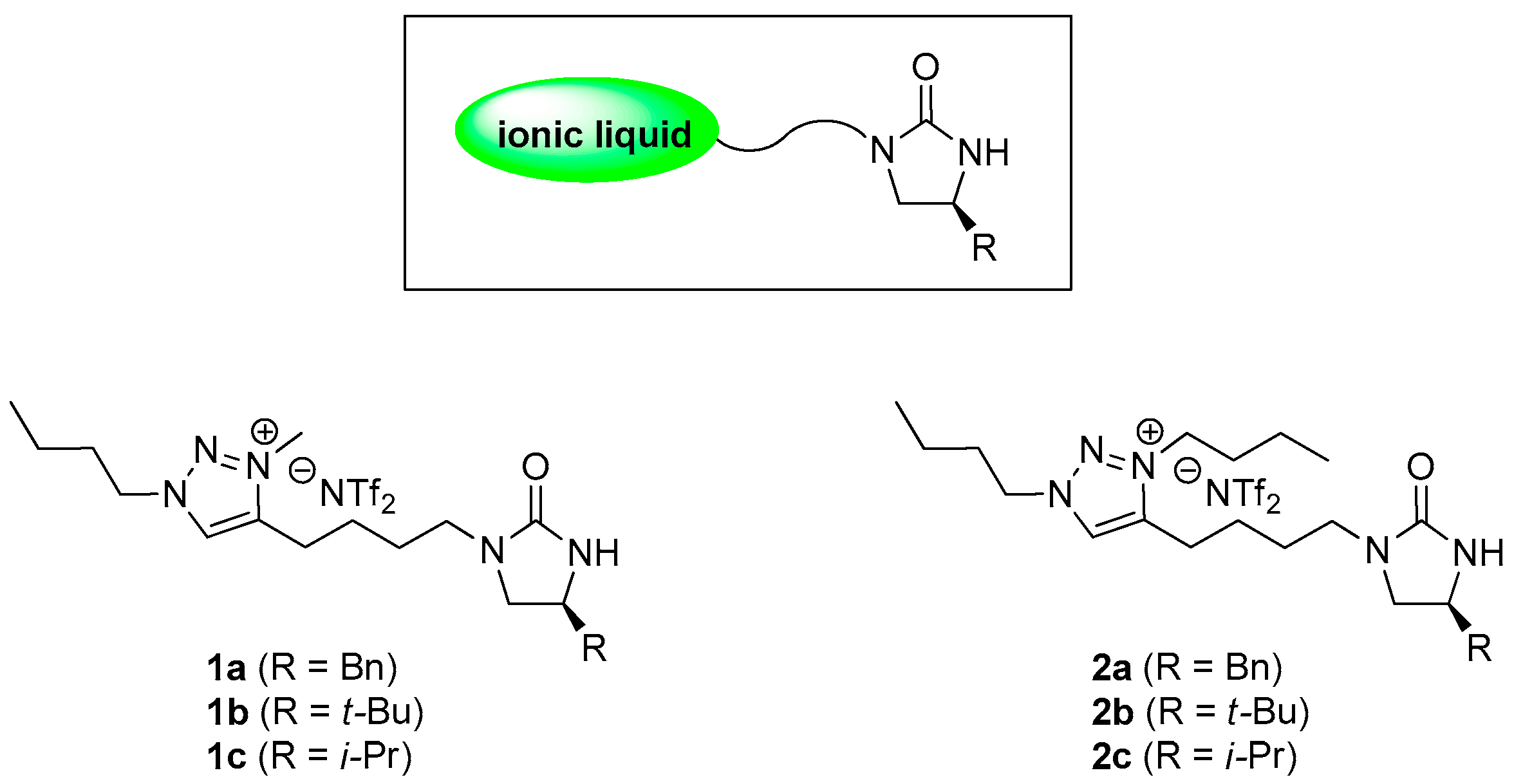
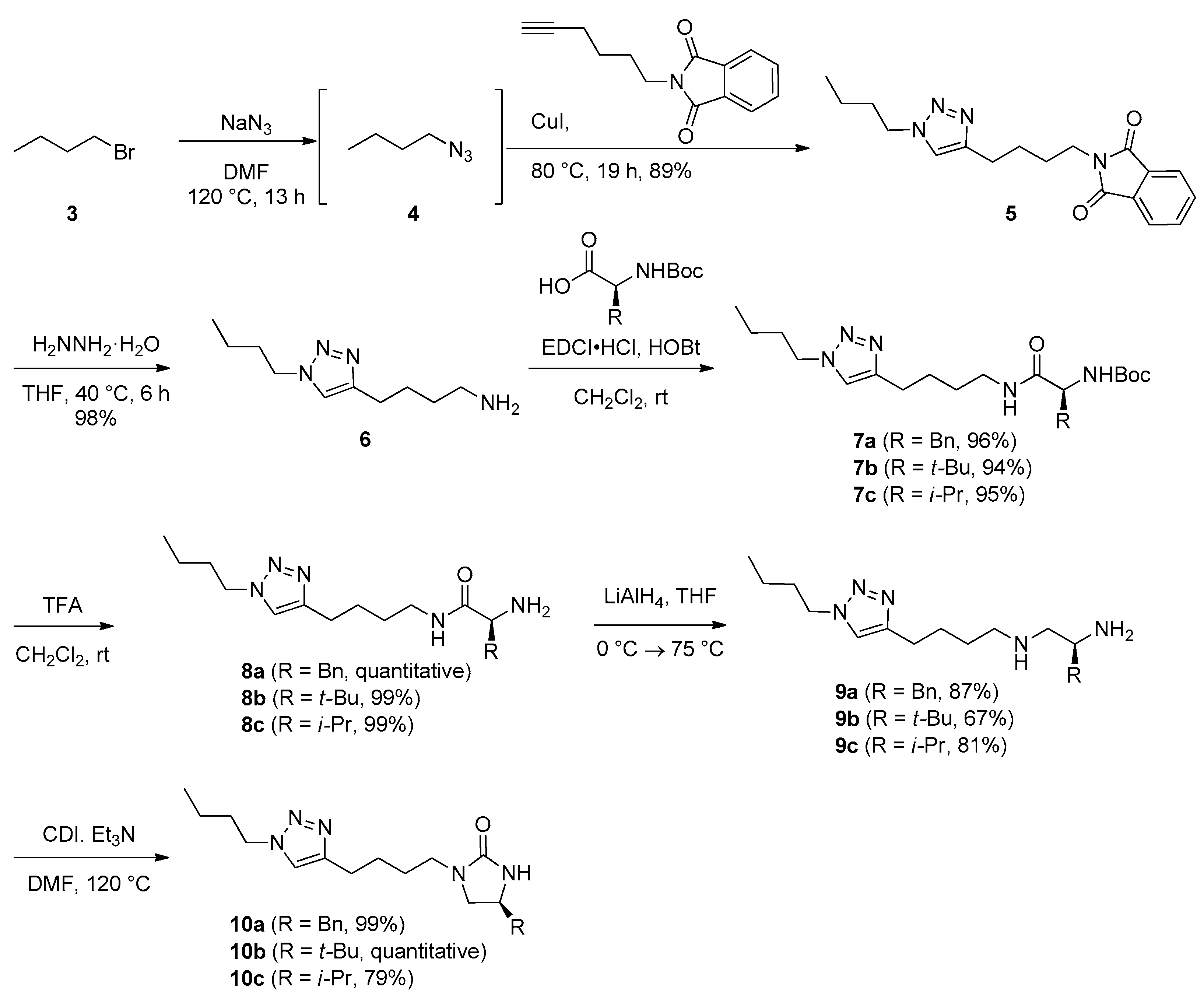
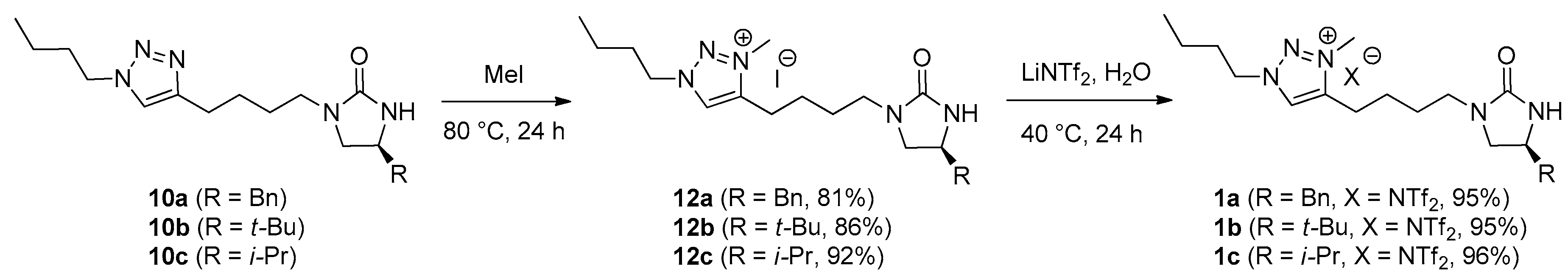
3.2. Synthesis
N-[4-(1-Butyl-1H-1,2,3-triazol-4-yl)butyl]phthalimide (5). In a screw-cap vial, a mixture of 1-bromobutane (3) (800 µL, 7.29 mmol) and sodium azide (949 mg, 14.6 mmol) in anhydrous DMF (11 mL) was stirred at 120 °C for 14 h. After the mixture was cooled to rt, N-(5-hexynyl)phthalimide (1.66 g, 7.29 mmol) and CuI (139 mg, 730 μmol) were added, and the solution was stirred at 80 °C for 19 h. Upon completion of the reaction, the reaction mixture was cooled to rt and concentrated in vacuo. The purification by column chromatography on silica gel (3:1 hexanes/EtOAc) afforded a desired triazole 5 (2.13 g, 89%) as a light-yellow solid. TLC: Rf 0.21 (3:1 hexanes/EtOAc). mp: 73.4–74.8 °C. 1H NMR (400 MHz, CDCl3): δ 7.84 (dd, J = 5.6, 2.8 Hz, 2H), 7.71 (dd, J = 5.6, 2.8 Hz, 2H), 7.28 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.72 (t, J = 6.8 Hz, 2H), 2.77 (t, J = 6.8 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.77–1.71 (m, 4H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 168.6, 147.6, 134.1, 132.3, 123.3, 120.7, 50.0, 37.8, 32.5, 28.3, 26.9, 25.3, 19.9, 13.6. LRMS (ESI) m/z (rel int): (pos) 327 ([M + H]+, 100), 282 ([M – C3H8]+, 100). HRMS (ESI) m/z calcd for C18H23N4O2+ ([M + H]+) 327.1816, found 327.1822.
4-(1-Butyl-1H-1,2,3-triazol-4-yl)butylamine (6). In a 100 mL roundbottom flask, phthalimide 5 (1.10 g, 3.38 mmol) was dissolved in anhydrous THF (5 mL) at rt. Hydrazine hydrate (1.41 mL, 28.9 mmol) was added and the mixture was stirred at 40 °C for 6 h. Upon completion of the reaction, the reaction mixture was diluted with Et2O (70 mL) and insoluble solid was filtered off. The filtrate was washed with aqueous 10% NaOH (3 mL) (pH 12), and dried over anhydrous Na2SO4, filtered, and concentrated in vacuo to yield analytically pure amine 6 as a colorless liquid (648 mg, 98%). TLC: Rf 0.02 (1:1 hexanes/EtOAc). 1H NMR (400 MHz, CDCl3): δ 7.26 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 2.73 (t, J = 7.6 Hz, 2H), 2.72 (t, J = 7.2 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.6 Hz, 2H), 1.52 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 147.9, 120.4, 49.8, 41.9, 33.3, 32.3, 26.7, 25.5, 19.7, 13.4. LRMS (ESI) m/z (rel int): (pos) 219 ([M + Na]+, 100), 180 ([M − NH2]+, 100). HRMS (ESI) m/z calcd for C10H20N4Na+ ([M + Na]+) 219.1580, found 219.1578.
(S)-tert-Butyl [1-((4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl)amino)-1-oxo-3-phenylpropan-2-yl]carbamate (7a). In a 50 mL roundbottom flask, amine 6 (261 mg, 1.33 mmol), Boc-l-Phe-OH (353 mg, 1.33 mmol), EDCI·HCl (286 mg, 1.46 mmol), and HOBt·hydrate (198 mg, 1.46 mmol) were dissolved in anhydrous CH2Cl2 (5.8 mL) at rt. The mixture was stirred at rt for 5 h. Upon completion of the reaction, 10% wt citric acid (11 mL) was added and the two layers were separated. The aqueous layer was extracted with CH2Cl2 (2 × 30 mL) and combined organic extracts were washed with saturated aqueous NaHCO3 (15 mL) and brine (15 mL) sequentially. It was then dried over anhydrous MgSO4, filtered, and concentrated in vacuo. The purification by column chromatography on silica gel (20:1 CH2Cl2/MeOH) afforded a desired amide 7a as a white foam (566 mg, 96%). TLC: Rf 0.19 (1:2 hexanes/EtOAc). mp: 98.8–100.8 °C. = +4.2 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.30–7.19 (m, 6H), 5.82 (br s, 1H), 5.05 (br s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 4.27 (q, J = 7.6 Hz, 1H), 3.19 (q, J = 6.4 Hz, 2H), 3.07 (dd, J = 13.6, 6.4 Hz, 1H), 3.02 (dd, J = 13.6, 7.6 Hz, 1H), 2.67 (t, J = 7.6 Hz, 2H), 1.88 (quintet, J = 7.6 Hz, 2H), 1.58 (quintet, J = 7.2 Hz, 2H), 1.45 (m, 2H), 1.40 (s, 9H), 1.36 (sextet, J = 7.2 Hz, 2H), 0.96 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 171.3, 155.6, 147.7, 137.0, 129.5, 128.8, 127.1, 120.7, 80.3, 56.2, 50.1, 39.3, 39.0, 32.5, 28.9, 28.4, 26.7, 25.2, 19.9, 13.7. LRMS (ESI) m/z (rel int): (pos) 444 ([M + H]+, 100), 388 ([M − C4H7]+, 75). HRMS (ESI) m/z calcd for C24H38N5O3+ ([M + H]+) 444.2969, found 444.2977.
(R)-tert-Butyl [1-((4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl)amino)-1-oxo-3-phenylpropan-2-yl]carbamate (7a′).7a′ was synthesized according to the procedure described for the synthesis of 7a. Amine 6 (719 mg, 3.66 mmol), Boc-d-Phe-OH (991 mg, 3.66 mmol), EDCI·HCl (772 mg, 4.03 mmol), and HOBt·hydrate (544 mg, 4.03 mmol) were used. Reaction time: 6 h. Yield: 1.60 g, 98%. White solid. TLC: Rf 0.19 (1:2 hexanes/EtOAc). mp: 107.1–109.1 °C. = −5.7 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.30–7.19 (m, 6H), 5.89 (br s, 1H), 5.08 (br s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 4.28 (q, J = 7.6 Hz, 1H), 3.19 (q, J = 6.8 Hz, 2H), 3.07 (dd, J = 13.6, 6.4 Hz, 1H), 3.02 (dd, J = 13.6, 7.6 Hz, 1H), 2.67 (t, J = 7.2 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.59 (quintet, J = 7.6 Hz, 2H), 1.44 (m, 2H), 1.40 (s, 9H), 1.36 (sextet, J = 7.2 Hz, 2H), 0.96 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 171.0, 155.4, 147.5, 136.9, 129.3, 128.6, 126.9, 120.5, 80.1, 56.1, 49.9, 39.1, 38.8, 32.3, 28.8, 28.3, 26.5, 25.1, 19.7, 13.5. LRMS (ESI) m/z (rel int): (pos) 444 ([M + H]+, 100), 388 ([M − C4H7]+, 23). HRMS (ESI) m/z calcd for C24H38N5O3+ ([M + H]+) 444.2969, found 444.2969.
(S)-tert-Butyl [1-((4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl)amino)-3,3-dimethyl-1-oxobutan-2-yl]carbamate (7b).7b was synthesized according to the procedure described for the synthesis of 7a. Amine 6 (813 mg, 4.14 mmol), Boc-tert-Leu-OH (931 mg, 3.95 mmol), EDCI·HCl (832 mg, 4.34 mmol), and HOBt·hydrate (587 mg, 4.34 mmol) were used. Reaction time: 14 h. Yield: 1.52 g, 94%. Colorless oil. TLC: Rf 0.43 (10:1 CH2Cl2/MeOH). = +1.3 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.27 (s, 1H), 5.78 (br s, 1H), 5.25 (br d, J = 8.8 Hz, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.75 (br d, J = 9.2 Hz, 1H), 3.29 (m, 2H), 2.73 (t, J = 7.2 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.2 Hz, 2H), 1.59 (m, 2H), 1.43 (s, 9H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.98 (s, 9H), 0.96 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 170.9, 155.8, 147.5, 120.6, 79.6, 62.5, 49.9, 39.1, 34.5, 32.3, 28.9, 28.3, 26.7, 26.6, 25.1, 19.7, 13.5. LRMS (ESI) m/z (rel int): (pos) 410 ([M + H]+, 100), 354 ([M – C4H7]+, 22). HRMS (ESI) m/z calcd for C21H40N5O3+ ([M + H]+) 410.3126, found 410.3129.
(S)-tert-Butyl [1-((4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl)amino)-3-methyl-1-oxobutan-2-yl]carbamate (7c).7c was synthesized according to the procedure described for the synthesis of 7a. Amine 6 (690 mg, 3.51 mmol), Boc-l-Val-OH (727 mg, 3.35 mmol), EDCI·HCl (706 mg, 3.68 mmol), and HOBt·hydrate (498 mg, 3.68 mmol) were used. The crude mixture was purified by column chromatography on silica gel (30:1 CH2Cl2/MeOH). Reaction time: 8 h. Yield: 1.26 g, 95%. White solid. TLC: Rf 0.52 (10:1 CH2Cl2/MeOH). mp: 81.5–83.5 °C. = −15.4 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.27 (s, 1H), 6.03 (br s, 1H), 5.02 (br s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.84 (dd, J = 8.8, 6.0 Hz, 1H), 3.29 (q, J = 6.8 Hz, 2H), 2.73 (t, J = 7.6 Hz, 2H), 2.14 (m, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.6 Hz, 2H), 1.59 (m, 2H), 1.44 (s, 9H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.96 (t, J = 7.2 Hz, 3H), 0.95 (d, J = 6.8 Hz, 3H), 0.90 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 171.6, 155.9, 147.6, 120.6, 79.8, 60.1, 49.9, 39.1, 32.3, 30.9, 28.9, 28.3, 26.7, 25.1, 19.7, 19.3, 17.8, 13.5. LRMS (ESI) m/z (rel int): (pos) 396 ([M + H]+, 100), 340 ([M − C4H7]+, 45). HRMS (ESI) m/z calcd for C20H38N5O3+ ([M + H]+) 396.2969, found 396.2961.
(S)-2-Amino-N-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]-3-phenylpropanamide (8a). In a 250 mL roundbottom flask, Boc protected amine 7a (1.87 g, 4.21 mmol) was dissolved in CH2Cl2 (33 mL) at rt under air. A solution of TFA in CH2Cl2 (20% v/v, 48 mL) was added dropwise for 2 h. Upon completion of the reaction, the reaction mixture was diluted with CH2Cl2 (30 mL), cooled to 0 °C, and basified with saturated aqueous NaHCO3 (800 mL). The aqueous layer was extracted with CH2Cl2 (3 × 100 mL). The combined organic extracts were then dried over anhydrous Na2SO4, filtered, and concentrated in vacuo to yield analytically pure amine 8a as a white solid (1.52 g, quantitative yield). TLC: Rf 0.65 (5:1 CH2Cl2/MeOH). mp: 57.2–59.2 °C. = −44.3 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.33–7.21 (m, 6H), 4.31 (t, J = 7.2 Hz, 2H), 3.62 (dd, J = 9.2, 4.0 Hz, 1H), 3.31–3.25 (m, 3H), 2.73 (t, J = 7.2 Hz, 2H), 2.71 (dd, J = 13.2, 9.2 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.70 (quintet, J = 7.2 Hz, 2H), 1.57 (m, 2H), 1.36 (sextet, J = 7.2 Hz, 2H), 0.96 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 174.3, 147.8, 138.1, 129.4, 128.8, 126.9, 120.7, 56.6, 50.0, 41.2, 38.9, 32.4, 29.2, 26.9, 25.3, 19.9, 13.6. LRMS (ESI) m/z (rel int): (pos) 344 ([M + H]+, 100), 180 ([M − n-Bu − NH2 − Bn]+, 33). HRMS (ESI) m/z calcd for C19H30N5O+ ([M + H]+) 344.2445, found 344.2442.
(R)-2-Amino-N-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]-3-phenylpropanamide(8a′).8a′ was synthesized according to the procedure described for the synthesis of 8a. Amine 7a′ (818 mg, 1.84 mmol) and a solution of TFA in anhydrous CH2Cl2 (20% v/v, 16 mL) were used. Reaction time: 1 h. Yield: 615 mg, 97%. White solid. TLC: Rf 0.65 (5:1 CH2Cl2/MeOH). mp: 62.5–64.5 °C. = −16.9 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.33–7.21 (m, 6H), 4.32 (t, J = 7.2 Hz, 2H), 3.59 (dd, J = 9.2, 4.0 Hz, 1H), 3.29 (t, J = 7.2 Hz, 2H), 3.27 (dd, J = 13.6, 4.0 Hz, 1H), 2.73 (t, J = 7.2 Hz, 2H), 2.68 (dd, J = 13.6, 9.2 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.70 (quintet, J = 7.2 Hz, 2H), 1.56 (m, 2H), 1.36 (sextet, J = 7.2 Hz, 2H), 0.96 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 174.2, 147.7, 138.0, 129.3, 128.7, 126.8, 120.6, 56.6, 49.9, 41.1, 38.8, 32.3, 29.1, 26.8, 25.2, 19.7, 13.5. LRMS (ESI) m/z (rel int): (pos) 344 ([M + H]+, 100), 180 ([M − C4H9 − NH2 − Bn]+, 37). HRMS (ESI) m/z calcd for C19H30N5O+ ([M + H]+) 344.2445, found 344.2445.
(S)-2-Amino-N-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]-3,3-dimethylbutanamide (8b). 8b was synthesized according to the procedure described for the synthesis of 8a. Amine 7b (1.16 g, 2.84 mmol) and a solution of TFA in anhydrous CH2Cl2 (20% v/v, 30 mL) were used. Reaction time: 1.5 h. Yield: 871 mg, 99%. White solid. TLC: Rf 0.17 (10:1 CH2Cl2/MeOH). mp: 45.0–47.0 °C. = +27.8 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.27 (s, 1H), 6.81 (br s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.29 (m, 2H), 3.09 (s, 1H), 2.74 (t, J = 7.2 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.73 (quintet, J = 7.2 Hz, 2H), 1.58 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.99 (s, 9H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 173.5, 147.6, 120.6, 64.4, 49.9, 38.7, 34.0, 32.3, 29.1, 26.8, 26.7, 25.2, 19.7, 13.5. LRMS (ESI) m/z (rel int): (pos) 310 ([M + H]+, 100), 180 ([M − n-Bu − NH2 − t-Bu]+, 37). HRMS (ESI) m/z calcd for C16H32N5O+ ([M + H]+) 310.2601, found 310.2605.
(S)-2-Amino-N-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]-3-methylbutanamide (8c).8c was synthesized according to the procedure described for the synthesis of 8a. Amine 7c (933 mg, 2.36 mmol) and a solution of TFA in anhydrous CH2Cl2 (20% v/v, 20 mL) were used. Reaction time: 2.5 h. Yield: 687 mg, 99%. Ivory solid. TLC: Rf 0.33 (10:1 CH2Cl2/MeOH). mp: 58.9–60.9 °C. = +5.9 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.36 (br s, 1H), 7.27 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.31 (m, 2H), 3.23 (d, J = 3.6 Hz, 1H), 2.74 (t, J = 7.6 Hz, 2H), 2.30 (m, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.73 (quintet, J = 7.6 Hz, 2H), 1.58 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.98 (d, J = 6.8 Hz, 3H), 0.95 (t, J = 7.2 Hz, 3H), 0.81 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 174.2, 147.7, 120.6, 60.2, 49.9, 38.7, 32.3, 30.8, 29.2, 26.8, 25.2, 19.8, 19.7, 16.1, 13.5. LRMS (ESI) m/z (rel int): (pos) 296 ([M + H]+, 100), 180 ([M − n-Bu − NH2 − i-Pr]+, 53). HRMS (ESI) m/z calcd for C15H30N5O+ ([M + H]+) 296.2445, found 296.2450.
(S)-N1-[4-(1-Butyl-1H-1,2,3-triazol-4-yl)butyl]-3-phenylpropane-1,2-diamine (9a). In a 100 mL roundbottom flask, amide 8a (1.40 g, 4.09 mmol) was dissolved in anhydrous THF (16 mL) at rt. The reaction mixture was cooled to 0 °C and LiAlH4 (931 mg, 24.5 mmol) was added. It was then stirred at 0 °C for 30 min and heated under reflux for 7 h. Upon completion of the reaction, the resulting grey suspension was cooled to 0 °C, quenched with saturated aqueous Na2SO4, and diluted with EtOAc (70 mL). The precipitate was filtered off, and the filtrate was concentrated by rotary evaporation. The residue was diluted with CH2Cl2 (100 mL), dried over anhydrous K2CO3, filtered, and concentrated in vacuo. The purification by column chromatography on silica gel (15:1 CH2Cl2/MeOH→10:1 CH2Cl2/MeOH→MeOH) afforded a desired diamine 9a as a pale yellow solid (1.17 g, 87%). TLC: Rf 0.04 (MeOH). mp: 38.1–40.1 °C. = +6.3 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.32−7.18 (m, 6H), 4.31 (t, J = 7.2 Hz, 2H), 3.09 (ddd, J = 13.6, 8.8, 4.8 Hz, 1H), 2.78 (dd, J = 13.6, 4.8 Hz, 1H), 2.73 (t, J = 7.2 Hz, 2H), 2.70 (dd, J = 11.6, 4.8 Hz, 1H), 2.64 (q, J = 7.2 Hz, 2H), 2.49 (dd, J = 13.6, 8.8 Hz, 1H), 2.45 (dd, J = 11.6, 8.8 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.2 Hz, 2H), 1.55 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CD3OD): δ 149.0, 140.2, 130.4, 129.6, 127.5, 123.1, 56.3, 53.3, 51.0, 50.5, 43.2, 33.4, 30.0, 28.3, 26.1, 20.7, 13.8. LRMS (ESI) m/z (rel int): (pos) 330 ([M + H]+, 100), 282 ([C15H32N5]+, 11), 180 ([M − C2H6N2 − Bn]+, 23). HRMS (ESI) m/z calcd for C19H32N5+ ([M + H]+) 330.2652, found 330.2659.
(R)-N1-[4-(1-Butyl-1H-1,2,3-triazol-4-yl)butyl]-3-phenylpropane-1,2-diamine(9a′).9a′ was synthesized according to the procedure described for the synthesis of 9a. Amide 8a′ (425 mg, 1.24 mmol) and LiAlH4 (299 mg, 7.88 mmol) were used. Reaction time: 6 h. Yield: 361 mg, 88%. White solid. TLC: Rf 0.04 (MeOH). mp: 47.2–49.2 °C. = −7.6 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.32–7.18 (m, 6H), 4.31 (t, J = 7.2 Hz, 2H), 3.09 (br s, 1H), 2.78 (dd, J = 13.2, 4.8 Hz, 1H), 2.73 (t, J = 7.6 Hz, 2H), 2.68 (m, 1H), 2.64 (q, J = 7.2 Hz, 2H), 2.50 (dd, J = 13.2, 8.8 Hz, 1H), 2.45 (dd, J = 11.2, 8.8 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.6 Hz, 2H), 1.55 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CD3OD): δ 149.0, 140.2, 130.4, 129.6, 127.5, 123.2, 56.4, 53.3, 51.0, 50.5, 43.2, 33.4, 30.0, 28.3, 26.1, 20.7, 13.8. LRMS (ESI) m/z (rel int): (pos) 330 ([M + H]+, 100), 282 ([C15H32N5]+, 48), 180 ([M − C2H6N2 − Bn]+, 22). HRMS (ESI) m/z calcd for C19H32N5+ ([M + H]+) 330.2652, found 330.2658.
(S)-N1-[4-(1-Butyl-1H-1,2,3-triazol-4-yl)butyl]-3,3-dimethylbutane-1,2-diamine (9b).9b was synthesized according to the procedure described for the synthesis of 9a. Amide 8b (2.30 g, 7.42 mmol) and LiAlH4 (1.69 g, 44.5 mmol) were used. Reaction time: 24 h. Yield: 1.47 g, 67% (brsm: 83%). Colorless oil. TLC: Rf 0.07 (MeOH). = +23.8 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.25 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 2.77 (dd, J = 11.2, 2.4 Hz, 1H), 2.74 (t, J = 7.6 Hz, 2H), 2.67 (dt, J = 11.2, 7.2 Hz, 1H), 2.59 (dt, J = 11.2, 7.2 Hz, 1H), 2.46 (dd, J = 11.2, 2.4 Hz, 1H), 2.21 (t, J = 11.2 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.73 (quintet, J = 7.6 Hz, 2H), 1.57 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H), 0.88 (s, 9H). 13C NMR (100 MHz, DMSO-d6): δ 146.8, 121.5, 59.1, 51.3, 49.2, 48.8, 33.3, 31.7, 29.3, 26.9, 26.3, 25.0, 19.1, 13.3. LRMS (ESI) m/z (rel int): (pos) 296 ([M + H]+, 100), 282 ([C15H32N5]+, 21). HRMS (ESI) m/z calcd for C16H34N5+ ([M + H]+) 296.2809, found 296.2814.
(S)-N1-[4-(1-Butyl-1H-1,2,3-triazol-4-yl)butyl]-3-methylbutane-1,2-diamine (9c).9c was synthesized according to the procedure described for the synthesis of 9a. Amide 8c (900 mg, 3.05 mmol) and LiAlH4 (482 mg, 18.3 mmol) were used. Reaction time: 24 h. Yield: 694 mg, 81% (brsm: 89%). Milky oil. TLC: Rf 0.05 (MeOH). = +16.3 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.25 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 2.73 (t, J = 7.6 Hz, 2H), 2.69–2.56 (m, 4H), 2.34 (dd, J = 11.6, 9.6 Hz, 1H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.72 (quintet, J = 7.6 Hz, 2H), 1.60–1.52 (m, 3H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H), 0.91 (d, J = 6.8 Hz, 3H), 0.89 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 148.1, 120.4, 56.6, 54.2, 49.9 (2C), 32.5, 32.3, 29.8, 27.3, 25.6, 19.8, 19.4, 17.8, 13.5. LRMS (ESI) m/z (rel int): (pos) 282 ([M + H]+, 82), 141 ([M − nBu − C2HN3 − NH2]+, 100). HRMS (ESI) m/z calcd for C15H32N5+ ([M + H]+) 282.2652, found 282.2662.
(S)-4-Benzyl-1-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]imidazolidin-2-one (10a). In a 100 mL roundbottom flask, diamine 9a (440 mg, 1.34 mmol) was dissolved in anhydrous DMF (1.3 mL) at rt. Et3N (470 µL, 3.34 mmol) and CDI (335 mg, 2.00 mmol) were added. The resulting reaction mixture was stirred at 120 °C for 13 h. Upon completion of the reaction, the reaction mixture was cooled to rt, and concentrated in vacuo. The purification by column chromatography on silica gel (15:1 CH2Cl2/MeOH) afforded a desired imidazolidinone 10a as a colorless liquid (470 mg, 99%). TLC: Rf 0.49 (10:1 CH2Cl2/MeOH). = −27.7 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.34–7.30 (m, 2H), 7.28–7.23 (m, 2H), 7.19–7.17 (m, 2H), 4.31 (t, J = 7.2 Hz, 2H), 4.30 (br s, 1H), 3.86 (m, 1H), 3.50 (t, J = 8.8 Hz, 1H), 3.20 (m, 2H), 3.13 (dd, J = 8.8, 6.0 Hz, 1H), 2.83 (dd, J = 13.2, 6.0 Hz, 1H), 2.77 (dd, J = 13.2, 8.8 Hz, 1H), 2.75 (t, J = 7.6 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.69 (m, 2H), 1.57 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 161.7, 147.8, 137.2, 129.2, 128.9, 127.0, 120.7, 51.4, 50.3, 50.0, 43.0, 42.1, 32.4, 27.2, 26.7, 25.3, 19.8, 13.6. LRMS (ESI) m/z (rel int): (pos) 356 ([M + H]+, 16), 282 ([C15H32N5]+, 100). HRMS (ESI) m/z calcd for C20H30N5O+ ([M + H]+) 356.2445, found 356.2440.
(R)-4-Benzyl-1-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]imidazolidin-2-one (10a′).10a′ was synthesized according to the procedure described for the synthesis of 10a. Diamine 9a′ (179 mg, 542 µmol), Et3N (190 µL, 1.36 mmol) and CDI (132 mg, 814 µmol) were used. Reaction time: 13 h. Yield: 175 mg, 91%. Colorless liquid. TLC: Rf 0.49 (10:1 CH2Cl2/MeOH). = +26.4 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.34–7.30 (m, 2H), 7.27–7.22 (m, 2H), 7.19–7.17 (m, 2H), 4.31 (t, J = 7.2 Hz, 2H), 4.29 (br s, 1H), 3.86 (m, 1H), 3.49 (t, J = 8.4 Hz, 1H), 3.23 (dq, J = 14.0, 7.2 Hz, 2H), 3.20 (m, 2H), 3.13 (dd, J = 8.8, 6.0 Hz, 1H), 2.82 (m, 2H), 2.75 (t, J = 7.6 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.69 (m, 2H), 1.56 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 161.7, 147.8, 137.3, 129.2, 129.0, 127.1, 120.7, 51.5, 50.4, 50.1, 43.1, 42.2, 32.5, 27.2, 26.8, 25.4, 19.9, 13.7. LRMS (ESI) m/z (rel int): (pos) 356 ([M + H]+, 100), 282 ([C15H32N5]+, 95). HRMS (ESI) m/z calcd for C20H30N5O+ ([M + H]+) 356.2445, found 356.2452.
(S)-4-(tert-Butyl)-1-[4-(1-butyl-1H-1,2,3-triazol-4-yl)butyl]imidazolidin-2-one (10b).10b was synthesized according to the procedure described for the synthesis of 10a. Diamine 9b (1.35 g, 4.55 mmol), Et3N (1.60 mL, 11.4 mmol) and CDI (1.11 g, 6.83 mmol) were used. Reaction time: 15 h. Yield: 1.46 g, quantitative yield. Colorless crystal. TLC: Rf 0.29 (30:1 CH2Cl2/MeOH (×2)). mp: 80.3–82.3 °C. = −6.9 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 7.27 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 4.30 (s, 1H), 3.41–3.34 (m, 2H), 3.25 (dt, J = 13.6, 7.2 Hz, 1H), 3.15 (dd, J = 13.6, 7.2 Hz, 1H), 3.13 (m, 1H), 2.75 (t, J = 7.6 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.68 (m, 2H), 1.58 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H), 0.87 (s, 9H). 13C NMR (100 MHz, CDCl3): δ 161.9, 147.8, 120.6, 58.8, 49.9, 46.4, 42.9, 33.2, 32.3, 27.0, 26.6, 25.2, 25.0, 19.7, 13.5. LRMS (ESI) m/z (rel int): (pos) 322 ([M + H]+, 100), 282 ([C15H32N5]+, 42). HRMS (ESI) m/z calcd for C17H32N5O+ ([M + H]+) 322.2601, found 322.2603.
(S)-4-Isopropyl-1-[4-(1-butyl-1,2,3-triazol-4-yl)butyl]imidazolidin-2-one (10c).10c was synthesized according to the procedure described for the synthesis of 10a. Diamine 9c (2.05 g, 7.30 mmol), Et3N (2.60 mL, 18.2 mmol) and CDI (1.77 g, 10.9 mmol) were used. Reaction time: 14.5 h. Yield: 1.78 g, 79%. White solid. TLC: Rf 0.29 (30:1 CH2Cl2/MeOH (×2)). mp: 109.7–111.7 °C. = −9.0 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 7.27 (s, 1H), 4.32 (s, 1H), 4.31 (t, J = 7.2 Hz, 2H), 3.46 (t, J = 8.4 Hz, 1H), 3.36 (ddd, J = 16.0, 7.2, 1.6 Hz, 1H), 3.26 (dt, J = 13.6, 7.2 Hz, 1H), 3.14 (dt, J = 13.6, 7.2 Hz, 1H), 3.06 (dd, J = 8.4, 7.2 Hz, 1H), 2.75 (t, J = 7.6 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.73–1.63 (m, 3H), 1.60 (m, 2H), 1.35 (sextet, J = 7.2 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H), 0.92 (d, J = 6.8 Hz, 3H), 0.88 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.0, 147.8, 120.6, 55.8, 49.9, 48.8, 43.0, 33.1, 32.3 27.1, 26.6, 25.3, 19.7, 18.4, 17.9, 13.5. LRMS (ESI) m/z (rel int): (pos) 308 ([M + H]+, 100), 282 ([C15H32N5]+, 25). HRMS (ESI) m/z calcd for C16H30N5O+ ([M + H]+) 308.2445, found 308.2450.
(S)-4-[4-(4-Benzyl-2-oxoimidazolidin-1-yl)butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium iodide (12a). In a screw-cap vial, a mixture of 10a (161 mg, 454 μmol) and MeI (28.3 μL, 454 μmol) was stirred at 80 °C. After 24 h, the reaction mixture was cooled to rt. The purification by column chromatography on silica gel (15:1 CH2Cl2/MeOH) afforded a desired compound 12a (183 mg, 81%) as a light-yellow sticky gum. TLC: Rf 0.15 (10:1 CH2Cl2/MeOH). = −16.0 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 9.33 (s, 1H), 7.31 (m, 2H), 7.25–7.19 (m, 3H), 4.68 (t, J = 7.6 Hz, 2H), 4.36 (s, 1H), 4.28 (s, 3H), 3.97 (m, 1H), 3.62 (t, J = 8.8 Hz, 1H), 3.32 (dt, J = 14.4, 7.2 Hz, 1H), 3.18 (dd, J = 8.8, 6.4 Hz, 1H), 3.16 (m, 1H), 3.04 (m, 2H), 2.87 (dd, J = 13.6, 5.6 Hz, 1H), 2.81 (dd, J = 13.6, 7.6 Hz, 1H), 2.03 (quintet, J = 7.6 Hz, 2H), 1.84 (m, 2H), 1.70 (m, 2H), 1.44 (sextet, J = 7.6 Hz, 2H), 0.98 (t, J = 7.6 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.1, 144.5, 137.1, 129.6, 129.3, 128.8, 126.8, 54.0, 51.3, 50.2, 42.0, 41.8, 38.9, 31.3, 26.6, 24.0, 23.2, 19.5, 13.5. LRMS (FAB) m/z (rel int): (pos) 370 ([C21H32N5O]+, 100), 278 ([C21H32N5O − Bn]+, 15). HRMS (FAB) m/z calcd for C21H32N5O+ 370.2601, found 370.2603.
(S)-4-[4-[4-(tert-Butyl)-2-oxoimidazolidin-1-yl])butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium iodide (12b).12b was synthesized according to the procedure described for the synthesis of 12a. 10b (1.46 g, 4.54 mmol) and MeI (290 µL, 4.54 mmol) were used. Reaction time: 24 h. Yield: 1.80 g, 86%. Yellow gum. TLC: Rf 0.47 (10:1 CH2Cl2/MeOH). = −8.1 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 9.41 (s, 1H), 4.70 (t, J = 7.6 Hz, 2H), 4.53 (br s, 1H), 4.26 (s, 3H), 3.51–3.45 (m, 2H), 3.36 (dt, J = 14.4, 6.8 Hz, 1H), 3.22–3.14 (m, 2H), 3.04 (td, J = 8.0, 2.8 Hz, 2H), 2.04 (quintet, J = 7.6 Hz, 2H), 1.86 (quintet, J = 7.6 Hz, 2H), 1.73 (sextet, J = 7.6 Hz, 2H), 1.44 (sextet, J = 7.6 Hz, 2H), 0.99 (t, J = 7.6 Hz, 3H), 0.88 (s, 9H). 13C NMR (100 MHz, CDCl3): δ 162.2, 144.5, 129.7, 58.8, 54.0, 46.5, 41.7, 38.5, 33.3, 31.3, 26.4, 25.1, 24.0, 22.9, 19.5, 13.4. LRMS (FAB) m/z (rel int): (pos) 336 ([C18H34N5O]+, 100), 278 ([C18H34N5O − t-Bu]+, 15). HRMS (FAB) m/z calcd for C18H34N5O+ 336.2758, found 336.2758.
(S)-4-[4-(4-Isopropyl-2-oxoimidazolidin-1-yl)butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium iodide (12c).12c was synthesized according to the procedure described for the synthesis of 12a. 10c (1.78 g, 5.78 mmol) and MeI (360 µL, 5.78 mmol) were used. Reaction time: 24 h. Yield: 2.39 g, 92%. Colorless oil. TLC: Rf 0.26 (10:1 CH2Cl2/MeOH). = −6.0 (c 0.50, MeOH). 1H NMR (400 MHz, CDCl3): δ 9.39 (s, 1H), 4.69 (t, J = 7.6 Hz, 2H), 4.52 (br s, 1H), 4.27 (s, 3H), 3.56 (t, J = 8.8 Hz, 1H), 3.46 (qm, J = 8.8 Hz, 1H), 3.36 (dt, J = 14.4, 7.2 Hz, 1H), 3.19 (dt, J = 14.4, 6.0 Hz, 1H), 3.11 (dd, J = 8.8, 7.6 Hz, 1H), 3.04 (m, 2H), 2.04 (quintet, J = 7.6 Hz, 2H), 1.86 (m, 2H), 1.73 (m, 1H), 1.68 (sextet, J = 7.6 Hz, 2H), 1.44 (sextet, J = 7.6 Hz, 2H), 0.99 (t, J = 7.6 Hz, 3H), 0.93 (d, J = 6.8 Hz, 3H), 0.89 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.5, 144.6, 129.8, 55.9, 54.0, 48.9, 41.8, 38.6, 33.1, 31.3, 26.4, 24.0, 23.1, 19.5, 18.5, 17.9, 13.4. LRMS (FAB) m/z (rel int): (pos) 322 ([C18H34N5O]+, 100), 278 ([C18H34N5O − i-Pr]+, 10). HRMS (FAB) m/z calcd for C18H34N5O+ 322.2601, found 322.2603.
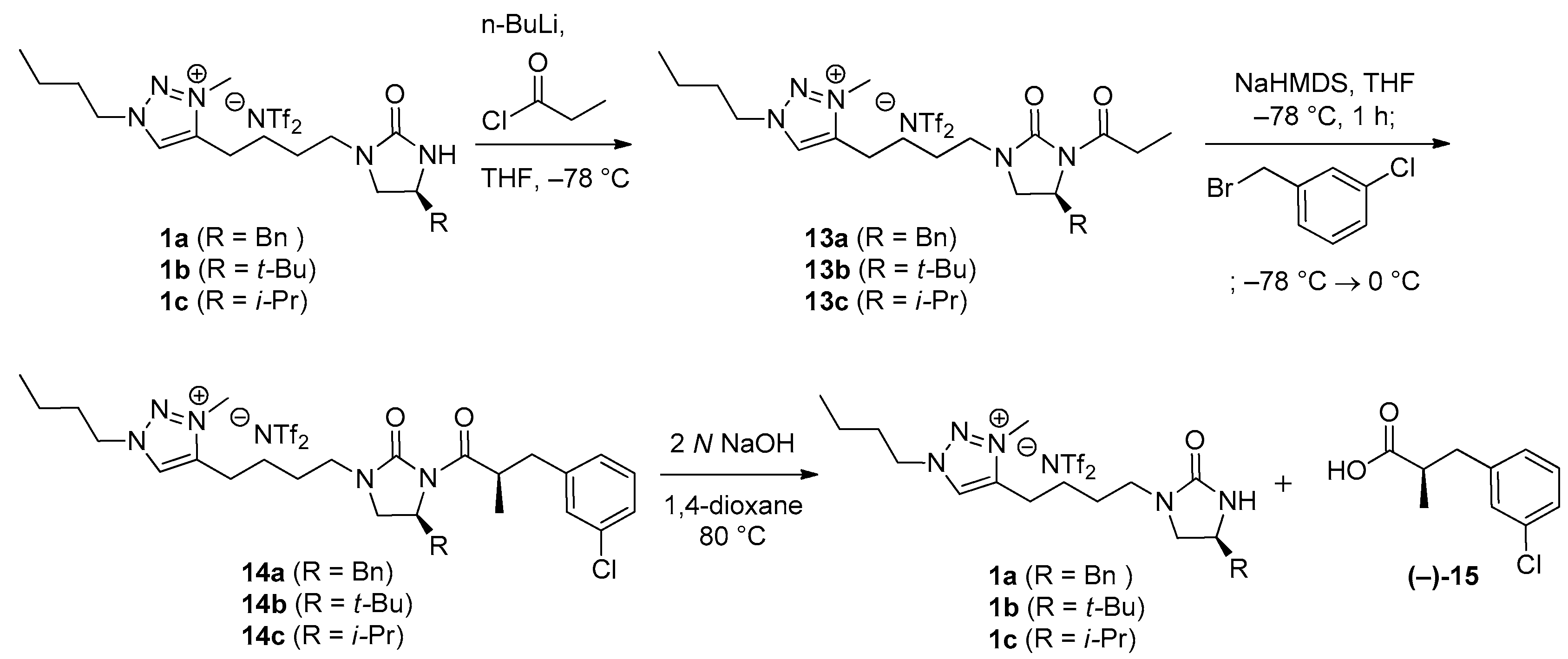
(S)-4-[4-(4-Benzyl-2-oxoimidazolidin-1-yl)butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (1a). In a 25 mL roundbottom flask, 12a (463 mg, 930 μmol) and LiNTf2 (267 mg, 930 μmol) were dissolved in deionized water (1.6 mL). The reaction mixture was stirred at 40 °C for 24 h. Upon completion of the reaction, the reaction mixture was cooled to rt, and extracted with CH2Cl2 (5 × 9 mL). The combined organic extracts were dried over anhydrous Na2SO4, and filtered. The concentration by rotary evaporation afforded a desired 1a (577 mg, 95%) as a light yellow liquid. TLC: Rf 0.10 (10:1 CH2Cl2/MeOH). = −6.5 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 8.40 (s, 1H), 7.31 (m, 2H), 7.26–7.17 (m, 3H), 4.53 (t, J = 7.6 Hz, 2H), 4.34 (br s, 1H), 4.21 (s, 3H), 3.94 (m, 1H), 3.57 (t, J = 8.8 Hz, 1H), 3.34 (dt, J = 14.4, 6.4 Hz, 1H), 3.18 (dt, J = 14.4, 6.0 Hz, 1H), 3.17 (dd, J = 8.8, 6.4 Hz, 1H), 2.93 (m, 2H), 2.87 (dd, J = 13.6, 5.6 Hz, 1H), 2.77 (dd, J = 13.6, 8.0 Hz, 1H), 1.99 (quintet, J = 7.6 Hz, 2H), 1.77 (m, 2H), 1.69 (m, 2H), 1.41 (sextet, J = 7.6 Hz, 2H), 0.99 (t, J = 7.6 Hz, 3H). 13C NMR (100 MHz, acetone-d6): δ 162.4, 145.9, 138.6, 130.2, 129.4, 129.1, 127.4, 121.1 (q, JC-F = 319.6 Hz), 54.4, 51.9, 50.4, 42.6, 42.5, 38.1, 32.0, 27.2, 24.7, 23.1, 20.0, 13.6. 19F NMR (376 MHz, CDCl3): δ −79.3. LRMS (FAB) m/z (rel int): (pos) 370 ([C21H32N5O]+, 100), 278 ([C21H32N5O − Bn]+, 25). HRMS (FAB) m/z calcd for C21H32N5O+ 370.2601, found 370.2602.
(S)-4-[4-[4-(tert-Butyl)-2-oxoimidazolidin-1-yl]butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (1b). 1b was synthesized according to the procedure described for the synthesis of 1a. 12b (331 mg, 714 mmol) and LiNTf2 (205 mg, 714 mmol) were used. Reaction time: 24 h. Yield: 417 mg, 95%. Colorless oil. TLC: Rf 0.56 (10:1 CH2Cl2/MeOH). = −5.7 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 8.45 (s, 1H), 4.54 (t, J = 7.6 Hz, 2H), 4.34 (s, 1H), 4.22 (s, 3H), 3.45–3.41 (m, 2H), 3.34 (dt, J = 14.4, 6.4 Hz, 1H), 3.20 (t, J = 6.0 Hz, 1H), 3.16 (m, 1H), 2.96 (m, 2H), 1.99 (quintet, J = 7.6 Hz, 2H), 1.79 (quintet, J = 7.6 Hz, 2H), 1.71 (m, 2H), 1.42 (sextet, J = 7.6 Hz, 2H), 0.99 (t, J = 7.6 Hz, 3H), 0.87 (s, 9H). 13C NMR (100 MHz, CDCl3): δ 162.2, 144.8, 128.3, 119.8 (q, JC-F = 319.7 Hz), 58.8, 53.9, 46.3, 41.6, 37.4, 33.2, 31.0, 26.3, 24.9, 23.7, 22.3, 19.4, 13.2. 19F NMR (376 MHz, CDCl3): δ −79.2. LRMS (FAB) m/z (rel int): (pos) 336 ([C18H34N5O]+, 100), 278 ([C18H34N5O − t-Bu]+, 13). HRMS (FAB) m/z calcd for C18H34N5O+ 336.2758, found 336.2760.
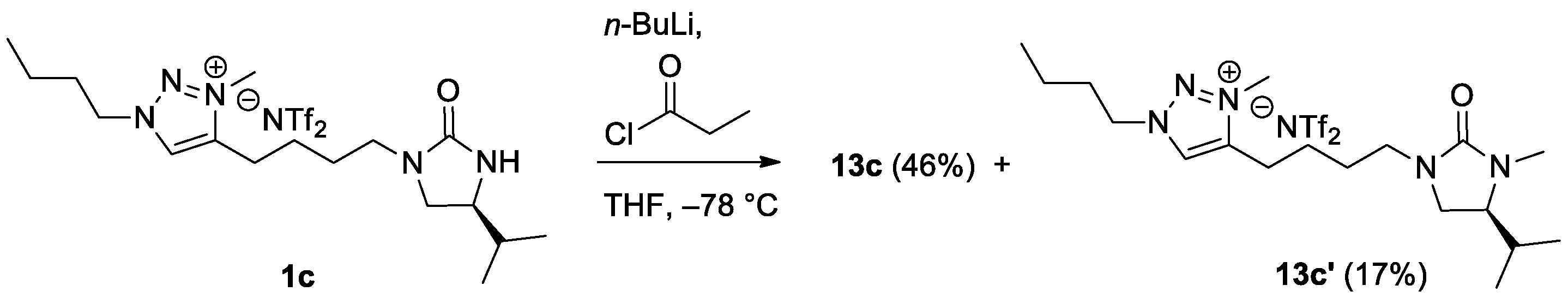
(S)-4-[4-(4-Isopropyl-2-oxoimidazolidin-1-yl)butyl]-1-butyl-3-methyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (1c).1c was synthesized according to the procedure described for the synthesis of 1a. 12c (1.92 g, 4.27 mmol) and LiNTf2 (1.23 g, 4.27 mmol) were used. Reaction time: 24 h. Yield: 2.46 g, 96%. Colorless oil. TLC: Rf 0.39 (10:1 CH2Cl2/MeOH). = −3.9 (c 1.0, MeOH). 1H NMR (400 MHz, CDCl3): δ 8.34 (s, 1H), 4.52 (t, J = 7.6 Hz, 2H), 4.46 (s, 1H), 4.21 (s, 3H), 3.52 (t, J = 8.8 Hz, 1H), 3.41 (qd, J = 8.8, 1.6 Hz, 1H), 3.33 (dt, J = 14.4, 6.4 Hz, 1H), 3.17 (dt, J = 14.4, 6.4 Hz, 1H), 3.09 (dd, J = 8.8, 7.6 Hz, 1H), 2.94 (m, 2H), 1.98 (quintet, J = 7.6 Hz, 2H), 1.77 (quintet, J = 7.6 Hz, 2H), 1.73–1.64 (m, 3H), 1.41 (sextet, J = 7.6 Hz, 2H), 0.99 (t, J = 7.6 Hz, 3H), 0.92 (d, J = 6.8 Hz, 3H), 0.88 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.4, 144.8, 128.2, 119.8 (q, JC-F = 319.7 Hz), 55.9, 53.9, 48.7, 41.7, 37.4, 33.0, 31.0, 26.3, 23.6, 22.4, 19.4, 18.2, 17.7, 13.2. 19F NMR (376 MHz, CDCl3): δ −79.3. LRMS (FAB) m/z (rel int): (pos) 322 ([C17H32N5O]+, 100), 278 ([C1H34N5O − i-Pr]+, 11), 57 ([C4H9]+, 62). HRMS (FAB) m/z calcd for C18H34N5O+ 322.2601, found 322.2607.
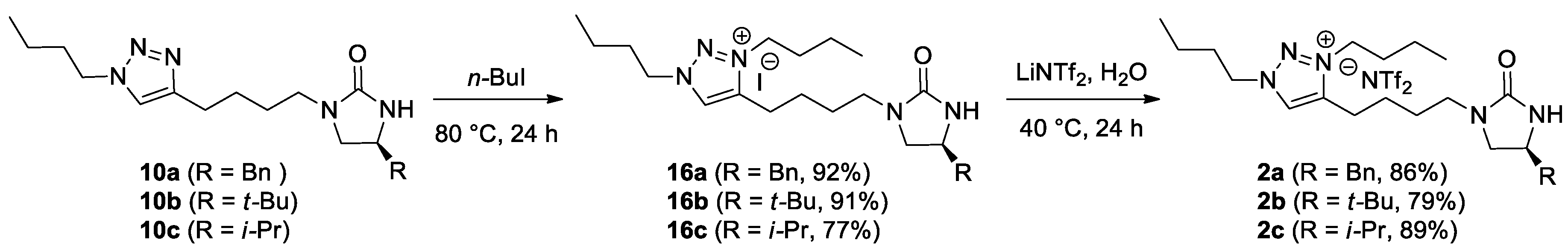
(S)-4-[4-(4-Benzyl-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium iodide (16a). In a screw-cap vial, a mixture of 10a (354 mg, 995 μmol) and n-BuI (115 μL, 995 μmol) in DMF (100 μL) was stirred at 100 °C. After 24 h, the reaction mixture was cooled to rt. The purification by column chromatography on silica gel (30:1 CH2Cl2/MeOH) afforded a desired triazolium iodide 16a (467 mg, 92%) as a yellow oil. TLC: Rf 0.36 (20:1 CH2Cl2/MeOH). = −37.2 (c 1.2, CHCl3). 1H NMR (400 MHz, CDCl3): δ 9.60 (s, 1H), 7.33–7.30 (m, 2H), 7.26–7.19 (m, 3H), 4.73 (t, J = 7.2 Hz, 2H), 4.48 (t, J = 7.2 Hz, 2H), 4.31 (br s, 1H), 3.97 (m, 1H), 3.64 (t, J = 8.8 Hz, 1H), 3.35 (dt, J = 14.4, 6.8 Hz, 1H), 3.22–3.14 (m, 2H), 2.98 (m, 2H), 2.90 (dd, J = 13.2, 5.2 Hz, 1H), 2.77 (dd, J = 13.2, 8.4 Hz, 1H), 2.04 (quintet, J = 7.6 Hz, 2H), 1.95 (m, 2H), 1.86 (quintet, J = 7.2 Hz, 2H), 1.74 (m, 2H), 1.42 (sextet, J = 7.2 Hz, 4H), 1.01 (t, J = 7.2 Hz, 3H), 0.99 (t, J = 7.2 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 161.8, 143.9, 137.1, 130.1, 129.1, 128.8, 126.9, 54.1, 51.4, 51.2, 50.4, 42.0, 41.6, 31.4, 31.0, 26.4, 24.2, 22.7, 19.6, 19.5, 13.4, 13.3. LRMS (ESI) m/z (rel int): (pos) 412 ([C24H38N5O]+, 100), 394 ([C24H38N5O − H2O]+, 3). HRMS (ESI) m/z calcd for C24H38N5O+ 412.3071, found 412.3071.
(S)-4-[4-(4-(tert-Butyl)-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium iodide (16b).16b was synthesized according to the procedure described for the synthesis of 16a. 10b (161 mg, 500 μmol) and n-BuI (58.0 μL, 500 μmol) in DMF (50 μL) were used. Reaction time: 38 h. Yield: 229 mg, 91%. Yellow oil. TLC: Rf 0.16 (20:1 CH2Cl2/MeOH). = −38.4 (c 0.82, CHCl3). 1H NMR (400 MHz, CDCl3): δ 9.63 (s, 1H), 4.74 (t, J = 7.2 Hz, 2H), 4.48 (t, J = 7.2 Hz, 2H), 4.34 (br s, 1H), 3.51–3.45 (m, 2H), 3.36 (m, 1H), 3.21–3.14 (m, 2H), 3.00 (td, J = 8.0, 5.2 Hz, 2H), 2.04 (quintet, J = 7.2 Hz, 2H), 1.95 (quintet, J = 7.2 Hz, 2H), 1.87 (quintet, J = 7.2 Hz, 2H), 1.74 (sextet, J = 7.6 Hz, 2H), 1.43 (sextet, J = 7.2 Hz, 2H), 1.42 (sextet, J = 7.2 Hz, 2H), 1.01 (t, J = 7.2 Hz, 3H), 0.99 (t, J = 7.2 Hz, 3H), 0.88 (s, 9H). 13C NMR (100 MHz, CDCl3): δ 162.1, 143.9, 130.1, 58.8, 54.1, 51.1, 46.5, 41.5, 33.3, 31.4, 31.0, 26.3, 25.1, 24.2, 22.6, 19.6, 19.5, 13.4, 13.3. LRMS (ESI) m/z (rel int): (pos) 378 ([C21H40N5O]+, 100), 361 ([C21H39N5O − H2O]+, 2). HRMS (ESI) m/z calcd for C21H40N5O+ 378.3227, found 378.3227.
(S)-4-[4-(4-Isopropyl-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium iodide (16c).16c was synthesized according to the procedure described for the synthesis of 16a. 10c (154 mg, 500 μmol) and n-BuI (58 μL, 500 μmol) in DMF (50 μL) were used. Reaction time: 34 h. Yield: 190 mg, 77%. Yellow oil. TLC: Rf 0.2 (10:1 CH2Cl2/MeOH). = −39.8 (c 0.54, CHCl3). 1H NMR (400 MHz, CDCl3): δ 9.64 (s, 1H), 4.74 (t, J = 7.2 Hz, 2H), 4.48 (t, J = 7.2 Hz, 2H), 4.35 (br s, 1H), 3.58 (t, J = 8.8 Hz, 1H), 3.45 (qm, J = 7.2 Hz, 1H), 3.35 (dt, J = 14.4, 2.8 Hz, 1H), 3.18 (dt, J = 14.4, 6.4 Hz, 1H), 3.11 (dd, J = 8.8, 7.6 Hz, 1H), 2.99 (m, 2H), 2.04 (quintet, J = 7.6 Hz, 2H), 1.95 (quintet, J = 7.2 Hz, 2H), 1.87 (m, 2H), 1.78–1.65 (m, 3H), 1.42 (sextet, J = 7.6 Hz, 4H), 1.01 (t, J = 7.2 Hz, 3H), 0.99 (t, J = 7.2 Hz, 3H), 0.93 (d, J = 6.8 Hz, 3H), 0.90 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.4, 144.1, 130.2, 56.0, 54.2, 51.3, 49.1, 41.7, 33.2, 31.5, 31.1, 26.5, 24.4, 22.8, 19.7, 19.6, 18.5, 18.1, 13.6, 13.5. LRMS (ESI) m/z (rel int): (pos) 364 ([C20H38N5O]+, 100), 387 ([C20H38N5O + Na]+, 1). HRMS (ESI) m/z calcd for C20H38N5O+ 364.3071, found 364.3062.
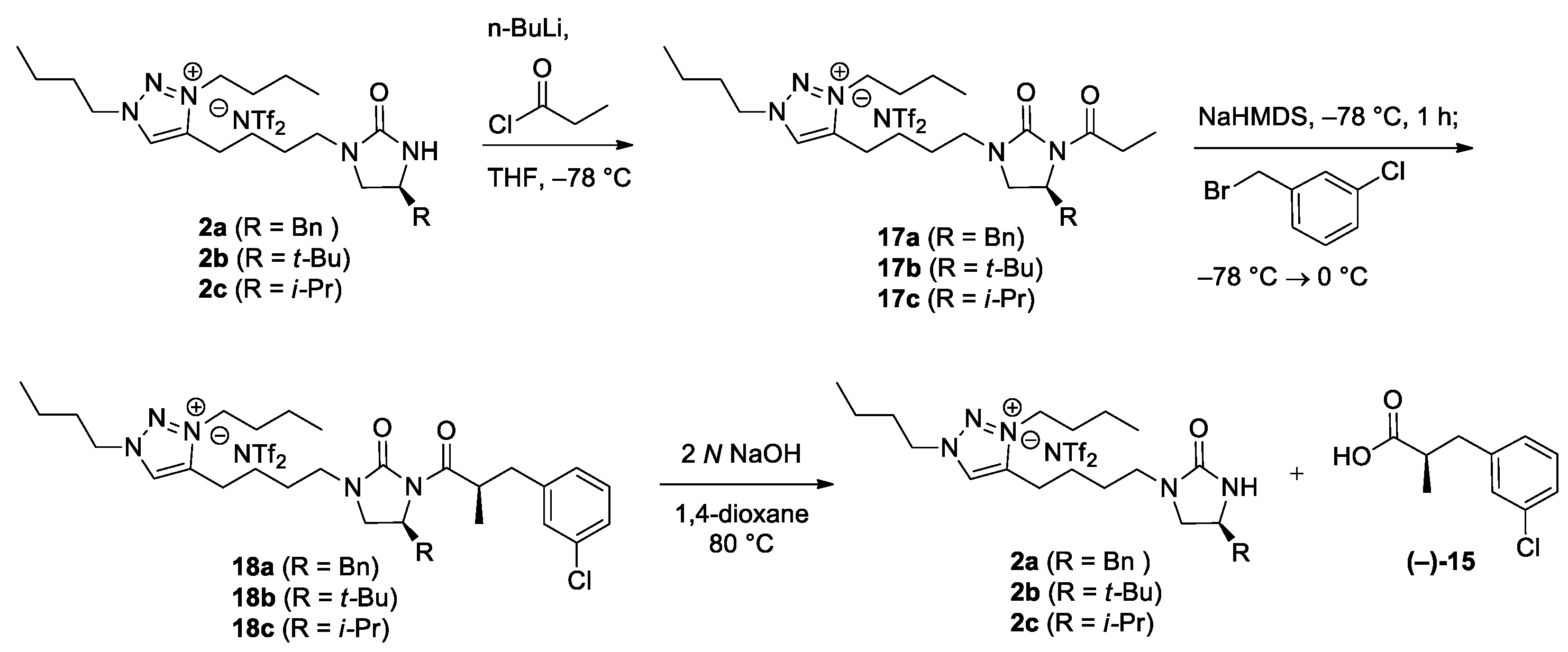
(S)-4-[4-(4-Benzyl-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (2a). In a screw-cap vial, 16a (164 mg, 321 μmol) and LiNTf2 (92.2 mg, 321 μmol) were dissolved in deionized water (0.54 mL). The reaction mixture was stirred at 40 °C for 24 h. Upon completion of the reaction, the reaction mixture was cooled to rt, and extracted with CH2Cl2 (3 × 3 mL). The combined organic extracts were dried over anhydrous MgSO4, and filtered. The concentration by rotary evaporation afforded the desired NTf2 salt 2a (192 mg, 86%) as a yellow oil. TLC: Rf 0.53 (15:1 CH2Cl2/MeOH). = −42.1 (c 1.1, CHCl3). 1H NMR (400 MHz, CDCl3): δ 8.43 (s, 1H), 7.33−7.30 (m, 2H), 7.26−7.17 (m, 3H), 4.54 (t, J = 7.6 Hz, 2H), 4.45 (t, J = 7.6 Hz, 2H), 3.96 (m, 1H), 3.58 (t, J = 8.8 Hz, 1H), 3.34 (dt, J = 14.4, 6.8 Hz, 1H), 3.22−3.14 (m, 2H), 2.92 (m, 2H), 2.87 (dd, J = 13.6, 5.2 Hz, 1H), 2.77 (dd, J = 13.6, 8.4 Hz, 1H), 1.99 (quintet, J = 7.6 Hz, 2H), 1.95 (quintet, J = 7.6 Hz, 2H), 1.78 (m, 2H), 1.69 (m, 2H), 1.42 (sextet, J = 7.6 Hz, 2H), 1.40 (sextet, J = 7.6 Hz, 2H), 1.00 (t, J = 7.6 Hz, 3H), 0.99 (t, J = 7.6 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 161.9, 144.3, 137.0, 129.1, 128.8, 128.6, 126.9, 119.8 (q, JC-F = 319.7 Hz), 54.0, 51.4, 50.9, 50.2, 41.9, 41.6, 31.1, 30.8, 26.3, 23.9, 22.3, 19.5, 19.4, 13.3, 13.2. 19F NMR (376 MHz, CDCl3): δ −79.2. LRMS (ESI) m/z (rel int): (pos) 412 ([C24H38N5O]+, 100), 394 ([C24H38N5O − H2O]+, 10). HRMS (ESI) m/z calcd for C24H38N5O+ 412.3071, found 412.3069.
(S)-4-[4-(4-(tert-Butyl)-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (2b).2b was synthesized according to the procedure described for the synthesis of 2a. 16b (199 mg, 393 μmol) and LiNTf2 (113 mg, 393 μmol) were used. Reaction time: 24 h. Yield: 204 mg, 79%. Light yellow oil. TLC: Rf 0.69 (10:1 CH2Cl2/MeOH (×2)). = −68.0 (c 0.40, CHCl3). 1H NMR (400 MHz, CDCl3): δ 8.43 (s, 1H), 4.54 (t, J = 7.6 Hz, 2H), 4.46 (t, J = 7.6 Hz, 2H), 3.52–3.46 (m, 2H), 3.37 (m, 1H), 3.22–3.14 (m, 2H), 2.92 (td, J = 8.0, 4.8 Hz, 2H), 1.99 (quintet, J = 7.6 Hz, 2H), 1.95 (quintet, J = 7.6 Hz, 2H), 1.79 (sextet, J = 7.6 Hz, 2H), 1.71 (m, 2H), 1.43 (sextet, J = 7.6 Hz, 2H), 1.40 (sextet, J = 7.6 Hz, 2H), 1.01 (t, J = 7.6 Hz, 3H), 0.99 (t, J = 7.6 Hz, 3H), 0.88 (s, 9H). 13C NMR (100 MHz, CDCl3): δ 162.5, 144.5, 128.8, 120.2 (q, JC-F = 318.9 Hz), 59.4, 54.2, 51.1, 46.7, 42.0, 33.4, 31.3, 31.0, 26.5, 25.1, 24.1, 22.6, 19.7, 19.6, 13.5, 13.4. 19F NMR (376 MHz, CDCl3): δ −79.2. LRMS (ESI) m/z (rel int): (pos) 378 ([C21H40N5O]+, 100), 383 ([C21H40N5O + Na − H2O]+, 1). HRMS (ESI) m/z calcd for C21H40N5O+ 378.3227, found 378.3237.
(S)-4-[4-(4-Isopropyl-2-oxoimidazolidin-1-yl)butyl]-1,3-dibutyl-1H-1,2,3-triazolium bis(trifluoromethanesulfonyl)imide (2c).2c was synthesized according to the procedure described for the synthesis of 2a. 16c (169 mg, 343 μmol) and LiNTf2 (98.6 mg, 343 μmol) were used. Reaction time: 24 h. Yield: 197 mg, 89%. Colorless oil. TLC: Rf 0.26 (10:1 CH2Cl2/MeOH). = −29.7 (c 1.0, CHCl3). 1H NMR (400 MHz, CDCl3): δ 8.44 (s, 1H), 4.55 (t, J = 7.2 Hz, 2H), 4.45 (t, J = 7.2 Hz, 2H), 3.56 (t, J = 8.8 Hz, 1H), 3.46 (dt, J = 8.8, 7.2 Hz, 1H), 3.37 (dt, J = 14.4, 7.2 Hz, 1H), 3.18 (dt, J = 14.4, 6.4 Hz, 1H), 3.12 (dd, J = 8.8, 7.6 Hz, 1H), 2.92 (m, 2H), 1.99 (quintet, J = 7.2 Hz, 2H), 1.95 (quintet, J = 7.2 Hz, 2H), 1.80 (m, 2H), 1.73–1.65 (m, 3H), 1.43 (sextet, J = 7.2 Hz, 2H), 1.40 (sextet, J = 7.2 Hz, 2H), 1.01 (t, J = 7.2 Hz, 3H), 0.99 (t, J = 7.2 Hz, 3H), 0.92 (d, J = 6.8 Hz, 3H), 0.89 (d, J = 6.8 Hz, 3H). 13C NMR (100 MHz, CDCl3): δ 162.3, 144.3, 128.5, 119.8 (q, JC-F = 319.6 Hz), 56.0, 53.9, 50.9, 48.8, 41.7, 33.0, 31.1, 30.8, 26.3, 24.0, 22.3, 19.5, 19.4, 18.2, 17.7, 13.3, 13.2. 19F NMR (376 MHz, CDCl3): δ −79.2. LRMS (ESI) m/z (rel int): (pos) 378 ([C20H38N5O]+, 100), 346 ([C20H38N5O − H2O]+, 1). HRMS (ESI) m/z calcd for C20H38N5O+ 364.3071, found 364.3066.