Phenolic Compounds as Phytochemical Tracers of Varietal Origin of Some Autochthonous Apple Cultivars Grown in Serbia
Abstract
1. Introduction
2. Results and Discussion
2.1. Spectrophotometric Antioxidant Assays of the Fruits and Leaves
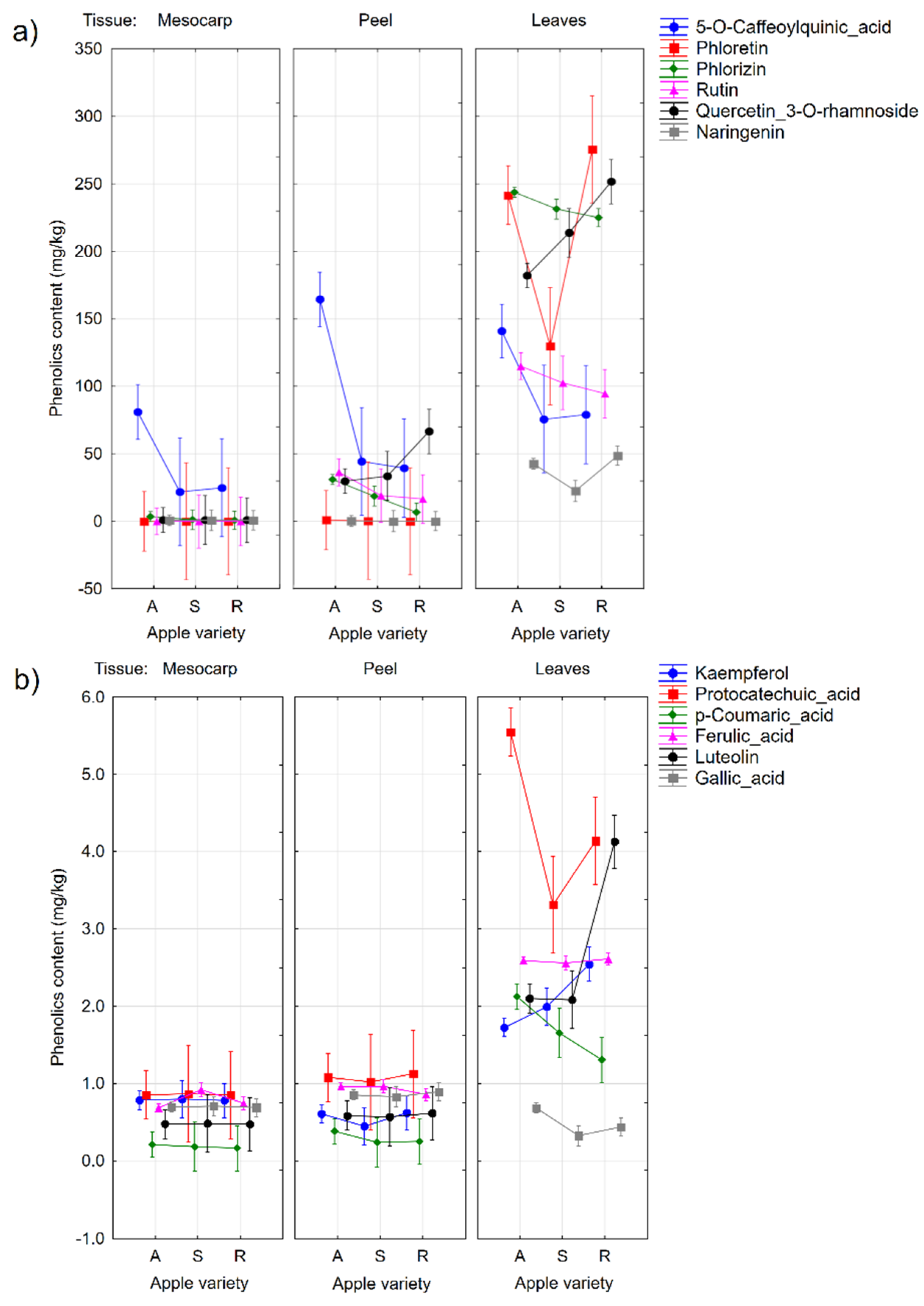
2.2. Quantification of the Individual Phenolic Compounds
2.3. Multivariate Analysis of Variance
2.4. Determination of the Phenolic Profile of the Mesocarp Using the UHPLC-LTQ Orbitrap MS4 Technique
3. Materials and Methods
3.1. Plant Material
3.2. Reagents and Standards
3.3. Extraction Procedures
3.4. Spectrophotometric Tests
3.4.1. Total Phenolic Content (TPC) Determination
3.4.2. Radical Scavenging Activity (RSA) Determination
3.5. UHPLC–DAD MS/MS Analysis of Phenolic Compounds
3.6. UHPLC—LTQ Orbitrap MS4
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cornille, A.; Giraud, T.; Smulders, M.J.M.; Roldán-Ruiz, I.; Gladieux, P. The Domestication and Evolutionary Ecology of Apples. Trends Genet. 2014, 30, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Kschonsek, J.; Wolfram, T.; Stöckl, A.; Böhm, V. Polyphenolic Compounds Analysis of Old and New Apple Cultivars and Contribution of Polyphenolic Profile to the In Vitro Antioxidant Capacity. Antioxidants 2018, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Jakobek, L.; García-Villalba, R.; Tomás-Barberán, F.A. Polyphenolic Characterisation of Old Local Apple Varieties from Southeastern European Region. J. Food Compos. Anal. 2013, 31, 199–211. [Google Scholar] [CrossRef]
- Šavikin, K.; Živković, J.; Zdunić, G.; Gođevac, D.; Ðorđević, B.; Dojčinović, B.; Đorđević, N. Phenolic and Mineral Profiles of Four Balkan Indigenous Apple Cultivars Monitored at Two Different Maturity Stages. J. Food Compos. Anal. 2014, 35, 101–111. [Google Scholar] [CrossRef]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of Polyphenols in Different Apple Varieties. J. Agric. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef]
- Jakobek, L.; Barron, A.R. Ancient Apple Varieties from Croatia as a Source of Bioactive Polyphenolic Compounds. J. Food Compos. Anal. 2016, 45, 9–15. [Google Scholar] [CrossRef]
- Le Bourvellec, C.; Bureau, S.; Renard, C.M.G.C.; Plenet, D.; Gautier, H.; Touloumet, L.; Girard, T.; Simon, S. Cultivar and Year Rather than Agricultural Practices Affect Primary and Secondary Metabolites in Apple Fruit. PLoS ONE 2015, 10, e0141916. [Google Scholar] [CrossRef]
- Mratinić, E.; Fotirić Akšić, M. Evaluation of Phenotipyc Diversity of Apple (Malus sp.) GermplasmThrough the Principle Component Analysis. Genetika 2011, 43, 331–340. [Google Scholar] [CrossRef]
- Petkovsek, M.M.; Slatnar, A.; Stampar, F.; Veberic, R. The Influence of Organic/Integrated Production on the Content of Phenolic Compounds in Apple Leaves and Fruits in Four Different Varieties over a 2-Year Period: Organic/Integrated Production and Content of Phenolic Compounds in Apple. J. Sci. Food Agric. 2010, 90, 2366–2378. [Google Scholar] [CrossRef]
- Petkovsek, M.M.; Stampar, F.; Veberic, R. Parameters of Inner Quality of the Apple Scab Resistant and Susceptible Apple Cultivars (Malus domestica Borkh.). Sci. Hortic. 2007, 114, 37–44. [Google Scholar] [CrossRef]
- Mayr, U.; Michalek, S.; Treutter, D.; Feucht, W. Phenolic Compounds of Apple and Their Relationship to Scab Resistance. J. Phytopathol. 1997, 145, 69–75. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Laskowski, P. Polyphenolic Compounds and Antioxidant Activity of New and Old Apple Varieties. J. Agric. Food Chem. 2008, 56, 6520–6530. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, D.; Laparra-Llopis, J.M.; Zielinski, H.; Szawara-Nowak, D.; Giménez-Bastida, J.A. Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients 2019, 11, 1173. [Google Scholar] [CrossRef]
- Fotirić Akšić, M.; Nešović, M.; Ćirić, I.; Tešić, Ž.; Pezo, L.; Tosti, T.; Gašić, U.; Dojčinović, B.; Lončar, B.; Meland, M. Polyphenolics and Chemical Profiles of Domestic Norwegian Apple (Malus × Domestica Borkh.) Cultivars. Front. Nutr. 2022, 9, 941487. [Google Scholar] [CrossRef] [PubMed]
- Mayr, U. Developmental Changes in the Phenol Concentrations of “Golden Delicious” Apple Fruits and Leaves. Phytochemistry 1995, 38, 1151–1155. [Google Scholar] [CrossRef]
- Mikulič Petkovšek, M.; Štampar, F.; Veberič, R. Accumulation of Phenolic Compounds in Apple in Response to Infection by the Scab Pathogen, Venturia Inaequalis. Physiol. Mol. Plant Pathol. 2009, 74, 60–67. [Google Scholar] [CrossRef]
- Bonarska-Kujawa, D.; Cyboran, S.; Oszmiański, J.; Kleszczyńska, H. Antioxidant Properties of Apple Leaves and Fruits Extracts from Apple Leaves and Fruits as Effective Antioxidants. J. Med. Plants Res. 2011, 5, 2339–2347. [Google Scholar] [CrossRef]
- Veberic, R.; Trobec, M.; Herbinger, K.; Hofer, M.; Grill, D.; Stampar, F. Phenolic Compounds in Some Apple (Malus domestica Borkh) Cultivars of Organic and Integrated Production. J. Sci. Food Agric. 2005, 85, 1687–1694. [Google Scholar] [CrossRef]
- Lu, Y.; Du, Y.; Qin, X.; Wu, H.; Huang, Y.; Cheng, Y.; Wei, Y. Comprehensive Evaluation of Effective Polyphenols in Apple Leaves and Their Combinatory Antioxidant and Neuroprotective Activities. Ind. Crop. Prod. 2019, 129, 242–252. [Google Scholar] [CrossRef]
- Kalinowska, M.; Bielawska, A.; Lewandowska-Siwkiewicz, H.; Priebe, W.; Lewandowski, W. Apples: Content of Phenolic Compounds vs. Variety, Part of Apple and Cultivation Model, Extraction of Phenolic Compounds, Biological Properties. Plant Physiol. Biochem. 2014, 84, 169–188. [Google Scholar] [CrossRef]
- Pantelić, M.; Dabić Zagorac, D.; Natić, M.; Gašić, U.; Jović, S.; Vujović, D.; Djordjević, J.P. Impact of Clonal Variability on Phenolics and Radical Scavenging Activity of Grapes and Wines: A Study on the Recently Developed Merlot and Cabernet Franc Clones (Vitis vinifera L.). PLoS ONE 2016, 11, e0163823. [Google Scholar] [CrossRef] [PubMed]
- Fotirić Akšić, M.; Dabić Zagorac, D.; Gašić, U.; Tosti, T.; Natić, M.; Meland, M. Analysis of Apple Fruit (Malus × Domestica Borkh.) Quality Attributes Obtained from Organic and Integrated Production Systems. Sustainability 2022, 14, 5300. [Google Scholar] [CrossRef]
- Cebulj, A.; Cunja, V.; Mikulic-Petkovsek, M.; Veberic, R. Importance of Metabolite Distribution in Apple Fruit. Sci. Hortic. 2017, 214, 214–220. [Google Scholar] [CrossRef]
- Feng, S.; Yi, J.; Li, X.; Wu, X.; Zhao, Y.; Ma, Y.; Bi, J. Systematic Review of Phenolic Compounds in Apple Fruits: Compositions, Distribution, Absorption, Metabolism, and Processing Stability. J. Agric. Food Chem. 2021, 69, 7–27. [Google Scholar] [CrossRef]
- Łata, B.; Trampczynska, A.; Paczesna, J. Cultivar Variation in Apple Peel and Whole Fruit Phenolic Composition. Sci. Hortic. 2009, 121, 176–181. [Google Scholar] [CrossRef]
- Raudone, L.; Raudonis, R.; Liaudanskas, M.; Janulis, V.; Viskelis, P. Phenolic Antioxidant Profiles in the Whole Fruit, Flesh and Peel of Apple Cultivars Grown in Lithuania. Sci. Hortic. 2017, 216, 186–192. [Google Scholar] [CrossRef]
- Gašić, U.M.; Natić, M.M.; Mišić, D.M.; Lušić, D.V.; Milojković-Opsenica, D.M.; Tešić, Ž.L.; Lušić, D. Chemical Markers for the Authentication of Unifloral Salvia Officinalis L. Honey. J. Food Compos. Anal. 2015, 44, 128–138. [Google Scholar] [CrossRef]
- Naish, M.; Clifford, M.N.; Birch, G.G. Sensory Astringency of 5-O-Caffeoylquinic Acid, Tannic Acid and Grape-Seed Tannin by a Time-Intensity Procedure. J. Sci. Food Agric. 1993, 61, 57–64. [Google Scholar] [CrossRef]
- Marks, S.C.; Mullen, W.; Crozier, A. Flavonoid and Chlorogenic Acid Profiles of English Cider Apples. J. Sci. Food Agric. 2007, 87, 719–728. [Google Scholar] [CrossRef]
- Belviso, S.; Scursatone, B.; Re, G.; Zeppa, G. Novel Data on the Polyphenol Composition of Italian Ancient Apple Cultivars. Int. J. Food Prop. 2013, 16, 1507–1515. [Google Scholar] [CrossRef]
- Slatnar, A.; Mikulic Petkovsek, M.; Halbwirth, H.; Stampar, F.; Stich, K.; Veberic, R. Enzyme Activity of the Phenylpropanoid Pathway as a Response to Apple Scab Infection. Ann. Appl. Biol. 2010, 156, 449–456. [Google Scholar] [CrossRef]
- Picinelli, A.; Dapena, E.; Mangas, J.J. Polyphenolic Pattern in Apple Tree Leaves in Relation to Scab Resistance. A Preliminary Study. J. Agric. Food Chem. 1995, 43, 2273–2278. [Google Scholar] [CrossRef]
- Skłodowska, M.; Mikiciński, A.; Wielanek, M.; Kuźniak, E.; Sobiczewski, P. Phenolic Profiles in Apple Leaves and the Efficacy of Selected Phenols against Fire Blight (Erwinia amylovora). Eur. J. Plant Pathol. 2018, 151, 213–228. [Google Scholar] [CrossRef]
- De Paepe, D.; Valkenborg, D.; Noten, B.; Servaes, K.; Diels, L.; De Loose, M.; Van Droogenbroeck, B.; Voorspoels, S. Variability of the Phenolic Profiles in the Fruits from Old, Recent and New Apple Cultivars Cultivated in Belgium. Metabolomics 2015, 11, 739–752. [Google Scholar] [CrossRef]
- Fotirić Akšić, M.; Dabić Zagorac, D.; Sredojević, M.; Milivojević, J.; Gašić, U.; Meland, M.; Natić, M. Chemometric Characterization of Strawberries and Blueberries According to Their Phenolic Profile: Combined Effect of Cultivar and Cultivation System. Molecules 2019, 24, 4310. [Google Scholar] [CrossRef] [PubMed]
- Kečkeš, S.; Gašić, U.; Veličković, T.Ć.; Milojković-Opsenica, D.; Natić, M.; Tešić, Ž. The Determination of Phenolic Profiles of Serbian Unifloral Honeys Using Ultra-High-Performance Liquid Chromatography/High Resolution Accurate Mass Spectrometry. Food Chem. 2013, 138, 32–40. [Google Scholar] [CrossRef]
- Jaiswal, R.; Kuhnert, N. How to Identify and Discriminate between the Methyl Quinates of Chlorogenic Acids by Liquid Chromatography-Tandem Mass Spectrometry. J. Mass Spectrom. 2011, 46, 269–281. [Google Scholar] [CrossRef]
- Park, E.K.; Ahn, S.R.; Kim, D.-H.; Lee, E.-W.; Kwon, H.J.; Kim, B.W.; Kim, T.H. Effects of Unripe Apple Polyphenols on the Expression of Matrix Metalloproteinase-1 and Type-1 Procollagen in Ultraviolet Irradiated Human Skin Fibroblasts. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 449–455. [Google Scholar] [CrossRef]
- Li, W.; Yang, R.; Ying, D.; Yu, J.; Sanguansri, L.; Augustin, M.A. Analysis of Polyphenols in Apple Pomace: A Comparative Study of Different Extraction and Hydrolysis Procedures. Ind. Crop. Prod. 2020, 147, 112250. [Google Scholar] [CrossRef]
- Li, H.; Subbiah, V.; Barrow, C.J.; Dunshea, F.R.; Suleria, H.A.R. Phenolic Profiling of Five Different Australian Grown Apples. Appl. Sci. 2021, 11, 2421. [Google Scholar] [CrossRef]
- Sanoner, P.; Guyot, S.; Marnet, N.; Molle, D.; Drilleau, J.-F. Polyphenol Profiles of French Cider Apple Varieties (Malus domestica sp.). J. Agric. Food Chem. 1999, 47, 4847–4853. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N.; Jaganath, I.B.; Ludwig, I.A.; Crozier, A. Chlorogenic Acids and the Acyl-Quinic Acids: Discovery, Biosynthesis, Bioavailability and Bioactivity. Nat. Prod. Rep. 2017, 34, 1391–1421. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.A.; Murillo, R.; Heinzmann, B.; Laufer, S.; Wray, V.; Merfort, I. Structural and Conformational Analysis of Proanthocyanidins from Parapiptadenia Rigida and Their Wound-Healing Properties. J. Nat. Prod. 2011, 74, 1427–1436. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Ku, K.; Jeong, M.; Kim, S.S.; Mitchell, A.E.; Lee, J. A Comparison of the Chemical Composition and Antioxidant Activity of Several New Early- to Mid-season Apple Cultivars for a Warmer Climate with Traditional Cultivars. J. Sci. Food Agric. 2019, 99, 4712–4724. [Google Scholar] [CrossRef]
- Chen, S.; Li, M.; Zheng, G.; Wang, T.; Lin, J.; Wang, S.; Wang, X.; Chao, Q.; Cao, S.; Yang, Z.; et al. Metabolite Profiling of 14 Wuyi Rock Tea Cultivars Using UPLC-QTOF MS and UPLC-QqQ MS Combined with Chemometrics. Molecules 2018, 23, 104. [Google Scholar] [CrossRef]
- Pavlović, A.V.; Dabić, D.Č.; Momirović, N.M.; Dojčinović, B.P.; Milojković-Opsenica, D.M.; Tešić, Ž.L.; Natić, M.M. Chemical Composition of Two Different Extracts of Berries Harvested in Serbia. J. Agric. Food Chem. 2013, 61, 4188–4194. [Google Scholar] [CrossRef]
- Fotirić Akšić, M.M.; Dabić, D.Č.; Gašić, U.M.; Zec, G.N.; Vulić, T.B.; Tešić, Ž.L.; Natić, M.M. Polyphenolic Profile of Pear Leaves with Different Resistance to Pear Psylla (Cacopsylla pyri). J. Agric. Food Chem. 2015, 63, 7476–7486. [Google Scholar] [CrossRef]
- Natić, M.M.; Dabić, D.Č.; Papetti, A.; Fotirić Akšić, M.M.; Ognjanov, V.; Ljubojević, M.; Tešić, Ž.L. Analysis and Characterisation of Phytochemicals in Mulberry (Morus alba L.) Fruits Grown in Vojvodina, North Serbia. Food Chem. 2015, 171, 128–136. [Google Scholar] [CrossRef]
- Gašić, U.; Kečkeš, S.; Dabić, D.; Trifković, J.; Milojković-Opsenica, D.; Natić, M.; Tešić, Ž. Phenolic Profile and Antioxidant Activity of Serbian Polyfloral Honeys. Food Chem. 2014, 145, 599–607. [Google Scholar] [CrossRef]

| 5-O-Caffeoylquinic Acid | Phloretin | Phlorizin | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 721,442 | 721,442 | 176.92 | <0.001 * | 671,792 | 671,792 | 138.98 | <0.001 * | 927,691 | 927,691 | 6835 | <0.001 * |
| F1 | 2 | 71,823 | 35,911 | 8.81 | <0.001 * | 1.33 × 106 | 667,048 | 138.00 | <0.001 * | 1.44 × 106 | 718,284 | 5292 | <0.001 * |
| F2 | 2 | 278,880 | 139,440 | 34.19 | <0.001 * | 43,735 | 21,867 | 4.52 | 0.012 * | 7188 | 3594 | 26.48 | <0.001 * |
| F3 | 1 | 44,856 | 44,856 | 11.00 | <0.001 * | 171.45 | 171 | 0.035 | 0.851 | 1918 | 1918 | 14.13 | <0.001 * |
| F1 × F2 | 4 | 36,913 | 9228 | 2.26 | 0.065 | 87,107 | 21,777 | 4.50 | 0.002 * | 2474 | 618 | 4.56 | 0.002 * |
| F1 × F3 | 2 | 49,047 | 24,523 | 6.01 | <0.001 * | 420.37 | 210 | 0.04 | 0.957 | 4113 | 2056 | 15.15 | <0.001 * |
| F2 × F3 | 2 | 61,802 | 30,901 | 7.58 | <0.001 * | 1063 | 531 | 0.11 | 0.896 | 45 | 22 | 0.17 | 0.846 |
| F1 × F2 × F3 | 4 | 90,284 | 22,571 | 5.54 | <0.001 * | 2237 | 559 | 0.12 | 0.977 | 638 | 159 | 1.18 | 0.323 |
| Rutin | Quercetin-3-O-Rhamnoside | Naringenin | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 236,066 | 236,066 | 236.66 | <0.001 * | 973,440 | 973,440 | 1163 | <0.001 * | 21,776 | 21,776 | 141 | <0.001 * |
| F1 | 2 | 256,056 | 128,028 | 128.35 | <0.001 * | 1.12 × 106 | 558,657 | 667.70 | <0.001 * | 40,385 | 20,192 | 130 | <0.001 * |
| F2 | 2 | 6105 | 3052 | 3.06 | 0.050 * | 34,919 | 17,459 | 20.87 | <0.001 * | 1408 | 704 | 4.55 | 0.012 * |
| F3 | 1 | 13,487 | 13,487 | 13.52 | <0.001 * | 1686 | 1686 | 2.01 | 0.158 | 0.226 | 0.226 | 0.00 | 0.969 |
| F1 × F2 | 4 | 3128 | 782 | 0.78 | 0.537 | 24,235 | 6058 | 7.24 | <0.001 * | 2806 | 701 | 4.54 | 0.002 * |
| F1 × F3 | 2 | 7595 | 3797 | 3.81 | 0.024 * | 7593 | 3796 | 4.54 | 0.012 * | 10.7 | 5.34 | 0.03 | 0.966 |
| F2 × F3 | 2 | 967 | 483 | 0.49 | 0.617 | 2029 | 1014 | 1.21 | 0.300 | 21.8 | 10.9 | 0.07 | 0.932 |
| F1 × F2 × F3 | 4 | 2352 | 588 | 0.59 | 0.671 | 1775 | 443 | 0.53 | 0.713 | 51.2 | 12.8 | 0.08 | 0.988 |
| Quercetin | Quercetin-3-O-glucoside | ||||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | ||||
| Intercept | 1 | 45,014 | 45,014 | 311.0 | <0.001 * | 2.78 × 106 | 2.78 × 106 | 2630 | <0.001 * | ||||
| F1 | 2 | 21,784 | 10,892 | 75.25 | <0.001 * | 2.68 × 106 | 1.34 × 106 | 1267 | <0.001 * | ||||
| F2 | 2 | 18.4 | 9.18 | 0.06 | 0.939 | 3056 | 1528 | 1.45 | 0.239 | ||||
| F3 | 1 | 200 | 200 | 1.38 | 0.241 | 46,937 | 46,937 | 44.39 | <0.001 * | ||||
| F1 × F2 | 4 | 749 | 187 | 1.29 | 0.275 | 6876 | 1719 | 1.63 | 0.170 | ||||
| F2 × F3 | 2 | 2973 | 1486 | 10.27 | <0.001 * | 73,995 | 36,997 | 34.99 | <0.001 * | ||||
| F2 × F3 | 2 | 31.6 | 15.8 | 0.11 | 0.897 | 628 | 314 | 0.30 | 0.743 | ||||
| F1 × F2 × F3 | 4 | 329 | 82.3 | 0.57 | 0.686 | 2525 | 631 | 0.60 | 0.665 | ||||
| p-Coumaric Acid | Protocatechuic Acid | Caffeic Acid | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 67.86 | 67.86 | 258.70 | <0.001 * | 564.05 | 564.05 | 571.60 | <0.001 * | 139.29 | 139.29 | 380.14 | <0.001 * |
| F1 | 2 | 61.25 | 30.63 | 116.76 | <0.001 * | 326.88 | 163.44 | 165.62 | <0.001 * | 69.83 | 34.91 | 95.29 | <0.001 * |
| F2 | 2 | 3.48 | 1.74 | 6.63 | 0.002 * | 16.49 | 8.24 | 8.35 | <0.001 * | 1.27 | 0.64 | 1.73 | 0.180 |
| F3 | 1 | 0.03 | 0.03 | 0.12 | 0.730 | 7.05 | 7.05 | 7.15 | 0.008 * | 0.10 | 0.10 | 0.28 | 0.599 |
| F1 × F2 | 4 | 3.67 | 0.92 | 3.49 | 0.009 * | 32.37 | 8.09 | 8.20 | <0.001 * | 4.44 | 1.11 | 3.03 | 0.019 * |
| F1 × F3 | 2 | 6.20 | 3.10 | 11.82 | <0.001 * | 82.05 | 41.02 | 41.57 | <0.001 * | 7.74 | 3.87 | 10.56 | <0.001 * |
| F2 × F3 | 2 | 0.26 | 0.13 | 0.50 | 0.604 | 0.95 | 0.48 | 0.48 | 0.617 | 5.65 | 2.82 | 7.71 | <0.001 * |
| F1 × F2 × F3 | 4 | 0.12 | 0.03 | 0.11 | 0.977 | 1.40 | 0.35 | 0.35 | 0.841 | 2.56 | 0.64 | 1.75 | 0.142 |
| Ferulic Acid | Gallic Acid | Luteolin | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 265.75 | 265.75 | 13,086 | <0.001 * | 59.81 | 59.81 | 1415 | <0.001 * | 211.86 | 211.86 | 588.07 | <0.001 * |
| F1 | 2 | 86.45 | 43.23 | 2128 | <0.001 * | 3.06 | 1.53 | 36.18 | <0.001 * | 144.00 | 72.00 | 199.85 | <0.001 * |
| F2 | 2 | 0.13 | 0.06 | 3.09 | 0.048 * | 0.41 | 0.21 | 4.89 | 0.009 * | 13.68 | 6.84 | 18.99 | <0.001 * |
| F3 | 1 | 21.29 | 21.29 | 1048 | <0.001 * | 1.77 | 1.77 | 41.83 | <0.001 * | 0.01 | 0.01 | 0.03 | 0.863 |
| F1 × F2 | 4 | 0.43 | 0.11 | 5.32 | <0.001 * | 0.95 | 0.24 | 5.63 | <0.001 * | 26.20 | 6.55 | 18.18 | <0.001 * |
| F1 × F3 | 2 | 12.43 | 6.22 | 306.09 | <0.001 * | 6.74 | 3.37 | 79.76 | <0.001 * | 8.50 | 4.25 | 11.79 | <0.001 * |
| F2 × F3 | 2 | 0.26 | 0.13 | 6.34 | 0.002 * | 0.04 | 0.02 | 0.47 | 0.627 | 0.59 | 0.30 | 0.82 | 0.441 |
| F1 × F2 × F3 | 4 | 0.19 | 0.05 | 2.28 | 0.063 | 0.04 | 0.01 | 0.21 | 0.933 | 1.39 | 0.35 | 0.96 | 0.430 |
| Apigenin | Eriodictyol | Naringin | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 18.02 | 18.02 | 585.96 | <0.001 * | 178.46 | 178.46 | 29,811 | <0.001 * | 232.89 | 232.89 | 117.58 | <0.001 * |
| F1 | 2 | 3.53 | 1.76 | 57.38 | <0.001 * | 85.97 | 42.98 | 7180 | <0.001 * | 76.44 | 38.22 | 19.30 | <0.001 * |
| F2 | 2 | 0.03 | 0.02 | 0.52 | 0.594 | 0.02 | 0.01 | 1.52 | 0.221 | 17.29 | 8.65 | 4.37 | 0.014* |
| F3 | 1 | 0.06 | 0.06 | 2.00 | 0.159 | 0.01 | 0.01 | 1.07 | 0.303 | 46.78 | 46.78 | 23.62 | <0.001 * |
| F1 × F2 | 4 | 0.08 | 0.02 | 0.63 | 0.642 | 0.03 | 0.01 | 1.07 | 0.373 | 22.06 | 5.52 | 2.78 | 0.028 |
| F1 × F3 | 2 | 1.68 | 0.84 | 27.28 | <0.001 * | 6.39 | 3.20 | 533.85 | <0.001 * | 97.00 | 48.50 | 24.49 | <0.001 * |
| F2 × F3 | 2 | 0.03 | 0.02 | 0.52 | 0.593 | 0.02 | 0.01 | 1.82 | 0.166 | 18.61 | 9.30 | 4.70 | 0.010* |
| F1 × F2 × F3 | 4 | 0.07 | 0.02 | 0.55 | 0.700 | 0.06 | 0.02 | 2.67 | 0.034 | 23.81 | 5.95 | 3.01 | 0.020* |
| Kaempherol | Kaempferol-7-O-glucoside | Isorhamnetin-3-O-glucoside | |||||||||||
| Factor | DF | SS | MS | F | p | SS | MS | F | p | SS | MS | F | p |
| Intercept | 1 | 169.55 | 169.55 | 1133 | <0.001 * | 421.15 | 421.15 | 383.12 | <0.001 * | 3992 | 3992 | 96.69 | <0.001 * |
| F1 | 2 | 58.51 | 29.25 | 195.44 | <0.001 * | 66.25 | 33.13 | 30.13 | <0.001 * | 1978 | 988.90 | 23.95 | <0.001 * |
| F2 | 2 | 2.11 | 1.055 | 7.05 | 0.001 * | 0.35 | 0.18 | 0.16 | 0.851 | 21.92 | 10.96 | 0.27 | 0.767 |
| F3 | 1 | 0.09 | 0.09 | 0.63 | 0.430 | 0.25 | 0.25 | 0.23 | 0.632 | 1053 | 1053 | 25.50 | <0.001 * |
| F1 × F2 | 4 | 4.34 | 1.09 | 7.26 | <0.001 * | 0.42 | 0.11 | 0.10 | 0.983 | 29.32 | 7.33 | 0.18 | 0.950 |
| F1 × F3 | 2 | 14.66 | 7.33 | 48.97 | <0.001 * | 18.96 | 9.48 | 8.62 | <0.001 * | 2394 | 1197 | 28.99 | <0.001 * |
| F2 × F3 | 2 | 0.17 | 0.09 | 0.58 | 0.560 | 0.31 | 0.15 | 0.14 | 0.870 | 20.37 | 10.18 | 0.25 | 0.782 |
| F1 × F2 × F3 | 4 | 0.43 | 0.11 | 0.71 | 0.584 | 0.38 | 0.10 | 0.09 | 0.986 | 47.17 | 11.79 | 0.29 | 0.887 |
| No. | Compound Name | tR, min | Molecular Formula, [M–H]− | Calculated Mass, [M–H]− | Exact Mass, [M–H]− | Δ ppm | MS2 Fragments, (% Base Peak) | MS3 Fragments, (% Base Peak) | MS4 Fragments, (% Base Peak) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Protocatechuic acid hexoside | 6.55 | C13H15O9− | 315.072156 | 315.07185 | 0.98 | 108 (10), 109 (11), 147(38), 152 (42), 153 (100), 163 (10), 165 (12) | 108 (21), 109 (100) | 81 (100) |
| 2 | Protocatechuic acid | 6.66 | C7H5O4− | 153.019332 | 153.01947 | −0.89 | 107 (89), 108 (33), 109 (100), 110 (25), 123 (41), 125 (79), 136 (24) | 65 (28), 67 (36), 81 (59), 83 (27), 91 (100) | |
| 3 | Protocatechuic acid pentosylhexoside | 6.72 | C18H23O13− | 447.114415 | 447.11415 | 0.60 | 152 (100), 153 (35), 163 (59), 177 (31), 179 (31), 271 (36), 315 (87) | 108 (100) | |
| 4 | Caffeic acid hexoside 1 | 6.84 | C15H17O9− | 341.087806 | 341.08804 | −0.68 | 147 (19), 153 (25), 161 (33), 179 (100), 180 (9), 203 (10), 251 (13) | 135 (100) | |
| 5 | p-Coumaric acid | 6.91 | C9H7O3− | 163.040068 | 163.04010 | −0.17 | 103 (82), 117 (68), 119 (96), 121 (61), 133 (71), 135 (100), 136 (76) | 75 (24), 107 (100), 108 (12) | |
| 6 | Caffeic acid hexoside 2 | 7.03 | C15H17O9− | 341.087806 | 341.08754 | 0.77 | 147 (19), 153 (25), 161 (33), 179 (100), 180 (9), 203 (10), 251 (13) | 135 (100) | |
| 7 | Caffeic acid hexoside 3 | 7.35 | C15H17O9− | 341.087806 | 341.08769 | 0.33 | 135 (9), 147 (42), 153 (26), 161 (37), 179 (100), 180 (10), 203 (7) | 135 (100) | 79 (40), 106 (100), 107 (52) |
| 8 | 5-O-Caffeoylquinic acid | 7.47 | C16H17O9− | 353.087806 | 353.08760 | 0.58 | 179 (3), 191 (100) | 85 (100), 93 (60), 111 (36), 127 (90), 171 (25), 173 (61) | 57 (100) |
| 9 | p-Coumaric acid hexoside | 7.84 | C15H17O8− | 325.092891 | 325.09274 | 0.48 | 119 (11), 145 (100), 146 (10), 163 (87), 187 (40), 265 (16), 289 (58) | 117 (100), 127 (3) | |
| 10 | 5-O-Caffeoylquinic acid isomer | 7.90 | C16H17O9− | 353.087806 | 353.08766 | 0.41 | 191 (100), 192 (3) | 85 (100), 93 (60), 111 (34), 127 (83), 171 (24), 173 (57) | 57 (100) |
| 11 | Methyl-3-O-caffeoylquinate | 8.03 | C17H19O9− | 367.103456 | 367.10338 | 0.22 | 135 (46), 161 (100), 162 (10), 179 (52), 320 (12), 321 (13), 329 (74) | 133 (100) | |
| 12 | 3-p-Coumaroylquinic acid | 8.10 | C16H17O8− | 337.092891 | 337.09273 | 0.48 | 163 (5), 173 (100) | 59 (7), 71 (20), 93 (100), 109 (7), 111 (49), 155 (11) | |
| 13 | Methyl-5-O-caffeoylquinate | 8.45 | C17H19O9− | 367.103456 | 367.10310 | 0.96 | 134 (3), 135 (47), 136 (4), 161 (12), 179 (100), 180 (9), 191 (22) | 135 (100) | 89 (38), 117 (13), 135 (100) |
| 14 | Caffeic acid | 8.54 | C9H7O4− | 179.034982 | 179.03507 | −0.49 | 135 (100) | 107 (100) | |
| 15 | Methyl-3-p-coumaroylquinate | 8.73 | C17H19O8− | 351.108541 | 351.10846 | 0.24 | 117 (5), 119 (9), 145 (100), 146 (5) | 117 (100), 145 (3) | |
| 16 | Methyl-5-O-caffeoylquinate isomer | 8.87 | C17H19O9− | 367.103456 | 367.10332 | 0.37 | 134 (3), 135 (47), 136 (4), 161 (10), 179 (100), 180 (8), 191 (20) | 135 (100) | 91 (68), 106 (29), 107 (81), 135 (100) |
| 17 | Methyl-5-p-coumaroylquinate | 8.93 | C17H19O8− | 351.108541 | 351.10856 | −0.06 | 119 (17), 145 (10), 163 (100), 164 (7) | 119 (100) | 93 (100) |
| 18 | Methyl-5-p-coumaroylquinate isomer | 9.38 | C17H19O8− | 351.108541 | 351.10865 | −0.31 | 119 (19), 145 (5), 163 (100), 164 (7) | 119 (100) | 93 (100), 119 (9), 135 (72) |
| 19 | Rosmarinic acid | 9.65 | C18H15O8− | 359.077241 | 359.07707 | 0.47 | 133 (5), 161 (100), 162 (9), 179 (22), 197 (20), 223 (7), 313 (4) | 133 (100) | |
| 20 | Caffeoyl-feruloylquinic acid | 10.22 | C26H25O12− | 529.135150 | 529.13492 | 0.43 | 161 (8), 179 (6), 349 (8), 367 (100), 368 (18) | 134 (5), 35 (60), 161 (76), 179 (100), 191 (20), 193 (5) | 135 (100) |
| 21 | Prodelphinidin B type ((epi)gallocatechin-(epi)catechin) | 6.39 | C30H25O13− | 593.130065 | 593.12993 | 0.22 | 289 (5), 315 (11), 441 (100), 442 (19) | 153 (34), 161 (9), 271 (14), 287 (14), 289 (50), 315 (100) | 151 (5), 153 (100), 161 (26), 193 (3), 297 (21) |
| 22 | (Epi)catechin-hexoside | 6.99 | C21H23O11− | 451.124585 | 451.12435 | 0.52 | 245 (12), 289 (100), 290 (16), 405 (3) | 179 (8), 203 (13), 205 (38), 231 (6), 245 (100), 247 (7) | 161 (20), 175 (17), 187 (33), 188 (20), 203 (100) |
| 23 | Procyanidin B type 1 (catechin-(epi)catechin) | 7.08 | C30H25O12− | 577.135150 | 577.13502 | 0.23 | 289 (24), 407 (61), 408 (13), 425 (100), 426 (16), 451 (26), 559 (8) | 273 (7), 381 (4), 407 (100) | 281 (93), 283 (34), 285 (100), 297 (35), 389 (31) |
| 24 | Epiafzelechin-3-O-gallate | 7.67 | C22H17O9− | 425.087806 | 425.08756 | 0.58 | 243 (21), 273 (11), 285 (6), 379 (7), 381 (10), 407 (100), 408 (33) | 256 (27), 281 (65), 283 (30), 285 (100), 297 (28), 389 (21) | 213 (19), 241 (4), 242 (8), 257 (100), 258 (13) |
| 25 | Procyanidin B type 2 (catechin-(epi)catechin) | 7.67 | C30H25O12− | 577.135150 | 577.13510 | 0.09 | 287 (6), 289 (17), 407 (51), 408 (10), 425 (100), 426 (11), 451 (18) | 273 (7), 381 (4), 407 (100) | 281 (87), 283 (30), 285 (100), 297 (27), 389 (30) |
| 26 | Catechin | 8.00 | C15H13O6− | 289.071762 | 289.07159 | 0.61 | 179 (12), 203 (10), 205 (38), 231 (6), 245 (100), 246 (10), 247 (6) | 161 (19), 175 (10), 187 (23), 188 (14), 203 (100), 227 (26) | 157 (10), 161 (33), 175 (100), 185 (16), 188 (48) |
| 27 | Procyanidin B type 3 (catechin-(epi)catechin) | 8.42 | C30H25O12− | 577.135150 | 577.13564 | −0.85 | 287 (11), 289 (16), 407 (45), 408 (9), 425 (100), 426 (14), 451 (19) | 273 (9), 381 (6), 407 (100) | 281 (100), 283 (34), 285 (84), 297 (29), 389 (34) |
| 28 | Epicatechin | 8.55 | C15H13O6− | 289.071762 | 289.07176 | 0.00 | 125 (19), 167 (35), 203 (8), 205 (30), 245 (100), 246 (9), 271 (18) | 161 (23), 185 (37), 187 (17), 201 (19), 203 (100), 227 (67) | |
| 29 | (Epi)catechin-methyl(epi)gallocatechin | 8.79 | C31H27O13− | 607.145715 | 607.14571 | 0.01 | 287 (79), 405 (46), 423 (33), 437 (37), 449 (22), 455 (95), 575 (100) | 245 (8), 287 (100), 405 (22), 413 (20), 423 (35), 449 (48) | 125 (100), 161 (5), 243 (12), 245 (5) |
| 31 | Quercetin-3-O-rhamnoside | 9.23 | C21H19O11− | 447.093285 | 447.09339 | −0.23 | 285 (3), 299 (4), 300 (28), 301 (100), 302 (11) | 151 (82), 179 (100), 257 (11), 72 (10), 273 (19), 283 (20) | 151 (100) |
| 32 | Kaempferol-3-O-pentoside | 9.56 | C20H17O10− | 417.082720 | 417.08261 | 0.26 | 255 (5), 283 (3), 284 (66), 285 (100), 286 (14), 327 (4) | 163 (20), 229 (45), 241 (29), 256 (46), 257 (100), 267 (42) | 163 (85), 212 (14), 213 (22), 229 (100), 239 (56) |
| 33 | Kaempferol-3-O-rhamnoside | 9.73 | C21H19O10− | 431.098371 | 431.09814 | 0.55 | 255 (6), 283 (7), 284 (40), 285 (100), 286 (16), 327 (5) | 213 (28), 229 (41), 241 (36), 256 (70), 257 (100), 267 (43) | 163 (63), 213 (19), 227 (14), 229 (100), 239 (22) |
| 34 | Phloretin 2′-O-(2′′-O-pentosylhexoside) | 9.00 | C26H31O14− | 567.171929 | 567.17197 | −0.07 | 167 (7), 273 (100), 274 (14) | 123 (4), 125 (4), 167 (100) | 123 (100), 125 (13), 151 (3) |
| 35 | Phloretin | 9.58 | C15H13O5− | 273.076847 | 273.07672 | 0.46 | 123 (4), 125 (4), 167 (100), 168 (7) | 123 (100), 125 (13), 151 (3) | 67 (3), 81 (100), 95 (55), 108 (3) |
| 36 | Phloretin-2′-O-hexoside (Phlorizin) | 9.59 | C21H23O10− | 435.129671 | 435.12950 | 0.39 | 273 (100), 274 (13) | 123 (5), 125 (3), 167 (100) | 123 (100), 125 (14), 151 (3) |
| Number | Sort | Peel Color | Cultivar Type |
|---|---|---|---|
| 1. | Red Delicious | Red | Standard |
| 2. | Granny Smith | Green | Standard |
| 3. | Idared | Yellow—red | Standard |
| 4. | Golden Delicious | Yellow | Standard |
| 5. | Jonagold | Yellow—red | Standard |
| 6. | Prima | Red—yellow | Resistant |
| 7. | Gala Galax | Yellow—red | Resistant |
| 8. | William’s Pride | Red | Resistant |
| 9. | Rewena | Red—yellow | Resistant |
| 10. | Topaz | Yellow—red | Resistant |
| 11. | Remura | Red—yellow | Resistant |
| 12. | Zaječarska duguljasta | Red | Autochthonous |
| 13. | MioničkaTikvara | Yellow—red | Autochthonous |
| 14. | Zaječarski delišes | Red—yellow | Autochthonous |
| 15. | Gružanjaska letnja kolačara | Red | Autochthonous |
| 16. | Sećeruša | Red | Autochthonous |
| 17. | Pamuklija | Yellow—red | Autochthonous |
| 18. | Demirka | Red—yellow | Autochthonous |
| 19. | Jesenji jablan | Yellow | Autochthonous |
| 20. | Kadumana | Red | Autochthonous |
| 21. | Buzlija | Yellow—red | Autochthonous |
| 22. | Krtajka | Red | Autochthonous |
| 23. | Hajdučica | Red | Autochthonous |
| 24. | Vrtiglavska slatkača | Yellow | Autochthonous |
| 25. | Kopaoničanka | Red—yellow | Autochthonous |
| 26. | Bela kalaćuša | Pale yellow—red | Autochthonous |
| 27. | Loznička tikvara | Yellow—red | Autochthonous |
| 28. | Šipura | Red—yellow | Autochthonous |
| 29. | Šipina | Pale red—yellow | Autochthonous |
| 30. | Kožara | Yellow—green | Autochthonous |
| 31. | Budimka | Pale yellow—green | Autochthonous |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horvacki, N.; Andrić, F.; Gašić, U.; Đurović, D.; Tešić, Ž.; Fotirić Akšić, M.; Milojković-Opsenica, D. Phenolic Compounds as Phytochemical Tracers of Varietal Origin of Some Autochthonous Apple Cultivars Grown in Serbia. Molecules 2022, 27, 7651. https://doi.org/10.3390/molecules27217651
Horvacki N, Andrić F, Gašić U, Đurović D, Tešić Ž, Fotirić Akšić M, Milojković-Opsenica D. Phenolic Compounds as Phytochemical Tracers of Varietal Origin of Some Autochthonous Apple Cultivars Grown in Serbia. Molecules. 2022; 27(21):7651. https://doi.org/10.3390/molecules27217651
Chicago/Turabian StyleHorvacki, Nikola, Filip Andrić, Uroš Gašić, Dejan Đurović, Živoslav Tešić, Milica Fotirić Akšić, and Dušanka Milojković-Opsenica. 2022. "Phenolic Compounds as Phytochemical Tracers of Varietal Origin of Some Autochthonous Apple Cultivars Grown in Serbia" Molecules 27, no. 21: 7651. https://doi.org/10.3390/molecules27217651
APA StyleHorvacki, N., Andrić, F., Gašić, U., Đurović, D., Tešić, Ž., Fotirić Akšić, M., & Milojković-Opsenica, D. (2022). Phenolic Compounds as Phytochemical Tracers of Varietal Origin of Some Autochthonous Apple Cultivars Grown in Serbia. Molecules, 27(21), 7651. https://doi.org/10.3390/molecules27217651








