Simultaneous Determination of Fifteen Constituents of Jitai Tablet Using Ultra High-Performance Liquid Chromatography Coupled with Triple Quadrupole Electrospray Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Sample Preparation
2.2. Optimization of UHPLC-ESI-MS/MS Conditions
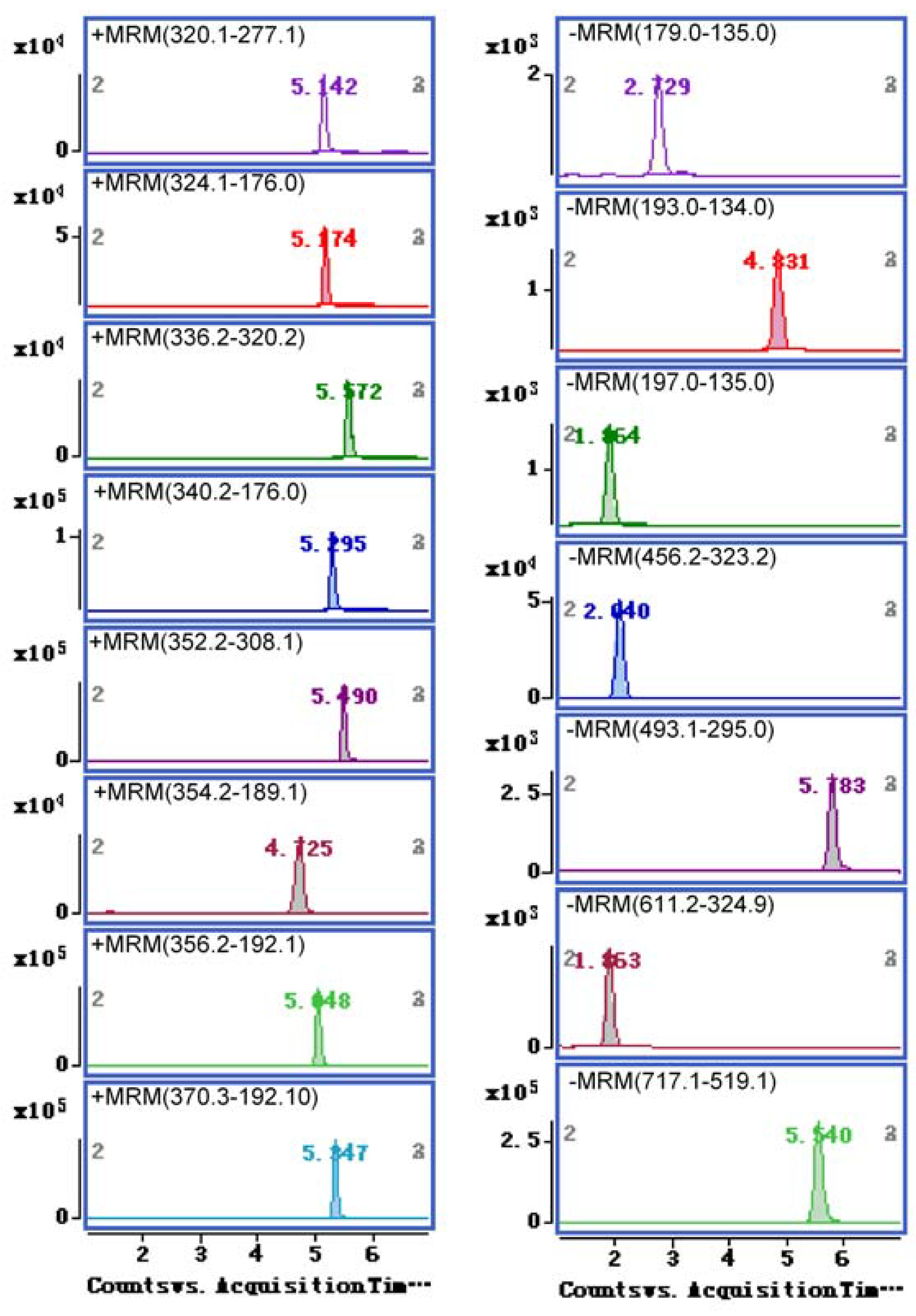
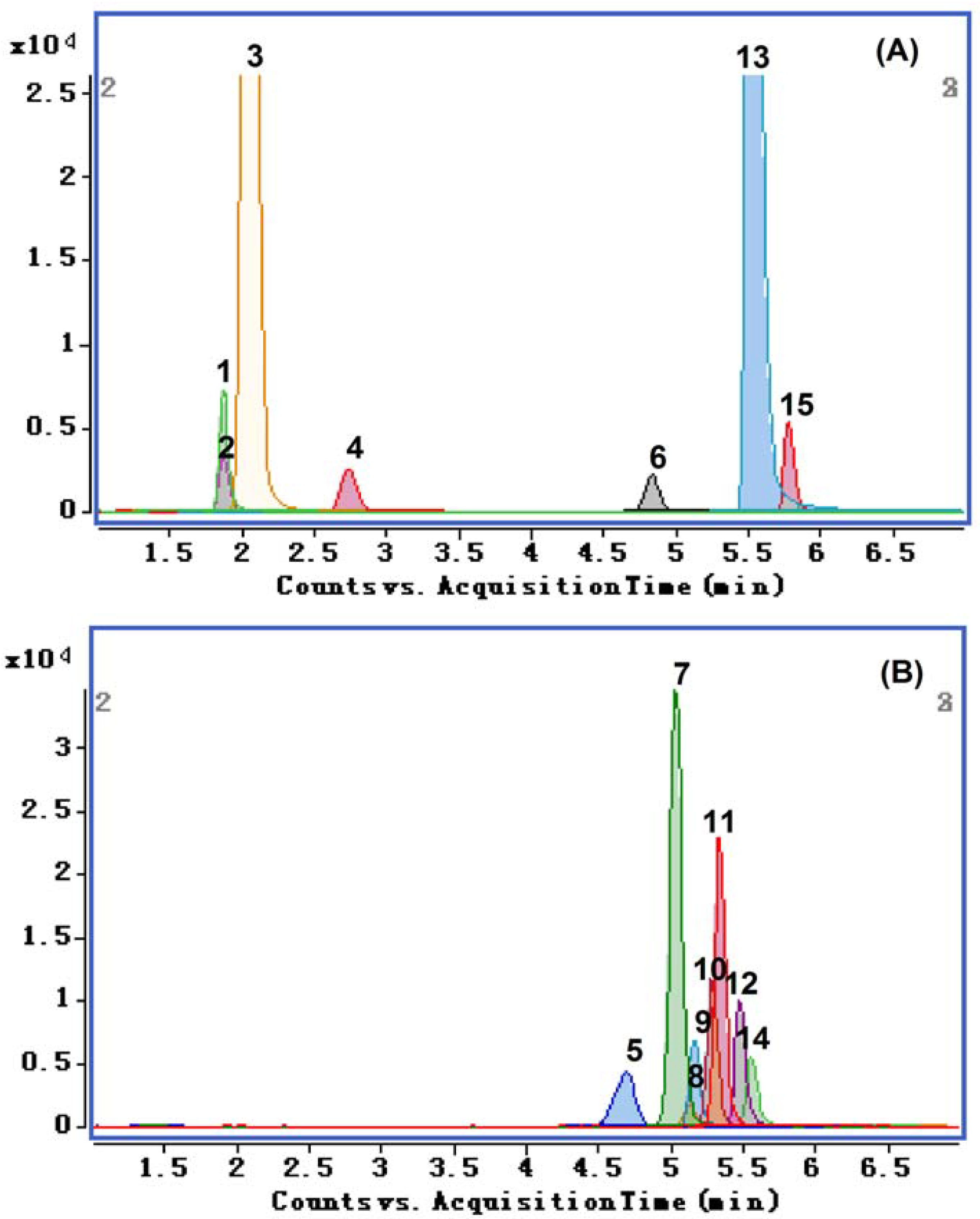
2.3. Method Validation
2.3.1. Linearity, Limit of Detection (LOD) and Limit of Quantification (LOQ)
2.3.2. Precision, Repeatability and Stability
2.3.3. Recovery
| Analyte | Molecular Weight (Da) | Ionization Mode | Retention Time (min) | Q1 Mass (m/z) | Q3 Mass (m/z) | Dwell Time (ms) | Fragmentor (V) | Collision Energy (V) |
|---|---|---|---|---|---|---|---|---|
| Hydroxysafflor yellow A | 612.2 | ESI− | 1.82 | 611.2 | 324.9 | 100 | 190 | 30 |
| Danshensu | 198.1 | ESI− | 1.86 | 197.0 | 135.0 | 100 | 90 | 4 |
| Amygdalin | 457.2 | ESI− | 2.04 | 456.2 | 323.2 | 100 | 160 | 6 |
| Caffeic acid | 180.0 | ESI− | 2.73 | 179.0 | 135.0 | 100 | 90 | 8 |
| Protopine | 353.1 | ESI+ | 4.73 | 354.2 | 189.1 | 100 | 130 | 30 |
| Ferulic acid | 194.1 | ESI− | 4.83 | 193.0 | 134.0 | 100 | 80 | 10 |
| Tetrahydropalmatine | 355.2 | ESI+ | 5.05 | 356.2 | 192.1 | 100 | 140 | 28 |
| Coptisine | 320.1 | ESI+ | 5.14 | 320.1 | 277.1 | 100 | 140 | 38 |
| Tetrahydrocoptisine | 323.1 | ESI+ | 5.17 | 324.1 | 176.0 | 100 | 150 | 30 |
| Tetrahydroberineper | 339.2 | ESI+ | 5.30 | 340.2 | 176.0 | 100 | 130 | 25 |
| Corydaline | 369.2 | ESI+ | 5.35 | 370.2 | 192.1 | 100 | 140 | 28 |
| Palmatine | 352.2 | ESI+ | 5.49 | 352.2 | 308.1 | 100 | 130 | 28 |
| Salvianolic acid B | 718.2 | ESI− | 5.54 | 717.1 | 519.1 | 100 | 150 | 13 |
| Berberine | 336.1 | ESI+ | 5.57 | 336.2 | 320.2 | 100 | 130 | 30 |
| Salvianolic acid A | 494.1 | ESI− | 5.78 | 493.1 | 295.0 | 100 | 130 | 10 |
| Analyte | LOD (ng/mL) | LOQ (ng/mL) | Linear Range (ng/mL | Slope (Mean ± SD) | Intercept (Mean ± SD) | r |
|---|---|---|---|---|---|---|
| Hydroxysafflor yellow A | 0.18 | 0.60 | 20.00–10,000.00 | 10.2902 ± 0.2564 | 2250.3307 ± 52.4467 | 0.9956 |
| Danshensu | 0.60 | 2.00 | 20.00–10,000.00 | 8.6778 ± 0.1746 | −456.6715 ± 9.8327 | 0.9992 |
| Amygdalin | 1.34 | 4.46 | 200.00–100,000.00 | 16.1377 ± 0.3781 | 4149.1033 ± 82.7875 | 0.9978 |
| Caffeic acid | 0.32 | 1.07 | 1.50–750.00 | 54.9048 ± 2.0422 | 385.0904 ± 8.8482 | 0.9991 |
| Protopine | 0.08 | 0.27 | 3.00–1500.00 | 1908.4269 ± 30.4657 | 975.8435 ± 17.3396 | 0.9990 |
| Ferulic acid | 2.21 | 7.36 | 15.00–7500.00 | 2.2024 ± 0.0462 | 1946.4735 ± 14.9437 | 0.9920 |
| Tetrahydropalmatine | 0.04 | 0.13 | 3.00–1500.00 | 6601.3733 ± 95.4892 | 2954.9081 ± 56.2518 | 0.9993 |
| Coptisine | 0.05 | 0.17 | 3.00–1500.00 | 491.4143 ± 9.3921 | 236.4471 ± 4.6437 | 0.9993 |
| Tetrahydrocoptisine | 0.06 | 0.20 | 1.50–750.00 | 2399.6703 ± 42.722 | 955.3523 ± 15.3732 | 0.9996 |
| Tetrahydroberineper | 0.02 | 0.07 | 1.50–750.00 | 8906.0226 ± 101.7735 | 1590.6877 ± 43.2583 | 0.9994 |
| Corydaline | 0.09 | 0.30 | 3.00–1500.00 | 4017.0902 ± 99.8759 | 537.0929 ± 10.2247 | 0.9994 |
| Palmatine | 0.02 | 0.07 | 3.00–1500.00 | 2643.7965 ± 55.2618 | −495.5594 ± 10.7284 | 0.9997 |
| Salvianolic acid B | 5.81 | 19.35 | 500.00–250000.00 | 30.7107 ± 1.2739 | 606660.6878 ± 1298.4689 | 0.9956 |
| Berberine | 0.01 | 0.03 | 1.50–750.00 | 3759.8114 ± 98.3726 | 2643.4459 ± 67.7739 | 0.9991 |
| Salvianolic acid A | 3.45 | 11.49 | 15.00–7500 | 7.4196 ± 0.5444 | 2354.9524 ± 78.1819 | 0.9916 |
| Analyte | Intra-day (RSD n = 6) | Inter-day (RSD n = 6) | Repeatability (RSD n = 6) | Stability (24 h) (RSD n = 6) |
|---|---|---|---|---|
| Hydroxysafflor yellow A | 2.03 | 3.60 | 2.90 | 3.28 |
| Danshensu | 0.71 | 3.01 | 3.27 | 2.09 |
| Amygdalin | 1.44 | 2.95 | 2.05 | 2.01 |
| Caffeic acid | 1.23 | 2.40 | 3.05 | 1.33 |
| Protopine | 0.49 | 0.68 | 1.45 | 2.19 |
| Ferulic acid | 1.44 | 3.08 | 3.09 | 4.48 |
| Tetrahydropalmatine | 0.61 | 0.95 | 1.85 | 3.82 |
| Coptisine | 0.74 | 0.94 | 1.79 | 3.75 |
| Tetrahydrocoptisine | 0.36 | 0.65 | 2.09 | 3.41 |
| Tetrahydroberineper | 0.41 | 1.24 | 1.86 | 2.99 |
| Corydaline | 0.30 | 0.95 | 1.89 | 2.75 |
| Palmatine | 1.00 | 0.84 | 1.57 | 2.37 |
| Salvianolic acid B | 1.82 | 3.61 | 3.23 | 4.47 |
| Berberine | 0.31 | 1.00 | 0.81 | 2.49 |
| Salvianolic acid A | 3.25 | 2.00 | 3.10 | 3.22 |
| Analyte | Original (ng) | Spiked (ng) | Detection (ng) | Recovery (%) | RSD (%) |
|---|---|---|---|---|---|
| Hydroxysafflor yellow A | 99.55 | 80 | 179.19 | 99.55 | 1.80 |
| 99.55 | 100 | 200.10 | 100.55 | 0.19 | |
| 99.55 | 120 | 219.01 | 99.55 | 0.64 | |
| Danshensu | 36,713.81 | 29,600 | 66,084.86 | 99.23 | 1.52 |
| 36,713.81 | 37,000 | 74,372.45 | 101.78 | 0.65 | |
| 36,713.81 | 44,400 | 80,770.39 | 99.23 | 1.19 | |
| Amygdalin | 606.20 | 480 | 1098.17 | 102.49 | 2.83 |
| 606.20 | 600 | 1192.46 | 97.71 | 1.42 | |
| 606.20 | 720 | 1342.21 | 102.22 | 1.84 | |
| Caffeic acid | 1018.24 | 800 | 1823.74 | 100.69 | 0.55 |
| 1018.24 | 1000 | 2025.88 | 100.76 | 1.80 | |
| 1018.24 | 1200 | 2251.24 | 102.75 | 1.78 | |
| Protopine | 173.99 | 140 | 311.99 | 98.57 | 0.51 |
| 173.99 | 180 | 346.86 | 96.04 | 0.79 | |
| 173.99 | 225 | 388.99 | 95.56 | 1.55 | |
| Ferulic acid | 10,800.76 | 8640 | 19,441.37 | 100.01 | 0.66 |
| 10,800.76 | 10,800 | 21,686.07 | 100.79 | 0.89 | |
| 10,800.76 | 12,960 | 23,761.68 | 100.01 | 0.58 | |
| Tetrahydropalmatine | 223.40 | 180 | 402.12 | 99.29 | 0.51 |
| 223.40 | 225 | 446.69 | 99.24 | 0.95 | |
| 223.40 | 270 | 489.48 | 98.55 | 0.79 | |
| Coptisine | 222.30 | 180 | 398.15 | 97.69 | 1.42 |
| 222.30 | 225 | 449.66 | 101.05 | 0.50 | |
| 222.30 | 270 | 497.07 | 101.77 | 0.25 | |
| Tetrahydrocoptisine | 96.60 | 80 | 175.89 | 99.11 | 0.13 |
| 96.60 | 100 | 198.22 | 101.62 | 0.71 | |
| 96.60 | 120 | 209.53 | 94.11 | 0.71 | |
| Tetrahydroberineper | 47.47 | 40 | 88.44 | 102.43 | 0.21 |
| 47.47 | 50 | 99.38 | 103.82 | 0.22 | |
| 47.47 | 60 | 108.43 | 101.60 | 0.80 | |
| Corydaline | 154.16 | 120 | 277.49 | 102.78 | 0.26 |
| 154.16 | 150 | 296.95 | 95.19 | 0.28 | |
| 154.16 | 180 | 339.16 | 102.78 | 0.23 | |
| Palmatine | 223.44 | 180 | 402.19 | 99.31 | 1.51 |
| 223.44 | 225 | 440.03 | 96.26 | 1.16 | |
| 223.44 | 270 | 491.56 | 99.30 | 0.44 | |
| Salvianolic acid B | 1495.32 | 1200 | 2691.58 | 99.69 | 0.77 |
| 1495.32 | 1500 | 2979.81 | 98.97 | 2.25 | |
| 1495.32 | 1800 | 3289.71 | 99.69 | 0.49 | |
| Berberine | 52.89 | 40 | 93.08 | 100.48 | 0.55 |
| 52.89 | 50 | 101.78 | 97.78 | 1.01 | |
| 52.89 | 60 | 113.77 | 101.47 | 1.01 | |
| Salvianolic acid A | 848.31 | 680 | 1526.95 | 99.80 | 1.64 |
| 848.31 | 850 | 1715.10 | 101.98 | 0.73 | |
| 848.31 | 1020 | 1866.28 | 99.80 | 0.62 |
| Compounds | Batch No. | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 50,302 | 50,303 | 50,401 | 50,601 | 50,602 | 70,101 | 70,102 | 70,501 | 70,502 | 90,401 | 91,201 | 100,101 | 100,102 | 110,701 | |
| Hydroxysafflor yellow A | 408.13 | 133.54 | 73.22 | 88.64 | 87.7 | 274.94 | 282.12 | 258.85 | 111.33 | 299.29 | 301.11 | 259.11 | 477.09 | 68.87 |
| Danshensu | 570.46 | 726.94 | 763.21 | 672.6 | 626.5 | 478.07 | 734.94 | 461.14 | 557.16 | 581.95 | 417.27 | 943.38 | 467.38 | 1406.02 |
| Amygdalin | 3566.33 | 3907.22 | 3928.31 | 4611.34 | 4533.97 | 4037.1 | 4618.81 | 5405.11 | 3782.52 | 7642.94 | 5825.54 | 5504.12 | 8527.1 | 4081.63 |
| Caffeic acid | 36.06 | 18.27 | 22.18 | 23.33 | 14.01 | 29.39 | 34.6 | 45.17 | 32.1 | 28.2 | 30.06 | 24.61 | 38.52 | 22.79 |
| Protopine | 135.37 | 87.81 | 90.38 | 87.33 | 85.6 | 108.84 | 94.09 | 80.63 | 81.92 | 36.42 | 69.17 | 68.25 | 35.8 | 66.78 |
| Ferulic acid | 155.6 | 82.59 | 98.93 | 103.46 | 85.93 | 157.09 | 172.73 | 211.41 | 151.8 | 230.84 | 273.67 | 259.4 | 330.46 | 192.62 |
| Tetrahydropalmatine | 177.55 | 152.42 | 164.02 | 112.17 | 119.27 | 160.45 | 109.38 | 119.25 | 102.69 | 43.33 | 81.81 | 64.45 | 38.96 | 81.2 |
| Coptisine | 253.52 | 141.21 | 149.96 | 132.52 | 110.31 | 184.39 | 138.94 | 121.61 | 123.71 | 36.43 | 75.06 | 64.15 | 33.94 | 52.13 |
| Tetrahydrocoptisine | 81.34 | 55.83 | 59.79 | 36.72 | 33.32 | 64.62 | 35.88 | 39.81 | 32.62 | 19.06 | 38.56 | 32.3 | 16.49 | 39.95 |
| Tetrahydroberineper | 22.81 | 19 | 23.05 | 12.58 | 18.69 | 26.86 | 12.25 | 13.1 | 11.08 | 5.64 | 11.99 | 10.18 | 4.3 | 11.35 |
| Corydaline | 132.19 | 98.41 | 122.47 | 51.16 | 67.91 | 124.4 | 45.23 | 61.93 | 40.66 | 17.06 | 57.79 | 50.51 | 17.15 | 51.77 |
| Palmatine | 193.22 | 104.11 | 98.49 | 131.12 | 93.58 | 146.23 | 163.81 | 136.34 | 135.15 | 45.25 | 79.9 | 80.54 | 46.17 | 75.24 |
| Salvianolic acid B | 20,674.32 | 2583.65 | 2655.05 | 15,835.48 | 2532.11 | 2135.44 | 14,172.63 | 9504.99 | 10,043.33 | 13,626.23 | 2166.91 | 5100.94 | 16,852.62 | 5460.88 |
| Berberine | 41.06 | 23.61 | 25.05 | 27.7 | 24.24 | 36.55 | 34.16 | 28.26 | 29.34 | 10.89 | 40.74 | 45.81 | 14.56 | 17.07 |
| Salvianolic acid A | 814.45 | 640.97 | 735.76 | 1017.09 | 998.76 | 666.93 | 1113.39 | 831.28 | 1112.67 | 717.89 | 329.76 | 196.28 | 509.48 | 385.02 |
| Total amount | 27,262.41 | 8775.58 | 9009.87 | 22,943.24 | 9431.9 | 8631.3 | 21,762.96 | 17,318.88 | 16,348.08 | 23,341.42 | 9799.34 | 12,704.03 | 27,410.02 | 12,013.32 |
2.4. Quantitative Analysis of Fifteen Compounds in JTT Samples
2.5. Marker Compound Selection for Quality Control of Complex TCMP
3. Experimental
3.1. Chemicals and Reagents
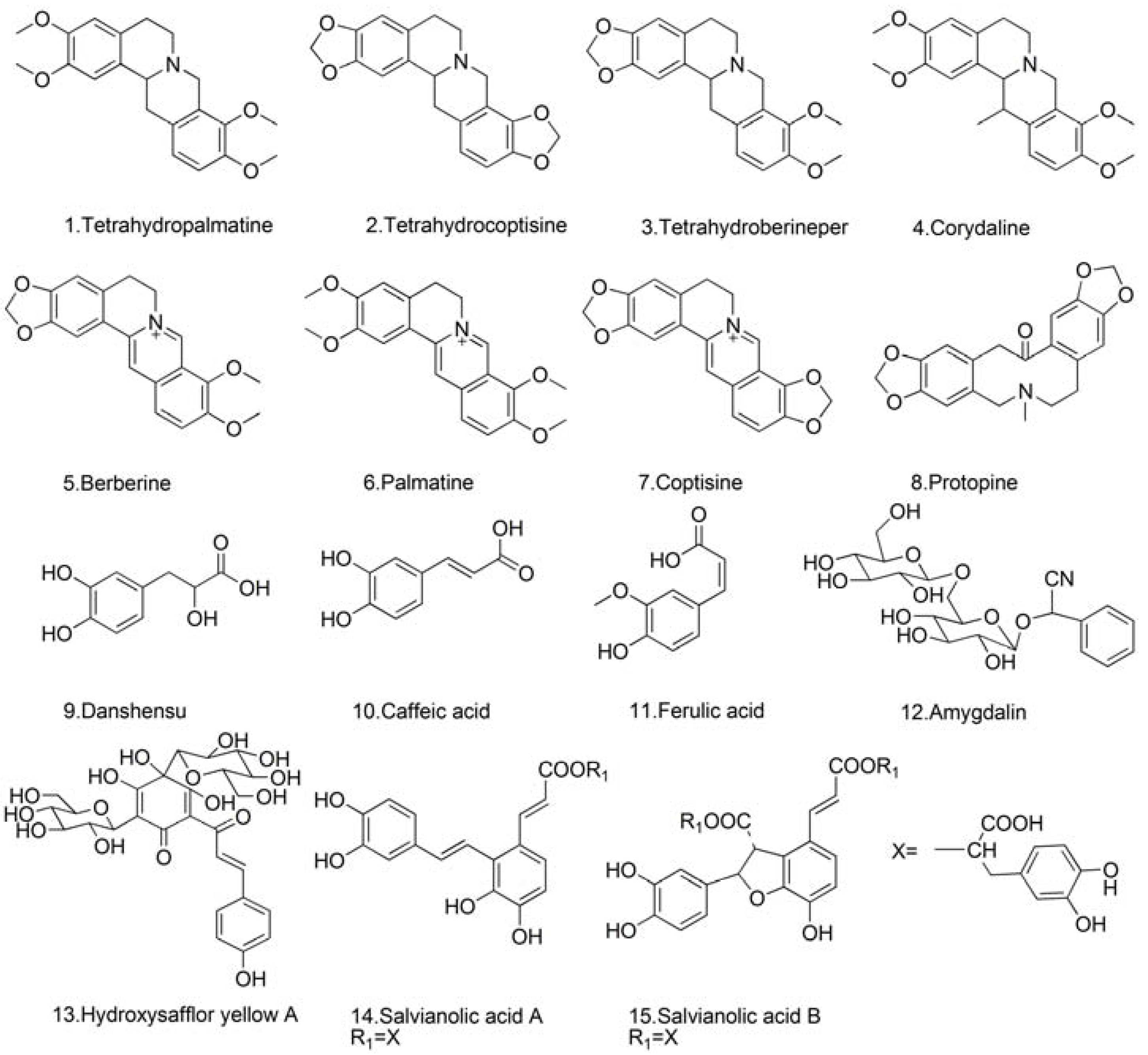
3.2. Sample Preparation
3.2.1. Preparation of the JTT Analytical Sample
3.2.2. Preparation of Standard Solutions
3.3. Liquid Chromatography Conditions
3.4. Mass Spectrometric Conditions
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Yin, L.H.; Lu, B.N.; Qi, Y.; Xu, L.N.; Han, X.; Xu, Y.W.; Peng, J.Y.; Sun, C.K. Simultaneous determination of 11 active components in two well-known traditional Chinese medicines by HPLC coupled with diode array detection for quality control. J. Pharm. Biomed. Anal. 2009, 49, 1101–1108. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, L.; Zhang, X.; Dai, W.X.; Li, H.Y.; Hu, L.W.; Liu, H.; Su, J.; Zhang, W.D. Qualitative and quantitative analysis of traditional Chinese medicine Niu Huang Jie Du Pill using ultra performance liquid chromatography coupled with tunable UV detector and rapid resolution liquid chromatography coupled with time-of-flight tandem mass spectrometry. J. Pharm. Biomed. Anal. 2010, 51, 565–571. [Google Scholar] [CrossRef]
- Zhang, Y.C.; Xu, H.Y.; Chen, X.M.; Chen, C.; Wang, H.J.; Meng, F.Y.; Yang, H.J.; Huang, L.Q. Simultaneous quantification of 17 constituents from Yuanhu Zhitong tablet using rapid resolution liquid chromatography coupled with a triple quadrupole electrospray tandem mass spectrometry. J. Pharm. Biomed. Anal. 2011, 56, 497–504. [Google Scholar] [CrossRef]
- Li, S.P.; Zhao, J.; Yang, B. Strategies for quality control of Chinese medicines. J. Pharm. Biomed. Anal. 2011, 55, 802–809. [Google Scholar] [CrossRef]
- Liang, X.M.; Jin, Y.; Wang, Y.P.; Jin, G.W.; Fu, Q.; Xiao, Y.S. Qualitative and quantitative analysis in quality control of traditional Chinese medicines. J. Chromatogr. A 2009, 1216, 2033–2044. [Google Scholar] [CrossRef]
- Jiang, Y.; David, B.; Tu, P.F.; Barbin, Y. Recent analytical approaches in quality control of traditional Chinese medicines-a review. Anal. Chim. Acta 2010, 657, 9–18. [Google Scholar] [CrossRef]
- Song, J.Z.; Li, S.L.; Zhou, Y.; Qiao, C.F.; Chen, S.L.; Xu, H.X. A novel approach to rapidly explore analytical markers for quality control of Radix Salviae Miltiorrhizae extract granules by robust principal component analysis with ultra-high performance liquid chromatography-ultraviolet-quadrupole time-of-flight mass spectrometry. J. Pharm. Biomed. Anal. 2010, 53, 279–286. [Google Scholar] [CrossRef]
- The European Medicines Agency: Reflection paper on markers used for quantitative and qualitative analysis of herbal medicinal products and traditional herbal medicinal products. Available online: http://www.emea.europa.eu/pdfs/human/hmpc/25362907en.pdf&refdoi=10.1186/1749-8546-3-7 (accessed on 27 January 2014).
- Li, L.J.; Xing, X.F.; Shao, H.X. Preparation of traditional Chinese herbs on addiction detoxification: recent progress. Chin. Med. 2003, 34, 20–22. [Google Scholar]
- Shi, J.; Liu, Y.L.; Fang, Y.X.; Xu, G.Z.; Zhai, H.F.; Lu, L. Traditional Chinese medicine in treatment of opiate addiction. Acta Pharmacol. Sin. 2006, 27, 1303–1308. [Google Scholar] [CrossRef]
- Xiong, J.G.; Xiao, Z.X.; Li, J.; Qin, D.S.; Min, M.S.; Yang, J.H.; Wang, Y.H.; Yan, L.; Shu, Q.L. Clinical effect of Jitai tablet combined with lofexidine in the treatment of heroin dependence. Chin. J. Drug Depend. 2001, 4, 290–292. [Google Scholar]
- Tu, Q.X.; Zhao, H.G.; Cheng, Y.P.; Chen, Y.M.; Huang, X.P.; Chen, Y.M.; Han, M. Comparision study on clinical efficacy of Jitai capsule with lofexidine in the treatment of opiate addicts. Chin. J. Drug Depend. 1999, 4, 285–287. [Google Scholar]
- Chinese Pharmacopeia Committee. Pharmacopeia of the People’s Republic of China; China Medical Science Press: Beijing, China, 2010; Volume 1, pp. 130–131. [Google Scholar]
- Wang, S.P.; Liu, L.; Wang, L.L.; Hu, Y.H.; Zhang, W.D.; Liu, R.H. Structural characterization and identification of major constituents in Jitai tablets by high-performance liquid chromatography/diode-array detection coupled with electrospray ionization tandem mass spectrometry. Molecules 2012, 17, 10470–10493. [Google Scholar] [CrossRef]
- Wang, J.B.; Mantsch, J.R. l-Tetrahydropalamatine: a potential new medication for the treatment of cocaine addiction. Future Med. Chem. 2012, 2, 177–186. [Google Scholar] [CrossRef]
- Xi, Z.X.; Gilbert, J.; Campos, A.C.; Kline, N.; Ashby, C.R., Jr.; Hagan, J.J.; Heidbreder, C.A.; Gardner, E.L. Blockade of mesolimbic dopamine D3 receptors inhibits stress induced reinstatement of cocaine-seeking in rats. Psychopharmacology (Berl) 2004, 176, 57–65. [Google Scholar]
- Liu, Y.L.; Liang, J.H.; Yan, L.D.; Su, R.B.; Wu, C.F.; Gong, Z.H. Effects of l-tetrahydropalmatine on locomotor sensitization to oxycodone in mice. Acta Pharmacol. Sin. 2005, 26, 533–538. [Google Scholar] [CrossRef]
- Chu, H.Y.; Jin, G.Z.; Friedman, E.; Zhen, X.C. Recent development in studies of tetrahydroprotoberberines: mechanism in antinociception and drug addiction. Cell Mol. Neurobiol. 2008, 28, 491–499. [Google Scholar] [CrossRef]
- Kinodhita, K.; Kawai, T.; Imaizumi, T.; Akita, Y.; Koyama, K.; Takahashi, K. Anti-emetic principles of Inula linariaefolia flowers and Forsythia suspensa fruits. Phytomedicine 1996, 3, 51–58. [Google Scholar] [CrossRef]
- Barone, E.; Calabrese, V.; Mancuso, C. Ferulic acid and its therapeutic potential as a hormetin for age-related diseases. Biogerontology 2009, 10, 97–108. [Google Scholar] [CrossRef]
- Lin, W.C.; Lin, J.Y. Five bitter compounds display different anti-inflammatory effects through modulating cytokine secretion using mouse primary splenocytes in vitro. J. Agric. Food Chem. 2011, 59, 184–192. [Google Scholar] [CrossRef]
- Sun, X.; Wei, X.B.; Qu, S.F.; Zhao, Y.X.; Zhang, X.M. Hydroxysafflor Yellow A suppresses thrombin generation and inflammatory responses following focal cerebral ischemia—reperfusion in rats. Bioorg. Med. Chem. Lett. 2010, 20, 4120–4124. [Google Scholar] [CrossRef]
- Janis, Y.X.Z.; Ken, Y.Z.Z.; Kevin, Y.Z.; Cathy, W.C.B.; Wendy, L.Z.; Crystal, Y.Q.D.; Qiang, F.; Tina, T.X.D.; Roy, C.Y.C.; Karl, W.K.T.; et al. Chemical and biological assessment of angelicae sinensis radix after processing with wine: an orthogonal array design to reveal the optimized conditions. J. Agric. Food Chem. 2011, 59, 6091–6198. [Google Scholar] [CrossRef]
- Xie, Y.; Jiang, Z.H.; Zhou, H.; Xu, H.X.; Liu, L. Simultaneous determination of six Aconitum alkaloids in proprietary Chinese medicines by high-performance liquid chromatography. J. Chromatogr. A 2005, 1093, 195–203. [Google Scholar] [CrossRef]
- Yang, J.; Chen, L.H.; Zhang, Q.; Lai, M.X.; Wang, Q. Quality assessment of Cortex cinnamomiby HPLC chemical fingerprint, principle component analysis and cluster analysis. J. Sep. Sci. 2007, 30, 1276–1283. [Google Scholar] [CrossRef]
- Fan, L.; Zhao, H.Y.; Xu, M.; Zhou, L.; Guo, H.; Han, J.; Wang, B.R.; Guo, D.A. Qualitative evaluation and quantitative determination of 10 major active components in Carthamus tinctorius L. by high-performance liquid chromatography coupled with diode array detector. J. Chromatogr. A 2009, 1216, 2063–2070. [Google Scholar] [CrossRef]
- Lu, G.H.; Chan, K.; Liang, Y.Z.; Leung, K.; Chan, C.L.; Jiang, Z.H.; Zhao, Z.Z. Development of high-performance liquid chromatographic fingerprints for distinguishing Chinese Angelica from related umbelliferae herbs. J. Chromatogr. A 2005, 1073, 383–392. [Google Scholar] [CrossRef]
- Yan, R.; Li, S.L.; Chung, H.S.; Tam, Y.K.; Lin, G. Simultaneous quantification of 12 bioactive components of Ligusticum chuanxiong Hort. by high-performance liquid chromatography. J. Pharm. Biomed. Anal. 2005, 37, 87–95. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the protopine, tetrahydropalmatine, coptisine, amygdalin, caffeic acid, danshensu, hydroxysafflor yellow A, ferulic acid, tetrahydrocoptisine, tetrahydroberineper, corydaline, palmatine, berberine, salvianolic acid A and salvianolic acid B are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, S.; Fu, P.; Liu, L.; Wang, L.; Peng, C.; Zhang, W.; Liu, R. Simultaneous Determination of Fifteen Constituents of Jitai Tablet Using Ultra High-Performance Liquid Chromatography Coupled with Triple Quadrupole Electrospray Tandem Mass Spectrometry. Molecules 2014, 19, 1635-1650. https://doi.org/10.3390/molecules19021635
Wang S, Fu P, Liu L, Wang L, Peng C, Zhang W, Liu R. Simultaneous Determination of Fifteen Constituents of Jitai Tablet Using Ultra High-Performance Liquid Chromatography Coupled with Triple Quadrupole Electrospray Tandem Mass Spectrometry. Molecules. 2014; 19(2):1635-1650. https://doi.org/10.3390/molecules19021635
Chicago/Turabian StyleWang, Shuping, Peng Fu, Lei Liu, Lingling Wang, Chengcheng Peng, Weidong Zhang, and Runhui Liu. 2014. "Simultaneous Determination of Fifteen Constituents of Jitai Tablet Using Ultra High-Performance Liquid Chromatography Coupled with Triple Quadrupole Electrospray Tandem Mass Spectrometry" Molecules 19, no. 2: 1635-1650. https://doi.org/10.3390/molecules19021635
APA StyleWang, S., Fu, P., Liu, L., Wang, L., Peng, C., Zhang, W., & Liu, R. (2014). Simultaneous Determination of Fifteen Constituents of Jitai Tablet Using Ultra High-Performance Liquid Chromatography Coupled with Triple Quadrupole Electrospray Tandem Mass Spectrometry. Molecules, 19(2), 1635-1650. https://doi.org/10.3390/molecules19021635




