Preparation of Lignin Carbon/Zinc Oxide Electrode Material and Its Application in Supercapacitors
Abstract
1. Introduction
2. Results and Discussion
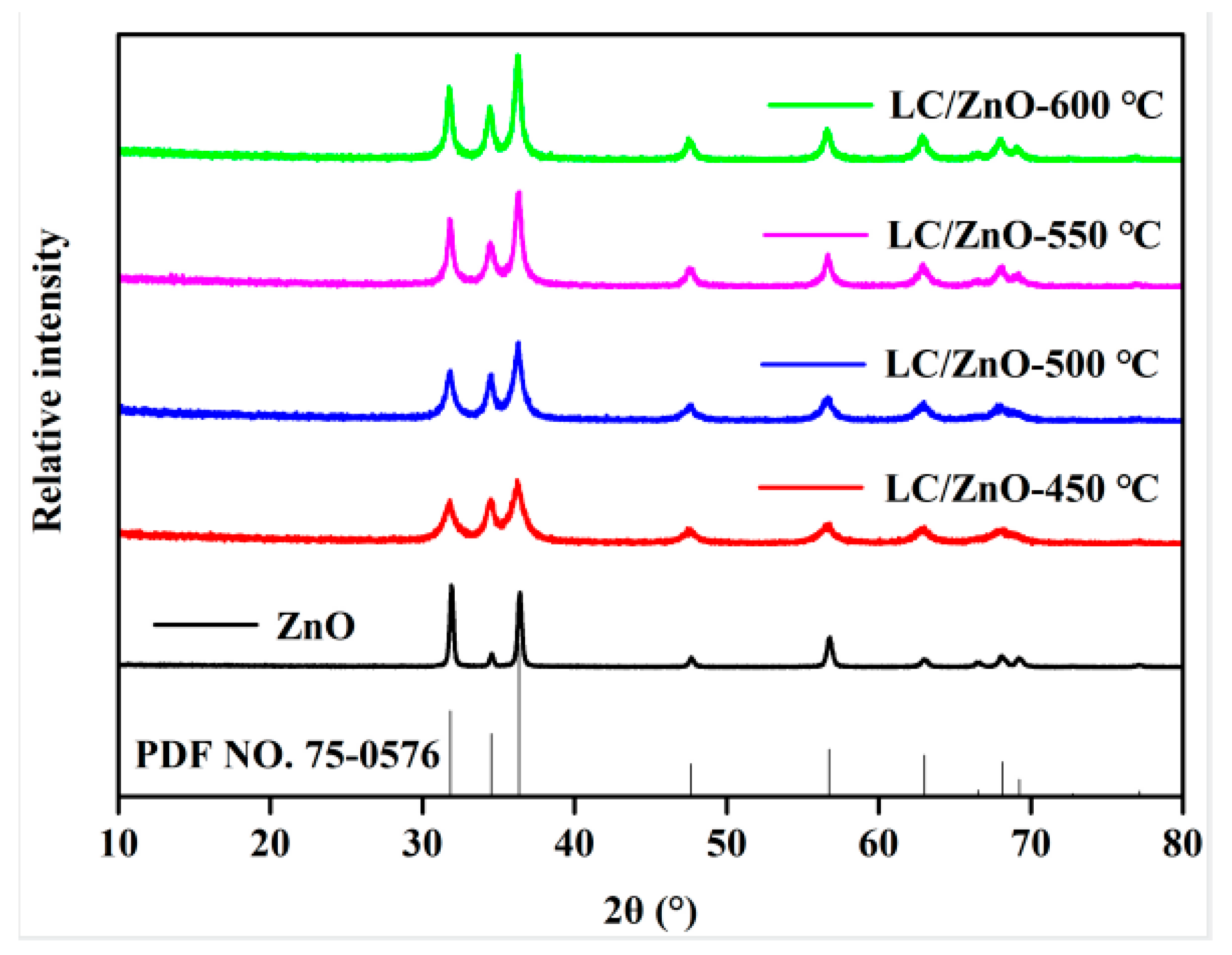
2.1. XRD Characterization Analysis
2.2. FESEM Characterization Analysis
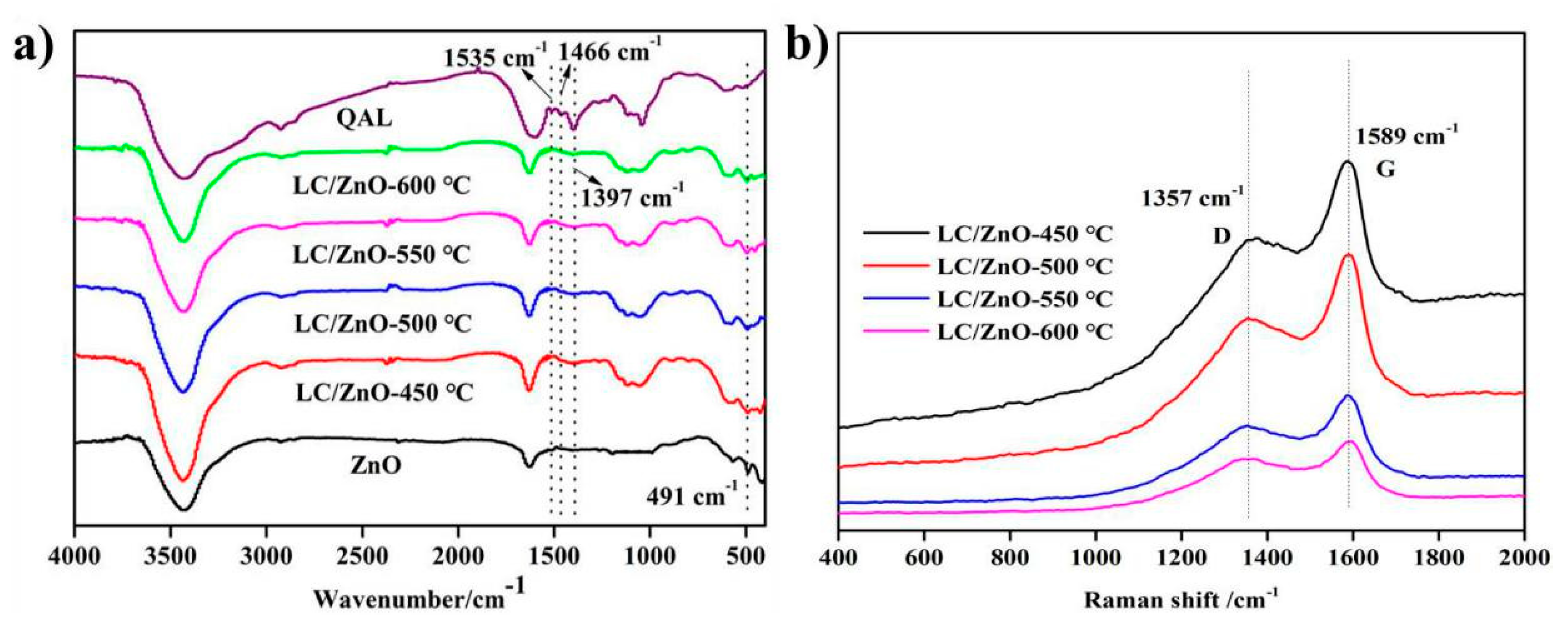
2.3. FT-IR and Raman Characterization Analysis
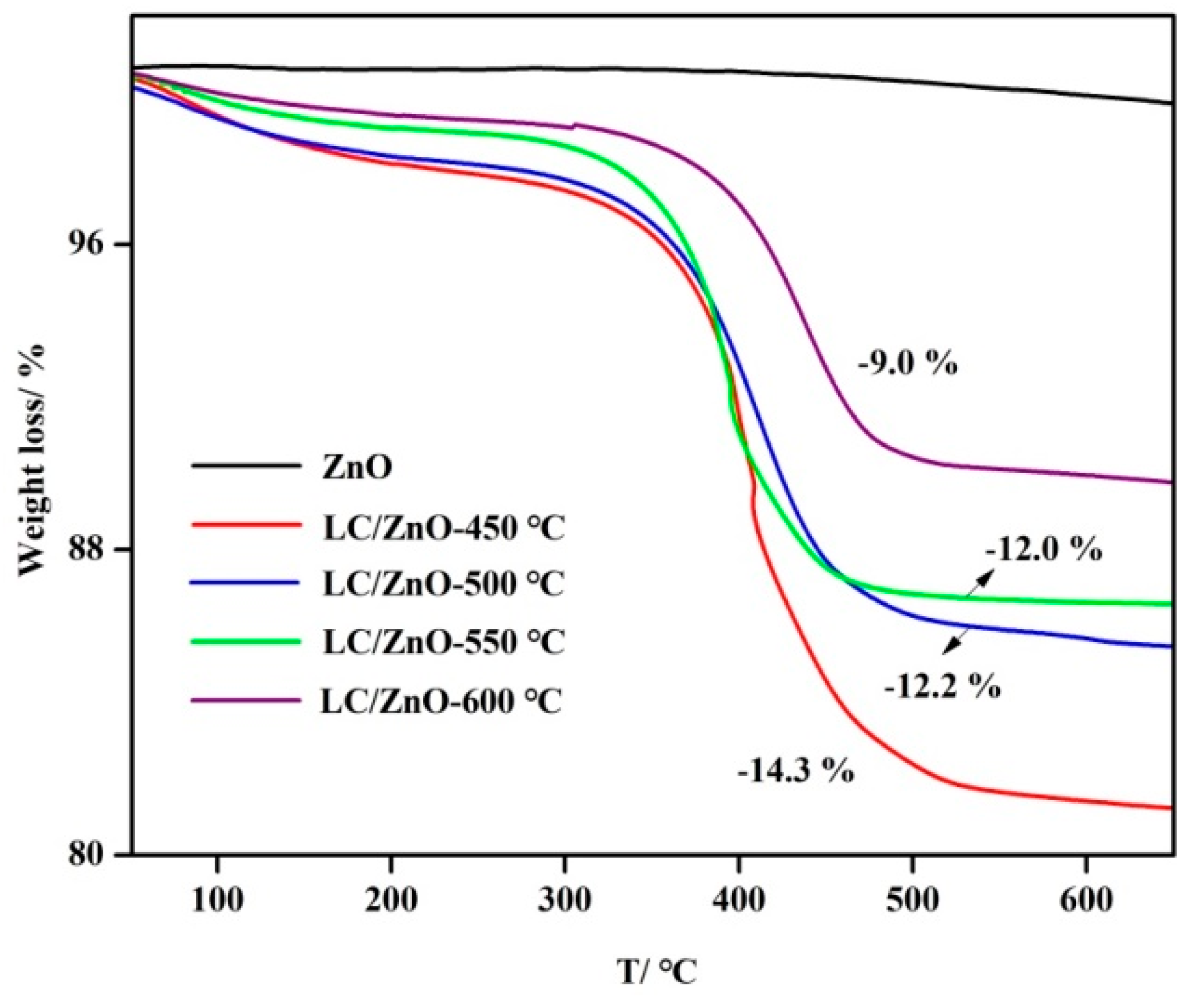
2.4. TG Characterization Analysis
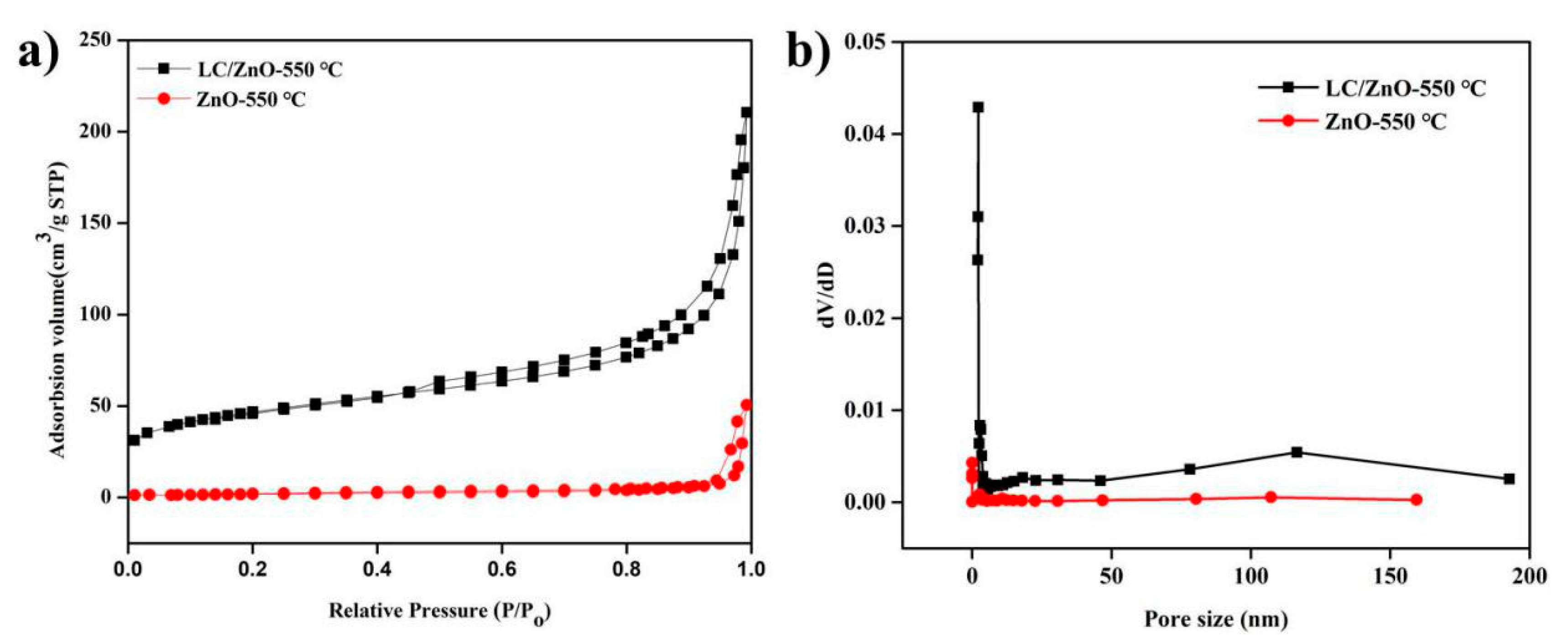
2.5. Characterization and Analysis of N2 Adsorption-Desorption
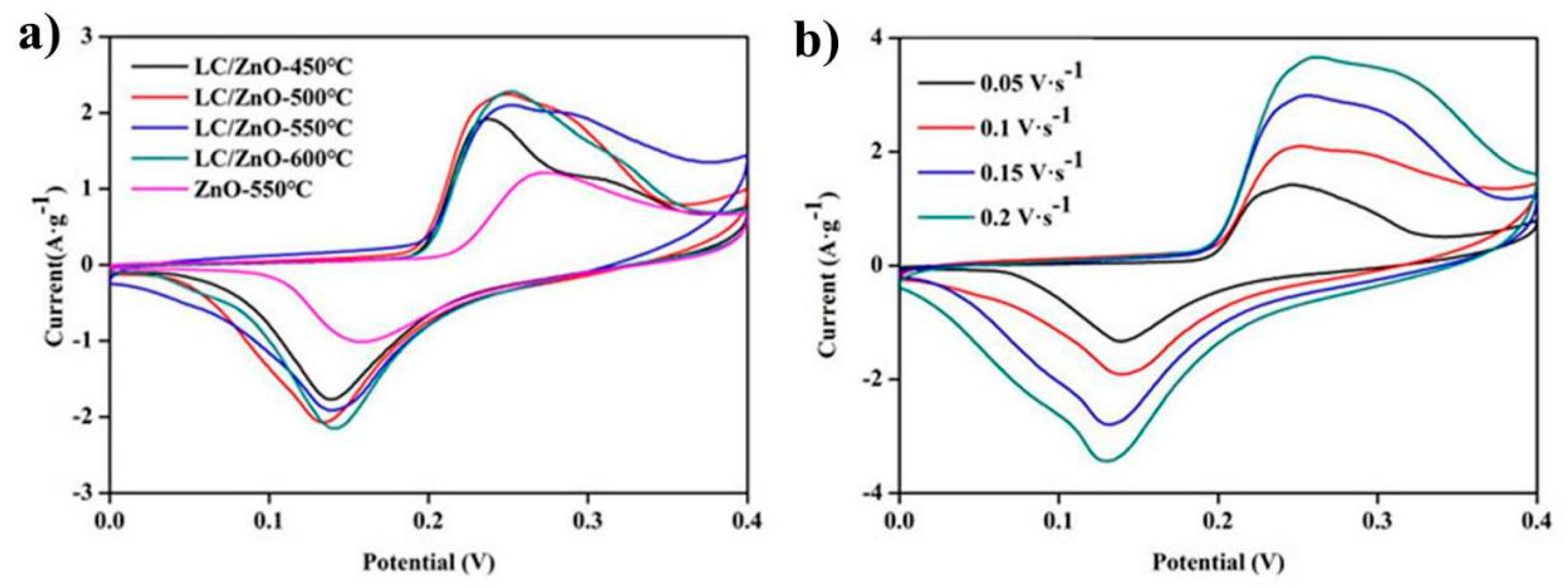
2.6. Cyclic Voltammetry
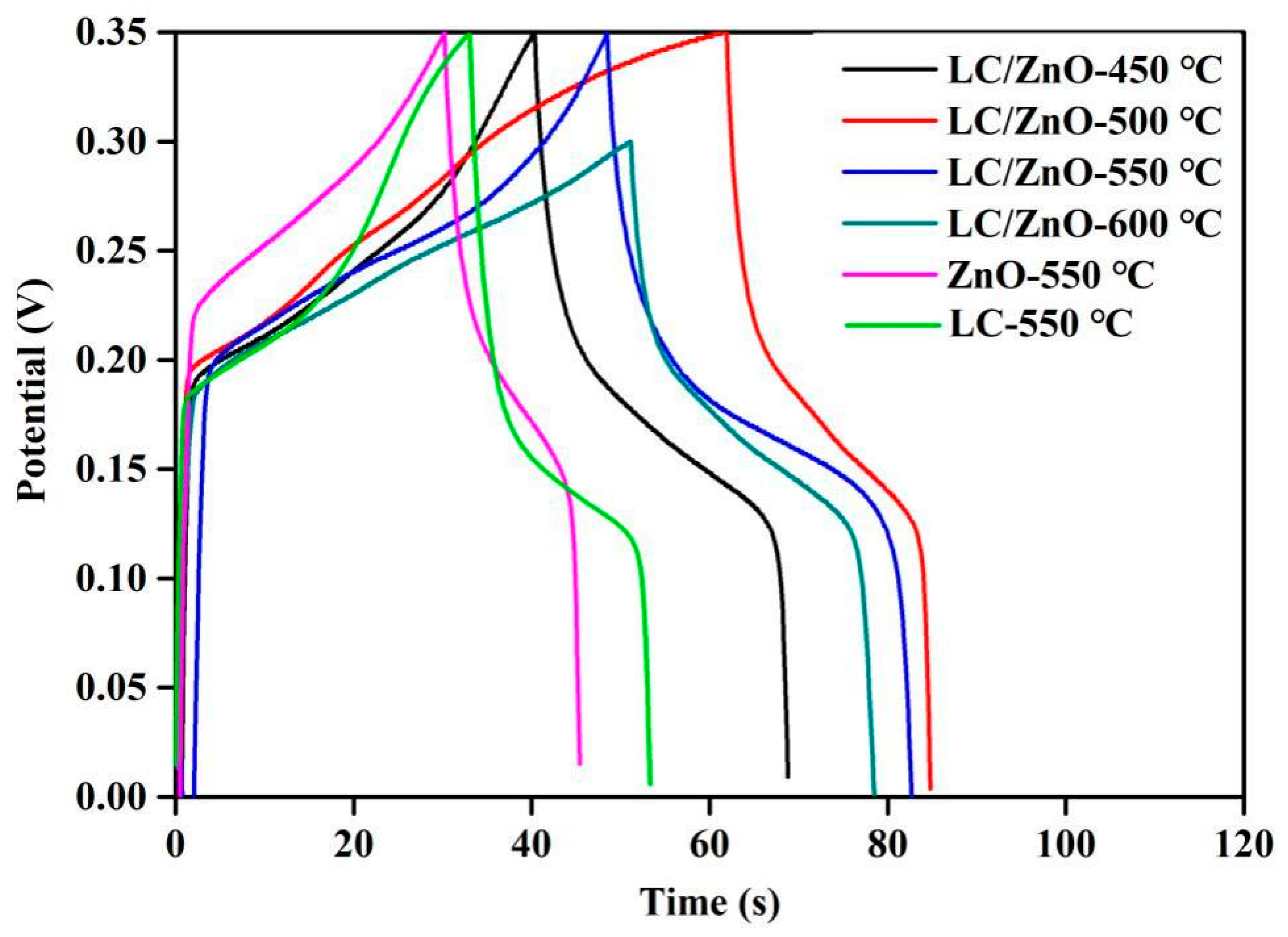
2.7. Galvanostatic Charge/Discharge
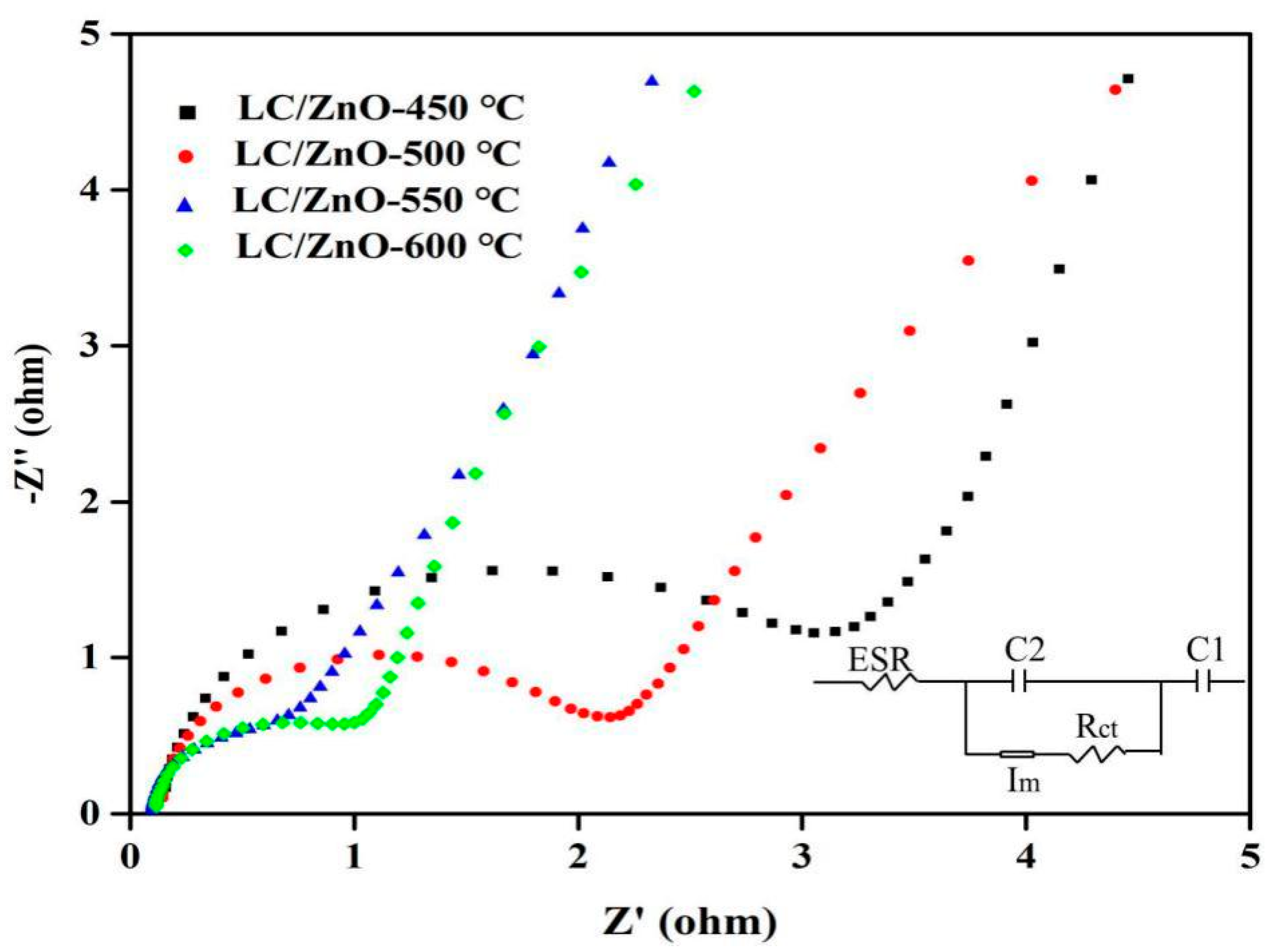
2.8. Electrochemical Impedance Spectroscopy
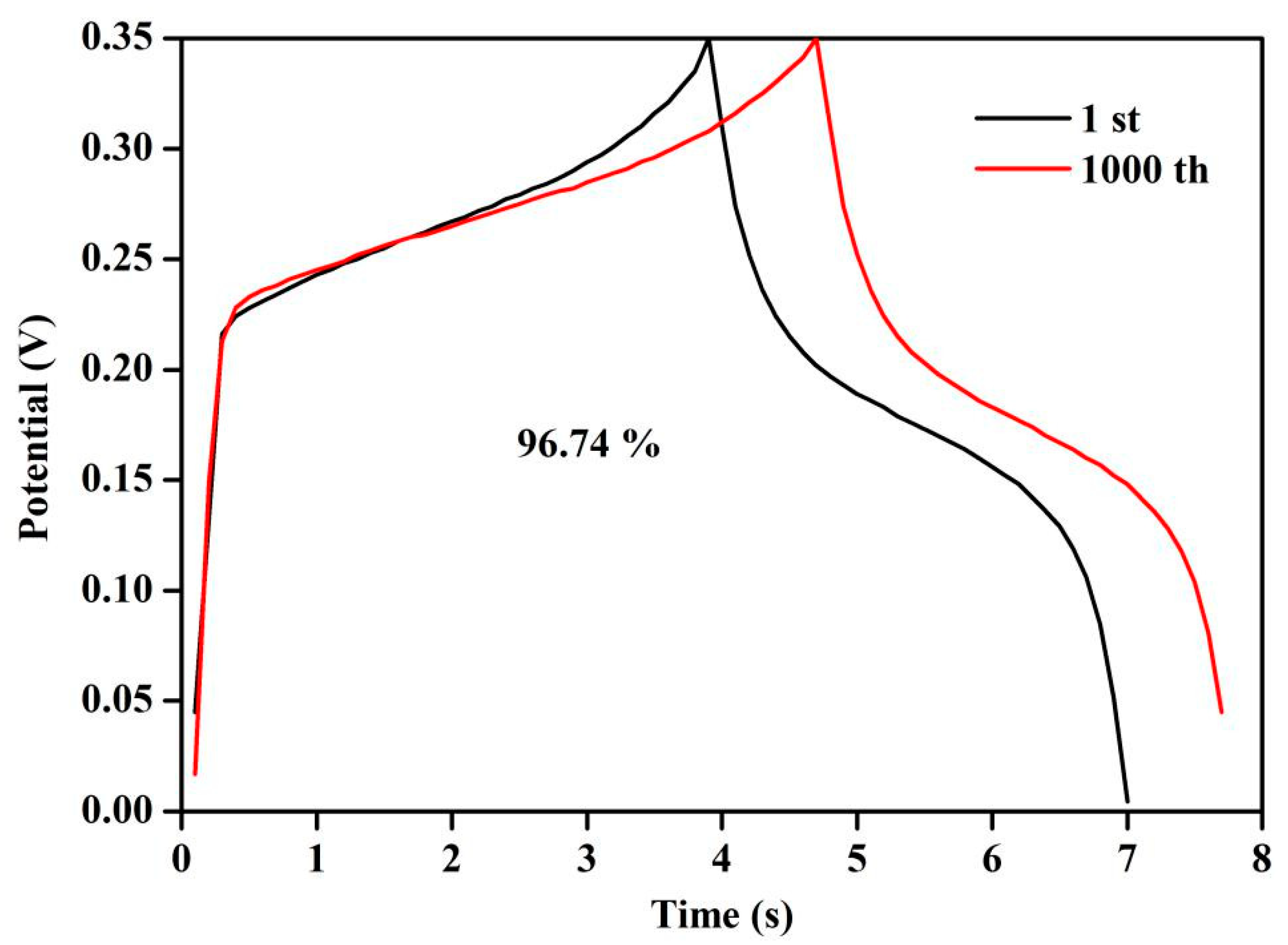
2.9. Cycle Performance Test
2.10. Mechanism Analysis of Electrochemical Performance
3. Materials and Methods
3.1. Preparation Method of Composite Material
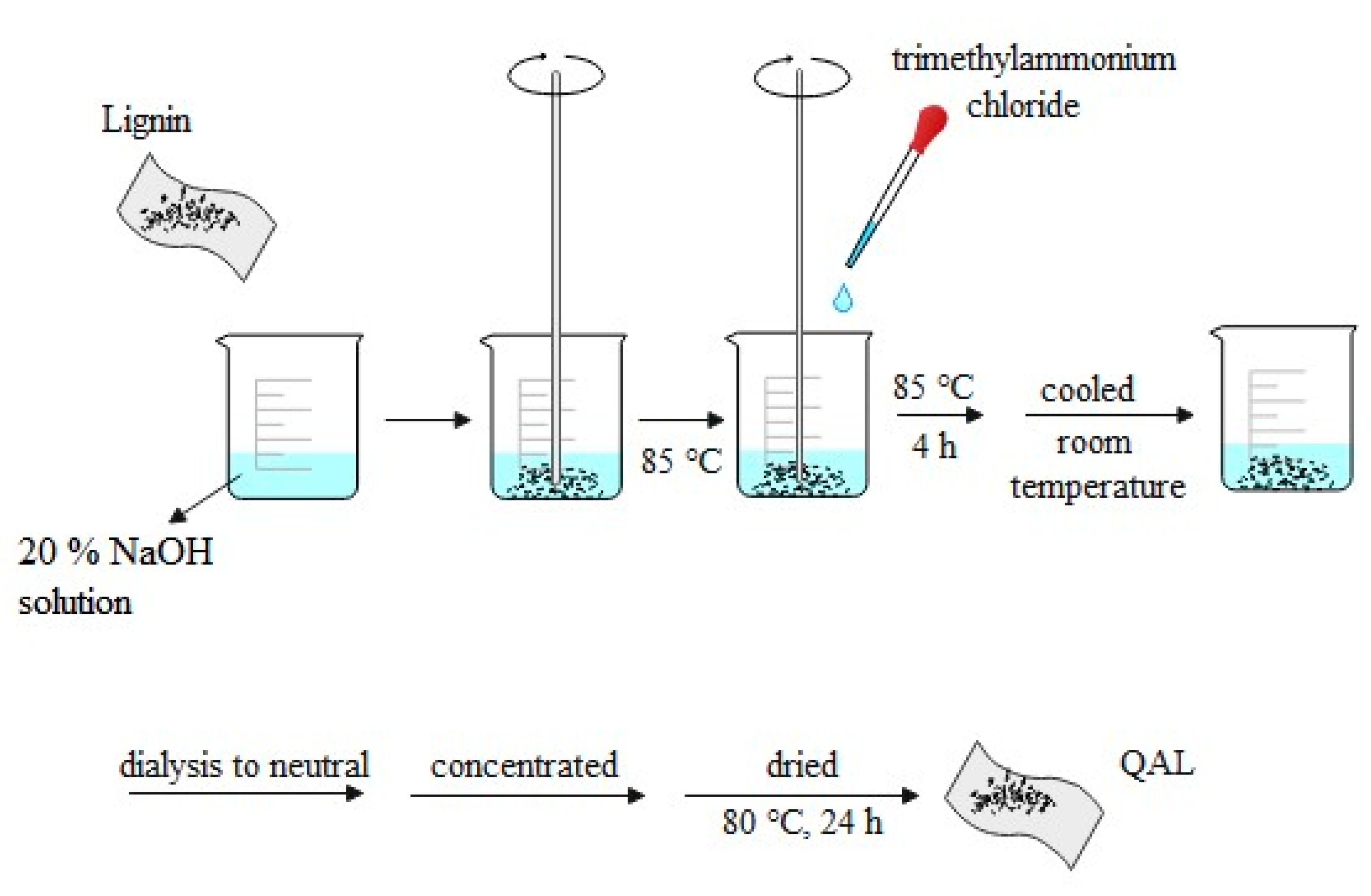
3.1.1. Activation of Alkali Lignin
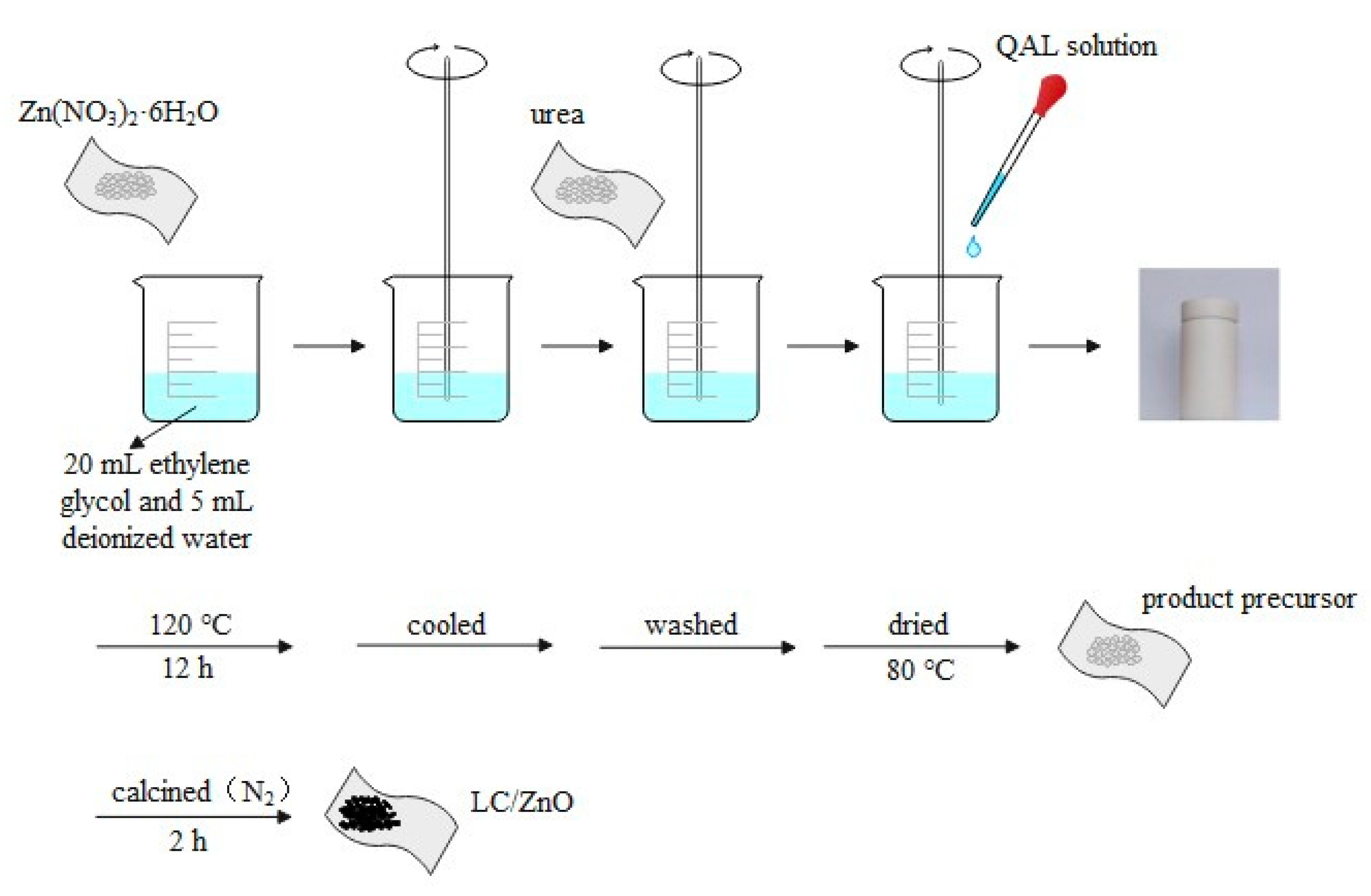
3.1.2. Preparation of Lignin Carbon/Zinc Oxide Composite Material
3.2. Characterization of Composite Materials
3.3. Electrochemical Performance Test
3.3.1. Production of Electrode Materials
3.3.2. Cyclic Voltammetry Curve Test
3.3.3. Determination of Specific Capacitance
3.3.4. Simulation of Equivalent Circuit and Determination of Dynamic Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Saito, T.; Brown, R.H.; Hunt, M.A.; Pickel, D.L.; Pickel, J.M.; Messman, J.M.; Baker, F.S.; Keller, M.; Naskar, A.K. Turning renewable resources into value-added polymer: Development of lignin-based thermoplastic. Green Chem. 2012, 14, 3295–3303. [Google Scholar] [CrossRef]
- Doherty, W.O.S.; Mousavioun, P.; Fellows, C.M. Value-adding to cellulosic ethanol: Lignin polymers. Ind. Crop. Prod. 2011, 33, 259–276. [Google Scholar] [CrossRef]
- Stewart, D. Lignin as a base material for materials applications: Chemistry, application and economics. Ind. Crop. Prod. 2008, 27, 202–207. [Google Scholar] [CrossRef]
- Xie, K.Y.; Wei, B.Q. Materials and Structures for Stretchable Energy Storage and Conversion Devices. Adv. Mater. 2017, 26, 3592–3617. [Google Scholar] [CrossRef]
- Daffos, B.; Taberna, P.L.; Gogotsi, Y.; Simon, P. Recent Advances in Understanding the Capacitive Storage in Microporous Carbons. Fuel Cells 2010, 10, 819–824. [Google Scholar] [CrossRef]
- Burke, A. R&D considerations for the performance and application of electrochemical capacitors. Electrochim. Acta 2007, 53, 1083–1091. [Google Scholar]
- Dubal, D.P.; Patil, S.V.; Gund, G.S.; Lokhande, C.D. Polyaniline-polypyrrole nanograined composite via electrostatic adsorption for high performance electrochemical supercapacitors. J. Alloys Compd. 2013, 552, 240–247. [Google Scholar] [CrossRef]
- Li, S.M.; Wang, Y.S.; Yang, S.Y.; Liu, C.H.; Chang, K.H.; Tien, H.W.; Wen, N.T.; Ma, C.C.M.; Hu, C.C. Electrochemical deposition of nanostructured manganese oxide on hierarchically porous grapheme-carbon nanotube structure for ultrahigh-performance electrochemical capacitors. J. Power Sources 2013, 225, 347–355. [Google Scholar] [CrossRef]
- Hou, Y.; Chen, L.Y.; Zhang, L.; Kang, J.; Fujita, T.; Jiang, J.; Chen, M. Ultrahigh capacitance of nanoporous metal enhanced conductive polymer pseudocapacitors. J. Power Sources 2013, 225, 304–310. [Google Scholar] [CrossRef]
- Islam, M.S.; Fisher, C.A.J. Lithium and sodium battery cathode materials: Computational insights into voltage, diffusion and nanostructural properties. Chem. Soc. Rev. 2014, 43, 185–204. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.S.; Parvez, K.; Feng, X.L. Graphene-based in-plane micro-supercapacitors with high power and energy densities. Nat. Commun. 2013, 4, 2487. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.B.; Yang, D.J.; Wang, H.; Qian, Y.; Yuan, F.; Zhong, J.; Qiu, X. Three-dimensional Porous Framework Lignin-Derived Carbon/ZnO Composite Fabricated by a Facile Electrostatic Self-Assembly Showing Good Stability for High-Performance Supercapacitors. ACS Sustain. Chem. Eng. 2019, 7, 16419–16427. [Google Scholar] [CrossRef]
- Yang, S.H.; Liu, Y.Y.; Hao, Y.F.; Yang, X.P.; Goddard, W.A.; Zhang, X.L.; Cao, B.Q. Oxygen-Vacancy Abundant Ultrafine Co3O4/Graphene Composites for High-Rate Supercapacitor Electrodes. Adv. Sci. 2018, 5, 1700659. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Chen, J.Z.; Zhou, Y.Y.; Song, C.; Tian, Q.H.; Xu, J.L.; Wong, C.P. Enhancing pseudocapacitive kinetics of nanostructured MnO2 through anchoring onto biomass-derived porous carbon. Appl. Surf. Sci. 2018, 440, 1027–1036. [Google Scholar] [CrossRef]
- Zhang, Y.P.; Sun, X.W.; Pan, L.K.; Li, H.B.; Sun, Z.; Sun, C.P.; Tay, B.K. Carbon nanotube−ZnO nanocomposite electrodes for supercapacitors. Solid State Ion. 2009, 180, 1525–1528. [Google Scholar] [CrossRef]
- Poonam Sharma, K.; Arora, A.; Tripathi, S.K. Review of supercapacitors: Materials and devices. J. Energy Storage 2019, 21, 801–825. [Google Scholar] [CrossRef]
- Lach, J.; Wróbel, K.; Wróbel, J.; Czerwinski, A. Applications of Carbon in Rechargeable Electrochemical Power Sources: A Review. Energies 2021, 14, 2649. [Google Scholar] [CrossRef]
- Majumder, M.; Choudhary, R.B.; Thakur, A.K. Hemispherical nitrogen-doped carbon spheres integrated with polyindole as high performance electrode material for supercapacitor applications. Carbon 2019, 142, 650–661. [Google Scholar] [CrossRef]
- Thakur, A.K.; Choudhary, R.B.; Majumder, M.; Gupta, G. In-Situ Integration of Waste Coconut Shell Derived Activated Carbon/Polypyrrole/Rare Earth Metal Oxide (Eu2O3): A Novel Step Towards Ultrahigh Volumetric Capacitance. Electrochim. Acta 2017, 251, 532–545. [Google Scholar] [CrossRef]
- Xu, S.W.; Zhao, Y.Q.; Xu, Y.X.; Chen, Q.H.; Zhang, G.Q.; Xu, Q.Q.; Zhao, D.D.; Zhang, X.; Xu, C.L. Heteroatom doped porous carbon sheets derived from protein-rich wheat gluten for supercapacitors: The synergistic effect of pore properties and heteroatom on the electrochemical performance in different electrolytes. J. Power Sources 2018, 401, 375–385. [Google Scholar] [CrossRef]
- Sevilla, M.; Ferrero, G.V.; Fuertes, A.B. Beyond KOH activation for the synthesis of superactivated carbons from hydrochar. Carbon 2017, 114, 50–58. [Google Scholar] [CrossRef]
- Qiu, D.P.; Guo, N.N.; Gao, A.; Zheng, L.; Xu, W.J.; Li, M.; Wang, F.; Yang, R. Preparation of oxygen-enriched hierarchically porous carbon by KMnO4 one-pot oxidation and activation: Mechanism and capacitive energy storage. Electrochim. Acta 2019, 294, 398–405. [Google Scholar] [CrossRef]
- Qiu, Z.P.; Wang, Y.S.; Bi, X.; Zhou, T.; Zhou, J.; Zhao, J.; Miao, Z.; Yi, W.; Fu, P.; Zhuo, S. Biochar-based carbons with hierarchical micro-meso-macro porosity for high rate and long cycle life supercapacitors. J. Power Sources 2018, 376, 82–90. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Fan, L.; Tao, Y.R.; Wang, W.; Wu, X.C.; Zhao, J.W. Pomelo Peel Derived Hierarchical Porous Carbon as Electrode Materials for High-Performance Supercapacitor. Chin. J. Inorg. Chem. 2018, 34, 1249–1260. [Google Scholar]
- Li, Y.M.; Liu, X. Activated carbon/ZnO composites prepared using hydrochars as intermediate and their electrochemical performance in supercapacitor. Mater. Chem. Phys. 2014, 148, 380–386. [Google Scholar] [CrossRef]
- Yun, S.I.; Kim, S.H.; Kim, D.W.; Kim, Y.A.; Kim, B.H. Facile preparation and capacitive properties of low-cost carbon nanofibers with ZnO derived from lignin and pitch as supercapacitor electrodes. Carbon 2019, 149, 637–645. [Google Scholar] [CrossRef]
- Chen, F.; Zhou, W.J.; Yao, H.F.; Fan, P.; Yang, J.T.; Fei, Z.D.; Zhong, M.Q. Self-assembly of NiO nanoparticles in lignin-derived mesoporous carbons for supercapacitor applications. Green Chem. 2013, 15, 3057–3063. [Google Scholar] [CrossRef]
- Lin, W.S.; Wang, H.; Yang, D.J.; Qiu, X.Q. Preparation of Lignin Carbon/Zinc Oxide Composite Material and Its Photocatalytic Performance. J. Chem. Eng. Chin. Univ. 2018, 32, 636–645. [Google Scholar]
- Wang, H.; Yi, G.; Zu, X.; Qin, P.; Tan, M.; Luo, H. Photoelectric characteristics of the p-n junction between ZnOnanorods and polyaniline nanowires and their application as a UV photodetector. Mater. Lett. 2016, 162, 83–86. [Google Scholar] [CrossRef]
- Demir, M.; Farghaly, A.A.; Decuir, M.J.; Collinson, M.M.; Gupta, R.B. Supercapacitance and Oxygen Reduction Characteristics of Sulfur Self-Doped Micro/Mesoporous Bio-Carbon Derived from Lignin. Mater. Chem. Phys. 2018, 216, 508–516. [Google Scholar] [CrossRef]
- Ma, S.Y.; Bu, J.Y.; Duan, Y.; Hou, K.H. Deactivation of Bulk Ni-Mo Catalysts in Selective Hydrodeoxygenation of Sulfolane. J. Chem. Eng. Chin. Univ. 2017, 31, 863–869. [Google Scholar]
- Tian, W.Q.; Gao, Q.M.; Tan, Y.L.; Yang, K.; Zhu, L.; Yang, C.; Zhang, H. Bio-inspired beehive-like hierarchical nanoporous carbon derived from bamboo-based industrial by-product as a high performance supercapacitor electrode material. J. Mater. Chem. A 2015, 3, 5656–5664. [Google Scholar] [CrossRef]

| Composites | LC/ZnO-450 °C | LC/ZnO-500 °C | LC/ZnO-550 °C | LC/ZnO-600 °C |
|---|---|---|---|---|
| Energy density(W·s/kg) | 3.98 | 4.96 | 6.19 | 5.64 |
| Power density(W/kg) | 0.173 | 0.177 | 0.177 | 0.176 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, G.; Zhou, Z.; Li, J.; Yan, H.; Li, F. Preparation of Lignin Carbon/Zinc Oxide Electrode Material and Its Application in Supercapacitors. Molecules 2021, 26, 3554. https://doi.org/10.3390/molecules26123554
Guo G, Zhou Z, Li J, Yan H, Li F. Preparation of Lignin Carbon/Zinc Oxide Electrode Material and Its Application in Supercapacitors. Molecules. 2021; 26(12):3554. https://doi.org/10.3390/molecules26123554
Chicago/Turabian StyleGuo, Gaijuan, Zijing Zhou, Jinda Li, Hong Yan, and Fen Li. 2021. "Preparation of Lignin Carbon/Zinc Oxide Electrode Material and Its Application in Supercapacitors" Molecules 26, no. 12: 3554. https://doi.org/10.3390/molecules26123554
APA StyleGuo, G., Zhou, Z., Li, J., Yan, H., & Li, F. (2021). Preparation of Lignin Carbon/Zinc Oxide Electrode Material and Its Application in Supercapacitors. Molecules, 26(12), 3554. https://doi.org/10.3390/molecules26123554





