Abstract
A variety of aminoisoquinoline-5,8-quinones bearing α-amino acids moieties were synthesized from 3-methyl-4-methoxycarbonylisoquinoline-5,8-quinone and diverse l- and d-α-amino acid methyl esters. The members of the series were evaluated for their cytotoxic activity against normal and cancer cell lines by using the (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (MTT) assay. From the current investigation, structure–activity relationships demonstrate that the location and structure of the amino acid fragment plays a significant role in the cytotoxic effects. Moderate to high cytotoxic activity was observed and four members, derived from l-alanine, l-leucine, l-phenylalanine, and d-phenylalanine, were selected as promising compounds by their IC50 ranging from 0.5 to 6.25 μM and also by their good selectivity indexes (≥2.24).
1. Introduction
Quinones (anthraquinones, naphthoquinones, and heterocyclic quinones) are important naturally occurring substances that are widely distributed in nature [1] and are known by their various physiological activities as antibiotics and anticancer agents. They form the second large class of antitumor agents approved for clinical use and several other antitumor members are currently in different stages of clinical and preclinical development. Their antitumor activity has been currently explained by their ability to generate reactive oxygen species (ROS) via a redox cycling process, by DNA intercalation and by covalent binding to essential proteins and/or DNA [2,3,4,5,6].
In this context, the therapeutic index of most anticancer therapeutic drugs is limited by the magnitude of the relative differences in the biological sensitivities of cancer cells and normal cells. Indeed, most anticancer drugs have a severe toxicity toward normal cells. Thus, the greatest challenge in this area is how to target the active species (e.g., quinones) and their cytotoxicity so that they will distinguish between normal healthy cells and damaged cells. Therefore, there is still an urgent need for new and more effective drugs and innovative drug design strategies.
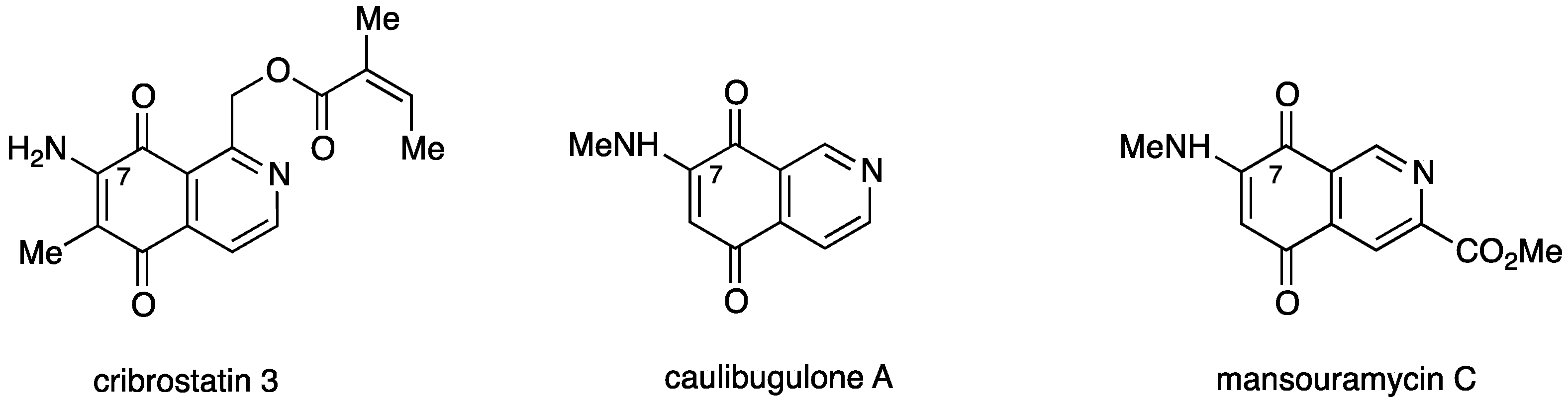
Among the broad variety of N-heterocyclic quinones with anti-cancer activity are the naturally occurring 7-aminoisoquinolinequinones such as cribrostatin 3 [7], caulibugulone A [8], and mansouramycin C [9] (Figure 1).
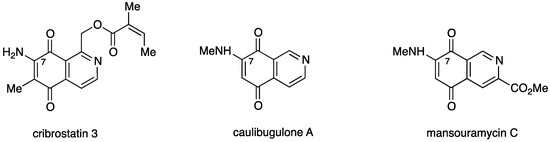
Figure 1.
Structures of naturally occurring 7-aminoisoquinolinequinones with anticancer activity.
Caulibugulone A was found to be a potent and selective inhibitor of the Cdc25B phosphatase, a potential human oncogene [10], by oxidizing and inactivating the catalytic cysteine of the enzyme through redox cycling and production of ROS [11,12]. In a previous work we reported that a series of synthetic amino- and alkylaminoisoquinolinequinones, structurally related to mansouramycin C, display significant in vitro cytotoxic activity on gastric adenocarcinoma, lung cancer, and human bladder carcinoma, but low selective index referred to human lung fibroblasts [13].
In order to add new members to this promising chemical series to define the critical structural elements required for potency and selectivity, we are interested in evaluating substances containing biologically relevant nitrogen substituents such as natural α-amino acids. Concerning the design of isoquinolinequinone-α-amino acid derivatives, it was based on the cytotoxic activity of α-amino acid-containing natural occurring 1,4-benzoquinones [14,15,16], 1,4-naphthoquinone [17,18,19], and 9,10-anthraquinone [20].
2. Results and Discussion
2.1. Chemistry
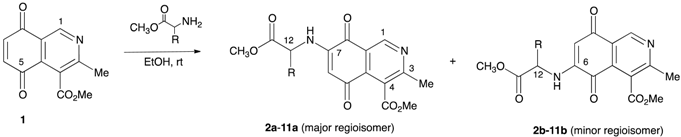
The preparation of the isoquinolinequinone-α-amino acid derivatives was carried out by reaction of the known isoquinolinequinone 1 [13] with a variety of α-amino acid methyl esters in ethanol, under aerobic conditions at room temperature (Table 1). In all cases the reaction gives a mixture of the respective regioisomers, as was observed by TLC and 1H-NMR. Pure samples were isolated by column chromatography of the respective mixtures, but efforts to isolate the minor regioisomers 2b, 5b, 9b, and 11b, were unsuccessful. The structures of the new compounds were fully established by IR, 1H-, 13C-NMR, and high resolution mass spectrometry (HRMS).

Table 1.
Reaction of isoquinolinequinone 1 with α-amino acid methyl esters.
The results show that the amination reaction of quinone 1 proceeds with regioselective preference to give the 7-substituted isomer as the main product. The control of the regioselectivity could be ascribed to the electron-withdrawing effect of the heterocyclic nitrogen atom in 1, which makes the C-5 carbonyl group more electron deficient, directing the nucleophilic attack of the amine to C-7.
It is worth mentioning that since there is no doubt of the inversion of configuration at chiral center, the absolute configuration of the l- and d-α-amino acid methyl ester is reflected in the corresponding aminoisoquinolinequinones. The optical rotations of the products could not be determined due to the darkening of solutions.
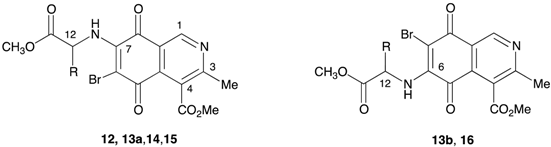
A number of bromine derivatives of the aminoisoquinolinequinones were prepared by reaction of the corresponding precursors with N-bromosuccinimide in methanol at room temperature (Table 2).

Table 2.
Yields of the synthesized bromine derivatives 12, 13a, 13b, 14, 15, and 16.
2.2. In Vitro Cytotoxic Activity of Isoquinolinequinone-Amino Acid Derivatives on Cancer Cell Lines
The series of isoquinolinequinone amino ester conjugates (Table 1) and the bromine derivatives (Table 2) were evaluated for their in vitro cytotoxic activities against normal human lung fibroblast (MRC-5) and three human cancer cells lines: human gastric adenocarcinoma (AGS), human lung cancer (SK-MES-1), and human bladder carcinoma (J82), in 72 h drug exposure assays. The cytotoxic activities of the new compounds were measured using conventional microculture tetrazolium reduction assays [21]. The cytotoxic activities of each of the quinones are expressed in terms of IC50. Etoposide, a clinically used anticancer agent, was taken as a positive control. The cytotoxic activity data are summarized in Table 3 and Table 4.

Table 3.
In vitro cytotoxic effect of compounds 2a–11a and 3b, 4b, 6b–8b, and 10b on human-derived tumor cell lines: AGS (gastric), SK-MES-1 (lung), J82 (bladder), and the non-tumor fibroblasts (MRC-5).

Table 4.
In vitro cytotoxic effect of the bromine compounds 12, 13a, 13b, 14, 15, and 16 on human-derived tumor cell lines: AGS (gastric), SK-MES-1 (lung), J82 (bladder), and the non-tumor fibroblasts (MRC-5).
The structure–activity relationship (SAR) will be firstly analyzed for aminoisoquinolinequinones 2a–11a and 3b, 4b, 6b–8b, and 10b. The data in Table 3 reveals that, in general, moderate to high cytotoxic activity was observed for the members of the series with IC50 values ranging from 0.58 to 15.43 μM, while the positive control, etoposide, exhibits IC50 values in the range of 0.58 to 3.49 μM. A differential sensitivity to the aminoisoquinolinequinones was shown by the three different cell lines. The most sensitive were the gastric-derived AGS cells, while the least sensitive was the urogenital J82 cell line. It may be possible that the J82 cells, because they have a mutated form of p53, are more resistant against an oxidant agent, as has been shown for cisplatin treatment [22]. It may be observed that the location of the amino acid fragment at C-6 (regioisomer b) enhanced the cytotoxic effect as compared to its regioisomer a, where the nitrogen fragment is at C-7. Among the series, compounds 3b, 4b, 8b, and 10b exhibited higher cytotoxic potencies (0.58–3.18 μM), comparable to that of etoposide (0.58–3.49 μM). Nevertheless, such increased activity was accompanied by a loss of selectivity.
Table 3 also shows the values of the Selective Index (SI), calculated as the ratio between IC50 values obtained for normal fibroblast cells and those IC50 values calculated for cancer cells. A high SI value (>2) of a compound gives a selective toxicity towards cancer cells. On the other hand, a compound with SI value <2 is considered to give general toxicity, i.e., it can also cause cytotoxicity on normal cells [23]. Based on this, compounds 2a, 6a, and 10a exhibit high cytotoxic activity on cancer cells (mean IC50 values <4.0 μM) and selective effect on cancer cells (MSI ≥ 2.24). Compound 10a, derived from d-phenylalanine, appears as the most active compound (mean IC50 = 2.83 μM) and selective on cancer cells (mean SI = 2.63). It is noteworthy that compound 7a, derived from l-tyrosine, is less potent and has a MSI ≤ 2 as compared to 6a, derived from l-phenylalanine, suggesting that the insertion of a p-hydroxyl group in the amino acid fragment of the latter decreases the cytotoxicity against cancer cells. Inspection of the data in Table 3 indicates that the size of the amino acid fragment is not a determining factor in the resultant cytotoxic activity. Also, it is worth noting that the enantiomeric pairs 2a/9a, 6a/10a, and 8a/11a exhibit differences in terms of their cytotoxic activity and selectivity index. These facts suggest that chiral recognition is probably involved in the mechanism of cytotoxic activity.
The cytotoxic properties of the bromine derivative series are summarized in Table 4. The data reveal that the bromine insertion at C-6 in precursors 2a and 3a (as in 12, 13a) and at C-7 in 3b (as in 13b) does not induce significant changes in the cytotoxic activity and selectivity. Now, by comparing them to each respective precursor, bromine substitution in compounds 12, 13a, and 15 enhances their cytotoxicity activity but they lose the selectivity shown by their precursors 2a, 3a, and 6a, respectively. It is noteworthy that the bromine insertion at C-6 in compound 4a (as in 14), dramatically influences the cytotoxic activity of the aminoisoquinolinequinone scaffold. In fact, compound 14 exhibits selective and potent cytotoxic activity on lung and bladder cancer cells (MSI > 45) but not on normal fibroblasts (SI > 100) or gastric cancer cells. This compound is a special case: indeed, it has shown extremely low activity against AGS cells, the cancer cell line of highest sensitivity, as well as against healthy fibroblasts (MRC-5). We do not have any explanation for such low cytotoxic activities.
In order to gain insight into the high selectivity of compound 14 on MRC-5 cells, we explored further its cytotoxic properties in other lines including T24 (bladder), DU-145 (prostate), MCF-7 (breast), and normal human fibroblasts (AG 1523). In these assays the anticancer drugs tamoxifen and 5-fluorouracil were used as positive controls (Table 5). The results show that compound 14 induces a high cytotoxic activity in cancer cells (IC50 0.50–0.67 μM) while it shows rather low activity in healthy fibroblasts (AG 1523). These results provide support to the absence of effect of 14 on fibroblasts (MRC-5) at IC50 level <100 μM, thus indicating that compound 14 exhibits a fairly high selective index.

Table 5.
In vitro cytotoxic effect of compound 14 on human-derived tumor cell lines: T24 (bladder), DU-145 (prostate), MCF7 (breast) and healthy non-tumor fibroblasts (AG 1523).
3. Experimental Section
3.1. General Information
All the solvents and reagents were purchased from different companies such as Aldrich (St. Louis, MO, USA) and Merck (Darmstadt, Germany) and were used as supplied. Melting points were determined on a Stuart Scientific SMP3 (Staffordshire, UK) apparatus and are uncorrected. The IR spectra were recorded on an FT IR Bruker spectrophotometer, model Vector 22 (Bruker, Rheinstetten, Germany), using KBr disks, and the wave numbers are given in cm−1. 1H-NMR spectra were recorded on a Bruker Avance-400 instrument (Bruker) in deuterochloroform (CDCl3). 13C-NMR spectra were obtained in CDCl3 at 100 MHz. Bidimensional NMR techniques and distortionless enhancement by polarisation transfer (DEPT) were used for signal assignment. Chemical shifts are expressed in ppm downfield relative to tetramethylsilane. Data for 1H-NMR spectra are reported as follows: s = singlet, d =doublet, m = multiplet and the coupling constants (J) in Hz. The HRMS spectra were obtained on a Thermo Finnigan spectrometer, model MAT 95XP and LTQ-Orbitrap mass spectrometer (Thermo-Fisher Scientific, Waltham, MA, USA) with the analysis performed using an atmospheric-pressure chemical ionization (APCI) source operated in positive mode. Silica gel Merck 60 (70–230 mesh, from Merck) was used for preparative column chromatography and TLC aluminum foil 60F254 for analytical thin layer chromatography (TLC). Isoquinolinequinone 1 was prepared according to the procedure previously reported [13].
3.2. Synthesis
3.2.1. Preparation of Isoquinolinequinone-Amino Acid Derivatives. General Procedure
Suspensions of isoquinolinequinone 1 (1 equiv.), l- or d-α-amino acid methyl esters hydrochloride (2 equiv.) and NaOAc (2 equiv.) in ethanol (15 mL) were left with stirring at room temperature (rt) after completion of the reaction as indicated by TLC. The solvent was removed under reduced pressure and the residue was purified by chromatography over silica gel (90:10 CH2Cl2/EtOAc) to yield mixtures of regioisomers, in ratios determined by 1H-NMR in CDCl3. Further column chromatography of the mixture of isomers over silica gel (CH2Cl2), provided pure samples of the regioisomers.
Methyl 6- and 7-(1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (2a, 2b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-alanine methyl ester hydrochloride (2 h). Compound 2a (less polar, 104 mg, 0.31 mmol, 53%): orange solid, mp: 141–143 °C; IR νmax: 3370 and 3324 (N-H), 2954 and 2930 (C-H), 1745 (C=O ester); 1687 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.57 (d, 3H, J = 6.8 Hz, 12-Me), 2.65 (s, 3H, 3-Me), 3.81 (s, 3H, 12-CO2Me), 4.03 (s, 3H, 4-CO2Me), 4.13 (m, 1H, 12-H), 5.70 (s, 1H, 6-H), 6.46 (d, 1H, J = 7.2 Hz, NH), 9.19 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 17.6 (CH3, 12-CH3), 23.1 (CH3, C-9), 50.7 (CH, C-12), 53.1 (CH3, C-10), 53.2 (CH3, C-14), 102.4 (CH, C-6), 122.0 (C, C-8a), 126.2 (C, C-4), 135.8 (C, C-4a), 146.4 (C, C-7), 148.3 (CH, C-1), 163.0 (C, C-3), 168.8 (C, C-11), 171.7 (C, C-13), 180.1 (C, C-5), 180.7 (C, C-8); HRMS (M+): m/z calcd. for C16H16N2O6: 332.10084; found: 332.10707. Attempts to isolate a pure sample of 2b were unsuccessful.
Methyl 6- and 7-(1-methoxy-3-methyl-1-oxobutan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydro-isoquinoline-4-carboxylate (3a, 3b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-valine methyl ester hydrochloride (2 h). Compound 3a (less polar, 128 mg, 0.36 mmol, 58%): orange solid, mp: 91.5–93 °C; IR νmax: 3368 (N-H), 2970 and 2930 (C-H), 1734 (C=O ester); 1686 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.02 (d, 3H, J = 6.8 Hz, 12-C-Me), 1.08 (d, 3H, J = 6.8 Hz, 12-C-Me), 2.31 (m, 1H, 12-C-CH), 2.65 (s, 3H, 3-Me), 3.80 (s, 3H, 12-CO2Me), 3.92 (m, 1H, 12-H), 4.03 (s, 3H, 4-CO2Me), 5.73 (s, 1H, 6-H), 6.48 (d, 1H, J = 8.0 Hz, NH), 9.19 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 18.4 (CH3, 12-CHCH3), 18.8 (CH3, 12-CHCH3), 22.9 (CH3, C-9), 31.2 (CH, 12-CH), 52.6 (CH3, C-14), 53.1 (CH3, C-10), 60.5 (CH, C-12), 102.2 (CH, C-6), 121.8 (C, C-8a), 126.0 (C, C-4), 135.6 (C, C-4a), 147.0 (C, C-7), 148.1 (CH, C-1), 162.8 (C, C-3), 168.6 (C, C-11), 170.5 (C, C-13), 180.0 (C, C-5), 180.5 (C, C-8); HRMS (M+): m/z calcd. for C18H20N2O6: 360.13214; found: 360.13813.
Compound 3b (more polar, 15 mg, 0.04 mmol, 7%): orange solid, mp: 81.5–83 °C; IR νmax: 3375 (N-H), 2959 and 2931 (C-H), 1739 (C=O ester); 1686 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.01 (d, 3H, J = 6.8 Hz, 12-C-Me), 1.06 (d, 3H, J = 6.8 Hz, 12-C-Me), 2.29 (m, 1H, 12-C-CH), 2.66 (s, 3H, 3-Me), 3.80 (s, 3H, 12-CO2Me), 3.89 (m, 1H, 12-H), 4.06 (s, 3H, 4-CO2Me), 5.72 (s, 1H, 7-H), 6.22 (d, 1H, J = 8.4 Hz, NH), 9.28 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 18.5 (CH3, 12-CHCH3), 18.9 (CH3, 12-CHCH3), 22.7 (CH3, C-9), 31.3 (CH, 12-CH), 52.7 (CH3, C-14), 53.4 (CH3, C-10), 60.6 (CH, C-12), 102.3 (CH, C-7), 122.7 (C, C-8a), 125.1 (C, C-4), 132.3 (C, C-4a), 146.9 (C, C-6), 148.8 (CH, C-1), 160.3 (C, C-3), 168.4 (C, C-11), 170.7 (C, C-13), 181.0 (C, C-5), 181.8 (C, C-8); HRMS (M+): m/z calcd. for C18H20N2O6: 360.13214; found: 360.13788.
Methyl 6- and 7-(1-methoxy-4-methyl-1-oxopentan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (4a, 4b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-leucine methyl ester hydrochloride (2 h). Compound 4a (less polar, 132 mg, 0.35 mmol, 56%): orange solid, mp: 90.5–92.5 °C; IR νmax: 3371 and 3326 (N-H), 2956 and 2929 (C-H), 1749 (C=O ester); 1693 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 0.85 (d, 3H, J = 6.4 Hz, 12-CH2-C-Me), 0.91 (d, 3H, J = 6.4 Hz, 12-CH2-C-Me), 1.68 (m, 1H, 12-CH2-CH), 1.72 (m, 2H, 12-CH2), 2.57 (s, 3H, 3-Me), 3.70 (s, 3H, 12-CO2Me), 3.95 (s, 4H, 4-CO2Me and 12-H), 5.64 (s, 1H, 6-H), 6.26 (d, 1H, J = 8.0 Hz, NH), 9.10 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.0 (CH3, 2-CHCH3), 22.5 (CH3, 12-CHCH3), 23.0 (CH3, C-9), 25.0 (CH, 12-CH2CH), 40.7 (CH, 12-CH), 52.8 (CH3, C-14), 53.1 (CH3, C-10), 53.6 (CH, C-12), 102.2 (CH, C-6), 121.8 (C, C-8a), 126.0 (C, C-4), 135.6 (C, C-4a), 146.8 (C, C-7), 148.1 (CH, C-1), 162.9 (C, C-3), 168.6 (C, C-11), 171.5 (C, C-13), 180.0 (C, C-5), 180.6 (C, C-8); HRMS (M+): m/z calcd. for C19H22N2O6: 374.14779; found: 374.15351.
Compound 4b (more polar, 17 mg, 0.04 mmol, 7%): orange solid, mp: 58.5–60.5 °C; IR νmax: 3367 (N-H), 2956 and 2928 (C-H), 1739 (C=O ester); 1687 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 0.93 (d, 3H, J = 6.0 Hz, 12-CH2-C-Me), 0.99 (d, 3H, J = 6.0 Hz, 12-CH2-C-Me), 1.76 (m, 3H, 12-CH2 and 12-CH2-CH), 2.66 (s, 3H, 3-Me), 3.78 (s, 3H, 12-CO2Me), 4.03 (s, 4H, 4-CO2Me and 12-H), 5.72 (s, 1H, 7-H), 6.01 (d, 1H, J = 8.0 Hz, NH), 9.28 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.1 (CH3, C-9), 22.7 (2CH3, 12-CHCH3), 25.0 (CH, 12-CH2CH), 40.9 (CH, 12-CH), 53.0 (CH3, C-14), 53.4 (CH3, C-10), 53.7 (CH, C-12), 102.4 (CH, C-7), 122.7 (C, C-8a), 125.0 (C, C-4), 132.3 (C, C-4a), 146.6 (C, C-6), 148.8 (CH, C-1), 160.4 (C, C-3), 168.4 (C, C-11), 171.7 (C, C-13), 181.0 (C, C-5), 181.9 (C, C-8); HRMS (M+): m/z calcd. for C19H22N2O6: 374.14779; found: 374.15335.
Methyl 6- and 7-(1-methoxy-4-(methylthio)-1-oxobutan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (5a, 5b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-methionine methyl ester hydrochloride (1 h 30 min). Compound 5a (less polar, 125 mg, 0.32 mmol, 52%): orange solid, mp: 66.5–68 °C; IR νmax: 3353 (N-H), 2951 and 2918 (C-H), 1738 (C=O ester); 1681 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.11 (s, 3H, Me-S), 2.23 (m, 2H, S-CH2), 2.60 (m, 2H, 12-CH2), 2.66 (s, 3H, 3-Me), 3.82 (s, 3H, 12-CO2Me), 4.03 (s, 3H, 4-CO2Me), 4.33 (m, 1H, 12-H), 5.830 (s, 1H, 6-H), 6.59 (d, 1H, J = 8.4 Hz, NH), 9.19 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 15.5 (CH3, S-CH3), 22.9 (CH3, C-9), 29.8 (CH2, 12-CH2), 30.6 (CH2, S-CH2), 53.0 (CH3, C-14), 53.1 (CH3, C-10), 53.6 (CH, C-12), 102.4 (CH, C-6), 121.8 (C, C-8a), 126.0 (C, C-4), 135.5 (C, C-4a), 146.7 (C, C-7), 148.0 (CH, C-1), 162.8 (C, C-3), 168.5 (C, C-11), 170.8 (C, C-13), 180.0 (C, C-5), 180.5 (C, C-8); HRMS (M+): m/z calcd. for C18H20N2O6S: 392.10421; found: 392.10987. Attempts to isolate a pure sample of 5b were unsuccessful.
Methyl 6- and 7-(1-methoxy-1-oxo-3-phenylpropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (6a, 6b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-phenylalanine methyl ester hydrochloride (2 h 30 min).Compound 6a (less polar, 149 mg, 0.37 mmol, 60%): orange solid, mp: 118–119.5 °C; IR νmax: 3353 and 3287 (N-H), 2947 and 2927 (C-H), 1735 (C=O ester); 1685 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.64 (s, 3H, 3-Me), 3.17 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.26 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.76 (s, 3H, 12-CO2Me), 4.02 (s, 3H, 4-CO2Me), 4.31 (m, 1H, 12-H), 5.68 (s, 1H, 6-H), 6.42 (d, 1H, J = 7.6 Hz, NH), 7.11 (m, 2H, arom), 7.28 (m, 3H, arom), 9.16 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 23.0 (CH3, C-9), 37.5 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.2 (CH3, C-10), 56.1 (CH, C-12), 102.5 (CH, C-6), 121.8 (C, C-8a), 126.1 (C, C-4), 127.8 (C, C-4’), 129.0 (2CH, C-2’ and C-6’), 129.1 (2CH, C-3’ and C-5’), 134.8 (C, 12-CH2, C-1’), 135.6 (C, C-4a), 146.4 (C, C-7), 148.2 (CH, C-1), 163.0 (C, C-3), 168.7 (C, C-11), 170.3 (C, C-13), 179.9 (C, C-8), 180.6 (C, C-5); HRMS (M+): m/z calcd. for C22H20N2O6: 408.13214; found: 408.13756.
Compound 6b (more polar, 17 mg, 0.04 mmol, 7%): orange solid, mp: 72–73.5 °C; IR νmax: 3350 and 3284 (N-H), 2946 and 2929 (C-H), 1735 (C=O ester); 1684 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.65 (s, 3H, 3-Me), 3.14 (dd, 1H, J = 6.8; 14 Hz, 12-CH2), 3.25 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.76 (s, 3H, 12-CO2Me), 4.04 (s, 3H, 4-CO2Me), 4.31 (m, 1H, 12-H), 5.67 (s, 1H, 7-H), 6.18 (d, 1H, J = 7.6 Hz, NH), 7.13 (m, 2H, arom), 7.29 (m, 3H, arom), 9.26 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.7 (CH3, C-9), 37.7 (CH2, 12-CH2), 53.0 (CH3, C-14), 53.4 (CH3, C-10), 56.3 (CH, C-12), 102.6 (CH, C-7), 122.7 (C, C-8a), 125.1 (C, C-4), 127.8 (C, C-4’), 129.1 (2CH, C-2’ and C-6’), 129.2 (2CH, C-3’ and C-5’), 132.3 (C, C-4a), 134.8 (C, 12-CH2, C-1’), 146.3 (C, C-6), 148.8 (CH, C-1), 160.4 (C, C-3), 168.4 (C, C-11), 170.5 (C, C-13), 180.8 (C, C-5), 181.8 (C, C-8); HRMS (M+): m/z calcd. for C22H20N2O6: 408.13214; found: 408.13762.
Methyl 6- and 7-(3-(4-hydroxyphenyl)-1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (7a, 7b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-tyrosine methyl ester hydrochloride (1 h 40 min). Compound 7a (less polar, 141 mg, 0.33 mmol, 56%): orange solid, mp: 93–94.5 °C; IR νmax: 3358 (N-H), 2953 and 2851 (C-H), 1738 (C=O ester); 1683 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.63 (s, 3H, 3-Me), 3.06 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.14 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.75 (s, 3H, 12-CO2Me), 4.01 (s, 3H, 4-CO2Me), 4.26 (m, 1H, 12-H), 5.68 (s, 1H, 6-H), 6.49 (d, 1H, J = 8.0 Hz, NH), 6.71 (d, 2H, J = 8.4 Hz, arom), 6.91 (d, 2H, J = 8.4 Hz, arom), 7.75 (br s, 1H, OH), 9.12 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.7 (CH3, C-9), 36.4 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.3 (CH3, C-10), 56.2 (CH, C-12), 102.1 (CH, C-6), 116.0 (2CH, C-3’ and C-5’), 122.0 (C, C-8a), 125.9 (C, C-4), 126.2 (C, 12-CH2, C-1’), 130.2 (2CH, C-2’ and C-6’), 135.8 (C, C-4a), 146.6 (C, C-7), 147.9 (CH, C-1), 155.9 (CH, C4’-OH), 162.7 (C, C-3), 168.7 (C, C-11), 170.5 (C, C-13), 179.5 (C, C-8), 180.6 (C, C-5); HRMS (M+): m/z calcd. for C22H20N2O7: 424.12705; found: 424.13256.
Compound 7b (more polar, 13 mg, 0.03 mmol, 5%): orange solid, mp: 76.5–78.5 °C; IR νmax: 3360 (N-H), 2953 and 2848 (C-H), 1739 (C=O ester); 1685 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.65 (s, 3H, 3-Me), 3.09 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.16 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.76 (s, 3H, 12-CO2Me), 4.04 (s, 3H, 4-CO2Me), 4.25 (m, 1H, 12-H), 5.23 (br s, 1H, OH), 5.67 (s, 1H, 7-H), 6.19 (d, 1H, J = 7.6 Hz, NH), 6.76 (d, 2H, J = 8.4 Hz, arom), 6.97 (d, 2H, J = 8.4 Hz, arom), 9.27 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.6 (CH3, C-9), 36.7 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.35 (CH3, C-10), 56.2 (CH, C-12), 102.3 (CH, C-7), 115.9 (2CH, C-3’ and C-5’), 122.6 (C, C-8a), 125.3 (C, C-4), 126.3 (C, 12-CH2, C-1’), 130.3 (2CH, C-2’ and C-6’), 134.9 (C, C-4a), 146.6 (C, C-6), 147.4 (CH, C-1), 155.9 (CH, C4’-OH), 162.8 (C, C-3), 168.6 (C, C-11), 170.5 (C, C-13), 179.5 (C, C-8), 179.6 (C, C-5); HRMS (M+): m/z calcd. for C22H20N2O7: 424.12705; found: 424.13239.
Methyl 6- and 7-(3-(1H-indol-3-yl)-1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (8a, 8b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and l-tryptophan methyl ester hydrochloride (2 h). Compound 8a (less polar, 214 mg, 0.48 mmol, 67%): orange solid, mp: 98.5–100 °C; IR νmax: 3360 (N-H), 2952 and 2927 (C-H), 1737 (C=O ester); 1681 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.63 (s, 3H, 3-Me), 3.35 (dd, 1H, J = 6.4; 15 Hz, 12-CH2), 3.40 (dd, 1H, J = 5.2; 15 Hz, 12-CH2), 3.70 (s, 3H, 12-CO2Me), 4.01 (s, 3H, 4-CO2Me), 4.33 (m, 1H, 12-CH), 5.64 (s, 1H, 6-H), 6.47 (d, 1H, J = 7.6 Hz, NH), 6.98 (m, 1H, CH), 7.07 (m, 1H, arom), 7.13 (m, 1H, arom), 7.28 (d, 1H, J = 8.0 Hz, arom), 7.45 (d, 1H, J = 8.0 Hz, arom), 8.45 (br s, 1H, NH), 9.10 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 23.0 (CH3, C-9), 27.5 (CH2, 12-CH2), 53.0 (CH3, C-14), 53.2 (CH3, C-10), 55.5 (CH, C-12), 102.2 (CH, C-6), 108.8 (C, 12-CH2C), 111.6 (CH, HN-CCH), 118.2 (CH, 12-CH2CCCH), 120.0 (CH, HN-CCHCH), 121.8 (C, C-8a), 122.6 (CH, CH2CCCHCH), 123.2 (CH, HNCH), 126.0 (C, C-4), 127.0 (C, 12-CH2CC), 135.7 (C, C-4a), 136.2 (C, HNC), 146.7 (C, C-7), 148.1 (CH, C-1), 162.8 (C, C-3), 168.8 (C, C-11), 170.8 (C, C-13), 179.8 (C, C-8), 180.6 (C, C-5); HRMS (M+): m/z calcd. for C24H21N3O6: 447.14304; found: 447.14845.
Compound 8b (more polar, 29 mg, 0.06 mmol, 9%): orange solid, mp: 89–91 °C; IR νmax: 3370 (N-H), 2952 and 2925 (C-H), 1737 (C=O ester); 1686 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.64 (s, 3H, 3-Me), 3.36 (dd, 1H, J = 6.4; 15 Hz, 12-CH2), 3.43 (dd, 1H, J = 5.2; 15 Hz, 12-CH2), 3.71 (s, 3H, 12-CO2Me), 4.01 (s, 3H, 4-CO2Me), 4.36 (m, 1H, 12-H), 5.66 (s, 1H, 7-H), 6.26 (d, 1H, J = 8.0 Hz, NH), 7.02 (m, 1H, CH), 7.12 (m, 1H, arom), 7.19 (m, 1H, arom), 7.35 (d, 1H, J = 8.0 Hz, arom), 7.50 (d, 1H, J = 8.0 Hz, arom), 8.23 (br s, 1H, NH), 9.24 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.7 (CH3, C-9), 27.7 (CH2, 12-CH2), 53.0 (CH3, C-14), 53.4 (CH3, C-10), 55.5 (CH, C-12), 102.4 (CH, C-7), 109.1 (C, 12-CH2C), 111.6 (CH, HN-CCH), 118.4 (CH, 12-CH2CCCH), 120.1 (CH, HN-CCHCH), 122.7 (C and CH, C-8a y CH2CCCHCH), 123.1 (CH, HNCH), 125.0 (C, C-4), 127.1 (C, 12-CH2CC), 132.3 (C, C-4a), 136.3 (C, HNC), 146.4 (C, C-6), 148.7 (CH, C-1), 160.3 (C, C-3), 168.4 (C, C-11), 170.9 (C, C-13), 180.8 (C, C-5), 181.8 (C, C-8); HRMS (M+): m/z calcd. for C24H21N3O6: 447.14304; found: 447.14818.
Methyl 6- and 7-(1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (9a, 9b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and d-alanine methyl ester hydrochloride (2 h). Compound 9a (less polar, 104 mg, 0.31 mmol, 50%): orange solid, mp: 140.5–142 °C; IR νmax 3370 and 3324 (N-H), 2954 and 2930 (C-H), 1745 (C=O ester); 1687 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.57 (d, 3H, J = 6.8 Hz, Me), 2.65 (s, 3H, 3-Me), 3.81 (s, 3H, 12-CO2Me), 4.03 (s, 3H, 4-CO2Me), 4.13 (m, 1H, 12-H), 5.70 (s, 1H, 6-H), 6.46 (d, 1H, J = 7.2 Hz, NH), 9.19 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 17.6 (CH3, 12-CH3), 23.1 (CH3, C-9), 50.7 (CH, C-12), 53.1 (CH3, C-10), 53.2 (CH3, C-14), 102.4 (CH, C-6), 122.0 (C, C-8a), 126.2 (C, C-4), 135.8 (C, C-4a), 146.4 (C, C-7), 148.3 (CH, C-1), 163.0 (C, C-3), 168.8 (C, C-11), 171.7 (C, C-13), 180.1 (C, C-5), 180.7 (C, C-8); HRMS (M+): m/z calcd. for C16H16N2O6: 332.10084; found: 332.10193. Attempts to isolate a pure sample of 9b were unsuccessful.
Methyl 6- and 7-(1-methoxy-1-oxo-3-phenylpropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (10a, 10b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and d-phenylalanine methyl ester hydrochloride (2 h 30 min).Compound 10a (less polar, 139 mg, 0.34 mmol, 58%): orange solid, mp: 118.5–120 °C; IR νmax: 3353 and 3287 (N-H), 2947 and 2927 (C-H), 1735 (C=O ester); 1685 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.64 (s, 3H, 3-Me), 3.17 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.26 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.76 (s, 3H, 12-CO2Me), 4.02 (s, 3H, 4-CO2Me), 4.31 (m, 1H, 12-H), 5.68 (s, 1H, 6-H), 6.42 (d, 1H, J = 7.6 Hz, NH), 7.11 (m, 2H, arom), 7.28 (m, 3H, arom), 9.16 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 23.0 (CH3, C-9), 37.5 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.2 (CH3, C-10), 56.1 (CH, C-12), 102.5 (CH, C-6), 121.8 (C, C-8a), 126.1 (C, C-4), 127.8 (C, C-4’), 129.0 (2CH, C2’ and C6’), 129.1 (2CH, C-3’ and C-5’), 134.8 (C, 12-CH2C1’), 135.6 (C, C-4a), 146.4 (C, C-7), 148.2 (CH, C-1), 163.0 (C, C-3), 168.7 (C, C-11), 170.3 (C, C-13), 179.9 (C, C-8), 180.6 (C, C-5); HRMS (M+): m/z calcd. for C22H20N2O6: 408.13214; found: 408.13759.
Compound 10b (more polar, 12 mg, 0.03 mmol, 5%): orange solid, mp: 71.5–73 °C; IR νmax: 3350 and 3284 (N-H), 2946 and 2929 (C-H), 1735 (C=O ester); 1684 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.65 (s, 3H, 3-Me), 3.14 (dd, 1H, J = 6.8; 14 Hz, 12-CH2), 3.25 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.76 (s, 3H, 12-CO2Me), 4.04 (s, 3H, 4-CO2Me), 4.31 (m, 1H, 12-H), 5.67 (s, 1H, 7-H), 6.18 (d, 1H, J = 7.6 Hz, NH), 7.13 (m, 2H, arom), 7.29 (m, 3H, arom), 9.26 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.7 (CH3, C-9), 37.7 (CH2, 12-CH2), 53.0 (CH3, C-14), 53.4 (CH3, C-10), 56.3 (CH, C-12), 102.6 (CH, C-7), 122.7 (C, C-8a), 125.1 (C, C-4), 127.8 (C, C-4’), 129.1 (2CH, C-2’ and C-6’), 129.2 (2CH, C-3’ and C-5’), 132.3 (C, C-4a), 134.8 (C, 12-CH2C1’), 146.3 (C, C-6), 148.8 (CH, C-1), 160.4 (C, C-3), 168.4 (C, C-11), 170.5 (C, C-13), 180.8 (C, C-5), 181.8 (C, C-8); HRMS (M+): m/z calcd. for C22H20N2O6: 408.13214; found: 408.13757.
Methyl 6- and 7-(3-(1H-indol-3-yl)-1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (11a, 11b). The mixture of regioisomers was prepared from 1 (200 mg, 0.87 mmol) and d-tryptophan methyl ester hydrochloride (2 h). Compound 11a (less polar, 205 mg, 0.46 mmol, 66%): orange solid, mp: 99–100.5 °C; IR νmax: 3360 (N-H), 2952 and 2927 (C-H), 1737 (C=O ester); 1681 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.63 (s, 3H, 3-Me), 3.35 (dd, 1H, J = 6.4; 15 Hz, 12-CH2), 3.40 (dd, 1H, J = 5.2; 15 Hz, 12-CH2), 3.70 (s, 3H, 12-Me), 4.01 (s, 3H, 4-CO2Me), 4.33 (m, 1H, 12-H), 5.64 (s, 1H, 6-H), 6.47 (d, 1H, J = 7.6 Hz, NH), 6.98 (m, 1H, CH), 7.07 (m, 1H, arom), 7.13 (m, 1H, arom), 7.28 (d, 1H, J = 8.0 Hz, arom), 7.45 (d, 1H, J = 8.0 Hz, arom), 8.45 (br s, 1H, NH), 9.10 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 23.0 (CH3, C-9), 27.5 (CH2, 12-CH2), 53.0 (CH3, C-14), 53.2 (CH3, C-10), 55.5 (CH, C-12), 102.2 (CH, C-6), 108.8 (C, 12-CH2C), 111.6 (CH, HN-CCH), 118.2 (CH, 12-CH2CCCH), 120.0 (CH, HN-CCHCH), 121.8 (C, C-8a), 122.6 (CH, CH2CCCHCH), 123.2 (CH, HNCH), 126.0 (C, C-4), 127.0 (C, 12-CH2CC), 135.7 (C, C-4a), 136.2 (C, HNC), 146.7 (C, C-7), 148.1 (CH, C-1), 162.8 (C, C-3), 168.8 (C, C-11), 170.8 (C, C-13), 179.8 (C, C-8), 180.6 (C, C-5); HRMS (M+): m/z calcd. for C24H21N3O6: 447.14304; found: 447.14867. Attempts to isolate a pure sample of 11b were unsuccessful.
3.2.2. Synthesis of Bromoisoquinolinequinones 12, 13a,b, 14, 15, 16. General Procedure
A solution of the required aminoisoquinolinequinone (1 equiv.), N-bromosuccinimide (NBS) (1.1 equiv.) and methanol (15 mL) was left with stirring at rt after completion of the reaction as indicated by TLC. The solvent was removed under reduced pressure and the residue was column chromatographed over silica gel (CH2Cl2/EtOAc 90:10) to yield the corresponding bromoisoquinolinequinones.
Methyl 6-bromo-7-(1-methoxy-1-oxopropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (12). Prepared from 2a (70 mg, 0.21 mmol) and NBS (20 min, 65 mg, 0.16 mmol, 76% yield): orange solid, mp: 120.5–122 °C; IR νmax: 3370 and 3320 (N-H), 2954 and 2924 (C-H), 1738 (C=O ester); 1685 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.62 (d, 3H, J = 6.8 Hz, 12-Me), 2.65 (s, 3H, 3-Me), 3.83 (s, 3H, 12-CO2Me), 4.05 (s, 3H, 4-CO2Me), 4.13 (m, 1H, 12-H), 6.55 (bs, 1H, NH), 9.17 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 20.2 (CH3, 12-CH3), 21.1 (CH3, C-9), 52.6 (CH, C-12), 53.1 (CH3, C-10), 53.3 (CH3, C-14), 121.3 (C, C-8a), 126.3 (C, C-4), 134.1 (C, C-4a), 148.7 (2C, C, C-7 and CH, C-1), 163.1 (C, C-3), 168.2 (C, C-11), 172.7 (C, C-13), 178.5 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C16H15BrN2O6: 410.01135; found: 410.01755.
Methyl 6-bromo-(1-methoxy-3-methyl-1-oxobutan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (13a). Prepared from 3a (100 mg, 0.28 mmol) and NBS (20 min, 101 mg, 0.23 mmol, 82% yield): orange solid, mp: 105.2–107 °C; IR νmax: 3368 (N-H), 2965 and 2934 (C-H), 1725 (C=O ester); 1680 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.02 (d, 3H, J = 6.8 Hz, 12-C-Me), 1.08 (d, 3H, J = 6.8 Hz, 12-C-Me), 2.33 (m, 1H, 12-CH), 2.65 (s, 3H, 3-Me), 4.03 (s, 3H, 12-CO2Me), 4.05 (s, 3H, 4-CO2Me), 4.13 (m, 1H, 12-H), 6.22 (bs, 1H, NH), 9.18 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 17.9 (CH3, 12-CHCH3), 18.4 (CH3, 12-CHCH3), 23.1 (CH3, C-9), 32.5 (CH, 12-CH), 52.7 (CH3, C-14), 53.3 (CH3, C-10), 60.5 (CH, C-12), 121.4 (C, C-8a), 126.4 (C, C-4), 135.6 (C, C-4a), 148.7 (2C, C-7 and CH, C-1), 163.1 (C, C-3), 168.2 (C, C-11), 171.2 (C, C-13), 178.6 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C18H19N2O6Br: 438.04265; found: 438.04810.
Methyl 7-bromo-(1-methoxy-3-methyl-1-oxobutan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (13b). Prepared from 3b (50 mg, 0.14 mmol) and NBS (30 min, 49 mg, 0.11 mmol, 78% yield): orange solid, mp: 105–106.6 °C; IR νmax: 3370 (N-H), 2962 and 2931 (C-H), 1743 (C=O ester); 1682 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 1.01 (d, 3H, J = 6.8 Hz, 12-C-Me), 1.07 (d, 3H, J = 6.8 Hz, 12-C-Me), 2.29 (m, 1H, 12-C-CH), 2.65 (s, 3H, 3-Me), 3.80 (s, 3H, 12-CO2Me), 4.04 (m, 1H, 12-H), 4.05 (s, 3H, 4-CO2Me), 6.30 (bs, 1H, NH), 9.29 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 17.8 (CH3, 12-CHCH3), 18.5 (CH3, 12-CHCH3), 22.8 (CH3, C-9), 32.5 (CH, 12-CH), 52.7 (CH3, C-14), 53.3 (CH3, C-10), 61.6 (CH, C-12), 121.8 (C, C-8a), 125.4 (2C, C-4 and C-4a), 146.9 (C, C-6), 149.0 (2C, CH, C-1 and C, C-3), 168.0 (C, C-11), 171.5 (C, C-13), 179.3 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C18H19N2O6Br: 438.04265; found: 438.04785.
Methyl 6-bromo-(1-methoxy-4-methyl-1-oxopentan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (14). Prepared from 4a (100 mg, 0.27 mmol) and NBS (30 min, 98 mg, 0.22 mmol, 80% yield): orange solid, mp: 104.3–106.2 °C; IR νmax: 3370 and 3326 (N-H), 2945 and 2914 (C-H), 1755 (C=O ester); 1710 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 0.98 (d, 3H, J = 6.4 Hz, 12-CH2-C-Me), 1.01 (d, 3H, J = 6.4 Hz, 12-CH2-C-Me), 1.81 (m, 3H, 12-CH2-CH and 12-CH2), 2.65 (s, 3H, 3-Me), 3.79 (s, 3H, 12-CO2Me), 4.07 (s, 4H, 4-CO2Me and 12-H), 6.21 (bs, 1H, NH), 9.17 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.5 (CH3, 2-CHCH3), 22.8 (CH3, 12-CHCH3), 23.0 (CH3, C-9), 25.1 (CH, 12-CH2CH), 42.6 (CH, 12-CH), 52.8 (CH3, C-14), 53.3 (CH3, C-10), 53.5 (CH, C-12), 121.3 (C, C-8a), 126.4 (C, C-4), 134.1 (C, C-4a), 148.7 (2C, C-7 and CH, C-1), 163.0 (C, C-3), 168.1 (C, C-11), 171.1 (C, C-13), 178.5 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C19H21N2O6Br: 452.05830; found: 452.06328.
Methyl 6-bromo-(1-methoxy-1-oxo-3-phenylpropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (15). Prepared from 6a (100 mg, 0.25 mmol) and NBS (40 min, 83 mg, 0.17 mmol, 68% yield): red solid, mp: 125.2–127.5 °C; IR νmax: 3350 and 3285 (N-H), 2944 and 2921 (C-H), 1746 (C=O ester); 1705 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.63 (s, 3H, 3-Me), 3.25 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.27 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.79 (s, 3H, 12-CO2Me), 4.03 (s, 4H, 4-CO2Me and 12-H), 6.31 (bs, 1H, NH), 7.13 (m, 2H, arom), 7.28 (m, 3H, arom), 9.09 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): δ 22.9 (CH3, C-9), 39.9 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.3 (CH3, C-10), 57.7 (CH, C-12), 121.5 (C, C-8a), 126.1 (C, C-4), 127.8 (C, C-4’), 129.1 (2CH, C-2’ and C-6’), 129.4 (2CH, C-3’ and C-5’), 134.6 (2C, 12-CH2C1’ and C, C-4a), 148.6 (2C, C-7 and CH, C-1), 163.0 (C, C-3), 168.2 (C, C-11), 171.3 (C, C-13), 178.5 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C22H19N2O6Br: 486.04265; found: 486.04732.
Methyl 6-bromo-(1-methoxy-1-oxo-3-phenylpropan-2-ylamino)-3-methyl-5,8-dioxo-5,8-dihydroisoquinoline-4-carboxylate (16). Prepared from 10a (80 mg, 0.19 mmol) and NBS (40 min, 63 mg, 0.13 mmol, 68% yield) red solid, mp: 126–127 °C; IR νmax: 3352 and 3285 (N-H), 2950 and 2927 (C-H), 1725 (C=O ester); 1715 (C=O quinone); 1H-NMR (400 MHz, CDCl3): δ 2.63 (s, 3H, 3-Me), 3.19 (dd, 1H, J = 6.4; 14 Hz, 12-CH2), 3.26 (dd, 1H, J = 5.6; 14 Hz, 12-CH2), 3.79 (s, 3H, 12-CO2Me), 4.05 (s, 4H, 4-CO2Me and 12-H), 6.29 (bs, 1H, NH), 7.13 (m, 2H, arom), 7.25 (m, 3H, arom), 9.08 (s, 1H, 1-H); 13C-NMR (100 MHz, CDCl3): 22.9 (CH3, C-9), 39.8 (CH2, 12-CH2), 52.9 (CH3, C-14), 53.2 (CH3, C-10), 57.7 (CH, C-12), 121.4 (C, C-8a), 126.1 (C, C-4), 127.7 (C, C-4’), 128.9 (2CH, C-2’ and C-6’), 129.4 (2CH, C-3’ and C-5’), 134.6 (2C, 12-CH2C1’ and C, C-4a), 148.5 (2C, C-7 and CH, C-1), 163.0 (C, C-3), 168.1 (C, C-11), 171.3 (C, C-13), 178.4 (2C, C-5 and C-8); HRMS (M+): m/z calcd. for C22H19N2O6Br: 486.04265; found: 486.04723.
3.3. In Vitro Cytotoxic Evaluation
Cytotoxicity assay (Table 3 and Table 4). Cell Lines and Culture Conditions: MRC-5 normal human lung fibroblasts (CCL-171), AGS human gastric adenocarcinoma cells (CRL-1739), SK-MES-1 human lung cancer cells (HTB-58), and J82 human bladder carcinoma cells (HTB-1) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). MRC-5, SK-MES-1, and J82 cells were grown in Eagle’s minimal essential medium (EMEM) containing 2 mM l-glutamine, 1 mM sodium pyruvate and 1.5 g/L sodium hydrogen carbonate. AGS cells were grown in Ham F-12 supplemented with 2 mM l-glutamine and 1.5 g/L sodium hydrogen carbonate. Finally, HL-60 cells were grown in RPMI medium. Media were supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 IU/mL penicillin and 100 μg/mL streptomycin and cell cultures were kept in a humidified incubator with 5% CO2 in air at 37 °C. For the cytotoxicity experiments, cells were seeded into 96-well plates at a density of 50,000 cells/mL. After reaching confluence, cells were incubated for three days with compounds at varied concentrations ranging from 0 up to 100 μM while untreated cells (medium containing 1% DMSO) were used as controls. Cytotoxicity was assessed using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) reduction assay. MTT was used at 1 mg/mL and the blue formazan crystals, formed during MTT reduction, were dissolved adding 100 μL of ethanol (acidified with HCl). The absorbance was measured at 550 nm using a Universal Microplate Reader (ELX 800, Bio-Tek Instruments Inc., Winooski, VT, USA). Values were the means of six replicates for each concentration and transformed to percentage of controls. The IC50 value was graphically obtained from the dose–response curves by adjusting them to a sigmoidal model (a + (b − a)/1 + 10(x − c)), where c = log IC50.
Cytotoxicity assay (Table 5). Cell Lines and Culture Conditions: Human cancer cells (T24, DU-145, MCF-7) and normal human fibroblasts (AG 1523 cells) were purchased from ATCC (Manassas, VA, USA) and cultured in high-glucose Dulbecco’s modified Eagle medium (Gibco, Grand Island, NY, USA) supplemented with 10% fetal calf serum, penicillin (100 U/mL), and streptomycin (100 μg/mL). All cultures were maintained in a humidified incubator at 37 °C under 5% CO2. Cells were incubated as monolayer at 37 °C for 48 h with DMSO (control conditions) and varied doses of compound 14, tamoxifen, and 5-fluorouracil. The cytotoxic effect of compounds on cell lines was assessed using the MTT assay as reported previously [24]. Briefly, 10,000 cells/well were plated for 24 h in 96-well plates and, after reaching confluence, the cells were incubated for 48 h with DMSO or in the presence of compounds. Cells were washed twice with warm phosphate-buffered saline (PBS) and further incubated for 2 h with MTT (0.5 mg/mL). The blue formazan crystals were then solubilized by adding 100 μL DMSO/well and optical density of colored solutions was read at 550 nm. Results are expressed as % of MTT reduction compared to untreated control conditions. The calculation of IC50 values was performed by using GraphPad Prism software (San Diego, CA, USA).
4. Conclusions
In conclusion, we have synthesized a series of isoquinolinequinone-amino acid derivatives from 3-methyl-4-methoxycarbonylisoquinoline-5,8-quinone 1 and diverse l- and d-α-amino acid methyl esters. The cytotoxic activity of these isoquinolinequinone-amino acid derivatives and a number of bromine derivatives was evaluated by using the MTT assay. From the current investigation, structure-activity relationships of the aminoisoquinolinequinones demonstrate that compounds 2a, 6a, 10a, and 14, derived from l-alanine, l-phenylalanine, d-phenylalanine, and l-leucine, respectively, are endowed with high cytotoxic potencies and selectivity against cancer cells (gastric, bladder, lung, prostate, and breast). Also, it was deduced that the structure of the amino acid fragment plays a role on the cytotoxic effects and selectivity index. The major incidence of this structural effect is observed in the enantiomeric pair S-2a/R-9a, derived from alanine. Among the tested compounds in this study, the bromine derivative 14, containing the l-leucine moiety at the 7-position, was found to be the most promising congener in the series, on account of its potency and selectivity to inhibit the growth of cancer cells. Compound 14 of novel structure, possessing significant cytotoxic activity on cancer cells, with relative fairly high MSI values, appears to be a new lead compound in the aminoisoquinolinequinone series.
Supplementary Materials
The following are available online at: http://www.mdpi.com/1420-3049/21/9/1199/s1, Figures S1–S7: 1H- and 13C-NMR of compounds 2a, 3a, 3b, 11a, 11b, 13b, and 15.
Acknowledgments
We thank the Fondo Nacional de Ciencia y Tecnología, Chile (Grant No. 1141307), the Fonds Spéciaux de Recherche (FSR), Université Catholique de Louvain, Belgium, and the FNRS (FRFC Grant 2.4555.08), Belgium for financial support to this study.
Author Contributions
J.A.V. proposed the subject and designed the study. V.D., S.S., and J.B. carried out the synthesis of the products. P.B.C. participated in the discussion of the biological results. C.T. and P.B.C. performed the biological evaluation. G.G.M. carried out the HRMS analysis. All the authors read and approved the final manuscript.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Thomson, R.H. Naturally Occurring Quinones IV Recent Advances, 4th ed.; Blackie: London, UK, 1997. [Google Scholar]
- Bolton, J.L.; Trush, M.A.; Penning, T.M.; Dryhurst, G.; Monks, T.J. Role of quinones in toxicology. Chem. Res. Toxicol. 2000, 13, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Powis, G. Metabolism and reactions of quinoid anticancer agents. Pharmacol. Ther. 1987, 35, 57–162. [Google Scholar] [CrossRef]
- O’Brien, P.J. Molecular mechanisms of quinone cytotoxicity. Chem. Biol. Interact. 1991, 80, 1–41. [Google Scholar] [CrossRef]
- Paz, M.M.; Das, A.; Palom, Y.; He, Q.-Y.; Tomasz, M. Selective activation of mitomycin A by thiols to form DNA, cross links and monoadducts: Biochemical basis for the modulation of mitomycin cytotoxicity by the quinone redox potential. J. Med. Chem. 2001, 44, 2834–2842. [Google Scholar] [CrossRef] [PubMed]
- Tudor, G.; Gutierrez, P.; Aguilera-Gutierrez, A.; Sausville, E.A. Cytotoxicity and apoptosis of benzoquinones: Redox cycling, cytochrome c release, and BAD protein expression. Biochem. Pharmacol. 2003, 65, 1061–1075. [Google Scholar] [CrossRef]
- Pettit, G.R.; Knight, J.C.; Collins, J.C.; Herald, D.L.; Pettit, R.K.; Boyd, M.R.; Young, V.G. Antineoplastic agents 430. Isolation and structure of cribrostatins 3, 4, and 5 from the republic of Maldives cribrochalina species. J. Nat. Prod. 2000, 63, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Milanowski, D.J.; Gustafson, K.R.; Kelley, J.A.; McMahon, J.B. Caulibugulones A-F, novel cytotoxic isoquinoline quinones and iminoquinones from the marine bryozoan Caulibugula intermis. J. Nat. Prod. 2004, 67, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Hawas, U.W.; Shaaban, M.; Shaaban, K.A.; Speitling, M.; Maier, A.; Kelter, G.; Fiebig, H.H.; Meiners, M.; Helmke, E.; Laatsch, H. Mansouramycins A–D, cytotoxic isoquinolinequinones from a marine streptomycete. J. Nat. Prod. 2009, 72, 2120–2124. [Google Scholar] [CrossRef] [PubMed]
- Kristjánsdóttir, K.; Rudolph, J. Cdc25 Phosphatases and Cancer. Chem. Biol. 2004, 11, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Wipf, P.; Joo, B.; Nguyen, T.; Lazo, J.S. Synthesis and biological evaluation of caulibugulones A–E. Org. Biomol. Chem. 2004, 2, 2173–2174. [Google Scholar] [CrossRef] [PubMed]
- Brisson, M.; Foster, C.; Wipf, P.; Joo, B.; Tomko, R.J.; Nguyen, T.; Lazo, J.S. Independent Mechanistic Inhibition of Cdc25 Phosphatases by a Natural Product Caulibugulone. Mol. Pharmacol. 2007, 71, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Delgado, V.; Ibacache, A.; Theoduloz, C.; Valderrama, J.A. Synthesis and in vitro cytotoxic evaluation of aminoquinones structurally related to marine isoquinolinequinones. Molecules 2012, 17, 7042–7056. [Google Scholar] [CrossRef] [PubMed]
- Shigemori, H.; Madono, T.; Sasaki, T.; Mikami, Y.; Kobayashi, J. Nakijiquinones A and B, new antifungal sesquiterpenoid quinones with an amino acid residue from an Okinawan marine sponge. Tetrahedron 1994, 50, 8347–8354. [Google Scholar] [CrossRef]
- Concepción Lozada, M.; Soria-Arteche, O.; Ramírez Apan, M.T.; Nieto-Camacho, A.; Enríquez, R.G.; Izquierdo, T.; Jiménez-Corona, A. Synthesis, cytotoxic and antioxidant evaluations of amino derivatives from perezone. Bioorg. Med. Chem. 2012, 20, 5077–5084. [Google Scholar] [CrossRef] [PubMed]
- Vilipić, J.; Novaković, I.; Stanojković, T.; Matić, I.; Šegan, D.; Kljajić, Z.; Sladić, D. Synthesis and biological activity of amino acid derivatives of avarone and its model compound. Bioorg. Med. Chem. 2015, 23, 6930–6942. [Google Scholar] [CrossRef] [PubMed]
- Rahimipour, S.; Weiner, L.; Fridkin, M.; Bade Shrestha-Dawadi, P.; Bittner, S. Novel naphthoquinonyl derivatives: Potential structural components for the synthesis of cytotoxic peptides. Lett. Pept. Sci. 1996, 3, 263–274. [Google Scholar] [CrossRef]
- Rahimipour, S.; Weiner, L.; Bade Shrestha-Dawadi, P.; Bittner, S.; Koch, Y.; Fridkin, M. Cytotoxic peptides: Naphthoquinonyl derivatives of luteinizing hormone-releasing hormone (LH-RH). Lett. Pept. Sci. 1998, 5, 421–427. [Google Scholar] [CrossRef]
- De Moraes, T.A.P.; Filha, M.J.S.; Camara, C.A.; Silva, T.M.S.; Soares, B.M.; Bomfim, I.S.; Pessoa, C.; Ximenes, G.C.; Silva, V.A., Jr. Synthesis and cytotoxic evaluation of a series of 2-amino-naphthoquinones against human cancer cells. Molecules 2014, 19, 13188–13199. [Google Scholar] [CrossRef] [PubMed]
- Janáky, T.; Juhász, A.; Bajusz, S.; Csernus, V.; Srkalovic, G.; Bokser, L.; Milovanovic, S.; Redding, T.W.; Rékási, Z.; Nagy, A. Analogues of luteinizing hormone-releasing hormone containing cytotoxic groups. Proc. Natl. Acad. Sci. USA 1992, 89, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar] [PubMed]
- Watanabe, J.; Nishiyama, H.; Matsui, Y.; Ito, M.; Kawanishi, H.; Kamoto, T.; Ogawa, O. Dicoumarol potentiates cisplatin-induced apoptosis mediated by c-Jun N-terminal kinase in p53 wild-type urogenital cancer cell lines. Oncogene 2006, 25, 2500–2508. [Google Scholar] [CrossRef] [PubMed]
- Badisa, R.B.; Darling-Reed, S.F.; Joseph, P.; Cooperwood, J.S.; Latinwo, L.M.; Goodman, C.B. Selective cytotoxic activities of two novel synthetic drugs on human breast carcinoma MCF-7 Cells. Anticancer Res. 2009, 29, 2993–2996. [Google Scholar] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 2a, 6a, 10a and 13a are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).