Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning
Abstract
1. Introduction
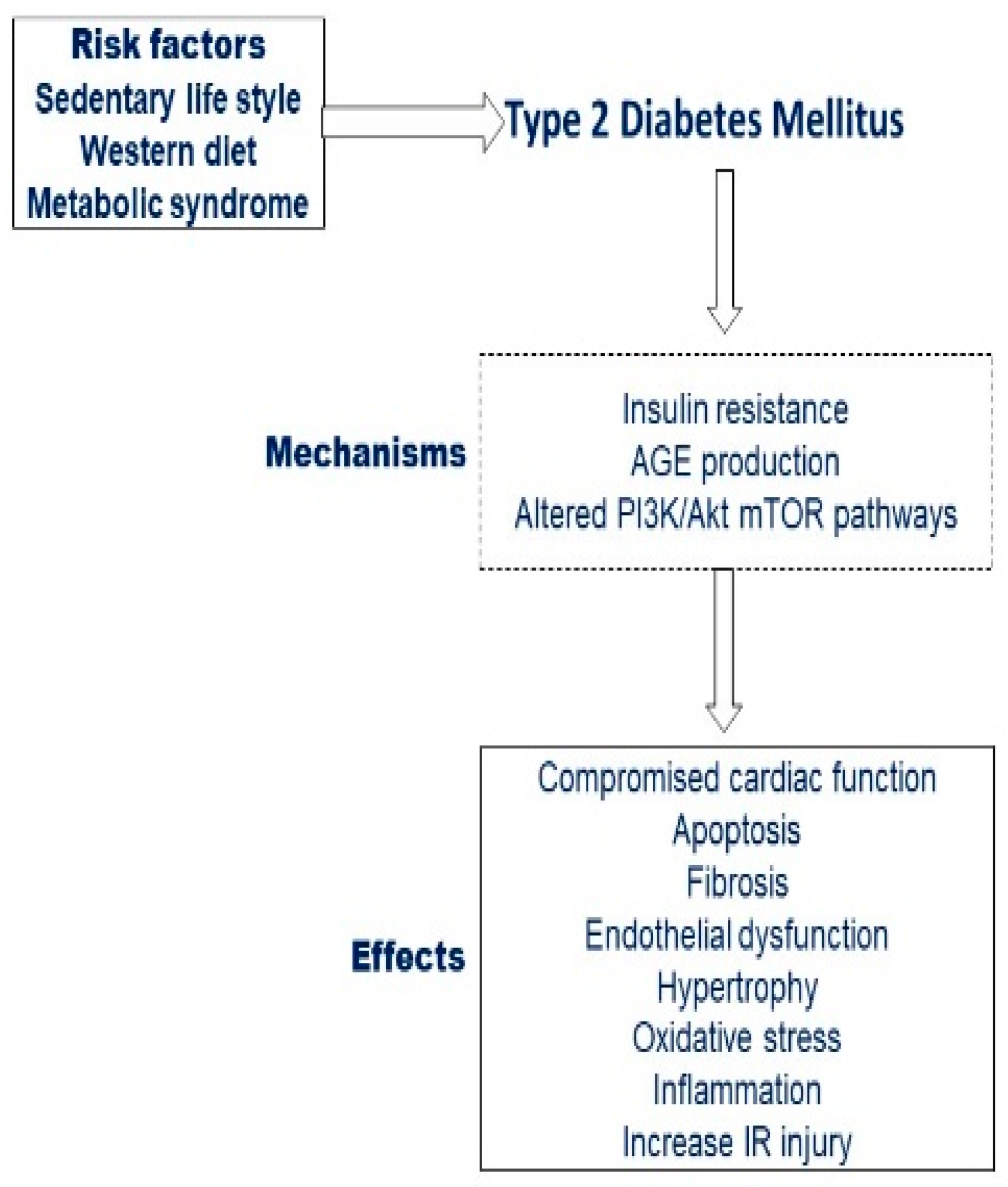
Diabetic/Metabolic Cardiomyopathy Definition
2. Myocardial Features of Diabetic Cardiomyopathy
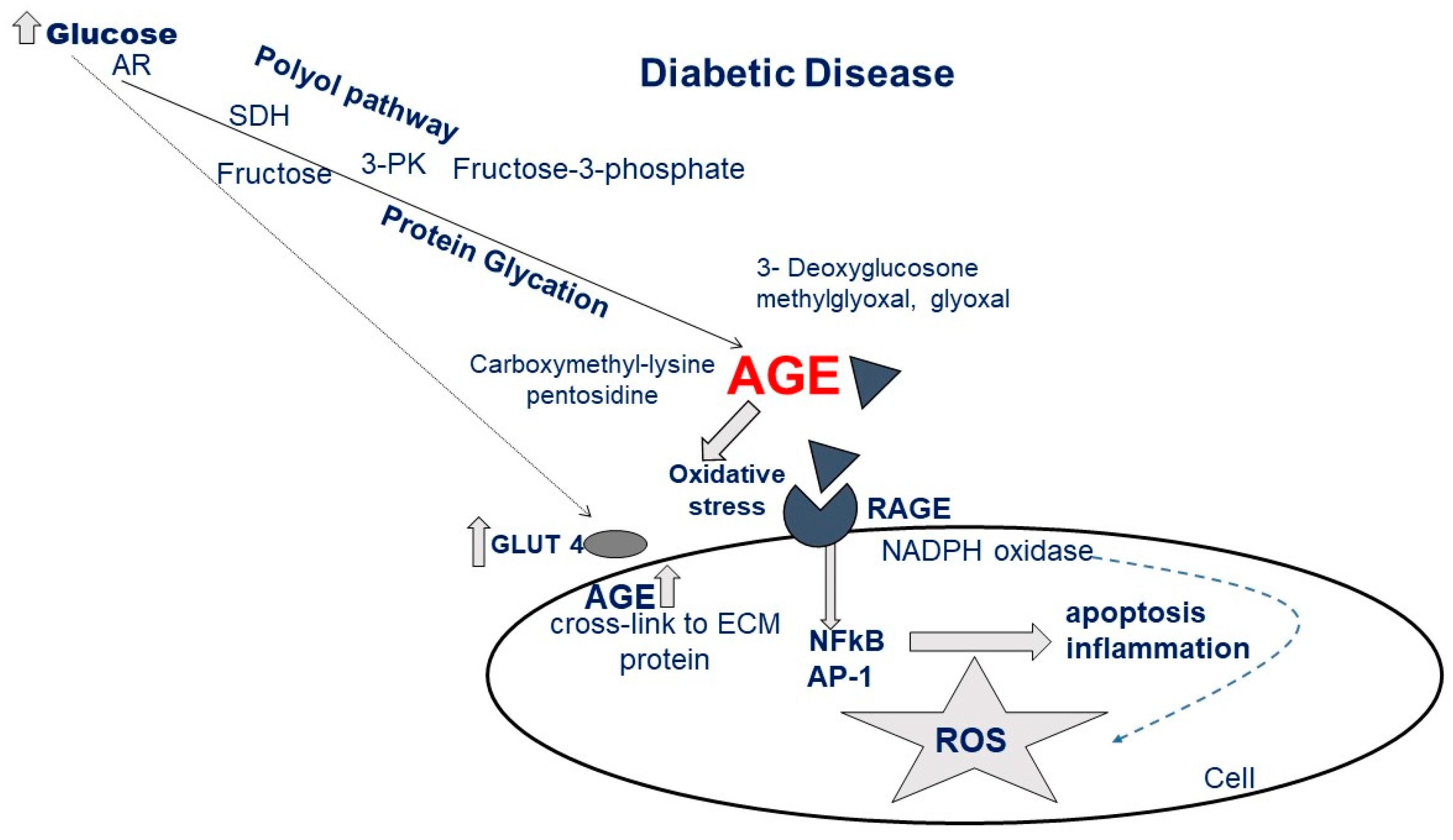
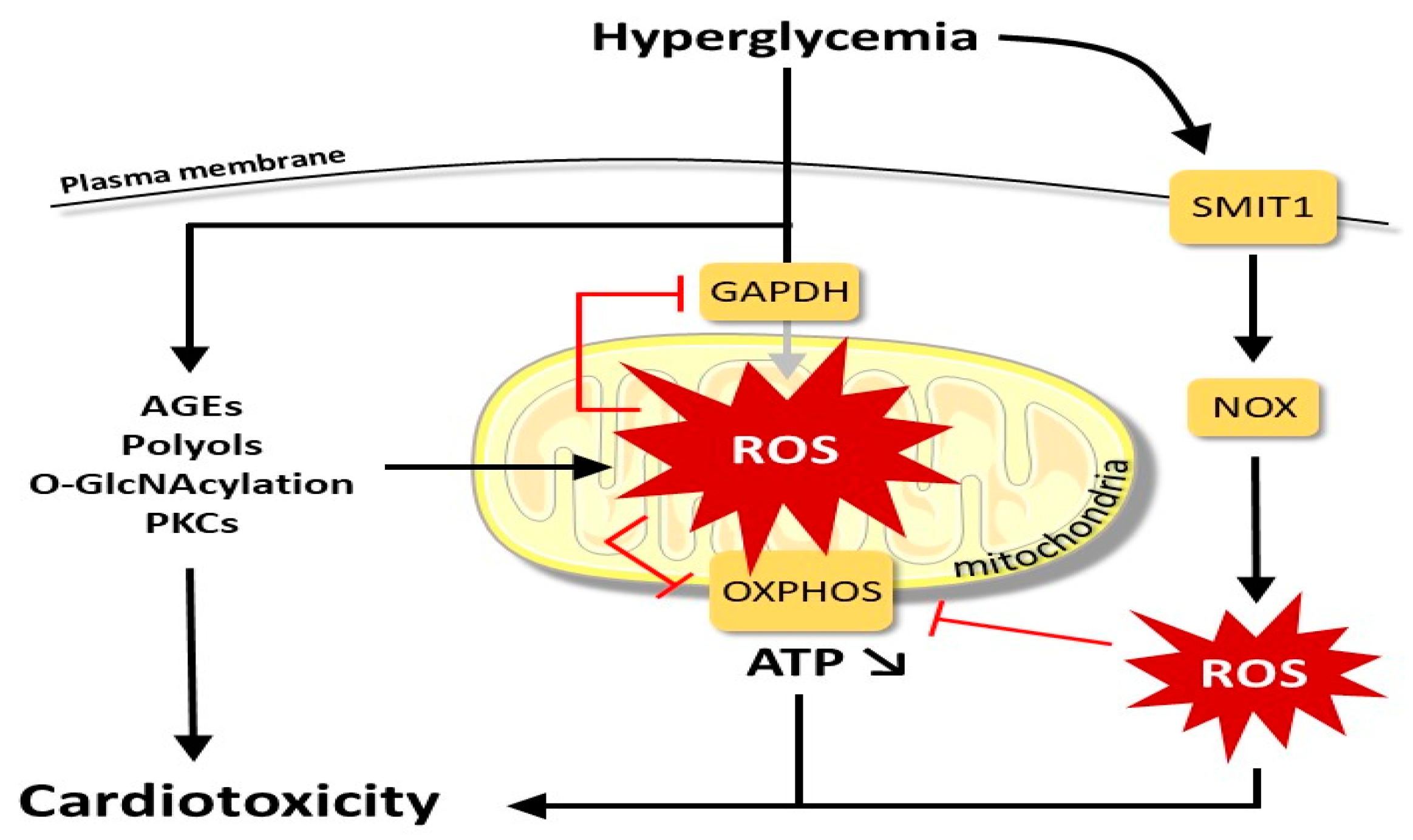
2.1. Molecular Alterations and Myocardial Damage Caused by Hyperglycemia
2.2. Diabetes Mellitus and Heart Failure, beyond the Established Risk Factor for Ischemic Heart Disease
3. Pathophysiology of Hyperglycemia and Diabetes in Cardiac Injury and Dysfunction
Effects of Hyperglycemia and Diabetes on Acute Myocardial Infarct Size
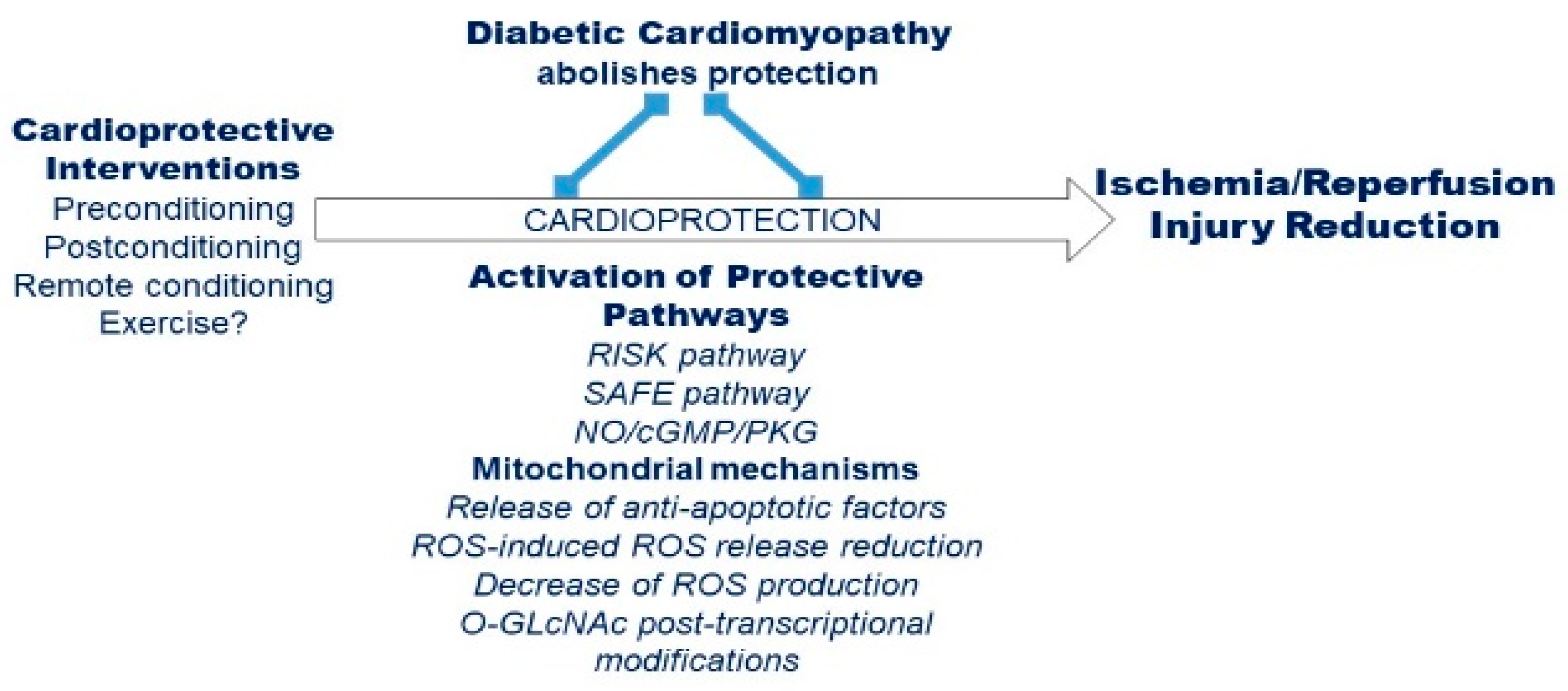
4. Effects of Remote Conditioning Protocols in Diabetes
5. Type 1 and 2 Diabetes Mellitus and the Cardiovascular Regulation during Exercise
5.1. Cardiovascular Dysregulation in T1DM
5.2. Cardiovascular Dysregulation in T2DM
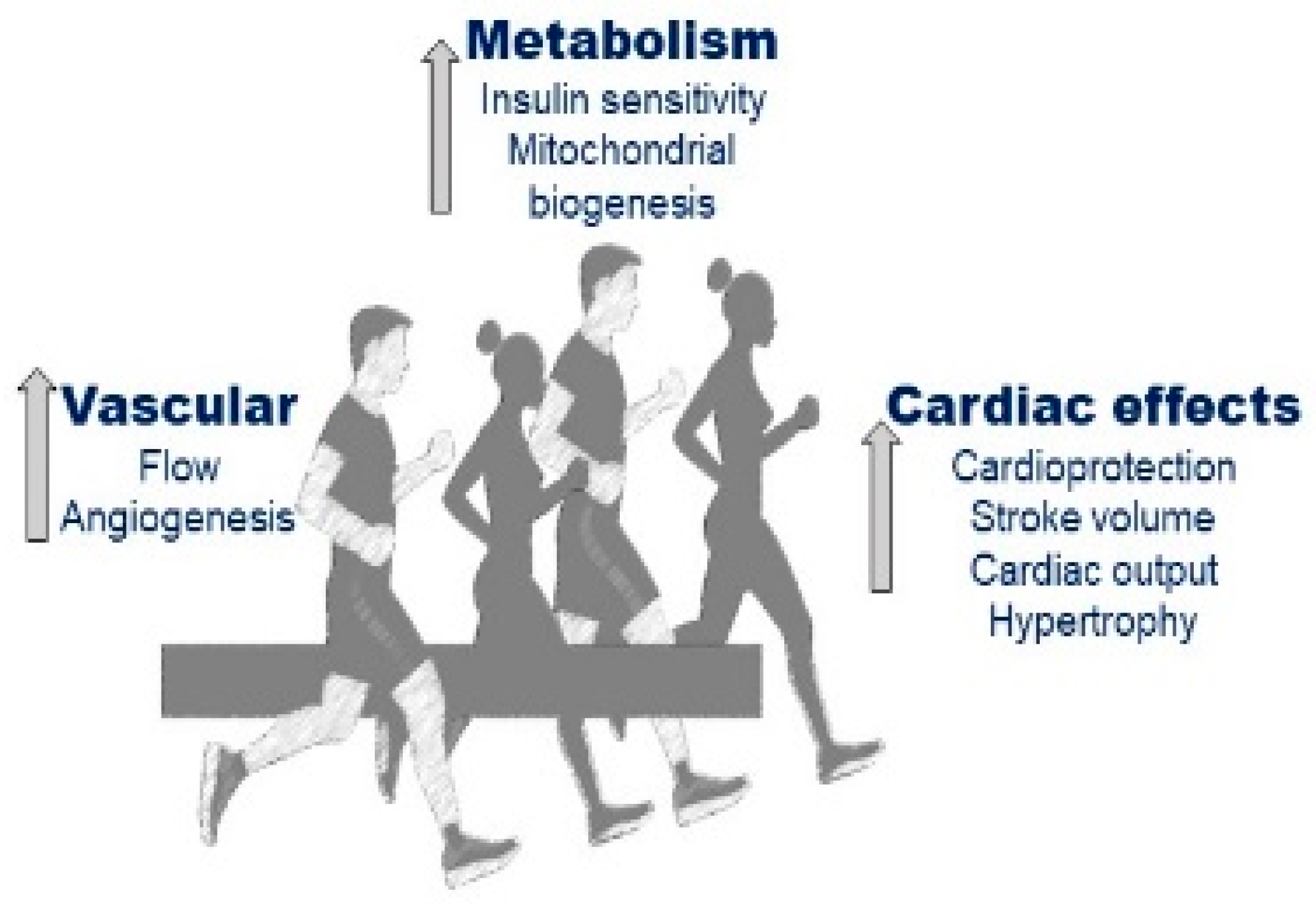
6. Exercise Training for the Management of Diabetes Mellitus: Classic Approaches
7. Exercise as Therapy for Diabetes Mellitus: Unconventional and Novel Exercise-Based Approaches
8. Conclusions and Future and Recommendations for Future Research
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-PK | phosphokinase |
| AET | aerobic exercise training |
| AGE | advanced glycation end-products |
| AR | aldose reductase |
| beta-MHC | beta-myosin heavy chain |
| BNP | type B natriuretic peptide |
| CK-MB | creatine kinase myocardial band |
| CO | cardiac output |
| DCM | diabetic cardiomyopathy |
| DM | diabetes mellitus |
| ECM | extracellular matrix |
| eNOS | endothelial nitric oxide synthase |
| ERK | extracellular signal-regulated kinases |
| ET-1 | endothelin-1 |
| FA | fatty acids |
| GLUT-4 | Glucose transporter type 4 |
| HAND | heart autonomic nervous system and neural crest derivatives |
| HbA1c | Hemoglobin A1c |
| HF | heart failure |
| HFpEF | heart failure with preserved ejection fraction |
| HFpEF | heart failure with reduced ejection fraction |
| HKII | hexokinase II |
| HR | heart rate |
| IR | Ischemia/Reperfusion |
| IRI | Ischemia Reperfusion Injury |
| JAK | Janus kinase |
| LV | left ventricle |
| MEF-2 | myogenic enhancer factor-2 |
| MS | metabolic syndrome |
| mTOR | mammalian target of rapamycin |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NO | nitric oxide |
| NOX2 | NADPH oxidase 2 |
| O-GlcNAc | O-Linked β-N-acetylglucosamine |
| PCI | percutaneous coronary intervention |
| per-RIC | perconditioning RIC |
| PKC | protein kinase C |
| PMI | periprocedural myocardial infarct |
| post-RIC | postconditioning RIC |
| pre-RIC | preconditioning RIC |
| PTEN | Phosphatase and tensin homolog deleted on chromosome 10 |
| RAGE | receptor for AGE |
| RIC | Remote ischemic conditioning |
| RISK | Reperfusion Injury Salvage Kinases |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen species |
| RT | resistance training |
| SAFE | Survivor Activating Factor Enhancement |
| SDH | sorbitol dehydrogenase |
| Smad | small mother against decapentaplegic |
| SMIT1 | sodium/myo-inositol cotransporter-1 |
| STAT-3 | signal transducer and activator of transcription 3 |
| SV | stroke volume |
| SVR | systemic vascular resistance |
| T1DM | type 1 diabetes mellitus |
| T2DM | type 2 diabetes mellitus |
| TGF-β | Transforming growth factor beta |
| TLR | Toll-like receptor |
| TNF-α | Tumor necrosis factor-α |
| VEGF | vascular endothelial growth factor |
References
- Rubler, S.; Dlugash, J.; Yuceoglu, Y.Z.; Kumral, T.; Branwood, A.W.; Grishman, A. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am. J. Cardiol. 1972, 30, 595–602. [Google Scholar] [CrossRef]
- Devereux, R.B.; Roman, M.J.; Paranicas, M.; O’Grady, M.J.; Lee, E.T.; Welty, T.K.; Fabsitz, R.R.; Robbins, D.; Rhoades, E.R.; Howard, B.V. Impact of diabetes on cardiac structure and function: The strong heart study. Circulation 2000, 101, 2271–2276. [Google Scholar] [CrossRef]
- Lee, M.; Gardin, J.M.; Lynch, J.C.; Smith, V.E.; Tracy, R.P.; Savage, P.J.; Szklo, M.; Ward, B.J. Diabetes mellitus and echocardiographic left ventricular function in free-living elderly men and women: The Cardiovascular Health Study. Am. Heart J. 1997, 133, 36–43. [Google Scholar] [CrossRef]
- Boudina, S.; Abel, E.D. Diabetic cardiomyopathy revisited. Circulation 2007, 115, 3213–3223. [Google Scholar] [CrossRef] [PubMed]
- Boudina, S.; Abel, E.D. Diabetic cardiomyopathy, causes and effects. Rev. Endocr. Metab. Disord. 2010, 11, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Bugger, H.; Abel, E.D. Molecular mechanisms of diabetic cardiomyopathy. Diabetologia 2014, 57, 660–671. [Google Scholar] [CrossRef] [PubMed]
- Bouthoorn, S.; Valstar, G.B.; Gohar, A.; den Ruijter, H.M.; Reitsma, H.B.; Hoes, A.W.; Rutten, F.H. The prevalence of left ventricular diastolic dysfunction and heart failure with preserved ejection fraction in men and women with type 2 diabetes: A systematic review and meta-analysis. Diab. Vasc. Dis. Res. 2018, 15, 477–493. [Google Scholar] [CrossRef]
- Nakamura, M.; Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat. Rev. Cardiol. 2018, 15, 387–407. [Google Scholar] [CrossRef]
- Jia, G.; Hill, M.A.; Sowers, J.R. Diabetic Cardiomyopathy: An Update of Mechanisms Contributing to This Clinical Entity. Circ. Res. 2018, 122, 624–638. [Google Scholar] [CrossRef]
- Singh, R.M.; Waqar, T.; Howarth, F.C.; Adeghate, E.; Bidasee, K.; Singh, J. Hyperglycemia-induced cardiac contractile dysfunction in the diabetic heart. Heart Fail. Rev. 2018, 23, 37–54. [Google Scholar] [CrossRef]
- Doliba, N.M.; Babsky, A.M.; Osbakken, M.D. The Role of Sodium in Diabetic Cardiomyopathy. Front. Physiol. 2018, 9, 1473. [Google Scholar] [CrossRef] [PubMed]
- Aon, M.A.; Tocchetti, C.G.; Bhatt, N.; Paolocci, N.; Cortassa, S. Protective mechanisms of mitochondria and heart function in diabetes. Antioxid Redox Signal. 2015, 22, 1563–1586. [Google Scholar] [CrossRef] [PubMed]
- Balteau, M.; Tajeddine, N.; de Meester, C.; Ginion, A.; Des Rosiers, C.; Brady, N.R.; Sommereyns, C.; Horman, S.; Vanoverschelde, J.L.; Gailly, P.; et al. NADPH oxidase activation by hyperglycaemia in cardiomyocytes is independent of glucose metabolism but requires SGLT1. Cardiovasc. Res. 2011, 92, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Van Steenbergen, A.; Balteau, M.; Ginion, A.; Ferté, L.; Battault, S.; Ravenstein, C.M.; Balligand, J.L.; Daskalopoulos, E.P.; Gilon, P.; Despa, F.; et al. Sodium-myoinositol cotransporter-1, SMIT1, mediates the production of reactive oxygen species induced by hyperglycemia in the heart. Sci. Rep. 2017, 7, 41166. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Pulinilkunnil, T.; Yuen, G.; Kewalramani, G.; An, D.; Qi, D.; Abrahani, A.; Rodrigues, B. Cardiomyocyte apoptosis induced by short-term diabetes requires mitochondrial GSH depletion. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H768–H776. [Google Scholar] [CrossRef]
- Ducheix, S.; Magré, J.; Cariou, B.; Prieur, X. Chronic O-GlcNAcylation and Diabetic Cardiomyopathy: The Bitterness of Glucose. Front. Endocrinol (Lausanne) 2018, 9, 642. [Google Scholar] [CrossRef]
- Qin, C.X.; Sleaby, R.; Davidoff, A.J.; Bell, J.R.; De Blasio, M.J.; Delbridge, L.M.; Chatham, J.C.; Ritchie, R.H. Insights into the role of maladaptive hexosamine biosynthesis and O-GlcNAcylation in development of diabetic cardiac. Pharm. Res. 2017, 116, 45–56. [Google Scholar] [CrossRef]
- Mailleux, F.; Gélinas, R.; Beauloye, C.; Horman, S.; Bertrand, L. O-GlcNAcylation, enemy or ally during cardiac hypertrophy development? Biochim. Biophys. Acta 2016, 1862, 2232–2243. [Google Scholar] [CrossRef]
- Fishman, S.L.; Sonmez, H.; Basman, C.; Singh, V.; Poretsky, L. The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: A review. Mol. Med. 2018, 24, 59. [Google Scholar] [CrossRef]
- Hegab, Z.; Mohamed, T.M.A.; Stafford, N.; Mamas, M.; Cartwright, E.J.; Oceandy, D. Advanced glycation end products reduce the calcium transient in cardiomyocytes by increasing production of reactive oxygen species and nitric oxide. FEBS Open Bio 2017, 7, 1672–1685. [Google Scholar] [CrossRef]
- Madonna, R.; Balistreri, C.R.; De Rosa, S.; Muscoli, S.; Selvaggio, S.; Selvaggio, G.; Ferdinandy, P.; De Caterina, R. Impact of Sex Differences and Diabetes on Coronary Atherosclerosis and Ischemic Heart Disease. J. Clin. Med. 2019, 8, 98. [Google Scholar] [CrossRef] [PubMed]
- Geraldes, P.; King, G.L. Activation of protein kinase C isoforms and its impact on diabetic complications. Circ. Res. 2010, 106, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Levelt, E.; Gulsin, G.; Neubauer, S.; McCann, G.P. MECHANISMS IN ENDOCRINOLOGY: Diabetic cardiomyopathy: Pathophysiology and potential metabolic interventions state of the art. Eur. J. Endocrinol 2018, 178, R127–R139. [Google Scholar] [CrossRef] [PubMed]
- Russo, I.; Frangogiannis, N.G. Diabetes-associated cardiac fibrosis: Cellular effectors, molecular mechanisms and therapeutic opportunities. J. Mol. Cell. Cardiol. 2016, 90, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.J.; Lee, A.S.; Chen, W.P.; Chang, W.L.; Tsai, Y.K.; Chiu, H.L.; Kuo, Y.H.; Su, M.J. Caffeic acid phenethyl amide ameliorates ischemia/reperfusion injury and cardiac dysfunction in streptozotocin-induced diabetic rats. Cardiovasc. Diabetol. 2014, 13, 98. [Google Scholar] [CrossRef]
- Aragno, M.; Mastrocola, R.; Medana, C.; Catalano, M.G.; Vercellinatto, I.; Danni, O.; Boccuzzi, G. Oxidative stress-dependent impairment of cardiac-specific transcription factors in experimental diabetes. Endocrinology 2006, 147, 5967–5974. [Google Scholar] [CrossRef]
- Aragno, M.; Mastrocola, R.; Alloatti, G.; Vercellinatto, I.; Bardini, P.; Geuna, S.; Catalano, M.G.; Danni, O.; Boccuzzi, G. Oxidative stress triggers cardiac fibrosis in the heart of diabetic rats. Endocrinology 2008, 149, 380–388. [Google Scholar] [CrossRef]
- Huynh, K.; Bernardo, B.C.; McMullen, J.R.; Ritchie, R.H. Diabetic cardiomyopathy: Mechanisms and new treatment strategies targeting antioxidant signaling pathways. Pharmacol. Ther. 2014, 142, 375–415. [Google Scholar] [CrossRef]
- Martín-Timón, I.; Sevillano-Collantes, C.; Segura-Galindo, A.; Del Cañizo-Gómez, F.J. Type 2 diabetes and cardiovascular disease: Have all risk factors the same strength? World J. Diabetes 2014, 5, 444–470. [Google Scholar] [CrossRef]
- Bernardo, B.C.; Weeks, K.L.; Pretorius, L.; McMullen, J.R. Molecular distinction between physiological and pathological cardiac hypertrophy: Experimental findings and therapeutic strategies. Pharmacol. Ther. 2010, 128, 191–227. [Google Scholar] [CrossRef]
- Galderisi, M.; Anderson, K.M.; Wilson, P.W.; Levy, D. Echocardiographic evidence for the existence of a distinct diabetic cardiomyopathy (the Framingham Heart Study). Am. J. Cardiol. 1991, 68, 85–89. [Google Scholar] [CrossRef]
- Feng, B.; Chen, S.; Chiu, J.; George, B.; Chakrabarti, S. Regulation of cardiomyocyte hypertrophy in diabetes at the transcriptional level. Am. J. Physiol. Endocrinol Metab. 2008, 294, E1119–E1126. [Google Scholar] [CrossRef] [PubMed]
- Morisco, C.; Marrone, C.; Trimarco, V.; Crispo, S.; Monti, M.G.; Sadoshima, J.; Trimarco, B. Insulin resistance affects the cytoprotective effect of insulin in cardiomyocytes through an impairment of MAPK phosphatase-1 expression. Cardiovasc. Res. 2007, 76, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Zuurbier, C.J.; Eerbeek, O.; Meijer, A.J. Ischemic preconditioning, insulin, and morphine all cause hexokinase redistribution. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H496-9. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Zhou, N.; Nan, Y.; Zhang, L.; Li, Y.; Hao, X.; Xiong, L.; Lau, W.B.; Ma, X.L.; Wang, H.; et al. Effective glycaemic control critically determines insulin cardioprotection against ischaemia/reperfusion injury in anaesthetized dogs. Cardiovasc. Res. 2014, 103, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Haffner, S.M.; Lehto, S.; Rönnemaa, T.; Pyörälä, K.; Laakso, M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N. Engl. J. Med. 1998, 339. [Google Scholar] [CrossRef]
- Donahoe, S.M.; Stewart, G.C.; McCabe, C.H.; Mohanavelu, S.; Murphy, S.A.; Cannon, C.P.; Antman, E.M. Diabetes and mortality following acute coronary syndromes. JAMA 2007, 298, 765–775. [Google Scholar] [CrossRef]
- Kannel, W.B.; Hjortland, M.; Castelli, W.P. Role of diabetes in congestive heart failure: The Framingham study. Am. J. Cardiol. 1974, 34, 29–34. [Google Scholar] [CrossRef]
- Rawshani, A.; Rawshani, A.; Franzén, S.; Sattar, N.; Eliasson, B.; Svensson, A.M.; Zethelius, B.; Miftaraj, M.; McGuire, D.K.; Rosengren, A.; et al. Risk Factors, Mortality, and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2018, 379, 633–644. [Google Scholar] [CrossRef]
- Mentz, R.J.; Kelly, J.P.; von Lueder, T.G.; Voors, A.A.; Lam, C.S.; Cowie, M.R.; Kjeldsen, K.; Jankowska, E.A.; Atar, D.; Butler, J.; et al. Non cardiac comorbidities in heart failure with reduced versus preserved ejection fraction. J. Am. Coll. Cardiol. 2014, 64, 2281–2293. [Google Scholar] [CrossRef]
- Kosiborod, M.; Rathore, S.S.; Inzucchi, S.E.; Masoudi, F.A.; Wang, Y.; Havranek, E.P.; Krumholz, H.M. Admission glucose and mortality in elderly patients hospitalized with acute myocardial infarction: Implications for patients with and without recognized diabetes. Circulation 2005, 111, 3078–3086. [Google Scholar] [CrossRef]
- Jun, J.H.; Jun, N.H.; Shim, J.K.; Shin, E.J.; Kwak, Y.L. Erythropoietin protects myocardium against ischemia-reperfusion injury under moderate hyperglycemia. Eur. J. Pharmacol. 2014, 745, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Deedwania, P.; Kosiborod, M.; Barrett, E.; Ceriello, A.; Isley, W.; Mazzone, T.; Raskin, P.; American Heart Association Diabetes Committee of the Council on Nutrition, Physical Activity, and Metabolism. Hyperglycemia and acute coronary syndrome: A scientific statement from the American Heart Association Diabetes Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation 2008, 117, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Eitel, I.; Hintze, S.; de Waha, S.; Fuernau, G.; Lurz, P.; Desch, S.; Schuler, G.; Thiele, H. Prognostic impact of hyperglycemia in nondiabetic and diabetic patients with ST-elevation myocardial infarction: Insights from contrast-enhanced magnetic resonance imaging. Circ. Cardiovasc. Imaging 2012, 5, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.; Miller, T.; Rutherford, B.D.; Gibbons, R.J.; Qureshi, M.; Kalynych, A.; Turco, M.; Schultheiss, H.P.; Mehran, R.; Krucoff, M.W.; et al. Comparison of myocardial reperfusion in patients undergoing percutaneous coronary intervention in ST-segment elevation acute myocardial infarction with versus without diabetes mellitus (from the EMERALD Trial). Am. J. Cardiol. 2007, 100, 206–210. [Google Scholar] [CrossRef]
- Przyklenk, K.; Maynard, M.; Greiner, D.L.; Whittaker, P. Cardioprotection with postconditioning: Loss of efficacy in murine models of type-2 and type-1 diabetes. Antioxid. Redox Signal. 2011, 14, 781–790. [Google Scholar] [CrossRef]
- Engbersen, R.; Riksen, N.P.; Mol, M.J.; Bravenboer, B.; Boerman, O.C.; Meijer, P.; Oyen, W.J.C.; Tack, C.; Rongen, G.A.; Smits, P. Improved resistance to ischemia and reperfusion, but impaired protection by ischemic preconditioning in patients with type 1 diabetes mellitus: A pilot study. Cardiovas Diabetol. 2012, 11, 124. [Google Scholar] [CrossRef]
- Ravingerová, T.; Neckar, J.; Kolar, F. Ischemic tolerance of rat hearts in acute and chronic phases of experimental diabetes. Mol. Cell Biochem. 2003, 249, 167–174. [Google Scholar] [CrossRef]
- Ravingerová, T.; Adameová, A.; Matejíková, J.; Kelly, T.; Nemčeková, M.; Kucharská, J.; Pecháňová, O.; Lazou, A. Subcellular mechanisms of adaptation in the diabetic myocardium: Relevance to ischemic preconditioning in the nondiseased heart. Exp. Clin. Cardiol. 2010, 15, 68–76. [Google Scholar]
- Yu, L.; Fan, C.; Li, Z.; Zhang, J.; Xue, X.; Xu, Y.; Zhao, G.; Yang, Y.; Wang, H. Melatonin rescues cardiac thioredoxin system during ischemia-reperfusion injury in acute hyperglycemic state by restoring Notch1/Hes1/Akt signaling in a membrane receptor-dependent manner. J. Pineal Res. 2017, 62. [Google Scholar] [CrossRef]
- Jonassen, A.K.; Sack, M.N.; Mjøs, O.D.; Yellon, D.M. Myocardial protection by insulin at reperfusion requires early administration and is mediated via Akt and p70s6 kinase cell-survival signaling. Circ. Res. 2001, 89, 1191–1198. [Google Scholar] [CrossRef]
- Kersten, J.R.; Schmeling, T.J.; Orth, K.G.; Pagel, P.S.; Warltier, D.C. Acute hyperglycemia abolishes ischemic preconditioning in vivo. Am. J. Physiol. 1998, 275, H721–H725. [Google Scholar] [CrossRef]
- Balakumar, P.; Sharma, N.K. Healing the diabetic heart: Does myocardial preconditioning work? Cell Signal. 2012, 24, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Gurel, E.; Ustunova, S.; Kapucu, A.; Yilmazer, N.; Eerbeek, O.; Nederlof, R.; Hollmann, M.W.; Demirci-Tansel, C.; Zuurbier, C.J. Hexokinase cellular trafficking in ischemia-reperfusion and ischemic preconditioning is altered in type I diabetic heart. Mol. Biol. Rep. 2013, 40, 4153–4160. [Google Scholar] [CrossRef]
- Femminò, S.; Pagliaro, P.; Penna, C. Obesity and cardioprotection. Curr. Med. Chem. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ferdinandy, P.; Hausenloy, D.J.; Heusch, G.; Baxter, G.F.; Schulz, R. Interaction of risk factors, comorbidities, and comedications with ischemia/reperfusion injury and cardioprotection by preconditioning, postconditioning, and remote conditioning. Pharmacol. Rev. 2014, 66, 1142–1174. [Google Scholar] [CrossRef] [PubMed]
- Gho, B.C.; Schoemaker, R.G.; van den Doel, M.A.; Duncker, D.J.; Verdouw, P.D. Myocardial protection by brief ischemia in noncardiac tissue. Circulation 1996, 94, 2193–2200. [Google Scholar] [CrossRef]
- Crimi, G.; Pica, S.; Raineri, C.; Bramucci, E.; De Ferrari, G.M.; Klersy, C.; Ferlini, M.; Marinoni, B.; Repetto, A.; Romeo, M.; et al. Remote ischemic post-conditioning of the lower limb during primary percutaneous coronary intervention safely reduces enzymatic infarct size in anterior myocardial infarction: A randomized controlled trial. JACC Cardiovasc. Interv. 2013, 6, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.B.; Liu, Y.; Zhu, Y.; Yin, G.L.; Wang, R.P.; Zhang, Y.; Zhu, J.; Jiang, W. Remote preconditioning, perconditioning, and postconditioning: A comparative study of their cardio-protective properties in rat models. Clinics 2013, 68, 263–268. [Google Scholar] [CrossRef]
- Tyagi, S.; Singh, N.; Virdi, J.K.; Jaggi, A.S. Diabetes abolish cardioprotective effects of remote ischemic conditioning: Evidences and possible mechanisms. J. Physiol. Biochem. 2019, 75, 19–28. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Yellon, D.M. New directions for protecting the heart against ischaemia-reperfusion injury: Targeting the Reperfusion Injury Salvage Kinase (RISK)-pathway. Cardiovasc. Res. 2004, 61, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Pagliaro, P.; Penna, C. Redox signalling and cardioprotection: Translatability and mechanism. Br. J. Pharmacol. 2015, 172, 1974–1995. [Google Scholar] [CrossRef]
- Rossello, X.; Yellon, D.M. The RISK pathway and beyond. Basic Res. Cardiol. 2017, 113, 2. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, M.; Tropak, M.; Diaz, R.J.; Suto, F.; Surendra, H.; Kuzmin, E.; Li, J.; Gross, G.; Wilson, G.J.; Callahan, J.; et al. Transient limb ischaemia remotely preconditions through a humoral mechanism acting directly on the myocardium: Evidence suggesting cross-species protection. Clin. Sci. 2009, 117, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.Y.; Yellon, D.M.; Hausenloy, D.J. The neural and humoral pathways in remote limb ischemic preconditioning. Basic Res. Cardiol. 2010, 105, 651–655. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.Y.; Baek, H.S.; Park, T.S. Morphologic Changes in Autonomic Nerves in Diabetic Autonomic Neuropathy. Diabetes Metab. J. 2015, 39, 461–467. [Google Scholar] [CrossRef][Green Version]
- Jensen, R.V.; Støttrup, N.B.; Kristiansen, S.B.; Bøtker, H.E. Release of a humoral circulating cardioprotective factor by remote ischemic preconditioning is dependent on preserved neural pathways in diabetic patients. Basic Res. Cardiol. 2012, 107, 285. [Google Scholar] [CrossRef]
- Kottenberg, E.; Thielmann, M.; Kleinbongard, P.; Frey, U.H.; Heine, T.; Jakob, H.; Heusch, G.; Peters, J. Myocardial protection by remote ischaemic pre-conditioning is abolished in sulphonylurea-treated diabetics undergoing coronary revascularisation. Acta Anaesthesiol. Scand. 2014, 58, 453–462. [Google Scholar] [CrossRef]
- Drenger, B.; Ostrovsky, I.A.; Barak, M.; Nechemia-Arbely, Y.; Ziv, E.; Axelrod, J.H. Diabetes blockade of sevoflurane postconditioning is not restored by insulin in the rat heart: Phosphorylated signal transducer and activator of transcription 3-and phosphatidylinositol 3-kinase-mediated inhibition. Anesthesiology 2011, 114, 1364–1372. [Google Scholar] [CrossRef]
- Raphael, J.; Gozal, Y.; Navot, N.; Zuo, Z. Hyperglycemia inhibits anesthetic-induced postconditioning in the rabbit heart via modulation of phosphatidylinositol-3-kinase/Akt and endothelial nitric oxide synthase signaling. J. Cardiovasc. Pharmacol. 2010, 55, 348–357. [Google Scholar] [CrossRef]
- Raphael, J.; Gozal, Y.; Navot, N.; Zuo, Z. Activation of Adenosine Triphosphate-regulated Potassium Channels during Reperfusion Restores Isoflurane Postconditioning-induced Cardiac Protection in Acutely Hyperglycemic Rabbits. Anesthesiology 2015, 122, 1299. [Google Scholar] [CrossRef] [PubMed]
- Davidson, S.M.; Selvaraj, P.; He, D.; Boi-Doku, C.; Yellon, R.L.; Vicencio, J.M.; Yellon, D.M. Remote ischaemic preconditioning involves signalling through the SDF-1α/CXCR4 signalling axis. Basic Res. Cardiol. 2013, 108, 377. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, M.M.; Yellon, D.M. PTEN, the Achilles’ heel of myocardial ischaemia/reperfusion injury? Br. J. Pharmacol. 2007, 150, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.P.; Nicholson, C.K.; Amin, H.; Amin, S.; Calvert, J.W. Hydrogen sulfide provides cardioprotection against myocardial/ischemia reperfusion injury in the diabetic state through the activation of the RISK pathway. Med. Gas Res. 2014, 4, 20. [Google Scholar] [CrossRef]
- Jensen, R.V.; Zachara, N.E.; Nielsen, P.H.; Kimose, H.H.; Kristiansen, S.B.; Bøtker, H.E. Impact of O-GlcNAc on cardioprotection by remote ischaemic preconditioning in non-diabetic and diabetic patients. Cardiovasc. Res. 2013, 97, 369–378. [Google Scholar] [CrossRef]
- Rohailla, S.; Clarizia, N.; Sourour, M.; Sourour, W.; Gelber, N.; Wei, C.; Li, J.; Redington, A.N. Acute, delayed and chronic remote ischemic conditioning is associated with downregulation of mTOR and enhanced autophagy signaling. PLoS ONE 2014, 9, e111291. [Google Scholar] [CrossRef]
- Baranyai, T.; Nagy, C.T.; Koncsos, G.; Onódi, Z.; Károlyi-Szabó, M.; Makkos, A.; Varga, Z.V.; Ferdinandy, P.; Giricz, Z. Acute hyperglycemia abolishes cardioprotection by remote ischemic perconditioning. Cardiovasc. Diabetol. 2015, 14, 151. [Google Scholar] [CrossRef]
- Das, A.; Durrant, D.; Koka, S.; Salloum, F.N.; Xi, L.; Kukreja, R.C. Mammalian target of rapamycin (mTOR) inhibition with rapamycin improves cardiac function in type 2 diabetic mice: Potential role of attenuated oxidative stress and altered contractile protein expression. J. Biol. Chem. 2014, 289, 4145–4160. [Google Scholar] [CrossRef]
- Zhang, M.; Sun, D.; Li, S.; Pan, X.; Zhang, X.; Zhu, D.; Li, C.; Zhang, R.; Gao, E.; Wang, H. Lin28a protects against cardiac ischaemia/reperfusion injury in diabetic mice through the insulin-PI3K-mTOR pathway. J. Cell. Mol. Med. 2015, 19, 1174–1182. [Google Scholar] [CrossRef]
- Cheung, M.M.; Kharbanda, R.K.; Konstantinov, I.E.; Shimizu, M.; Frndova, H.; Li, J.; Holtby, H.M.; Cox, P.N.; Smallhorn, J.F.; Van Arsdell, G.S.; et al. Randomized controlled trial of the effects of remote ischemic preconditioning on children undergoing cardiac surgery: First clinical application in humans. J. Am. Coll. Cardiol. 2006, 47, 2277–2282. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Mwamure, P.K.; Venugopal, V.; Harris, J.; Barnard, M.; Grundy, E.; Ashley, E.; Vichare, S.; Di Salvo, C.; Kolvekar, S.; et al. Effect of remote ischaemic preconditioning on myocardial injury in patients undergoing coronary artery bypass graft surgery: A randomised controlled trial. Lancet 2007, 370, 575–579. [Google Scholar] [CrossRef]
- Karuppasamy, P.; Chaubey, S.; Dew, T.; Musto, R.; Sherwood, R.; Desai, J.; John, L.; Shah, A.M.; Marber, M.S. Remote intermittent ischemia before coronary artery bypass graft surgery: A strategy to reduce injury and inflammation? Basic Res. Cardiol. 2011, 106, 511–519. [Google Scholar] [CrossRef] [PubMed]
- McCrindle, B.W.; Clarizia, N.A.; Khaikin, S.; Holtby, H.M.; Manlhiot, C.; Schwartz, S.M.; Caldarone, C.A.; Coles, J.G.; Van Arsdell, G.S.; Scherer, S.W.; et al. Remote ischemic preconditioning in children undergoing cardiac surgery with cardiopulmonary bypass: A single-center double-blinded randomized trial. J. Am. Heart Assoc. 2014, 3, e000964. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Kharbanda, R.K.; Møller, U.K.; Ramlall, M.; Aarøe, J.; Butler, R.; Bulluck, H.; Clayton, T.; Dana, A.; Dodd, M.; et al. Effect of remote ischaemic conditioning on clinical outcomes in patients with acute myocardial infarction (CONDI-2/ERIC-PPCI): A single-blind randomised controlled trial. Lancet 2019, 394, 1415–1424. [Google Scholar] [CrossRef]
- Przyklenk, K. Efficacy of cardioprotective ‘conditioning’ strategies in aging and diabetic cohorts: The co-morbidity conundrum. Drugs Aging 2011, 28, 331–343. [Google Scholar] [CrossRef]
- Randhaw, P.K.; Bali, A.; Virdi, J.K.; Jaggi, A.S. Conditioning-induced cardioprotection: Aging as a confounding factor. Korean J. Physiol. Pharm. 2018, 22, 467–479. [Google Scholar] [CrossRef]
- Lee, T.M.; Su, S.F.; Chou, T.F.; Lee, Y.T.; Tsai, C.H. Loss of preconditioning by attenuated activation of myocardial ATP-sensitive potassium channels in elderly patients undergoing coronary angioplasty. Circulation 2002, 105, 334–340. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Candilio, L.; Evans, R.; Ariti, C.; Jenkins, D.P.; Kolvekar, S.; Knight, R.; Kunst, G.; Laing, C.; Nicholas, J.; et al. Remote Ischemic Preconditioning and Outcomes of Cardiac Surgery. N. Engl. J. Med. 2015, 373, 1408–1417. [Google Scholar] [CrossRef]
- Iliodromitis, E.K.; Cohen, M.V.; Dagres, N.; Andreadou, I.; Kremastinos, D.T.; Downey, J.M. What is Wrong With Cardiac Conditioning? We May be Shooting at Moving Targets. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 357–369. [Google Scholar] [CrossRef]
- Varga, Z.V.; Giricz, Z.; Bencsik, P.; Madonna, R.; Gyongyosi, M.; Schulz, R.; Mayr, M.; Thum, T.; Puskas, L.G.; Ferdinandy, P. Functional Genomics of Cardioprotection by Ischemic Conditioning and the Influence of Comorbid Conditions: Implications in Target Identification. Curr. Drug Targets 2015, 16, 904–911. [Google Scholar] [CrossRef]
- Moretti, C.; Cerrato, E.; Cavallero, E.; Lin, S.; Rossi, M.L.; Picchim, A.; Sanguineti, F.; Ugo, F.; Palazzuoli, A.; Bertaina, M.; et al. The EUROpean and Chinese cardiac and renal Remote Ischemic Preconditioning Study (EURO-CRIPS CardioGroup I): A randomized controlled trial. Int. J. Cardiol. 2018, 257, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cavallero, E.; Bertaina, M.; Cerrato, E.; D’Ascenzo, F. Reply to: Cardiac protection by remote ischemic preconditioning in patients with diabetes status. Int. J. Cardiol. 2018, 267, 56. [Google Scholar] [CrossRef] [PubMed]
- Rehni, A.K.; Dave, K.R. Ameliorative potential of conditioning on ischemia-reperfusion injury in diabetes. Cond. Med. 2018, 1, 105–115. [Google Scholar] [PubMed]
- Roberto, S.; Crisafulli, A. Consequences of Type 1 and 2 Diabetes Mellitus on the Cardiovascular Regulation During Exercise: A Brief Review. Curr. Diabetes Rev. 2017, 13, 560–565. [Google Scholar] [CrossRef][Green Version]
- Crisafulli, A.; Marongiu, E.; Ogoh, S. Cardiovascular Reflexes Activity and Their Interaction during Exercise. Biomed. Res. Int. 2015, 2015, 394183. [Google Scholar] [CrossRef]
- Nóbrega, A.C.L.; O’Leary, D.S.; Silva, B.M.; Marongiu, E.; Piepoli, M.F.; Crisafulli, A. Neural regulation of cardiovascular response to exercise: Role of central command and peripheral afferents. Biomed. Res. Int. 2014, 2014, 478965. [Google Scholar] [CrossRef]
- Padilla, J.; Olver, T.D.; Thyfault, J.P.; Fadel, P.J. Role of habitual physical activity in modulating vascular actions of insulin. Exp. Physiol. 2015, 100, 759–771. [Google Scholar] [CrossRef]
- Roberto, S.; Marongiu, E.; Pinna, M.; Angius, L.; Olla, S.; Bassareo, P.P.; Tocco, F.; Concu, A.; Milia, R.; Crisafulli, A. Altered hemodynamics during muscle metaboreflex in young, type 1 diabetes patients. J. Appl. Physiol. 2012, 113, 1323–1331. [Google Scholar] [CrossRef]
- Ahlborg, G.; Lundberg, J.M. Exercise-induced changes in neuropeptide Y, noradrenaline and endothelin-1 levels in young people with type I diabetes. Clin. Physiol. 1996, 16, 645–655. [Google Scholar] [CrossRef]
- Galassetti, P.; Mann, S.; Tate, D.; Neill, R.A.; Costa, F.; Wasserman, D.H.; Davis, S.N. Effects of antecedent prolonged exercise on subsequent counter regulatory responses to hypoglycemia. Am. J. Physiol. 2001, 280, E908–E917. [Google Scholar] [CrossRef]
- Koponen, A.S.; Peltonen, J.E.; Päivinen, M.K.; Aho, J.M.; Hägglund, H.J.; Uusitalo, A.L.; Lindholm, H.J.; Tikkanen, H.O. Low total haemoglobin mass, blood volume and aerobic capacity in men with type 1 diabetes. Eur. J. Appl. Physiol. 2013, 113, 1181–1188. [Google Scholar] [CrossRef] [PubMed]
- Rissanen, A.P.; Tikkanen, H.O.; Koponen, A.S.; Aho, J.M.; Peltonen, J.E. Central and peripheral cardiovascular impairments limit VO(2peak) in type 1 diabetes. Med. Sci. Sports Exerc. 2015, 47, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Carnethon, M.R.; Craft, L.L. Autonomic regulation of the association between exercise and diabetes. Exerc. Sport Sci. Rev. 2008, 36, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Manzella, D.; Paolisso, G. Cardiac autonomic activity and Type II diabetes mellitus. Clin. Sci. 2005, 108, 93–99. [Google Scholar] [CrossRef]
- Roberto, S.; Milia, R.; Doneddu, A.; Pinna, V.; Palazzolo, G.; Serra, S.; Orrù, A.; Hosseini Kakhak, S.A.; Ghiani, G.; Mulliri, G.; et al. Hemodynamic Abnormalities During Muscle Metaboreflex Activation in Patients With Type 2 Diabetes Mellitus. J. Appl. Physiol. 2019, 126, 444–453. [Google Scholar] [CrossRef]
- Vianna, L.C.; Deo, S.H.; Jensen, A.K.; Holwerda, S.W.; Zimmerman, M.C.; Fadel, P.J. Impaired dynamic cerebral autoregulation at rest and during isometric exercise in type 2 diabetes patients. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H681–H687. [Google Scholar] [CrossRef][Green Version]
- Milia, R.; Velluzzi, F.; Roberto, S.; Palazzolo, G.; Sanna, I.; Sainas, G.; Pusceddu, M.; Mulliri, G.; Loviselli, A.; Crisafulli, A. Differences in hemodynamic response to metaboreflex activation between obese patients with metabolic syndrome and healthy subjects with obese phenotype. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H779–H789. [Google Scholar] [CrossRef]
- Smith, M.M.; Minson, C.T. Obesity and adipokines: Effects on sympathetic overactivity. J. Physiol. 2012, 590, 1787–1801. [Google Scholar] [CrossRef]
- Eringa, E.C.; Stehouwer, C.D.; Roos, M.H.; Westerhof, N.; Sipkema, P. Selective resistance to vasoactive effects of insulin in muscle resistance arteries of obese Zucker (fa/fa) rats. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1134–E1139. [Google Scholar] [CrossRef]
- Lalande, S.; Hofman, P.L.; Baldi, J.C. Effect of reduced total blood volume on left ventricular volumes and kinetics in type 2 diabetes. Acta Physiol. 2010, 199, 23–30. [Google Scholar] [CrossRef]
- Poitras, V.J.; Hudson, R.W.; Tschakovsky, M.E. Exercise Intolerance in Type 2 Diabetes: Is There a Cardiovascular Contribution? J. Appl. Physiol. 2018, 124, 1117–1139. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.; Vaccaro, O.; Neaton, J.D.; Wentworth, D. Diabetes, Other Risk Factors, and 12-Yr Cardiovascular Mortality for Men Screened in the Multiple Risk Factor Intervention Trial. Diabetes Care 1993, 16, 434–444. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 4. Lifestyle management: Standards of medical care in diabetes-2018. Diabetes Care 2018, 41, S38–S50. [Google Scholar] [CrossRef] [PubMed]
- Hansen, D.; Coninx, K.; Dendale, P. The EAPC EXPERT tool. Eur. Heart J. 2017, 38, 2318–2320. [Google Scholar] [CrossRef]
- IDF Epidemiology Task Force Consensus Group. The metabolic syndrome- a new world-wide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in atherosclerosis. Nature 2002, 420, 868–874. [Google Scholar] [CrossRef]
- Kraniou, G.N.; Cameron-Smith, D.; Hargreaves, M. Acute exercise and GLUT4 expression in human skeletal muscle: Influence of exercise intensity. J. Appl. Physiol. 2006, 101, 934–937. [Google Scholar] [CrossRef]
- Zierath, J.R.; He, L.; Gumà, A.; Odegoard Wahlström, E.; Klip, A.; Wallberg, -H.H. Insulin action on glucose transport and plasma membrane GLUT4 content in skeletal muscle from patients with NIDDM. Diabetologia 1996, 39, 1180–1189. [Google Scholar] [CrossRef]
- Hawley, J.A.; Lessard, S.J. Exercise training-induced improvements in insulin action. Acta Physiol. 2008, 192, 127–135. [Google Scholar] [CrossRef]
- Holloszy, J.O. Exercise-induced increase in muscle insulin sensitivity. J. Appl. Physiol. 2005, 99, 338–343. [Google Scholar] [CrossRef]
- Mul, J.D.; Stanford, K.I.; Hirshman, M.F.; Goodyear, L.J. Exercise and Regulation of Carbohydrate Metabolism. Prog. Mol. Biol. Transl. Sci. 2015, 135, 17–37. [Google Scholar] [CrossRef] [PubMed]
- Quirk, H.; Blake, H.; Tennyson, R.; Randell, T.L.; Glazebrook, C. Physical activity interventions in children and young people with Type 1 diabetes mellitus: A systematic review with meta-analysis. Diabet. Med. 2014, 31, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.; Freeman, J.A.; Janssen, I. Exercise alone is an effective strategy for reducing obesity and related comorbidities. Exerc. Sport Sci. Rev. 2000, 28, 165–170. [Google Scholar] [PubMed]
- Lakka, T.A.; Laaksonen, D.E. Physical activity in prevention and treatment of the metabolic syndrome. Appl. Physiol. Nutr. Metab. 2007, 32, 76–88. [Google Scholar] [CrossRef]
- Oliveira, C.; Simões, M.; Carvalho, J.; Ribeiro, J. Combined exercise for people with type 2 diabetes mellitus: A systematic review. Diabetes Res. Clin. Pract. 2012, 98, 187–198. [Google Scholar] [CrossRef]
- Hussey, S.E.; McGee, S.L.; Garnham, A.; Wentworth, J.M.; Jeukendrup, A.E.; Hargreaves, M. Exercise training increases adipose tissue GLUT4 expression in patients with type 2 diabetes. Diabetes Obes. Metab. 2011, 13, 959–962. [Google Scholar] [CrossRef]
- Kirwan, J.P.; Sacks, J.; Nieuwoudt, S. The essential role of exercise in the management of type 2 diabetes. Cleve Clin. J. Med. 2017, 84, S15–S21. [Google Scholar] [CrossRef]
- Colberg, S.R.; Sigal, R.J.; Yardley, J.E.; Riddell, M.C.; Dunstan, D.W.; Dempsey, P.C.; Horton, E.S.; Castorino, K.; Tate, D.F. Physical Activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016, 39, 2065–2079. [Google Scholar] [CrossRef]
- Dunstan, D.W.; Daly, R.M.; Owen, N.; Jolley, D.; De Courten, M.; Shaw, J.; Zimmet, P. High-Intensity Resistance Training Improves Glycemic Control in Older Patients With Type 2 Diabetes. Diabetes Care 2002, 25, 1729–1736. [Google Scholar] [CrossRef]
- Balducci, S.; Leonetti, F.; Di Mario, U.; Fallucca, F. Is a long-term aerobic plus resistance training program feasible for and effective on metabolic profiles in type 2 diabetic patients? Diabetes Care 2004, 27, 841–842. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Missbach, B.; Dias, S.; König, J.; Hoffmann, G. Impact of different training modalities on glycaemic control and blood lipids in patients with type 2 diabetes: A systematic review and network meta-analysis. Diabetologia 2014, 57, 1789–1797. [Google Scholar] [CrossRef] [PubMed]
- Jelleyman, C.; Yates, T.; O’Donovan, G.; Gray, L.Y.; King, J.A.; Khunti, K.; Davies, M.J. The effects of high-intensity interval training on glucose regulation and insulin resistance: A meta-analysis. Obes. Rev. 2015, 16, 942–961. [Google Scholar] [CrossRef] [PubMed]
- Mayor, S. Insulin pumps improve glucose control in children with type 1 diabetes, study finds. BMJ 2015, 351, h5998. [Google Scholar] [CrossRef] [PubMed]
- McKnight, J.A.; Wild, S.H.; Lamb, M.J.; Cooper, M.N.; Jones, T.W.; Davis, E.A.; Hofer, S.; Fritsch, M.; Schober, E.; Svensson, J.; et al. Glycaemic control of Type 1 diabetes in clinical practice early in the 21st century: An international comparison. Diabet Med. 2015, 32, 1036–1050. [Google Scholar] [CrossRef]
- Ogata, H.; Nakamura, K.; Sato, M.; Tokuyama, K.; Nagasaka, S.; Ebine, N.; Kiyono, K.; Yamamoto, Y. Lack of negative correlation in glucose dynamics by nonexercise activity thermogenesis restriction in healthy adults. Med. Sci. Sports Exerc. 2013, 45, 60–66. [Google Scholar] [CrossRef]
- Camacho, R.C.; Galassetti, P.; Davis, S.N.; Wasserman, D.H. Glucoregulation during and after exercise in health and insulin-dependent diabetes. Exerc. Sport Sci. Rev. 2005, 33, 17–23. [Google Scholar]
- Coker, R.H.; Kjaer, M. Glucoregulation during exercise: The role of the neuroendocrine system. Sports Med. 2005, 35, 575–583. [Google Scholar] [CrossRef]
- Al Khalifah, R.A.; Suppère, C.; Haidar, A.; Rabasa-Lhoret, R.; Ladouceur, M.; Legault, L. Association of aerobic fitness level with exercise-induced hypoglycaemia in Type 1 diabetes. Diabet Med. 2016, 33, 1686–1690. [Google Scholar] [CrossRef]
- Harmer, A.R.; Chisholm, D.J.; McKenna, M.J.; Hunter, S.K.; Ruell, P.A.; Naylor, J.M.; Maxwell, L.J.; Flack, J.R. Sprint training increases muscle oxidative metabolism during high-intensity exercise in patients with type 1 diabetes. Diabetes Care 2008, 31, 2097–2102. [Google Scholar] [CrossRef]
- Štotl, I.; Kambič, T.; Hadžić, V.; Zdolšek, A. Different Types of Physical Activity and Metabolic Control in People With Type 1 Diabetes Mellitus. Front. Physiol. 2019, 10, 1210. [Google Scholar] [CrossRef]
- Cugusi, L.; Manca, A.; Bassareo, P.P.; Crisafulli, A.; Deriu, F.; Mercuro, G. Supervised aquatic-based exercise for men with coronary artery disease: A meta-analysis of randomised controlled trials. Eur. J. Prev. Cardiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cugusi, L.; Manca, A.; Bergamin, M.; Di Blasio, A.; Yeo, T.J.; Crisafulli, A.; Mercuro, G. Working Group of Gender Cardiovascular Disease of the Italian Society of Cardiology. Zumba Fitness and Women’s Cardiovascular Health: A systematic review. J. Cardiopulm. Rehabil. Prev. 2019, 39, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Cugusi, L.; Manca, A.; Yeo, T.J.; Bassareo, P.P.; Mercuro, G.; Kaski, J.C. Nordic walking for individuals with cardiovascular disease: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Prev. Cardiol. 2017, 24, 1938–1955. [Google Scholar] [CrossRef] [PubMed]
- Nuhu, J.M.; Maharaj, S.S. Influence of a mini-trampoline rebound exercise program on insulin resistance, lipid profile and central obesity in individuals with type 2 diabetes. J. Sports Med. Phys. Fitness. 2018, 58, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Rees, J.L.; Johnson, S.T.; Boulé, N.G. Aquatic exercise for adults with type 2 diabetes: A meta-analysis. Acta Diabetol. 2017, 54, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Thind, H.; Lantini, R.; Balletto, B.L.; Donahue, M.L.; Salmoirago-Blotcher, E.; Bock, B.C.; Scott-Sheldon, L.A.J. The effects of yoga among adults with type 2 diabetes: A systematic review and meta-analysis. Prev. Med. 2017, 105, 116–126. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhou, R.; Li, K.; Zhu, Y.; Zhang, Z.; Luo, Y.; Luan, R. Effects of tai chi on physiology, balance and quality of life in patients with type 2 diabetes: A systematic review and meta-analysis. J. Rehabil. Med. 2019, 51, 405–417. [Google Scholar] [CrossRef]
- Da Silva, M.F.; Pelúzio Mdo, C.; Amorim, P.R.; Lavorato, V.N.; Santos, N.P.; Bozi, L.H.; Penitente, A.R.; Falkoski, D.L.; Berfort, F.G.; Natali, A.J. Swimming training attenuates contractile dysfunction in diabetic rat cardiomyocytes. Arq. Bras. Cardiol. 2011, 97, 33–39. [Google Scholar] [CrossRef]
- Da Silva, E.; Natali, A.J.; Silva, M.F.; Gomes, G.J.; Cunha, D.N.; Ramos, R.M.; Toledo, M.M.; Drummond, F.R.; Belfort, F.G.; Novaes, R.D.; et al. Ventricular remodeling in growing rats with experimental diabetes: The impact of swimming training. Pathol. Res. Pract. 2013, 209, 618–626. [Google Scholar] [CrossRef]
- Da Silva, E.; Natali, A.J.; da Silva, M.F.; Gomes Gde, J.; da Cunha, D.N.; Toledo, M.M.; Drummond, F.R.; Ramos, R.M.; Dos Santos, E.C.; Novaes, R.D.; et al. Swimming training attenuates the morphological reorganization of the myocardium and local inflammation in the left ventricle of growing rats with untreated experimental diabetes. Pathol. Res. Pract. 2016, 212, 325–334. [Google Scholar] [CrossRef]
- Suntraluck, S.; Tanaka, H.; Suksom, D. The Relative Efficacy of Land-Based and Water-Based Exercise Training on Macro- and Microvascular Functions in Older Patients with Type 2 Diabetes. J. Aging Phys. Act. 2017, 25, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Cugusi, L.; Cadeddu, C.; Nocco, S.; Orrù, F.; Bandino, S.; Deidda, M.; Caria, A.; Bassareo, P.P.; Piras, A.; Cabras, S.; et al. Effects of an aquatic-based exercise program to improve cardiometabolic profile, quality of life, and physical activity levels in men with type 2 diabetes mellitus. PM&R 2015, 7, 141–148. [Google Scholar] [CrossRef]
- Ring, M.; Eriksson, M.J.; Fritz, T.; Nyberg, G.; Östenson, C.G.; Krook, A.; Zierath, J.R.; Caidahl, K. Influence of physical activity and gender on arterial function in type 2 diabetes, normal and impaired glucose tolerance. Diab. Vasc. Dis. Res. 2015, 12, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Fiodorenko-Dumas, Z.; Dumas, I.; Mastej, K.; Adamiec, R. Physical activity related changes in ADMA and vWF levels in patients with type 2 diabetes: A preliminary study. Adv. Clin. Exp. Med. 2017, 26, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Tokar, T.N.; Boylan, M.M.; Griffin, K.; Feng, D.; Mcmurry, L.; Esperat, C.; Cooper, J.A. Zumba® dance improves health in overweight/obese or type 2 diabetic women. Am. J. Health Behav. 2015, 39, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Borges, L.; Passos, M.E.P.; Silva, M.B.B.; Santos, V.C.; Momesso, C.M.; Pithon-Curi, T.C.; Gorjão, R.; Gray, S.R.; Lima, K.C.A.; de Freitas, P.B.; et al. Dance Training Improves Cytokine Secretion and Viability of Neutrophils in Diabetic Patients. Mediat. Inflamm. 2019, 2924818. [Google Scholar] [CrossRef]
- Melo, K.C.B.; Araújo, F.S.; Cordeiro Júnior, C.C.M.; de Andrade, K.T.P.; Moreira, S.R. Pilates Method Training: Functional and Blood Glucose Responses of Older Women With Type 2 Diabetes. J. Strength Cond. Res. 2018. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crisafulli, A.; Pagliaro, P.; Roberto, S.; Cugusi, L.; Mercuro, G.; Lazou, A.; Beauloye, C.; Bertrand, L.; Hausenloy, D.J.; Aragno, M.; et al. Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning. Int. J. Mol. Sci. 2020, 21, 2896. https://doi.org/10.3390/ijms21082896
Crisafulli A, Pagliaro P, Roberto S, Cugusi L, Mercuro G, Lazou A, Beauloye C, Bertrand L, Hausenloy DJ, Aragno M, et al. Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning. International Journal of Molecular Sciences. 2020; 21(8):2896. https://doi.org/10.3390/ijms21082896
Chicago/Turabian StyleCrisafulli, Antonio, Pasquale Pagliaro, Silvana Roberto, Lucia Cugusi, Giuseppe Mercuro, Antigone Lazou, Christophe Beauloye, Luc Bertrand, Derek J. Hausenloy, Manuela Aragno, and et al. 2020. "Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning" International Journal of Molecular Sciences 21, no. 8: 2896. https://doi.org/10.3390/ijms21082896
APA StyleCrisafulli, A., Pagliaro, P., Roberto, S., Cugusi, L., Mercuro, G., Lazou, A., Beauloye, C., Bertrand, L., Hausenloy, D. J., Aragno, M., & Penna, C. (2020). Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning. International Journal of Molecular Sciences, 21(8), 2896. https://doi.org/10.3390/ijms21082896






