Abstract
Heterosis has been widely accepted as an effective strategy to increase yields in plant breeding. Notably, the chemical hybridization agent SQ-1 induces male sterility in wheat, representing a critical potential tool in hybrid seed production. However, the mechanisms underlying the male sterility induced by SQ-1 still remain poorly understood. In this study, a cyclin-dependent kinase inhibitor gene, TaICK1, which encodes a 229 amino acid protein, was identified as a potential contributor to male sterility in common wheat. The expression of TaICK1 was upregulated during the development of anthers in Xinong1376 wheat treated with SQ-1. Meanwhile, the seed setting rate was found to be significantly decreased in TaICK1 transgenic rice. Furthermore, we identified two cyclin proteins, TaCYCD2;1 and TaCYCD6;1, as interactors through yeast two-hybrid screening using TaICK1 as the bait, which were validated using bimolecular fluorescence complementation. Subcellular localization revealed that the proteins encoded by TaICK1, TaCYCD2;1, and TaCYCD6;1 were localized in the cell nucleus. The expression levels of TaCYCD2;1 and TaCYCD6;1 were lower in Xinong1376 treated with SQ-1. A further analysis demonstrated that the expression levels of OsCYCD2;1 and OsCYCD6;1 were lower in transgenic TaICK1 rice lines as well. Taken together, these results suggest that the upregulation of TaICK1, induced by SQ-1, may subsequently suppress the expression of TaCYCD2;1 and TaCYCD6;1 in anthers, resulting in male sterility. This study provides new insights into the understanding of SQ-1-induced wheat male sterility, as well as the developmental mechanisms of anthers.
1. Introduction
Bread wheat (Triticum aestivum L.) is one of the most stable cereal crops and feeds nearly 40% of the world population, contributing to approximately 20% of the global total caloric intake [1,2,3]. In China, wheat is the third most important food crop after rice and maize, in terms of either total production or sown area. In 2017, the area of sown wheat was more than 24.5 million hectares, with a total output of 1.34 hundred million tons (http://www.stats.gov.cn/tjsj/ndsj/2018/indexch.htm, China Statistical Yearbook 2018). However, with the continued increase in the global population, reductions in cultivatable land (19% less in the wheat cultivated area in 2016 compared to 1998), frequent abnormal climate changes, and other factors, a gap still remains between the total output of wheat and its demand [4,5]. Therefore, there is still an urgent need to improve wheat yields [3,6].
Heterosis is one of the most effective ways to improve the yield and quality of wheat [7,8]. At present, two main wheat heterosis strategies are available: a genetic male sterility system (e.g., CMS, cytoplasmic male sterility; or PTMS, photo-thermo-sensitive male sterility) and a physiological male sterility (PHYMS) system (e.g., CIMS, chemically induced male sterility) [9]. For the genetic male sterile system, the creation of excellent male-sterile lines, maintainer lines, and restorer lines remains a roadblock for the production of hybrid seed [10]. By contrast, hybrid seed production using the CIMS system does not have these limitations [11].
Previous studies have indicated that the chemical hybridization agent (CHA) SQ-1 is an effective male-killing agent for wheat. It is worth noting that there are no evident interaction effects between most genotype varieties and SQ-1, nor negative effects on agronomic traits [12,13,14]. These results demonstrate that SQ-1 has tremendous application potential in wheat hybrid seed production. However, the degree of male sterility induced by SQ-1 in some fine varieties is unsatisfactory for hybrid seed production [15,16]. As a result, some fine varieties cannot be used as parents in hybrid seed production. Elucidation of the molecular mechanism underlying SQ-1-induced male sterility has the potential to improve the prospects for utilizing CHA. Nevertheless, the molecular mechanism underlying SQ-1-induced wheat male sterility remains relatively unknown, representing an impediment to the application of SQ-1-induced wheat male sterility. Previous research has indicated that pollen abortion may be associated with impaired energy metabolism after spraying of SQ-1 [17,18,19,20,21,22,23]. For example, SQ-1-induced sterility lines showed lower expression levels of the PDH-E1α gene, which is essential for regulation of the tricarboxylic acid (TAC) cycle [24]. This is consistent with a study showing that the inhibition of PDH-E1α expression in tobacco leads to male sterility [25]. In addition, pyruvate dehydrogenase kinase (PDK) is a core regulator of the cell cycle and can block the function of protein PDH-E1α through phosphorylation [26,27]. Meanwhile, TaPCNA is also an important regulator of PDK in wheat [28].
In our previous study, the protein TaICK1 was identified in a yeast two-hybrid assay using TaPCNA as the bait [29]. In plants, ICKs [30] are inhibitors of CDKs (cyclin-dependent kinases), which play a central role in regulating the cell cycle in plants [31]. To date, seven ICK proteins have been identified in the model plant Arabidopsis thaliana: ICK1/KRP1, ICK2/KRP2, KRP3, KRP4, KRP5, KRP6, and KRP7 [32]. Simultaneously, in other plants, increasingly more ICKs have also been identified, such as NtKIS1a and NtKIS1b in tobacco [33], Zeama;KRP;1 and Zeama;KRP;2 in maize [34], LeKRP1 and LeKRP2 in tomato [35], and Orysa;KRP1 in rice [36].
It was reported that ICKs play an important role in the regulation of plant growth and development. ICKs can bind CDK–Cycd protein complexes directly to regulate the activity of CDKs [37] and, thus, regulate cell development [38]. Hence, a high level of ICKs can inhibit the CDK activity. For example, the overexpression of NtKIS1a, an ICK-like inhibitor isolated from tobacco, inhibits nuclear re-replication and reduces CDK activity in Arabidopsis thaliana, which demonstrates that plant organ size and shape as well as organ cell number and cell size might be controlled by the modulation of the activity of a single NtKIS1a gene [39]. Wang et al. (2000) reported that the growth of transgenic 35S::ICK1 Arabidopsis plants is significantly inhibited, and most of their organs are smaller [40]. Similar results have been reported in other studies [39,40,41,42,43]. On the other hand, downregulation of both ICK4/KRP6 and ICK5/KRP7 can affect pollen development [32,44]. Furthermore, some members of the ICK protein family can interact with D-type cyclins (Cycds), which involves a number of CDK–Cycd protein complexes and plays a crucial role in the cell cycle [45]. For example, excess ICK2 can interact with CYCD2;1, thereby inhibiting lateral root formation [46]. Additionally, increased expression of CYCD can suppress the negative effects of excess ICK/KRP levels on plant growth [39,47]. These results suggest that ICKs may play an important role in the development of anther. Although there are some studies on the functions of ICK1 in anther development, relatively little is known about this process in common wheat. As wheat is one of the most important plants for humans, basic knowledge regarding the molecular biology of anther development will promote a better understanding of male sterility and, moreover, promote the utilization of heterosis.
In this study, we confirmed the full-length coding sequence of TaICK1 in Xinong1376 (XN1376) wheat. The results show that TaICK1 mRNA could be detected in roots, stems, leaves, developing seeds, and anthers. Remarkably, the transcript was most abundant in anthers, especially at the late uninucleate stage of anthers. Similar expression patterns of TaICK1 were detected in the male sterile lines Xinong1376-CIMS (XN1376-CIMS), but the detected expression level was higher than that in XN1376 for all stages of anther development except at stage Bn. Meanwhile, the overexpression of TaICK1 in rice may block the expression of cyclin (e.g., OsCYCD6;1 and OsCYCD2;1). In addition, the seed setting rate significantly decreased in overexpressed TaICK1 transgenic rice. This study provides a theoretical basis for further exploring the regulatory pathways related to energy metabolism in the process of SQ-1-induced male sterility in wheat and lays a foundation for eventually revealing the mechanism underlying wheat physiological male sterility induced by SQ-1.
2. Results
2.1. Morphological and Cytological Characteristics
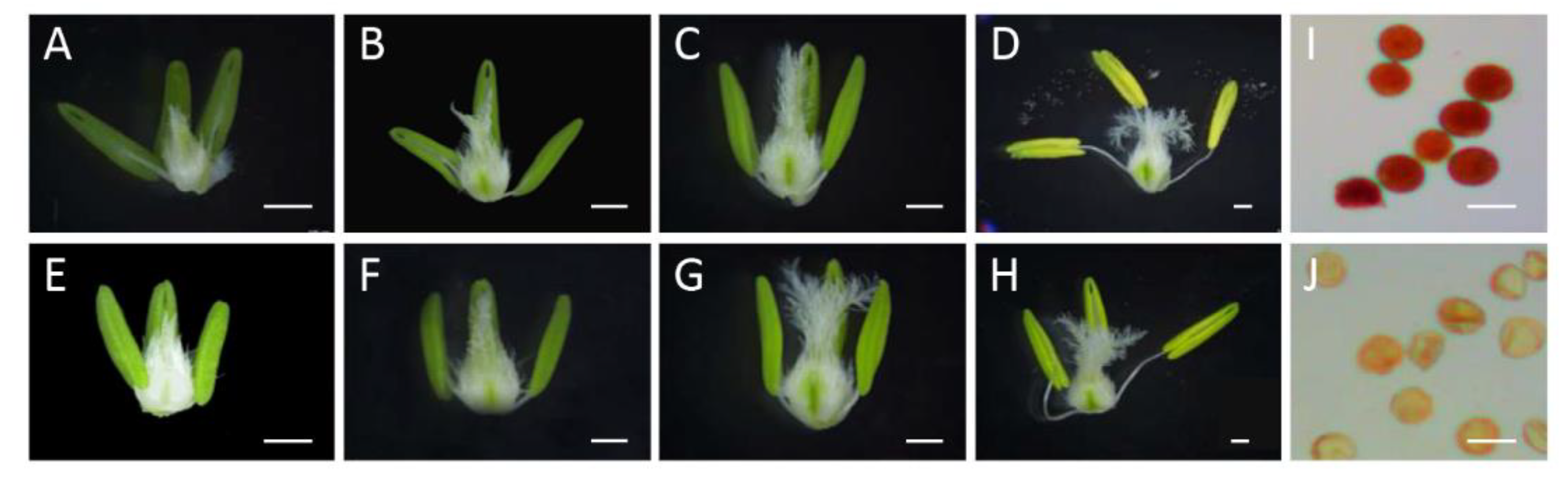
In this study, the development of wheat anthers was divided into four stages (e.g., Eun, the early uninucleate stage; Lun, the late uninucleate stage; Bn, the binucleate stage; and Tn, the trinucleate stage). As shown in Figure 1, the pistil tissue showed the normal development of all four stages of XN1376 and XN1376-CIMS. By contrast, the anthers from XN1376-CIMS showed a significant difference compared to XN1376 from the Eun stage to the Tn stage. For example, the anthers from XN1376-CIMS plants were smaller and lighter-colored than those from XN1376 plants (Figure 1A–H). More importantly, the anthers of XN1376-CIMS were shriveled and indehiscent, and less pollen could be deeply stained using the 1% KI–I2 solution compared to the anthers of XN1376 (Figure 1I,J). These results indicate that CHA SQ-1 had a negative effect on anther development and destroyed the activity of pollen grains.
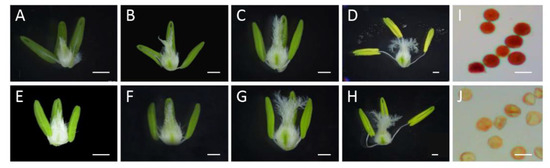
Figure 1.
Anther phenotype and KI–I2 staining in XN1376 (A–D,I) and XN1376-CIMS (E–H,J). (A,E) Eun, early uninucleate stage; (B,F) Lun, late uninucleate stage; (C,G) Bn, binucleate stage; (D,H) Tn, trinucleate stage; and (I,J) KI–I2 staining. Scale bars are 1 mm in (A–H) and 50 µm in (I,J).
The surface characteristics of the anthers and pollen grains at the Tn stage in XN1376-CIMS and XN1376 were analyzed using scanning electron microscopy (SEM) (Figure 2A,B,E,F,I,J). The anther surface characteristics were more irregular in shape in XN1376-CIMS compared to those in XN1376 (Figure 2E,F). In addition, the pollen grains exhibited a severely malformed, shrunken extine pattern, with small and shrunken germination apertures in XN1376-CIMS (Figure 2J). By contrast, the pollen grains from XN1376 looked round and did not shrink (Figure 2I).

Figure 2.
SEM observations of the mature pollen and phenotypes of transgenic rice plants. (A) Anthers of XN1376. (B) Anthers of XN1376-CIMS. (C) Anthers of wild-type Nipponbare. (D) Anthers of transgenic TaICK1 rice plants. (E) The outer epidermal cells of XN1376. (F) The outer epidermal cells of XN1376-CIMS. (G) The outer epidermal cells of wild-type Nipponbare. (H) The outer epidermal cells of transgenic TaICK1 rice plants. (I) The microspores of XN1376. (J) The microspores of XN1376-CIMS. (K) The microspores of wild-type Nipponbare. (L) The microspores of transgenic TaICK1 rice plants. (M) The phenotype of 35S::TaICK1 overexpression plants in young seedlings compared to the wild type. The left is the wild type, and the right is the transgenic plant. (N) The phenotype of TaICK1 overexpression plants in the heading period compared to the wild type. The left is the wild type and the right is the transgenic plant. Scale bars are 500 μm in A–D; 10 μm in E–H; and 50 μm in I–L.
2.2. Structure of the Gene TaICK1
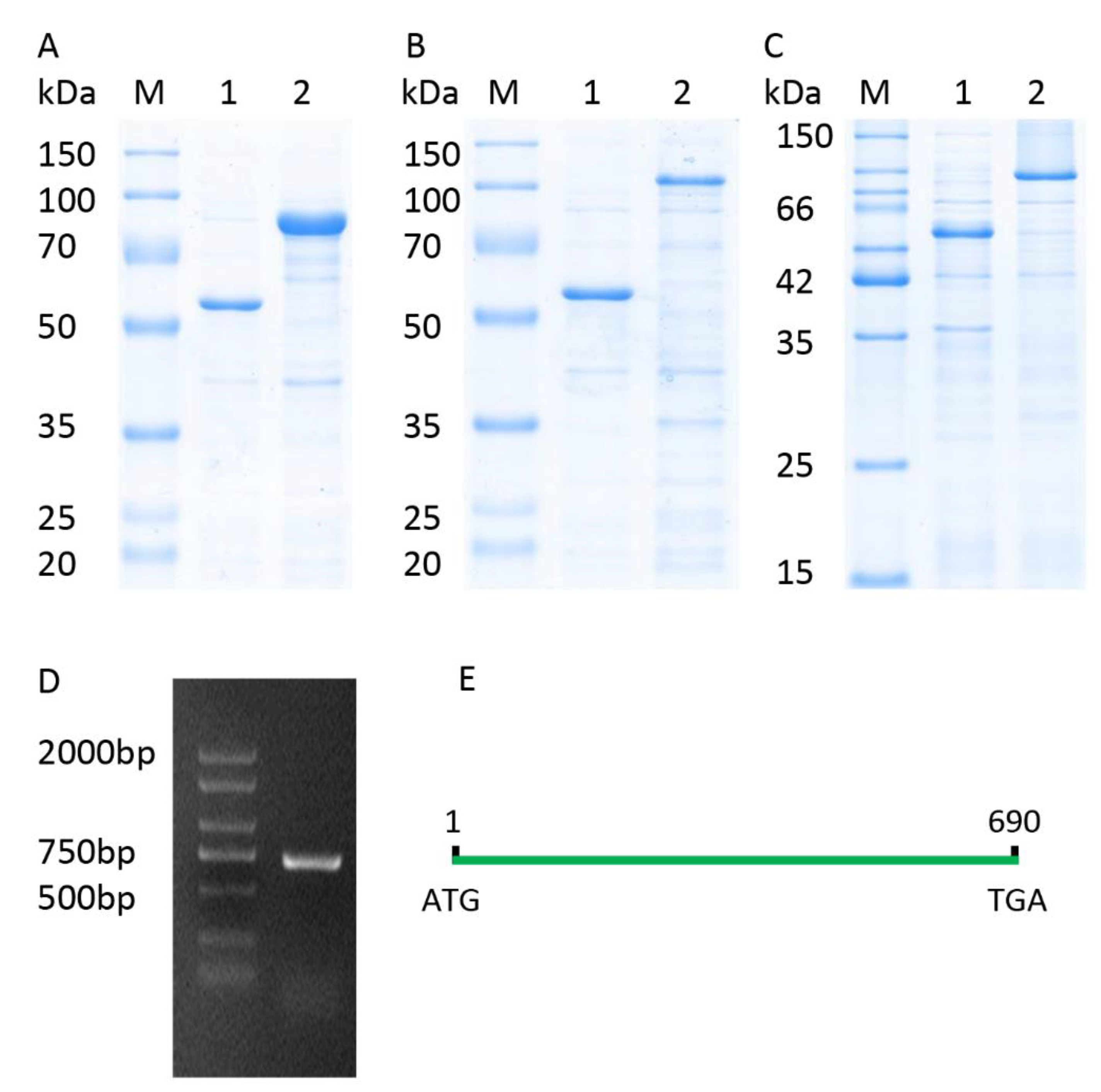
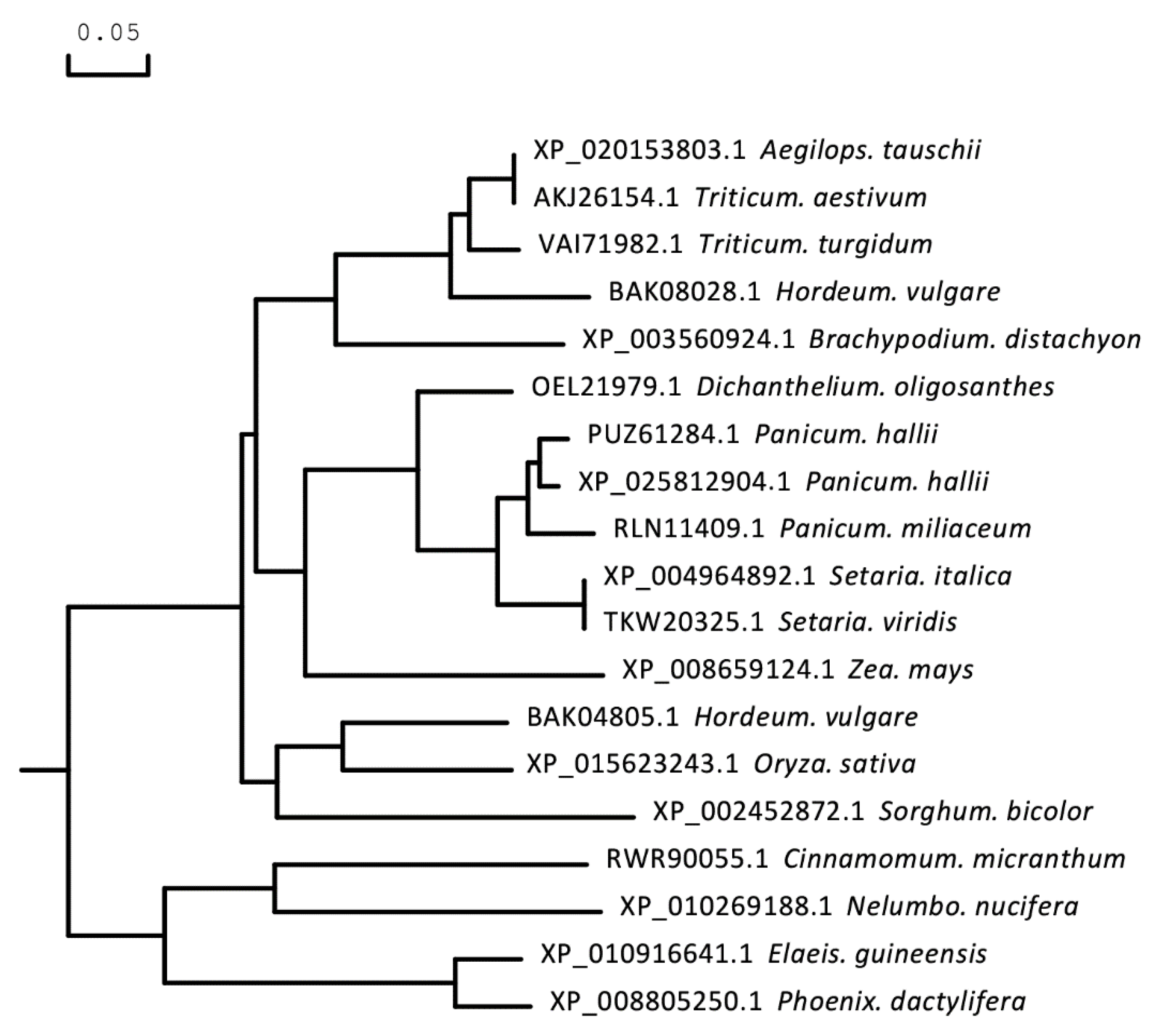
The full-length CDS of TaICK1 was identified from XN1376 using PCR. The results of agarose electrophoresis show the presence of only one DNA band corresponding to a specific product (Figure 3D). Sequencing revealed that the gene TaICK1 contained a 690 bp (Figure 3E) open reading frame that encoded 229 amino acid residues. The homology analysis indicated that the TaICK1 amino acid residue sequence has a high similarity with that of Aegilops tauschii (GenBank XP_020153803.1) and Hordeum (GenBank BAK08028.1)—99.56% and 83.26%, respectively (Figure 4).
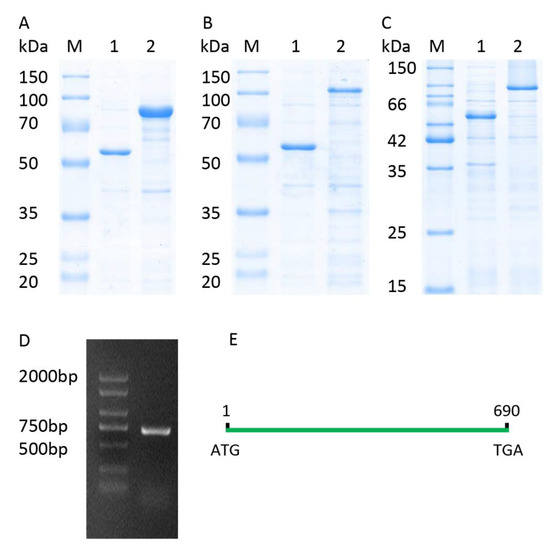
Figure 3.
Recombinant expression analysis and full-length CDS of TaICK1. (A) Lane M: protein marker; lane 1: pCold-TF; lane 2: TaICK1 recombinant protein. (B) Lane M: protein marker; lane 1: pCold-TF; lane 2: TaCYCD2;1 recombinant protein. (C) Lane M: protein marker; lane 1: pCold-TF; lane 2: TaCYCD6;1 recombinant protein. (D) The full-length CDS of TaICK1 was cloned by the reverse transcription polymerase chain reaction of the RNA from wheat XN1376 anthers. M: marker; 1: PCR product of TaICK1. (E) TaICK1 gene structure.
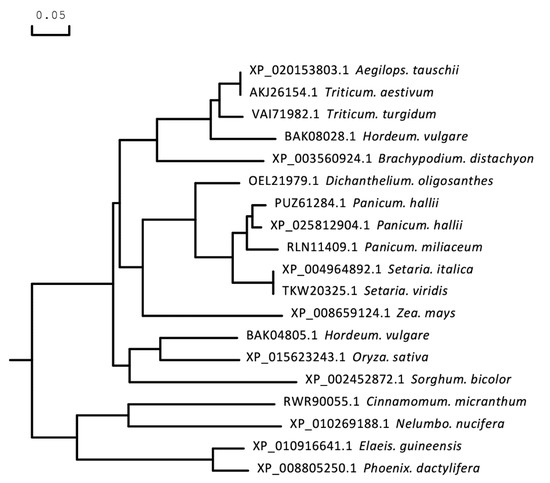
Figure 4.
Phylogenetic tree based on the amino acid sequence of the protein TaICK1. A molecular phylogenetic analysis of the protein sequences of TaICK1 using the maximum likelihood method with bootstrap confidence values exceeding 80% from 1000 replicates. The number before the Latin name is the accession number from NCBI.
2.3. Expression Level of TaICK1 in XN1376-CIMS and XN1376
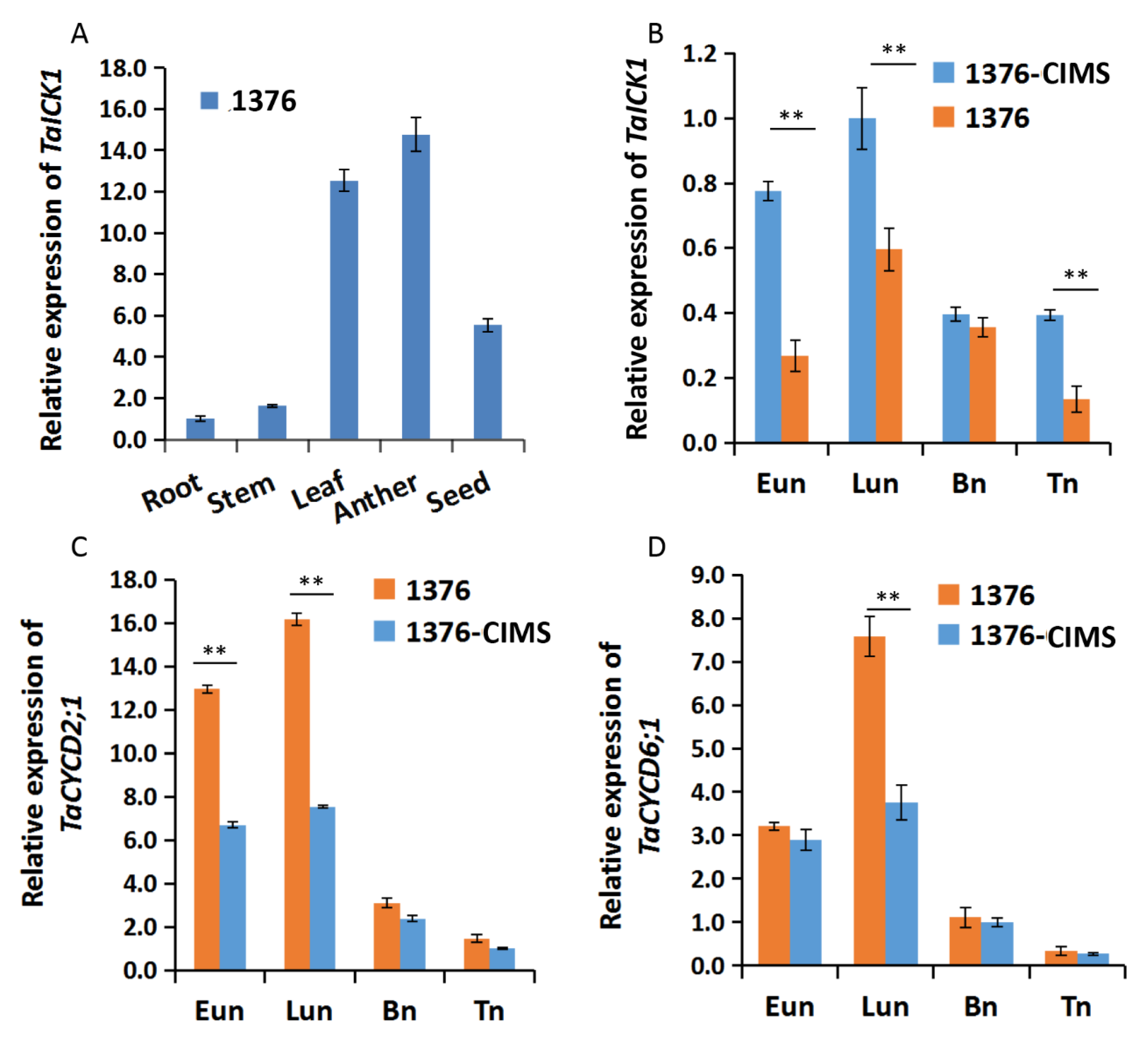
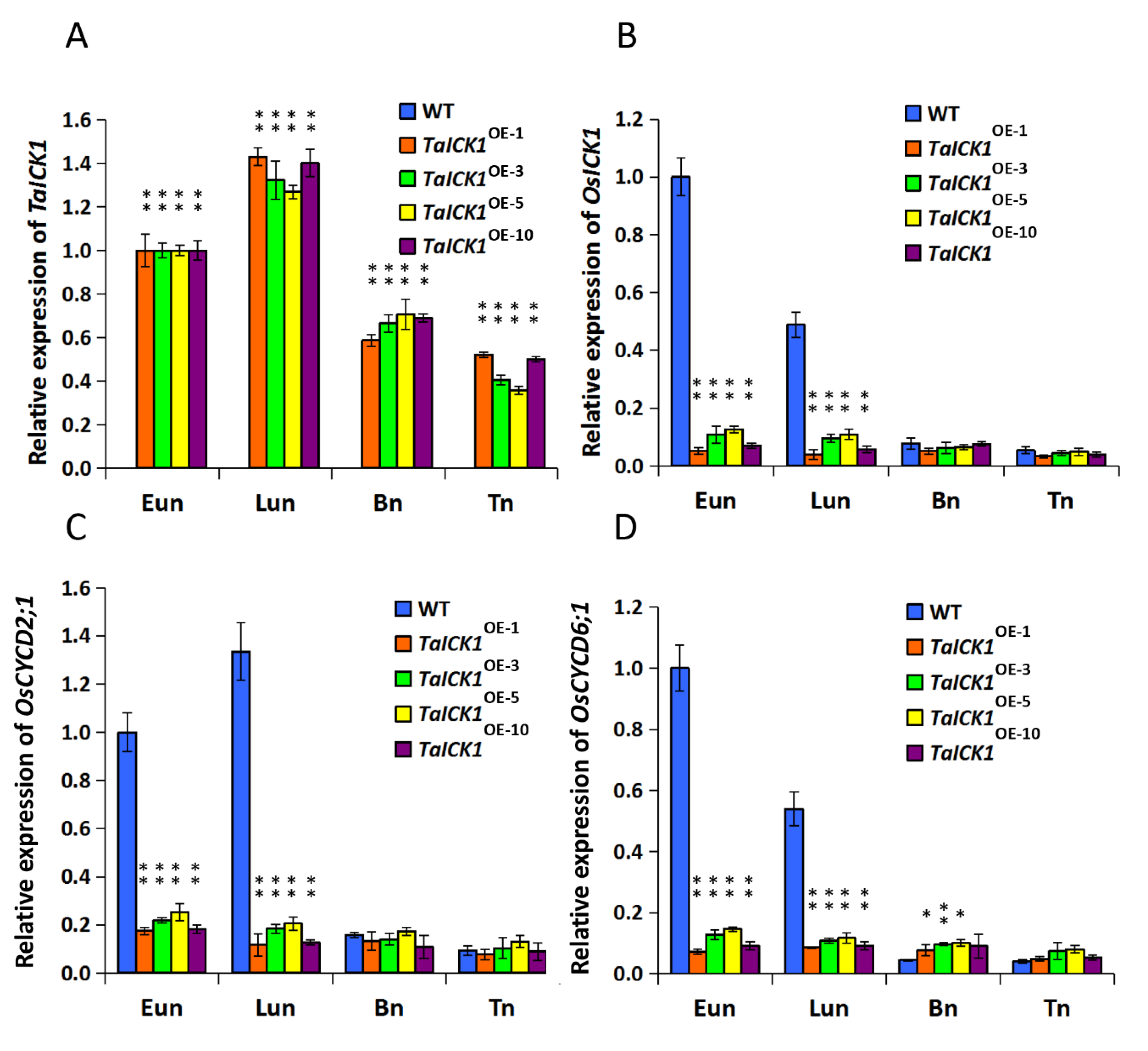
The expression patterns of TaICK1 were first studied in various tissues of XN1376. Meanwhile, the expression patterns were further measured during the stages of Eun, Lun, Bn, and Tn in XN1376-CIMS and XN1376. The expression of TaICK1 significantly differed depending on the tissue. As shown in Figure 5A, the most abundant cDNA of TaICK1 was detected in anthers, followed by leaves and developing seeds. The root and stem presented less TaICK1 cDNA compared to other tissues. Especially in root, the expression level was lower than 14 times that in anthers (Figure 5A). These results indicate that the expression pattern in wheat is organ-specific. In addition, the relative expression of TaICK1 was further analyzed in the anthers of XN1376-CIMS and XN1376 and showed an increase from the Eun to Lun stage followed by a continuous decrease from the Lun to Tn stage in XN1376-CIMS. XN1376 also exhibited a similar expression pattern: the highest expression level was detected at the Lun stage (Figure 5B). Notably, the expression of TaICK1 was higher in XN1376-CIMS than that in XN1376 at stages Eun, Lun, and Tn (Figure 5B).
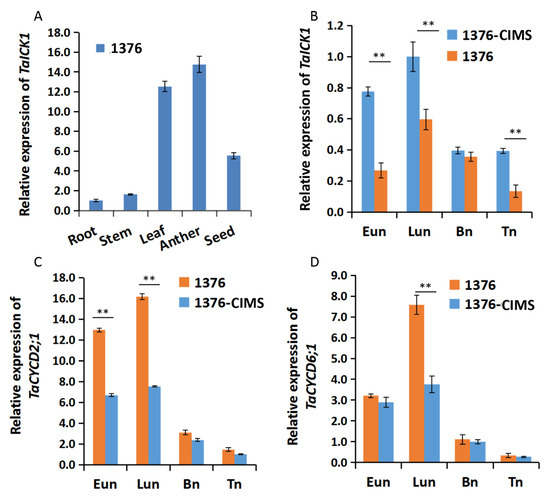
Figure 5.
The expression patterns of TaICK1. (A) The expression pattern in various tissues of XN1376. (B) The expression levels of TaICK1 in XN1376-CIMS and XN1376 at different stages of anther development. (C) The expression levels of TaCYCD2;1 in XN1376-CIMS and XN1376 at different stages of anther development. (D) The expression levels of TaCYCD6;1 in XN1376-CIMS and XN1376 at different stages of anther development. Data are presented as the means ± SD; mean and error bars indicate SD with three technical replicates and three independent biological replicates. The significance of differences was assessed using Student’s t-test. * p < 0.05, ** p < 0.01.
2.4. The Phenotype of TaICK1 Transgenic Rice Plants
The full-length CDS sequence of TaICK1 was cloned into the expression vector pCAMBIA1301, which drives the expression of inserts through the 35S promoter. The vector was then introduced into the rice variety Nipponbare. In total, 15 independent TaICK1 transgenic T0 plants were identified. Homozygous transgenic plants were obtained from the T2 lines. In young seedlings, there were no observable differences between the wild-type Nipponbare and transgenic plants (Figure 2M). By contrast, at later stages, one striking change was observed: the heading period was delayed by 7–10 days in transgenic plants compared to control plants (Figure 2N). In addition, the seed setting rates of transgenic plants and wild-type plants were investigated. The average seed setting rate was 46.5% in the T1 generation, which was significantly lower than that in the wild type (Table 1). To confirm the results, the homozygote T2 generation transgenic plants were selected for further analysis. Similar to the T1 generation, the average seed setting rate of the T2 generation was 50.2%, which was also significantly lower than that of the wild-type plants (Table 1).

Table 1.
The seed-setting rate of the TaICK1 over-expressing plants and wild-type plants.
SEM was used to observe the outside epidermal cells and pollen grains from transgenic and wild-type plants. The results showed that the anthers from the wild type were large and full (Figure 2C). By contrast, the anthers from the transgenic T2 generation were short and thin (Figure 2D). Meanwhile, the outside epidermal cells showed a roughly uniform distribution in wild type (Figure 2G), which was abnormal with relatively irregular distribution in the transgenic lines (Figure 2H). Further observations showed that the pollen grains of the wild type were plump, round, and spherical (Figure 2K), while the pollen grains of the transgenic plants were sunken into other irregular shapes (Figure 2L).
2.5. Expression Patterns of TaICK1 and OsICK1 in Transgenic Rice
The expression patterns of TaICK1 and OsICK1 were examined in transgenic and wild-type rice plant anthers using qRT-PCR analyses (Figure 6A,B). The expression pattern of the OsICK1 gene revealed that OsICK1 had relatively higher expression levels in wild-type plants than those in transgenic plants during the Eun and Lun stages (Figure 6B). As shown in Figure 6A, the cDNA of TaICK1 was detected only in the anthers of TaICK1 transgenic plants.
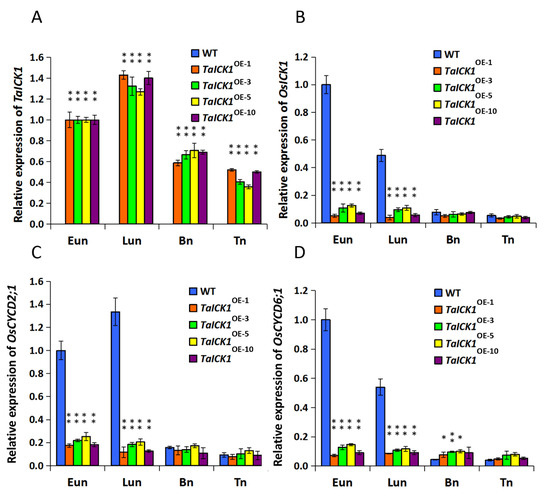
Figure 6.
The expression levels of the genes TaICK1, OsICK1, OsCYCD2;1, and OsCYCD6;1 in TaICK1 transgenic rice and wild-type plants. (A) The expression levels of TaICK1. (B) The expression levels of OsICK1. (C) The expression levels of OsCYCD2;1. (D) The expression levels of OsCYCD6;1. Data are presented as the mean ± SD; the mean and error bars indicate SD from OE-1, OE-3, OE-5, and OE-10 with three independent biological and three technical replicates. The mRNA of TaICK1 was not detected in the anthers of wild-type plants; OE: overexpression plants of T2; the number following OE is a code referring to an independent T2 overexpression plant. The significant differences were assessed using Student’s t-test. * p < 0.05, ** p < 0.01.
2.6. Subcellular Localization Assay of Protein TaICK1
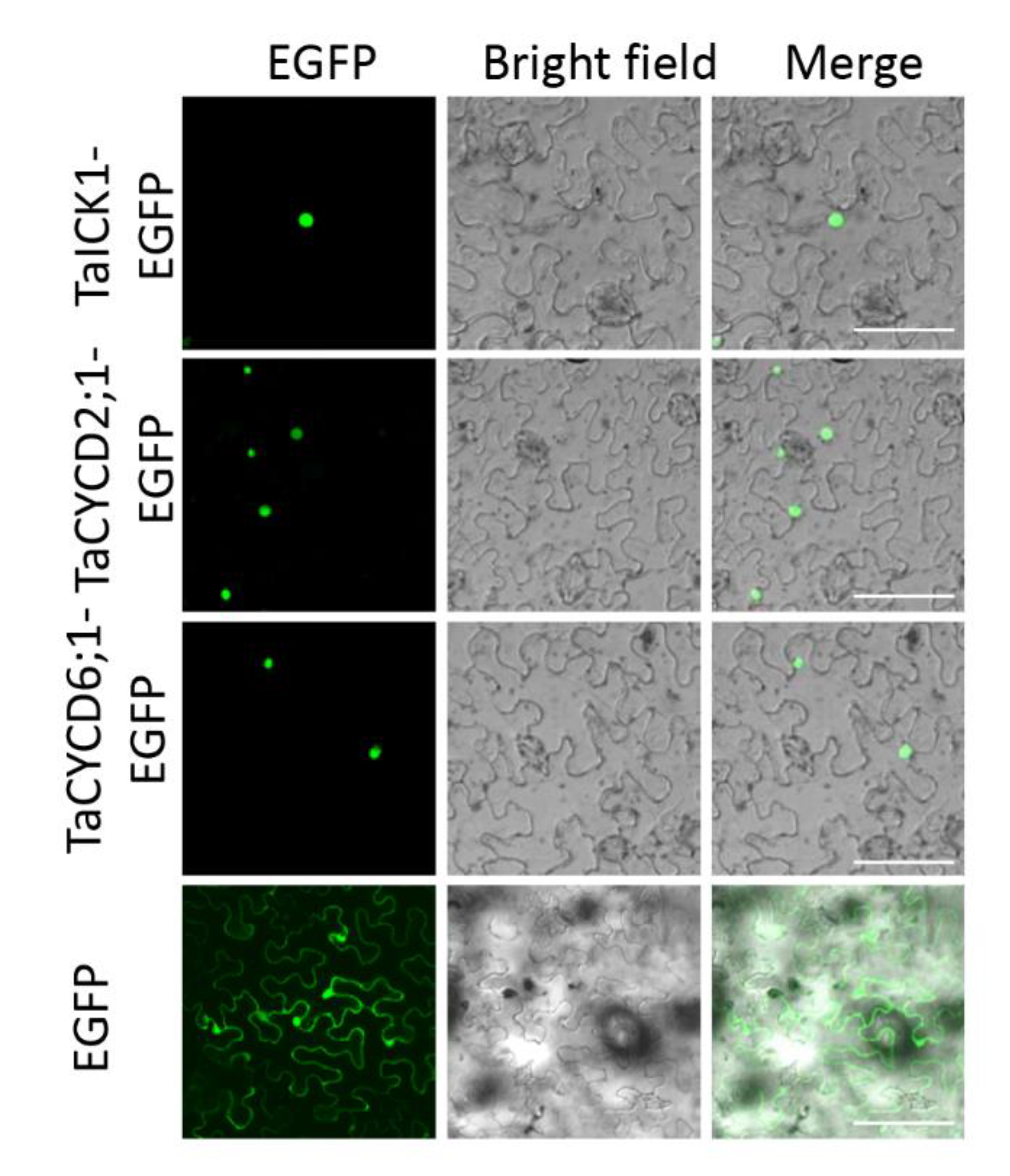
To determine the intracellular distribution of protein TaICK1, the TaICK1-EGFP construct was transformed into the leaf of Nicotiana benthamiana. Signals of the EGFP protein alone, as a positive control, were distributed throughout the cell (Figure 7). By contrast, the green fluorescence of the TaICK1-EGFP fusion protein was only detected in the nucleus (Figure 7).
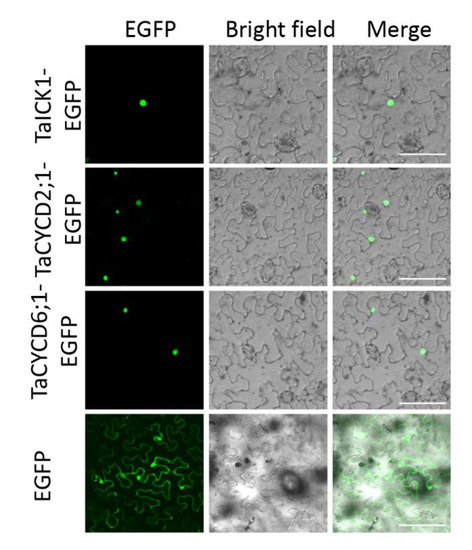
Figure 7.
Subcellular localization of TaICK1, TaCYCD2;1, and TaCYCD6;1 in N. benthamiana cells. The fluorescence (EGFP), bright field, and merged images were obtained using a confocal microscope. The scale bars are 50 μm.
2.7. Prokaryotic Expression of TaICK1 in E. coli
The expected molecular weight of TaICK1 is 24 kDa based on the coding sequence. The fusion protein TF-TaICK1 was expressed in the E. coli strain BL21 (DE3). As shown in Figure 3A, the migration of fusion protein corresponded with a molecular weight of about 75 kDa according to SDS-PAGE. Hence, the objective protein was approximately 27 kDa after subtracting 48 kDa corresponding to the fused trigger factor protein tag. This is approximate with the prediction based on the coding sequence.
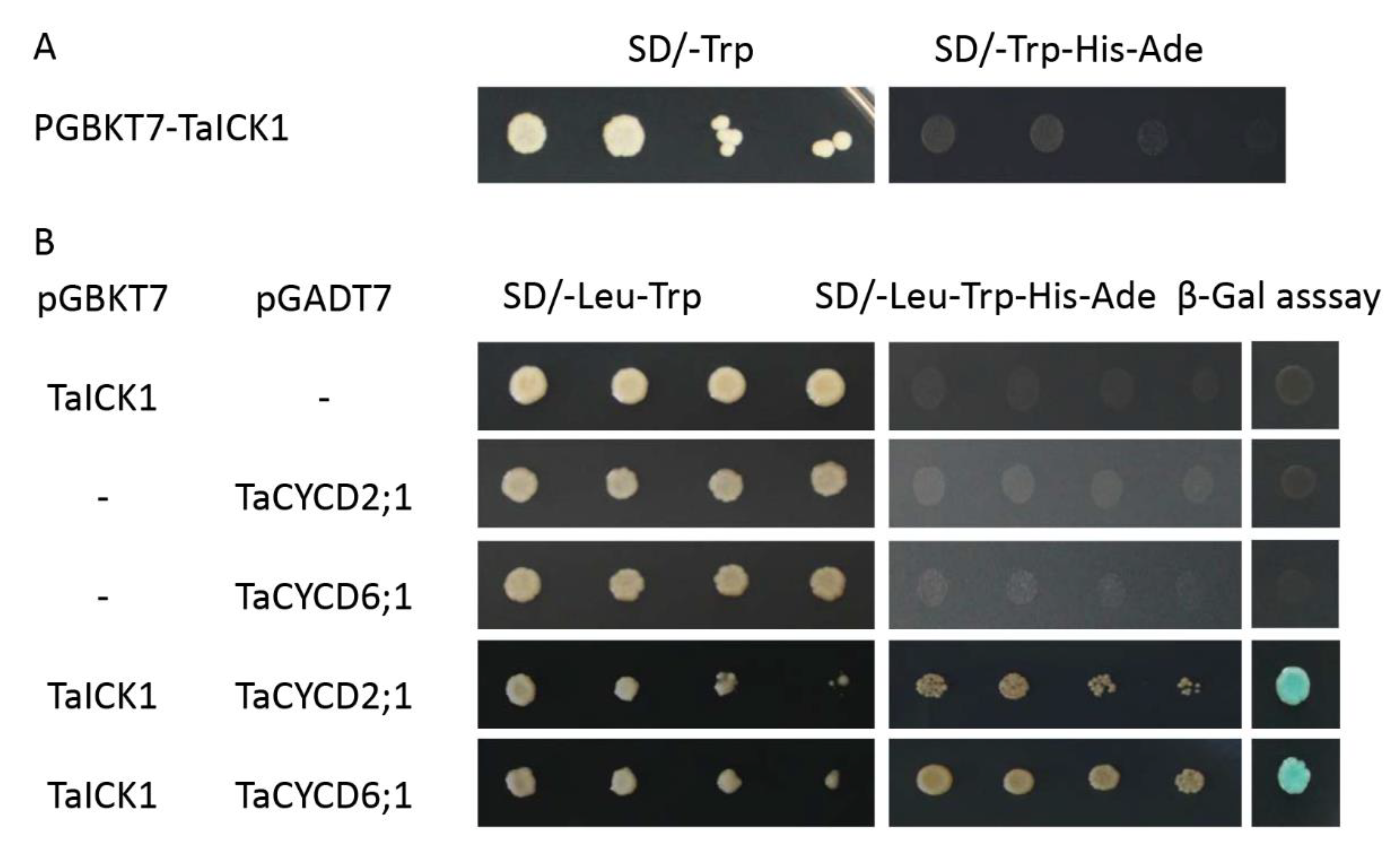
2.8. TaICK1 Interacts with TaCYCD2;1 and TaCYCD6;1
The pGBKT7-TaICK1 plasmid failed to autonomously activate reporter genes in the yeast strain Y2H Gold (Figure 8A). Hence, this plasmid could be used to screen the yeast library. Ultimately, a total of 24 individual yeast clones were obtained, in which AF512432.1 (NCBI) and AK450777.1 (NCBI) were identified and annotated as TaCYCD2;1 and TaCYCD6;1, respectively. TaCYCD2;1 and TaCYCD6;1, which have been identified as being involved in the regulation of the cell cycle, were selected for further analysis. In addition, both the TaCYCD2;1-EGFP and TaCYCD6;1-EGFP fusion proteins were localized to the nucleus in the epidermis leaf cells of Nicotiana benthamiana (Figure 7). The respective protein molecular weights of TaCYCD2;1 and TaCYCD6;1 were approximately 38 and 33 kDa after subtracting the 48 kDa trigger factor protein tag (Figure 3B,C).
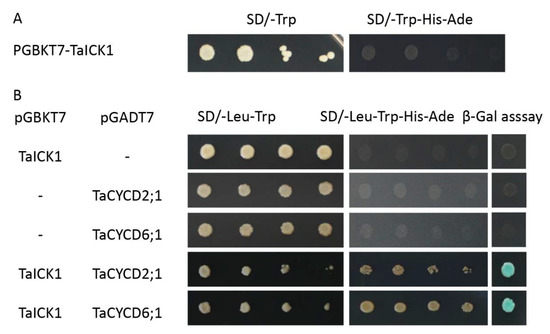
Figure 8.
The interaction of TaICK1 with CYCD2;1 and CYCD6;1 in a yeast two-hybrid assay. (A) Transcriptional activation analysis of the TaICK1 in yeast. (B) The interaction of TaICK1 with TaCYCD2;1 and TaCYCD6;1 in the yeast two-hybrid assays.
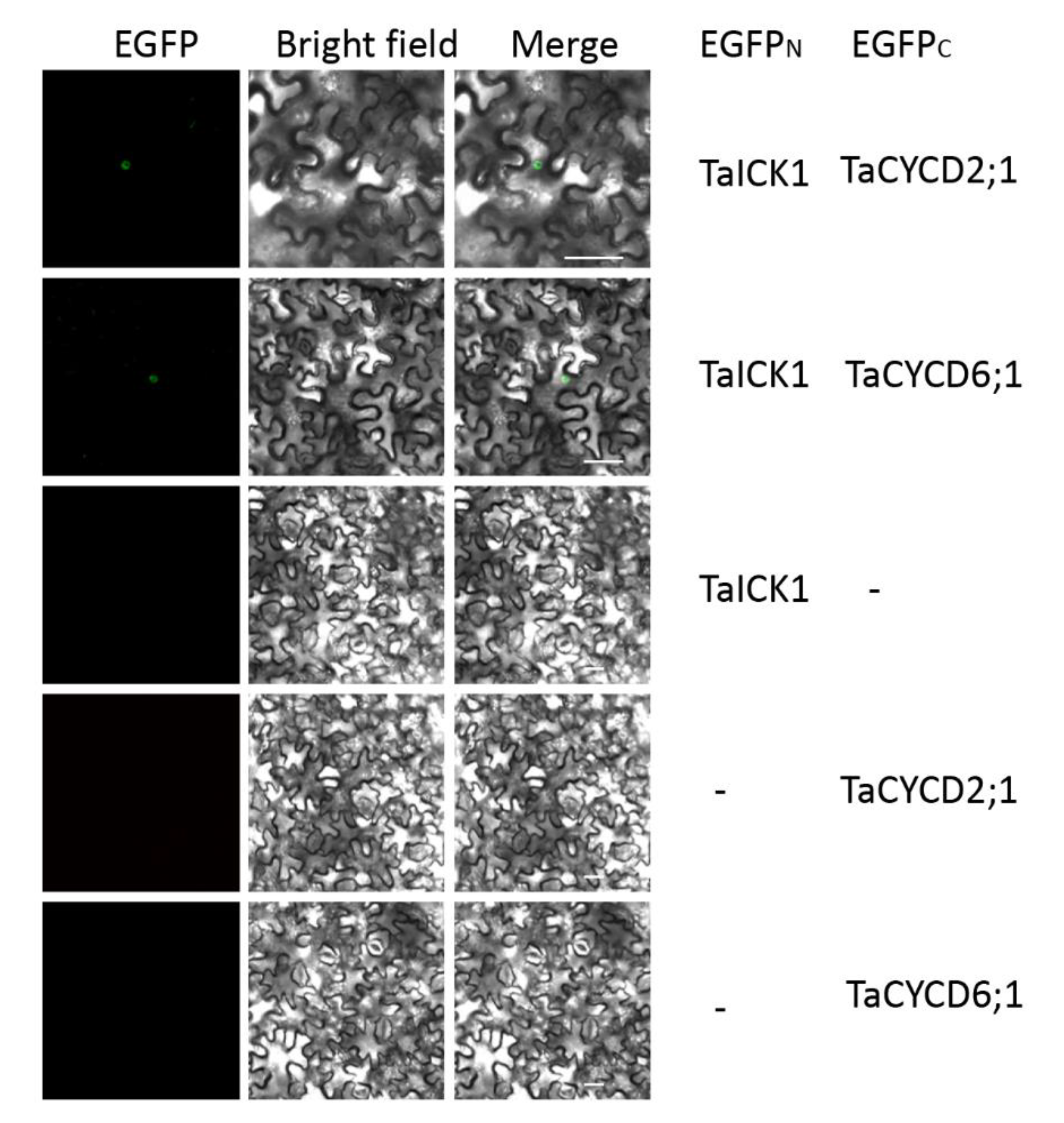
The interactions between TaICK1 and TaCYCD2;1 and TaCYCD6;1 were confirmed in yeast (Figure 8B). The interactions between TaICK1 and CYCD2;1 and TaCYCD6;1 were further validated using a BiFC assay (Figure 9). pCAMBIA1302-35S::TaICK1-EGFPN1–155 and pCAMBIA1302-35S::TaCYCD2;1 -EGFPC156–237 or pCAMBIA1302-35S::TaCYCD6;1-EGFPC156–237 were co-expressed in Nicotiana benthamiana leaf cells. These results not only demonstrate that TaICK1 interacted with TaCYCD2;1 and TaCYCD6;1 but also suggest that the protein complexes of TaICK1 and TaCYCD2;1, as well as that of TaCYCD6;1, may act functionally in the nucleus.
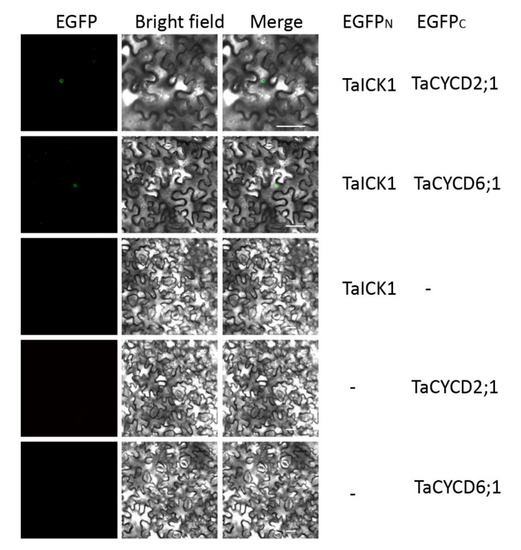
Figure 9.
The interaction of TaICK1 with CYCD2;1 and CYCD6;1 in the BiFC system. Fluorescence (EGFP), bright field, and merged images were obtained using a confocal microscope. The scale bars are 50 µm.
2.9. The Expression Patterns of CYCD2;1 and CYCD6;1 in Wheat and Transgenic Rice
The expression levels of CYCD2;1 and CYCD6;1 were analyzed in the wheat and transgenic rice plants. As shown in Figure 5C, the expression level of TaCYCD2;1 was lower in XN1376-CIMS than that in XN1376, especially at the Eun and Lun stages. In total, the trend of expression increased initially and then decreased from the Eun to Tn stage (Figure 5C). The expression of TaCYCD6;1 was similar to that of TaCYCD2;1 (Figure 5D). Furthermore, the expression levels of OsCYCD2;1 and OsCYCD6;1 were measured in transgenic and wild-type rice plants. The expression of OsCYCD2;1 was higher in transgenic rice plants and wild-type plants at the Eun and Lun stages (Figure 6C), as was OsCYCD6;1 (Figure 6D). Notably, in rice plants overexpressing TaICK1, the expression of OsCYCD2;1 and OsCYCD6;1 was significantly lower than that in wild-type plants (Figure 6C,D).
3. Discussion
CHA SQ-1 can effectively induce male sterility in most wheat varieties, reaching rates of up to 95%. Meanwhile, the seed setting rate of artificial saturation pollination can reach 98% [15]. This result shows the great potential of utilizing heterosis. However, a small amount of self-fertility will affect the purity of hybrid F1 seeds to some extent. Hence, it is urgently required to ameliorate the male sterility induced by SQ-1. Thereafter, we could improve the induction efficiency of male sterility to reduce this self-fertility.Therefore, several studies have investigated the molecular mechanism behind the sterility induced by SQ-1, including characterization of its physiological and biochemical metabolism characteristics [48,49] as well as the genes related to pollen grain development [19,24,50] and proteome [16,22,23,51] and epigenetic [52,53,54,55] profiles. However, the molecular mechanism underlying SQ-1-induced male sterility remains unclear.
In our study, we confirmed that TaICK1 is a potential contributor to wheat male sterility induced by SQ-1. A previous study indicated that ICK proteins are inhibitors of CDK proteins and can block CDK activity by binding, generating CDK–Cycd protein complexes [56]. Some studies have indicated that ICK1 expression level can be induced by ABA [37] or salt [57]. Interestingly, the expression level of TaICK1 in XN1376-CIMS was significantly higher than that in the control plant XN1376 during the Eun, Lun, and Tn stages (Figure 5B). Especially at the Eun and Lun stages, the expression level of XN1376-CIMS was 2.8 and 1.7 times that of XN1376. These results report, for the first time, that CHA SQ-1 may be able to increase the expression of TaICK1. Moreover, as shown in Figure 1J, the pollen grains of XN1376-CIMS were only lightly stained while the pollen grains of XN1376 were deeply stained (Figure 1I). This is consistent with the results of the OsiICK6 overexpression, which showed that the normal pollen grain ratio was only 22% in transgenic plants, which is lower than that in control plants [58]. Similar results were also observed in the Arabidopsis ICK1 overexpression lines [59] and for overexpression of Orysa;KRP1 in rice [36]. Together, these results suggest that the ICK1 gene may act as a key potential contributor to male sterile formation. Meanwhile, previous studies have indicated that the expression levels of ICKs are crucially related to plant development. For example, increased ICK1 expression resulted in reduced CDK activity and also a reduction in the overall number of cells in transgenic Arabidopsis ICK1 plants [40]. A similar phenotype was observed in the transgenic Arabidopsis plants overexpressing KRP2 (the leaves of mature transgenic plants are serrated with enlarged cells) [41]. Similar results were also obtained in our study. The development of TaICK1 transgenic plants showed a difference compared with control rice plants. For example, the seed setting rate of TaICK1OE-1 decreased by 67% compared to the control plants (Table 1). This result is similar to the experiment showing that the transgenic lines overexpressing OsiICK6 have a seed setting rate of only 27.7% on average [58]. Furthermore, the heading period was markedly delayed in TaICK1 transgenic lines (Figure 2N). To the best of our knowledge, this is the first time that ICK genes have been implicated in the regulation of the heading period. Taken together, these results demonstrate that an excess of ICKs greatly impairs the seed setting rate [31,60]. Hence, these results also show that a higher level of TaICK1 in XN1376-CIMS is closely related to male sterility.
Generally, cell division in a plant is controlled by CDK–cyclin protein complexes [56]. In the current study, the expression patterns of TaICK1 in XN1376-CIMS after spraying with SQ-1 suggest that TaICK1 might serve a key role in the process of SQ-1-induced physiological male sterility. Thus, it is necessary to determine whether TaICK1 can interact with the cyclins in wheat. Therefore, a yeast two-hybrid system was used to identify the possible interacting proteins by screening the yeast libraries. Interestingly, TaCYCD2;1 and TaCYCD6;1 were identified from common wheat by using the protein TaICK1 as the bait. It is known that D-type cyclins (Cycds) control the G1/S transition of the mitotic cell cycle in mammals and plants [61], as well as cell growth [62]. CYCD2;1 increases cell formation in leaf [63] and accelerates the rate of leaf initiation [64], suggesting that CYCD2;1 exerts a positive function on cell development. Furthermore, the increased expression of CYCD can suppress the negative effects of excessive ICK/KRP levels on plant growth [39,47]. In XN1376-CIMS, the expression levels of TaCYCD2;1 and TaCYCD6;1 were significantly lower than in XN1376 (Figure 5C,D). One possible explanation is that SQ-1 increased the expression level of TaICK1 and also reduced the TaCYCD2;1 and TaCYCD6;1 expression levels in XN1376-CIMS. However, as shown in Figure 6C,D, the expression levels of OsCYCD2;1 and OsCYCD6;1 were also lower in TaICK1 transgenic rice plants than in control plants. This result demonstrates that reduced TaCYCD2;1 and TaCYCD6;1 expression is not related to SQ-1.
In total, excess TaICK1 impaired the sterility of anthers in XN1376-CIMS. In addition, TaICK1 might suppress the expression level of TaCYCD2;1 and TaCYCD6;1 in the anthers of XN1376-CIMS, which might then impair the fertility and seed setting rate. This study offers new insights into SQ-1-related sterility in common wheat. However, more studies should be carried out to determine, in detail, the molecular mechanism underlying wheat sterility.
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
The wheat cultivar XN1376 was planted in the experimental field of Northwest A&F University, Yangling, Shaanxi, China (108°E, 34°15′N) from 2015 to 2017 (as usual). Trenching was manual and planting was single. The plants were evenly distributed using this approach. SQ-1, a novel CHA, was readily dissolvable in water [49]. A total of 5.0 kg/ha SQ-1 was sprayed on leaves of wheat XN1376 at the 8.5 stage of the Feekes scale to produce male sterile XN1376-CIMS lines [48]. Meanwhile, XN1376 was treated with water to produce control lines. Cytological characteristics were investigated at the different microsporogenesis phases of male sterility and male fertility development. Fresh anthers from four different developmental stages (e.g., Eun, the early uninucleate stage; Lun, the late uninucleate stage; Bn, the binucleate stage; and Tn, the trinucleate stage) were collected and stored at −80 °C.
4.2. Phenotype Characterization of Wheat Anther Development
Photographs of the fresh anthers at each sequential stage were obtained from XN1376 and XN1376-CIMS with a Nikon E995 digital camera (Nikon, Tokyo, Japan) fixed firmly to a Motic K400 dissecting microscope (Preiser Scientific, Louisville, KY, United States). Pollen grains were also analyzed via 1% iodine–potassium iodide (1% KI–I2) staining to determine the viability of the mature pollen, as previously described [65]. Scanning electron microscopy was used to characterize the surface characteristics of the anthers. Before the experiment, fresh anthers and pollen grains were fixed in 4% glutaraldehyde and then treated with an alcohol gradient, dried, and broken in sequence. Finally, the anthers and pollen grains were mounted on a stub with colloidal silver and photographed using a JSM-6360LV scanning electron microscope (JEOL, Tokyo, Japan) [66].
4.3. RNA Extraction and qRT-PCR Analysis
Total RNA was extracted from the anthers of four developmental stages (i.e., Eun, Lun, Bn, and Tn) using TRIzol reagent (Takara Bio, Tokyo, Japan). The integrity of the RNA was assessed using electrophoresis on a 1.5% agarose gel. Meanwhile, the concentration and purity of the RNA was further determined using NanoDrop (Thermo Scientific, Wilmington, DE, USA). A PrimeScriptTM RT reagent Kit with a gDNA Eraser (Takara Bio, Tokyo, Japan) was used to synthesize the first-strand cDNA. 2× RealStar Green Power Mixture (GenStar BioSolutions Co., Ltd., China) was used to perform qRT-PCR. The reaction mixture was used according to the manufacturer’s protocols. The data were collected using a QuantStudioTM Real-Time PCR System (Thermo Fisher, Waltham, MA, USA Applied Biosystems, USA) under the following PCR program: 95 °C for 10 min, followed by 40 cycles at 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 30 s. All experiments were carried out with three technical replicates and three independent biological replicates. The relative expression levels of the genes were computed using the 2−ΔΔCt method, as described by Livak and Schmittgen [67].
4.4. Primer Design
The gene TaICK1 was identified from leaf cDNA of XN1376 based on the nucleotide sequence of the ICK1 in Aegilops tauschii (NCBI reference sequence: XM_020298214.1). The primers used in this study were designed using Oligo7 software. The forward primer sequence was 5′-GCGAAGATGAGGAAGCAG-3′, and the reverse primer sequence was 5′-CATCATGCTCTGCTCACACGG-3′. Gene-specific primers for the genes TaCYCD2;1 (GenBank AF512432.1) and TaCYCD6;1 (GenBank XM_020345750.1) were also designed (Supplementary Table S1). The specific primers used for qRT-PCR are listed in Supplementary Table S2.
4.5. Plasmid Constructs
Plasmid pCAMBIA1301-35S::TaICK1, containing a hygromycin selection marker, was constructed to generate transgenic rice plants. A cauliflower mosaic virus 35S (35S) promoter was used to drive the expression of TaICK1. The pGBKT7-TaICK1 construct was used as the bait to screen the yeast cDNA library of the wheat anthers. In order to confirm the interaction of TaICK1 with TaCYCD2;1 or TaCYCD6;1, the coding sequences of TaCYCD2;1 and TaCYCD6;1 were cloned into pGADT7 as the prey. To generate the subcellular localization vector, the full-length coding sequence of gene TaICK1 (without the termination codon) was cloned into the expression vector backbone pCAMBIA1302 under the control of the 35S promoter to create a pCAMBIA1302-2×35S::TaICK1-EGFP construct to express the TaICK1-EGFP fusion protein. Likewise, the pCAMBIA1302-2×35S::TaCYCD2;1-EGFP and pCAMBIA1302-2×35S::TaCYCD6;1-EGFP constructs were also created. For BiFC vector construction, the full-length coding sequence of TaICK1 was ligated with DNA of EGFPN1–155 to produce the pCAMBIA1302-TaICK1-EGFPN1–155 construct for fusion protein expression driven by the 35S promoter. The pCAMBIA1302-TaCYCD2;1-EGFPC156–237 and pCAMBIA1302-TaCYCD6;1-EGFPC156–237 constructs were similarly generated for fusion protein expression. The prokaryotic expression vector pCold-TF (TaKaRa, Japan) was used to express the proteins TaICK1, TaCYCD2;1, and TaCYCD6;1, which generates a fusion protein with an extra 48kD trigger factor protein tag. The details of primers used for vector construction are given in Supplementary Table S1.
4.6. Generation of the TaICK1 Overexpression Rice Plants
To generate transgenic plants, the plasmid pCAMBIA1301-35S::TaICK1 was introduced into Agrobacterium tumefaciens strain EHA105 using electroporation [68], and positive transformants were identified using PCR assay. Nipponbare rice plants were transformed as described [69]. The transgenic seedlings were initially identified using PCR with the primers described in Supplemental Table S2. The transgenic lines were then further screened on an identification buffer containing 50 mg L−1 of hygromycin and 0.5 mg L−1 6-BA [70]. The transgenic rice leaves were able to remain green in this buffer, while the leaves from the non-transgenic rice showed a chlorotic phenotype. The T2 plants derived from at least three independent T1 transgenic plants were used to characterize the phenotype and assay gene expression using three independent biological replicates.
4.7. Yeast Two-Hybrid Assay
The yeast cDNA library of the anther tissue was introduced into the yeast strain Y187. The analyses were performed according the manufacturer’s instructions (Clontech, USA). The transcription activation activity of the bait protein was analyzed through the yeast growth status and an α-galactosidase activity assay on medium SD (−Trp/−Ade/−His) plates. For yeast cDNA library screening, Y2H colonies containing the pGBKT7-TaICK1 vector were mated with yeast strain Y187 (cDNA library of Anthers tissue), according to the instructions of the Two-Hybrid System 3 (Clontech, USA). Then, the mating type was selected on a high-stringency medium SD (−Ade/−His/−Leu/−Trp). Finally, after cloning the full length CDS of the prey gene, the prey genes were reconstructed into pGADT7. To further verify the protein interactions, the pGBKT7-bait vector and pGADT7-prey vector were co-transformed into Y2H Gold yeast cells (yeast strain Y2H, Clontech, USA), and the positive clones were selected on the SD medium (−Leu/−Trp). Positive yeast clones were picked and spread on the SD medium (−Ade/−His/−Leu/−Trp/X-α-Gal) to assay for protein interactions.
4.8. Subcellular Localization and BiFC Assay
Subcellular localization vectors were transformed into Agrobacterium tumefaciens strain GV3101, which was cultured in an LB medium with appropriate antibiotics (50 mg L−1 Kan, 25 mg L−1 gentamycin, and 25 mg L−1 rifampicin) under shaking at 200 rpm at 28 °C for about 36 h. Agrobacteria tumefaciens was then centrifuged at 5000g for 5 min to collect the bacteria when the OD600 reached 0.6. The cells were resuspended in resuspension buffer (containing 10 mM MES, 10 mM MgCl2, and 200 mM acetosyringone) and incubated for 2 h at room temperature. The suspension of Agrobacteria tumefaciens was then used to infiltrate 4- to 6-week-old Nicotiana benthamiana leaves as previously described [71,72]. Similarly, for the BiFC assay, pairs of EGFPN and EGFPC fusion proteins were transiently co-expressed in the leaves of N. benthamiana. Infiltrated leaves were observed using a laser confocal scanning microscope Olympus IX83 confocal microscope (Olympus, Tokyo, Japan). The excitation and detection wavelengths for EGFP were 514 and 527 nm, respectively.
4.9. Recombinant Protein Expression in E. coli
Plasmids pCold-TF-TaICK1, pCold-TF-TaCYCD2;1, and pCold-TF-TaCYCD6;1 were introduced into the E. coli BL21 (DE3) strain (TransGen Biotech, China) via the heat shock method, as described by the manufacturer. Colonies of verified transformants were incubated in LB [73] medium containing 50 mg L−1 carbenicillin at 37 °C for 8–12 h until the optical density OD600 reached 0.6–0.8. IPTG (Isopropyl β-D-Thiogalactoside) was then added into the medium at a final concentration of 0.5 mM, followed by incubation at 16 °C overnight with shaking at 100 rpm. The control culture was treated using the same protocol without IPTG. After incubation, the crude cell extracts were prepared as described [74]. Then, the crude proteins were analyzed using SDS-PAGE and dyed with Coomassie brilliant blue R-250 using the protocol of Sambrook and Russell [74].
5. Conclusions
In this study, the wheat TaICK1 gene was isolated and characterized. Moreover, the chemical hybridization agent SQ-1 was shown to induce male sterility in common wheat, as well as a higher expression level of TaICK1 in the anthers of sterile wheat lines. More importantly, the overexpression of TaICK1 suppressed the expression of TaCYCD2;1 and TaCYCD6;1 and reduced the fertility seed setting rates. These results provide new insights into the molecular mechanism of wheat male sterility induced by the chemical hybridization agent SQ-1.
Supplementary Materials
Supplementary materials can be found at https://www.mdpi.com/1422-0067/21/7/2468/s1. Table S1. Primers for gene-specific and plasmid construction in this study; Table S2. Primers used for quantitative real-time RT-PCR analysis.
Author Contributions
L.Z. and G.Z. conceived and designed the study. L.Z., C.W., Y.Y., Y.Z. (Yamin Zhang), Z.L. (Zheng Li), Y.Z. (Yanfang Zhang), X.G., D.L., and Z.L. (Ziliang Li) participated in experiments. L.Z. wrote the manuscript. L.Z., C.W., Y.S., S.W., S.M., J.Z., H.Z., and G.Z. discussed the results and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
The study was sponsored by the National Support Program of China (No. 2015BAD27B01), the Technological Innovation and Over Planning Projects of Shaanxi Province (No. 2014KTZB02-01-02).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gill, B.S.; Appels, R.; Botha-Oberholster, A.; Buell, C.R.; Bennetzen, J.L.; Chalhoub, B.; Chumley, F.; Dvorák, J.; Iwanaga, M.; Keller, B.; et al. A workshop report on wheat genome sequencing: International Genome Research on Wheat Consortium. Genetics 2004, 168, 1087–1096. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Mir, R.R.; Mohan, A.; Kumar, J. Wheat genomics: Present status and future prospects. Int. J. Plant Genom. 2008, 2008, 896451. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, J.; Scott, P.; Brinton, J.; Mestre, T.C.; Bush, M.; Del, B.A.; Dubcovsky, J.; Uauy, C. A splice acceptor site mutation in TaGW2-A1 increases thousand grain weight in tetraploid and hexaploid wheat through wider and longer grains. Theor. Appl. Genet. 2016, 129, 1099–1112. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.C.; Zhang, X.K.; Yin, G.H.; Wang, L.N.; Han, Y.L.; Chen, L.; Huang, F.; Tang, J.W.; Xia, X.C.; He, Z.H. Genetic gains in grain yield, net photosynthesis and stomatal conductance achieved in Henan Province of China between 1981 and 2008. Field Crop. Res. 2011, 122, 225–233. [Google Scholar] [CrossRef]
- Ray, D.K.; Mueller, N.D.; West, P.C.; Foley, J.A. Yield Trends Are Insufficient to Double Global Crop Production by 2050. PLoS ONE 2013, 8, e66428. [Google Scholar] [CrossRef]
- Yang, Z.; Bai, Z.; Li, X.; Wang, P.; Wu, Q.; Yang, L.; Li, L.; Li, X. SNP identification and allelic-specific PCR markers development for TaGW2, a gene linked to wheat kernel weight. Theor. Appl. Genet. 2012, 125, 1057–1068. [Google Scholar] [CrossRef]
- Kindred, D.R.; Gooding, M.J. Heterosis for yield and its physiological determinants in wheat. Euphytica 2005, 142, 149–159. [Google Scholar] [CrossRef]
- Zhang, Q. Utilization of crop heterosis: A review. Euphytica 2014, 2, 161–173. [Google Scholar]
- Singh, S.P.; Srivastava, R.; Kumar, J. Male sterility systems in wheat and opportunities for hybrid wheat development. Acta Physiol. Plant. 2014, 37, 1713. [Google Scholar] [CrossRef]
- Hohn, C.E.; Lukaszewski, A.J. Engineering the 1BS chromosome arm in wheat to remove the Rf multi locus restoring male fertility in cytoplasms of Aegilops kotschyi, Ae. uniaristata and Ae. mutica. Theor. Appl. Genet. 2016, 129, 1769–1774. [Google Scholar] [CrossRef][Green Version]
- Cheng, Y.; Wang, Q.; Li, Z.; Cui, J.; Hu, S.; Zhao, H.; Chen, M. Cytological and comparative proteomic analyses on male sterility in Brassica napus L. induced by the chemical hybridization agent monosulphuron ester sodium. PLoS ONE 2013, 8, e80191. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.W.; Zhang, G.S.; Wang, J.W.; Wang, X.L.; Fang, Z.W. Effect of male sterility on different wheat genotype induced by SQ-1. J. Northwest Univ. Nat. Sci. Ed. 2003, 31, 15–18. [Google Scholar]
- Zhang, G.; Liu, H.; Wang, J. New variety of hybrid wheat with high resistance, quality and yield. Agric. New Technol. 2003, 4, 25–26. [Google Scholar]
- Chen, C.; Lu, Y.; Zhang, G.; Zhang, Z.; Li, P.; Zhang, B. The synthesis of genesis and the research of bioactive chemicals. Chin. J. Pestic. 2004, 43, 315–317. [Google Scholar]
- Wang, S.; Zhang, G.; Song, Q.; Zhang, Y.; Li, Y.; Guo, J.; Chen, Z.; Niu, N.; Ma, S.; Wang, J. Programmed cell death, antioxidant response and oxidative stress in wheat flag leaves induced by chemical hybridization agent SQ-1. J. Integr. Agr. 2016, 15, 76–86. [Google Scholar] [CrossRef]
- Song, Q.; Wang, S.; Zhang, G.; Li, Y.; Li, Z.; Guo, J.; Niu, N.; Wang, J.; Ma, S. Comparative proteomic analysis of a membrane-enriched fraction from flag leaves reveals responses to chemical hybridization agent SQ-1 in wheat. Front. Plant Sci. 2015, 6, 669. [Google Scholar] [CrossRef]
- Chen, R.; Ye, J.; Zhang, G.; Wang, J.; Niu, N.; Ma, S.; Zhao, J.; Zhu, J. Differential proteomic analysis of anther proteins between cytoplasmic-nuclear male sterility line and its maintainer in wheat (Triticum aestivum L.). Prog. Biochem. Biophys. 2009, 36, 431–440. [Google Scholar]
- Wang, J.; Zhang, G.; Yuan, L.; Zhang, M.; Niu, N.; Ma, S.; Ye, J. Metabolism of Reactive Oxygen Species of Physiological Male Sterile Anther Induced by Chemical Hybrid Agent in Wheat. Acta Bot. Boreali Occident. Sin. 2009, 29, 1351–1357. [Google Scholar]
- Wei, M.; Wang, J.; Zhang, G.; Zhang, L.; Yang, Z.; Sun, R.; Ye, J.; Niu, N.; Ma, S.; Li, H. Relationship between the expression of GAPDH gene and anther abortion of physiological male sterile of wheat. Mol. Plant Breed. 2009, 7, 679–684. [Google Scholar]
- Ye, J.; Zhang, G.; Wang, S.; Chen, R.; Wang, J.; Niu, N.; Ma, S.; Li, H.; Zhu, J. Differential proteomic studies on pollen grain proteins of wheat male sterile line induced by chemical hybridizing agent SQ-1. Chin. J. Biochem. Mol. Biol. 2009, 25, 949–957. [Google Scholar]
- Wang, S.; Zhang, Y.; Song, Q.; Fang, Z.; Chen, Z.; Zhang, Y.; Zhang, L.; Zhang, L.; Niu, N.; Ma, S.; et al. Mitochondrial Dysfunction Causes Oxidative Stress and Tapetal Apoptosis in Chemical Hybridization Reagent-Induced Male Sterility in Wheat. Front. Plant Sci. 2018, 8, 2217. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, G.; Wang, J.; Li, J.; Song, Y.; Qiao, L.; Niu, N.; Wang, J.; Ma, S.; Li, L. Chemical hybridizing agent SQ-1-induced male sterility in Triticum aestivum L.: A comparative analysis of the anther proteome. BMC Plant Biol. 2018, 18, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, Y.; Fang, Z.; Zhang, Y.; Song, Q.; Hou, Z.; Sun, K.; Song, Y.; Li, Y.; Ma, D.; et al. Cytological and Proteomic Analysis of Wheat Pollen Abortion Induced by Chemical Hybridization Agent. Int. J. Mol. Sci. 2019, 20, 1615. [Google Scholar] [CrossRef]
- Zhang, L.; Yuan, L.; Yang, S.L.; Zhang, G.S.; Wang, J.S.; Song, Y.L.; Zhao, Z.J.; Niu, N.; Ma, S.C. Expression characteristic on Ta PDC- E1α gene and its regulatory enzymes gene in male sterile line of wheat(Triticum aestivum L.). Acta Agron. Sin. 2011, 37, 620–628. [Google Scholar] [CrossRef]
- Yui, R.; Iketani, S.; Mikami, T.; Kubo, T. Antisense inhibition of mitochondrial pyruvate dehydrogenase E1α subunit in anther tapetum causes male sterility. Plant J. 2003, 34, 57–66. [Google Scholar] [CrossRef] [PubMed]
- de Kok, A.; Hengeveld, A.F.; Martin, A.; Westphal, A.H. The pyruvate dehydrogenase multi-enzyme complex from Gram-negative bacteria. Biochim. Biophys. Acta BBA Protein Struct. Mol. Enzymol. 1998, 1385, 353–366. [Google Scholar] [CrossRef]
- Tovar-Méndez, A.; Miernyk, J.A.; Randall, D.D. Regulation of pyruvate dehydrogenase complex activity in plant cells. Eur. J. Biochem. 2003, 270, 1043–1049. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Zhao, X.L.; Liu, H.Z.; Song, Y.L.; Zhang, G. Proliferating cell nuclear antigen (TaPCNA), a pyruvate dehydrogenase kinase (TaPDK)-interacting protein is involved in the regulation of microspore cell cycle in wheat (Triticum aestivum). J. Agric. Biotechnol. 2013, 21, 1045–1051. [Google Scholar]
- Yu, Y. Study on Expression Analysis of TaPCNA from Regulation of Microspore Cell Cycle and Its Interactive Protein of Physiological Male- Sterility in Wheat; Northwest A&F university: Xianyang, China, 2015. [Google Scholar]
- Wang, H.; Fowke, L.C.; Crosby, W.L. A plant cyclin-dependent kinase inhibitor gene. Nature 1997, 386, 451–452. [Google Scholar] [CrossRef]
- Lui, H.; Wang, H.; DeLong, C.; Fowke, L.C.; Crosby, W.L.; Fobert, P.R. The Arabidopsis Cdc2a-interacting protein ICK2 is structurally related to ICK1 and is a potent inhibitor of cyclin-dependent kinase activity in vitro. Plant J. 2000, 21, 379–385. [Google Scholar] [CrossRef]
- Gusti, A.; Baumberger, N.; Nowack, M.; Pusch, S.; Eisler, H.; Potuschak, T.; De Veylder, L.; Schnittger, A.; Genschik, P. The Arabidopsis thaliana F-box protein FBL17 is essential for progression through the second mitosis during pollen development. PLoS ONE 2009, 4, e4780. [Google Scholar] [CrossRef] [PubMed]
- Jasinski, S.; Perennes, C.; Bergounioux, C.; Nathalie, G. Comparative Molecular and Functional Analyses of the Tobacco Cyclin-Dependent Kinase Inhibitor NtKIS1a and Its Spliced Variant NtKIs1b. Plant Physiol. 2002, 130, 1871–1882. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.M.; Dante, R.A.; Sabelli, P.A.; Sun, Y.; Dilkes, B.P.; Gordon-Kamm, W.J.; Larkins, B.A. Cyclin-Dependent Kinase Inhibitors in Maize Endosperm and Their Potential Role in Endoreduplication. Plant Physiol. 2005, 138, 2323–2336. [Google Scholar] [CrossRef] [PubMed]
- Bisbis, B.; Delmas, F.; Joubès, J.; Sicard, A.; Hernould, M.; Inzé, D.; Mouras, A.; Chevalier, C. Cyclin-dependent Kinase (CDK) Inhibitors Regulate the CDK-Cyclin Complex Activities in Endoreduplicating Cells of Developing Tomato Fruit. J. Biol. Chem. 2006, 281, 7374–7383. [Google Scholar] [CrossRef] [PubMed]
- Barrôco, R.M.; Peres, A.; Droual, A.; De Veylder, L.; Nguyen, L.S.L.; De Wolf, J.; Mironov, V.; Peerbolte, R.; Beemster, G.T.S.; Inzé, D.; et al. The cyclin-dependent kinase inhibitor Orysa;KRP1 plays an important role in seed development of rice. Plant Physiol. 2006, 142, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Qi, Q.; Schorr, P.; Cutler, A.J.; Crosby, W.L.; Fowke, L.C. ICK1, a cyclin-dependent protein kinase inhibitor from Arabidopsis thaliana interacts with both Cdc2a and CycD3, and its expression is induced by abscisic acid. Plant J. 1998, 15, 501–510. [Google Scholar] [CrossRef]
- Torres Acosta, J.A.; Fowke, L.C.; Wang, H. Analyses of phylogeny, evolution, conserved sequences and genome-wide expression of the ICK/KRP family of plant CDK inhibitors. Ann. Bot. (Lond.) 2011, 107, 1141–1157. [Google Scholar] [CrossRef]
- Jasinski, S.; Riou-Khamlichi, C.; Roche, O.; Perennes, C.; Bergounioux, C.; Glab, N. The CDK inhibitor NtKIS1a is involved in plant development, endoreduplication and restores normal development of cyclin D3; 1-overexpressing plants. J. Cell Sci. 2002, 115, 973–982. [Google Scholar]
- Wang, H.; Zhou, Y.; Gilmer, S.; Whitwill, S.; Fowke, L.C. Expression of the plant cyclin-dependent kinase inhibitor ICK1 affects cell division, plant growth and morphology. Plant J. 2000, 24, 613–623. [Google Scholar] [CrossRef]
- De Veylder, L.; Beeckman, T.; Beemster, G.T.; Krols, L.; Terras, F.; Landrieu, I.; van der Schueren, E.; Maes, S.; Naudts, M.; Inzé, D. Functional analysis of cyclin-dependent kinase inhibitors of Arabidopsis. Plant Cell 2001, 13, 1653–1668. [Google Scholar] [CrossRef]
- Zhou, Y.; Fowke, L.; Wang, H. Plant CDK inhibitors: Studies of interactions with cell cycle regulators in the yeast two-hybrid system and functional comparisons in transgenic Arabidopsis plants. Plant Cell Rep. 2002, 20, 967–975. [Google Scholar] [CrossRef]
- Schnittger, A.; Weinl, C.; Bouyer, D.; Schöbinger, U.; Hülskamp, M. Misexpression of the cyclin-dependent kinase inhibitor ICK1/KRP1 in single-celled Arabidopsis trichomes reduces endoreduplication and cell size and induces cell death. Plant Cell 2003, 15, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Oh, S.A.; Brownfield, L.; Hong, S.H.; Ryu, H.; Hwang, I.; Twell, D.; Nam, H.G. Control of plant germline proliferation by SCFFBL17 degradation of cell cycle inhibitors. Nature 2008, 455, 1134–1137. [Google Scholar] [CrossRef] [PubMed]
- Oakenfull, E.A.; Riou-Khamlichi, C.; Murray, A.H. Plant D—Type cyclins and the control of G1 progression. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2002, 357, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Sanz, L.; Dewitte, W.; Forzani, C.; Patell, F.; Nieuwland, J.; Wen, B.; Quelhas, P.; De Jager, S.; Titmus, C.; Campilho, A.; et al. The Arabidopsis D-type cyclin CYCD2;1 and the inhibitor ICK2/KRP2 modulate auxin-induced lateral root formation. Plant Cell 2011, 23, 641–660. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, H.; Gilmer, S.; Whitwill, S.; Fowke, L.C. Effects of co-expressing the plant CDK inhibitor ICK1 and D-type cyclin genes on plant growth, cell size and ploidy in Arabidopsis thaliana. Planta 2003, 216, 604–613. [Google Scholar] [CrossRef]
- Song Ba, Q.; Sheng Zhang, G.; Sheng Wang, J.; Xue Che, H.; Liu, H.; Niu, N.; Cai Ma, S.; Wei Wang, J. Relationship between metabolism of reactive oxygen species and chemically induced male sterility in wheat (Triticum aestivum L.). Can. J. Plant Sci. 2013, 93, 675–681. [Google Scholar]
- Ba, Q.; Zhang, G.; Che, H.; Liu, H.; Ng, T.; Zhang, L.; Wang, J.; Sheng, Y.; Niu, N.; Ma, S.; et al. Aliphatic Metabolism during Anther Development Interfered by Chemical Hybridizing Agent in Wheat. Crop Sci. 2014, 54, 1458–1467. [Google Scholar] [CrossRef]
- Zhang, M.; Yuan, L.; Wang, J.; Zhang, G.; Guo, W.; Zhang, L. Expressing Analysis of cACO Gene in Male Sterile Wheat Induced by Chemical Hybridizing Agent SQ-1. Chin. J. Biochem. Mol. Biol. 2010, 26, 740–748. [Google Scholar]
- Wang, S.; Zhang, G.; Zhang, Y.; Song, Q.; Chen, Z.; Wang, J.; Guo, J.; Niu, N.; Wang, J.; Ma, S. Comparative studies of mitochondrial proteomics reveal an intimate protein network of male sterility in wheat (Triticum aestivum L.). J. Exp. Bot. 2015, 66, 6190–6203. [Google Scholar] [CrossRef]
- Wang, J.; Yuan, L.; Zhang, M.; Zhang, G.; Niu, N.; Ma, S.; Li, H. Expression of Ubiquitin-26S Proteasome Pathway in Physiological Male Sterility of Wheat Induced by Chemical Hybrid Agents SQ-1. J. Agric. Biotechnol. 2010, 18, 695–701. [Google Scholar]
- Sang, Q.; Liu, H.; Zhang, G.; Zhu, Q.; Zhu, W.; Zhang, L.; Che, H. Study on the Relationship between the Ubiquitination of Histones on Chromosomes and the Physiological Male Sterility Induced by Chemical Hybridizing Agent SQ-1 in Wheat (Triticum aestivum L.). J. Agric. Biotechnol. 2013, 21, 396–406. [Google Scholar]
- Ba, Q.; Zhang, G.; Wang, J.; Niu, N.; Ma, S.; Wang, J. Gene expression and DNA methylation alterations in chemically induced male sterility anthers in wheat (Triticum aestivum L.). Acta Physiol. Plant. 2014, 36, 503–512. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, G.; Zhu, W.; Wu, W.K.K.; Ba, Q.; Zhang, L.; Zhang, L.; Niu, N.; Ma, S.; Wang, J. Differential proteomic analysis of polyubiquitin-related proteins in chemical hybridization agent-induced wheat (Triticum aestivum L.) male sterility. Acta Physiol. Plant. 2014, 36, 1473–1489. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Fowke, L.C. The emerging importance of cyclin-dependent kinase inhibitors in the regulation of the plant cell cycle and related processesThis review is one of a selection of papers published in the Special Issue on Plant Cell Biology. Can. J. Bot. 2006, 84, 640–650. [Google Scholar] [CrossRef]
- Pettkó-Szandtner, A.; Mészáros, T.; Horváth, G.V.; Bakó, L.; Csordás-Tóth, É.; Blastyák, A.; Zhiponova, M.; Miskolczi, P.; Dudits, D. Activation of an alfalfa cyclin-dependent kinase inhibitor by calmodulin-like domain protein kinase. Plant J. 2006, 46, 111–123. [Google Scholar] [CrossRef]
- Yang, R.; Tang, Q.; Wang, H.; Zhang, X.; Pan, G.; Wang, H.; Tu, J. Analyses of two rice (Oryza sativa) cyclin-dependent kinase inhibitors and effects of transgenic expression of OsiICK6 on plant growth and development. Ann. Bot. 2011, 107, 1087–1101. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, H.; Gilmer, S.; Whitwill, S.; Keller, W.; Fowke, L.C. Control of petal and pollen development by the plant cyclin-dependent kinase inhibitor ICK1 in transgenic Brassica plants. Planta 2002, 215, 248–257. [Google Scholar] [CrossRef]
- Sherr, C.; Roberts, J. CDK inhibitors: Positive and negative regulators of G1-phase progression. Genes Dev. 1999, 13, 1501–1512. [Google Scholar] [CrossRef]
- Zheng, T.; Zhuo, X.; Li, L.; Cheng, T.; Zhang, Q. Genome-Wide Analysis of the D-type Cyclin Gene Family Reveals Differential Expression Patterns and Stem Development in the Woody Plant Prunus mume. Forests 2019, 10, 147. [Google Scholar] [CrossRef]
- Menges, M.; Samland, A.K.; Planchais, S.; Murray, J.A.H. The D-Type Cyclin CYCD3;1 Is Limiting for the G1-to-S-Phase Transition inArabidopsis. Plant Cell 2006, 18, 893–906. [Google Scholar] [CrossRef] [PubMed]
- Qi, R.; John, P.C.L. Expression of genomic AtCYCD2;1 in Arabidopsis induces cell division at smaller cell sizes: Implications for the control of plant growth. Plant Physiol. 2007, 144, 1587–1597. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cockcroft, C.E.; den Boer, B.G.W.; Healy, J.M.S.; Murray, J.A.H. Cyclin D control of growth rate in plants. Nature 2000, 405, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Gong, L.; Yuan, W.; Li, X.; Chen, G.; Li, X.; Zhang, Q.; Wu, C. Replication Protein A (RPA1a) Is Required for Meiotic and Somatic DNA Repair But Is Dispensable for DNA Replication and Homologous Recombination in Rice. Plant Physiol. 2009, 151, 2162–2173. [Google Scholar] [CrossRef]
- Liu, M.; Li, H.; Su, Y.; Li, W.; Shi, C. G1/ELE Functions in the Development of Rice Lemmas in Addition to Determining Identities of Empty Glumes. Front. Plant Sci. 2016, 7, 1006. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Wu, Y.; Williams, M.; Bernard, S.; Driouich, A.; Showalter, A.M.; Faik, A. Functional Identification of Two Nonredundant Arabidopsis α(1,2) Fucosyltransferases Specific to Arabinogalactan Proteins. J. Biol. Chem. 2010, 285, 13638–13645. [Google Scholar] [CrossRef]
- Liu, X.; Bai, X.; Wang, X.; Chu, C. OsWRKY71, a rice transcription factor, is involved in rice defense response. J. Plant Physiol. 2007, 164, 969–979. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, X.; Wang, X.; Peng, L.; Gu, M. A Rapid Simple Method of Assaying Hygromycin Resistance in Transgenic Rice Plants. J. Agric. Biotechnol. 2001, 9, 264. [Google Scholar]
- Waadt, R.; Schmidt, L.K.; Lohse, M.; Hashimoto, K.; Bock, R.; Kudla, J. Multicolor bimolecular fluorescence complementation reveals simultaneous formation of alternative CBL/CIPK complexes in planta. Plant. J. 2008, 56, 505–516. [Google Scholar] [CrossRef]
- Sparkes, I.A.; Runions, J.; Kearns, A.; Hawes, C. Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat. Protoc. 2006, 1, 2019–2025. [Google Scholar] [CrossRef] [PubMed]
- Schatz, P.J.; Beckwith, J. Genetic Analysis of Protein Export in Escherichia Coli. Annu. Rev. Genet. 1990, 24, 215–248. [Google Scholar] [CrossRef] [PubMed]
- Beliveau, C.; Potvin, C.; Trudel, J.; Asselin, A.; Bellemare, G. Cloning, sequencing, and expression in Escherichia coli of a Streptococcus faecalis autolysin. J. Bacteriol. 1991, 173, 5619–5623. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).