Abstract
A one-pot preparation of pyridyl isothiocyanates (ITCs) from their corresponding amines has been developed. This method involves aqueous iron(III) chloride-mediated desulfurization of a dithiocarbamate salt that is generated in situ by treatment of an amine with carbon disulfide in the present of DABCO or sodium hydride. The choice of base is of decisive importance for the formation of the dithiocarbamate salts. This one-pot process works well for a wide range of pyridyl ITCs. Utilizing this protocol, some highly electron-deficient pyridyl and aryl ITCs are obtained in moderate to good yields.
1. Introduction
Isothiocyanates (ITCs) constitute an important class of natural products that are abundant in many cruciferous vegetables [1]. ITCs have versatile biological activities, ranging from anticancer and chemoprotective properties [2,3,4] to agrochemical activities [5,6,7], and they are also useful intermediates for the synthesis of various sulfur- and nitrogen-containing organic compounds [8], especially for heterocycles [9,10,11,12].
Numerous methods for preparing ITCs have been developed using different starting materials such as amines [13,14,15,16,17,18,19], tertiary alcohols [20], halides [21,22], nitrile oxides [23], azides [5], isocyanides [24,25]. Among these starting materials, amines are usually employed because of their broad availability and versatility. Most reported methods are highly effective for the synthesis of alkyl and electron-rich aryl ITCs, but their applicability to pyridyl-substituted ITCs is limited due to the lower nucleophilicity of pyridyl amines. In fact, the synthesis of ITCs from pyridyl amines proved to be more difficult than that from aryl amines.
There are two main methods to convert substituted aminopyridines into the corresponding ITC analogue (Scheme 1). The most well-known method is based on thiophosgene [9], and later refinements of ‘thiocarbonyl transfer’ reagents such as thiocarbonyl-diimidazole [26] and dipyridyl-thionocarbonate [27]. The high toxicity and incompatability of thiophosgene with many functional groups limit its general use, furthermore, these ‘thiocarbonyl transfer’ reagents are not readily available and often do not work as desired due to the formation of thiourea byproducts. Another two-step approach, based on reagent-promoted decomposition of dithiocarbamate salts into ITCs, was first reported by Le Count [28] in 1977. The intermediate dithiocarbamate salts are generated by treatment of amines with carbon disulfide and Et3N. Although some desulfurylating reagents for this approach were developed [17,28], the first step, preparing the N-pyridyldithiocarbamate salts, was often neglected. Most of these methods are efficient only for electron-rich pyridyl ITCs, because electron-deficient aminopyridines lack enough reactivity to form dithiocarbamate salts, which results in low yield or excess (hundredfold) use of carbon disulfide. Thus, so far few efficient and general methods have been reported for the preparation of pyridyl ITCs, especially for those with highly electron-withdrawing groups. Therefore, research into an improved method for pyridyl ITCs, which can be used for a broad range of substituents, remains a topic of considerable interest.
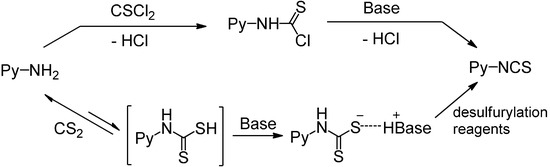
Scheme 1.
Methods for conversion of amines to pyridyl ITCs.
Scheme 1.
Methods for conversion of amines to pyridyl ITCs.
2. Results and Discussion
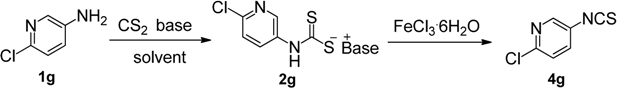
In Le Count’s work, iron(III) chloride has been proved to be effective for the decomposition of dithiocarbamate salts, but the preparation of N-pyridyldithiocarbamate salts was seldom investigated, so it became crucial for us to improve their preparation, because once the dithicarbamates were obtained, the desulfurylation step proceeded smoothly [13,16]. In the initial study, 3-amino-6-chloropyridine (1g) was chosen as a model substrate to prepare ITCs in a one-pot process (Table 1). At first, the effect of various bases was evaluated by performing the model reaction in tetrahydrofuran (entries 1–9). When inorganic bases (K2CO3, KOH) and organic bases like 1,8-bis(dimethylamino)naphthalene (Proton Sponge™) or pyridine were employed, the conversion of 1g was rather low, even after 12 h, giving less than 30% of 4g (entries 1–4). When triethylamine and potassium tert-butoxide was used, the conversion was significantly improved after 12 h (entries 5–6), however, a large amount of thiourea was formed in the case of t-BuOK. To our delight, when 1,8-diazabicyclo[5.4.0]-undec-7-ene (DBU), 4-dimethylaminopyridine (DMAP) or 1,4-diazabicyclo[2.2.2]octane (DABCO) were used as base, the conversion was complete within 4 h and 4g was obtained in excellent yield (entries 7–9). The results was summarized in Table 1 and could not be explained by the strength of the base (pKa), for example, the substrate 1g reacted with CS2 in the presence of DABCO (pKa 8.7) and Et3N (pKa 10.7), but it did not in the presence of pyridine (pKa 5.4) and Proton Sponge™ (pKa 12.1). The pKa values for protonated base are determined in polar solvents (water, MeCN, DMSO), in which they are dissociated as free ions [29]. However, THF is a nonpolar solvent and has a low dielectric constant, thus, the corresponding ammonium salts in nonpolar solvents are present entirely as ion pairs rather than free ions. To measure ion pairs basicity of some amines in THF, Streitwieser introduced the concept of pKip [30], which refers to the equilibrium between the base and the acid with the H-bonded ion pair, and found that the pKip values are inconsistent with their corresponding pKa values [31].

Table 1.
Optimization of reaction conditions for the synthesis of 2-chloro-5-isothiocyanatopyridine a.

| Entry | Solvent | Base | PKa b | PKip c | Conversion of 1g (%) | Overall Yield (%) |
|---|---|---|---|---|---|---|
| 1 | THF | K2CO3 | 10.3 | 31 | 11 | |
| 2 | THF | KOH | 15.7 | 46 | 25 | |
| 3 | THF | pyridine | 5.4 | 2.2 | 0 | trace |
| 4 | THF | Proton sponge | 12.1 | 0 | trace | |
| 5 | THF | Et3N | 10.7 | 2.1 | 85 | 77 |
| 6 | THF | t-BuOK | 29.0 | 78 | trace | |
| 7 | THF | DABCO | 8.7 | 0.8 | 100 | 96 |
| 8 | THF | DBU | 11.6 | −3.8 | 100 | 90 |
| 9 | THF | DMAP | 9.9 | 0.61 | 100 | 90 |
| 10 | DMF | DABCO | 95 | 87 | ||
| 11 | acetone | DABCO | 86 | 70 | ||
| 12 | MeCN | DABCO | 84 | 70 | ||
| 13 | EtOH | DABCO | 0 | trace | ||
| 14 | CH2Cl2 | DABCO | 60 | 48 |
Notes: a Reaction conditions: 1g (1 equiv), CS2 (3 equiv), base (2 equiv), solvent, r.t.; FeCl3·6H2O (2 equiv), r.t., 1 h; b The dissociation constant of the protonated base in water. Values were collected from refs [32,33]; c The equilibrium between the base and acidic indicator hydrocarbons InH with the H-bonded ion pairs. pKip = −logKip [30].
A possible mechanism for the formation of pyridyl dithiocarbamate salts is proposed in Scheme 1. The first step, the attack of amine on carbon disulfide to form dithiocarbamic acid, is likely reversible. The driving force of the reaction is most likely the reaction of the dithiocarbamic acid with base to generate the stable dithiocarbamate salts. A greater ion pair basicity corresponds to a tighter ion pair, which facilitates the generation of dithiocarbamates, the ion pair basicities of Et3N (pKip 2.1) and DABCO (pKip 0.8) agree with their observed different reactivity. When we used DABCO as the base, an examination of different solvents showed that THF was the best solvent compared with DMF, acetone, MeCN, EtOH, CH2Cl2 (entries 10–14). Finally, with the optimized conditions for the formation of 2g, we then found that upon addition of aqueous FeCl3 to unpurified 2g in one-pot, complete conversion to 4g was observed in about 1 h at room temperature.
Under the reaction conditions outlined above (Table 1, entry 7), the substrate scope of various aminopyridines was examined next (Table 2). The electronic effect of the substituents has a significant influence on the reaction outcome.

Table 2.
Preparation of aromatic ITCs a.

| Entry | Amines | Product | CS2 (equiv) | Time (h) b | Overall Yield (%) |
|---|---|---|---|---|---|
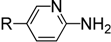 |  | ||||
| 1 | R = H | 4a | 3 | 4 | 87 |
| 2 | R = Me | 4b | 3 | 4 | 88 |
 | 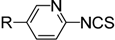 | ||||
| 3 | R = F | 4c | 3 | 12 | 76 |
| 4 | R = Cl | 4d | 10 | 12 | 81 |
| 5 | R = Br | 4e | 10 | 12 | 83 |
| 6 | R = CF3 | 4f | 20 | 24 | 42 |
| 7 | 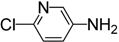 | 4g | 3 | 4 | 96 |
| 8 | 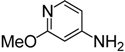 | 4h | 3 | 2 | 91 |
| 9 | 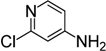 | 4i | 10 | 12 | 73 |
 |  | ||||
| 10 | R = CN | 4j | 4 | 12 | 87 |
| 11 | R = NO2 | 4k | 5 | 24 | 77 |
| 12 | R = CF3 | 4l | 4 | 12 | 85 |
| 13 | 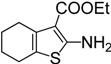 | 4m | 4 | 12 | 66 |
Notes: a Reaction conditions: 1 (8.0 mmol), CS2 (excess), DABCO (16.0 mmol), THF (10 mL), r.t.; FeCl3·6H2O (16.0 mmol), r.t., 1 h; b The reaction time for the first step.
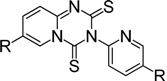
For example, aminopyridines containing electron donating groups (Me, OMe) afforded good yields of 87%–91% in a relatively short reaction time (entries 2 and 8). Incidently, the corresponding ITCs from 2-aminopyridine and 2-amino-5-methylpyridine have been obtained as dimers, and such dimers slowly dissociate to monomers in hot organic solvent [34,35]. When the 2- or 4-aminopyridines contained halides (entries 3–5, 9), longer reaction times and more equivalents of CS2 were required to access 2, but the corresponding ITCs were still obtained in moderate to good yields, ranging from 73% to 83%. Meanwhile, the position of the amino group on the pyridine also exerted an influence on the reaction outcome; for example, the overall yield of C6H3ClN2S varies for 2-(3-or 4-)aminopyridines (entries 4, 7, 9), and a greater yield was obtained when the amino group is at the meta position with respect to the nitrogen atom in the pyridine (96%, entry 7). To our delight, several anilines with strong electron-withdrawing groups, such as NO2, CN, and CF3 (entries 10–12), were also smoothly converted into the desired ITCs in 77%–87% yields. The approach also worked well for the five-membered heterocyclic substrate (entry 13). However, the desired ITCs could not be detected when highly electron-deficient aminopyridines (such as those with NO2, CN, CO2Me substituents) were used. Only 5-trifluoromethylpyridyl-2-amine afforded the corresponding ITC in a low yield (42%, entry 6), even after prolonged reaction time and with excess CS2. For halide substituents in the ortho position of the amino group, no corresponding ITCs were observed. Thus, additional investigations are necessary to develop methods for the preparation of some highly electron-deficient pyridyl ITCs.

Table 3.
Preparation of highly electron-deficient pyridyl ITCs a.

| Entry | Amine | Product | Overall Yield (%) |
|---|---|---|---|
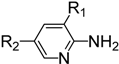 | 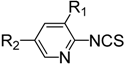 | ||
| 1 | R1 = H, R2 = CN | 4n | 51 |
| 2 | R1 = H, R2 = NO2 | 4o | 31 |
| 3 | R1 = H, R2 = CO2Me | 4p | 63 |
| 4 | R1 = Cl, R2 = Cl | 4q | 77 |
| 5 | R1 = Cl, R2 = H | 4r | 84 |
| 6 | R1 = F, R2 = H | 4s | 72 |
| 7 | 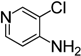 | 4t | 49 |
Notes: a Reaction conditions: 1 (8.0 mmol), CS2 (32.0 mmol), NaH (9.6 mmol), DMF (8 mL), r.t., 6 h; Et3N (8.0 mmol), FeCl3·6H2O (16.0 mmol), r.t., 1 h.
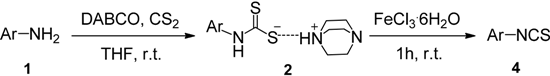
The observed deficiencies in the synthesis of highly electron-deficient pyridyl ITCs inspired us to further optimize the process. The difficulty in the generation of dithiocarbamates is likely due to the weaker nucleophilicity of these amine substrates. In an effort to improve the reactivity, higher reaction temperatures in a variety of solvents were tested. Using methyl 6-aminonicotinate as a test substrate, we found that after 20 h of reflux in THF or DMF, only trace amounts of the corresponding ITCs were observed. We therefore investigated next the use of the strong base NaH to generate the more nucleophilic amide anions prior to CS2 addition. After testing various solvents, the use of NaH in DMF was found to be the best choice. The amines was treated with NaH in DMF at 0 °C, then CS2 was added, and after 6 h at room temperature, when the amines were fully consumed as monitored by TLC, the reaction mixtures were slowly treated with aqueous FeCl3. Using this process, we were able to obtain reasonable yields of several pyridyl ITCs with strong electron-withdrawing groups, such as NO2, CN, CO2Me, and 3,5-Cl2 (Table 3, entries 1–4, 31%–77% yield). This method was also effective for substrates bearing halide substituents in the ortho position of the amino moiety (entries 5–7, 49%–84% yield).
3. Experimental Section
3.1. General Information
Tetrahydrofuran was redistilled in the presence of sodium/benzophenone. Unless otherwise stated, all reagents were commercially available and were used without purification. TLC was performed on pre-coated silica gel glass plates. Flash column chromatography was performed using flash silica gel (200–300 mesh) (Qingdao Haiyang, Qingdao, China). HPLC analyses were performed on an Agilent 1200 Series instrument (Santa Clara, CA, USA, column: Agilent Eclipse XDB-C18, 5 μm, 4.6 × 150 mm). Melting points were determined using a Stuart melting point apparatus and were uncorrected. 1H- and 13C-NMR spectra were recorded with a 300 MHz spectrometer (Bruker, Fallanden, Switzerland). HRMS and GC-MS were recorded on an Agilent mass spectrometer by the ESI and EI techniques, respectively. All yields given refer to isolated yields.
3.2. General Procedure for the Preparation of Isothiocyanates 4a–m
To a solution of amine 1 (8.0 mmol) and DABCO (16 mmol) in anhydrous THF (10 mL) was added dropwise a certain amount of CS2. The resulting mixture was stirred at r.t. for several hours until completion by TLC analysis. Then a solution of FeCl3·6H2O (16 mmol) in water (15 mL) was added rapidly to the well suspended dithiocarbamate 2, and stirring was continued for 1 h. The aqueous layer was separated and extracted with EtOAc (2 × 10 mL). The combined organic phase was washed with water (2 × 10 mL), and dried over MgSO4. After removal of the solvent, the product was purified by flash column chromatography (petroleum ether–EtOAc) to give the corresponding ITCs 4.
3.3. General Procedure for the Preparation of Isothiocyanates 4n–t
To an ice-cold stirred solution of amine 1 (8.0 mmol) in DMF (8 mL) was added NaH (60% in mineral oil; 9.6 mmol) in two portions. After the evolution of gas from the reaction mixture ceased, CS2 (32 mmol) was added via syringe pump over about 30 min. The resulting mixture was brought up to r.t. and kept for 6 h, then the mixture was cooled on an ice bath. Et3N (8.0 mmol) and a solution of FeCl3·6H2O (16 mmol) in water (15 mL) were successively added to the dithiocarbamate 3. After the additions, the mixture was stirred at r.t. for 1 h. The subsequent operations were the same as the workup in the experimental procedure described above.
3.4. Characterization Data
3-(Pyridin-2-yl)-2H-pyrido[1,2-a][1,3,5]triazine-2,4(3H)-dithione (4a) [34]. The crude product purified by column chromatography (petroleum ether/CHCl3 = 5:1~1:1, v/v), affording the dimer of 2-pyridyl isothiocyanate as a brick-red solid; yield: 0.95 g (3.48 mmol, 87%); m.p. 110.2–111.1 °C (lit. [28] 112 °C); 1H-NMR (CDCl3) δ 9.28–9.25 (m, 1H), 8.68–8.66 (m, 1H), 7.95–7.77 (m, 2H), 7.44–7.30 (m, 3H), 7.01–6.96 (m, 1H); 13C-NMR (CDCl3) δ 179.17 (C=S), 172.26 (C=S), 155.36, 150.34, 146.68, 142.16, 139.03, 132.75, 125.03, 124.14, 123.48, 115.62; HRMS (ESI): m/z [M+H]+ calcd for C12H9N4S2: 273.0269; found: 273.0272.
7-Methyl-3-(5-methylpyridin-2-yl)-2H-pyrido[1,2-a][1,3,5]triazine-2,4(3H)-dithione (4b) [28]. Brick-red solid, purified by column chromatography (petroleum ether/CHCl3 = 5:1~1:1, v/v); yield: 1.06 g (3.52 mmol, 88%); m.p. 137.0–137.4 °C; 1H-NMR (CDCl3) δ 9.10–9.09 (m, 1H), 8.50 (d, J = 2.3 Hz, 1H), 7.78–7.61 (m, 2H), 7.29–7.22 (m, 2H), 2.44 (s, 3H), 2.35 (s, 3H); 13C-NMR (CDCl3) δ 179.23 (C=S), 172.37 (C=S), 153.31, 150.60, 145.80, 145.07, 139.66, 134.18, 129.95, 126.01, 124.64, 122.71, 18.29, 18.17; HRMS (ESI): m/z [M+H]+ calcd for C14H13N4S2: 301.0582; found: 301.0585.
5-Fluoro-2-isothiocyanatopyridine (4c). Red solid purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v); yield: 0.94 g (6.08 mmol, 76%); m.p. 21.2–22.4 °C; 1H-NMR (CDCl3) δ 8.28 (d, J = 3.0 Hz, 1H), 7.46 (ddd, J = 8.7, 7.3, 3.0 Hz, 1H), 7.12 (dd, J = 8.7, 3.9 Hz, 1H); 13C-NMR (DMSO-d6) δ 158.03 (d, 1JC-F = 253.3Hz), 141.14 (d, 4JC-F = 2.7Hz), 139.59 (NCS), 137.98 (d, 2JC-F = 26.3 Hz), 126.71 (d, 2JC-F = 20.6 Hz), 121.57 (d, 3JC-F = 5.8 Hz); GC-MS (EI): m/z = 154 [M+].
5-Chloro-2-isothiocyanatopyridine (4d). White solid purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v); yield: 1.10 g (6.48 mmol, 81%); m.p. 43.5–44.5 °C (lit. [28] 41–43 °C); 1H-NMR (CDCl3) δ 8.38 (dd, J = 2.6, 0.5 Hz, 1H), 7.68 (dd, J = 8.5, 2.6 Hz, 1H), 7.05 (dd, J = 8.5, 0.5 Hz, 1H); 13C-NMR (CDCl3) δ 148.77, 144.61, 142.93 (NCS), 138.32, 130.22, 120.21; HRMS (ESI): m/z [M+H]+ calcd for C6H4ClN2S: 170.9784; found: 170.9766.
5-Bromo-2-isothiocyanatopyridine (4e). White solid purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v); yield: 1.42 g (6.64 mmol, 83%); m.p. 73.3–74.2 °C (lit. [28] 74–76 °C); 1H-NMR (CDCl3) δ 8.48 (d, J = 2.5 Hz, 1H), 7.82 (dd, J = 8.4, 2.5 Hz, 1H), 7.00 (d, J = 8.4 Hz, 1H); 13C-NMR (CDCl3) δ 151.02, 145.09, 143.00 (NCS), 141.15, 120.69, 118.46; HRMS (ESI): m/z [M + H]+ calcd for C6H4BrN2S: 214.9279; found: 214.9268.
2-Isothiocyanato-5-(trifluoromethyl)pyridine (4f) [36]. Red oil purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v) ; yield: 0.69 g (3.36 mmol, 42%); 1H-NMR (CDCl3) δ 8.73–8.66 (m, 1H), 8.02–7.92 (m, 1H), 7.23–7.20 (m, 1H); 13C-NMR (CDCl3) δ 149.69, 147.19 (q, 3JC-F = 4.1 Hz), 144.58 (NCS), 135.95 (q, 3JC-F = 3.4 Hz), 124.89 (q, 2JC-F = 33.7 Hz), 122.98 (q, 1JC-F = 270.7 Hz), 119.07; HRMS (ESI): m/z [M+H]+ calcd for C7H4F3N2S: 205.0047; found: 205.0047.
2-Chloro-5-isothiocyanatopyridine (4g) [37]. White solid purified by column chromatography (petroleum ether/EtOAc = 20:1, v/v); yield: 1.31 g (7.68 mmol, 96%); m.p. 56.0–57.9 °C; 1H-NMR (CDCl3) δ 8.31 (dd, J = 2.7, 0.7 Hz, 1H), 7.51 (dd, J = 8.5, 2.7 Hz, 1H), 7.35 (dd, J = 8.5, 0.7 Hz, 1H); 13C-NMR (CDCl3) δ 148.53, 146.29, 140.14 (NCS), 134.76, 128.41, 124.66; HRMS (ESI): m/z [M+H]+ calcd for C6H4ClN2S: 170.9784; found: 170.9784.
4-Isothiocyanato-2-methoxypyridine (4h). White solid purified by column chromatography (petroleum ether/EtOAc = 20:1, v/v); yield: 1.21 g (7.28 mmol, 91%); m.p. 32.4–33.5 °C; 1H-NMR (CDCl3) δ 8.12 (d, J = 5.5 Hz, 1H), 6.71 (dd, J = 5.5, 1.8 Hz, 1H), 6.52 (d, J = 1.8 Hz, 1H), 3.93 (s, 3H); 13C-NMR (CDCl3) δ 165.28, 148.18, 141.40, 139.83 (NCS), 113.79, 106.78, 53.75; HRMS (ESI): m/z [M+H]+ calcd for C7H7N2OS: 167.0279; found: 167.0273.
2-Chloro-4-isothiocyanatopyridine (4i) [17]. White solid purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v); yield: 0.99 g (5.84 mmol, 73%); m.p. 44.0–44.9 °C, 1H-NMR (CDCl3) δ 8.37 (dd, J = 5.4, 0.6 Hz, 1H), 7.16 (dd, J = 1.8, 0.6 Hz, 1H), 7.04 (dd, J = 5.4, 1.8 Hz, 1H); 13C-NMR (DMSO-d6) δ 151.47, 151.27, 141.03, 139.32 (NCS), 120.80, 120.11; HRMS (ESI): m/z [M+H]+ calcd for C6H4ClN2S: 170.9784; found: 170.9806.
4-Isothiocyanatobenzonitrile (4j). White solid purified by column chromatography (petroleum ether/EtOAc = 20:1, v/v); yield: 1.11 g (6.96 mmol, 87%); m.p. 121.4–122.5 °C (lit. [16] 121–122 °C); 1H-NMR (CDCl3) δ7.67 (d, J = 8.7 Hz, 2H), 7.30 (d, J = 8.7 Hz, 1H); 13C-NMR (CDCl3) δ 139.73 (NCS), 136.05, 133.55, 126.39, 117.79, 110.62; HRMS (ESI): m/z [M+H]+ calcd for C8H5N2S: 161.0173; found: 161.0158.
1-Isothiocyanato-4-nitrobenzene (4k). White solid purified by column chromatography (petroleum ether/EtOAc = 20:1, v/v); yield: 1.11 g (6.16 mmol, 77%); m.p. 109.4–110.2 °C (lit. [18] 108–109 °C); 1H-NMR (CDCl3) δ 8.25 (d, J = 9.0 Hz, 2H), 7.36 (d, J = 9.0 Hz, 2H); 13C-NMR (CDCl3) δ 145.80, 140.31 (NCS), 137.90, 126.32, 125.23; HRMS (ESI): m/z [M+H]+ calcd for C7H5N2O2S: 181.0072; found: 181.0054.
1-Isothiocyanato-4-(trifluoromethyl)benzene (4l). White solid purified by column chromatography (petroleum ether/EtOAc = 20:1, v/v); yield: 1.38 g (6.80 mmol, 85%); m.p. 40.1–41.2 °C (lit. [16] 40–41 °C); 1H-NMR (CDCl3) δ 7.61 (d, J = 8.3 Hz, 2H), 7.30 (d, J = 8.3 Hz, 2H); 13C-NMR (CDCl3) δ 138.47 (NCS), 135.00, 129.05 (q, 2JC-F = 32.9 Hz), 126.76 (q, 3JC-F = 3.7 Hz), 125.92, 123.55 (q, 1JC-F = 270.7 Hz); GC-MS (EI): m/z = 203 [M+].
Ethyl 2-isothiocyanato-4,5,6,7-tetrahydrobenzo[b]-thiophene-3-carboxylate (4m). Yellow solid purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 1.41 g (5.28 mmol, 66%); m.p. 45.3–45.7 °C (lit. [17] 45–46 °C); 1H-NMR (CDCl3) δ 4.34 (q, J = 7.1 Hz, 2H), 2.77 (t, J = 5.7 Hz, 2H), 2.64 (t, J = 5.7 Hz, 2H), 1.83–1.76 (m, 4H), 1.40 (t, J = 7.1 Hz, 3H); 13C-NMR (CDCl3) δ 161.89, 137.33 (NCS), 134.72, 132.55, 131.96, 126.49, 60.66, 26.07, 24.88, 22.62, 22.21, 14.35; GC-MS (EI): m/z = 267 [M+].
6-Isothiocyanatonicotinonitrile (4n). Yellow solid purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 0.66 g (4.08 mmol, 51%); m.p. 68.3–69.5 °C; 1H-NMR (CDCl3) δ 8.72 (dd, J = 2.3, 0.8 Hz, 1H), 7.99 (dd, J = 8.3, 2.3 Hz, 1H), 7.18 (dd, J = 8.3, 0.8 Hz, 1H); 13C-NMR (CDCl3) δ 153.14, 149.86, 145.93 (NCS), 141.78, 119.30, 115.85, 107.82; HRMS (ESI): m/z [M+H]+ calcd for C7H4N3S: 162.0126; found: 162.0109.
2-Isothiocyanato-5-nitropyridine (4o). Yellow solid purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 0.45 g (2.48 mmol, 31%); m.p. 50.3–51.0 °C; 1H-NMR (CDCl3) δ 9.27 (d, J = 2.8 Hz, 1H), 8.51 (dd, J = 8.7, 2.8 Hz, 1H), 7.22 (d, J = 8.7 Hz, 1H); 13C-NMR (CDCl3) δ 151.57, 146.46, 146.06, 142.11 (NCS), 134.07, 119.08; HRMS (ESI): m/z [M+H]+ calcd for C6H4N3O2S: 182.0024; found: 182.0019.
Methyl 6-isothiocyanatonicotinate (4p). White solid purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 0.98 g (5.04 mmol, 63%); m.p. 87.2–88.2 °C; 1H-NMR (CDCl3) δ 9.03 (dd, J = 2.3, 0.8 Hz, 1H), 8.31 (dd, J = 8.3, 2.3 Hz, 1H), 7.15 (dd, J = 8.3, 0.8 Hz, 1H), 3.96 (s, 3H); 13C-NMR (CDCl3) δ 164.62, 151.51, 149.80, 143.86 (NCS), 139.76, 124.34, 118.96, 52.46; HRMS (ESI): m/z [M+H]+ calcd for C8H7N2O2S: 195.0228; found: 195.0263.
3,5-Dichloro-2-isothiocyanatopyridine (4q). White solid purified by column chromatography (petroleum ether/EtOAc = 15:1, v/v); yield: 1.26 g (6.16 mmol, 77%); m.p. 51.5–52.6 °C; 1H-NMR (CDCl3) δ 8.27 (d, J = 2.3 Hz, 1H), 7.77 (d, J = 2.3 Hz, 1H); 13C-NMR (CDCl3) δ 146.37, 144.32 (NCS), 142.07, 137.92, 129.92, 127.89; HRMS (ESI): m/z [M+H]+ calcd for C6H3Cl2N2S: 204.9394; found: 204.9374.
3-Chloro-2-isothiocyanatopyridine (4r). White oil purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 1.14 g (6.72 mmol, 84%); 1H-NMR (CDCl3) δ 8.32 (dd, J = 4.7, 1.6 Hz, 1H), 7.78 (dd, J = 8.0, 1.6 Hz, 1H), 7.21 (dd, J = 8.0, 4.7 Hz, 1H); 13C-NMR (CDCl3) δ 147.36, 143.36, 142.91 (NCS), 138.37, 127.65, 122.82; HRMS (ESI): m/z [M+H]+ calcd for C6H4ClN2S: 170.9784; found: 170.9775.
3-Fluoro-2-isothiocyanatopyridine (4s). Red oil purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 0.89 g (5.76 mmol, 72%); 1H-NMR (CDCl3) δ 8.11–8.09 (m, 1H), 7.46–7.40 (m, 1H), 7.20–7.15 (m, 1H); 13C-NMR (CDCl3) δ 153.85 (d, 1JC-F = 262.7 Hz), 144.90 (NCS), 144.40 (d, 3JC-F = 5.8 Hz), 134.95 (d, 2JC-F = 13.3 Hz), 124.13 (d, 2JC-F = 16.8 Hz), 123.19 (d, 4JC-F = 3.0 Hz); HRMS (ESI): m/z [M+H]+ calcd for C6H4FN2S: 155.0079; found: 155.0051.
3-Chloro-4-isothiocyanatopyridine (4t). White solid purified by column chromatography (petroleum ether/EtOAc = 10:1, v/v); yield: 0.67 g (3.92 mmol, 49%); m.p. 30.4–31.5 °C; 1H-NMR (CDCl3) δ 8.62 (d, J = 0.5 Hz, 1H), 8.45 (d, J = 5.2 Hz, 1H), 7.11 (dd, J = 5.2, 0.5 Hz, 1H); 13C-NMR (CDCl3) δ 150.36, 148.81, 143.11 (NCS), 137.78, 128.99, 120.14; HRMS (ESI): m/z [M+H]+ calcd for C6H4ClN2S: 170.9784; found: 170.9771.
4. Conclusions
In summary, we have developed a facile and environmentally friendly method for the preparation of various pyridyl ITCs from amines via a one-pot process. In comparison to existing methods, our procedure for the synthesis of highly electron-deficient pyridyl ITCs without using dangerous thiophosgene is simple yet efficient. The employed reagents are inexpensive and of low toxicity and the procedure is operationally simple, affording a wide range of pyridyl ITCs in moderate to excellent yields. Based on these characteristics, we envision that this method will be useful to the synthetic community.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/19/9/13631/s1.
Supplementary Files
Supplementary File 1Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 21172256), the National Basic Research Program of China (No. 2010CB126104), the National S&T Pillar Program of China (No. 2012BAK25B03) and China Agriculture University Scientific Fund (No. 2013YJ010).
Author Contributions
Hao Zhang and Shang-Zhong Liu conceived of this study and carried out most of compounds synthesis as well as manuscript preparation. Rui-Quan Liu, Ke-Chang Liu participated in compounds synthesis. Qi-Bo Li and Qing-Yang Li assisted in characterization experiments. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pedras, M.S.C.; Zheng, Q.-A.; Gadagi, R.S. The first naturally occurring aromatic isothiocyanates, rapalexins a and b, are cruciferous phytoalexins. Chem. Commun. 2007, 368–370. [Google Scholar] [CrossRef]
- Wu, X.; Zhou, Q.-H.; Xu, K. Are isothiocyanates potential anti-cancer drugs? Acta Pharmacol. Sin. 2009, 30, 501–512. [Google Scholar] [CrossRef]
- Adsule, S.; Banerjee, S.; Ahmed, F.; Padhye, S.; Sarkar, F.H. Hybrid anticancer agents: Isothiocyanate-progesterone conjugates as chemotherapeutic agents and insights into their cytotoxicities. Bioorg. Med. Chem. Lett. 2010, 20, 1247–1251. [Google Scholar] [CrossRef]
- Galuppo, M.; Nicola, G.; Iori, R.; Dell'Utri, P.; Bramanti, P.; Mazzon, E. Antibacterial activity of glucomoringin bioactivated with myrosinase against two important pathogens affecting the health of long-term patients in hospitals. Molecules 2013, 18, 14340–14348. [Google Scholar] [CrossRef]
- Wu, H.; Feng, J.-T.; Lin, K.-C.; Zhang, X. Synthesis and herbicidal activity of substituted pyrazole isothiocyanates. Molecules 2012, 17, 12187–12196. [Google Scholar] [CrossRef]
- Azaiez, I.; Meca, G.; Manyes, L.; Fernández-Franzón, M. Antifungal activity of gaseous allyl, benzyl and phenyl isothiocyanate in vitro and their use for fumonisins reduction in bread. Food Control 2013, 32, 428–434. [Google Scholar] [CrossRef]
- Santos, J.C.; Faroni, L.R.A.; Sousa, A.H.; Guedes, R.N.C. Fumigant toxicity of allyl isothiocyanate to populations of the red flour beetle tribolium castaneum. J. Stored Prod. Res. 2011, 47, 238–243. [Google Scholar] [CrossRef]
- Sun, Z.-H.; Huang, W.; Gong, Y.-Y.; Lan, J.; Liu, X.-H.; Weng, J.-Q.; Li, Y.-S.; Tan, C.-X. Synthesis and herbicidal activity of new 1,3,4-thiadizols sulfourea derivative. Chin. J. Org. Chem. 2013, 33, 2612–2617. [Google Scholar] [CrossRef]
- Mukerjee, A.K.; Ashare, R. Isothiocyanates in the chemistry of heterocycles. Chem. Rev. 1991, 91, 1–24. [Google Scholar] [CrossRef]
- Hemdan, M.M.; Fahmy, A.F.; Ali, N.F.; Hegazi, E.; Abd-Elhaleem, A. Synthesis of some new heterocycles derived from phenylacetyl isothiocyanate. Chin. J. Chem. 2008, 26, 388–391. [Google Scholar] [CrossRef]
- Wu, X.-L.; Zhu, C.-F.; Lv, Z.-D.; Wei, C.-S.; Liao, X.-C. Synthesis of sulfur ethers containing 1,3,4-oxadiazole and 1,3,4-thiadiazole. Chin. J. Org. Chem. 2011, 31, 824–831. [Google Scholar]
- Karpyak, V.; Obushak, M.; Ganushchak, M. Synthesis of 2-(2-R1-hydrazino)-5-(R2-benzyl)-2-thiazolines on the basis of meerweins arylation products of allyl isothiocyanate. Molecules 2003, 8, 263–268. [Google Scholar] [CrossRef]
- Nath, J.; Ghosh, H.; Yella, R.; Patel, B.K. Molecular iodine mediated preparation of isothiocyanates from dithiocarbamic acid salts. Eur. J. Org. Chem. 2009, 2009, 1849–1851. [Google Scholar]
- Wong, R.; Dolman, S.J. Isothiocyanates from tosyl chloride mediated decomposition of in situ generated dithiocarbamic acid salts. J. Org. Chem. 2007, 72, 3969–3971. [Google Scholar] [CrossRef]
- Munch, H.; Hansen, J.S.; Pittelkow, M.; Christensen, J.B.; Boas, U. A new efficient synthesis of isothiocyanates from amines using di-tert-butyl dicarbonate. Tetrahedron Lett. 2008, 49, 3117–3119. [Google Scholar] [CrossRef]
- Sun, N.; Li, B.; Shao, J.-P.; Mo, W.-M.; Hu, B.-X.; Shen, Z.-L.; Hu, X.-Q. A general and facile one-pot process of isothiocyanates from amines under aqueous conditions. Beilstein J. Org. Chem. 2012, 8, 61–70. [Google Scholar] [CrossRef]
- Liu, P.-F.; Li, C.-Y.; Zhang, J.-W.; Xu, X.-Y. Facile and versatile synthesis of alkyl and aryl isothiocyanates by using triphosgene and cosolvent. Synth. Commun. 2013, 43, 3342–3351. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Ma, H.-Z.; Han, C.; Xi, H.-T.; Meng, Q.; Chen, X.; Sun, X.-Q. Synthesis of isothiocyanates by reaction of amines with phenyl chlorothionoformate via one-pot or two-step process. Synthesis 2013, 45, 1667–1674. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, H.-H.; Tan, Y.-J. A simple and straightforward synthesis of phenyl isothiocyanates, symmetrical and unsymmetrical thioureas under ball milling. RSC Adv. 2013, 3, 16940–16944. [Google Scholar] [CrossRef]
- Pronin, S.V.; Reiher, C.A.; Shenvi, R.A. Stereoinversion of tertiary alcohols to tertiary-alkyl isonitriles and amines. Nature 2013, 501, 195–199. [Google Scholar] [CrossRef]
- Zhong, B.; Al-Awar, R.S.; Shih, C.; Grimes, J.H.; Vieth, M.; Hamdouchi, C. Novel route to the synthesis of 4-quinolyl isothiocyanates. Tetrahedron Lett. 2006, 47, 2161–2164. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Yusof, M.S.M.; Amin, N.M. Anti-amoebic properties of carbonyl thiourea derivatives. Molecules 2014, 19, 5191–5204. [Google Scholar] [CrossRef]
- Burkett, B.A.; Fu, P.; Hewitt, R.J.; Ng, S.L.; Toh, J.D.W. Purification-free, small-scale synthesis of isothiocyanates by reagentless fragmentation of polymer-supported 1,4,2-oxathiazoles. Eur. J. Org. Chem. 2014, 2014, 1053–1058. [Google Scholar]
- Kobayashi, K.; Kobayashi, A.; Ezaki, K. One-pot synthesis of 2-sulfanyl-3-sulfinyl(or sulfonyl)-1H-indoles via cyclization of 1-isothiocyanato-2-[sulfinyl(or sulfonyl)methyl]benzenes with sodium hydride. Tetrahedron 2013, 69, 7936–7942. [Google Scholar] [CrossRef]
- Fujiwara, S.; Shin-Ike, T.; Sonoda, N.; Aoki, M.; Okada, K.; Miyoshi, N.; Kambe, N. Novel selenium catalyzed synthesis of isothiocyanates from isocyanides and elemental sulfur. Tetrahedron Lett. 1991, 32, 3503–3506. [Google Scholar] [CrossRef]
- Larsen, C.; Steliou, K.; Harpp, D.N. Organic sulfur chemistry. 25. Thiocarbonyl transfer reagents. J. Org. Chem. 1978, 43, 337–339. [Google Scholar] [CrossRef]
- Kim, S.; Yi, K.Y. Di-2-pyridyl thionocarbonate. A new reagent for the preparation of isothiocyanates and carbodiimides. Tetrahedron Lett. 1985, 26, 1661–1664. [Google Scholar] [CrossRef]
- Le Count, D.J.; Dewsbury, D.J.; Grundy, W. An improved synthesis of pyridyl isothiocyanates and thioureas. Synthesis 1977, 582–583. [Google Scholar] [CrossRef]
- Ding, F.-Z.; Smith, J.M.; Wang, H.-B. First-principles calculation of pKa values for organic acids in nonaqueous solution. J. Org. Chem. 2009, 74, 2679–2691. [Google Scholar] [CrossRef]
- Streitwieser, A.; Kim, Y.-J. Ion pair basicity of some amines in THF: Implications for ion pair acidity scales. J. Am. Chem. Soc. 2000, 122, 11783–11786. [Google Scholar] [CrossRef]
- Cecchi, L.; de Sarlo, F.; Machetti, F. 1,4-diazabicyclo [2.2.2]octane (DABCO) as an efficient reagent for the synthesis of isoxazole derivatives from primary nitro compounds and dipolarophiles: The role of the base. Eur. J. Org. Chem. 2006, 2006, 4852–4860. [Google Scholar] [CrossRef]
- Perrin, D.D. Dissociation Constants of Organic Bases in Aqueous Solution; Butterworths: London, UK, 1965. [Google Scholar]
- Haynes, W.M. Crc Handbook of Chemistry and Physics, 95th ed; Lide, D.R., Ed.; CRC Press/Taylor and Francis: Boca Raton, FL, USA, 2014. [Google Scholar]
- Nair, V.; Kim, K.H. The reaction of 1-azirines with 2-pyridyl isothiocyanate: Possible approaches to benzodiazepine and benzodiazepine derivatives. J. Heterocycl. Chem. 1976, 13, 873–876. [Google Scholar] [CrossRef]
- L'abbé, G.; Allewaert, K.; Toppet, S. A rare case of three rearrangements during the cycloaddition-elimination reaction of 4-methyl-5-phenylimino-Δ2-1,2,3,4-thiatriazoline with 2-pyridyl isothiocyanate. J. Heterocycl. Chem. 1988, 25, 1459–1462. [Google Scholar] [CrossRef]
- Ramachandran, S.; Shahul, H.P.; Srivastava, A.; Shanbhag, G.; Morayya, S.; Rautela, N.; Awasthy, D.; Kavanagh, S.; Bharath, S.; Reddy, J.; et al. N-Aryl-2-aminobenzimidazoles: Novel, efficacious, antimalarial lead compounds. J. Med. Chem. 2014, 57, 6642–6652. [Google Scholar] [CrossRef]
- Altenbach, R.J.; Bai, H.; Brioni, J.D.; Carroll, W.A.; Gopalakrishnan, M.; Gregg, R.J.; Holladay, M.W.; Huang, P.P.; Kincaid, J.F.; Kort, M.E.; et al. Potassium Channel Openers. U.S. Patent 2002/0028836A1; filed 05 February 2001, and issued 07 March 2002,
- Sample Availability: Samples of the compounds 4a–t are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
