperature under nitrogen atmosphere. A solution of L-tryptophan methyl ester hydrochloride, L-phenylalanine methyl ester hydrochloride or benzylamine (1.5 equivalents) and DMAP (0.3 equivalent) in DMF (2 mL) was then added dropwise to the reaction mixture at 0 °C. The reaction mixture was stirred at room temperature overnight. After the completion of the reaction, the reaction mixture was poured into water (50 mL). The white precipitant was collected via vacuum filtration, redissolved in DCM (25 mL) and washed with NaHCO3 (2 × 50 mL), 2 M HCl (2 × 50 mL) and, finally, brine. The organic phase was then dried over anhydrous sodium sulfate and concentrated in vacuo to afford the product.
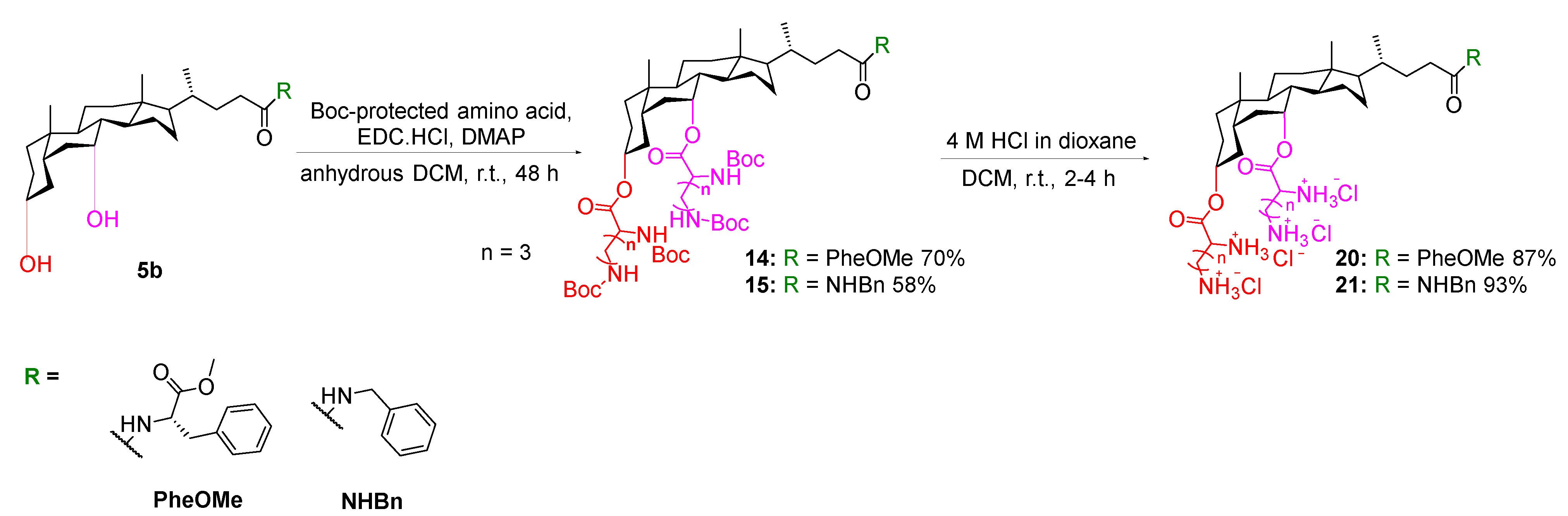
3.1.2. General Synthetic Procedure B for N-Boc-Protected Esters
To a solution of substituted cholic acid derivatives (1 equivalent) in DCM (5 mL), EDC.HCl (1.5–8 equivalents), Boc-protected amino acids (1.5–8 equivalents) and DMAP (1.5–8 equivalents) was added portion-wise at room temperature under nitrogen atmosphere. The resulting suspension was stirred at room temperature for 16–96 h. After completion of reaction, water (100 mL) was added to the reaction mixture. The organic layer was extracted with DCM (3 × 25 mL), washed with brine (50 mL), dried over anhydrous sodium sulfate and concentrated in vacuo to provide the crude product. The crude product was then purified by flash column chromatography on silica using ethyl acetate/chloroform (25:75) as eluents to afford the N-Boc-protected esters.
3.1.3. General Synthetic Procedure C for HCl Salts
To the Boc-protected product in DCM (10 mL), 4 M HCl in dioxane (3 mL) was added and the reaction mixture was stirred at room temperature for 2–4 h. After completion of reaction, the reaction mixture was concentrated in vacuo, triturated with diethyl ether and freeze-dried to afford the product as a HCl salt.
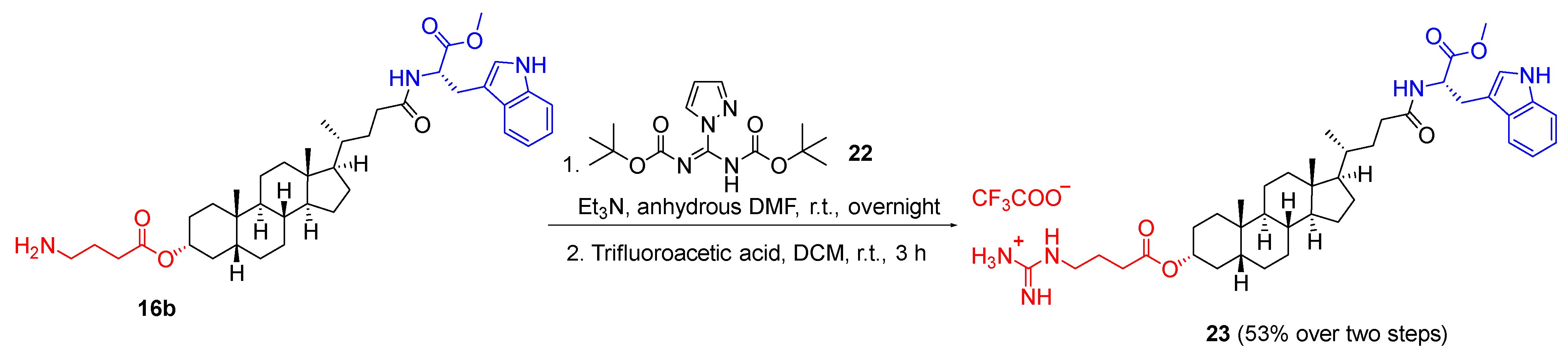
3.1.4. General Synthetic Procedure D for Guanidinium Salt Compound
To a solution of GABA-substituted lithocholic ester (1.0 equivalent) and N,N′-di-Boc-1H-pyrazole-1-carboxamidine (1.3 equivalents) in anhydrous DMF (2 mL), triethylamine (2.5 equivalents) was added dropwise at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature overnight. After the completion of the reaction, the reaction mixture was added into an ice-water mixture (50 mL) and the precipitate formed was collected by vacuum filtration to provide the crude product. The crude was then purified by flash column chromatography on silica using ethyl acetate/n-hexane (20–50%) to afford the Boc-protected product. To the Boc-protected product in DCM (5 mL), TFA (5 mL) was added at 0 °C. The reaction was stirred at room temperature for 3 h. After completion of reaction, the reaction mixture was concentrated in vacuo, triturated with diethyl ether and freeze-dried to afford the product as a TFA salt.
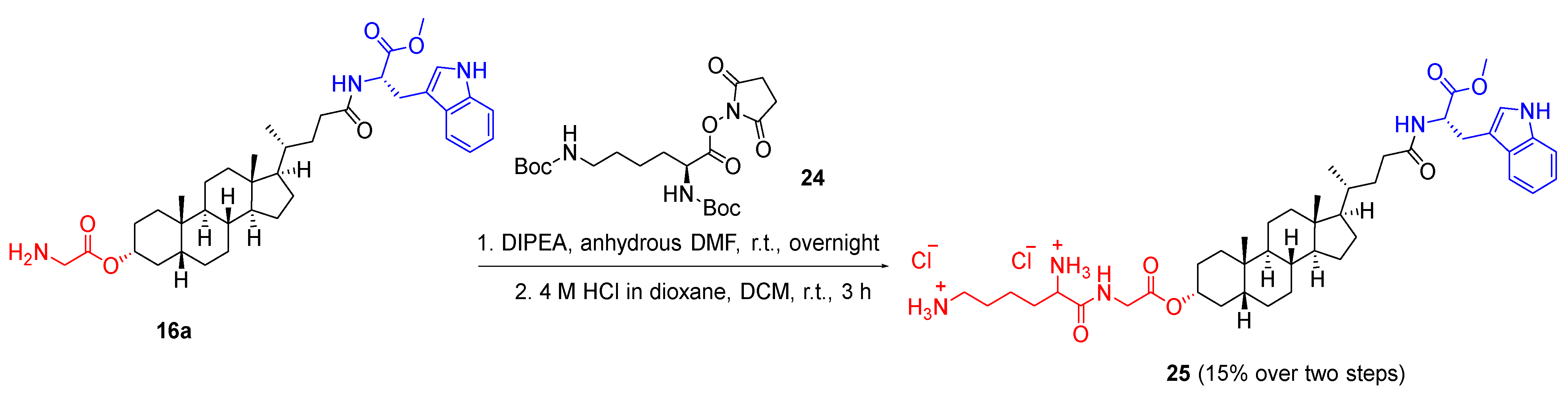
3.1.5. General Synthetic Procedure E for Glycine-Lysine Monoester
To a solution of glycine-substituted lithocholic ester (1.0 equivalent) in DMF (3 mL), N,N′-di-Boc-L-lysine hydroxysuccinimide ester (1.2 equivalents) and DIPEA (4.0 equivalents) was added dropwise at 0 °C under nitrogen atmosphere. The reaction mixture was then stirred at room temperature overnight. After the completion of reaction, the reaction mixture was added to water (50 mL) and the white precipitant formed was collected via vacuum filtration, redissolved in DCM (25 mL), washed with brine (50 mL), dried over anhydrous sodium sulfate and concentrated in vacuo to provide the crude product. The crude product was purified by flash column chromatography on silica using ethyl acetate/n-hexane (25:75) to yield the Boc-protected product. To the Boc-protected product in DCM (10 mL), 4 M HCl in dioxane (3 mL) was added and stirred at room temperature for 4 h. After completion of reaction, the reaction mixture was concentrated in vacuo, triturated with diethyl ether and freeze-dried to afford the product as a HCl salt.
3.1.6. General Synthetic Procedure F for Glycine-Valine Dipeptide
To a solution of glycine-substituted cholic acid triester (1.0 equivalent) in anhydrous DCM (3 mL), Boc-Val-OH (6.0 equivalents), EDC.HCl (6.0 equivalent), HOBt (3.0 equivalents) and DIPEA (5.0 equivalents) was added portion-wise at 0 °C under nitrogen atmosphere. The reaction was stirred at room temperature for 48 h. After the completion of the reaction, water (100 mL) was added to the reaction mixture and the product was extracted into DCM (3 × 25 mL), washed with saturated NaHCO3 (50 mL), 2 M HCl (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate and concentrated in vacuo to give the crude product. The crude was purified by flash chromatography on silica using methanol/DCM (3%) as eluent to afford the Boc-protected product. To the Boc-protected product in DCM (10 mL), 4 M HCl in dioxane (3 mL) was added and stirred at room temperature for 4 h. After completion of the reaction, the reaction mixture was concentrated in vacuo, triturated with diethyl ether and freeze-dried to afford the product as a HCl salt.
3.1.7. 2-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-2-oxoethan-1-aminium chloride (16a)
The title compound was synthesised from 10a (0.300 g, 0.458 mmol) following general synthetic procedure C. The product 16a was obtained as a white solid (0.195 g, 71%); mp 228.1 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.4 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 0.96–3.13 (m, CH, CH2 steroid), 2.97–3.18 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.76 (q, J = 5.6 Hz, 2H, -CO-CH2-NH3), 4.48 (td, J = 8.1, 5.8 Hz, 1H, -CO-CH-NH), 4.76 (tt, J = 11.1, 4.7 Hz, 1H, OCH), 6.98 (ddd, J = 8.0, 6.9, 1.1 Hz, 1H, ArH), 7.07 (ddd, J = 8.1, 6.9, 1.2 Hz, 1H, ArH), 7.14 (d, J = 2.4 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.49 (d, J = 7.8 Hz, 1H, ArH), 8.25 (d, J = 7.5 Hz, 1H, NH), 8.38 (d, J = 5.8 Hz, 3H, NH3), 10.91 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1, 167.5, 136.6, 127.5, 124.1, 121.4, 118.8, 118.5, 111.9, 110.1, 76.1, 66.8, 56.4, 56.1, 53.6, 52.2, 42.7, 41.5, 40.0, 39.8, 35.8, 35.4, 34.8, 34.6, 32.5, 32.2, 31.8, 28.2, 27.5, 27.0, 26.6, 26.4, 24.3, 23.4, 20.9, 18.8, 12.3; νmax 3389, 2930, 2863, 2627, 2343, 1734, 1519, 1439, 1369, 1119, 1044, 973, 910, 745 cm−1; HRMS (+ESI): Found m/z 634.4214, [M]+, C38H56N3O5 required 634.4215.
3.1.8. 4-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-4-oxobutan-1-aminium chloride (16b)
The title compound was synthesised from 10b (0.273 g, 0.358 mmol) following general synthetic procedure C. The product 16b was obtained as a white solid (0.198 g, 79%); mp 161.3 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.4 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 0.95–2.79 (m, CH, CH2, steroid), 3.02–3.14 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 4.48 (td, J = 8.1, 5.8 Hz, 1H, -CO-CH-NH), 4.65 (td, J = 11.3, 10.6, 4.9 Hz, 1H, -O-CH), 6.98 (ddd, J = 8.0, 7.0, 1.1 Hz, 1H, ArH), 7.07 (ddd, J = 8.1, 6.9, 1.2 Hz, 1H, ArH), 7.14 (d, J = 2.4 Hz, 1H, ArH), 7.34 (dt, J = 8.1, 0.9 Hz, 1H, ArH), 7.49 (d, J = 7.8 Hz, 1H, ArH), 8.00 (s, 3H, NH3), 8.24 (d, J = 7.6 Hz, 1H, NH), 10.90 (d, J = 2.5 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1 170.2, 136.6, 127.5, 124.1, 121.4, 118.8, 118.5, 111.9, 110.1, 74.8, 56.3, 56.1, 55.4, 53.6, 52.2, 42.7, 41.7, 40.0, 35.8, 35.3, 35.1, 34.9, 34.6, 32.5, 32.3, 32.1, 31.8, 31.0, 28.2, 27.3, 27.1, 26.7, 26.4, 24.3, 23.5, 20.9, 18.8, 12.3; νmax 3390, 2928, 2863, 2321, 1934, 1721, 1665, 1521, 1440, 1356, 1294, 1204, 1118, 1020, 858 cm−1; HRMS (+ESI): Found m/z 662.4527, [M]+, C40H60N3O5 required 663.4528.
3.1.9. (S)-5-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-5-oxopentane-1,4-diaminium chloride (16c)
The title compound was synthesised from 10c (0.350 g, 0.393 mmol) following general synthetic procedure C. The product 16c was obtained as a white solid (0.180 g, 67%); mp 185.7 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.3 Hz, 3H, CH3), 0.92 (s, 3H, CH3), 0.97–2.79 (m, CH, CH2, steroid), 2.99–3.17 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.97 (t, J = 6.3 Hz, 1H, -CO-CH-NH3), 4.48 (td, J = 8.1, 5.7 Hz, 1H, -CO-CH-CH2), 4.75 (tt, J = 11.2, 4.6 Hz, 1H, OCH), 6.98 (t, J = 7.4 Hz, 1H, ArH), 7.06 (t, J = 7.5 Hz, 1H, ArH), 7.14 (d, J = 2.3 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.48 (d, J = 7.8 Hz, 1H, ArH), 8.25 (s, 1H, NH), 8.33 (m, 6H, NH), 10.93 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1, 169.3, 136.6, 127.5, 124.1, 121.4, 118.8, 118.4, 111.9, 110.0, 76.3, 56.4, 56.1, 53.6, 52.2, 52.2, 51.9, 42.7, 41.6, 40.2, 38.5, 35.8, 35.4, 34.8, 34.6, 32.5, 32.2, 31.8, 28.2, 27.7, 27.5, 27.1, 26.6, 26.5, 24.3, 23.4, 23.1, 20.9, 18.8, 12.3; IR (ATR): νmax 3389, 2929, 2861, 2647, 2320, 2192, 2087, 1869, 1729, 1664, 1521, 1456, 1356, 1231, 1775, 1097, 933, 860 cm−1. HRMS (+ESI): Found m/z 692.4828, [M]+2, C41H62N4O5 required 692.4866.
3.1.10. (S)-6-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-6-oxohexane-1,5-diaminium chloride (16d)
The title compound was synthesised from 10d (0.527 g, 0.582 mmol) following general synthetic procedure C. The product 16d was obtained as a white solid (0.416 g, 96%); mp 205.7 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.4 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 1.02–2.75 (m, CH, CH2, steroid), 2.98–3.17 (m, 2H, -CO-CH-CH2), 3.48 (s, 3H, OCH3), 3.92 (q, J = 5.8 Hz, 1H, -CO-CH-NH3), 4.48 (td, J = 8.1, 5.8 Hz, 1H, -CO-CH-NH), 4.76 (tt, J = 11.0, 4.6 Hz, 1H, OCH), 6.94–7.02 (m, 1H, ArH), 7.07 (ddd, J = 8.2, 6.9, 1.2 Hz, 1H, ArH), 7.14 (d, J = 2.3 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.48 (d, J = 7.9 Hz, 1H, ArH), 8.10 (d, J = 7.5 Hz, 3H, NH3), 8.26 (d, J = 7.5 Hz, 1H, NH), 8.57–8.67 (m, 3H, NH3), 10.93 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (75 MHz, DMSO-d6) δ 173.2, 173.1, 169.3, 136.6, 127.5, 124.1, 121.4, 118.8, 118.4, 111.9, 110.0, 76.2, 66.8, 56.4, 56.1, 53.6, 52.2, 52.1, 52.1, 42.2, 41.6, 40.0, 38.6, 35.8, 35.4, 34.8, 34.6, 32.5, 32.2, 31.8, 29.8, 28.2, 27.5, 27.5, 27.1, 26.6, 24.3, 23.4, 21.6, 20.9, 18.8, 12.3; IR (ATR): νmax 2857, 2281, 2171, 2109, 1722, 619, 1494, 1439, 1364, 1223, 1124, 1067, 1001, 892 cm−1; HRMS (+ESI): Found m/z 705.4953, [M]2+, C42H64N4O5 required 705.4950.
3.1.11. 2,2′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7-diyl)bis(oxy))bis(2-oxoethan-1-aminium) chloride (17a)
The title compound was synthesised from 11a (0.300 g, 0.331 mmol) following general synthetic procedure C. The product 17a was obtained as a white solid (0.218 g, 85%); mp 253.1 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.61 (s, 3H, CH3), 0.87 (d, J = 6.4 Hz, 3H, CH3), 0.92 (s, 3H, CH3), 1.02–2.11 (m, CH, CH2, steroid), 2.97–3.17 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.75–3.93 (m, 4H, 2 × -CO-CH2-NH3), 4.47 (td, J = 8.2, 5.7 Hz, 1H, -CO-CH-NH), 4.63 (dd, J = 10.5, 5.6 Hz, 1H, OCH), 4.92 (d, J = 3.6 Hz, 1H, OCH), 6.94–7.02 (m, 1H, ArH), 7.07 (ddd, J = 8.2, 7.0, 1.2 Hz, 1H, ArH), 7.14 (d, J = 2.3 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.48 (d, J = 7.8 Hz, 1H, ArH), 8.25 (d, J = 7.5 Hz, 1H, NH), 8.44 (d, J = 9.9 Hz, 6H, 3 × NH3), 10.91 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1, 167.7, 167.4, 136.6, 127.5, 124.1, 121.4, 118.8, 118.4, 111.9, 110.1, 75.8, 55.9, 53.6, 52.2, 50.1, 42.8, 37.5, 35.3, 34.8, 34.0, 27.5, 22.6, 18.8, 12.0; IR (ATR): νmax 3181, 2928, 2861, 2621, 1734, 1651, 1498, 1434, 1367, 1230, 1118, 892 cm−1; HRMS (+ESI): Found m/z 354.2230, [M]2+, C40H58N4O7 required 354.2226.
3.1.12. 4,4′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7-diyl)bis(oxy))bis(4-oxobutan-1-aminium) chloride (17b)
The title compound was synthesised from 11b (0.760 g, 0.789 mmol) following general synthetic procedure C. The product 17b was obtained as a white solid (0.467 g, 71%); mp 149.0 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.3 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 1.09–2.51 (m, CH, CH2, steroid), 2.80 (t, J = 6.9 Hz, 4H, 2 × -CH2-NH3), 2.98–3.17 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 4.50 (dtd, J = 21.5, 9.1, 8.0, 4.8 Hz, 2H, -OCH, -CO-CH-NH), 4.80 (q, J = 3.0 Hz, 1H, -OCH), 6.98 (t, J = 7.4 Hz, 1H, ArH), 7.02–7.11 (m, 1H, ArH), 7.15 (d, J = 2.4 Hz, 1H, ArH), 7.34 (d, J = 8.0 Hz, 1H, ArH), 7.48 (d, J = 7.8 Hz, 1H, ArH), 8.15 (d, J = 8.9 Hz, 6H, 2 × NH3), 8.28 (d, J = 7.5 Hz, 1H, NH), 10.93 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1, 172.1, 172.0, 136.6, 127.5, 124.1, 121.4, 118.8, 118.4, 111.9, 110.1, 74.0, 71.4, 65.4, 55.8, 53.6, 52.2, 50.6, 42.7, 40.6, 40.4, 40.2, 40.0, 39.8, 39.6, 39.4, 38.6, 38.5, 37.6, 35.3, 34.8, 34.8, 34.2, 32.4, 31.6, 31.3, 27.5, 23.0, 22.8, 18.8, 15.6, 12.0; νmax 3745, 2954, 2832, 2475, 2285, 2103, 2177, 2103, 2047, 1995, 1944, 1719, 1589, 1492, 1236, 1068, 1128, 991, 916 cm−1; HRMS (+ESI): Found m/z 763.5000, [M]+, C44H66N4O7 required 763.5004.
3.1.13. (4S,4′S)-5,5′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7-diyl)bis(oxy))bis(5-oxopentane-1,4-diaminium) chloride (17c)
The title compound was synthesised from 11c (0.511 g, 0.418 mmol) following general synthetic procedure C. The product 17c was obtained as a white solid (0.305 g, 75%); mp 235.9 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.87 (d, J = 6.2 Hz, 3H, CH3), 0.92 (d, J = 3.9 Hz, 3H, CH3), 1.23– 2.18 (m, CH, CH2, steroid), 2.83 (m, 4H, 2 × -CH2-NH3), 2.97–3.19 (m, 2H, CH2-CH-NH), 3.57 (d, J = 2.8 Hz, 3H, OCH3), 3.96–4.13 (m, 2H, 2 × CH-NH3), 4.45 (ddd, J = 11.8, 8.1, 6.1 Hz, 1H, CO-CH-NH), 4.55–4.70 (m, 1H, OCH), 4.87 (q, J = 2.9 Hz, 1H, OCH), 6.95–7.01 (m, 1H, ArH), 7.03–7.10 (m, 1H, ArH), 7.15 (dd, J = 5.0, 2.4 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.48 (q, J = 8.6, 7.6 Hz, 1H, ArH), 8.27–8.34 (m, 6H, 2 × NH3), 8.70–8.92 (m, 7H, 2 × NH3, NH), 10.97 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.1, 169.3, 136.6, 129.5, 127.5, 124.2, 121.4, 121.0, 118.8, 118.7, 112.0, 110.0, 76.5, 76.2, 74.3, 73.8, 65.4, 55.9, 55.8, 53.6, 52.6, 52.2, 52.0, 51.9, 51.8, 49.9, 42.9, 42.8, 38.7, 37.7, 34.9, 34.8, 29.8, 23.7, 23.2, 23.1, 22.6, 22.5, 20.8, 18.8, 15.6, 12.0, 12.0; IR (ATR): νmax 3745, 2868, 2653, 2284, 2171, 2041, 1730, 1596, 1496, 1456, 1365, 1223, 1139, 1048, 1001, 890 cm−1; HRMS (+ESI): Found m/z 411.2809, [M]4+, C46H74N6O7 required 411.2804.
3.1.14. (5S,5′S)-6,6′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7-diyl)bis(oxy))bis(6-oxohexane-1,5-diaminium) chloride (17d)
The title compound was synthesised from 11d (0.061 g, 0.048 mmol) following general synthetic procedure C. The product 17d was obtained as a white solid (0.041 g, 85%); mp 244.2 °C; 1H NMR (600 MHz, DMSO-d6) δ 0.60 (s, J = 5.0 Hz, 3H, CH3), 0.87 (d, J = 6.1 Hz, 3H, CH3), 0.92 (s, 3H, CH3), 0.98–2.07 (m, steroid), 2.75–2.81 (m, 4H, 2 × -CH2-NH3), 3.03–3.15 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.90 (d, J = 5.7 Hz, 1H, -CH-NH3), 4.05 (t, J = 5.8 Hz, 1H, -CH-NH3), 4.46 (td, J = 8.0, 5.8 Hz, 1H, -CO-CH-NH), 4.60 (dt, J = 11.3, 6.4 Hz, 1H, OCH), 4.89 (t, J = 3.3 Hz, 1H, OCH), 6.98 (t, J = 7.5 Hz, 1H, ArH), 7.07 (t, J = 7.5 Hz, 1H, ArH), 7.15 (dd, J = 7.7, 2.4 Hz, 1H, ArH), 7.35 (d, J = 8.1 Hz, 1H, ArH), 7.48 (d, J = 8.0 Hz, 1H, ArH), 8.07–8.19 (m, 6H, 2 × NH3), 8.30 (dd, J = 28.8, 7.5 Hz, 1H, NH), 8.63–8.73 (m, 6H, 2 × NH3), 10.93 (d, J = 2.5 Hz, 1H, NH); 13C NMR (150 MHz, DMSO-d6) δ 173.4, 173.1, 169.2, 136.6, 127.5, 124.2, 124.1, 121.4, 118.9, 118.4, 112.0, 110.0, 76.1, 74.1, 72.7, 72.6, 71.0, 66.8, 65.4, 60.7, 60.6, 53.7, 52.6, 52.3, 52.2, 50.4, 44.1, 42.8, 42.7, 38.7, 38.6, 35.4, 34.8, 34.6, 34.5, 33.9, 32.6, 29.8, 28.0, 27.5, 26.7, 26.7, 23.7, 22.7, 22.5, 18.8, 15.6, 12.0; IR (ATR): νmax 3365, 3236, 2930, 2865, 2072, 1733, 1638, 1510, 1457, 1218, 1137, 1065, 999 cm−1; HRMS (+ESI): Found m/z 425.2962, [M]4+, C48H72N6O7 required 425.2961.
3.1.15. 2,2′-(((3R,5R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,12-diyl)bis(oxy))bis(2-oxoethan-1-aminium) chloride (18a)
The title compound was synthesised from 12a (0.173 g, 0.191 mmol) following general synthetic procedure C. The product 18a was obtained as a white solid (0.130 g, 87%); mp 242.9 °C; 1H NMR (300 MHz, DMSO-d6) δ 0.69 (s, 3H, CH3), 0.75 (d, J = 6.2 Hz, 3H, CH3), 0.90 (s, 3H, CH3), 1.01– 2.19 (m, CH, CH2, steroid), 2.98–3.20 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.74 (d, J = 5.2 Hz, 2H, -CO-CH2-NH3), 3.83–3.95 (m, 2H, -CO-CH2-NH3), 4.47 (td, J = 8.1, 5.8 Hz, 1H, -CO-CH-NH), 4.72 (tt, J = 10.8, 5.1 Hz, 1H, OCH), 5.13 (d, J = 2.8 Hz, 1H, OCH), 6.98 (td, J = 7.4, 7.0, 1.1 Hz, 1H, ArH), 7.07 (ddd, J = 8.1, 7.0, 1.2 Hz, 1H, ArH), 7.16 (d, J = 2.4 Hz, 1H, ArH), 7.34 (d, J = 8.0 Hz, 1H, ArH), 7.48 (d, J = 7.8 Hz, 1H, ArH), 8.28 (d, J = 7.5 Hz, 1H, ArH), 8.47–8.63 (m, 6H, 2 × NH3), 10.97 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (75 MHz, DMSO-d6) δ 173.3, 173.1, 167.5, 167.4, 136.6, 127.5, 124.1, 121.4, 118.8, 118.4, 111.9, 110.0, 77.6, 76.2, 65.4, 53.6, 52.2, 49.0, 47.4, 45.1, 41.5, 40.0, 40.0, 35.6, 34.9, 34.6, 34.2, 32.4, 32.1, 31.5, 27.5, 27.0, 26.5, 26.0, 25.7, 23.5, 23.1, 17.8, 15.6, 12.4; IR (ATR): νmax 3237, 2923, 2863, 2321, 1920, 1735, 1640, 1509, 1435, 1367, 1236, 1120, 1049, 967, 913 cm−1; HRMS (+ESI): Found m/z 354.2226, [M]2+, C40H58N4O7 required 354.2226.
3.1.16. 2,2′-(((3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-3-(2-aminoacetoxy)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-7,12-diyl)bis(oxy))bis(2-oxoethan-1-aminium) chloride (19a)
The title compound was synthesised from 13a (0.500 g, 0.559 mmol) following general synthetic procedure C. The product 19a was obtained as a white solid (0.426 g, 87%); mp 221.4 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.69 (s, 3H), 0.76 (d, J = 6.3 Hz, 3H), 0.92 (s, 3H), 1.03–3.17 (m, CH, CH2, steroid), 3.57 (s, 3H, OCH3), 3.70–4.03 (m, 6H, 3 × -CO-CH2-NH3), 4.46 (td, J = 8.0, 5.6 Hz, 1H, -CO-CH-NH), 4.59 (tq, J = 9.0, 4.3 Hz, 1H, OCH), 4.89–4.96 (m, 1H, OCH), 5.13 (t, J = 2.9 Hz, 1H, OCH), 6.98 (t, J = 7.4 Hz, 1H, ArH), 7.07 (t, J = 7.5 Hz, 1H, ArH), 7.16 (d, J = 2.4 Hz, 1H, ArH), 7.35 (d, J = 8.1 Hz, 1H, ArH), 7.48 (d, J = 7.9 Hz, 1H, ArH), 8.27 (d, J = 7.4 Hz, 1H, NH), 8.44–8.70 (m, J = 7.5, 7.1 Hz, 9H, 3 × NH3), 10.97 (d, J = 2.4 Hz, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 124.1, 121.4, 118.9, 118.4, 111.9, 77.3, 76.0, 73.2, 72.7, 72.7, 69.7, 66.8, 66.8, 60.7, 52.2, 47.4, 43.0, 40.6, 40.4, 40.3, 40.2, 40.0, 39.8, 37.2, 34.8, 28.6, 27.5, 22.5, 17.7, 12.2; νmax 3194, 2936, 2859, 2616, 2342, 1734, 1640, 1597, 1508, 1435, 1368, 1232, 1117, 1039, 742 cm−1; HRMS (+ESI): Found m/z 390.7312, [M]3+, C42H63N5O9 required 390.7307.
3.1.17. 4,4′,4″-(((3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7,12-triyl)tris(oxy))tris(4-oxobutan-1-aminium) chloride (19b)
The title compound was synthesised from 13b (0.138 g, 0.551 mmol) following general synthetic procedure C. The product 19b was obtained as a white solid (0.094 g, 81%); mp 212.2 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.64–0.69 (m, 3H, CH3), 0.73 (d, J = 6.2 Hz, 3H, CH3), 0.88 (d, J = 15.8 Hz, 3H, CH3), 1.01– 2.64 (m, CH, CH2, steroid), 2.78–2.91 (m, 6H, 3 × CH2-NH3), 3.02–3.15 (m, 2H, CO-CH-CH2), 3.57 (s, 3H, OCH3), 4.48 (dtd, J = 15.8, 9.0, 8.1, 4.7 Hz, 2H, OCH, CO-CH-NH), 4.76–4.86 (m, 1H, OCH), 4.99 (dt, J = 7.0, 2.9 Hz, 1H, OCH), 6.98 (t, J = 7.4 Hz, 1H, ArH), 7.06 (t, J = 7.5 Hz, 1H, ArH), 7.17 (q, J = 2.6 Hz, 1H, ArH), 7.34 (d, J = 8.0 Hz, 1H, ArH), 7.50 (dd, J = 17.0, 7.8 Hz, 1H, NH), 8.12–8.31 (m, 9H, NH3), 10.90–10.98 (m, 1H, NH-indole); 13C NMR (100 MHz, DMSO-d6) δ 124.2, 121.4, 118.8, 118.4, 111.9, 75.2, 73.8, 71.0, 66.8, 66.8, 53.6, 52.2, 47.3, 43.5, 40.6, 40.5, 40.3, 40.0, 38.7, 38.6, 37.3, 34.8, 34.5, 32.2, 31.7, 31.6, 31.4, 31.2, 31.0, 28.7, 27.5, 27.1, 26.9, 25.5, 23.2, 23.0, 22.9, 22.8, 22.6, 17.9, 12.3; νmax 2932, 2071, 1934, 1719, 1639, 1510, 1438, 1379, 1191, 1113, 1057, 1005, 749 cm−1; HRMS (+ESI): Found m/z 288.8543, [M]3+, C48H76N5O9 required 288.8542.
3.1.18. (4S,4′S,4″S)-5,5′,5″-(((3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7,12-triyl)tris(oxy))tris(5-oxopentane-1,4-diaminium) chloride (19c)
The title compound was synthesised from 13c (0.200 g, 0.129 mmol) following general synthetic procedure C. The product 19c was obtained as a white solid (0.113 g, 75%); mp 241.6 °C; 1H NMR (600 MHz, DMSO-d6) δ 0.70 (d, J = 1.9 Hz, 3H, -CH3), 0.75 (dd, J = 16.3, 6.6 Hz, 3H, CH3), 0.93 (d, J = 7.8 Hz, 3H, CH3), 1.02–2.41 (m, CH, CH2, steroid), 2.88 (d, J = 32.7 Hz, 4H, 2 × -CH2-NH3), 3.00–3.18 (m, 4H, -CH2-NH3, -CO-CH-CH2), 3.57 (d, J = 1.5 Hz, 3H, -OCH3), 4.15 (d, J = 5.5 Hz, 1H, -CO-CH-NH3), 4.32 (s, 1H, -CO-CH-NH3), 4.45 (m, J = 7.5, 5.9, 3.5 Hz, 2H, -OCH, -CO-CH-NH3), 4.59 (t, J = 5.6 Hz, 1H, -CO-CH-NH), 5.05 (dq, J = 20.3, 3.0 Hz, 1H, OCH), 5.15 (q, J = 2.9 Hz, 1H, OCH), 6.98 (ddd, J = 8.0, 7.0, 1.1 Hz, 1H, ArH), 7.07 (ddd, J = 8.1, 6.9, 1.2 Hz, 1H, ArH), 7.17 (q, J = 4.8 Hz, 1H, ArH), 7.34–7.36 (m, 1H, ArH), 7.48 (d, J = 7.9 Hz, 1H, ArH), 8.27–8.41 (m, 10H, -NH, 3 × NH3), 8.69–8.87 (m, 9H, 3 × NH3), 10.93–10.99 (m, 1H, NH-indole); 13C NMR (150 MHz, DMSO-d6) δ 173.3, 173.1, 169.3, 169.2, 169.1, 167.7, 136.6, 127.5, 124.2, 121.4, 118.8, 118.4, 112.0, 110.0, 77.6, 76.7, 73.0, 53.6, 52.2, 51.9, 51.9, 51.8, 49.3, 47.4, 45.3, 45.3, 45.2, 42.6, 41.3, 38.8, 38.6, 38.4, 37.6, 35.0, 34.7, 32.6, 31.6, 31.5, 28.4, 27.8, 27.4, 27.1, 23.5, 22.9, 22.8, 22.4, 22.2, 20.7, 17.7, 12.3, 12.2; νmax 3373, 2863, 2079, 1948, 1731, 1599, 1509, 1457, 1365, 1226, 1128, 1004, 894 cm−1; HRMS (+ESI): Found m/z 317.8809, [M]6+, C51H85N8O9 required 317.8808.
3.1.19. (5S,5′S,5″S)-6,6′,6″-(((3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7,12-triyl)tris(oxy))tris(6-oxohexane-1,5-diaminium) chloride (19d)
The title compound was synthesised from 13d (0.282 g, 0.177 mmol) following general synthetic procedure C. The product 19d was obtained as a white solid (0.185 g, 86%); mp 243.3 °C; 1H NMR (600 MHz, DMSO-d6) δ 0.70 (d, J = 3.0 Hz, 3H, CH3), 0.73–0.79 (m, 3H, CH3), 0.92 (dd, J = 6.6, 2.6 Hz, 3H, CH3), 1.05–2.26 (m, CH, CH2, steroid), 2.77–2.84 (m, 6H, 3 × -CH2-NH3), 3.01–3.15 (m, 2H, -CO-CH-CH2), 3.38 (d, J = 7.0 Hz, 3H, 3 × -CO-CH-NH3), 3.57 (s, 3H, OCH3), 3.74–5.19 (m, 4H, -OCH, -CO-CH-NH), 6.95–7.01 (m, 1H, ArH), 7.06 (ddd, J = 8.1, 6.9, 1.1 Hz, 1H, ArH), 7.17 (q, J = 3.5, 2.9 Hz, 1H, ArH), 7.35 (d, J = 8.1 Hz, 1H, ArH), 7.47 (d, J = 7.9 Hz, 1H, ArH), 8.04–8.14 (m, 3H, NH3), 8.19–8.25 (m, 6H, 2 × NH3), 8.33 (dt, J = 7.9, 3.2 Hz, 1H, NH), 8.65–8.90 (m, 9H, 3 × NH3), 10.97 (d, J = 2.2 Hz, 1H, NH-indole). 13C NMR (150 MHz, DMSO-d6) δ 173.4, 173.3, 173.1, 172.5, 169.3, 136.6, 127.5, 124.2, 121.4, 118.8, 118.4, 112.0, 110.0, 77.6, 76.7, 73.0, 65,4, 53.7, 52.2, 45.3, 45.3, 45.2, 42.8, 38.8, 38.8, 38.6, 38.4, 37.5, 35.0, 34.8, 32.6, 34.5, 34.3, 32.7, 31.7, 31.5, 31.1, 30.8, 29.8, 29.8, 29.3, 28.3, 27.6, 27.4, 26.7, 26.5, 25.6, 22.4, 22.4, 21.7, 17.7, 15.6, 12.3, 12.2; νmax 3377, 2920, 2866, 2045, 1732, 1602, 1509, 1456, 1364, 1224, 1129, 1068, 994, 893 cm−1; HRMS (+ESI): Found m/z 331.8966, [M]6+, C54H91N8O9 required 331.8964.
3.1.20. (4S,4′S)-5,5′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-1-Methoxy-1-oxo-3-phenylpropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7-diyl)bis(oxy))bis(5-oxopentane-1,4-diaminium) chloride (20)
The title compound was synthesised from 14 (0.783 g, 0.662 mmol) following general procedure C. The product 20 was obtained as a white solid (0.532 g, 87%); mp 236.6 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.85 (d, J = 6.3 Hz, 3H, CH3), 0.92 (d, J = 4.2 Hz, 3H, CH3), 1.05–2.92 (m, CH, CH2, steroid), 2.96–3.06 (m, 2H, -CO-CH-CH2), 3.58 (d, J = 1.0 Hz, 3H, OCH3), 3.89–4.19 (m, 2H, CH, CH-NH3), 4.36–4.70 (m, 3H, 2 × OCH, -CO-CH-CH2), 4.87 (d, J = 3.2 Hz, 1H, CH), 7.21 (d, J = 2.0 Hz, 1H, ArH), 7.23 (d, J = 2.0 Hz, 2H, ArH), 7.26 (s, 1H, NH), 7.27 (d, J = 1.4 Hz, 1H, ArH), 7.30 (t, J = 3.2 Hz, 1H, ArH), 8.26–8.38 (m, 6H, NH3), 8.81 (d, J = 35.9 Hz, 6H, NH3); 13C NMR (100 MHz, DMSO-d6) δ 173.2, 173.2, 172.7, 171.8, 169.4, 169.3, 169.1, 168.8, 137.9, 137.4, 129.6, 129.5, 128.8, 128.7, 127.2, 127.0, 76.5, 55.9, 54.7, 54.1, 52.6, 52.2, 52.0, 51.2, 49.8, 42.8, 42.8, 38.7, 38.5, 38.1, 37.7, 37.1, 36.8, 35.4, 34.9, 34.8, 34.4, 33.4, 23.2, 22.6, 22.5, 18.8, 12.0, 12.0; IR (ATR): νmax 3374, 2929, 2865, 2061, 1733, 1639, 1510, 1449, 1366, 1223, 1138, 1065, 997; HRMS (+ESI): Found m/z 391.7755, [M]4+, C44H73N5O7 required 391.7745.
3.1.21. (4S,4′S)-5,5′-(((3R,5S,7R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(Benzylamino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene-3,7-diyl)bis(oxy))bis(5-oxopentane-1,4-diaminium) chloride (21)
The title compound was synthesised from 15 (0.520 g, 0.468 mmol) following general procedure C. The product 21 was obtained as a white solid (0.374 g, 93%); mp 208.3 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.61 (s, 3H, CH3), 0.88–0.94 (m, 6H, 2 × CH3), 1.02–2.23 (m, CH, CH2, steroid), 2.82 (dq, J = 12.8, 6.4 Hz, 4H, 2 × CH2-NH3), 3.95–4.31 (m, 5H, OCH, 2 × CH-NH3, NH-CH2), 4.53–4.71 (m, 1H, OCH), 7.21–7.32 (m, 5H, ArH), 8.21–8.34 (m, 6H, NH3), 8.42 (dt, J = 14.5, 6.0 Hz, 1H, NH), 8.78 (dd, J = 26.3, 12.0 Hz, 6H, NH3); IR (ATR): νmax 3369, 2927, 2862, 1942, 1733, 1509, 1601, 1509, 1450, 1378, 1226, 1138, 1065, 962, 916; HRMS (+ESI): Found m/z 355.7646, [M]4+, C41H69N5O5 required 355.7644.
3.1.22. ((4-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-4-oxobutyl)amino)(amino)methaniminium trifluoroacetate (23)
The title compound was synthesised from 16b (0.213 g, 0.305 mmol), N,N′-di-Boc-1H-pyrazole-1-carboxamidine (0.125 g, 0.396 mmol) and triethylamine (1 mL, 0.762 mmol) following general synthetic procedure D. The product 23 was obtained as a white solid (0.105 g, 53%); mp 247.9 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.3 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 1.04–2.55 (m, CH, CH2, steroid), 3.02–3.12 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 4.48 (q, J = 7.2 Hz, 1H, -CO-CH-NH), 4.56–4.73 (m, 1H, OCH), 6.81–7.72 (m, 10H, ArH, NH2, NH), 8.22 (d, J = 7.6 Hz, 1H, NH), 10.87 (s, 1H, NH-indole); 13C NMR (75 MHz, DMSO-d6) δ 173.2, 173.1, 172.3, 170.9, 157.3, 136.6, 127.6, 124.1, 121.4, 118.8, 118.5, 111.9, 110.1, 56.4, 56.1, 53.6, 52.2, 42.7, 41.7, 40.8, 40.5, 40.3, 40.0, 39.7, 39.1, 35.8, 35.4, 35.0, 34.7, 33.9, 32.5, 31.3, 27.5, 27.1, 26.4, 24.5, 24.3, 23.5, 20.9, 18.8, 12.3; IR (ATR): νmax 3275, 3184, 2930, 2863, 2320, 1654, 1533, 1439, 1357, 1175, 1132, 800 cm−1; HRMS (+ESI): Found m/z 704.4746, [M]+, C41H61N5O5 required 704.4746.
3.1.23. (S)-6-((2-(((3R,5R,8R,9S,10S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthren-3-yl)oxy)-2-oxoethyl)amino)-6-oxohexane-1,5-diaminium chloride (25)
The title compound was synthesised from 16a (0.150 g, 0.224 mmol), Nε-Z-Nα-Boc-L-lysine hydroxysuccinimide ester (0.119 g, 0.269 mmol) and DIPEA (0.116 g, 0.895 mmol) following general synthetic procedure E. The product 25 was obtained as a white solid (0.027 g, 15%); mp 247.9 °C; 1H NMR (400 MHz, DMSO-d6) δ 0.60 (s, 3H, CH3), 0.86 (d, J = 6.4 Hz, 3H, CH3), 0.91 (s, 3H, CH3), 1.01–2.15 (m, steroid), 2.75 (q, J = 8.3, 7.2 Hz, 2H, -CH2-NH3), 3.02–3.13 (m, 2H, -CO-CH-CH2), 3.57 (s, 3H, -OCH3), 3.82–3.90 (m, 2H, -CH2-NH), 3.96 (dd, J = 17.5, 5.9 Hz, 1H, -CH-NH3), 4.48 (td, J = 8.1, 5.8 Hz, 1H, -CO-CH-NH), 4.62–4.72 (m, 1H, -OCH), 6.98 (t, J = 7.4 Hz, 1H, ArH), 7.07 (t, J = 7.5 Hz, 1H, ArH), 7.14 (d, J = 2.4 Hz, 1H, ArH), 7.34 (d, J = 8.1 Hz, 1H, ArH), 7.49 (d, J = 7.9 Hz, 1H, ArH), 8.05 (s, 3H, NH3), 8.25 (d, J = 7.6 Hz, 1H, NH), 8.35 (d, J = 5.3 Hz, 3H, NH3), 9.05 (t, J = 5.8 Hz, 1H, NH), 10.90 (d, J = 2.3 Hz, 1H, NH-indole); 13C NMR (75 MHz, DMSO-d6) δ 173.2, 173.1, 169.6, 169.3, 136.6, 127.5, 124.1, 121.4, 118.8, 118.5, 111.9, 110.1, 75.1, 66.8, 66.8, 65.4, 56.4, 56.1, 53.6, 52.2, 52.1, 42.7, 41.6, 40.8, 40.5, 40.3, 40.0, 39.7, 39.4, 39.1, 38.7, 35.8, 35.4, 34.9, 34.6, 30.8, 27.5, 26.7, 23.5, 21.3, 20.9, 18.7, 15.6, 12.3; IR (ATR): νmax 3229, 3016, 2926, 2863, 1733, 1651, 1509, 1439, 1207, 1097, 1009, 976 cm−1; HRMS (+ESI): Found m/z 762.5160, [M]2+, C44H67N5O6 required 762.5164.
3.1.24. 1,1′,1″-(((((3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-17-((R)-5-(((S)-3-(1H-Indol-3-yl)-1-methoxy-1-oxopropan-2-yl)amino)-5-oxopentan-2-yl)-10,13-dimethylhexadecahydro-1H-cyclopenta[α]phenanthrene-3,7,12-triyl)tris(oxy))tris(2-oxoethane-2,1-diyl))tris(azanediyl))tris(3-methyl-1-oxobutan-2-aminium) chloride (27)
The title compound was synthesised from 19a (0.250 g, 0.281 mmol), Boc-Val-OH (0.371 g, 1.687 mmol), EDC.HCl (0.323 g, 1.687 mmol), 1-hydroxybenzotriazole (HOBt) hydrate (0.153 g, 0.843 mmol) and DIPEA (0.25 mL, 1.41 mmol) following general procedure F. The product 27 was obtained as a white solid (0.029 g, 10%); mp 242.9 °C; 1H NMR (600 MHz, DMSO-d6) δ 0.69 (s, 3H, CH3), 0.72–0.76 (d, 3H, CH3), 0.91 (s, 3H, CH3), 1.00 (d, J = 8.2 Hz, 18H, 3 × CH3-valine side chain), 1.04–3.57 (m, CH, CH2, steroid), 3.12 (d, 2H, -CO-CH-CH2), 3.57 (s, 3H, OCH3), 3.69 (s, 3H, 3 × -CO-CH-NH3), 3.83 (s, 2H, -NH-CH2-CO), 3.99–4.07 (m, 4H, 2 × -NH-CH2-CO), 4.45–4.49 (m, 1H, -CO-CH-NH), 4.54 (s, 1H, -OCH), 4.85 (s, 1H, -OCH), 5.07 (s, 1H, -OCH), 6.96–7.01 (m, 1H, ArH), 7.04–7.10 (m, 1H, ArH), 7.15 (s, 1H, ArH), 7.35 (d, J = 8.0 Hz, 1H, ArH), 7.48 (d, J = 7.9 Hz, 1H, ArH), 8.24–8.33 (m, 10H, 3 × NH3, NH), 9.01 (s, 1H, NH), 9.03–9.07 (m, 1H, NH), 9.10–9.14 (m, 1H, NH), 10.93 (s, 1H, NH-indole); 13C NMR (150 MHz, DMSO-d6) δ 173.2, 173.1, 169.2, 169.2, 168.9, 168.8, 136.6, 127.5, 124.1, 121.4, 118.9, 118.4, 112.0, 110.0, 76.1, 74.9, 72.0, 65.4, 57.7, 57.6, 53.7, 52.2, 47.5, 45.2, 44.8, 43.6, 41.4, 41.2, 37.3, 34.8, 34.7, 34.6, 34.5, 34.4, 32.6, 31.6, 31.2, 30.3, 30.3, 30.2, 29.8, 28.9, 28.1, 27.5, 27.4, 26.8, 25.8, 22.7, 22.6, 22.5, 18.6, 18.4, 18.2, 17.9, 15.7, 12.5, 12.4; IR (ATR): νmax 3223, 3052, 2936, 2343, 2095, 1730, 1674, 1509, 1378, 1196, 1097, 1004 cm−1; HRMS (+ESI): Found m/z 359.8915, [M]3+, C57H88N8O12 required 359.8913.