Abstract
Cardio-renal syndrome (CRS) is a term referring to a bidirectional group of disorders in which there is a concomitant compromise of both organs, the heart and the kidney, leading to a significant increase in morbidity and mortality. In recent years, numerous publications have addressed this complex entity from different points of view. For better understanding, five subtypes have been established: depending on its form of presentation, acute or chronic; the organ initially affected; and whether there is another responsible systemic disease. CRS represents a complex interaction between both organs with several neurohormonal, inflammatory, and hemodynamic pathophysiological mechanisms involved. Because of its heterogeneous presentation and complex management, CRS requires a multidisciplinary approach targeting multiple pathogenic mechanisms. Throughout this review, we will analyze all relevant aspects of CRS from its classification to current diagnosis and treatment. Accordingly, this review provides a holistic overview of cardio-renal syndrome, emphasizing integrated pathophysiological mechanisms, contemporary diagnostic approaches, and their relevance for clinical management.
1. Introduction
A substantial proportion of patients exhibit varying degrees of cardiac and renal dysfunction, where a primary injury in one of these organs may lead to secondary disorder in the other [1,2,3]. Cardio-renal syndrome (CRS) is characterized by the coexistence of cardiac and renal impairment within a bidirectional and dynamic interaction, in which heart failure (HF) precipitates or exacerbates renal functional decline and vice versa [3,4,5]. In this process, dysfunction of either organ can initiate or exacerbate injury in the other through complex neurohormonal feedback mechanisms [2,6]. The lack of a clear definition has contributed to uncertainty in the diagnosis and management of this syndrome for a long time. For this reason, a subdivision of CRS into five subtypes has been proposed to allow a more specific approach for each case [2]. However, identifying the initial injury and subsequent events is often challenging in clinical practice, as phenotypic overlap frequently occurs during disease progression [7]. In this context, the present review addresses cardio-renal syndrome as a complex and heterogeneous clinical entity, integrating current knowledge on pathophysiology and diagnostic evaluation to support a more comprehensive and clinically oriented approach to patient management. An integrated overview of the different cardio-renal syndrome subtypes and their clinical implications is provided in Figure 1.
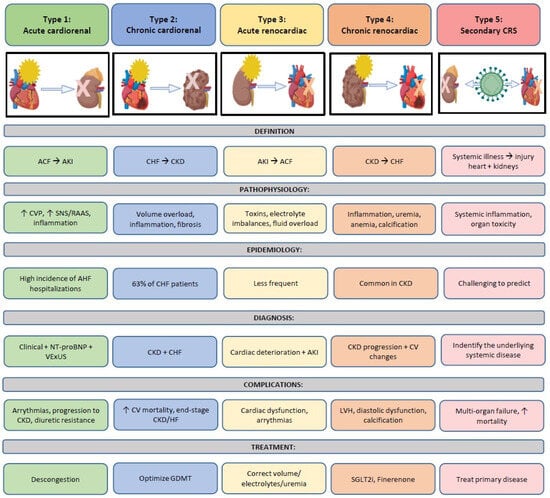
Figure 1.
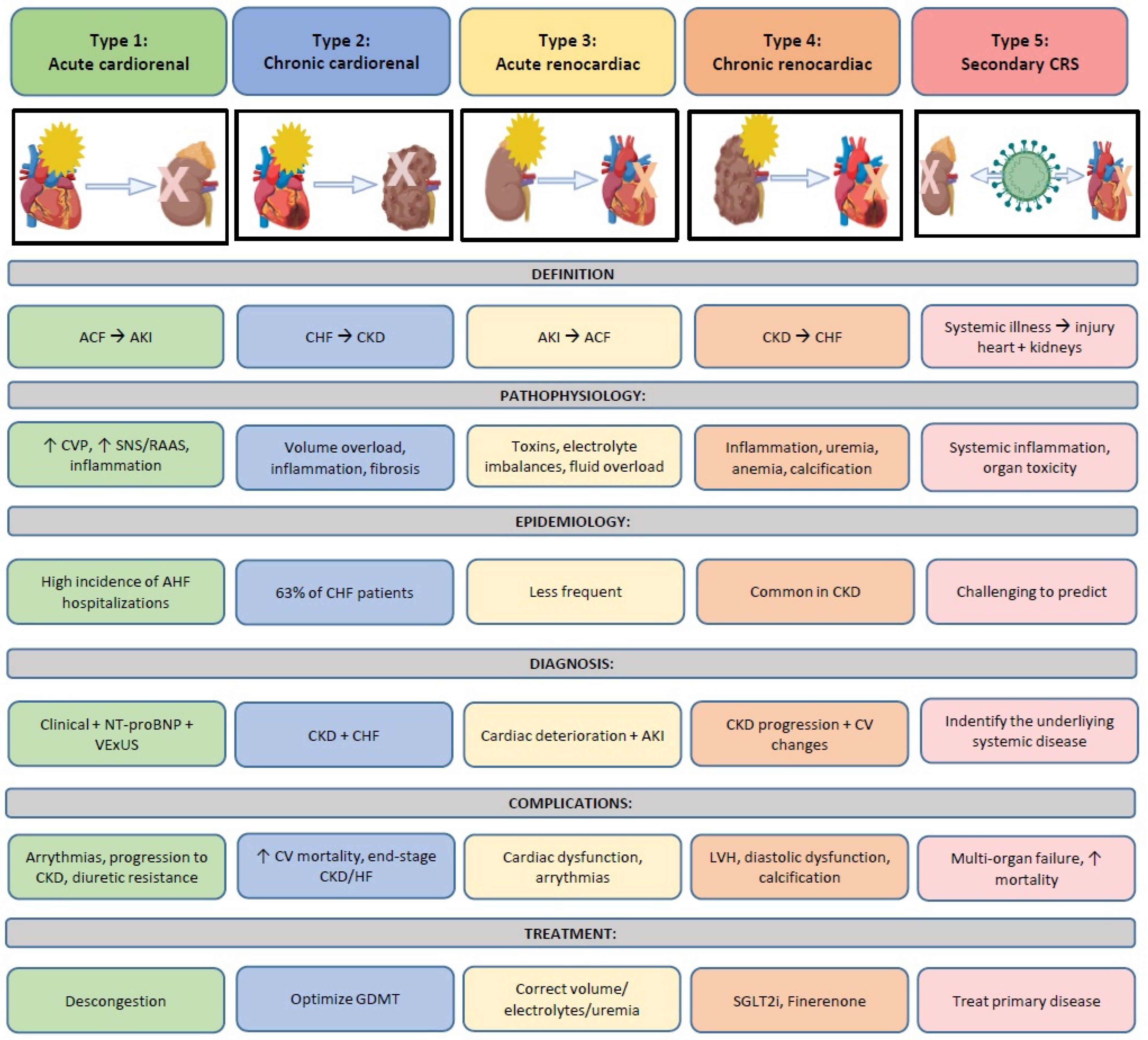
Global management of CRS from subtypes to treatment. This central figure summarizes the five types of cardio-renal syndrome, illustrating their definitions, underlying pathophysiological mechanisms, epidemiology, diagnostic features, major complications, and therapeutic principles. The figure provides an integrated and clinically oriented overview of the bidirectional interactions between the heart and the kidneys across acute and chronic settings, reflecting the conceptual framework developed throughout the manuscript. ACF: Acute cardiac failure; AKI: Acute kidney injury; CHF: Chronic heart failure; CKD: Chronic kidney disease; CRS: Cardio-renal syndrome; CV: Cardiovascular; CVP: Central venous pressure; GDMT: guideline-directed medical therapy; HF: Heart failure; NT-ProBNP: N-terminal brain natriuretic peptide; SGLT2i. SGLT2 inhibitors; SNS: Sympathetic nervous system, AVP: Arginine–vasopressin; VExUS: Venous Excess Ultrasound Score.
2. Classification of Cardio-Renal Syndrome and Phenotypes: What Are We Talking About
First description of CRS is attributed to the English physician Robert Bright, who in 1836 described the relationship between the kidney and the heart in his classic paper published in Volume 1 Guy’s Hospital Reports entitled “Cases and observations illustrative of renal disease accompanied by the secretion of albuminous urine”, in which the significant cardiac structural changes seen in patients with advanced kidney disease were revealed [8].
Years later and, as we have previously mentioned, the pioneering definition of CRS was established by the Working Group of the National Heart, Lung, and Blood Institute in 2004, defining the CRS as the result of interactions between the kidneys and other circulatory compartments that increase circulating volume, which exacerbates the symptoms of heart failure (HF) and disease progression [9,10].
Therefore, it represents the confluence of heart–kidney interactions across several interfaces. Later, in September 2008, a consensus conference was held in Venice, sponsored by the Acute Dialysis Quality Initiative (ADQI), with experts and leaders in the fields of nephrology, critical care, cardiac surgery, and cardiology. This meeting resulted in a consensus document that established the definition and classification of CRS into five phenotypes as we know it today, depending on the organ initially injured and the acute or chronic nature of the damage [11]. The definitions, pathophysiological mechanisms, diagnostic features, and therapeutic principles of the different CRS subtypes are summarized in Figure 1.
The relevance of CRS lies in the fact that the coexistence of heart and kidney disease has been linked to a worse medium and long-term prognosis, due to a higher risk of readmissions and mortality in patients [12]. Understanding the symbiosis between both organs and the early recognition of damage to one of them can help to stop the deterioration in the other one earlier [11].
According to the proposed classification, the five types of CRS are defined as follows.
2.1. Acute Cardio-Renal Syndrome (CRS1)
Acute cardio-renal syndrome, also known as CRS type 1, is defined as an abrupt worsening of cardiac function that occurs in at least 30% of patients with acute decompensated heart failure and can lead to the development of acute kidney injury. Traditionally, acute kidney injury (AKI) in acute heart failure (AHF) patients is defined by worsening renal function (WRF) during hospitalization, which has been broadly defined as serum creatinine changes ranging from 0.3 to 0.5 mg/dL. However, there is a great disagreement between heart failure and nephrology guidelines on the best criteria for WRF [13].
We summarize in Table S1 RIFLE (Risk, Injury, Failure, Loss of Kidney, Function, and End-Stage Kidney Disease), AKIN (Acute Kidney Injury Network), KDIGO (Kidney Disease: Improving Global Outcomes), and WRF (Worsening Renal Function) criteria for the definition of acute kidney injury [14,15,16].
2.2. Chronic Cardio-Renal Syndrome (CRS2)
Cardio-renal syndrome type 2 is described by chronic cardiac abnormalities resulting in renal damage and or dysfunction. This syndrome is common and has been reported in 63% of patients hospitalized with congestive heart failure (CHF) [17,18].
Two conditions have been proposed to make a diagnosis of CRS type 2: that CHF and chronic kidney disease (CKD) coexist in the patient and that CHF causally provokes the CKD. Sometimes it results in difficulty in determining which of the two pathologies is primary versus secondary; for this reason, the term CRS type 2/4 has been suggested [19].
2.3. Acute Reno-Cardiac Syndrome (CRS3)
This subtype refers to heart injury and/or dysfunction secondary to acute worsening of kidney function. There are a number of potential contributing causes for AKI that may predispose to the development of CRS type 3, such as fluid overload, metabolic acidosis, retention of uremic toxins, electrolyte disturbances, accumulation of myocardial depressant factors, neurohormonal activation, and systemic inflammation. All of them have been postulated to lead to cardiac dysfunction [13]. The development of AKI may also adversely impact cardiac function by contributing to alterations in drug pharmacokinetics and pharmacodynamics [20,21].
2.4. Chronic Reno-Cardiac Syndrome (CRS4)
It is the result of chronic kidney disease (CKD) leading to heart injury, disease, and/or dysfunction. This subtype refers to a disease or dysfunction of the heart occurring secondary to CKD. Different examples of this subtype are: diabetic nephropathy, chronic glomerular disease, or autosomal dominant polycystic kidney disease. They all can promote the progression of chronic HF (with preserved or reduced ejection fraction [EF]) [22], ventricular hypertrophy, diastolic dysfunction, and increased risk of adverse cardiovascular events [23,24].
Type 4 of CRS is associated with high morbidity and mortality, and its incidence is increasing with population aging.
2.5. Secondary Cardio-Renal Syndrome (CRS5)
It refers to systemic conditions leading to simultaneous injury and/or dysfunction of the heart and kidney, for example, sepsis, lupus, amyloidosis, or inflammatory conditions [23].
2.6. Cardiovascular–Kidney–Metabolic Syndrome
Cardiovascular–kidney–metabolic syndrome represents a novel conceptual framework that integrates metabolic, renal, and cardiovascular dysfunction into a single systemic disorder. Unlike traditional approaches that consider these conditions separately, this syndrome emphasizes the interconnected pathophysiology linking excess or dysfunctional adiposity, chronic kidney disease, and cardiovascular damage. Adiposity acts as a central driver, promoting inflammation, insulin resistance, and organ injury [25].
In Table 1, we summarize the current 5 CRS subtypes.

Table 1.
Subtypes of cardio-renal syndrome.
3. Epidemiology: A Syndrome with High Incidence and Prevalence
The estimated prevalence of chronic kidney disease (CKD) is around 10–20% in many countries. It is well established that these patients carry a high cardiovascular (CV) risk, resulting in an increased incidence of CV events, including CV mortality [2,5]. Data from the United States show that over 60% of patients with CKD have CV disease, and the degree of CV disease is closely correlated with the severity of CKD. Moreover, the impact of CKD on CV disease becomes even more pronounced in patients requiring dialysis [3].
Additionally, it is estimated that the prevalence of HF reaches approximately 64 million adults worldwide, among whom CKD is present in more than 50% [1,5]. Furthermore, the National Registry of Acute Decompensated Heart Failure has shown that 30% of patients hospitalized because of HF decompensation exhibit impaired renal function [3,4,6].
Acute kidney injury (AKI) is an ominous predictor of mortality in cardiogenic shock and has been associated with in-hospital mortality rates of approximately 50% across several observational studies [10].
Regarding each subtype of CRS, type 1 precipitates the need for hospitalization in over one million people per year in the United States, with an estimated incidence between 27% and 45%. In addition, the prevalence of type 2 CRS is approximately 25% [2].
Type 3 CRS appears to be less frequent than type 1, probably due to limited research on this subtype and, as referred in the consensus document, the challenge in understanding the epidemiology of type 3 CRS is that its incidence and associated risk factors fail to consider the inciting event for CSA-AKI as either primarily AKI-related or heart-related [2]. Regarding chronic reno-cardiac syndrome (type 4), as we have previously mentioned, cardiac disease is common in patients with CKD, and this is associated with a high increase in both cardiovascular morbidity and mortality. Finally, epidemiological data on type 5 CRS, secondary to other diseases, are difficult to obtain precisely due to the wide range of systemic illnesses involved.
4. Pathophysiological Mechanism in Cardio-Renal Syndrome: Complex and Multifactorial
Pathophysiology of CRS is complex and multifactorial; different processes are involved, and each of them contributes to a different extent depending on the patient’s clinical situation [26]. We briefly review these mechanisms:
4.1. Cardiac Output and Cardiac Index: Low Cardiac Output-Renal Hypoperfusion
Among hospitalized patients, worsening renal failure (WRF) is common, and it is associated with increased risk of death and prolonged hospitalization, as proven in different studies [22]. Hemodynamic factors like hypotension play a role, but it is not the only one. Low cardiac output and central fluid redistribution with renal hypoperfusion activate compensatory mechanisms such as stimulation of the sympathetic nervous system, renin–angiotensin–aldosterone system (RAAS), and vasopressin secretion, which lead to enhanced water and sodium reabsorption. As a result of the renal afferent arteriolar vasoconstriction and decrease in glomerular perfusion, renal function finally worsens [27]. This response also induces fibrosis, apoptosis, and ventricular remodeling [26]. In this context, one might think that increasing cardiac output and blood pressure with inotropes would be sufficient to improve renal and other organ perfusion. However, the results of the ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) found no correlation between renal function and cardiac output, and improvements in cardiac output did not translate into better renal function, reduced mortality, or fewer hospitalizations. Therefore, these results suggest that decreased cardiac output and hemodynamic failure are not solely responsible for the progressive decline in renal function in patients with heart failure [28]. As a conclusion, pathophysiology and management are far more complicated than previously thought.
4.2. Increase in Central Venous Pressure
Carl Ludwig, the first scientist to describe the process of glomerular filtration, and F. R. Winton, in 1931, reached the same conclusion: the increase in venous pressure was responsible for a reduction in urine flow. Although the mechanisms are not clearly established, the increase in central venous pressure affects the kidney by increasing its afterload and intra-renal pressure [29,30]. This increase leads to a decrease in glomerular filtration rate (GFR) and an increase in water and sodium retention; therefore, relieving decongestion is clearly relevant in the management of patients with heart failure and CRS [27]. Otherwise, and also related to venous congestion, the study published by Maeder et al. showed that patients with at least a moderate tricuspid regurgitation had lower estimated GFR and that a linear relationship existed between the severity of tricuspid regurgitation and the degree of GFR impairment, probably secondary to elevation of central and renal venous pressure [31].
4.3. Neurohormonal Dysregulation and Sympathetic Activity
The sympathetic nervous system (SNS) is activated in both acute kidney injury and heart failure. The direct effects of catecholamines on cardiac myocytes can contribute to both normal physiological adaptation and pathological remodeling, and may be associated with cellular hypertrophy, apoptosis, and alterations in contractile function. Furthermore, stimulation of the SNS involves stimulation of the RAAS. Angiotensin II (AT II) induces apoptosis and fibrosis of myocytes and is also involved in oxidative stress, inflammation, and extracellular matrix regulation, which ultimately lead to renal parenchymal damage [32,33]. RAAS inhibition is a cornerstone of heart failure treatment, offering benefits through reduced adrenergic tone, improved endothelial function, and prevention of myocardial fibrosis. Furthermore, ACE inhibitors and angiotensin receptor blockers also have a renoprotective effect in hypertensive and diabetic patients with nephropathy [27].
4.4. Stress Oxidative and Endothelial Dysfunction
There is growing evidence supporting the link between oxidative stress and progressive cardiac and renal damage. ATII plays a significant role in this process, exerting a deleterious effect by activating NADPH oxidase and NADH oxidase [28]. The activation of these two enzymes is responsible for oxidative stress in renal tubule cells and cardiac myocytes. Since both primary cardiac failure and primary renal failure lead to the development of the RAAS, activation of oxidases by AT-II in one organ has the potential to lead to progressive dysfunction in the other one through the generation of reactive oxygen species [27].
4.5. Inflammatory Response
All the aforementioned factors, such as activation of the sympathetic nervous system (SNS) and renin–angiotensin–aldosterone system (RAAS), venous congestion, and oxidative stress, act as triggers for the initiation and propagation of the inflammatory cascade. Thus, both heart failure and chronic kidney disease are inflammatory states that lead to the generation of pro-inflammatory biomarkers, resulting in damage to both organs with cell death and fibrosis. Several cytokines are involved, such as tumor necrosis factor-α (TNF-α), members of the interleukin-1 (IL-1) family, and interleukin-6 (IL-6), all of which have been associated with heart failure as well as chronic kidney disease (CKD). Otherwise, inflammation seems to be largely associated with inadequate renal perfusion pressures, peritubular edema, pathological reduction in glomerular filtration, and tubular damage [26,34]. Some of these inflammatory biomarkers also have prognostic implications, such as IL-6, which independently predicted overall and cardiovascular mortality in patients at different stages of chronic kidney disease and soluble ST2, a member of the IL-1 family [35]. Moreover, chronic inflammation associated with chronic kidney disease predicts all-cause and cardiovascular mortality in hemodialysis patients. In the same way, high C-Reactive protein (CRP) in hemodialysis patients also predicts left ventricular dysfunction, cardiac hypertrophy, and mortality [35,36].
4.6. Renal Failure and Anemia: The Cardio-Renal Anemia Syndrome
Anemia, as defined by hemoglobin levels < 13 g/dL in men and <12 g/dL in women, plays an important role in the pathogenesis of CRS. It is common in congestive heart failure (CHF) and is associated with increased mortality, morbidity, and progressive kidney failure. The most common causes of anemia in CHF include chronic kidney disease (CKD), which reduces erythropoietin production (EPO), and systemic inflammation, which impairs both erythropoietin synthesis and bone marrow response. This set of interactions is called cardio-renal–anemia syndrome [37].
In patients with CKD, anemia is associated with poor quality of life, progression of kidney disease, cardiovascular comorbidities, and higher mortality. There are several mechanisms by which anemia is involved in the pathogenesis of CRS. Ischemic insult secondary to lack of oxygen in both heart and kidney can result in progressive cell death in both organs, with myocardial necrosis and tubular damage. Anemia can also cause ischemia in other organs and tissues with peripheral vasodilatation and activation of SNS and RAAS, which finally leads to chronic renal venous congestion and progressive nephron loss and fibrosis [26]. Managing anemia is a complex therapeutic target. There are no definitive guidelines, and treatment should address HF, CKD, and anemia concurrently. A proposed algorithm includes correcting iron deficiency, initiating sodium–glucose cotransporter inhibitors, and considering erythropoiesis-stimulating agents if hemoglobin remains <10 g/dL [38].
4.7. Adiposity and Renal Failure: Cardio-Renal–Metabolic Syndrome
Excess visceral and ectopic fat promotes chronic low-grade inflammation, insulin resistance, and dyslipidemia. Adipokines and inflammatory cytokines contribute to endothelial dysfunction, oxidative stress, and organ injury, as previously described. Moreover, hyperglycemia, hypertension, and systemic inflammation impair renal hemodynamics and promote glomerular and tubular injury [25].
5. Diagnosis: Integrated Approach, Testing, and Expected Results
5.1. General Principles of Diagnosis
Diagnosis begins with clinical suspicion: acute or subacute worsening of renal function in the context of heart failure, or cardiac deterioration in a patient with chronic kidney disease without another evident explanation. Worsening renal function is commonly defined as an increase in serum creatinine of ≥0.3 mg/dL within 48 h or a ≥25% decline in estimated glomerular filtration rate (eGFR) from baseline, while persistent increases in albumin-to-creatinine ratio (ACR) may reflect structural renal injury. Because creatinine and eGFR are late and volume-dependent markers, CRS diagnosis requires a multimodal approach integrating clinical assessment, biomarkers, imaging, and functional testing. In most clinical studies, WRF has been defined as a change in serum creatinine or eGFR over time, but there is great variability in the specific markers used (serum creatinine vs. eGFR vs. cystatin C) and in the thresholds considered significant. Some studies use an absolute rise in creatinine, while others use a relative change in eGFR or different cutoffs, and there is no single consensus definition adopted across international heart failure or nephrology guidelines. This heterogeneity makes it difficult to compare results between studies and generate consistent clinical interpretations. Another source of controversy is that similar changes in serum creatinine may reflect different underlying processes. Because serum creatinine levels can also be influenced by factors such as baseline renal function, muscle mass, inflammation, or fluid shifts, the same numeric change can have different clinical implications in different patients [39,40,41].
5.2. Clinical and Biomarker Assessment in CRS
Diagnostic evaluation should distinguish between acute (CRS type 1) and chronic (CRS type 2) cardiac patients, and CRS type 3–4, which primarily affect the kidney.
- CRS1 and CRS2 (acute/chronic cardiac):
History and physical examination: monitor diuretics, weight changes, nephrotoxic drugs, signs of congestion (Figure 2, pathway 2: jugular venous distention, edema, and hepatomegaly), and hypoperfusion (Figure 2, pathway 1: oliguria and hypotension). Systemic congestion predicts renal deterioration [42,43].
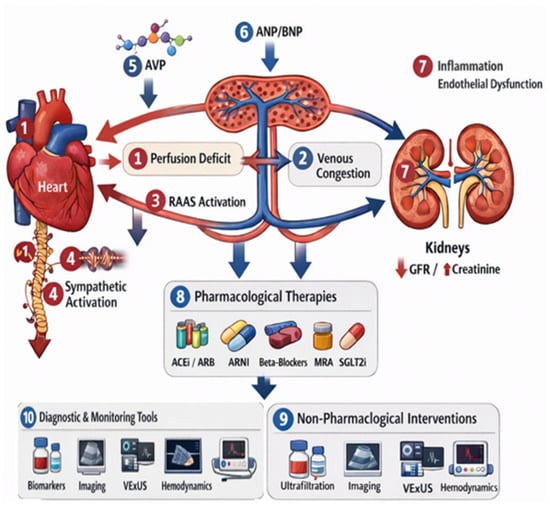
Figure 2.
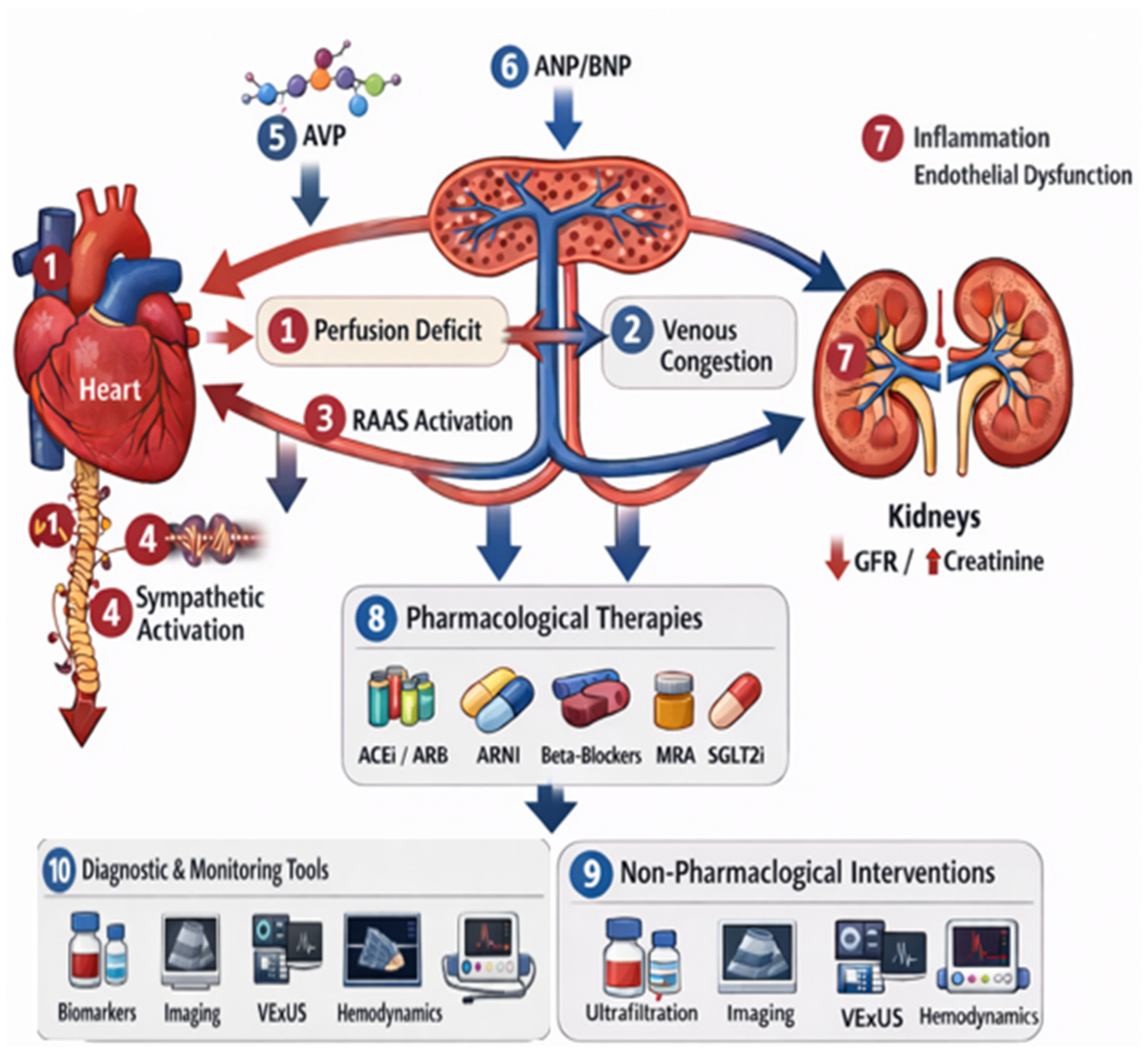
Pathophysiological mechanisms, diagnostic tools, and therapeutic targets in CRS. Numbered pathways correspond to mechanisms or interventions discussed in the text: (1) reduced cardiac output; (2) venous congestion; (3) RAAS activation; (4) sympathetic nervous system activation; (5) AVP release; (6) natriuretic peptides (ANP/BNP); (7) inflammation and endothelial dysfunction; (8) pharmacological therapies (ACEi, ARB, ARNI, beta-blockers, MRA, SGLT2 inhibitors, and diuretics); (9) non-pharmacological interventions (ultrafiltration, peritoneal dialysis, and inotropes); and (10) diagnostic tools (biomarkers, imaging, VExUS, and hemodynamic assessment). A detailed explanation of each pathway is provided in the main text. CRS: Cardio-renal syndrome; RAAS: Renin–angiotensin–aldosterone system; AVP: Arginine–vasopressin; ANP/BNP: Atrial/brain natriuretic peptide; ARBs: Angiotensin receptor blockers; ACEi: Angiotensin-converting enzyme inhibitor; ARNI: Angiotensin receptor–neprilysin inhibitor; MRAs: Mineralocorticoid receptor antagonists; VExUS: Venous Excess Ultrasound Score.
- Basic monitoring: Creatinine, urea, electrolytes, hourly diuresis, fluid balance, and sodium/potassium; use an ECG to rule out ischemia/arrhythmias [44].
- Natriuresis: urine sodium after IV diuretics predicts diuretic response; low values indicate need for escalation (Figure 2, pathways 8–9) [48,49].
- CRS3 and CRS4 (primary kidney):
Patients are at risk of heart failure and coronary artery disease due to shared risk factors (hypertension, diabetes, and vascular calcification) and endothelial dysfunction (Figure 2, pathways 1–2, 7) [42,43,44].
- Assessment includes renal function (creatinine and eGFR), electrolytes, and cardiac evaluation (ECG, troponin, and imaging) to detect subclinical CAD and guide therapy (Figure 2, pathways 1–2, 10).
- Biomarkers and natriuresis remain critical to identify tubular injury and guide therapy (Figure 2, pathways 8–10) [46,47,48,49,50].
- Interpretation guide (all CRS types):
Elevated NT-proBNP + congestion → predominant venous component (Figure 2, pathways 2,6).
Low natriuresis + abnormal VExUS → renal congestion and diuretic resistance (Figure 2, pathways 2,10).
Positive renal biomarkers + stable creatinine → early tubular injury (Figure 2, pathway 10).
5.3. Imaging and Hemodynamic Testing
- ▪
- Lung ultrasound (B-lines): Correlates with interstitial edema and lung fluid. Monitors response to diuretics (Figure 2, pathway 10) [51].
- ▪
- Echocardiography (2D/3D + strain): Evaluates systolic/diastolic function, filling pressures, pericardial effusion, and cardiopathies. Reduced ejection fraction or severe diastolic dysfunction increases CRS risk (Figure 2, pathway 1) [52].
- ▪
- Global longitudinal strain (GLS): Detects subclinical dysfunction preceding biomarkers and renal deterioration (Figure 2, pathway 1).
- ▪
- Venous Doppler and VExUS: Quantifies systemic venous congestion. High scores correlate with AKI risk, poor diuretic response, and rehospitalization (Figure 2, pathway 2) [53].
- ▪
- Intrarenal Doppler/RVSI: Pulsatile/monophasic renal venous flow indicates significant renal congestion. It correlates with reduced diuresis and worse prognosis (Figure 2, pathway 2) [54].
- ▪
- Right-heart catheterization: In selected patients, measures fill pressures to confirm predominant venous congestion. It guides ultrafiltration or advanced therapy (Figure 2, pathways 2, 10) [55].
5.4. Functional Evaluation and Applied Diagnostic Algorithms
An efficient diagnostic algorithm typically integrates: clinical assessment, natriuresis/biomarkers, POCUS/VExUS, and transthoracic echocardiography. The goal is to classify CRS into subphenotypes (predominant venous congestion, predominant hypoperfusion, primary tubular injury, or mixed forms) (Figure 2, pathways 1–2, 10) that determine the intervention (aggressive decongestion, hemodynamic adjustment, or specific renal-protective therapy) (Figure 2, pathways 8–9) [2,7,56].
6. Complications of Cardio-Renal Syndrome: Pathophysiological Mechanisms
CRS complications (arrhythmias, worsening ventricular function, progression to CKD, diuretic resistance, and electrolyte disturbances) arise from intertwined hemodynamic, neurohormonal, inflammatory, and structural mechanisms (Figure 2, pathways 1–7). Understanding the “why” behind each complication guides preventive and therapeutic strategies. Figure 1 represents mechanisms and treatment for CRS.
6.1. Arrhythmias: Why They Increase in CRS
Arrhythmias (atrial fibrillation, ventricular tachycardias, and bradyarrhythmias) are frequent and have multiple converging causes:
- Pressure and volume overload: Atrial and ventricular dilation from overload increases reentrant substrate and electrical heterogeneity [57,58]. Venous congestion and elevated left atrial pressure favor atrial fibrillation through anatomical and electrical remodeling.
- Myocardial fibrosis and remodeling: Chronic inflammation, neurohormonal activation (RAAS, norepinephrine), and oxidative stress induce interstitial fibrosis, creating areas of slow conduction and conduction block [59,60].
- Ionic and electrolyte disturbances: Hypokalemia (diuretics), hypomagnesemia, or hyperkalemia (renal failure or RAAS inhibition) alter repolarization and predispose to both tachyarrhythmias and bradyarrhythmias; hyponatremia is also associated with poor prognosis [61].
- Myocardial ischemia and cellular injury: Coronary hypoperfusion in hypotension or coexisting coronary disease can generate irritative foci and elevated troponin with arrhythmic risk [62].
- Pharmacologic effects and drug accumulation.
- Systemic inflammation and sympathetic stress [63].
6.2. Worsening Ventricular Function and Progression to Heart Failure
- Reduced output from systolic or diastolic dysfunction leads to renal hypoperfusion; lower renal perfusion worsens sodium/water retention, increasing congestion [64].
- Chronic neurohormonal activation (RAAS, aldosterone, sympathetic tone) causes vasoconstriction, fibrosis, and remodeling that perpetuate HF and renal injury [65].
- Myocardial injury from uremia and uremic toxins: In advanced CKD, accumulation of metabolites promotes myocardial dysfunction (uremic cardiomyopathy) [66].
- Inflammation and oxidative stress accelerate myocyte loss and replacement with fibrotic tissue [67].
6.3. Diuretic Resistance and Failure of Depletive Therapy
- Pharmacokinetic mechanisms: Reduced renal perfusion may diminish diuretic delivery to the tubule [68].
- Shift in reabsorption site: RAAS and aldosterone activation increase distal sodium reabsorption, counteracting loop inhibition [69].
- Proximal sodium recovery and tubular adaptation (braking phenomenon) [70].
- Intrarenal congestion (elevated renal venous pressure) reduces filtration gradient, and volume depletion fails to produce natriuresis; correcting venous congestion is often more effective than intensifying diuretics [46,48].
6.4. Electrolyte Disorders and Therapeutic Limitations
Hyperkalemia from reduced GFR or RAAS inhibitor use limits evidence-based HF therapies; hyponatremia reflects severe neurohormonal activation and correlates with poor prognosis [52,71].
6.5. Thrombotic Risk and Other Systemic Complications
Venous stasis, chronic inflammation–associated hypercoagulability, and endothelial dysfunction increase thrombotic risk (deep vein thrombosis and pulmonary embolism) [72].
7. Practical Implications and How to Translate Diagnosis into Prevention of Complications
Early diagnosis and subphenotyping: Implementing protocols that combine early natriuresis, renal biomarkers, and VExUS allows identification of patients with intrarenal congestion and intervention before established AKI develops [46,49,53].
Prioritized correction of venous congestion when present: Guided decongestion (natriuresis, VExUS) is more effective than indiscriminate increases in diuretics; consider sequential nephron blockade and, in refractory cases, advanced therapies [2,50].
Management of arrhythmias and correction of precipitants: Strict electrolyte control, review of accumulated drugs, optimization of hemodynamics, and ischemia prevention are recommended [57,61,62].
Preservation of guideline-directed medical therapies: When possible, use strategies (closer monitoring, adjunctive treatments) that allow maintaining RAAS inhibitors, beta-blockers, and SGLT2 inhibitors while protecting renal function [55,65,73].
Integrated care models: Cardiology, nephrology, clinical pharmacy, and specialized nursing improve adherence to diagnostic and therapeutic strategies and enable proactive follow-up [56].
8. Prevention of Cardio-Renal Syndromes
Before addressing the treatment of CRS, it is essential to highlight the importance of prevention. The first step is to identify patients at risk of developing the different types of cardio-renal syndrome and initiate preventive measures early as a strategy to reduce the likelihood of occurrence and improve outcomes should they develop.
8.1. Acute Cardio-Renal Syndrome (CRS1)
Predisposing risk factors for CRS 1 include [23,74,75]:
- ○
- Non-modifiable risk factors:
- History of diabetes mellitus;
- Previous hospitalizations for heart failure;
- History of acute myocardial infarction;
- Severe ventricular dysfunction;
- Baseline renal impairment.
- ○
- Modifiable risk factors:
- Use of high doses of diuretics;
- Vasodilator therapy;
- Drugs promoting sodium and volume retention, such as NSAIDs, thiazolidinediones, and corticosteroids;
- Radiographic contrast agents.
Several risk-prediction tools exist for estimating the likelihood of acute kidney injury in specific contexts, such as heart failure [22] (exposure to iodinated contrast during interventional procedures [76,77,78]) or after cardiac surgery [79,80,81]. These tools incorporate clinical variables [22,77,79,80], procedural characteristics [76,77,78,79,80,81], and novel approaches including machine learning [82,83], enabling accurate identification of at-risk patients and facilitating preventive and early diagnostic strategies for CRS.
Key preventive measures for CRS type 1 include [6,23,74,75]:
- Comprehensive control of cardiovascular risk factors: smoking cessation, blood pressure optimization, lipid management, intensified glycemic control, and weight management.
- Avoidance of excessive or unnecessary use of radiographic contrast and application of renal protection protocols when contrast is required. Risk factors for contrast-induced nephropathy include pre-existing CKD, diabetes, chronic heart failure, hypotension, advanced age, and high contrast volumes. Evidence supports the use of isotonic fluid administration as the most effective preventive measure, along with low-osmolar, non-ionic contrast agents such as iopamidol [84].Additional strategies include statins, which reduce the risk of contrast-induced nephropathy and slow CKD progression in select populations, although heterogeneity in study designs limits generalizability [85], and N-acetylcysteine, which in recent meta-analyses has shown benefit in reducing contrast nephropathy but without impact on dialysis or mortality [86].
- Avoidance of nephrotoxic medications, or use at the minimum necessary dose and duration.
- Avoidance of sodium-retaining drugs.
- Implementation of cardiovascular therapies, pharmacological and invasive, appropriate for the underlying cardiac disease, with the aim of improving cardiac function and preventing heart failure decompensation: optimization of hemodynamic and volume status; modulators of neurohormonal and stress responses; treatment of anemia or malnutrition; cardiac resynchronization therapy or defibrillator implantation when indicated; mechanical circulatory support or transplantation in end-stage HF or end stage renal disease (ESRD); optimal renal replacement therapy; and disease-specific therapies for CVD, CKD, or HF.
- Close monitoring of renal function and electrolytes in patients with cardiac decompensation.
8.2. Chronic Cardio-Renal Syndrome (CRS2)
All patients with chronic heart disease, particularly those with chronic heart failure, carry a potential risk of CRS2, especially individuals older than 75 years, those with diabetes, or those with baseline renal dysfunction [23,74]. In this setting, therapies proven to improve the natural history of heart failure with preserved ejection fraction (SGLT2 inhibitors (SGLT2i)) or reduced ejection fraction (renin–angiotensin system inhibitors (RASis), beta-blockers, SGLT2i, mineralocorticoid receptor antagonists (MRAs), cardiac resynchronization therapy, or defibrillator therapy in indicated cases, wherein mechanical circulatory support or transplantation in patients with end-stage HF and other specific therapies for CVD serve as preventive strategies for CRS2 [6,7,23,87].
8.3. Acute Reno-Cardiac Syndrome (CRS3)
Predisposing factors for CRS3 include [74,88,89]:
- ○
- Non-modifiable risk factors:
- Age > 75 years;
- History of diabetes mellitus;
- Chronic heart failure;
- Peripheral vascular disease;
- Liver disease;
- Sepsis.
- ○
- Modifiable risk factors:
- Use of nephrotoxic and cardiotoxic medications;
- Hypotension;
- Electrolyte and fluid imbalances.
Preventive measures for CRS3 focus on avoiding or minimizing the use of potentially nephrotoxic or cardiotoxic drugs, ensuring adequate organ perfusion, and maintaining appropriate hydration while avoiding hypervolemia to prevent cardiac decompensation. Additionally, careful management of uremic changes, hyperkalemia, and pro-inflammatory mediators with adverse cardiac consequences is essential [23,74,88,89].
8.4. Chronic Reno-Cardiac Syndrome (CRS4)
CRS4 is a common manifestation in the natural history of CKD progression. Preventive strategies, therefore, aim at optimizing control of factors associated with CKD progression, such as proteinuria, diabetes, hypertension, atherosclerosis, left ventricular hypertrophy, systolic and diastolic dysfunction, and prothrombotic or proinflammatory states. Slowing CKD progression may reduce the incidence of CRS type 4 [6,7,14,23,74].
8.5. Secondary Cardio-Renal Syndrome (CRS5)
Finally, prevention of CRS5 relies on the early and appropriate management of the primary multisystem disease responsible for simultaneous cardiac and renal involvement [7,23].
9. Treatment of Cardio-Renal Syndrome
Management of CRS is challenging due to its pathophysiological heterogeneity, five subtypes, and frequent comorbidities influencing treatment response. Therefore, therapy should be individualized, focused on the dominant pathology, and often multidisciplinary [1,23]. Specific evidence is limited, as most drugs have not been tested in CRS-specific trials, and patients with reduced glomerular filtration were typically excluded. No dedicated CRS guidelines exist, so management relies on HF and acute or chronic kidney disease recommendations [7,44,90,91]. Drugs improving HF prognosis and slowing renal progression are central. Optimal treatment of underlying cardiac disease is key in CRS types 1 and 2, while controlling baseline nephropathy is critical in types 3 and 4. In CRS type 5, focus is on the primary systemic disease [2]. To facilitate interpretation of the evidence discussed in this section, Table 2 summarizes the major randomized clinical trials supporting the pharmacological treatment of cardio-renal syndrome, including kidney exclusion criteria, key findings, and clinical implications for each therapeutic class across different CRS subtypes.
9.1. Acute Cardio-Renal Syndrome (CRS1)
In CRS1, associated with acute HF, persistent congestion and renal deterioration worsen prognosis, making effective decongestion while preserving renal perfusion the main goal [1,23]. Management includes oxygen therapy, congestion control, hemodynamic support, discontinuation of nephrotoxics, and arrhythmia management [74].
Loop diuretics are first-line; high-dose IV accelerates decongestion, with transient renal impairment generally considered hemodynamic without prognostic impact [50,74]. Dose should be adjusted early according to urine output or urinary sodium [1,44,90,91]. If response is insufficient, combinations with thiazides, acetazolamide, high-dose MRA, or tolvaptan may be used safely, though without additional cardio-renal prognostic benefit. In contrast, iSGLT2 provides provencardio-renall benefit [5,92]. Ultrafiltration shows heterogeneous results. While some studies demonstrated effective decongestion [93,94], others reported greater renal deterioration and adverse effects [95]; these mixed findings highlight that the effectiveness, safety, and optimal use of ultrafiltration remain unsettled, particularly in differing clinical contexts and CRS subtypes. Therefore, current guidelines reserve this technique for volume overload refractory to optimal medical therapy [44,74,90]. Peritoneal dialysis may be considered in selected patients with advanced CKD and refractory HF, with good hemodynamic tolerance [1].
Vasodilators may help with normal or high blood pressure but carry a risk of hypotension [23,74]. In hypotension or cardiogenic shock, inotropes and mechanical circulatory support are critical [1,23,74].
After stabilization, early initiation and optimization of therapies with proven HF benefit is essential, with close outpatient follow-up for titration and monitoring of volume status and renal function [5,91].
9.2. Chronic Cardio- Renal Syndrome (CRS2)
Management of chronic HF in CRS2 targets underlying etiology and symptom control to prevent progression and decompensation of HF and CKD, requiring a comprehensive patient-centered approach including adherence, lifestyle, and guideline-directed drugs and devices [2,5,6,7,23].
In LVEF < 40%, RASi (ACEi, ARBs, sacubitril/valsartan), beta-blockers, MRA, and iSGLT2 are first-line, reducing morbidity and mortality. In diabetic or obese patients, aGLP-1 adds cardiovascular benefit. In patients with LVEF > 40%, only iSGLT2 and, in diabetic or obese patients, aGLP-1 have demonstrated significant cardiovascular benefit. Diuretics improve symptoms without affecting mortality. Finerenone reduces HF hospitalizations in CKD with diabetes [5,6,23,44,74,90,91]. CRS2 patients often exhibit hypervolemia, requiring intensive loop diuretics, often combined. Initiation and titration of RASi and MRA may cause transient renal impairment or hyperkalemia, requiring close monitoring and sometimes new potassium binders. RASi, iSGLT2, finerenone, and aGLP-1 reduce proteinuria, and iSGLT2 slows CKD progression. The correction of anemia and iron deficiency improves symptoms and prognosis [1,7,23,74].
Devices such as cardiac resynchronization therapy, implantable defibrillators, and advanced therapies should be considered in selected patients, though less effective in advanced CRS [1,7,23,74].
In patients with CRS type 2, evidence supporting cardio-renal protection derives from multiple clinical settings, including patients with cardiovascular disease and diabetes, as well as those with HF with reduced ejection fraction, HF with preserved ejection fraction, and HF with mildly reduced ejection fraction. In these populations, renin–angiotensin system inhibitors, SGLT2 inhibitors, GLP-1 receptor agonists, and non-steroidal mineralocorticoid receptor antagonists such as finerenone have demonstrated beneficial effects on cardiovascular and renal outcomes across different randomized trials. While the magnitude of benefit may vary according to the underlying phenotype, these therapies collectively represent the cornerstone of an integrated cardio-renal protective strategy. Key supporting trials are summarized in Table 2.
9.3. Acute Cardio-Renal Syndrome (CRS3)
Prevention is key, particularly in CRS3 associated with contrast nephrotoxicity or cardiac surgery. Once established, management focuses on treating AKI, controlling systemic complications, and close hemodynamic monitoring. Proper volume management, avoiding hypo- or hypervolemia, and correcting uremia, acid-base, and electrolyte disturbances are critical, as these can exacerbate cardiac dysfunction, arrhythmias, hemodynamic compromise, and catecholamine resistance. If conventional therapy fails, renal replacement therapy should be considered early [23,74,89].
9.4. Chronic Reno-Cardiac Syndrome (CRS4)
Management focuses on comprehensive CKD care and mitigating cardiovascular impact. Lifestyle measures (sodium, potassium, and protein restriction) and glycemic control are essential. Blood pressure should be managed with RASi (ACEi or ARBs) and beta-blockers for cardio- and renoprotection, though use is limited in renal replacement therapy [14,74].
iSGLT2 improves renal and cardiovascular outcomes even in non-diabetic CKD [96]. Finerenone reduces renal progression and CV events in CKD with type 2 diabetes [97], and aGLP-1 improves metabolic control and cardiovascular risk, potentially benefiting renal outcomes in diabetic or obese patients [98].
Metabolic acidosis should be corrected with bicarbonate or citrate, associated with slower CKD progression and improved survival. Anemia management is controversial: iron and erythropoiesis-stimulating agents improve cardiac and renal function, but overcorrection increases CV events; target Hb 11–12 g/dL, avoiding > 13 g/dL and excluding iron deficiency. Calcium phosphate metabolism control is crucial to prevent vascular calcification [14,23,74].
9.5. Secondary Cardio-Renal Syndrome (CRS5)
Management is based on treating the primary disease and supporting renal and cardiovascular function. Early hypotension correction with volume resuscitation and, if needed, inotropes or vasopressors is critical to prevent organ dysfunction. Hemodynamic monitoring guides interventions. Persistent renal deterioration may require continuous renal replacement therapy to maintain function and modulate inflammation via cytokine removal. Avoid nephrotoxics and adjust medications according to perfusion and renal function, using an early multidisciplinary approach to optimize outcomes [2,23,74].

Table 2.
Evidence table of clinical outcomes with therapeutic interventions in cardio-renal syndromes.
Table 2.
Evidence table of clinical outcomes with therapeutic interventions in cardio-renal syndromes.
| Class of Drug/Intervention | Drug or Strategy | Mechanism of Action | Trial (Year) | No. of Patients | Intervention | Kidney Exclusion Criteria | Key Findings | Clinical Use in Cardio-Renal Syndrome |
|---|---|---|---|---|---|---|---|---|
| ACE inhibitor | Enalapril | RAAS inhibition | SOLVD-Treatment (1991) [99] | 2569 | Enalapril vs. placebo | Creatinine > 2.5 mg/dL | Reduced mortality and HF hospitalization | Core therapy in CRS type 2 with HFrEF |
| SOLVD-Prevention (1992) [100] | 4228 | Enalapril vs. placebo | Creatinine > 2.5 mg/dL | Delayed HF development | Early CRS type 2 with HFrEF | |||
| ARB | Valsartan | AT1 receptor blockade | Val-HeFT (2001) [101] | 5010 | Valsartan vs. placebo | Creatinine > 2.5 mg/dL | Reduced HF hospitalization | ACEi intolerance in CRS type 2 and CRS type 4 |
| Candesartan | CHARM (2003) [102] | 7601 | Candesartan vs. placebo | Creatinine ≥ 3.0 mg/dL | Reduced CV death and HF hospitalization | CRS type 2 with HFrEF and CRS type 4 | ||
| CHARM-Preserved (2003) [103] | 3023 | Candesartan vs. placebo | Creatinine ≥ 3.0 mg/dL | Reduced HF hospitalization | Selected CRS with HFpEF | |||
| ARNI | Sacubitril/valsartan | Neprilysin inhibition + ARB | PARADIGM-HF (2014) [104] | 8442 | ARNI vs. enalapril | eGFR < 30 mL/min | Reduced CV death and HF hospitalization | Preferred RAAS therapy in CRS type 2 with HFrEF |
| PARAGON-HF (2019) [105] | 4822 | ARNI vs. valsartan | eGFR < 30 mL/min | Trend toward fewer HF hospitalizations | Selected CRS type 2 with HFpEF | |||
| Beta-blocker | Bisoprolol | β1-adrenergic blockade | CIBIS-II (1999) [106] | 2647 | Bisoprolol vs. placebo | Severe CKD excluded | Reduced all-cause mortality | Stable CRS type 2 with HFrEF |
| Metoprolol CR/XL | MERIT-HF (1999) [107] | 3991 | Metoprolol vs. placebo | Severe CKD excluded | Reduced mortality and HF hospitalization | CRS type 2 with stable hemodynamics | ||
| Carvedilol | COPERNICUS (2002) [108] | 2289 | Carvedilol vs. placebo | Creatinine > 2.8 mg/dL | Improved survival | Advanced CRS type 2 with careful titration | ||
| Various | J-DHF (2013) [109] | 245 | Beta-blocker vs. standard care | Advanced CKD excluded | No mortality benefit | Limited role in CRS type 2 with HFpEF | ||
| MRA | Eplerenone | Selective aldosterone blockade | EPHESUS (2003) [110] | 6632 | Eplerenone post-MI | Creatinine > 2.5 mg/dL | Reduced mortality | CRS type 2 with HFrEF with potassium monitoring |
| EMPHASIS-HF (2011) [111] | 2737 | Eplerenone vs. placebo | eGFR < 30 mL/min | Reduced CV death and HF hospitalization | Early CRS type 2 with HFrEF | |||
| Spironolactone | Aldosterone receptor antagonism | RALES (1999) [112] | 1663 | Spironolactone vs. placebo | Creatinine > 2.5 mg/dL | Reduced mortality | CRS type 2 with HFrEF with potassium monitoring | |
| Aldosterone receptor antagonism | TOPCAT (2014) [113] | 3445 | Spironolactone vs. placebo | Creatinine ≥ 2.5 mg/dL | Reduced HF hospitalization | Selected CRS type 2 with HFpEF | ||
| ATHENA-HF (2017) [114] | 360 | High-dose spironolactone (100 mg/day) vs. usual care in acute decompensated HF | eGFR < 30 mL/min/1.73 m2 or serum potassium > 5.0 mmol/L | No improvement in NT-proBNP levels or clinical congestion at 96 h | Limited role for early high-dose spironolactone in acute CRS type 1 | |||
| Non-steroidal MRA | Finerenone | Selective mineralocorticoid receptor antagonism | FIDELITY (2022) [115] | 13,171 | Finerenone vs. placebo | eGFR < 25 mL/min | Reduced renal and CV outcomes | CRS type 2 and 4 with diabetic CKD |
| FINEARTS-HF (2024) [116] | ~6000 | Finerenone vs. placebo | eGFR < 25 mL/min | Reduced HF events | CRS type 2 with HFpEF | |||
| Loop diuretics | Furosemide | Na-K-2Cl cotransporter inhibition | DOSE (2011) [50] | 308 | High vs. low IV dose | Creatinine > 3.0 mg/dL | Similar efficacy, transient renal dysfunction | First-line decongestion in CRS type 1 |
| Thiazides | Hydrochlorothiazide | Distal NaCl cotransporter inhibition | CLOROTIC (2023) [117] | 230 | Add-on thiazide | eGFR < 20 mL/min | Improved decongestion | Diuretic resistance in CRS type 1 |
| Carbonic anhydrase inhibitor | Acetazolamide | Proximal bicarbonate reabsorption inhibition | ADVOR (2022) [92] | 519 | Add-on acetazolamide | eGFR < 20 mL/min | Faster and more complete decongestion | CRS type 1 with volume overload |
| Vasopressin antagonist | Tolvaptan | V2 receptor antagonism | EVEREST (2007) [118] | 4133 | Tolvaptan vs. placebo | Severe CKD excluded | Improved congestion, no mortality benefit | Hyponatremic or refractory CRS type 1 |
| SGLT2 inhibitor | Dapagliflozin | DECLARE-TIMI 58 (2019) [119] | 17,160 | Dapagliflozin vs. placebo | eGFR < 60 mL/min/1.73 m2 at entry | Reduced hospitalization for heart failure; renal composite benefit, including reduced sustained eGFR decline | Diabetic CRS type 2 | |
| DAPA-HF (2019) [73] | 4744 | Dapagliflozin vs. placebo | eGFR < 30 mL/min | Reduced CV death and HF hospitalization | CRS type 2 with HFrEF and CRS type 4 | |||
| DAPA-CKD (2020) [120] | 4304 | Dapagliflozin vs. placebo | eGFR < 25 mL/min/1.73 m2 | Reduced progression to kidney failure and cardiovascular death in patients with and without diabetes | CRS type 4 regardless of diabetic status | |||
| Proximal sodium–glucose cotransport inhibition | DAPA ACT HF–TIMI 68 (2025) [121] | ~2400 | Early dapagliflozin vs. placebo in hospitalized HF | eGFR < 25 mL/min | Not significantly reduce the risk of cardiovascular death or worsening HF | CRS type 1 during HF hospitalization | ||
| Empagliflozin | EMPA-REG OUTCOME (2015) [122] | 7020 | mpagliflozin vs. placebo | eGFR < 30 mL/min/1.73 m2 | Reduced cardiovascular mortality; slower progression of kidney disease, and reduced incidence or worsening nephropathy | Diabetic CRS type 2 and CRS type 4 with established cardiovascular disease | ||
| EMPEROR-Reduced (2020) [123] | 3730 | Empagliflozin vs. placebo | eGFR < 20 mL/min | Reduced CV death or HF hospitalization. Slower eGFR decline | CRS type 2 with HFrEF and CRS type 4 | |||
| EMPEROR-Preserved (2021) [124] | 5988 | Empagliflozin vs. placebo | eGFR < 20 mL/min | Reduced HF hospitalization | CRS type 2 with HFpEF | |||
| EMPA-KIDNEY (2022) [125] | 6609 | Empagliflozin vs. placebo | eGFR < 20 mL/min/1.73 m2 | Reduced risk of kidney disease progression or cardiovascular death in CKD patients with and without diabetes | CRS type 4 independent of diabetes | |||
| EMPULSE (2022) [126] | 530 | Empagliflozin vs. placebo during AHF | eGFR < 20 mL/min | Improved clinical composite outcome | CRS type 1 | |||
| GLP-1 receptor agonist | Semaglutide | GLP-1 receptor activation | STEP-HFpEF (2023) [127] | 529 | Semaglutide vs. placebo | eGFR < 30 mL/min | Improved symptoms and weight loss | Obese CRS type 2 with HFpEF |
| SUSTAIN-6 (2016) [128] | 3297 | Semaglutide vs. placebo | eGFR < 30 mL/min | Reduced CV events | Diabetic CRS type 2 | |||
| FLOW (2024) [129] | 3533 | Semaglutide vs. placebo | eGFR < 25 mL/min | Reduced kidney failure and CV events | CRS type 4 with diabetic CKD | |||
| Ultrafiltration | Mechanical ultrafiltration | Extracorporeal fluid removal | UNLOAD (2007) [94] | 200 | Ultrafiltration vs. IV diuretics | Severe CKD excluded | Greater weight loss | Refractory CRS type 1 |
| RAPID-CHF (2005) [93] | 40 | Early ultrafiltration vs. standard diuretic therapy | Severe CKD excluded | Greater early fluid and weight loss without worsening renal function | Refractory CRS type 1 | |||
| CARRESS-HF (2012) [95] | 188 | Ultrafiltration vs. stepped pharmacologic care | Advanced CKD excluded | Worse renal outcomes | Refractory CRS type 1 | |||
| AVOID-HF (2016) [130] | 224 | Adjustable ultrafiltration vs. adjustable IV diuretics | Advanced CKD excluded | Trend toward fewer HF rehospitalizations; higher catheter-related adverse events | Refractory CRS type 1 | |||
| Peritoneal dialysis | Chronic peritoneal dialysis | Continuous volume control | Courivaud et al. (2014) [131] | 126 | PD in refractory HF | ESRD | Reduced HF hospitalizations | End-stage CRS type 2 and 4 |
| Bertoli et al. (2014) [132] | 48 | PD | ESRD | Symptom improvement | End-stage CRS type 2 and 4 | |||
| Cardiac resynchronization | CRT-D | Biventricular pacing | MADIT-CRT (2009) [133] | 1820 | CRT-D vs. ICD | Severe CKD excluded | Reduced HF events | CRS type 2 with HFrEF and electrical dyssynchrony |
| RAFT (2010) [134] | 1798 | CRT-D vs. ICD | Severe CKD excluded | Reduced mortality and HF hospitalization | CRS type 2 with HFrEF and electrical dyssynchrony | |||
| Implantable defibrillator | ICD | Sudden cardiac death prevention | SCD-HeFT (2005) [135] | 2521 | ICD vs. placebo/amiodarone | Advanced CKD excluded | Reduced all-cause mortality | CRS type 2 with HFrEF |
| Inotropes | Dopamine/Nesiritide | Increased cardiac output | ROSE-AHF (2014) [136] | 360 | Inotropes vs. placebo | Advanced CKD excluded | No renal benefit | Refractory CRS type 1 |
| Levosimendan | Calcium sensitization | REVIVE (2013) [137] | 600 | Levosimendan vs. placebo | Severe CKD excluded | Rapid and sustained symptomatic improvement; increased cardiovascular adverse events | Refractory CRS type 1 | |
| Anemia and iron therapy | Ferric carboxymaltose | Iron repletion | FAIR-HF (2009) [138] | 459 | IV iron vs. placebo | Severe CKD excluded | Improved symptoms and exercise capacity | CRS type 2 and 4 with iron deficiency |
| AFFIRM-AHF (2020) [139] | 1132 | IV iron vs. placebo | Severe CKD excluded | Reduced HF rehospitalization | CRS type 2 and 4 with iron deficiency | |||
| Darbepoetin alfa | Erythropoiesis stimulation | RED-HF (2013) [140] | 2278 | Darbepoetin vs. placebo | Severe CKD excluded | No CV benefit | Not recommended in CRS |
ACEi: Angiotensin-converting enzyme inhibitor; AHF: Acute heart failure; ARB: Angiotensin receptor blocker; ARNI: Angiotensin receptor–neprilysin inhibitor; β1: Beta-1 adrenergic receptor; CKD: Chronic kidney disease; CRS: Cardio-renal syndrome; CRT-D: Cardiac resynchronization therapy with defibrillator; CV: Cardiovascular; DAPA-HF: Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure trial; eGFR: Estimated glomerular filtration rate; ESRD: End-stage renal disease; HF: Heart failure; HFrEF: Heart failure with reduced ejection fraction; HFpEF: Heart failure with preserved ejection fraction; ICD: Implantable cardioverter–defibrillator; IV: Intravenous; MRA: Mineralocorticoid receptor antagonist; NT-proBNP: N-terminal pro-B-type natriuretic peptide; PD: Peritoneal dialysis; RAAS: Renin–angiotensin–aldosterone system; SGLT2 inhibitor: Sodium–glucose cotransporter 2 inhibitor; GLP-1 receptor agonist: Glucagon-like peptide-1 receptor agonist; V2 receptor: Vasopressin type 2 receptor.
9.6. Innovative Treatments in CRS
In recent years, basic and translational research has increasingly focused on emerging and experimental therapeutic pathways targeting the complex pathophysiology of CRS. These include strategies aimed at reducing oxidative stress and inflammation through targeted modulation of the inhibitor of kappa B (IκB)/nuclear factor kappa B (NF-κB) pathway, the Kelch-like ECH-associated protein 1 (Keap1)/nuclear factor erythroid 2–related factor 2 (Nrf2) pathway, and their downstream genes. Other approaches focus on remodeling the gut microbiota. Linaclotide, a guanylate cyclase C agonist, has been shown to restore microbial dysbiosis in murine models of chronic kidney disease. In addition, modulation of non-coding RNAs has emerged as a potential therapeutic strategy. Clofibrate, a peroxisome proliferator-activated receptor alpha (PPARα) agonist, improves left ventricular function in rats with type 4 CRS [87]. Overall, this is a rapidly expanding field, and advances in translational medicine may enable the incorporation of novel therapeutic strategies for CRS in the coming years.
Numerous studies are currently evaluating new agents for the treatment of CRS. These include pharmacological therapies designed to induce vasodilation and monoclonal antibodies targeting inflammatory cytokines. Other strategies aim to enhance innate and humoral immunity to mitigate inflammation and endothelial dysfunction. Diuretic resistance represents another major area of investigation. Ongoing trials are assessing pharmacological agents that increase diuretic responsiveness, such as lysine chloride, as well as non-diuretic approaches to remove salt and water, including sodium-free peritoneal dialysate. Finally, several device-based therapies are under investigation. These devices aim to reduce cardiac preload and afterload, increase renal preload by improving renal artery perfusion, and decrease renal afterload either by facilitating renal venous outflow or by reducing intracapsular renal pressure [1].
10. Conclusions
CRS management poses multiple clinical challenges. Classification into five types is not always straightforward, and distinguishing true AKI from hemoconcentration-related functional changes can be difficult [1,2,7,23,74]. Assessing fluid overload, individualizing diuretic therapy, and determining optimal decongestion rates remain challenging, though controlled rapid decongestion does not worsen renal prognosis or increase mortality [1,2,23,74].
Patient evaluation should integrate renal function with symptoms, vital signs, urine output, and CKD history, while excluding secondary causes such as persistent hypotension, nephrotoxics, or renovascular disease [1,2,23,74]. Multidisciplinary cardio-renal units and bedside echocardiography are key tools for prevention, early detection, and timely management of CRS [99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,120,122,123,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140].
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jcrm2010004/s1, Table S1: RIFLE, AKIN, KDIGO, and WRF classification.
Author Contributions
Conceptualization, M.M. and J.R.; resources, M.F. and L.P.B.; investigation, M.F. and L.P.B.; writing—original draft preparation, M.M., M.F., L.P.B. and J.R.; writing—review and editing, M.M., M.F., L.P.B. and J.R.; visualization, M.M., M.F., L.P.B. and J.R.; supervision, M.M. and J.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article. All the sources are listed in the references.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript
| CRS | Cardio-Renal Syndrome |
| HF | Heart Failure |
| CKD | Chronic Kidney Disease |
| ADQI | Acute Dialysis Quality Initiative |
| CV | Cardiovascular |
| AKI | Acute Kidney Injury |
| AHF | Acute Heart Failure |
| WRF | Worsening Renal Function |
| RIFLE | Risk Injury Failure Loss of Kidney Function and End-Stage Kidney Disease |
| AKIN | Acute Kidney Injury Network |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| CHF | Congestive Heart Failure |
| EF | Ejection Fraction |
| RAAS | Renin–Angiotensin–Aldosterone System |
| GFR | Glomerular Filtration Rate |
| SNS | Sympathetic Nervous System |
| ATII | Angiotensin II |
| ACE | Angiontensin-Converting Enzyme |
| TNF-α | Tumor necrosis factor-α |
| ILE-1 | Interleukin-1 |
| ILE-6 | Interleukin-6 |
| CRP | C- Reactive protein |
| EPO | Erythropoietin |
| eGFR | Estimated Glomerular Filtration Rate |
| ECG | Electrocardiogram |
| GLS | Global Longitudinal Strain |
| PCPW | Pulmonary Capillary Wedge Pressure |
| IVC | Inferior Vena Cava |
| RVSI | Renal Venous Stasis Index |
| AVP | Arginin–Vasopresin |
| ANP | Atrial Natriuretic Peptide |
| BNP | Brain Natriuretic Peptide |
| VExUS | Venous Excess Ultrasound Score |
| POCUS | Point of Care Ultrasound |
| ESRD | End Stage Renal Disease |
| iSGLT2s | SGLT 2 Inhibitors |
| RASis | Renin–Angiotensin System Inhibitors |
| ACEis | Angiotensin-Converting Enzyme Inhibitors |
| ARBs | Angiotensin Receptor Blockers |
| MRAs | Mineralocorticoid Receptor Antagonists |
| aGLP-1s | Glucagon-Like Peptide 1 Agonists |
| RRRT | Renal Replacement Therapy |
| NSAiDs | Non Steroidal Antiinflamatory Drugs |
| LVEF | Left Ventricle Ejection Fraction |
References
- McCallum, W.; Sarnak, M.J. Cardiorenal Syndrome in the Hospital. Clin. J. Am. Soc. Nephrol. 2023, 18, 933–945. [Google Scholar] [CrossRef]
- Ronco, C.; Haapio, M.; House, A.A.; Anavekar, N.; Bellomo, R. Cardiorenal Syndrome. J. Am. Coll. Cardiol. 2008, 52, 1527–1539. [Google Scholar] [CrossRef]
- Gallo, G.; Lanza, O.; Savoia, C. New Insight in Cardiorenal Syndrome: From Biomarkers to Therapy. Int. J. Mol. Sci. 2023, 24, 5089. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.P.; Katsurada, K.; Zheng, H. Cardiorenal Syndrome: The Role of Neural Connections Between the Heart and the Kidneys. Circ. Res. 2022, 130, 1601–1617. [Google Scholar] [CrossRef]
- McCallum, W.; Testani, J.M. Updates in Cardiorenal Syndrome. Med. Clin. N. Am. 2023, 107, 763–780. [Google Scholar] [CrossRef]
- Schefold, J.C.; Filippatos, G.; Hasenfuss, G.; Anker, S.D.; von Haehling, S. Heart Failure and Kidney Dysfunction: Epidemiology, Mechanisms and Management. Nat. Rev. Nephrol. 2016, 12, 610–623. [Google Scholar] [CrossRef] [PubMed]
- Rangaswami, J.; Bhalla, V.; Blair, J.E.A.; Chang, T.I.; Costa, S.; Lentine, K.L.; Lerma, E.V.; Mezue, K.; Molitch, M.; Mullens, W.; et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e840–e878. [Google Scholar] [CrossRef]
- Bright, R. Cases and Observations Illustrative of Renal Disease, Accompanied with the Secretion of Albuminous Urine. Med. Chir. Rev. 1836, 25, 23–35. [Google Scholar]
- Bongartz, L.G.; Cramer, M.J.M.; Braam, B. The Cardiorenal Connection. Hypertension 2004, 43, e14. [Google Scholar] [CrossRef]
- Sheikh, O.; Nguyen, T.; Bansal, S.; Prasad, A. Acute Kidney Injury in Cardiogenic Shock: A Comprehensive Review. Catheter. Cardiovasc. Interv. 2021, 98, E91–E105. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C.; House, A.A.; Haapio, M. Cardiorenal Syndrome: Refining the Definition of a Complex Symbiosis Gone Wrong. Intensive Care Med. 2008, 34, 957–962. [Google Scholar] [CrossRef]
- Valika, A.A.; Costanzo, M.R. The Acute Cardiorenal Syndrome Type I: Considerations on Physiology, Epidemiology, and Therapy. Curr. Heart Fail. Rep. 2014, 11, 382–392. [Google Scholar] [CrossRef]
- Núñez, J.; Miñana, G.; Santas, E.; Bertomeu-González, V. Cardiorenal Syndrome in Acute Heart Failure: Revisiting Paradigms. Rev. Esp. Cardiol. 2015, 68, 426–435. [Google Scholar] [CrossRef]
- Lameire, N.; Eknoyan, G. Kidney Disease: Improving Global Outcomes (KDIGO) KDIGO Clinical Practice Guidelines for the Prevention, Diagnosis, Evaluation, and Treatment of Hepatitis C in Chronic Kidney Disease. Kidney Int. 2008, 73, S1–S2. [Google Scholar] [CrossRef]
- Mehta, R.L.; Kellum, J.A.; Shah, S.V.; Molitoris, B.A.; Ronco, C.; Warnock, D.G.; Levin, A. Acute Kidney Injury Network Acute Kidney Injury Network: Report of an Initiative to Improve Outcomes in Acute Kidney Injury. Crit. Care 2007, 11, R31. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, R.; Ronco, C.; Kellum, J.A.; Mehta, R.L.; Palevsky, P. Acute Dialysis Quality Initiative workgroup Acute Renal Failure—Definition, Outcome Measures, Animal Models, Fluid Therapy and Information Technology Needs: The Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit. Care 2004, 8, R204–R212. [Google Scholar] [CrossRef] [PubMed]
- Heywood, J.T.; Fonarow, G.C.; Costanzo, M.R.; Mathur, V.S.; Wigneswaran, J.R.; Wynne, J.; ADHERE Scientific Advisory Committee and Investigators. High Prevalence of Renal Dysfunction and Its Impact on Outcome in 118,465 Patients Hospitalized with Acute Decompensated Heart Failure: A Report from the ADHERE Database. J. Card. Fail. 2007, 13, 422–430. [Google Scholar] [CrossRef]
- Jois, P.; Mebazaa, A. Cardio-Renal Syndrome Type 2: Epidemiology, Pathophysiology, and Treatment. Semin. Nephrol. 2012, 32, 26–30. [Google Scholar] [CrossRef]
- De Vecchis, R.; Baldi, C. Cardiorenal Syndrome Type 2: From Diagnosis to Optimal Management. Ther. Clin. Risk Manag. 2014, 10, 949–961. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yap, S.C.; Lee, H.T. Acute Kidney Injury and Extrarenal Organ Dysfunction: New Concepts and Experimental Evidence. Anesthesiology 2012, 116, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.J. Distant Effects of Experimental Renal Ischemia/Reperfusion Injury. J. Am. Soc. Nephrol. 2003, 14, 1549–1558. [Google Scholar] [CrossRef]
- Forman, D.E.; Butler, J.; Wang, Y.; Abraham, W.T.; O’Connor, C.M.; Gottlieb, S.S.; Loh, E.; Massie, B.M.; Rich, M.W.; Stevenson, L.W.; et al. Incidence, Predictors at Admission, and Impact of Worsening Renal Function among Patients Hospitalized with Heart Failure. J. Am. Coll. Cardiol. 2004, 43, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C.; McCullough, P.; Anker, S.D.; Anand, I.; Aspromonte, N.; Bagshaw, S.M.; Bellomo, R.; Berl, T.; Bobek, I.; Cruz, D.N.; et al. Cardio-Renal Syndromes: Report from the Consensus Conference of the Acute Dialysis Quality Initiative. Eur. Heart J. 2010, 31, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Clementi, A.; Virzì, G.M.; Brocca, A.; de Cal, M.; Vescovo, G.; Granata, A.; Ronco, C. Cardiorenal Syndrome Type 4: Management. Blood Purif. 2013, 36, 200–209. [Google Scholar] [CrossRef]
- Cases, A.; Broseta, J.J.; Marqués, M.; Cigarrán, S.; Julián, J.C.; Alcázar, R.; Ortiz, A. Cardiovascular-Kidney-Metabolic Syndrome Definition and Its Role in the Prevention, Risk Staging, and Treatment. An Opportunity for the Nephrology. Nefrología 2024, 44, 771–783. [Google Scholar] [CrossRef]
- Kumar, U.; Wettersten, N.; Garimella, P.S. Cardiorenal Syndrome: Pathophysiology. Cardiol. Clin. 2019, 37, 251–265. [Google Scholar] [CrossRef] [PubMed]
- Bock, J.S.; Gottlieb, S.S. Cardiorenal Syndrome: New Perspectives. Circulation 2010, 121, 2592–2600. [Google Scholar] [CrossRef]
- Nohria, A.; Hasselblad, V.; Stebbins, A.; Pauly, D.F.; Fonarow, G.C.; Shah, M.; Yancy, C.W.; Califf, R.M.; Stevenson, L.W.; Hill, J.A. Cardiorenal Interactions: Insights from the ESCAPE Trial. J. Am. Coll. Cardiol. 2008, 51, 1268–1274. [Google Scholar] [CrossRef]
- Gnanaraj, J.F.; von Haehling, S.; Anker, S.D.; Raj, D.S.; Radhakrishnan, J. The Relevance of Congestion in the Cardio-Renal Syndrome. Kidney Int. 2013, 83, 384–391. [Google Scholar] [CrossRef]
- Verbrugge, F.H.; Dupont, M.; Steels, P.; Grieten, L.; Malbrain, M.; Tang, W.H.W.; Mullens, W. Abdominal Contributions to Cardiorenal Dysfunction in Congestive Heart Failure. J. Am. Coll. Cardiol. 2013, 62, 485–495. [Google Scholar] [CrossRef]
- Maeder, M.T.; Holst, D.P.; Kaye, D.M. Tricuspid Regurgitation Contributes to Renal Dysfunction in Patients with Heart Failure. J. Card. Fail. 2008, 14, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Kawano, H.; Do, Y.S.; Kawano, Y.; Starnes, V.; Barr, M.; Law, R.E.; Hsueh, W.A. Angiotensin II Has Multiple Profibrotic Effects in Human Cardiac Fibroblasts. Circulation 2000, 101, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Iwao, H. Molecular and Cellular Mechanisms of Angiotensin II-Mediated Cardiovascular and Renal Diseases. Pharmacol. Rev. 2000, 52, 11–34. [Google Scholar] [CrossRef] [PubMed]
- Ronco, C.; Cicoira, M.; McCullough, P.A. Cardiorenal Syndrome Type 1: Pathophysiological Crosstalk Leading to Combined Heart and Kidney Dysfunction in the Setting of Acutely Decompensated Heart Failure. J. Am. Coll. Cardiol. 2012, 60, 1031–1042. [Google Scholar] [CrossRef]
- Barreto, D.V.; Barreto, F.C.; Liabeuf, S.; Temmar, M.; Lemke, H.-D.; Tribouilloy, C.; Choukroun, G.; Vanholder, R.; Massy, Z.A. European Uremic Toxin Work Group (EUTox) Plasma Interleukin-6 Is Independently Associated with Mortality in Both Hemodialysis and Pre-Dialysis Patients with Chronic Kidney Disease. Kidney Int. 2010, 77, 550–556. [Google Scholar] [CrossRef]
- Kim, B.-S.; Jeon, D.S.; Shin, M.J.; Kim, Y.O.; Song, H.C.; Lee, S.H.; Kim, S.Y.; Choi, E.J.; Chang, Y.S.; Bang, B.K. Persistent Elevation of C-Reactive Protein May Predict Cardiac Hypertrophy and Dysfunction in Patients Maintained on Hemodialysis. Am. J. Nephrol. 2005, 25, 189–195. [Google Scholar] [CrossRef]
- Silverberg, D.S.; Wexler, D.; Blum, M.; Wollman, Y.; Schwartz, D.; Sheps, D.; Keren, G.; Iaina, A. The Interaction between Heart Failure, Renal Failure and Anemia—The Cardio-Renal Anemia Syndrome. Blood Purif. 2004, 22, 277–284. [Google Scholar] [CrossRef]
- Marques Vidas, M.; Portolés, J.; Cobo, M.; Gorriz, J.L.; Nuñez, J.; Cases, A. Anemia Management in the Cardiorenal Patient: A Nephrological Perspective. J. Am. Heart Assoc. 2025, 14, e037363. [Google Scholar] [CrossRef]
- Damman, K.; Valente, M.A.E.; Voors, A.A.; O’Connor, C.M.; van Veldhuisen, D.J.; Hillege, H.L. Renal Impairment, Worsening Renal Function, and Outcome in Patients with Heart Failure: An Updated Meta-Analysis. Eur. Heart J. 2014, 35, 455–469. [Google Scholar] [CrossRef]
- Pliquett, R.U. Cardiorenal Syndrome: An Updated Classification Based on Clinical Hallmarks. J. Clin. Med. 2022, 11, 2896. [Google Scholar] [CrossRef]
- McCullough, P.A. Cardiorenal Syndromes: Pathophysiology to Prevention. Int. J. Nephrol. 2010, 2011, 762590. [Google Scholar] [CrossRef]
- Damman, K.; van Deursen, V.M.; Navis, G.; Voors, A.A.; van Veldhuisen, D.J.; Hillege, H.L. Increased Central Venous Pressure Is Associated with Impaired Renal Function and Mortality in a Broad Spectrum of Patients with Cardiovascular Disease. J. Am. Coll. Cardiol. 2009, 53, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.L. Fluid Volume Overload and Congestion in Heart Failure: Time to Reconsider Pathophysiology and How Volume Is Assessed. Circ. Heart Fail. 2016, 9, e002922. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Logeart, D.; Thabut, G.; Jourdain, P.; Chavelas, C.; Beyne, P.; Beauvais, F.; Bouvier, E.; Solal, A.C. Predischarge B-Type Natriuretic Peptide Assay for Identifying Patients at High Risk of Re-Admission after Decompensated Heart Failure. J. Am. Coll. Cardiol. 2004, 43, 635–641. [Google Scholar] [CrossRef]
- Beaubien-Souligny, W.; Rola, P.; Haycock, K.; Bouchard, J.; Lamarche, Y.; Spiegel, R.; Denault, A.Y. Quantifying Systemic Congestion with Point-Of-Care Ultrasound: Development of the Venous Excess Ultrasound Grading System. Ultrasound J. 2020, 12, 16. [Google Scholar] [CrossRef]
- Kashani, K.; Al-Khafaji, A.; Ardiles, T.; Artigas, A.; Bagshaw, S.M.; Bell, M.; Bihorac, A.; Birkhahn, R.; Cely, C.M.; Chawla, L.S.; et al. Discovery and Validation of Cell Cycle Arrest Biomarkers in Human Acute Kidney Injury. Crit. Care 2013, 17, R25. [Google Scholar] [CrossRef]
- Nijst, P.; Martens, P.; Dupont, M.; Tang, W.H.W.; Mullens, W. Intrarenal Flow Alterations During Transition From Euvolemia to Intravascular Volume Expansion in Heart Failure Patients. JACC Heart Fail. 2017, 5, 672–681. [Google Scholar] [CrossRef] [PubMed]
- ter Maaten, J.M.; Valente, M.A.E.; Damman, K.; Hillege, H.L.; Navis, G.; Voors, A.A. Diuretic Response in Acute Heart Failure-Pathophysiology, Evaluation, and Therapy. Nat. Rev. Cardiol. 2015, 12, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Felker, G.M.; Lee, K.L.; Bull, D.A.; Redfield, M.M.; Stevenson, L.W.; Goldsmith, S.R.; LeWinter, M.M.; Deswal, A.; Rouleau, J.L.; Ofili, E.O.; et al. Diuretic Strategies in Patients with Acute Decompensated Heart Failure. N. Engl. J. Med. 2011, 364, 797–805. [Google Scholar] [CrossRef]
- Gargani, L.; Pang, P.S.; Frassi, F.; Miglioranza, M.H.; Dini, F.L.; Landi, P.; Picano, E. Persistent Pulmonary Congestion before Discharge Predicts Rehospitalization in Heart Failure: A Lung Ultrasound Study. Cardiovasc. Ultrasound 2015, 13, 40. [Google Scholar] [CrossRef] [PubMed]
- Melenovsky, V.; Borlaug, B.A.; Rosen, B.; Hay, I.; Ferruci, L.; Morell, C.H.; Lakatta, E.G.; Najjar, S.S.; Kass, D.A. Cardiovascular Features of Heart Failure with Preserved Ejection Fraction versus Nonfailing Hypertensive Left Ventricular Hypertrophy in the Urban Baltimore Community: The Role of Atrial Remodeling/Dysfunction. J. Am. Coll. Cardiol. 2007, 49, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Rola, P.; Miralles-Aguiar, F.; Argaiz, E.; Beaubien-Souligny, W.; Haycock, K.; Karimov, T.; Dinh, V.A.; Spiegel, R. Clinical Applications of the Venous Excess Ultrasound (VExUS) Score: Conceptual Review and Case Series. Ultrasound J. 2021, 13, 32. [Google Scholar] [CrossRef]
- Iida, N.; Seo, Y.; Sai, S.; Machino-Ohtsuka, T.; Yamamoto, M.; Ishizu, T.; Kawakami, Y.; Aonuma, K. Clinical Implications of Intrarenal Hemodynamic Evaluation by Doppler Ultrasonography in Heart Failure. JACC Heart Fail. 2016, 4, 674–682. [Google Scholar] [CrossRef]
- Baldetti, L.; Cosenza, M.; Galdieri, C.; Gallone, G.; Ricchetti, G.; Gaspardone, C.; Peveri, B.; Gramegna, M.; Cianfanelli, L.; Calvo, F.; et al. Invasive Hemodynamic Monitoring in Acute Heart Failure and Cardiogenic Shock. Rev. Cardiovasc. Med. 2025, 26, 27034. [Google Scholar] [CrossRef]
- House, A.A.; Anand, I.; Bellomo, R.; Cruz, D.; Bobek, I.; Anker, S.D.; Aspromonte, N.; Bagshaw, S.; Berl, T.; Daliento, L.; et al. Definition and Classification of Cardio-Renal Syndromes: Workgroup Statements from the 7th ADQI Consensus Conference. Nephrol. Dial. Transplant. 2010, 25, 1416–1420. [Google Scholar] [CrossRef]
- Ling, L.-H.; Kistler, P.M.; Ellims, A.H.; Iles, L.M.; Lee, G.; Hughes, G.L.; Kalman, J.M.; Kaye, D.M.; Taylor, A.J. Diffuse Ventricular Fibrosis in Atrial Fibrillation: Noninvasive Evaluation and Relationships with Aging and Systolic Dysfunction. J. Am. Coll. Cardiol. 2012, 60, 2402–2408. [Google Scholar] [CrossRef]
- Park, J.; Joung, B.; Uhm, J.-S.; Shim, C.Y.; Hwang, C.; Hyoung Lee, M.; Pak, H.-N. High Left Atrial Pressures Are Associated with Advanced Electroanatomical Remodeling of Left Atrium and Independent Predictors for Clinical Recurrence of Atrial Fibrillation after Catheter Ablation. Heart Rhythm. 2014, 11, 953–960. [Google Scholar] [CrossRef]
- De Boer, R.A.; Pinto, Y.M.; Van Veldhuisen, D.J. The Imbalance between Oxygen Demand and Supply as a Potential Mechanism in the Pathophysiology of Heart Failure: The Role of Microvascular Growth and Abnormalities. Microcirculation 2003, 10, 113–126. [Google Scholar] [CrossRef]
- González, A.; Schelbert, E.B.; Díez, J.; Butler, J. Myocardial Interstitial Fibrosis in Heart Failure: Biological and Translational Perspectives. J. Am. Coll. Cardiol. 2018, 71, 1696–1706. [Google Scholar] [CrossRef] [PubMed]
- Gheorghiade, M.; Abraham, W.T.; Albert, N.M.; Gattis Stough, W.; Greenberg, B.H.; O’Connor, C.M.; She, L.; Yancy, C.W.; Young, J.; Fonarow, G.C.; et al. Relationship between Admission Serum Sodium Concentration and Clinical Outcomes in Patients Hospitalized for Heart Failure: An Analysis from the OPTIMIZE-HF Registry. Eur. Heart J. 2007, 28, 980–988. [Google Scholar] [CrossRef]
- Metra, M.; Nodari, S.; Parrinello, G.; Bordonali, T.; Bugatti, S.; Danesi, R.; Fontanella, B.; Lombardi, C.; Milani, P.; Verzura, G.; et al. Worsening Renal Function in Patients Hospitalised for Acute Heart Failure: Clinical Implications and Prognostic Significance. Eur. J. Heart Fail. 2008, 10, 188–195. [Google Scholar] [CrossRef]
- Kaye, D.M.; Lefkovits, J.; Jennings, G.L.; Bergin, P.; Broughton, A.; Esler, M.D. Adverse Consequences of High Sympathetic Nervous Activity in the Failing Human Heart. J. Am. Coll. Cardiol. 1995, 26, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Verbrugge, F.H.; Guazzi, M.; Testani, J.M.; Borlaug, B.A. Altered Hemodynamics and End-Organ Damage in Heart Failure: Impact on the Lung and Kidney. Circulation 2020, 142, 998–1012. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.A.; Braunwald, E. Ventricular Remodeling after Myocardial Infarction. Exp. Obs. Clin. Implic. Circ. 1990, 81, 1161–1172. [Google Scholar] [CrossRef]
- London, G.M.; Pannier, B.; Guerin, A.P.; Marchais, S.J.; Safar, M.E.; Cuche, J.L. Cardiac Hypertrophy, Aortic Compliance, Peripheral Resistance, and Wave Reflection in End-Stage Renal Disease. Comparative Effects of ACE Inhibition and Calcium Channel Blockade. Circulation 1994, 90, 2786–2796. [Google Scholar] [CrossRef]
- Mak, S.; Newton, G.E. The Oxidative Stress Hypothesis of Congestive Heart Failure: Radical Thoughts. Chest 2001, 120, 2035–2046. [Google Scholar] [CrossRef]
- Ellison, D.H. Diuretic Resistance: Physiology and Therapeutics. Semin. Nephrol. 1999, 19, 581–597. [Google Scholar]
- Sparks, M.A.; Crowley, S.D.; Gurley, S.B.; Mirotsou, M.; Coffman, T.M. Classical Renin-Angiotensin System in Kidney Physiology. Compr. Physiol. 2014, 4, 1201–1228. [Google Scholar] [CrossRef]
- Wilcox, C.S.; Testani, J.M.; Pitt, B. Pathophysiology of Diuretic Resistance and Its Implications for the Management of Chronic Heart Failure. Hypertension 2020, 76, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Rossi, J.; Bayram, M.; Udelson, J.E.; Lloyd-Jones, D.; Adams, K.F.; Oconnor, C.M.; Stough, W.G.; Ouyang, J.; Shin, D.D.; Orlandi, C.; et al. Improvement in Hyponatremia during Hospitalization for Worsening Heart Failure Is Associated with Improved Outcomes: Insights from the Acute and Chronic Therapeutic Impact of a Vasopressin Antagonist in Chronic Heart Failure (ACTIV in CHF) Trial. Acute Card. Care 2007, 9, 82–86. [Google Scholar] [CrossRef]
- Piazza, G.; Goldhaber, S.Z. Venous Thromboembolism and Atherothrombosis: An Integrated Approach. Circulation 2010, 121, 2146–2150. [Google Scholar] [CrossRef]
- McMurray, J.J.V.; Solomon, S.D.; Inzucchi, S.E.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; Bělohlávek, J.; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, B.; Santamaría Olomo, R.; Gorostidi, M. Síndrome Cardiorrenal. Nefrología al día. (Online). Sociedad Española de nefrología. 2023. Available online: https://nefrologiaaldia.org/es-articulo-sindrome-cardiorrenal-555 (accessed on 1 November 2025).
- Goldstein, S.L.; Jaber, B.L.; Faubel, S.; Chawla, L.S. Acute Kidney Injury Advisory Group of American Society of Nephrology AKI Transition of Care: A Potential Opportunity to Detect and Prevent CKD. Clin. J. Am. Soc. Nephrol. 2013, 8, 476–483. [Google Scholar] [CrossRef]
- Mehran, R.; Owen, R.; Chiarito, M.; Baber, U.; Sartori, S.; Cao, D.; Nicolas, J.; Pivato, C.A.; Nardin, M.; Krishnan, P.; et al. A Contemporary Simple Risk Score for Prediction of Contrast-Associated Acute Kidney Injury after Percutaneous Coronary Intervention: Derivation and Validation from an Observational Registry. Lancet 2021, 398, 1974–1983. [Google Scholar] [CrossRef]
- Mehran, R.; Aymong, E.D.; Nikolsky, E.; Lasic, Z.; Iakovou, I.; Fahy, M.; Mintz, G.S.; Lansky, A.J.; Moses, J.W.; Stone, G.W.; et al. A Simple Risk Score for Prediction of Contrast-Induced Nephropathy after Percutaneous Coronary Intervention: Development and Initial Validation. J. Am. Coll. Cardiol. 2004, 44, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Uzendu, A.; Kennedy, K.; Chertow, G.; Amin, A.P.; Giri, J.S.; Rymer, J.A.; Bangalore, S.; Lavin, K.; Anderson, C.; Wang, T.Y.; et al. Contemporary Methods for Predicting Acute Kidney Injury After Coronary Intervention. JACC Cardiovasc. Interv. 2023, 16, 2294–2305. [Google Scholar] [CrossRef] [PubMed]
- Thakar, C.V.; Arrigain, S.; Worley, S.; Yared, J.-P.; Paganini, E.P. A Clinical Score to Predict Acute Renal Failure after Cardiac Surgery. J. Am. Soc. Nephrol. 2005, 16, 162–168. [Google Scholar] [CrossRef]
- Wijeysundera, D.N.; Karkouti, K.; Dupuis, J.-Y.; Rao, V.; Chan, C.T.; Granton, J.T.; Beattie, W.S. Derivation and Validation of a Simplified Predictive Index for Renal Replacement Therapy After Cardiac Surgery. JAMA 2007, 297, 1801–1809. [Google Scholar] [CrossRef]
- Lee, C.P.-T.; Ng, K.H.-L.; Choi, G.Y.-S.; Wong, W.-T.; Wong, H.M.K.; Ho, K.-M.; Ling, L.; Wong, R.H.-L. Performance of Cleveland, Mehta, and Simplified Renal Index Scores for Predicting Dialysis-Requiring Acute Kidney Injury after Aortic vs. Non-Aortic Cardiac Surgery (2006–2023, 6160 Patients). Ren. Fail. 2025, 47, 2592437. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, A.; Xu, Z.; Yin, Z.; Wang, C.; Zhang, J.; Bian, L. Analysis of Correlative Factors of Female Coronary Slow-Flow Phenomenon: A Retrospective Study. Medicine 2024, 103, e38262. [Google Scholar] [CrossRef]
- Petrosyan, Y.; Mesana, T.G.; Sun, L.Y. Prediction of Acute Kidney Injury Risk after Cardiac Surgery: Using a Hybrid Machine Learning Algorithm. BMC Med. Inform. Decis. Mak. 2022, 22, 137. [Google Scholar] [CrossRef]
- Lameire, N.; Kellum, J.A. KDIGO AKI Guideline Work Group Contrast-Induced Acute Kidney Injury and Renal Support for Acute Kidney Injury: A KDIGO Summary (Part 2). Crit. Care 2013, 17, 205. [Google Scholar] [CrossRef] [PubMed]
- Chikatimalla, R.; Trivedi, Y.V.; Ruhela, N.; Singh, S.; Lal, A.; Pawa, A.; Singh, B.; Jain, R. Statins and Kidney Health: Exploring Cardiovascular Benefits, Renal Protection, and Risks in Chronic Kidney Disease. Postgrad. Med. 2025, 137, 588–600. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Zheng, R.; Deng, B.; Liu, P.; Wang, Y. Association of N-Acetylcysteine Use with Contrast-Induced Nephropathy: An Umbrella Review of Meta-Analyses of Randomized Clinical Trials. Front. Med. 2023, 10, 1235023. [Google Scholar] [CrossRef]
- Zhao, B.-R.; Hu, X.-R.; Wang, W.-D.; Zhou, Y. Cardiorenal Syndrome: Clinical Diagnosis, Molecular Mechanisms and Therapeutic Strategies. Acta Pharmacol. Sin. 2025, 46, 1539–1555. [Google Scholar] [CrossRef]
- Kellum, J.A.; Lameire, N. KDIGO AKI Guideline Work Group Diagnosis, Evaluation, and Management of Acute Kidney Injury: A KDIGO Summary (Part 1). Crit. Care 2013, 17, 204. [Google Scholar] [CrossRef] [PubMed]
- Khwaja, A. KDIGO Clinical Practice Guidelines for Acute Kidney Injury. Nephron Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2022, 79, e263–e421. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2023, 44, 3627–3639. [Google Scholar] [CrossRef]
- Mullens, W.; Dauw, J.; Martens, P.; Verbrugge, F.H.; Nijst, P.; Meekers, E.; Tartaglia, K.; Chenot, F.; Moubayed, S.; Dierckx, R.; et al. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N. Engl. J. Med. 2022, 387, 1185–1195. [Google Scholar] [CrossRef]
- Bart, B.A.; Boyle, A.; Bank, A.J.; Anand, I.; Olivari, M.T.; Kraemer, M.; Mackedanz, S.; Sobotka, P.A.; Schollmeyer, M.; Goldsmith, S.R. Ultrafiltration versus Usual Care for Hospitalized Patients with Heart Failure: The Relief for Acutely Fluid-Overloaded Patients with Decompensated Congestive Heart Failure (RAPID-CHF) Trial. J. Am. Coll. Cardiol. 2005, 46, 2043–2046. [Google Scholar] [CrossRef]
- Costanzo, M.R.; Guglin, M.E.; Saltzberg, M.T.; Jessup, M.L.; Bart, B.A.; Teerlink, J.R.; Jaski, B.E.; Fang, J.C.; Feller, E.D.; Haas, G.J.; et al. Ultrafiltration versus Intravenous Diuretics for Patients Hospitalized for Acute Decompensated Heart Failure. J. Am. Coll. Cardiol. 2007, 49, 675–683. [Google Scholar] [CrossRef]
- Bart, B.A.; Goldsmith, S.R.; Lee, K.L.; Givertz, M.M.; O’Connor, C.M.; Bull, D.A.; Redfield, M.M.; Deswal, A.; Rouleau, J.L.; LeWinter, M.M.; et al. Ultrafiltration in Decompensated Heart Failure with Cardiorenal Syndrome. N. Engl. J. Med. 2012, 367, 2296–2304. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.U.; Mancini, G.B.J.; Fitzpatrick-Lewis, D.; Connelly, K.A.; O’Meara, E.; Zieroth, S.; Sherifali, D. The Effectiveness of Sodium-Glucose Co-Transporter 2 Inhibitors on Cardiorenal Outcomes: An Updated Systematic Review and Meta-Analysis. Cardiovasc. Diabetol. 2024, 23, 72. [Google Scholar] [CrossRef]
- Rai, B.; Dar, S.F.; Alowami, M.; Sajjad, R.B.; Agarwal, T.; Khan, M.H.; Mubarak Ali, S.R.; Kamran, G.; Thomas, R.P.; Mudiyanselage, N.S.B.A.; et al. Finerenone and Its Cardiorenal Protective Effects: A Meta-Analysis of 21,731 Patients From Randomized Trials. Cardiol. Rev. 2025. Epub ahead of print. [Google Scholar] [CrossRef]
- Sasaki, T.; Giang, S.M.; Wu, J.; Yokoo, T.; Gallagher, M.; Bellomo, R.; Wang, A.Y. The Effect of GLP-1 Receptor Agonists on Renal Outcomes: A Systematic Review and Meta-Analysis. Nephrol. Dial. Transplant. 2025. Epub ahead of print. [Google Scholar] [CrossRef]
- SOLVD Investigators; Yusuf, S.; Pitt, B.; Davis, C.E.; Hood, W.B.; Cohn, J.N. Effect of Enalapril on Survival in Patients with Reduced Left Ventricular Ejection Fractions and Congestive Heart Failure. N. Engl. J. Med. 1991, 325, 293–302. [Google Scholar] [CrossRef]
- SOLVD Investigators; Yusuf, S.; Pitt, B.; Davis, C.E.; Hood, W.B.; Cohn, J.N. Effect of Enalapril on Mortality and the Development of Heart Failure in Asymptomatic Patients with Reduced Left Ventricular Ejection Fractions. N. Engl. J. Med. 1992, 327, 685–691. [Google Scholar] [CrossRef]
- Cohn, J.N.; Tognoni, G. Valsartan Heart Failure Trial Investigators A Randomized Trial of the Angiotensin-Receptor Blocker Valsartan in Chronic Heart Failure. N. Engl. J. Med. 2001, 345, 1667–1675. [Google Scholar] [CrossRef]
- Pfeffer, M.A.; Swedberg, K.; Granger, C.B.; Held, P.; McMurray, J.J.V.; Michelson, E.L.; Olofsson, B.; Ostergren, J.; Yusuf, S.; Pocock, S.; et al. Effects of Candesartan on Mortality and Morbidity in Patients with Chronic Heart Failure: The CHARM-Overall Programme. Lancet 2003, 362, 759–766. [Google Scholar] [CrossRef]
- Yusuf, S.; Pfeffer, M.A.; Swedberg, K.; Granger, C.B.; Held, P.; McMurray, J.J.V.; Michelson, E.L.; Olofsson, B.; Ostergren, J. CHARM Investigators and Committees Effects of Candesartan in Patients with Chronic Heart Failure and Preserved Left-Ventricular Ejection Fraction: The CHARM-Preserved Trial. Lancet 2003, 362, 777–781. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.V.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.C.; Solomon, S.D.; Swedberg, K.; et al. Angiotensin-Neprilysin Inhibition versus Enalapril in Heart Failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef]
- Solomon, S.D.; McMurray, J.J.V.; Anand, I.S.; Ge, J.; Lam, C.S.P.; Maggioni, A.P.; Martinez, F.; Packer, M.; Pfeffer, M.A.; Pieske, B.; et al. Angiotensin-Neprilysin Inhibition in Heart Failure with Preserved Ejection Fraction. N. Engl. J. Med. 2019, 381, 1609–1620. [Google Scholar] [CrossRef]
- Lechat, P.; Brunhuber, K.W.; Hofmann, R.; Kuhn, P.; Nesser, H.J.; Slany, J.; Weihs, W.; Wiedermann, C.; Wimmer, H.; van Mieghem, W.; et al. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): A Randomised Trial. Lancet 1999, 353, 9–13. [Google Scholar] [CrossRef]
- MERIT-HF Study Group. Effect of Metoprolol CR/XL in Chronic Heart Failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999, 353, 2001–2007. [Google Scholar] [CrossRef]
- Packer, M.; Fowler, M.B.; Roecker, E.B.; Coats, A.J.S.; Katus, H.A.; Krum, H.; Mohacsi, P.; Rouleau, J.L.; Tendera, M.; Staiger, C.; et al. Effect of Carvedilol on the Morbidity of Patients with Severe Chronic Heart Failure: Results of the Carvedilol Prospective Randomized Cumulative Survival (COPERNICUS) Study. Circulation 2002, 106, 2194–2199. [Google Scholar] [CrossRef]
- Yamamoto, K.; Origasa, H.; Hori, M. J-DHF Investigators Effects of Carvedilol on Heart Failure with Preserved Ejection Fraction: The Japanese Diastolic Heart Failure Study (J-DHF). Eur. J. Heart Fail. 2013, 15, 110–118. [Google Scholar] [CrossRef]
- Pitt, B.; Remme, W.; Zannad, F.; Neaton, J.; Martinez, F.; Roniker, B.; Bittman, R.; Hurley, S.; Kleiman, J.; Gatlin, M.; et al. Eplerenone, a Selective Aldosterone Blocker, in Patients with Left Ventricular Dysfunction after Myocardial Infarction. N. Engl. J. Med. 2003, 348, 1309–1321. [Google Scholar] [CrossRef]
- Zannad, F.; McMurray, J.J.V.; Krum, H.; van Veldhuisen, D.J.; Swedberg, K.; Shi, H.; Vincent, J.; Pocock, S.J.; Pitt, B. EMPHASIS-HF Study Group Eplerenone in Patients with Systolic Heart Failure and Mild Symptoms. N. Engl. J. Med. 2011, 364, 11–21. [Google Scholar] [CrossRef]
- Pitt, B.; Zannad, F.; Remme, W.J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The Effect of Spironolactone on Morbidity and Mortality in Patients with Severe Heart Failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 1999, 341, 709–717. [Google Scholar] [CrossRef]
- Pitt, B.; Pfeffer, M.A.; Assmann, S.F.; Boineau, R.; Anand, I.S.; Claggett, B.; Clausell, N.; Desai, A.S.; Diaz, R.; Fleg, J.L.; et al. Spironolactone for Heart Failure with Preserved Ejection Fraction. N. Engl. J. Med. 2014, 370, 1383–1392. [Google Scholar] [CrossRef]
- Butler, J.; Anstrom, K.J.; Felker, G.M.; Givertz, M.M.; Kalogeropoulos, A.P.; Konstam, M.A.; Mann, D.L.; Margulies, K.B.; McNulty, S.E.; Mentz, R.J.; et al. Efficacy and Safety of Spironolactone in Acute Heart Failure: The ATHENA-HF Randomized Clinical Trial. JAMA Cardiol. 2017, 2, 950–958. [Google Scholar] [CrossRef]
- Agarwal, R.; Filippatos, G.; Pitt, B.; Anker, S.D.; Rossing, P.; Joseph, A.; Kolkhof, P.; Nowack, C.; Gebel, M.; Ruilope, L.M.; et al. Cardiovascular and Kidney Outcomes with Finerenone in Patients with Type 2 Diabetes and Chronic Kidney Disease: The FIDELITY Pooled Analysis. Eur. Heart J. 2022, 43, 474–484. [Google Scholar] [CrossRef]
- Solomon, S.D.; McMurray, J.J.V.; Vaduganathan, M.; Claggett, B.; Jhund, P.S.; Desai, A.S.; Henderson, A.D.; Lam, C.S.P.; Pitt, B.; Senni, M.; et al. Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N. Engl. J. Med. 2024, 391, 1475–1485. [Google Scholar] [CrossRef]
- Trullàs, J.C.; Morales-Rull, J.L.; Casado, J.; Carrera-Izquierdo, M.; Sánchez-Marteles, M.; Conde-Martel, A.; Dávila-Ramos, M.F.; Llácer, P.; Salamanca-Bautista, P.; Pérez-Silvestre, J.; et al. Combining Loop with Thiazide Diuretics for Decompensated Heart Failure: The CLOROTIC Trial. Eur. Heart J. 2023, 44, 411–421. [Google Scholar] [CrossRef]
- Konstam, M.A.; Gheorghiade, M.; Burnett, J.C.; Grinfeld, L.; Maggioni, A.P.; Swedberg, K.; Udelson, J.E.; Zannad, F.; Cook, T.; Ouyang, J.; et al. Effects of Oral Tolvaptan in Patients Hospitalized for Worsening Heart Failure: The EVEREST Outcome Trial. JAMA 2007, 297, 1319–1331. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Heerspink, H.J.L.; Stefánsson, B.V.; Correa-Rotter, R.; Chertow, G.M.; Greene, T.; Hou, F.-F.; Mann, J.F.E.; McMurray, J.J.V.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- Berg, D.D.; Patel, S.M.; Haller, P.M.; Cange, A.L.; Palazzolo, M.G.; Bellavia, A.; Kuder, J.F.; Desai, A.S.; Inzucchi, S.E.; McMurray, J.J.V.; et al. Dapagliflozin in Patients Hospitalized for Heart Failure: Primary Results of the DAPA ACT HF-TIMI 68 Randomized Clinical Trial and Meta-Analysis of Sodium-Glucose Cotransporter-2 Inhibitors in Patients Hospitalized for Heart Failure. Circulation 2025, 152, 1411–1422. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Pocock, S.J.; Carson, P.; Januzzi, J.; Verma, S.; Tsutsui, H.; Brueckmann, M.; et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N. Engl. J. Med. 2020, 383, 1413–1424. [Google Scholar] [CrossRef]
- Anker, S.D.; Butler, J.; Filippatos, G.; Ferreira, J.P.; Bocchi, E.; Böhm, M.; Brunner-La Rocca, H.-P.; Choi, D.-J.; Chopra, V.; Chuquiure-Valenzuela, E.; et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N. Engl. J. Med. 2021, 385, 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- The EMPA-KIDNEY Collaborative Group; Herrington, W.G.; Staplin, N.; Wanner, C.; Green, J.B.; Hauske, S.J.; Emberson, J.R.; Preiss, D.; Judge, P.; Mayne, K.J.; et al. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Biegus, J.; Voors, A.A.; Collins, S.P.; Kosiborod, M.N.; Teerlink, J.R.; Angermann, C.E.; Tromp, J.; Ferreira, J.P.; Nassif, M.E.; Psotka, M.A.; et al. Impact of Empagliflozin on Decongestion in Acute Heart Failure: The EMPULSE Trial. Eur. Heart J. 2023, 44, 41–50. [Google Scholar] [CrossRef]
- Kosiborod, M.N.; Abildstrøm, S.Z.; Borlaug, B.A.; Butler, J.; Rasmussen, S.; Davies, M.; Hovingh, G.K.; Kitzman, D.W.; Lindegaard, M.L.; Møller, D.V.; et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. N. Engl. J. Med. 2023, 389, 1069–1084. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L.; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef]
- Perkovic, V.; Tuttle, K.R.; Rossing, P.; Mahaffey, K.W.; Mann, J.F.E.; Bakris, G.; Baeres, F.M.M.; Idorn, T.; Bosch-Traberg, H.; Lausvig, N.L.; et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. N. Engl. J. Med. 2024, 391, 109–121. [Google Scholar] [CrossRef]
- Costanzo, M.R.; Negoianu, D.; Jaski, B.E.; Bart, B.A.; Heywood, J.T.; Anand, I.S.; Smelser, J.M.; Kaneshige, A.M.; Chomsky, D.B.; Adler, E.D.; et al. Aquapheresis Versus Intravenous Diuretics and Hospitalizations for Heart Failure. JACC Heart Fail. 2016, 4, 95–105. [Google Scholar] [CrossRef]
- Courivaud, C.; Kazory, A.; Crépin, T.; Azar, R.; Bresson-Vautrin, C.; Chalopin, J.-M.; Ducloux, D. Peritoneal Dialysis Reduces the Number of Hospitalization Days in Heart Failure Patients Refractory to Diuretics. Perit. Dial. Int. 2014, 34, 100–108. [Google Scholar] [CrossRef]
- Bertoli, S.V.; Musetti, C.; Ciurlino, D.; Basile, C.; Galli, E.; Gambaro, G.; Iadarola, G.; Guastoni, C.; Carlini, A.; Fasciolo, F.; et al. Peritoneal Ultrafiltration in Refractory Heart Failure: A Cohort Study. Perit. Dial. Int. 2014, 34, 64–70. [Google Scholar] [CrossRef]
- Moss, A.J.; Hall, W.J.; Cannom, D.S.; Klein, H.; Brown, M.W.; Daubert, J.P.; Estes, N.A.M.; Foster, E.; Greenberg, H.; Higgins, S.L.; et al. Cardiac-Resynchronization Therapy for the Prevention of Heart-Failure Events. N. Engl. J. Med. 2009, 361, 1329–1338. [Google Scholar] [CrossRef]
- Tang, A.S.L.; Wells, G.A.; Talajic, M.; Arnold, M.O.; Sheldon, R.; Connolly, S.; Hohnloser, S.H.; Nichol, G.; Birnie, D.H.; Sapp, J.L.; et al. Cardiac-Resynchronization Therapy for Mild-to-Moderate Heart Failure. N. Engl. J. Med. 2010, 363, 2385–2395. [Google Scholar] [CrossRef] [PubMed]
- Bardy, G.H.; Lee, K.L.; Mark, D.B.; Poole, J.E.; Packer, D.L.; Boineau, R.; Domanski, M.; Troutman, C.; Anderson, J.; Johnson, G.; et al. Amiodarone or an Implantable Cardioverter-Defibrillator for Congestive Heart Failure. N. Engl. J. Med. 2005, 352, 225–237. [Google Scholar] [CrossRef]
- Chen, H.H.; Anstrom, K.J.; Givertz, M.M.; Stevenson, L.W.; Semigran, M.J.; Goldsmith, S.R.; Bart, B.A.; Bull, D.A.; Stehlik, J.; LeWinter, M.M.; et al. Low-Dose Dopamine or Low-Dose Nesiritide in Acute Heart Failure with Renal Dysfunction: The ROSE Acute Heart Failure Randomized Trial. JAMA 2013, 310, 2533–2543. [Google Scholar] [CrossRef] [PubMed]
- Packer, M.; Colucci, W.; Fisher, L.; Massie, B.M.; Teerlink, J.R.; Young, J.; Padley, R.J.; Thakkar, R.; Delgado-Herrera, L.; Salon, J.; et al. Effect of Levosimendan on the Short-Term Clinical Course of Patients with Acutely Decompensated Heart Failure. JACC Heart Fail. 2013, 1, 103–111. [Google Scholar] [CrossRef]
- Anker, S.D.; Comin Colet, J.; Filippatos, G.; Willenheimer, R.; Dickstein, K.; Drexler, H.; Lüscher, T.F.; Bart, B.; Banasiak, W.; Niegowska, J.; et al. Ferric Carboxymaltose in Patients with Heart Failure and Iron Deficiency. N. Engl. J. Med. 2009, 361, 2436–2448. [Google Scholar] [CrossRef]
- Ponikowski, P.; Kirwan, B.-A.; Anker, S.D.; McDonagh, T.; Dorobantu, M.; Drozdz, J.; Fabien, V.; Filippatos, G.; Göhring, U.M.; Keren, A.; et al. Ferric Carboxymaltose for Iron Deficiency at Discharge after Acute Heart Failure: A Multicentre, Double-Blind, Randomised, Controlled Trial. Lancet 2020, 396, 1895–1904. [Google Scholar] [CrossRef] [PubMed]
- Swedberg, K.; Young, J.B.; Anand, I.S.; Cheng, S.; Desai, A.S.; Diaz, R.; Maggioni, A.P.; McMurray, J.J.V.; O’Connor, C.; Pfeffer, M.A.; et al. Treatment of Anemia with Darbepoetin Alfa in Systolic Heart Failure. N. Engl. J. Med. 2013, 368, 1210–1219. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.