Abstract
First-generation nonsteroidal androgen receptor (AR) antagonists, such as flutamide (2a) and bicalutamide (3), are effective for most prostate cancer patients, but resistance often appears after several years due to the mutation of AR. Second-generation AR antagonists are effective against some of these castration-resistant prostate cancers, but their structural variety is still limited. In this study, we designed and synthesized 4-methyl-7-(N-alkyl-arylcarboxamido)coumarins as AR antagonist candidates and evaluated their growth-inhibitory activity toward androgen-dependent SC-3 cells. Coumarinamides with a secondary amide bond did not show inhibitory activity, but their N-methylated derivatives exhibited AR-antagonistic activity. Especially, 19b and 31b were more potent than the lead compound 7b, which was comparable to hydroxyflutamide (2b). Conformational analysis showed that the inactive coumarinamides with a secondary amide bond have an extended structure with a trans-amide bond, while the active N-methylated coumarinamides have a folded structure with a cis-amide bond, in which the two aromatic rings are placed face-to-face. Docking study suggested that this folded structure is important for binding to AR. Selected coumarinamide derivatives showed AR-antagonistic activity toward LNCaP cells with T877A AR, and they had weak progesterone receptor (PR)-antagonistic activity. The folded coumarinamide structure appears to be a unique pharmacophore, different from those of conventional AR antagonists.
1. Introduction
Androgen receptor (AR) is a ligand-dependent transcription factor belonging to the nuclear receptor superfamily [1,2], and its endogenous ligands, so-called androgens, are testosterone (1a) (see Supplementary Materials) and dihydrotestosterone (DHT, 1b) (Figure 1). These androgens have various roles in the differentiation, growth, and maintenance of the male reproductive organs [3,4]. Based on its physiological and pharmacological functions, AR is recognized as an important target for drug discovery. For example, since androgen plays an important role in the progression of prostate cancer, AR antagonists have been used clinically for the treatment of prostate cancer [5,6,7]. However, AR antagonists bearing a steroid core structure have undesirable side effects due to their cross-activity with other steroid hormone receptors. Consequently, nonsteroidal AR antagonists have been developed, and some of them, such as flutamide (2a) [8] and bicalutamide (BIC, 3, Figure 1) [9,10], are in clinical use to treat prostate cancer. These first-generation nonsteroidal AR antagonists are effective for most prostate cancer patients, but castration-resistant prostate cancer (CRPC) often develops after a few years, due to the mutation of AR [11,12,13]. For example, T877A is the most common mutation of AR, and hydroxyflutamide (2b), an active metabolite of flutamide (2a), acts as an agonist toward T877A AR [14]. Bicalutamide (3) acts as antagonist toward T877A AR, but as an agonist toward W741C AR [15].
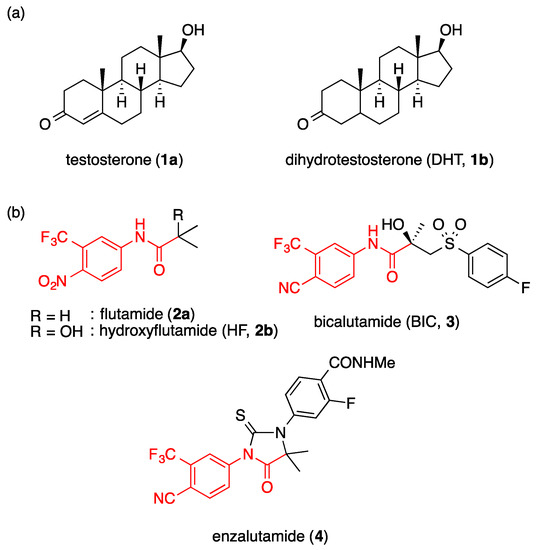
Figure 1.
Structures of (a) endogenous androgens and (b) typical nonsteroidal AR antagonists.
Second-generation AR antagonists effective against CRPC have been developed, such as enzalutamide (4) [16,17], apalutamide [18,19], and darolutamide [20]. Structurally, these AR antagonists have the same pharmacophore as the lead compounds, flutamide (2a) and bicalutamide (3), and they contain an anilide structure with electron-withdrawing functional groups on the phenyl ring (Figure 1). Recently, non-anilide type AR antagonists have also been reported, but variation in the chemotype is still limited [21].
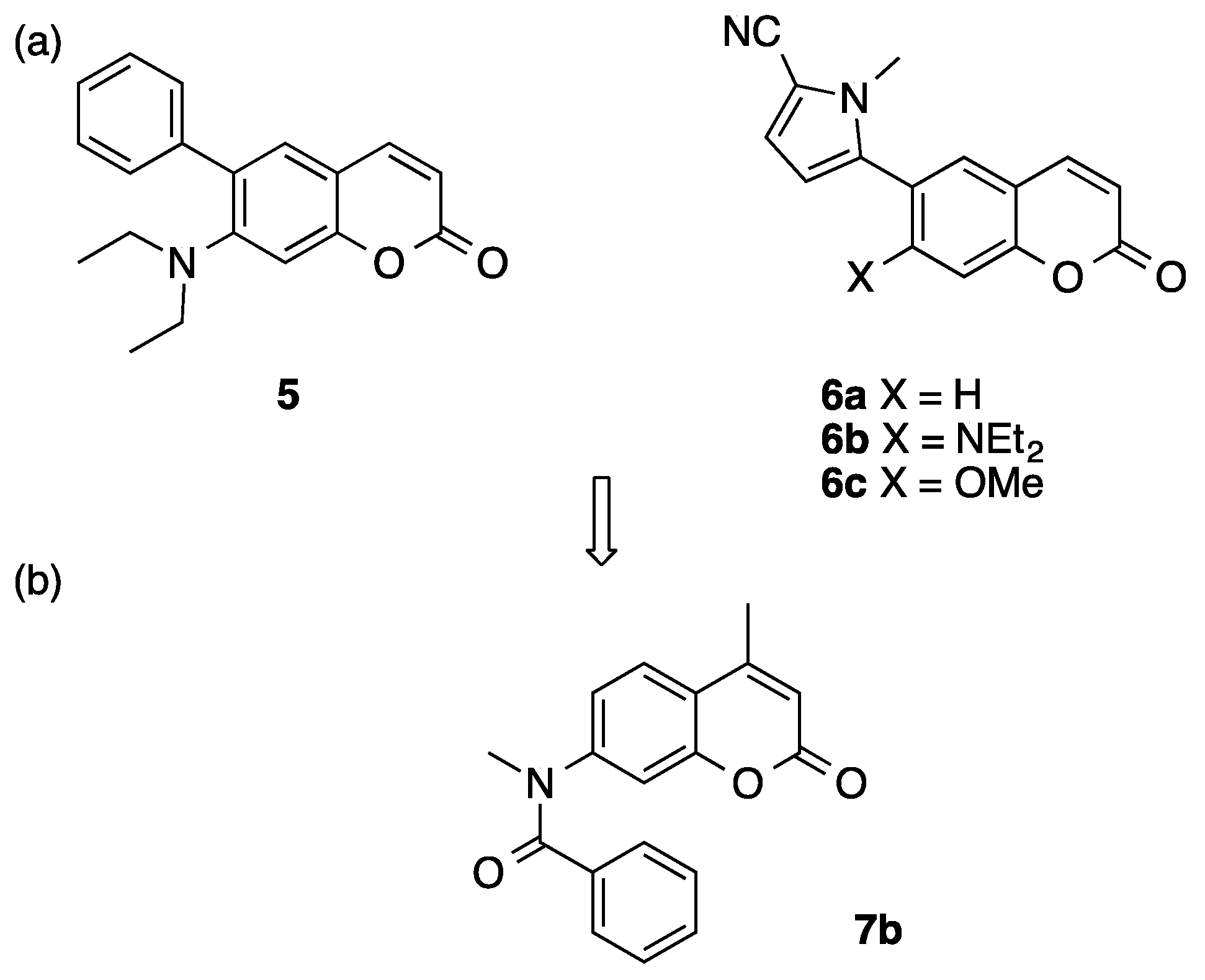
We have reported 6-arylcoumarin derivatives such as 5 and 6 as novel nonsteroidal progesterone receptor (PR) antagonists (Figure 2) [22,23]. In these compounds, the coumarin ring provides an alternative hydrophobic core structure to the sterane ring of steroidal derivatives or to the bicyclic anilide scaffold of known nonsteroidal PR ligands (i.e., benzoxadinone, and benzimidazolone derivatives bearing a nitrogen atom in the bicyclic heterocyclic skeleton). Among them, compounds 5, 6b, and 6c showed moderate AR-antagonistic activity in the androgen-dependent SC-3 cell growth assay [24]. These results suggested that the coumarin ring could mimic the anilide scaffold of AR antagonists. In addition, we reported 4-benzyl-1-(2H)-phthalazinone derivatives as AR antagonists with a novel core structure [25]. With this background, we decided to search for compounds with AR-antagonistic activity among our coumarin compound library, and we found that compound 7b bearing a 7-(N-methyl-benzoylamino) moiety showed AR-antagonistic activity with rather low PR-antagonistic activity (Figure 2). Compound 7b has a unique core structure that is different from those of conventional AR antagonists. In this study, we examined the structure–activity relationship of coumarinamide derivative 7b as an AR antagonist and identified a unique pharmacophore, consisting of an aromatic folded core structure that can interact with amino acid residues in the ligand-binding pocket of AR.
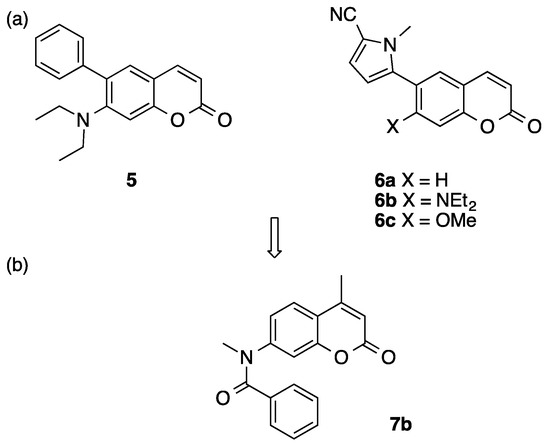
Figure 2.
Structures of (a) 6-arylcoumarin derivatives 5 and 6 with progesterone receptor (PR)-antagonistic activity and (b) androgen receptor (AR)-antagonistic coumarin 7.
2. Results and Discussion
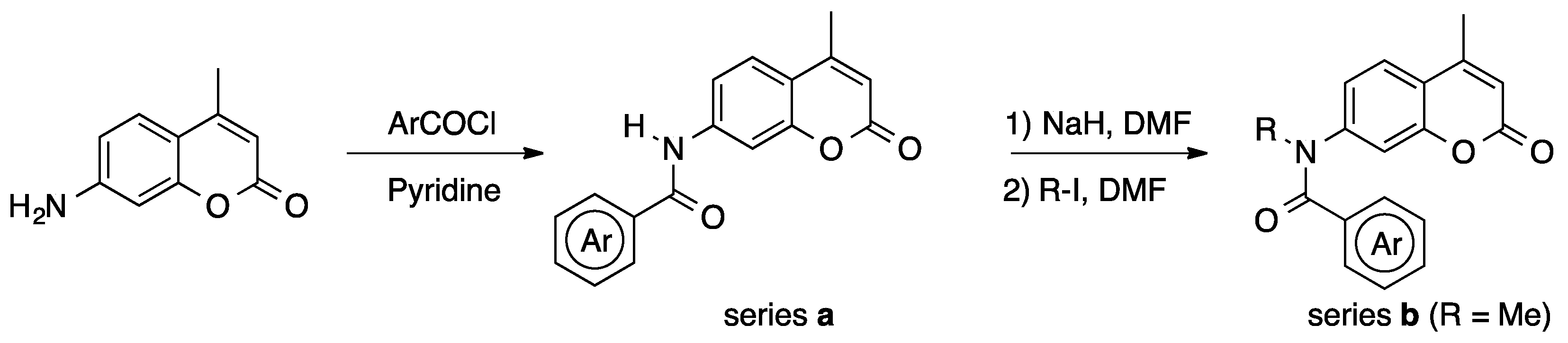
Various 4-methyl-7-(N-methyl-arylcarboxamido)coumarins with different substituents on the phenyl ring or amide nitrogen atom were synthesized from 7-amino-4-methylcoumarin by N-acylation, followed by N-alkylation (Scheme 1); their structures are shown in Table 1, Table 2 and Table 3. In order to clarify the electronic and steric properties of the amide bond, the biological activities of the derivatives bearing a secondary amide bond (series a) were also examined.
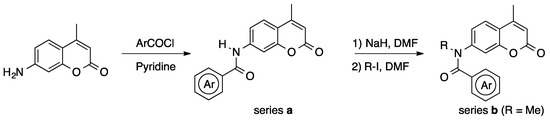
Scheme 1.
Synthesis of coumarinamides.

Table 1.
AR-antagonistic activity of coumarinamide derivatives in SC-3 cell assay a.

Table 2.
Effect of N-substituents of coumarinamide derivatives on AR-antagonistic activity in SC-3 cell assay a.

Table 3.
AR-antagonistic activity of coumarinamide derivatives in SC-3 cell assay a.
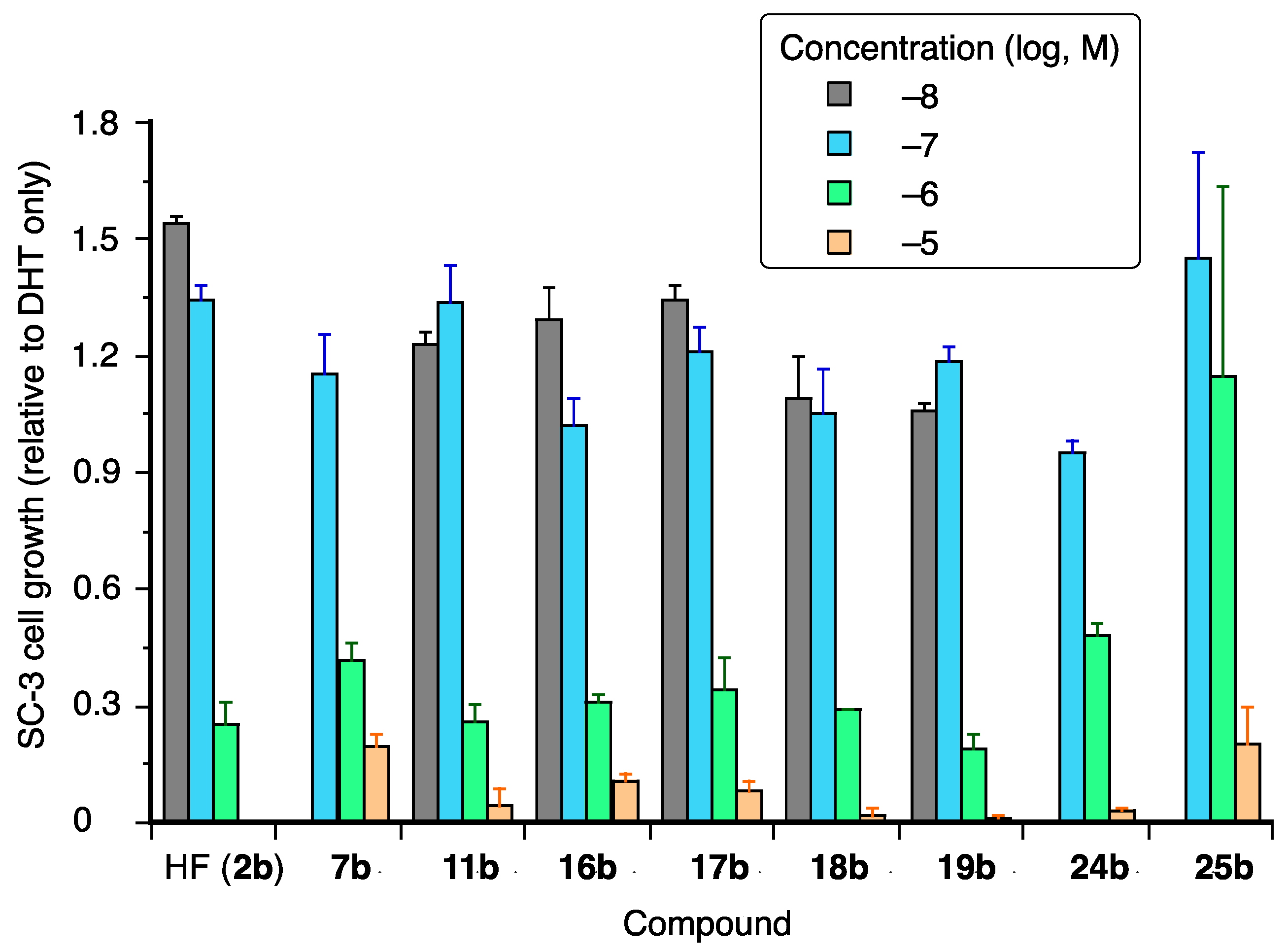
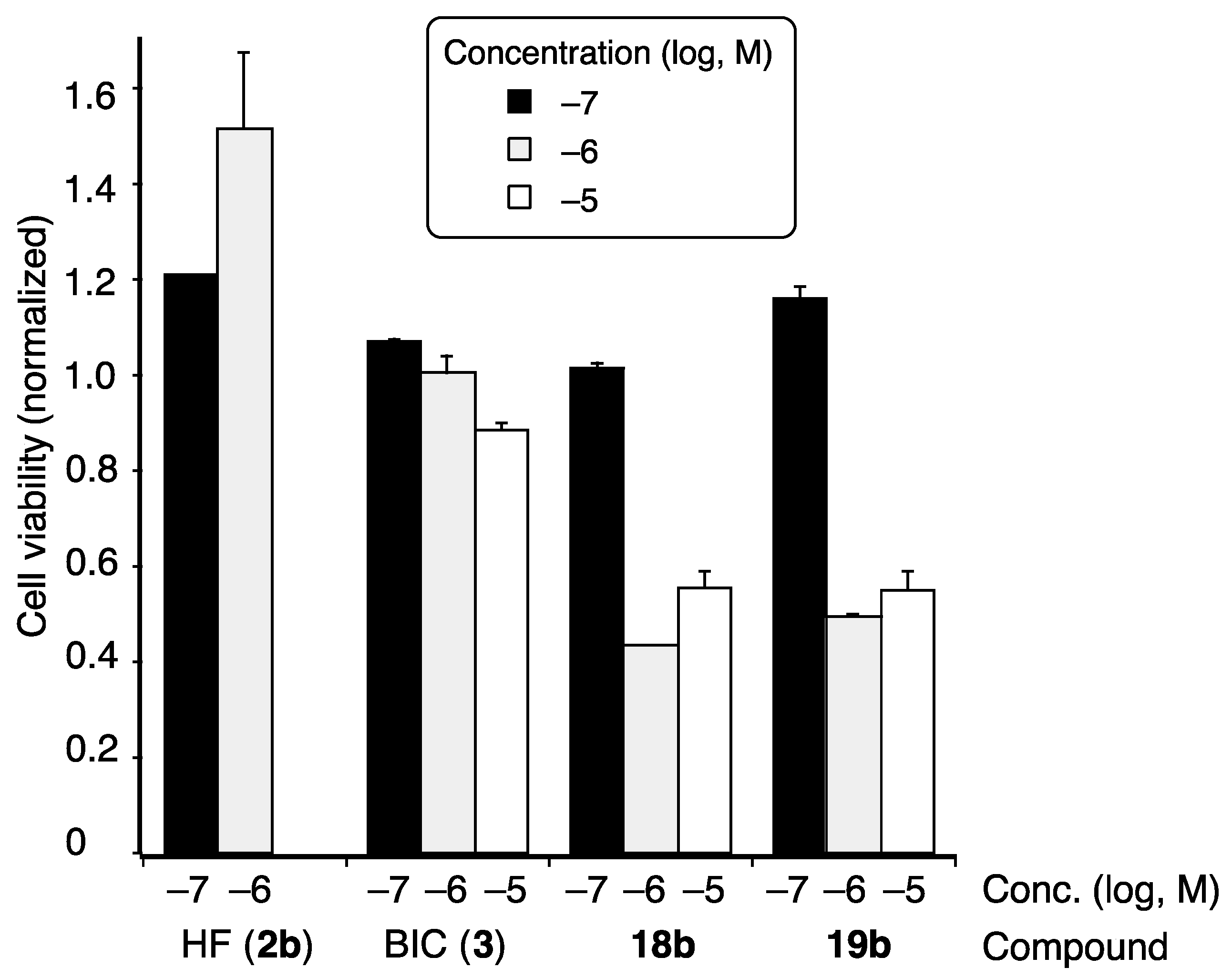
AR-agonistic and antagonistic activities of the synthesized coumarinamide derivatives were evaluated in terms of growth-inhibitory activity toward SC-3 cells bearing wild-type (wt) AR [24]. These cells show androgen-dependent cell proliferation, which is one of the typical and reliable assays for AR modulators [26]. None of the coumarinamide derivatives examined affected the proliferation of SC-3 alone at a concentration below 10−5 M (data not shown), which means they do not act as AR agonists. The antagonistic activity of the test compounds was examined in terms of their effect on 1 nM DHT-dependent proliferation of SC-3 cells. The IC50 values of test compounds are shown in Table 1, and the dose-dependency of the selected active compounds are shown in Figure 3. Compound 7b, the lead compound in this study, inhibited the DHT-induced proliferation of SC-3 cells with the IC50 value of 1.34 µM, and its activity is nearly one-fifth that of hydroxyflutamide (2b; IC50 = 0.29 μM), the positive control.
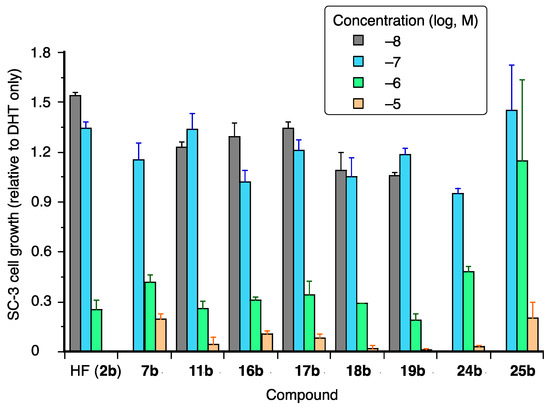
Figure 3.
Dose-dependent growth inhibition of selected coumarinamides in the presence of 1 nM DHT. The vertical scale is SC-3 cell growth, normalized to that of DHT-treated cells without the test compound, which is taken as 1.
First, we examined the substituent effect on the phenyl ring of 7b. Among the synthesized compounds, most of the coumarinamides with a secondary amide bond (series a) did not affect SC-3 cell growth, except for compounds 16a and 19a, which showed weak inhibitory activity only at 10 μM. On the other hand, some of the N-methylated compounds (series b) dose-dependently inhibited the DHT-induced proliferation of SC-3 cells. The introduction of a substituent at the para position of the phenyl group was not effective, and only compound 11b bearing a small fluorine atom exhibited inhibitory activity, being more potent than the lead compound without a substituent on the phenyl ring, and comparable in potency to hydroxyflutamide (2b). The introduction of a substituent at the ortho position was also ineffective, while compounds bearing a meta-substituent showed potent inhibitory activity. The introduction of a meta-cyano group, which is an electron-withdrawing group common to the structures of bicalutamide (3) and enzalutamide (4), decreased the activity compared to the lead compound 7b. The introduction of a halogen atom or halogen-containing substituent at the meta position increased the activity, and compounds 16b, 17b, and 18b showed more potent activity than 7b. Among the compounds with a meta-substituent, compound 19b with the m-methyl group was most active in SC-3 assay, showing an IC50 value of 0.49 μM. Introduction of a second meta-substituent was not effective, and compounds 24b and 25b with m,m’-dichloro or m,m’-dimethyl groups, respectively, are slightly less active than the corresponding monosubstituted compounds 18b and 19b.
Secondly, the effect of N-substituents was examined for the selected compounds 18b and 19b (Table 2). In both cases, as the N-substituent became larger, the inhibitory activity decreased. Thus, the c and d compounds with a N-ethyl or N-n-propyl group showed moderate inhibitory activity, while the e and f compounds with the N-substituent containing an aromatic ring were less active.
Thirdly, we replaced the phenyl group of compound 7b with other ring structures (Table 3). Compound 26b with a saturated cyclic alkyl group instead of the phenyl ring was inactive, which was probably due to the bulkiness of the nonplanar cyclohexyl ring. Interestingly, compound 27b with a 1-naphthyl group is more potent than the lead compound 7b, while compound 28b with a 2-naphthyl group, an isomer of 27, exhibited weaker inhibitory activity. Thus, the orientation of the naphthyl group is important for the activity. The derivatives of 7b with para-substituents were almost inactive (Table 1), although the para position of compound 28b could be regarded as substituted by a benzo group. Replacement of the phenyl ring of 7b with the basic pyridine ring diminished the activity, while replacement with 5-membered heterocyclic aromatics such as furyl and thiophenyl groups increased the activity. Compound 31b had potent inhibitory activity with the IC50 value of 0.50 µM, which was comparable to that of hydroxyflutamide (2b).
Next, we examined the activity of the selected compounds 18b and 19b toward LNCaP cells bearing mutated T877A AR (Figure 4) [27,28]. Bicalutamide (3) inhibited the proliferation of LNCaP cells, whereas hydroxyflutamide (2b) did not. Compounds 18b and 19b inhibited the DHT-dependent cell growth of LNCaP cells, and their inhibitory activities were higher than that of bicalutamide (3). Thus, coumarinamide derivatives can act as AR antagonists toward mutated T877A AR.
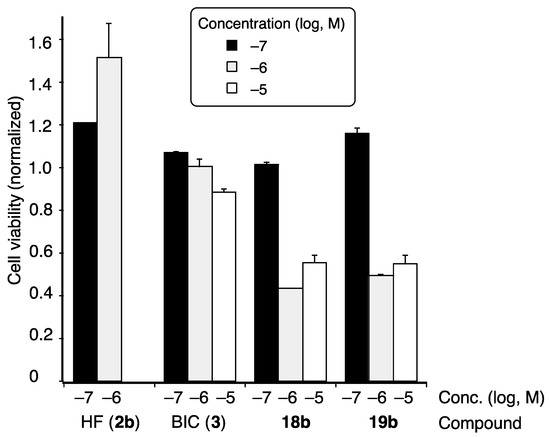
Figure 4.
Inhibitory activities of selected coumarinamide derivatives on the proliferation of LNCaP cells with T877A AR. Cell viability was determined by the WST method and normalized to that of vehicle-treated cells, taken as 1.
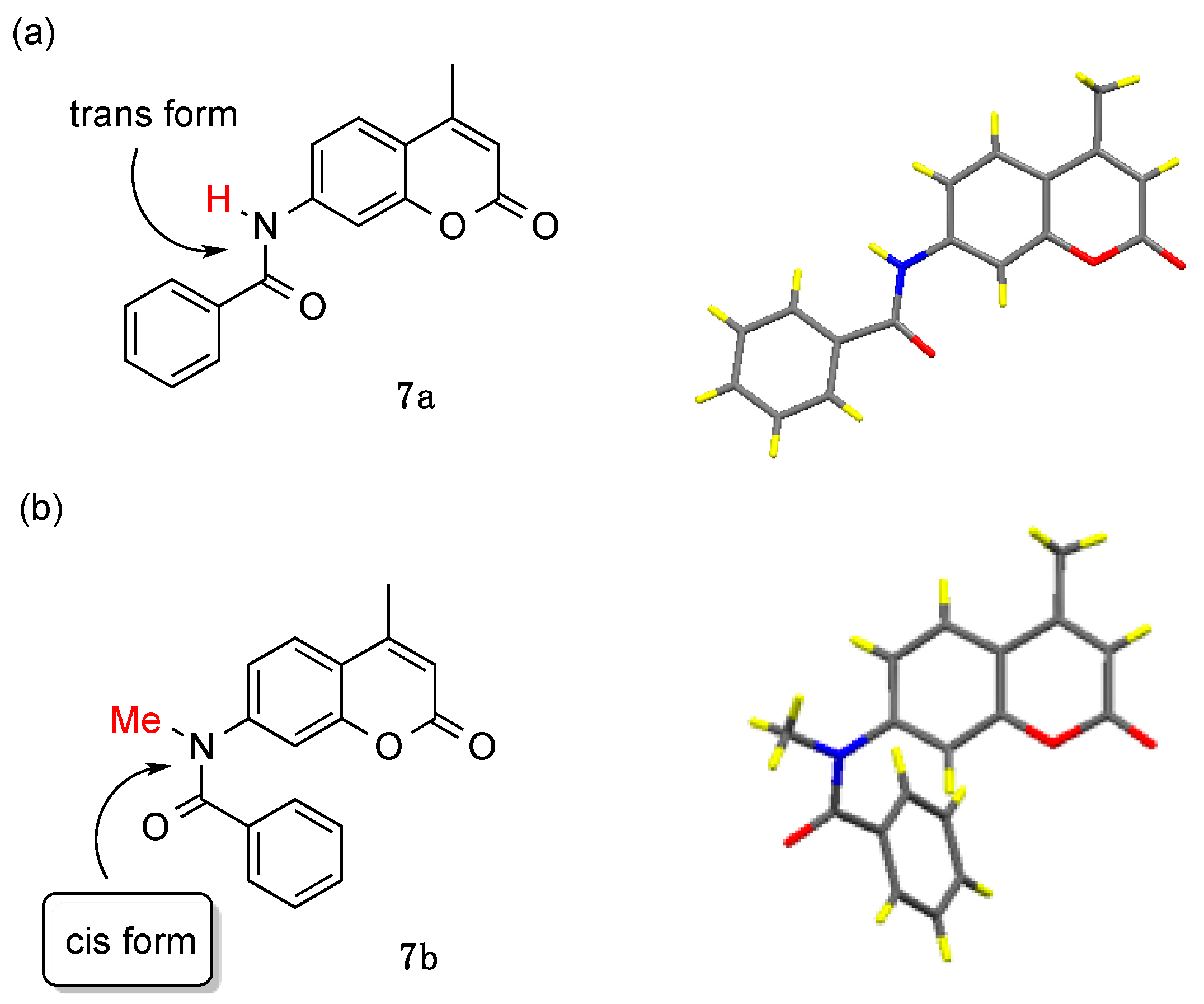
As discussed above, the coumarinamide derivatives (series a) with a secondary amide bond (R = H) showed little or no activity, while their N-methylated derivatives (series b) exhibited potent AR-antagonistic activity. We have previously reported on the unique conformational properties of aromatic amide compounds [28,29]. Thus, secondary amides such as benzanilide existed in trans form, while their N-methylated amides exist in cis form in the crystal and predominantly in cis form in solution. The cis conformational preference is a general property of N-methylated amides, including those bearing N-heterocyclic aromatics such as pyridine, pyrrole, and imidazole. Therefore, we assumed that the difference in AR-antagonistic activity between secondary and N-methylated compounds would result from the difference in their conformational properties. Then, we examined the crystal structures of compounds 7a and 7b (Figure 5). As expected, compound 7a exists in the extended structure with a trans-amide form, in which the phenyl and coumarin rings are located on opposite sides of the molecule. On the other hand, compound 7b exists in a folded structure with cis-amide, in which the two aromatic rings take a face-to-face position. Comparison of the 1H NMR chemical shifts of 7a and 7b indicated that these compounds exist in solution in conformations similar to those found in the crystal. Thus, compounds 7b shows aromatic proton signals at a higher field than compound 7a in the 1H NMR spectra, due to the anisotropic effects between two aromatic rings. A similar tendency was observed with the other coumarinamide derivatives. These results indicate that the folded structure with a cis-amide bond is important for AR-antagonistic activity.
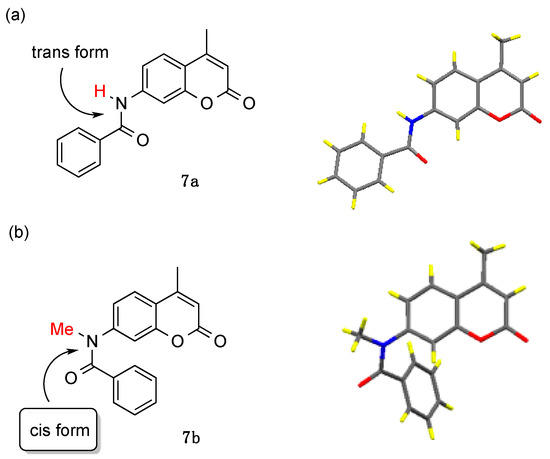
Figure 5.
Crystal structures of compounds (a) 7a and (b) 7b.
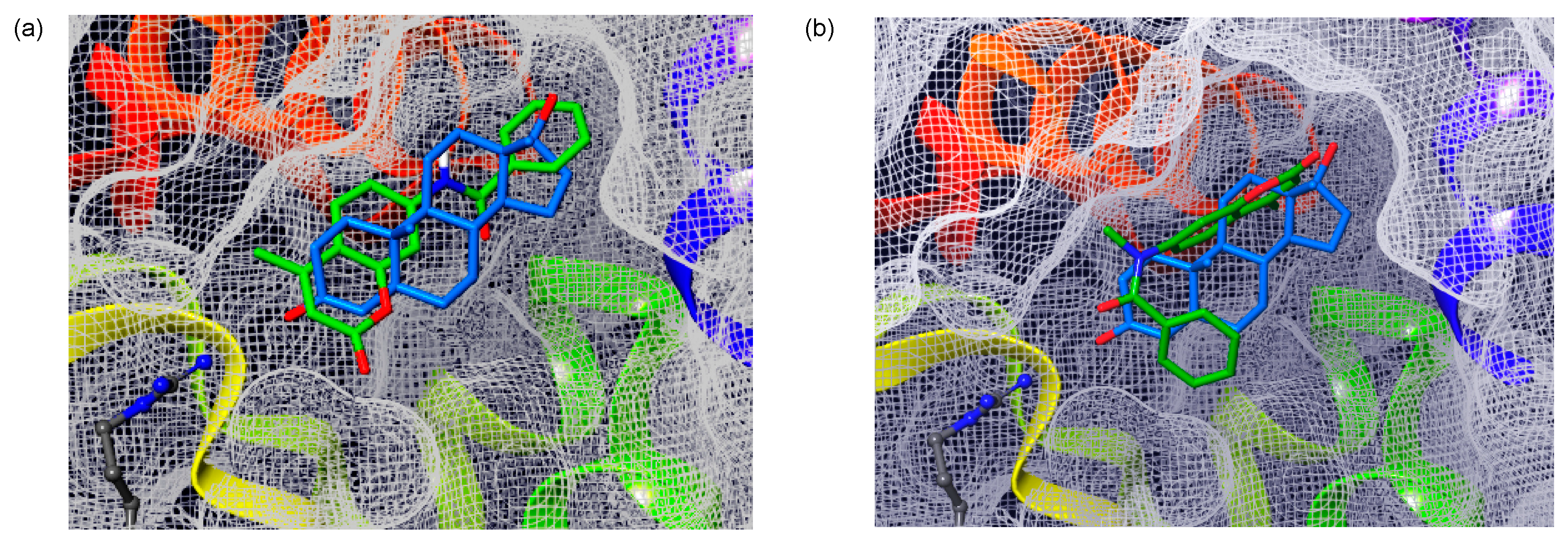
In order to analyze the binding features of coumarinamide derivatives with AR, we conducted a docking study of compounds 7a and 7b with the AR ligand-binding domain (LBD) by using the AutoDock program. For the docking, we used the crystal structure of the AR LBD in the complex with DHT (1b) (PDB ID: 2AMA) and the crystal structures of 7a and 7b, as shown in Figure 6. Notably, 7b with the folded structure can dock with the AR LBD in a similar position to that of DHT (1b). In this binding structure, the carbonyl group of the cis-amide bond of compound 7b lies in the same direction as that of DHT (1b), and the phenyl group of 7b is placed in the hydrophobic region. On the other hand, compound 7a with an extended structure can bind to the ligand-binding pocket, but there is no strong interaction with the surrounding amino acid residues. The binding energy of 7a with AR LBD is –7.55 kcal/mol, which is lower in energy than that of 7b (−9.04 kcal/mol). Thus, the docking study also supports the significance of the folded coumarin amide structure for AR-antagonistic activity.
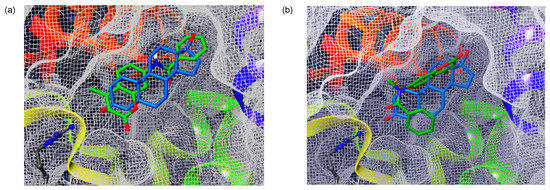
Figure 6.
Docking study of coumarin derivatives (a) 7a and (b) 7b with the AR ligand-binding domain (LBD) (PDB ID: 2AMA) using AutoDock 4.2, superposed with the structure of DHT (1b) in the crystal. The CPK colored residue is Arg752. Coumarin derivatives and DHT (1b) are shown in green and blue, respectively.
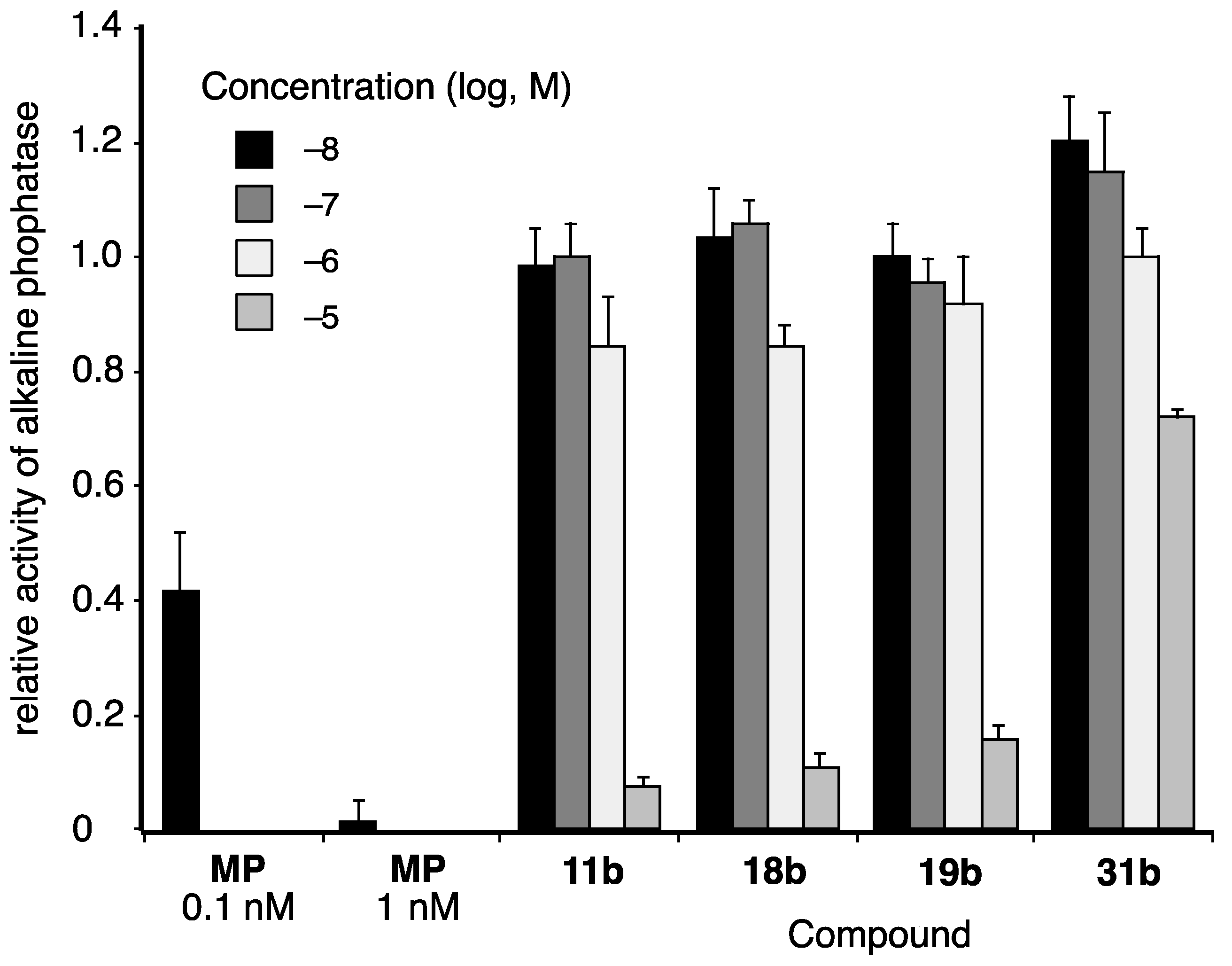
Finally, we examined the activity of selected coumarinamide derivatives toward progesterone receptor (PR), since some coumarin derivatives act as PR antagonists. The PR-antagonistic activities were evaluated by means of alkaline phosphatase (AP) assay using the T-47D human breast carcinoma cell line (Figure 7) [30]. All the selected coumarinamide derivatives examined showed PR-antagonistic activity only at 10 µM, and they did not affect the progesterone (1 nM)-induced AP activity at concentrations below 1 µM. In particular, the PR-antagonistic activity of compound 31b was very weak, and the compound only slightly inhibited AP activity, even at 10 µM. Thus, the coumarinamide derivatives show selectivity as AR antagonists over PR.
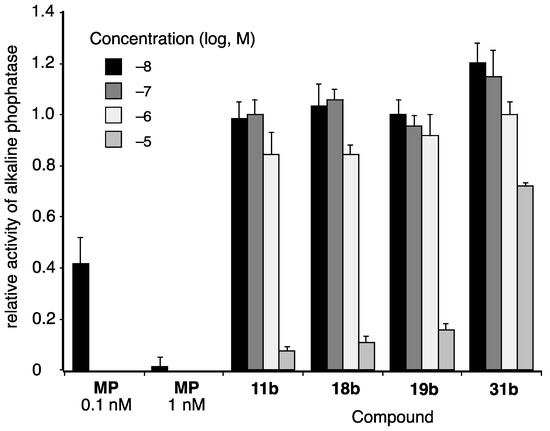
Figure 7.
PR-antagonistic activities of selected coumarinamide derivatives, evaluated by means of alkaline phosphatase (AP) assay using T-47D cells. The vertical axis is the activity of alkaline phosphatase in the presence of 1 nM progesterone and test compound, normalized to progesterone alone as 1. MP: mifepristone (PR antagonist).
3. Materials and Methods
3.1. Chemistry
3.1.1. General
All reagents were purchased from Sigma-Aldrich Chemical Co. (Tokyo, Japan), Tokyo Kasei Kogyo Co. (Tokyo, Japan), Wako Pure Chemical Industries (Tokyo, Japan), or Kanto Kagaku Co., Inc. (Tokyo, Japan). Silica gel for column chromatography was purchased from Kanto Kagaku Co., Inc (Tokyo, Japan). 1H and 13C NMR spectra were recorded on a JEOL ECA 600 (Tokyo, Japan), or Bruker 600 spectrometer (Tokyo, Japan). Mass spectral data were obtained on a Bruker Daltonics microTOF-2focus (Tokyo, Japan), Thermo Scientific Q-Exactive or Waters Q-TOF Premier in the positive ion detection mode (Tokyo, Japan).
3.1.2. Synthesis of 23a (General Procedure for Coumarinamides with Secondary Amide Bond)
ο-Anisoyl chloride (120 mg, 0.70 mmol) was added to a solution of 7-amino-4-methylcoumarin (101 mg, 0.58 mmol) in pyridine (3 mL), and the mixture was stirred at 50 °C for 2 h. The mixture was poured into 2 M HCl (30 mL), and the precipitates were collected, washed with water, and a mixture of chloroform and methanol to give 23a (171 mg, 96%).
3.1.3. Synthesis of 23b (General Procedure for N-Alkylated Coumarinamides)
Sodium hydride (24 mg, 0.60 mmol) was washed with n-hexane. A solution of 23a (61 mg, 0.20 mmol) in DMF (1 mL) was added to a suspension of sodium hydride in DMF (2 mL). After 20 min, iodomethane (83 mg, 0.58 mmol) was added to the mixture. After 5 h, the reaction mixture was poured into water and extracted with ethyl acetate. The organic layer was washed with brine, dried over magnesium sulfate, and concentrated. The residue was purified by silica gel column chromatography (ethyl acetate:hexane = 1:1) to give 23b (21.0 g, 32%).
3.2. SC-3 Growth Inhibition Assay
SC-3 cell growth inhibition assay was performed as the previous report [24,26]. SC-3 cells were seeded in 96-well plates at a concentration of 2000 cells per well in 100 μL of MEMα medium supplemented with 2% FBS (fetal bovine serum), and the plates were incubated for 24 h at 37 °C under 5% CO2. Then, 10 μL of medium was removed from each well and replaced with 10 μL of drug solution supplemented with serial dilutions of test compound or DMSO as a dilution control in the presence of 1 nM DHT, and the plates were incubated for 3 days. The cell number was determined using a Cell Counting Kit-8 (Dojindo, Kumamoto, Japan) according to the manufacturer’s instruction. This parameter is proportional to the number of living cells in the culture. IC50 (Table 1, Table 2 and Table 3) is the concentration of test compound that reduces DHT-induced cell growth to 50% of the control. All experiments were performed in triplicate or more.
3.3. LNCaP Cell Proliferation Assay
LNCaP cell growth inhibition assay was performed as the previous report [27,28,31]. Briefly, LNCaP cells were seeded in 96-well plates at a concentration of 2000 cells per well in 100 μL of RPMI-1640 medium supplemented with 2% charcoal-stripped FBS, and the plates were incubated for 24 h at 37 °C under 5% CO2. Then, 10 μL of medium was removed from each well and replaced with 10 μL of drug solution supplemented with serial dilutions of test compound or DMSO as a dilution control in the presence or absence of 10 nM DHT. Cells were incubated for 6 days, and half of the medium was removed and replaced once after 3 days with medium containing test compound or DMSO as a dilution control. At the end of the incubation, proliferation was evaluated using a Cell Counting Kit-8 (Dojindo) according to the manufacturer’s instruction. This parameter is proportional to the number of living cells in the culture. All experiments were performed in triplicate or more.
3.4. Alkaline Phosphatase Assay Using T-47D Cells
Alkaline phosphatase assay was performed based on the previous report [22,32]. Briefly, T-47D cells were plated in 96-well plates at 10,000 cell per well and incubated at 37 °C under 5% CO2 for 24 h. Then, 10 μL of medium was removed from each well and replaced with 10 μL of drug solution supplemented with serial dilutions of test compound or DMSO as a dilution control in the presence or absence of 1 nM progesterone, and incubation was continued for 24 h. Then, the medium was aspirated, and the cells were fixed with 100 μL of 1.8% formalin in PBS. The fixed cells were washed with PBS, and 75 μL of assay buffer (1 mg/mL p-nitrophenol phosphate in diethanolamine water solution, pH 9.0, 2 mM MgCl2) was added. The mixture was incubated at room temperature with shielding from light for 2 h, and then the reaction was terminated by the addition of 100 μL of NaOH. The absorbance at 405 nm was measured with a microplate reader.
4. Conclusions
We synthesized a series of 4-methyl-7-(N-alkyl-arylcarboxamido)coumarins as AR antagonist candidates, using compound 7b as a lead compound. The AR-antagonistic activity of the synthesized coumarinamide derivatives was examined in terms of growth-inhibitory activity toward SC-3 cells. Coumarinamides with a secondary amide bond were inactive, but their N-alkylated derivatives exhibited AR-antagonistic activity. Among them, compounds 19b and 31b showed more potent AR-antagonistic activity than the lead compound 7b, and they were comparable in potency to hydroxyflutamide (2b). Conformational analysis and docking study revealed that the folded structure of coumarinamides associated with the cis-amide bond is important for binding to AR. The selected coumarinamide derivatives showed AR-antagonistic activity toward LNCaP cells with T877A AR and had weak PR-antagonistic activity. The detailed action mechanism of novel coumarinamides as AR modulators including AR-binding ability and transcriptional regulation should be elucidated in the future works, as the folded coumarinamide is a different pharmacophore from those of conventional AR antagonists, and these compounds may be promising candidates for the treatment of prostate cancer.
Supplementary Materials
The following are available online at https://www.mdpi.com/1422-0067/21/15/5584/s1. Compound Data: Development of novel androgen antagonistic coumarinamides bearing unique aromatic folded structure.
Author Contributions
H.K. (Hitomi Koga), M.N., M.K. synthesized the compounds. S.F., S.M., M.I.-Y., E.K. examined the biological activities. H.K. (Hiroyuki Kagechika) and A.T. planed and supervised the scientific works, and wrote the manuscript. All authors have read and edited the manuscript, and agreed to the published version of the manuscript.
Funding
This work was partly supported by JSPS KAKENHI Grant No. 20K06963 (to A.T.), 17H03887 and 19K22486 (to S.F.), JSPS Core-to-Core Program, A. Advanced Research Networks, and Japan Agency for Medical Research and Development (AMED) under Grant Number JP20am0101098 (Platform Project for Supporting Drug Discovery and Life Science Research, BINDS). A part of this research involved a Cooperative Research Project of the Research Center for Biomedical Engineering. A.T. thanks The Naito Foundation, the Cosmetology Research Foundation, and the Tokyo Biochemical Research Foundation.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Abbreviations
| AR | androgen receptor |
| DHT | dihydrotestosterone |
| BIC | bicalutamide |
| CRPC | castration-resistant prostate cancer |
| PR | progesterone receptor |
| NMR | ligand-binding domain |
| LBD | keratinocyte growth factor |
References
- Gao, W.; Bohl, C.E.; Dalton, J.T. Chemistry and structural biology of androgen receptor. Chem. Rev 2005, 105, 3352–3370. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.Z.; Wardell, S.E.; Burnstein, K.L.; DeFranco, D.; Fuller, P.J.; Giguère, V.; Hochberg, R.B.; McKay, L.; Renoir, J.-M.; Weigel, N.L.; et al. International Union of Pharmacology. LXV. The Pharmacology and Classification of the Nuclear Receptor Superfamily: Glucocorticoid, Mineralocorticoid, Progesterone, and Androgen Receptors. Pharmacol. Rev. 2006, 58, 782–797. [Google Scholar] [CrossRef] [PubMed]
- Mooradian, A.D.; Morley, J.E.; Korenman, S.G. Biological Actions of Androgens. Endocr. Rev. 1987, 8, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Bagatell, C.J.; Bremner, W.J. Androgens in Men—Uses and Abuses. N. Engl. J. Med. 1996, 334, 707–715. [Google Scholar] [CrossRef]
- Heinlein, C.A.; Chang, C. Androgen receptor in prostate cancer. Endocr. Rev. 2004, 25, 276–308. [Google Scholar] [CrossRef]
- Chen, Y.; Sawyers, C.L.; I Scher, H. Targeting the androgen receptor pathway in prostate cancer. Curr. Opin. Pharmacol. 2008, 8, 440–448. [Google Scholar] [CrossRef]
- Taplin, M.-E. Androgen receptor: Role and novel therapeutic prospects in prostate cancer. Expert Rev. Anticancer. Ther. 2008, 8, 1495–1508. [Google Scholar] [CrossRef]
- Neri, R.; Florance, K.; Koziol, P.; Van Cleave, S. A Biological Profile of a Nonsteroidal Antiandrogen, SCH 13521 (4′–Nitro–3′–Trifluoromethylisobutyranilide). Endocrinology 1972, 91, 427–437. [Google Scholar] [CrossRef]
- Schellhammer, P. An update on bicalutamide in the treatment of prostate cancer. Expert Opin. Investig. Drugs 1999, 8, 849–860. [Google Scholar] [CrossRef]
- Fradet, Y. Bicalutamide (Casodex®) in the treatment of prostate cancer. Expert Rev. Anticancer. Ther. 2004, 4, 37–48. [Google Scholar] [CrossRef]
- Watson, P.A.; Arora, V.K.; Sawyers, C.L. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat. Rev. Cancer 2015, 15, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Yap, T.A.; Smith, A.D.; Ferraldeschi, R.; Al-Lazikani, B.; Workman, P.; De Bono, J.S. Drug discovery in advanced prostate cancer: Translating biology into therapy. Nat. Rev. Drug Discov. 2016, 15, 699–718. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, C.M.; Gao, A.C. Drug resistance in castration resistant prostate cancer: Resistance mechanisms and emerging treatment strategies. Am. J. Clin. Exp. Urol. 2015, 3, 64–76. [Google Scholar] [PubMed]
- Steketee, K.; Timmerman, L.; Der Made, A.C.Z.-V.; Doesburg, P.; Brinkmann, A.O.; Trapman, J. Broadened ligand responsiveness of androgen receptor mutants obtained by random amino acid substitution of H874 and mutation hot spot T877 in prostate cancer. Int. J. Cancer 2002, 100, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Bohl, C.E.; Gao, W.; Miller, D.D.; Bell, C.E.; Dalton, J.T. Structural basis for antagonism and resistance of bicalutamide in prostate cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 6201–6206. [Google Scholar] [CrossRef]
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; Wongvipat, J.; Smith-Jones, P.M.; Yoo, N.; Kwon, A.; et al. Development of a Second-Generation Antiandrogen for Treatment of Advanced Prostate Cancer. Science 2009, 324, 787–790. [Google Scholar] [CrossRef]
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.-E.; Sternberg, C.N.; Miller, K.; De Wit, R.; Mülders, P.; Chi, K.N.; Shore, N.D.; et al. Increased Survival with Enzalutamide in Prostate Cancer after Chemotherapy. N. Engl. J. Med. 2012, 367, 1187–1197. [Google Scholar] [CrossRef]
- Clegg, N.J.; Wongvipat, J.; Joseph, J.D.; Tran, C.; Ouk, S.; Dilhas, A.; Chen, Y.; Grillot, K.; Bischoff, E.D.; Cai, L.; et al. ARN-509: A novel antiandrogen for prostate cancer treatment. Cancer Res. 2012, 72, 1494–1503. [Google Scholar] [CrossRef]
- Smith, M.R.; Saad, F.; Chowdhury, S.; Oudard, S.; Hadaschik, B.; Graff, J.N.; Olmos, D.; Mainwaring, P.N.; Lee, J.Y.; Uemura, H.; et al. Apalutamide Treatment and Metastasis-free Survival in Prostate Cancer. N. Engl. J. Med. 2018, 378, 1408–1418. [Google Scholar] [CrossRef]
- Fizazi, K.; Albigès, L.; Loriot, Y.; Massard, C. ODM-201: A new-generation androgen receptor inhibitor in castration-resistant prostate cancer. Expert Rev. Anticancer. Ther. 2015, 15, 1007–1017. [Google Scholar] [CrossRef]
- Fujii, S.; Kagechika, H. Androgen receptor modulators: A review of recent patents and reports (2012-2018). Expert Opin. Ther. Patents 2019, 29, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Sakai, H.; Hirano, T.; Mori, S.; Fujii, S.; Masuno, H.; Kinoshita, M.; Kagechika, H.; Tanatani, A. 6-Arylcoumarins as Novel Nonsteroidal Type Progesterone Antagonists: An Example with Receptor-Binding-Dependent Fluorescence. J. Med. Chem. 2011, 54, 7055–7065. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; Negishi, M.; Sakai, H.; Hirano, T.; Mori, S.; Fujii, S.; Kagechika, H.; Tanatani, A. Development of 6-arylcoumarins as nonsteroidal progesterone antagonists. Structure–activity relationships and fluorescence properties. Bioorganic Med. Chem. 2016, 24, 5602–5610. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, D.; Nakamura, N.; Nishizawa, Y.; Uchida, N.; Noguchi, S.; Matsumoto, K.; Sato, B. Inhibitory and stimulatory effects of glucocorticoid on androgen- induced growth of murine shionogi carcinoma 115 in vivo and in cell culture. Cancer Res. 1987, 47, 6560–6564. [Google Scholar]
- Inoue, K.; Urushibara, K.; Kanai, M.; Yura, K.; Fujii, S.; Ishigami-Yuasa, M.; Hashimoto, Y.; Mori, S.; Kawachi, E.; Matsumura, M.; et al. Design and synthesis of 4-benzyl-1-(2H)-phthalazinone derivatives as novel androgen receptor antagonists. Eur. J. Med. Chem. 2015, 102, 310–319. [Google Scholar] [CrossRef]
- Veldscholte, J.; Berrevoets, C.A.; Brinkmann, A.O.; Grootegoed, J.A.; Mulder, E. Anti-androgens and the mutated androgen receptor of LNCaP cells: Differential effects on binding affinity, heat-shock protein interaction, and transcription activation. Biochemistry 1992, 31, 2393–2399. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, B.; Geng, G.; Wu, J.H. Study of the impact of the T877A mutation on ligand-induced helix-12 positioning of the androgen receptor resulted in design and synthesis of novel antiandrogens. Proteins Struct. Funct. Bioinform. 2009, 78, 623–637. [Google Scholar] [CrossRef] [PubMed]
- Kagechika, H.; Himi, T.; Kawachi, E.; Shudo, K. Retinobenzoic acids. 4. Conformation of aromatic amides with retinoidal activity. Importance of trans-amide structure for the activity. J. Med. Chem. 1989, 32, 2292–2296. [Google Scholar] [CrossRef] [PubMed]
- Tanatani, A.; Yokoyama, A.; Azumaya, I.; Takakura, Y.; Mitsui, C.; Shiro, M.; Uchiyama, M.; Muranaka, A.; Kobayashi, N.; Yokozawa, T. Helical Structures ofN-Alkylated Poly(p-benzamide)s. J. Am. Chem. Soc. 2005, 127, 8553–8561. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, D.; Albertini, A.; Zava, D. Progestin regulation of alkaline phosphatase in the human breast cancer cell line T47D. Cancer Res. 1991, 51, 4470–4475. [Google Scholar]
- Yamada, A.; Fujii, S.; Mori, S.; Kagechika, H. Design and Synthesis of 4-(4-Benzoylaminophenoxy)phenol Derivatives As Androgen Receptor Antagonists. ACS Med. Chem. Lett. 2013, 4, 937–941. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ohta, K.; Goto, T.; Fujii, S.; Suzuki, T.; Ohta, S.; Endo, Y. Design and synthesis of carborane-containing androgen receptor (AR) antagonist bearing a pyridine ring. Bioorg. Med. Chem. 2008, 16, 8022–8028. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).