Abstract
The aim of this study was to assess the beneficial inhibitory effect of silver nanoparticles immobilized on SiO2 or TiO2 on biofilm formation by Pseudomonas aeruginosa—one of the most dangerous pathogens isolated from urine and bronchoalveolar lavage fluid of patients hospitalized in intensive care units. Pure and silver doped nanoparticles of SiO2 and TiO2 were prepared using a novel modified sol-gel method. Ten clinical strains of P. aeruginosa and the reference PAO1 strain were used. The minimal inhibitory concentration (MIC) was determined by the broth microdilution method. The minimal biofilm inhibitory concentration (MBIC) and biofilm formation were assessed by colorimetric assay. Bacterial enumeration was used to assess the viability of bacteria in the biofilm. Silver nanoparticles immobilized on the SiO2 and TiO2 indicated high antibacterial efficacy against P. aeruginosa planktonic and biofilm cultures. TiO2/Ag0 showed a better bactericidal effect than SiO2/Ag0. Our results indicate that the inorganic compounds (SiO2, TiO2) after nanotechnological modification may be successfully used as antibacterial agents against multidrug-resistant P. aeruginosa strains.
1. Introduction
Infections caused by antibiotic-resistant bacterial strains pose a great challenge to modern medicine. The greatest threat is posed by microorganisms collectively referred to as “ESKAPE” [1]. This name is an acronym from the first letters of the names of the following pathogens: Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter spp. Most of these are multi-drug resistant isolates, which are the main cause of nosocomial infections worldwide. It is clinically significant that ESKAPE pathogens are often hypervirulent and are characterized by the presence of numerous mechanisms of resistance to antimicrobial drugs, which directly affects therapeutic failures as well as the prolongation of therapy and, consequently, an increase in treatment costs [2,3].
P. aeruginosa infections are a source of serious problems in patients, especially those in intensive therapy units and/or patients with burns or wounds, and/or after endoscopy. P. aeruginosa strains may cause infections of soft tissues, the urinary tract, the respiratory system, and keratitis [4]. These pathogens can cause gastrointestinal disorders, dermatitis, and bacteremia [5,6]. The severity of infection depends on the production of different extracellular and cell-associated virulence factors which contribute to the various aspects of pathogenesis including biofilm formation [7]. Bacteria living in biofilm consortia are a frequent cause of chronic and recurrent infections due to their increased resistance to the antibiotics used [8]. Therefore, new substances with high antibacterial effectiveness are sought. One of such compounds is silver nanoparticles (AgNPs) [9].
The formation of the biofilm structure is a multi-stage process including bacterial adhesion, maturation, and dispersion. The first and most important step in biofilm formation is the attachment of bacteria to the biotic or abiotic surfaces [10,11,12]. The surface structures of bacteria (fimbrial and non-fimbrial adhesins), the hydrophobic nature of the cell surface, the presence of cilia, as well as substances secreted outside the cell (polysaccharides and proteins) play a key role in this process. Therefore, the most valuable strategy in inhibiting biofilm formation is to reduce the adhesion of bacteria cells on the surface. This can be achieved by inhibiting cell proliferation at the first stage of biofilm formation [13,14,15] by the use of antibiofilm materials and equipment, preferably doped with AgNPs. The phenomenon of “quorum sensing” plays a significant role in the process of biofilm formation and maturation. It is a communication system between microorganisms with the participation of chemical compounds (autoinducers), regulated by specific genes in response to the size of the microbial population. After exceeding the threshold concentration of autoinducers, the expression of genes encoding various virulence factors, including those responsible for the formation of biofilm (lasl, lasR, rhlI, rhlR, ambD, ambE, pqsA, pqsB, lecA, pelA, lasB in P. aeruginosa strains), is changed [16].
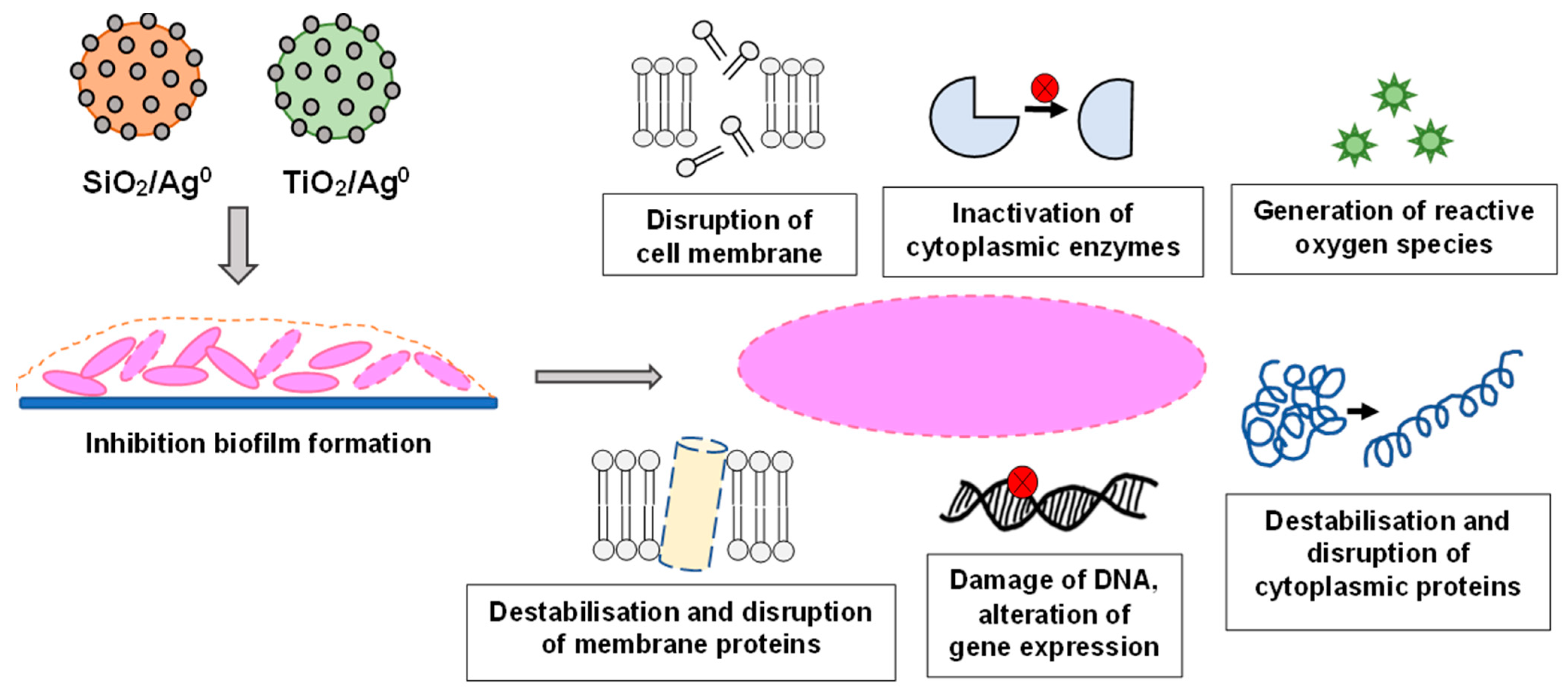
AgNPs can adhere to the surface of bacteria and change the membrane permeability, whilst inside the bacterial cell, they can lead to DNA damage, disruption of cytoplasmic proteins, and inhibition of enzymes by binding to their active center. An important aspect of the action of AgNPs on bacteria is their participation in the formation of free radicals and the generation of metabolic disorders caused by an excessive amount of reactive oxygen forms [9,17,18] (Figure 1).
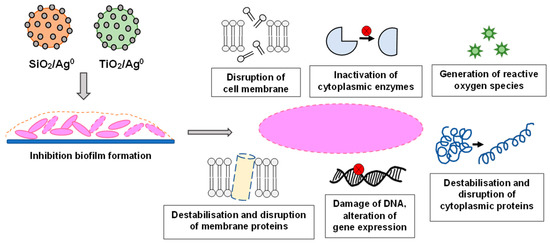
Figure 1.
The various antibacterial targets of nanoparticles.
Nanotechnology enables maximization of the surface of silver particles by depositing them on organic or inorganic carriers (e.g., titanium dioxide or silicon dioxide). TiO2 and SiO2 as carriers of silver nanoparticles make it possible to maintain the dispersion of the nanometal, thus causing the development on the surface active and more effective antimicrobial action at lower concentrations [19]. SiO2 is used in the food industry as an agent preventing “clumping” of loose products; it is added to beverages as a clarifying agent and prevents their foaming [20]. TiO2 is used in cosmetics as a pigment, thickener, and UV absorber. TiO2 enables the osseointegration of artificial medical implants and bone [21]. Synthetic amorphous silica has been used as a common food additive for several decades [22].
The results of some studies showed that the biofilms created by P. aeruginosa strains were resistant to AgNPs [23,24,25]. However, it should be noted that the lack of the effect was not caused by the resistance of bacteria living on biofilm or the extent of its structure, but rather due to the aggregation of nanoparticles, which resulted in the formation of particle clusters with sizes up to 40 times larger than the original nanoparticles. Therefore, embedding silver nanoparticles on inorganic carriers helps to prevent their aggregation. The immobilization of AgNPs on the inorganic carrier surface leads to an extension of the nanoform’s surface area, which results in its enhanced antibacterial activity. The inorganic carrier ensures a higher dispersion of silver particles, prevents their aggregation, and intensifies antibacterial activity [26,27,28,29,30].
Therefore, the aim of this study was to evaluate the antibacterial activity of AgNPs immobilized on SiO2 or TiO2 against multidrug-resistant P. aeruginosa strains isolated from bronchoalveolar lavage fluid and urine of hospitalised patients.
2. Results
2.1. Silver Nanoparticles Immobilized on SiO2 and TiO2
Used in experiments, silver nanoparticles embodied on the inorganic carrier (SiO2/Ag0 and TiO2/Ag0) are shown in Figure 2. Their characteristics are presented in Table 1.

Figure 2.
TEM images of amorphous SiO2/Ag0 (A) and TiO2/Ag0 (B).

Table 1.
Characteristic of the used nanoforms: SiO2/Ag0 and TiO2/Ag0.
2.2. Antibiotic Susceptibility
All the examined P. aeruginosa isolates were classified as multidrug-resistant (MDR) strains because they were resistant or intermediately resistant to at least three drugs from a variety of antibiotic classes (Table 2). Strains isolated from bronchoalveolar lavage fluid showed greater resistance than those isolated from urine. Bacteria showed the greatest resistance to IPM—eight out of eleven strains were resistant, and three were intermediately resistant. Seven out of all P. aeruginosa strains included in the test were resistant to TZP whilst four were intermediately resistant. A similar proportion was established with respect to CAZ and CIP, seven out of eleven isolates being resistant to CAZ and CIP, three strains being intermediately resistant, and one strain showing susceptibility to these drugs. Five P. aeruginosa strains were susceptible to AN while six isolates were resistant to this antibiotic. Five out of all tested strains were resistant to MEM with three isolates showing intermediate resistance and two showing sensitivity. It is worth noting that only one strain, P. aeruginosa 0024, was intermediately resistant to CL, while the remaining were susceptible. The presence of MBL-type carbapenemase was detected in the P. aeruginosa 3 strain isolated from bronchoalveolar lavage fluid.

Table 2.
Antibiotics’ susceptibility of P. aeruginosa strains.
2.3. Prevalence of silE Gene in Tested Bacterial Strains
As shown on Figure 3, the silE gene was only found in the reference P. aeruginosa PAO1 strain (line 15 on the electrophoregram). Clinical isolates did not possess this gene (line 4–14 on the electrophoregram).

Figure 3.
Agarose gel electrophoresis of PCR products for presence of silE gene (220 bp) in P. aeruginosa strains. Lanes: 1 and 16 molecular size markers (1000 bp, Fermentas), 2—negative control, 3—positive control, E. coli J53, 4—P. aeruginosa 0013, 5—P. aeruginosa 0024, 6—P. aeruginosa 3, 7—P. aeruginosa 472, 8—P. aeruginosa 669, 9—P. aeruginosa 124, 10—P. aeruginosa 137, 11—P. aeruginosa 300, 12—P. aeruginosa 328, 13—P. aeruginosa 407, 15—P. aeruginosa PAO1.
2.4. Activity of SiO2/Ag0, TiO2/Ag0, SiO2, and TiO2 against Planktonic Forms (Minimal Inhibitory Concentration (MIC) Determination)
Silver nanoforms were tested as potential inhibitory factors affecting the growth of the planktonic form of P. aeruginosa. The antibacterial activities of SiO2/Ag0 and TiO2/Ag0 were determined and compared with those of SiO2 and TiO2. The results are shown in Table 3. The silver nanoparticles immobilized on the SiO2 and TiO2 were indicative of high antibacterial efficacy. SiO2/Ag0 showed a significant inhibitory effect on bacteria both in the bronchoalveolar lavage fluid isolates (p < 0.05) and urine isolates (p < 0.05). The MICs obtained for TiO2/Ag0 were also significantly lower than the MIC values for TiO2 both with respect to the bronchial tree material (p < 0.05) and urine material (p < 0.05). The MIC values for SiO2 and TiO2 were >8192 µg/mL. It means that at this concentration, no activity against the bacterial growth was observed in the nanoforms without silver ions in contrast to those doped with Ag0. The reference P. aeruginosa PAO1 strain showed much lower sensitivity to immobilized silver particles than the clinical strains. The MIC values of SiO2/Ag0 and TiO2/Ag0 were 256 µg/mL.

Table 3.
Minimal inhibitory concentrations (MICs) of SiO2/Ag0, TiO2/Ag0, SiO2, TiO2 against P. aeruginosa strains.
2.5. Activity of SiO2/Ag0 and TiO2/Ag0 against Biofilm Forms (Minimal Biofilm Inhibitory Concentration (MBIC) Determination)
The MBIC results of SiO2/Ag0 and TiO2/Ag0 are presented in Table 4. The statistical analysis showed that MICs and MBIC values significantly differed among bacterial species (p < 0.05). The MBIC values of nanoparticles were higher than the MICs for all the bacterial strains. The MBICs of SiO2/Ag0 were 2 to 4 times greater than their MICs while the MBICs of TiO2/Ag0 were 2 to 32 times greater than their MICs. The MIC and MBIC values of SiO2/Ag0 against clinical strains demonstrated ranges of 16–128 μg/mL and 32–256 μg/mL, respectively, whereas those of TiO2/Ag0 were 16–64 μg/mL and 64–512 μg/mL, respectively. The highest MIC was established at 128 μg/mL of SiO2/Ag0 for P. aeruginosa 124 isolated from urine whereas the lowest value was 16 μg/mL for the P. aeruginosa 328 urine isolate and P. aeruginosa 669 isolated from bronchoalveolar lavage fluid. The highest MIC was established at 64 μg/mL of TiO2/Ag0 for two strains, P. aeruginosa 124 and 300, where both were isolated from urine. The highest MBIC value of SiO2/Ag0 was 256 μg/mL for P. aeruginosa 124 whereas the lowest value was determined at 32 μg/mL for P. aeruginosa 669. The highest MBIC value of TiO2/Ag0 was 512 μg/mL for two strains, P. aeruginosa 0024 and 3, where both were isolated from bronchoalveolar lavage fluid. The PAO1 strain was less sensitive to SiO2/Ag0 and TiO2/Ag0 compared to tested clinical strains.

Table 4.
Minimal biofilm inhibitory concentrations (MBICs) of SiO2/Ag0, TiO2/Ag0 against P. aeruginosa strains.
2.6. Antibiofilm Activity of SiO2/Ag0 and TiO2/Ag0
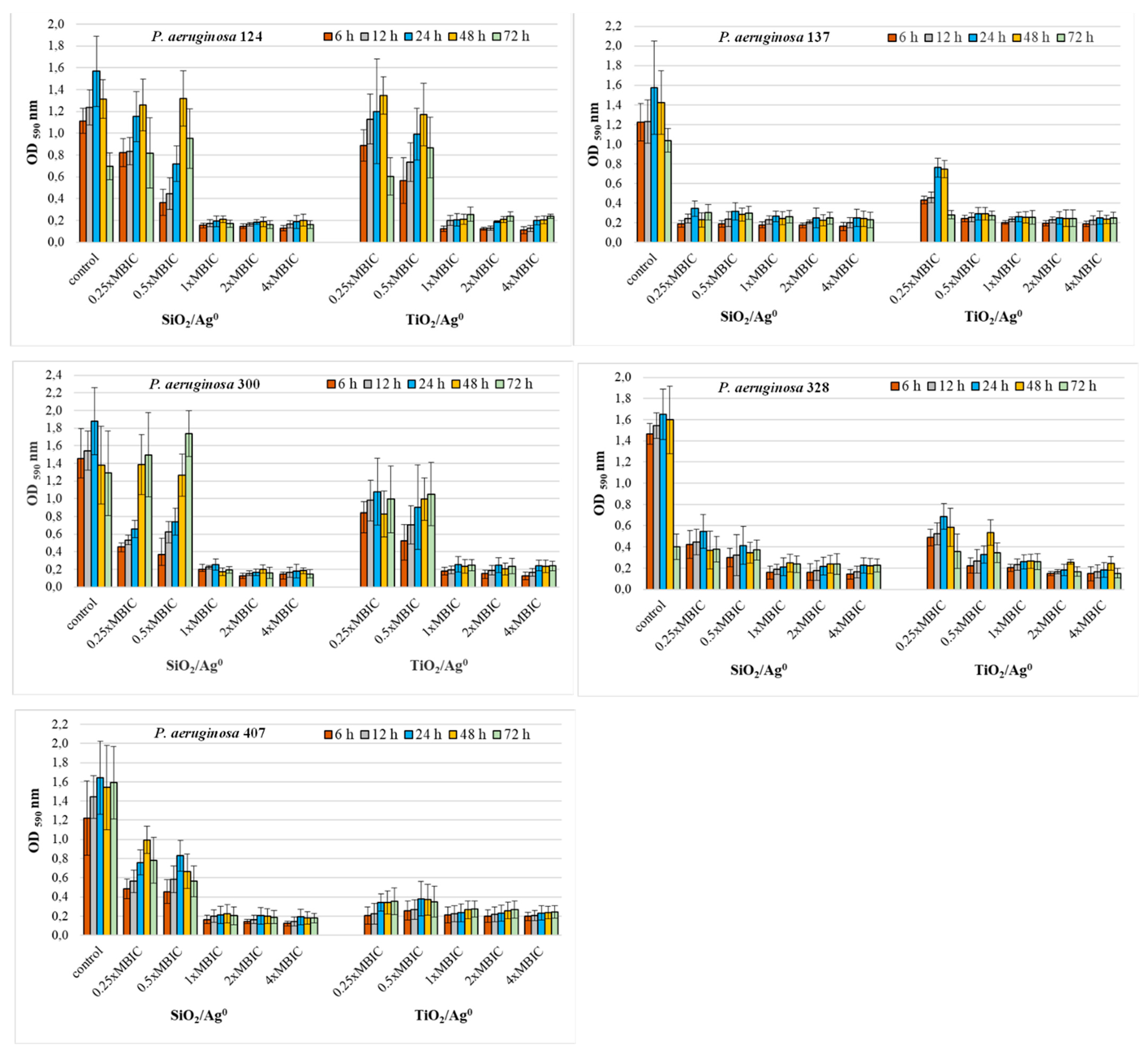
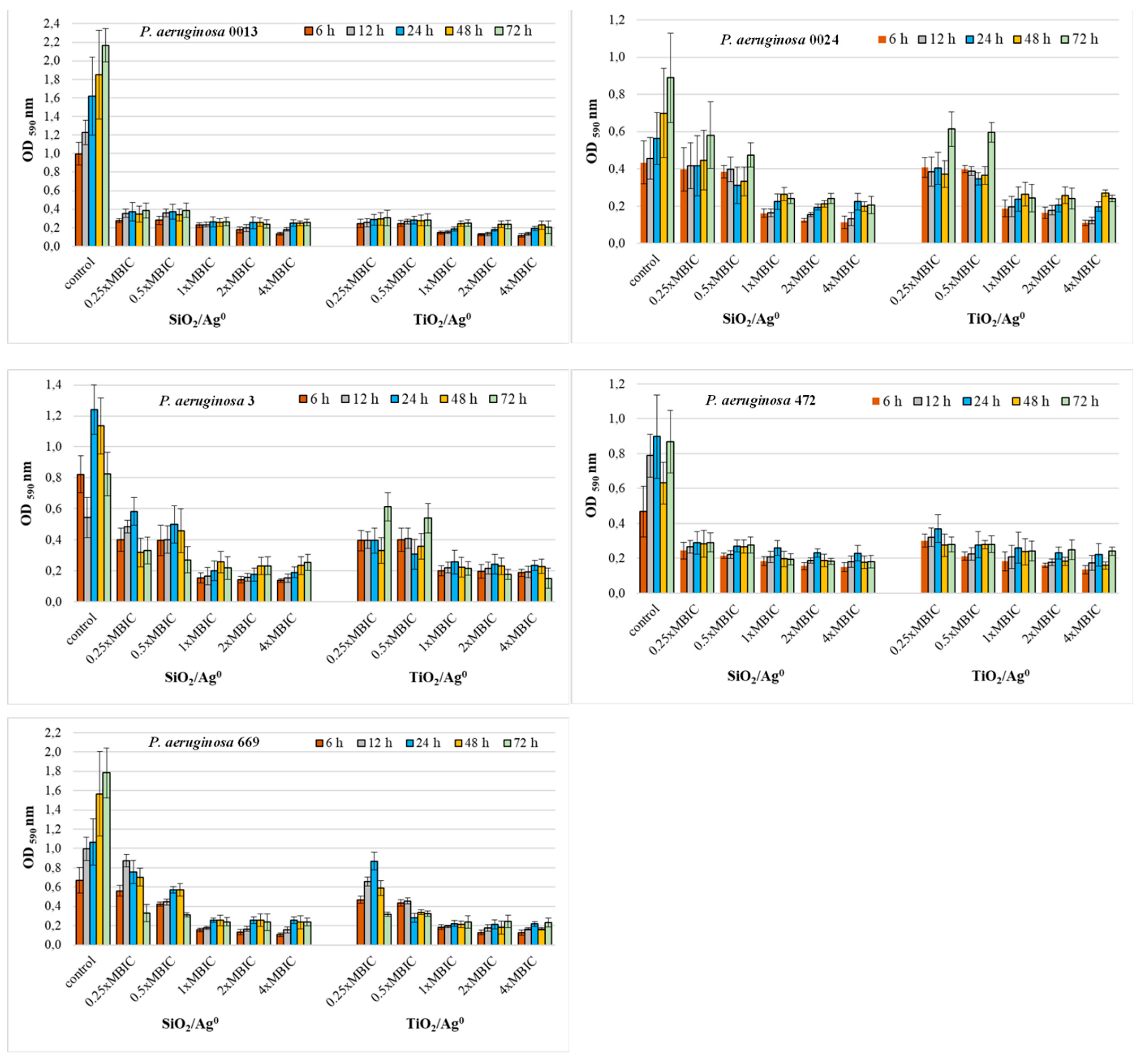
The antibiofilm activity of SiO2/Ag0 and TiO2/Ag0 on clinical P. aeruginosa strains was tested at selected MBIC values: 0.25 × MBIC, 0.5 × MBIC, 1 × MBIC, 2 × MBIC, and 4 × MBIC. Figure 4 and Figure 5 illustrate the inhibitory effects of nanoparticles on biofilm formation. According to the established criteria [28], the OD value ≤ 0.096 is indicative of an inhibited biofilm production; weak biofilm formation is reflected by OD values between 0.096 and 0.192; OD values between 0.193 and 0.384 indicate moderate biofilm production. A strong biofilm synthesis and no inhibition by nanoparticles are reflected by OD values above 0.384. All the untreated P. aeruginosa strains produced a strong biofilm with the OD values more than 0.384.
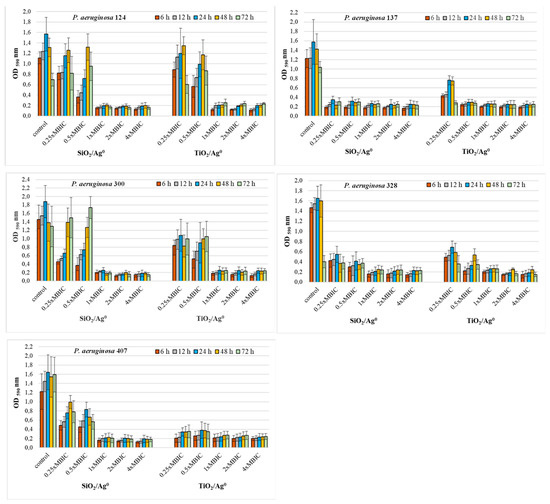
Figure 4.
Effect of SiO2/Ag0 and TiO2/Ag0 on biofilm formation by P. aeruginosa strains isolated from urine. Interpretation of the results: OD ≤ 0.096—non-biofilm producer; 0.096 < OD ≤ 0.192—weak-biofilm producer; 0.192 < OD ≤ 0.384—moderate-biofilm producer; OD > 0.384—strong-biofilm producer.
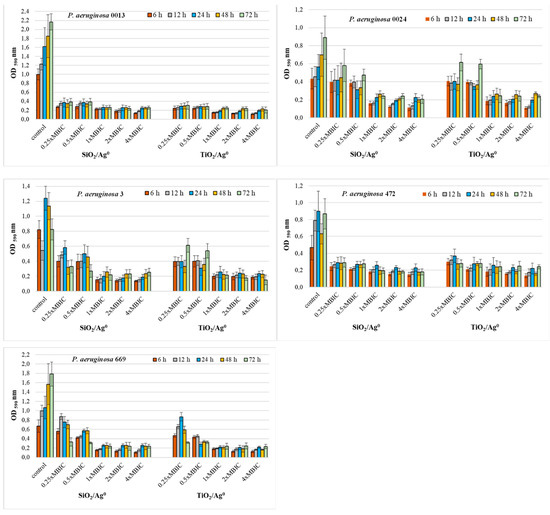
Figure 5.
Effect of SiO2/Ag0 and TiO2/Ag0 on biofilm formation by P. aeruginosa strains isolated from bronchoalveolar lavage fluid. Interpretation of the results: OD ≤ 0.096—non-biofilm producer; 0.096 < OD ≤ 0.192—weak-biofilm producer; 0.192 < OD ≤ 0.384—moderate-biofilm producer; OD > 0.384—strong-biofilm producer.
Where the concentrations of SiO2/Ag0 and TiO2/Ag0 (1, 2, and 4 × MBIC) were at the highest, the biofilm production in P. aeruginosa strains isolated from urine was significantly lower than the control strains grown in the absence of the nanoparticles (p < 0.05) (Figure 4). Under these conditions, the bacteria produced moderate (0.192 < OD ≤ 0.384) or weak (0.096 < OD ≤ 0.192) biofilm. Two of the five strains (P. aeruginosa 137 and 328) were also sensitive to lower SiO2/Ag0 and TiO2/Ag0 concentrations (0.25, 0.5 × MBIC). In the presence of these concentrations, both strains produced significantly less biofilm than untreated bacteria.
Both the P. aeruginosa strains isolated from both bronchoalveolar lavage fluid and urine had a similar sensitivity to SiO2/Ag0 and TiO2/Ag0 (Figure 5). Two strains, P. aeruginosa 0013 and 472, showed significantly decreased biofilm formation (0.096 < OD ≤ 0.384) in the presence of all the tested nanoparticles’ concentrations (p < 0.05). Three strains, P. aeruginosa 0024, 3, and 669, were more sensitive only to the highest concentrations (1, 2, and 4 × MBIC).
These results show that SiO2/Ag0 and TiO2/Ag0 can reduce the amount of biofilm formed but cannot completely inhibit its production.
2.7. The Viability of P. aeruginosa Clinical Strains in the Biofilm in the Presence of SiO2/Ag0 and TiO2/Ag0
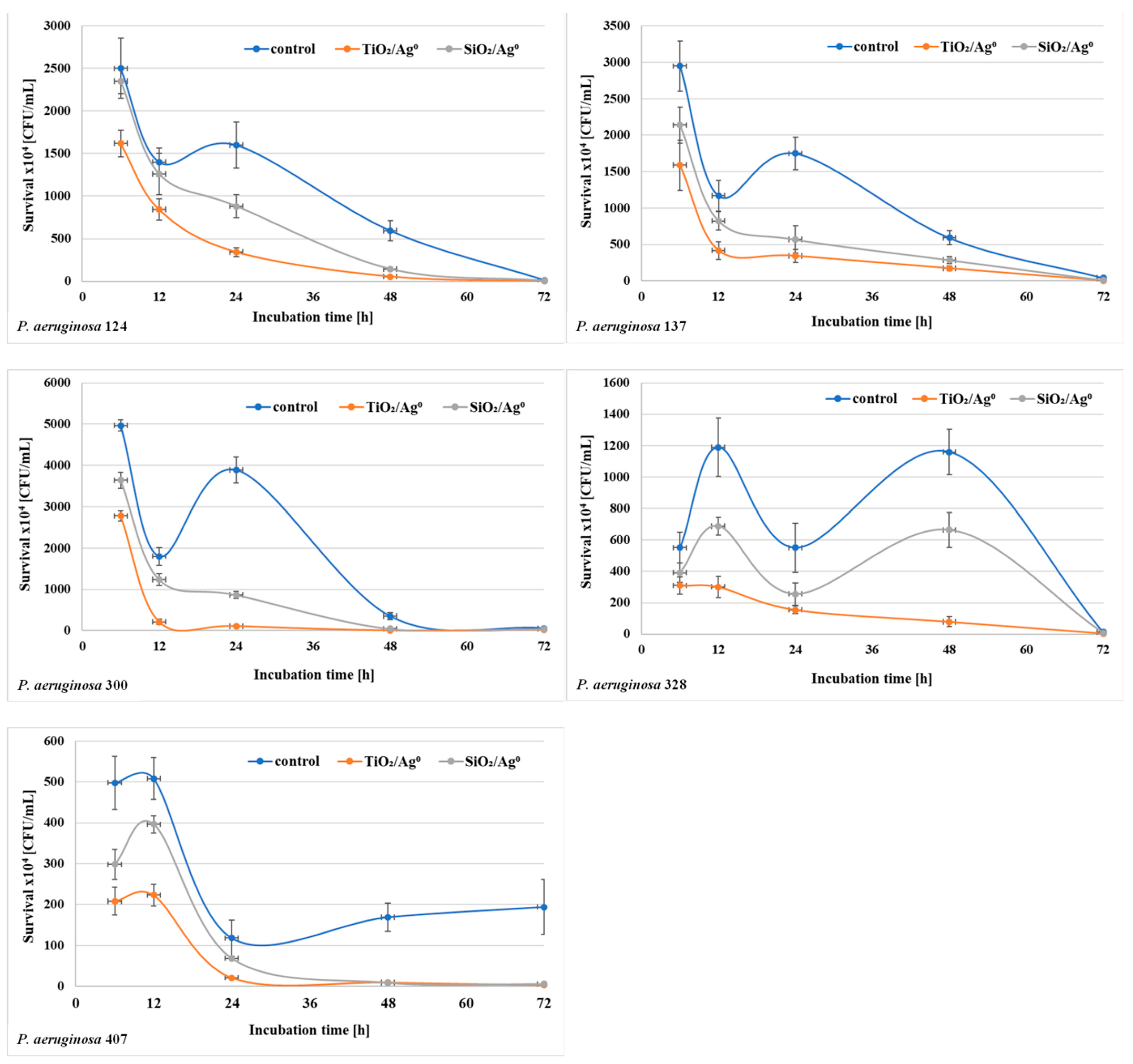
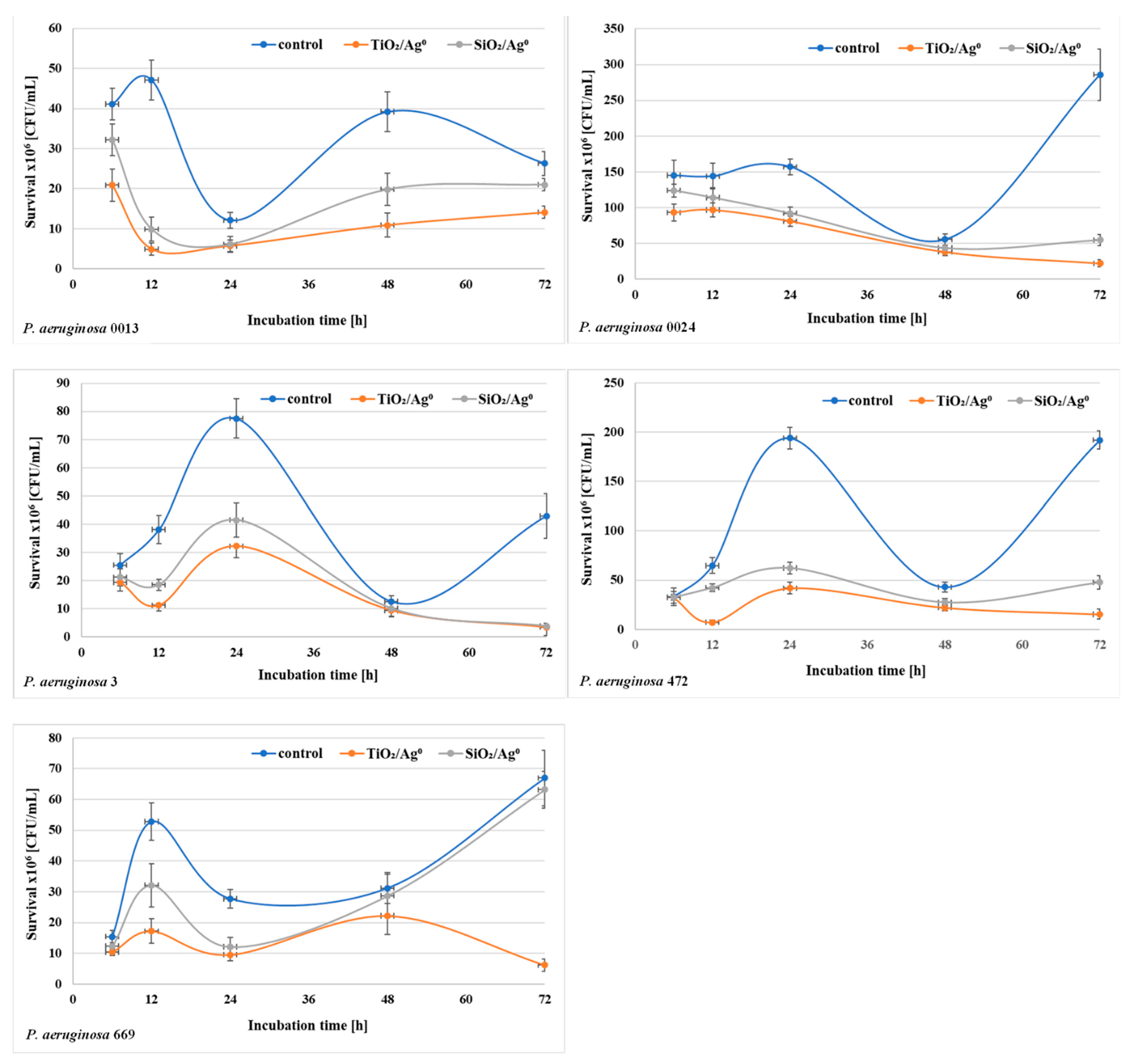
The bacterial survival (the number of CFU/mL) in biofilm mass was determined after each period of incubation (6–72 h) at a concentration of 0.5 × MBIC. The results shown in Figure 6 and Figure 7 indicate that the number of viable bacteria in samples containing SiO2/Ag0 or TiO2/Ag0 was lower compared to the controls. It is also worth noting that TiO2/Ag0 showed a better bactericidal effect than SiO2/Ag0. Detailed information about the number of CFU/mL are provided in Table S1 in Supplementary Materials.
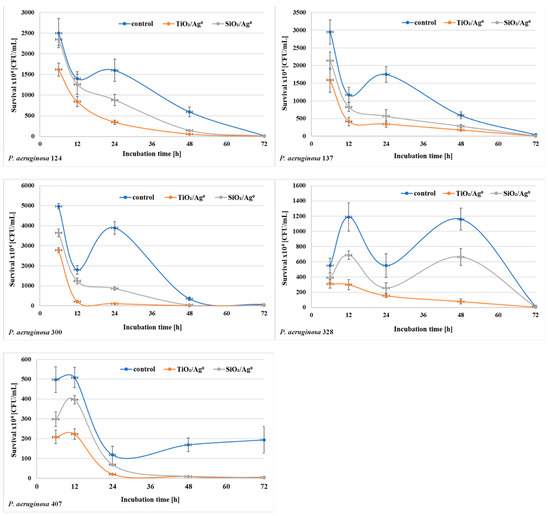
Figure 6.
Effect of SiO2/Ag0 and TiO2/Ag0 on the survival of P. aeruginosa strains isolated from urine in biofilm mass.
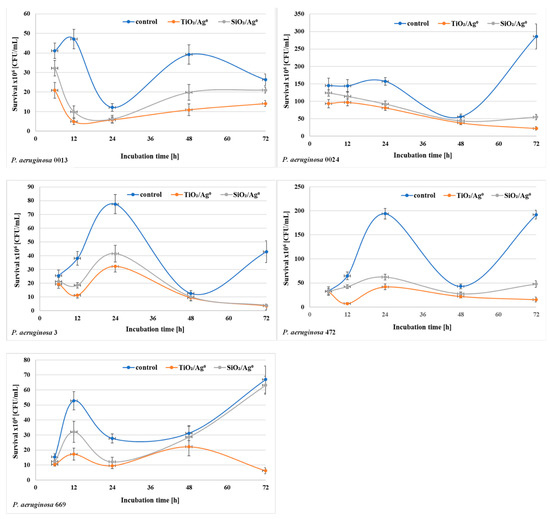
Figure 7.
Effect of SiO2/Ag0 and TiO2/Ag0 on the survival of P. aeruginosa strains isolated from bronchoalveolar lavage fluid in biofilm mass.
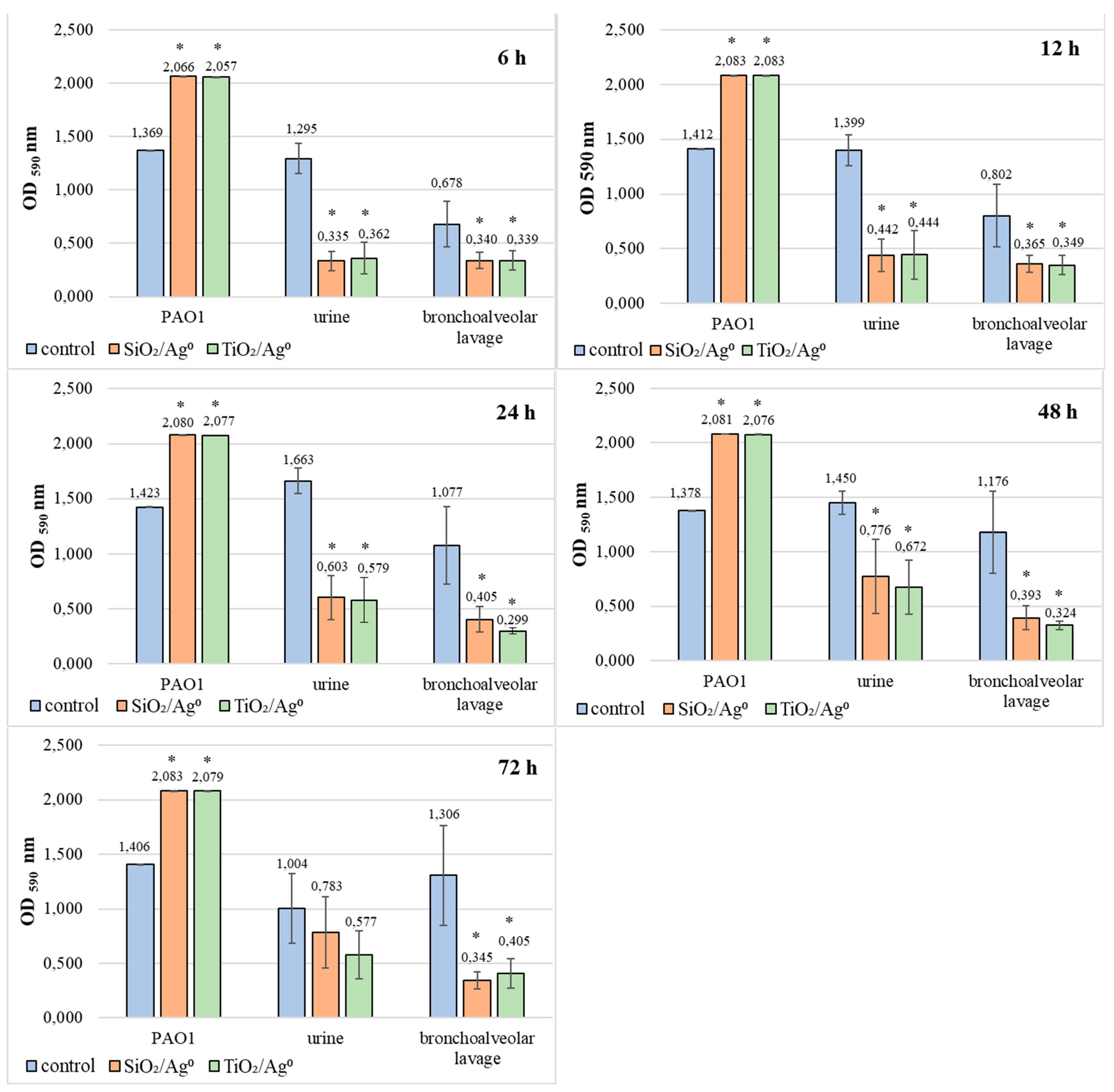
2.8. Comparison of SiO2/Ag0 and TiO2/Ag0 Antibiofilm Activity against the Reference PAO1 Strain and Clinical Strains of P. aeruginosa
Based on the results presented in Figure 8, it can be concluded that SiO2/Ag0 and TiO2/Ag0 significantly reduce the average amount of biofilm formation by bacteria isolated from both urine and bronchoalveolar lavage fluid (p < 0.05). The most effective action of both nanoparticles against these bacteria was recorded in 6-h biofilms. In these biofilms, the mean OD values ranged from 0.335 to 0.362 and indicated the average production of biofilm mass.
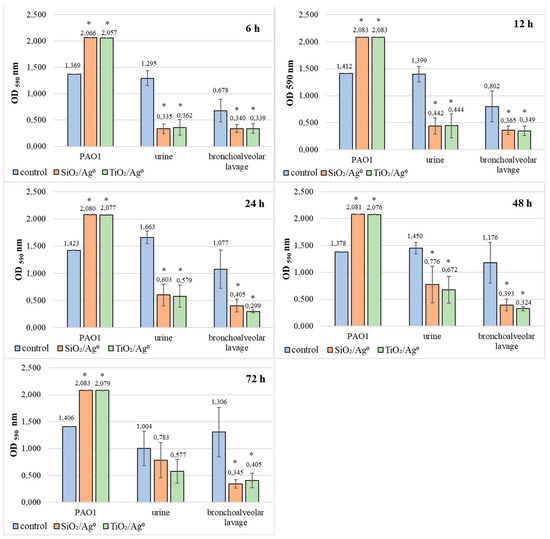
Figure 8.
Effect of SiO2/Ag0 and TiO2/Ag0 at a concentration of 0.5 × MBIC on biofilm formation by PAO1 and clinical strains of P. aeruginosa. Values represent the mean ± SD for five strains isolated from urine and five from bronchoalveolar lavage fluid. * p ≤ 0.05 compared with control. Interpretation of the results: OD ≤ 0.096—non-biofilm producer; 0.096 < OD ≤ 0.192—weak-biofilm producer; 0.192 < OD ≤ 0.384—moderate-biofilm producer; OD > 0.384—strong-biofilm producer.
Completely different effects of SiO2/Ag0 and TiO2/Ag0 were noted in biofilms formed by PAO1. In this case, regardless of the incubation time, the amount of biofilm mass produced was always higher when compared to the control sample (without silver nanoparticles).
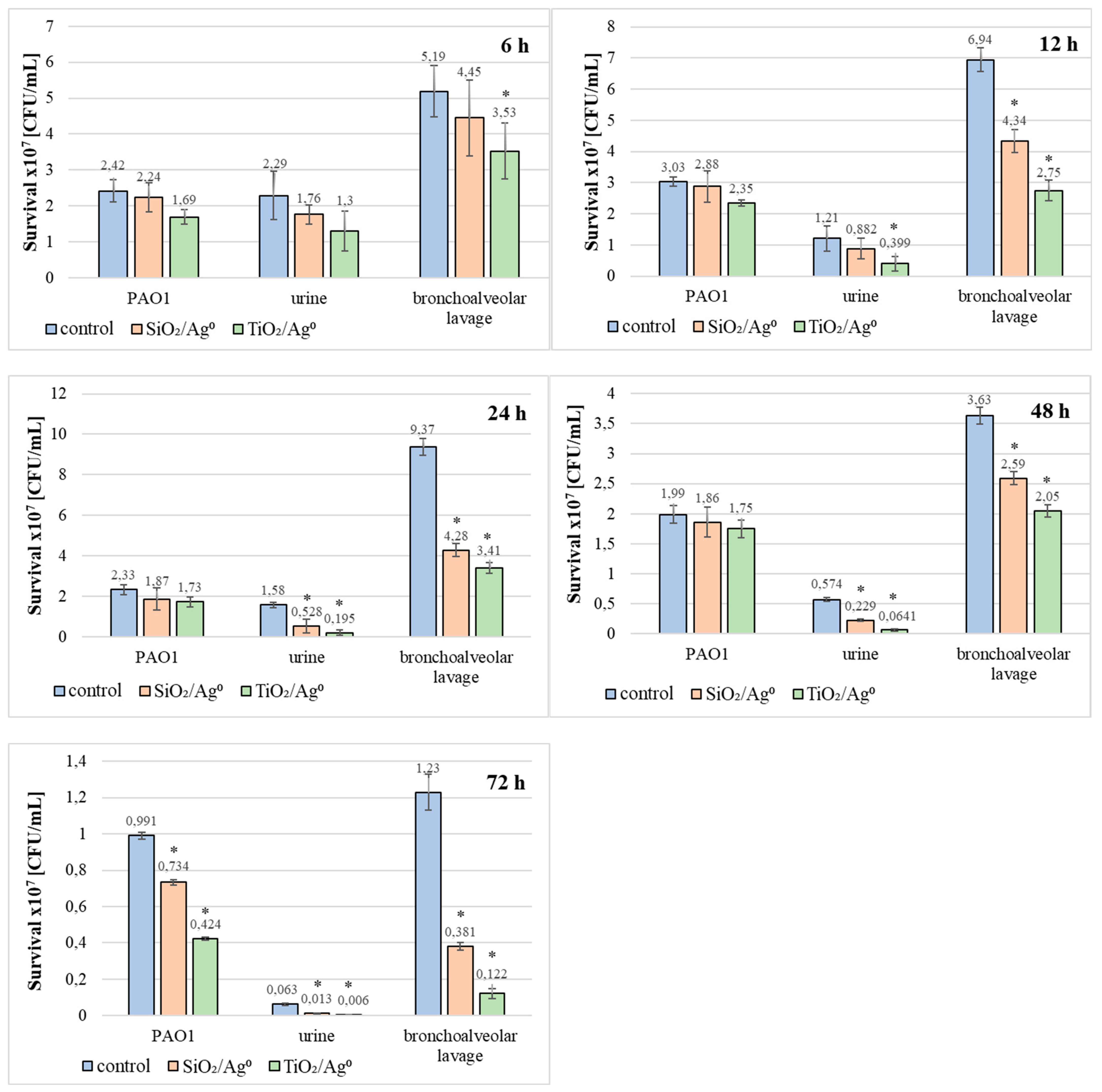
Additionally, the influence of SiO2/Ag0 and TiO2/Ag0 on the survival of PAO1 and clinical strains living in biofilms has been tested (Figure 9). TiO2/Ag0, regardless of the incubation time, significantly decreased the average number of viable bacteria in biofilms formed by strains isolated from bronchoalveolar lavage fluid (p < 0.05). The anti-growth effect of TiO2/Ag0 on urine-derived strains was observed in 12–72 h biofilms (p < 0.05). SiO2/Ag0 was slightly less effective than TiO2/Ag0. A statistically significant reduction in the number of viable bacterial cells in the presence of SiO2/Ag0 was noted in young (12-h) and mature (24-, 48-, and 72-h) biofilms formed by the bronchoalveolar-lavage-derived strains and in the 24-, 48-, and 72-h biofilms formed by the urine-derived strains. Contrary to the clinical strains, the average number of viable cells of the reference PAO1 strain decreased significantly as a result of exposure to SiO2/Ag0 and TiO2/Ag0 only in the oldest biofilm tested (72 h).
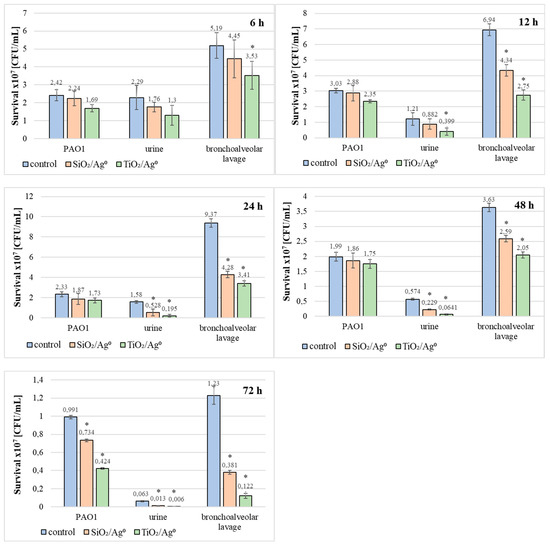
Figure 9.
Effect of SiO2/Ag0 and TiO2/Ag0 at a concentration of 0.5 × MBIC on survival PAO1 and clinical strains of P. aeruginosa in biofilms. Values represent the mean ±SD for five strains isolated from urine and five from bronchoalveolar lavage fluid. * p ≤ 0.05 compared with control.
3. Discussion
Silver nanoparticles (AgNPs) are the nanomaterials most widely used as antimicrobial agents [31]. It is worth noting that they are used in low concentrations and do not generate bacterial resistance. The mechanism of AgNPs’ action on bacterial plankton forms has been described. It is known that silver nanoparticles destroy bacterial membranes and cell walls. They damage DNA and inactivate many proteins, including enzyme proteins involved in the respiratory process [32].
It is known that the size of AgNPs directly influences the activity of nanoparticles: the smaller the particle size, the greater the surface contact area between AgNPs and a microorganism. In our research, two sizes of AgNPs immobilized on the inorganic carriers, 16.57 (±5.01) nm (TiO2/Ag0) and 17.91 (±3.37) nm (SiO2/Ag0), were used. It is likely that the slight difference in the size of the silver particles caused SiO2/Ag0 and TiO2/Ag0 to exhibit the same antibacterial activity against 7 of the 11 tested strains. In these cases, the MIC values were the same. Arokiyaraj et al. [33] tested the activity of AgNPs with a size of 121 nm and obtained a MIC result of 15 μg/mL against the P. aeruginosa strain. The MIC value was lower and amounted to 12.5 μg/mL when silver nanoparticles with smaller sizes of 14–48 mm were used against P. aeruginosa [34]. Singh et al. [35] used AgNPs with a size of 20–40 nm and noted that the MIC value was 6.25 μg/mL against the P. aeruginosa strain. Mann et al. [36] showed that the MIC value of AgNPs against P. aeruginosa was only 3 μg/mL when the size of these nanoparticles was 2 nm.
In our study, the MBIC values (the lowest concentrations inhibiting biofilm formation) of SiO2/Ag0 and TiO2/Ag0 were also determined. These values ranged from 32 and 512 μg/mL and were even 32 times higher than the MIC values (the lowest concentrations inhibiting the growth of planktonic forms of bacteria). Such large differences result from the presence of exopolysaccharide present in biofilm consortia, which makes it difficult for the penetration of antibacterial particles [37]. Therefore, to inhibit the growth of bacteria growing in the biofilm, much higher concentrations of the antibacterial compound should be used.
Our preliminary research showed that the MIC values of SiO2/Ag0 and TiO2/Ag0 ranged from 16 to 128 µg/mL against all clinical strains. On the other hand, PAO1 used as the control strain showed less susceptibility (MIC = 256 µg/mL) as compared to the clinical strains. Such a significant difference in sensitivity can be explained by the fact that the PAO1 strain had a silver resistance gene (silE) in contrast to our tested clinical strains. The mechanism of bacterial resistance to silver ions has been known and described in Gram-negative bacteria: E. coli, K. pneumoniae, E. cloacae [23,38,39,40]. It is related to pMG101 plasmid, encoding 9 sil genes gathered in three transcriptional units (silRS, silE, and silCFBAGP), each controlled by a separate promoter [38,39]. SilE is a periplasmic chaperone which captures and delivers silver ions to the SilCFBA protein-a structure spanning both membranes, responsible for ejecting silver ions out of the cell (from the cytoplasm or periplasmic space). Based on literature data, it can be concluded that resistance to silver (conditioned by the presence of the pMG101 plasmid) among bacteria is relatively rare. Percival et al. [23] reported that among 112 bacteria isolated from wounds, the silver resistance genes were only found in 1.8% of the strains (including 9 strains of P. aeruginosa). The study by Finley et al. [24] was carried out on a large sample consisting of 859 clinical isolates from 60 different species, most of them belonging to the genera Staphylococcus, Escherichia, Pseudomonas, Klebsiella, Enterococcus, and Enterobacter. It was noted that of the 32 isolates containing the sil genes, the majority belonged to the genus Enterobacter and Klebsiella. This study showed the prevalence of genes found in hospital isolates at a relatively low level (3.6%) [24]. Hosny et al. [25] pinpointed that among 150 bacteria strains isolated from burns and wounds (including 148 strains as multidrug-resistant), nineteen silver-resistant bacterial isolates (12.6%) including P. aeruginosa were detected.
This study and the previous research [27,30] show that a suitable modification of the nanomaterials (e.g., the stabilization of the silver ions or nanoparticles on the surface of an inorganic carrier) protects Ag+ or Ag0 against mutual aggregation, promotes the increase of the surface area of nanocompounds, and improves long-term antibacterial efficacy against planktonic E. coli, S. aureus and K. pneumoniae, and P. aeruginosa [18,27,30,33,41,42]. Bugla-Płoskońska et al. [43] and Jasiorski et al. [44] suggested that SiO2 is an appropriate carrier for silver nanoparticles (Ag0). With its antibacterial efficacy (against E. coli and S. aureus) which is enhanced within 24 h, it may also be used as powder and a supplement in textiles. An inhibitory effect was also reported in silver doped HAp (hydroxyapatites) over 24 h treatments [30]. Kedziora et al. [27,45] suggested that TiO2 and GO (graphene oxide) may serve as excellent carriers for silver ions (Ag+), silver nanoparticles (Ag0) showing efficacy against E. coli, K. pneumoniae, and S. aureus within 24 h. Furthermore, the biofunctionalization of the GO surface with silver nanoparticles and phthalocyanines effectively stopped the growth of the P. aeruginosa within 24 h following near-infrared irradiation [46]. The author of this work suggests that these compounds have a potential and pivotal role as antibiofilm factors (silver nanoparticles immobilized on the SiO2 and TiO2) in nanomaterials thanks to their proven efficacy. Kulshrestha et al. [47] confirmed that the subinhibitory concentration of silver nanoparticles immobilized onto graphene oxide (graphene-oxide silver nanocomposite) reduces the biofilm formation in Enterobacter cloacae (Gram-negative bacteria) and Streptococcus mutans (Gram-positive bacteria). They described the mode of GO-Ag inhibiting action in biofilms. The disruption of the bacterial cell membrane and ROS production were also observed. Kulshrestha et al. [47] showed that comDE, spaP, and vicR genes crucial in quorum sensing cascade are downregulated in the presence of GO-Ag. As a result, less EPS was produced, and a thinner layer of biofilm was observed within 24 h.
The primary interaction of Ag–TiO2 nanostructures with the bacteria is probably an electrostatic attraction between the nanostructure surface and positively charged regions of the extracellular domain of integral membrane proteins on the cell surface [48]. The nanostructures penetrate the outer and inner bacterial membranes. Protrusions, pits, or holes of the bacterial cell wall could be associated with internalized particles [48,49].
The most commensal and pathogenic bacteria in the human body live in a biofilm state. Biofilms are involved in many persistent and chronic infections in humans and increase resistance to antimicrobials. The inhibition of the bacterial cell’s adhesion or the reduction in bacterial growth and proliferation with the use of nanocomposites is the most valuable strategy in preventing biofilm formation.
In the next stage of our research, the influence of SiO2/Ag0 and TiO2/Ag0 on the formation of biofilm by P. aeruginosa strains was determined. It was shown that the amount of biofilm mass decreased with an increasing concentration of nanoforms. Fatima et al. [50] and Altaf et al. [51] obtained similar results. Fatima et al. [50] showed that exposure of oral bacterial strains (Georgenia sp., Staphylococcus saprophyticus, and Rothia mucilaginosa) to titanium dioxide nanoparticles (TiO2-NPs) significantly reduced biofilm formation in a concentration-dependent manner. The presence of TiO2-NPs at concentrations of 8, 16, 32, and 64 μg/mL inhibited the development of the biofilm of P. aeruginosa PAO1, E. coli ATCC25922, and S. aureus MTCC3160 [51]. The results of the research presented by Maurer-Jones et al. [52] showed that TiO2-NPs caused a slower growth of biofilm formed by Shewanella oneidensis. The results obtained by Guo et al. [53] show that biofilms created by P. aeruginosa cells lost their ability to develop biofilm in a concentration-dependent manner when they were exposed to AgNPs at a concentration higher than MIC. Palanisamy et al. [54] investigated the effect of AgNPs on the formation of biofilm in multidrug resistant strains of P. aeruginosa. The inhibitory activity of silver nanoparticles was highest at the concentration of 20 μg/mL.
In our study, we noted an interesting result regarding the influence of sublethal doses of SiO2/Ag0 and TiO2/Ag0 (0.5 × MBIC) on biofilm formation by the PAO1 strain. It produced more biofilm mass in the presence of nanoforms when compared to the control samples, regardless of the incubation time. The amount of biofilm depends not only on the number of bacterial cells in the biofilm but also on the amount of exopolysaccharide (EPS) produced. The production of EPS depends on the quorum sensing and expression of genes involved in its synthesis [55]. Bacteria living in the biofilm, under the influence of nanoparticles, communicate and produce more EPS. It is a defensive reaction of bacteria. The greater amount of EPS makes the penetration of antimicrobial molecules difficult. Similar results were obtained by Xu et al. [56], who showed that bacteria in the presence of sublethal concentrations of cerium oxide nanoparticles produced more biofilm compared to control samples. This reaction was caused by oxidative stress and the greater production of reactive oxygen species.
The penetration of nanoparticles into biofilm mass depends primarily on biofilm age, its surface, the amount of extracellular exopolysaccharide, nanoparticles size, their surface charge, and concentration [18]. The literature does not offer any accurate/compelling reports/accounts on how the own-synthesized silver nanoforms influence biofilm formation in P. aeruginosa strains. Kamaraj et al. [57] showed that the total number of viable Pseudomonas sp. and Bacillus sp. cells in the 6 h biofilm treated with TiO2–Ag was significantly lower compared to that of biofilms treated with the polished Ti or TiO2. Yao et al. [58] investigated the bactericidal effect of the Ag/TiO2-coated silicon catheters on the survival of P. aeruginosa, E. coli, and S. aureus strains. These bacteria showed different sensitivities to the bactericidal effect of the Ag/TiO2 coating. The viability of E. coli was reduced to a virtually zero level within only 20 min. P. aeruginosa and S. aureus, however, respectively, required 60 and 90 min to reach a similar reduction in survival. Thuptimdang et al. [12] determined that unlike immature biofilms, mature biofilms and EPS reduce the susceptibility of the Pseudomonas putida biofilm to AgNPs. Kalishwaralal et al. [14] confirmed a high antibiofilm efficacy (P. aeruginosa and Staphylococcus epidermidis) following a 24 h treatment with silver nanoparticles. In our research, the greatest anti-biofilm effect in relation to the tested clinical strains was also noted after 24 h of incubation of bacteria in the presence of SiO2/Ag0 and TiO2/Ag0. The amount of biofilm formed by the strains isolated from urine decreased to 36% (SiO2/Ag0) and 35% (TiO2/Ag0). The reduction in biofilm mass formed by the strains isolated from the bronchoalveolar lavage fluid was 38% (SiO2/Ag0) and 28% (TiO2/Ag0).
Nonetheless, there are several publications focusing on the antibacterial activity of modified silver nanocomposites. Naik et al. [59] proved that the AgCl-TiO2-coated surface inhibits the biofilm formation of P. aeruginosa at a concentration of 125 µg/cm2. Flores et al. [60] showed that the surface coated with citrate-capped silver nanoparticles immobilized on TiO2 prevented the attachment of P. aeruginosa cells.
We proved four benefits of silver nanoforms’ usage as an antibacterial factor: (1) the antibacterial efficacy of the tested SiO2/Ag0 and TiO2/Ag0 samples is higher than the control SiO2 and TiO2 samples; (2) no differences in susceptibility were detected between the bronchial and urinary isolates’ P. aeruginosa strains; (3) both SiO2/Ag0 and TiO2/Ag0 indicate a high level of antibiofilm activity at the concentration of 1, 2, and 4 × MBIC; (4) TiO2/Ag0 more strongly inhibits the biofilm mass production and the survival of bacteria in the biofilm than SiO2/Ag0. With such properties and their favorable and inhibitory impact on P. aeruginosa biofilm formation, these nanoforms offer an excellent solution as components of materials used in medical equipment manufacturing (catheters, intubation tubes, endoscopes).
4. Materials and Methods
4.1. Bacterial Strains
In this work, the reference PAO1 strain (P. aeruginosa ATCC 15692) and ten clinical strains of P. aeruginosa were used. The clinical strains were obtained from the DiaLab Medical Laboratory in Wroclaw. The preliminary identification of these strains included an analysis of their phenotypic-properties-growth type and colony morphology on Columbia blood agar and MacConkey agar. Next, species affiliation was determined by manual cytochrome oxidase assay and an evaluation of biochemical properties using an automated VITEK 2 compact system (bioMérieux, France). The GN VITEK 2 compact card (Biomerieux, France) was used to identify non-fermenting bacilli.
The clinical P. aeruginosa strains were sourced from patients hospitalized in intensive care units. Five of them were isolated from the bronchoalveolar lavage fluid of patients with respiratory system infections (No 0013, 0024, 3, 472, and 669), the remaining five from the urine of patients suffering from urinary tract infections (No 124, 137, 300, 328, and 407).
4.2. Silver Nanoforms (SiO2/Ag0, TiO2/Ag0)
In the sol–gel process, TiO2 was prepared by the following steps: hydrolysis and polycondensation of titanium alkoxides (Ti(OR)n). The sol–gel reaction occurred in an acetone environment, in the presence of a catalyst (ammonium hydroxide) and hydrofluoric acid. Titanium n-butoxide and hydrofluoric acid were added drop wise to acetone with stirring. The gel was then washed in methyl alcohol and water and centrifuged at 4000 rpm. Ready gel was dried at 80 °C. The silica carrier was prepared the same way, but silica alkoxide (TEOS) was used in ethanolic solution. In order to synthesize silver nanoparticles, inorganic carriers (SiO2 and TiO2) were impregnated in a solution of diammina silver [Ag(NH3)2]+ (0.2M) and a reducing factor (glucose, 6.8%). The diamminesilver(I) nitrate solution (Tollen’s reagent) was prepared according to Kędziora et al. [27]. The silver content in these carriers is 10% of Ag0.
4.3. Antibiotic Susceptibility
Susceptibility of isolates to different antibiotics was determined by the Kirby–Bauer method according to the recommendations of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) [61]. Antibiotic disks (BBL Sensi-Disc, Becton Dickinson, Warsaw, Poland) tested were amikacin (AN, 30 μg), ceftazidime (CAZ, 30 μg), ciprofloxacin (CIP, 5 μg), imipenem (IPM, 10 μg), meropenem (MEM, 10 μg), and piperacillin/tazobactam (TZP, 100 μg/10 μg). According to the EUCAST guidelines, the sensitivity of bacteria to colistin (CL, 10 μg) was determined by the broth microdilution method [45]. A synergistic test with Imipenem-EDTA Double-Disk was used to denote the ability to produce beta-lactamase strains [62].
4.4. Prevalence of Silver Resistance in Tested Bacteria Strains
To verify the silver resistance among tested P. aeruginosa strains, the prevalence of the silE gene located on the pMG101 plasmid was determined [40]. Briefly, the plasmid was isolated using the Plasmid Mini Kit (A&A Biotechnology, Gdańsk, Poland) according to the attached instructions from an overnight culture of P. aeruginosa strains in Luria-Broth (Biomaxima, Lublin, Poland). The E. coli J53 strain was used as a positive control. The reaction mixture was as follows: 10 µL Phusion Flash High Fidelity PCR Master Mix (Thermo Scientific, Waltham, MA, USA), 1 µL forward primer silE (AGGGGAAACGGTCTGACTTC, Genomed), 1 µL reverse primer silE (ATATCCATGAGCGGGTCAAC, Genomed), 2.5 µL isolated plasmid, and the required volume of ultrapure water (A&A Biotechnology). The final volume of all reactions was 20 μL. The positive (with plasmid from E. coli J53) and negative (sample without any of the plasmid) controls were also performed (and were marked on the image). The following steps were established for final products of PCR: 10 s at 98 °C (initial denaturation) followed by 30 cycles of 98 °C for 5 s (denaturation), 62 °C for 5 s (annealing), 72 °C for 5 s (elongation), and 72 °C for 1 min (final elongation). PCR was carried out on a T100 Thermal Cycler (BioRad, Warsaw, Poland). A total of 15 μL of each sample and 5 μL of Loading buffer for agarose electrophoresis (A&A Biotechnology) were loaded to a 2% agarose Tris–acetate–EDTA (TAE, Thermo Scientific) gel containing 0.002% Midori Green Advance DNA Stain (Nippon Genetics, Tokyo, Japan); ectrophoresis was carried out during 1.5 h at 7.5 V/cm. A 5 μL DNA Ladder marker (0.1 μg/mL. 100–1000 bp, A&A Biotechnology) was applied on the gel. Images of the gels were carried out on a Gel Doc XR + (BioRad).
4.5. Preparation of Bacterial Suspensions
The P. aeruginosa strains were grown overnight on Mueller–Hinton agar (MHA, Biocorp, Warsaw, Poland), and then bacterial cells were transferred to fresh Mueller–Hinton broth (MHB, Biocorp, Warsaw, Poland) and incubated at 37 °C for 5 h in the shaking water bath (Julabo SW22). Following incubation, while P. aeruginosa cells were in log phase growth, the bacterial suspensions were centrifuged (4000 rpm for 20 min) and suspended in phosphate-buffered saline (PBS, POCH, Gliwice, Poland) to reach the final density 0.5 in McFarland (108 CFU/mL). The bacterial suspensions prepared in this way were used in the subsequent stages of the research.
4.6. Determination of the Minimal Inhibitory Concentration (MIC)
The MIC was determined by the broth microdilution method with the reference to the Clinical and Laboratory Standards Institute guidelines [63]. For this purpose, initial inoculums of bacteria (0.5 × 105 CFU/mL) in MHB were exposed to a series of concentrations (0.5–8192 μg/mL) of SiO2/Ag0, TiO2/Ag0, SiO2, TiO2 and incubated for 24 h at 37 °C. The experiments were conducted on 96-well microtiter polystyrene plates with a final volume of 100 μL. The MIC was taken as the lowest concentration at which there was a noticeable inhibition in the growth of bacteria.
4.7. Determination of the Minimal Biofilm Inhibitory Concentration (MBIC)
Biofilm formation was assessed by the standard crystal violet colorimetric assay using P. aeruginosa strains [64]. In brief, 100 µL of the bacterial suspension in MHB of each strain (106 CFU/mL) were transferred into wells of U-bottom 96-well microtiter polystyrene plates. To these suspensions, SiO2/Ag0 and TiO2/Ag0 were added to reach their concentrations of 8, 16, 32, 64, 128, 256, 512, 1024, 2048, 4096, or 8192 µg/mL. Wells containing bacterial suspensions without SiO2/Ag0 and TiO2/Ag0 served as controls. The plates were incubated at 37 °C for 24 h. At the end of the culture period, the content of the plates was removed by inverting the plates, and then, washing the wells twice with 200 µL PBS solution. Then, the microplate wells were stained with 125 µL of 0.1% (w/v) crystal violet for 15 min at 37 °C. The excess stain was rinsed off by placing the microplate under running PBS solution. The microplate was air-dried, and the dye bound to the adherent cells was resolubilized with 200 µL of 96% (v/v) ethanol (POCH, Warsaw, Poland) per well. A suspension of bacteria in MHB was used as a control. The intensity coloration was measured at a wavelength of 595 nm in a microplate reader (Asys Hitachi 340, Driver Version: 4.02; Biogenet, Józefów, Poland). The MBIC was defined as the lowest concentration which inhibited at least 90% biofilm formation. Each test was performed in quadruplicate with three independent repetitions.
4.8. Effects of SiO2/Ag0 and TiO2/Ag0 on Biofilm Formation
The effect of SiO2/Ag0 and TiO2/Ag0 on biofilm formation was evaluated in 96-well polystyrene U-bottom plates [64]. Wells of the microplate were filled with 100 µL of MHB containing SiO2/Ag0 or TiO2/Ag0 at concentrations of 0.25 × MBIC, 0.5 × MBIC, 1 × MBIC, 2 × MBIC, and 4 × MBIC. Wells containing MHB without nanoparticles were used as controls. The overnight bacterial cultures in MHB were diluted to a final bacterial concentration of 106 CFU/mL. Next, 10 µL the diluted bacterial culture were added to each well of the microtiter plates. Plates were incubated aerobically for 6, 12, 24, 48, and 72 h at 37 °C. After incubation, the supernatant was removed, and each well was twice washed with 200 µL PBS solution to remove free-floating cells. Then, the microplate wells were stained with 125 µL of 0.1% (w/v) crystal violet for 15 min at 37 °C. The excess stain was rinsed off by placing the microplate under running PBS solution. Finally, the dye bound to the cells was solubilized by adding 200 μL of 95% (v/v) ethanol to each well, and after 15 minutes the optical density (OD) was measured at 595 nm using a microplate reader. All experiments were performed in triplicate and repeated three times. Based on acquired readings, standard deviations were calculated, and a cut-off value (ODc) was established. ODc is defined as three standard deviations (SD) above the mean OD of the negative control (medium): ODc = average OD of negative control + (3 × SD of negative control). The ODc value was 0.096. Each isolate was categorized according to the following criteria: non-biofilm producer (OD ≤ ODc); weak-biofilm producer (ODc < OD ≤ 2 × ODc); moderate-biofilm producer (2 × ODc < OD ≤ 4 × ODc); strong-biofilm producer (4 × ODc < OD) [65].
4.9. Effects of SiO2/Ag0 and TiO2/Ag0 on the Count of Live Bacteria in Biofilm
In this assay, SiO2/Ag0 and TiO2/Ag0 were tested at a concentration of 0.5 × MBIC. It is the subinhibitory concentration of MBIC which does not inhibit biofilm formation. The appropriate concentrations of SiO2/Ag0 and TiO2/Ag0 were added to microtiter wells containing bacterial suspensions. The survival of bacteria in biofilms was established after 6, 12, 24, 48, and 72 h incubation. After these times, the contents of the wells were washed with PBS to remove nonadherent bacteria. Then, biofilms were manually scraped using a sterile spatula, transferred into microtubes containing PBS, and vortexed for 3 min to disperse the biofilm uniformly in water. The CFU (colony forming unit) values in 1 mL were assessed by plating serial dilutions on MHA.
4.10. Statistical Analysis
Differences in MICs values between tested nanoforms with silver (SiO2/Ag0, TiO2/Ag0) and their control (SiO2, TiO2) in the whole group as well as divided by the isolation place of bacteria were analyzed using the non-parametric Mann–Whitney test. Statistical differences between bacterial strains exposed to SiO2/Ag0, TiO2/Ag0, and unexposed (controls) were analyzed by the non-parametric Kruskal–Wallis test followed by a Dunn’s multiple comparison test. All statistical analysis was performed using STATISTICA v. 13.0 (StatSoft, Krakow, Poland). The results were considered statistically significant for p values < 0.05. All tests were carried out in triplicate, and the results were averaged.
5. Conclusions
Antibiotic resistance of microorganisms is a serious threat to human health. The times when antibiotics were the answer to all problems are gone, and only a very good understanding of structures such as biofilm and the mechanisms occurring in them will allow us to avert the danger posed by multidrug-resistant bacteria. The use of innovative methods to combat bacterial biofilm, including silver nanoforms, maybe the first step to creating another line of defense against microorganisms.
Our results indicate that the inorganic compounds (SiO2, TiO2) after nanotechnological modification (e.g., embodied with silver nanoparticles Ag0) may be used with satisfactory results as antibacterial agents against multidrug resistant P. aeruginosa strains. SiO2/Ag0 and TiO2/Ag0 significantly inhibited the process of biofilm formation and reduced the number of viable cells in biofilm consortia. This type of nanoform can be used as a material for covering catheters and implants, which will reduce the risk of their colonization by bacterial strains.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms23010284/s1.
Author Contributions
Conceptualization, K.K., A.K. and D.W.; methodology, K.K., A.K. and D.W.; validation, K.K., A.K., B.S., G.B.-P. and D.W.; formal analysis, K.K., A.K., B.S. and D.W.; investigation, K.K., A.K. and B.S.; writing—original draft preparation, K.K., A.K., B.S. and D.W.; writing—review and editing, K.K., A.K., B.S., G.B.-P. and D.W.; visualization, A.K. and D.W.; project administration, K.K.; funding acquisition, K.K. and G.B.-P. All authors have read and agreed to the published version of the manuscript.
Funding
Publication of this article was financially supported by the Excellence Initiative – Research University (IDUB) programme for the University of Wroclaw.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding authors.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Michiels, J.E.; Van den Bergh, B.; Verstraeten, N.; Fauvart, M.; Michiels, J. In vitro emergence of high persistence upon periodic aminoglycoside challenge in the ESKAPE pathogens. Antimicrob. Agents Chemother. 2016, 60, 4630–4637. [Google Scholar] [CrossRef]
- Rice, L.B. Federal funding for the study of antimicrobial resistance in nosocomial pathogens: No ESKAPE. J. Infect. Dis. 2008, 19, 1079–1081. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.B. Progress and challenges in implementing the research on ESKAPE pathogens. Infect. Control Hosp. Epidemiol. 2010, 31, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Streeter, K.; Katoul, M. Pseudomonas aeruginosa: A review of their pathogenesis and prevalence in clinical settings and the environment. Infect. Epidemiol. Med. 2016, 2, 25–32. [Google Scholar] [CrossRef]
- Fakhkharia, P.; Tajeddinb, E.; Azimiradb, M.; Salmanzadeh-Ahrabi, S.; Abdi-Alia, A.; Nikmaneshd, B.; Eshratie, B.; Gouya, M.M.; Owliaf, P.; Zalig, M.R.; et al. Involvement of Pseudomonas aeruginosa in the occurrence of community and hospital acquired diarrhea, and its virulence diversity among the stool and the environmental samples. Int. J. Environ. Health Res. 2020, 19, 1–11. [Google Scholar] [CrossRef]
- Alhazmi, A. Pseudomonas aeruginosa—Pathogenesis and pathogenic mechanisms. Int. J. Biol. 2015, 7, 44–67. [Google Scholar] [CrossRef]
- Huang, Z.; Jiang, Y.; Liang, J. Pathogenesis could be one of the anti-cheating mechanisms for Pseudomonas aeruginosa society. Med. Hypotheses 2011, 76, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus biofilm: An emerging battleground in microbial communities. Antimicrob. Resist. Infect. Control 2019, 8, 76. [Google Scholar] [CrossRef]
- Mikhailova, E.O. Silver nanoparticles: Mechanism of action and probable bio-application. J. Funct. Biomater. 2020, 11, 84. [Google Scholar] [CrossRef]
- O’Loughlin, C.T.; Miller, L.C.; Siryaporn, A.; Martin, K.; Semmelhack, F.; Bassler, B.L. A quorum-sensing inhibitor blocks Pseudomonas aeruginosa virulence and biofilm formation. Proc. Natl. Acad. Sci. USA 2013, 110, 17981–17986. [Google Scholar] [CrossRef]
- Ross, A.S.; Baliga, C.; Verma, P.; Duchin, J.; Gluck, M.A. Quarantine process for the resolution of uodenoscope-associated transmission of multidrug-resistant Escherichia coli. Gastrointest. Endosc. 2015, 82, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Thuptimdang, P.; Limpiyakorn, T.; McEvoy, J.; Prüß, B.M.; Khan, E. Effect of silver nanoparticles on Pseudomonas putida biofilms at different stages of maturity. J. Hazard. Mater. 2001, 290, 127–133. [Google Scholar] [CrossRef]
- Arancibia, F.; Bajer, T.T.; Ewig, S.; Mensa, J.; Gonzalez, J.; Niederman, M.S.; Torres, A. Community-acquired pneumonia due to gram-negative bacteria and Pseudomonas aeruginosa: Incidence, Risk and Prognosis. Arch. Intern. Med. 2002, 162, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- Kalishwaralal, K.; BarathManiKanth, S.; Pandian, S.R.; Deepak, V.; Gurunathan, S. Silver nanoparticles impede the biofilm formation by Pseudomonas aeruginosa and Staphylococcus epidermidis. Colloids Surf. B Biointerfaces 2010, 79, 340–344. [Google Scholar] [CrossRef]
- Sullivan, M.; Folytn-Arfa Kia, A.; Long, M.; Walsh, M.; Kavanagh, K.; McClean, S.; Creaven, B.S. Isolation and characterization of silver(I) complexes of substituted coumarin-4-carboxylates which are effective against Pseudomonas aeruginosa biofilms. Polyhedron 2014, 67, 549–559. [Google Scholar] [CrossRef]
- Parasuraman, P.; Devadatha, B.; Sarma, V.V.; Ranganathan, S.; Ampasala, D.R.; Reddy, D.; Kumavath, R.; Kim, I.W.; Patel, S.K.S.; Kalia, V.C.; et al. Inhibition of microbial quorum sensing mediated virulence factors by Pestalotiopsis sydowiana. J. Microbiol. Biotechnol. 2020, 30, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Dhanalekshmi, K.I.; Meena, K.S. Comparison of antibacterial activities of Ag@TiO2 and Ag@SiO2 core—Shell nanoparticles. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 128, 887–890. [Google Scholar] [CrossRef]
- Joshi, A.S.; Singh, P.; Mijakovic, I. Interactions of gold and silver nanoparticles with bacterial biofilms: Molecular interactions behind inhibition and resistance. Int. J. Mol. Sci. 2020, 21, 7658. [Google Scholar] [CrossRef]
- Otari, S.V.; Patel, S.K.S.; Kalia, V.C.; Kim, I.W.; Lee, J.K. Antimicrobial activity of biosynthesized silver nanoparticles decorated silica nanoparticles. Indian J. Microbiol. 2019, 59, 379–382. [Google Scholar] [CrossRef]
- Fletcher, P.D.; Holt, B.L. Controlled silanization of silica nanoparticles to stabilize foams, climbing films, and liquid marbles. Langmuir 2011, 27, 12869–12876. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, W.; Xue, C.; Zhou, S.; Lan, F.; Bi, L.; Xu, H.; Yang, X.; Zeng, F.D. Toxicity and penetration of TiO2 nanoparticles in hairless mice and porcine skin after subchronic dermal exposure. Toxicol. Lett. 2009, 191, 1–8. [Google Scholar] [CrossRef]
- Athinarayanan, J.; Periasamy, V.S.; Alsaif, M.A.; Al-Warthan, A.A.; Alshatwi, A.A. Presence of nanosilica (E551) in commercial food products: TNF-mediated oxidative stress and altered cell cycle progression in human lung fibroblast cells. Cell Biol. Toxicol. 2014, 30, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.L.; Woods, E.; Nutekpor, M.; Bowler, P.; Radford, A.; Cochrane, C. Prevalence of silver resistance in bacteria isolated from diabetic foot ulcers and efficacy of silver-containing wound dressings. Ostomy Wound Manag. 2008, 54, 30–40. [Google Scholar]
- Finley, P.J.; Norton, R.; Austin, C.; Mitchell, A.; Zank, S.; Durham, P. Unprecedented silver resistance in clinically isolated Enterobacteriaceae: Major implications for burn and wound management. Antimicrob. Agents Chemother. 2015, 59, 4734–4741. [Google Scholar] [CrossRef] [PubMed]
- Hosny, A.E.M.; Rasmy, S.A.; Aboul-Magd, D.S.; Kashef, M.T.; El-Bazza, Z.E. The increasing threat of silver-resistance in clinical isolates from wounds and burns. Infect. Drug Resist. 2019, 12, 1985–2001. [Google Scholar] [CrossRef]
- Kędziora, A.; Gorzelańczyk, K.; Bugla-Płoskońska, G. Positive and negative aspects of silver nanoparticles usage. Biol. Int. 2013, 53, 67–76. [Google Scholar]
- Kędziora, A.; Stręk, W.; Kepiński, L.; Bugla-Płoskońska, G.; Doroszkiewicz, W. Synthesis and antibacterial activity of novel titanium dioxide doped with silver. J. Solgel. Sci. Technol. 2012, 62, 79–86. [Google Scholar] [CrossRef]
- Li, L.; Zhao, C.; Zhang, Y.; Yao, J.; Yang, W.; Hu, Q.; Wang, C.; Cao, C. Effect of stable antimicrobial nano-silver packaging on inhibiting mildew and in storage of rice. Food Chem. 2017, 215, 477–482. [Google Scholar] [CrossRef]
- Mohandas, A.; Krishnan, A.G.; Biswas, R.; Menon, D.; Nair, M.B. Antibacterial and cytocompatible nanotextured surface incorporating silver via single step hydrothermal processing. Mat. Sci. Eng. C Mater. Biol. Appl. 2017, 75, 115–124. [Google Scholar] [CrossRef]
- Wiglusz, R.J.; Kędziora, A.; Łukowiak, A.; Doroszkiewicz, W.; Stręk, W. Hydroxyapatites and europium (III) doped hydroxyapatites as a carrier of silver nanoparticles and their antimicrobial activity. J. Biomed. Nanotechnol. 2012, 8, 605–612. [Google Scholar] [CrossRef]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver nanoparticles as potential antibacterial agents. Molecules 2015, 18, 8856–8874. [Google Scholar] [CrossRef]
- Yin, I.X.; Zhao, I.S.; Mei, M.L.; Li, Q.; Yu, O.Y.; Chu, C.H. Use of silver nanomaterials for caries prevention: A concise review. Int. J. Nanomedicine. 2020, 6, 3181–3191. [Google Scholar] [CrossRef]
- Arokiyaraj, S.; Vincent, S.; Saravanan, M.; Lee, Y.; Oh, Y.K.; Kim, K.H. Green synthesis of silver nanoparticles using Rheum palmatum root extract and their antibacterial activity against Staphylococcus aureus and Pseudomonas aeruginosa. Artif. Cell Nanomed. 2017, 45, 372–379. [Google Scholar] [CrossRef]
- Shah, S.; Gaikwad, S.; Nagar, S.; Kulshrestha, S.; Vaidya, V.; Nawani, N.; Pawar, S. Biofilm inhibition and anti-quorum sensing activity of phytosynthesized silver nanoparticles against the nosocomial pathogen Pseudomonas aeruginosa. Biofouling 2019, 35, 34–49. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Pandit, S.; Garnæs, J.; Tunjic, S.; Mokkapati, V.R.S.S.; Sultan, A.; Thygesen, A.; Mackevica, A.; Mateiu, R.V.; Daugaard, A.E.; et al. Green synthesis of gold and silver nanoparticles from Cannabis sativa (industrial hemp) and their capacity for biofilm inhibition. Int. J. Nanomed. 2018, 13, 3571–3591. [Google Scholar] [CrossRef]
- Mann, R.; Holmes, A.; McNeilly, O.; Cavaliere, R.; Sotiriou, G.A.; Rice, S.A.; Gunawan, C. Evolution of biofilm-forming pathogenic bacteria in the presence of nanoparticles and antibiotic: Adaptation phenomena and cross-resistance. J. Nanobiotechnol. 2021, 19, 291. [Google Scholar] [CrossRef] [PubMed]
- de Lacerda Coriolano, D.; de Souza, J.B.; Bueno, E.V.; Medeiros, S.M.F.R.D.S.; Cavalcanti, I.D.L.; Cavalcanti, I.M.F. Antibacterial and antibiofilm potential of silver nanoparticles against antibiotic-sensitive and multidrug-resistant Pseudomonas aeruginosa strains. Braz. J. Microbiol. 2021, 52, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Silver, S. Bacterial silver resistance: Molecular biology and uses and misuses of silver compounds. FEMS Microbiol. Rev. 2003, 27, 341–353. [Google Scholar] [CrossRef]
- Silver, S.; Gupta, A.; Matsui, K.; Lo, J.F. Resistance to Ag(I) cations in bacteria: Environments, genes and proteins. Met. Based Drugs 1999, 6, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Kedziora, A.; Wernecki, M.; Korzekwa, K.; Speruda, M.; Gerasymchuk, Y.; Lukowiak, A.; Bugla-Płoskońska, G. Consequences of long-term bacteria’s exposure to silver nanoformulations with different physico-chemical properties. Int. J. Nanomed. 2020, 15, 199–213. [Google Scholar] [CrossRef]
- Kiedrowski, M.R.; Horswill, A.R. New approaches for treating staphylococcal biofilm infections. Ann. N. Y. Acad. Sci. 2011, 1241, 104–121. [Google Scholar] [CrossRef] [PubMed]
- Alemayehu, D.; Casey, P.G.; McAuliffe, O.; Guinane, C.M.; Martin, J.G.; Shanahan, F.; Coffey, A.; Ross, R.P.; Hill, C. Bacteriophages ϕMR299-2 and ϕNH-4 can eliminate Pseudomonas aeruginosa in the murine lung and on cystic fibrosis lung airway cells. mBio 2012, 3, e00029-12. [Google Scholar] [CrossRef]
- Bugla-Płoskońska, G.; Leszkiewicz, A.; Borak, B.H.; Jasiorski, M.; Drulis-Kawa, Z.; Baszczuk, A.; Maruszewski, K.; Doroszkiewicz, W. Bactericidal properties of silica particles with silver islands located the surface. Int. J. Antimicrob. Agents 2007, 29, 746–748. [Google Scholar] [CrossRef] [PubMed]
- Jasiorski, M.; Leszkiewicz, A.; Brzeziński, S.; Bugla-Płoskońska, G.; Malinowska, G.; Borak, B.; Karbownik, I.; Baszczuk, A.; Stręk, W.; Doroszkiewicz, W. Textile with silver silica spheres: Its antimicrobial activity against Escherichia coli and Staphylococcus aureus. J. Solgel. Sci. Technol. 2009, 51, 330–334. [Google Scholar] [CrossRef]
- Kędziora, A.; Korzekwa, K.; Strek, W.; Pawlak, A.; Doroszkiewicz, W.; Bugla-PLoSkonska, G. Silver nanoforms as a therapeutic agent for killing Escherichia coli and certain ESKAPE pathogens. Curr. Microbiol. 2016, 73, 139–147. [Google Scholar] [CrossRef][Green Version]
- Gerasymchuk, Y.; Lukowiak, A.; Wedzynska, A.; Kedziora, A.; Bugla-PLoSkonska, G.; Piatek, D.; Bachanek, T.; Chernii, V.; Tomachynski, L.; Strek, W. New photosensitive nanometric graphite oxide composites as antimicrobial material with prolonged action. J. Inorg. Biochem. 2016, 159, 142–148. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Qayyum, S.; Khan, A.U. Antibiofilm efficacy of green synthesized graphene oxide-silver nanocomposite using Lagerstroemia speciosa floral extract: A comparative study on inhibition of gram-positive and gram-negative biofilms. Microb. Pathog. 2017, 103, 167–177. [Google Scholar] [CrossRef]
- Ravishankar, R.V.; Jamuna, B.A. Nanoparticles and their potential application as antimicrobials. In Science against Microbial Pathogens: Communicating Current Research and Technological Advances; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2011; pp. 197–209. [Google Scholar]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramirez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef]
- Fatima, S.; Ali, K.; Ahmed, B.; Al Kheraif, A.A.; Syed, A.; Elgorban, A.M.; Musarrat, J.; Lee, J. Titanium dioxide nanoparticles Induce inhibitory effects against planktonic cells and biofilms of human oral cavity isolates of Rothia mucilaginosa, Georgenia sp. and Staphylococcus saprophyticus. Pharmaceutics 2021, 13, 1564. [Google Scholar] [CrossRef]
- Altaf, M.; Zeyad, M.T.; Hashmi, M.A.; Manoharadas, S.; Hussain, S.A.; Abuhasile, M.S.A.; Almuzainid, M.A.M. Effective inhibition and eradication of pathogenic biofilms by titanium dioxide nanoparticles synthesized using Carum copticum extract. RSC Adv. 2021, 11, 19248. [Google Scholar] [CrossRef]
- Maurer-Jones, M.A.; Gunsolus, I.L.; Meyer, B.M.; Christenson, C.J.; Haynes, C.L. Impact of TiO2 nanoparticles on growth, biofilm formation, and flavin secretion in Shewanella oneidensis. Anal. Chem. 2013, 85, 5810–5818. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Qin, S.; Wei, Y.; Liu, S.; Peng, H.; Li, Q.; Luo, L.; Lv, M. Silver nanoparticles exert concentration-dependent influences on biofilm development and architecture. Cell Prolif. 2019, 52, e12616. [Google Scholar] [CrossRef]
- Palanisamy, N.K.; Ferina, N.; Amirulhusni, A.N.; Mohd-Zain, Z.; Hussaini, J.; Ping, L.J.; Durairaj, R. Antibiofilm properties of chemically synthesized silver nanoparticles found against Pseudomonas aeruginosa. J. Nanobiotechnol. 2014, 12, 2. [Google Scholar] [CrossRef]
- Poulin, M.B.; Kuperman, L.L. Regulation of Biofilm Exopolysaccharide Production by Cyclic Di-Guanosine Monophosphate. Front. Microbiol. 2021, 12, 730980. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, C.; Hou, J.; Wang, P.; You, G.; Miao, L. Effects of cerium oxide nanoparticles on bacterial growth and behaviors:induction of biofilm formation and stress respons. Environ. Sci. Pollut. Res. 2019, 26, 9293–9304. [Google Scholar] [CrossRef] [PubMed]
- Kamaraj, K.; George, R.P.; Anandkumar, B.; Parvathavarthini, N.; Kamachi Mudali, U. A Silver nanoparticle loaded TiO2 nanoporous layer for visible light induced antimicrobial applications. Bioelectrochemistry 2015, 106, 290–297. [Google Scholar] [CrossRef]
- Yao, Y.; Ohko, Y.; Sekiguchi, Y.; Fujishima, A.; Kubota, Y. Self-sterilization using silicone catheters coated with Ag and TiO2 nanocomposite thin film. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 2, 453–460. [Google Scholar] [CrossRef]
- Naik, K.; Kowshik, M. Anti-Biofilm efficacy of low temperature processed AgCl–TiO2 nanocomposite coating. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 34, 62–68. [Google Scholar] [CrossRef]
- Flores, C.Y.; Diaz, C.; Rubert, A.; Benítez, G.A.; Moreno, M.S.; Fernández Lorenzo de Mele, M.A.; Salvarezza, R.C.; Schilardi, P.L.; Vericat, C. Spontaneous adsorption of silver nanoparticles on Ti/TiO2 surfaces. Antibacterial effect on Pseudomonas aeruginosa. J. Colloid Interface Sci. 2010, 350, 402–408. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Routine and extended internal quality control for MIC determination and disk diffusion as recommended by EUCAST. Ver-sion 11.0. 2021. Available online: http://www.eucast.org (accessed on 9 December 2021).
- Lee, K.; Lim, Y.S.; Yong, D.; Yum, J.H.; Chong, Y. Evaluation of the Hodge test and the imipenem-EDTA double-disk synergy test for differentiating metallo-beta-lactamase producing isolates of Pseudomonas spp. and Acinetobacter spp. J. Clin. Microbiol. 2003, 41, 4623–4629. [Google Scholar] [CrossRef]
- National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Disc Susceptibility Tests, 6th ed.; National Committee for Clinical Laboratory Standards: Malvern, PA, USA, 1997. [Google Scholar]
- O’Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signaling pathways: A genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Stepanovic, S.; Vulcovic, D.; Hola, V.; Bonaventura, G.D.; Djukić, S.; Cirković, I.; Ruzicka, F. Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 2007, 13, 891–899. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).