1. Introduction
The increasing levels of air pollution and environmental degradation in urban areas pose serious challenges to sustainable development, human health, and the longevity of civil infrastructure [
1,
2,
3]. Urban surfaces such as pavements, façades, and concrete structures tend to accumulate particulate matter, organic pollutants, and microorganisms, resulting not only in aesthetic deterioration but also in accelerated chemical and physical degradation [
2,
4,
5]. Addressing these challenges requires innovative multifunctional materials that combine structural performance with environmental self-cleaning and pollutant degradation capabilities [
6,
7,
8].
In this context, photocatalytic materials—particularly titanium dioxide (TiO
2)—have emerged as promising candidates due to their stability, non-toxicity, abundance, and strong oxidative potential under light irradiation [
1,
9,
10,
11]. When exposed to ultraviolet (UV) or visible light, TiO
2 generates reactive oxygen species (ROS) capable of decomposing a wide range of organic and inorganic contaminants, including volatile organic compounds (VOCs), nitrogen oxides (NO
x), and hydrocarbons [
4,
10,
11,
12,
13]. This photocatalytic mechanism has been successfully applied in environmental cleanup, air purification, and wastewater treatment processes [
7,
14,
15,
16,
17]. In addition, TiO
2-based materials have shown potential in energy-related applications, such as hydrogen production from organic feedstocks, highlighting their versatility in pollutant degradation and resource transformation [
18]. Recent advancements in heterogeneous photocatalysis have significantly improved the design and efficiency of TiO
2-based materials. Modifications such as metal and non-metal doping (e.g., nitrogen, sulfur, carbon), surface sensitization, and hybrid composite formation have extended TiO
2 activity into the visible light spectrum, which is crucial for real-world applications where UV radiation is limited [
9,
14,
19,
20,
21]. Moreover, innovative deposition techniques such as sol–gel processing, spray and dip coating, plasma treatment, and nanostructured film formation have enhanced TiO
2 adhesion, dispersion, and long-term durability on construction surfaces [
22,
23,
24,
25]. These developments have positioned TiO
2 as a cornerstone material in the emerging field of photocatalytic construction composites, where functionality extends beyond mechanical strength to include environmental remediation and sustainability [
12,
26,
27,
28,
29].
Concrete, as the most widely used construction material worldwide, provides an ideal substrate for TiO
2 integration due to its abundance, surface exposure, and structural versatility [
19,
23,
30]. However, its inherently porous microstructure promotes the accumulation of dust, smog particles, and organic pollutants, leading to discoloration, surface weakening, and reduced service life [
24,
31,
32]. The incorporation of TiO
2 into concrete—either as a surface coating or a bulk additive—offers a viable solution to mitigate these effects. Upon illumination, TiO
2 promotes photocatalytic degradation of pollutants, thus reducing maintenance needs, improving hydrophilicity and self-cleaning properties, and extending the lifespan of concrete elements [
2,
7,
12,
33]. Furthermore, TiO
2-based coatings have been shown to improve the depolluting efficiency of urban infrastructure by reducing atmospheric concentrations of NO
x, CO, and VOCs in both indoor and outdoor environments [
13,
18,
34,
35,
36].
Recent studies have expanded the functionality of TiO
2-modified construction materials. Nitrogen-doped TiO
2 composites have shown improved visible-light activity, enabling efficient photocatalytic performance even in shaded or low-light urban environments [
10,
14]. Hybrid systems such as TiO
2/SiO
2, Cu–TiO
2/SiO
2, and black TiO
2 have been developed to enhance light absorption, charge carrier separation, and overall photocatalytic stability [
24,
25]. Investigations by Khannyra et al. [
17] demonstrated that appropriately engineered TiO
2 coatings retain high self-cleaning efficiency and mechanical integrity even after long-term exposure to environmental stressors. Moreover, research by Kalinowski et al. [
33] and Jenim et al. [
12] highlighted the potential of nano-TiO
2-modified cementitious composites to achieve carbon-negative performance through simultaneous air purification and CO
2 reduction. These findings emphasize the dual environmental and structural benefits of TiO
2 integration in cementitious materials.
In addition to environmental reactivity, TiO
2 incorporation improves mechanical behavior and durability of cementitious composites. Studies indicate that optimal nano-TiO
2 dosages (1–6 wt.%) increase compressive and flexural strength, refine pore structure, and enhance resistance to freeze–thaw cycles and abrasion [
34,
35,
36]. When combined with recycled aggregates such as reclaimed asphalt pavement, CRT glass, or industrial by-products, TiO
2 improves interfacial bonding while preserving photocatalytic activity, contributing to more sustainable composite design [
29,
30,
32]. Recent research highlights the development of TiO
2 enhanced building materials for environmental purification [
37] and demonstrates the effectiveness of hydrophilic polymer dispersions that improve TiO
2 dispersion uniformity, resulting in enhanced surface activation and NOx degradation [
38], while comparative analyses of spray- and dip-coated layers underscore the importance of the deposition method for long-term self-cleaning performance [
39]. Additional advances include immobilization strategies for TiO
2 on reusable substrates and reactor systems that enhance photocatalytic efficiency and enable catalyst recovery [
40], as well as progress in visible-light-activated TiO
2 materials relevant to pollutant degradation in alkaline environments typical of cementitious systems [
41]. Based on these developments, the present study investigates TiO
2-coated concrete paving units intended for environmental remediation and long-term durability in urban conditions. The experimental program encompasses photocatalytic degradation of organic contaminants and NO
x [
6,
10,
14], surface self-cleaning ability [
17,
24], and changes in surface wettability associated with TiO
2 coating and dispersion mechanisms [
38,
39]. Measurements of water and oil absorption, contact angles, and photodegradation efficiency are conducted under natural and artificial illumination, while UV exposure, moisture cycling, and abrasion tests follow methodologies used in durability studies of photocatalytic building materials [
12,
19,
25]. These analyses provide a comprehensive understanding of the potential of TiO
2-modified concrete to enhance air quality, surface functionality, and material lifespan within urban infrastructure, contributing to the development of sustainable, multifunctional construction materials.
2. Materials and Methods
2.1. Materials and Chemicals
All reagents used in this study were of analytical grade and applied without further purification. Deionized water was employed for all solution preparations. Concrete paving blocks were fabricated using ordinary Portland cement (CEM I 42.5R), industrial sand (0–2 mm), and gravel (2–8 mm), following standard construction practice to ensure reproducibility and mechanical stability. The detailed composition of the concrete mixture is presented in
Table 1.
Concrete cubes with dimensions of 5 × 5 × 5 cm3 were cast in steel molds and cured under controlled laboratory conditions (18 ± 2 °C, relative humidity > 90%) for 14 days. The impregnation agent (Feda, Warsaw, Poland) was primarily composed of Tetrahydro-1,3,4,6-tetrakis(hydroxymethyl)imidazo[4,5-d]imidazole-2,5(1H,3H)-dione, stabilized with a mixture of 5-chloro-2-methyl-4-isothiazolin-3-one and 2-methyl-4-isothiazolin-3-one (3:1). Titanium dioxide (TiO2, anatase phase, <100 nm, Sigma-Aldrich, Saint Louis, MO, USA) was used as the photocatalyst.
The impregnation system was designed to utilize the four hydroxymethyl (–CH2OH) groups in Feda compound, which form hydrogen bonds with hydroxyl groups on the TiO2 surface. These interactions enhance the adhesion of nanoparticles to the concrete matrix and limit their potential leaching under aqueous conditions.
2.2. Preparation of TiO2-Coated Concrete Cubes
The preparation of TiO2-coated concrete cubes followed a multi-step procedure to ensure uniform coating and reproducible photocatalytic performance. Initially, dry components—cement, sand, and gravel—were thoroughly mixed until a homogeneous composition was achieved. Deionized water was then gradually added while mixing to reach a workable consistency. The fresh mixture was poured into molds (5 × 5 × 5 cm3), compacted to remove air voids, and leveled to achieve surface uniformity. After casting, the molds were placed in a curing chamber maintained at 18 ± 2 °C and relative humidity above 90% for 14 days, ensuring proper hydration and strength development.
Following the curing process, the TiO
2/Feda coating was applied via immersion. TiO
2 nanoparticles were dispersed in deionized water containing 2 wt.% of Feda agent and magnetically stirred for 1 h to ensure complete homogenization. The coating process is schematically illustrated in
Figure 1.
The cured concrete cubes were immersed in this dispersion for 10 min, drained, and dried at 60 °C for 2 h. This procedure was repeated up to three times to obtain multilayer coverage, providing enhanced surface uniformity and photocatalytic performance. After coating, the samples were stored under ambient conditions for 24 h before testing. The parameters applied during the coating process are summarized in
Table 2.
2.3. Analysis of Water Absorption and Wettability
The water absorption behavior of the cementitious samples was analyzed using a gravimetric method based on indirect contact. Prior to testing, all concrete cubes (both TiO
2/Feda-coated and uncoated reference samples) were dried in a laboratory oven at 105 °C for 3 h to remove physically adsorbed moisture. Each cube was subsequently placed on a polyethylene (PE) foam pad saturated with deionized water, ensuring uniform water delivery to the sample surface (
Figure 2). The mass of each sample was recorded at regular intervals using a standard analytical balance (accuracy ±0.1 mg). This setup is distinct from a contact angle measurement, which assesses surface wettability rather than absorption kinetics.
The wetting rate Nw was calculated according to Equation (1):
where:
Nw—wetting rate [g/(m2·h)];
Δm = mt − m0—difference in the mass of the sample at time t(mt) and the initial mass (m0) [g];
A—surface area of the sample in contact with the sponge, through which absorption occurs [m2];
Δt—time interval during which absorption is measured [h].
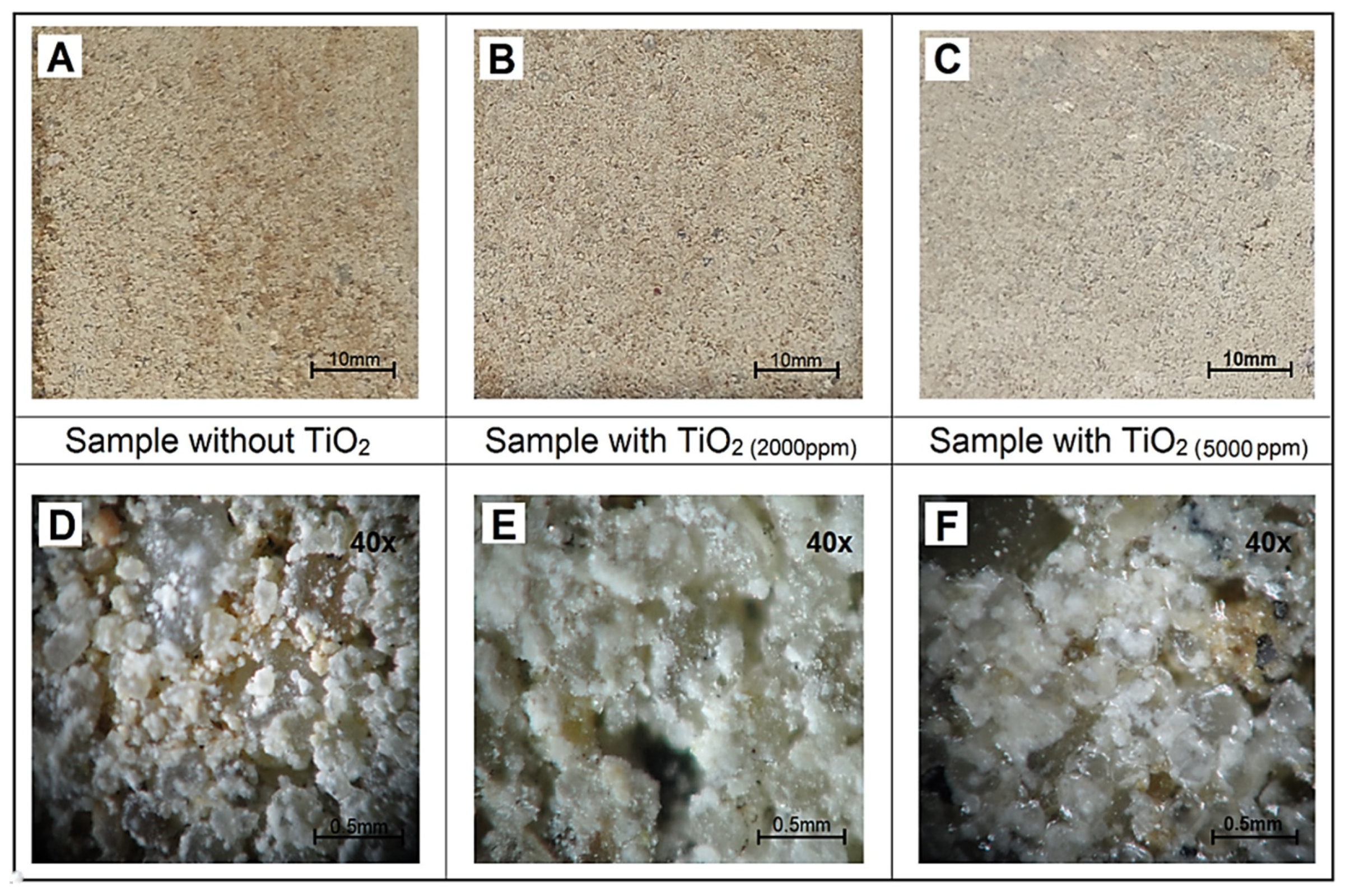
Contact angle measurements were carried out using a Goniometer OCA 25 (DataPhysics Instruments, Filderstadt, Germany) to determine surface wettability (10 μL water droplets). Microscopic observations were performed using a Bresser Researcher Bino Binocular Microscope 40×–1000× (Bresser, Rhede, Germany) to examine surface morphology and coating uniformity. The oven drying parameters were 105 °C for 3 h.
This experimental setup allows reproducible evaluation of water absorption kinetics and wettability of both TiO2/Feda-coated and uncoated concrete samples, providing the basis for subsequent quantitative analysis in the Results and Discussion section.
2.4. Mechanical Properties of TiO2-Modified Concrete Cubes
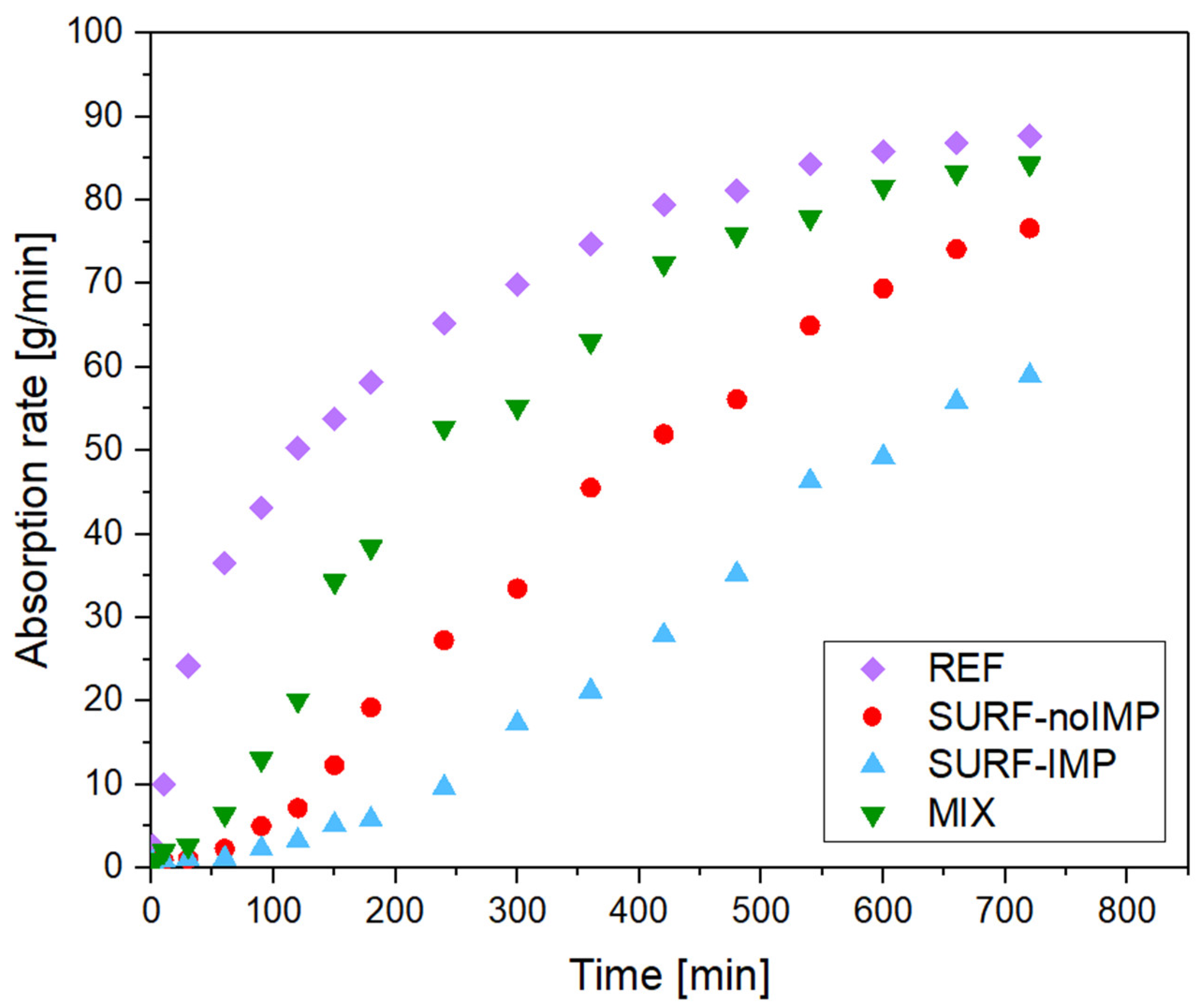
The mechanical properties of the concrete cubes were evaluated using compressive strength testing. Concrete specimens with dimensions of 50 × 50 × 40 mm were prepared and classified into four groups:
REF—reference concrete cube without TiO2;
SURF-noIMP—concrete cube with TiO2 (P25) applied on the surface, without impregnation;
SURF-IMP—concrete cube with TiO2 (P25) applied on the surface with impregnation;
MIX—concrete cube with TiO2 (P25) distributed throughout the concrete mass (0.5 wt.%).
All samples were cured for 28 days under humid conditions. Prior to testing, the specimens were dried to constant mass. Compressive strength measurements were conducted using a MEGA 100 universal testing machine (ViaTeco, Warszawa, Poland). For each sample type, three replicates (n = 3) were tested, and the mean values with standard deviations (SD) were reported.
2.5. Photocatalytic Activity Tests
The photocatalytic performance of TiO
2-coated concrete cubes was evaluated using methylene blue (MB) as a model organic pollutant. Each concrete cube was uniformly coated with a 10 mg/L aqueous solution of MB and allowed to rest in the dark for 1 h to reach adsorption–desorption equilibrium. Subsequently, the samples were irradiated under a Xe-arc lamp (4 × 75 W, spectral range 320–780 nm, light intensity 55 W/m
2 at a distance of 20 cm) for 4 h. The experimental setup for the photocatalytic tests is presented in
Figure 3.
The concentration of residual methylene blue (MB) was measured spectrophotometrically at 664 nm, the wavelength of maximum absorbance of the dye. All measurements were performed in triplicate (
n = 3) to ensure reproducibility. The main experimental parameters for the photocatalytic degradation tests, including TiO
2 loading, sample geometry, irradiation conditions, and solution volume, are summarized in
Table 3. The photocatalytic degradation process was monitored by recording changes in MB absorbance at regular time intervals throughout the irradiation period.
4. Conclusions
The results of this study demonstrate that the method use for TiO2 incorporation exerts a decisive influence on both the photocatalytic efficiency and surface performance of concrete paving blocks. Among all tested configurations, the surface application of TiO2 combined with a functional impregnation treatment proved to be the most effective modification strategy. This system achieved the highest pollutant degradation efficiency—over sixteen times greater than that of the unmodified reference sample—confirming the pronounced photocatalytic activity of the treated surface.
In addition to superior photocatalytic performance, the impregnated TiO2 coating significantly reduced water absorption and increased the contact angle values, indicating enhanced hydrophobicity and improved resistance to environmental factors such as moisture ingress and surface fouling. These characteristics are essential for extending the service life, self-cleaning capability, and long-term durability of concrete elements exposed to outdoor conditions. Conversely, the incorporation of TiO2 nanoparticles within the bulk of the concrete matrix yielded markedly lower photocatalytic activity. This is primarily attributed to the partial encapsulation of TiO2 particles within the cementitious microstructure, which limits their accessibility to ultraviolet radiation and atmospheric pollutants. Consequently, the spatial distribution and stabilization of TiO2 on the concrete surface are key determinants of its functional effectiveness.
Overall, the application of TiO2 through surface coating combined with impregnation presents a practical and scalable approach for producing multifunctional, photocatalytically active concrete materials. Such surfaces not only enhance the environmental performance of urban infrastructure—by contributing to air purification and self-cleaning effects—but also improve durability and reduce maintenance demands. Therefore, this technology represents a promising pathway toward the development of sustainable and intelligent construction materials for next-generation urban environments.