Abstract
A new class of 4H-imidazoles was synthesized starting from fused-ring aromatic dinitriles. Strong bathochromic shifts of the longest wavelength absorptions were observed in the corresponding UV/vis spectra due to a conversion of the merocyanine chromophores into cyanines/(aza)oxonoles upon protonation/deprotonation of the 4H-imidazoles. Novel boratetraazapentalenes were synthesized via a cyclization reaction with boron trifluoride. These mesoionic species bearing a cyanine chromophore not only show NIR-fluorescence, they also participate as part of a quasi-reversible multi-step redox system. Large calculated semiquinone formation constants KSEM (3x1010 to 5x1011) indicate a high thermodynamic stability of the corresponding radical anions (SEM).
Introduction
Over the last several years a great number of organic substances with extraordinary absorption, emission or electrochemical properties have been developed. In conjunction with materials science, many of these substances were employed in new materials such as displays, organic LED’s or other, mostly electronic devices. Only a few organic systems combine all the desirable properties for all the mentioned application fields, thus making research on polyfunctional dyes very popular.
4H-Imidazoles 1 are well known for their long wavelength UV/vis absorptions and high extinction coefficients (log ▯ > 4) [1]. As part of a two-electron redox system, the reduction of the deeply coloured 4H-imidazoles 1 (OX) leads to nearly colourless, fluorescent 1H-imidazoles 2 (RED) via radical-ion intermediates (SEM) (Scheme 1). Cyclization of derivatives 1 with boron compounds yields red fluorescent boratetraazapentalenes 3, which on the other hand may be regarded as electron-poor heterocycles. The radical anions of this new class of electrophores show unusual high KSEM values (up to 1015) [2].
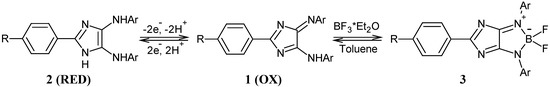
Scheme 1.
Redox behaviour and cyclization of 4H-imidazoles.
As part of a four-electron redox system, in which the radical state is replaced by more stable closed-shell moieties, bis-4H-imidazoles show almost identical properties as their monofunctional parent compounds [3]. Due to their long wavelength absorptions in the UV/vis spectra, correlated with high extinction coefficients, together with their interesting redox behaviour, 4H-imidazoles are promising candidates for the synthesis of functional dyes [4].
Therefore we sought to prepare 4H-imidazoles which are connected to other chromophores. The involvement of ring-fused aromatic systems such as anthracene or naphthalene, should lead to bathochromic shifts of the absorptions/emissions of the corresponding boratetraazapentalenes. Due to the switch between merocyanine and cyanine upon protonation of 4H-imidazoles, their application as acidochromic dyes also should be of interest.
Results and Discussion
As the starting material required for bis-4H-imidazoles, dinitriles of type 4 were synthesized via a condensation reaction of o-xylylenedicyanide with the corresponding 1,2-diketones in the presence of sodium ethoxide [5,6]. The naphthalene derivative 4d was obtained starting from 1,5-naphthalenediol [7,8]. All reported dinitriles are yellow-greenish fluorescent solids and, with the exception of derivative 4d, sparingly soluble in organic solvents. Reactions of the dinitriles 4 with lithium hexamethyldisilazide (LiHMDS) yielded the corresponding amidinates, which were converted into the persilylated amidines by heating with chlorotrimethylsilane under reflux. As depicted in Scheme 2, all attempts to interconvert both nitrile groups failed. This failure can be attributed to considerable delocalization of the negative charge over the aromatic system which significantly lowers the electrophilicity of the second cyano group. The observed decrease of reactivity is in accordance with the synthesis of a series of substituted aromatic amidines [9]. Unfortunately, the alternative Pinner-synthesis route was not successful, due to poor solubility of dinitriles 4 in ethanol.
The 4H-imidazoles of type 5 were synthesized by a deprotection-cyclization sequence using bis-imidoylchlorides derived from oxalic acid in the presence of KF/18-crown-6-ether. The products 5 were purified by column chromatography of the reaction mixture in 15 up to 30% yield (Scheme 2).
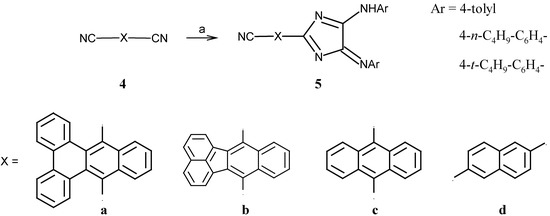
Scheme 2.
Synthesis of 4H-imidazoles 5.
The newly synthesized 4H-imidazoles have essentially the same properties as the phenyl-substituted derivatives [1]. Their longest wavelength absorption in the UV/vis spectra is approximately 515 to 540 nm, with log(ε) values higher than 4.0; the infrared spectra show two characteristic bands at about 3400 cm-1 (NH-group) and 2220 cm-1 for the cyano-group. In the NMR spectra two doublets were detected between 7 and 8 ppm, which are characteristic for the aromatic protons of the arylamino/arylimino moieties.
We recently demonstrated that 4H-imidazoles and other cyclic amidines (e.g. tetraazafulvalenes) behave as electrophores which can easily be switched between oxidized and reduced forms [2,10]. As a first step, most likely the corresponding radical anion is generated. The second electron transfer step then leads to the formation of the dianion, which is immediately protonated in the presence of water to yield the 1H-imidazoles of type 6. Now, both consecutive single-electron transfer processes could be recorded by electrochemical measurements. Employing difference pulse polarographic measurements, two peaks can clearly be ascribed to two different single-electron transfer steps. The quasi-reversibility of the reduction was confirmed by cyclovoltammetric measurements (Δ ERED,OX1,2 > 0,059V). The redox potentials and semiquinone formation constants KSEM of 4H-imidazoles 5 are listed in Table 1.

Table 1.
Redox potentials and semiquinone formation constants of 4H-imidazoles 5.
| Comp. | Ar | E1Red, E2Red | KSEM |
|---|---|---|---|
| 5a | 4-tolyl | -0.72V, -1.05V | 3.9*105 |
| 5a | 4-n-C4H9-C6H4- | -0.85V, -1.16V | 1.8*105 |
| 5a | 4-t-C4H9-C6H4- | -0.87V, -1.17V | 1.2*105 |
| 5b | 4-n-C4H9-C6H4- | -1.15V, -1.48V | 3.9*105 |
| 5b | 4-t-C4H9-C6H4- | -1.02V, -1.34V | 2.7*105 |
| 5c | 4-tolyl | -0.84V, -1.12V | 5.6*104 |
| 5c | 4-n-C4H9-C6H4- | -0.75V, -1.12V | 1.9*106 |
| 5c | 4-t-C4H9-C6H4- | -0.79V, -1.13V | 5.8*105 |
| 5d | 4-tolyl | -0.90V, -1.31V | 8.9*106 |
| 5d | 4-n-C4H9-C6H4- | -0.94V, -1.39V | 4.2*107 |
Chemically induced reduction could be achieved by treating a solution of the 4H-imidazole 5 with sodium dithionite in THF. The colour of the reaction mixture changed from red to light yellow, which indicates the formation of 1H-imidazoles 6. These nearly colourless, but fluorescent products (λmax,em : 439 up to 665nm), immediately reoxidize when exposed to air (Scheme 3).
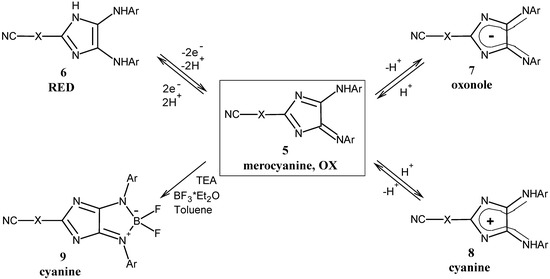
Scheme 3.
Modifications of 4H-imidazoles.
A further interesting feature of 4H-imidazoles 5 is the protonation-deprotonation reaction with acids or bases (Scheme 3). While the neutral species is based on a merocyanine system, the formation of the protonated or deprotonated 4H-imidazole causes an alteration of their chromophoric systems and consequently of their long wavelength absorption in UV/vis spectra. Compared to the neutral species, both the protonated (cyanine) and the deprotonated 4H-imidazole (azaoxonole) display a bathochromic shift in their UV/vis spectra (Table 2).

Table 2.
Absorption and emission maxima of reduced 6, deprotonated 7 and protonated 8.
| Comp. | Ar | λmax/type 5 log ε | λmax/type 6 log ε | λmax,em/type 6 λmax,exc. | λmax/type 7 log ε | λmax/type 8 log ε |
| 5-8a | 4-tolyl | 519 (4.1) | 409 (4.2) | 439 (401) | 533 (4.1) | 619 (4.2) |
| 5-8a | 4-n-C4H9-C6H4- | 524 (4.0) | 411 (4.2) | 455 (418) | 540 (4.1) | 623 (4.2) |
| 5-8a | 4-t-C4H9-C6H4- | 521 (4.1) | 412 (4.2) | 475 (429) | 542 (4.1) | 625 (4.2) |
| 5-8b | 4-n-C4H9-C6H4- | 523 (4.3) | 406 (4.2) | 535 (457) | 535 (4.1) | 620 (4.2) |
| 5-8b | 4-t-C4H9-C6H4- | 517 (4.3) | 412 (4.2) | 570 (461) | 538 (4.1) | 614 (4.2) |
| 5-8c | 4-tolyl | 520 (4.2) | 409 (4.2) | 448 (408) | 535 (4.1) | 614 (4.2) |
| 5-8c | 4-n-C4H9-C6H4- | 525 (4.2) | 409 (4.2) | 471 (427) | 538 (4.1) | 617 (4.3) |
| 5-8c | 4-t-C4H9-C6H4- | 526 (4.1) | 410 (4.2) | 470 (428) | 540 (4.1) | 618 (4.2) |
| 5-8d | 4-tolyl | 536 (4.1) | 415 (4.2) | 651 (432) | 549 (4.1) | 631 (4.2) |
| 5-8d | 4-n-C4H9-C6H4- | 540 (4.2) | 421 (4.2) | 665 (451) | 555 (4.1) | 637 (4.2) |
UV/vis and fluorescence spectra were recorded in THF-solution.
Usually the cyanines 8 and the oxonoles 7 display bathochromic shifts of approximately 100 nm and 20 nm, respectively, with regards to the merocyanine 5 (Figure 1). The isosbestic point in the UV/vis spectra during protonation or deprotonation of the 4H-imidazole 5c indicates a first order reaction (Figure 2). Thus, 4H-imidazoles are well suited for the application as acidochromic dyes, especially in sensor systems.
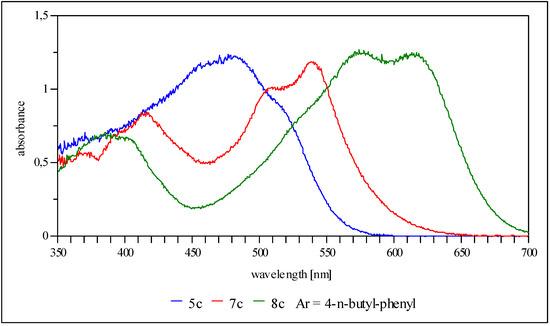
Figure 1.
UV/vis spectra (THF) of anthracene derivative 5c, deprotonated 7c and protonated 5c species.
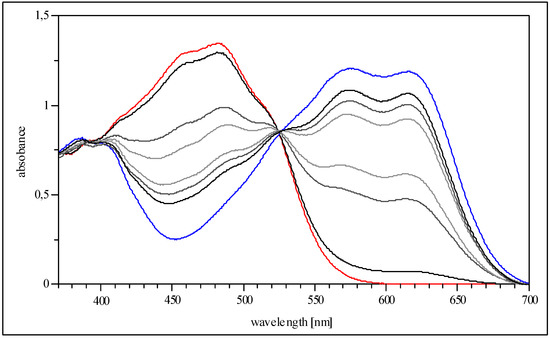
Figure 2.
Isosbestic point (at 524nm, THF) during protonation of 5c.
The mesoionic boracycles 9 were obtained in about 50% yield by cyclization of 4H-imidazoles 5 with boron trifluoride diethyl ether complex in the presence of triethylamine. These deep blue reaction products were purified by column chromatography and identified by MS-, IR-, NMR- and UV/vis spectroscopy. Cyclic voltammetric measurements showed the quasi-reversibility of the reduction step. The very large calculated semiquinone formation constants KSEM (3x1010 to 5x1011) indicate a high thermodynamic stability of the radicalanions (SEM) (Table 3).

Table 3.
Electrochemical properties of the 1,3,2-diazaborolidines 9.
| Comp. | Ar | E1Red, E2Red | KSEM |
|---|---|---|---|
| 9a | 4-n-C4H9-C6H4- | -0.45V, -1.14V | 5.0*1011 |
| 9a | 4-t-C4H9-C6H4- | -0.48V, -1.16V | 3.4*1011 |
| 9b | 4-t-C4H9-C6H4- | -0.88V, -1.50V | 3.2*1010 |
| 9c | 4-n-C4H9-C6H4- | -0.42V, -1.05V | 4.8*1010 |
| 9d | 4-tolyl | -0.82V, -1.47V | 1.0*1011 |
Compared with the phenylogous boracycles 3, the absorptions of 1,3,2-diazaborolidines 9 are shifted bathochromically (between 600 and 620nm). An unusually high stokes-shift of about 250 to 300 nm leads to the longest wavelength emissions in the area of NIR (Φ < 20%), exemplified by derivative 9d which is emitting at 870 nm (Figure 3).
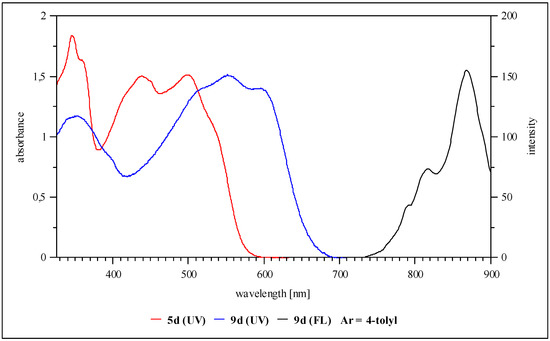
Figure 3.
UV/vis- and fluorescence spectra of derivative 9d (solvent THF).
Conclusions
We have prepared the new 4H-imidazoles 5 and mesoionic 1,3,2-diazaborolidines 9, and have characterized them by elemental analysis, MS and NMR. Strong bathochromic shifts of the longest wavelength absorptions were observed in the corresponding UV/vis spectra due to a conversion of the merocyanine chromophores into cyanines/(aza)oxonoles upon protonation/deprotonation. The mesoionic boracycles 9 bearing a cyanine chromophore not only show NIR-fluorescence with an unusually high Stokes-shift, but they also participate as part of a quasi-reversible multi-step redox system. Large calculated semiquinone formation constants indicate a high thermodynamic stability of the corresponding radical anions. Thus, derivatives of 4H-imidazole are well suited for the applications in the functional dyes field.
Experimental
General
All reactions were monitored by TLC, carried out on 0.25 mm Merck silica gel plates (60F254) using UV light for visualization. 1H- and 13C-NMR spectra were recorded with a Bruker DRX 400 or Bruker AC 250 spectrometer. Melting points are measured with a Galen TM 3 apparatus and are uncorrected. UV/vis spectra were recorded on a Perkin-Elmer Lambda 19 spectrophotometer. MS spectra were taken from measurements on a Finnigan MAT SAQ 710 mass spectrometer. Elemental analyses were carried out in-house with an automatic analyzer LECO CHNS 932. Electrochemical measurements were carried out in dichloromethane with a Metrohm 663VA Stand using mercury or platinum electrodes (reference electrode SCE) and tetrabutylammonium hexafluorophosphate as conductive salt. The fluorescence spectra were recorded with a JASCO P-6500 instrument. Dinitriles 4a, 4b [5], 4c [6] and 4d [7,8] were prepared according to the published literature procedures.
9,14-Dicyanobenz[b]triphenylene (4a). Yield 91%, yellow solid; mp 244-246°C (acetic acid); IR cm-1: 2216 (CN); MS (EI): m/z (%): 328 (M+, 100), 299 (20), 164 (15), 150 (18), 136 (20); UV/vis (THF) λmax (log ε): 256 nm (4.6), 307 (4.6), 320 (4.4), 395 (4.0), 417 (3.9); fluorescence (THF, 325 nm): λmax,em: 487 nm.
7,12-Dicyanobenz[k]fluoranthene (4b). Yield 85%, light yellow solid; mp 357-359 °C (tetrachloro-ethane); Anal. calc. for C22H10N2: 87.4% C, 3.3% H, 9.3% N; found 87.2% C, 3.4% H, 9.4% N; IR cm−1: 2220 (CN); MS (EI): m/z (%): 302 (M+, 100), 273 (10), 151 (23), 123 (13), 111 (5), 28 (5); UV/vis (THF) λmax (log ε): 246 nm (3.7), 302 (3.3), 315 (3.6), 329 (3.8), 371 (4.1), 407 (3.8); fluorescence (THF, 363 nm): λmax,em: 440 nm.
9,10-Anthracenedicarbonitrile (4c). Yield 70%, yellow needles; mp 340-341°C (p-xylene); IR cm-1: 2215 (CN); MS (DEI): m/z (%): 228 (M+, 100), 201 (6), 174 (4); UV/vis (THF) λmax (log ε): 261 nm (4.5), 359 (3.6), 377 (4.0), 401 (4.0), 425 (4.0); fluorescence (THF, 333 nm): λmax,em: 458 nm; CV (DMF): ERED = -0.81V.
2,6-Dicyano-1,5-di-n-hexyloxynaphthalene (4d). Yield 60% (three steps), orange solid; mp 87°C (lit. [8] mp 88°C); IR cm-1: 2227 (CN); MS (DEI): m/z (%): 378 (M+, 10), 294 (27), 223 (18), 209 (100), 85 (20), 43 (84); UV/vis (THF) λ max (log ε): 285 nm (3.7), 295 (3.8), 308 (3.7), 347 (3.7), 363 (3.7), 440 (3.9), 469 (3.7); 1H-NMR (250 MHz, CDCl3) δ (ppm): 0.85 (t, 3J = 6.3 Hz, 6H), 1.28-1.51 (m, 12H), 1.87 (m, 4H), 4.40 (t, 3J = 6.3 Hz, 4H), 7.45 (d, 3J = 10.0 Hz, 2H), 7.65 (d, 3J = 10.0 Hz, 2H); fluorescence (THF, 470 nm): λmax,em: 511 nm.
General procedure for the synthesis of 4H-imidazoles 5:
Dinitrile 4 (1 mmol) was dissolved or suspended in dry THF (25 mL). After the addition of 1.0M Li-HMDS solution in THF or n-hexane (5 mL, 5 mmol), the reaction mixture was stirred at room temperature for 4 days and then evaporated to dryness in vacuo. The residue was dissolved in dry toluene (20 mL) and of chlorotrimethylsilane (0.75 mL, 6.0 mmol) was added. The reaction mixture was heated to 110 °C for 12 h and then the solvent was removed in vacuo. The silylated amidine was dissolved in dry THF (20 mL) and cyclized by addition of KF (0.35 g, 6.0 mmol), 18-crown-6-ether (1.58 g, 6.0 mmol) and the appropriate bis-imidoylchloride (1.5 mmol; Ar = 4-tol, 4-n-C4H9-C6H4-, 4-t-C4H9-C6H4- [11]), whereby the colour changed from yellow to deep red. The mixture was stirred for 3-4 hours at about 40 to 50 °C to ensure completion of the reaction. The 4H-imidazoles 5 were obtained by filtration and column chromatography of the crude product (SiO2, toluene/acetone 50:1).
9-{5-p-Tolylamino-4-[p-tolylimino]-4H-imidazol-2-yl}-benzo[b]triphenylene-14-carbonitrile (5a, Ar = 4-tolyl): Yield 21%, red solid, practically insoluble in all organic solvents tested; IR cm-1: 3283 (NH), 2210 (CN); MS (DEI): m/z (%): 577 (M+, 100), 486 (28), 459 (88), 326 (20), 215 (12), 106 (6), 91 (9); UV/vis (THF) λ max (log ε): 408 nm (4.2), 453 (4.2), 482 (4.3), 519 (4.1); CV: ERED1 = -0.42 V (CN), ERED2 = -0.72 V, ERED3 = -1.05 V.
9-{5-p-n-Butylphenylamino-4-[p-n-butylphenylimino]-4H-imidazol-2-yl}-benzo[b]triphenylene-14-carbonitrile (5a, Ar = 4-n-butylphenyl): Yield 28%, red solid; IR cm-1: 3480 (NH), 2210 (CN); MS (DEI): m/z (%): 661 (M+, 20), 351 (75), 175 (68), 106 (100); UV/vis (THF) λ max (log ε): 423 nm (4.1), 455 (4.1), 485 (4.2), 524 (4.0); 1H-NMR (250 MHz, THF-d8) δ (ppm): 0.89 (t, 6H), 1.55 (m, 4H), 2.56 (t, 4H), 7.31 (t, 2H), 7.52 (t, 2H), 7.71 (m, 6H), 7.88 (d, 3J = 8.8 Hz, 4H), 8.52 (d, 3J = 8.8 Hz, 2H), 8.61 (d, 3J = 8.8 Hz, 2H), 10.07 (s, 1H); CV: ERED1 = -0.47 V (CN), ERED2 = -0.85 V, ERED3 = -1.16 V.
9-{5-p-t-Butylphenylamino-4-[p-t-butylphenylimino]-4H-imidazol-2-yl}-benzo[b]triphenylene-14-carbonitrile (5a, Ar = 4-t-butylphenyl): Yield 30%, red solid; IR cm-1: 3338 (NH), 2212 (CN); MS (EI): m/z (%): 661 (M+, 10), 352 (60), 337 (95), 328 (12), 175 (46), 160 (100), 134 (92); UV/vis (THF) λ max (log ε): 211 nm (4.8), 298 (4.7), 386 (4.0), 481 (4.2), 521 (4.1); 1H-NMR (250 MHz, THF-d8) δ (ppm): 1.31 (s, 18H), 7.37 (d, 3J = 8.8 Hz, 4H), 7.3-7.4 (m, 6H), 7.76 (d, 3J = 8.8 Hz, 4H), 7.91 (d, 3J = 8.8 Hz, 2H), 8.52 (t, 2H), 8.60 (t, 2H), 10.12 (s, 1H); CV: ERED1 = -0.51 V (CN), ERED2 = -0.87 V, ERED3 = -1.17 V.
12-{5-p-n-Butylphenylamino-4-[p-n-butylphenylimino]-4H-imidazol-2-yl}-benzo[k]fluoranthene-7-carbonitrile (5b, Ar = 4-n-butylphenyl): Yield 15%, red solid; IR cm-1: 3298 (NH), 2224 (CN); MS (DEI): m/z (%): 635 (M+, 8), 578 (10), 508 (40), 352 (25), 306 (50), 263 (83), 175 (59), 132 (100), 106 (66); UV/vis (THF) λ max (log ε): 315 nm (4.2), 403 (4.3), 487 (4.4), 523 (4.3); 1H-NMR (250 MHz, THF-d8) δ (ppm): 0.95 (t, 6H), 1.35 (m, 4H), 1.58 (m, 4H), 2.62 (m, 4H), 6.98-7.33 (m, 14H), 7.73 (d, 3J = 8.3 Hz, 4H); 13C-NMR (100 MHz, THF-d8) δ (ppm): 13.8, 21.8, 31.5, 31.9, 34.8, 35.0, 101.4, 116.8, 117.6, 120.9, 121.2, 122.5, 125.5, 126.1, 126.3, 126.4, 126.8, 127.2 128.1, 128.2, 128.6, 129.3, 131.2, 132.0, 132.5, 132.8, 132.9, 133.1, 134.0, 135.4, 136.2, 138.4, 141.0, 148.0, 158.9; CV: ERED1 = -1.15 V, ERED2 = -1.48 V.
12-{5-p-t-Butylphenylamino-4-[p-t-butylphenylimino]-4H-imidazol-2-yl}-benzo[k]fluoranthene-7-carbonitrile (5b, Ar = 4-t-butylphenyl): Yield 18%, red solid; IR cm-1: 3315 (NH), 2221 (CN); MS (DEI): m/z (%): 635 (M+, 30), 578 (35), 477 (58), 352 (100), 337 (94), 302 (26), 160 (39), 132 (28); UV/vis (THF) λ max (log ε): 286 nm (4.2), 406 (4.2), 490 (4.4), 517 (4.3); 1H-NMR (250 MHz, THF-d8) δ (ppm): 1.28 (s, 18H), 7.07-7.18 (m, 6H), 7.36-7.40 (m, 8H), 7.74 (d, 3J = 8.3 Hz, 4H); 13C-NMR (100 MHz, THF-d8) δ (ppm): 31.4, 31.6, 34.8, 35.0, 101.4, 116.8, 117.6, 120.9, 121.2, 122.5, 125.5, 126.1, 126.3, 126.4, 126.8, 127.2 128.1, 128.2, 128.6, 129.3, 131.2, 132.0, 132.5, 132.8, 132.9, 133.1, 134.0, 135.4, 136.2, 138.4, 141.0, 148.0, 158.9; CV: ERED1 = -1.02 V, ERED2 = -1.50 V.
10-{5-p-Tolylamino-4-[p-tolylimino]-4H-imidazol-2-yl}-anthracene-9-carbonitrile (5c, Ar = 4-tolyl): Yield 19%, red solid; IR cm-1: 3545 (NH), 2222 (CN); MS (DEI): m/z (%): 477 (M+, 100), 462 (10), 359 (4), 228 (10), 221 (2), 106 (2); UV/vis (THF) λmax (log ε): 390 nm (4.1), 410 (4.1), 452 (4.2), 480 (4.3), 520 (4.2); 1H-NMR could not be recorded (almost insoluble in organic solvents); CV: ERED1 = -0.44 V (CN), ERED2 = -0.84 V, ERED3 = -1.12 V.
10-{5-p-n-Butylphenylamino-4-[p-n-butylphenylimino]-4H-imidazol-2-yl}-anthracene-9-carbonitrile (5c, Ar = 4-n-butylphenyl): Yield 27%, red solid; IR cm-1: 3257 (NH), 2221 (CN); MS (DEI): m/z (%): 561 (M+, 100), 504 (68), 402 (20), 228 (10); UV/vis (THF) λmax (log ε): 393 nm (4.1), 411 (4.2), 455 (4.2), 482 (4.3), 525 (4.2); 1H-NMR (250 MHz, CD2Cl2) δ (ppm): 0.82 (t, 3J = 7.5 Hz, 6H), 1.24 (m, 4H), 1.47 (m, 4H), 2.48 (q, 3J = 7.5 Hz, 4H), 7.12 (d, 3J = 8.3 Hz, 4H), 7.52 (d, 3J = 7.8 Hz, 2H), 7.68 (d, 3J = 7.8 Hz, 2H), 7.77 (d, 3J = 8.3 Hz, 4H), 8.42 (t, 3J = 7.3 Hz, 2H), 8.52 (t, 3J = 7.3 Hz, 2H); CV: ERED1 = -0.44 V (CN), ERED2 = -0.75 V, ERED3 = -1.12 V.
10-{5-p-t-Butylphenylamino-4-[p-t-butylphenylimino]-4H-imidazol-2-yl}-anthracene-9-carbonitrile (5c, 4-t-butylphenyl): Yield 29%, red solid; IR cm-1: ▯3456 (NH), 2220 (CN); MS (DEI): m/z (%): 561 (M+, 8), 504 (1), 436 (3), 379 (4), 351 (100), 336 (19), 296 (16), 175 (21), 160 (21), 134 (17), 91 (23); UV/vis (THF) λmax (log ε): 453 nm (4.3), 484 (4.3), 526 (4.1); 1H-NMR (250 MHz, THF-d8) δ (ppm): 1.30 (s, 18H), 7.36 (d, 3J = 8.5 Hz, 2H), 7.41 (d, 3J = 8.5 Hz, 4H), 7.61 (t, 2H), 7.69 (d, 3J = 8.5 Hz, 2H), 7.80 (t, 2H), 7.99 (d, 3J = 8.5 Hz, 4H), 10.01 (s, 1H); CV: ERED1 = -0.47 V (CN), ERED2 = -0.79 V, ERED3 = -1.13 V.
1,5-Di-n-hexyloxy-6-{5-p-tolylamino-4-[p-tolylimino]-4H-imidazol-2-yl}-naphthalene-2-carbonitrile (5d, Ar = 4-tolyl): Yield 20%, deep red solid; IR cm-1: 3278 (NH), 2227 (CN); MS (DEI): m/z (%): 627 (M+, 16), 268 (100), 205 (20), 177 (18), 133 (25), 106 (72); UV/vis (THF) λmax (log ε): 345 nm (4.2), 440 (4.4), 500 (4.3), 536 (4.1); 1H-NMR (250 MHz, THF-d8) δ (ppm): 0.89 (t, 6H), 1.29 (m, 8H), 1.40 (m, 4H), 1.96 (m, 4H), 2.37 (s, 6H), 4.46 (t, 4H), 7.24 (d, 3J = 8.8 Hz, 4H), 7.60 (d, 3J = 8.8 Hz, 1H), 7.70 (d, 3J = 8.8 Hz, 2H), 7.98 (d, 3J = 8.8 Hz, 4H), 8.70 (d, 3J = 8.8 Hz, 1H); CV: ERED1 = -0.90 V, ERED2 = -1.31 V.
1,5-Di-n-hexyloxy-6-{5-p-n-butylphenylamino-4-[p-n-butylphenylimino]-4H-imidazol-2-yl}-naphtha-lene-2-carbonitrile (5d, Ar = 4-n-butylphenyl): Yield 27%, red solid; IR cm-1: 3296 (NH), 2227 (CN); MS (DEI): m/z (%): 711 (M+, 20), 483 (24), 378 (28), 352 (20), 210 (100), 149 (10), 106 (21); UV/vis (THF) λmax (log ε): 345 nm (4.2), 415 (4.3), 503 (4.3), 540 (4.2); 1H-NMR (250 MHz, THF-d8) δ (ppm): 0.93 (t, 3J = 7.0 Hz, 12H), 1.29 (m, 4H), 1.40 (m, 8H), 1.60 (m, 8H), 1.95 (m, 4H), 2.59 (t, 3J = 7.5 Hz, 4H), 4.46 (t, 3J = 6.5 Hz, 4H), 7.15 (d, 3J = 8.5 Hz, 2H), 7.69 (m, 6H), 8.02 (d, 3J = 8.5 Hz, 4H), 10.08 (s, broad, 1H); CV: ERED1 = -0.94 V, ERED2 = -1.39 V.
Boratetraazapentalenes 9 were prepared according to literature [2].
9-[2-Difluoro-1,3-bis-(4-t-butylphenyl)-1,2-dihydro-1,3,4,6-tetraaza-2-borapentalen-5-yl]-benzo[b]-triphenylene-14-carbonitrile (9a, Ar = 4-t-butylphenyl): Yield 52%, dark violet solid; IR cm-1: 2212 (CN); MS (DEI): m/z (%): 709 (M+, 18), 661 (6), 488 (24), 462 (26), 328 (100), 253 (14); UV/vis (THF) λmax (log ε): 372 nm (4.1), 529 (4.2), 572 (4.2), 614 (4.2); 1H-NMR (400 MHz, THF-d8) δ (ppm): 1.33 (s, 18H), 7.42 (d, 3J = 8.0 Hz, 4H), 7.40-7.65 (m, 6H), 7.85 (d, 3J = 8.0 Hz, 4H), 8.02 (d, 3J = 8.0 Hz, 2H), 8.62 (t, 2H), 8.67 (t, 2H); Fluorescence (THF, 475 nm); λmax,em: 681 nm; CV: ERED1 = -0.48 V, ERED2 = -1.16 V, KSEM = 3.4*1011.
9-[2-Difluoro-1,3-bis-(4-n-butylphenyl)-1,2-dihydro-1,3,4,6-tetraaza-2-borapentalen-5-yl]-benzo[b]-triphenylene-14-carbonitrile (9a, Ar = 4-n-butylphenyl): Yield 54%, dark violet solid; IR cm-1: 2209 (CN); MS (DEI): m/z (%): 709 (M+, 20), 661 (2), 451 (28), 369 (20), 328 (56), 221 (24), 111 (23), 97 (34), 85 (64), 71 (87), 57 (100); UV/vis (THF) λmax (log ε): 375 nm (4.1), 531 (4.1), 573 (4.3), 616 (4.2); 1H-NMR (400 MHz, THF-d8) δ (ppm): 0.97 (t, 6H), 1.38 (m, 4H), 1.62 (m, 4H), 2.66 (m, 4H), 7.02-7.39 (m, 14H), 7.79 (d, 3J = 8.0 Hz, 4H); Fluorescence (THF, 441 nm): λmax,em: 699 nm, (toluene, 440nm): λmax,em: 706 nm; CV: ERED1 = -0.45 V, ERED2 = -1.14 V, KSEM = 5.0*1011.
12-[2-Difluoro-1,3-bis-(4-t-butylphenyl)-1,2-dihydro-1,3,4,6-tetraaza-2-borapentalen-5-yl]-benzo[k]-fluoranthene-7-carbonitrile (9b, Ar = 4-t-butylphenyl): Yield 48%, blue-violet solid; IR cm-1: 2220 (CN); MS (DEI): m/z (%): 683 (M+, 18), 635 (36), 578 (64), 302 (100), 134 (20), 91 (25), 57 (48); UV/vis (THF) λmax (log ε): 407 nm (4.3), 524 (4.4), 571 (4.3), 608 (4.1); 1H-NMR (400 MHz, THF-d8) δ (ppm): 1.31 (s, 18H), 7.10-7.22 (m, 6H), 7.38-7.43 (m, 8H), 7.76 (d, J = 8.0Hz, 4H); Fluorescence (toluene, 405 nm): λmax,em: 885 nm; CV: ERED1 = -0.88 V, ERED2 = -1.50 V, KSEM = 3.2*1010.
10-[2-Difluoro-1,3-bis-(4-t-butylphenyl)-1,2-dihydro-1,3,4,6-tetraaza-2-borapentalen-5-yl]-anthra-cene-9-carbonitrile (9c, Ar = 4-t-butylphenyl): Yield 40%, dark violet solid; IR cm-1: 2219 (CN); MS (EI): m/z (%): 609 (M+, 20), 561 (84), 552 (100), 504 (78), 228 (69), 134 (71); UV/vis (THF) λmax (log ε): 388 nm (4.1), 403 (4.1), 528 (4.2), 567 (4.2), 602 (4.2); 1H-NMR (400 MHz, THF-d8) δ (ppm): 1.30 (s, 18H), 7.41 (t, 2H), 7.54 (d, 3J = 8.0 Hz, 4H), 7.76 (t, 2H), 7.80-7.95 (m, 4H), 8.08 (d, 3J = 8.0 Hz, 4H); Fluorescence (THF, 462 nm): λmax,em: 676 nm; CV: ERED1 = -0.42 V, ERED2 = -1.05 V, KSEM = 4.8*1010.
1,5-Di-n-hexyloxy-6-[2-Difluoro-1,3-bis-(4-tolyl)-1,2-dihydro-1,3,4,6-tetraaza-2-borapentalen-5-yl]-naphthalene-2-carbonitrile (9d, Ar = 4-tolyl): Yield 52%, dark violet solid; IR cm-1: 2227 (CN); MS (DEI): m/z (%): 678 (M+, 21), 627 (38), 268 (100), 205 (20), 177 (18), 133 (25), 106 (72); UV/vis (THF) λmax (log ε): 352 nm (4.1), 511 (4.2), 553 (4.3), 596 (4.2); 1H-NMR (400 MHz, THF-d8) δ (ppm): 0.91 (t, 6H), 1.32 (m, 8H), 1.44 (m, 4H), 1.99 (m, 4H), 2.42 (s, 6H), 4.50 (t, 4H), 7.36 (d, 3J = 8.0 Hz, 4H), 7.81 (d, 3J = 8.0 Hz, 1H), 7.93 (d, 3J = 8.0 Hz, 2H), 8.18 (d, 3J = 8.0 Hz, 4H), 8.90 (d, 3J = 8.0 Hz, 1H); Fluorescence (THF, 394 nm): λmax,em: 870 nm; CV: ERED1 = -0.82 V, ERED2 = -1.47 V, KSEM = 1.0*1011.
References
- Fabian, J.; Beckert, R.; Atzrodt, J. Fünfring Cycloamidine – Neue farbige Heterocyclen mit ungewöhnlichen Eigenschaften. J. Prakt. Chem./Chem.-Ztg. 1997, 339, 729–741. [Google Scholar] [CrossRef]
- Gebauer, T.; Beckert, R.; Weiß, D.; Knop, K.; Käpplinger, C.; Görls, H. Mesoionic boratetraaza-pentalenes – fully reversible two step redox systems. Chem. Comm. 2004, 16, 1860–1861. [Google Scholar]
- Matschke, M.; Käpplinger, C.; Beckert, R. Bis-4H-imidazoles – tetraazafulvalenes – 2,2’-bi-imidazoles: three variations of one redox system. Tetrahedron 2006, 62, 8586–8590. [Google Scholar]
- Kühn, C.; Beckert, R.; Grummt, U.-W.; Käpplinger, C.; Birckner, E. 1,4,5,8-Tetraazafulvalene – Darstellung schwefelhaltiger Derivate und Zuordnung des Chromophors. Z. Naturforsch. 2004, 59b, 406–413. [Google Scholar]
- Hinsberg, O. Synthetische Versuche mit o-Xylylendicyanid. Chem. Ber. 1910, 43, 1360–1363. [Google Scholar]
- Rio, G.; Sillion, B. Reactions of substitution, 1,6-addition, and alkylation of substituted 9-Cyano-anthracenes. Bull. Soc. Chim. France 1961, 831–834. [Google Scholar]
- Wheeler, A.S.; Ergle, D.R. Naphthol studies – Bromination of 1,5-Dihydroxynahthalene. J. Am. Chem. Soc. 1930, 52, 4872–4878. [Google Scholar]
- Segura, J.L.; Nazzario, M.; Hanack, M. Oligo-2,6-naphthylenevinylenes – New building blocks for the preparation of photoluminescent polymeric materials. Eur. J. Org. Chem. 1999, 3, 643–652. [Google Scholar]
- Boere, R.T.; Oakley, R.T.; Reed, R.W. Preparaion of N,N,N-tris(trimethylsilyl)amidines; a convenient route to unsubstituted amidines. J. Organomet. Chem. 1987, 331, 161–167. [Google Scholar]
- Matschke, M.; Käpplinger, C.; Weiß, D.; Beckert, R. The redox behaviour of cyclic tetra-aminoethenes derived from 2,2’-biimidazole. Tetrahedron Lett. 2005, 46, 8249–8251. [Google Scholar]
- Lindauer, D.; Beckert, R.; Döring, M.; Feeling, P.; Görls, H. Zur Aminolyse von Bis-imidoyl-chloriden der Oxalsäure. J. Prakt. Chem. 1995, 337, 143–152. [Google Scholar]
- Sample availability: Most of the described substances are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.