Abstract
Background/Objectives: Stereotactic Body Radiation Therapy (SBRT) is increasingly used for localized prostate cancer (PCa), but evidence supporting its use in high-risk PCa (HRPC) remains limited. Standard management continues to favor conventional or moderately hypofractionated radiotherapy combined with long-course androgen deprivation therapy (ADT). This systematic review aimed to synthesize current data on SBRT biochemical outcomes, toxicity, and technical aspects in localized HRPC. Methods: A systematic PubMed search was conducted on 1 May 2024, following PRISMA 2020 guidelines (PROSPERO ID CRD420251235649). Studies reporting biochemical control (BC) for HRPC treated definitively with SBRT, with or without ADT, were included. Studies not meeting these criteria or including ≤10 HRPC patients were excluded. Risk of bias was assessed through qualitative appraisal of study methodology. Substantial heterogeneity across study design, SBRT schedules, cohort composition, and ADT integration precluded a meta-analysis; data were synthesized descriptively. Results: Thirty studies contributed biochemical control data after prostate SBRT for 1354 patients meeting inclusion criteria. SBRT was delivered using diverse platforms and dose-fractionation schemes, frequently in combination with ADT. Across studies, BC was generally favorable, though follow-up duration varied widely. Toxicity profiles were acceptable, with most reports describing predominantly grade 1–2 events and low rates of severe toxicity. Marked variability was observed in target volume definition, focal-boost strategies, urethra-sparing techniques, and the use of rectal spacers. Conclusions: Although current evidence is heterogeneous and largely derived from non-randomized studies, BC and toxicity outcomes are consistently promising, supporting SBRT as a potentially effective strategy for localized HRPC. Randomized prospective trials are needed to confirm these findings and refine optimal SBRT regimens and the role of ADT. This review received no funding.
1. Introduction
Prostate cancer (PCa) is the second most common malignancy and the fifth leading cause of cancer-related mortality in men worldwide [1]. The majority of patients with PCa are diagnosed with localized disease [2] and are stratified into risk categories that guide treatment decisions [3]. Definitive Radiation Therapy (RT), typically combined with long-course androgen deprivation therapy (ADT), is one of the curative treatment options for localized high-risk PCa (HRPC) [3]. RT can be administered using either conventional or moderately hypofractionated regimens [4].
Compared to conventional fractionation, hypofractionated RT delivers a higher dose per fraction, allowing for a reduction in total treatment time, increased patient comfort, and lower healthcare costs [5,6,7,8]. It may also provide a theoretical therapeutic advantage in terms of tumor control and toxicity, considering the lower α/β of PCa compared to surrounding healthy tissues [9,10,11].
In recent years, the development of image-guided RT (IGRT) and Intensity-Modulated RT (IMRT), along with the introduction of stereotactic techniques, has enabled the clinical implementation of Stereotactic Body Radiation Therapy (SBRT) [12,13,14]. SBRT delivers extremely hypofractionated radiation schedules with high precision and conformality [15,16]. The safety and efficacy of SBRT in the definitive management of localized PCa have been demonstrated in the PACE-B and HYPO-RT-PC trials [17,18]. These randomized phase III trials compared SBRT with standard fractionation, showing similar tumor control, low toxicity rates, and a favorable patient-reported Quality of Life (QoL) profile. However, these results primarily reflect the outcomes of patients with low- and intermediate-risk PCa, as PACE-B did not enroll HRPC patients and HYPO-RT-PC included only 11% of high-risk cases. Considering the paucity of randomized data on SBRT for HRPC and the evolving landscape of RT towards hypofractionation in this setting, the objective of this systematic review was to summarize the available evidence on SBRT for HRPC, with a primary focus on biochemical control, and to describe toxicity outcomes and technical/planning considerations when reported.
2. Materials and Methods
We conducted a systematic review of the literature on SBRT for the definitive treatment of localized HRPC, following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 guidelines(Figure 1; Supplementary File S1). This review protocol was registered in PROSPERO (ID: CRD420251235649) and no amendments were made after registration.
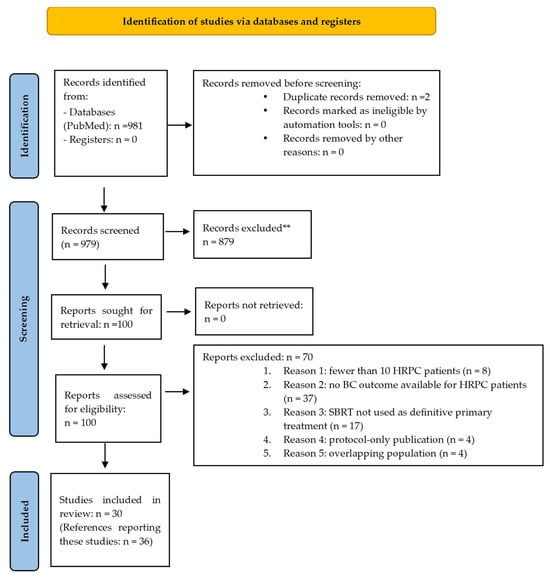
Figure 1.
PRISMA flow diagram. **: Reviews or meta-analyses; study protocols without patient data; editorials, letters, and case reports; conference abstracts; non-English articles; not prostate SBRT; not organ-confined disease; HRPC patients not included; dosimetric/planning-only studies.
2.1. Search Strategy
A systematic literature search was performed using the PubMed database for articles published in English from 1 January 2000 to 1 May 2024, based on a predefined combination of keywords: prostate cancer, prostatic neoplasm, primary, localized, stereotactic body radiotherapy, and SBRT. These terms were combined using a predefined search query without MeSH expansion or additional filters or limits. The final search was conducted on 1 May 2024. PubMed was selected as the sole database due to resource optimization considerations and the expected overlap with Embase and the Cochrane Library. Given the clinical focus of the present review and the well-indexed nature of SBRT prostate cancer studies in PubMed, this approach was considered sufficient to address the study objectives. The exact PubMed search used was “(prostate cancer OR prostatic neoplasm) AND (primary OR localized) AND (stereotactic body radiotherapy OR SBRT)”. Reference lists of included studies were not systematically searched.
The keywords and search results are summarized in Table 1.

Table 1.
Summary of keywords and search results in PubMed.
2.2. Eligibility Criteria
We initially identified 981 studies in the PubMed database. The inclusion criteria comprised original articles that reported on Biochemical Control (BC) for localized HRPC, defined according to D’Amico or NCCN criteria [19,20], treated primarily with SBRT, with or without androgen deprivation therapy (ADT). Studies that also included very-high-risk PCa (VHRPC) patients were eligible as long as BC outcomes were reported for the HRPC subgroup. VHRPC outcomes were extracted and presented when available but were not required for inclusion. Studies including fewer than 10 HRPC patients or exclusively other treatment modalities than SBRT (i.e., SBRT boost, IMRT, others) were excluded. Case reports, letters, editorials, congress abstracts, newspaper articles, reviews, and meta-analyses were excluded. The exclusion criteria included studies where HRPC patients were not included or where BC was not specifically reported for them, node-positive or metastatic prostate cancer, recurrent disease, or SBRT primarily used as a boost or in combination with other radiation techniques. For the purpose of data synthesis, the studies were grouped by study design for efficacy outcomes, while toxicity and technical parameters were synthesized descriptively. All outcomes and variables were predefined in accordance with the review protocol.
2.3. Study Selection
For article selection, two reviewers independently screened all titles and abstracts without the use of automation tools to identify studies reporting on BC for high-risk patients. To minimize the risk of omitting relevant studies, abstracts that did not clearly specify the inclusion of HRPC or the reporting of the outcome of interest, were labeled as “potentially eligible” and screened in full text. A consensus meeting was conducted to resolve discrepancies and finalize study selection. After removing duplicates and excluding articles not relevant to the scope of the review, 30 studies were deemed eligible and reviewed in full for data collection. When multiple publications referred to the same cohort, only the most recent or comprehensive report was used for data extraction, and the study was counted once in the final number. Earlier reports were not analyzed separately, although multiple references may appear within a single entry to indicate continuity of reporting. When separate trials were later reported together in a pooled publication that also preserved trial-specific results, the pooled publication was retained for data extraction and cited alongside the original reports. When the population of smaller trials was entirely incorporated into a larger pooled analysis based on individual patient data, only the pooled analysis was included to avoid overlapping populations. Such pooled analysis could integrate additional prospective cohorts not published separately. In rare cases of suspected but unconfirmed cohort overlap, studies were retained if they met inclusion criteria and reported non-identical data.
2.4. Data Extraction
Two reviewers extracted all data independently and in duplicate, without the use of automation tools. Extracted data included BC and all the available information on other efficacy outcomes such as Local Failure (LF), regional failure, Distant Metastasis (DM), Distant Metastasis-Free Survival (DMFS), Cancer-Specific Survival (CSS), Overall Survival (OS), patterns of failure, acute and late toxicity rates and scales, dosimetric and technical aspects of SBRT, as well as ADT use and specifications. When a publication referred to an SBRT protocol previously described elsewhere, we extracted the corresponding technical details from the cited reports and indicated this accordingly. All outcomes and variables were collected exactly as defined and reported by the authors for each study, including all available time points without restrictions nor applying standardized definitions or selecting preferred measures. No assumptions were made for missing or unclear data, except for the calculation of percentages when exact proportions were provided and all information was recorded exactly as reported by each study. Information on study funding or conflicts of interest was not collected.
2.5. Risk of Bias
The risk of bias was assessed independently by two reviewers through qualitative appraisal of each study’s methodology and reporting, without the use of automation tools or formal risk-of-bias instruments, as comparator arms and preregistered protocols were generally absent among the included studies. The appraisal considered domains relevant to observational designs, including cohort definition, inclusion criteria, outcome definitions, reporting consistency, potential confounding such as variations in SBRT regimens, ADT use, and technical parameters, the duration and adequacy of follow-up, and the extent of outcome reporting. No specific assessment of reporting bias was performed due to the observational and predominantly non-comparative nature of the included studies.
2.6. Statistical Analyses and Synthesis Methods
HRPC classification was accepted as defined in each individual study, using established clinical risk stratification criteria, including D’Amico or NCCN classifications [19,20]. No reclassification or harmonization across definitions was performed, and data were synthesized as reported by the original authors. The data are presented using descriptive statistics. Medians were calculated for variables for which numerical aggregation was clinically and methodologically appropriate (absolute number and percentage of HRPC patients, and EQD2), whereas other outcomes were summarized using reported percentages due to heterogeneity in follow-up, study design, and reporting time points. EQD2 values were calculated when not explicitly reported, assuming an α/β of 1.5 Gy. Crude proportions were used as the effect measure for all efficacy and toxicity outcomes. Heterogeneity was assessed qualitatively by comparing study design, patient characteristics, SBRT regimens, ADT use, toxicity grading systems, follow-up time points and planning and delivery parameters across studies. No additional statistical analyses were performed because the substantial methodological and clinical heterogeneity across the included studies precluded a meaningful quantitative synthesis or meta-analysis. No methods were applied to explore heterogeneity due to the descriptive nature of the synthesis, and no data conversions, subgroup analyses, or sensitivity analyses were performed. Results were tabulated and visually summarized across the main tables.
2.7. Certainty of Evidence
Certainty of evidence was not assessed due to non-comparative nature and methodological heterogeneity of the included studies.
3. Results
3.1. Study, Patients, and Treatment Characteristics
We identified 30 studies reporting on BC after prostate SBRT for 1354 patients meeting the inclusion criteria (results summarized in Table 2). All included studies were published between 2011 and 2024 and comprised one phase 1a–b trial, seven phase 2 trials, eight prospective studies (seven single-institution and one multi-institutional), two retrospective analyses of prospectively collected individual patient data collected from phase 2 studies and institutional registries, and twelve retrospective studies (nine single-institution, two multi-institutional, and one based on a large clinical database). The study selection process, including full-text exclusions (n = 70) and the reasons for exclusion, are presented in the PRISMA flow diagram (Figure 1).

Table 2.
Summary of the studies.
Most studies included a mixture of disease risk groups, with only two studies exclusively enrolling high-risk patients (n = 344 and n = 59) [44,52,53,54]. The proportion and absolute number HRPC patients varied considerably across studies, with a median of 42.55 patients (range, 12–344) and median percentage of 41 (range, 6.1–100%). The proportion of VHRPC patients was reported in only four studies, refs. [27,32,37,45] where they comprised a minority of the cohort (range, 2–26 patients and 4.6–19.5%).
Different fractionation schedules were used, with a median prescribed EQD2 dose of 90.6 Gy (range, 46.43–135 Gy). Focal dose escalation strategies targeting the prostate were reported in five studies [35,38,46,49,55]. Four of these [35,46,49,55] used a simultaneous integrated boost (SIB) to a dominant intraprostatic lesion (DIL) identified on MRI with or without PET/CT PSMA, delivering prescribed EQD2 doses ranging from 135 to 164.3 Gy. Two studies implemented an alternative approach by defining an intraprostatic high-dose PTV, cropped at 3 mm from the urethra, bladder, and rectum, which received either a D50% of at least 164.3 Gy EQD2 or a prescription dose of 50 Gy in five fractions [31,38]. Elective pelvic irradiation was administered in only four studies [37,43,44,52], with 18 to 66 patients per trial, all receiving a concomitant dose of 25 Gy in five fractions (EQD2 46.4 Gy). The most common treatment delivery schedule was daily consecutive fractions (eight studies, 26.7%), while seven studies (23.3%) allowed either consecutive or alternate-day regimens. Six studies (20%) used exclusively alternate-day treatment. Other schedules included once- or twice-weekly fractions, a sandwich strategy with a 2-day break, and variable intervals of at least 40 h or treatment every 2 to 6 days, with a maximum of two fractions a week. Treatment frequency was not reported in two studies [26,32].
Androgen deprivation therapy (ADT) was allowed per the protocol in 28 studies (93.3%) [21,22,23,24,26,27,28,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,55], though only 12 reported the proportion of high-risk patients who received it, ranging from 0% to 100% [21,23,26,33,35,39,42,44,46,48,52,55]. Five studies administered ADT to all HRPC patients, refs. [21,42,48,52,55], while in one study, despite protocol allowance, all high-risk subgroup declined hormonal treatment [35]. Data on ADT duration among HRPC patients were available in eight studies [21,23,39,42,44,48,52,55] and showed substantial variation. Reported durations ranged from median suppression periods of 4 to 9 months, refs. [23,42,44,55], fewer specific intervals spanning from under 7 months to over 24 months or from 26 months to over 26 months, refs. [21,39], as well as fixed treatment durations of 12, 18, or 42 months [48,52].
Individual study results for efficacy and toxicity outcomes are presented in Table 2 and Table 3 using the information provided in the original publications. Although biochemical control was consistently defined, efficacy outcomes were reported at different follow-up time points, and confidence intervals were not consistently provided. Toxicity reporting was heterogeneous across studies in terms of grading systems and the extent of acute and late toxicity assessments. Consequently, standardized effect estimates could not be calculated, and results are summarized narratively based on the available data.

Table 3.
Acute and late toxicity.
3.2. Efficacy
3.2.1. Biochemical Control
The BC rates for HRPC were reported mainly as subgroup analyses across different studies. Overall, BC outcomes varied widely according to study design, risk stratification, and follow-up (FUP) duration. All studies used the Phoenix definition to define biochemical failure [21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57].
Retrospective studies demonstrated a broad range of BC rates, from 66% to 100%, refs. [33,45], with several studies including long-term outcomes [21,23,24,25,26,32,33,34,39,40,41,42,44,45,51]. Among those with follow-up periods of 5 years or more, BC rates ranged from 74.1% to 95.2% [23,32,34,39,51]. In line with these findings, the only retrospective study exclusively including HRPC patients used individual patient data collected prospectively from institutional registries and phase 2 trials, and reported a BC rate of 81.7% at 4 years [44]. Subgroup analyses separating high-risk and very-high-risk groups were reported in two studies, revealing lower BC very-high-risk patients (86.3% vs. 61.6% at 5 years or 2/48 vs. 2/12 at a median follow-up of 45.53 months) [32,45], while another study which reported outcomes for patients with Gleason Score ≥ 8 showed a BC rate of 66.7% at 64 months [39].
Among prospective trials, one trial reported a BC rate of 100% at 13 months, with ADT planned for a duration of 18 months when administered to this subgroup [43]. Across four additional studies with follow-up periods of 2.5 to 3 years [22,27,38,46], one of which included mixed HRPC and VHRPC outcomes [27], BC ranged from 82% to 94%. Only two prospective studies reported long-term results, showing a BC of 72.8% at 7 years or a bDFS of 65% at 8 years [28,47].
Similarly, phase 2 studies demonstrated early BC rates of 100% in two studies at 2 [50] and 2.5 years [55]. Consistent with prospective and retrospective studies with medium-term follow-up, two trials showed a BC of 78.3% and 90% at 3 and 4 years, respectively [37,48]. Long-term outcomes were available in two studies: one reported PSA-control of 96.7% at 5 years for high-risk and unfavorable intermediate-risk (UIR) [36], while another showed an overall BC of 69.7%, with 40% of failures involving patients with stage T3a/b and/or Gleason Score ≥ 8 [49]. The only phase 2 study including exclusively HRPC patients reported BC rates of 68.8% and 74.2% at 5 years in both arms [49].
3.2.2. Distant Metastasis-Free Survival, Overall Survival, and Cancer-Specific Survival
Among studies reporting on DM, those with shorter follow-up periods showed control rates ranging from 97.3% at 15 months to 100% at 3 years [27,37,42]. Longer-term follow-up at 5 years demonstrated slightly lower DMFS rates, ranging from 87.9% to 93.3% [51,52]. Notably, one phase II study reported a cumulative rate of DM of 2.7% at 5 years, although it included both UIR and HRPC patients [36].
Regarding survival, two retrospective analyses showed OS rates ranging from 88.2% to 100% at 5 years [39,51], while three phase II trials documented rates between 100% at 2 years and 75–87.9% at 5 years [37,50,52]. Similarly, CSS ranged from 87.5% to 100% at 5 years [21,52].
3.3. Local Control and Patterns of Failure
Three studies reported on LC and failure patterns in HRPC patients [27,36,38]. Fan et al. reported 100% LC at 3 years [27]. In the study by Parsai et al., one HRPC patient (5.5%) developed isolated prostatic recurrence at 4 years, while two others (11.1%) experienced bone metastases without local relapse [38]. Similarly, Macias et al. [36] noted that among the three individuals from the UIR and HRPC cohort who recurred at 5 years, LC remained 100%. The observed failure patterns included bone metastases, either alone or in combination nodal relapse (two HRPC patients and one with unclassifiable risk, respectively) [36].
3.4. Toxicity
Toxicity was reported in 26 studies (86.7%), either using RTOG (n = 9), RTOG/EORTC (n = 1), CTCAE scales (n = 15, including trials that reported as NCI-CTC, NCI-CTC-AE or NCIC-CTC, versions 3–5), or a combination of both (n = 1) [21,22,24,26,27,28,31,32,33,34,35,36,37,38,41,42,43,44,45,46,47,48,49,50,51,52,55]. One study utilized an institutional scale for self-reported toxicity [46]. Data are summarized in Table 3.
Most patients experienced Grade (G) 1 or G2 toxicities, in both acute and late settings [21,22,24,26,27,28,29,30,31,32,33,34,35,36,37,38,41,42,43,44,45,47,48,49,50,51,52,53,54,55]. Grade 3 genitourinary (GU) toxicity was infrequent, reported acutely in four studies (0.8% to 2%) and late in eight trials (0.2% to 2%) [22,24,28,33,36,41,44,45,47,48,51,55]. Severe gastrointestinal (GI) events were generally rare, with no studies documenting acute Grade 3 side effects and seven trials reporting low incidence in the late setting (0.5% to 2.9%) [33,34,38,41,44,48,51].
An exception to these findings was observed by Tang et al., who reported only late toxicity outcomes and noting substantially higher rates of Grade 3 or higher events in the cohort receiving pelvic irradiation, with rates reaching 25% for GU and 31.3% for GI toxicity [52]. Only one study recorded Grade 4 acute and late events in both GI and GU domains (n = 1 patient, 2.9% for each domain) [38]. No Grade 5 acute or late toxicities were reported.
A single narrative synthesis encompassing all efficacy and toxicity outcomes was performed. Across the included studies, efficacy and toxicity outcomes were described with generally similar directional patterns, with high rates of BC and limited severe toxicity reported. No quantitative meta-analysis was conducted for this synthesis because the included studies showed substantial clinical and methodological heterogeneity, including differences in design, risk classification, SBRT regimens, target volumes, focal-boost strategies, androgen deprivation therapy use, follow-up duration, and toxicity reporting. For the same reasons, no formal assessments of statistical heterogeneity or sensitivity analyses were conducted.
3.5. Radiotherapy Planning and Delivery
Radiotherapy was delivered using CyberKnife® systems (n = 14) [21,22,23,24,25,27,28,32,34,35,39,42,45,55], LINAC-based platforms (n = 10) [31,33,37,38,46,47,49,50,51,52], TomoTherapy® units (n = 2) [36,48], MR-LINAC (n = 1) [43], or a combination of modalities (n = 3) [26,41,44]. Image-guided radiotherapy (IGRT) was employed in all studies, most commonly utilizing orthogonal X-ray imaging [21,22,23,24,25,27,28,32,34,35,39,42,44,45,49,55] or cone-beam CT (CBCT) [31,33,37,38,44,47,50,52]. Fiducial markers were implanted in 23 studies (76.7%) [21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,38,39,40,41,42,43,44,45,46,47,48,51,52,53,54,55].
Other devices were applied less frequently and included hydrogel spacers (n = 3; at least two studies used SpaceOAR™) [42,43,46], biodegradable rectal spacers (n = 2; BioProtect Balloon Implant™ System) [35,49], rectal balloons (n = 2) [36,48], and bladder catheters (n = 5; one employing the Calypso® system and Soft Tissue and Surface Beacon® transponders) [22,36,38,47,55]. Simulation and treatment preparation details are summarized in Table 4. Most studies adopted a full bladder and empty rectum protocol [22,25,33,35,36,37,42,43,44,45,46,47,48,49,50,52,53,54,55], while an empty bladder approach was used exclusively in the study by Fan et al. [27].

Table 4.
Radiotherapy Technical Aspects.
MRI fusion for target volume definition was reported in 60% of studies (n = 18) [22,24,25,26,28,32,33,34,35,38,39,42,43,45,46,47,49,55], in contrast to PSMA PET/CT, which was employed in only one study [45]. The clinical target volume (CTV) was described in 29 studies (96.7%) [21,22,23,24,25,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55], and generally encompassed the entire prostate and proximal seminal vesicles [22,25,28,31,32,33,36,38,39,40,41,42,43,44,45,46,47,48,51,52,53,54,55]. In three studies, the CTV also included a 3–5 mm margin around the prostate or the involved prostatic side [36,41,42]. The whole seminal vesicles (SVs) were treated in all high-risk patients in seven trials [21,23,27,35,37,49,50], and in selected cases with T3b disease or based on institutional preference in three others [41,43,44]. Additionally, one study targeted the involved area of the SVs based on MRI findings [39], while another trial irradiated the SVs based on risk stratification but without specifying the policy followed for HRPC patients [34]. In one study enrolling 12 HRPC patients, the SVs were excluded from the CTV in all but two cases [24].
Urethral-sparing techniques, defined as delivering less than the prescribed dose (or DIL-PTV dose) to the urethra, were reported in five studies [46,47,49,50,55]. An additional eight studies included urethral planning objectives such as limiting the high-dose exposure through specific constraints, avoiding hot-spots, or recommending contouring of the urethra, though not mandatory [22,24,25,31,35,36,38,42,45].
In the five studies that actively implemented urethral-sparing approaches, T2-weighted MRI sequences or a Foley urinary catheter were used for delineation of the urethra [46,47,49,50,55]. Among these, although all delivered SBRT in five fractions, ranging from 35 to 50 Gy to the high-dose region, the urethral dose constraints varied. Examples included Dmax up to 30 Gy, D1cc up to 36 Gy, Dmax 36.25 Gy, D1cc up to 39 Gy, and V42 < 50% [46,47,49,50,55].
3.6. Bias Assessment and Heterogeneity
Each study was qualitatively evaluated for risk of bias without the use of formal instruments, according to the defined approach. Overall, the risk of bias across the included studies was considered moderate to high, reflecting their non-randomized, single-arm design and substantial methodological variability. In addition, a possible overlap in patient populations between two studies could not be determined from the available data [24,32]. Only two publications enrolled exclusively HRPC patients [44,52,53,54], while in the remaining studies this subgroup represented only a small proportion of the overall cohort. Reporting of key clinical variables, particularly the use and duration of ADT, was inconsistent, and definitions of high-risk disease differed. Toxicity outcomes were assessed using multiple grading systems, with variation in whether acute, late, or grouped toxicity events were reported. Technical aspects of SBRT also varied across studies, including dose and fractionation schedules, target volume definitions, urethral-sparing techniques, focal-boost approaches, organ spacers, imaging modalities for contouring, and simulation protocols. Although all studies used the Phoenix definition for biochemical failure, follow-up duration and the time points at which outcomes were reported were not uniform. Together, these elements describe the methodological and clinical heterogeneity observed across the included studies.
Risk of bias was therefore assessed qualitatively at the evidence-body level, considering domains such as patient selection, potential confounding assessed descriptively through variability in ADT use and duration, risk-group definitions and SBRT technical parameters, and outcome measurement. A formal assessment of reporting bias was not performed, as no quantitative syntheses were conducted and tools designed for meta-analytic evaluation were not applicable.
3.7. Certainty of Evidence Assessment
As prespecified in the Methods, no formal certainty-of-evidence assessment was performed because the non-comparative design and substantial heterogeneity among studies prevented a structured evaluation of confidence in the evidence.
4. Discussion
Our systematic review underscores the absence of randomized data evaluating the efficacy of SBRT for HRPC and the limited availability of prospective studies. Most of the current evidence derives from retrospective series, and only two studies were specifically designed to include HRPC patients [44,52]. In the remaining trials, BC outcomes were primarily obtained through subgroup analysis. In this context, early biochemical control endpoints in HRPC, particularly in the setting of prolonged ADT, provide limited information on durable disease control and should be interpreted cautiously.
When focusing on studies with longer follow-up, long-term BC outcomes appear favorable despite the aforementioned methodological limitations. Retrospective studies documented five-year PSA-control rates ranging from 74.1% to 95.2% [23,34,36,39,51]. Prospective evidence from three phase II trials [36,49,52,53,54] and two single-institution cohort [28,29,30,47] reported BC rates between 65% and 96.7% at five to eight years. Notably, only Tang et al. enrolled exclusively HRPC patients, reporting BC rates of 68.8% and 74.2% at five years in each arm [52,53,54]. In contrast, the other phase II studies either included combined outcomes of UIR and HRPC [36], or reported a global BC rate across all risk groups with at least 40% of failures being of high-risk [49]. These factors highlight two key limitations when interpreting long-term biochemical control in HRPC: heterogeneity in cohort composition and the fact that long-term estimates are informed by a limited subset of studies within a broader evidence base characterized by variable follow-up duration. Inclusion of mixed risk populations may dilute conclusions specific to HRPC, and interpretation of long-term outcomes should also be made with caution, as only a subset of the included studies reported mature follow-up of five years or longer.
Within this context, trials exclusively enrolling HRPC patients provide additional insight into SBRT efficacy in this subgroup. The consortium analysis by Van Dams et al., which pooled individual-level data from prospective institutional registries and phase II trials, reported a four-year BC rate of 81.7% [44]. Together with the study by Tang et al., this large pooled dataset strengthens the evidence supporting SBRT in HRPC patients [44,52,53,54].
Collectively, the long-term BC outcomes observed in our review align with those reported in contemporary trials employing conventional fractionation (CFRT) and moderate hypofractionation (HFRT) in high-risk populations [6,60,61]. For instance, the recent phase III PCS5 study by Niazi et al., which treated HRPC patients with dose-escalated RT using either CFRT or HFRT combined with prophylactic pelvic irradiation and 28 months of ADT, reported a five-year BC of 85.2% in both arms [60]. While PCS5 demonstrated slightly higher BC rates than those seen in exclusively HRPC SBRT cohorts [44,52,53,54], these differences may be explained by variations in treatment protocols and study designs. Importantly, no direct comparisons are currently available between SBRT and CFRT or HFRT for HRPC patients. Overall, the available evidence supports SBRT as a promising treatment alternative for HRPC.
Although phase III SBRT trials have largely focused on lower-risk patients, they provide useful reference points. The PACE-B trial evaluated a low and intermediate-risk PCa population, most of whom had UIR disease. In this context, SBRT to the prostate without ADT achieved excellent five-year BC rates of 95.8% [62]. In contrast, the HYPO-RT-PC trial included a higher-risk population, with 11% presenting with HRPC. This study reported lower BC rates at five years, at 84% in both the SBRT and CFRT arms. Importantly, despite SBRT dose being escalated to 92.7 Gy EQD2 (α/β = 1.5 Gy), ADT was not permitted [18]. Given that ADT is a standard component of treatment for HRPC, its omission in this higher-risk population may have contributed to the lower BC rates observed when compared to PACE-B. Multiple randomized trials have confirmed the benefit of combining EBRT with long-term ADT in HRPC patients [63,64]. This issue was addressed in the studies included in our review, as most of them considered the use of hormone therapy, despite differences in treatment schedules, duration, ADT protocols, or indication (fixed vs. physician’s discretion) [21,22,23,24,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,50,51,52,53,54,55].
However, the marked heterogeneity in ADT use, indication, and duration across the included studies should be considered when interpreting BC outcomes. Variations ranging from no ADT to prolonged courses exceeding 24 months, as well as differences in whether ADT was administered systematically or at physician discretion, may have influenced reported BC rates and contribute to between-study variability, since longer durations of ADT are associated with improved BC in HRPC [63,64]. While overall BC outcomes appear favorable and comparable to those reported with CFRT and HFRT, this heterogeneity limits the comparability of BC results across studies and should be taken into account when interpreting the existing evidence. This consideration is supported by the divergent findings reported in the two pooled analyses of phase II trials. While Van Dams et al. found a significant association between ADT use and reduced risk of biochemical failure (p = 0.009 by log-rank), King et al. reported no benefit in BC at five years for HRPC patients (80.2% vs. 82.5%, p = 0.5) [23,44].
Evidence regarding the impact of dose on BC in SBRT for definitive treatment of HRPC remains mixed. In our review, two studies revealed an association between dose and BC [24,44], while three studies found no significant difference between the high- and low-dose levels [23,28,52]. Oliai et al. found that all patients with biochemical failure at 3 years were in the dose cohorts of 35 Gy or 36.25 Gy in five fractions (EQD2 85–90.6 Gy) in contrast to 37.5 Gy (96.4 Gy) [24]. Additionally, subgroup analyses in Van Dams et al.’s trial revealed that a dose per fraction of 7 Gy vs. 8 Gy, for a total dose of 35 to 40 Gy, was associated with significantly higher risk of biochemical failure (HR 2.15, p = 0.03) [44]. This relationship between dose and BC has been previously documented. Zelesky et al. reported that when the SBRT dose is <40 Gy in five fractions (EQD2 108.6 Gy), the likelihood of a positive post-treatment biopsy increases (OR 2.34, p = 0.008), which is significantly associated with biochemical failure at 5 years (57% with positive biopsy vs. 7% with negative biopsy, p = 0.001) [65]. Interestingly, Tang et al. found no significant differences in BC at 5 years between 35 and 40 Gy in five fractions (p = 0.97), although different target volumes (including prophylactic pelvic RT in one arm) and different ADT durations were used (12 months vs. 18 months for prostate-only) in both arms [52]. This variability in treatment factors, including ADT duration and target volumes, likely contributed to these findings, underscoring the complex interplay of radiation dose and therapeutic variables in determining BC outcomes.
In most of the studies in our review, the prescription dose aimed for a PTV coverage of at least 95% [21,22,23,24,25,27,28,29,30,32,33,34,35,36,37,38,39,40,41,42,43,45,46,47,48,49,51,52,53,54,55], with substantial heterogeneity allowed (prescription dose normalization ranging from 65% to 90%) [21,22,24,25,27,31,32,34,39,40,41,42,45,51]. Apart from this, some studies included specific target coverage objectives to escalate the prescription dose [47], while some others used a simultaneous integrated boost strategy to a high-risk area or DIL to pursue the same aim [31,35,38,46,49,55].
In the study by Greco et al., a PTV mean dose of at least 45 Gy (EQD2 135 Gy) and a D98% >36 Gy (EQD2 89.5 Gy) were pursued [47]. Similarly, Parsai et al. [38] defined a low-dose PTV (LD-PTV) consisting of the prostate and 2 cm of the SVs receiving 36.25 Gy (EQD2 90.6 Gy) and a high-dose PTV (HD-PTV), consisting of the LD-PTV cropped at 3 mm from the urethra, bladder, and rectum. The HD-PTV was intended to receive a mean dose of 50 Gy or more or a coverage of at least 50% with a dose of 50 Gy (EQD2 164.3 Gy) [38]. Similarly, Kotecha et al. defined a LD-PTV and HD-PTV, receiving 36.25 Gy and 50 Gy in five fractions, respectively [31]. In four additional studies [35,46,49,55], a SIB was prescribed to a DIL ± margin. In three studies, the target area was defined using a mp-MRI [35,48,54], while one trial complemented this with PET/CT PSMA [46]. In these studies, the dose to the DIL ranged from 40 Gy to 50 Gy (EQD2 108 to 164.3 Gy). In all the studies using dose-escalation strategies and follow-up of at least 2 years, the reports on BC were good, ranging from 82.3% to 100% at 2.5–3 years, especially when ADT was used consistently [46,55].
Longer-term outcomes using dose escalation with dose painting were reported by Greco et al., showing a BC of 72.8% at 7 years [47]. This control rate is lower compared to the randomized phase III trial ASCENDE-RT, which reported a biochemical control of 86% and 75% at 7 years in the arms using dose-escalated radiotherapy with LDR-brachytherapy boost and EBRT [66]. Notably, although Greco’s trial used an EQD2 dose nearly double that of the EBRT arm in the ASCENDE-RT study, (135 Gy vs. 78 Gy, respectively), BC was lower. Moreover, when compared to the LDR-boost arm, which escalated the dose to about 161 Gy, BC rates were substantially lower [47,66]. Different factors, such as the most favorable profile of the ASCENDE-Trial that included 30.7% of intermediate-risk patients and the consistent use of 12 months of ADT, compared to only 36.2% of patients in Greco’s study at physicians’ discretion, with a median duration of 6 months, may partially explain these findings. Interestingly, this highlights the hypothesis that even in the setting of dose escalation using ultra-hypofractionated regimens, ADT plays a crucial role in HRPC populations and cannot be replaced by further dose escalation. It remains to be evaluated in randomized studies if dose-escalated SBRT associated with a longer course of hormone therapy can further improve the outcomes for HRPC patients.
Regarding toxicity, studies generally reported a favorable safety profile despite variations in assessment scales. However, interpretation of toxicity outcomes across studies should be made with caution, as variations in assessment scales (including RTOG, CTCAE, and institutional questionnaires) limit direct cross-study comparability and may influence the precise estimation and reporting of toxicity rates. Across the included studies, most patients presented with G1 or 2 events, or no toxicity at all in both the acute and late setting [21,22,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,41,42,43,44,45,47,51,55]. One study found that ADT use significantly correlated with acute and late G2+ GU toxicity (OR 4.1, p = 0.02; OR 4.34, p = 0.002) and late GI G2+ side effects (OR 0.11, p = 0.009) [44]. Similarly, dose per fraction of 7 Gy vs. 8 Gy was significantly associated with acute and late GU G2+ (OR 0.09, p = 0.005; OR 0.18, p = 0.002) toxicity but not with GI side effects [44]. Severe side effects in the GU and GI domains were uncommon in most trials, with acute and late G3 rates being generally either not observed or below 2%, G4 events being extremely rare and present in only one case in the study by Parsai et al., and G5 toxicity not seen across studies [21,22,25,26,27,28,29,30,31,32,33,34,35,36,37,38,41,42,43,45,47,48,49,50,51]. Three studies reported slightly higher rates of G3 toxicity, with rates of acute and late GU and GI events of 4–5% and 2.3–3% respectively [24,44,55].
Notably, the 40 Gy arm in the study by Tang et al. [52] presented an exceptionally opposite trend, revealing greatly increased rates of late G3+ GU and GI toxicity at 25% and 31.3%, respectively. These excessive 6-month toxicity rates led to early closure of this arm. The authors amended the protocol by decreasing the prescribed dose to 35 Gy in five fractions, reducing the posterior PTV margin, omitting elective nodal irradiation and using more restrictive constraints for organs at risk. These changes improved the dosimetric sparing of the rectum and bladder, which translated into lower rates of severe GU toxicity and significantly reduced G3+ GI side effects. Interestingly, the authors underlined that the reduced severe GI toxicity was likely due to a decrease in rectum volume exposed to a high radiation dose rather than a smaller bowel volume receiving a medium RT dose after omitting elective pelvic RT [31].
In our review, several studies employed urethral-sparing techniques or at least aimed to reduce the dose received [22,24,25,35,36,38,42,45,46,47,49,50,55]. In the five studies performing urethral sparing, the late GU G2 toxicity ranged from 2% to 16.7%, with most of the studies reporting rates between 2% and 4.5% [46,47,49,50,55]. Among these, the highest observed rates of late G2 toxicity were reported by Cloitre et al. and Yasar et al. [49,55] at 12.1% and 16.7%, respectively. Both trials used a SIB approach, with a DIL prescribed dose up to 47.5 Gy and 50 Gy [49,55]. Conversely, Greco et al. [47] reported a rate of 4.5% of late G2 GU side effects, with a cumulative rate of 5.3% of G2+ at 7 years. This study, prescribing a dose of 45 Gy in five fractions, used an inverse dose-painting planning to perform urethra sparing, with a D1cc up to 36 Gy [47]. Several studies in the literature have highlighted the importance of limiting the urethral dose [67,68,69]. Helou et al. reported that late urinary toxicity G2+ increased to 48% in patients receiving 40 Gy vs. 35 Gy in five fractions (OR 16.69, p < 0.001) [67]. Similarly, urethral V42 Gy > 2 cc has been associated with higher grade 2+ late urinary toxicity, and other studies have shown that urinary side effects increase progressively with dose escalation [67,68]. Moreover, a systematic review by Le Guevelou et al. reported that a Dmax < 90 Gy EQD2 (α/β = 3 Gy) to the urethra is associated with low rates of acute and late GU side effects [69]. Consistently, we observed in the study by Gregucci et al. a significant correlation in the univariate analysis between the use of urethra-sparing and GU toxicity (OR 0.39, p = 0.05) [50].
Overall, the toxicity findings suggest that urethral sparing and strict adherence to dose constraints are crucial strategies to mitigate GU and GI side effects in prostate SBRT. Although the general toxicity profile reported was favorable, the observed increase in severe toxicities at higher doses in certain protocols underscores the importance of optimizing treatment planning to balance treatment efficacy with patient safety. Further prospective studies are needed to confirm these findings and to refine dose constraints, particularly in the context of advanced techniques such as SIB to DIL and dose painting in the SBRT setting.
Across the studies included in our review, different treatment delivery systems were used, including CyberKnife, TomoTherapy, LINAC, and MR-LINAC. We did not observe any clear correlations between toxicity rates or efficacy outcomes and the type of treatment delivery system used, indicating that all these modalities are feasible and effective options. Importantly, IGRT was used in all studies and 23 of them (76.6%) included fiducial markers [21,22,23,24,25,26,27,28,32,34,35,38,39,40,41,42,44,45,46,47,48,50,51,55], which likely contributed to the low rates of severe toxicity and favorable BC profile observed across trials. As reported by Dang et al., IG-RT allows accounting for daily changes in target anatomy and positioning, reducing geometric uncertainties and geographic misses, as well as granting smaller PTV margins that translate into an improved toxicity profile [70]. Interestingly, we observed similar outcomes between trials using a conventional treatment preparation (e.g., full bladder and empty rectum) and those using additional devices (tissue spacers, rectal balloon, Foley catheter for bladder filling) [22,35,36,38,42,43,46,47,49,55]. Although a direct comparison was not made in our study due to limited data, further investigation is needed to assess the potential benefit of these devices in the context of prostatic SBRT.
The CTV consistently included the whole prostate in all studies plus a variable portion of the SVs, aligned with contouring guidelines for HRPC receiving definitive radiotherapy [71,72]. Whole seminal vesicle irradiation was used in seven studies (25%) as the standard protocol [21,23,27,35,37,49,50], and given in selected cases such as cT3b disease in another three [41,43,44]. This approach appeared feasible as no particularly higher toxicity rates were reported for these patients. In addition, some trials included an extra CTV margin for the prostate [36,41,42], while in others a gross tumor volume (GTV) based on MRI ± PET/CT PSMA findings was contoured for the effects of dose escalation [35,46,49,55]. MRI fusion was widely used for prostate volume definition and/or DIL delineation, refs. [22,24,25,26,30,32,33,34,35,38,39,42,43,45,46,47,49,55] with one study including a pre- and post-ADT MRI fusion to more accurately identify the GTV in the context of target changes associated with the use of hormone therapy [55]. Use of MRI for prostate contouring can result in smaller target volumes and reduce inter-observer variation compared to CT [73,74,75]. Moreover, MRI-based prostate SBRT has proven to significantly reduce moderate acute toxicity and to improve patient-reported Quality of Life compared to CT-based SBRT in the context of reduced PTV margins [76]. Indeed, in our review, the study by Poon et al. [43] using MRI-guided SBRT found no cases of acute G3 GU and one case (2%) of severe acute GI side effects.
Although few studies included the pelvis for elective nodal irradiation [37,43,44,52], the reported tolerability did not vary substantially from the rest of the studies, supporting the feasibility of this approach. Regarding the study of Tang presenting higher G3 toxicity rates in the arm receiving pelvic irradiation, this was likely due to an increased volume of rectum in the high-dose area, and not because of a higher volume of bowel receiving a moderate dose of radiation in the context of pelvic RT [52]. Indeed, Van Dams et al. reported in their multivariate analysis that nodal irradiation was not predictive of acute or late GU and GI toxicity [52]. Therefore, when deemed appropriate, this approach warrants further study, especially considering the benefit for HRPC in DMFS that has been reported in randomized trials of RT [77].
One limitation of this review is the restriction of the search to a single database (PubMed). While PubMed offers extensive coverage of biomedical literature, the exclusion of other databases may have led to the exclusion of studies not indexed in PubMed, potentially leading to selection bias. Nonetheless, given the high concentration of prostate cancer research in PubMed-indexed journals and the transparent reporting of our strategy, we believe the risk of significant omission is low. In addition, the search strategy did not include gray literature sources, which may further limit the comprehensiveness of the evidence capture. Although this approach was consistent with the predefined protocol and the use of two independent reviewers for screening and data extraction helps mitigate procedural error, the absence of supplementary searches remains a potential source of selection bias.
Second, as mentioned in the bias assessment section, there is considerable risk of bias among the included studies as they were all non-randomized comparisons, methodological designs varied, and no control arms were used in any of the trials. Also, follow-up periods were variable and redundancy may be introduced due to potential overlap between patient cohorts in two studies [24,32]. Importantly, HRPC were only a subgroup in most of the studies, and substantial heterogeneity on treatment characteristics such as SBRT dose, fractionation and planning, ADT schedule, and toxicity assessment, among others, was seen across the included trials. Therefore, conducting more precise comparisons and further analysis than the ones presented in our review was precluded. Taken together, these methodological and clinical differences reduce the comparability of results across studies and limit the certainty with which HRPC-specific conclusions can be drawn.
Nonetheless, our systematic review is, to our knowledge, the first to provide a comprehensive synthesis of biochemical control outcomes of SBRT in HRPC, incorporating updated evidence and restricting inclusion to studies reporting results specifically for this subgroup. Despite the limitations described, it provides a critical overview of the current evidence base. The favorable results observed across studies suggest that SBRT, in combination with ADT, may be a promising approach for selected high-risk patients. Ongoing prospective randomized trials, such as ASCENDE-SBRT and PACE-C [78,79], are expected to provide more definitive data and further clarify the role of SBRT in this population.
Beyond the need for forthcoming randomized evidence, future work should also focus on improving the consistency of SBRT-specific reporting to enable more robust cross-study comparisons. From a policy and implementation standpoint, the favorable therapeutic ratio observed across studies suggests that SBRT may be a promising option to help reduce treatment burden in selected high-risk patients, although decisions regarding broader adoption should remain context-specific and cautious until stronger data are available.
5. Conclusions
The evidence supporting the use of SBRT for HRPC is heterogeneous and derives primarily from retrospective cohorts and small phase 2 trials. Nonetheless, the reported outcomes in terms of BC and toxicity appear favorable, supporting further investigation of this treatment approach. Although the use and scheduling of ADT varied across studies, it still appears to play an important role in the management of high-risk patients, even when dose escalation and ultra-hypofractionated schedules are used.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/uro6010006/s1. PRISMA 2020 Checklist and PRISMA 2020 for Abstracts Checklist are included as supplementary files [80].
Author Contributions
Conceptualization, R.L., R.C., C.F. and M.S.; methodology, R.L. and R.C.; validation, R.L., R.C., C.F., A.G., R.M. and S.A.; data curation R.L., R.C., L.L.F., L.D.C., G.R.D. and M.B.; writing—original draft preparation R.L. and R.C.; writing—review and editing, R.L., R.C., C.F., L.L.F., L.D.C., G.R.D., A.G., R.M., S.A. and M.S.; supervision, M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Supporting data are available upon request to authors. No analytic code was generated and no additional materials were produced beyond the data extracted from published studies.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADT | Androgen deprivation therapy |
| BC | Biochemical control |
| CBCT | Cone-beam computed tomography |
| CCS | Cancer-specific survival |
| CFRT | Conventionally fractionationed radiation therapy |
| CT | Computed tomography |
| CTV | Clinical target volume |
| DIL | Dominant intraprostatic lesion |
| DM | Distant metastasis |
| DMFS | Distant metastasis-free survival |
| EBRT | External beam radiation therapy |
| FUP | Follow-up |
| G | Grade |
| GI | Gastrointestinal |
| GU | Genitourinary |
| HD-PTV | High-dose planning targetvolume |
| HFRT | Hypofractionationated radiation therapy |
| HRPC | High-risk prostate cancer |
| IGRT | Image-guided radiation therapy |
| IMRT | Intensity-modulated radiation therapy |
| LD-PTV | Low-dose planning targetvolume |
| LF | Local failure |
| MRI | Magnetic resonance imaging |
| Mp-MRI | Multiparametric magnetic resonance imaging |
| OS | Overall survival |
| P | Prostate |
| PCa | Prostate cancer |
| PET | Positron emission tomography |
| PSMA | Prostate-specific membrane antigen |
| PTV | Planning target volume |
| QoL | Quality of life |
| RT | Radiation therapy |
| SBRT | Stereotactic body radiation therapy |
| SIB | Simultaneous integrated boost |
| SVs | Seminal vesicles |
| UIR | Unfavorable intermediate-risk |
| VHRPC | Very high-risk prostate cancer |
References
- World Health Organization. Prostate Cancer Fact Sheet Globocan 2020; World Heal Organ: Geneva, Switzerland, 2020; Volume 419, pp. 119–120. [Google Scholar]
- Luz, M.A.; Guimarães, G.C.; Nardi, A.C.; Pompeo, A.S.F.L.; Sarkis, Á.S.; Nowier, A.; Pompeo, A.C.L.; Nardozza, A.; Adamy, A.; Carneiro, A.; et al. Consensus on Prostate Cancer Treatment of Localized Disease with Very Low, Low, and Intermediate Risk: A Report from the First Prostate Cancer Consensus Conference for Developing Countries (PCCCDC). JCO Glob. Oncol. 2021, 7, 523–529. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer. 2023. Available online: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf (accessed on 1 April 2023).
- Morgan, S.C.; Hoffman, K.; Loblaw, D.A.; Buyyounouski, M.K.; Patton, C.; Barocas, D.; Bentzen, S.; Chang, M.; Efstathiou, J.; Greany, P.; et al. Hypofractionated Radiation Therapy for Localized Prostate Cancer: Executive Summary of an ASTRO, ASCO and AUA Evidence-Based Guideline. J. Urol. 2019, 201, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Zaorsky, N.G.; Shaikh, T.; Murphy, C.T.; Hallman, M.A.; Hayes, S.B.; Sobczak, M.L.; Horwitz, E.M. Comparison of outcomes and toxicities among radiation therapy treatment options for prostate cancer. Cancer Treat. Rev. 2016, 48, 50–60. [Google Scholar] [CrossRef]
- Dearnaley, D.; Syndikus, I.; Mossop, H.; Khoo, V.; Birtle, A.; Bloomfield, D.; Graham, J.; Kirkbride, P.; Logue, J.; Malik, Z.; et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016, 17, 1047–1060. [Google Scholar] [CrossRef]
- Arcangeli, G.; Saracino, B.; Arcangeli, S.; Gomellini, S.; Petrongari, M.G.; Sanguineti, G.; Strigari, L. Moderate Hypofractionation in High-Risk, Organ-Confined Prostate Cancer: Final Results of a Phase III Randomized Trial. J. Clin. Oncol. 2017, 35, 1891–1897. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.; Mauldon, E.; Anderson, N. Cost-containment in hypofractionated radiation therapy: A literature review. J. Med. Radiat. Sci. 2018, 65, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Miralbell, R.; Roberts, S.A.; Zubizarreta, E.; Hendry, J.H. Dose-fractionation sensitivity of prostate cancer deduced from radiotherapy outcomes of 5969 patients in seven international institutional datasets: α/β = 1.4 (0.9–2.2) Gy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, e17–e24. [Google Scholar] [CrossRef] [PubMed]
- Fowler, J.F. The radiobiology of prostate cancer including new aspects of fractionated radiotherapy. Acta Oncol. 2005, 44, 265–276. [Google Scholar] [CrossRef]
- Ritter, M. Rationale, conduct and outcome using hypofractioned radiotherapy in prostate cancer. Semin. Radiat. Oncol. 2008, 23, 249–256. [Google Scholar] [CrossRef]
- Aznar, M.; Méndez Romero, A.; Heijmen, B.J. The role of technology in clinical trials using stereotactic body radiotherapy. Br. J. Radiol. 2017, 90, 20160930. [Google Scholar] [CrossRef]
- Udrescu, C.; Jalade, P.; de Bari, B.; Michel-Amadry, G.; Chapet, O. Evaluation of the respiratory prostate motion with four-dimensional computed tomography scan acquisitions using three implanted markers. Radiother. Oncol. 2012, 103, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Staffurth, J.; Radiotherapy Development Board. A review of the clinical evidence for intensity-modulated radiotherapy. Clin. Oncol. (R. Coll. Radiol.) 2010, 22, 643–657. [Google Scholar] [CrossRef]
- Lucchini, R.; Panizza, D.; Colciago, R.R.; Vernier, V.; Daniotti, M.C.; Faccenda, V.; Arcangeli, S. Treatment outcome and compliance to dose-intensified linac-based SBRT for unfavorable prostate tumors using a novel real-time organ-motion tracking. Radiat. Oncol. 2021, 16, 180. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Dean, C.; Patel, R.; Webster, G.; Eaton, D.J. Multi-center evaluation of dose conformity in stereotactic body radiotherapy. Phys. Imaging Radiat. Oncol. 2019, 11, 41–46. [Google Scholar] [CrossRef]
- Brand, D.H.; Tree, A.C.; Ostler, P.; van der Voet, H.; Loblaw, A.; Chu, W.; Ford, D.; Tolan, S.; Jain, S.; Martin, A.; et al. Intensity-modulated fractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-B): Acute toxicity findings from an international, randomised, open-label, phase 3, non-inferiority trial. Lancet Oncol. 2019, 20, 1531–1543. [Google Scholar] [CrossRef]
- Widmark, A.; Gunnlaugsson, A.; Beckman, L.; Thellenberg-Karlsson, C.; Hoyer, M.; Lagerlund, M.; Kindblom, J.; Ginman, C.; Johansson, B.; Björnlinger, K.; et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet 2019, 394, 385–395. [Google Scholar] [CrossRef]
- D’Amico, A.V.; Whittington, R.; Malkowicz, S.B.; Schultz, D.; Blank, K.; Broderick, G.A.; Tomaszewski, J.E.; Renshaw, A.A.; Kaplan, I.; Beard, C.J.; et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998, 280, 969–974. [Google Scholar] [CrossRef]
- Schaeffer, E.M.; Srinivas, S.; Adra, N.; An, Y.; Barocas, D.; Bitting, R.; Bryce, A.; Chapin, B.; Cheng, H.H.; D’Amico, A.V.; et al. Prostate Cancer, Version 4.2023, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2023, 21, 1067–1096. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.K.; Cho, C.K.; Choi, C.W.; Yoo, S.; Kim, M.S.; Yang, K.; Yoo, H.; Kim, J.H.; Seo, Y.S.; Lee, D.H.; et al. Image-guided stereotactic body radiation therapy for localized prostate cancer. Tumori 2011, 97, 43–48. [Google Scholar] [CrossRef]
- Bolzicco, G.; Favretto, M.S.; Satariano, N.; Scremin, E.; Tambone, C.; Tasca, A. A single-center study of 100 consecutive patients with localized prostate cancer treated with stereotactic body radiotherapy. BMC Urol. 2013, 13, 49. [Google Scholar] [CrossRef]
- King, C.R.; Freeman, D.; Kaplan, I.; Fuller, D.; Bolzicco, G.; Collins, S.; Meier, R.; Wang, J.; Kupelian, P.; Steinberg, M.; et al. Stereotactic body radiotherapy for localized prostate cancer: Pooled analysis from a multi-institutional consortium of prospective phase II trials. Radiother. Oncol. 2013, 109, 217–221. [Google Scholar] [CrossRef]
- Oliai, C.; Lanciano, R.; Sprandio, B.; Yang, J.; Lamond, J.; Arrigo, S.; Good, M.; Mooreville, M.; Garber, B.; Brady, L.W. Stereotactic body radiation therapy for the primary treatment of localized prostate cancer. J. Radiat. Oncol. 2013, 2, 63–70. [Google Scholar] [CrossRef]
- Bhattasali, O.; Chen, L.N.; Woo, J.; Park, J.W.; Kim, J.S.; Moures, R.; Yung, T.; Lei, S.; Collins, B.T.; Kowalczyk, K.; et al. Patient-reported outcomes following stereotactic body radiation therapy for clinically localized prostate cancer. Radiat. Oncol. 2014, 9, 52. [Google Scholar] [CrossRef]
- Davis, J.; Sharma, S.; Shumway, R.; Perry, D.; Bydder, S.; Simpson, C.K.; D’Ambrosio, D. Stereotactic Body Radiotherapy for Clinically Localized Prostate Cancer: Toxicity and Biochemical Disease-Free Outcomes from a Multi-Institutional Patient Registry. Cureus 2015, 7, e395. [Google Scholar] [CrossRef]
- Fan, C.Y.; Chao, H.L.; Huang, W.Y.; Lin, C.S.; Chen, C.M.; Lo, C.H. Stereotactic ablative radiotherapy with CyberKnife in the treatment of locally advanced prostate cancer: Preliminary results. Tumori 2015, 101, 684–691. [Google Scholar] [CrossRef]
- Katz, A.; Formenti, S.C.; Kang, J. Predicting Biochemical Disease-Free Survival after Prostate Stereotactic Body Radiotherapy: Risk-Stratification and Patterns of Failure. Front. Oncol. 2016, 6, 168. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.; Kang, J. Stereotactic body radiotherapy with or without external beam radiation as treatment for organ confined high-risk prostate carcinoma: A six year study. Radiat. Oncol. 2014, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.J.; Santoro, M.; Diblasio, F.; Ashley, R. Stereotactic body radiotherapy for localized prostate cancer: Disease control and quality of life at 6 years. Radiat. Oncol. 2013, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Kotecha, R.; Djemil, T.; Tendulkar, R.D.; Reddy, C.A.; Thousand, R.A.; Vassil, A.; Stovsky, M.; Berglund, R.K.; Klein, E.A.; Stephans, K.L. Dose-Escalated Stereotactic Body Radiation Therapy for Patients with Intermediate- and High-Risk Prostate Cancer: Initial Dosimetry Analysis and Patient Outcomes. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 960–964. [Google Scholar] [CrossRef]
- Ricco, A.; Manahan, G.; Lanciano, R.; Hanlon, A.; Yang, J.; Arrigo, S.; Lamond, J.; Feng, J.; Mooreville, M.; Garber, B.; et al. The Comparison of Stereotactic Body Radiation Therapy and Intensity-Modulated Radiation Therapy for Prostate Cancer by NCCN Risk Groups. Front. Oncol. 2016, 6, 184. [Google Scholar] [CrossRef] [PubMed]
- Marvaso, G.; Riva, G.; Ciardo, D.; Gandini, S.; Fodor, C.; Zerini, D.; Colangione, S.P.; Timon, G.; Comi, S.; Cambria, R.; et al. “Give me five” ultra-hypofractionated radiotherapy for localized prostate cancer: Non-invasive ablative approach. Med. Oncol. 2018, 35, 96. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Park, H.J.; Jang, W.I.; Jeong, B.K.; Kim, H.-J.; Chang, A.R. Long-term results and PSA kinetics after robotic SBRT for prostate cancer: Multicenter retrospective study in Korea (Korean radiation oncology group study 15–01). Radiat. Oncol. 2018, 13, 230. [Google Scholar] [CrossRef] [PubMed]
- Herrera, F.G.; Valerio, M.; Berthold, D.; Tawadros, T.; Meuwly, J.Y.; Vallet, V.; Baumgartner, P.; Thierry, A.-C.; De Bari, B.; Jichlinski, P.; et al. 50-Gy Stereotactic Body Radiation Therapy to the Dominant Intraprostatic Nodule: Results from a Phase 1a/b Trial. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 320–334. [Google Scholar] [CrossRef]
- Macias, V.A.; Barrera-Mellado, I. Ultra-hypofractionated radiation therapy for unfavourable intermediate-risk and high-risk prostate cancer is safe and effective: 5-year outcomes of a phase II trial. BJU Int. 2020, 125, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Mallick, I.; Arunsingh, M.; Chakraborty, S.; Arun, B.; Prasath, S.; Roy, P.; Dabkara, D.; Achari, R.; Chatterjee, S.; Gupta, S. A Phase I/II Study of Stereotactic Hypofractionated Once-weekly Radiation Therapy (SHORT) for Prostate Cancer. Clin. Oncol. (R. Coll. Radiol.) 2020, 32, e39–e45. [Google Scholar] [CrossRef]
- Parsai, S.; Juloori, A.; Sedor, G.; Reddy, C.A.; Thousand, R.; Magnelli, A.; Berglund, R.K.; Stovsky, M.M.; Klein, E.A.; Tendulkar, R.D.; et al. Heterogenous Dose-escalated Prostate Stereotactic Body Radiation Therapy for All Risk Prostate Cancer: Quality of Life and Clinical Outcomes of an Institutional Pilot Study. Am. J. Clin. Oncol. 2020, 43, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Vuolukka, K.; Auvinen, P.; Tiainen, E.; Palmgren, J.E.; Heikkilä, J.; Seppälä, J.; Aaltomaa, S.; Kataja, V. Stereotactic body radiotherapy for localized prostate cancer—5-year efficacy results. Radiat. Oncol. 2020, 15, 173. [Google Scholar] [CrossRef]
- Koskela, K.; Palmgren, J.E.; Heikkilä, J.; Virsunen, H.; Sailas, L.; Auvinen, P.; Seppälä, J.; Kataja, V. Hypofractionated stereotactic body radiotherapy for localized prostate cancer—First Nordic clinical experience. Acta Oncol. 2017, 56, 978–983. [Google Scholar] [CrossRef]
- Ishiyama, H.; Tsumura, H.; Nagano, H.; Watanabe, M.; Mizuno, E.; Taka, M.; Kobayashi, H.; Eriguchi, T.; Imada, H.; Inaba, K.; et al. Multi-institutional retrospective analysis of ultrahypofractionated radiotherapy for Japanese prostate cancer patients. Sci. Rep. 2021, 11, 13194. [Google Scholar] [CrossRef]
- Ito, M.; Yoshioka, Y.; Takase, Y.; Suzuki, J.; Matsunaga, T.; Takahashi, H.; Takeuchi, A.; Adachi, S.; Abe, S.; Oshima, Y.; et al. Stereotactic body radiation therapy for Japanese patients with localized prostate cancer: 2-year results and predictive factors for acute genitourinary toxicities. Jpn. J. Clin. Oncol. 2021, 51, 1253–1260. [Google Scholar] [CrossRef]
- Poon, D.M.C.; Yuan, J.; Wong, O.L.; Yang, B.; Tse, M.Y.; Lau, K.K.; Chiu, S.T.; Chiu, P.K.-F.; Ng, C.F.; Chui, K.L.; et al. One-year clinical outcomes of MR-guided stereotactic body radiation therapy with rectal spacer for patients with localized prostate cancer. World J. Urol. 2024, 42, 97. [Google Scholar] [CrossRef]
- Van Dams, R.; Jiang, N.Y.; Fuller, D.B.; Loblaw, A.; Jiang, T.; Katz, A.J.; Collins, S.P.; Aghdam, N.; Suy, S.; Stephans, K.L.; et al. Stereotactic Body Radiotherapy for High-Risk Localized Carcinoma of the Prostate (SHARP) Consortium: Analysis of 344 Prospectively Treated Patients. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 731–737. [Google Scholar] [CrossRef]
- Zhao, X.; Ye, Y.; Yu, H.; Jiang, L.; Cheng, C.; Guo, X.; Ju, X.; Zhu, X.; Zhang, H. Five-year outcomes of stereotactic body radiation therapy (SBRT) for prostate cancer: The largest experience in China. J. Cancer Res. Clin. Oncol. 2021, 147, 3557–3564. [Google Scholar] [CrossRef]
- Eade, T.; Kneebone, A.; Hruby, G.; Booth, J.; Hsiao, E.; Le, A.; Kwong, C.; Atyeo, J.; Brown, C.; Hunter, J.; et al. Early Outcomes and Decision Regret Using PSMA/MRI-Guided Focal Boost for Prostate Cancer SBRT. Pract. Radiat. Oncol. 2022, 12, e201–e206. [Google Scholar] [CrossRef] [PubMed]
- Greco, C.; Pares, O.; Pimentel, N.; Louro, V.; Nunes, B.; Kociolek, J.; Stroom, J.; Vieira, S.; Mateus, D.; Cardoso, M.J.; et al. Urethra Sparing with Target Motion Mitigation in Dose-Escalated Extreme Hypofractionated Prostate Cancer Radiotherapy: 7-Year Results from a Phase II Study. Front. Oncol. 2022, 12, 863655. [Google Scholar] [CrossRef]
- Kawakami, S.; Tsumura, H.; Satoh, T.; Tabata, K.; Sekiguchi, A.; Kainuma, T.; Nakano, M.; Iwamura, M.; Ishiyama, H. A phase II trial of stereotactic body radiotherapy in 4 fractions for patients with localized prostate cancer. Radiat. Oncol. 2022, 17, 67. [Google Scholar] [CrossRef] [PubMed]
- Cloitre, M.; Valerio, M.; Mampuya, A.; Rakauskas, A.; Berthold, D.; Tawadros, T.; Meuwly, J.-Y.; Heym, L.; Duclos, F.; Vallet, V.; et al. Toxicity, quality of life, and PSA control after 50 Gy stereotactic body radiation therapy to the dominant intraprostatic nodule with the use of a rectal spacer: Results of a phase I/II study. Br. J. Radiol. 2023, 96, 20220803. [Google Scholar] [CrossRef] [PubMed]
- Gregucci, F.; Carbonara, R.; Surgo, A.; Ciliberti, M.P.; Curci, D.; Ciocia, A.; Branà, L.; Ludovico, G.M.; Scarcia, M.; Portoghese, F.; et al. Extreme hypofractionated stereotactic radiotherapy for elderly prostate cancer patients: Side effects preliminary analysis of a phase II trial. Radiol. Med. 2023, 128, 501–508. [Google Scholar] [CrossRef]
- Pei Yuin, J.L.; Jia Shin, J.T.; Jing, C.B.; Mun, T.L.; Balasubramaniam, M.A.; Ibrahim Wahid, D.M. Retrospective Analysis of Clinical Outcomes of Stereotactic Body Radiation Therapy for Localized Prostate Cancer at an Asian Cancer Specialist Centre. Asian Pac. J. Cancer Prev. 2023, 24, 545–550. [Google Scholar] [CrossRef]
- Tang, T.; Rodrigues, G.; Warner, A.; Bauman, G. Long-Term Outcomes Following Fairly Brief Androgen Suppression and Stereotactic Radiation Therapy in High-Risk Prostate Cancer: Update from the FASTR/FASTR-2 Trials. Pract. Radiat. Oncol. 2024, 14, e48–e56. [Google Scholar] [CrossRef]
- Callan, L.; Bauman, G.; Chen, J.; Lock, M.; Sexton, T.; D’Souza, D.; Rodrigues, G. A Phase I/II Trial of Fairly Brief Androgen Suppression and Stereotactic Radiation Therapy for High-Risk Prostate Cancer (FASTR-2): Preliminary Results and Toxicity Analysis. Adv. Radiat. Oncol. 2019, 4, 668–673. [Google Scholar] [CrossRef]
- Bauman, G.; Ferguson, M.; Lock, M.; Chen, J.; Ahmad, B.; Venkatesan, V.M.; Sexton, T.; D’Souza, D.; Loblaw, A.; Warner, A.; et al. A Phase 1/2 Trial of Brief Androgen Suppression and Stereotactic Radiation Therapy (FASTR) for High-Risk Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 856–862. [Google Scholar] [CrossRef]
- Yasar, B.; Suh, Y.E.; Chapman, E.; Nicholls, L.; Henderson, D.; Jones, C.; Morrison, K.; Wells, E.; Henderson, J.; Meehan, C.; et al. Simultaneous Focal Boost with Stereotactic Radiation Therapy for Localized Intermediate- to High-Risk Prostate Cancer: Primary Outcomes of the SPARC Phase 2 Trial. Int. J. Radiat. Oncol. Biol. Phys. 2024, 120, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, L.; Suh, Y.E.; Chapman, E.; Henderson, D.; Jones, C.; Morrison, K.; Sohaib, A.; Taylor, H.; Tree, A.; van As, N. Stereotactic radiotherapy with focal boost for intermediate and high-risk prostate cancer: Initial results of the SPARC trial. Clin. Transl. Radiat. Oncol. 2020, 25, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Roach, M., III; Hanks, G.; Thames, H., Jr.; Schellhammer, P.; Shipley, W.U.; Sokol, G.H.; Sandler, H. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: Recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 965–974. [Google Scholar] [CrossRef]
- Lei, S.; Piel, N.; Oermann, E.K.; Chen, V.; Ju, A.W.; Dahal, K.N.; Hanscom, H.N.; Kim, J.S.; Yu, X.; Zhang, G.; et al. Six-dimensional correction of intra-fractional prostate motion with CyberKnife stereotactic body radiation therapy. Front. Oncol. 2011, 1, 48. [Google Scholar] [CrossRef]
- Chen, L.N.; Suy, S.; Uhm, S.; Oermann, E.K.; Ju, A.W.; Chen, V.; Hanscom, H.N.; Laing, S.; Kim, J.S.; Lei, S.; et al. Stereotactic body radiation therapy (SBRT) for clinically localized prostate cancer: The Georgetown University experience. Radiat. Oncol. 2013, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Niazi, T.; Nabid, A.; Malagon, T.; Tisseverasinghe, S.; Bettahar, R.; Dahmane, R.; Martin, A.G.; Jolicoeur, M.; Yassa, M.; Barkati, M.; et al. Hypofractionated Dose Escalation Radiotherapy for High-Risk Prostate Cancer: The survival analysis of the Prostate Cancer Study-5 (PCS-5), a GROUQ-led phase III trial. Eur. Urol. 2025, 87, 314–323. [Google Scholar] [CrossRef]
- de Vries, K.C.; Wortel, R.C.; Oomen-de Hoop, E.; Heemsbergen, W.D.; Pos, F.J.; Incrocci, L. Hyprofractionated Versus Conventionally Fractionated Radiation Therapy for Patients with Intermediate- or High-Risk, Localized, Prostate Cancer: 7-Year Outcomes from the Randomized, Multicenter, Open-Label, Phase 3 HYPRO Trial. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 108–115. [Google Scholar] [CrossRef]
- van As, N.; Tree, A.; Patel, J.; Ostler, P.; Van Der Voet, H.; Loblaw, D.; Chu, W.; Ford, D.; Tolan, S.; Jain, S.; et al. 5-Years Outcomes from PACE B: An International phase III Randomized Controlled Trial Comparing Stereotactic Body Radiotherapy vs. Conventionally Fractionated or Moderately Hypo Fractionated External Beam Radiotherapy for Localized Prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2023, 117, e2–e3. [Google Scholar] [CrossRef]
- Bolla, M.; Van Tienhoven, G.; Warde, P.; Dubois, J.B.; Mirimanoff, R.O.; Storme, G.; Bernier, J.; Kuten, A.; Sternberg, C.; Billiet, I.; et al. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 2010, 11, 1066–1073. [Google Scholar] [CrossRef]
- Cartes, R.; Karim, M.U.; Tisseverasinghe, S.; Tolba, M.; Bahoric, B.; Anidjar, M.; McPherson, V.; Probst, S.; Rompré-Brodeur, A.; Niazi, T. Neoadjuvant Versus Concurrent Androgen Deprivation Therapy in Localized Prostate Cancer Treated with Radiotherapy: A Systematic Review of the Literature. Cancers 2023, 15, 3363. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zelefsky, M.J.; Goldman, D.A.; Hopkins, M.; Pinitpatcharalert, A.; McBride, S.; Gorovets, D.; Ehdaie, B.; Fine, S.W.; Reuter, V.E.; Tyagi, N.; et al. Predictors for post-treatment biopsy outcomes after prostate stereotactic body radiotherapy. Radiother. Oncol. 2021, 159, 33–38. [Google Scholar] [CrossRef]
- Morris, W.J.; Tyldesley, S.; Rodda, S.; Halperin, R.; Pai, H.; McKenzie, M.; Duncan, G.; Morton, G.; Hamm, J.; Murray, N. Androgen Suppression Combined with Elective Nodal and Dose Escalated Radiation Therapy (the ASCENDE-RT Trial): An Analysis of Survival Endpoints for a Randomized Trial Comparing a Low-Dose-Rate Brachytherapy Boost to a Dose-Escalated External Beam Boost for High- and Intermediate-risk Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2017, 98, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Helou, J.; D’Alimonte, L.; Quon, H.; Deabreu, A.; Commisso, K.; Cheung, P.; Chu, W.; Mamedov, A.; Davidson, M.; Ravi, A.; et al. Stereotactic Ablative Radiotherapy in the Treatment of Low and Intermediate Risk Prostate Cancer: Is There an Optimal Dose? Radiother. Oncol. 2017, 123, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Johnson, J.; Gottschalk, A.R.; Chang, A.J.; Hsu, I.C.; Roach, M.; Seymour, Z.A. Receiver Operating Curves and Dose-Volume Analysis of Late Toxicity with Stereotactic Body Radiation Therapy for Prostate Cancer. Pract. Radiat. Oncol. 2017, 7, 109–116. [Google Scholar] [CrossRef]
- Le Guevelou, J.; Bosetti, D.G.; Castronovo, F.; Angrisani, A.; de Crevoisier, R.; Zilli, T. State of the art and future challenges of urethra-sparing stereotactic body radiotherapy for prostate cancer: A systematic review of literature. World J. Urol. 2023, 41, 3287–3299. [Google Scholar] [CrossRef]
- Dang, A.; Kupelian, P.A.; Cao, M.; Agazaryan, N.; Kishan, A.U. Image-guided radiotherapy for prostate cancer. Transl. Androl. Urol. 2018, 7, 308–320. [Google Scholar] [CrossRef]
- Salembier, C.; Villeirs, G.; De Bari, B.; Hoskin, P.; Pieters, B.R.; Van Vulpen, M.; Khoo, V.; Henry, A.; Bossi, A.; De Meerleer, G.; et al. ESTRO ACROP consensus guideline on CT- and MRI-based target volume delineation for primary radiation therapy of localized prostate cancer. Radiother. Oncol. 2018, 127, 49–61. [Google Scholar] [CrossRef]
- Patel, K.R.; van der Heide, U.A.; Kerkmeijer, L.G.W.; Schoots, I.G.; Turkbey, B.; Citrin, D.E.; Hall, W.A. Target Volume Optimization for Localized Prostate Cancer. Pract. Radiat. Oncol. 2024, 14, 522–540. [Google Scholar] [CrossRef]
- Rasch, C.; Barillot, I.; Remeijer, P.; Touw, A.; van Herk, M.; Lebesque, J.V. Defintion of the prostate in CT and MRI: A multi-observer study. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Sander, L.; Langkilde, N.; Holmberg, M.; Carl, J. MRI target delineation may reduce long-term toxicity after prostate radiotherapy. Acta Oncol. 2014, 53, 809–814. [Google Scholar] [CrossRef]
- Hentschel, B.; Oehler, W.; Straus, D.; Ulrich, A.; Malich, A. Definition of the CTV prostate in CT and MRI by using CT–MRI image fusion in IMRT planning for prostate cancer. Strahlenther. Onkol. 2011, 187, 183–190. [Google Scholar] [CrossRef]
- Kishan, A.U.; Ma, T.M.; Lamb, J.M.; Casado, M.; Wilhalme, H.; Low, D.A.; Sheng, K.; Sharma, S.; Nickols, N.G.; Pham, J.; et al. Magnetic Resonance Imaging-Guided vs. Computed Tomography-Guided Stereotactic Body Radiotherapy for Prostate Cancer: The MIRAGE Randomized Clinical Trial. JAMA Oncol. 2023, 9, 365–373. [Google Scholar] [CrossRef]
- Murthy, V.; Maitre, P.; Kannan, S.; Panigrahi, G.; Krishnatry, R.; Bakshi, G.; Prakash, G.; Pal, M.; Menon, S.; Phurailatpam, R.; et al. Prostate-Only Versus Whole-Pelvic Radiation Therapy in High-Risk and Very High-Risk Prostate Cancer (POP-RT): Outcomes from Phase III Randomized Controlled Trial. J. Clin. Oncol. 2021, 39, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.clinicaltrials.gov/study/NCT06235697 (accessed on 19 March 2025).
- Tree, A.C.; Hinder, V.; Chan, A.; Tolan, S.; Ostler, P.; van der Voet, H.; Kancherla, K.; Loblaw, A.; Naismith, O.; Jain, S.; et al. PACE Investigators. Intensity-modulated moderately hypofractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-C): Early toxicity results from a randomised, open-label, phase 3, non-inferiority trial. Lancet Oncol. 2025, 26, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.