- Article
Melatonin Receptor 1 and Melatonin Receptor 2 Expression During Human Kidney Development and Their Association with CAKUT
- Ann-Kathrin Schmitt,
- Victoria Tjora and
- Anita Racetin
- + 6 authors
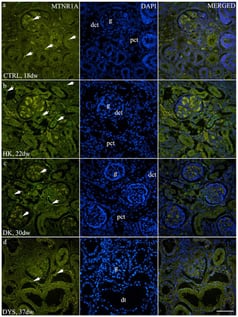
Background/Objectives: Growing evidence indicates that melatonin contributes to kidney development and function, while disruptions of fetal circadian signaling have been linked to congenital anomalies of the kidney and urinary tract (CAKUT). This study aimed to characterize the developmental and spatial expression patterns of melatonin receptors MTNR1A and MTNR1B in normal human fetal kidneys and in CAKUT phenotypes. Methods: This study analyzed 40 human fetal kidney specimens, including healthy controls and CAKUT cases (horseshoe kidneys, duplex kidneys, and dysplastic kidneys), obtained from spontaneous abortions and pregnancy terminations. Samples were classified into developmental phases Ph2–Ph4 according to established morphological criteria. Immunofluorescence staining was used to visualize MTNR1A and MTNR1B expression. Quantitative analysis was performed using ImageJ, measuring the fluorescence area percentage. Statistical comparisons were conducted using a two-way ANOVA. Results: In control kidneys, MTNR1A expression was predominantly observed in glomeruli and interstitial cells and showed a descending trend across developmental stages, whereas MTNR1B was localized to glomeruli and strongly to the apical membranes of tubules, particularly distal tubules, without substantial developmental variation. CAKUT phenotypes exhibited higher expression of both receptors compared to controls. Significant phase-dependent differences in MTNR1A expression were observed in horseshoe, duplex, and dysplastic kidneys. MTNR1B expression decreased across developmental stages in dysplastic kidneys and differed significantly between Ph3 and Ph4 in duplex kidneys. At Ph3, duplex kidneys showed the highest MTNR1B expression. Conclusions: Altered developmental expression patterns of MTNR1A and MTNR1B in CAKUT suggest an association between melatonin signaling and abnormal human kidney development.
15 April 2026